Abstract
Extracellular vesicles (EVs) are lipid bilayer-bound vesicles secreted by cells. Subtypes of EVs such as microvesicles and exosomes are further categorized mainly by their different biogenesis mechanisms. EVs have been revealed to play an important role in disease diagnosis and intercellular communication. Despite the wide interest in EVs, the technologies for the purification and enrichment of EVs are still in their infancy. The isolation of EVs, especially exosomes, is inherently challenging due to their small size and heterogeneity. In this review, we mainly introduce the advances of techniques in isolating microvesicles and exosomes according to their approaches. Also, we discuss the limitations of currently reported technologies in terms of their specificity and efficiency, and provide our thoughts about future developments of EV purification and enrichment technology.
Introduction
Extracellular vesicles (EVs) are lipid bilayer-bound vesicles generated by cells. While EVs were initially regarded as cellular debris, 1 they are now recognized to play an important role in intercellular communication. Because of their tiny size (50–1000 nm), 2 EVs possess a strong ability to invade and disperse in the human body. Consequently, EVs are present in almost all biological fluids. 3 Meanwhile, EVs have been demonstrated to contain abundant proteins, nucleic acids, and lipids. 4 Hence, the effects of EVs in intercellular signaling and the application of EVs as body status biomarkers have been of great interest in biological and clinical research.
EVs are usually classified into three main types based on their different biogenesis mechanisms ( Fig. 1 ). 5 The first category is exosomes that are vesicles formed in multivesicular bodies (MVBs) and secreted by cells when MVBs fuse with the cytoplasmic membrane. Exosomes usually have sizes ranging from 50 to 150 nm and densities from 1.15 to 1.19 g/mL. 6 Exosomes have been reported to participate in the signaling of disease, such as tumor progression and metastasis.7–14 In contrast, microvesicles (MVs) are generated through budding of the cell membrane. MVs are believed to have heterogeneous sizes ranging from 50 to 1000 nm and densities from 1.12 to 1.16 g/mL. 15 MVs usually carry different sets of proteins and nucleic acids from exosomes; however, they are also reported to be cell–cell messengers.16–20 Apoptotic bodies may also be regarded as a subtype of EVs, and they are formed during the cell apoptotic process. In this review, we do not discuss these apoptotic bodies.
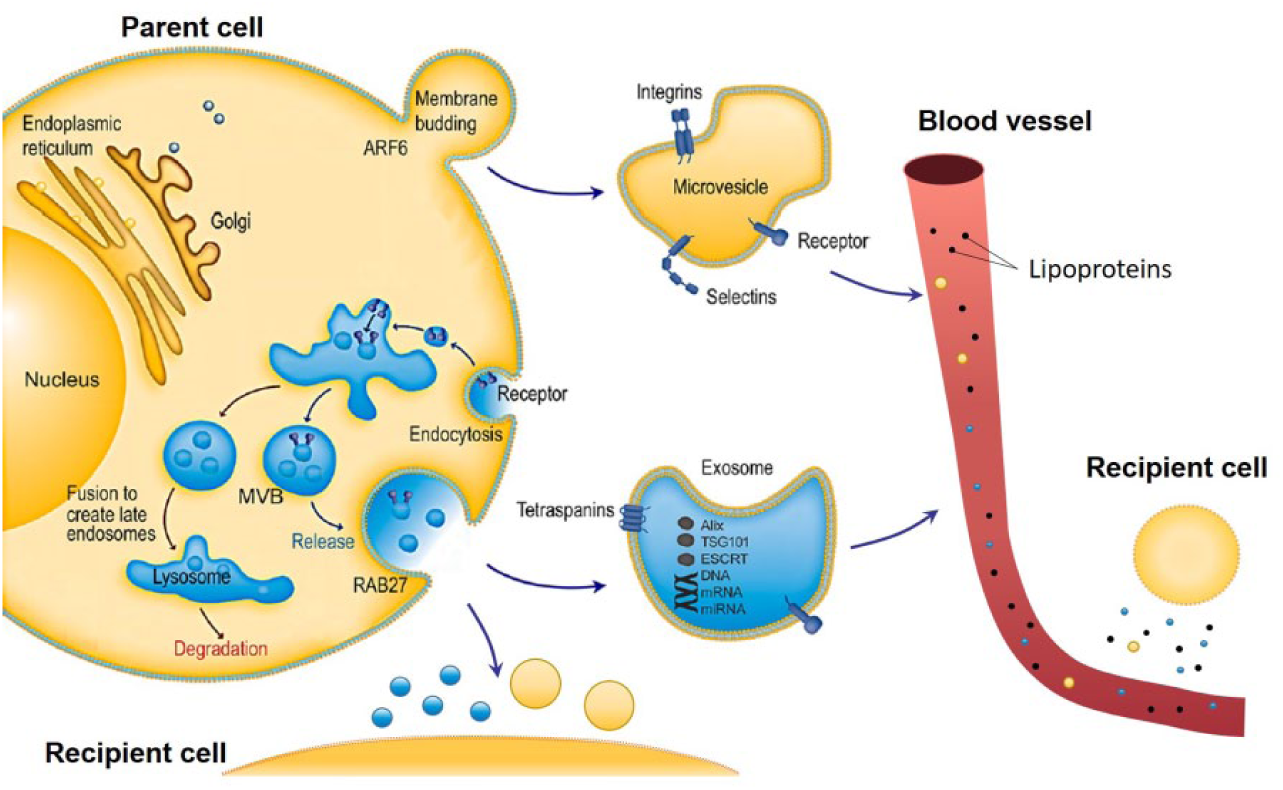
EV biogenesis and role in intercellular communications. Exosomes are vesicles formed in MVBs and secreted by cell when MVBs fuse with cthe ytoplasmic membrane. In contrast, MVs are generated through budding of the cell membrane. EVs can deliver proteins and nucleic acids to recipient cells in proximity, or reach distant recipient cells through the blood circulation. Reprinted with permission from Shao et al. 21 Copyright 2018 American Chemical Society.
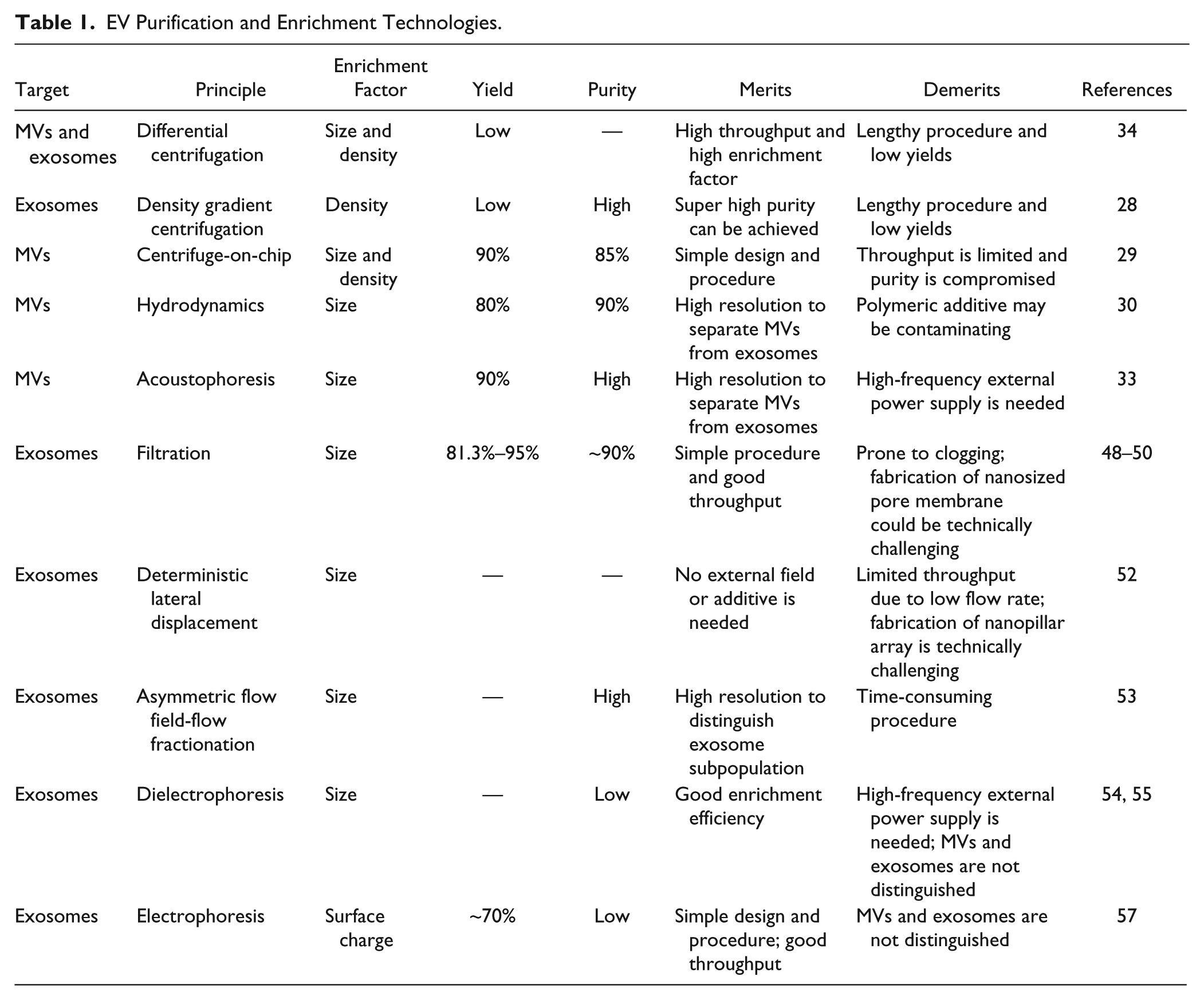
Despite the exponentially growing interest in EVs, it is still technically challenging to acquire EV samples with high yield and high purity. This article particularly reviews recent advances in methods and technologies that were developed for the enrichment and purification of EVs. These technologies were classified according to the physical laws they were based on. The yield, purity, merits, and demerits of these methods are summarized in Table 1 . For more information about EV analysis technologies 21 and biology, 22 we recommend other reviews focusing on these specific topics.
EV Purification and Enrichment Technologies.
Microvesicles Purification and Enrichment Technology
The isolation of EVs is important before downstream diagnostic or therapeutic approaches can be performed. Without this step, information obtained may be flawed owing to the cross-contamination of other bioparticles present in the fluids. Crescitelli et al. 23 demonstrated this importance in their study, which found distinct RNA profiles in subpopulations of EVs. However, obtaining pure, enriched EV subpopulations is not trivial because of their small size and heterogeneity. 24 We noticed that new technologies have been developed to separate larger MVs. In this section, we analyze the different techniques in obtaining MVs between sizes of 100 and 1000 nm, noting their advantages and drawbacks.
Centrifugation
The most common way to retrieve MVs in biofluids is centrifugation. Centrifugation is typically used to pellet larger particulate matter in solution. For example, in whole blood samples, cells were isolated and removed by pelleting with centrifugation at 300
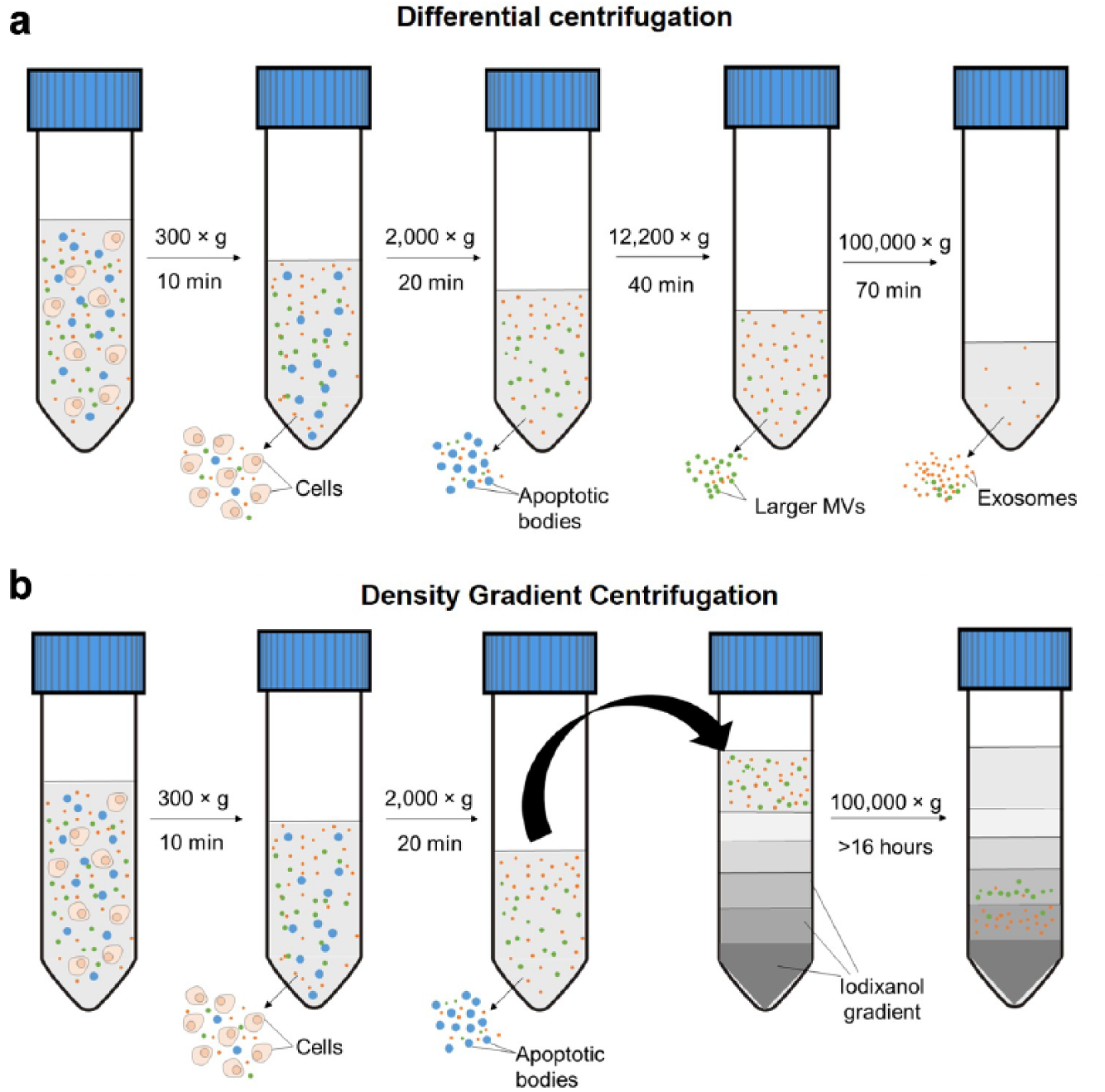
Schematic illustration for centrifugation techniques to obtain submicron particles. (
An alternative approach to isolate MVs is the use of density gradient centrifugation.
Figure 2b
illustrates the steps required for density gradient centrifugation. Briefly, EV mixtures could be added onto a discontinuous density gradient and ultracentrifuged at high speeds such that the particles may be fractionated according to their densities. For example, one protocol suggested the use of iodixanol gradients to separate vesicles from exosomes.
28
Compared with the former approach, density gradient centrifugation increases separation resolution significantly and removes the contaminating proteins. However, the procedures are lengthy and an extremely strong centrifugation force is required to overcome the viscous media. For example, centrifugation at 100,000
Size-Based Methods
Owing to the shortcomings of the above-mentioned approaches, researchers have embarked on the use of different principles to separate MVs. In recent years, size-based MV isolation technologies have been developed and have shown higher efficiency and effectiveness.
Centrifuge-on-a-Chip
In conventional centrifugation, MVs are required to migrate along the length of the Falcon tube or microtube, typically in centimeter scale. Considering the particle size and migration distance, sustained high forces are necessary to ensure pelleting of the MVs. Centrifugal microfluidics possess the obvious advantages of reducing the migration distance for the MVs. Furthermore, centrifugal hydrodynamics has been demonstrated to be effective in applying centrifugal pressure to manipulate liquids to move sample volumes within different compartments of the microfluidic channel. In our lab, we combined both advantages within a single microfluidic chip, which has been shown to be effective in obtaining MVs and exosomes in different outlets. 29 In fact, a gentler centrifugal process may be applied for shorter durations, ensuring the high viability of the exosomes for downstream processing. Figure 3a shows the setup of the centrifugal microfluidic chip to obtain different sized particles in separate outlets.
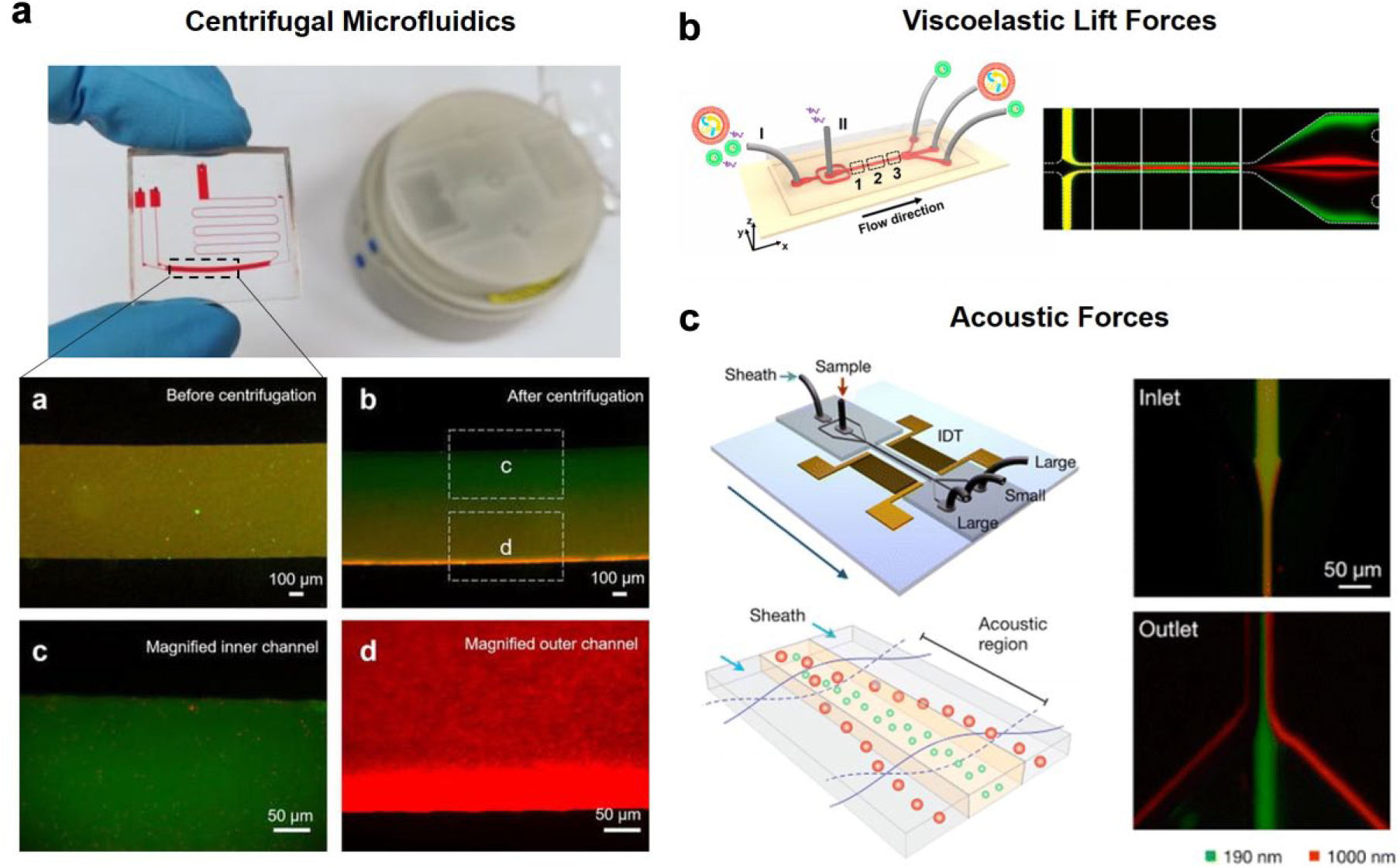
Microfluidics for larger MV enrichment. (
Hydrodynamics
Besides gravitational force, other microhydrodynamic forces may be applied to these nanosized bodies. For example, viscoelastic lift forces can be realized through the addition of poly(oxyethylene) (PEO) solution—a viscoelastic medium. In Poiseuille flow, elastic lift forces control the lateral position of submicron particles within the microfluidic channel. A collaborative research group at the University of Chinese Academy of Sciences experimented with this concept and demonstrated the possibility of separating MVs from exosomes ( Fig. 3b ). 30
External Fields
Apart from hydrodynamic fluid manipulation, externally applied fields using electrophoresis, magnetics, 31 and others 32 have been demonstrated for submicron particles. For example, the use of acoustic forces has been demonstrated in Lee’s group. Figure 3c illustrates the acoustic principle of fractionating submicrometer MVs according to their size. Briefly, interdigitated transducer (IDT) electrodes were patterned on the microfluidic chip to generate surface acoustic waves across the microchannel. Particles passing through the microchannel experienced an ultrasonic radiation energy, resulting in a deflection from their intended path. Importantly, the migration from their initial path was mainly dependent on their size. As such, MVs are directed to the sides of channels, while exosomes stay in the middle of the channel. 33
Exosomes Purification and Enrichment Technology
Ultracentrifugation
Differential ultracentrifugation or density gradient ultracentrifugation is the current gold standard to enrich and purify exosomes. The differential ultracentrifugation protocol was established by Théry et al.
34
in 2006. In this well-established protocol, exosomes were sedimented by ultracentrifugation at 100,000
Label-Based Methods
Surface markers that are frequently used for exosome isolation include but are not limited to CD9, CD63, CD81, and Ep-CAM.24,36–38 The reliability of these markers is further discussed in the Discussion section. The antibody of these markers is usually coated on beads39–44 or functionalized on specific features of the channel ( Fig. 4 ).45–47 When the antibody is functionalized on channel features, a wavy channel surface is generally employed to increase the capture efficiency. To do so, Wang et al. 46 placed pillars at the bottom and Kang et al. 45 combined two layers of extruded patterns. In such a design, it would be convenient to conduct on-chip imaging-based quantification of immobilized exosomes. If the antibody is instead coated on beads, either magnetic39–42,44 or not, 43 exosomes are supposed to be captured when mixed with the beads. Next, the beads could carry enriched exosomes to downstream analysis such as on-chip enzyme-linked immunosorbent assay (ELISA)39,43 or lysis for exosomal proteins and nucleic acid detection.40–42,44
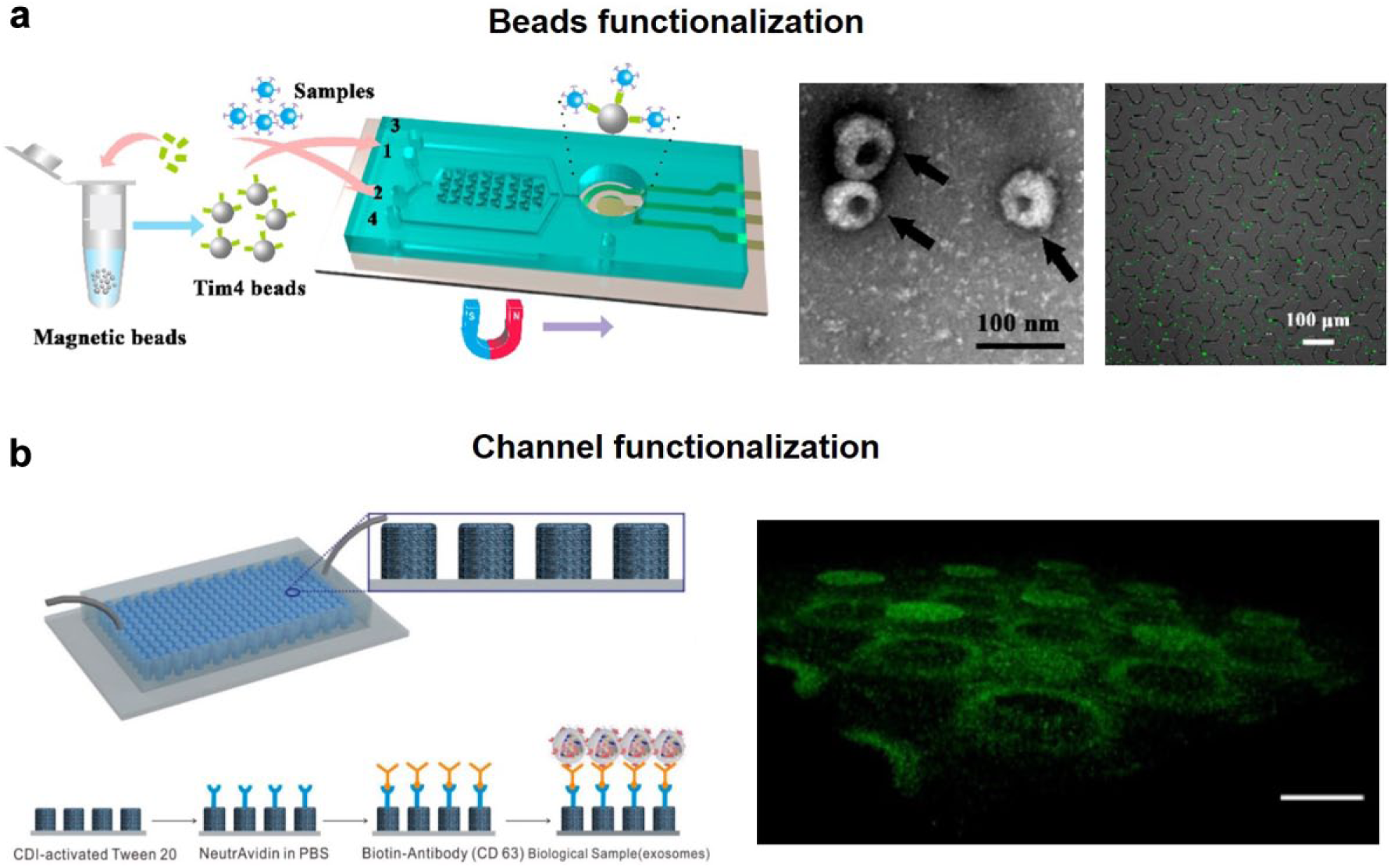
Label-based exosome isolation platforms. (
Size-Based Methods
The widely accepted size range of exosomes is 50–150 nm, which distinguishes them from larger MVs and smaller molecules. Different size-based principles including filtration, deterministic lateral displacement (DLD), and asymmetric flow field-flow fractionation (AF4) have been applied to exosome purification.
Filtration
In order to obtain a highly purified exosome sample from either cell culture media or human body liquid by filtration, membranes with two different cutoff sizes are generally implemented. One membrane with a larger pore size of around 150 nm can segregate large contaminates when the exosome can pass through the membrane. Another membrane with a smaller cutoff size of around 50 nm is needed to collect exosomes while allowing the contaminating soluble proteins and nucleic acids to be washed away. Woo et al. 48 implemented the 600 and 20 nm filtration membranes into a centrifuge-on-chip device. In this device, centrifugal force drove the loaded sample through two filtration membranes connected by channels ( Fig. 5a ). Similarly, Liang et al. 49 embedded 200 and 30 nm filtration membranes on two wells and flowed sample through the membranes to purify exosomes. Liu et al. 50 used a series of double-filtration devices and fractioned exosomes into 30–50, 50–80, 80–100, and 100–200 nm portions. Quantification of exosomes in these segregations showed different size distributions of exosomes from different cell lines.
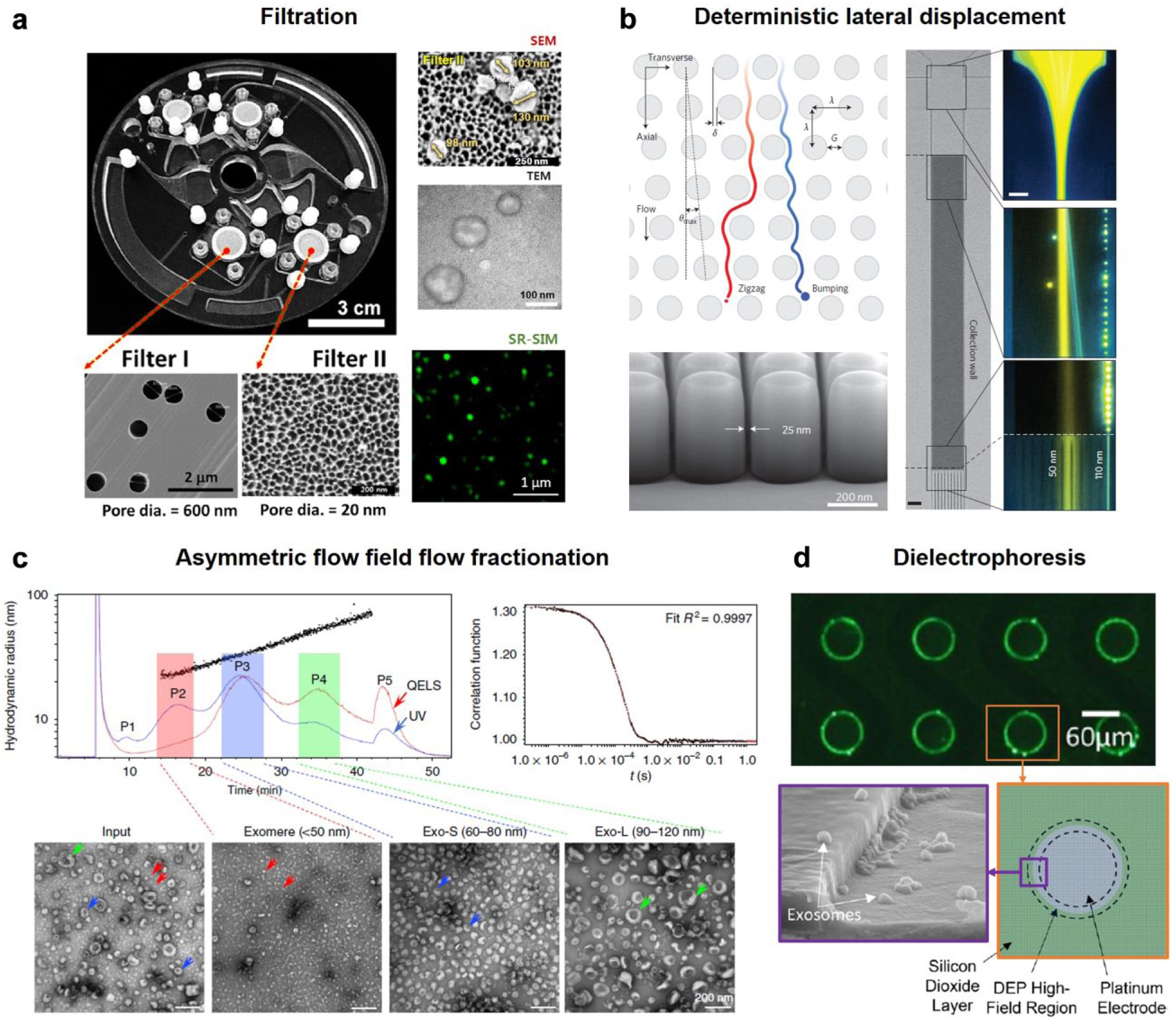
Size-based exosome isolation platforms. (
Deterministic Lateral Displacement
DLD pillar arrays were first reported in 2004. 51 The gap distance of pillars within rows and the displacement of pillars between rows determine a cutoff size. When passing through the pillar arrays, large particles are bumped to one side of the channel while small particles remain in the original path or trajectory. Wunsch et al. 52 managed to fabricate pillar arrays with a gap distance down to 25 nm. As such, ~110 nm particles can be bumped and isolated from <50 nm particles ( Fig. 5b ).
Asymmetric Flow Field-Flow Fractionation
In a typical AF4 system, particles are first focused to one side of the channel wall by external fields. Due to the difference in size, particles will stay at different distances from the wall. And because of the parabolic velocity profile of flow in the channel, smaller particles that are farther from the side wall will experience higher transport velocity and thus be eluted out earlier than larger particles. In this way, targeted particles can be isolated and collected at specific time sections. Zhang et al. 53 applied the AF4 method to successfully fractionate exosomes from tissue culture medium. As a result, exosomes are found to have a bimodal size distribution with one peak at 60–80 nm and another at 90–120 nm, implying the existence of an exosome subpopulation ( Fig. 5c ).
Dielectrophoresis
Similar to dielectric particles, cells and EVs can be manipulated by a polarization force produced by a nonuniform electric field. The intensity of the force is dependent on particle size, particle effective polarizability, electric field intensity gradient, and medium permittivity. Although the force is determined by multiple factors, dielectrophoresis (DEP) is usually used to sort the particles by size in an inhomogeneous electric field. DEP has been demonstrated to be able to enrich exosomes.54,55 In their design, multilayer disk-like electrodes were arranged at the bottom of the channel. These electrodes generated high-field regions that could attract exosomes when 15 kHz of alternative voltage was applied with a voltage of 10 Vpp ( Fig. 5d ).
Charge-Based Methods
As exosomes are enveloped by cellular membrane, it is not surprising that exosomes show strong negative zeta-potential 56 at physiological pH. Therefore, exosomes could possibly be manipulated by electrophoresis.
Electrophoresis
Electrophoresis means driving charged particles by simply applying an electric field. The speed of particles or molecules subject to electrophoresis is determined by electric field intensity, the charge of the particles or molecules, and drag forces the subject may experience when moving in media. Marczak et al. 57 have created an exosome electrophoresis microfluidic device. Exosomes were carried through a channel by flow and attracted to the anode, which was embedded in agarose gel. According to the results, an electric field with an intensity of 100 V/cm is enough to enrich exosomes, regardless of the purity.
Discussion
Specific Protein Marker for EVs?
Researchers have been seeking specific protein markers for EVs so that isolation and verification would be more straightforward. Unfortunately, no single protein has been demonstrated to be uniquely and ubiquitously present on EVs. For now, the reported markers for EVs include but are not limited to ESCRT (endosomal sorting complexes required for transport) system components (such as TSG101 and Alix), tetraspanin proteins (such as CD9, 58 CD63, 59 and CD8124), and some Rab small GTPase (such as Rab27a,60–62 Rab27b,60,61 and Rab35 63 ) because they have all been reported to participate in the biogenesis of EVs or are frequently enriched in EVs. In particularly, tetraspanins have been widely used for label-based EV isolation as they are surface proteins. However, tetraspanin marker-based isolation of EVs may be of concern because increasing evidence has shown that these markers may be poorer than expected in terms of both specificity and ubiquity.23,24,60,64
Insufficient Specificity to Distinguish EVs
Tetraspanins are present not only on exosomes but also on other EVs, like MVs and apoptotic bodies. Bobrie et al. 60 claimed that CD9 was detected in vesicles larger than 150 nm in diameter and heavier than 1.20 g/mL. CD9 was shown to be positive in MVs isolated by ultracentrifugation, and its level in vesicles was not affected by RAB27a silencing. Crescitelli et al. 23 detected the presence of CD9, CD63, and CD81 by flow cytometry. The three tetraspanins were demonstrated to be present in all EVs, including exosomes, MVs, and apoptotic bodies ( Fig. 6a ). Therefore, tetraspanins were insufficient to distinguish the different groups of EVs.
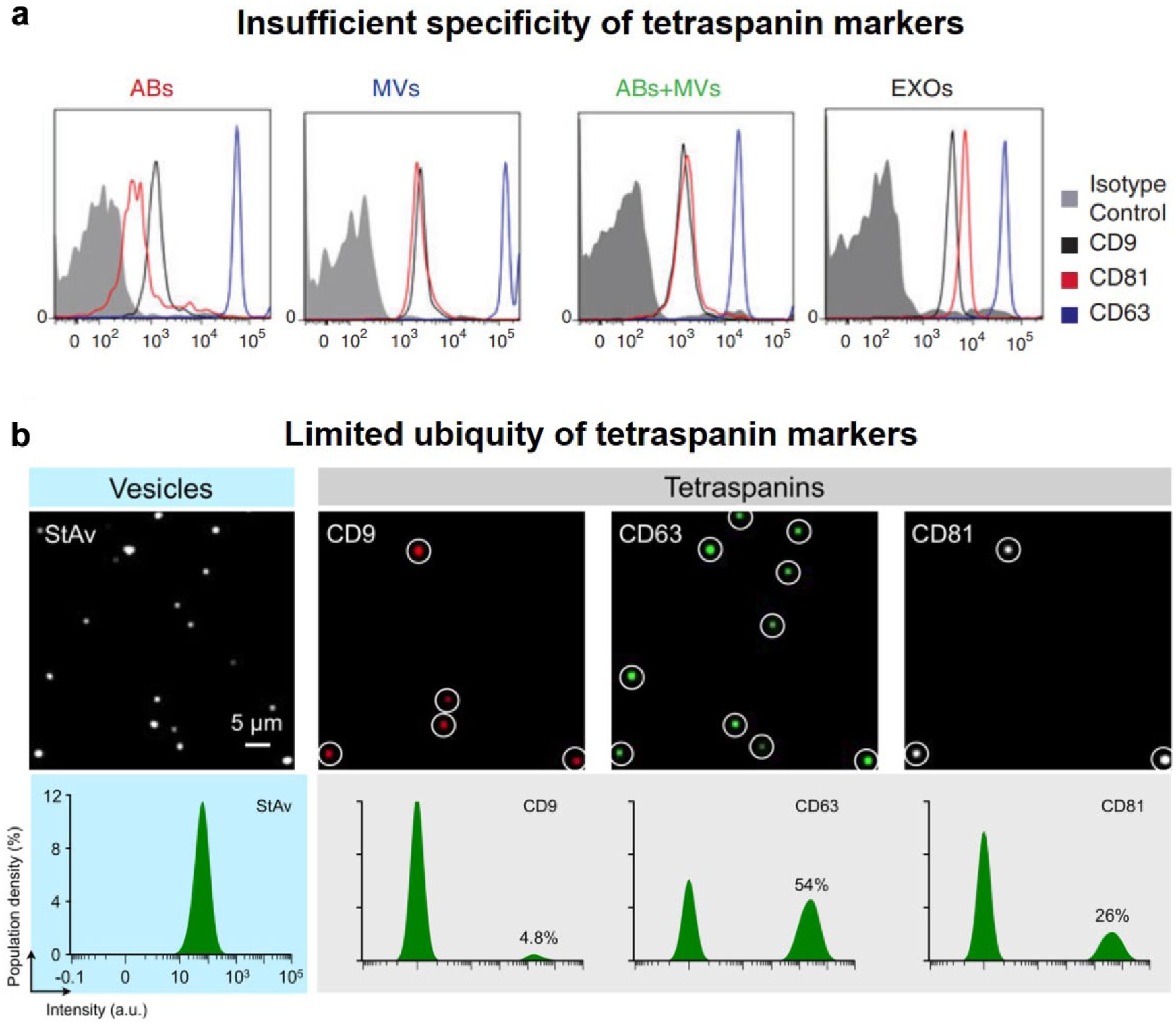
Limited specificity and ubiquity of tetraspanin as EV markers. (
Limited Ubiquity within EVs
Although it is not clear yet how many subpopulations of exosomes exist, it is believed that exosomes are heterogeneous. Lee et al. 64 managed to stain EVs after immobilization; thus, different surface markers on single EVs could be detected by imaging. Among 800 single vesicles, CD9, CD63, and CD81 were present on 4.8%, 54%, and 26% of vesicles, respectively ( Fig. 6b ). In other words, tetraspanins are not universally present on EVs. Consequently, the great portion of EVs that do not carry the tetraspanin markers may be systematically missed if EVs are isolated by immunoaffinity. Other proteomic studies also reported that EVs isolated by CD9, CD63, and CD81, respectively, would present different proteomic profiles, implying that each of them was representing only a subpopulation of EVs. 24 In conclusion, tetraspanins only identify a part of EVs. Caution must be taken to consider tetraspanins as general EV markers.
Rising Concern of Lipoprotein Contamination
Although much effort has been devoted to analyze the EVs from patient blood to identify disease biomarkers, those results might be biased as lipoprotein contamination has not been inspected until recent years.
The concern for lipoprotein contamination is reasonable because the amount of lipoprotein particles (1012–1015/mL)65,66 is overwhelming compared with the typical EV quantity (107–109/mL) 67 in healthy individuals. In fact, different groups of lipoprotein could be co-isolated with EVs using different isolation technologies. For example, high-density lipoprotein (HDL) occupies a similar density range to EVs and may contaminate an EV sample obtained from density gradient ultracentrifugation. 68 Transmission electron microscopy (TEM) imaging revealed that in a 1.063–1.21 g/mL fraction the ratio of HDL to the EV quantity was 100:1. Apo A-I was also found to be enriched in this fraction. In addition to HDL, low-density lipoprotein (LDL), very low-density lipoprotein (VLDL), and chylomicrons (CMs) have size ranges that overlap with EVs. LDL contamination in EVs has actually been observed and evaluated. 69 The measured particle concentration within the EV size range was affected by food consumption. Lipoprotein markers like Apo B were also detected and shown to be predominant compared with EV markers. All this evidence indicated that lipoproteins can be abundant in the EV sample separated from serum by either density-based-only or size-based-only methods. And because lipoproteins have a membrane-bound structure, it can be hard to distinguish them from EVs when imaged using TEM. 70 From the cryo-TEM image provided by Yuana et al., it can be observed that EVs have various morphologies and some frequent lipoprotein-like vesicles show similar a size and structure to EVs.
The presence of lipoprotein not only biases the quantification of EVs but may also complicate the proteomic and nucleic acid analysis of EVs. Lipoproteomic studies have identified lists of proteins that were carried by HDL71–73 and LDL/VLDL 74 additionally to lipoprotein markers. As for nuclei acid, microRNA (miRNA) has been popular to study in the EV research field. 75 However, Vickers et al. demonstrated that HDL delivered abundant miRNA to recipient cells. A more comprehensive review of lipoprotein miRNA can be found in Michell and Vickers. 76 Exosomal mRNA 77 quantification may be relatively less threatened by lipoprotein contamination as mRNA is not reported to be with lipoproteins yet.
Consequently, the lipoprotein contamination can be a severe issue in blood EV studies. The abundant and dynamic lipoproteins that carry proteins and miRNA can significantly deviate the EV analysis result and thus potentially lead to wrong disease biomarkers. Isolating EVs from lipoproteins requires a combination of multiple isolation factors (e.g., size and density). Also, good resolution of the separation is necessary because the density or size of the lipoproteins can be very close to that of EVs.
Cumulatively, EVs deliver diverse molecules to recipient cells and facilitate important changes in these cells. However, due to the small size and the existence of subtypes, acquiring pure and enriched EVs has been technically difficult, weakening the consistency of EV downstream analysis. In this article, we reviewed the advances in technologies for EV enrichment and purification. Most of the approaches separate MVs or exosomes by immunoaffinity or size, and various principles have been applied. These new platforms provided (1) high resolution to distinguish subtypes of EVs, (2) a simple and fast procedure, or (3) an integrated system that combines downstream analysis.
Despite the progress in EV purification and enrichment technologies, the obtained EVs may still be subject to limited purity and impartiality due to the issues discussed above. As a result, EV studies are likely to have biased and inconsistent results, impeding our understanding of EVs in biological activity and applications of EVs in disease diagnosis and therapeutics. Thus, new EV isolation technologies that do not rely on EV markers and can overcome the lipoprotein contamination are strongly expected.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
