Abstract
Label-free in vitro potency assays are an emerging field in drug discovery to enable more physiological conditions, to improve the readout quality, and to save time. For this approach mass spectrometry (MS) is a powerful technology to directly follow physiological processes. The speed of this methodology, however, was for a long time not compatible with chemiluminescence- or fluorescence-based assays. Recent advances in matrix-assisted laser desorption/ionization (MALDI) instrumentation paved the way for high-throughput MS analysis of label-free assays for large compound libraries, whereas electrospray ionization (ESI)-based mass spectrometers equipped with RapidFire autosamplers were limited to medium throughput. Here we present a technological advancement of the RapidFire device to enable cycle times of 2.5 s per sample. This newly developed BLAZE-mode substantially boosted the ESI-MS analysis speed, providing an alternative technology for label-free high-throughput screening.
Introduction
Efficient drug discovery requires the identification of potent hits from large compound libraries in a reasonable time frame with low false discovery rates. Prerequisites are predictive assays with high-throughput readouts to determine the potency of the compounds. Label-based assays built on chemiluminescent or fluorescent detection provide a very high throughput. However, test substances can interfere with the readout, or the labeled assay substrate may alter enzyme kinetics. 1
In recent years, mass spectrometry (MS) has become competitive in terms of speed while providing the potential for more physiologically relevant assays. This technology allows the direct detection of analytes without having a tracer.
Matrix-assisted laser desorption/ionization (MALDI) MS was introduced recently, as a powerful and fast tool (<1 s per sample) to perform high-throughput analysis. 2 Combined with advances in automated preparation of MALDI plates, this becomes interesting for high-throughput screening (HTS). 3 For protein and peptide analytes, MALDI-MS is a particularly sensitive analyzer, as previously demonstrated for kinases,4,5 histone demethylases, 2 phosphatases, 6 histone lysine methyltransferases, 7 and the ubiquitin system,8,9 whereas electrospray ionization (ESI) MS is commonly used for small molecules and offers a versatile technology to detect a broad range of physicochemical diverse substances. However, the HTS-compatible supply of samples to the ESI-MS is still a technological challenge. The Agilent RapidFire platform is a very fast autosampler, which includes desalting and enrichment of the sample on a cartridge ( Fig. 1A ). The analysis with this setup usually takes 7–13 s per sample and is widely used in drug discovery for the profiling of compounds toward their ADME (absorption, distribution, metabolism, excretion) properties.10–12 The technology is also applied to the profiling of drug potencies with label-free assays.13–19 In the classical RapidFire setting, the sample becomes aspirated onto a solid-phase extraction (SPE) cartridge. This allows the enrichment and washing of the sample, which improves sensitivity and peak shape. Subsequently, the analytes get eluted from the cartridge with a back-flush. This step is very powerful to concentrate the analytes and submit the sample to the mass analyzer. 10 However, as outlined before, this setup is time-consuming, limiting the speed of this technology, and vice versa its applicability for HTS.
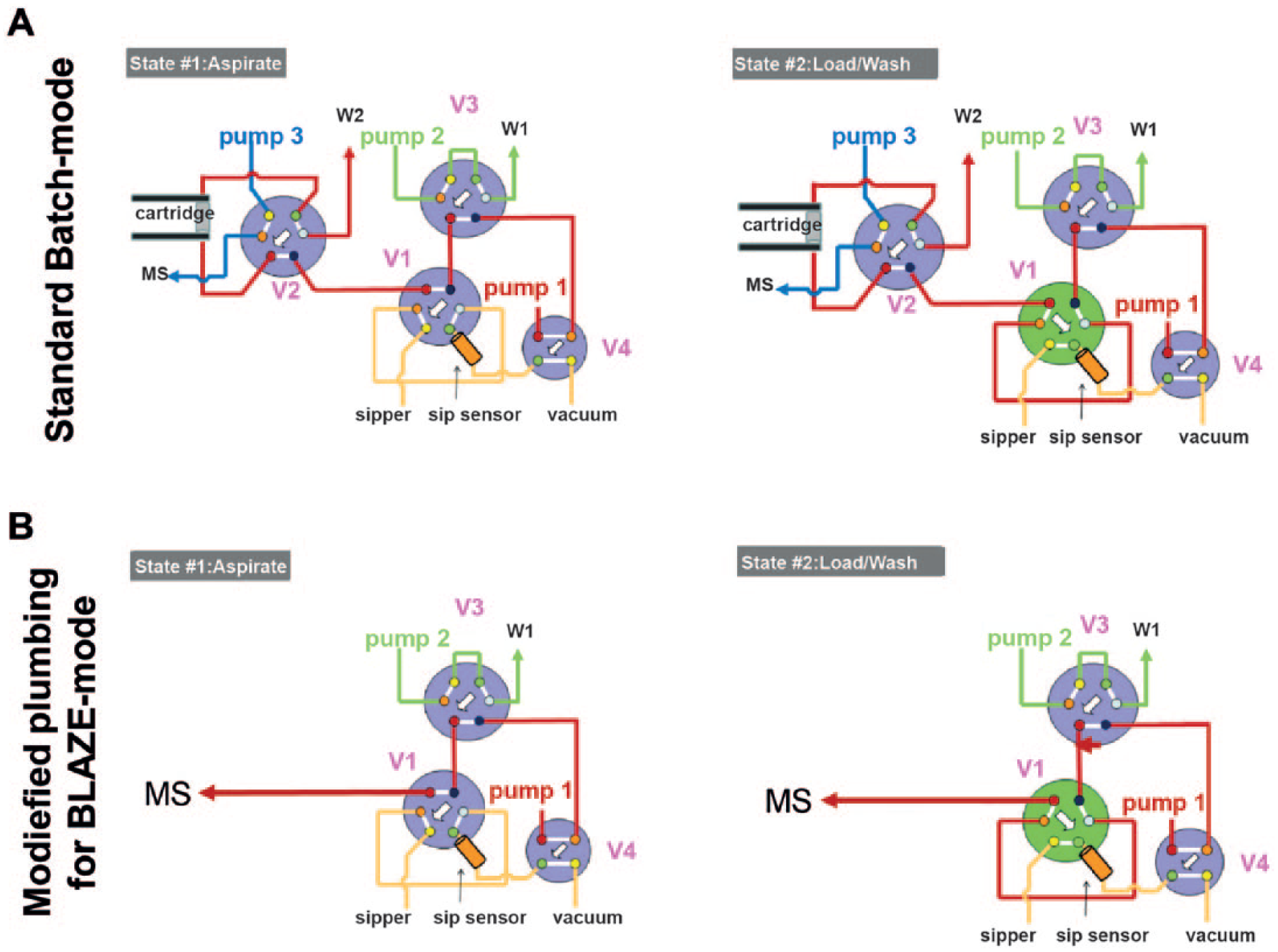
Plumbing of the RapidFire valves in (
Here, we present a new mode for the RapidFire hardware, which allows cycle times of 2.5 s per sample. We demonstrate the feasibility of this setting using an assay for the detection of trimethylamine (TMA) originating from a bacterial incubation with choline. TMA was recently discovered as a xenobiotic, which is generated by the gut microbiome.
20
TMA becomes oxidized to trimethylamine-
In this report, we describe the technological adaptions to the RapidFire instrument to enable HTS for the identification of bacterial inhibitors of the TMA production ( Fig. 1 ). This advancement transforms every RapidFire-MS platform into a fast cartridge-free system for direct sample injection. This novel BLAZE-mode boosts ESI-MS toward a widely applicable, HTS-compatible readout for label-free assays within drug discovery campaigns, but also for other research areas requiring ultrafast MS-based detection.
Materials and Methods
All used solvents chemicals and standards were purchased from Sigma-Aldrich (Munich, Germany). Reaction tubes and plates were from Eppendorf (Hamburg, Germany).
RapidFire Settings
An API 6500 mass spectrometer (ABSciex, Toronto, Canada) was equipped with a binary pump (Agilent 1290, Santa Clara, CA), a plate hotel (made in-house), and a RapidFire sample injector (Agilent). This setup was controlled by an in-house developed LabView-based master software. Samples were aspirated from 384-well plates by negative pressure at −50 kPa into a 10 µL sample loop.
Chromatograms were recorded by Analyst 1.6.2 (ABSciex) and DiscoveryQuant 2.3 (ABSciex).
The Turbo V ion source conditions were set to ion spray voltage = 4200 V, gas 1 = 65 psi, gas 2 = 80 psi, temperature = 550 °C, curtain gas = 35 psi, and collision gas = medium.
The mass spectrometer was operating in positive mode recording the following multiple reaction monitoring (MRM) transitions: derivatized d9-TMA 183.1/127.3 and derivatized internal standard 13C3, 15 N-TMA 178.2/122.0 both at a DP of 46 V and CE of 20 V. For nonderivatized d9-TMA, settings of 69.0/49.0 at a DP of 40 V and CE of 25 V and 13C3, 15 N-TMA 64.0/64.0 at a DP of 40 V and CE of 10 V were used.
Batch-Mode
After aspiration of the sample for 250 ms into a 10 µL injection loop, valve 1 was actuated, flushing the sample onto a H5 12 µL HILIC cartridge (Optimize Technologies, Oregon City, OR). After a 4 s wash, valve 2 was actuated, resulting in a back-flush of the analytes to the MS detector for an additional 4 s, after which both valves were actuated to the home position and the cartridge was equilibrated for an additional 0.5 s prior to the aspiration of the next sample.
Solvent A (wash solvent) was 70/30 isopropanol/hexane and solvent B (elution solvent) was methanol supplemented with 0.1% formic acid. Pump 1, delivering solvent A, was set to a flow rate of 1.1 mL/min, while pumps 2 and 3, delivering solvent B, were set to flow rates of 1.25 and 0.5 mL/min, respectively.
The sipper capillary tube was washed between the injections at the wash port, once with water and once with methanol.
BLAZE-Mode
Some basic modifications were made to the RapidFire system to enable BLAZE-mode direct injection. Specifically, the tubing connecting valves 1 and 2 was disconnected and replaced with a direct connection to the mass spectrometer ( Fig. 1B ). The RapidFire control software was modified within the configuration software such that valves 2 and 3 were not actuated by setting states 3 and 4 to [0, 0, 0, 0]. The settings for states 1 and 2 were unchanged and the valve timings could be adjusted through the normal user interface. A BLAZE-mode configuration file can be saved to quickly switch between normal and direct injection modes.
For the TMA-based assay, solvent A consisting of 0.1% trifluororacetic acid (TFA) in water was used, which was delivered by pump 1 at a flow rate of 2 mL/min. For the ATX assay, a solvent composition of 40% acetonitrile in water, supplemented with 10 mM ammonium acetate, was used.
Even though pumps 2 and 3 are not used in BLAZE-mode, the RapidFire system requires all 3 pumps to be running, in order to avoid an error message by the instrument. BLAZE-mode operation requires pumps 2 and 3 to be turned on, but the pumps were set to minimal flow rates (10 µL/min) and diverted to waste by opening the purge valves.
For BLAZE-mode operation, sample was aspirated into the 10 µL injection loop of valve 1 for 250 ms, after which the valve was actuated, delivering the sample to the mass spectrometer for an additional 300 ms. States 3 and 4 (used for elution and reequilibration in conventional RapidFire operation) were set to 0 ms and were unused in direct injection analysis. BLAZE-mode enables sample-to-sample injection times below 3 s per sample, increasing the overall throughput of the RapidFire system by three- to fivefold compared with conventional operation when the SPE cartridge is used, enabling a 384-well plate to be analyzed in 16 min.
TMA Assay
d9-TMA was generated from a bacterial incubation of
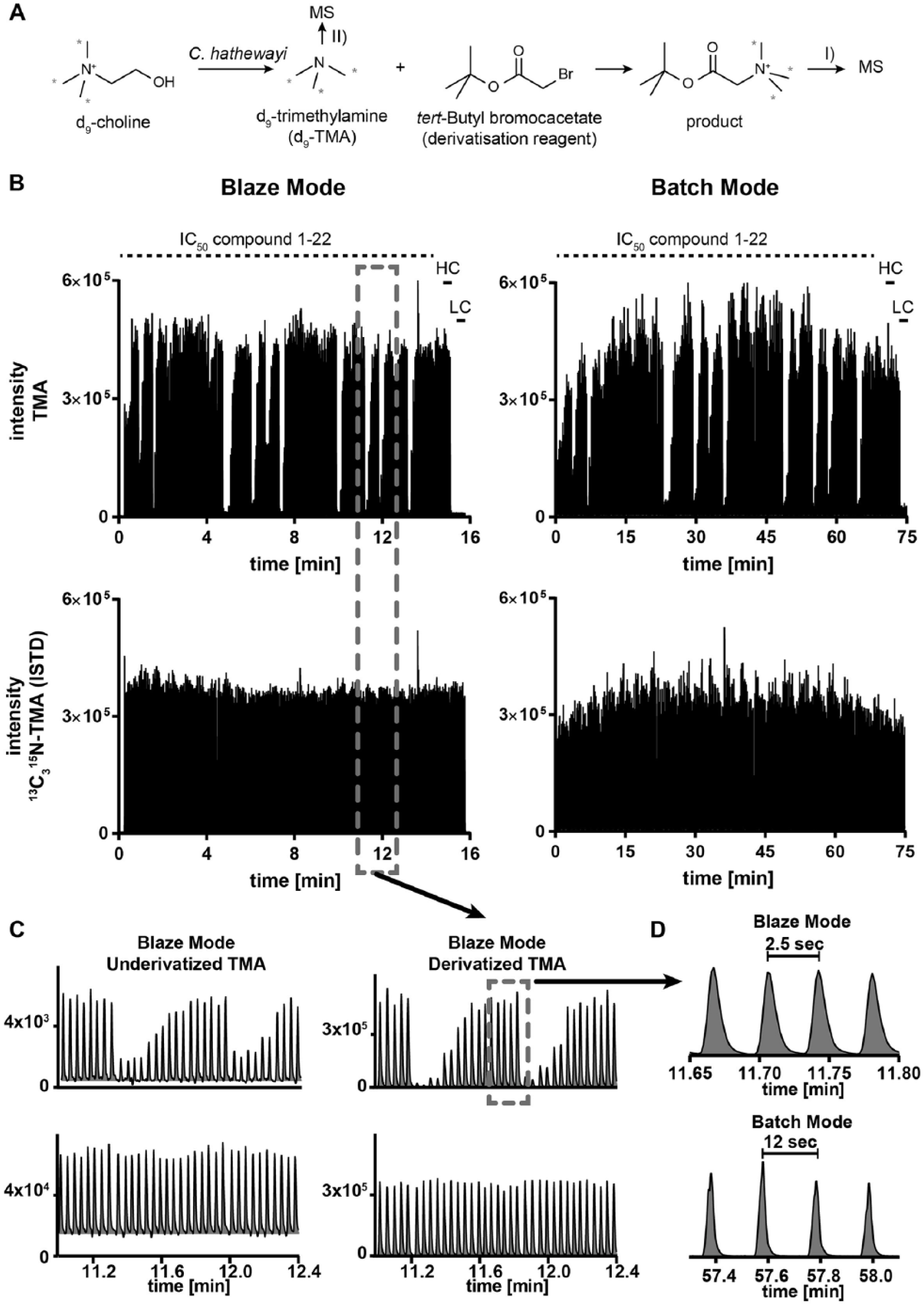
(
For the analysis of nonderivatized d9-TMA, the same protocol was applied without the addition of the derivatization agent.
ATX Assay
The assay was performed as described previously. 13 The BLAZE-mode settings are described above.
Results and Discussions
BLAZE-Mode Technology
The overall throughput of a conventional RapidFire analysis is rate limited by the SPE washing and enrichment of the sample. High concentrations of salts and/or buffers commonly used in biochemical assays result in ESI ion suppression and source contamination, requiring the sample washing step. However, if compromises can be made in assay buffer selection to minimize concentrations of ESI-incompatible buffers, the SPE step may be eliminated. Alternatively, if the analyte concentration is sufficiently high, the sample can be diluted such that the interfering salt and buffer concentrations are lowered to a point where ESI ion suppression and source contamination become acceptable.
With the elimination of the SPE step in BLAZE-mode, the rate-limiting step becomes the autosampler cycle and ultimately the width of the analyte peaks in the MS. To minimize the autosampler cycle time, the sip height was set to the maximum possible level (typically 5 mm), while the safe-Z height was set to the minimum level, keeping motion in the Z axis as small as possible. This approach additionally helped to reduce carryover, as the needle only has minimal contact with the sample. The required sample volume is not impacted by this and has the same requirements as for the Batch-mode.
To further minimize the autosampler cycle time, the two-stage sipper wash between injections was eliminated in BLAZE-mode. In conventional RapidFire analysis, the sipper wash can take as long as 5 s but is performed simultaneously with the SPE step and does not add any time to the sample analysis. In the TMA assay, elimination of the sipper wash resulted in unacceptable carryover. The addition of 0.1% TFA to the mobile phase helped greatly reduce nonspecific binding, and a carryover of 1.2% could be achieved. In Batch-mode, we applied a hydrophilic interaction liquid chromatography (HILIC)-based SPE approach with 0.1% formic acid in methanol as solvent B. In BLAZE-mode, methanol resulted in broader peaks compared with the aqueous buffer. Nevertheless, the combination of the analyte and its solvent used in BLAZE-mode requires individual adaption, in order to achieve good peak shape, high sensitivity, and less carryover. We recommend starting with the composition of solvent B, which is usually used in Batch-mode.
Similar to BLAZE-mode, high-throughput MS approaches by MALDI do not incorporate any means of sample cleanup or fractionation and rely on compromises to be made in buffer selection and/or sample dilution for successful analyses. The RapidFire platform can be easily switched between conventional analysis using the SPE sample cleanup and high-throughput BLAZE-mode using direct injection. A hit identified in an HTS run in an assay buffer optimized for direct injection can be quickly followed up in a physiologically relevant assay buffer on the same instrument with the same mass spectrometer.
BLAZE-Mode versus Batch-Mode
In order to compare the classical RapidFire Batch-mode with the BLAZE-mode, we used a TMA-based assay for qualification (
Fig. 2A
). TMA is generated by gut microorganisms by metabolizing dietary choline. TMA is absorbed within the gut and becomes transformed into TMAO, which is involved in a variety of diseases, including arteriosclerosis and hyperglycemia. Targeting the TMA production is of interest for studying the microbiome–mammalian axis. We tested various inhibitors of TMA production and applied the BLAZE-mode as readout. TMA was derivatized with
For the test shown in Figure 2B , we injected samples from the same incubation head-to-head in Batch-mode and BLAZE-mode, respectively. For each mode, 10 µL of sample was injected. The graph is presenting the MS signal, which was recorded from a 384-well plate. The plate layout contains 22 potential inhibitors of the TMA production. Each compound was tested in eight different concentrations in duplicate to determine IC50 values. Moreover, 16 high controls (no test compound) and 16 low controls (no bacterial background) were measured to assess the carryover. In BLAZE-mode, a carryover of 1.2% was detected, which is comparable to the Batch-mode (0.8%).
For the signal of the internal standard, we achieved for derivatized TMA an average peak height of 3.8 × 105 counts in the BLAZE-mode and 3.8 × 105 counts in the Batch-mode. This indicates that the sensitivity of the BLAZE-mode is comparable to that of conventional RapidFire analysis for this assay. Notably, the coefficient of variation for the internal standard signal in the Batch-mode was 13.3%, whereas in BLAZE-mode it was 7.5%. In addition, the signal for the internal standard in the Batch-mode shows a recurring variability pattern (see Fig. 2B lower right panel). Nonetheless, this fluctuation appears in the signal of the analyte too, which becomes perfectly balanced by the internal standard (see peak area ratio between analyte and internal standard in Suppl. Fig. S1 ). In BLAZE-mode, these variations were not observed, demonstrating the robustness of this newly developed methodological setup. The IC50 values determined by Batch-mode were compared with the BLAZE-mode approach and showed an excellent correlation ( Fig. 3A ).
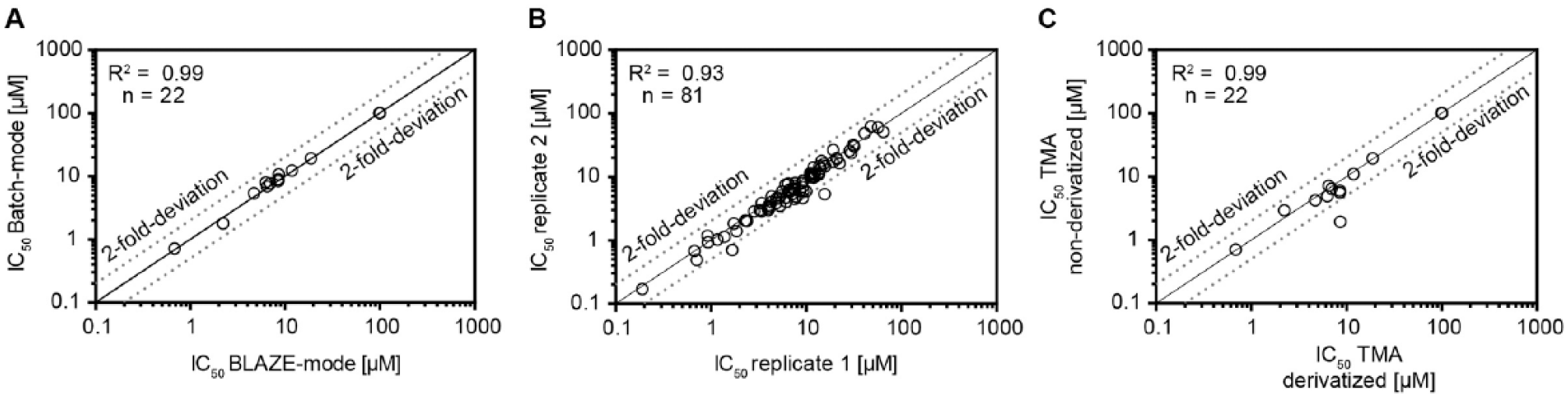
Comparability of assay results. (
To assess the reproducibility of the TMA assay with BLAZE-mode readout, we performed two independent runs with 81 test compounds and compared their IC50 values. The correlation of both runs is shown in
Figure 3B
. The
In BLAZE-mode, less method development effort was required. In particular, the development of the enrichment step, including cartridge selection, solvent, and method optimization, becomes dispensable. In addition, for very small molecules like TMA, derivatization becomes beneficial to enable retention on the cartridge. As this step is skipped in BLAZE-mode, we were interested in seeing whether TMA detection without derivatization would give reasonable results.
TMA Detection without Derivatization
To study the BLAZE-mode for the applicability of measuring nonderivatized TMA, we took an aliquot of the samples before derivatization. A head-to-head comparison of the signals between derivatized and nonderivatized TMA is shown in
Figure 2C
. We observed an increase in the baseline and a decrease in peak intensity for both the analyte and the internal standard. Nevertheless, the assay window is still apparent and allowed us to determine IC50 values. We compared the IC50 values of a set of 22 compounds between both experimental setups. For both the nonderivatized and derivatized measurements, we determined comparable values, as shown in
Figure 3C
. The correlation between both runs correlated with an
BLAZE-Mode for Screening Purposes
Running a 384-well plate in Batch-mode took 77 min, whereas in BLAZE-mode the samples were analyzed in 16 min. The cycle time from sample to sample in Batch-mode was 12 s and in BLAZE-mode 2.5 s, as shown in Figure 2D . The peak width at half maximum was increased in the BLAZE-mode from 0.6 to 1.5 s compared with Batch-mode. This is a result of the omitted enrichment step onto the cartridge, which results in peak broadening. Nevertheless, in BLAZE-mode full baseline separation of the peaks was achieved, allowing accurate baseline integration.
Subsequently, we aimed to apply the novel BLAZE-mode technology to our previously published assay for the screening of autotaxin (ATX) inhibitors by tracking lysophosphatidic acid (LPA) generation.
13
Cycle time in Batch-mode required 13 s per sample being significantly reduced to 2.5 s per sample in the established BLAZE-mode (see
Suppl. Fig. S2
). We analyzed 29 individual compounds in both Batch-mode and BLAZE-mode and determined comparable (
The increased speed for both assays by approximately a factor of 5 with the BLAZE-mode facilitates the application to run a full-deck screen with an ESI-based MS system in a reasonable time frame. For instance, a typical drug discovery screen with 500,000 compounds at single concentration takes approximately 2 weeks of measurement in BLAZE-mode, whereas in Batch-mode this would require more than 2 months.
The BLAZE-mode developed here is a simple modification of existing instruments and can immediately be applied in all labs operating RapidFire systems without further investment. The engineering of the RapidFire toward a high-throughput reader accelerated the speed for the shown assay examples by a factor of 5, hence decreasing cycle times and ultimately increasing efficiency.
We demonstrated that the data quality, based on sensitivity, peak shape, carryover, and reproducibility of IC50 values in BLAZE-mode, is comparable to that of the Batch-mode. The observed fluctuations in the signal intensities in the Batch-mode indicate suboptimal chromatographic conditions. In contrast, this issue was avoided in BLAZE-mode by bypassing the SPE cartridge. Consequently, the setup of BLAZE-mode-based assays is assumed to require less optimization steps, such as retaining small analytes on the SPE cartridge, and less consumables, such as cartridges. Therefore, the BLAZE-mode is very attractive for the integration into screening environments. Although the speed of the BLAZE-mode is typically slower than that of MALDI-based assays,2,4–9 it is especially attractive for low-molecular-mass analytes, which are typically more difficult to detect by MALDI due to matrix interferences and sensitivity limitations. The power of RapidFire-MS in this domain has been demonstrated with a variety of analytes.13–19 Besides the application for label-free screenings, the BLAZE-mode can also boost the throughput for the in vitro ADME profiling of drug candidates (e.g., permeability and microsomal stability assay).10–12 Therefore, the BLAZE-mode provides a versatile tool along the drug discovery value chain and overcomes the throughput limitations of conventional ESI-based MS platforms.
With this report we present a label-free high-throughput assay for the identification of inhibitors of TMA production from large compound libraries to study the TMA-TMAO axis in the host–microbiome interaction. Furthermore, we applied this technology for the detection of LPA, which is one of our recently published RapidFire-based assays. 13 Applying the BLAZE-mode shortened the analysis of a 384-well plate from the previous 77 min to 16 min.
Assay buffer components (e.g., salts, detergents, and media components) can reduce ESI-MS efficiency, which are typically removed with the cartridge-based Batch-mode approach. 25 For BLAZE-mode, we applied a preprocessing of the samples, which is dilution for the TMA-based assay and liquid–liquid extraction for the ATX assay, respectively. Both setups are readily automatable, and we demonstrated that this approach is a simple way to achieve a sensitive and robust BLAZE-mode analysis. Even samples with higher matrix complexity (e.g., plasma) may benefit from this procedure to be compatible with BLAZE-mode. Nevertheless, this procedure requires individual adaption to the respective analyte and assay composition.
Further optimization of the BLAZE-mode speed can be achieved by truncating the sipper capillary to shorten the way to the sample loop. Additional acceleration can be achieved by using a smaller injection loop, resulting in a sharper peak at the expense of some signal intensity. However, the ability to quickly change between high-throughput BLAZE-mode direct injection of samples in an optimized buffer system and conventional RapidFire with SPE cleanup, enabling analysis of samples in physiologically relevant buffers on the same platform using identical MS settings, is the primary benefit of this technology.
Further developments in fast supply of samples to ESI-based MS systems are on the way by applying acoustic dispensing. Comparable to the BLAZE-mode, this concept directly injects the sample into the MS, without having a desalting/enrichment step. The high frequency of acoustic droplet ejection systems allows high-speed analysis. Acoustic-mist ionization MS26,27 and open-port probe MS 28 were recently presented as potential future “autosamplers” for the delivery of samples to the MS within less than 1 s. Acoustic-mist MS uses a transfer line to submit droplets produced by an ECHO acoustic dispenser to the MS. Although this setup is faster (3 samples per second) than BLAZE-mode, so far it does not reach classical ESI-MS sensitivity and is quite sensitive to matrix effects. 27 Moreover, currently these acoustic-MS devices are prototypes and not yet commercially available. Therefore, the customer-based adaption of RapidFire instruments toward the BLAZE-mode technology is the fastest sample delivering system for ESI-based MS, which is readily available.
Supplemental Material
2018-10-22_RapidFire_BLAZE_ready_for_submission_SI – Supplemental material for RapidFire BLAZE-Mode Is Boosting ESI-MS Toward High-Throughput-Screening
Supplemental material, 2018-10-22_RapidFire_BLAZE_ready_for_submission_SI for RapidFire BLAZE-Mode Is Boosting ESI-MS Toward High-Throughput-Screening by Tom Bretschneider, Can Ozbal, Markus Holstein, Martin Winter, Frank H. Buettner, Sven Thamm, Daniel Bischoff and Andreas H. Luippold in SLAS Technology
Footnotes
Acknowledgements
We thank Danielle Fowler, Christina Barlett, Ralp Binetti, and John Broadwater for setting up the bacterial TMA assay; Daniel Grabowski and Sarah Chandler for delivering samples from the bacterial TMA assay; Jörg T. Kley for providing test substances for the bacterial TMA assay; and Gerald Roth and Hannes Koolman for providing test substances for the ATX assay.
Supplemental material is available online with this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
