Abstract
Radiosynthesis of [1-11C]acetate is well described in literature, but all syntheses either require adaptations in complex commercial synthesizers or rely on closed-source hardware and software control. Arduino microcontrollers are ideal for the compact, flexible, and inexpensive control of low-complexity hardware, making them particularly suited for radiochemistry where operation in space-limited shielded hot cells is mandatory. We established a [1-11C]acetate radiosynthesis module for combination with a [11C]MeI module available in almost every lab working with 11C. Its small footprint even enables back-to-back production in a hot cell already occupied by other modules. Using this setup, we achieved a reliable and flexible supply of this tracer, with radiochemical yields of 51.4 ± 28.2% and radiochemical purities (RCPs) of 94.4 ± 6.7% (n = 9) in a synthesis time of 15 minutes. Positron emission tomography (PET) and biodistribution analysis demonstrated low background uptake in healthy mice, with highest uptake in liver and kidneys. Arduino microcontrollers have become valuable and versatile tools in our lab for the automatization of low-complexity procedures not requiring full-blown commercial radiochemistry synthesizers, as showcased here for the production of [1-11C]acetate.
Introduction
[11C]Acetate has received strong and enduring attention as a metabolic positron emission tomography (PET) tracer for preclinical and clinical application due to its complementarity to [18F]fluorodeoxyglucose (FDG). While FDG uptake reflects glucose consumption and glycolytic activity, acetate can be either oxidatively catabolized to CO2 in the tricarboxylic acid cycle or used anabolically for the biosynthesis of, for example, ketone bodies and fatty acids. Both catabolic and anabolic roles have been utilized for PET imaging, 1 mainly in oncology2,3 and cardiology.4–7
The broad interest in [11C]acetate as a PET tracer is reflected in the literature by a multitude of radiosynthesis and purification strategies,8–22 each adapted to the specific infrastructure of the individual production laboratory. The synthesis of [1-11C]acetate is accomplished by the reaction of cyclotron-produced [11C]CO2 with a Grignard reagent (MeMgCl or MeMgBr), either in solution or on the surface of a coated loop. The product is then hydrolyzed by acidification (e.g., carrier acetic acid or ion exchange resin) and acetate is purified from educts and byproducts ([11C]bicarbonate, [11C]acetone, [11C]tert-butanol) using different strategies (
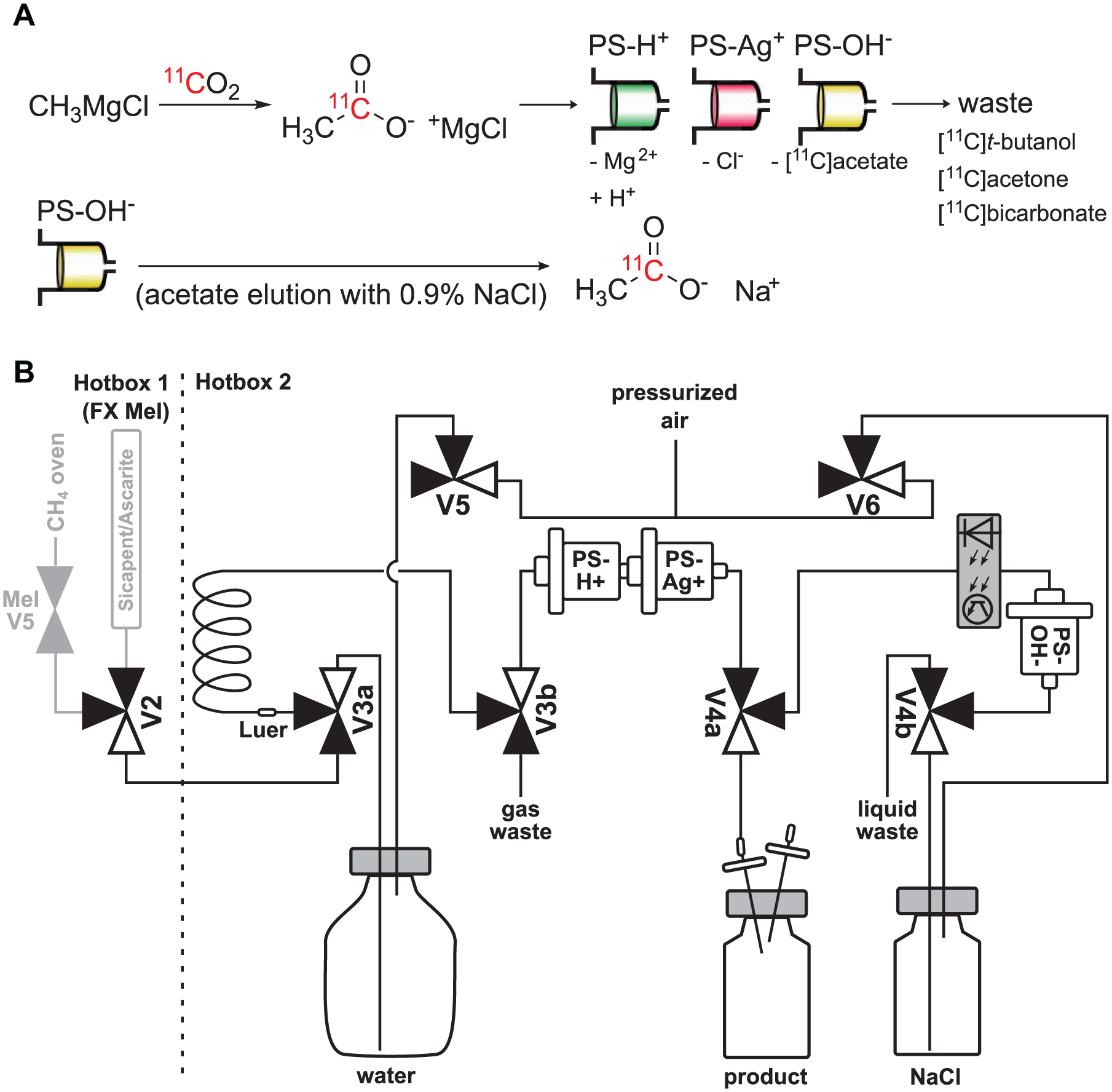
Reaction and purification scheme for [1-11C]acetate (
For example, in the method of Soloviev et al., 9 the [11C]CO2 is first captured from the target gas in a cooled steel tube and then transferred to a loop coated with Grignard reagent. The reaction mixture is then transferred to a stack of two cation exchange cartridges for acidification and reduction of chloride content, and acetate is trapped on an anion exchange cartridge. Isotonic saline solution is used for recovery and formulation of the tracer. This strategy avoids addition of carrier acetate and is simple enough to avoid dedicating a complex radiosynthesis module10,18,22 or even a complete hot cell infrastructure (for commercial acetate module) to acetate synthesis. However, the implementation of Soloviev et al. is not described in detail and thus not directly transferrable to other labs. The same holds true for essentially all dedicated synthesis setups for [1-11C]acetate described so far,9,11–17,19 impeding the adaptation of such syntheses to local requirements. Basically, while many authors call their synthesis “simple” or “easy” because of the low chemical complexity, implementation of these strategies usually requires significant investments in the infrastructure (adding more dedicated hot cells), which is a critical obstacle for establishing this tracer in a research lab. Flexible synthesizers with their respective control boxes and computers need a lot of precious lab space, and manual modifications of existing setups like the Tracerlab FX C Pro synthesizer10,18 are a critical source of errors when other tracers are also routinely produced on a synthesis module.
Arduino (https://www.arduino.cc/) is an open-source platform for physical computing, typically featuring Atmel 8-bit AVR series microcontrollers with a user-friendly periphery and an integrated development environment (IDE). Its documented, robust, and affordable hardware design, combined with a high availability of libraries written by the companies behind Arduino and a broad community, enables quick familiarization and makes the Arduino platform ideal for educational as well as productive or scientific application.23–26
The development of Arduino-based 3D printers was a substantial milestone and catalyst of the so-called “maker culture,”27–29 inspired by the FabLab of the Center for Bits and Atoms at the Massachusetts Institute of Technology. Currently, the number of FabLabs worldwide is rapidly growing, bringing the maker culture and access to modern fabrication methods such as 3D printing, computer numerical control (CNC) machining, and lasercutting to the public.
In this work, we used an Arduino microcontroller to build a simple and inexpensive module to synthesize [1-11C]acetate from [11C]CO2 prepurified by a Tracerlab FX MeI or FX C Pro module (GE Healthcare, Uppsala, Sweden). The setup can be replicated at other sites (with cyclotron, MeI synthesizer, and adequate radiation protection), since the additional hardware parts either can be manufactured by 3D printing and lasercutting in a FabLab or are commercially available (valves, tubes, fittings, vials, etc.). In this study, we put focus on transparent documentation of hardware, software, and the synthesis procedures, to foster replication by chemists less skilled with microcontrollers or programming.
We demonstrate successful radiosynthesis and quality control of [1-11C]acetate for the first in vivo experiments with this tracer in our group, paving the road for now ongoing oncological studies.
Materials and Methods
Electronics
As a control unit, we used an Arduino/Genuino Uno, which is easily available from commercial suppliers (e.g., RS Components, Conrad, Mouser). To connect the control unit to the peripherals, such as valves, switches, and power supply, we developed an adapter circuit (so-called shield) on a perfboard. The schematic and photographs of the shield are shown in the supplemental data (
Mechanical Parts
The base plate holding the valves and vials was designed in OpenSCAD 2011.12 (http://www.openscad.org) and 3D printed with black acrylonitrile butadiene styrene (ABS) on a PRotos V2 (German RepRap, Feldkirchen, Germany) FDM printer with the following parameters: 0.5 mm nozzle, 0.3 mm layer height, 110 °C bed temperature, 235 °C extruder temperature, and 20% rectilinear fill. The G-code used was generated by Slic3r 1.2.9 (http://slic3r.org). A rendered image of the plate is shown in the supplemental data.
The housing of the electronics was designed with makercase (http://www.makercase.com) and Inkscape 0.91 (https://inkscape.org) and cut from 3 mm medium-density fiberboard (MDF) using a 100 W CO2 lasercutting machine (1390, AM Laserpoint, Hamburg, Germany).
Fluidic Parts
Seven Type 0127 three-way solenoid valves (120433, Bürkert fluid control systems, Ingelfingen, Germany) were salvaged from the annual maintenance of our commercial radiosynthesizers. Six of them were screwed to the 3D-printed base plate using M3×16 mm screws and the respective nuts, while the remaining valve was installed between V5 and the Ascarite/Sicapent trap of the Tracerlab FX MeI module. To avoid clogging, 1/8-inch tubing was used to connect the loop to the gas waste (no. 1648, P-300, P-308, IDEX Health&Science, Erlangen, Germany), while standard 1/16-inch ETFE tubing with the respective ¼-inch -28 fittings (1528, P-200, P-208, IDEX Health&Science) was used to interconnect the remaining parts. The cartridges, the needles, and the loop (no. 1159.10, Vygon, Aachen, Germany) were attached using Luer connectors (P-655-01, P-658, IDEX Health&Science).
The 3D-printed base plate was designed with two cylindrical wells to accommodate the liquid reservoirs for water and phosphate-buffered saline (PBS), a squared well for the liquid detector printed circuit board (PCB) and a front plate with holes to guide the 1/16-inch tubing. Since the authors replaced clogging-prone parts of the tubing with 1/8-inch tubing during the development, not all these holes are actually used in the latest setup described here. To allow for larger tubing, V3b was mounted on the base plate at the outer left position, followed by V3a, V4a, and V4b.
General Radiochemistry
All solvents and reagents were obtained from commercial suppliers in at least analytical quality. Anhydrous tetrahydrofuran (THF) and methyl magnesium chloride (3 M in THF) were obtained from Sigma-Aldrich (Taufkirchen, Germany) and used without further purification. Chromafix PS-OH– (200 mg), PS-H+ (230 mg), and PS-Ag+ (240 mg) cartridges (Macherey-Nagel, Düren, Germany) were conditioned with 5 mL of ethanol and 5 mL of water. NaOH-coated thin-layer chromatography (TLC) plates were prepared by soaking the plates (POLYGRAM SIL G/UV254, Macherey-Nagel) in a saturated solution of sodium hydroxide in methanol, and stored in a sealed bag after vacuum drying at ~10 mbar for 30 min. Autoradiography of TLC plates was performed using the Cyclone Plus Storage Phosphor System (PerkinElmer, Waltham, MA).
Radio-high-performance liquid chromatography (radio-HPLC) was performed on a 1260 Infinity HPLC system (Agilent Technologies, Böblingen, Germany) equipped with an autosampler, an NaI(Tl) scintillator setup, and the multiwavelength detector set to 214 nm.
Preparation of the Module for Synthesis
The module was cleaned and dried between syntheses with water, acetone, and THF according to the protocol (see SOP in supplemental data,
Before each synthesis, the CH4 oven was conditioned at 350 °C with hydrogen and helium for 15 min each, and then cooled to <35 °C before end of bombardment (EOB).
Reagent vials were filled with water and saline, respectively, according to the attached SOP. The product vial was filled with citrate buffer, and the needle was situated at the bottom of the vial. The conditioned cartridges were placed between the respective Luer connectors, and the loop was coated with methyl magnesium chloride by filling and retracting 2 mL of a 2 M solution in THF. After the loop was closed, an unblocked flow path was verified from the flow regulator reading of the FX MeI module at 10 mL/min.
Radiosynthesis of [1-11C]Acetate
[11C]CO2 was prepared by bombardment of 14N2 target gas containing 1% O2 with 70 µA of 16.5 MeV protons using a PETtrace 860 cyclotron (GE Healthcare). Activity was transferred to the CH4 oven of an FX MeI module and then released by heating to 380 °C. The released [11C]CO2 was transferred to the Acetuino module with a helium stream (40 mL/min) and trapped in the synthesis loop coated with Grignard reagent (“CO2 trapping” step, Table 1 ). After a reaction time of 3 min, the [1-11C]acetate was transferred through a PS-H+ and a PS-Ag+ cartridge to a PS-OH– cartridge using 40 mL of water (“loop elution” step). When the liquid detector indicated completion of this step, the PS-OH– cartridge was eluted through a sterile filter into the product vial with 4 mL of 0.9% NaCl (“acetate elution” step). This procedure was executed fully automated from the time point of delivery from the cyclotron, using the trigger connected to V9 (FX MeI) and the liquid detector preceding the PS-OH– cartridge ( Table 1 ).
Overview of the Automated Synthesis and Cleaning Steps and the Respective LED and Valve States.
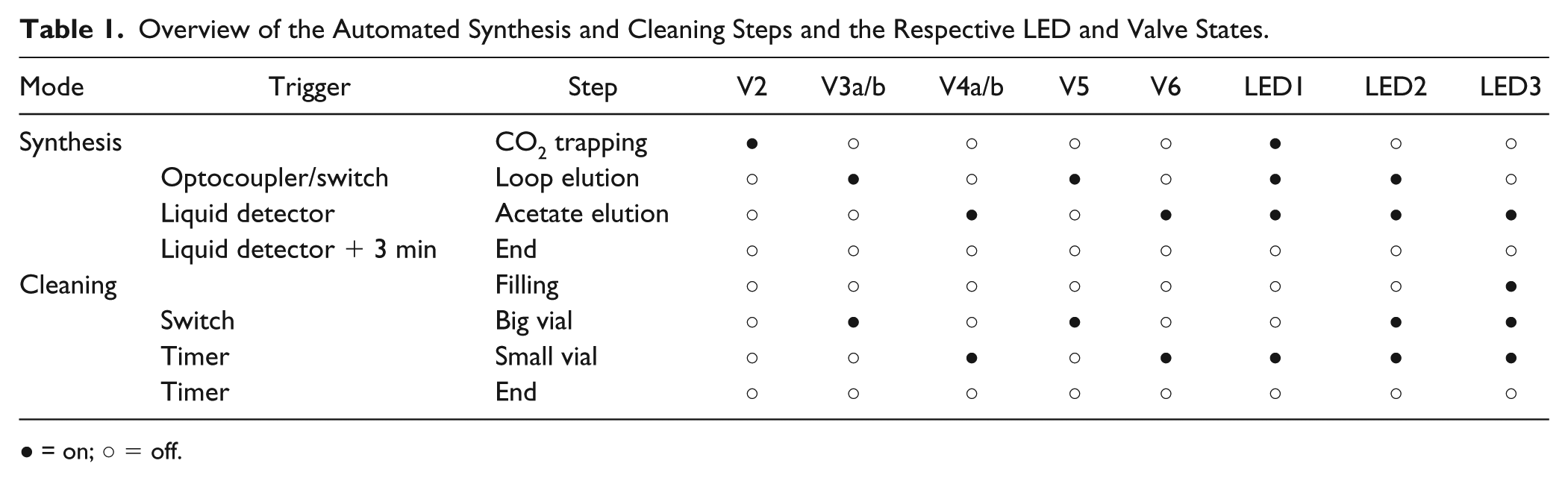
= on; ○ = off.
Quality Control
The radiochemical purity (RCP) of the product was assessed by TLC using 0.5 µL of the product solution on NaOH-impregnated silica plates with methanol as the mobile phase as described before. 8 [11C]Acetate had Rf values of 0.8–0.9, while the contaminant near the baseline (Rf from 0.0 to 0.1) was considered [11C]bicarbonate.
In the first experiments, radiochemical identity and chemical purity were analyzed by radio-HPLC on a Dionex CarboPak PA1 column (4 × 250 mm; Thermo Fisher Scientific, Waltham, MA) with a flow of 0.8 mL/min of 0.1 M carbonate-free NaOH (Sigma-Aldrich no. 43617, protected from atmospheric CO2 by an Ascarite II trap).
Since this column completely retains the main potential contamination, [11C]bicarbonate, we later used an Aminex HPX-87H column (300 × 7.8 mm; Bio-Rad, Munich, Germany) with 1 mM sulfuric acid at a flow of 0.6 mL/min for analysis of radiochemical identity and radiochemical and chemical purity.
pH was analyzed using pH indicator strips (MColorpHast pH 0-6.0, Merck, Darmstadt, Germany) and by visual evaluation.
Imaging
All experiments involving animals were conducted in accordance with the German animal protection laws after approval by the local authorities (Regierungspräsidium Tübingen).
Male C57BL/6 mice were obtained from Charles River (Sulzfeld, Germany) at an age of 6 weeks and kept in individually vented cages under standardized environmental conditions. Food and water were provided ad libitum. Animals anesthetized with 1.5% isoflurane (Abbott, Wiesbaden, Germany) evaporated in oxygen at a flow of 0.5 L/min were placed on an Inveon dedicated small animal PET scanner (Siemens Preclinical Solutions, Knoxville, TN). Fiducial markers were placed in proximity to the animals for better alignment of PET and magnetic resonance (MR) images. Five seconds after initiating the PET scan, ~13 MBq [1-11C]acetate was injected via a tail vein catheter. Body temperature was controlled with a rectal temperature sensor and maintained at 37 °C with a heating pad. The PET scan was stopped after a total scan time of 3600 s, followed by a transmission scan for attenuation correction using the integrated Co-57 source. Subsequently, the animals were transferred into a 7 T BioSpec MR scanner (Bruker, Ettlingen, Germany) and anatomic images were acquired. After the MR scan, animals were killed and several organs were collected, weighed, and measured in a γ-counter (Wizard Wallac 1480, PerkinElmer) to determine the uptake in percent of injected dose per gram of tissue (%ID/g). PET images were histogrammed in 39 frames (12 × 5, 6 × 10, 6 × 30, 5 × 60, 10 × 300 s) and reconstructed with 2 iterations of the ordered subset expectation maximization 3D (OSEM3D) algorithm, followed by 18 iterations of the fast maximum a posteriori algorithm. No scatter correction was performed. Dynamic PET and MR images were aligned and analyzed in Inveon Research Workplace (Siemens Preclinical Solutions) and time–activity curves (TACs) were generated for the percent injected dose per cubic centimeter (%ID/cc) for several organs.
Results and Discussion
Acetuino Module Setup
The custom assembled Acetuino module described in this work (for photographs, see
Radiosynthesis, Purification, and Quality Control
We used the synthesis and purification procedure described by Soloviev et al. 9 as a starting point of our work, automated the procedure, and combined it with [11C]CO2 trapping and purification in our FX MeI module. In short, the Grignard reagent methyl magnesium chloride dissolved in THF was deposited on the loop walls by brief flushing. Since residual moisture could cause clogging, we found it practical to perform this as the last step of the preparation and to verify the absence of obstructions by analyzing the flow of helium carrier gas from the Tracerlab module after filling.
After irradiation of the target gas, [11C]CO2 was trapped in the CH4 oven of the Tracerlab FX MeI, followed by elution at an elevated temperature of 380 °C in a flow of helium. In contrast to the recommendations by the manufacturer of the MeI module, in our experience conditioning the oven with hydrogen and helium (at 350 °C for 15 min each) between syntheses led to a more stable [1-11C]acetate yield. However, we did not thoroughly follow up on this hypothesis due to our scheduled regular productions of 11C-methylated tracers requiring daily conditioning of the oven. We therefore included this in our protocol for acetate production.
As expected and reported by Soloviev et al., trapping of the activity on the loop coated with Grignard reagent was almost quantitative, as no appreciable activity (<50 MBq) was found on Ascarite traps mounted to the gas waste tube. We calibrated the activity detector of the Tracerlab CH4 oven by replacing the Acetuino loop with two subsequent Ascarite traps and measuring the radioactivity on both traps after release from the CH4 oven using a dose calibrator. No activity reached the second trap; the activity on the first trap was thus used to calculate the radioactivity released from the CH4 oven.
After transfer of [11C]CO2 to the loop and reaction with the Grignard reagent, the purification program flushed the loop with water; by this the reaction solution was guided over a stack of a PS-H+ and a PS-Ag+ cartridge for the hydrolysis and reduction of halide (here, chloride from the Grignard reagent) content, respectively. Afterwards, the flow was guided to the PS-OH– cartridge where the product was trapped, and in the next step eluted into the product vial with isotonic sodium chloride solution.
After the first syntheses, we discovered that the “unknown anionic impurity” that Soloviev et al. observed also occurred in our setup in unpredictable amounts (Rf of 0.0–0.1 on TLC, not eluted on radio-HPLC with the CarboPak system). When we acidified the tracer solution and bubbled air, helium, or argon through the solution, the impurity was removed (
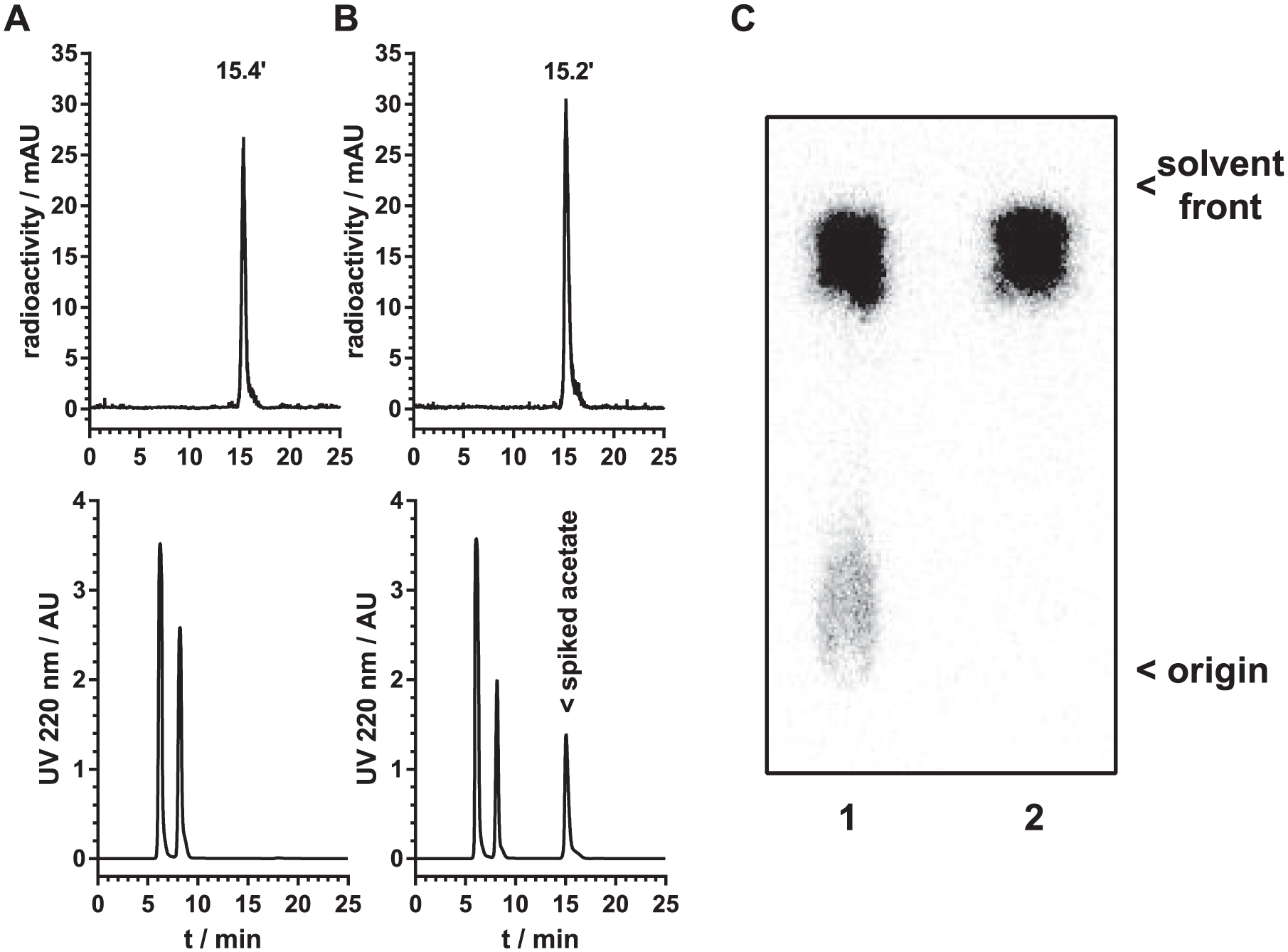
Quality control of [1-11C]acetate by HPLC on an Aminex HPX-87H column before (
For estimation of the RCP as well as the detection and identification of contaminants, we relied on both TLC and CarboPak-HPLC since the different literature-known contaminants, [11C]bicarbonate, [11C]acetone, and [11C]tert-butanol, are not covered by only one of these methods. The latter two contaminants are separated from [11C]acetate by HPLC,
9
but these contaminants were efficiently removed by the cartridge purification and not detected in the final tracer. The more problematic contaminant, [11C]bicarbonate (as it was not always completely removed), was not detectable using this column since it is completely retained by the stationary phase. TLC in turn separated carbonate well from acetate, making both methods complementary for estimation of the RCP. The Aminex HPX-87H column used by Kruijer et al. and Le Bars et al. is reportedly able to resolve all the abovementioned compounds,13,16 making this system an attractive option and justifying the extended analysis time of 30 min instead of 10 min, and the additional time needed to ramp up the flow rate according to the manufacturer’s instructions. We thus switched to this column for routine quality control of [1-11C]acetate (
In summary, after optimization of the procedure, we were able to reliably produce [1-11C]acetate in suitable amounts (3.2 ± 1.3 GBq at EOS) and quality (typically >95% RCP) for small animal imaging from 5 min target irradiations at 70 µA in only 15 min of synthesis and purification time. Radiochemical yields (relative to [11C]CO2 released from the CH4 oven) were in the range of previously reported procedures (51.4 ± 28.2%, decay corrected).
Imaging and Ex Vivo Analysis
In our in vivo experiments, [1-11C]acetate showed a strong perfusion peak in heart and kidneys. The radioactivity was quickly washed out, possibly after oxidative metabolism to bicarbonate. Already, ~400 s after tracer injection the liver became the organ with the highest uptake. A stable equilibrium of the tracer distribution was reached 1800 s after tracer injection. In the last time frame analyzed after the start of the PET scan (3300–3600 s), values of 0.5 ± 0.1, 1.5 ± 0.4, 0.9 ± 0.2, and 0.5 ± 0.2 %ID/cc for muscle, liver, kidney, and heart, respectively, were determined in the PET images (
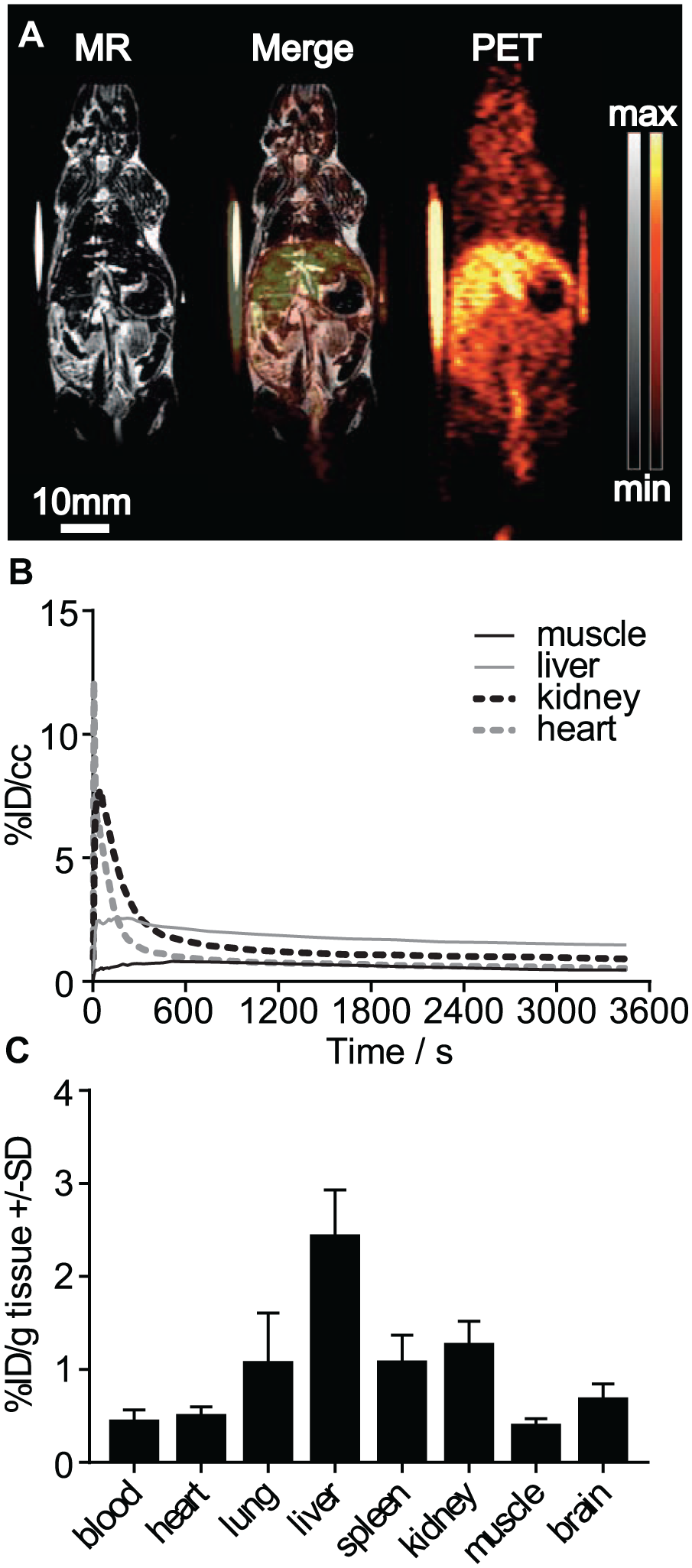
Results of in vivo experiments in healthy animals. (
Here we describe a low-cost and low-footprint synthesizer for the production of [1-11C]acetate for preclinical imaging. It is small enough to fit into an already-occupied hot cell, and we are using this setup routinely to start the synthesis of a 11C-methylated tracer (e.g., for neuroimaging), directly followed (and eventually outpaced) by [1-11C]acetate for oncological studies.
Numerous synthesizers for [1-11C]acetate have been described, but most of them rely on closed-source hardware and software for controlling the valves and lack crucial information about their automation. Automation solutions often require the use of separate hot cells, hampering their use in research labs with limited space and resources. Our setup aims to be easily replicated at other sites using inexpensive equipment by anyone with basic skills in electronics. The growing network of FabLabs worldwide27,28 aims to enable access to the production methods we used in this work, such as 3D printing, lasercutting, and soldering, without significant additional investments.
Due to the simplicity of the synthesis and purification task, we chose not to include a display and instead only used LEDs to indicate the progress of the synthesis. We did not include a radioactivity detector into the setup because the actual benefit would be low in such a rapid synthesis. We also chose to rely on existing infrastructure for [11C]CO2 trapping and release (Tracerlab FX MeI) instead of using nitrogen-cooled steel loops for direct connection to the cyclotron, since automated CO2 purification capabilities are typically already available at cyclotron facilities.
Our device is able to produce no-carrier-added [1-11C]acetate with high radiochemical yield and purity. This is not only shown by chemical analysis with TLC and HPLC, but also supported by the in vivo and ex vivo distribution of the tracer, as shown in dynamic PET scans and γ-counter studies, which show a similar biodistribution of our [1-11C]acetate to what has been reported by others. 30
We thus claim that the use of microcontrollers like the Arduino platform enables versatile construction and control of radiochemical synthesizers. Their low cost and small footprint allow dedication of the module for individual tasks. Setups based on this platform have become valuable tools in our lab, automating processes where full radiochemistry synthesizers are too bulky and uneconomical.
Supplemental Material
Supplemental_Material_for_Acetuino_by_Maurer_et_al – Supplemental material for Acetuino—A Handy Open-Source Radiochemistry Module for the Preparation of [1-11C]Acetate
Supplemental material, Supplemental_Material_for_Acetuino_by_Maurer_et_al for Acetuino—A Handy Open-Source Radiochemistry Module for the Preparation of [1-11C]Acetate by Andreas Maurer, Gregory Bowden, Jonathan Cotton, Christoph Parl, Marcel A. Krueger and Bernd J. Pichler in SLAS Technology
Footnotes
Acknowledgements
We thank Johannes Kinzler, Elena Kimmerle, and Sandro Aidone for excellent technical assistance. Fablab Neckar-Alb e.V. supported the construction of Acetuino with their lasercutter.
Supplemental material is available online with this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research leading to these results was funded by FOR 2314 of the Deutsche Forschungsgemeinschaft (DFG).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
