Abstract
Human induced pluripotent stem cells (HiPSCs) provide several advantages for drug discovery, but principally they provide a source of clinically relevant tissue. Furthermore, the use of HiPSCs cultured in three-dimensional (3D) systems, as opposed to traditional two-dimensional (2D) culture approaches, better represents the complex tissue architecture in vivo. The use of HiPSCs in 3D spheroid and organoid culture is now growing, but particularly when using myocardial, intestinal enteric nervous system, and retinal cell lines. However, organoid cell culture is perhaps making the most notable impact in research and drug discovery, in which 3D neuronal cell cultures allow direct modeling of cortical cell layering and neuronal circuit activity. Given the specific degeneration seen in discrete neuronal circuitry in Alzheimer’s disease (AD) and Parkinson’s disease (PD), HiPSC culture systems are proving to be a major advance. In the present review, the second part of a two-part review, we discuss novel methods in which 3D cell culture systems (principally organoids) are now being used to provide insights into disease mechanisms. (The use of HiPSCs in target identification was reviewed in detail in Part 1.)
Introduction
Over the last decade, numerous approaches have been developed to culture and investigate cellular function in a three-dimensional (3D) environment.1–9 In parallel, the adoption of human induced pluripotent stem cells (HiPSCs), for both basic research and drug discovery, is increasing (see Part 1 of this review). Since 3D culture systems provide a more natural tissue environment to study cell function—and patient-derived HiPSCs can model human disease pathophysiology better than most animal or other model systems—the use of HiPSC differentiated cell types in 3D culture systems provides numerous advantages: (1) study tissue and organ complexity in vivo; (2) provide the potential to be experimentally tractable, notably for compound screening; and (3) allow patient-specific responses, prior to clinical evaluation, to be monitored. 1
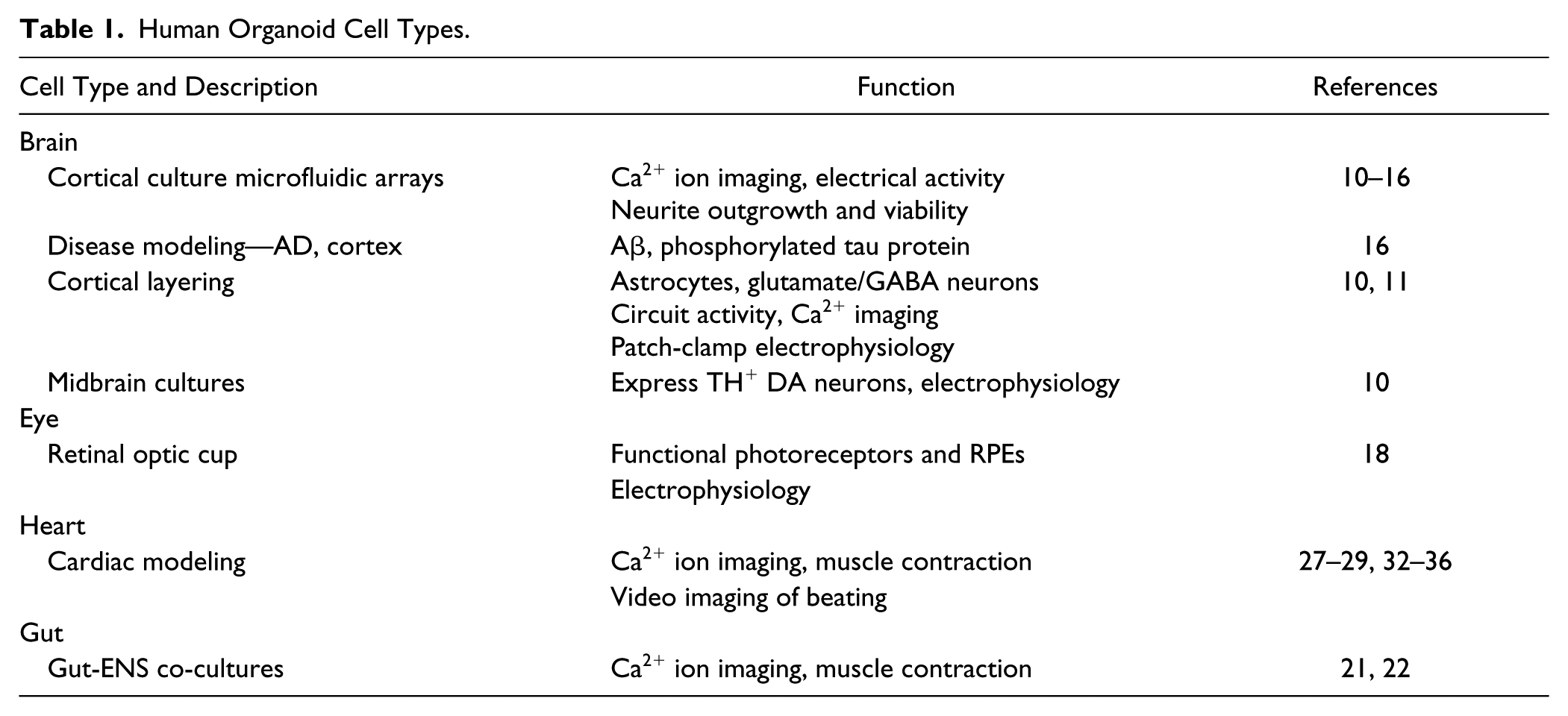
In summary, HiPSC-derived organoids, as with other cells cultured in 3D, enable the generation of multiple organ-specific cell types, each possessing appropriate cells: cell organizations resembling those found in native human tissues and therefore exhibiting pathophysiological characteristics more like cells found in the patient population. Several organlike structures, termed organoids, have now been generated from HiPSCs, including brain,10–16 eye,17–20 intestine,21,22 liver,23–25 lung, 26 heart,27,28 and several other tumors, organs, and tissues29–31 (summarized in Table 1 ). It is therefore unsurprising that the adoption of HiPSC-derived cells, particularly when cultured in organoids, is growing for use in both fundamental research and drug discovery.1–3,6,7,31
Human Organoid Cell Types.
Cancer Cell 3D Models
One of the first applications of 3D systems for drug discovery involved cancer cells.29,31,37,38 Cancer cell organoids are generated from primary cells rather than from HiPSCs. Yet the 3D system can provide a better model of solid tumors than 2D systems because they generate cancer cells in a spatially relevant manner, encouraging cell–cell and cell–matrix interactions that closely mimic the native environment of solid tumors.39,40 They also cause the cancer cells to acquire morphological and cellular characteristics relevant to in vivo tumors. 41 3D cancer cell systems mimic solid tumor gene and protein expression, as well as metabolic activity, cell stress response, structure, signal transduction, and cellular transport proteins, better than 2D-cultured cells.42,43 Furthermore, the 3D cultures show the variability and resistance of drug uptake and metabolism, like that found in solid tumors but not seen in 2D cultures, in which all the cells are equally exposed to drugs.1,39,43
Cancer cells in 3D culture can be monitored for activity using a variety of assay forms5,44–46 and exhibit differences in drug sensitivity when compared with 2D cultures of the same cells. For example, breast tumor 3D cultures express a higher sensitivity to trastuzumab than 2D cultured cells, as the cells of the 3D cultures display increased activation and dependence on Her2 and Her3 signaling.42,43 Trastuzumab blocked Her2 and Her3 activation and proliferation of 3D cultures but not 2D-cultured cells. 42 RAF and MEK inhibitors block the invasion of thyroid carcinoma 3D cultures but have no effect on the migration of monolayer cells. 43 The disparities in pharmacological activity between 3D-cultured and 2D-cultured cells are related to differences in drug pharmacokinetics, generation of hypoxia and ROS, enhanced expression of multi-drug resistant genes, and activation of cell survival pathways.42–44
The 3D cultures can also reproduce several parameters of the tumor microenvironment, including oxygen and nutrient gradients, as well as the development of dormant tumor regions. 40 Tumor cells become dormant and stop cycling if their access to oxygen and nutrients is restricted. In fact, one of the critical targets of cancer research and drug discovery is cancer stem cells that self-renew and differentiate into different cell types, contributing to tumor heterogeneity. 41 These cells generally do not proliferate in the tumor and have lower metabolic activity than surrounding proliferating cancer cells. Their lower activity makes them relatively resistant to cytostatic drugs that mainly target proliferating cells. The 3D culture provides a system to target these dormant cells to enhance the efficacy of therapies to treat solid tumors. 44
Methods now exist to visualize the dormant cells in 3D cultures.46,47 Thus, cancer cells grown in 3D can be labeled with nucleoside analog. The nucleoside analog becomes diluted in dividing, proliferating cells but is retained in the quiescent, nondividing cancer cells. The dormant cancer cells can then be visualized, quantified, and distinguished from the surrounding proliferating cancer cells by confocal microscopy.46,47
Wenzel et al. 47 generated T47D breast cancer cells in 3D cultures and used imaging technologies to distinguish cells in the inner core of the cultures from those in the surrounding outer core. The inner core cells had less access to oxygen and nutrients and showed reduced metabolic activity compared with the outer core cells. By screening small molecule libraries against these 3D cultures, these authors 47 identified nine compounds that selectively killed the inner core cancer cells without affecting the more active proliferating outer core cancer cells. The drugs primarily inhibited the respiratory chain pathway, consistent with the change in metabolic activity of the oxygen-deprived cells from aerobic to anaerobic metabolism. 47 Thus, the 3D culture system may provide approaches to develop new therapeutics to target those cancer cells that resist cytostatic anticancer drugs. In fact, Wenzel et al. 47 showed that the compounds that selectively target the dormant cancer cells greatly enhance the efficacy of commonly used cytostatic anticancer drugs. Taken together, these, and numerous other studies,40–51 demonstrate the advantages of using 3D culture systems for in vitro oncology studies, bridging the gap between 2D culture models and in vivo whole-animal systems.1,3,31
HiPSC-Derived Liver Tissue in 3D Cell Culture
3D culture systems have been developed for HiPSC-derived hepatocytes,23–25,52 and the liver organoids can be monitored for activity using several different detection and imaging systems ( Table 1 ).4,23,52 These cells have significant value in evaluating the potential of drugs for exhibiting liver toxicity since in many cases human hepatocytes have different sensitivities to drugs than hepatocytes derived from animals or liver tumor cell lines, as described extensively in Part 1 of this review. While human hepatocytes are used for testing drugs for toxicity in preclinical development studies, they are generally limited in availability, and thus HiPSC-derived hepatocytes provide a greater source of tissue for compound evaluation. 52
Liver cells in organoids have advantages over the use of similar cells grown in 2D since they provide a more relevant cell–cell interaction due to the polarity of the hepatocytes, which is critical for cell metabolism, a feature essential for drug metabolism. Most importantly, human liver organoids respond differently to drugs than similar cells grown in 2D culture. 23 Interestingly, because of differences in expression of metabolizing P450 enzymes between individuals, one can envision individual drugs being screened against large populations of HiPSC-derived hepatocytes in 3D to provide a better preclinical assessment of a drug’s potential to induce toxicity than presently available with any other in vitro screening system. 52
HiPSC-Derived Myocardial Tissue in 3D Cell Culture
Cardiac myocytes have been cultured in 3D systems to develop in vitro models of myocardial function ( Table 1 ).53–56 Previously, approaches to culturing myocyte organoids have required that the cells be cultured and embedded within ring-shaped molds, either using rigid cantilever structures or by being interspersed into rigid meshes. 55 These rigid boundaries thus anchor the underlying substrate and provide a mechanical load against which the myocardial organoids contract. Subsequently, myocardial contraction induces sarcomere assembly and cell alignment, such that the tissue architecture begins to resemble that found in vivo. Recently, Heubsch et al. 28 developed a 3D cell culturing system using HiPSC-derived cardiac myocyte organoids, in which cells were arrayed to form elongated muscle fibers, but without an ECM. The human cardiac organoids formed in a tissue syncytium much like cardiac tissue in vivo in humans. Using video imaging, these authors then monitored cell contraction in the array fibers, as well as Ca2+ ion transients, using the biosensor reporter, GCaMP6F. These authors 28 observed that the rate of myocardial beating in 3D culture was less variable, on a well-to-well basis, than that observed in 2D myocyte culture. Pharmacologically, cultured fibers showed positive inotropic responses to β adrenoceptor agonists, as well as an inhibition of contraction by the Ca2+ channel inhibitor, verapamil. These authors 28 also monitored the contractility of human cultured muscle fibers and showed an increased twitch response when exogenously stretched. This approach may therefore be adapted to a high-throughput screening (HTS) format for the compound screening of human cardiac muscle fibers, both to evaluate myocardial toxicity and to identify novel therapeutics for cardiac diseases.
HiPSC-Derived Gut and Enteric Nervous System in 3D Cell Culture
3D cell culture using HiPSCs can model the enteric nervous system (ENS), gut function, and the functional interactions between them ( Table 1 ).21,57 Physiologically, the ENS controls gastrointestinal motility, secretion, blood flow, epithelial barrier permeability, and fluid exchange.21,22,57 Dysfunction in the gut ENS occurs in numerous digestive diseases, including Crohn’s or inflammatory bowel disease (IBD), while impaired function frequently occurs in Parkinson’s disease (PD) 58 and diabetes mellitus. 59 The bowel also harbors a major portion of the human gut microbiome, which interacts with the epithelial cells, and so influences nutrient transport, as well as contributing to the etiology of several diseases. Nonetheless, despite this important physiological function, few human cell culture systems exist to model the interaction between the ENS and gut cells. To address this deficit, several groups have now developed human intestinal organoids using HiPSCs.21,22,60,61 HiPSCs, differentiated to intestinal cells, self-organize to form 3D crypt-villus structures that mimic intestinal organization and physiology, and display several intestinal functions, including motility, absorption, and secretion. 3D HiPSC-derived colon organoids have begun to be used for drug screening and discovery.61,62 Use of these cell systems has many potential important uses, especially when the iPSCs are derived from patients with different gut disorders.
To develop intestinal innervation models, Workman et al. 21 have developed a culture system consisting of gut organoids innervated by a functional ENS, and used HiPSCs to generate neural crest cells and intestinal organoids. They combined these two cell types and cultured them under 3D culture conditions and observed that the neural crest cells migrated into the gut mesenchyme and differentiated into neurons and glia. By contrast, neurons were not found in HiPSC-derived intestinal organoids alone. They suggest 21 that the combined neural crest cells and intestinal tissue resembled human fetal intestinal tissue and observed that neural crest cells differentiated into multiple types of neurons, including those with acetylcholine, serotonin, and catecholamine neuronal phenotypes. Through the use of GCaMP6f, an ultrafast Ca2+ ion biosensor, selectively expressed in ENS cells, it was shown that the intestinal neuronal inputs were spontaneously active, demonstrating rapid increases in Ca2+ ion transients in response to K+ ion depolarization. Electrical stimulation produced sustained intestinal muscle contractions, as monitored by video imaging, that were dependent on neuronal inputs, and activation of neurons by nicotinic agonists, or veratridine, induced intestinal muscle relaxation. Interestingly, this was seen in the ENS–intestinal organoid complex but not in intestinal organoids alone, suggesting that the neuronal input actively regulated gut contractility. In support of this hypothesis, the ENS–intestinal organoids secreted nitric oxide, since the relaxant response nicotinic receptor stimulation was inhibited by nitric oxide synthase (NOS) inhibitors.
Taken together, this novel HiPSC-based, human ENS–gut system appears to be an advance, given that there are numerous differences between human and rodent intestinal systems, from both developmental and functional perspectives.60–64 This organoid system may therefore provide one of the first demonstrations of human PSC-derived intestinal tissue with a functional ENS that may be employed in compound screening.
Cell culture systems such as these may also allow the study of motility disorders of the human gastrointestinal (GI) tract in other diseases, including PD. Recent reports have shown that the ENS–intestinal organoids express tyrosine hydroxylase (TH)-positive neurons.58,65,66 These neurons in the gut play a prominent role in PD because early in the disease they begin to express Lewy bodies and α-synuclein, critical early neuropathological features of the disease. 57 The appearance of Lewy bodies in the gut can occur decades before the appearance of motor symptoms in the disease. Consequently, culturing ENS–intestinal organoids, using cells from PD patients, may provide a unique system to study the earliest pathogenic mechanisms in this disease—and help identify novel therapeutics targeting the gut, rather than the brain, to mitigate PD progression. 66
HiPSC-Derived Retinal Organoids in 3D Cell Culture
HiPSC culture technology has also been used to develop 3D retinal cell “cups” that express most of the major retinal cell types, including photoreceptors, and are physiologically arranged in discrete layers ( Table 1 ).17–20 A major advantage of 3D retinal cell culture is the formation of outer-segment discs that elicit a response to light. 19 This property in a cellular model is important, given that most retinal degenerative diseases show dysfunction and, ultimately, death of photoreceptors, leading to vision impairment and blindness.67–69 However, despite extensive efforts, no human model of a functioning human retina has yet been developed, and partly as a result, few effective treatments of retinal diseases are currently available. The 3D retinal cell culture therefore provides an approach to develop in vitro models for human ocular diseases, supporting the discovery of therapeutics to ameliorate progression of diseases such as acute macular degeneration (AMD) or retinitis pigmentosa.20,70–72
One key aspect of organoid development is that HiPSCs can be differentiated in such a manner that recapitulates, both spatially and temporally, the main developmental steps in the human retina. These cells can be induced to form 3D retinal cups containing the relevant retinal cell phenotypes, including retinal pigmented epithelium (RPE). 17 To demonstrate that the HiPSC-derived photoreceptors in these retinal cups retain the ability to respond to light, Zhong et al. 18 used perforated-patch recordings, in the voltage-clamp mode, to monitor the activity of rod cells. In this model, the cells responded to a light flash with an increase in the outward current and, as such, resembled a human retinal cup both morphologically and functionally.
A 3D cell culture model of a retinal cup can be used to better understand the pathophysiology of human retinal disease. RPE and photoreceptor disease models have been reported using iPSCs from patients with AMD and two types of RPE dystrophies, Best disease (vitelliform macular degeneration) and gyrate atrophy.20,70–72 In these models, RPEs are dysfunctional and express drusen-related proteins and inflammatory factors, all of which are pathological markers in the human disease. HiPSC-derived RPEs taken from Best disease patients also show abnormal function by a delayed degradation of photoreceptor outer segments, a critical function of RPEs. 72 Collectively, the development of 3D cell culture models of retinal cups using HiPSC from patients may provide functional human models to identify new therapeutics to treat retinal diseases.
HiPSC-Derived Neuronal Organoid 3D Culture Systems
Although monolayers of HiPSC-derived neurons have been used in compound screening, “flat” 2D culture systems do not fully recapitulate the spatial architecture of neural circuits or their complexity or functionality seen in vivo. This deficit has led several groups ( Table 1 ) to develop 3D neuronal cell culture systems for generating organoid cultures that contain mixed derivatives of ectodermal cells. These methods may recapitulate several aspects of brain development, and clearly display a level of self-organization beyond that found in 2D cultures ( Table 1 ).10–16 To optimize culture formats for compound screening, Wevers et al. 73 cultured HiPSC-derived neurons and glia in Matrigel using a microfluidic system, specifically in an ECM scaffold in tissue chips embedded in a 96-well microtiter plate. Compounds were then screened for potential effects on neuronal electrical activity, neurite outgrowth, and viability.
To develop more consistent methods for monitoring 3D cultures of HiPSC-derived neuronal cells, Jorfi et al. 16 generated 3D neuronal cultures in large-scale arrays and used imaging to measure neurite outgrowth. These authors showed that HiPSC-derived neurons from patients with familial Alzheimer’s disease (AD) mutations could be grown in 3D cultures for at least 8 weeks. They further showed that diseased neurons expressed several key proteins involved in AD neurodegeneration, including Aβ and phosphorylated tau protein. Taken together, cultured human neuronal arrays may provide a screening format to identify compounds acting to reduce the production of these AD disease-causing biomarkers.
Lee et al. 14 generated 3D cultures of HiPSC-derived neurons from five different patients with familial AD and monitored the expression of Aβ. They assessed the cell responsiveness by measuring the action of known inhibitors of β- and γ-secretase (enzymes responsible for generating neuronal Aβ). They 3D cultured the neurons for up to 9 weeks to generate relatively mature neurons for drug testing. In the neurons in the 3D culture, the addition of β- and γ-secretase inhibitors lowered Aβ levels in the neurons, but their efficacy was markedly less than that in studies using 2D cultures of the same cells. It is likely that the decreased compound efficacy in 3D neuronal cultures was partly due to reduced bioavailability. In addition, it was shown that neurons from an AD patient in which the β-secretase inhibitors were ineffective lacked key proteins required for enzyme inhibition.74,75
These data, and other reports, clearly suggest that the variability in the cell genome and proteome from different patients affects drug efficacy. More generally, a major reason for using HiPSC-derived neurons from AD patients is that many nonhuman models, including animal models of AD, are poor models of drug efficacy. Since 3D cell cultures generally express a greater structural similarity for the neuronal microenvironment than that which occurs in 2D cell culture, it is probable that 3D culture will provide a better predictor of human drug efficacy in vivo. This is particularly relevant given that β-secretase inhibitors have now all failed to show efficacy in late-stage clinical trials. 76 This lack of efficacy of secretase inhibitors in reducing neuronal Aβ in AD patient neurons in 3D culture also illustrates the importance of screening HiPSC-derived neurons from multiple patients to estimate variations in clinical efficacy.
A novel approach to study neuronal circuitry involving the use of a novel microfluidic system has been described by Takeda et al. 76 These authors developed a microfluidic culture platform consisting of three interconnected chambers in which neurons, grown in the first chamber, extend neurites to contact neurons grown in an adjacent but separate chamber. The neurites in the second chamber were then extended to neuronal cell bodies in a third chamber. This model was used to measure uptake of the protein tau, as well as its transfer from one neuron to another. Tau is a critical protein involved in AD neurodegeneration, with the spread of brain pathogenesis probably involving the release and uptake of tau from one neuron to another in specific brain circuits. Indeed, using this microfluidic system, these authors showed that tau either from transgenic mouse expressing AD-causing genes or in cells from AD patients was released and propagated from one neuron to another. Consequently, this approach may be adapted to study the circuitry of HiPSC-derived neurons cultured in 3D, and potentially used to investigate mechanisms of disease spreading. Importantly, it may be employed to identify compounds that act to reduce neuronal tau uptake and potentially slow disease progression. The approaches may also have other applications to study in vitro synapse formation, as well as circuit connectivity and reinforcement. Although techniques using brain slices or two-photon microscopies are available in this regard, a neuronal organoid microfluidic chamber could provide a better method to monitor those neurons connecting under defined conditions. 74
While 3D HiPSC-derived neuronal cultures create more physiologically relevant systems, many models lack the environmental complexity of neuronal tissue in vivo, specifically as cortical neurons develop in defined layered networks when cultured in concert with other cell types. However, in many 2D culture systems cortical lamination is absent, and furthermore, co-expression of astrocytes with supportive cells types is also lacking. 77 Most importantly, robust synaptogenesis and spontaneous synaptic activity are not apparent. Therefore, the use of 3D cultures that more effectively recapitulate specific human brain regions, with high reproducibility and scalability, is an essential goal when developing models to study neuronal circuitry and function in vitro—particularly when establishing human disease models.78–80
To address this point, Pasca et al. 11 reported a novel 3D culture system using HiPSC-derived neurons that represent the laminated neuronal structure seen in the human brain. These cultures were free floating, in that the cells were not cultured on embedded structures or scaffolds but were maintained for up to 9 months in vitro. Transcriptional profiling demonstrated that the 3D cultures expressed genes resembling those found in the developing human cortex during the late to midfetal period. These cultures expressed both neuronal markers, including those for human forebrain neurons, astrocytes, and glia. The organoids could be sectioned and analyzed morphologically and showed similar cytoarchitectures to those found in developing human cortex. By using immunocytochemistry techniques, these authors 11 distinguished between superficial and deep cortical layers. These organoid neuronal cultures were functionally active and amenable to recording responses from electrically stimulated neurons. Using Ca2+ ion imaging, they showed that spontaneously active neurons and patch-clamp recording demonstrated transient inward currents sensitive to tetrodotoxin. Using markers for pre- and postsynaptic elements, they also showed that neurons formed synapses—glutaminergic synapses resembling the primary synapses found in human forebrain cortex. Excitatory postsynaptic activity at these glutamate synapses was spontaneous and blocked by glutamate receptor antagonists. Taken together, Pasca et al. 11 showed, in 3D culture of HiPSC neurons, physiologically relevant activity of network circuits, a characteristic difficult to demonstrate using other in vitro or in vivo systems.
Birey et al. 80 generated 3D culture systems of HiPSC-derived neurons from subregions of the human cortex, using either the dorsal or ventral forebrain expressing glutamate or GABAergic (gamma-aminobutyric acid) neurons, respectively. The ventral forebrain is of importance given that during development GABAergic interneurons migrate from this region to the dorsal forebrain. Development disorders may therefore involve impaired neuronal migration and subsequent formation of dysfunctional circuitry. These authors reported that ventral forebrain neuronal cultures expressed GABA interneurons and, by using patch-clamp electrophysiology of ventral forebrain organoid slices, identified a large population of neurons expressing spontaneous chloride-dependent inhibitory postsynaptic currents. These currents were abolished by the GABAA receptor antagonist, gabazine. In co-cultures of the dorsal and ventral forebrain 3D HiPSC-derived neurons, confocal imaging was used to visualize migration of GABA interneurons from ventral to dorsal forebrain regions, a feature seen in vivo only during development. In summary, these authors suggested that they had recreated a unique model that mimicked the development activity of the human cortex needed for GABAergic inhibitory control. These authors further generated dorsal and ventral forebrain cultures from HiPSCs of Timothy syndrome patients—a neurodevelopmental disorder caused by mutations in the Cav1.2 calcium ion channel. They co-cultured the different 3D organoids and showed impaired migration of the interneurons. This novel 3D culture system of HiPSC-derived neurons, in summary, potentially provides a novel platform with which to study numerous human central nervous system (CNS) diseases involving cortical circuitry dysfunction, such as AD or epilepsies, as well as developmental disorders, such as autism and schizophrenia.
The generation of 3D cultures from cells taken from specific human brain regions provides unique opportunities to develop innovative models of neurodegenerative diseases, such as PD, involving gradual dysfunctional and consequent loss of nigrostriatal dopamine (DA) neurons. Jo et al. 10 recently developed 3D cultures of human midbrain, a region intimately involved in basal ganglia function and motor control and, notably, the major site of degeneration in PD. Midbrain 3D cultures were generated from human embryonic stem cells, and it is likely that they could be adapted to HiPSC technology. The 3D cultures consisted of neurons that expressed DA uptake sites and TH, key proteins involved in DA neuronal function. Of interest, the 3D neuronal cultures expressed DA neurons containing the pigment neuromelanin. In humans, these neurons gradually die in PD. Generally, it has been difficult to generate neuromelanin containing neurons in vitro in culture. TH+ neurons also expressed the G protein gated inwardly rectifying K+ channel (GIRK2), a target, enriched in human brain nigrostriatal DA neurons. Patch-clamp recordings of 3D organoid slices showed neurons to be spontaneously active and responsive to DA receptor agonists. The cultures also expressed neuronal circuitry that may be studied by electrical stimulation.
These 3D human brain organoids should be amenable to study the basic disease mechanisms involved in PD, particularly as most of the research on the disease involves animal models that in many cases do not recapitulate the human disease well. 79 The 3D organoid model can also be employed to study the morphological changes in the nigrostriatal human neurons as they relate to changes in their physiology. By using HiPSC as a tissue source, they can also be employed to study differences in nigrostriatal DA neurons from patients with familial PD from those with the more prevalent sporadic form of the disease. Furthermore, adaption to HTS formats may provide novel opportunities to discover drugs to slow the degenerative processes of this disease.
One problem of developing 3D cultures of neurons, or for that matter any cell type, is a general inability to simulate the vascularization required to support tissue survival and function. 80 Vascularization is also critical in evaluating drug effects, since the drugs normally access the tissue via transit through the blood. To begin to address this issue, Mansour et al. 12 differentiated human embryonic stem cells into cerebral cortical organoids and implanted them into the brains of adult severe combined immunodeficient (SCID) mice. The human neurons showed gradual maturation and growth into the host brain tissue. They also found vascularization in the human organoid and formation of neuronal networks both with the organoid and between the human tissue and rodent brain neurons. Thus, for the first time it may be possible to develop human neuronal tissue with its blood supply intact. This will be important when studying the way vascularization could impact neuronal survival, especially in disease; since many of the problems of developing CNS effective drugs concern appropriate bioavailability and penetration of the blood–brain barrier, this model could provide a system to better replicate in vivo drug access to human target tissues.
Beyond testing for compounds that affect neuronal activity, brain organoids have also been developed to test for viral infectivity, infection by Zika virus.81,82 These studies are particularly important because there are few systems that can assess viral infectivity of human brain tissue in vitro in an HTS format. These authors employed a novel brain organoid screening assay to test 6000 compounds for block Zika virus infection and identified a class of cyclin-dependent kinase inhibitors that effectively block cell death caused by the virus. This provides a new approach to develop small-molecule antiviral agents that may be useful in treating several brain disorders caused by virus and other organisms.
Summary
A major advance in drug discovery is the culture of human HiPSC-derived tissues in 3D spheroid1,3,5 or organoid2,6–8 systems that can be used for both drug safety evaluation and drug discovery. The human origin of the cells is an important feature given that many characteristics of human diseases cannot be replicated by nonhuman cultures in vitro or in rodent modeling in vivo. This provides one reason why many drugs that show preclinical efficacy in rodent disease models fail in clinical testing. 31 The advantages of human 3D model systems for drug safety and discovery can be summarized as follows: First, 3D cell cultures provide more architecturally relevant barriers for compounds to traverse than present in 2D cell cultures. This is an important fact since tissue absorption is a major factor in determining efficacy and many compounds have similar access to all cells in flat 2D cultures, resulting in overestimation of efficacy.1,30,31 Absorptive pharmacokinetics is particularly important in developing anticancer drugs, many of which exhibit cytotoxic efficacy on cells cultured in 2D but fail clinically due to lack of access to the inner regions of tumors, where cancer stem cells reside. 38 Indeed, comparison of cells cultured in 2D versus 3D formats shows differential sensitivities to several chemotherapeutic agents. Second, 3D culture systems allow better modeling of the cell–cell interaction seen in tissue than is possible with 2D culture systems. This is most apparent in complex tissues, such as the brain, but also occurs in tissues such as cardiac and gut tissues; here, 3D culture systems have been developed in which neuronal inputs to the organ or organoid are maintained.8,45 These neuronal organ cultures allow for screening compounds at multiple targets, that is, the site of neuronal input, the end organ, and the neuronal organ synapse. An example of this approach is human neuromuscular 3D cultures in which compounds can be screened for effects on the motor neuron activity; the skeletal muscle contractility, independent of the neuronal input; and the muscular junction. Third, in many cases 3D cultures are long lived, in some cases for months. This temporal aspect of cell culture is important when modeling diseases slow to develop, such as neurodegenerative diseases. The relatively long life of the cell culture provides a system to identify pathological defects emerging in the tissue over time.
Nonetheless, while 3D cultures present many advantages in drug discovery, they may also pose hurdles to overcome before their widespread adoption. First, they can be expensive to culture since specialized growth factors are needed to differentiate and maintain iPSCs into specific cell types. This is likely to be less of an issue over time as newer, more efficient methods are developed, particularly as fluid volumes are miniaturized in specialized vessels. Second, developments in imaging and detection techniques are needed to monitor the tissues in 3D culture, particularly as simultaneous detection of cells at the inner core, as well as those at the surface of 3D cultures, will be needed. Finally, an issue in terms of the use of HiPSC-derived cells is that they often express properties of immature cells, notably neurons. However, the ability of 3D cell cultures to survive for several months provides a means to allow maturation in culture and thus study cellular development in vitro.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
