Abstract
Infertile couples needing assisted reproduction are increasing, so a fundamental understanding of motile sperm migration is required. This paper presents an advanced microfluidic device for sperm motion analysis utilizing chemotaxis and thermotaxis simultaneously for the first time. The proposed device is a transparent polydimethylsiloxane- and glass-based microfluidic chip system providing a low-cost, useful, and disposable platform for sperm analysis. The concentration gradient of the chemoattractant (acetylcholine) and the temperature difference are formed along the microchannel. The temperature gradient is generated and controlled by a microheater and microsensor. Thermotactic and chemotactic responses of mouse sperm were examined using the proposed device. Experimental results show that motile mouse sperm are attracted more sensitively under integrated conditions of chemotaxis and thermotaxis rather than individual conditions of chemotaxis and thermotaxis. This sperm analysis device is expected to be a useful tool for the study of mammalian sperm migration and the improvement of artificial insemination techniques.
Introduction
The number of infertile couples worldwide is increasing due to environmental hormones, stress, irregular eating habits, and the trend of delaying childbearing. 1 In particular, male fertility factors are of increasing concern. 2 Couples diagnosed with infertility, that is, who are unable to conceive naturally, can undergo artificial insemination via assisted reproduction technology, including intrauterine insemination (IUI), in vitro fertilization (IVF), and intracytoplasmic sperm injection (ICSI). 3 For these artificial insemination treatments, except ICSI, sperm processing is required to select motile sperm. 4 All conventional sperm separation methods involve separating the sperm from other semen components by centrifugation, which is highly damaging to the sperm. 5 Also, most of these separation techniques are time-consuming and laborious. New methods of efficiently selecting only good sperm from male semen samples of infertile couples are urgently needed. 6 As this demand has emerged, sperm separation and analysis using microfluidics have been studied. 7 Microfluidic devices enable only small amounts of sample to be used, thereby increasing the sperm recovery rate compared with conventional methods. Moreover, microfluidic devices can shorten the analysis or selection time required because the spermatozoa are treated or separated on the microlevel. 8 While the mechanism of internal fecundation has been investigated, the precise processes involved have not yet been clarified. According to recent studies, sperm find the egg via chemotaxis, thermotaxis, and rheotaxis. 9 Thermotaxis describes the ability of a cell to move by sensing a temperature gradient formed by a temperature difference. 10 The spermatozoa that arrive at the isthmus uteri sense the temperature gradient between the uterus and the oviducts, where the egg resides, and move toward the warmer egg position. While the precise temperature at which the egg resides and the temperature difference range are not well understood in humans, the temperature range has been reported to be 1–2 °C in rabbits and pigs. 11 Chemotaxis refers to cell migration that occurs when a concentration gradient due to a concentration difference is formed. 12 When a spermatozoon approaches the egg through the oviduct, it detects a concentration gradient of the chemoattractant secreted by the egg and moves to the egg. In this trajectory, the concentration of the chemical increases gradually. Rheotaxis is a type of taxis caused by fluid flow stimulation that has been shown to play an important role in motile sperm migration.13,14 When the fluid secreted by the oviduct increases, spermatozoa capacitated in the isthmic sperm reservoir swim against the oviductal fluid flow and reach the fertilization site.
To date, microfluidic devices have only separated sperm using sperm self-movement,15,16 laminar flow,17,18 a chemoattractant,19,20 or a thermal gradient. 21 In vivo, however, the motile sperm that reach the tubal isthmus of the fallopian tubes move toward higher temperatures. This movement occurs in response to oviductal fluid flow and the temperature gradient due to the temperature difference between the isthmus uteri and the ampulla of the uterine tube. As fertile sperm approach the region of the oviduct in which the egg resides, the concentration gradient of the chemoattractant is detected and the sperm move toward the egg. Finally, a fertile spermatozoon fertilizes the egg. 22 Hence, it is believed that spermatozoa will achieve higher success rates in artificial insemination if motile sperm are selected under conditions similar to those in the intrauterine environment. 23 However, no studies have been reported on the sperm motion and separation in an environment in which thermotaxis and chemotaxis are combined.
The goal of this study was to develop a device that can analyze sperm migration under conditions as close as possible to the actual fertilization process, representing a substantial improvement over the existing methods. To this end, a novel microfluidic system is proposed in which the temperature gradient and the attractant concentration gradient are integrated. Based on a previous study of sperm separation via chemotaxis, 20 a microfluidic device suitable for sperm migration study was designed and constructed, mimicking the in vivo environment of a long-range temperature gradient and a short-range chemoattractant gradient. Utilizing the combined thermotaxis and chemotaxis approach, sperm motility was then evaluated with a mouse sperm sample in the microchannel of the proposed device. The mouse sperm were experimentally validated to display thermotaxis and chemotaxis. The optimal conditions for attracting capacitated sperm in the microfluidic channel were also examined.
Materials and Methods
Device Design and Fabrication
A conceptual schematic diagram of the system that was constructed to concurrently induce sperm chemotaxis and thermotaxis is shown in Figure 1 . The microfluidic device of the system consists of a sperm migration microchannel chip and an underlying microheater glass substrate. A three-dimensional rendering of the microfluidic device is shown in Figure 2 . The migration has a single microchannel with one sperm sample inlet and one outlet ( Fig. 2a ). Straight channels were adopted so that chemotactic attractant concentration and temperature gradients are generated linearly. As in our previous chemotaxis study, 20 the chemoattractant was injected into the sample outlet of the channel containing the buffer solution. The chemoattractant was then allowed to diffuse, such that the concentration of the chemoattractant was high at the outlet and low at the inlet. The channel is 13 mm long, 500 μm wide, and 100 μm tall. The diameters of the outlet and inlet are 2 and 4 mm, respectively. Given a mouse sperm speed of approximately 1–1.2 mm/min, the channel length was designed so that the sperm can reach the outlet during the experiment time of 13 min.
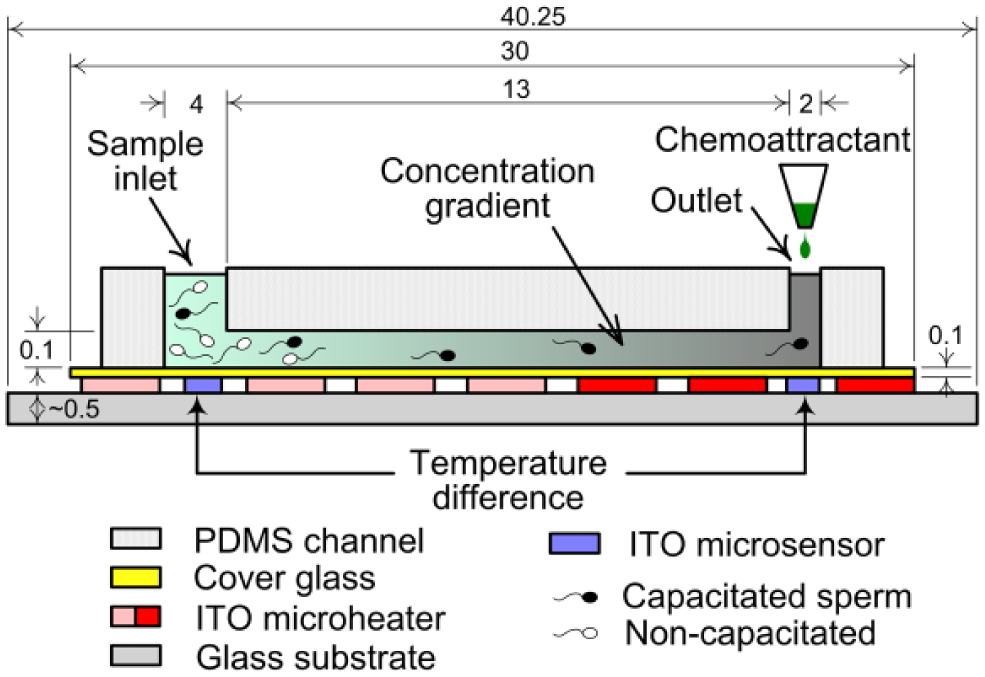
Schematic diagram of the bioenvironmental mimetic integrated system for analysis of motile sperm (dimensions in mm).
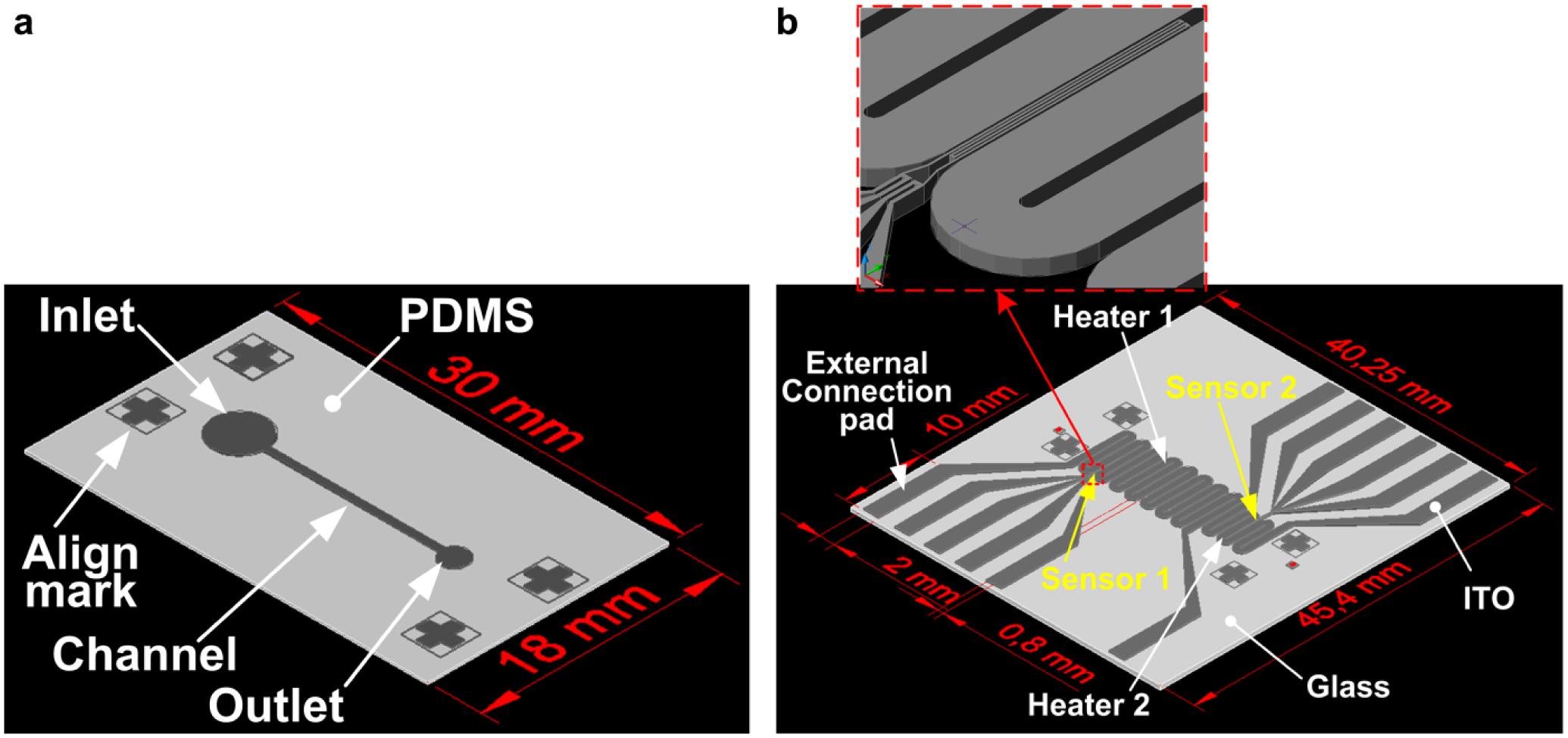
Schematic diagrams of the sperm-sorting microfluidic device. (
The glass substrate has two microheaters and two temperature microsensors ( Fig. 2b ). In order to form a linear temperature gradient in the microchannel from a temperature difference between the inlet (lower temperature) and the outlet (higher temperature), microheaters capable of maintaining different temperatures were placed under the inlet side channel and under the outlet side channel. The microheater is a thin-film resistor that acts as a heater due to the joule heat generated when voltage is applied. In addition, microsensors for measuring the temperature of the buffer solution in real time were placed under the centers of the inlet and outlet. A four-wire-type resistance temperature detector (RTD) was applied as the temperature microsensor. The microheater line was 800 μm wide, and the lines were placed at 100 μm intervals. The microsensor was 20 μm wide, and the microsensors were placed at 20 μm intervals. The total areas of heater 1 (inlet side) and heater 2 (outlet side) were 170.7 and 92.1 mm2, respectively. In addition, a connection pad connectable to the socket was constructed for temperature control.
The microchannel in the sperm migration chip was fabricated with polydimethylsiloxane (PDMS) on a cover glass by a replica molding technique, according to the PDMS–glass hybrid chip (a typical biochip type) manufacturing method.
24
The microheaters and thermal sensors were fabricated using 185 nm thick indium tin oxide (ITO)-deposited glass (Eagle 2000TM, Corning Inc., Corning, NY). The conductivity of ITO is similar to that of metal. Moreover, ITO is transparent and can be used as a heater and a sensor material. The ITO heater and sensor were patterned on the glass wafer by the traditional photolithography and wet etching process. With a 1.5 µm thick mask of positive photoresist (AZ1512, Clariant Corp., Pratteln, Switzerland), ITO thin film was etched at 21.5 °C by wet etchant (LCE-12K, Cyanteck Corp., Kailua, HI). Due to the significant manufacturing cost of the microheater glass substrate, the substrate was designed for repeated use, that is, as a semipermanent device component. The sperm migration chip, which comes into direct contact with the sample, is made of economical PDMS–glass chips to facilitate single use. The disposable sperm migration chip was aligned with the microheater substrate connected to the temperature control system by the alignment marks, after which sperm attraction was performed. Each experiment was performed on a fresh chip. The final fabricated microfluidic device is shown in
Preparation of Sperm and Reagents and Experimental Setup
For sample preparation, mice cervical vertebrae were dislocated. The epididymis was dissected from each dead mouse, after which the caudal epididymis was gently squeezed to release sperm. Sperm cells were dispensed into four 1.5 mL tubes with 0.1 mL of human tubal fluid (HTF) medium and stored at 37 °C. Further details are provided in a previous paper on chemotaxis-based separation. 20 Sperm from six mice were used in the experiments. Acetylcholine was used as a chemoattractant and prepared as in the same chemotaxis paper. 20
Motile sperm attraction via both chemotaxis and thermotaxis was performed as follows: First, to measure the density of the mouse sperm stored in a 1.5 mL microtube, 1 μL of the sample was extracted from the tube, diluted in water, and added dropwise onto a hemocytometer. Spermatozoa were enumerated using a microscope. Of the four total tubes, the tube with the highest sperm concentration was selected and used in the experiment. After identifying the most highly concentrated sample, approximately 15 μL of HTF medium was injected into the sperm migration chip using a micropipette, after which the chip was put onto a microheater and maintained at a specific temperature. The number of sperm injected into the inlet was about 1554 ± 143. For approximately 5 min, the temperature gradient was formed inside the chip and stabilized. After stabilization, 2 μL of acetylcholine solution diluted to a predefined concentration was ejected using a micropipette to form a tiny drop on the tip, and the drop was brought close to the outlet. The acetylcholine solution was then sucked into the outlet by the attractive force of the water molecules. This type of injection method minimizes the fluid flow generated during the injection. If there is a difference in the evaporation rate of the HTF medium at the inlet and the outlet, a pressure difference due to the height difference between the inlet and outlet may develop, leading to fluid flow in the channel.25,26 Hence, after injecting the acetylcholine solution, 0.1 μL of mineral oil (Sigma-Aldrich, St. Louis, MO) was dropped on the outlet using a micropipette to prevent fluid flow by evaporation of HTF medium. The sperm migration chip was then kept on the microheater substrate for 6 min 30 s or 3 min to form a concentration gradient of the acetylcholine (8.125 or 3.75 g/mL, respectively) solution in the channel. After forming the concentration gradient, 1 μL of sperm was injected into the inlet and 0.3 μL of mineral oil was dropped onto the inlet. The sample was then left for 13 min. Finally, the numbers of spermatozoa reaching the outlet were counted using an optical microscope. As a control, HTF medium was injected into the sperm migration chip and left at the appropriate temperature. R software (version 3.4.1) was used for all statistical evaluations of the experimental results. The statistical significance of the difference between experimental results was calculated using the Mann–Whitney U test or one-way ANOVA with post hoc Tukey’s honest significant difference (HSD) test.
Temperature Control System
Voltage was applied to the microheater to generate heat; the temperature reached in the microchannel due to the heater was estimated from the resistance change of the microsensor. A control system is needed to regulate the voltage applied to the heater so that the measured temperature can reach the desired temperature. To utilize an ITO thin-film-type RTD sensor as a temperature sensor, the microheater substrate was first placed in a natural convection oven (OF-02, Jeio Tech, Daejeon, Korea). The resistance value of the sensor was then measured as the temperature was increased from 30 to 90 °C in 10 °C intervals, demonstrating that resistance and temperature exhibit a linear relationship. The temperature control system consisted of a power supply (E3631A, Agilent Technology, Santa Clara, CA) for the microheater, a signal conditioner (SC2345, National Instruments, Austin, TX) that passed constant current to the sensor and treated the signal from the sensor, a computer data acquisition board (PCI-6024E, National Instruments) that collected the signals from the sensor, and a control program (LabVIEW 7.0, National Instruments) that adjusted the output voltage of the power supply by comparing the sensor signal with the setting temperature. The temperature inside the channel was controlled to within ± 0.5 °C of the set temperature value by a proportional integral derivative (PID) controller.
Heat Transfer Simulation
To predict the temperature distribution inside the channel during the operation of the device, a heat transfer analysis of the microheater substrate and the sperm migration chip in the aligned state was performed. A computational fluid dynamics program (CFD ACE+, ESI Group, Paris, France) was used for the heat transfer analysis. The simplified three-dimensional model for the analysis is shown in
The analysis area was modeled as a three-dimensional unstructured grid containing 67,221 total nodes and 99,154 total volume elements. As in the numerical method, the upwind scheme was applied to the space difference of the velocity field and the enthalpy analysis. The calculation used the conjugate gradient squared and preconditioning. The same method was used to calculate the pressure field. For this calculation, the microchannel was assumed to contain water. The convection heat transfer inside the microchannel was considered; natural convection took into account heat transfer between the chip and the outside atmosphere. The heat transfer coefficient between the outside air and the device (consisting of the microheater substrate and the migration chip) was assumed to be 10 W/m2·K. In the simulation, the voltages applied to heaters 1 and 2 were 25 and 14 V, respectively, and the resistances of the heater were 1450 and 590.5 Ω, respectively. The outside air temperature and the device initial temperature were both set to 20 °C. The material properties required for the analysis are listed in
Results and Discussion
Temperature Characteristics of the Microfluidic Device
The results of the heat transfer analysis of the model in which the sperm migration chip was aligned with the microheater are shown in
Figure 3a
. To investigate the temperatures of the sample inside the microchannel, the temperature distribution was analyzed longitudinally along the channel (
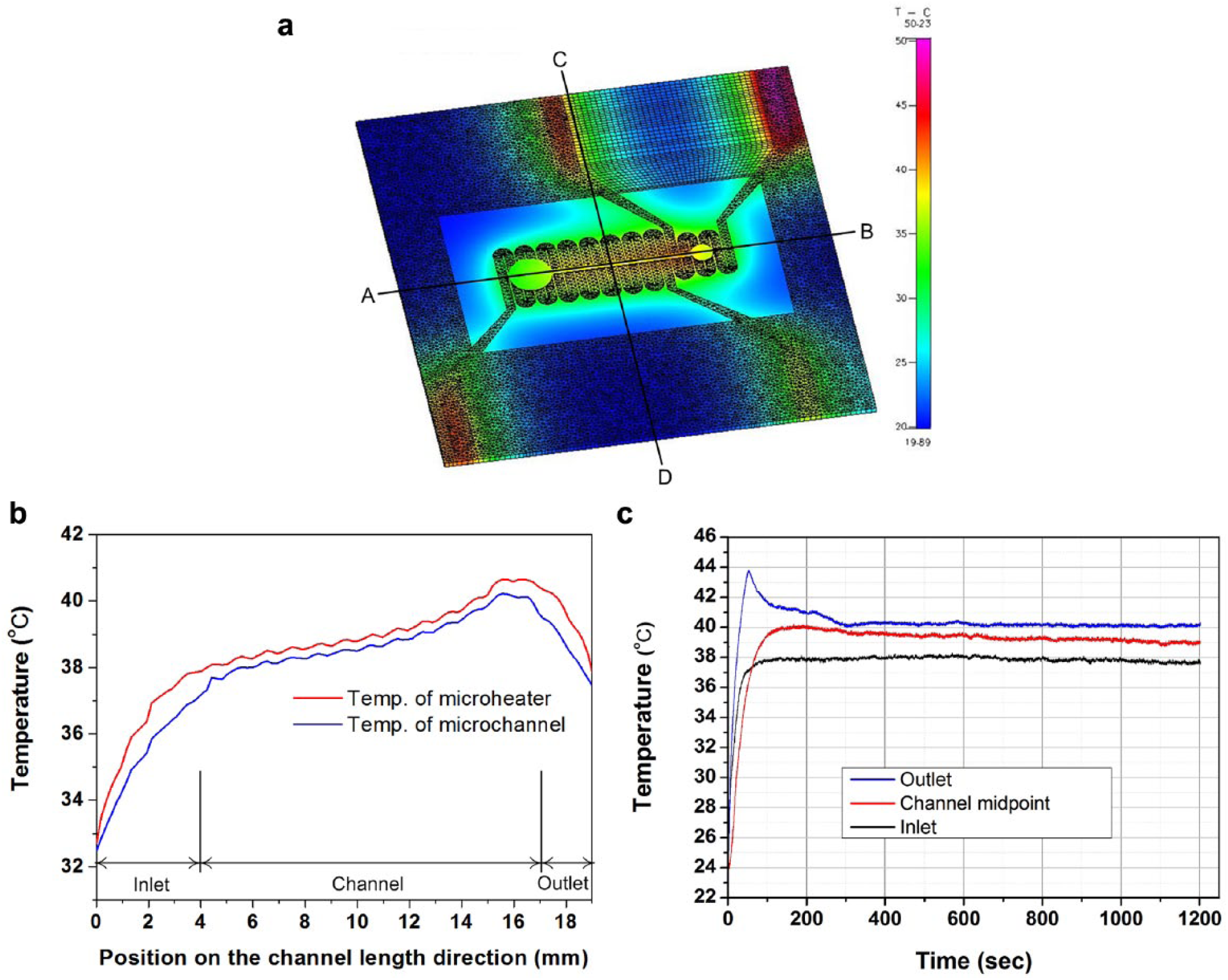
(
An experiment was next conducted to determine whether the temperature inside the sperm migration chip, which is placed on the microheater substrate, forms a linear temperature gradient. As shown in
Thermotaxis Analysis for Motile Sperm Attraction
A photomicrograph of the same position 5 and 7 s after placing the chip containing the PDMS debris on the microheater is shown in
Two different experiments were next performed to determine whether mouse sperm react with thermotaxis to the temperature gradient on the chip. In the first experiment, the temperature of the control sample was kept constant at 36 °C and no temperature gradient was used. For the experimental group, the chip inlet and outlet temperatures were set to 34/36 °C (inlet/outlet) and 36/38 °C. The second experiment was the same as the first, except that the control sample was maintained at a constant temperature of 37 °C and the set temperatures of the experimental were 35/37 °C and 37/39 °C, respectively. Due to the resolution limit of the module reading the voltage signal measured by the sensor, the temperature difference between the inlet and outlet was set to 2 °C. Temperatures of the inlet and outlet were changed by altering the set temperatures with a verified temperature control system ( Fig. 3C ), and 10 replicates of each experiment were performed; the results are shown in Figure 4a . Significantly more sperm in the experimental group were attracted to the outlet with a temperature gradient of 0.154 °C/mm than in the absence of a temperature gradient (p < 0.01, Mann–Whitney U test). These findings confirm that sperm in the mouse samples perform thermotaxis in the proposed device.
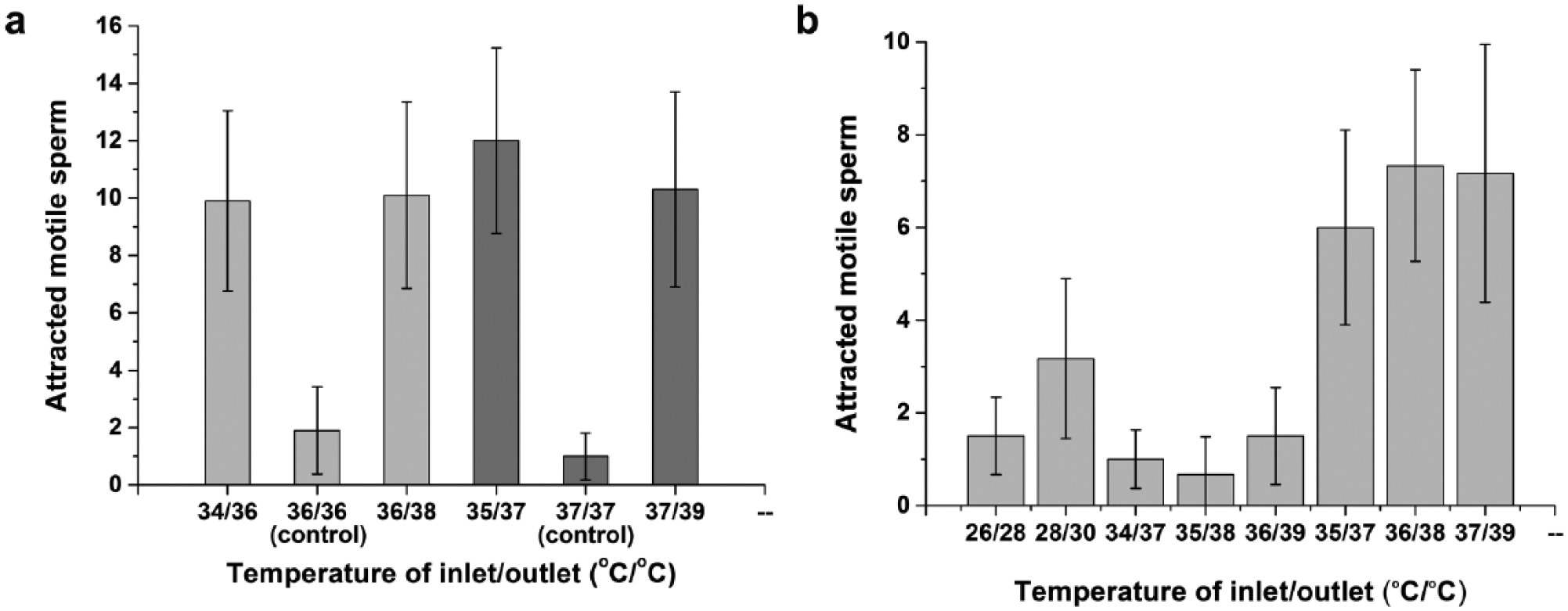
Number of sperm observed at the outlet in the case of (
Next, an experiment was conducted to define the optimal temperature conditions at which mouse sperm show the strongest thermotaxis and at which motile sperm can best be attracted using the temperature gradient formed in the channel. The temperature conditions for the experiment were set so that the temperature difference between the microchannel inlet and outlet was 2 or 3 °C, with temperatures ranging from 26 to 39 °C. Thus, the temperature gradient was set at 0.154 or 0.231 °C/mm, respectively. Since the body temperature of a mouse is about 36.5 °C, the experimental temperature was set based on this value. The eight inlet/outlet temperatures for the experiment were set at 26/28 °C, 28/30 °C, 34/37 °C, 35/38 °C, 36/39 °C, 35/37 °C, 36/38 °C, and 37/39 °C. Six replicates were performed under each condition, and the number of sperm reaching the outlet under each condition was counted ( Fig. 4b ). Significantly more (p < 0.01) sperm arrived at the outlet when the temperature difference between the inlet and outlet was 2 °C (temperature gradient of 0.154 °C/mm) than when it was 3 °C (temperature gradient of 0.231 °C/mm), according to one-way ANOVA with post hoc Tukey’s HSD test. The experiment was also performed when a temperature gradient of 0.308 °C/mm was formed by a temperature difference of 4 °C, but almost no sperm arrived at the outlet under these conditions. Sperm migration due to thermotaxis increases as the temperature difference increases; 27 however, these findings show that when the temperature difference exceeds a certain limit, thermotaxis is actually decreased. These findings suggest that an optimum temperature gradient exists. Further studies should investigate whether the temperature gradient at which the sperm thermotaxis response is highest coincides with the temperature gradient in the mouse body.
When the temperature difference was 2 °C and the temperature gradient was 0.154 °C/mm, more sperm reached the outlet for temperatures of 35/37 °C, 36/38 °C, and 37/39 °C than for temperatures of 26/28 °C and 28/30 °C, all of which are lower than the body temperature range. The difference between these two cases, except between 28/30 °C and 35/37 °C, was statistically significant (p < 0.05) according to one-way ANOVA with post hoc Tukey’s HSD test. While the temperature gradient was the same, more spermatozoa arrived at the outlet when the temperature increased. A likely explanation for this finding is that motile sperm move faster at higher temperatures, 27 thus increasing the number of sperm arriving at the outlet within a given time.
Motile Sperm Migration Assay Incorporating Both Chemotaxis and Thermotaxis
Motile sperm migration experiments involving simultaneous thermotaxis and chemotaxis were carried out. Five different versions of the experiment were performed, including a negative control. The first version (case 1) was a chemotaxis-based test. This experiment was carried out on the novel device in the same manner as described previously in our study of sperm chemotaxis. 20 In the previous chemotaxis study, the diffusion of dye (rhodamine B) whose molecular mass is similar to that of the acetylcholine used here showed a linear chemical concentration profile in the microchannel, and the profile remained linear with time. The sperm separation chip used in the previous study had a microchannel length of 10 mm. The motile sperm were best separated when acetylcholine (6.25 g/mL) was injected into the channel outlet and a concentration gradient was formed by diffusion for 5 min. The microchannel chip made for the motile sperm migration analysis combining thermotaxis and chemotaxis was 13 mm long. Therefore, for the optimum chemoattractant concentration derived from the chemotaxis study to be achieved in the proposed chip, the acetylcholine must be injected at a concentration of 8.125 g/mL and a diffusion time of 6 min 30 s is required. The chemotaxis experiment was carried out with this injection concentration and diffusion time. The second version (case 2) was a thermotaxis-based test. The experiments were carried out at a temperature gradient of 0.154 °C/mm, which is formed when the inlet and outlet temperatures are 36 and 38 °C, respectively. These are the optimal temperature conditions obtained from the thermotaxis experiment. The third version (case 3) involved both thermotaxis and chemotaxis. In this version, the chemoattractant required for chemotaxis was allowed to diffuse only until reaching the middle of the microchannel (6 mm from the outlet). From the outlet to the middle of the channel, the optimum concentration gradient derived from the chemotaxis study should be formed. Therefore, the acetylcholine must be injected at a concentration of 3.75 g/mL and the sample must be kept on the microheater substrate for 3 min. At this concentration gradient and with a temperature gradient of 0.154 °C/mm (formed by 36/38 °C [inlet/outlet temperature], as in version 2), the motile sperm attraction experiment was performed with both chemotaxis and thermotaxis. In the fourth version (case 4), the acetylcholine was injected at a concentration of 8.125 g/mL and allowed to diffuse until reaching the end of the microchannel. In addition, similar to cases 2 and 3, a temperature gradient of 0.154 °C/mm (formed by 36/38 °C) was generated inside the microchannel. The last version (case 5) was a test with the control sample. After HTF medium was injected into the microchannel, 1 µL of sperm sample was injected. The microchannel and sample were then both kept at room temperature (26 °C) for 13 min. In this version, no thermotactic or chemotactic stimuli were given to the sperm. Each version of the experiment was repeated 10 times. The number of motile sperm arriving at the outlet for each version of the experiment are shown in Figure 5 .
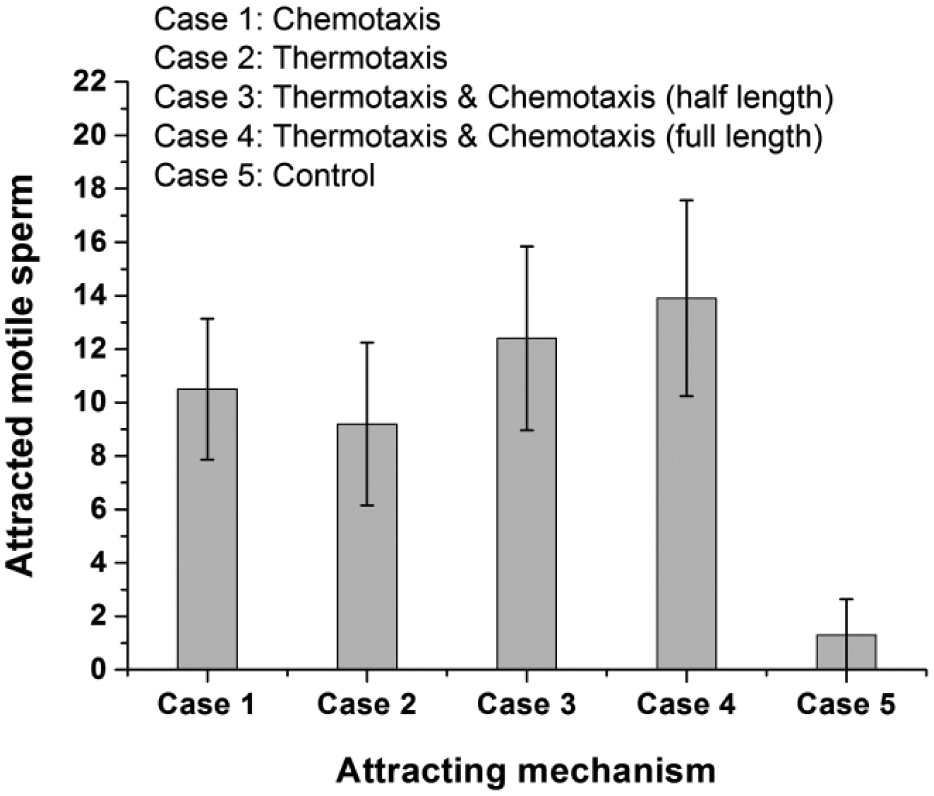
Number of sperm observed at the outlet in the five cases (n = 10, mean ± SD).
The results showed that significantly more sperm arrived at the outlet in all versions of the experiment compared with the control test (p < 0.01, Mann–Whitney U test). This finding confirms that the sample sperm displayed both chemotaxis and thermotaxis. When comparing the results of the chemotaxis test (case 1) and the thermotaxis test (case 2), slightly more sperm were detected at the outlet in the chemotaxis test than in the thermotaxis test. However, this difference was not significant when analyzed by the one-way ANOVA with post hoc Tukey’s HSD test. Although mouse sperm seem to be slightly more sensitive to chemical stimuli (e.g., a concentration gradient of a chemical attractant) than to physical stimuli (e.g., a temperature gradient), this difference was not clear. We could not conclude the thermotaxis effect would be stronger than the chemotaxis effect. For the experiments in which thermotaxis and chemotaxis were performed in combination, slightly more sperm arrived at the outlet when the attractant diffused until the end of the channel than when it diffused until only the middle of the channel. However, this difference was not statistically significant. On the other hand, more spermatozoa were detected at the outlet when thermotaxis and chemotaxis were both performed compared with when the experiment was performed with only thermotaxis or chemotaxis. When the attractant was spread until the channel inlet along with the temperature gradient, significantly more sperm were detected at the outlet than when only thermotaxis was used (p < 0.05, one-way ANOVA with post hoc Tukey’s HSD test). The differences between the other cases were not statistically significant. This finding reflects that the combination of thermotaxis and chemotaxis is more similar to the in vivo environment than when only thermotaxis or chemotaxis occurs. This finding is associated with the fact that when sperm approach the ovum, the sperm begin to receive chemotactic and thermotactic guidance concurrently rather than receiving separate types of guidance in different regions. 22
The acetylcholine used in this study is a putative chemoattractant for mouse sperm. 12 For greater accuracy, confirmed chemoattractants for the target mammalian spermatozoa would be preferable. Because the chemotactic gradient is created by simply applying the chemoattractant at one end of the channel and letting it diffuse, it is not uniform and changes continuously, contributing to lack of precision in experimental conditions. Device technology is needed to provide a more controlled gradient. Analysis of motile sperm from the proposed microfluidic device takes 24 min 30 s. The approximate separation times of conventional sperm processing techniques are as follows: swim-up method, 40 min to 1 h; density gradient centrifugation, 30 min. 28 While conventional methods use approximately 1 mL of semen, it was possible to evaluate motile sperm with only 1 μL of semen. Thus, the experiment time is slightly shorter or similar, but the amount of sample required can be reduced 1000-fold. More importantly is that this method provides a means to select better spermatozoa for intracytoplasmic single-sperm insertion 29 because only capacitated spermatozoa are chemotactically and thermotactically responsive, 30 and capacitation is an excellent reflection of the overall physiological process of spermatozoa. 31 The temperatures and gradients used with this device for the sperm assay do not exactly correspond to actual physiological conditions. In their application to motile sperm sorting, the temperature and gradient should be adjusted to physiological conditions. However, this new device was developed as an assay to help identify the sperm guidance mechanism. Hence, this device is not suitable for motile sperm selection as used in artificial insemination and traditional IVF, which require many more sperm. In this study, only the functionality of the proposed device was verified, so larger numbers of sperm would be needed in the actual sperm analysis.
Conclusion
An integrated system mimicking the in vivo environment was constructed. This system simultaneously utilizes thermotaxis and chemotaxis. To analyze motile sperm by thermotaxis, a microfluidic channel chip and an ITO thin-film-type microheater with a temperature sensor were designed. The feasibility of the design was verified by carrying out heat transfer analysis using a computational fluid dynamics program. The designed chip and heater were fabricated and a temperature control system was constructed to regulate the temperature of the microfluidic chip through a heater and a sensor. The optimum chemoattractant concentration gradient drawn from a previous chemotaxis study was further applied to this microfluidic sperm analysis device, on which thermotaxis experiments can also be performed. To test its performance, a control group was set up and the thermotactic and chemotactic tests were performed together, analyzed, and compared. The analysis showed that utilizing the combination of thermotaxis and chemotaxis was more advantageous than utilizing only one of the motion types to attract motile sperm. In this study, motile sperm were quantitatively and efficiently attracted by using both thermotaxis and chemotaxis, which are properties of capacitated sperm, instead of the conventional highly subjective and empirical methods. For this purpose, the analysis device was precisely miniaturized using microfluidic technology and a micromachining fabrication process. Therefore, the proposed system can be applied to mammalian sperm motility analysis using chemotaxis and thermotaxis and also to motile sperm separation research for IVF.
Supplemental Material
Supplemental_Info_for_Design,_Fabrication,_Testing_by_Ko,_et_al._revised – Supplemental material for Thermotaxis and Chemotaxis Assays of Sperm
Supplemental material, Supplemental_Info_for_Design,_Fabrication,_Testing_by_Ko,_et_al._revised for Thermotaxis and Chemotaxis Assays of Sperm by Yong-Jun Ko, Joon-Ho Maeng, Seung Yong Hwang and Yoomin Ahn in SLAS Technology
Footnotes
Supplemental material is available online with this article.
Authors’ Note
Yong-Jun Ko’s current affiliation is R&D Center, LG Innotek, Ltd., Ansan, Gyeonggi-do, Republic of Korea. Joon-Ho Maeng’s current affiliation is Biometrix Technology Inc., Chuncheon-si, Gangwon-do, Republic of Korea.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Science, ICT and Future Planning (NRF-2015R1A2A2A01006088) and by the NRF grant funded by the Korean government (MSIT) (no. 2018R1A2B6003661).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
