Abstract
A physiologically relevant environment is essential for successful long-term cell culturing in vitro. Precise control of temperature, one of the most crucial environmental parameters in cell cultures, increases the fidelity and repeatability of the experiments. Unfortunately, direct temperature measurement can interfere with the cultures or prevent imaging of the cells. Furthermore, the assessment of dynamic temperature variations in the cell culture area is challenging with the methods traditionally used for measuring temperature in cell culture systems. To overcome these challenges, we integrated a microscale cell culture environment together with live-cell imaging and a precise local temperature control that is based on an indirect measurement. The control method uses a remote temperature measurement and a mathematical model for estimating temperature at the desired area. The system maintained the temperature at 37±0.3 °C for more than 4 days. We also showed that the system precisely controls the culture temperature during temperature transients and compensates for the disturbance when changing the cell cultivation medium, and presented the portability of the heating system. Finally, we demonstrated a successful long-term culturing of human induced stem cell–derived beating cardiomyocytes, and analyzed their beating rates at different temperatures.
Introduction
Cell culturing in vitro is one of the cornerstones of modern biology. It is known that mammalian cells are very sensitive to the properties of their environment; for example, temperature, oxygen concentration, mechanical stimulation, and pH. Therefore, it is crucial to provide a proper microenvironment for successful long-term cell culturing. Furthermore, a physiologically relevant cell culture environment is key for achieving the reliable data required in, for example, stem cell–based disease-modeling studies.1–6
Compared to traditional cell-culturing methods using bioreactors, the use of microfabricated microfluidic systems is a fascinating approach due to its better control over the physiological culturing conditions.7–9 Other advantages of using these devices are their faster response times, lower fabrication costs and power requirements, and smaller reagent consumption. Even though several commercial microbioreactors exist, they are typically expensive, do not provide uniform environmental conditions for cells, and have only a limited number of designs available.10–12 Therefore, there have been many studies on creating cost-effective microbioreactors for cell cultures in vitro. Typically, these devices are fabricated from polydimethylsiloxane (PDMS) using a so-called soft-lithography technique, due to this being an easy, fast, and cheap fabrication method. Furthermore, because PDMS is transparent, biocompatible, and gas-permeable, it is well suited for cell-culturing systems.4,5,13–17
Proper temperature is one of the most important microenvironmental parameters in cell culturing because the intrinsic properties of fluids and cells are affected by temperature variations. Hence, temperature should be controlled carefully to provide optimal circumstances for cell growth and differentiation. 18 Also, temperature has a strong effect on the emergent network activity and membrane potential at the cellular level. 19 Precise control of the environmental temperature was required for microrheology measurements; activity of the cells slowed, and they became softer when the temperature was lowered. 20 Several methods and techniques used in cell culturing, however, cause temperature variations in the culture. For example, cardiac cell research can include such analysis methods as patch clamp, multielectrode arrays, fluorescent imaging, impedance assays, video microscopy, and live-cell imaging, to name a few. 21 During the culturing, it is common to move the cultured cells from one measuring instrument to another, such as from the incubator to live-cell imaging and analysis. Unfortunately, without a proper portable heating system, these movements of the cell cultures can create significant temperature variations within the cultures, resulting in unwanted stimulation to the cells. Therefore, a control system that can minimize temperature variations is very important to successfully culture cells in vitro. Another potential source of temperature variations is changing of the cell culture medium, which is typically performed every 2–3 days. If the medium is not carefully preconditioned, the medium change can cause a temperature stress to the cells.
A typical problem with accurate temperature control is that it requires a precise temperature measurement from the cell culture. This direct measurement brings some challenges; for example, the sensor can interfere with the cells and prevent microscopic inspection. In many cases, it is significantly more difficult to place sensors in the region of interest than elsewhere, such as outside the cell culture chamber. Also, if one is placing the temperature sensor inside the chamber, close to the cells, a larger cell culture chamber is often required.2,10 Therefore, an indirect temperature measurement is preferable to a direct measurement in the cell area.
Temperature sensors have been placed outside the chamber in many cell culture studies. Solutions include placing sensors together with the heating element,22–24 close to the inlet of the chip or the culture chamber,25,26 downstream and upstream from the culture chamber, 27 or next to the device. 28 None of these cases, however, can guarantee the exact temperature of the cell area. Typically reported temperature differences between the measured temperature and the temperature of the cell area have been up to 2−3 °C. 28 This difference is typically too large for cell culture studies.19,21,29 For instance, it was shown that temperature variation between 37 and 39 °C altered cardiomyocyte beating characteristics. 21 Also, the firing rate during up states in the cortical network was modulated when temperatures varied between 36 and 38 °C. 19 To minimize temperature variations, a sensor has been placed in a separate reference chamber;30–32 however, this method requires more space because one chamber is used only for temperature logging. When using only a sensor placed outside, an extremely good insulating system is required for precise temperature control. One demonstrated solution to provide a uniform temperature profile has been to build a large, complex, insulated device in which a water bath surrounds the chamber.19,32,33 Unfortunately, this typically leads to a longer temperature settling time during the heating phase, such as approximately 60 min, 32 or a minimum time of 5 min to change the temperature by 1 °C. 19 Moreover, with this technique, it can still be difficult to avoid excessively large temperature differences between the central part of the chamber and the surrounding parts. 10
Optical methods have also been proposed for measuring temperature locally. Measurement from the cell culture area using infrared cameras is challenging because the PDMS material shields these signals. 34 Fluorescent labels have been used for direct temperature measurement in microfluidic devices.35–37 With this method, fluorescent dyes are mixed into the working fluid. Although the method can be used with glass-based materials, it presents significant problems with porous materials, such as PDMS, because accurate temperature measurements are prevented by the adsorption of dye particles into the material. Some solutions have been demonstrated to overcome this adsorption problem; 35 however, measurement performance is still reduced. Furthermore, typical temperature measurement precision (approximately 2.5 °C at 37 °C 36 ) is not sufficient for cell culture studies.
To solve the aforementioned problems, we previously developed an indirect temperature measurement method to monitor temperature in the cell culture area using a system identification approach. 38 We used a commercial heater and a cell culture device made in-house, and we showed that our method precisely estimated the cell culture temperature. We also presented a simulation study to demonstrate how the method could be used for temperature control purposes. In this study, we extend this approach by integrating an indirect temperature control into a developed cell culture system. The control method combines an external temperature measurement and a numerical model to calculate an approximation of the temperature at the desired location. This estimate is then compared to the desired temperature set-point, thereby providing an indirect measurement and control method for the cell culture temperature. Furthermore, the developed heating system is portable; it has a power source to maintain the desired temperature for more than 1 h for applications in which transportation of the system is needed. The system is also suitable for long-term microscopy and cell imaging, because it includes a gas supply for the maintenance of the proper carbon dioxide (CO2) and oxygen (O2) concentrations in the cell culture media.
In this study, we show how accurate temperature measurement in the cell culture area can be performed without disturbing the cells or preventing imaging. First, we demonstrate how the heating system compensates disturbances arising from the ambient room temperature variations. We also illustrate how the cell culture temperature is restored to the desired level after opening of the device or liquid change. Finally, using beating cardiomyocytes, we demonstrate successful long-term cell culturing and a temperature stress study.
The rest of the article is organized as follows. First, we explain the working principle of the system, describe details on the experimental setups used, and present developed models. Next are experimental data and control results: the models are developed, their performances are compared to the measured data, and finally experiments with the developed closed-loop control system are presented. Then, we discuss the results from this article, before providing conclusions and possible future work.
Materials and Methods
The main components of the cell culture system are an indium tin oxide (ITO) plate as a heating element (70 × 70 × 0.7 mm boro-aluminosilicate glass plate from UniversityWafer, Inc., Boston, MA, USA, with a resistivity of 8–10 Ω
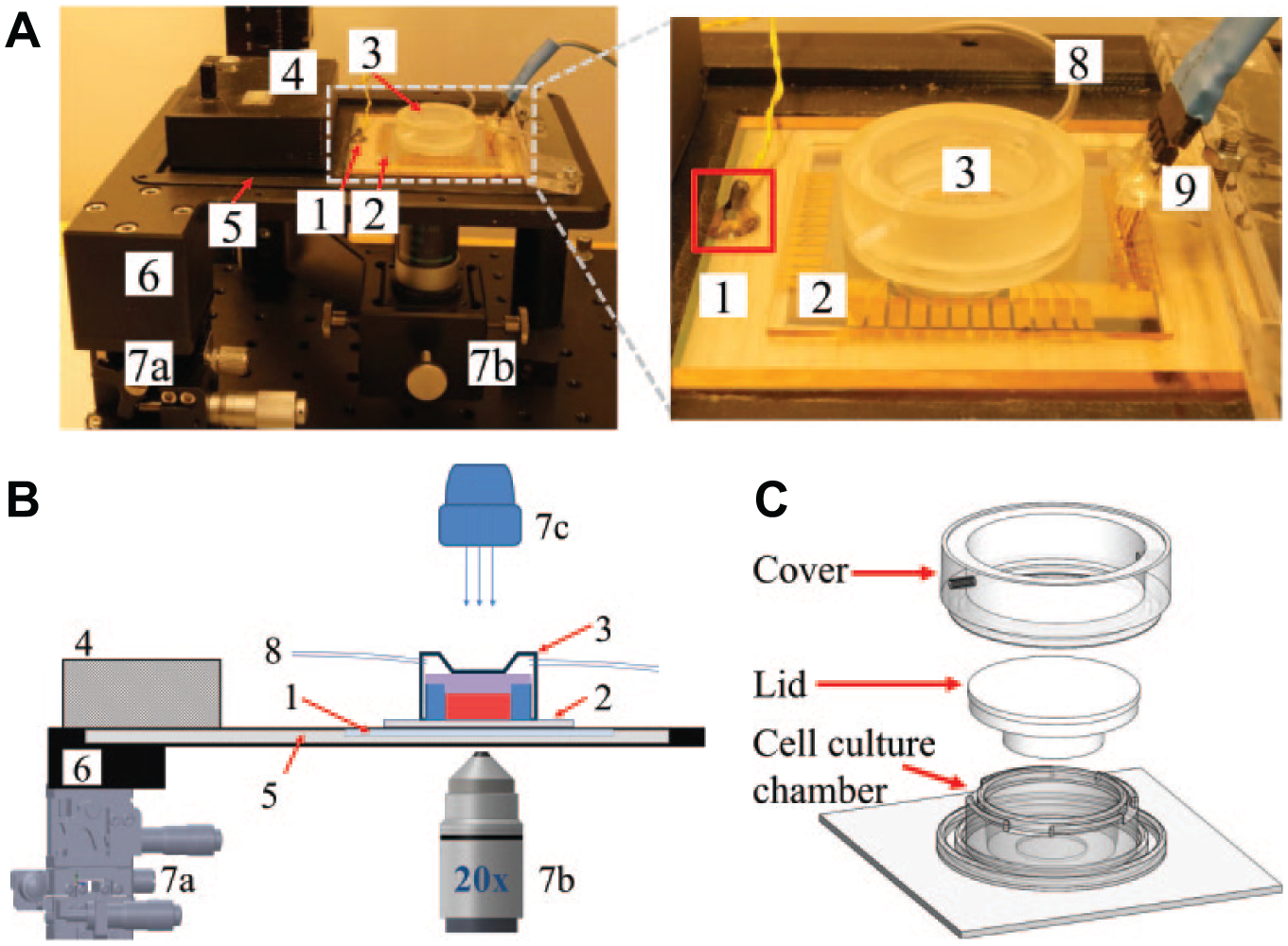
A portable microscale cell culture system. (
Working Principle of Indirect Temperature Control
The temperature estimate is made using a mathematical model and a temperature measurement; the measured temperature outside the desired location (
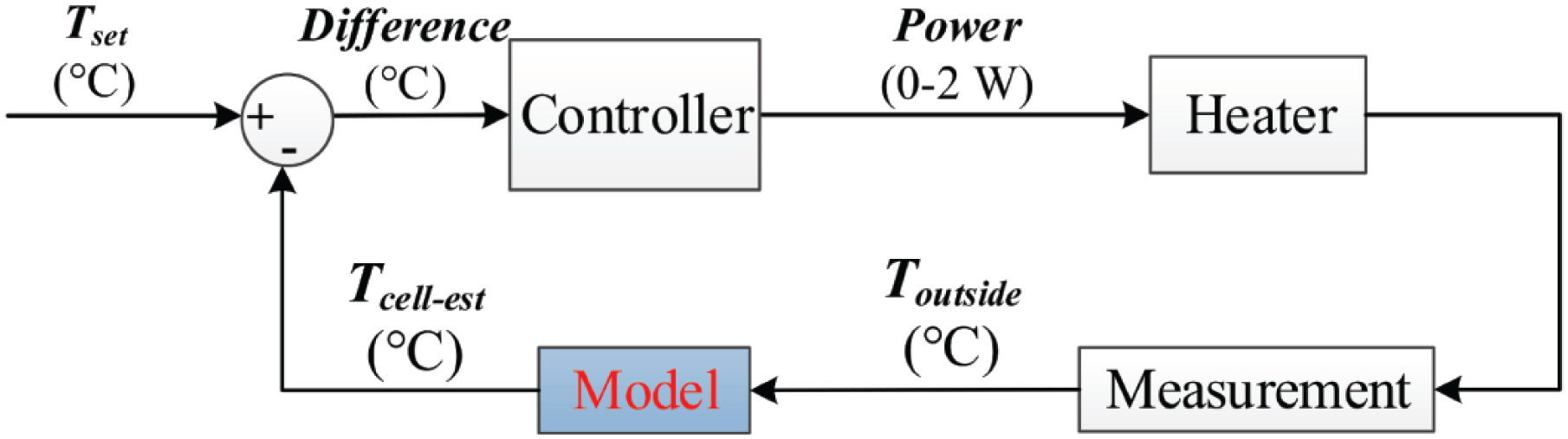
Working principle of indirect cell culture temperature measurement and control.
We developed two temperature estimation models for different purposes. The difference between the models was that different measurements were used as
Temperature logging: (
The sensor required in Model 1 is easy to place, and the location of the sensor can be chosen rather freely. Also, there is no need to use the sensor plate for temperature control. Because this method does not measure temperature inside the cell culture chamber, however, the estimated temperature is less accurate. Therefore, Model 1 is not capable of fully compensating for temperature changes inside the chamber caused by, for instance, variations in the ambient room temperature. Furthermore, this approach does not work properly in cases when the temperature inside the culture chamber changes without changes in
For the aforementioned reasons, we also developed Model 2, which measures the temperature inside the chamber close to the cell culture area and therefore can better predict temperature changes in the cell area. Model 2 can compensate for the residual thermal disruptions caused by microscopy imaging and changes in the ambient temperature, both of which affect the cell culture temperature. The drawbacks of this approach are that the sensor is typically more fragile, the sensor is more difficult to position because it needs to be in a certain location, and the sensor plate is required. To summarize, the two models have their preferred applications depending on the requirements.
Experimental Setups
Overall Device Description
The measurement system included the ITO heater, TSP, PDMS-based cell culture device, gas supply, illumination and optics, frames and stages, and electronics required for temperature measurement and control. We designed all the custom-made mechanical parts in the setup using SolidWorks (SolidWorks, Cambridge, UK). The setup is shown in Figure 1 , which also includes the cell-imaging arrangement. The cell imaging was carried out using an invert-upright convertible compact microscopy system as previously described. 39 In our case, prior to time-lapse video microscopy (20×, 1.3 MP camera BFLY-U3-13S2M-CS, Point Grey Research, Inc., Richmond, BC, Canada), initial sample positioning and coerce focusing were performed using the system’s manual xyz stage. Precise focusing and timely focus corrections in long-term microscopy were carried out with the motorized focusing stage. A custom-written MATLAB (MathWorks, Inc., Natick, MA, USA) user interface software was implemented for controlling the illumination, stage heater, and time-lapse data logging (uncompressed .avi videos with a frame rate of 50 frames per second and temperature sensors). 39
Electronic circuits made in-house (
Although the diagram shows only one channel, the device has two channels, allowing the user to easily switch between two different temperature feedback locations. This simple arrangement designed for the Pt100 sensor was not sufficient for the TSP sensors, because they can have significant and varying wiring and contact resistances. To support the use of the TSP sensors, the circuit was modified to a four-wire measurement compensating for the wiring resistance, as shown in
To minimize the interference caused by the heating element current, a constant-current drive scheme was adopted (
The actual controller was built around an 8-bit microcontroller (ATMega328, Microchip Technology, Inc., Chandler, AZ, USA), which measures the sensor resistances, performs simple gain and offset compensations, and converts the results to temperatures either by using a standard (second-order) Pt100 equation or by using a user-entered, first-degree polynomial fit. This temperature reading is supplied either directly or through the identified model to the PID controller. The temperature measurement, identification, and PID controller run at a 5 Hz frequency. The device has a USB port for configuring the device and for logging temperature data. It also contains a 3.1 Ah, 18650-size lithium ion battery and an associated USB charger (LTC4098, Analog Devices).
Heating System
A heating system that provides a uniform temperature profile is very important for successful indirect temperature control and long-term cell culture studies. Also, optical microscopy is typically required in these studies. Therefore, an ITO plate was chosen as a heating element, because it is not only electrically conductive but also optically transparent; Joule heating can be generated when electrical power is applied, and at the same time, ITO does not obscure illumination. To supply the heating current uniformly over the ITO plate, and thus to equalize temperature on top of the plate, we e-beam evaporated copper pads 900 nm thick (3.5 × 70 mm) onto the ends of the ITO plate. These copper lines are shown in Figure 1a . Furthermore, a 300 nm silicon nitride (Si3N4) insulator layer was deposited over the ITO using a plasma-enhanced chemical vapor deposition (PECVD) process.
Frame Design
A proper design of the frame (see
Fig. 1
, item 5) for the ITO plate is important for achieving a uniform temperature profile. We tested three different frame structures. Frame 1, manufactured by Saloteam Oy (Salo, Finland), was made of aluminum and mounted to an xyz stage (see
To compare the temperature distribution in the different frames, we used thermal imaging. Because direct thermal imaging of the ITO glass was not possible due to reflection issues, we placed a glass plate (50 × 50 × 0.5 mm) on top of the ITO plate and only considered temperature measurement on this area of the plate (marked with dashed rectangles in
It is clear from
Temperature Sensor Plate
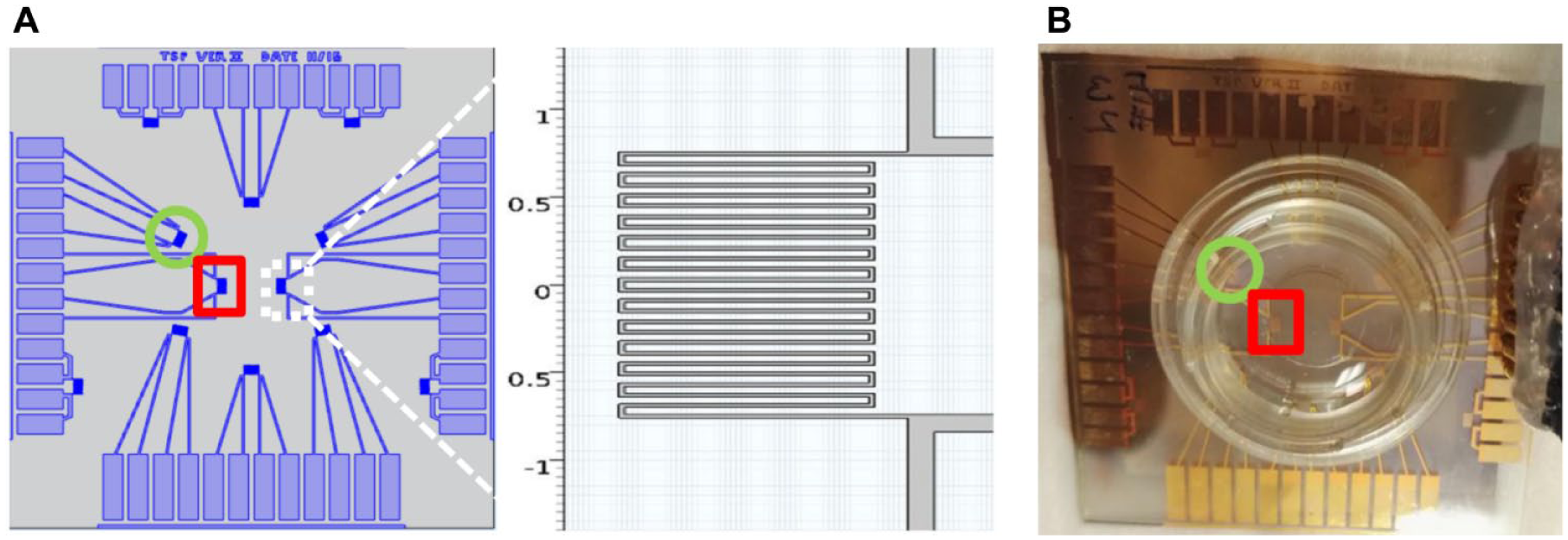
We designed the TSP for logging the temperature inside the cell culture chamber. The TSP was built on a glass plate and included 14 identical temperature sensors, as shown in Figure 3 . The TSP consisted of resistors, tracks, and contact pads patterned using photolithography on an e-beam evaporated copper layer that was 275 nm thick. The resistor and the track area were electrically insulated using 100 nm silicon dioxide (SiO2) and 500 nm Si3N4 layers with PECVD. The width of the resistor line in the design was 20 µm.
Temperature sensors were calibrated in a temperature-controlled oven by measuring their electrical resistances using a four-wire method at several different temperatures from 24 to 38 °C after the plate was thermally stabilized. A typical room-temperature resistance was approximately 105 Ω. Because a good linear relationship between electrical resistance and temperature was obtained, a linear interpolation was used to estimate temperature from the measured resistance.
Cell Culture
The cell culture device, its fabrication procedure, and the main working principle have been described previously.39–41 Therefore, we present here only the main steps and details of study-specific differences. The design goals were to enable online microscopy (therefore, transparent materials were chosen) and to keep the cell culture alive for several days in the cell culture device.
The structure of the cell culture device is shown in Figure 1c with three main parts: a cell culture chamber, a lid, and a cover. The lid was machined from polycarbonate (PC; Saloteam Oy), and the cover was 3D-printed (Shapeways, Eindhoven, the Netherlands). The lid made a watertight seal on the culture chamber and prevented contamination from entering the chamber. This setup also enabled the use of a dry gas supply without significant evaporation of the culture medium. The same technology has been shown to keep the cell culture alive and vivid outside an incubator for 3 days. 41
The cell culture chamber was cast in-house from PDMS (Sylgard 184, Dow Corning, Auburn, MI, USA) using standard soft-lithography techniques. Cells were plated on a round opening area (diameter of 10 mm) that was punched in the bottom of the chamber. During experiments, the cell culture chamber was reversibly bonded on the TSP and filled with 1 mL deionized water or cell culture medium before the chamber lid was closed. The desired gas environment is provided to the cell area by placing the cover on the cell culture chamber. With the cover placed atop the lid and the chamber, the gas supply pipe was connected to provide the desired gas environment (CO2 and O2 concentrations) inside the chamber. In cell culture studies, we used a motorized inverted microscopy system, made in-house, 39 for cell imaging as described previously.
Cardiomyocytes (CMs) derived from the human induced pluripotent stem (iPS) cell line UTA.04602WT, as described previously, 42 were cultured in the developed system. Differentiated beating iPS-CMs were used in the present study. The iPS cells, derived from a healthy person, have been differentiated into CMs with the standard method used in our laboratory. The differentiated iPS-CMs have been characterized with multiple molecular biology and functional methods, and the characterization data have been published in our earlier studies.43–45 For sterilization, TSPs were immersed in 70% ethanol and dried under sterilized conditions. The PDMS culture chamber was mounted directly on the sterilized TSP. The beating iPS-CM aggregates were plated at the bottom of the cell chamber, which was first hydrophilized with fetal bovine serum (FBS) and then coated with 0.1% gelatin type A (Sigma-Aldrich, St. Louis, MO, USA). The iPS-CMs were cultured in knockout Dulbecco’s modified Eagle’s medium (KO-DMEM; Lonza, Basel, Switzerland) with 20% FBS (Lonza), 1% non-essential amino acids (Cambrex, East Rutherford, NJ, USA), 2 mM GlutaMAX (Invitrogen, Carlsbad, CA, USA), and 50 U/mL penicillin/streptomycin (Lonza). For each cell chamber, three to four iPS-CM aggregates were plated. After plating, the iPS-CM aggregates were cultured for 24 h in an incubator (37 °C, 5% CO2, 19% O2, 76% N2) for initial stabilization. After that, the device was removed from the incubator, closed with the lid and the cover, and placed on the preheated (37 °C) ITO heater. The gas environment around the cell culture area was created by flushing with a gas mixture (5% CO2, 19% O2, 76% N2) at a constant flow rate of 5 mL/min.
Temperature Estimation Models
In the study, we used a so-called black-box technique to develop models using only input and output data, regardless of the physical system. 46 To develop our models, we used a prediction error 47 that made a prediction that is as close as possible to the true system if it was known. We compared models using a fit number, which is based on a normalized root-mean-square error criterion. It can be calculated (as a percentage) using the following equation: 48
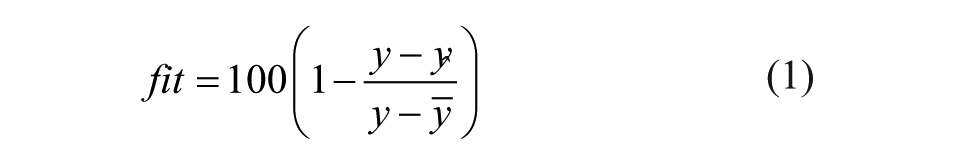
where
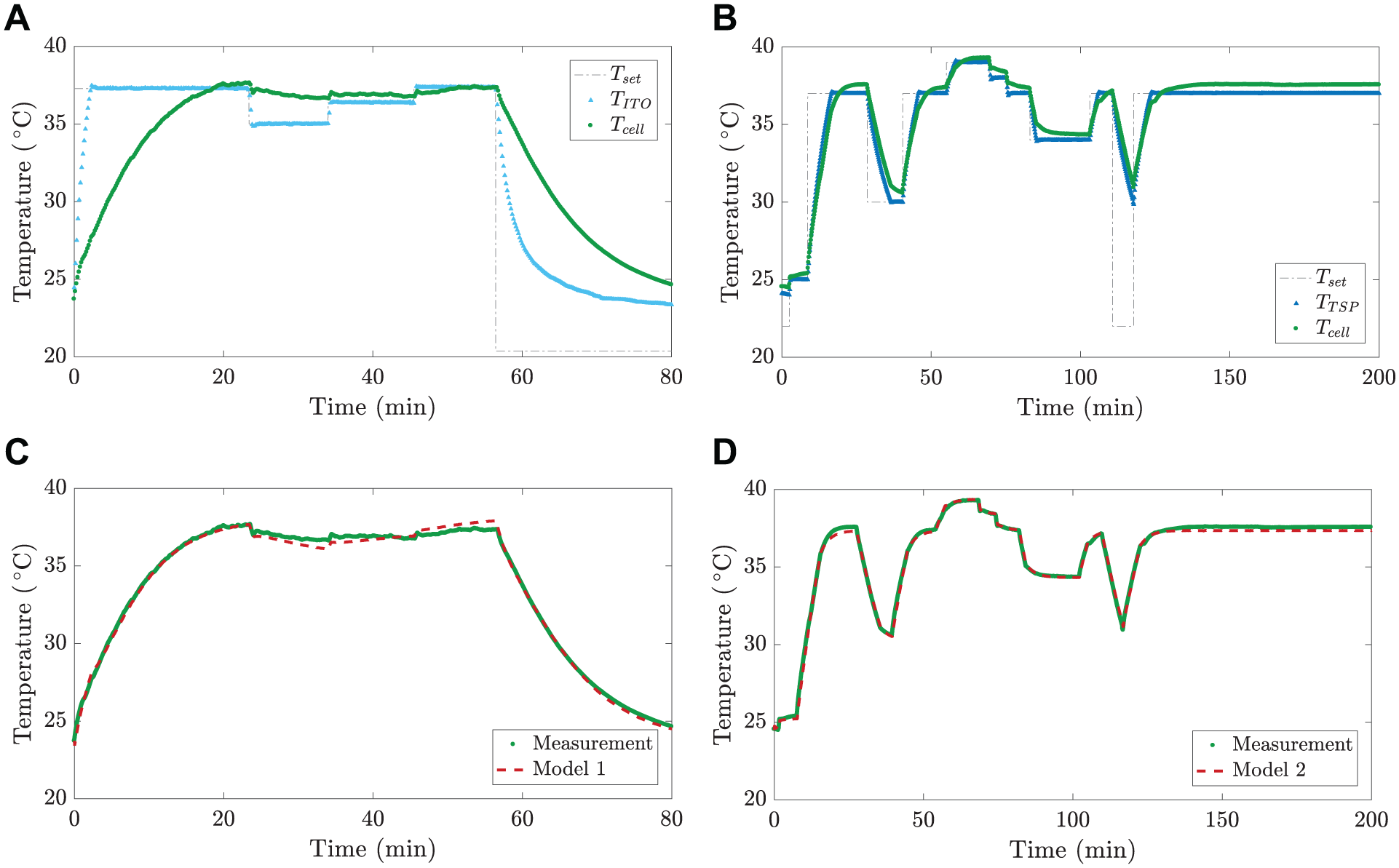
Model development: model estimation experiments for (
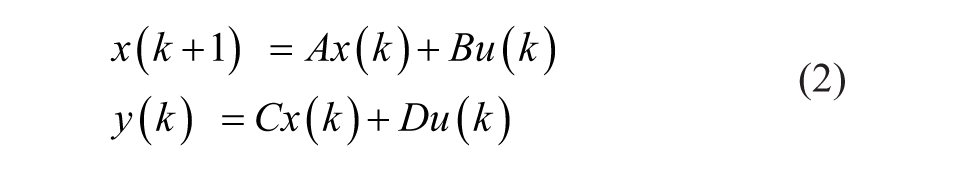
where matrices
Results
Model Development
To develop the temperature estimation models, temperature was controlled using
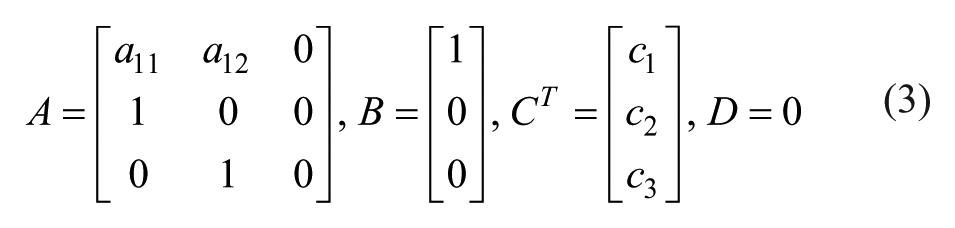
Because the coefficient of matrix
Model 1:
Model 2:
The measured temperatures and the temperatures simulated using the models with the aforementioned parameter values are compared in
Figure 4c
and
4d
. The calculated model fit numbers were 94.2 and 94.8% for Model 1 and Model 2, respectively, indicating that the models are suitable for estimating the cell culture temperature. In the following sections, we use the outputs of these models—the estimated temperatures (
Comparison of Different Control Strategies to Maintain Constant Temperature
In this section, we compare different controller strategies to illustrate the benefits of the developed indirect control system. As mentioned, to obtain optimal cell growth and differentiation, precisely controlled temperature is required. Therefore, the purpose of this experiment is to show how variations in the ambient room (marked as
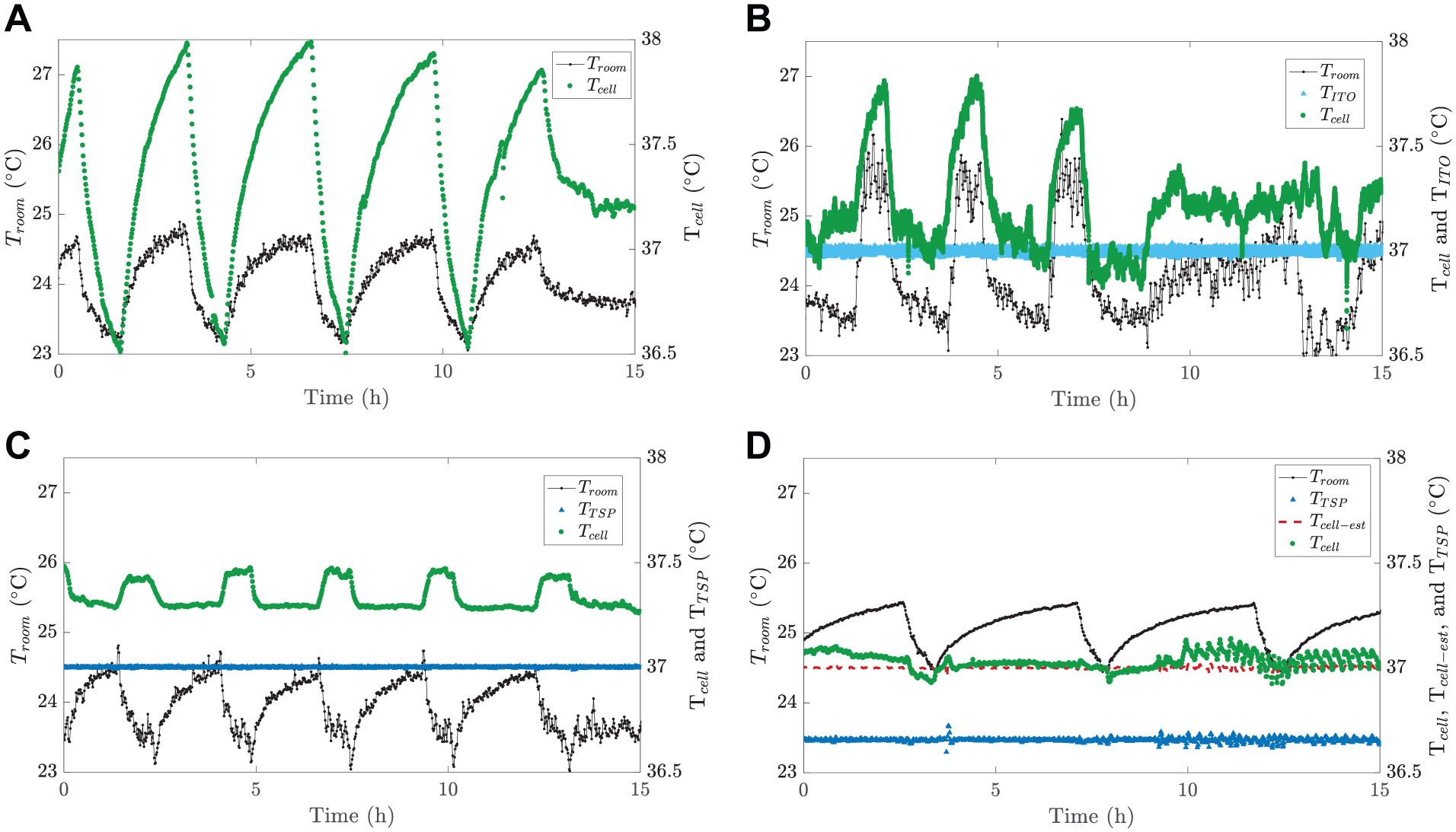
Comparison of the different temperature controller strategies: (
In the open-loop system, a constant heating power was used; no measurement was used to control the heating power. Three closed-loop systems used the same PI controller (
At the beginning of each experiment, we added 1 mL deionized water to the cell culture chamber and preheated the system so that
Precise Long-Term Temperature Control
The indirect control system provided very good results, as presented in the previous section. Therefore, a long-term temperature control test was performed. The purpose is to support long-term cell growth by precisely regulating the cell culture temperature to 37 °C. We again added 1 mL deionized water to the cell culture chamber and calculated
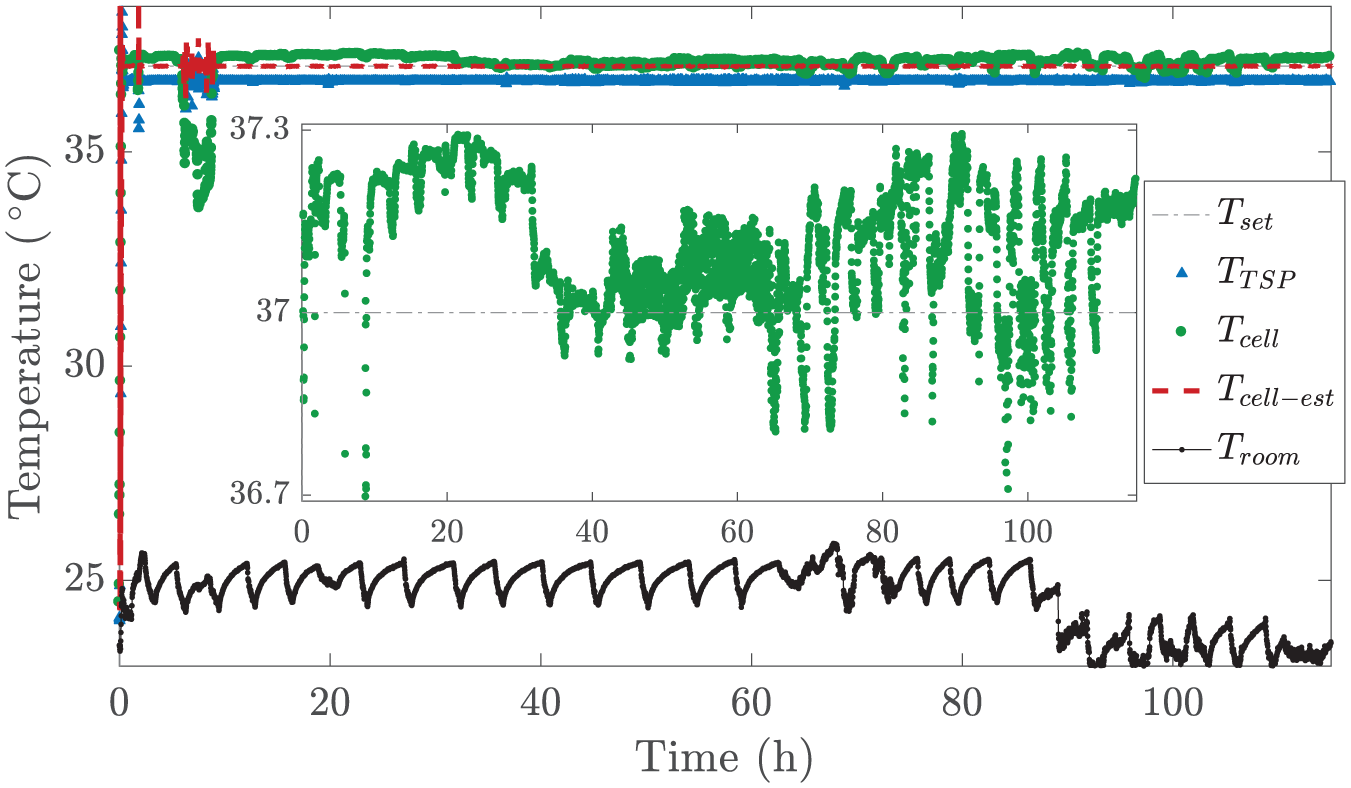
Long-term temperature control using Model 2. Inset presents only
Controlled Temperature Steps
A precisely controllable cell culture temperature provides several opportunities for temperature-dependent cell behavior studies. For example, it enables one to characterize transient behaviors of the cell cultures during the heating and cooling phases around the physiological temperature,
2
or to determine a temperature threshold for the activation of ion channels.
49
Our system can precisely monitor and control cell culture temperature
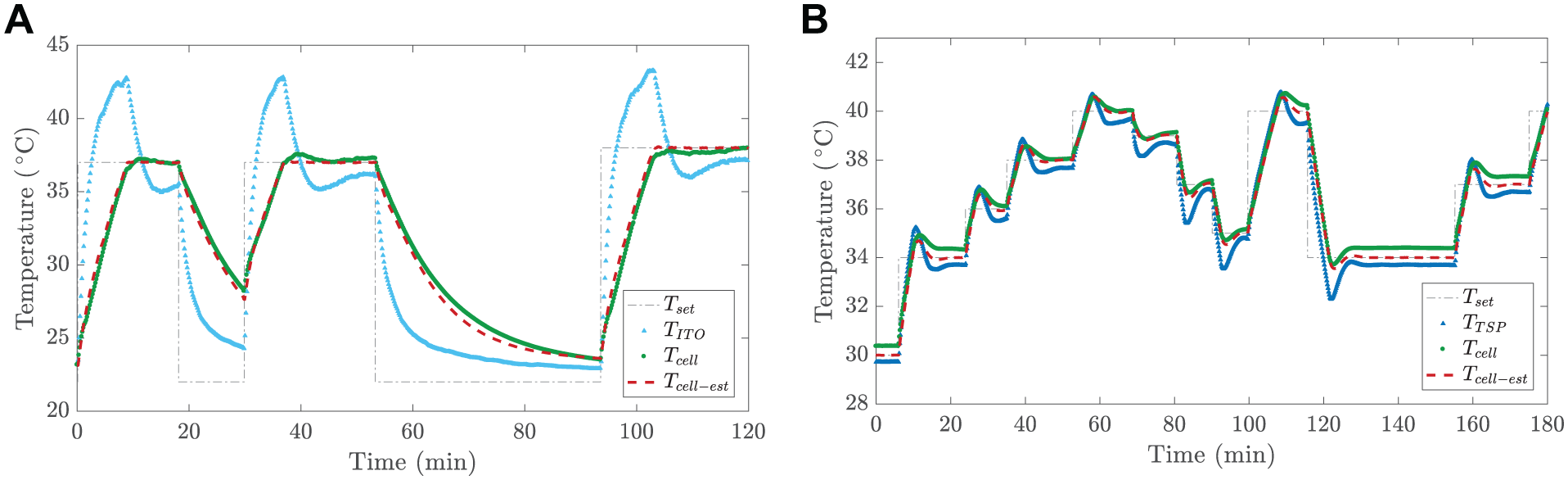
Transient temperature control using (
Disturbance Compensation during Liquid Changes
The performance of the temperature control system during a liquid change, mimicking the change of a cell culture medium, is demonstrated using Model 2. Here, we changed liquid that was heated to 37 °C with liquid stabilized to the ambient air temperature (approximately 24 °C). This is an extreme case; in a typical application, the fresh cell cultivation liquid would be close to 37 °C. Therefore, this demonstration overemphasizes the effect of a typical temperature drop, thus imposing a higher requirement on the heating system. Because Model 1 uses the temperature measured from the ITO heater, the result with Model 1 would not have been satisfactory, because the temperature mainly changes inside the cell culture chamber.
The results (
Portable Heating System
In the case of the portable heating system, which would be beneficial while moving the device, the temperature control was directly based on
Cell Experiments
To demonstrate the capability of the system for long-term cell culturing in vitro, the iPS-CM aggregates were cultured in the system, and the beating behavior of the iPS-CMs was assessed for more than 100 h. We used Model 1 for temperature control and the imaging system presented in
Figure 1
. We recorded 60 s videos with a frame rate of 50 frames per second, once a day, starting 24 h after the cells were initially plated to the device. The iPS-CMs remained functional when cultured in the system; analyzed beating rates on the first day of culturing and 110 h later were 44 and 36 beats per minute (BPM), respectively, using a video image-based method. This non-invasive method, presented by Ahola et al.,
44
has proved to be a reliable and fast approach to monitor the mechanical beating behavior of cardiomyocytes.
21
Snapshot images of the initial and final videos are presented in
Another set of the iPS-CM aggregates was exposed to different temperatures. We varied temperature between 37 and 25 °C in several steps. We waited 25 min before a video recording to stabilize the cell culture temperature. After the experiment, using the same video image-based analysis, 44 we calculated the beating rates at different temperatures. The results are shown in Figure 8 . As can be seen in Figure 8a , we were able not only to change the beating rate of the cardiomyocytes but also to recover the beating rate when the temperature was returned to 37 °C. The calculated average beating rate at 37 °C was 54.8 ± 3.2 BPM based on 11 measurement points presented in Figure 8b . Furthermore, average beating rates at 35 and 34 °C were 44.5 and 36.0 BPM, respectively; however, it should be highlighted here that these latter results are from only four measurement points, and more comprehensive studies are required.
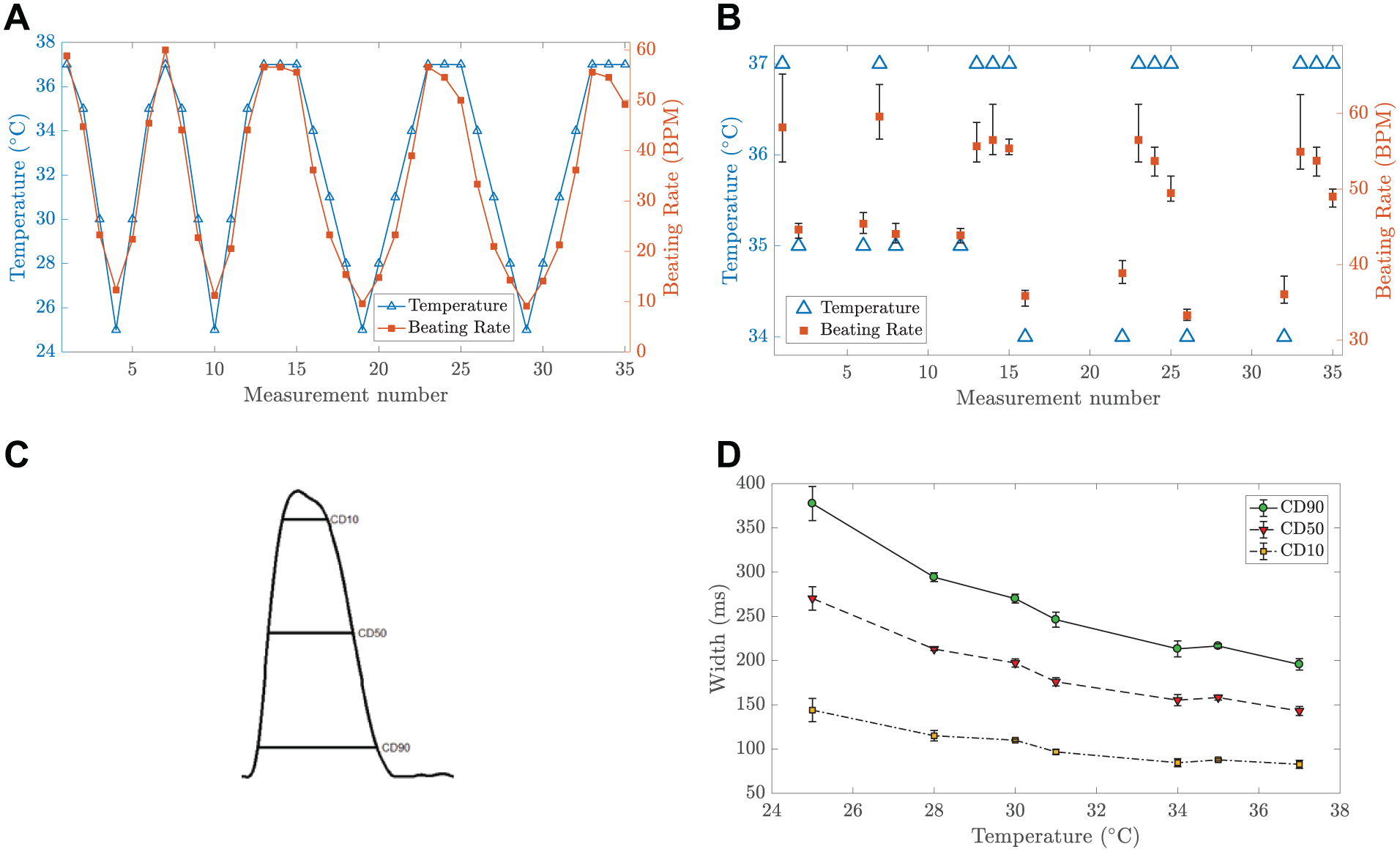
Study of beating cardiomyocytes at different temperatures. (
Using the recorded videos, we further parameterized the contractile motion of the iPS-CM aggregates using the previously presented method with a Fridericia QT correction.50,51 In brief, the motion is characterized by three parameters related to the widths of the contraction transient peaks at different heights, as presented in Figure 8c . The widths are defined at the heights of 10%, 50%, and 90% from the transient maximum. The results shown in Figure 8d demonstrate the temperature dependence of the contractile motion. The results also show that cells return to their normal beating mechanics after the stress induced by the temperature change. Finally, these results highlight the importance of maintaining a stable temperature during cell experiments, because temperature fluctuations influence the contraction durations.
Discussion
We demonstrated a microscale cell culture device together with a unique indirect temperature control method. To our best knowledge, this is the first time that the cell culture temperature has been precisely maintained and regulated without measuring the temperature directly from the cell culture area. Especially, our system can precisely control the cell culture during temperature transients without placing a sensor inside the cell culture area; this has typically not been possible in the systems used in previous studies. As a summary, we have demonstrated that the developed system can precisely control the cell culture temperature when (1) ambient room temperature is changing, (2) the system is moved, (3) the cell culture device is opened, and (4) liquid is changed, all without direct measurement. Furthermore, the system is suitable for temperature-dependent cell behavior studies; this was demonstrated by studying the beating iPS-CM aggregates.
Our system maintained the temperature at 37 °C for more than 4 days. The measured temperature variation, ±0.3 °C, was similar to that in other studies; for instance, variations of ±0.2 °C,27,31,32,52,53 ±0.25 °C, 18 ±0.26 °C, 10 ±0.3 °C, 54 ±0.4 °C, 55 ±0.5 °C,56,57 and ±0.8 °C 58 have been reported. Because the intrinsic properties of fluids and cells are temperature-dependent, it is highly beneficial to prevent large temperature variations in the cell studies. For this reason, it is crucial that the heating system can compensate for typical temperature disturbances related to the cell cultures; for example, variations in ambient room temperature, opening of the device, and changes of medium. Unfortunately, this is typically not possible with the previously presented devices without direct measurement from the cell culture area, whereas we demonstrated that these disturbances can be indirectly monitored and compensated for using the developed heating system. This allows us to minimize the undesired stimulation of cells due to the temperature variations. In addition, we presented the portability of the system. With the battery-operated heating, a constant cell culture temperature can be maintained for about an hour, for instance while moving the device or during live-cell imaging.
With the slight temperature variation of our heating system, a successful cell culture could be carried out. We demonstrated this by culturing the beating iPS-CM aggregates for more than 4 days and using the video image-based beating rate analysis. We also combined this analysis and controlled temperature variations to demonstrate how the beating rate varied at different temperatures. We achieved two main results from this experiment; first, we were able to return the beating rate when the temperature was set back to 37 °C, as is shown in
Figure 8a
. Second, even though more tests are needed, the initial results in
Figure 8
clearly show the importance of the precise temperature control. For example, if we consider the closed-loop heating system based on the direct control of
Conclusion
We developed a portable microscale cell culture system device, including a precise heating system based on an indirect control method. The method combines a numerical model and a temperature measurement to maintain and control the temperature in the desired area, without the need to place a sensor in that area. Using the method, we precisely maintained a constant temperature throughout 100 hours. We also presented accurate temperature control during temperature changes. The model-based control system was also able to compensate for temperature disturbances caused by variations in the ambient temperature, for instance during liquid changes. Furthermore, we cultured the beating iPS-CM aggregates in the developed system for more than four days. The system was capable of providing a stable and cell-friendly environment for the cell cultures. In addition, a temperature-dependent beating rate was demonstrated with the iPS-CM aggregates.
One future step is to implement oxygen sensing in the system, similar to that presented previously. 61 Integration of a zigzag-shaped heater and a sensor on a single ITO plate could be possible through conventional photolithography methods.55,62 Cell cultures in vitro in this study were on a plane. Unfortunately, two-dimensional culture models poorly mimic tissues in vivo. 63 In the future, designing a three-dimensional cell culture environment would offer a biologically more relevant in vitro model and would better reproduce in vivo culturing conditions and cell–cell interactions.
Supplemental Material
DS_TECH768710 – Supplemental material for A Portable Microscale Cell Culture System with Indirect Temperature Control
Supplemental material, DS_TECH768710 for A Portable Microscale Cell Culture System with Indirect Temperature Control by Antti-Juhana Mäki, Jarmo Verho, Joose Kreutzer, Tomi Ryynänen, Dhanesh Rajan, Mari Pekkanen-Mattila, Antti Ahola, Jari Hyttinen, Katriina Aalto-Setälä, Jukka Lekkala and Pasi Kallio in SLAS Technology
Footnotes
Supplemental material is available online with this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Tekes, the Finnish Funding Agency for Technology and Innovation (Decision no. 40346/11), the Finnish Culture Foundation, the Finnish Foundation for Cardiovascular Research, and the Pirkanmaa Hospital District, and was carried out within the Human Spare Parts 2 project.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
