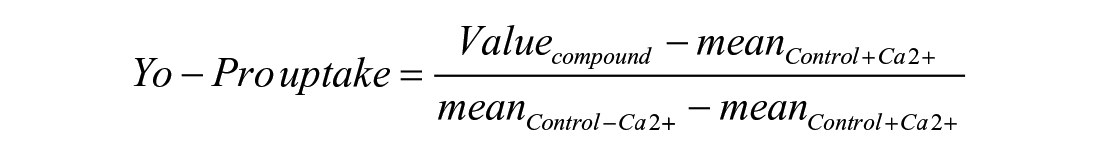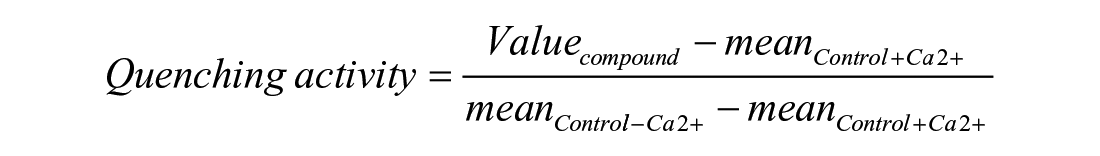Abstract
In vertebrates, intercellular communication is largely mediated by connexins (Cx), a family of structurally related transmembrane proteins that assemble to form hemichannels (HCs) at the plasma membrane. HCs are upregulated in different brain disorders and represent innovative therapeutic targets. Identifying modulators of Cx-based HCs is of great interest to better understand their function and define new treatments. In this study, we developed automated versions of two different cell-based assays to identify new pharmacological modulators of Cx43-HCs. As HCs remain mostly closed under physiological conditions in cell culture, depletion of extracellular Ca2+ was used to increase the probability of opening of HCs. The first assay follows the incorporation of a fluorescent dye, Yo-Pro, by real-time imaging, while the second is based on the quenching of a fluorescent protein, YFPQL, by iodide after iodide uptake. These assays were then used to screen a collection of 2242 approved drugs and compounds under development. This study led to the identification of 11 candidate hits blocking Cx43-HC, active in the two assays, with 5 drugs active on HC but not on gap junction (GJ) activities. To our knowledge, this is the first screening on HC activity and our results suggest the potential of a new use of already approved drugs in central nervous system disorders with HC impairments.
Keywords
Introduction
Cell-to-cell communication is mediated by a family of transmembrane proteins, called connexins (Cx). Cx43 is the most abundant and widespread isoform and can form both gap junctions (GJs) and hemichannels (HCs) on the cell membrane. More specifically, Cx43 HCs allow exchange between the intra- and extracellular environments of small molecules and ions. 1 HC opening or closure probabilities are regulated by inflammation, hypoxia, changes in intra- and extracellular osmolarity concentrations, and cytosolic pH. 2 In physiological situations, HCs are mostly closed. 3 However, in pathological situations, HC pores are opened more frequently than usual, leading to so-called “leaky hemichannels” 4 and resulting in the excessive release of ATP and glutamate, with damage to neighboring cells, 5 or in the unnecessary influx of ions, leading to ionic imbalance. 6 Mutations in Cx-encoding genes have also been identified in disorders of the skin, ear, heart, eye lens, and brain. 4 HCs containing Cx43 are also upregulated in different pain models, for example, neurodegenerative disorders such as Alzheimer’s disease, cerebral ischemia, amyotrophic lateral sclerosis, and cancer. 7
Modulation of Cx43 HCs has been successfully achieved using pharmacological approaches8,9 or mimetic peptide application. 10 Meanwhile, experimental approaches on animal models using those peptides or RNA interference using silencing or short-hairpin RNA oligonucleotides,11,12 and ongoing clinical trials, 13 have confirmed that HCs represent innovative—yet largely unexplored—therapeutic targets, notably in central nervous system (CNS) disorders. 7
Identifying new modulators of Cx-based HCs is therefore a necessity in this therapeutic perspective. Classical methods to assess HC function include the quantification of ATP release 14 and uptake or leakage of fluorescent dyes.14,15 Additional techniques have been further developed in bacteria deficient in K+ uptake and genetically modified to express functional human Cx, 16 or using passive uptake of iodide and the quenching of a iodide-sensor protein (iodide-sensitive yellow fluorescent protein [YFPQL]). 17 Previous high-content (HCS) and high-throughput (HTS) screening methods have been described to monitor the GJ function using microfluidic chip technology 18 or automated fluorescence imaging assay.19–21 Here, we developed automated versions of two different cell-based assays14,15 to specifically identify new modulators of HC function in cell culture. Both assays exploit the sensitivity of HCs to calcium to control channel opening. While high external calcium maintains the HCs in a closed conformation, depletion of extracellular Ca2+ increases the probability of opening of HCs. 22 The first assay is based on the incorporation of Yo-Pro, a fluorescent dye that enters the cells via open HCs. Previous work on HCs using Yo-Pro incorporation correlated the uptake of Yo-Pro and the total amount of Cx43. 23 In the second assay, iodide uptake through open HCs is indirectly followed by measurement of the iodide-dependent quenching of a fluorescent protein, YFPQL. 17 These assays were validated with known pharmacological inhibitors of HCs and used for the screening of a focused collection of 2242 approved drugs or compounds under preclinical and clinical development. As far as we know, this study is the first systematic screening of HC modulators using chemical libraries dedicated to the identification of HC modulators for CNS disorders.
Materials and Methods
Reagents, Cell Culture, and Chemical Compounds
Opti-MEM culture media, 0.05% trypsin-EDTA, penicillin/streptomycin, Hank’s Balanced Salt Solution (HBSS), fetal bovine serum (FBS), Yo-Pro-1, iodide, and other chemical compounds were purchased from Invitrogen (Saint-Aubin, France), Sigma-Aldrich (St. Louis, MO), or GE Healthcare Life Science (Velizy-Villacoublay, France). IncuCyte Cytotox Red Reagent was purchased from Sartorius (Royston Hertfordshire, UK). Human glioblastoma U251 cells were purchased from Sigma-Aldrich and Ln215-YFPQL cells were licensed by Yonsei University, Seoul, Korea. 21
An in-house selection of 2242 Food and Drug Administration (FDA)- or European Medicines Agency (EMA)-approved drugs and compounds under clinical assays was generated from two commercial sources, including 1280 compounds from the Prestwick Chemical Library (Illkirch, France) and 962 additional drugs that were selected in the tebu-bio library (Le Perray-en-Yvelines, France). Compounds were dissolved in 10 mM DMSO.
Cx43 Silencing Procedure
As presented in Picoli et al., 24 silencing of Cx43 in U251 cells was efficiently achieved by stable transfection with shRNA-Cx43 TRCN0000344843 (shCx43-843) (targets 30-UTR region) from the Mission Sigma shRNA Library, according to the manufacturer’s instructions. Cells stably transfected with nonrelevant shRNA (indicated as shNE) were used as controls.
Cytotoxicity Assay
U251 and Ln215-YFPQL cells were seeded at 10,000 and 24,000 cells/well, respectively, in 100 µL of fresh culture medium in 96-well tissue culture microplates. Cellular viability was assessed using the IncuCyte Cytotox Red reagent according to the manufacturer’s protocol, in the presence of Yo-Pro (5 µM) for U251 cells or iodide (140 mM) for Ln215-YFPQL cells, in the presence or absence of Ca2+. Four images per well (covering 27% of the well surface) were automatically acquired using IncuCyte Zoom (Essen BioScience, Ann Arbor, MI) every 5 min for 1 h in phase contrast and in the red fluorescence channel with a 10× objective. A threshold based on the fluorescence intensity of Cytotox Red staining was applied for the selection of the dead cells.
Monitoring of Yo-Pro Incorporation in U251 Cells by Real-Time Imaging
A Yo-Pro incorporation assay has been adapted from Orellana et al. 25 in 96-well plates. Briefly, U251 cells were seeded at 10,000 cells/well in 96-well tissue culture microplates (Greiner, Les Ulis, France, no. 655090) in Opti-MEM medium supplemented with 10% FBS and allowed to grow for 24 h. Cells were then washed with HBSS buffer with or without Ca2+ and treated simultaneously with 5 µM Yo-Pro and chemical compounds. The microplates were transferred into an automated imaging system (IncuCyte Zoom). Four images per well were acquired in phase contrast and in the green fluorescence channel with a 4× objective, 10, 15, and 20 min after compound addition. A threshold based on the fluorescence intensity of Yo-Pro green staining was applied for the selection of the Yo-Pro-positive (Yo-Pro+) cells. Yo-Pro uptake activity was subsequently calculated and expressed as a percentage of maximal uptake obtained in the absence of calcium, according to the following formula:
where
Automated Monitoring of YFPQL Quenching by Iodide Incorporation in Ln215 Cells
The YFPQL quenching assay has been here adapted from Lee et al. to a 96-well plate format. 21 Briefly, YFPQL-expressing Ln215 cells were seeded at 24,000 cells/well in 96-well tissue culture microplates (Greiner, no. 655090) in DMEM supplemented with 10% FBS and allowed to grow for 24 h. Cells were then washed with HBSS buffer with or without Ca2+ and treated with chemical compounds for 10 min at 37 °C. Immediately after the subsequent addition of 140 mM sodium iodide, the YFPQL fluorescent signal was monitored using an Infinite M1000 multimode reader (Tecan, Switzerland) with acquisitions every 80 s for 560 s. Results were expressed in percentage of quenching activity according to the following formula:
Results
Setup of a Real-Time Microscopy-Based Assay to Quantify HC Function by Yo-Pro Uptake
Yo-Pro incorporation by U251 cells was analyzed in a 96-well format from 10 to 20 min after calcium depletion (
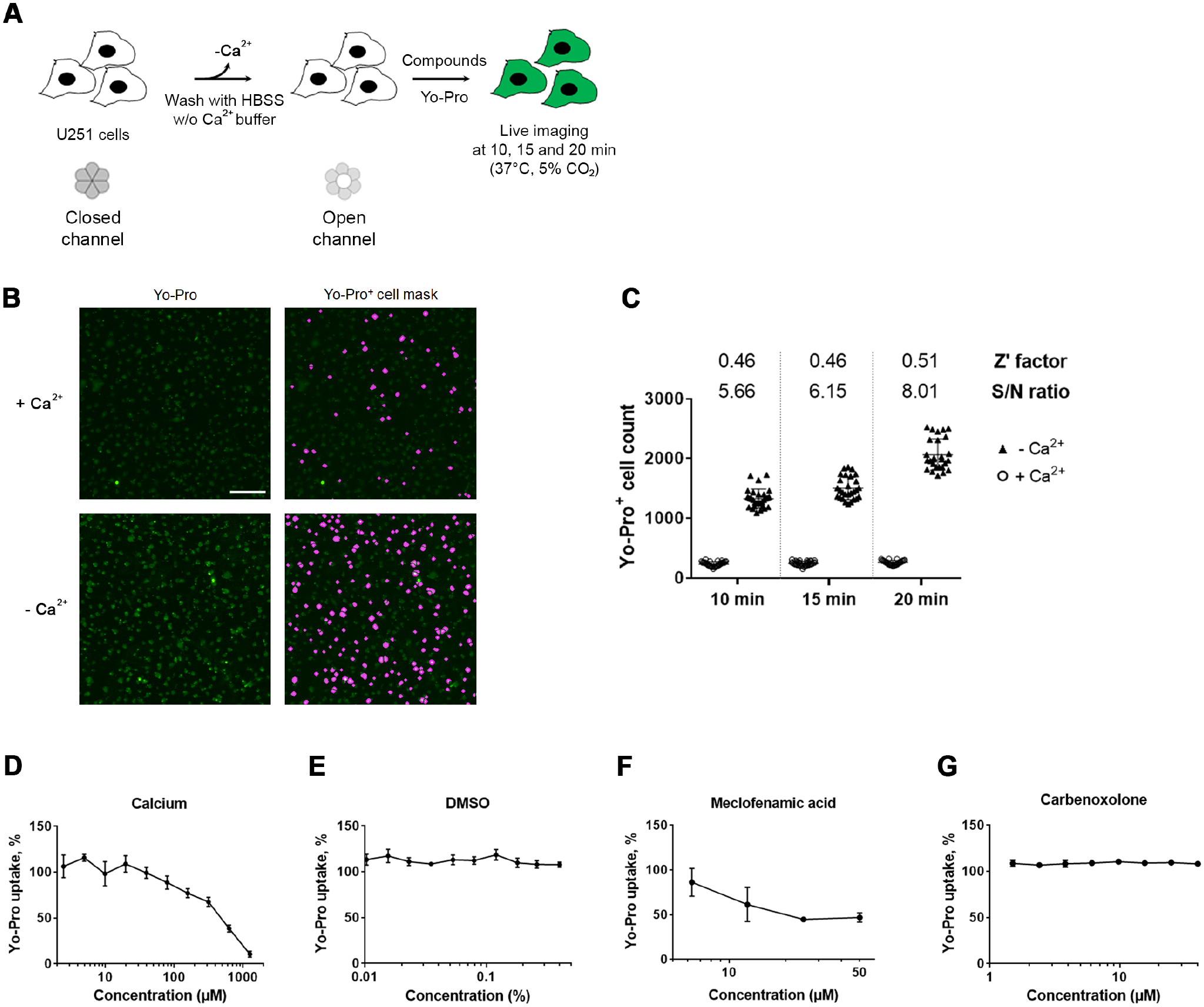
Cell-based assay setup to follow the incorporation of the Yo-Pro fluorescent marker in U251 cells by real-time imaging. (
Setup of an Automated Assay to Quantify HC Function through the Rapid YFPQL Fluorescence Quenching after Iodide Incorporation
As described previously, HC function can be monitored through the HC-dependent passive uptake of iodide revealed by the quenching of the YFPQL, stably expressed in Ln215 cells.
21
We set up the conditions to monitor the quenching of YFPQL fluorescence following Ca2+ depletion in 96-well microplates (
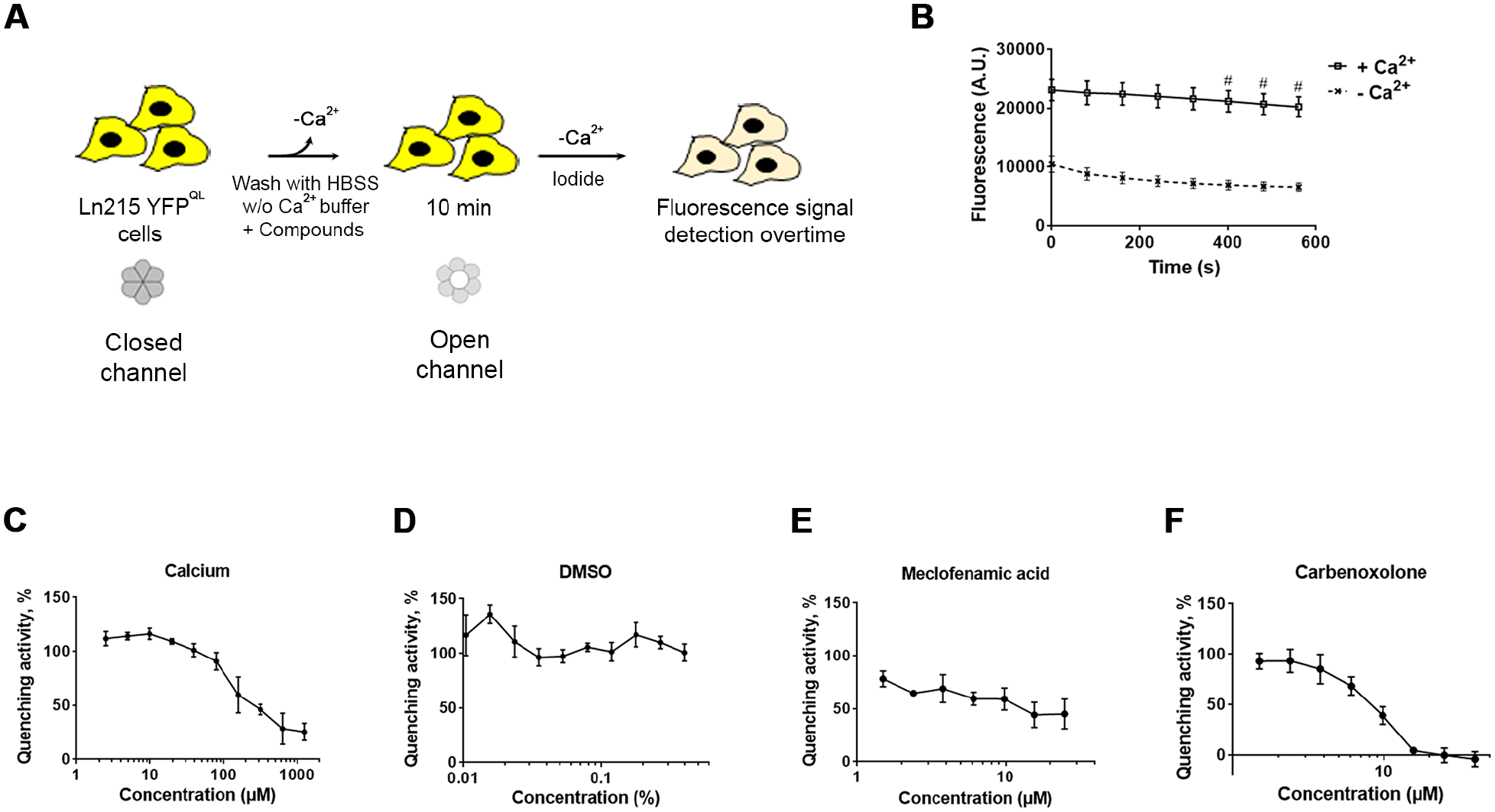
Cell-based assay for automated monitoring of YFPQL quenching by iodide incorporation. (
Screening of an In-House Collection of FDA-Approved Drugs
To identify putative modulators of HCs, an in-house collection of 2242 compounds was screened using the Yo-Pro incorporation cell-based assay by real-time imaging (
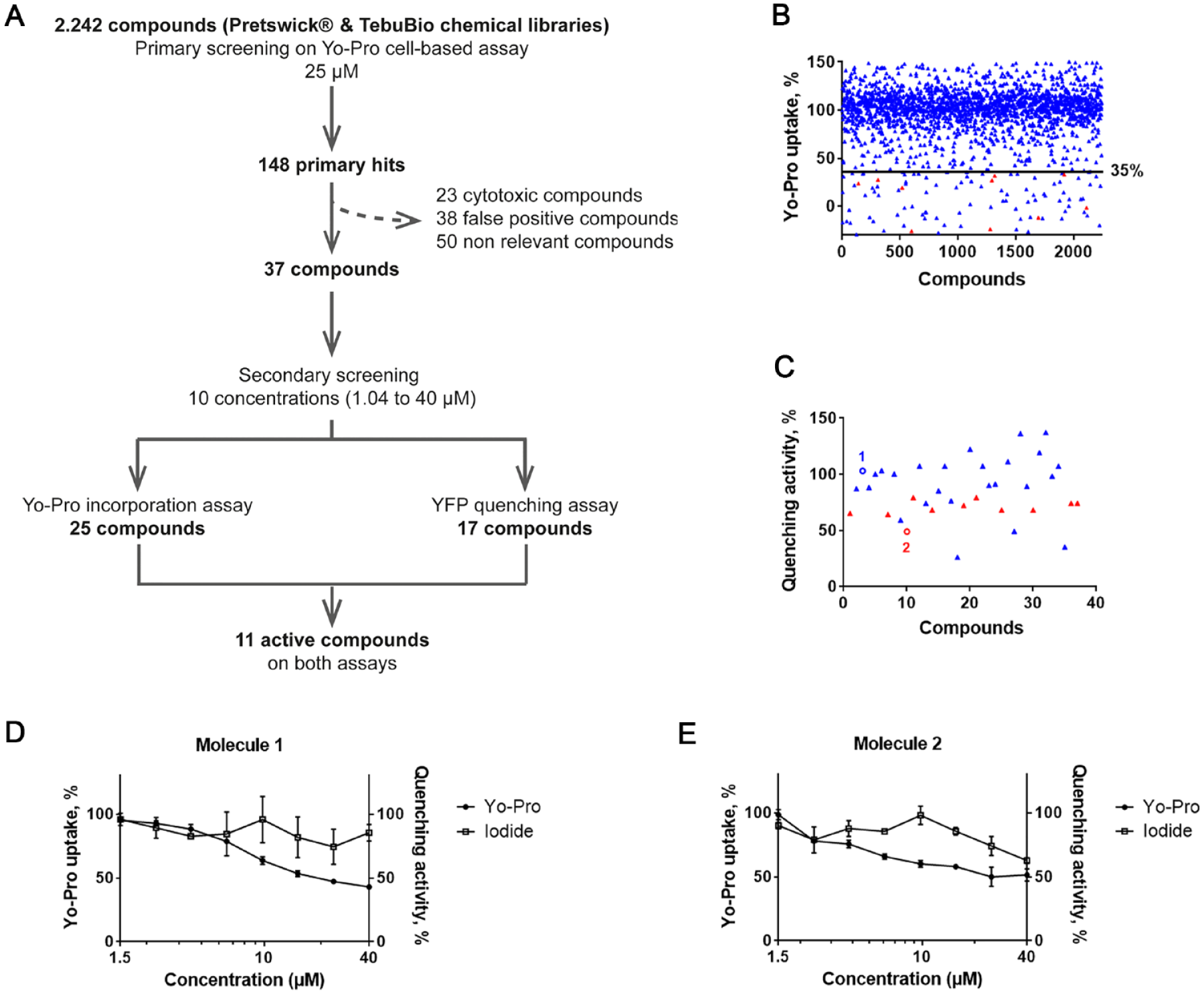
Screening workflow and results. (
Comparison of the Activity of Primary Hits on Yo-Pro and Iodide Incorporation Assays
The selected 37 primary hits were tested at 10 concentrations in simplicate, together with meclofenamic acid and carbenoxolone, used here as controls on both the Yo-Pro incorporation assay and the iodide-induced YFPQL quenching assay. The ability to inhibit Yo-Pro incorporation was confirmed for 25 compounds, with at least one efficient concentration (not shown). In this assay, meclofenamic acid displayed an IC50 above 5 µM (not shown), a value consistent with our previous results obtained during the setup of the assay (see
Discussion
Cx, and notably Cx43, are dysregulated in numerous indications 7 and have been considered as therapeutic targets 27 in sleep disorders,28–30 neuropathic pain, 9 skin and corneal wounds, or cardiac ischemic injury. 31 Marketed drugs routinely used in patients, such as antidepressants, 32 have been revealed to act on Cx43-based HCs. Conclusively, investigating a library of FDA- and EMA-approved drugs using HC-targeting assays may therefore identify compounds with so far unsuspected HC activity-modulating properties and prove particularly fruitful.
Cx43 GJ function has been previously monitored in cell culture through the quantification of cell–cell transfer of fluorescent dyes, 33 calcium, 34 or iodide. 17 Assays have been adapted to HTS, using microfluidic chips 35 and automated fluorescence imaging assays. 36 However, those assays are valid to measure GJ but not HC function, as it is largely admitted that under regular culture conditions, the contribution of those HCs is minor37,38—hence the need to use nonphysiological conditions to specifically monitor their activity. In physiological conditions, HCs are actually mostly in their closed conformation, due to extracellular Ca2+ and Mg2+ in the millimolar range, negative membrane potentials, or posttranslational modifications. 3 Depletion of extracellular Ca2+ is hence classically used in cell culture to assess HC function.14,15,17
We present here the adaptation to automated screening of two previously described cell-based HC assays. 15 The two cell lines have been selected due to endogenous expression of Cx43 in U25117 and the conclusive use of Ln215 in GJ and HC assays.17,21,39 Supporting previous reports, extracellular calcium depletion increased HC-mediated uptake of the probes in a dose-dependent manner, and DMSO, the solvent used to solubilize compounds, was revealed to be inactive. Finally, meclofenamic acid, a broad Cx blocker,30,40 confirmed the sensitivity of the assays. Surprisingly, carbenoxolone displayed inconsistent effects in the two assays, potentially correlating with its previously described relative potency on Cx43.41,42 The two assays have been described as dependent upon Cx43 HC function;17,43 this has been further demonstrated in the present study using RNA silencing in U251, and elsewhere in Ln215 using CRISPR-Cas9 KO. 39 In addition, the Yo-Pro incorporation assay notably depends on the transfer of Yo-Pro from the cytoplasm to the nucleus after uptake, while the YFP quenching assay notably requires an active quenching of YFP by iodide after iodide uptake. We chose to focus on the search for compounds active on both assays to be considered as properly active against HCs.
These two assays were used for the screening of a chemical collection and further hit characterization. An in-house chemical library composed of 2242 approved drugs was screened on the Yo-Pro incorporation assay (Z′ > 0.5). A threshold was fixed at 35% activity relative to the control, as it allowed the clear discrimination between bioactive and bioinactive controls. A total of 148 primary hits were identified with Yo-Pro uptake under this threshold. Nonrelevant (poor ability to cross the blood-brain barrier, low oral bioavailability, adverse effect profile, etc.) and toxic compounds were eliminated from this first set, leading to a subset of 25 confirmed hits. Among them, 11 also inhibited the quenching of YFPQL by iodide incorporation after calcium depletion, representing a discovery rate of 0.49%.
Besides, a subset of 1280 compounds of this collection have already been screened on Cx43 GJ function using an automated assay monitoring dye transfer; 20 3 out of 11 compounds efficient on both HC assays also inhibited GJ function. Interestingly, five compounds were active on HCs but not on GJs, and additional studies should confirm their specificity toward HCs and not GJs and evaluate their effects using a newly developed in vivo imaging technique based on manganese-enhanced magnetic resonance. 44
To our knowledge, this study represents the first large screening on HC activity and suggests a new use for already approved drugs in CNS disorders with HC impairments.
Supplemental Material
Figure_S1_v4 – Supplemental material for Quantitative Automated Assays in Living Cells to Screen for Inhibitors of Hemichannel Function
Supplemental material, Figure_S1_v4 for Quantitative Automated Assays in Living Cells to Screen for Inhibitors of Hemichannel Function by Emmanuelle Soleilhac, Marjorie Comte, Anaelle da Costa, Caroline Barette, Christèle Picoli, Magda Mortier, Laurence Aubry, Franck Mouthon, Marie-Odile Fauvarque and Mathieu Charvériat in SLAS Discovery
Supplemental Material
Figure_S2_v3 – Supplemental material for Quantitative Automated Assays in Living Cells to Screen for Inhibitors of Hemichannel Function
Supplemental material, Figure_S2_v3 for Quantitative Automated Assays in Living Cells to Screen for Inhibitors of Hemichannel Function by Emmanuelle Soleilhac, Marjorie Comte, Anaelle da Costa, Caroline Barette, Christèle Picoli, Magda Mortier, Laurence Aubry, Franck Mouthon, Marie-Odile Fauvarque and Mathieu Charvériat in SLAS Discovery
Supplemental Material
Figure_S3_v3 – Supplemental material for Quantitative Automated Assays in Living Cells to Screen for Inhibitors of Hemichannel Function
Supplemental material, Figure_S3_v3 for Quantitative Automated Assays in Living Cells to Screen for Inhibitors of Hemichannel Function by Emmanuelle Soleilhac, Marjorie Comte, Anaelle da Costa, Caroline Barette, Christèle Picoli, Magda Mortier, Laurence Aubry, Franck Mouthon, Marie-Odile Fauvarque and Mathieu Charvériat in SLAS Discovery
Footnotes
Acknowledgements
This work has notably been performed at the CMBA screening platform (http://irig.cea.fr/drf/irig/english/Pages/Platform/CMBA.aspx) supported by the GRAL LabEX GRAL, ANR-10-LABX-49-01, financed within the University Grenoble Alpes graduate school (Ecoles Universitaires de Recherche), CBH-EUR-GS (ANR-17-EURE-0003). The CMBA is a member of the Chembiofrance research infrastructure (www.chembiofrance.fr) and the GAP2D consortium supported by the GIS-IBiSA (![]() ). Ln215 YFPQL cells were produced by Dr. Jinu Lee and licensed by Yonsei University (Seoul, Korea).
). Ln215 YFPQL cells were produced by Dr. Jinu Lee and licensed by Yonsei University (Seoul, Korea).
Supplemental material is available online with this article.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: A.D., C.P., F.M., and M.C. are full-time employees of Theranexus Company.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the CEA, Theranexus, and a research grant from the Auvergne-Rhone-Alpes district (Pack Ambition 2017, no. 17 011104 01-8748).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.