Abstract
Autosomal dominant leukodystrophy (ADLD) is a fatal, progressive adult-onset disease characterized by widespread central nervous system (CNS) demyelination and significant morbidity. The late age of onset together with the relatively slow disease progression provides a large therapeutic window for the disorder. However, no treatment exists for ADLD, representing an urgent and unmet clinical need. We have previously shown that ADLD is caused by duplications of the lamin B1 gene causing increased expression of the lamin B1 protein, a major constituent of the nuclear lamina, and demonstrated that transgenic mice with oligodendrocyte-specific overexpression of lamin B1 exhibit temporal and histopathological features reminiscent of the human disease. As increased levels of lamin B1 are the causative event triggering ADLD, approaches aimed at reducing lamin B1 levels and associated functional consequences represent a promising strategy for discovery of small-molecule ADLD therapeutics. To this end, we have created an inducible cell culture model of lamin B1 overexpression and developed high-content analysis in connection with multivariate analysis to define, analyze, and quantify lamin B1 expression and its associated abnormal nuclear phenotype in mouse embryonic fibroblasts (MEFs). The assay has been optimized to meet high-throughput screening (HTS) criteria in multiday variability studies. To control for batch-to-batch variation in the primary MEFs, we have implemented a screening strategy that employs sentinel cells to avoid costly losses during HTS. We posit the assay will identify bona fide suppressors of lamin B1 pathophysiology as candidates for development into potential therapies for ADLD.
Keywords
Introduction
Autosomal dominant leukodystrophy (ADLD) is an ultrarare, fatal, progressive adult-onset disease. It occurs in the fourth or fifth decade of life, and the primary pathology is widespread central nervous system (CNS) demyelination. 1 Symptoms usually begin with signs of autonomic dysfunction followed by loss of fine motor control. This is followed by spasticity, ataxia, tremor, and paralysis of upper and lower extremities. ADLD is thus associated with significant morbidity before patients ultimately succumb to the disease, usually within 10–20 years after the onset. 2 Robust genetic testing, the late age at onset, and the relatively slow progression of the disease provide a large window for therapeutic intervention and thus make ADLD an ideal candidate for the identification of potential drugs. However, no cure or treatment exists for the disease, representing an urgent and unmet clinical need.
ADLD is caused by heterozygous duplications of the lamin B1 gene and genomic deletions upstream of lamin B1, both resulting in increased LMNB1 protein expression.3–6 Lamin B1 is an intermediate filament protein that is an integral part of the nuclear lamina, a fibrous meshwork found adjacent to the inner nuclear membrane in all cells. In vertebrates, there are of two main types of lamins: A and B type. The A type lamins, lamins A and C, are alternatively spliced products of the same gene, LMNA. Two separate genes, LMNB1 and LMNB2, code for the major B type lamins.7–10 While originally identified as having a structural role in maintaining nuclear architecture, the nuclear lamina is now thought to play critical roles in multiple cellular processes, including transcription, DNA replication, DNA repair, and epigenetic regulation.7,10,11 Nuclear abnormalities have been reported in ADLD patient fibroblasts, fibroblasts from ataxia telangiectasia patients exhibiting increased lamin B1 levels, and HEK293 cells transfected with lamin B1 constructs.3,12,13 Mouse models overexpressing lamin B1 in oligodendrocytes, the cell types that produce myelin in the CNS, recapitulate the late age at onset and many of the histopathological features of ADLD, further strengthening the link between lamin B1 overexpression and the demyelination phenotype.14,15 As an increased level of lamin B1 is the causative agent triggering ADLD, we reasoned that approaches aimed at reducing lamin B1 levels, rather than downstream targets, whose roles in ADLD are currently poorly understood, would represent a potent and innovative strategy that could be exploited for therapeutic discovery. This study describes the development of a high-content analysis (HCA) platform to identify suppressors of lamin B1 overexpression that may serve as potential therapeutic molecules for the treatment of ADLD.
Materials and Methods
Generation of TRE-LMNB1 Cell Lines
TRE-FLAG-LMNB1 hemizygous mice, described previously, 14 were independently generated at the University of Pittsburgh transgenic core on an FVB/N background and were mated with Rosa26-rtTA homozygous mice (Jackson Laboratories) to produce TRE-FLAG-LMNB1;Rosa-rtTA and wild-type (WT) offspring ( Fig. 1A ). Primary mouse embryonic fibroblasts (MEFs) were isolated from 13.5-day-old embryos as previously described. 16 All animals were housed at University of Pittsburgh animal facilities in accordance with Institutional Animal Care and Use Committee (IACUC) guidelines.
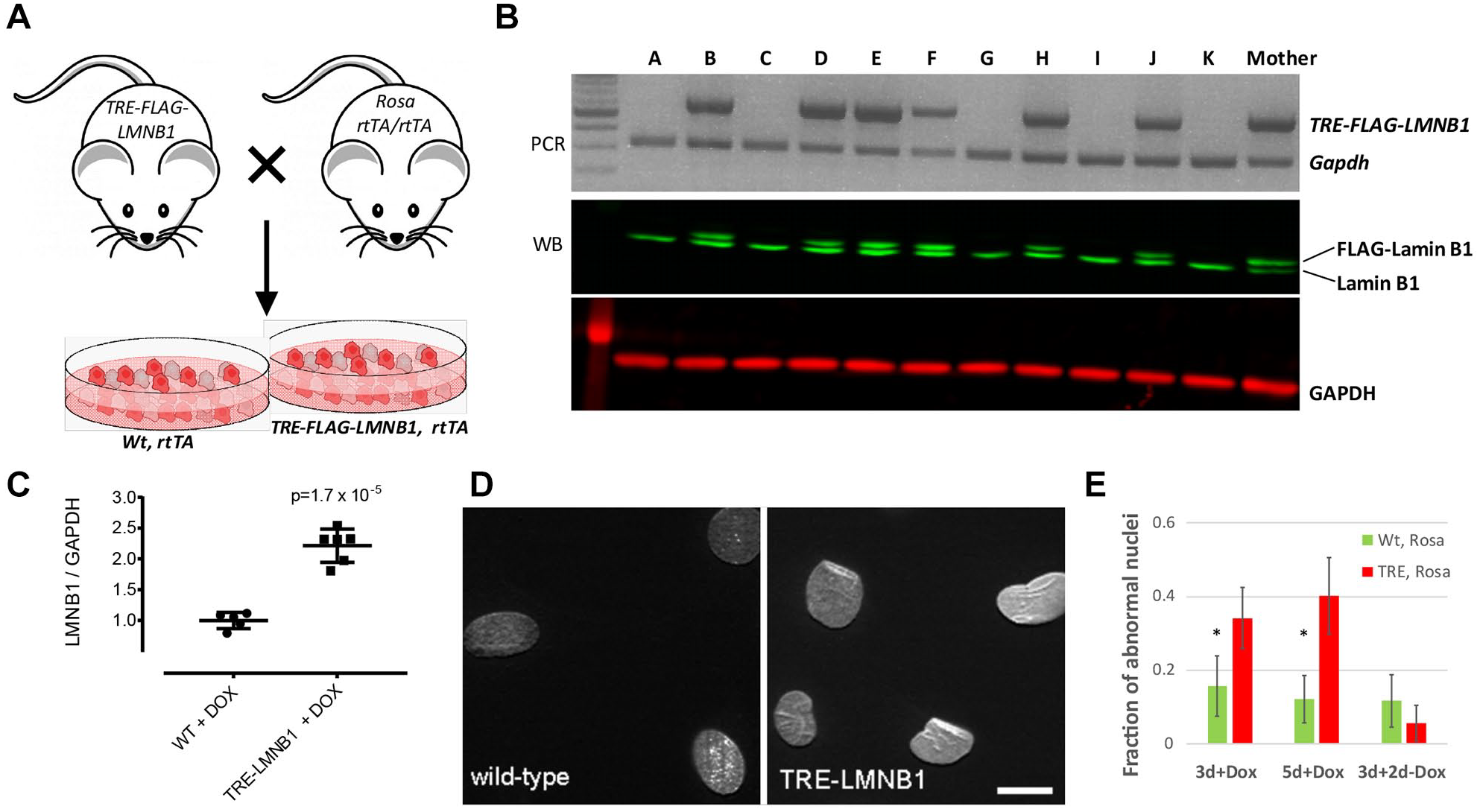
Generation of a mouse model of inducible lamin B1 expression. (
Cell Culture
Primary MEFs from WT and TRE-FLAG-LMNB1;Rosa-rtTA embryos were cultured in complete Dulbecco’s modified Eagle’s medium (high-glucose DMEM; Corning) supplemented with 10% fetal bovine serum (FBS; Fisher), 2 mM L-glutamine (Millipore), and 1% penicillin streptomycin (Hyclone) in a humidified chamber at 37 °C and 5% CO2. When cells reached 90%–100% confluence, they were trypsinized, diluted, and replated using fresh media. Lamin B1 overexpression was induced in TRE-FLAG-LMNB1;Rosa-rtTA transgenic MEFs by adding 2 μg/mL doxycycline (DOX) in growth medium. Primary human skin fibroblasts from an ADLD patient were isolated as previously described 6 and cultured in complete DMEM in a humidified chamber at 37 °C and 5% CO2.
Preliminary Imaging and Quantification
Cells seeded on glass coverslips were washed with Dulbecco’s phosphate-buffered saline (DPBS; Sigma-Aldrich, St. Louis, MO) and then fixed with 4% formaldehyde (Ted Pella) and simultaneously blocked and permeabilized in PBS + 5% normal donkey serum (Jackson ImmunoResearch) + 0.3% Triton X-100 (Sigma-Aldrich) for 1 h at room temperature. Cells were probed overnight at 4 °C with primary antibodies anti-lamin B1 and anti-lamin A/C diluted 1:500 each in PBS + 1% bovine serum albumin (BSA; Sigma-Aldrich) + 0.3% Triton X-100. The next morning, cells were washed in PBS at room temperature, then probed with anti-rabbit and anti-mouse secondary antibodies each diluted 1:350 in PBS + 1% BSA for 1 h at room temperature in the dark, washed again in PBS, and then mounted onto glass microscope slides using Vectashield antifade mounting medium with DAPI (Vector Laboratories). Wide-field microscopy images were acquired of immunofluorescently labeled nuclei on a Leica DM5000B upright microscope with a 40× 0.75 NA objective, a 63× 1.40 NA oil immersion objective, and a Leica DFC310 FX digital camera. Preliminary quantification of abnormal nuclei was performed manually by counting nuclei with excessive surface folding, contortions, convolutions, or invaginations of the nuclear envelope. For each experiment, cells from 9 to 17 imaging fields were counted and assigned as “normal” and “abnormal” by a human observer. Fractions of abnormal nuclei in each field were averaged and analyzed for significance using the modified Wald method 17 in GraphPad Prism.
Reagents
The antibodies used were anti-lamin B1 (Abcam AB16048), anti-lamin A/C (Cell Signaling Technology 4777), donkey-anti-rabbit-Cy3 (711-165-150), donkey-anti-mouse-Cy5 (715-175-150) for MEFs, and donkey-anti-rabbit-FITC (711-095-152, all from Jackson ImmunoResearch) for ADLD patient cells. All other reagents including DOX (D-9891) and BSA (A-2153) were from Sigma-Aldrich.
High-Content Analysis
Tetracycline responsive element (TRE) MEFs were plated in collagen-coated 384-well microplates (Greiner 781956), fixed with formaldehyde (4%), permeabilized with Triton X-100 (0.3%)/BSA (1%) in PBS, and stained with anti-lamin B1 and anti-lamin A/C antibodies overnight at 4 °C, followed by Cy3- and Cy5-conjugated secondary antibodies, respectively. Nuclei were counterstained with Hoechst 33342. Plates were washed with PBS and sealed, and images acquired on a Thermo Fisher Cellomics ArrayScan II high-content reader using a 10× 0.75 NA objective. One thousand cells per well were acquired and analyzed for lamin expression and texture using the Target Activation Bioapplication. Nuclear shape measurements were made on Hoechst labeling. All measurements were made in an area defined by Hoechst staining. The Bioapplication generated 24 parameters ( Suppl. Table S1 ). To quantify lamin B1 intensity distributions, three heterogeneity indices (Kolmogorov–Smirnov [KS_Norm], quadratic entropy [QE], and percent outliers) were calculated as described 18 based on lamin B1 staining.
Higher-resolution images were acquired on a Molecular Devices ImageXpress Ultra confocal high-content screening (HCS) reader with a 40× objective (to illustrate cell population heterogeneity) or as three-dimensional z stacks (20 planes, 0.3 µm) using a 60× objective followed by 3D reconstruction in MetaXpress (to document the nuclear abnormalities’ phenotype).
Sentinel Cell Analysis
From a single vial of MEFs, two 10% aliquots are set aside and treated with DOX or vehicle (0.001% ethanol) for 3 days. The remaining 80% of cells are plated into a single T-150 flask and expanded for large-scale high-throughput screening (HTS). The small aliquots are trypsinized on day 3, plated into medium with or without DOX, and immunostained with anti-lamin B1 and anti-lamin A/C antibodies. HCA is performed to determine DOX responsiveness of the particular vial. Batches that do not meet HTS criteria in the sentinel cells are discarded; otherwise, cells are used for experiments.
Multivariate Analysis
To enable measurements of nuclear abnormalities with sufficient statistical rigor for HTS, we employed linear discriminant analysis (LDA) to integrate multiple readouts into the Z′ factor as previously described by us19,20 and others.21,22 The details of the method have been previously documented 20 and are only briefly reiterated here. From the multidimensional high-content data set, 27 parameters and the three heterogeneity indices described above were reduced to a single statistical parameter (the LDA value) using the LDA function included in the MASS package of the R statistical software program (https://cran.r-project.org). Z′ factors were then calculated as described 23 using LDA values from WT and TRE-LMNB1 MEFs treated with DOX (2 µg/mL) as minimum (MIN) and maximum (MAX) controls, respectively.
Results
Generation and Characterization of an Inducible Cellular Primary Mouse Fibroblast Model of Lamin B1 Overexpression
Fibroblasts from ADLD patient cells express higher levels of lamin B1 (Giorgio et al. 6 and Suppl. Fig. S1 ), but ADLD patient cells cannot be generated in numbers large enough for HTS, and donor-to-donor variability constitutes a problem. We therefore established a tractable cell culture model for the study of nuclear alterations that would also be suited for HTS and generated a MEF system overexpressing lamin B1.14,24 We have generated inducible transgenic mice where the overexpression of a FLAG-tagged human–lamin B1 gene is under the control of a TRE. These mice are then mated to a second mouse strain (Rosa-rtTA), which contains the reverse tetracycline transactivator (rtTA).25,26 MEFs derived from bi-transgenic embryos (i.e., Rosa-rtTA:TRE-LMNB1, henceforth referred to as TRE-LMNB1) ( Fig. 1A ) overexpress FLAG-tagged lamin B1 upon addition of DOX, a more stable derivative of tetracycline ( Fig. 1B ). The levels of lamin B1 overexpression are ~2-fold at the protein level ( Fig. 1C ), comparable to what has been observed in ADLD patient fibroblasts.6,12 The lamin B1 overexpressing MEFs also exhibit structural abnormalities similar to those that we and others have observed in ADLD patient fibroblasts,12,13 and show significant increases in misshapen nuclei ( Fig. 1D ). These alterations were reversible, as subsequent to the removal of DOX and the cessation of lamin B1 overexpression, the nuclear structure returned to normal ( Fig. 1E ). Our results are consistent with a previous report demonstrating that RNAi mediated knockdown of lamin B1 levels in ADLD patient fibroblasts and cells derived from ADLD patient induced pluripotent stem cells (iPSCs) restore nuclear rigidity and morphology to those indistinguishable from control cells. 12 Taken together, these results strongly suggest that normalizing lamin B1 levels can functionally rescue the lamin B1 overexpression phenotype. As the transgene in the MEFs is stably integrated into the genome of these cells, they have a uniform and persistent lamin B1 overexpression in the presence of DOX. Furthermore, we can generate MEFs in the large numbers that are required for an HTS assay, an approach that would not be possible using primary ADLD patient fibroblasts.
Quantifying Lamin B1 Expression and Associated Nuclear Abnormalities by HCA
The human eye can detect lamin B1 overexpression-mediated nuclear abnormalities, but the phenotype is subtle, heterogeneous, and not observed in all cells. We therefore developed single-cell, multiplexed HCA to define and quantify lamin B1 overexpression and associated nuclear morphology changes.TRE-LMNB1 MEFs were treated with DOX for 5 days and stained with anti-lamin B1-FITC and anti-lamin A/C-Cy3 primary/secondary antibody pairs. Negative controls included DOX-treated WT and noninduced TRE-LMNB1 cells. Nuclei were counterstained with Hoechst 33342. We decided to use an anti-LMNB1 antibody and not an anti-FLAG antibody because it detects both endogenously and exogenously expressed lamin B1, and reducing either one could lead to a rescue of the nuclear abnormalities. In contrast, a FLAG antibody would only be identifying compounds that reduce the artificially overexpressed FLAG-tagged human protein. These might not work on LMNB1 expressed from an endogenous promoter, which is the ultimate objective in patient cells.
Cells were imaged on an Array Scan II high-content reader and analyzed using the Target Activation Bioapplication for lamin B1 and lamin A/C expression, as well as changes in nuclear morphology and texture. Figure 2 shows that differences in WT and TRE-LMNB1 MEFs were quantifiable by HCA. Lamin B1 overexpression was two- to threefold, mirroring that seen in ADLD patients. 6 DOX response was specific to lamin B1 because lamin A/C levels and DNA content as measured by Hoechst staining remained unchanged. The assay also quantified lamin B1 nuclear abnormalities as changes in nucleus size, shape, and lamin B1 texture ( Fig. 2 ; see Suppl. Table S1 for a detailed description of measured parameters). The negative controls (WT + DOX and unstimulated TRE-LMNB1) showed no significant differences in lamin B1 or nuclear morphology, although DOX in WT cells appeared to cause slight changes in nuclear size ( Fig. 2 ). Thus, HCA quantified both lamin B1 expression and associated nuclear morphology changes. The response seen with some parameters (lamin B1 levels, signal-to-background [S/B] ratio = 2.7, 5.8% coefficient of variation [CV]; lamin B1 texture, S/B ratio = 2.2, 4.4% CV) indicated that individual lamin B1 response parameters were moderately elevated but tightly distributed, suggesting that the assay could conceivably be developed to HTS robustness, but that further improvements were necessary.
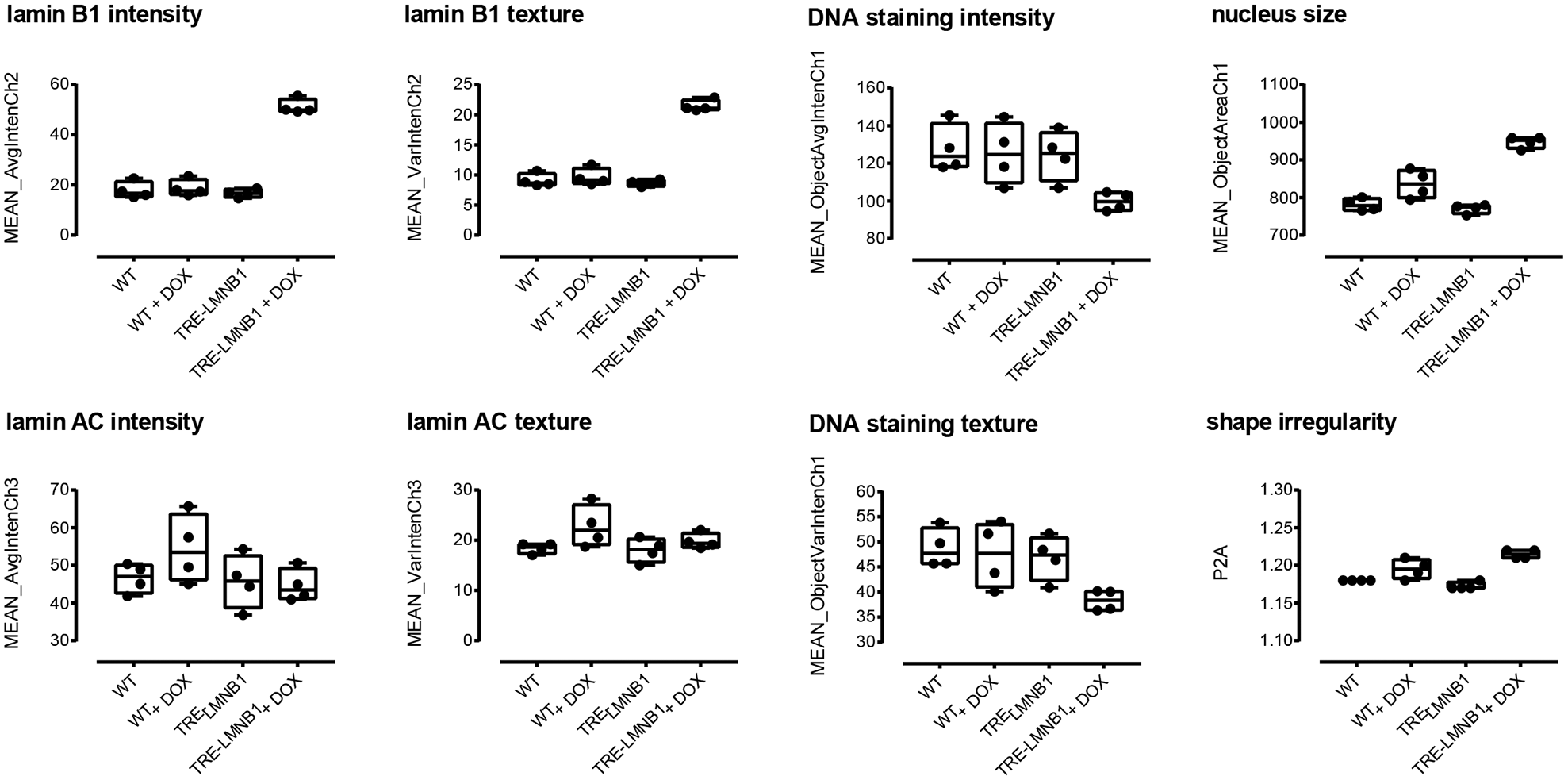
Initial quantification of lamin B1 expression and nuclear morphology by HCA. WT and TRE-LMNB1 MEFs were cultured in 96-well plates for 3 days in the presence or absence of DOX and immunostained with anti-lamin B1 and anti-lamin A/C antibodies. Plates were scanned on an ArrayScan II HCS reader and analyzed for lamin expression and nuclear texture by the Target Activation Bioapplication as described in Materials and Methods. Each box plot shows the mean, first and third quartiles, and range of four wells from a single experiment that has been repeated at least three times.
Cell Density Dependence
Because cell morphology is dependent on environmental conditions and context, we first performed a cell density dependence study. WT and TRE-LMNB1 MEFs were plated at different seeding densities, stimulated for 3 days with DOX, and stained with anti-lamin B1-Cy3 and anti-lamin A/C-Cy5 primary/secondary antibody pairs. Figure 3 shows that lamin B1 expression and associated nuclear abnormalities were highly dependent on cell density, although the separation of stimulated and unstimulated samples was qualitatively maintained across densities. In particular, cell size and length-to-width ratio (LWR) decreased substantially as cells reached confluency, most likely through crowding effects. Because S/B ratios of the most relevant parameters (lamin B1 intensity and texture, nuclear shape) between WT and TRE-LMNB1 MEFs were small by HTS standards and further decreased with increasing cell density, maintaining a good separation between positive and negative controls requires a substantial tightening of the data populations at the higher densities. This could be a problem for HTS given the overall heterogeneous phenotype caused by lamin B1 overexpression and the fact the assay is based on primary cells, which could vary substantially from batch to batch. Because the influence of cell density was of similar magnitude to or even exceeded that of the primary parameters measured, these data indicated that cell seeding density would have to be tightly controlled.
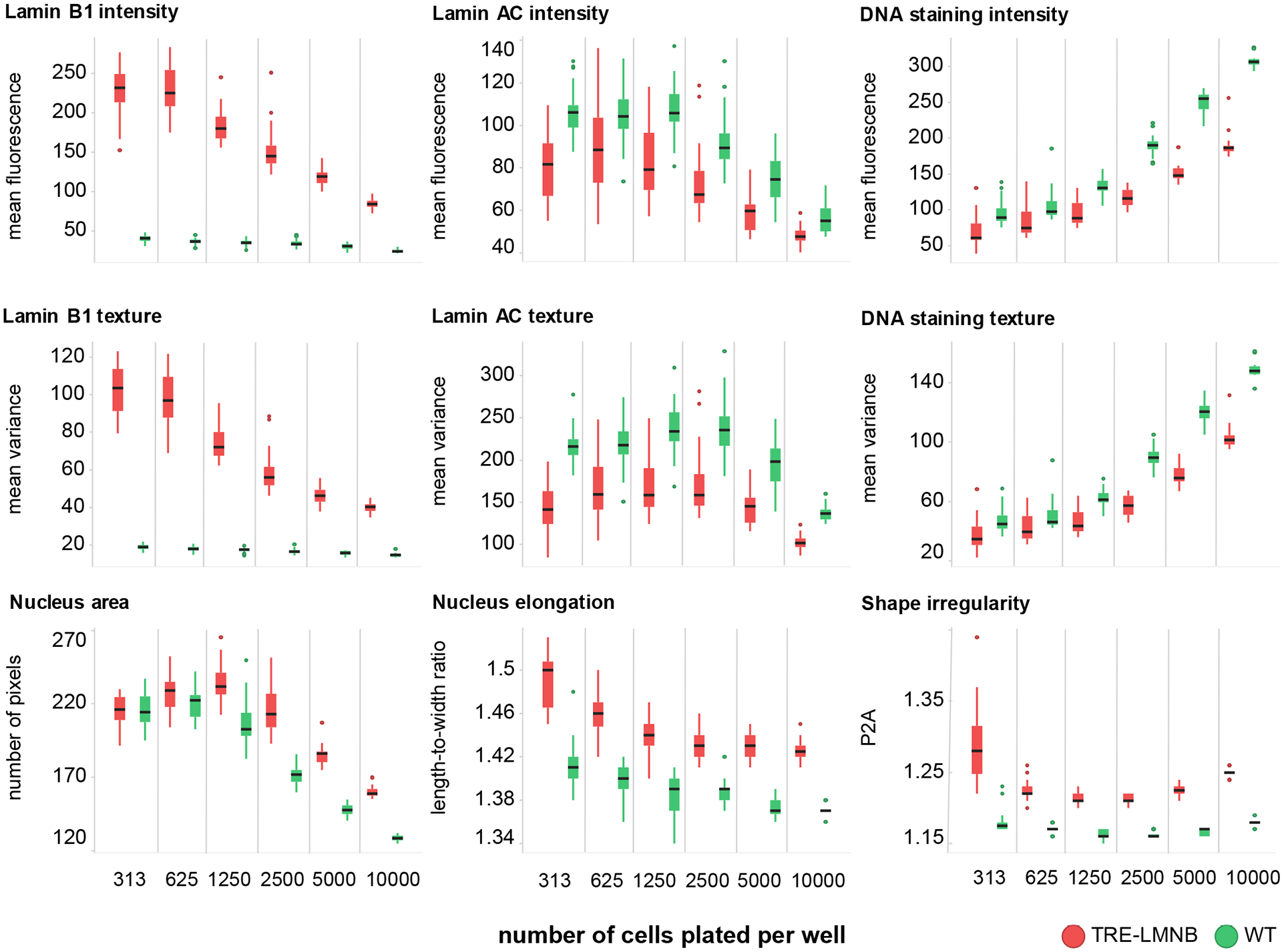
Effects of lamin B1 overexpresson are dependent on cell density. WT or TRE-LMNB1 MEFs were seeded at different densities and analyzed by HCA 3 days after DOX stimulation. Most parameters were strongly affected by cell density; however, for some differences between WT and lamin B1 overexpressing MEFs were retained. Each box plot shows the mean, first and third quartiles, and range of 7–14 technical replicates from a single experiment that has been repeated twice. Green, WT; red, TRE-LMNB1 MEFs.
Assay Optimization
We next performed a formal HTS assay implementation and validation according to universally accepted guidelines. 27 We first investigated the minimum amount of time needed for lamin B1 levels to return to background after DOX withdrawal in order to recapitulate conditions to be encountered during compound screening. Because there are currently no small molecules that selectively reduce lamin B1, we established reversal of lamin B1 overexpression after DOX withdrawal as a positive control. TRE-LMNB1 MEFs at passage 1 were stimulated for 3 days with DOX (2 µg/mL) and plated in 384-well plates in the presence or absence of DOX. Cells were fixed and stained every day thereafter with anti-lamin B1 and anti-lamin A/C antibodies and analyzed for lamin B1/AC expression by HCA. Supplemental Figure S2 shows that in the presence of DOX, lamin B1 overexpression was maintained for at least 4 days (left panel, red symbols). After DOX withdrawal, lamin B1 expression returned to near basal levels after 2 days (left panel, green symbols), which indicates that for screening a 2-day exposure to compounds would be necessary and sufficient to measure a significant reduction in lamin B1. In contrast, DOX withdrawal did not affect lamin A/C or DNA staining intensity ( Suppl. Fig. S2 , middle and right panels).
Improving Assay Robustness and Performance through Multivariate Analysis
Nuclear abnormalities caused by lamin B1 expression present as a highly heterogeneous phenotype, with varying expression levels and shape changes across cell populations. The phenotype of lamin B1-induced nuclear abnormalities is a composite of many different features including lamin B1 expression, but also the appearance of indentations, invaginations, or contortions, and excessive surface folding ( Fig. 1D ). A human observer processes these observations based on experience and context, and thus can classify objects as “normal” or “abnormal” ( Fig. 1E ). For screening this is obviously not feasible. On the other hand, when measured quantitatively, the primary parameter of interest, lamin B1 expression, on occasion yielded HTS performance (i.e., Z factors > 0.5), but this was not observed consistently. To more robustly quantify the cellular phenotype of lamin B1 expression and associated nuclear morphologies, we developed a multivariate analysis methodology (LDA, 28 similar to what we previously developed for zebrafish 19 and yeast 20 ). Figure 4 illustrates the extent to which LDA improved the analysis. Lamin B1-mediated nuclear abnormalities can be seen with the naked eye but are present in both positive (TRE-LMNB1) and negative (WT) samples, do not present uniformly across the image, and occur only in a subset of cells. “Histo-box” plots that we developed for heterogeneity analysis of cell populations 18 expose quantitative differences in the distribution of several cell features ( Fig. 4B ), suggesting that the analysis would benefit from heterogeneity indices that we recently developed to capture biologically relevant heterogeneity in cell populations. 18
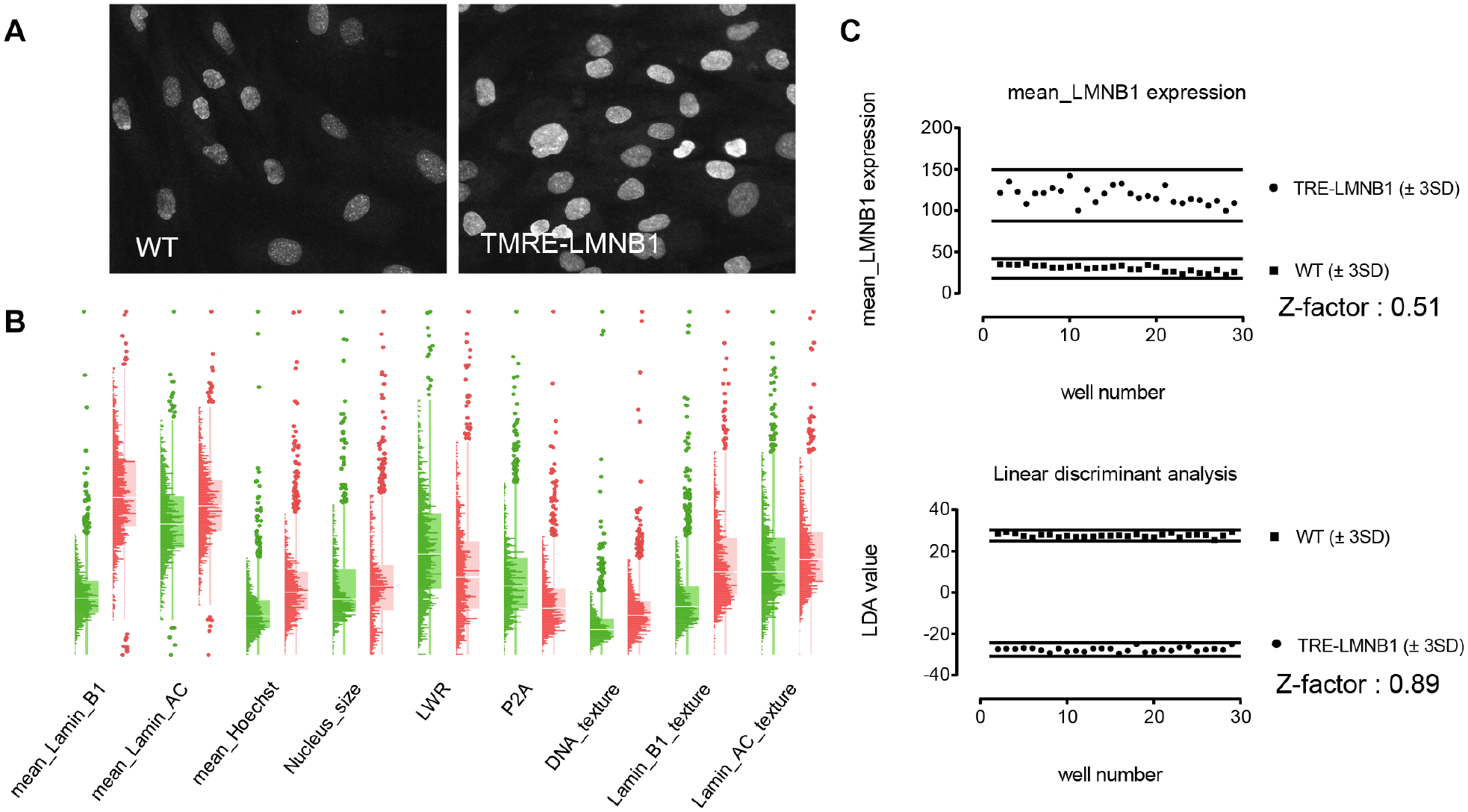
Exploiting multiple features to fully capture complexity and heterogeneity of lamin B1 overexpression and associated nuclear abnormalities. (
The final parameter set encompassed nine intensity and texture measurements (mean, total, and variance of lamin B1, lamin A/C, and Hoechst), three shape measurements (area, circularity, and elongation), 12 cell-to-cell intensity distribution parameters (prefix SD_), and three heterogeneity measurements (KS_norm, denoting deviation from normal distribution; percent outliers; and QE, denoting differences in the shape of data distributions
18
) (
Fig. 4C
Final HTS Implementation
Based on the results from the optimization studies, the assay was subjected to a formal 3-day variability assessment consisting of two full microplates of positive (+ DOX) and negative controls (2 days of DOX withdrawal to mimic compound treatment) on 3 different days. The assay met accepted HTS standards on all 3 days with the same level of statistical significance, documenting screening readiness ( Suppl. Fig. S3 ). We also devised a strategy to deal with batch-to-batch variability, which is based on analysis of “sentinel” cells the week before a large-scale run is performed ( Fig. 5 ). This design ensures that the particular batch of primary cells to be used in HTS responds to DOX with lamin B1 expression in a manner to anticipate screenability. The strategy reduces the risk of losing an entire experimental run due to failure of cells to respond to DOX.
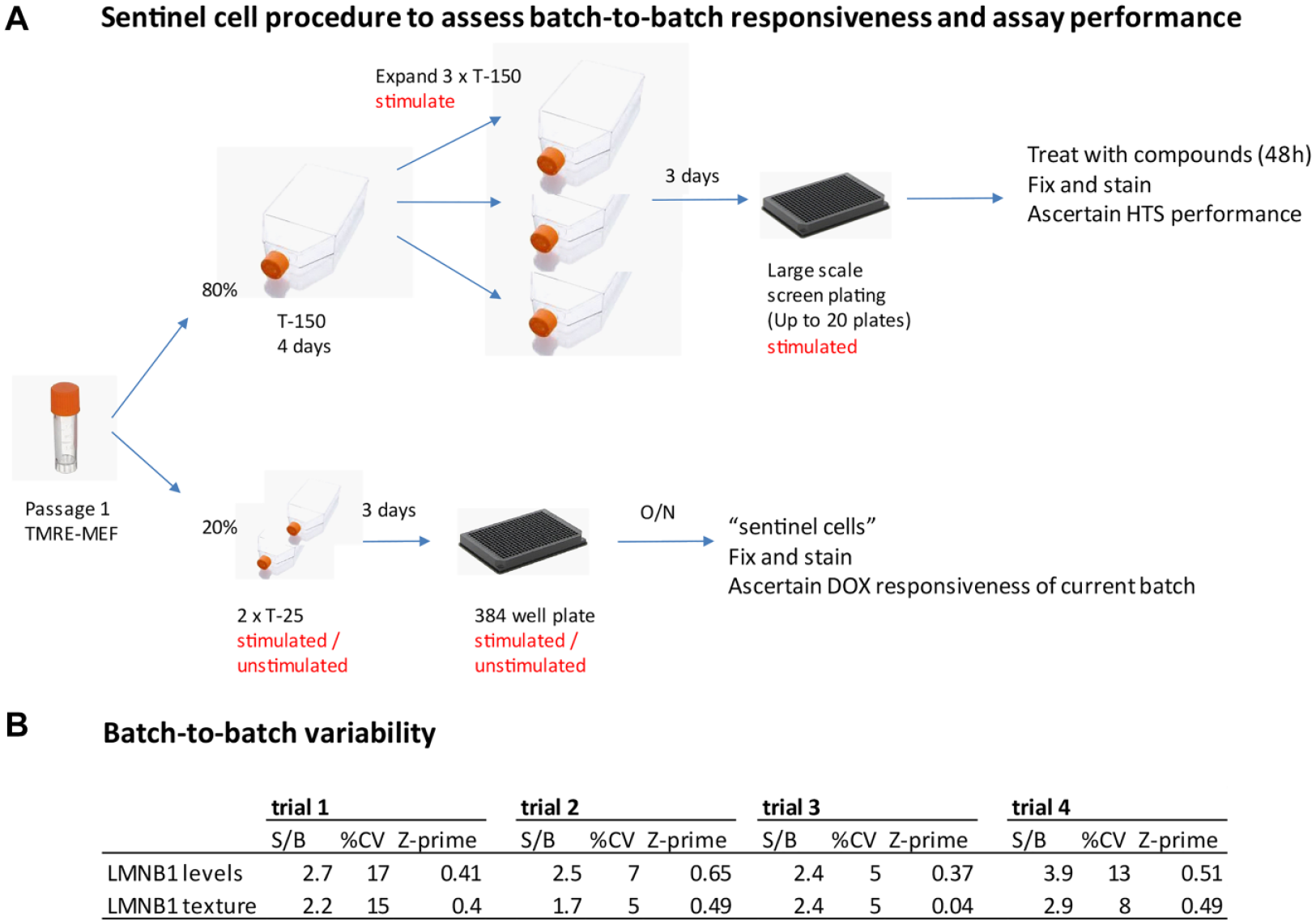
Sentinel cell design to ascertain batch-to-batch responsiveness and assay performance and to eliminate risk of failure during HTS. (
Discussion
We have developed an HTS-ready cellular assay to detect lamin B1 overexpression and associated nuclear abnormalities. Lamin B1 overexpression is the genetic cause for ADLD, a debilitating demyelinating disease for which no treatments exist. While the precise mechanisms linking the genetic defect to demyelination and the biological consequences of nuclear abnormalities are not known, changes in nuclear architecture are a direct result of lamin B1 overexpression because they revert to normal when lamin B1 levels are reduced, either by siRNA or, in our model, by withdrawing the overexpression stimulus. ADLD is a late-onset disease, which presents a large window of opportunity for therapeutic intervention. We posit that small molecules that reduce lamin B1 levels and revert associated nuclear abnormalities could prevent disease onset and/or progression.
There are some obstacles in the development of an assay measuring a reduction in lamin B1 levels. First, the extent of lamin B1 overexpression in ADLD patient fibroblasts is very small when measured on a well average level ( Suppl. Fig. S1 ). Second, the relevant cell type responsible for the demyelinating defect is oligodendrocytes, which are not available for screening. Third, the phenotype of lamin B1-mediated nuclear abnormalities in patient-derived cells is extremely heterogeneous and only seen in a small percentage of cells. We therefore developed a mouse model of DOX-inducible lamin B1 overexpression (TRE-LMNB1), which allows for the generation of an unlimited number of cells for screening. Fibroblasts from TRE-LMNB1 mice overexpress pathophysiologically relevant levels of lamin B1 and show nuclear abnormalities seen in ADLD patients. Importantly, the phenotype reverses after DOX withdrawal; both lamin B1 overexpression and nuclear abnormalities return to levels seen in normal cells. Therefore, the mouse model has good face and construct validity. However, we recognize that an inherent weakness of our cell culture model is that it does not include the endogenous LMNB1 promoter driving the overexpression. Compounds that we identify will be only those that can reduce LMNB1 at the protein level and will not include molecules that reduce LMNB1 expression levels through transcriptional inhibition.
The phenotype of lamin B1-mediated nuclear abnormalities can be visualized by immunocytochemistry and quantified by a human observer. This is an extemely laborious process that limits the utility of the method to small numbers of samples. We used HCS to automatically acquire images and perform preliminary measurements on lamin B1 and nuclear morphology. Initial experiments showed a two- to threefold increase in lamin B1 ( Fig. 2 ), confirming the results of Western blots ( Fig. 1 ). DOX stimulation did not change levels of lamin A/C or DNA content, documenting specificity of the system.
Changes in nuclear morphology were highly dependent on cell density. This was not surprising as the environment in which cells are grown is known to determine cell size and shape. However, we found that not all parameters were equally affected. Regardless of stimulation, cells at higher densities assumed a more rounded shape, became more compact and regularly shaped, and showed decreased levels of lamin B1 and lamin A/C as measured by immunocytochemistry, but increased DNA staining. DOX stimulation caused a substantial difference in lamin B1-associated measurements and nuclear morphology that remained measurable for some parameters (lamin B1 intensity and texture, nucleus elongation, and shape irregularity). Differences in nucleus size became apparent only at high density, whereas differences in lamin A/C levels and texture (if any) remained constant. Importantly, the magnitude of changes due to cell density was comparable to that caused by lamin B1 overexpression, thereby obscuring, counteracting, canceling, or even outweighing changes caused by lamin B1 expression. Furthermore, at a high initial cell seeding density DOX was unable to sustain ectopic lamin B1 expression on days 3 and 4 (data not shown), likely because of cells attaining quiescence due to contact inhibition. This indicated that too high a seeding density might be unsuitable for screening because of a collapse in assay window. On the other hand, very low densities present a challenge for HTS as the time required to collect meaningful numbers of cells could substantially limit throughput. Therefore, cell density had to be carefully optimized and tightly controlled.
In order to overcome heterogeneity, we developed a multiparametric analysis that attempts to fully capture the heterogeneous nature of lamin expression, nuclear morphoplogy, and distribution across cell populations. To quantify the phenotype as a human observer would, we developed a numerical measure to classify nuclei as “normal” and “abnormal.” Our method of choice was LDA, which separates cell populations into two classes through the linear combination of multiple features. Because LDA extracts maximum information from fluorescence images, it has the potential to dramatically improve the separation of classes compared with individual features alone.
We measured 27 individual parameters derived from cell segmentation. No single parameter reliably and repeatedly yielded HTS performance, but some parameters came close in some experiments and described the phenotype better than others. In the sample data set summarized in Supplemental Table S1 , The best differentiating parameter was QE, which measures differences in the shape of data distributions. 18 This parameter therefore quantified the visually apparent increase in cellular heterogeneity upon DOX stimulation. Lamin B1 levels and texture were the parameters that best differentiated cells based on direct lamin B1 measurements. Those parameters also contributed substantially to LDA weighting (note that absolute numbers indicate the degree to which the parameter contributes to the classifier, with larger values denoting larger contributions), indicating that LDA captured the most important biological events. Other parameters showed decent Z factors but lacked informativeness as their S/B ratios were small (perimeter-to-area ratio [P2A] and LWR, 5% increase). Others had large S/B ratios but small Z factors, suggesting high variability (e.g., cell-to-cell distribution of lamin B1 texture). Importantly, LDA weighting and the extent of separation of stimulated and unstimulated cell populations for individual parameters changed from experiment to experiment. LDA consolidated the collective changes into a single “lamin B1-mediated nuclear abnormalites” parameter, much like a human observer would, and rendered the assay reliable and robust enough to satisfy HTS criteria in multiday variability assessments ( Suppl. Fig. S3 ). We should note that the ultimate proof of utility will be how well the method performs under conditions used in screening, and pilot library screening is currently underway. Because the 3-day variability assessments represent an extremely rigorous analysis, we expect the assay will be a suitable plate quality control criterion separating positive and negative controls during screening, when numbers of control wells are much smaller.
ADLD is a devastating and debilitating disease for which no treatments exist. It is a monogenic disease caused by lamin B1 gene duplication and protein overexpression, suggesting that reducing lamin B1 levels could be a viable therapeutic strategy. To discover small molecules that reduce lamin B1 expression, we created a cell culture model that recapitulates the underlying cause of the disease, and developed a robust HCS assay that meets stringent HTS requirements. It is our hope that this assay, when executed in HTS, will identify compounds that reduce lamin B1 levels and associated nuclear abnormalities. We posit that such agents could be valuable starting points for the development of ADLD therapeutics.
Supplemental Material
LMNB1_Assay_Development_paper_REV2_Supplemental – Supplemental material for Development and Optimization of a High-Content Analysis Platform to Identify Suppressors of Lamin B1 Overexpression as a Therapeutic Strategy for Autosomal Dominant Leukodystrophy
Supplemental material, LMNB1_Assay_Development_paper_REV2_Supplemental for Development and Optimization of a High-Content Analysis Platform to Identify Suppressors of Lamin B1 Overexpression as a Therapeutic Strategy for Autosomal Dominant Leukodystrophy by Bruce Nmezi, Laura L. Vollmer, Tong Ying Shun, Albert Gough, Harshvardhan Rolyan, Fang Liu, Yumeng Jia, Quasar S. Padiath and Andreas Vogt in SLAS Discovery
Footnotes
Acknowledgements
We thank Harold Takyi for technical assistance.
Supplemental material is available online with this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by NIH grants R33NS106087 to A.V. and Q.S.P. and R01NS095884 and R21NS104384 to Q.S.P.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
