Abstract
OCTN is a small subfamily of membrane transport proteins that belongs to the larger SLC22 family. Two of the three members of the subfamily, namely, OCTN2 and OCTN1, are present in humans. OCTN2 plays a crucial role in the absorption of carnitine from diet and in its distribution to tissues, as demonstrated by the occurrence of severe pathologies caused by malfunctioning or altered expression of this transporter. These findings suggest avoiding a strict vegetarian diet during pregnancy and in childhood. Other roles of OCTN2 are related to the traffic of carnitine derivatives in many tissues. The role of OCTN1 is still unclear, despite the identification of some substrates such as ergothioneine, acetylcholine, and choline. Plausibly, the transporter acts on the control of inflammation and oxidative stress, even though knockout mice do not display phenotypes. A clear role of both transporters has been revealed in drug interaction and delivery. The polyspecificity of the OCTNs is at the base of the interactions with drugs. Interestingly, OCTN2 has been recently exploited in the prodrug approach and in diagnostics. A promising application derives from the localization of OCTN2 in exosomes that represent a noninvasive diagnostic tool.
Introduction
Organic Cation Transporters Novel (OCTN) is a small subfamily of membrane transporters, which includes three members: OCTN1, 2, and 3. This subfamily, together with other five subfamilies, fall in the larger SLC22 family counting many proteins, mostly catalyzing organic cation and anion transport. The proteins of the SLC22 family are characterized by 12 hydrophobic membrane-spanning segments, with an α-helical secondary structure, connected by hydrophilic amino acid loops. Two of the hydrophilic loops are much larger than the others; one of them protrudes toward the extracellular environment, and the other protrudes toward the intracellular space. Predicted glycosylation sites are present in the extracellular loop, whereas the intracellular loop may have a role in regulation. 1 As for most of the transport proteins of higher eukaryotes, the three-dimensional (3D) structures of the SLC22 members, including the three OCTN transporters, are still not available. From the functional point of view, SLC22 transporters regulate the level of key metabolites and signaling molecules. Most members of the SLC22 family are considered polyspecific because they also recognize many organic compounds with different structures, including drugs.1–5 For this reason, several transporters of the SLC22 family and in particular the organic cation transporters (OCTs) and organic anion transporters (OATs), as well as OCTN1 and OCTN2, have been included in the catalog of transporters responsible for interaction with drugs by the International Transporter Consortium (ITC). This is a group of industrial and academic scientists with expertise in drug metabolism, transport, and pharmacokinetics. In this context, ITC listed the transporters that are known to influence the pharmacokinetics and drug-drug interactions. 6 Therefore, the possible interactions with the listed transporters have to be taken into account for designing new drugs. 1 OCTNs represent special members because, on one hand, OCTNs are described as very specific for one or few substrates, and on the other hand, they are emerging as polyspecific transporters, exhibiting affinity for many compounds and drugs as well. Therefore, these members are extremely interesting for pathophysiology. The OCTN transporters are described in vertebrates. Thus, these transporters seem to arise later in evolution. 7 The relationships of these members with OCTs of lower organisms are, however, unclear. Some traces of OCTN function can be also found in lower organisms. Indeed, Caenorhabditis elegans and Drosophila melanogaster have SLC22 homologs that are similar to OCTNs.8–10 An intriguing member of this family is the third one, namely, OCTN3. It has been identified as SLC22A21 in some organisms, such as mouse and zebrafish,11,12 but it is not annotated in the human genome, where it seems to be lost.12–14 Therefore, in humans, the OCTN subfamily is limited to OCTN1 (SLC22A4) and OCTN2 (SLC22A5), which share a sequence identity of 76%. 15 The role of the OCTN transporters in the animal organisms, including humans, has been studied since their first identification, 16 and many efforts have been made to understand the physiological function of the two transporters.12,17–19 The involvement of OCTN2 in carnitine transport in several tissues is clearly assessed and unequivocally demonstrated by the association of OCTN2 polymorphisms with a human pathology called primary carnitine deficiency (PCD; OMIM 212140). 20 Very recently, the presence of this transporter in exosomes has been reported, thus suggesting this protein as a novel biomarker in human diagnostics. 21 On the contrary, the physiological role of OCTN1 is not unequivocally defined. After the identification of tetraethylammonium (TEA) as the prototype substrate, which gave the name to OCTN1, another substrate was identified: ergothioneine, a mushroom metabolite 22 whose role in humans is not clear. Subsequently, some human endogenous organic cations, such as acetylcholine and choline, were described as OCTN1 substrates.16,23–25 Despite the uncertainties, correlations between OCTN1 expression and neural tissue development have been recently reported.26–29 Thus, the status artis on the role of the two transporters in cell metabolism is quite different. A clear and common aspect of the two transporters is the involvement in drug delivery and interaction.5,17–19,30 Therefore, we expect that unveiling the missing information on the two transporters will give a further drive to human drug discovery. A major bottleneck is the still-lacking 3D structure of the transporters for performing suitable structure/function analyses for investigating the molecular basis of transport and the defects underlying pathologies. The solution of these issues will allow for the design of novel therapies as well as novel diagnostics. In this review, we dedicate our effort in assembling the puzzle on OCTNs in an attempt to report a hopefully readable picture of these intriguing transporters.
Genes
The SLC22A4 gene, located at 5q31.1, counts 49,823 nucleotides with 11 exons (https://www.ncbi.nlm.nih.gov/gene/6583). Orthologs of this gene have been found in 107 different vertebrates. Three transcripts are reported in the Ensembl database for the SLC22A4 human gene, one of which is coding for a protein. The gene encodes a 551 amino acid protein, with a molecular mass of 62,155 Da, known as OCTN1. The SLC22A5 gene has the same chromosomal localization of SLC22A4 and counts 25,911 nucleotides with 11 exons (https://www.ncbi.nlm.nih.gov/gene/6584). Orthologs of this gene have been found in 111 different vertebrates. Two verified transcripts, originating from this gene, have been reported either in the Ensembl or in the NCBI database. The longest transcript isoform (isoform a, NM_001308122.1) is characterized by 11 exons and codes for a 587 amino acid protein, known as OCTN2vt. No information is available on the existence or function of this hypothetical protein. The other isoform (b), NM_003060.3, arises from 10 exons and codes for a 557 amino acid protein known as OCTN2.
Function and Metabolic Roles
The knowledge of the function and the cellular roles of the transporters is the prerequisite for assessing their involvement in drug delivery as well as their employment as a target of inhibitors for silencing transporter-linked metabolic pathways. The functional characterization of OCTN2 is at a more advanced stage with respect to OCTN1, whereas OCTN3 has been studied for some time. The interest in this protein has decreased because of its absence in the human genome.
OCTN2 (SLC22A5) Functional Properties
OCTN2 is a carnitine transporter. It has been well characterized using different experimental models, that is, intact cells, brush border vesicles, and proteoliposomes. 17 It catalyzes an electrogenic cotransport of Na+ with carnitine or other carnitine derivatives.17,31,32 The crucial functional groups of carnitine are the ammonium and the carboxylate.17,33 Among the substrates that satisfy these requirements, there is γ-butyrobetaine, a precursor of carnitine biosynthesis, and other nonphysiological molecules that are used or have been used in human therapy. Esterification of the carnitine hydroxyl group by an acyl derivative does not impair recognition by the transporter. Thus, endogenous as well as exogenous carnitine derivatives are accepted as substrates. Among the physiological derivatives, there are acetyl-carnitine and acyl-carnitines with various acyl chain lengths.33–37 This feature is quite similar to that of the mitochondrial carnitine acyl-carnitine transporter (CAC), whose substrate binding site has been mapped by bioinformatics, site-directed mutagenesis, and chemical targeting. The binding site of CAC involves a large moiety of the protein to allocate all the different substrates. 38 A similar large substrate binding site of OCTN2 may correlate with its capacity to transport and/or interact with a large number of drugs,17,8 in line with the properties of the transporters belonging to the SLC22 family. 5
The orthologs from rat and mouse also show an antiport mode of transport (i.e., exchange of a countersubstrate, which is transported in the opposite direction with respect to Na+ and carnitine).33,35 This may be related to a specific role in simultaneously excreting carnitine derivatives and reabsorbing carnitine in kidney tubules (
Fig. 1
). This different transport mode should be linked to a different functional form of the protein. It cannot be excluded that in humans too, such a functional variant might be active, deriving from different transcription or posttranslational modifications modulating the transport function. The electrogenic nature of the carnitine-sodium cotransport39–43 allows OCTN2 to maintain a carnitine gradient of about two orders of magnitude between plasma and the intracellular compartment40,44–47 (
Fig. 1
). On the basis of the available information, it can be inferred that the transporter also mediates the efflux of carnitine and carnitine derivatives that may be driven by a favorable gradient of these molecules.33,48 Thus, Na+ may not be involved in the energy coupling of the efflux process (
Figs. 1
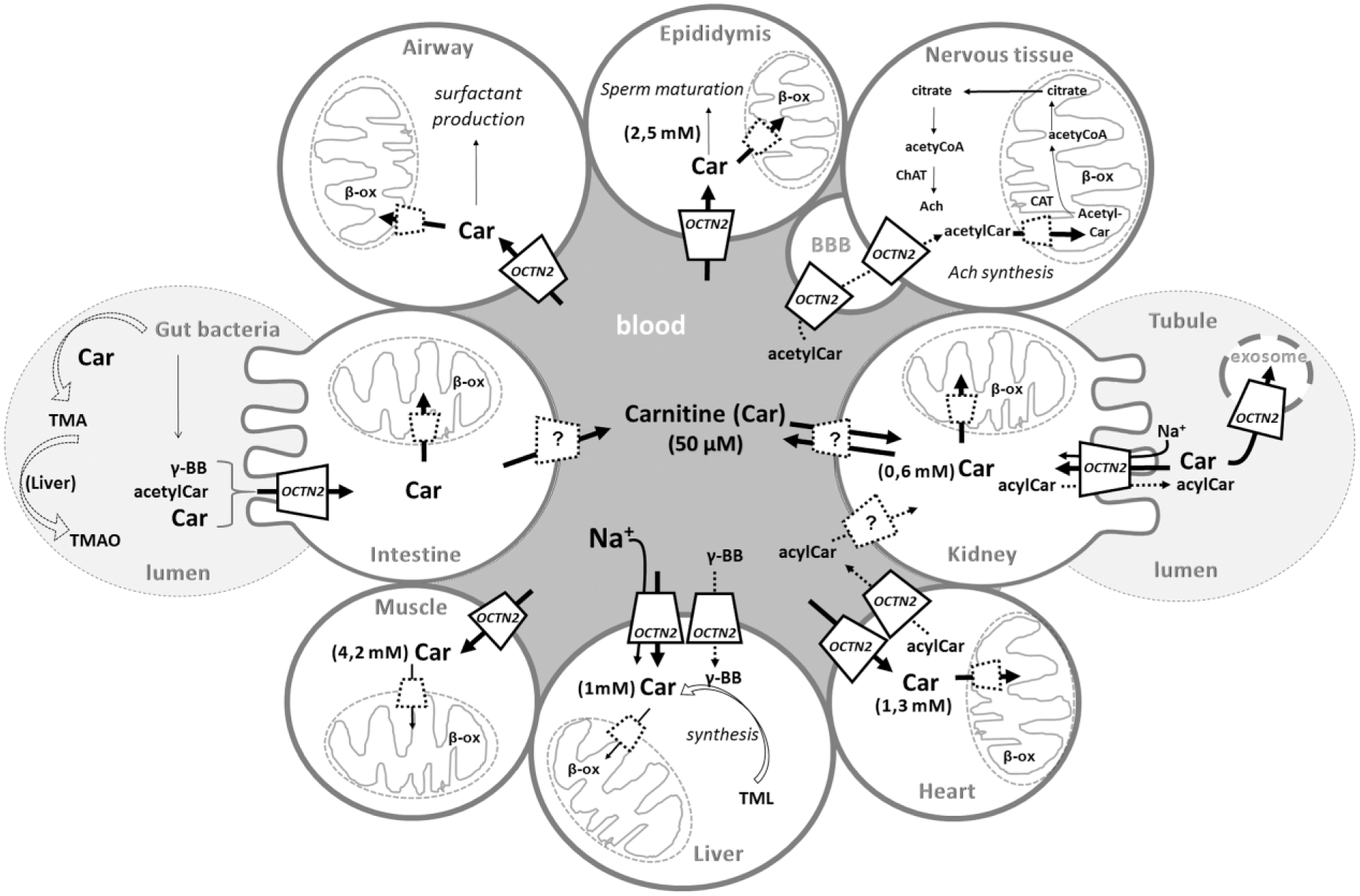
Involvement of OCTN2 in carnitine homeostasis and description of its different tissue-specific roles. Epithelial polarized and other tissues are depicted as “brushed” or “normal” circles. Intestine on the left, kidney on the right; airway, epididymis, and nervous tissue with blood-brain barrier (BBB) on the top; the primary sites of carnitine storage: muscle, liver, and heart on the bottom. OCTN2-mediated carnitine (Car) transport is described by continuous arrows; dotted arrows refer to the transport of other OCTN2 substrates (acetylcarnitine, acetylCar; acylcarnitine, acylCar; γ-butyrobetaine, γ-BB). The transport mechanism is reported in the liver and kidney as an example for all tissues. Dotted lines represent transporters that are different by OCTN2. The question mark is for the still undefined transporter. Intestine and tubular lumens are depicted as white-gray dotted circles and blood as a dark-gray circle. Mitochondria are depicted by sketches. Simplified human and bacteria metabolic pathways are depicted: gut bacteria carnitine metabolism leading first to trimethylamine (TMA) and concluding in the liver with the trimethylamine oxide (TMAO) production; acetylcholine synthesis involving, among others, the enzymes carnitine acetyltransferase (CAT) and choline acetyltransferase (ChAT); surfactant production; sperm maturation; carnitine biosynthesis starting by trimethyllysine (TML); mitochondrial oxidation (β-ox).
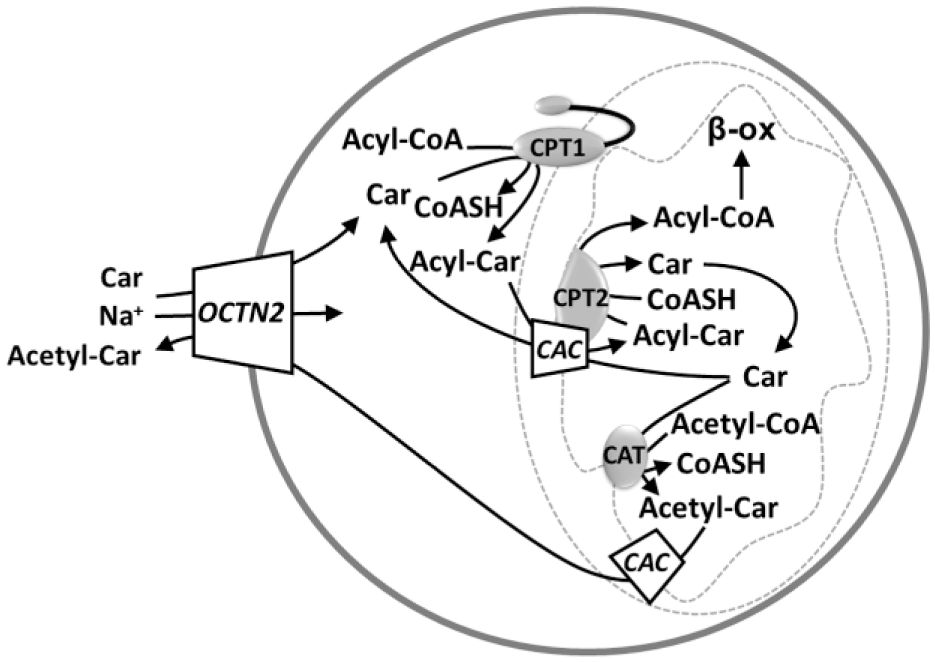
OCTN2 mediates the sodium-dependent cellular uptake of carnitine. Carnitine palmitoyltransferase I (CPT1) transfers the acyl groups from coenzyme A (CoASH) to carnitine (Car), forming acylcarnitine (AcylCar), which can enter into the mitochondrial matrix thanks to carnitine/acylcarnitine carrier (CAC) that operates in an antiport mode. Indeed, it catalyzes the exchange of acylcarnitine (AcylCar) with free carnitine. At this point, carnitine palmitoyltransferase II (CPT2) transfers the acyl moiety from carnitine to coenzyme A, forming acyl-CoA, which will be subjected to beta-oxidation. CAC is also able to mediate the mitochondrial export of acetylcarnitine (AcetylCar), while the cellular export of this compound could be mediated by OCTN2. The acetylcarnitine formation is catalyzed by CAT.
For translational medicine, information on the human transporter is necessary. The information on the human transporter function mostly derives from experiments in cancer cell lines.17,50–55 Differences in the affinity for carnitine are highlighted between the mouse/rat and the human proteins. The Km of the human transporter is one order of magnitude lower than the Km of the rat transporter, 56 as measured in different experimental systems.11,57–59 The internal Km, which could be measured using the proteoliposome approach, for the rat OCTN2, is much higher than the external one. Interestingly, the difference between the external and internal Km values reflects the difference between the extracellular and intracellular carnitine concentration, respectively. The internal Km for the human isoform is still not available. More detailed information on the human OCTN2 kinetics may derive by future studies using the protein overexpressed in bacteria. 60
OCTN2 in Human Metabolism
The major metabolic function of OCTN2 is that of maintaining the carnitine homeostasis. Other parallel and/or apparently minor functions have been proposed and, in some cases, demonstrated. The maintenance of carnitine homeostasis is an organism-level process ( Fig. 1 ). It depends on the balance among the absorption from the diet, the endogenous biosynthesis, the tissue distribution, and the renal reabsorption. 47 Although few tissues allow carnitine replenishment to blood, many “carnitine not-producing tissues” take up carnitine. OCTN2 performs most of these jobs from absorption/reabsorption to distribution because of its ubiquitous expression ( Fig. 1 ). This is definitively demonstrated by the observation that defects of the transporter strongly compromise the function of several tissues leading to PCD. Other transporters, namely, ATB0+ (SLC6A14) and OCTN1, which transport carnitine with lower affinity with respect to OCTN2, are not able, per se, to maintain the homeostasis, but they can only partially compensate OCTN2 defects. Another carnitine-specific transporter is CT2 (SLC22A16), also known as FLIPT2, which is expressed mainly in testis. 61 FLIPT1 (SLC22A15) is a broadly expressed putative transporter, which shows similarity with the OCTNs, indicating that it might also participate in carnitine transport. 62 Carnitine homeostasis guarantees the functioning of the mitochondrial carnitine system, which, in turn, allows translocation of fatty acyl units deriving from cytosolic Acyl-CoA to the mitochondrial matrix for the β-oxidation process ( Fig. 2 ). Carnitine also allows extrusion of acetyl (and acyl) residues, deriving from amino acids or lipid catabolism, avoiding the trapping of intramitochondrial CoA, which is caused by formation of acyl- or acetyl-CoA, under conditions of low oxaloacetate level and, hence, scarce citrate formation and efflux from mitochondria for fueling the adenosine triphosphate (ATP) citrate lyase. By the action of the mitochondrial carnitine acetyl transferase (CAT), the acetyl units are transferred back from acetyl-CoA to carnitine, forming acetyl-carnitine,63,64 which can be exported from mitochondria via the CAC. 65 Then, OCTN2 mediates cell excretion ( Fig. 2 ). Carnitine derivatives, in the form of acyl-carnitines of various chain lengths, are present in cells and in plasma, and their concentrations can change depending on nutrition states. It has been observed that the plasma concentrations of these derivatives are mainly regulated by efflux from myocardium and uptake from other tissues ( Fig. 1 ). The efflux process, which is Na+ independent, occurs when the acyl-carnitines in myocardium exceed the plasma concentration. On the basis of the function of OCTN2, it can be asserted that the regulation of acyl-carnitine fluxes is mediated by OCTN2. 66 Interestingly, a total of 117 different acyl-carnitines 67 were detected in plasma and urine samples, confirming the role of carnitine as a scavenger of acyl groups.33,68,69
OCTN2 in Nutrition
Some specific nutritional states could impair carnitine homeostasis. Carnitine absorption in humans under a regular diet regimen (i.e., consuming either meat, fish, dairy products, or vegetables) accounts for up to 75% of the body’s carnitine content. The endogenous synthesis in these conditions accounts for less than 25%. The low contribution of biosynthesis is caused by insufficient enzyme activity and by limited transport of some precursors, such as γ-butyrobetaine,70,71 which is transported by OCTN2 72 ( Fig. 1 ). A variable fraction of excreted carnitine is reabsorbed at the kidney level by OCTN2 for homeostasis completion44,73–75 ( Fig. 1 ). Indeed, defective carnitine transport is responsible for carnitine loss in urine, low levels of blood carnitine (0–8 µM), and reduced carnitine accumulation inside cells. 74
Under a strict vegetarian diet, the absorption dramatically falls from 75% to even much less than 10% 44 of that under a normal diet regimen because vegetables contain much less carnitine than meat and fish. 76 This deficiency is, at least partially, compensated by increased renal reabsorption.44,77–79 Much lower, if any, is the contribution of increased biosynthesis. 79 Other reports indicate that the amount of body carnitine deriving from biosynthesis is not different between vegetarians and omnivores.44,80 Some discrepancies are present in reports concerning carnitine level in the muscles of vegetarians. Altered or equal levels with respect to omnivores are reported. 77 Thus, the role of OCTN2 is again fundamental. It may be hypothesized that the increased kidney reabsorption is mediated by control of OCTN2 expression, as it occurs in muscle. 81 No apparent consequences related to the reduced carnitine levels are observed, except of that in the case of exercise. Thus, whether or not reduced muscle carnitine content occurring in vegetarians may affect physiologic functions remains to be established. This issue is important in light of the role of carnitine concentration in limiting the fatty acid β-oxidation.77,81,82 Serum carnitine levels are markedly depressed in vegetarian children and in premature infants who do not receive a dietary source of carnitine. This could be also ascribed to the immaturity of the biosynthetic conversion machinery of trimethyllysine into carnitine.44,80,83,84 Other authors show that dietary carnitine is probably sufficient for normal function and, possibly, only a strict vegetarian diet in combination with a defect in carnitine biosynthesis would generate symptoms of carnitine deficiency. 82 Moreover, it is known that an adequate supply of carnitine in suckling neonates is guaranteed by breast milk, where carnitine accumulates by the action of OCTN2 and ATB0+.80,85,86 Thus, it can be assumed that a vegetarian diet in mothers could result in a carnitine defect in suckling neonates. It is then evident that the information on carnitine homeostasis under a strict vegetarian diet is not unequivocal. It is clear, however, that at least in a cohort of the vegetarian population, a lower carnitine level is detected both in plasma and in tissues. Because there are no parallel studies on possible OCTN2 polymorphisms associated to the carnitine defect in this population, it is not possible to assess with certainty whether or not the vegetarian diet is a risk factor per se. On the basis of the present literature concerning studies on pregnant women and infants, our opinion is that a strict vegetarian diet has to be avoided during pregnancy, lactation, and in early-age children, in whom, in addition, some of the carnitine-producing enzymes are still immature. 44 Carnitine is not metabolized by humans and it is excreted as such. Gut bacteria metabolize a fraction of carnitine before absorption ( Fig. 1 ), converting it into trimethylamine (TMA) that, after absorption, is rapidly oxidized by a hepatic enzyme to trimethylamine N-oxide (TMAO). 87 This molecule has been recently the object of investigations for its association with atherosclerosis and cardiovascular risk. However, TMAO-rich seafood has been considered beneficial for the circulatory system. Therefore, the role of TMAO in health and disease remains to be established.88–90 The mechanism of transport of TMAO is still unclear.84,91 It seems that TMAO is not transported by OCTN2 but is transported by OCT2 (SLC22A2). 92
OCTN2 in Epithelia
Concerning carnitine transport in polarized epithelia, some experimental pieces of evidence highlighted that different carnitine transporters are present in brush-border membranes or basolateral membranes of the kidney. This correlates with the different transport mechanisms across the two membranes.93–95 More recent data on localization of transporters highlights that OCTN2, as well as OCTN1, are localized in the apical membranes.39,96 Thus, the flux of carnitine through the basolateral membrane from the intestine to the blood or vice versa is not mediated by OCTN2 ( Fig. 1 ). It was previously hypothesized that OCTN3 could play such a role in the intestine or kidney. 31 However, the OCTN3 coding gene is not present in the human genome.12–14 Among transporters that may cooperate with OCTN2 in the flux of carnitine through the epithelial cells, there is OCT2. 5 This transporter is localized in basolateral membranes and may transport carnitine although with a very low affinity. 31 In summary, the issue of carnitine flux through the basolateral membranes is still not solved. The expression of OCTN2 has been demonstrated in airways97,98 ( Fig. 1 ), where transport of carnitine has been measured. In this tissue, carnitine is used for surfactant production and lung maturation in the fetus. 54 OCTN2-mediated carnitine transport has also been measured in cultured human limbal corneal and conjunctival epithelial cells in which carnitine exerts an osmoregulatory role. 99 The OCTN2 function has also been recently investigated in epididymal epithelial cells, where it would play an important role in sperm maturation by modulating the pathway of spermatogenesis. 100 However, it cannot be excluded that another transporter for carnitine (CT2) may play a relevant role in this cell type, where this transporter is mainly expressed. 101 However, little information is available on this carnitine transporter.
OCTN2 in Neurons
OCTN2 seems important not only for the tissues that require carnitine for fatty acid β-oxidation, such as muscle, heart, and liver, but also for the nervous tissue, where it may play some different roles. OCTN2 may be involved in the transport of carnitine derivatives, such as acetyl-carnitine, for supporting the synthesis of neurotransmitters in the brain ( Fig. 1 ). This hypothesis correlates well with the expression of OCTN2 in neurons and with the finding that acetyl-carnitine administration leads to increased level of acetylcholine.102–105 In this respect, OCTN2 could mediate transport of acetyl-carnitine deriving from other tissues or by the therapeutic administration. This pathway explains, at the molecular level, the possible efficacy of acetyl-carnitine in neuroprotection. 104 Moreover, acetyl units deriving from acetyl-CoA are in part incorporated into brain lipids and/or used for energy production.104,106 These functions might be relevant to Alzheimer’s disease, in which some beneficial effects of acetyl-carnitine have been described. 17 It has to be stressed that OCTN2 is also expressed at the blood-brain barrier (BBB; Fig. 1 ), thus allowing carnitine derivatives to reach neuronal cells. 55 This correlates well with the very low rate, or absence, of carnitine biosynthesis in the nervous tissue.68,82
OCTN1 (SLC22A4) Functional Properties
Unlike OCTN2, OCTN1 is not a carnitine transporter. 58 Various experimental systems (i.e., intact cells, Xenopus laevis oocytes, membrane vesicles, brush-border membrane vesicles, and proteoliposomes23,107–109) have been exploited to investigate the functional properties of OCTN1. In terms of efficiency, TEA is a good substrate, whereas carnitine is a poor substrate. 109 The nonphysiological metabolite ergothioneine is a good substrate 22 as well as the physiological cation acetylcholine. 24 We will resume the latest findings, which add both novel information and open questions about this transporter. In past years, it was confirmed by some studies with cell and mouse models110–112 that ergothioneine is transported by OCTN1. The ergothioneine level in human blood depends on the expression of OCTN1. In fact, it drastically falls in OCTN1 knockout mice. However, human cells cannot metabolize ergothioneine; thus, it must be excreted as such probably via OCTN1 itself ( Fig. 3 ), even though no experimental evidence is present so far. To date, the role of ergothioneine in humans remains ambiguous. The most plausible factor is the antioxidant function in tissues that are exposed to reactive oxygen species (ROS). 113 However, there are no reports of symptoms caused by ergothioneine deficiency. As in the case of ergothioneine, the capacity of OCTN1 to mediate acetylcholine transport has been confirmed by recent studies using the proteoliposome model as well as different cell lines such as primary mesothelial and HeLa cells. Uptake of acetylcholine is inhibited by external sodium, whereas efflux is sodium insensitive. 114 Additional substrates have been identified (i.e., choline and acetyl-carnitine) by studies in cells and in proteoliposome experimental models.25,115 The proteoliposome tool has been revealed to be particularly useful for assessing substrate specificity, as it is an up-to-date model that can unequivocally characterize the function of a single transporter.116–121 Notably, the transport specificity of OCTN1 correlates well with the adaptation of the transported molecules to the binding site, predicted on the basis of the homology model ( Fig. 4 ). Indeed, according to a previous hypothesis on the structure of the binding site formed by three subsites, 17 three main residues tether the substrates, as shown in Figure 5 .
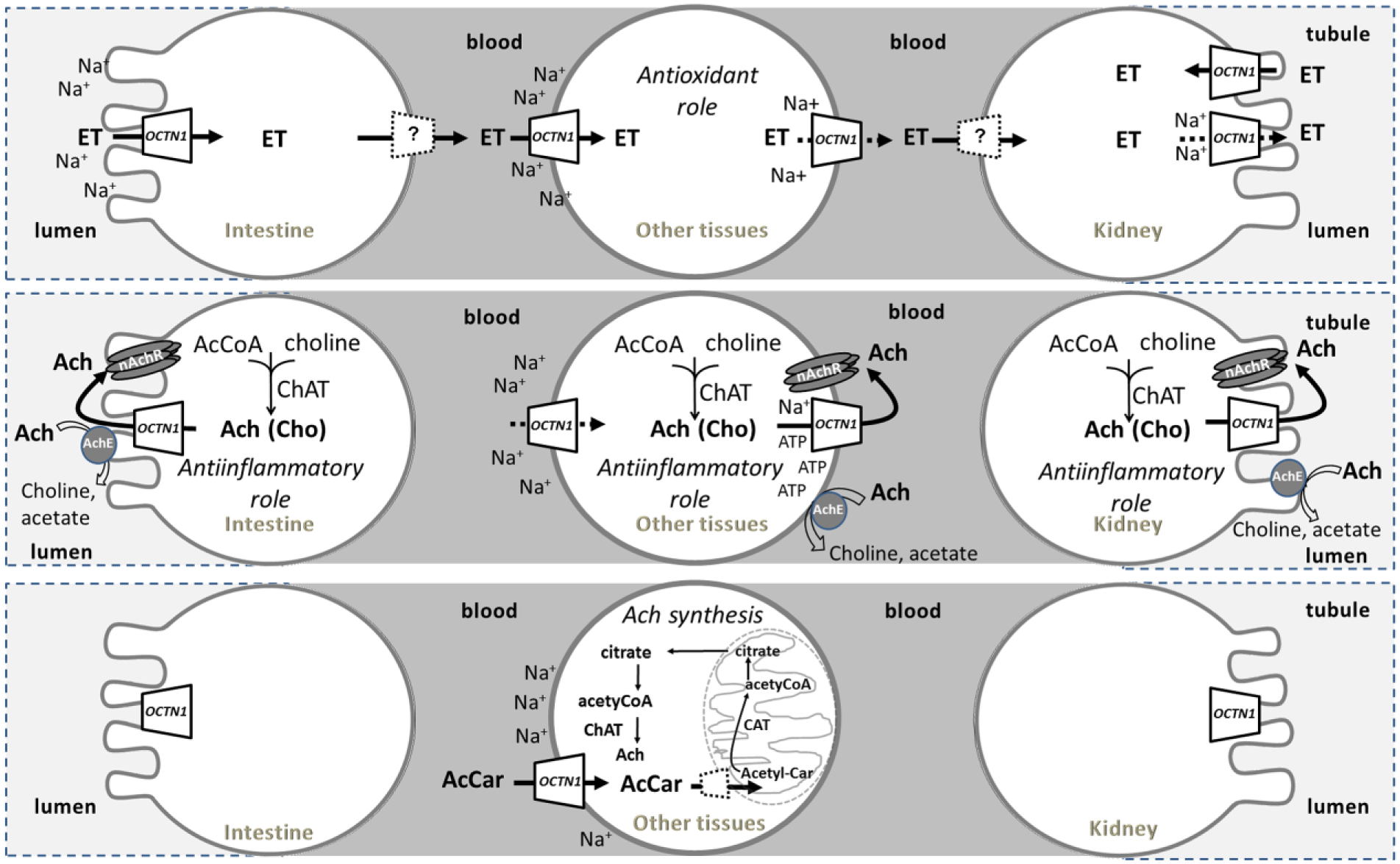
The OCTN1 hypothesized substrates, metabolism, and cellular role. Epithelial polarized and other tissues are depicted as brushed or normal circles. Intestine on the left, kidney on the right; other tissues in the middle. Intestine and tubular lumens are depicted in light gray. Blood is depicted in dark gray. Continuous arrows describe the OCTN1 transport of specific substrates: ergothioneine, ET; acetylcholine, Ach; choline, Cho; acetylcarnitine, AcetylCar. Dotted arrows refer to a very low transport rate.22,114,208 Dotted lines represent transporters different by OCTN1. The mitochondrion is depicted by a sketch. Simplified human metabolic pathways are depicted: acetylcholine synthesis, involving the enzymes carnitine acetyltransferase (CAT) and choline acetyltransferase (ChAT); Ach catabolism mediated by acetylcholinesterase (AchE; gray circle); receptor interaction (nAchR; dark-gray barrel shape). Transport regulation by sodium and adenosine triphosphate is reported.
Homology models of hOCTN1 and hOCTN2 proteins. (
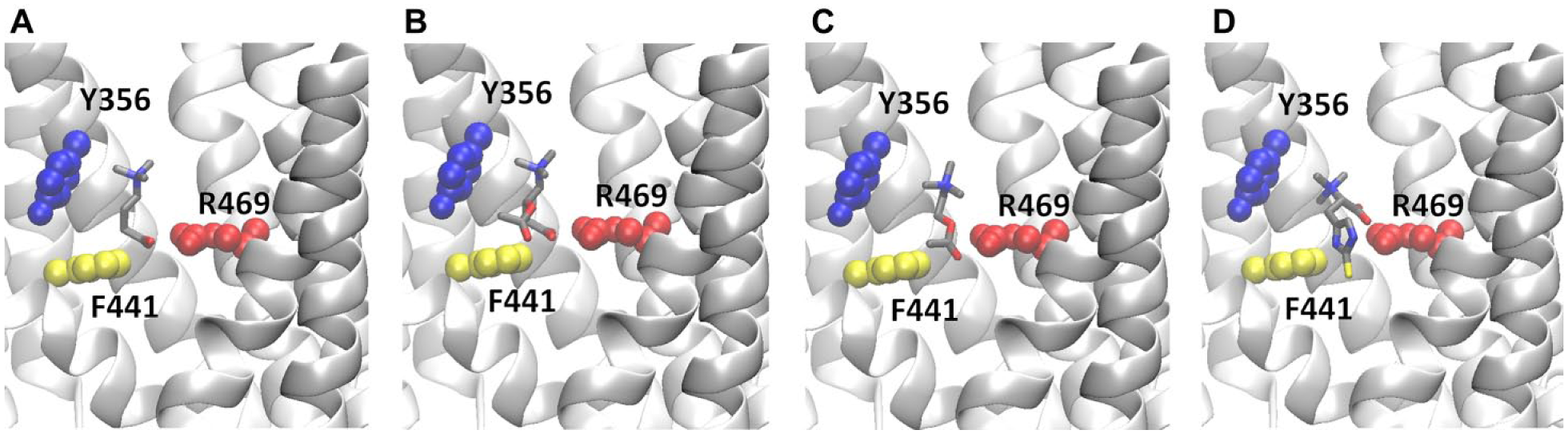
An enlarged view of the interaction of (
OCTN1 in Human Metabolism
After about 20 years of studies on OCTN1, 16 the issue of its role in human metabolism is still not clear. The above-described findings highlight that OCTN1-expressing cells perform a sodium-regulated acetylcholine export that may be part of the nonneuronal cholinergic system. This ubiquitous pathway plays a great number of cell functions, among which is the control of inflammation.25,122 In this regard, a crucial role should be played by OCTN1 in immune cells, in which the nonneuronal cholinergic system is expressed 123 but no transporters with some specificity for acetylcholine, other than OCTN1, are present. 124
OCTN1 in the Immune System
The possible role of OCTN1 in immune cells could be linked to the transport of acetylcholine.17,24,114 SLC22A4 maps within a cytokine gene cluster that is in a susceptibility locus for autoimmunity. This suggests a role of OCTN1 in immunity. The immune system includes many cell types, located in different body tissues in which OCTN1 expression has been demonstrated.27,112,125–127 These cells express the nonneuronal cholinergic system as well. 98 Some authors have shown that OCTN1 may have a role in intestinal macrophage immune function, which is related to ergothioneine transport. 112 Ergothioneine may suppress the activation of proinflammatory macrophages. Thus, OCTN1 up-regulation may be a feedback mechanism of the body to increase ergothioneine absorption for preventing inflammation. The role of OCTN1 in exporting acetylcholine may be important as well ( Fig. 3 ). OCTN1 knockout mice, apparently, do not show any phenotype. It is probable, however, that alterations related to acetylcholine transport could be observed in a medium-long term, after exposure to prolonged inflammatory status. In the lung, where OCTN1 is exposed to environmental pollutants, microbial agents and so forth would be involved in immune defense. This correlates with the expression of OCTN1 in alveolar macrophages.127,128
OCTN1 in Brain
OCTN1 is strongly expressed in brain but not in the BBB. 129 Thus, it cannot play the role of providing neuronal cells with ergothioneine or acetylcholine. Thus, if ergothioneine has a role in this organ, another ergothioneine transporter should be required. 130 This transporter could be OCTN2, which is expressed in the BBB and is reported to transport ergothioneine although with low affinity. 54
Microglia cells, which are nervous tissue immune cells, express OCTN1 ( Fig. 3 ). Microglia play an important role in the maintenance of brain homeostasis 27 by regulating the expression of the inflammatory cytokine interleukin-1β (IL1β). Indeed, chronic overactivation of these cells is involved in the development of various neurodegenerative disorders. 27 Ergothioneine minimally affects the induction of IL1β mRNA. Thus, a substrate different from ergothioneine is required to regulate the expression of inflammatory cytokines via interaction with nicotinic receptors. This substrate could be acetylcholine. 131
OCTN1 in Epithelia
The expression of OCTN1 in airway epithelia may be related to the role of acetylcholine. In this tissue, classical cholinergic nerve fibers coexist with nonneuronal cholinergic cells. Thus, it is intrinsically difficult to identify the two different pathways of secretion of acetylcholine and, hence, the effects deriving from neurons and/or other cells. In this regard, novel implications of acetylcholine are emerging in remodeling and in inflammatory processes that are not exhaustively explainable by the neuronal system. Importantly, alveolar macrophages express nicotinic and muscarinic receptors on their surfaces, which are involved in anti-inflammatory or proinflammatory effects.122,123 Thus, expression of OCTN1 in the surrounding cells may mediate acetylcholine release responsible for the receptor activation. The presence of preneuronal cholinergic signaling has been demonstrated in regions of airway epithelium, such as the trachea, where sensory nonneuronal and neuronal cells are nearby. The nonneuronal sentinel cells taste the presence of bacterial products and then release acetylcholine-activating sensory neurons. In this intricate cell protection system, OCTN1 could be involved in releasing acetylcholine. 98 On the basis of previous findings concerning the specificity of OCTN1 toward bacterial metabolites,23,24 the transporter could be involved in chemical sensing as well. OCTN1 has been also found in human mesothelial cells and in rat peritoneum. 25 In this serous membrane, the transporter catalyzes acetylcholine release as well as acetyl-carnitine transport ( Fig. 3 ). These fluxes are very probably related to the control of inflammation, which is very important during peritoneal dialysis. 25
Relevance to Human Pathology
Missense variants of OCTN1 and OCTN2 present in the dbSNP database (https://www.ncbi.nlm.nih.gov/snp) are reported in Supplemental Table S1 . A total of 459 variants of the NM_003060.3 gene coding for OCTN2 are reported, 171 of which are confirmed by at least one validation method ( Suppl. Table S1 ; Suppl. Fig. S1 ). About 24% of the variants have clinical significance. Most of these are associated with carnitine transport defects; conflicting or uncertain significance is reported in other cases. A total of 366 variants are reported for NM_003059.2 coding for OCTN1, 120 of which are validated. Only one variant, leading to L503F substitution, shows clinical significance and is associated with increased susceptibility to Crohn’s disease. 125
OCTN2
OCTN2 provides tissues with carnitine to perform β-oxidation (
Figs. 1
Another consequence of OCTN2 defect might be the development of respiratory distress in premature infants due to reduced surfactant action of carnitine derivatives, which are able to decrease the surface tension of the alveoli. 133
Carnitine plays an antioxidant role, acting as a free radical scavenger 134 also in neurons. Therefore, impairment of OCTN2 activity causes a reduced supply of carnitine to the brain through the BBB, where OCTN2 is normally expressed. In fact, carnitine deficiency has also been observed in patients with autism, confirming that carnitine and carnitine derivatives have important roles in nervous tissue, especially in the early stages of life during brain development. 74 Another defect of the OCTN2 gene has been recently described. The gene analysis revealed a premature stop codon and an in-frame deletion. This defect is associated with attention deficit/hyperactivity disorder. 103 Furthermore, OCTN2 is linked to inflammatory processes; in fact, OCTN2 variants and carnitine deficiency are associated with inflammatory bowel disease, Crohn’s disease, and asthma.125,135–138 Apart from the OCTNs, the carnitine transporter SLC22A16 (CT2 or FLIPT2) has relevance to pathology. It is up-regulated in gastric cancer 139 and is involved in the response to chemotherapy. 140
OCTN1
OCTN1 plays a major role in the control of inflammation. In this respect, acetylcholine-mediated transport may explain, even better than ergothioneine, the role of OCTN1 in inflammation-based diseases, such as inflammatory bowel disease, according to the nonneuronal cholinergic system. 123 Interestingly, the L503F variant of OCTN1, associated with the Crohn’s disease, loses the function of exporting acetylcholine. 24 To evaluate the consequence of ergothioneine deficiency, an OCTN1 knockout mouse has been generated. In this model, a significant decrease of ergothioneine levels in tissues is detected, even though the animals are viable with no apparent phenotype. The most noticeable effect of OCTN1 ablation is a slight but significant decrease in tolerance to intestinal oxidative stress. This finding may contribute to explain the Crohn’s disease inflammatory state of the intestine, typical of human patients. 141 Another mice model of chronic kidney disease was used to gain insights into the possible role of ergothioneine and the relationships between alterations of kidney functionality and ergothioneine intestinal absorption via OCTN1. These mice show an impaired intestinal absorption of ergothioneine due to a decreased transport by OCTN1. The transport defect is not caused by reduced expression of the transporter but by a reduced expression of PDZ domain containing 1, a scaffold protein responsible for stabilization of OCTN1 in the membrane. On the contrary, the same mice showed an increased kidney ergothioneine concentration. 110 These findings may suggest a role of the transporter in interorgan cross-talk. 142 Such a mechanism suggests an involvement of OCTN1 in a response to altered redox state and might explain the apparent lack of phenotypes associated with the knockout of OCTN1. We may suppose that metabolic alterations arise only after medium-/long-time ablation of the transporter and/or exposure to stress conditions. Very interestingly, a role for OCTN1 is proposed in the nervous system after confirming the neuronal expression of this transporter26,28,29,143 that was not described in the first reports. 11 Taking into consideration that many neurological diseases are caused by oxidative stress, the ergothioneine transport mediated by OCTN1 is plausibly linked to the antioxidant property of the mushroom metabolite ( Fig. 3 ). It has to be highlighted that the capacity of OCTN1 to transport acetylcholine and choline may be involved also in neurodegenerative disorders. In good agreement, the expression of OCTN1 in neurons is linked to differentiation by reducing proliferation, as proposed by Nakamichi et al.26,29 Since the molecular mechanism and the involvement of ergothioneine in neuronal differentiation are still not clarified, it can be also hypothesized that, in this cell type, acetylcholine acting as a hormone and not only as a neurotransmitter may affect neuronal growth and development.26,28,29 Finally, an association between neurogenesis and antidepressant effects has been suggested, linking OCTN1 activity to depression. 130
Regulation of the OCTNs
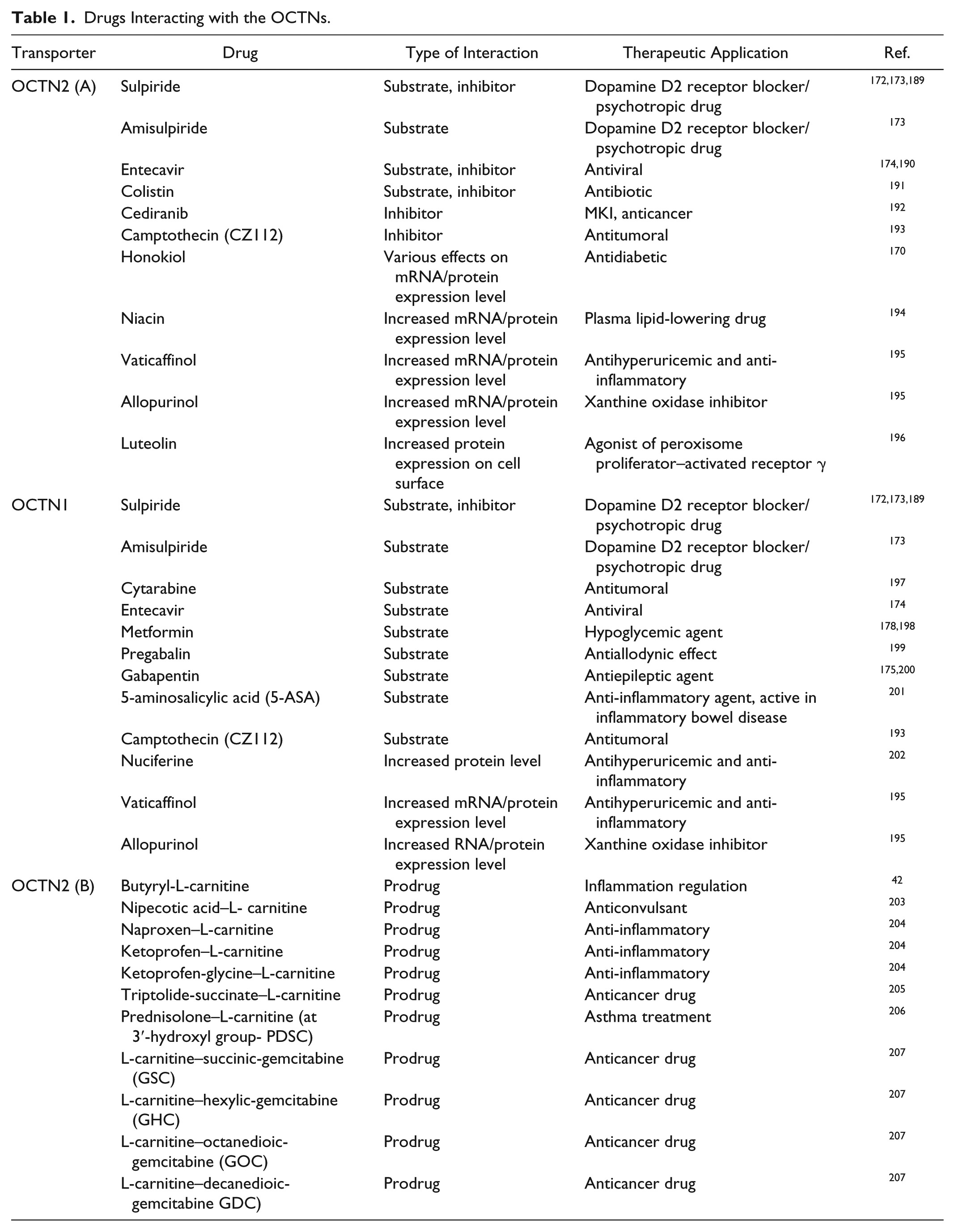
OCTN2 expression is closely related to its function in carnitine homeostasis and also in drug uptake. Information on OCTN2 regulation has been previously reported. 17 Some advancements on this aspect are reported in this section. It has been described that the peroxisome proliferator–activated receptor alpha (PPARα), together with PPARβ and PPARδ, are responsible for the modulation of OCTN2.144,145 The level of plasma-free carnitine in mice, subjected to acute exercise, decreases. The rapid return at a normal level is due to an increased reabsorption at renal level induced by both mRNA and protein level increase. 146 Recently, carnitine has been identified as a modulator of the immune response. Indeed, during the transition of monocyte to macrophage, the rate of carnitine uptake increases. This is due to a higher level of OCTN2 expression. During the macrophage differentiation induced by the monocyte colony-stimulating factor, cytokines activate a mammalian target of rapamycin (mTOR) kinase leading to the activation of the signal transducer and activator of transcription 3 (STAT3), which, in turn, regulates the OCTN2 expression. 147 OCTN2 is also involved in mediating the uptake of different drugs, among which are antineoplastic agents ( Table 1 ). Furthermore, the expression of OCTs and especially OCTN2 in bronchial-epithelial tissue is involved in the uptake of bronchodilators. The expression of OCTN2 is induced after physical injury or treatment with lipopolysaccharide. 148 Both free and total carnitine concentrations are elevated in the liver upon exercise. This is likely related to an increased uptake since the mRNA of the carnitine transporter Slc22a5/OCTN2 is acutely up-regulated. 64 The Granulocyte-macrophage colony-stimulating factor–mTOR-STAT axis induces an increase of L-carnitine transport by increasing OCTN2 expression during macrophage differentiation. It has been hypothesized that carnitine could serve to provide mature cells with fuel to meet the increased demand for energy for exerting their immune functions. 147
Drugs Interacting with the OCTNs.
OCTN2 forms a complex with the protein phosphatase 2 and phosphorylation by protein kinase C (PKC) of one of the regulatory subunits of this enzyme, and its release from the complex, facilitates OCTN2 transfer from the cytoplasm to the plasma membrane of astrocytes. Upon PKC activation, OCTN2 binds more caveolin-1, further facilitating the localization of the transporter to the plasma membrane. 149 Lastly, it has been postulated that PKC-dependent phosphorylation of the Zonula occludens-1 (ZO-1) leads to ZO-1 dissociation from OCTN2, decreasing transporter activity. 150 Therefore, it is not the fully phosphorylated ZO-1 that keeps OCTN2 in its active state. Thus, activation of PKC, although not leading to transporter phosphorylation, regulates OCTN2 at different steps of its trafficking and controls its activity through affecting proteins that interact with the transporter.
Hypermethylation of the OCTN2 promoter is inversely correlated with the levels of OCTN2 in cancer cells. 151 Application of a demethylating agent increases OCTN2 expression, leading to enhanced uptake of oxaliplatin. 152
Much less new information is available on OCTN1 regulation. 17 mRNA of OCTN1 is subject to variations induced by some drugs, as reported in the next section.
Structural Information
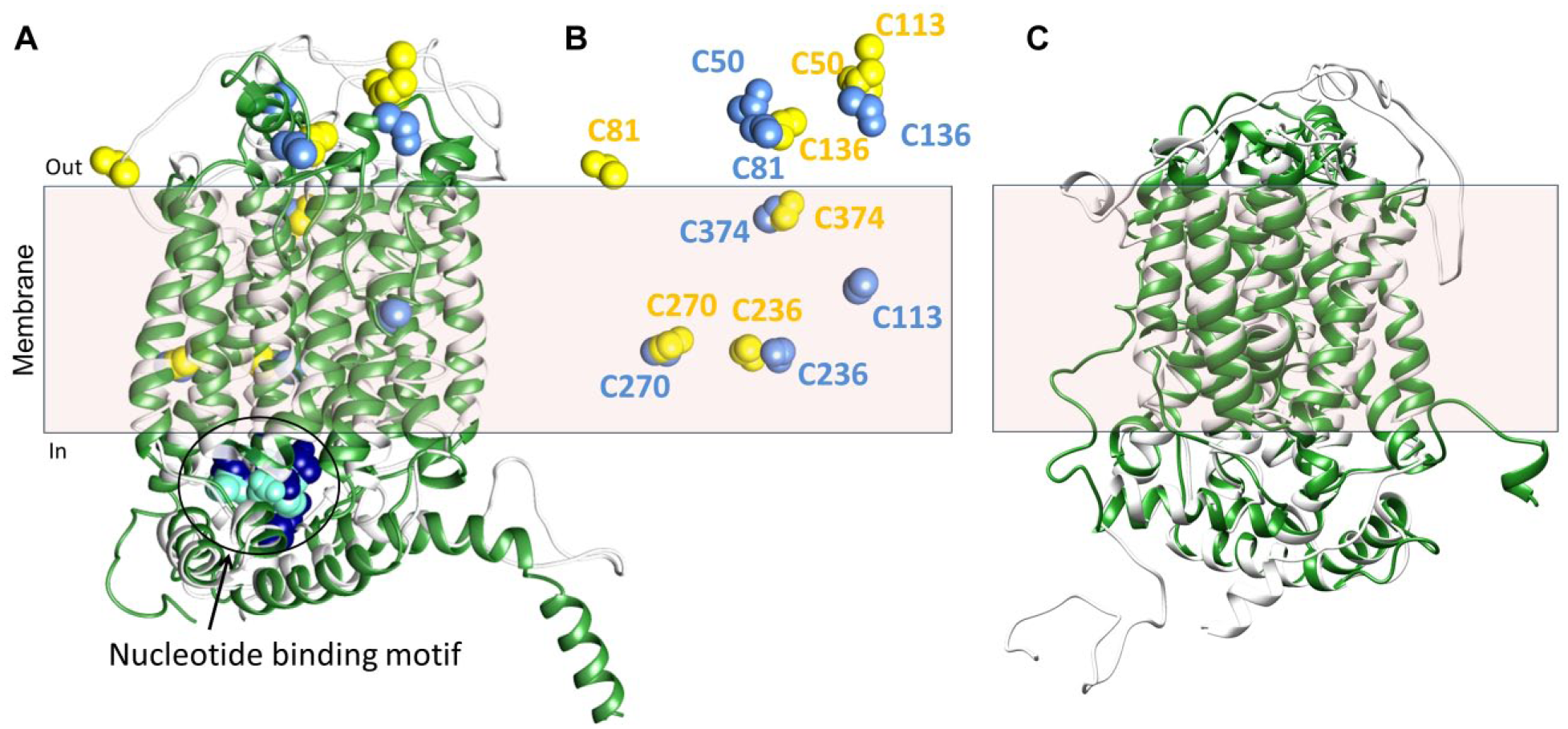
The main challenge in obtaining the structure of membrane transporters is the intrinsic difficulty in crystallizing these proteins for two main reasons: the high hydrophobicity, which hampers the production of highly concentrated pure proteins, and the mobility, which hampers the growth of ordered crystals. Bioinformatics has been employed to obtain information on the OCTN structures by in silico methodologies. After preliminary structural analysis highlighting the topology of the transporters, 15 attempts have been made to obtain homology structural models. However, the structures to be used as possible templates show a low degree of homology with the OCTN sequences (i.e., <20%). The first homology model of the OCTN1 protein, obtained in 2012 using a de novo prediction EVfold membrane algorithm, lacks the first 142 amino acid residues, which include the first transmembrane domain and the large extracellular hydrophilic loop. 153 In 2013, another homology model was reported, which was obtained by comparative modeling using Modeller 9v9 software with the glycerol-3-phosphate transporter (1PW4) as a template. In this case, the amino acid residues 43–141 were absent. 154 A novel model has been obtained using the Phyre2 server that identified, as the best template, the eukaryotic phosphate transporter from Piriformospora indica (4J05) solved in 2013.155–157 In this model, the hydrophilic loop was present, even though it had a random coiled structure. Finally, the I-TASSER automatic server that includes comparative modeling, threading, and ab initio modeling strategies has been employed by Dakal et al. using the human glucose transporter GLUT3 158 as a template. 159 To validate the existing homology models, we employed an approach based on the accessibility of Cys residues of the transporter to specific hydrophilic SH reagents. The variation of accessibility was tested on site-directed mutants, in which each of the seven Cys residues was substituted with Ala. The accessible Cys residues are C50 and C136, as assayed in proteoliposomes. 156 Comparison of the data with the two more recent structures highlights that the two reactive Cys residues are indeed accessible in both models ( Fig. 4B ). The relationships of the Cys with the membrane and hence the accessibility of the thiol residues in each model are highlighted in Figure 4B , showing the single Cys residues in the same position held in the protein models. The superposition of the two models ( Fig. 4A , B ) highlights the small differences in the transmembrane domains as confirmed by the superposition of the intermembrane Cys residues (i.e., C236, C270, C374). An ATP binding site, predicted by ScanProsite, is present in the intracellular loop ( Fig. 4A ). The localization in the internal side of the transporters correlates well with the regulatory role of the nucleotide. Figure 4C depicts the superposition of the two available OCTN2 models built by the Phyre2 server (Galluccio et al., unpublished results) and by Dakal et al.157,159 Some improvement is needed to correctly model the large hydrophilic loops. A similar experimental approach, based on site-directed mutagenesis and targeting assays, should be assessed in the future also for OCTN2 model validation. As it is well known, the 3D structure of a protein is the crucial starting point for drug design using the docking approach. 160 In the case of the OCTN members, whose interest in pharmacology is increasing a lot, the homology models could be initially used for allocating substrates and then for starting the design of potential interactors ( Fig. 5 ).
OCTNs in Drug Discovery
Plasma membrane transporters are attractive as drug targets for several reasons. Their location on the cell membrane makes transporters the first-level targets for drugs. Indeed, transporters located in the apical membrane of intestinal epithelium are accessible to administered drugs, while internal tissues are accessible from the bloodstream (
Figs. 1
Up-to-Date Methodologies
The most widely used methodologies for identifying interactors of transporters include in silico procedures, which make use of computational approaches and are often the first step toward the screening and identification of lead compounds. This is mainly due to the much lower costs with respect to the biochemical and molecular biology methodologies and to the possibility of screening a very large number of molecules, up to several hundred thousand. A number of excellent reviews summarize these methodologies in their applications to transporters.160,161 To use in silico approaches, the structure of the transporters or, at least, a refined homology model must be available. The basic features of the active site and interaction with the putative substrates of OCTN1 can be summarized for OCTN1, as highlighted in the enlarged view of the model ( Fig. 5 ). Hopefully, this represents a starting point for in silico studies. Validation of the interaction of chemicals, either substrates or designed inhibitors, with the transporters must be performed with biochemical approaches. To this aim, transport can be followed using different up-to-date techniques both in cell systems and in proteoliposomes. The preferred method, which gives the highest level of detection sensitivity, is the use of radioactively labeled substrates. The same procedure is also used for inhibition studies adding chemical compounds together with the tracer substrates. 162 Mass spectrometry is used in some cases to trace the movement of substrates from one compartment to the other of cells. Mass spectrometry also provides structural identification of the individual components with high molecular specificity but with a lower detection sensitivity with respect to the radioactivity-based methodologies. The dose-response analysis of inhibition of the transport activity by chemicals can be performed by the two methodologies to derive the IC50. A good indication of a well-conceived inhibitor is given by more or less coincident IC50 values as derived from the in silico docking analysis and from the biochemical approach.162,163 In general, procedures for high-throughput screening (HTS) in the case of OCTNs cannot be generated before pointing out low-cost and radioactive-free methodologies for transport assays. Indeed, for HTS apparatus, ultraviolet or visible detection methods are preferred if not exclusive. Developing standard spectrofluorometric procedures for organic molecules is a frontier research topic that will give, in the future, a strong advancement to the transporter-based HTS. The most interesting advancements on the OCTNs-drug interaction of the past years are reported in Table 1 , classified on the basis of the three types of interactions with the transporters underneath (i.e., inhibitors, transported substrates, or action on mRNA/protein levels). The results obtained so far on transported drugs have been collected in cell systems. In these models, the proofs of action of a specific transporter are given by the transient or stable transfection of the transporter. As a proof of concept, in some cases, transporters are also silenced, as in the case of metformin ( Table 1 ). In this respect, the technique of genome editing, namely, CRISPR/Cas9, will give further advancements because of the possibility of stably knocking-out or knocking-in a gene. 164
OCTNs and Physiological Drugs
A peculiarity of the two human OCTNs is that some of their substrates are drugs themselves. As an example, carnitine and some derivatives are commonly used as drugs for specific pathologies.
63
Indeed, carnitine at high doses, used in treatment of PCD, can be distributed to tissues via a defective OCTN2, although with a lower efficiency. In the case of a complete loss of OCTN2 function, carnitine can be taken up via the low-affinity transporters ATB0+ or OCTN1 if administered at high concentrations. Acetyl-carnitine represents another example. OCTN2 is involved in absorption of administered acetyl-carnitine and delivery to the blood first and then to the central nervous tissue through the BBB. This sequence of transport processes allows acetyl-carnitine to improve the acetylcholine synthesis in neurons (
Figs. 1
Interaction with Exogenous Compounds
The interaction of OCTN2 and OCTN1 with exogenous compounds concerns three further aspects:
Inhibition of physiological transport
Capacity of transporting specific chemical compounds and enabling drug delivery
Effect on expression of the transporter acting at the transcription/translation
Searching for an inhibitor is normally performed through the design of chemical compounds sharing structural similarity with the substrates. This strategy has been widely employed for amino acid transporter inhibitors. 165 In this regard, an additional advantage for OCTNs is their polyspecificity, which allows a wide collection of possible chemical compounds interacting with the transporters and inhibiting their activity. Indeed, the specificity of these proteins toward many exogenous compounds has been already well documented and summarized in previous reviews.5,17,18 Thanks to the improvement of the methodologies for studying transport in cells and/or artificial systems and of silencing specific transporters, in the past 5 y, the involvement of OCTN2 and OCTN1 in drug interaction has been more precisely assessed ( Table 1 ).
Among the possible roles of the OCTNs related to drug transport, there is the uptake of inhibitors of specific metabolic pathways. This may have benefits in pathological states, as it occurs in the case of amino acid transporters, which are overexpressed in cancer. 166 As an example, silencing the β-oxidation of fatty acids by inhibition of OCTN2 will result in switching the metabolism toward glucose utilization. This metabolic switch is considered beneficial in ischemia and reperfusion. To this aim, mildronate, a typical competitive inhibitor because of its structural similarity to carnitine, has been exploited in the past. This drug, besides interacting with OCTN2, also has the effect of inhibiting enzymes involved in carnitine biosynthesis. However, after revealing dangerous side effects, it has been removed from use in human therapy. 167 The search for novel inhibitors of OCTN2 is therefore an important issue in the field of pharmacological treatment of ischemic injuries.
Concerning the issue of drug delivery, many compounds have been demonstrated or suggested to be delivered through OCTN2 or OCTN1, even though these compounds are substrate unrelated ( Table 1 ).168,169 Clear structure-function relationships cannot be drawn from the few available data. It seems that some requisites such as the presence of carbonyl, carboxyl, or amino groups may be important for being an OCTN1 substrate. These functional groups may resemble the functional groups of acetylcholine or ergothioneine. Larger structures, such as cediranib or campthothecin, behave exclusively as inhibitors, indicating a size limitation.
Interestingly, together with synthetic chemical compounds, some natural compounds also have been reported to act on OCTNs with a third type of interaction: regulation of mRNA or protein levels. Some of these compounds down-regulate the mRNA level, and others lead to opposite effects, as reported in Table 1 . In some cases, more complex effects are described as in the case of honokiol, which down-regulates mRNA expression of renal OCTN2 in rats with type 2 diabetes whereas it up-regulates the mRNA of hepatic OCTN2. 170 An aspect of OCTN2 pharmacology that deserves attention is linked to the role that this protein may play in cancer according to the metabolic switch typical of this disease. Interestingly, OCTN2 expression is generally repressed in cancers by epigenetic mechanisms of methylation of its promoter.151,152 Given the possibility for OCTN2 to transport drugs that can have also anticancer effects, pretreatment of cells with demethylating reagents that reactivates OCTN2 expression may represent a possible strategy for optimization of anticancer drugs delivery.
Moreover, because OCTN2 is a critical regulator of carnitine homeostasis, lipid metabolism, and cell proliferation, the transporter has been suggested as a potential therapeutic target for breast cancer. Indeed, OCTN2 is up-regulated in estrogen-responsive breast cancer, which is different from many other cancers. 171
Using different approaches, the capacity of OCTN2 and OCTN1 to transport many drugs has been documented. Despite their own specificity for the substrate, in some cases the two transporters share the specificity for some drugs ( Table 1 ) such as sulpiride, amisulpiride,172,173 or entecavir. 174 These drugs could improve the survival rates of cancer patients. In the case of gabapentin, which is transported by OCTN1, ergothioneine trans-stimulates the secretion of the drug, resulting in an increased clearance. 175
Last, but not least, polymorphisms of OCTN1 and OCTN2 genes may represent other factors affecting pharmacokinetics and pharmacodynamics. 176 Thus, genotyping may be an important predictor of time to progression in patients with gastrointestinal stromal tumors who are receiving imatinib therapy. 177
The L503F OCTN1 variant transports biguanides, especially metformin, more efficiently than wild type. 178 However, polymorphisms of OCTN1 show no significant impact on glycemic response to metformin. 179
Prodrug Approach
The transport mechanism and the tissue distribution of OCTN2 have been exploited also in the prodrug approach.
180
This is characterized by the employment of a poorly active or inactive compound that contains the parental drug. Upon metabolic transformation of the prodrug, the active molecule is delivered, allowing therapeutic effects to occur. Two major groups of prodrugs are classically identified: bioprecursor and carrier-linked prodrugs. In other words, the bioprecursors are normally inactive compounds that are enzymatically or chemically activated upon a redox reaction, as an example. Carrier-linked compounds are characterized by the linkage of a drug to a molecule that increases the solubility and/or bioavailability of the drug of interest (
Table 1
). The carrier molecule, indeed, is chosen for either increasing the water solubility of hydrophobic drugs that cannot be otherwise administered or for ameliorating tissue distribution. In this case, the carrier molecule is used as a Trojan horse to cross biological membranes via transporters able to recognize the carrier molecule as a substrate. This strategy is particularly important for reaching those tissues normally resistant to pharmacological treatment; as an example, the BBB normally constitutes a strong biological hindrance for drugs targeting the brain. In the context of prodrug approach, OCTN2 that is a major carnitine transporter in several body tissues (
Figs. 1
OCTNs in Diagnostics
OCTN2 has also been exploited for diagnostic purposes. The positron emission tomography tracer for tumor imaging, 3-18F-L-a-methyl-tyrosine ([18F] FAMT), accumulates specifically in malignant tumors by means of amino acid transporters. It has been shown that OCTN2 is in part responsible for the renal accumulation of [18F] FAMT. Therefore, inhibitors of this transporter may be used as tools for reducing the [18F] FAMT background. 185
An absolutely novel location and role of OCTN2 has been recently demonstrated. It is present in exosomes isolated from both human urine and human embryonic kidney 293 (HEK293) cell media. Exosomes are vesicles with an endosomic origin, released by several cell types. 186 It is now well assessed that these vesicles play a pivotal role in the modulation of several physiological and pathological processes, such as inflammation, immune response, angiogenesis, cell death, neurodegenerative diseases, and cancer progression. In this regard, OCTN2 represents a novel player of cell-cell signaling, linked to both inflammation and kidney physiology. Interestingly, several studies show that carnitine is associated with inflammation-related diseases, including diabetes mellitus, chronic renal failure, cardiomyopathy, cirrhosis, sepsis,80,187 and Crohn’s disease.125,135,188 In good agreement with the link of OCTN2 and inflammation, treatment of HEK293 with the proinflammatory cytokine interferon gamma increases OCTN2 expression in entire cells as well as in exosomes. Interestingly, OCTN2 derived from HEK293 cells and from human urine exosomes is correctly folded and fully active. The exosomal transporter could represent a vehicle for transferring functional OCTN2 to the inflammation site for recovering the carnitine transport capacity of altered renal cells and controlling the inflammatory state. Therefore, the level of OCTN2 in exosomes may represent a novel biomarker for inflammation. 21 Moreover, the exosomal location of OCTN2 is in line with the interorgan cross-talk hypothesis put forward for OCTN1 as well.
Conclusions
The interaction among membrane transporters and existing or newly designed drugs is a frontier research topic. In this regard, the polyspecificity feature of OCTNs makes these transporters particularly versatile and, hence, eminent targets in this area. Indeed, as occurs for OCTN1, the core of the protein is structured in a way that can tether structurally different substrates (see Fig. 5 ). The capacity to interact with different substrates reflects the ability of this small transporter subfamily to recognize a sizable number of organic cations with different structures as well. Therefore, OCTNs have a central role in drug delivery. Because these transporters have no counterparts in bacteria, but they seem to have arisen in vertebrates, the capacity of recognizing exogenous molecules might be a side effect of evolution. Notwithstanding this consideration, at least in the case of OCTN1, the pharmacologically related aspect is better defined than the physiological role. Thus, from a functional point of view, much work is still required to answer the question: What is the role of the transporter in the human organism? On the other side, OCTN2’s role in metabolism is much better defined. However, besides the major involvement in carnitine homeostasis, additional information has emerged in recent years regarding its involvement in other processes such as inflammation and its exocellular localization. Thus, again in line with versatility, OCTN2 seems to be a promising candidate in noninvasive human diagnostics. Overall, even though significant advancements in the knowledge of these transporters have been achieved, many dark sides have still to be unraveled to reach a satisfying picture in terms of physiopathology and then to fully exploit these proteins for biotechnological applications in drug discovery.
Supplemental Material
Supplemental_Material – Supplemental material for OCTN: A Small Transporter Subfamily with Great Relevance to Human Pathophysiology, Drug Discovery, and Diagnostics
Supplemental material, Supplemental_Material for OCTN: A Small Transporter Subfamily with Great Relevance to Human Pathophysiology, Drug Discovery, and Diagnostics by Lorena Pochini, Michele Galluccio, Mariafrancesca Scalise, Lara Console and Cesare Indiveri in SLAS Discovery
Footnotes
Supplemental material is available online with this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by funds from MIUR (Ministery of Instruction, University and Research): Programma Operativo Nazionale [01_00937] “Modelli sperimentali biotecnologici integrati per lo sviluppo e la selezione di molecole di interesse per la salute dell’uomo.”
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
