Abstract
In recent years, the ability to unambiguously identify complex mixtures of analytes with high accuracy and resolving power in a label-free format continues to expand the application of mass spectrometry (MS) in the drug discovery process. This advantage combined with improved instrumentation makes MS suitable for targets with limited alternative assays for high-throughput screening (HTS). We describe a novel screening format using Self-Assembled Monolayers and matrix-assisted laser Desorption Ionization (SAMDI) technology. SAMDI enables affinity capture of a target protein for use in a small-molecule–binding assay format. Subsequent ionization enables the inferred identification of noncovalent compound interactions. SAMDI technology overcomes shot-to-shot variability by uniformly saturating the surface with captured protein, thereby minimizing matrix crystallization “hot spots.” Furthermore, the combination with high-resolution matrix-assisted laser desorption/ionization time of flight significantly reduces interference of small-molecule detection from salt, detergent, and matrix. By using a pooled library format, the SAMDI assay can significantly improve the throughput of MS-based screening irrespective of enzyme activity. Finally, we demonstrate binding affinity rank ordering from a pool of compounds that correlates with potency data from a biochemical assay.
Introduction
The drug discovery process executes conventional compound library screenings using fluorescence- and chemiluminescence-based detection that affords screening throughput of more than 100,000 compounds per day. However, these detection readouts are prone to interference with compound libraries and can lead to high false-positive rates. The process of confirming real hits and eliminating false-positives can require a number of dose-response triage assays that demand additional time, reagents, and human resources.
Mass spectrometry (MS) can unambiguously identify complex mixtures of analytes with high accuracy and resolving power in a label-free detection format. This advantage makes MS suitable for screening small-molecule compound libraries as new target classes emerge with increasing complexity and limited availability of labeled detection reagents. Limitations on the existing MS front-end technology, however, constrains the sampling rate and throughput despite the fact that modern mass spectrometers can scan as rapidly as tens of microseconds. For electrospray, Agilent RapidFire MS 1 remains state-of-the-art technology, allowing medium throughput in 384-well format and has been implemented as high-throughput screening (HTS) triage where samples can be analyzed in as fast as 15 s. However, this throughput is insufficient for screening libraries that can well surpass 1 million compounds. For reference, a RapidFire MS point screen covers <5% of a typical small-molecule compound library in the same amount of time that a fluorescent-based assay can be executed.
As an alternative to electrospray, 2 matrix-assisted laser desorption/ionization (MALDI) 3 is amenable to speeds required for ultra-high-throughput screening (i.e., >100,000 samples per day).4-8 While MALDI can be more tolerable to salt and detergent, sample cleanup is critical to ensure a high-quality MS response. 9 While cleanup has historically been achieved through solid phase extraction, affinity capture has been previously used in conjunction with MALDI.10,11 The SAMDI technology12,13 pioneered by the Mrksich lab, enables specific capture of analytes for high-throughput sample preparation with a throughput of >100,000 compounds per day. 13 SAMDI uses self-assembled monolayers on gold arrays in 384 or 1536 formats that are compatible with state-of-the-art MALDI time-of-flight (TOF) instrumentation and liquid handling. The monolayers are capped with polyethylene glycol for compatibility with biological molecules and a chemical moiety, such as biotin, to enable capture of a biotinylated-protein target via immobilized neutravidin. This unique MS capture approach imparts several advantages for MALDI assay workflows. First, SAMDI simplifies sample preparation and enables unprecedented purification of up to 1536 samples in <10 s, while reducing MALDI acquisition time to 1 to 2 s per sample. This efficient purification of analyte minimizes ion suppression caused by buffer components, such as salts and detergents. SAMDI also provides uniform monolayers as a foundation for consistent matrix application onto sample spots in the array. Furthermore, sample purification can also minimize charging in the ion optics and significantly reduces source contamination that results in improved robustness.
Although SAMDI technology has been implemented for label-free screening of compounds that modulate functional activities of target enzymes,13–16 application to screening small-molecule binding to protein targets through noncovalent interactions is unprecedented. Native MS17–19 or affinity selection MS20–22 have enabled MS screening of small-molecule drug binding. In the former, detecting a mass shift of the protein intact mass under native conditions can indicate the presence of small-molecule binding. Alternatively for the latter, we infer hit identification from MS detection of the compound mass after isolation of the protein target incubated with a mixture of compounds under native conditions and subsequent dissociation of bound compounds using denatured MS. Aside from lower resolution and sensitivity for native MS, a key challenge associated with either screening strategy is lower throughput compared with conventional fluorescence-based screening assays. Furthermore, affinity selection MS using the current process of protein isolation employing liquid chromatography or plate-based filtration suffers from low retention of compounds with rapid off rates.
Here we describe the application of affinity selection MS of noncovalent compound binders to a protein target using SAMDI that is amenable to HTS. We demonstrate a workflow and proof of concept for a novel screening format of a pooled compound mixture using affinity capture and purification by SAMDI paired with the high sampling speed of MALDI with a 1 kHz laser.
Materials and Methods
Matrix Selection for Small-Molecule Detection
Three representative compounds with molecular weights (MW) in the 200 to 600 Da range were prepared in a serial dilution ranging from 20 to 0.625 µM compound and tested with a panel of 13 matrices ( Suppl. Table S1 ; Suppl. Fig. S1 ). The compound serial dilutions were diluted 1:1 with matrix, and 1 µL was spotted to a MALDI target plate.
Protein and Compounds
Biotinylated protein, β-secretase 22 (BACE) or Dual Leucine Zipper Kinase 23 (DLK; MAP3K12, Carna Biosciences, Natick, MA), no protein control, and the desired concentration of small molecule(s) were mixed in buffer containing 20 mM sodium acetate pH 4.4, 0.002% Tween 20, and 0.1% bovine skin gelatin (BSG; Sigma Aldrich, St. Louis, MO) for BACE or Tris buffer pH 8, 0.002% Tween 20, and a range of 1-0.25% DMSO for DLK.23, 24 BSG and Tween were optimized for the SAMDI process to minimize nonspecific compound binding and spreading on the SAMDI plate ( Suppl. Fig. S2A BSG optimization; Suppl. Fig. 2B Tween optimization at 0.1% BSG). Compounds with a range of inhibitory activities were made in-house from historical small-molecule projects and pooled at 10 mM in DMSO to yield a final assay concentration of 10 µM for BACE and 5 µM for DLK for each individual compound.
Preparation of Protein with Compounds on SAMDI Plates
To minimize shot-to-shot variability and ensure quality and homogeneous signal, the surface of the SAMDI plate should be saturated with biotinylated-protein. Compounds were bound to target proteins either in “solution” or “solid” phase ( Suppl. Fig. S3 ). For solution-phase SAMDI capture, biotinylated-protein samples were incubated with compound prior to affinity capture on SAMDI plates. Compound titrations of a two-fold dilution series starting at 40 µM compound, eight points, were first incubated with 1 µM biotinylated BACE in its respective assay buffer for up to 30 min at room temperature and transferred to a 384-spot SAMDI array that presents a monolayer of neutravidin immobilized onto a self-assembled monolayer comprising 3% (biotin-ethylene glycol)6-SH and 97% (ethylene glycol)3-SH and incubated at room temperature for 60 min. In the solid phase capture format, 1 µM biotinylated BACE protein was captured to the neutravidin-coated SAMDI plate first for 30 min at room temperature and then washed with assay buffer; compounds were then added to allow compound binding to the captured protein on the SAMDI plate for 60 min at room temperature. The solution-phase method was used for individual compound binding EC50 determination, plate uniformity, and pooled compound experiments. Mixtures of either 1 µM biotinylated BACE or 650 nM biotinylated DLK with 10 µM or 5 µM compound pools, respectively, were incubated in a humidified chamber at room temperature for up to 30 min prior to applying 2.5 µL of the solution to a 384-spot SAMDI array. Surfaces were then washed gently three times with 20 mL of water (total 60 mL) and dried using compressed air. Unless otherwise specified, a matrix mixture of 20 mg/mL α-cyano-4-hydroxycinnamic acid (CHCA; Sigma Aldrich, St. Louis, MO) in acetonitrile, 25 mg/mL dihydroammonium citrate (Sigma Aldrich), 0.3% trifluoroacetic acid was applied by dispensing approximately 50 nL on each spot in the array.
MS Detection of Small Molecules Bound to Target Proteins on SAMDI Plates
An AB SCIEX 5800 MALDI TOF-TOF (AB Sciex, Framingham, MA) or a Bruker UltraFleXtreme MALDI TOF-TOF (Bruker Daltonics, Bremen, Germany) was used to acquire data in the 100 to 600 m/z range in reflector positive mode. Specifically, the AB Sciex 5800 MALDI TOF-TOF mass spectrometer was operated with a 400 Hz YAG laser (power 4000, 600 laser shots). Similarly, for the Bruker UltraFleXtreme MALDI-TOF-TOF mass spectrometer, a 1 kHz laser (66% power, 500 laser shots) was used.
Binding of small molecules was quantitated using the area under the curve (AUC) of the small molecule compound peak and the AUC of the (ethylene glycol)3-SH (EG3 thiol) monolayer component with MW 335.2 Da. The compound peak AUC data were normalized to the EG3 thiol monolayer peak AUC, and MS data were analyzed with Genedata Expressionist software (Genedata, Basel, Switzerland) in batch load mode. Compounds were identified and annotated in the spectra based on experimental versus theoretical input mass through database searching by referencing a database file containing compound masses and chemical formulas loaded into Genedata Expressionist. Samples within a given pool of compounds are compared with and without target protein. Compounds with equivalent signal in the presence and absence of protein were identified as nonspecific compound binders and eliminated as hits.
Results and Discussion
SAMDI Workflow for Affinity Capture and MS Detection
The schematic of the SAMDI workflow to identify noncovalent compound binders to a protein target is shown in Figure 1 . The first step involves incubating the compounds with biotinylated proteins in solution to allow complex formation. The solution is subsequently spotted onto neutravidin-coated SAMDI plates for affinity capture of the biotinylated protein with bound and unbound compounds. Plates are washed to remove buffer components and enrich samples, followed by matrix application and finally small-molecule detection with SAMDI-MS for identification of inferred binders.
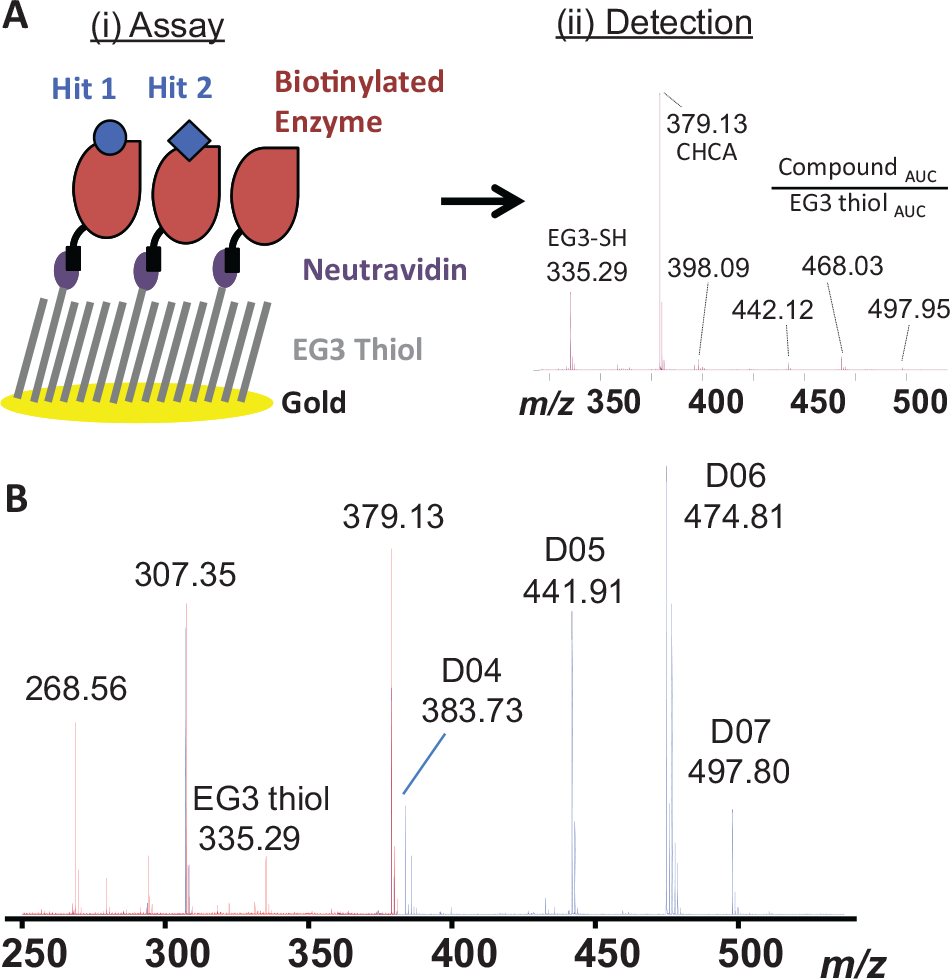
Self-Assembled Monolayers and matrix-assisted laser Desorption Ionization (SAMDI) small-molecule binding workflow. (
Matrix Selection for Small-Molecule MALDI-TOF Detection
Historically, identifying small-molecule compounds with MALDI has been challenging because of interference from the matrix peaks in the low mass range. 25 Recent improvements in MS performance, specifically in mass accuracy and resolving power, have greatly improved small-molecule MALDI detection. To demonstrate this, representative MALDI mass spectra comprising CHCA matrix alone or co-crystallized with a five-component compound mixture (2 µM each) are shown in Figure 1B . One of the five compounds tested did not show sufficient signal, suggesting that poor detection of individual compounds can contribute to false-negatives in this assay format. Poor compound ionization in reflector positive mode or compatibility of the compound with the choice of matrix is one of the likely causes of false-negatives. Compared with electrospray, having higher false-negatives is the intrinsic nature of performing MALDI, and unfortunately for screening a broad class of compounds with wide differences in chemical properties, this may be unavoidable unless new matrices or an improved MALDI process is developed. However, the detection bias can be improved if the screen may be performed for a certain class of molecules with a more specific MALDI preparation.
To assess the impact of different matrices on the ability of MALDI MS to detect small molecules, a panel of 13 matrices was evaluated for low background and high signal to noise (S/N) ratio for the low mass range ( Suppl. Fig. S1A ). Eight of the 13 matrices tested yielded S/N > 100 and mean AUC > 5 × 105 for the tested compounds at concentrations as little as 625 nM ( Suppl. Fig. S1B ). Co-crystallizing with 2,5-DHB showed promising data with low background in the low mass range; however, the signal intensity for each compound also suffered. Although some matrices provided higher S/N and overall intensities, CHCA had fewer impurities in the spectrum that potentially overlap with, and complicate the analysis of, the small molecules of interest. Overall, the CHCA matrix achieved a good balance between S/N and total compound intensity and was therefore selected for small-molecule detection using SAMDI for the remainder of this study.
Solution- versus Solid-Phase Capture for SAMDI Small-Molecule Binding
In the SAMDI small-molecule binding assay workflow, we first tested individual compounds known to bind and inhibit BACE either in a solution-phase or solid-phase capture to determine the best format for retaining specific small-molecule binding to a protein target. In the solution-phase format, compounds and BACE were incubated to form specific complexes in an assay plate and then transferred to a neutravidin-coated SAMDI plate followed by a washing step. In contrast, for the solid-phase capture format, the biotinylated BACE first bound directly onto the neutravidin-coated SAMDI plate followed by a washing step before compound addition for binding to occur.
An n = 4 for each of four compounds of varying molecular weights and affinities were compared individually between solution-phase and solid-phase capture ( Fig. 2A ). The solution-phase capture exhibited approximately twofold higher signal when compared with the solid-phase capture. Although the solid-phase capture would result in a higher local concentration of protein and may yield better detection of weak binders, the solution-phase capture will allow more efficient compound-protein complex formation prior to SAMDI capture, as seen by the overall increase in signal and reduced variability within each sample. Based on these results, the solution-phase capture was considered the ideal format for this work.
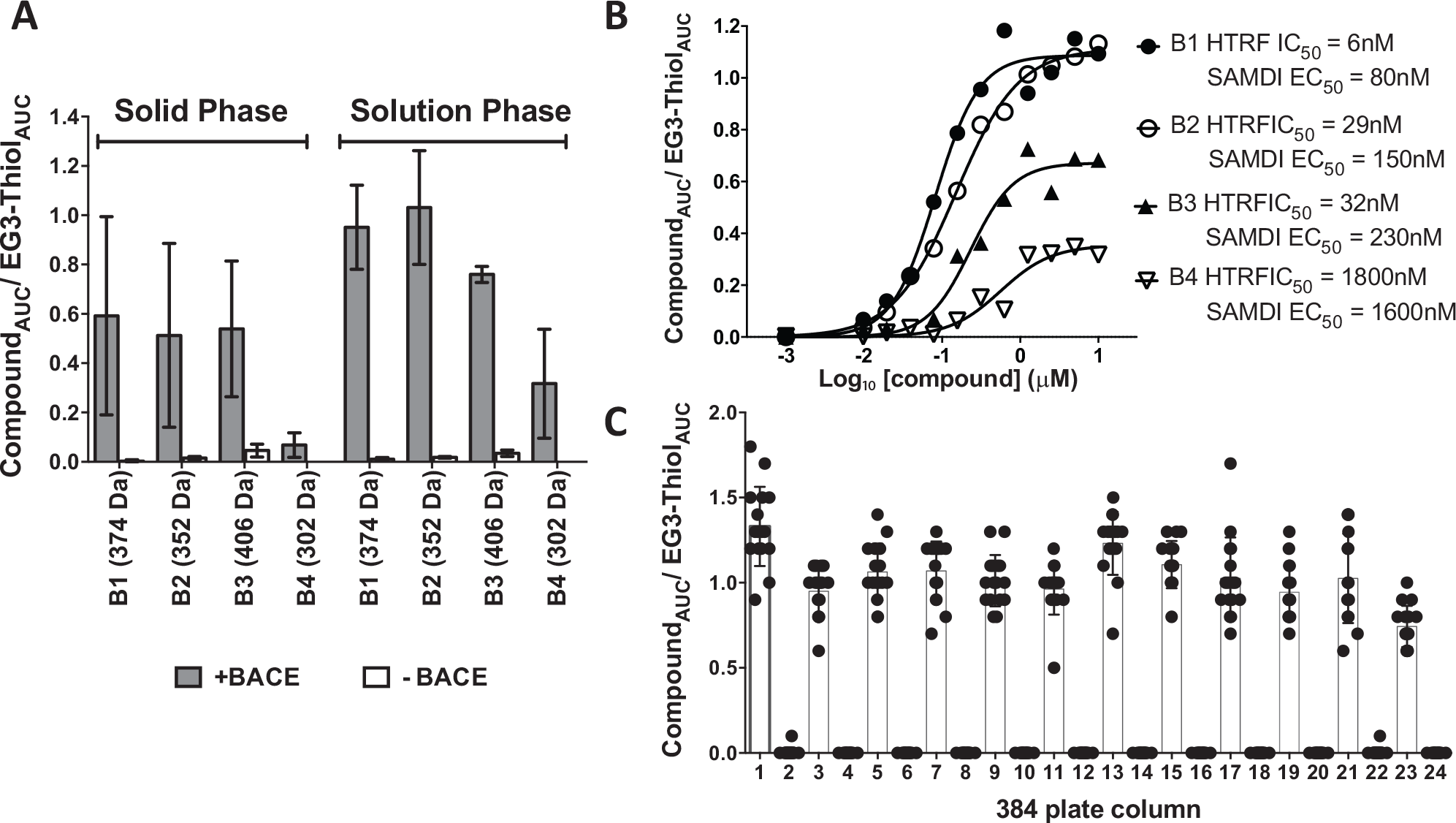
Screening feasibility. (
Compound Dose Response Detected by SAMDI Small-Molecule Assay
Using the solution-phase capture approach, four compounds from a chemical series with varying affinities were titrated individually and incubated with biotinylated BACE. The binding activity of these compounds was previously characterized using BACE time-resolved fluorescence resonance energy transfer (TR-FRET, HTRF CisBio) biochemical assay. 24 The four compounds showed that the SAMDI binding EC50 values (mean values plotted of n = 4 for each compound) ranked in a similar order with the BACE HTRF activity data ( Fig. 2B ). In addition, the maximum percentage bound at 10 µM compound concentration in the SAMDI binding assay correlated with the biochemical activity data, where the weakest compound, B4 at 1.8 µM in the biochemical HTRF assay, showed a maximum percentage bound of 25% and an EC50 of 1.6 µM in the SAMDI binding format. The most potent compound tested, B1 with an HTRF IC50 of 6 nM, showed 100% bound at 10 µM and the highest binding affinity EC50 value of 80 nM in the SAMDI binding format. The lower maximal bound of low-affinity compounds may be the result of faster off-rate compounds dissociating from the protein during the wash step. Optimization of the wash conditions for each protein target will likely improve the retention of weak binders.
Plate Uniformity
For HTS of compound libraries, data uniformity determines the reproducibility of the assay format. Because of the spread in kinetic energies after ionization, MALDI can have signal variation for the same sample. We assessed the signal variation by alternating columns of samples by incubating 1 µM compound, B1 (MW = 374 Da, HTRF IC50 = 6 nM), with and without 1 µM of biotinylated-BACE protein, across a 384-well microplate ( Fig. 2C ), which allowed assessment of consistency of signal across the plate. The mean AUC for compound B1 for samples without BACE was <1000, consistent with the absence of small-molecule detection. In contrast, samples with BACE added resulted in a compound mean AUC of 3 × 105 for compound B1. This represents a 300-fold compound signal difference in samples with BACE compared with samples without BACE. Despite the large AUC associated with target-specific compound signal, the variability of the AUC values was significant with a coefficient of variation (CV) = 48%. To address this issue, we incorporated a normalization step that took into account the variability introduced at any given sample-processing step and from MS detection. Because EG3 thiol comprises the major species of the SAMDI monolayer with a MW of 335 Da, similar to the MW range of a typical small molecule, it can serve as an internal control for each well. Normalizing to the EG3 thiol signal minimized signal variability introduced by the MALDI ionization process and decreased the standard deviation for the compound signal. After EG3 thiol normalization ( Fig. 2C ), the %CV of the samples with BACE decreased from 48% to 22%, and the S/N increased from 300-fold to a window of 400-fold. Future work to further reduce variation will involve analyzing more sample spots to address spot-to-spot variability (possible with faster laser speed). Optimization of washing procedures such as volume and timing of wash cycles may also contribute to improvement in CVs. Despite the apparent higher CV relative to the values typically observed in a functional assay HTS, the hit selection with this type of binding screens is gated primarily on a gain of signal above a defined threshold and thus in theory can tolerate higher CV. By comparing the MS signals of samples with and without protein, we can simultaneously and reliably determine true hits and eliminate nonspecific binding interactions. In cases in which compounds may show only minimal difference between samples with and without protein, additional characterization data defining clear hit-calling criteria prior to a screen will aid in decision making to include or eliminate these compounds from the hit set.
The EG3 thiol mass also serves as a quality control tool to assess assay performance over time because it is present in every well. Tracking this mass over time can show trends both intra- and interplate to indicate changes in the detection sensitivity for the duration of a screen. This set of data can also serve as an early indicator to initiate MS source cleaning to ensure we maintain the highest data quality throughout the screening campaign. If used in parallel with control compounds, the normalized data can also inform potential complications with the automation process during sample preparation such as incorrect transfer of analytes from the assay plate to the SAMDI plate.
Overall, uniformity and reproducibility performed well for the SAMDI small-molecule binding format. Data normalization to EG3 thiol helped minimize signal variation and improved the %CV. Normalizing data and comparing the signal difference in the samples with and without protein helps eliminate false-positives and identify true hits for this proposed HTS format.
Proof-of-Concept Application of SAMDI in Pooled Compound Binding Screen
For this proof-of-concept experiment, we pooled three compounds with varying inhibitory potencies against BACE together at a fixed concentration, incubated with biotinylated BACE, captured by SAMDI, and then analyzed in the same solution-phase capture process described above. We rank ordered the AUC for each compound showing good correlation with the HTRF activity data ( Fig. 3A ). Compounds with more potent biochemical HTRF IC50 values correlated with a larger AUC compared with the weaker binding compounds. The format detected potent inhibitors in the hundreds of nM range with 25-fold signal over the no-protein control samples.
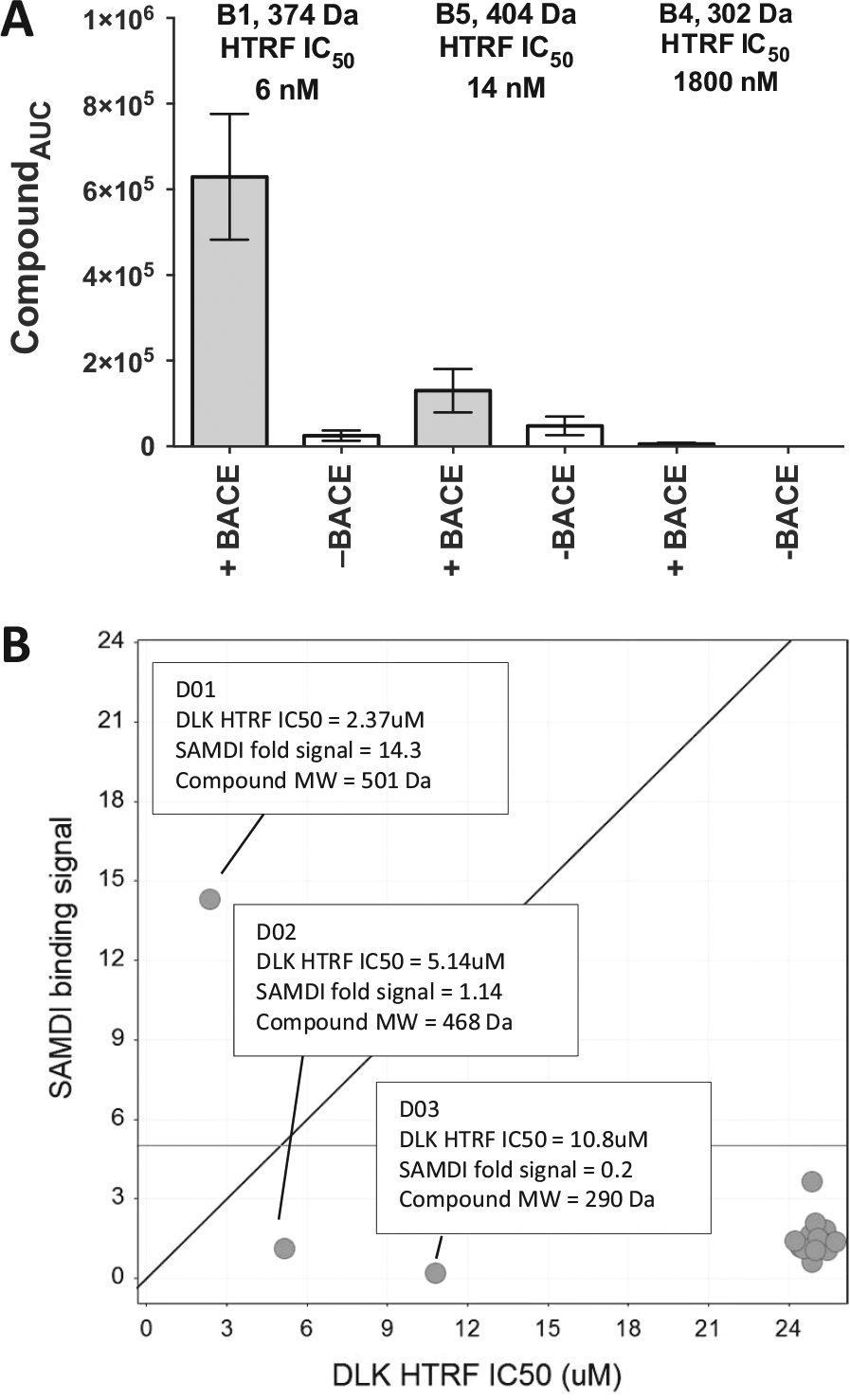
Self-Assembled Monolayers and matrix-assisted laser Desorption Ionization (SAMDI) detection of compound binders from pooled compounds. (
Similar to other affinity capture methodologies, saturating the capture surface with target protein on SAMDI (typically in the range of 500 nM to single-digit micromolar) will maximize the detection sensitivity and uniformity. The need for high protein concentration is balanced by the small assay volume and even smaller volume for the actual detection on SAMDI (as low as 300 nL). To understand the sensitivity limit of the SAMDI small-molecule binding assay in detecting weak binders from a mixture, 650 nM biotinylated DLK and a chemically diverse set of compounds were used to mimic conditions of a screen against a randomized and unbiased library. 23 We pooled three ATP-competitive DLK inhibitors, D01, D02, and D03, that ionize efficiently using straight MALDI and had weak biochemical IC50 values of 2 µM, 5 µM, and 10 µM, respectively, in a total of 20 inactive compounds with IC50 values all >25 µM. The concentration of each compound in the pool was 5 µM. Compound D01 (DLK-HTRF IC50 = 2 µM) revealed a 15-fold signal increase in samples with protein compared with samples without protein and was easily identified as a positive hit from the pool of 20 compounds ( Fig. 3B ). However, the two weaker DLK compounds with HTRF IC50 = 5 µM and 10 µM did not show significant signal in the samples with DLK compared with samples without DLK. This result was consistent with the hypothesis that the DLK protein was fully occupied by the most potent inhibitor in the pool, given that the weaker compounds present were known to bind to the same site of the protein and were not detected as binders. Although these data demonstrated that hits with single-digit micromolar binding potency were readily identified in this binding assay format from a compound pool, thoughtful planning of the assay conditions that consider the expected hit rate and anticipated potencies of hits would minimize false-negatives in a screen due to competitive binding of multiple compounds in a pool.
Compound binders with single-digit micromolar affinity are common starting points for new chemical matter identified from HTS. Experimental parameters to test prior to a screen that may maximize detection of weaker binding compounds, especially in a compound mixture, include (1) incubating the plates at 4 °C for a longer time period rather than at room temperature to preserve weak complexes, (2) increasing the screening compound concentration if the anticipated hit rate and potency are low, and (3) decreasing the compound pool size to reduce the competition for available binding sites for low-affinity molecules to the protein targets.
In summary, affinity capture of small-molecule binders using SAMDI potentially applies to a wide variety of protein target classes, especially those with limited reagent options or if the screen aims to capture hits with diverse binding epitopes on the target protein. In this study, we demonstrated proof of concept that the SAMDI platform can identify binders with affinities in the micromolar range and below, the output was robust and reproducible, and data correlated with biochemical activity for two different protein targets. Given the ability for MS to accurately identify individual hits from a mixture based on the mass identity, screening compound libraries in a pooled mixture using SAMDI can further increase the throughput. Because not all compounds ionize well and can lead to false-negatives, this format would not preclude data generated in other screening formats but rather offers an alternative for targets that are difficult to procure reagents for functional screens. The described proof of concept supports future work that will involve further customization of automating compound tracking (i.e., mass IDs), optimization of compound binding and wash procedures, and use of standard automation platforms. The combination of using a pooled compound library, sensitive MALDI instrumentation, and automation will enable screening of >100,000 compounds/day.
Footnotes
Acknowledgements
The authors would like to thank and acknowledge Amy Gustafson for developing the DLK HTRF functional assay and providing IC50 data for this study.
Supplementary material is available online with this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
