Abstract
Monoacylglycerol acyltransferase enzymes (MGAT1, MGAT2, and MGAT3) convert monoacylglycerol to diacylglycerol (DAG). MGAT1 and MGAT2 are both implicated in obesity-related metabolic diseases. Conventional MGAT enzyme assays use radioactive substrates, wherein the product of the MGAT-catalyzed reaction is usually resolved by time-consuming thin layer chromatography (TLC) analysis. Furthermore, microsomal membrane preparations typically contain endogenous diacylglycerol acyltransferase (DGAT) from the host cells, and these DGAT activities can further acylate DAG to form triglyceride (TG). Our mass spectrometry (liquid chromatography–tandem mass spectrometry, or LC/MS/MS) MGAT2 assay measures human recombinant MGAT2-catalyzed formation of didecanoyl-glycerol from 1-decanoyl-rac-glycerol and decanoyl-CoA, to produce predominantly 1,3-didecanoyl-glycerol. Unlike 1,2-DAG, 1,3-didecanoyl-glycerol is proved to be not susceptible to further acylation to TG. 1,3-Didecanoyl-glycerol product can be readily solubilized and directly subjected to high-throughput mass spectrometry (HTMS) without further extraction in a 384-well format. We also have established the LC/MS/MS MGAT activity assay in the intestinal microsomes from various species. Our assay is proved to be highly sensitive, and thus it allows measurement of endogenous MGAT activity in cell lysates and tissue preparations. The implementation of the HTMS MGAT activity assay has facilitated the robust screening and evaluation of MGAT inhibitors for the treatment of metabolic diseases.
Introduction
The monoacylglycerol acyltransferase enzyme MGAT2 is highly expressed in the small intestine, and it catalyzes the synthesis of diacylglycerol (DAG) from free fatty acids (FFA) and sn-monoacylglycerol (MG), the two major hydrolysis products of dietary fat. 1 MGAT2-deficient mice have delayed absorption of FFA and MG from the proximal to distal intestine, which in turn leads to enhanced incretin release and improvement in glucose metabolism. 2 The loss of MGAT2 reduces the absorption rate of dietary fat, leading to its partitioning toward energy expenditure rather than storage in white adipose tissue. As a consequence, MGAT2-deficient mice show resistance to obesity, glucose intolerance, hypercholesterolemia, and hepatic steatosis induced by a high-fat diet. 2 MGAT2-deficient mice demonstrate increased energy expenditure as compared to wild-type mice, and the differences are most pronounced during the feeding period, suggesting that MGAT2 modulates diet-induced thermogenesis.2–3 Elevation of glucagon-like peptide-1 (GLP1) is observed in MGAT2-deficient mice. 2 The mouse genetic data therefore support the concept of pharmaceutical inhibition of MGAT2 for the treatment of obesity and its related metabolic diseases. Several pharmaceutical companies have initiated campaigns and reported the identification of MGAT2 inhibitors.4–6 The pharmacological profile of MGAT2 inhibitor JTP-103237 recapitulates the results observed in the MGAT2-deficient mice. 6
In addition, the expression of MOGAT1, which encodes MGAT1, and MGAT activity are significantly increased in diet-induced obese (DIO) and ob/ob mice.7–8 The silencing of hepatic MGAT1 resulted in a dramatic improvement in blood glucose levels and hepatic steatosis, suggesting that the increase of MGAT1 expression in liver is responsible for the early onset of type 2 diabetes associated with hepatic steatosis and obesity.7–8 Therefore, targeting hepatic MGAT activity by pharmacologic means may be a viable therapeutic approach for the treatment of diabetes-related metabolic diseases.
The use of unique or stable isotope-labeled substrates and the analysis of its metabolism products by mass spectrometry (MS) play important roles in assay development, target engagement, and translational medicine biomarker discovery. Conventional MGAT enzyme assays use radioactive substrates. The product of the MGAT-catalyzed reaction is usually resolved by TLC analysis, following a cumbersome organic solvent extraction procedure.1–8 Thus, there is a need to develop a nonradioactive, high-throughput, liquid chromatography–tandem mass spectrometry (LC/MS/MS) enzymatic assay. Furthermore, MGATs also possess DGAT activity, 1 and the microsomal membrane preparations usually contain endogenous DGAT from the host cells. These DGAT activities can further acylate the DAG to form TG; thus, quantification of both radiolabeled DAG and TG products in the TLC plate is required to calculate the MGAT enzymatic reaction product formation. 1 We explored decanoyl-CoA and 1-decanoyl-rac-glycerol as alternate substrates in our MGAT reaction. We observed that the majority of the reaction product was 1.3-didecanoyl-glycerol, which was not susceptible to acylation to TG containing three short fatty acyl chains. Therefore, such reaction eliminated the need to quantify the TG product. In addition, didecanoyl-glycerol was fully soluble and forms one phase after addition of three volumes of acetonitrile to stop the MGAT reaction. Hence, the product could be subjected directly to LC/MS/MS in a 384-well format. Our MGAT LC/MS/MS assay was robust, allowing us to determine total MGAT activity in native tissue preparations.
To adapt the LC/MS/MS assay for MGAT2 HTS, a high-throughput mass spectrometry (HTMS) assay using the Agilent RapidFire 300 system (part no. G9203-80107; Agilent Technologies, Santa Clara, CA) was developed in a large-scale screening campaign for the human MGAT2 inhibitors. Our screening results showed that both the assay quality and hit-finding capability were sufficiently high. Several MGAT2 inhibitors of different chemotypes were identified in our HTS campaign.
Materials and Methods
Materials and Reagents
1-Decanoyl-rac-glycerol, decanoyl coenzyme A, and fatty acid–free bovine serum albumin (FAF-BSA) were purchased from Sigma-Aldrich (St. Louis, MO). Verapamil was purchased from Enzo Life Sciences (Farmingdale, NY). 1,2-Didecanoyl-glycerol and 1,3-didecanoyl-glycerol were purchased from Nu-Chek Prep (Waterville, MN). Pooled CD-1 mouse, Sprague Dawley rat, beagle dog, cynomolgus monkey, and human intestinal microsomes were obtained from XenoTech (Kansas City, KS).
Enzyme and Substrate Mixture Preparation
Recombinant human MGAT2 was produced in the baculovirus expression system. Sf9 insect cells were infected for 72 h. Cell pellets were resuspended in homogenization buffer (0.1 M sucrose, 50 mM KCl, 40 mM KH2PO4, 30 mM EDTA, pH 7.2). After centrifugation at 2500×g for 15 min at 4 °C, the pellet was resuspended in 500 ml lysis buffer, and total cell membranes were collected by ultracentrifugation at 100,000×g for 60 min at 4 °C. The collected membranes were resuspended in homogenization buffer and stored in aliquots at −80 °C. 1-Decanoyl-rac-glycerol was dissolved in 100% acetone at a stock concentration of 20 mM and stored in aliquots at −20 °C. Decanoyl-coenzyme A was dissolved in 0.1 M sodium acetate, pH 4.0, at 10 mM and stored in aliquots at −20 °C.
Recombinant MGAT2 Assay
In this assay, the MGAT2 enzyme catalyzed the production of didecanoyl-glycerol starting from 1-decanoyl-rac-glycerol and decanoyl-CoA. Didecanoyl-glycerol was detected by LC/MS/MS in a 384-well format. Briefly, 100 nl of compounds was dispensed to a 384-well polypropylene plate by the
Intestinal Microsome MGAT LC/MS/MS Assay
MGAT activity in intestinal microsomes of mice, rats, dogs, monkeys, and humans was assayed in 50 mM KH2PO4 (pH 7.4), 0.02% BSA, 1 mM DTT, indicated concentrations of human MGAT (hMGAT) or intestinal microsomes, 50 µM decanoyl-CoA, 70 µM 1-decanoyl-rac-glycerol, and 0.5% acetone, in a total reaction volume of 20 µl. The reaction was stopped by adding 60 µl acetonitrile containing 0.8 nM verapamil as an internal standard. After a brief mixing, the plate was centrifuged at 800×g for 5 min. The reaction product 1,3-didecanoyl-glycerol was quantified by LC/MS/MS as described below.
Product Detection by LC/MS/MS
The reaction product 1,3-didecanoyl-glycerol is quantified by LC/MS/MS using an Agilent 1100 HPLC system interfaced with a Waters Micromass Quattro Ultima triple quadrupole mass spectrometer (Waters, Milford, MA) equipped with an electrospray ionization source. The experimental conditions included the following parameters. A Zorbax SB-C8 column (2.1 × 50 mm and 3.5 µm particle size) was used. The column temperature was 60 °C. Buffer A consisted of 4 mM ammonium formate and 0.1% formic acid in water. Buffer B consisted of acetonitrile–water (90:10) containing 4 mM ammonium formate and 0.1% formic acid. The flow rate was 0.6 ml/min. A gradient elution had an initial ratio of 60:40 (A:B), then ramped to 100% B in 0.5 min, held for 1.5 min, and returned to initial conditions for a total run time of 3 min. A positive-ion mode was used for this multiple reaction monitoring (MRM). The mass analyzer was set for a selected reaction monitoring (SRM) transition for analyte of m/z 383 to 155 with a dwell time of 0.1 sec per transition. The internal standard used was verapamil (0.8 nM), with an SRM transition of m/z 455 to 165. The sample size used was 5 µL. The collision voltage was +25 V, and the capillary voltage was +3.1 kV. AUC (area under the curve) for the product was reported using MassLynx 4.0 software. Calibration curves were generated using synthetic 1,3-didecanoyl-glycerol or 1,2-didecanoyl-glycerol as standards (range: 0.004 µM to 48 µM).
For separation of the 1,2-didecanoyl glycerol from 1,3-didecanoyl glycerol ( Figure 1B ), the following LC conditions were used. Buffer solutions A and B, column temperature, flow rate, and mass spectral parameters are as described above. The column used was a Zorbax 300SB-C8, 2.1 × 150 mm with 3.5 µm particle size. The gradient elution used had an initial ratio of 30% B, then ramped to 75% B in 27 min, then returned stepwise to 30% B for 30 min.
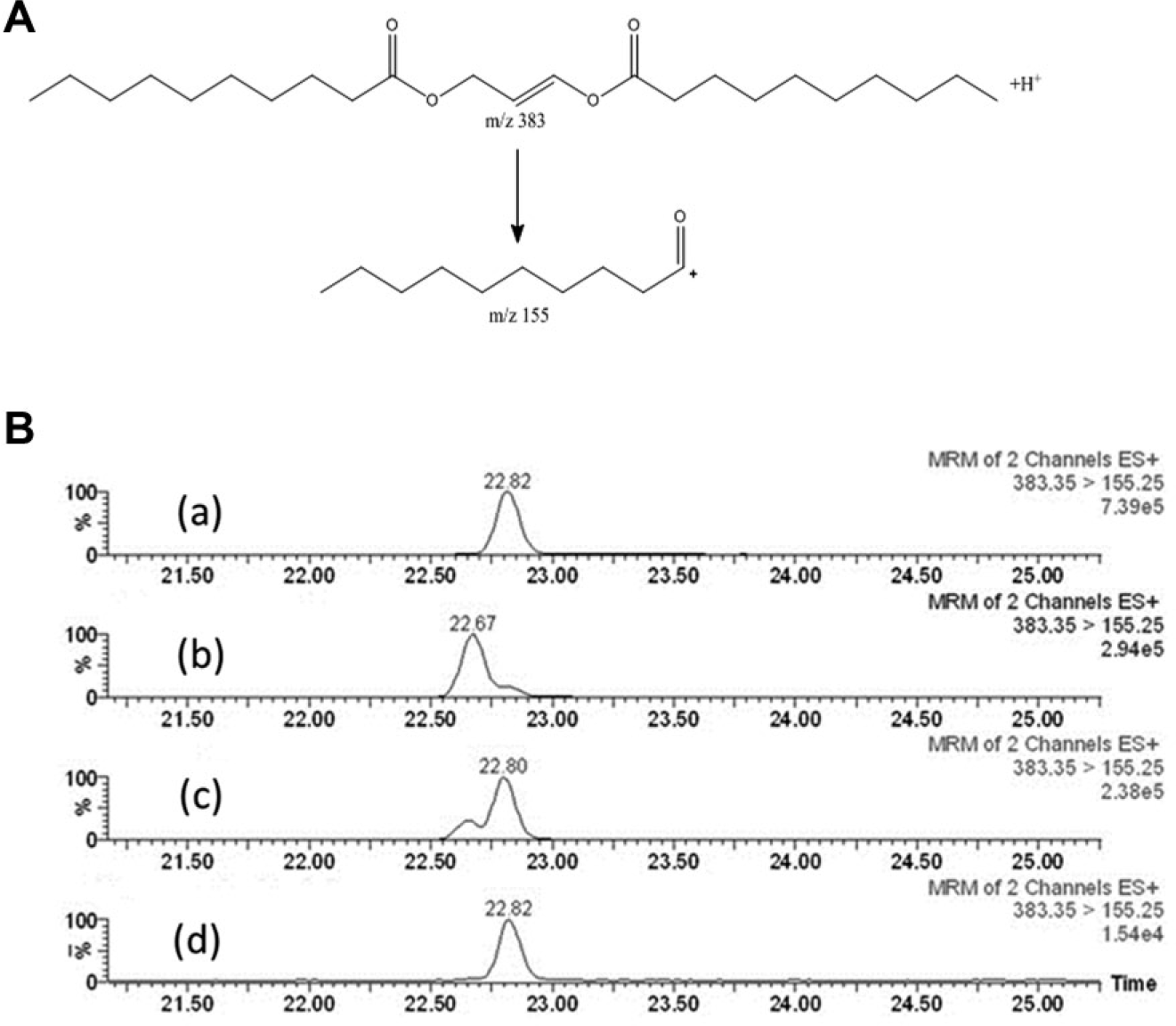
(
Product Detection by HTMS
The reaction product 1,3-didecanoyl-glycerol was quantified using a RapidFire 300 inlet system (Agilent) coupled to a Sciex 4000 triple quadrupole mass spectrometer (Sciex, Toronto, Canada). The mobile phase was 15 mM ammonium formate and 0.1% formic acid in acetonitrile–water (1:1). The wash solvent used was isopropanol–water (1:1). The wash between samples was 500 ms. The sample aspiration time (state 1) was 200 ms (600 ms with sip sensor), and the sample wash time (state 2) was 3000 ms. The sample elution time (state 3) was 3000 ms. The column equilibration time (state 4) was 500 ms. The flow rates used were pump 1 = 1.5 mL/min, and pump 2 and 3 = 1.25 mL/min. The Sciex 4000 mass spectrometer parameters included an SRM transition for didecanoyl glycerol at m/z 418.6 to 229 with 100 ms dwell time, declustering potential = 50 V, entrance potential = 5 V, collision energy = 20 V, and collision cell exit potential = 20 V; for the internal standard (verapamil), the SRM used was m/z 455 to 165 with a dwell time of 100 ms, a declustering potential of 100 V, entrance potential = 9 V, collision energy = 40 V, and collision cell exit potential = 15 V. Other parameters included: positive-ion mode; curtain gas = 20; collisionally activated dissociation gas = medium; ion spray voltage = 5500 V; source temperature = 550 °C; ion source gas 1 = 50; ion source gas 2 = 50; and resolution for Q1/Q3 = low/low. Note that the SRM transition m/z 418 to 229 was a result of neutral loss of decanoic acid from the ammonium adduct ion of 1,3-didecanoyl glycerol.
Results and Discussion
Development of LC/MS/MS-Based MGAT2 Assays
Zhang et al. recently reported a LC/MS/MS-based DGAT1 membrane activity assay by combining two DGAT1 substrates, di-oleoyl-glycerol and oleoyl-CoA. The product triolein was detected by multiparallel liquid chromatography coupled with LC/MS/MS in a 384-well format.
9
We have previous published DGAT1 and DGAT2 activity assays by measuring DGAT-catalyzed formation of TG using 1,2-dicapryl-sn-glycerol and oleoyl-CoA as substrates. The product carrying two capryl side chains and one oleoyl side chain, TG-(C10:0, C10:0, C18:1), was detected by HTMS in a 384-well format.
10
To date, no MGAT LC/MS/MS assay was reported in the literature. The MGAT activity has been determined routinely by quantifying radiolabeled DAG.1>–8 Microsomal membrane preparations typically contained endogenous DGAT from the host cells, and these DGAT activities could further acylate the radiolabeled DAG to form radiolabeled TG. A conventional MGAT2 assay using radiolabeled decanoyl-CoA with 2-oleoyl-glycerol or 1-decanoyl-glycerol is shown in
We also explored the combination of other short-chain acyl-CoA (e.g., octanoyl-CoA, decanoyl-CoA, and lauroyl-CoA) and short-chain monoacylglycerols (e.g., 1-decanoyl-rac-glycerol and 1-octanoyl-rac-glycerol) as MGAT2 substrates. MGAT2 enzyme products of 1-decanoyl-3-lauroyl glycerol (C10:0-C12:0), 1-decanoyl-3-octanoyl glycerol (C10:0-C8:0), or 1-octanoyl-3-lauroyl glycerol (C8:0-C12:0) were successfully detected by LC/MS/MS (
Development and Optimization of an MGAT2 HTMS Assay
Although our MGAT2 LC/MS/MS assay was in a 384-well format, it required 2 min run time for each well. In the HTMS assay, MGAT2-catalyzed formation of didecanoyl-glycerol was quantified by HTMS detection in positive mode using the SRM m/z 418 to 229. The use of HTMS shortened MS detection time to 10 sec for a well. Note that the SRM transition m/z 418–229 was a result of neutral loss of decanoic acid from the ammonium adduct ion of 1,3-didecanoyl glycerol. It was observed that the most abundant transition for 1,3-didecanolyl glycerol was different between the LC/MS/MS and HTMS assays depending on which spectrometer was used. This is likely due to a combination of differences in ion source and collision cell design and operating parameters between the Waters Quattro Ultima and Sciex 4000 mass spectrometers.
Figure 2A
shows the Km determination of decanoyl-CoA and 1-decanoyl-glycerol in the MGAT2-catalyzed enzymatic reaction. The substrate Km values for 1-decanoyl-rac-glycerol and decanoyl-CoA in MGAT2 HTMS were 40 µM and 18 µM, respectively. These Km values for 1-decanoyl-rac-glycerol and decanoyl-CoA are close to the observed Km values for 2-monooleoyl-glycerol and oleoyl-CoA in the radiolabeled MGAT2 TLC assay.
1
The reaction kinetic and enzyme titrations are shown in
Figure 2B
and
Figure 2C
. A linear time course was observed at least up to 40 min. From these experiments, the enzyme concentration and the reaction time were set at 40 ng/ml and 30 min, respectively.
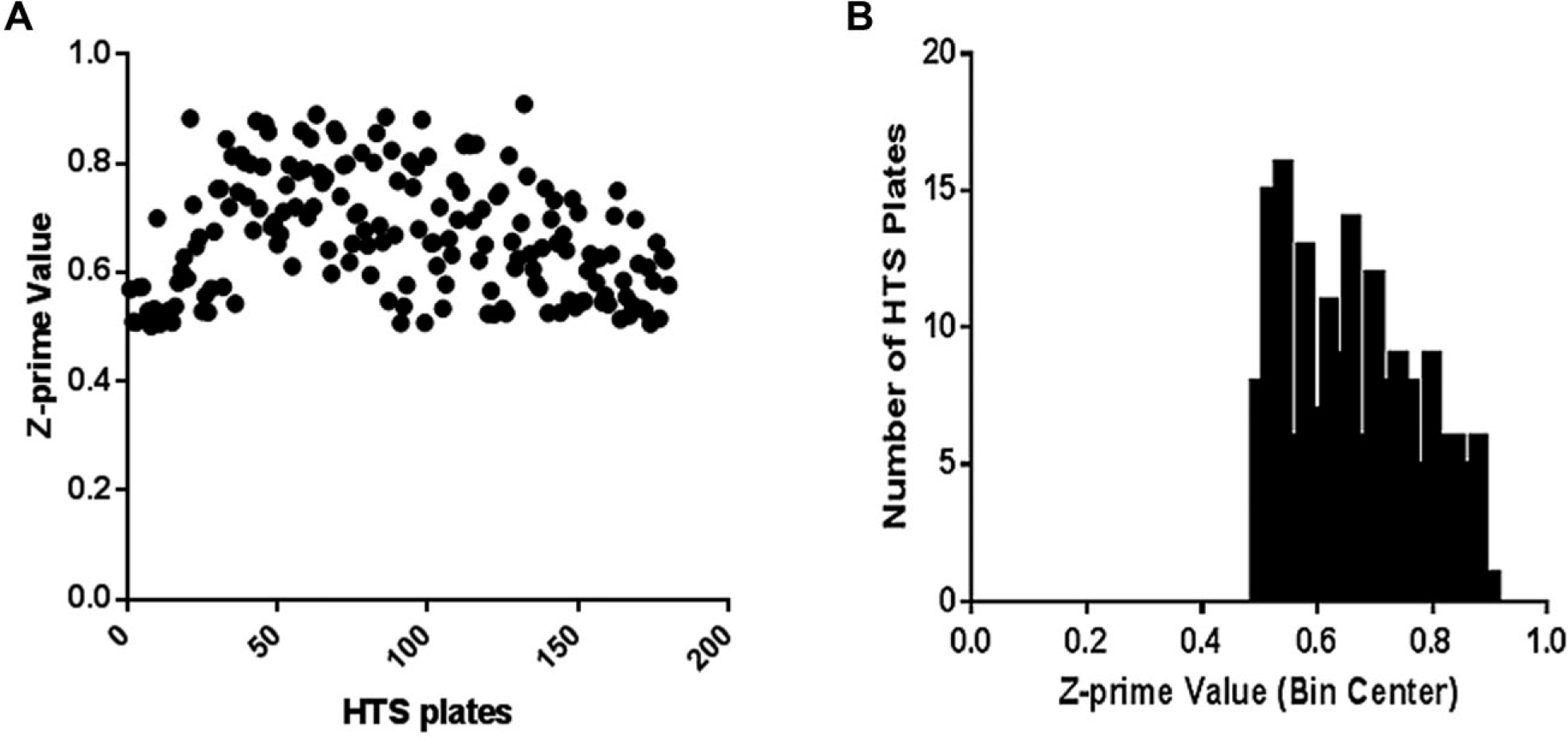
Figure 3
shows Z prime values and their distribution of the HTS plates. The signal-to-background ratio was 8. This MGAT2 HT/MS has been applied successfully in an MGAT2 HTS campaign with nearly 310,000 compounds with a hit rate of 0.47% at 7 µM screening concentration, and a confirmation rate was 49%. Several chemotypes of MGAT2 inhibitors were discovered. As an example, the inhibition of the enzyme product DAG (C10:0, C10:0) by two MGAT2 inhibitors was demonstrated (
Figure 3
). The IC50 values of JNJ-MGAT2-A and JNJ-MGAT2-B were determined as 40.7 ± 17.2 nM (
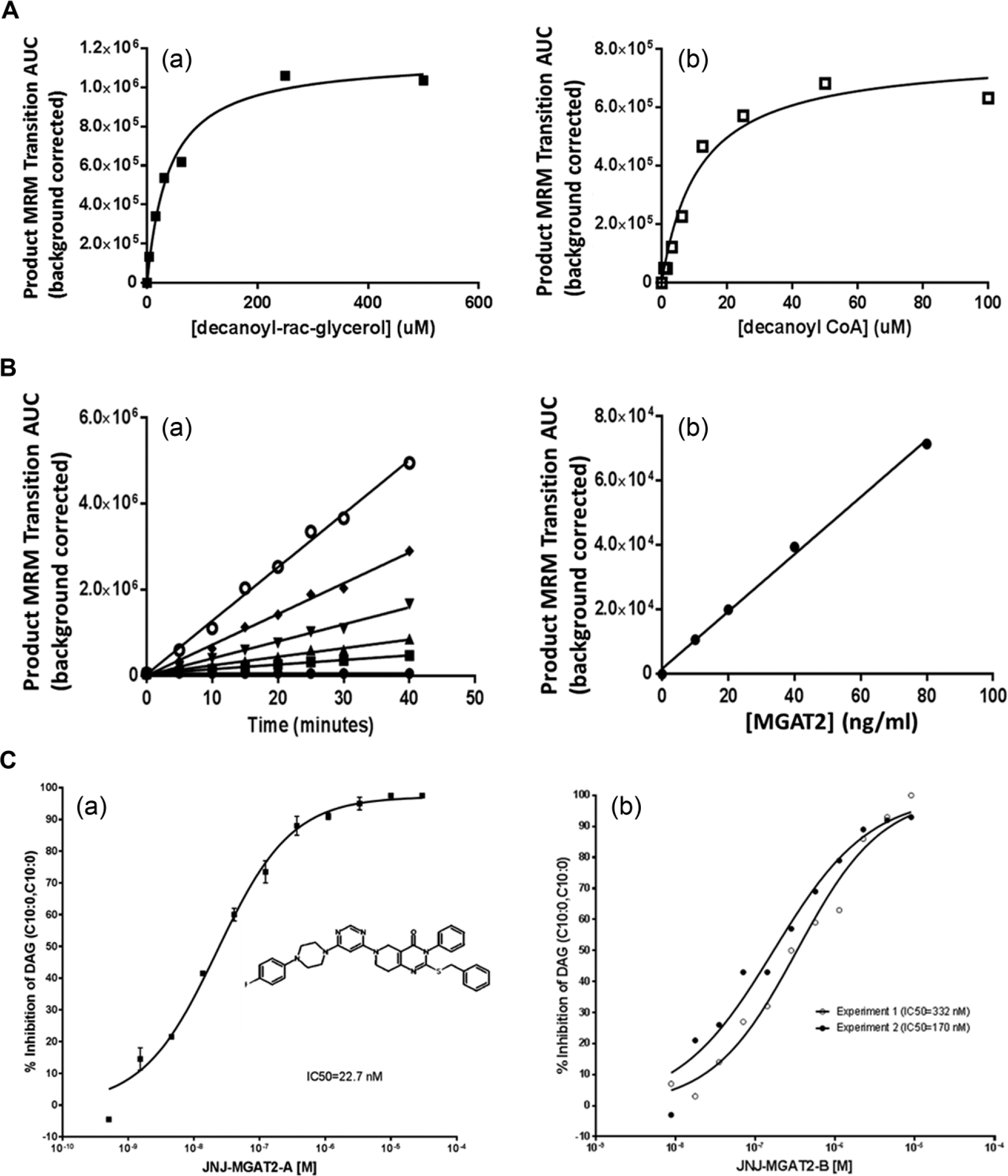
(
(
Intestinal Microsome MGAT LC/MS/MS Assay
Because MGAT activity is highest in the intestines, we evaluated MGAT activity in the intestinal microsomes from mice, rats, dogs, monkeys, and humans using decanoyl-CoA and 1-decanoyl-glycerol as substrates. The robust MGAT activity was found in the intestinal microsomes of all these species (
Figure 4
). As an example, the inhibition of DAG (C10:0, C10:0) in the MGAT assay with the mouse, rat, dog, and human intestinal microsomes was shown, and ~90% inhibition of total MGAT activity was achieved at 10 µM, the highest concentration tested (
Figure 4
). The calculated IC50 values of JNJ-MGAT2-B in the MGAT assay with mouse or rat intestinal microsomes were 109 ± 14 nM (
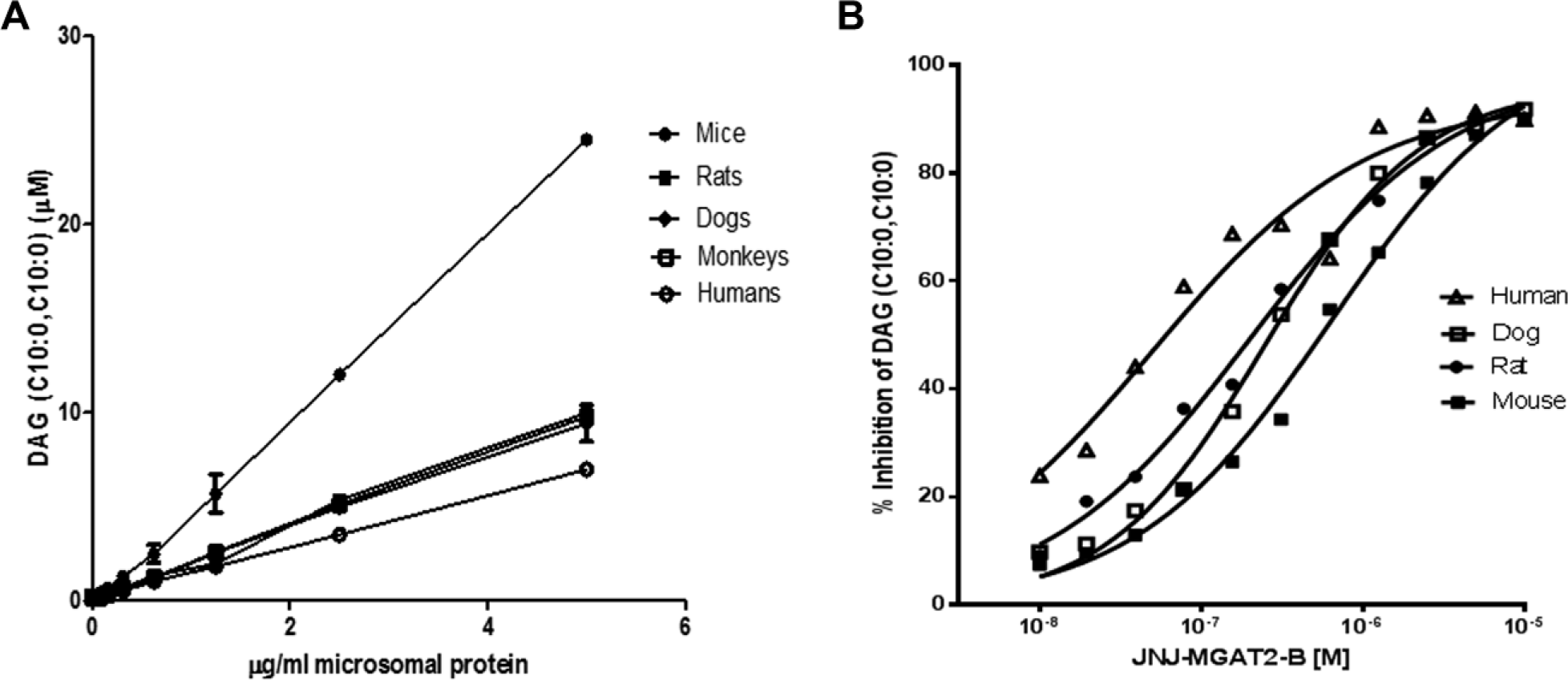
(
Recent studies suggest that the significant increase of MGAT1 (MOGAT1) expression in liver and hepatic MGAT activity was responsible for the early onset of type 2 diabetes associated with hepatic steatosis and obesity.7–8 The silencing of hepatic MGAT1 resulted in a dramatic improvement in blood glucose levels and hepatic steatosis.7–8 In addition, there was evidence suggesting that MGAT2 and MGAT3 were overexpressed in the livers of patients with nonalcoholic fatty liver disease (NAFLD). 11 JTP-103237, an MGAT2 inhibitor, was shown to prevent fatty liver and suppress both hepatic TG synthesis and de novo lipogenesis in a carbohydrate-induced mouse fatty liver model. 12 In that report, the inhibition of hepatic MGAT activity by JTP-103237 was assessed using 2-oleoyl and [1-14C]-oleoyl-CoA as substrates. The radioactivity of the MGAT product, [1-14C] diacylglycerol, was separated using TLC and analyzed as MGAT activity. Because the liver microsomal membrane preparations contain endogenous DGATs, the DGAT activities can further acylate the hepatic MGAT product, [1-14C] diacylglycerol, to form [1-14C]-TG. The use of decanoyl-CoA and 1-decanoyl-rac-glycerol in our MGAT LC/MS/MS assay will produce mostly 1,3-didecanoyl-glycerol as a final MGAT-mediated reaction product, which is not susceptible to acylation to TG. Our MGAT LC/MS/MS assay should aid in the evaluation of the regulation of native MGAT activity in these liver tissue preparations.
Footnotes
Acknowledgements
We thank Fany Bonilla and Wenyu Li for their support and critical review of the manuscript.
Supplementary material is available online with this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
