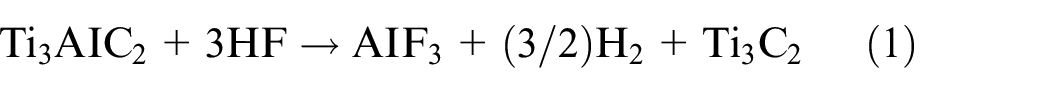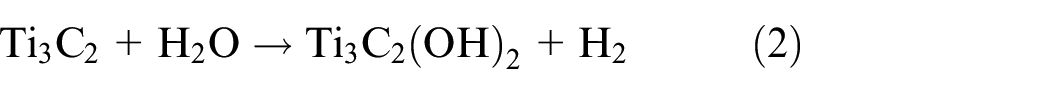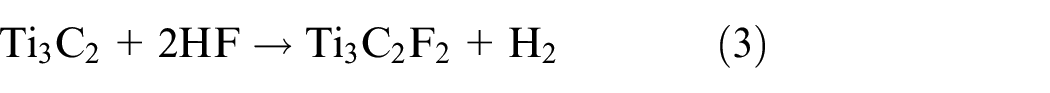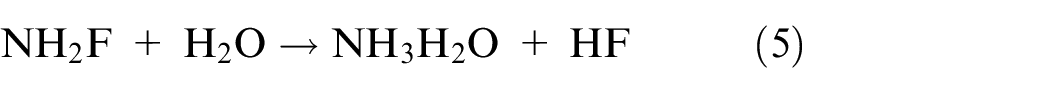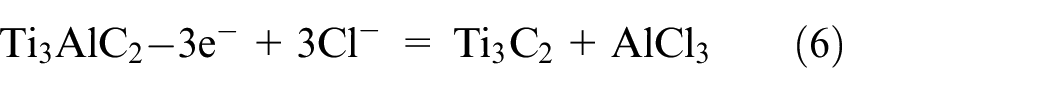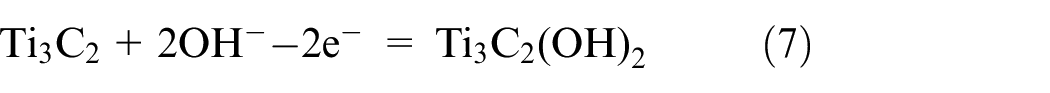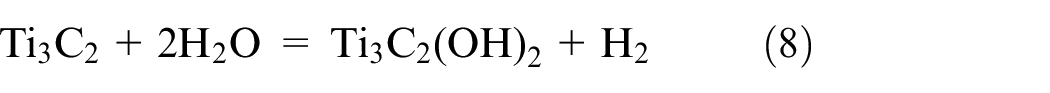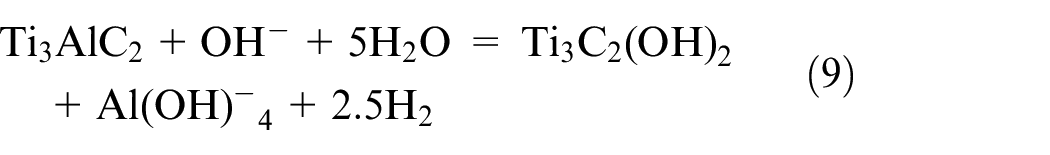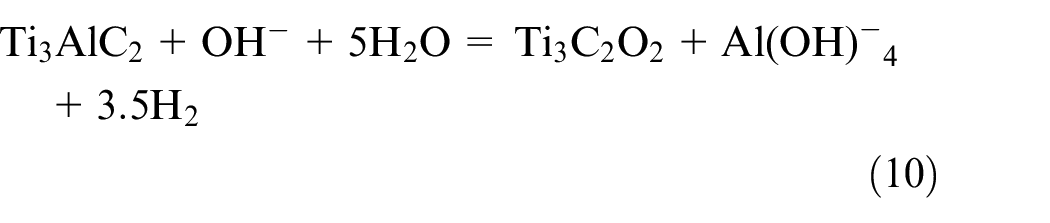Abstract
The revelation of MXenes as two-dimensional (2D) transition metal carbides and nitrides, in the midst of their exceptional features like rich surface chemistry, tunable terminations, metallic conductivity, and exceptional processability, has completely changed the realm of material chemistry research. The introduction of MXene has been contributive to the design and development of various textile e-devices. Among these, textile-based supercapacitors (TSCs), which encompass fiber, yarn, and fabric supercapacitors, are developed and have been manifested to be useful in powering wearable electronics. The present article summarizes historical developments of energy storage devices, types of super-capacitors like double-layered capacitors, hybrid capacitors, and pseudo-capacitors, their relative merits and demerits. Various top-down and bottom-up approaches of MXene synthesis processes are discussed with their scopes and limitations. Different process of coating/loading of MXene onto textile substrates viz. dip coating, electro-spinning and wet spinning are described and morphological cum surface characteristics of the developed coated textiles are highlighted. Mechanical and electrical properties of MXene based textiles have been summarized and compared with various other electro-conductive textiles prepared from conductive polymers, CNTs, and rGO. The performance of MXene-based textiles for energy storage applications is critically reviewed and summarized.
Introduction
Textiles are among the most widely used and necessities for day-to-day living. Their benefits of flexibility, porosity, durability, and softness make them suitable skeletal materials for the next generation wearable electronic gadgets other than clothing. 1 Traditional textiles have undergone a significant transformation as a result of the rapid advancement of wearable technologies by incorporating their inherent flexibility and unique capabilities. A variety of novel electro-conductive textile sensors have been developed, 2 energy storage devices, 3 electrodes, thermal regulators, microwave absorbers, electromagnetic interference (EMI) shielding materials, etc.4–7 Attainment of novel functionalities while maintaining basic properties of textiles, such as flexibility, breathability, ambient stability, and washability, is one of the primary challenges for the development of multifunctional textiles. 8 Owing to the fundamental prerequisite of conductivity, there are three main types of conductive materials that are extensively employed for the preparation of electro-conductive textiles, viz. conductive polymers (e.g. polyaniline, polythiophene, polypyrrole, poly (3,4-ethylenedioxythiophene), etc.); 9 carbon-based materials (graphene and carbon nanotubes); and metallic nanoparticles or wires (for example, Ag nanoparticles, Cu nanoparticles, Cu nanowires, etc.). 10 Unfortunately, the above-mentioned electronic textiles have limited practical applications since manufacturing of metalized textiles is expensive, conductive polymers have poor electrical conductivity, 11 and weak interfacial adhesion of carbon and CNTs toward textiles. 12
The rapidly expanding class of two-dimensional layered metal carbides or nitrides known as MXene. It was created by etching and exfoliating bulk precursors of the MAX phase (where M stands for an early transition metal, A for an IIIA/IVA element, and X for carbon and/or nitrogen). 13 MXene is excellent at storing energy. Its storage capacity may be increased by the incorporation of suitable components to create nanocomposites. Conductivity, specific capacitance, cyclability, capacitive retention, etc., all affect the storage capacity. It is a good antimicrobial and anticancer material. 14 It is a good contrast agent because of its high photothermal conversion and absorption capacities. It is very successful in the fabrication of nanocomposite sensors for biomedical applications like biosensors, physical sensors, etc. 15
History of Energy Storage Devices and Developments of Supercapacitors
Capacitors are one of the essential electrical circuit components and can be employed in a variety of ways for energy storage. 16 The proficiency and adequacy of the capacitors have moreover been upgraded, and capacitance has increased from milli-Farads (mF) to thousands of Farads (F), beginning with the foremost essential electrostatic capacitors and advancing to electrolytic capacitors, then supercapacitors. 17 Figure 1 illustrates a few important stages of historical development of capacitors starting from year 1746 to date. In 1746, a Leyden jar was invented as a real capacitor which could store electrical charge. The drawback of the Leyden jar was its larger size as well higher inductance and resistance. After that, in 1853 the first double-layer (DL) capacitors were developed with an important characteristic of the electrical double layer which appears at the interface between a solid electrode surface and a fluid (for example, between a conductive electrode and an adjacent liquid electrolyte). However, the DL capacitor had very low internal resistance that allowed extremely fast discharge. After that an electrolytic capacitor using aluminum electrode and a liquid electrolyte was developed. In this type of capacitor, an electrolyte-soaked paper was sandwiched between two metal foils that had been coated with a non conducting oxide layer. When compared to electrostatic capacitors, the thin oxide coatings on electrodes served as dielectric elements and provided a larger capacitance per unit volume. Electrolytic capacitors were commonly made of aluminum, tantalum, niobium, and niobium oxides, with the range of relative permittivity between 9.6 and 41 and capacitance varying from a few micro Farad to hundreds of mili Farad.16,18 However, the electrolytic capacitors suffer from high possibility of leakage currents, value tolerances, equivalent series resistance, short lifespan, etc. Afterwards, Bell laboratory had developed solid Tantalum capacitor that consisted of a pellet of porous tantalum metal as an anode, covered by an insulating oxide layer that formed the dielectric, surrounded by liquid or solid electrolyte as a cathode. The failure of tantalum capacitors may lead to fires, thermal runaway and smaller explosions which were the main drawbacks. At later stage in 1957 electrochemical DL capacitors (EDLC) were developed by the General Electric Company. For an EDLC the electrolyte is either an aqueous or non-aqueous liquid, or a solid material like a conducting polymer. In any case, the primary patent was not issued until 1957, the year when a carbon-based capacitor with a large surface area was demonstrated. 19 In 1971, a commercial disk shaped DL capacitor was conceptualized. The first supercapacitor was invented in 1982 with significantly lower internal resistance for the military applications. Supercapacitors provide tens to hundreds of times more specific energy than conventional basic electrostatic and electrolytic capacitors. Additionally, compared to many different types of batteries, supercapacitors can deliver better specific power, even though their specific energy is generally lower. 20
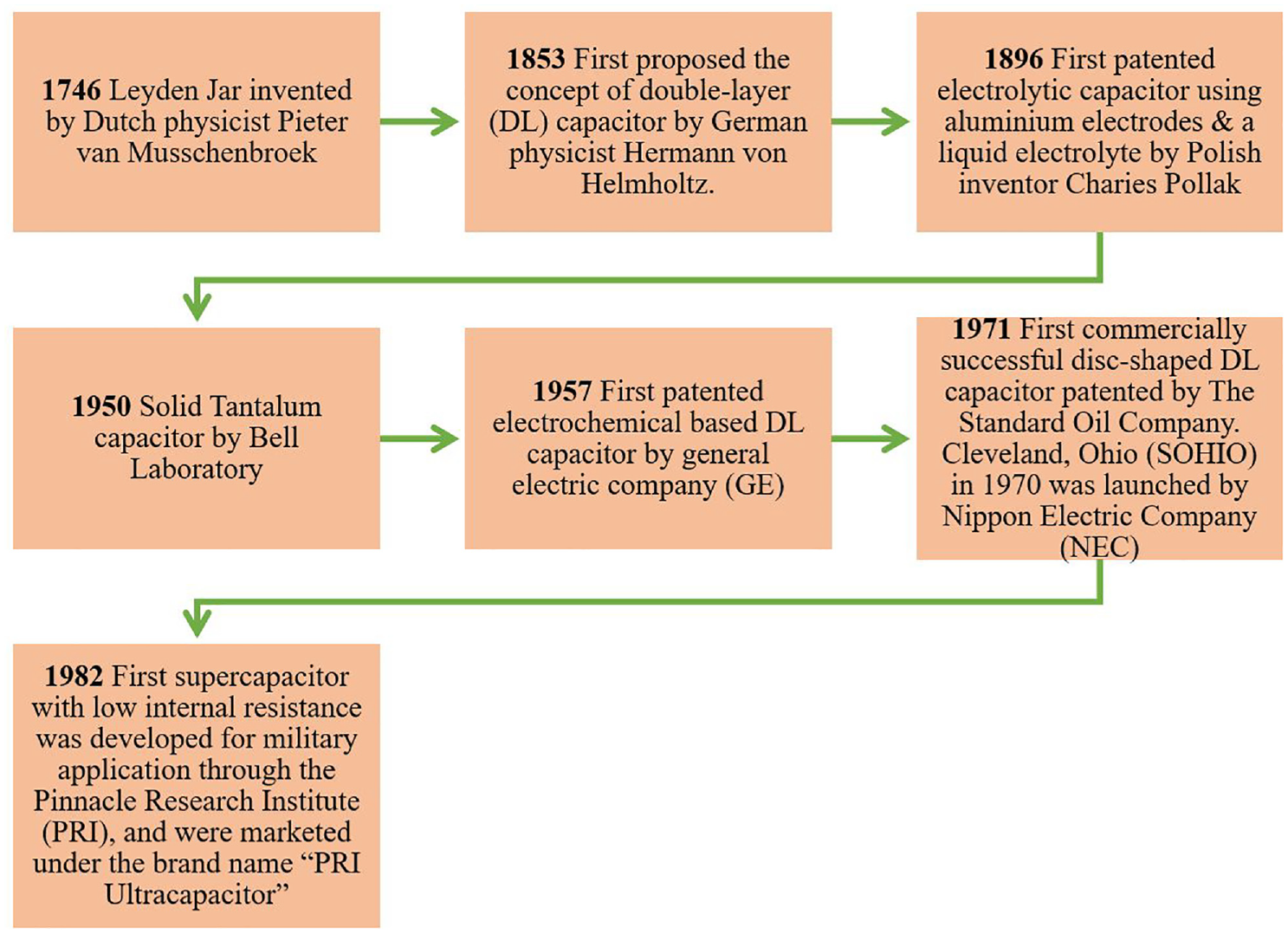
Various stages of development of capacitor.
Supercapacitors are credited with holding the key to bridging the energy storage gap between batteries and capacitors, establishing quick charging energy-storage systems, and supplying intermediate specific energy. Because of their highly reversible charge-storage process, supercapacitors have much longer life cycles and can respond quickly during both the charging and discharging processes. This generates a great deal of interest in its use in a variety of consumer devices, industrial power management systems, and hybrid cars that run on both electric and conventional fuels.17,20
Types of Supercapacitors
As per charging methods, the supercapacitors are classified into three types, as depicted in Figure 2: (i) double-layer capacitors, (ii) pseudo capacitors, and (iii) hybrid capacitors. It was reported that the two layers of polarizing electrodes in a double-layer capacitor are filled with electrolytes and encased in a separator, serving as an energy storage device. 16 In the context of redox reactions, energy is stored primarily through redox material in pseudo capacitors, where the rapid chemical reaction serves as capacitance. 17 The lithium-ion capacitor is a common example of a hybrid capacitor, which can exhibit both reversible Faradaic-type charge transfer and electrostatic reactions. The electrodes can be doped or supplemented with materials like transitional metal oxides, conducting polymers, or even active carbon.
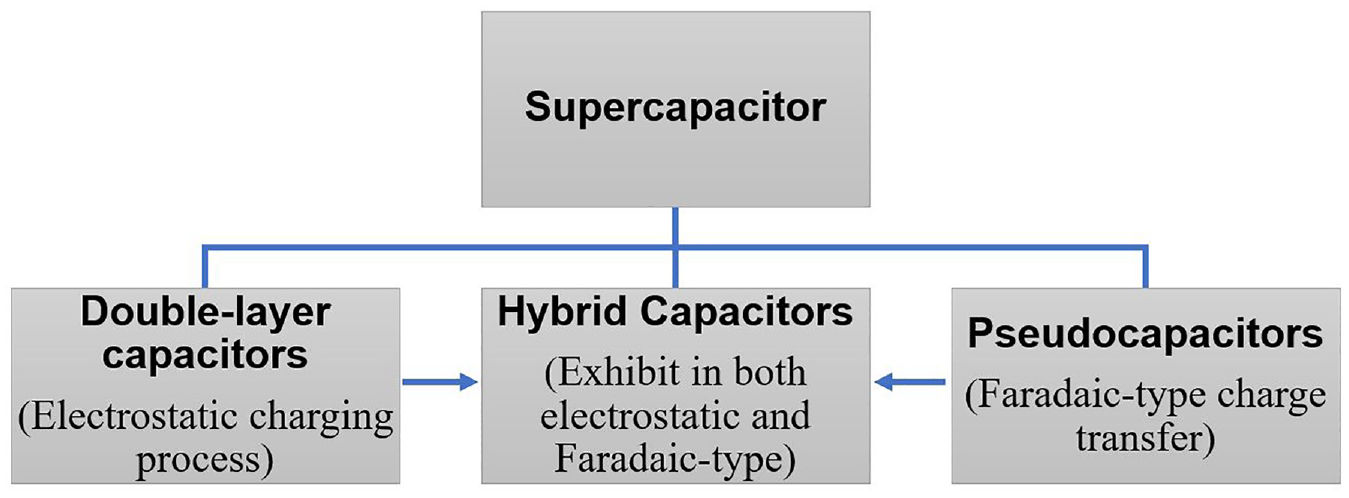
The classification of supercapacitors.
Mechanism of Energy Storage in Supercapacitors
Two well known energy storage devices such as supercapacitors and batteries have been extensively used in a variety of industries.21,22 Two electrodes, an electrolyte, and a separator are the basic building blocks of both batteries and supercapacitors. 23 The active materials used in electrodes can be free-standing or linked to current collectors, and their properties have a significant impact on how much energy they can store. Usually, solutions having the appropriate ionic conductivity are called electrolytes; however, in recent years, the majority of researchers have been concentrating on the development of solid-state electrolytes and hydrogel electrolytes (quasi-solid-state), with the provision of removal of liquid that can improve the safety concerns caused by flammable and toxic solutions, enhance cyclic durability and resistance to leakage of electrolytic solutions, and eliminate the need for separators. Separators are employed to keep two electrodes apart because if they touch each other, a short circuit will result. Despite having similar configurations, batteries and supercapacitors have different energy storage methods. Batteries rely on relatively fast faradaic reactions, whereas supercapacitors use electrical double-layer capacitance and/or pseudo-capacitance mechanisms.24,25 Supercapacitors are classified into electrical-double-layer capacitors (EDLCs) and pseudo capacitors based on how they function, as mentioned above. At the electrode/electrolyte interfaces, charges are stored in electrostatic double layer capacitors (EDLCs) without using a faradaic process. As a result, the large specific surface area of materials that are good for ion adsorption, like activated carbon (AC), graphene, and carbon nanotubes (CNTs), have been employed a lot in EDLCs.22,23 Pseudo capacitors, on the other hand, store charge through quick and reversible faradaic processes that take place at or near the surface area of electrodes. Pseudo capacitors have substantially larger capacitance than EDLCs because of their faradaic features. Still, the power density of the pseudo capacitors is relatively low due to an inherently slower faradaic process than a non-faradaic process. Various conducting polymers like polyaniline, polypyrrole and polythiophene, and various metal oxides, such as MnO2, Co3O4, RuO2, etc., are noteworthy examples of electrode materials for pseudo-capacitors.26,27 The working mechanisms of electrical-double-layer capacitors (EDLCs) and pseudo-capacitors are described in the following sections.
Electrical Double-Layer Capacitor (EDLC)
Similar to the conventional parallel capacitor, energy is stored in double-layer capacitor by the mechanism of charge separation. But compared to a conventional capacitor, it can store a lot more energy. An electrical double layer allows for the establishment of an interphase between a certain electrode and its neighboring electrolyte since the charge separation occurs over a comparatively short distance in this scenario. A larger amount of energy can also be retained on an electrode with a big surface area and plenty of pores. Due to the straightforward migration of ions to and from the electrode surface, as depicted in Figure 3, this energy storage mechanism may react quickly.16–19
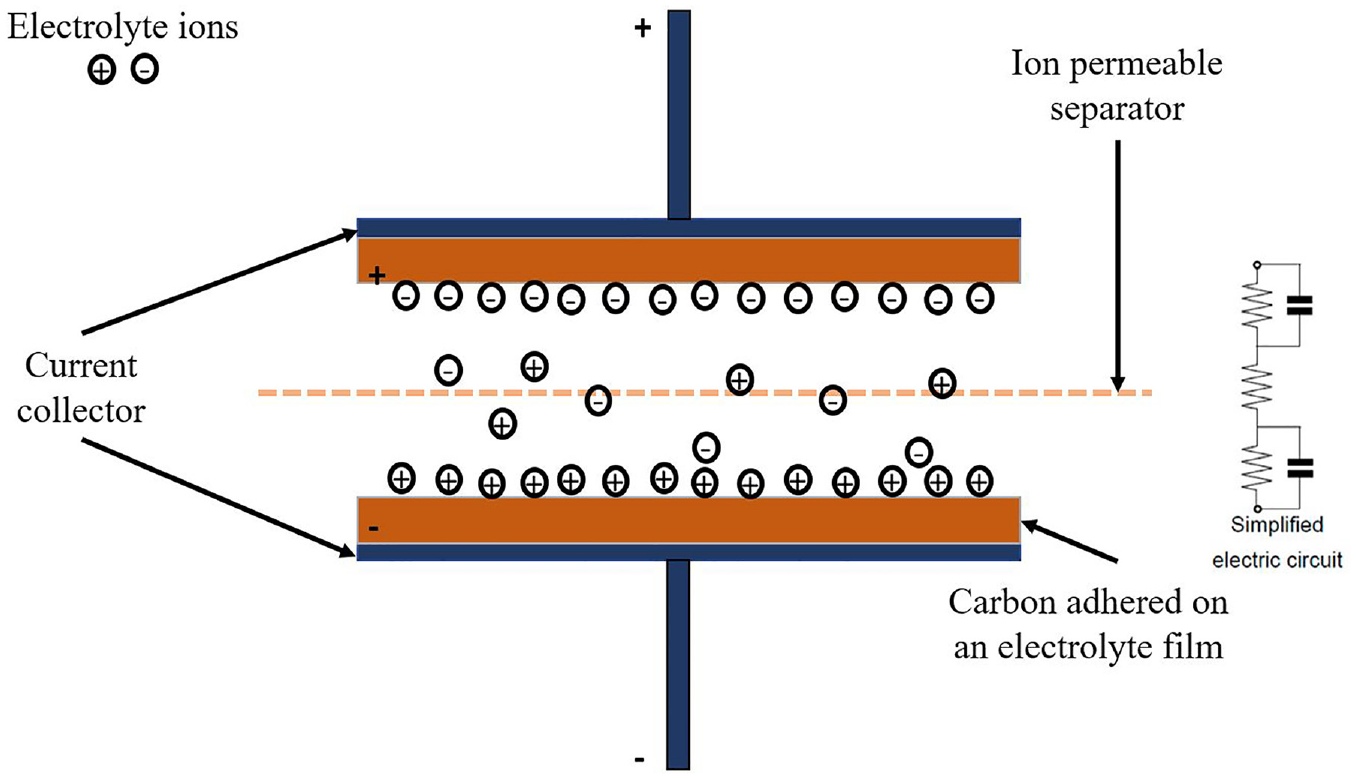
An electrochemical double layer capacitor in its charged condition.
Pseudo Capacitors
Pseudo capacitance is characterized by comparatively high capacitance under a non-electrostatic basis, which is caused by a reversible charge transfer of the Faradaic type. The production of capacitance associated with an electrochemical charge-transfer process is also possible with a finite number of active materials or effective surfaces. Recent research focuses on conductive polymers like polyaniline (PAni), polypyrrole (PPy), and derivatives of polythiophene (PTh), such as poly (3,4-ethylenedioxythiophene), or PEDOT, as well as transition metal oxides like manganese oxide (MnO2). This pseudo capacitor is also known as a redox supercapacitor because it performs a typical charge-transferring process of Cl− ions in conducting polymer matrix, as illustrated in Figure 4.16–19
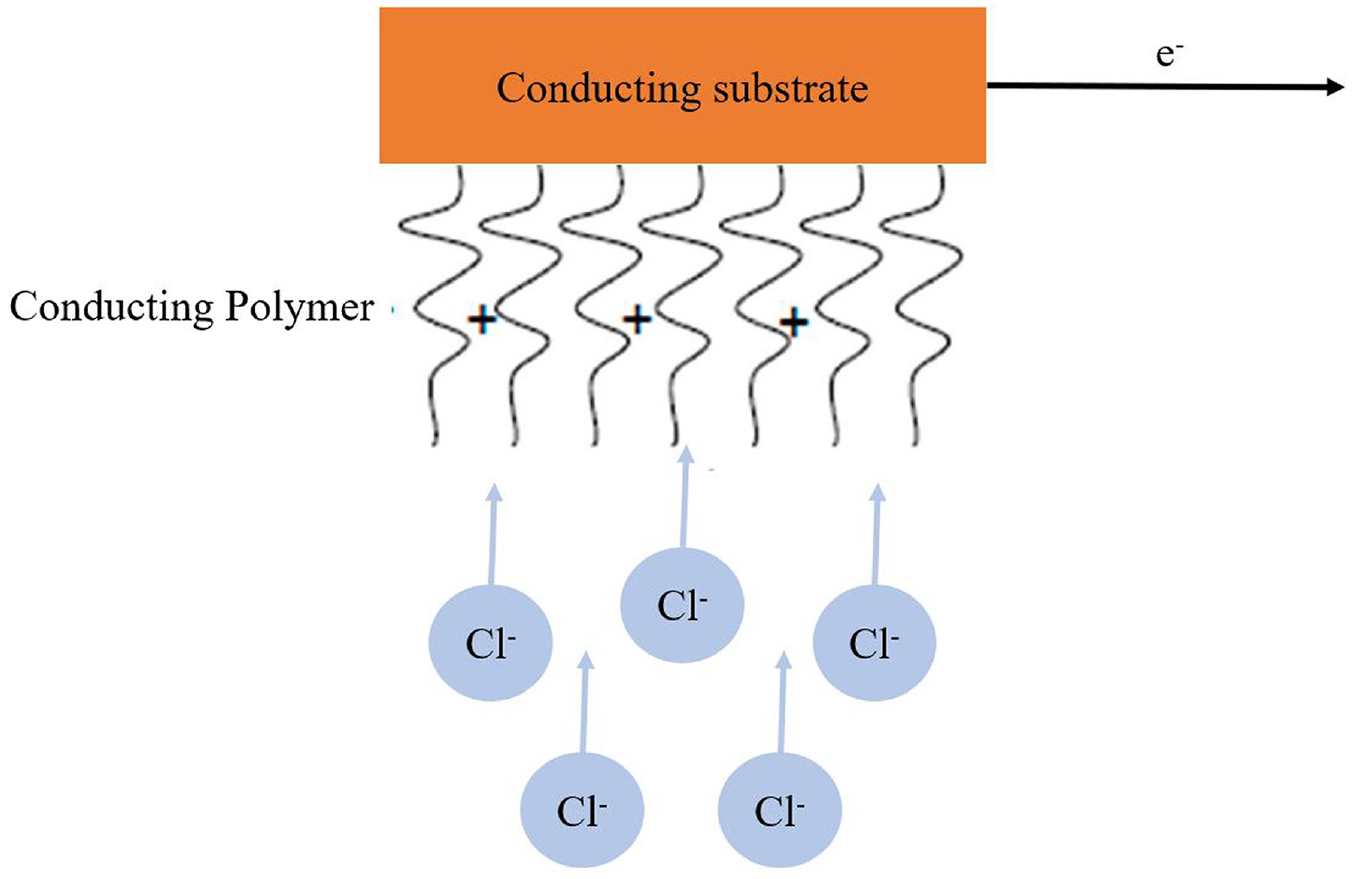
Mechanism of pseudo capacitance by a conducting polymer.
Configurations of Energy Storage Textiles
Energy storage application is defined by the process of gathering energy generated during a specific time and storing it for later use under a specific circumstances with the least amount of loss. Energy storage can take many forms, but the primary topics of discussion are mechanical, electrical, electrochemical, thermal, and chemical energy storage methods. 28 The textile energy storage devices seem to be classified in two ways, the thermal and electrical energy storage devices which are discussed in Figure 5.
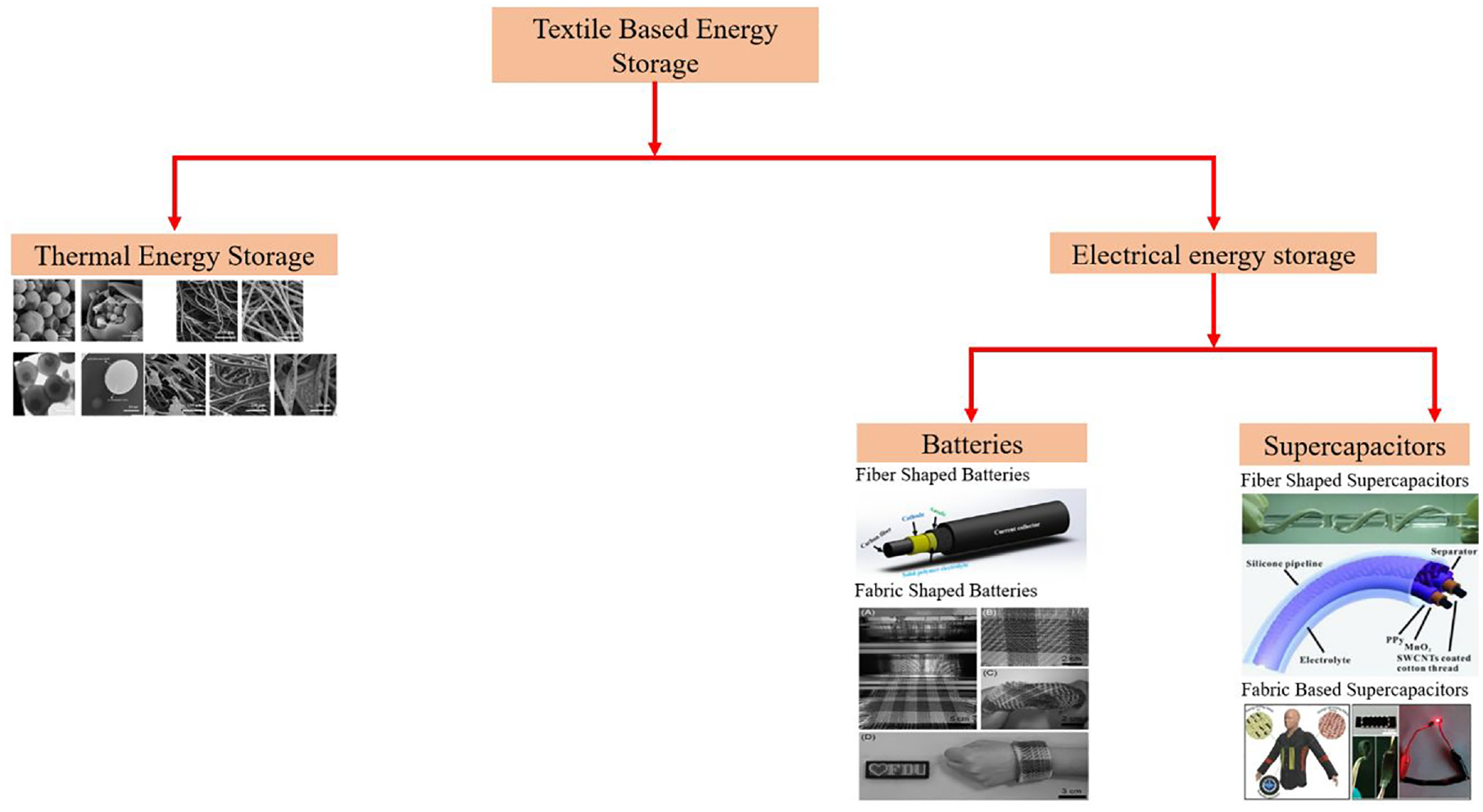
Summary of textile-based energy storage devices. 30
In essence, there are two classes of thermal energy storage mechanisms. “Sensible heat” is the first one. 28 This mechanism results in an increase in material temperature as energy is applied to the material. Different heat transfer mechanisms (such as radiation, convection, or conduction) move the heat stored in a material that is getting hotter to a cooler material or environment. The process by which substances absorb or release heat from their surroundings during phase transition is the basis of the second type of thermal storage mechanism, known as “latent heat.” Materials that undergo inorganic or organic phase changes are employed in this way.28,30
The electrical energy storage devices can store electric charge and disseminate during the requirements. Batteries and supercapacitors are the two classes of such electrical energy storage devices as shown in Figure 5. Supercapacitors and batteries are integrated into flexible textile substrates in the latest Energy Storage Textiles (EST) fabrication designs. The raw ingredients used to create all textile items are fibers. Bundles of filament or staple fibers are spun into long, continuous strands known as “yarns,” which can then be further knitted or woven into textile fabrics. Without weaving or knitting, fibers link together using chemical, mechanical, heat, or solvent treatment to create nonwoven fabrics. 31 Textiles offer the advantage of excellent tensile strength and flexibility that allows them to withstand cyclic deformations with quick recovery capabilities in comparison to papers, plastics, and metal sheets. 32 There are currently two main types of EST designs available, viz. top-down and bottom-up approaches. According to Huang et al. (2016) 33 , the bottom-up method involves converting fibers and yarns into conductive materials for manufacturing fiber and yarn electrodes, which are then arranged parallel or twisted form to produce entire supercapacitors or batteries. After that, these fiber/yarn supercapacitors or batteries might be further converted to ESTs by being woven or knitted, or even just stitched into current fabrics as a minor fraction of the overall textiles. This strategy is adaptable and can be used for a variety of interesting applications. The top-down approach, in contrast to the bottom-up approach, uses existing textiles to build electrodes, which are then sandwiched together with the electrolyte and separator (if required) to produce ESTs. This method has the advantage of the typical rough surfaces, holes, and gaps seen in textile products, which favors extensive mass loading of active components and a greater interface between the electrodes and electrolyte. 34 Prior to loading the active materials, it is necessary to convert the nonconductive character of ordinary fibers/yarn and textile substrates into electrically conductive materials. This can be done through carbonization or a variety of coating/depositing techniques. Classification of textile-based energy devices is shown in Figure 5.
Synthesis and Surface Chemistry of MXenes
MXene is a relatively young member of the 2D family of nanomaterials and has shown great promise for a variety of uses. Several members of the MXene family have been developed, and the idea of using these materials to create new products is gaining popularity. 35 Although, MXenes can be synthesized by either top-down (from the MAX phase) or bottom-up approaches (from M and C atoms), the latter is more common. The MAX phase, which is the starting material of the top down approach, is composed of stacked ternary carbides and nitrides which are denser than MXene.
Bottom-up Approach
The bottom-up approach frequently begins with small organic or inorganic molecules or atoms, and is followed by crystal growth that can be arranged to create 2D ordered layers. 36 Mostly, the chemical vapor deposition (CVD) technique is used for this approach. High-quality thin films can be produced on a variety of substrates by this method (Figure 6(a)). Multilayer (up to six layers) thin film can be successfully prepared by this CVD method. 37 As a first CVD approach, a high-quality ultrathin Mo2C (molybdenum carbide) film was produced on a Cu/Mo (copper/molybdenum) foil using methane gas (CH4) as the carbon source at a temperature above 1085°C (Figure 6(a)). A variety of films with lateral sizes ranging from 10 to 100 μm were produced through the optimization of temperature and time for growth. The absence of surface functional groups may be indicated by the defect-free and highly crystalline Mo2C films that were synthesized. 38 MXene synthesized using this method lacks a surface functional group, which makes it unsuitable for coating textile materials in order to create electro-conductive textiles. As a result, it is not substantive to textile substrates. Also, it has been reported that this approach is not ideal for biomedical applications because the size of MXene flakes (μm) is too large for cell penetration. The surface engineering of the MXene nanosheet coating over textile substrates should be well functionalized. The chemical vapor deposition-based bottom-up strategy showed promise for generating MXene with minimal defects in comparison to the top-down approach, but further research is necessary to fully grasp this method’s potential for producing very pure MXene. 39
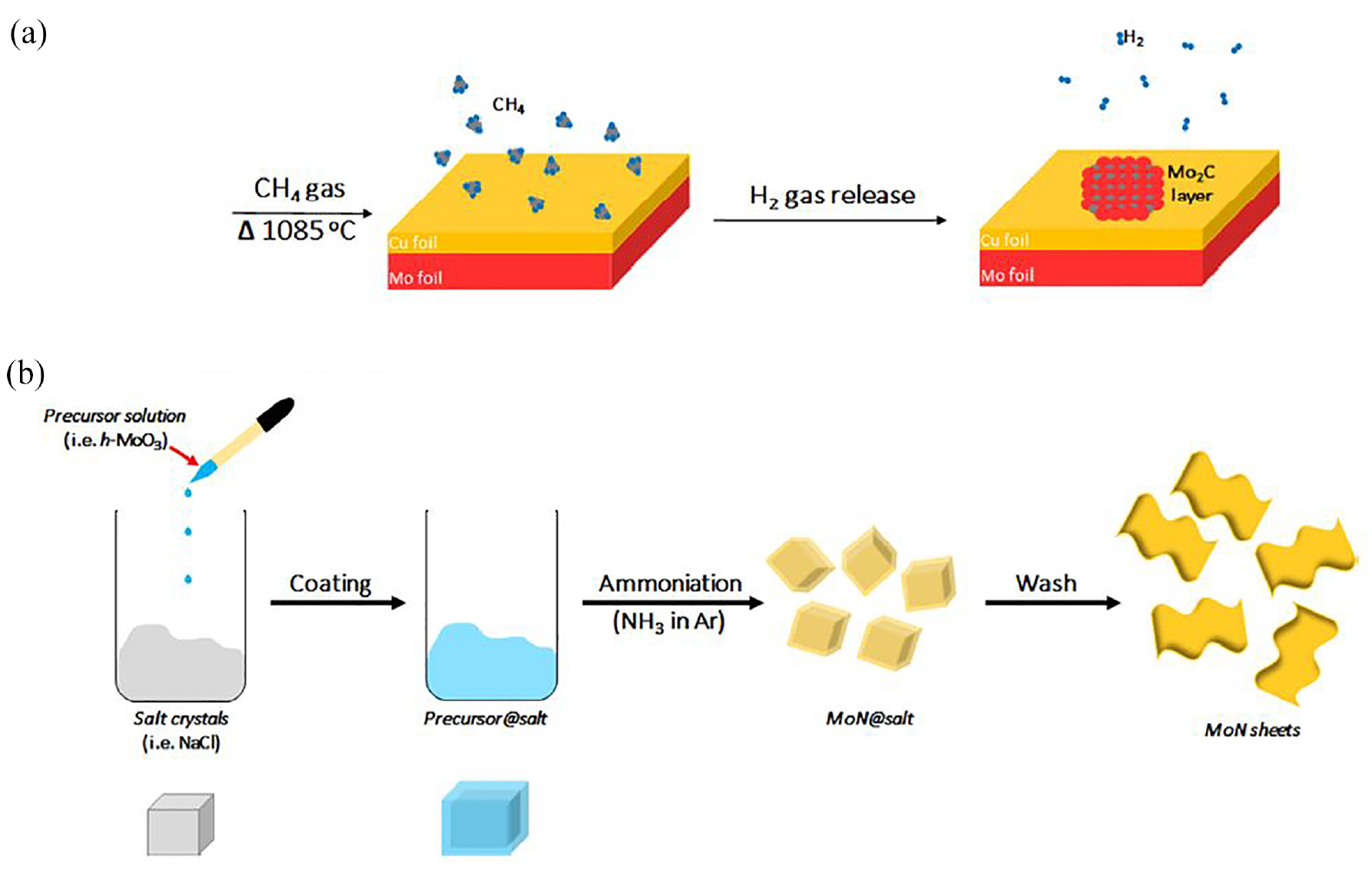
Diagram showing the bottom-up approach for MXene synthesis: (a) chemical vapor deposition of Mo and C to form Mo2C thin film; and (b) salt-template synthesis of MoN nanosheets. 40
The template synthesis method and plasma-enhanced pulsed laser deposition (PELPD) method of MXene synthesis are also explored.41,42 The first PELPD-synthesized ultrathin molybdenum carbide (Mo2C) films reacted with the Mo vapor produced by the pulsed laser using methane plasma as a carbon source. This reaction was conducted on a sapphire surface at 700°C to yield a high-quality Mo2C film with tailorable thickness by adjustment of applied laser pulse rate. Additional modifications were reported, 43 like the synthesis of 2D nitrides (MoN, V2N, and W2N) using a scalable salt template by ammonia reduction of their corresponding 2D hexagonal oxides. They discovered that 2D metal oxide precursors could be generated by salt-templating, and the resulting MoN nanosheets were hydrophilic and had a thickness of less than 1 nm when dissolved in water (Figure 6(b)). 41 It has been demonstrated that this technique works with other metal oxides, including W2N and V2N nanosheets. However, there is still space for improvement because there is currently little information available regarding the bottom-up synthesis of biocompatible MXene. 44 However, no research group has found evidence of the presence of surface functional groups, and there is a dearth of knowledge regarding the coating of textile substrates using this technique. 40
Top-down Approaches
Hydrofluoric Acid-Based Etching of MAX Phases By
The MAX phase precursors, of which there are currently more than 150 MAX available, are shown in Figure 7(a). The selective etching of A-element layers from the MAX phase is caused by the MX bond, which is less chemically reactive than the MA bond.39,45,46 A loosely layered, multilayer MXene is created when the A element is taken out of the MAX phase. Fewer than four stack MXene layers are known as a few-layered MXene, and it necessitates additional degradation. The production of the first MXene (Ti3C2T x ) was reported in 2011 through the selective etching of the aluminum layer from the titanium aluminum carbide MAX phase precursor with the assistance of hydrofluoric acid (HF). 47 Hydrofluoric acid is frequently used as etching agent for carbide-based MXenes, and the concentration and duration of treatment with HF solution influence the MXenes’ characteristics. Figure 7(b) depicts the exfoliation of MAX phase and formation of MXene, while equations (1)–(3) illustrate the etching reactions. 48
Al is removed during the first reaction step (reaction 1), exposing Ti atoms that are extremely reactive and forming Ti3C2. The aqueous HF solution and the Ti atoms easily combine to form hydroxyl (-OH), oxygen (=O), or fluorine (-F) functional groups on the surface of MXene (equations (2) and (3)). The surface functional groups can be changed to give exceptional features of MXenes, like tuned bandgaps, increased electron movement at ambient temperature, half metallicity, two-dimensional ferromagnetism, etc. 49
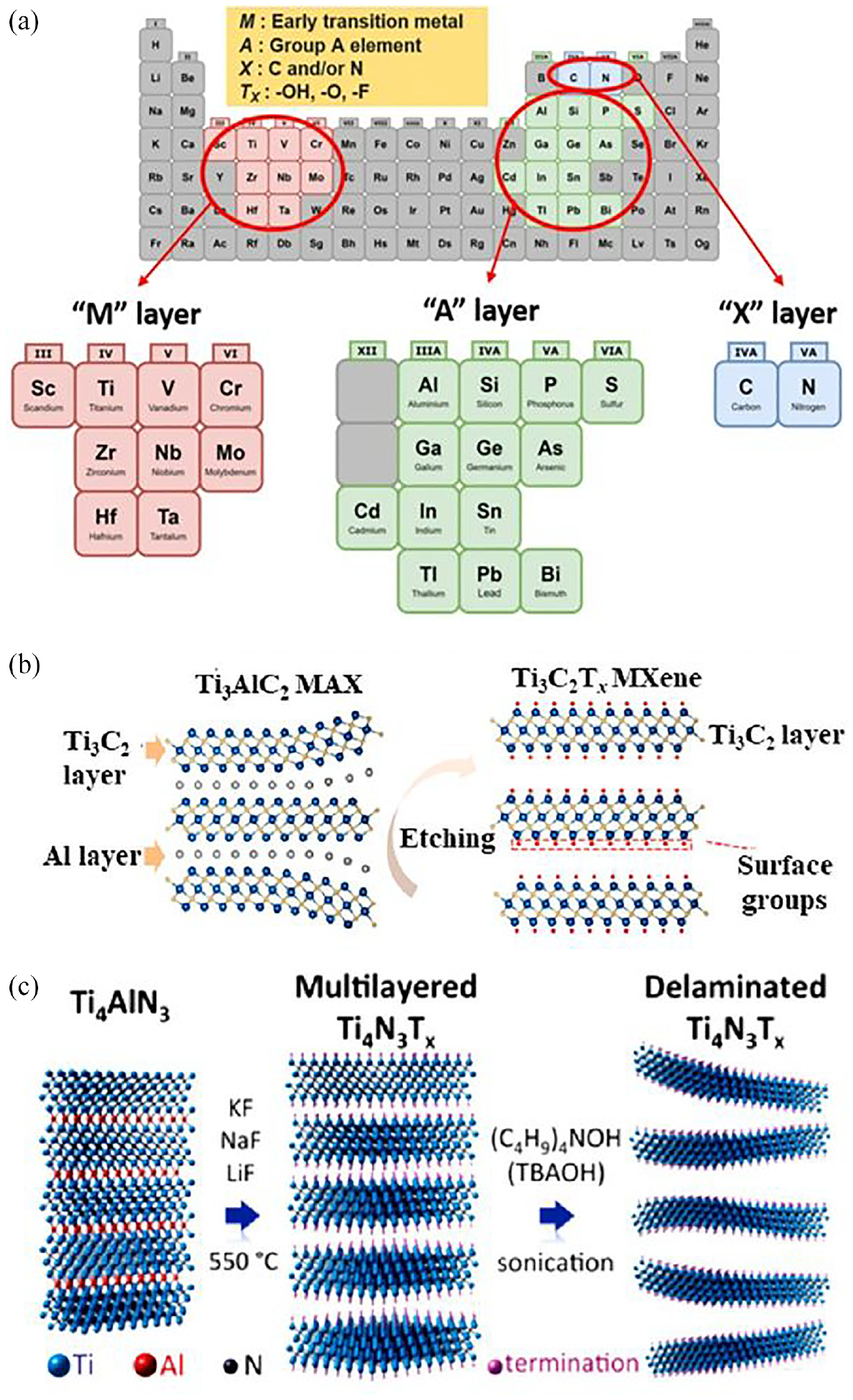
Chemistry of MXene and MAX Phase. (a) The MAX phase composition shown in the periodic table of the elements. 44 (b) Schematic illustration of synthesis of MXene from its MAX phase precursor. Reprinted with permission from Lin et al. 45 (c) Schematic illustration of synthesis of MXene by etching with molten salt. Reprinted with permission from Urbankowski et al. 50
Using Fluoride Salts for Etching
Since hydrofluoric acid is quite dangerous, various alternative acidic etchants that combine hydrochloric acid (HCl) and fluoride-containing chemicals have been proposed. When HCl and fluoride salts are used, the conductivity is increased and MXene is produced with fewer flaws. Crucially, a wide range of fluoride salts are easily accessible, including sodium fluoride (NaF), potassium fluoride (KF), lithium fluoride (LiF), calcium fluoride (CaF2), and ferric fluoride (FeF3).51–54 This procedure replaces the direct use of dangerous hydrofluoric acid.
Researchers have also looked into other difluoride solutions that contain ammonium bifluoride (NH4HF2), sodium bifluoride (NaHF2), and potassium bifluoride (KHF2) as softer etching agents.55,56 However, etching of MXene with hydrochloric acid and fluoride salts produces hydrofluoric acid in-situ during the following processes (equations (4) and (5)). 57 When compared to direct HF etching, this procedure is safer.
Mostly lithium fluoride with hydrochloric acid is used in-situ hydrofluoric etchant, and MXene’s hydrophilic properties are due to its functional group.58,59
Using Molten Salts for Etching
Preparation of MXene (Tin+1Nn) from its MAX phase (Tin+1AlNn) by etching in HF acidic solution is a high energy consuming process. Additionally, MXene containing nitrogen is less stable in hydro fluoride solutions due to the reduced cohesive energy of nitride. 5 It is noted that the mixture of molten salt (KF, NaF, and LiF) at 550°C in Ar to synthesize the first nitride MXene Ti4N3 (Figure 7(c)). 50 ZnCl2 salts were used as the etchant to create halide-terminated MXene from the Zn-based MAX phase precursor. 52 Molten salts are thought to be the most efficient etchant for nitride-based MXenes 60 and have been created recently to etch MAX phases that include a variety of A elements, including aluminum, silicon, zinc, and gallium. 61
Intercalation and Delamination Approaches for Synthesis of Few/Single Layered MXene
Exfoliation is necessary for the synthesis of single or multilayer MXene and can be accomplished through the intercalation of various polar inorganic and organic compounds. When fluoride salts dissolved in HCl further etched, no further delamination agents are required because of in situ delamination by cations. 62 By substituting Al layers for the NH3 and NH4 molecules, MXene interlayers might be delaminated. 52 It has been reported that pre-intercalation of organic molecules into the MXene interlayers, such as urea, dimethyl sulfoxide, isopropyl amine, hydrazine, or large organic base molecules, is effective when followed by mechanical vibration or sonication.39,46 Exfoliation can also be brought on by ionic compounds (metal hydroxide and halide salts), metal cations, and mild mechanical vibration at 7 pH 39 .
Electrochemical Etching Process
In the process of electrochemical etching, the atomic layer of Al is selectively removed from the MAX phase at a particular voltage. Sodium chloride, hydrogen chloride, and hydrogen fluoride are used as the electrolyte. 63 By breaking the M-A bond during a typical electrochemical etching process, the A-layer in the MAX phase can be eliminated utilizing cyclic voltammograms between 0 and 2.5 V (Figure 8(a)). The M-layer is gradually removed by a gradual increase in voltage, leaving the amorphous carbon behind. The spectra of Raman spectroscopy verifies that, in various electrolytes, the Ti3AlC2, Ti3SiC2, and Ti2AlC MAX phases successfully transformed into CDCs (Figure 8(b)). In this type, by adjusting the range of etching voltage in the reaction potential between A and M layers and the proper etching time, selective removal of A atoms can be achieved, allowing fine control of produced MXenes. The etching process is initially performed on the surface of the MAX electrode since the MAX phase normally constitutes the working electrode. During the process the formation of surface CDCs is usually occurred that obstruct the subsequent etching process. Therefore, the key to successfully etching the MAX phase should be the modulation of the etching voltage. Using a three-electrode setup, the MAX phase was electrochemically etched at 0.6 V (vs Ag/AgCl). The working electrodes were thin cuboid blocks cut from the bulk Ti2AlC, and the reference, counter electrode, and electrolytic system were silver (Ag)/silver chloride (AgCl), platinum (Pt), and hydrochloric acid (HCl), respectively. The electrochemical etching process is schematically represented in Figure 8(c). It shows how Ti2AlC transforms into Ti2CTx and CDC layers. The CDC layers on the Ti2AlC surface prevents the further etching, resulting in a MAX covered in MXene. The MAX phase gradually changed into CDC due to the increased electrochemical etching. Further, the effects of electrolyte concentration and electrochemical etching time on the final products are investigated. The morphology of Ti2AlC and its products under various etching conditions are shown in Figure 8(d). It is evident that incomplete etching was the outcome of the procedure using a 1 M HCl electrolyte. After the optimization of etching time and etchant concentration to 120 h and 2 M HCl, respectively, the products’ morphology evolved into an accordion-like structure. When the etching time was increased to 14 days the characteristic of CDC morphology was easily seen, suggesting that the majority of the Ti2AlC was converted to CDC. The electrochemical approach is frequently criticized for producing a low yield due to the concurrent CDC layer over MXenes, despite having various benefits such as low reaction temperature, low energy consumption, and minimal corrosive acid usage. Yang et al. used a two-electrode setup in which the electrolytes were FeCl3, HNO3, H2SO4, NH4Cl, NaOH, and Ti3AlC2 MAX pieces, which were used simultaneously as the working electrode and counter electrode (Figure 8(e,f)). The study emphasized how various electrolytes affect the etching procedure. 64
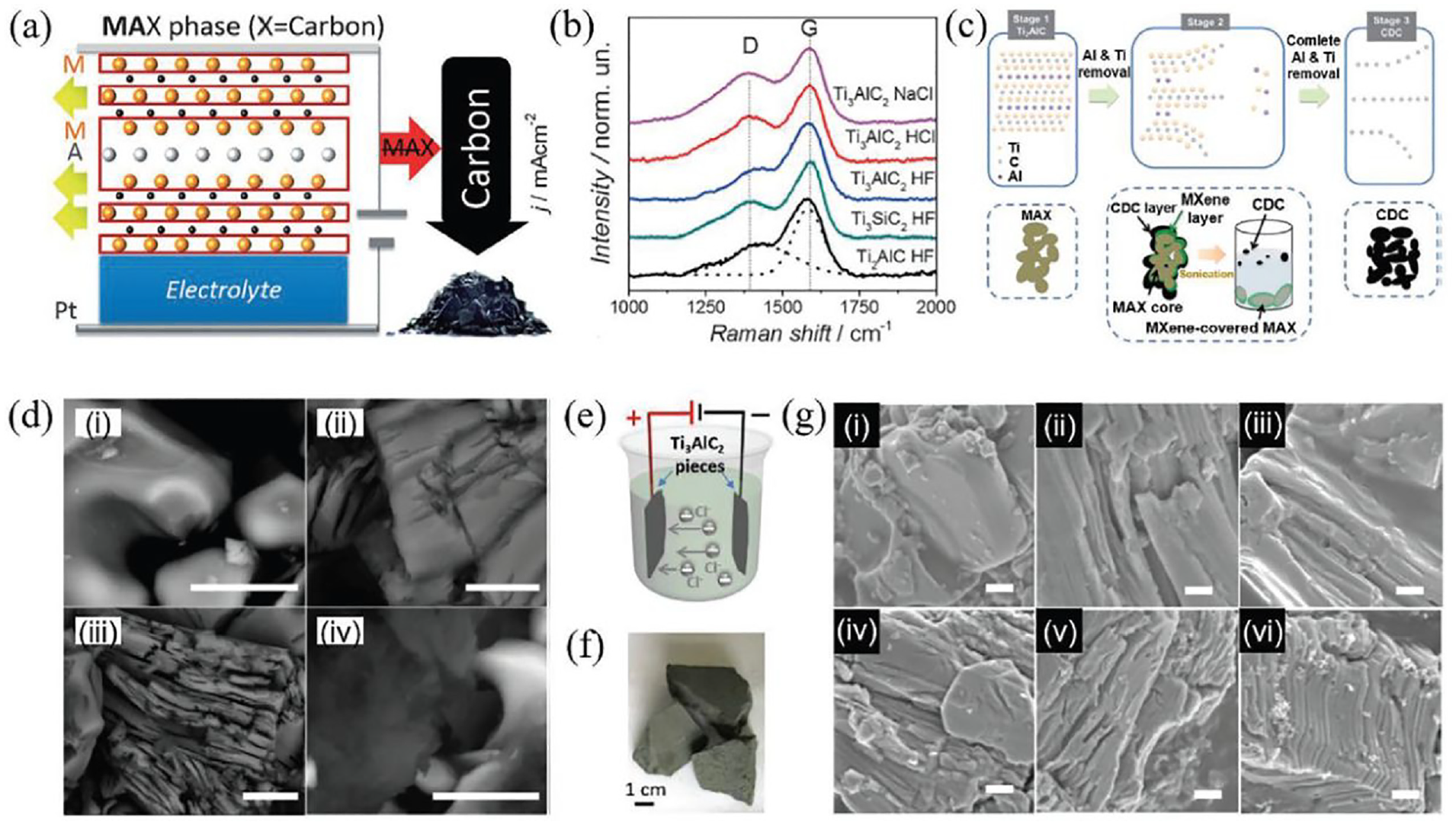
Working methods of electrochemical etching for synthesis of MXene: (a) diagrammatic representation of electrochemical etching of MXene; (b) Raman spectra of various MXene synthesis; (c) the suggested mechanism of electrochemical etching of Ti2AlC in an aqueous HCl electrolyte; (d) MXene prepared by different etching technique and analyzed through SEM; (e) the electrochemical cell for MXene synthesis; and (f) SEM images of MXenes prepared with different etching conditions. Reprinted with permission from ref. 65
Chlorine-free acids like H2SO4 and HNO3 can effectively corrode aluminum foil, but in an electrochemical system, these acids are unable to remove the aluminum atomic layer from the MAX phase. On the other hand, proper etching of Al layer in MAX phase is due to the strong interactions of Al and Cl containing electrolytes. In this case, based on the weight of the product to be etched and the weight of the precursor, we can estimate an etching yield of approximately 40%. Intercalators can be used to extend the interlayer spacing of the MAX phase and allow for continuous diffusion of the electrolyte ions. Intercalating the MAX phase will increase the internal availability of the phase, allowing for proper contact between the phase and ensuring the electrolyte a constant etching reaction. 0.2 M TMAOH and 1 M NH4Cl are one example of a mixture of electrolyte used to obtain MXene (Ti3C2T x ) at 5 V (vs SCE) for a 5 h etching time. 66 The interlayers of the MAX phase could easily intercalated by TMAOH, increasing the electrolyte accessibility of the Al layer. The efficient etching of the MAX phase was made possible by the combination of Al3+ and Cl−, which can break the titanium aluminum (Ti–Al) bond. Consequently, the addition of noncharged NH4OH may also widen the edges of Ti3AlC2 and encourage the etching of the internal MAX phases. The following definition applies to the etching reactions:
While this method can lessen CDC layer interference during the etching process, intercalator toxicity poses a risk to experimental safety. A new thermo-assisted electrochemical etching technique has been reported that could be used to effectively etch without the use of intercalators. Different MAX phases (i.e. Ti2AlC, V2AlC, and Cr2AlC) were etched using a three-electrode system with 1 M hydrochloric acid (HCl), platinum (Pt), and a calomel electrode serving as the electrolyte, counter electrode, and reference electrode, respectively. 66 The etching of Ti2AlC MAX phase and morphology of the products at different temperatures, reaction times and HCl concentrations are displayed in Figure 8(g). The product exhibits no discernible stratification as the temperature drops to 25°C after 9 h of etching, but when the etching time is between 3 and 9 h, the accordion-like structure can be seen at 50°C. With increased efficiency, 9 h of etching time can produce a minimum of one CDC layer.
An environmentally friendly, safe and energy-efficient synthetic method is electrochemical etching. Beyond the inadequate yield, there is still a barrier to overcome due to the CDC layer. Even though the MAX phase can be reused as an electrode multiple times, standard etching methods yield a low amount of MXenes, making them unsuitable for large-scale preparation.
Alkali Etching
In fact, the selective etching of the MAX phase is also anticipated to be realized by the alkali. The surface etching of the MAX phase into Ti3C2T x was achieved through a two-step etching process described by Xie et al. 67 This involves soaking the Ti3AlC2 in 1 M sodium hydroxide solution for 100 h and then in 1 M sulfuric acid solution for 2 h at 80°C. 67 The final morphology of the MXene and the process are depicted in Figure 9(a). Aluminum (Al) atoms from MAX phase layers were extracted using an alkali in this procedure, whereas Al atoms that were exposed to the surface were extracted using H2SO4. Using a low concentration of alkali as an etchant, the process effectively etched the MAX phase; however, the yield of MXene produced when etching the surface layer of the MAX phase was only very low. The main challenging task is to separate etched MXene from its precursor. In alkali etching some oxide/hydroxide layers formed on the MAX phase. A core–shell MAX@K2Ti8O17 composite was created in one instance when 2 M KOH was used to etch Ti3SiC2 at 200°C using hydrothermal reaction. 68 On the other hand, employing NaOH hindered the production of pure MXenes by causing the surface of the MAX phase to form Na2Ti7O15.
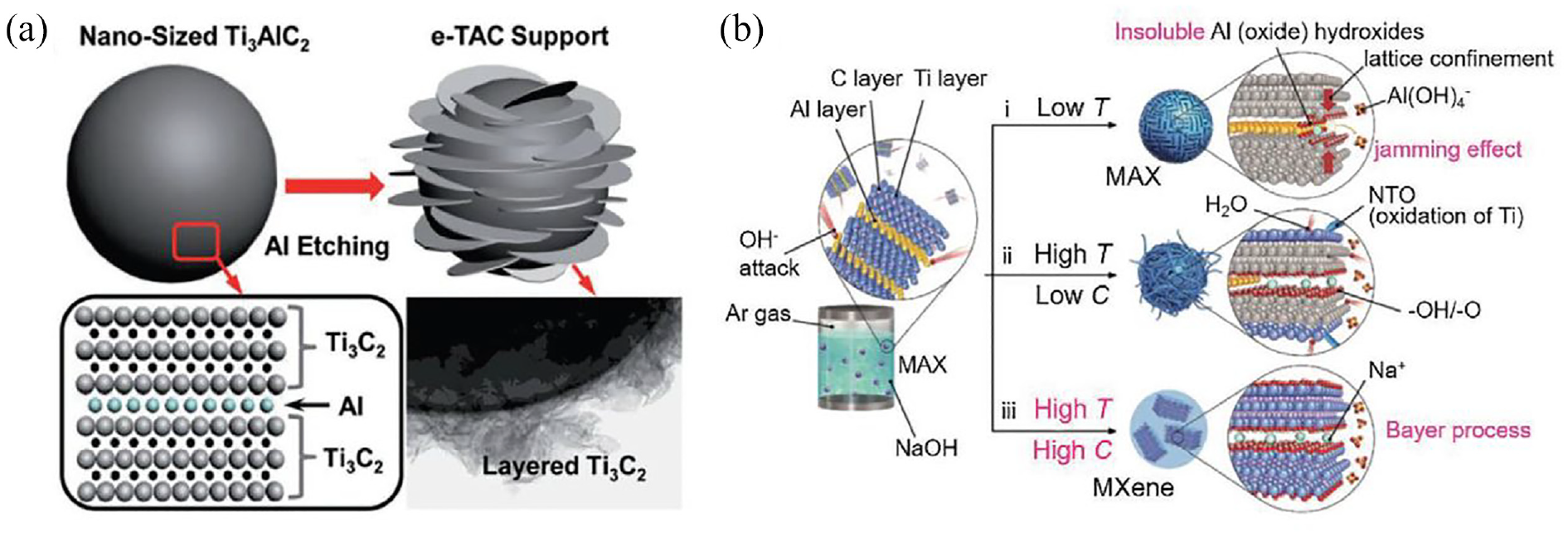
The characterizations and morphological structure of MXene which is obtained by alkali etching: (a) schematic for the formation of layered Ti3C2. In the product interior the unetched Ti3AlC2 is present; and (b) the reaction between NaOH and Ti3AlC2 under different conditions. Reprinted with permission from ref. 65
Once the temperature and alkali concentration are maintained at a certain point, there will be some qualitative changes in the reaction between the MAX phase and alkali. For instance, Ti3AlC2 could have its Al layer effectively removed with 27.5 M sodium hydroxide at 270°C to produce Ti3C2T x with a 92% yield. 69 The corresponding alkali etching process reaction and schematic diagram are displayed in Figure 9(b). The primary reaction pathway involved the conversion of Al into Al (oxide) hydroxides and its subsequent dissolution in the alkaline medium. The following reactions took place during the etching process:
In this instance, fluorine-free MXenes could be easily formed by dissolving Al (oxide) hydroxides at high temperatures and with concentrated NaOH. More alcohol (-OH) and oxygen (=O) terminations groups were made possible by the fluorine-free environment, which improved the overall performance of supercapacitors. In this instance, the 52 μm thick Ti3C2T x film electrode produced a volumetric capacitance of 511 F cm−3 at 2 mV s−1 and a gravimetric capacitance of 314 F g−1 in 1 m H2SO4 electrolyte.
In a similar manner, Li et al. used 0.35 g potassium hydroxide (KOH) in 0.05 mL water to etch 0.1 g Ti3AlC2 at 180°C for 24 h in a hydrothermal reactor. 70 Here, large lateral dimension 2D Ti3C2(OH)2 nanosheets can be formed by substituting -OH groups for Al atoms. The alkali concentration in this method reached up to 87.5 weight percent. In a similar vein, The hydrothermal etching of Ti3AlC2 at 180°C for 24 h with a high concentration of KOH (approximately 93.3 wt%) yielded MXene nanoribbons. 71
Etching the MAX phase with concentrated alkali is a very efficient technique that produces good hydrophilic products with F-free terminations. However, the risks involved in using highly concentrated alkali and high temperatures limit its applicability for large-scale MXene preparation. Moreover, the end products are usually accordion-shaped multilamellar MXenes, which require additional intercalation and delamination to become single-layer MXene nanosheets.
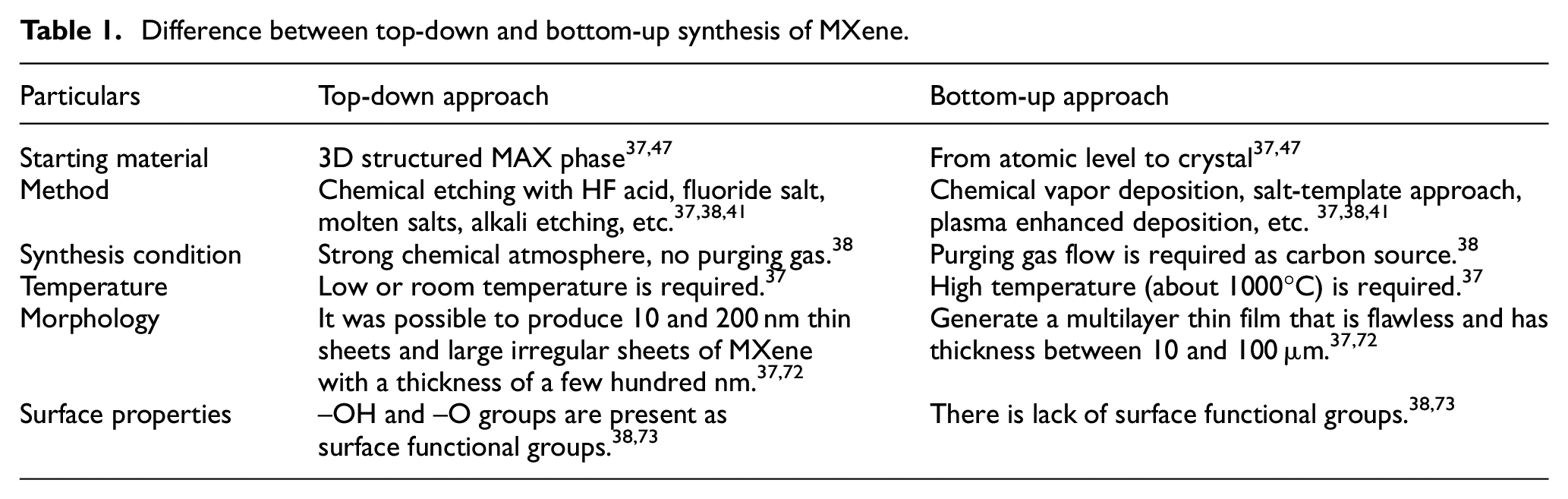
A comparative assessment between the top-down and bottom-up synthesis approaches of MXene and the material properties obtained are summarized in Table 1. Among the top down approaches, the electrochemical etching is most suitable for textile applications owing to its green and safe synthesis method, low energy consumption, and minimal usage of corrosive acid.
Difference between top-down and bottom-up synthesis of MXene.
Preparation of MXene-Based Textile
Coating
Coating textile substrates with conductive nanoparticles is one of the methods that has received the most attention because of its simple, scalable, and inexpensive procedure. Coating textile fibers, yarns, or textiles with a range of nanomaterials, including metallic nanowires, conductive polymers, CNTs, and graphene like MXene, is a common practice. In this regard, a variety of natural and synthetic textile substrates, such as cotton, nylon, polyamide, PET, and other fibers or yarns have been coated with MXene.74–76 The coating strategy usually involves making a homogenous MXene dispersion first, then soaking, spraying, or drop-casting the textile substrate and evaporating the solvents to remove them (Figure 10). Ti3C2T x MXene has been coated onto pure cellulose fibers to produce an MXene loading of 77%. After 45 h of washing cycles at 80°C with constant stirring, the barely noticeable changes in resistance showed that MXene had adhered to the cotton fibers to a good extent. Furthermore, without losing their electrical conductivity, MXene-coated cellulosic yarns have good knit ability using a range of stitch patterns. 77 Air laid nonwoven textiles are coated with MXene by the dip coating method to prepare multifunctional textiles suitable for applications such as personal heating systems, electromagnetic interference shielding, and pressure sensing. 78
Schematic diagram of preparation of MXene coated smart textiles. 79
In another study, MXene was drop-cast onto nylon fibers that had been plated with silver. The SEM micrograph showed that the MXene flakes were coated evenly on each fiber’s surface. 76 MXene-coated fabrics also exhibit this morphology.79,80 Although coating made it possible to create electroactive and elastic fibers without interfering with the host polymer’s stretchable network, multiple coating cycles were required to achieve high mass loading.80,81 A vivid presence of MXene flakes on cotton, hemp and nylon surface is observed under SEM micrograph as shown in Figure 11 which reveals a uniform deposition on MXene on these fiber surfaces.

SEM image of (a) uncoated cotton, (b) uncoated hemp, (c) uncoated nylon, (d) MXene coated cotton, (e) MXene coated hemp, and(f) MXene coated nylon fibers under 1,000 × (left) and 10,000 × (right) magnifications. 82
Electrospinning
Electrospinning is a useful technique for creating nanoscale-diameter fibers from a composite solution based on polymers. Graphene, conducting polymers, and metallic nanoparticles are among the many forms of nanomaterials that have been added to spinning dope to create a vast array of composite fibers. When the electroactive particles are included there is an increase in the solution’s net charge density observed, which has a substantial effect on the fibers’ spinnability. While electrospinning is thought to be a scalable method for producing stretchable polymeric nanofiber yarns, wearable electronics applications have not used it as much as other applications. This could be because the solution is extremely sensitive to electrospinning, and with high loadings of the electroactive material, hundreds of nanofibers induced beading in the fiber formation due to particle agglomeration and restacking.
For the general electrospinning process to produce nanofibers, a syringe with a polymer solution capped with a needle tip and a grounded metal collector are required (Figure 12). When the surface tension of the solution is overcome by the electrostatic force, the needle tip extrudes a jet of polymer, causing fibers to gather across the metal plate. This process is initiated by a high voltage. For electrospinning of composite polymeric fibers based on MXene, different solvent systems of homogeneous MXene dispersions need to be prepared. In order to achieve this, numerous researchers have created nanofibers by adding MXene to fiber spinning solutions, such as PET/Ti3C2Tx fibers, 83 MXene/CNT@polycaprolactone (PCL) fiber mats, and poly(vinyl alcohol) (PVA)/Ti3C2Tx fibers. 84 For example, carbonization after polyacrylonitrile (PAN)/MXene electrospinning was used to obtain MXene nanofibers which have MXene loading of 35%, which is the highest among all reported MXene loadings for electrospun fibers. However, recently, MXene-infiltrated nanowires with up to 90% MXene loading were created from nylon and polyurethane using one-step bath electrospinning. 81
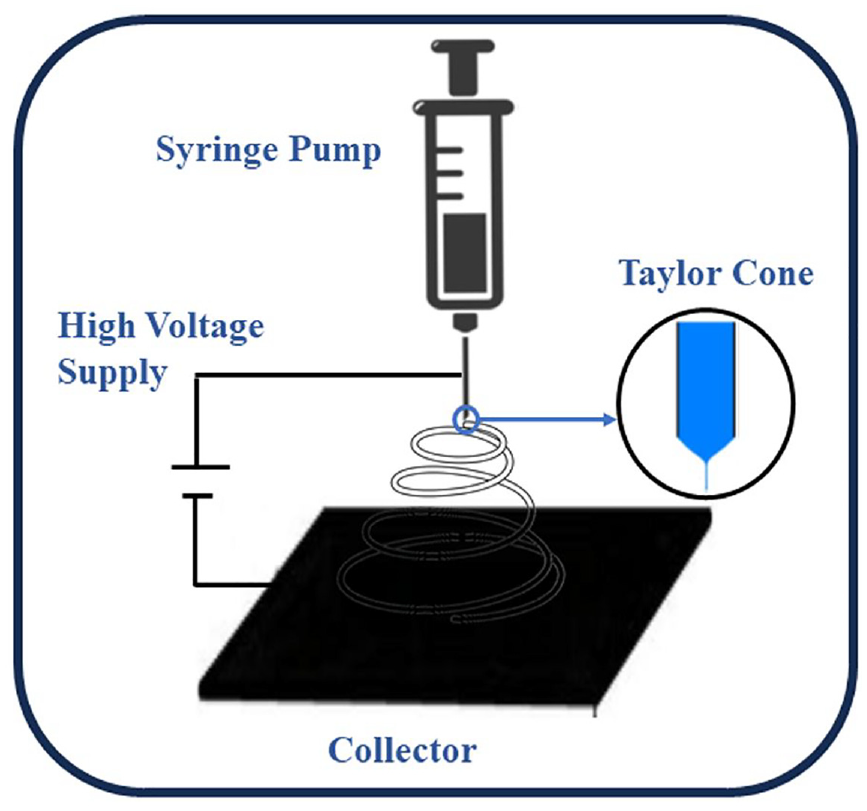
Schematic diagram of preparation of MXene based textiles through electrospinning. 79
The ideal interaction between nanofibers and MXene pieces was accomplished by creating an extraordinary mix of damp turning and electrospinning (current collector). The highest electronic conductivity to date was demonstrated by nylon/MXene nanoyarns, reaching 1195 S cm−1. The amazing use of MXene with its high electrical conductivity and satisfactory fiber properties (such as cell adhesion and proliferation to promote skin regeneration, which is often used as a dressing to promote wound healing after surgery) 85 have made this manufacturing strategy successful. SEM micrograph images of PVA/MXene electrospun nanofiber and EDX spectra of same are shown in Figure 13. It can be seen that the nanofibers have a smooth surface with uniform morphology. EDX spectra shows the presence of Ti, Cu, C, O etc. in the materials.

SEM images of PVA/MXene nanofibers: (a) 500×, (b) 2500×, (c) EDX spectra of PVA/MXene fiber. 85
Wet Spinning
Wet spinning has gained popularity as a continuous, scalable, and effective method of producing long fibers. Utilizing this strategy, a number of useful fibers which are produced by conductive and carbonaceous materials have as of now been developed. The wet spinning method involves injecting a polymer solution into a coagulation bath through a spinneret, resulting in coagulation and solidification of the manufactured fiber (Figure 14). The fiber is then taken out of the bath, allowed to dry, and then continuously gathered onto a winder. Due to MXene’s superior dispersion into a wide range of polar solvents, a number of materials have been combined with MXene in spinning dope to create hybrid fibers thus far. A fiber-shaped hybrid supercapacitor anode is developed by the wet-spinning technology doping rGO and MXene in fiber spinning solution. Electrochemical performance and excellent electrical conductivity (743.1 S cm−1) were achieved at 60% of MXene loading. 87 By employing this method, the hybrid fiber assembly can attain a high MXene loading (10−98%). Furthermore, using the wet spinning method pure MXene fibers can be spun owing to their exceptional liquid crystalline behavior, pseudoplastic rheological characteristics, and dispersibility in a variety of solvents. MXene fibers are successfully developed by solution spinning method and the morphology of the fibers is shown in Figure 15. The cross-sectional and longitudinal view of the fiber depicts that the fiber resembles viscose fiber like morphology with irregular serrated shape.
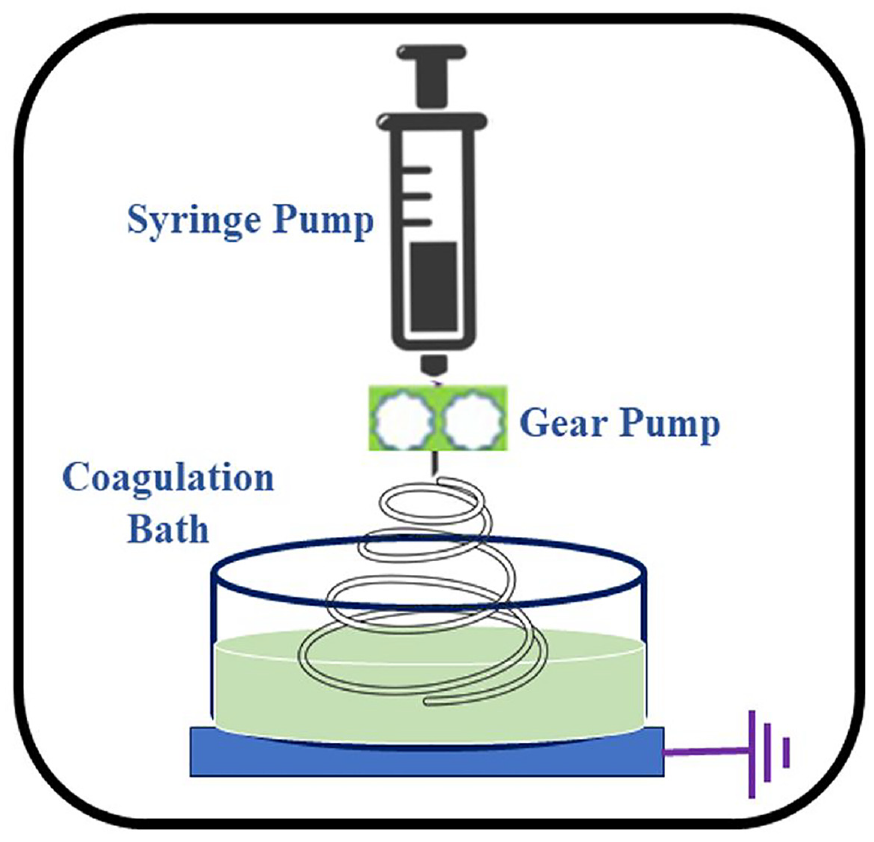
Schematic diagram of preparation of MXene based textiles through wet spinning. 79

SEM images of Ti3C2Tx MXene fiber: (a) overall, (b) in cross-section, and (c) longitudinal views. 88
Enhancing the Adhesion of MXene on Textiles Substrate
The adhesion of MXene on textile substrate is essential for successful surface deposition during the coating process by using nip-and-dip technology, which is the widely used technique due to its simplicity and ease of application.
The presence of hydrophilic groups on the surface of MXene, as was explained in the preceding section, aids in its processability in the solution route. This enables them to be processed into membrane devices, composite paper, or thin films using spray coating, 89 vacuum filtration, 90 or composite paper. 91 However, due to the weak interlayer contact and thin sheet size between MXene sheets, coating MXene directly onto the fiber still presents difficulties. 92 Despite having a lower flexibility and stickiness, which are essential for wearable applications, higher conductivity is obtained when the textile substrate is coated with a greater loading percentage with large flake size Figure 16. 77

The cross-sectional analysis of cotton yarn: (a) pristine, (b) coated with S-Ti3C2 MXene flakes, and (c) coated with S-Ti3C2 and L Ti3C2 MXene flakes. SEM images of cosssection of (d) pristine cotton fibers, (e) cotton fibers coated with S-Ti3C2 MXene flakes, and (f) S-Ti3C2 and L-Ti3C2 MXene coated cotton yarn. SEM images of longitudinal view of cotton yarn: (g) pristine, (h) coated with S-Ti3C2 MXene flakes, and (i) coated with S-Ti3C2 and L-Ti3C2 MXene flakes. Reprinted with permission from ref. 77
To solve the adhesion issue, MXene has been grafted with other conducting materials such as poly(3,4-ethylenedioxythiophene) polystyrene sulfonate (PEDOT: PSS). According to Zhang et al., 112 PEDOT: PSS works as a binder, fixing together all the MXene sheets and improving the MXene’s adherence to the fiber surface. Moreover, the incorporation of PEDOT: PSS improves the coating proficiency by preventing MXene from washing off carbon fiber substrates. 93 Oxygen containing groups functionalize the fiber surface when plasma treatment is done. The plasma treatment of textiles is another strategy to enhance the adherence of MXene to the fiber surface. 76 As an illustration, carbonized silk fibers were found to have a hydrophilic fiber surface after being exposed to oxygen plasma. 94 This further improved the adhesion of MXene onto the fiber surface. Furthermore, by functionalizing the PET surface with 3-aminopropyl tri ethoxy silane (APTES), the PET surface can be altered to affect the engagement between hydrophobic polyethylene terephthalate (PET) yarns and MXene. In order to create amine functional groups, PET fabrics were submerged in a 2% (v/v) APTES solution for 30 min. The MXene dispersion was then dipped into the functionalized textiles, improving the absorption of MXene flakes onto the fiber surface. 95 The positively charged APTES-functionalized PET and the negatively charged Ti3C2T x interact electrostatically to cause MXene flakes to self-assemble on the individual PET fibers, creating a densely packed PET surface.
Performance of MXene-based Textiles for Energy Storage Applications
Textile materials like yarns and fabrics can absorb water and swell in the colloidal solution of MXene due to the presence of rich hydrophilic groups, like hydroxyl, anime, carboxylic etc., and porous structure. Therefore, using a simple dipping-coating technique, Ti3C2Tx nanosheets possessing strong metallic conductivity and hydrophilicity can be coated onto the textile surface uniformly. The pseudo capacitance material PPy, possessing sufficient environmental stability, can be electrochemically coated onto the surface of the MXene-coated textile to enhance its capacitive performance even further and that prevents oxidation of the exposed Ti3C2T x . Additionally, the symmetrical solid-state supercapacitors (SCs) were created by sandwiching two PPy–MXene coated textiles, where H2SO4/PVA gel was used as the electrolyte, between two macroporous textiles (as the separator). Further, to stop the above SCs from completely dehydrating, a thin film of PE (polyethylene) was laminated and fixed over them (Figure 17).
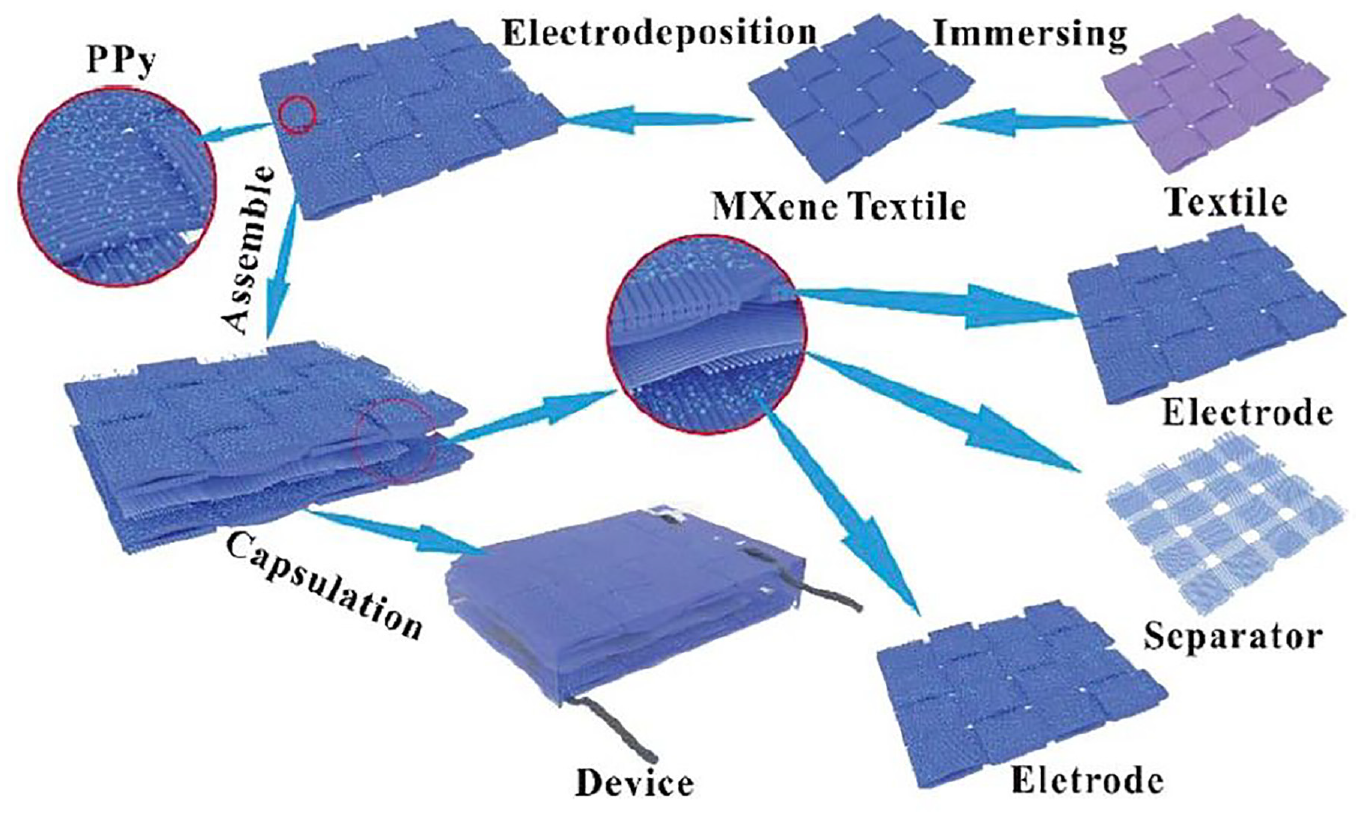
Schematic diagrams of PPy/MXene coated textile supercapacitors. 96
Comparison of Volumetric Capacitance of MXene
MXene’s high conductivity (up to 9880 Scm−1), good hydrophilicity, and superior ion intercalation behavior have made it very promising as a possible material for electrochemical energy storage.93,95–101 In fact, Ti3C2T x MXene has already been shown to be a very good electrode material when used in interdigitated coplanar micro-supercapacitors,102,103 fiber SCs, 97 and sandwiched structure SCs. 104 In contrast to carbon materials, Ti3C2T x “clay” films have volumetric capacitances of up to 900 F cm−3101 Figure 16.
In fact, it has already been demonstrated that Ti3C2T x MXene is a superior electrode material for use in sandwiched structured supercapacitors (SCs), 97 fiber SCs 104 and coplanar interdigitated micro-supercapacitors.102,103 As illustrated in Figure 18, Michael Ghidiu et al. found that the volumetric capacitance of Ti3C2T x clay films (∼700 Fcm−3) is highest among other materials. 100
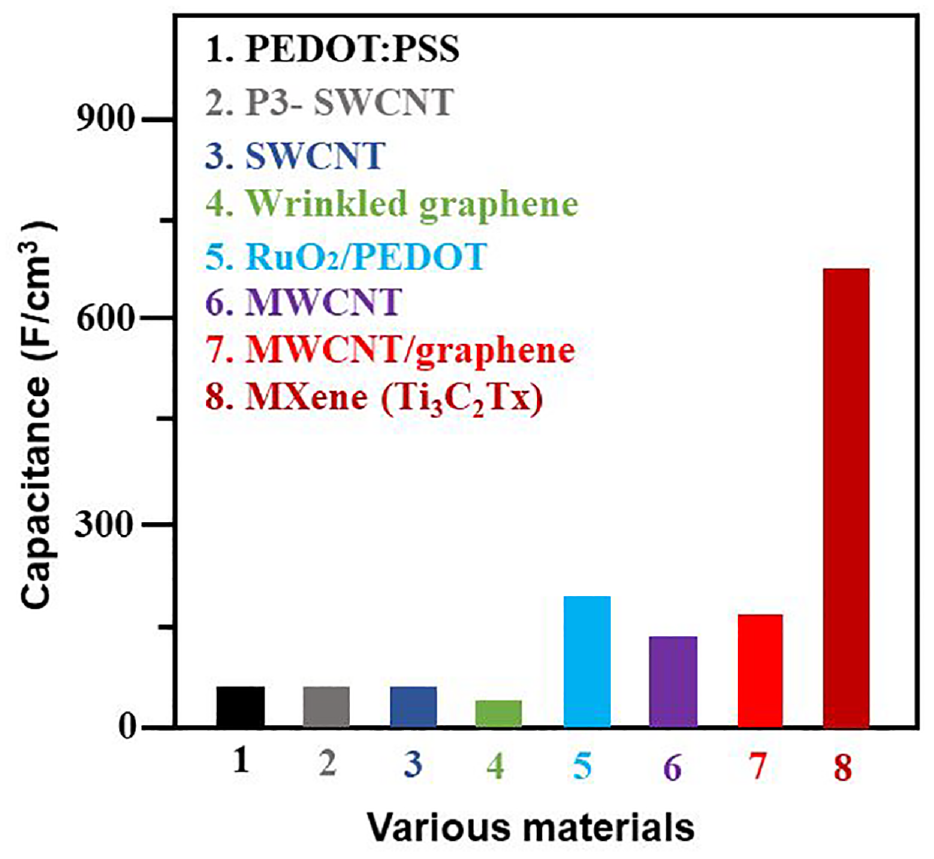
Volumetric capacitance of Ti3C2T x MXene and other materials. Reprinted with permission from ref. 97
Moreover, Xu et al. found that the Ti3C2T x based asymmetrical micro-supercapacitors is prepared through screen-printing method, possessing with a high energy density of about 8.84 µW h/cm2 that can operate a force sensor to detect variations in applied pressure. 105 Many efforts have been made to build various MXene electrode architectures. In an effort, performance and ion accessibility of Ti3C2T x MXene were further enhanced by creating more atctive sites. 106 For example, mechanical shearing of a discotic lamellar liquid-crystal phase of Ti3C2T x was used to achieve the vertical alignment of MXene nanosheets. This method is independent of film thickness and can effectively improve ion transport. 106 Even though research on Ti3C2T x -based SCs has made a number of advances, further optimization of their preparation method is still necessary for stability and commercialization (primarily for low-cost, easy preparation). However, restacking and oxidation of Ti3C2T x MXene during use must be prevented, as these actions will impair stability and relative capacitance. 96
CV and GCD Curve of MXene Coated Textiles
Polypyrrole (PPy) was deposited on the surface of MXene-based textiles electrochemically to enhance the capacitive performance of the MXene-based electrode and prevent oxidation of MXene. As illustrated in Figure 19, the CV and GCD curves verified that after the PPy coating the capacitive performance of the MXene-based electrode was remarkably improved. The PPy-MXene coated textile electrode has high specific capacitance (343.2 F g−1), and the enclosed area of the CV curve from this electrode was roughly three times larger than that from the MXene-based electrode. The CV curves are observed to have a large enclosed area and a good symmetrical rectangular shape at scan rates between 5 and 100 mV s−1. Additionally, the GCD curves display an isosceles triangle’s shape. With a current density of 1.0 A cm−2, the calculated capacitance from GCD curves reached a maximum of 275.2 F g−1.
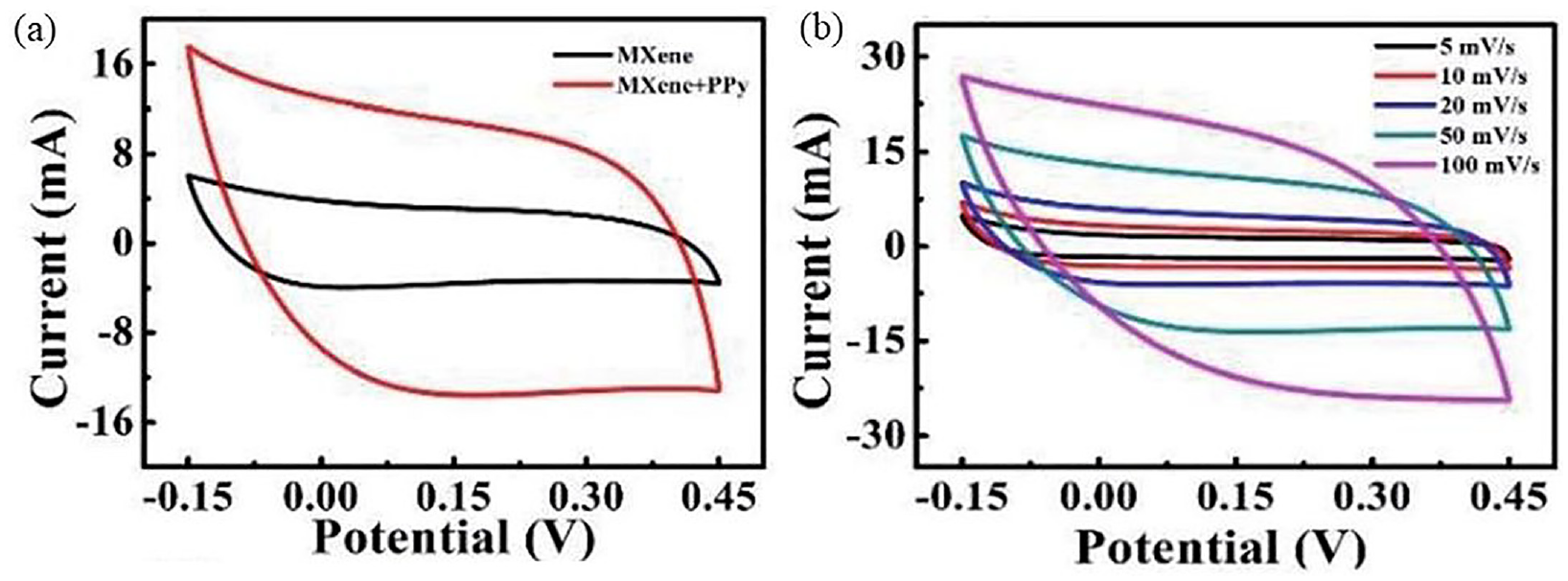
Capacitive performance of MXene and MXene coated textiles: (a) CV curve; and (b) GCD curve. 96
Mechanical Performance of MXene Integrated Textiles
Many polymer matrices have MXenes added to them to improve the mechanical characteristics of composite materials. Different MXene materials are efficient reinforcement agents for polymer composites due to their large elastic moduli (500 GPa). 48 With respect to MXene nanocomposites prepared through cellulosic material, the extraordinary nanostructured MXene worked as an effective nanofiller, enhancing the moduli and mechanical strength of the composites.91,107 It was discovered that the use of extremely small amounts of MXene might produce a reinforcing effect that was more effective than the use of raw ingredients. Cao et al. 90 utilized a vacuum-assisted filtration approach and made a composite paper for EMI shielding by embedding two-dimensional Ti3C2T x into a one-dimensional cellulosic nanofiber (CNF) framework. There is a significant enhancement in Young’s modulus (3.8 ± 0.3 GPa) and mechanical strength (135.4 ± 6.9 MPa) of the coated CNF paper, which were about 2.5 and 2 times greater, respectively, than those of pristine CNF paper, at an optimized MXene loading (50 wt%) as shown in Figure 20.
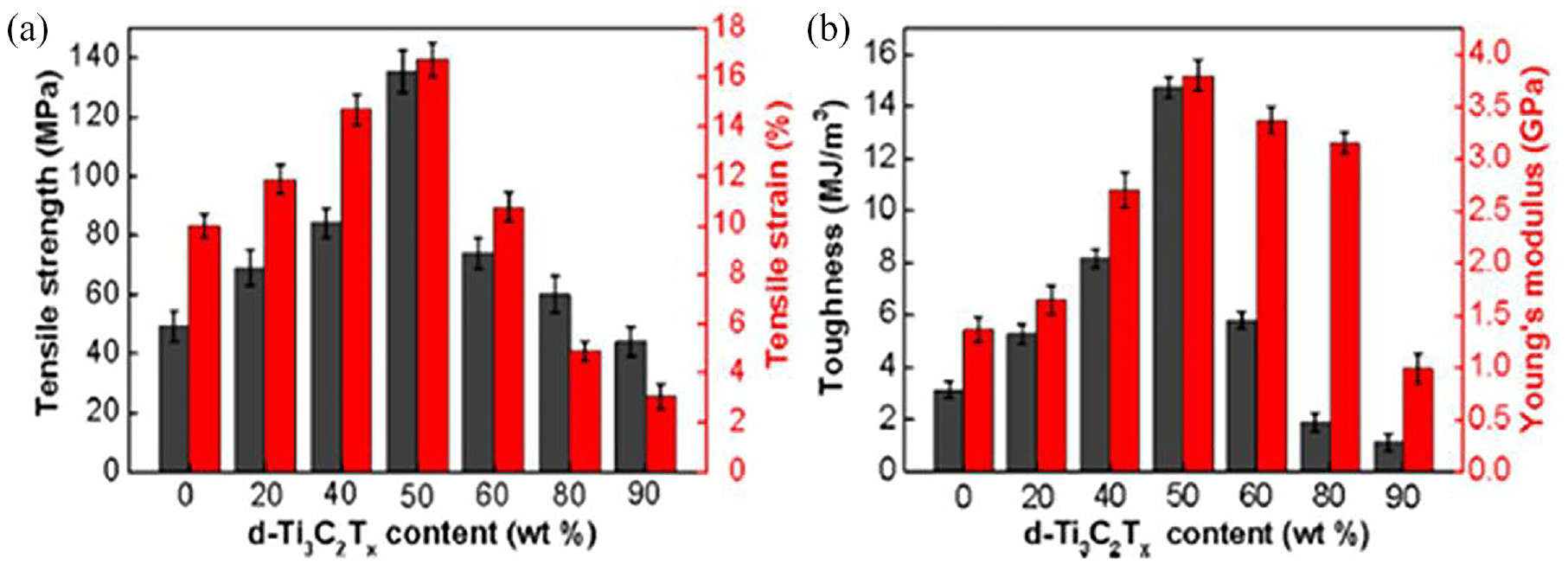
Tensile properties of MXene coated cellulose: (a) tensile strength and tensile strain of the MXene/CNF composite paper with different d-Ti3C2T x contents; and (b) toughness and Young’s modulus of the MXene/CNF composite paper with different d-Ti3C2T x contents. 90
According to an SEM study on the fracture morphology of the MXene and CNF paper, the arrangement of a vigorous interconnection between MXene nanosheets and CNFs within the composite structure permitted the maintenance of a closely staked layered structure. In order to investigate the necessary mechanical characteristics of materials for specific applications, a number of MXene/cellulose composites have been created. It’s interesting to note that for identical applications, this mechanical property improvement was better than that of its 2D cousin graphene and others.108–110 Another case is the higherachially aligned MXene/CNF composite film developed by Zhan et al. 89 It has been reported that Ti3C2T x layers are intercalated into CNF, as shown by the authors’ analysis of the XRD spectra, which was attributable to the broadening and displacement of the peak (Figure 21), which produced an extremely strong composite film with superior tensile capabilities. The study reported the highest ever values for Young’s modulus (7 GPa) and remarkable tensile strength (212 MPa) for other MXene functionalized materials. Although the tensile strength and modulus of MXene/cellulose membranes improved, this reinforcement is frequently determined by a number of variables, including the MXene flake size, interactions between the filler and the matrix, the degree of filler dispersion in the matrix, the tendency of the filler to aggregate, and the orientation of the fibril modulus.
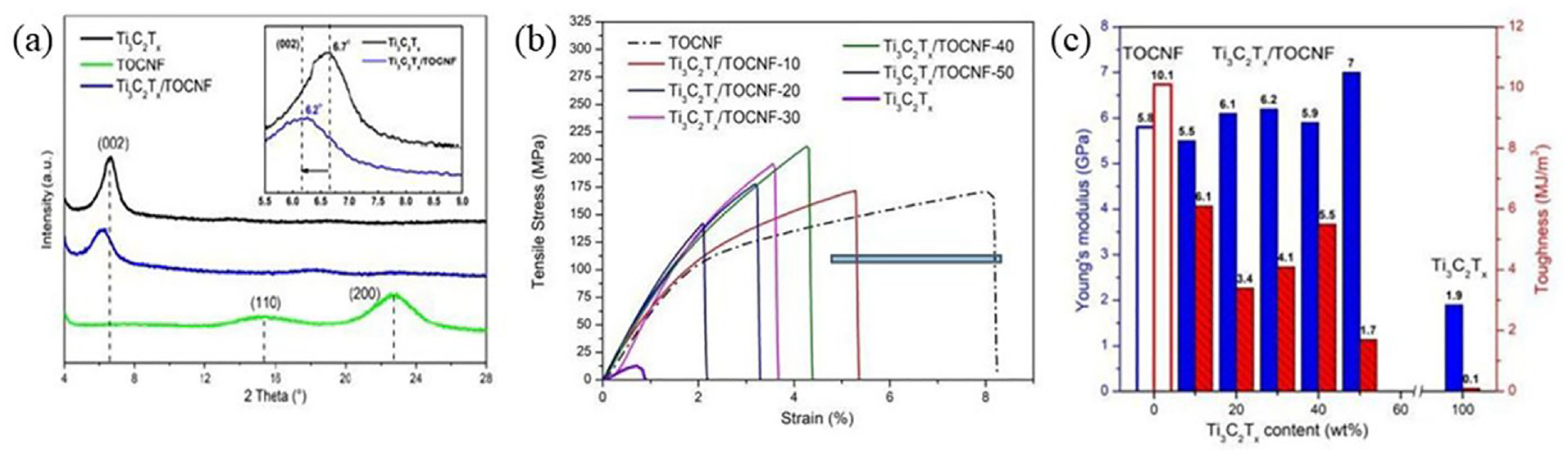
Physical properties of MXene coated cellulose: (a) XRD patterns; (b) stress–strain curves; (c) Young’s modulus and toughness histograms. Reprinted with permission from ref. 111
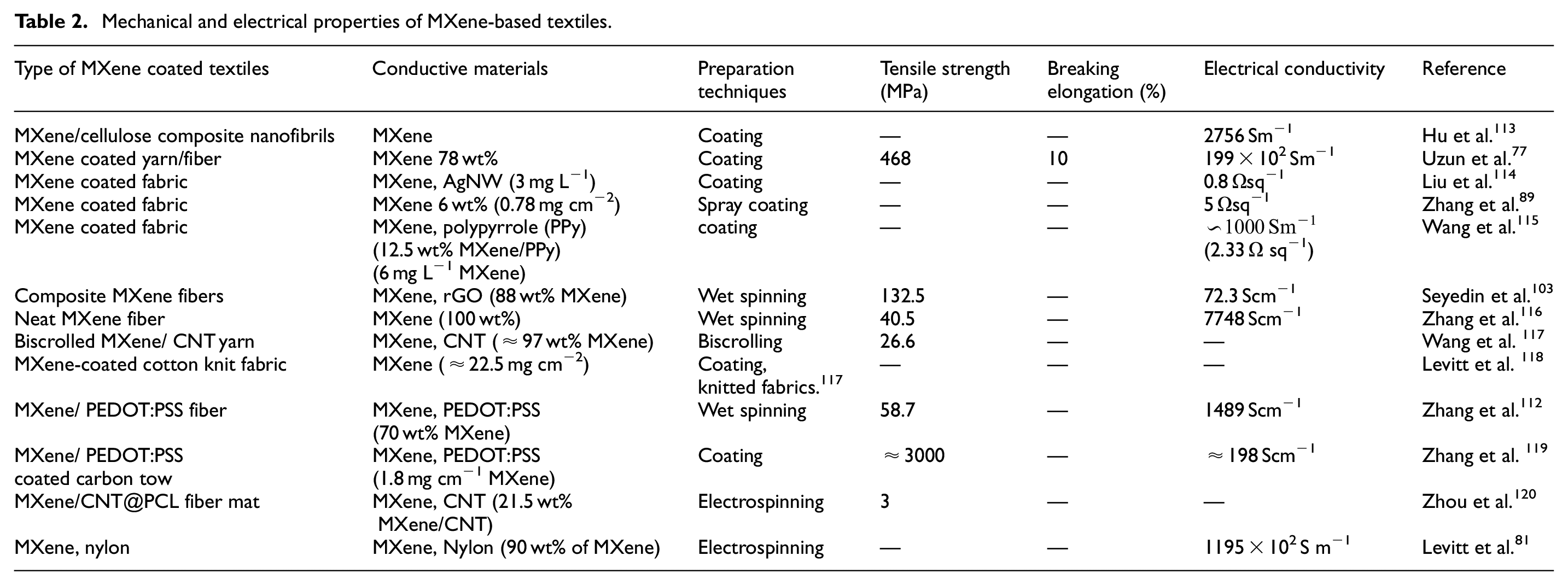
On the other hand, the interaction between the MXene flakes and substrate fibers results in the wet spun MXene composite fibers that have exhibited exceptional mechanical performance properties. A recent study showed that at 15 wt% MXene loading, the wet spun MXene and GO fiber composite yielded flawless rGO filaments with enhanced mechanical strength of up to 132.5 MPa, a Young’s modulus of up to 11.3 GPa, and a strain at break of up to 2.9%. When MXene loading is increased to 88 wt%, the spun fiber showed a modest reduction in the strain at the break. According to several studies,92,104,112 at a high MXene loading (>70 wt%) the MXene composite fiber often exhibits a failure to strain of less than 5%, which may be a result of the smaller MXene flakes’ decreased inter-sheet sliding. Therefore, maintaining mechanical integrity is crucial in order to convert these fibers into textiles further through weaving or knitting. In comparison to neat spun MXene fibers, it was discovered when a small amount of active materials, such as PEDOT:PSS or rGO was added to MXene containing spinning solution, the tensile strength of the fibers was greatly increased. Compared to pristine cotton yarn (334 MPa), 79 wt% MXene loading cellulose fibers displayed excellent tensile strength of 468 MPa. When MXene was used to coat linen and bamboo fibers, improved mechanical characteristics were also obtained. 77 Table 2 provides a review of the mechanical and electrical characteristics of materials based on MXene.
Mechanical and electrical properties of MXene-based textiles.
Numerous researchers have reported on the tensile strength of textiles coated with MXene and other conductive materials. The literature was consulted in order to compile, summarize, and plot the percentage increase in the coated textiles’ tensile strength, as shown in Figure 22. It can be seen that the increment of tensile strength of MXene coated textile is maximum (∼90%) in comparison to other conductive materials.
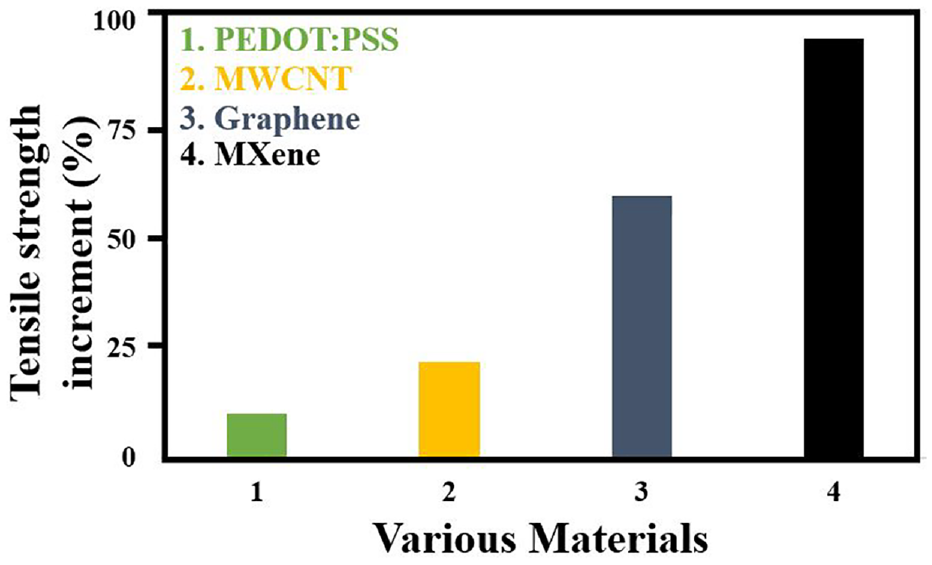
Electrical Performance of MXene Integrated Textiles
MXene is a desirable material for electronic applications due to its exceptional electrical properties. Among the solution-processable 2D materials, MXene is an intriguing material that has the highest conductivity. It is thought to be a potential nanomaterial for the production of MXene-based fibers, membranes, functional films, and other textile goods. The electrical characteristics of textile fabrics based on MXene are listed in Table 2.
It was discovered that the electrical characteristics of the textile substrate significantly increase when a tiny mass loading of MXene is added. Vacuum-assisted assembly of cellulose composite papers containing 50 weight percent Ti3C2T x MXene resulted in excellent electrical conductivity of 2837 Sm−1, which is attributable to Ti3C2T x nanoflakes’ assistance of an effective flow of conductive electrons and orientation. After 1.89 vol% of MXene loading on cellulosic filter paper by a dip-coated method, nearly the same conductivity was achieved. 113 Additionally, Xin et al. created MXene-silver-CNF composite membranes utilizing a vacuum-assisted filtration technique, utilizing MXene-reduced silver particles. 107 Electrical conductivity was found to rise 43 times more with the addition of MXene-reduced silver nanoparticles than it did with a composite membrane made just of MXene and cellulose. 79 This is most likely due to the improved mobility of conducting free electron supported by the improved interfacial interaction between silver nanoparticles and MXene.
According to Zhang et al., 89 MXene fibers spun through a pure wet-spinning process have the highest electrical conductivity of any MXene-spun fibers to date, reaching 7748 S cm−1. The size of the MXene flake, however, has a remarkable effect on the electrical conductivity of these fibers. As a result, clean MXene fibers treated from small flake size had half the electrical conductivity of fibers processed from big flake size. 116 The electrical conductivity of composite spun fibers based on MXene followed a similar pattern. The type of solvent employed also affects the electrical conductivity of MXene spun composite fibers. When MXene loading was increased from 2 to 6 wt% in cotton fabrics, it was discovered that the sheet resistance decreased from 48 to 5 Ωsq−1 88 . This shows that highly electroconductive smart fabrics can be created using even a small amount of MXene.
The SEM study reveals that the surface topology of MXene coated polyester fabric is comparable to that of uncoated polyester fabrics (Figure 23(a,b)). Figure 23(c) shows that as the coating time increased, the electrical resistance of the MXene coated fabric reduced and the surface temperature increased. As the electric field increased from 2.0 to 3.3 V/cm, the yarn heater engraved on cotton gloves for thermotherapy exhibited a progressive rise in temperature, reaching a maximum value of 53.5°C, as shown in Figure 23(d, e). Additionally, a flexible fabric heater made from a knitted structure demonstrated exceptional heating capabilities across a range of geometries and extreme deformations. Figure 23(f, g) shows that the MXene fabric heater has excellent potential for application in wearable thermotherapy because it can successfully raise body temperature when attached to a human wrist by dispersing heat energy throughout the arm without causing harm to the skin. 79 These experiments sufficiently evidenced the excellent electrical performance of electro-conductive MXene coated textiles.
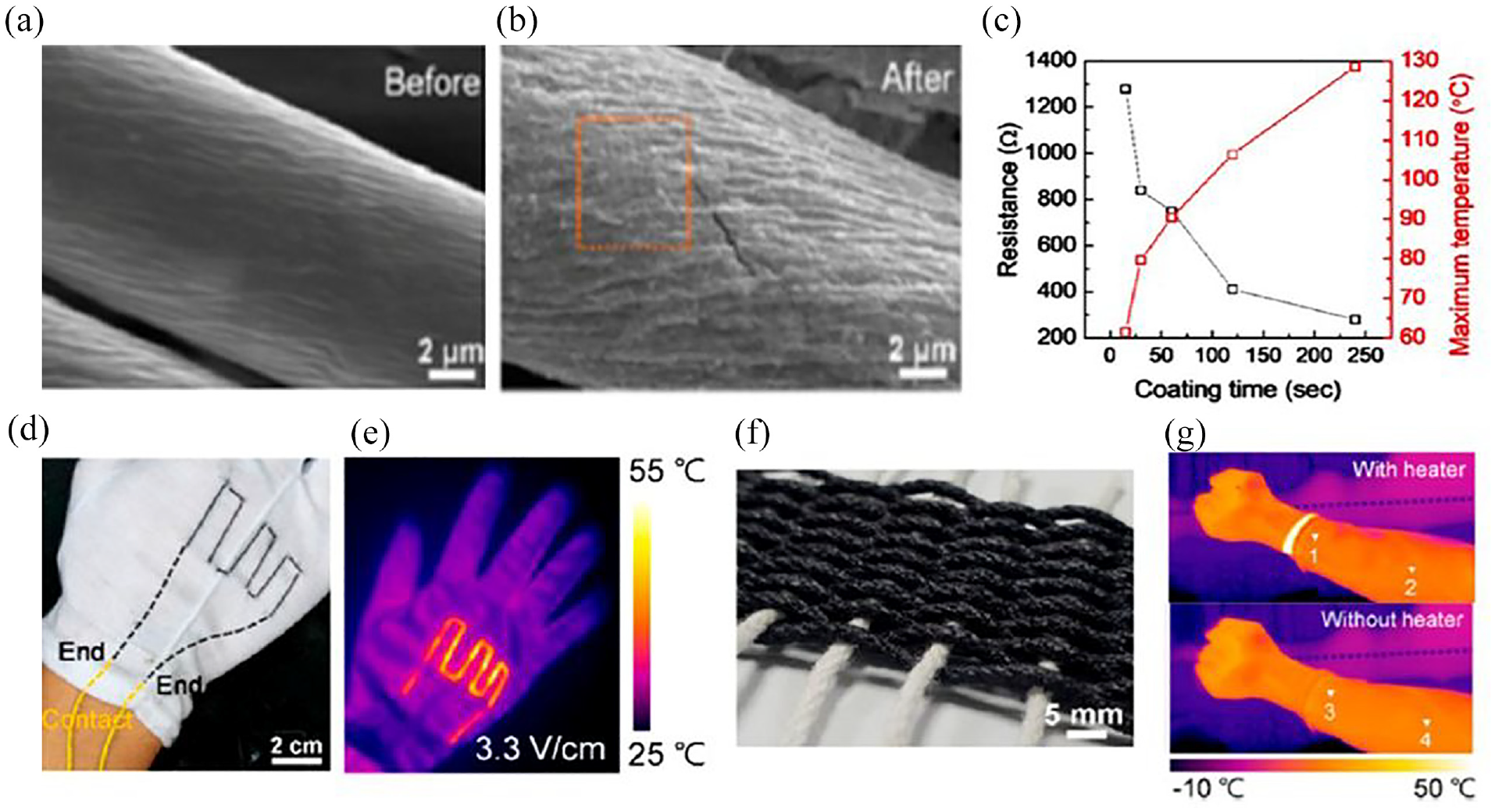
MXene coated polyester textiles for heat generation application: (a) SEM image of uncoated polyester; (b) MXene coated polyester; (c) curves showing temperature and resistance as a function of coating time for the MXene thread heater; (d) image of a cotton glove that was sewn using the MXene thread heater and its (e) thermal infrared pictures at the appropriate voltages applied; (f) images of the MXene fabric heater that is woven; and (g) thermal images of the arm-worn MXene fabric heater heated to a freezing temperature. Reprinted with permission from ref. 95
Conclusion and Future Perspectives
In the last five years, energy storage application of textile substrates has been a thriving research area. In order to materialize the real-life applications of the textile energy storage devices, researchers have so far put up a variety of designs. The design of textile energy storage devices must first be adjusted to match the anticipated energy storage capacity. It is believed that supercapacitors are the secret to creating fast charging energy-storage systems, providing intermediate specific energy, and bridging the energy storage gap between batteries and capacitors. Supercapacitors have much longer life cycles and can react quickly during both the charging and discharging processes due to their highly reversible charge-storage process. This sparks a lot of interest in its application in various consumer electronics, commercial power management systems, and hybrid vehicles that run on both conventional and electric fuels. MXene’s high conductivity (up to 9880 S cm−1), good hydrophilicity, and superior ion intercalation behavior have made it a very promising material for electrochemical energy storage. By adding the right ingredients to make nanocomposites, its storage capacity could be raised. The storage capacity is affected by various factors such as conductivity, specific capacitance, cyclability, capacitive retention, etc. Although there are two ways to synthesize MXenes: top-down (from the MAX phase) and bottom-up (from M and C atoms), the latter method is more frequently used. MXene sysnthesis from its MAX phase by electro-chemical etching is more sustainable because of its environment friendly, safe, and energy-efficient synthesis method. Other acid, salt and alakali etching processes are chemical intensive and require harsh conditions. MXene-loaded fibers or filaments can be prepared using electrospinning and wet spinning techniques, and MXene flakes can be surface-deposited on textile yarn and fabric surfaces using the coating process. SEM images reveal the manufacture of MXene loaded filaments of uniform diameter by electrospinning, and with an irregular serrated shape by solution spinning technique. The deposition of MXene flakes over textile substrates is found to be uniform. The adhesion of MXene on textile surface can be improved by various approaches like use of PEDOT: PSS like a binder, oxygen plasma treatment, silane treatment, etc.
MXene can be coupled with other conductive materials like PPy, PEDOT:PSS, CNTs etc. to enhance energy storage capacity. The mechanical and electrical properties of the textiles are found to increase remarkably after MXene loading. About a 90% increment in tensile strength of textile fabrics is observed after MXene coating. MXene fibers spun through the wet-spinning process have an electrical conductivity of 7748 Scm−1. Only 6% loading of MXene on cotton fabric reduces its electrical resistivity to as low as 5 Ω sq−1. These observations show that highly electroconductive smart fabrics can be developed using even a small amount of MXene.
Wearability is an important factor to take into account when designing any textile energy storage device. The standards for wearability of textile energy storage devices have not yet been clearly established. Some fundamental factors to take into account are mechanical strength, flexibility, abrasion resistance, flame resistance, stretchability, moisture penetrability, toxicity, fatigue lifetime, thermal conductivity, mass density, and wettability. 124 It is observed that little study has been done on the fatigue lifetime of textile energy storage devices when worn on a daily basis. To produce completely wearable textile energy storage systems, substantial research will be needed. 125 It is more likely that textile energy storage devices that cannot be washed will be created initially. Because washing conditions are difficult mechanically and chemically, washing textiles poses a number of technological problems.
In recent years, textile energy storage devices have now added more features to them in addition to energy storage. These functionalities include stretchability,126–130 and color tenability.131,132 These extra features could aid in the development of new markets and applications for textile energy storage devices. It is important to keep in mind that the “primary function” of a textile energy storage device continues to be energy storage. 133 The added functionality shouldn’t substantially or dramatically reduce the gravimetric or volumetric capacity as well as the power capability. This review article gives a critical guideline of how new research studies need to be conducted to determine the correct balance between the energy storage functionality and the other such features of wearable textiles. There are still many fascinating challenges ahead of us that we have identified before we can fully realize the diverse range of uses for textile energy storage technologies.
Footnotes
Credit Authorship Contribution Statement
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author [Subhankar Maity], upon reasonable request.