Abstract
In this study, the characteristic properties and biological activities of the electrospun nanofibers made of polymers including chitosan (CS), polyethylene glycol (PEG), and polylactic acid (PLA) loaded with 15% of a natural anti-inflammatory and analgesic agent, ethyl
Introduction
Electrospinning is a simple, low-cost, and effective technique for fabricating fibers with diameters ranging from micrometers to nanometers using a high-voltage power supply. 1 First introduced in 1900, 2 the electrospinning method has been developed and has undergone many revolutions over the past two decades, 3 from the single-fluid blending process4–6 to bi-fluid coaxial electrospinning,7,8 side-by-side electrospinning, 9 multiple-fluid triaxial, 10 tri-layer Janus,11,12 and their combinations for producing new types of nanofibers.13,14 One merit of those complex nanostructures is the ability to tailor the multiple components for a certain joint effect. However, traditional single-fluid electrospinning can also be easilty encapsulated with multiple components under reasonable selections of polymeric matrices, active ingredients and additives, and holds advantages for scale-up productions. Certainly, these multiple-component nanofibers can also be designed into multi-chamber nanofibers in the future. 15
Electrospun polymer nanofibers have been widely applied in various fields including drug delivery systems,16–19 tissue engineering,20–22 biosensors,23–26 wound dressings,27–30 filtration,31,32 and environmental engineering.33,34 One of the most important and extensively studied areas of electrospun polymer nanofibers is drug delivery. 33 A wide variety of drugs such as anti-inflammatory (ibuprofen, 34 naproxen, 35 indomethacin, griseofulvin, 36 meloxicam, 37 ketoprofen 38 ), anti-microbial (amoxicillin, 39 ciprofloxacin, 40 and tetracycline hydrochloride 41 ), anti-cancer (doxorubicin 42 and cisplatin 43 ), and anti-histamine (chlorpheniramine maleate 44 ) drugs have been loaded into the electrospun nanofibers to improve their bioavailability or to achieve controlled release.
Numerous organic polymers, including both natural and synthetic polymers, have been successfully explored for solution electrospinning to directly produce nanofibers.
45
Chitosan (CS), polylactic acid (PLA), and polyethylene glycol (PEG) are popular polymers that have been used for the fabrication of electrospun nanofibers. Chitosan is a natural polymer with biodegradable, biocompatible, anti-bacterial and anti-tumor effects, wound healing properties, and nontoxicity. The cationic nature of chitosan due to the presence of amino groups causes its characteristic properties and controlled drug release. Therefore, chitosan is currently being utilized in targeted drug delivery methods for the treatment of diverse diseases, including cancer, Crohn’s disease, Alzheimer’s disease, and Parkinson’s disease. Some applications of chitosan involve drug delivery systems such as scaffolds, nanoparticles, hydrogels, nanocomposites, and fibers.46,47 However, the process of electrospinning pure chitosan presents significant difficulties and challenges due to various reasons including its restricted solubility in the majority of organic solvents, broad distribution of molecular weights, and three-dimensional networks of hydrogen bonds.4,33 Polylactic acid (PLA) is a polyester obtained from renewable resources such as beet, corn and sugar. Similar to chitosan, PLA is also biodegradable, biocompatible, bio-absorbable and non-toxic. PLA is one of the most promising materials used in the biomedical field, approved by the Food and Drug Administration for use in microcapsules, surgical sutures, microspheres, and implant materials. In contrast to chitosan, PLA has excellent mechanical strength and good fiber-forming ability. Some papers have shown that adding CS to PLA helps reduce the diameter of nanofibers by electrospinning technique.4,33 PEG is a hydrophilic, biocompatible polymer that has been extensively studied for its potential in various biomedical applications.
48
Nowadays, PEG has been widely utilized in drug delivery to enhance the stability and solubility of drugs
Nonsteroidal anti-inflammatory drugs (NSAIDs) are used widely to control inflammation and pain in different acute or chronic pain conditions such as osteoarthritis, rheumatoid arthritis, and other inflammatory diseases.56,57 However, NSAIDs can trigger various adverse effects on the gastrointestinal, hepatic, renal, and cardiovascular systems.56–59 Therefore, natural anti-inflammatory compounds like EPMC have become promising candidates for the treatment of inflammatory diseases due to their safety and lack of no side effects.60,61
To create a new drug delivery system carrying the natural anti-inflammatory, analgesic agent EPMC for the treatment of inflammatory diseases, in the present study, we report the results of investigating the characteristic properties, anti-inflammatory and anti-bacterial effects, and safety of electrospun nanofibers made from polymers including CS, PEG, PLA loaded with 15% of EPMC.
Experimental
Materials
Chitosan (CS, Mw≈ 100 kDa; degree of deacetylation = 99%) was purchased from Sigma-Aldrich (USA). Polylactic acid (PLA, Mw = 200 kDa), Polyethylene glycol (PEG, Mw = 600 Da), acetic acid (≥99.5%), chloroform (≥99.5%), and ethanol (≥99.5%) were purchased from Merck (Germany). EPMC (≥95%) (obtained from
Vero cells originated from the American Type Culture Collection (ATCC no. CCL-81.4), trichloroacetic acid (Alpha Chemika, India), sulforhodamine B (Sigma-Aldrich, USA), tris base solution (Alpha Chemika, India), ellipticine (Sigma-Aldrich, USA), and some other necessary chemicals were used for the cytotoxicity assay.
RAW 264.7 cells provided by Professor Domenico Delfino (Perugia University, Italy), lipopolysaccharides (LPS) from
Bacterial strains including
Healthy white mice, weighing about 22–25 g, regardless of breed, were raised at the Institute of Biotechnology VAST, Vietnam under standard conditions of temperature and light for an acute toxicity assay.
Fabrication of Ethyl p-Methoxycinnamate-Loaded CS/PEG/PLA Nanofibers by Electrospinning
Preparation of Polymer Solutions for Each Electrospinning Batch
CS (0.1g) was completely dissolved in 5 mL of 90% acetic acid solution at 50oC to give CS solution at a concentration of 2% (w/v) (solution 1). PLA (0.75 g) and PEG (0.05 g) were dissolved in chloroform at 40oC to obtain a solution with concentrations of PLA and PEG of 15% and 1% (w/v), respectively (solution 2). A 0.159 g aliquot of EPMC was then added to solution 2 to achieve solution 3. Solution 1 was slowly added to solution 3 with stirring to obtain a homogeneous solution for electrospinning (solution 4).
Electrospinning Process
The horizontal electrospinning process was carried out using an electrospinning machine. Solution 4 was put into a 5 mL syringe attached to a metal needle that was connected to a high voltage of 20 kV. The feeding rate of the solution was 1 mL/h. The distance from the needle to the drum collector was set at 15 cm. The thickness of the nanofibers was controlled through the electrospinning time. The produced nanofibers containing 15% EPMC were dried by a vacuum drying machine at 50oC for 12 h to completely remove the solvents.
Characterization of the Nanofibers
Morphological Analysis
The morphological characteristics of the electrospun nanofibers were observed using a scanning electron microscope (Hitachi, S-4800). The diameter of nanofibers was determined using image processing software (ImageJ). The average diameter was then calculated randomly from about 100 nanofibers using MS Excel software.
Chemical Analysis
Chemical characterization of the nanofibers was observed using an FT-IR spectrometer (GXPerkinElmer, USA).
Mechanical Analysis
The tensile strength of the nanofibers was measured using a material testing machine Zwick Z2.5 (Germany). Each sample was cut to dimensions of 15 mm in width and 50 mm in length. The sample’s thickness was the mean thickness value at three different positions on the sample measured by a digital micrometer. The test speed was 10 mm/min, and the gauge length was 30 mm. The measurements were repeated three times.
Wetting Study
The wetting properties of the nanofibers were tested by the sessile drop method using optical contact angle equipment Dataphysics OCA 50 (Germany). A small droplet (5 µL) of deionized water was placed on the surface of the nanofibers. The contact angle was measured using a digital camera at 60 s after the droplet touched the nanofiber surface. This measurement was repeated three times.
In Vitro EPMC Release Study
The
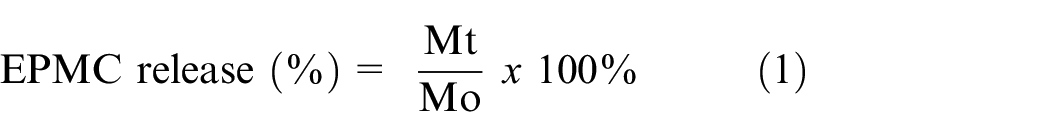
where
Nitrite Assay
Initially, RAW264.7 cells were cultured in DMEM medium supplemented with 10% FBS in 96-well plates with a density of 2 × 105 cells per well. The cells were then incubated in a humidified atmosphere with 5% CO2 at 37°C for 24 h. Subsequently, the medium was replaced with DMEM (free FBS) and incubated for an additional 3 h. The cells were treated with test samples for 2 h, followed by treatment with 1 µg/mL of LPS for 24 h. To assess the production of nitric oxide, the accumulated nitrite in the culture medium was measured using the Griess reaction. In brief, 100 µL of the culture medium was mixed with 100 µL of Griess reagent which consisted of 50 μL of 1% (w/v) sulfanilamide in 5% (v/v) phosphoric acid and 50 μL of 0.1% (w/v)
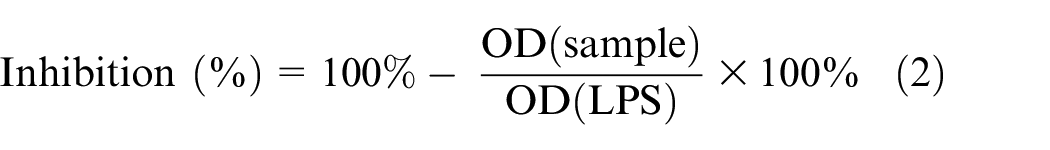
where OD (sample) is the absorbance in wells treated with the test sample and OD (LPS) is the absorbance in wells of LPS-stimulated nitric oxide (NO) production.
The test was repeated three times to ensure accuracy. The IC50 value was determined using TableCurve 2Dv4 software. The DMEM (free FBS) medium was used as a blank, and
Anti-bacterial Assay
The anti-bacterial activity of the nanofibers was conducted on 96-well microplates according to the methods of Vlietinck
63
and Mckane and Kandel.
64
Test bacterial strains included
Cytotoxicity Assay
Vero cells were cultured at 37oC in DMEM medium supplemented with 10% FBS, 100 U/mL penicillin, and 100 µg/mL streptomycin in a 5% CO2 incubator. The
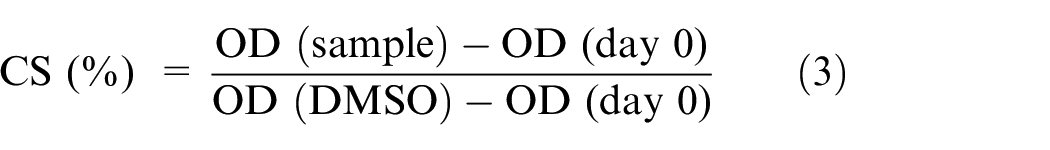
where OD (sample) is the absorbance in wells treated with the test sample, OD (DMSO) is the absorbance in wells treated with DMSO, and OD (day 0) was the absorbance in the control well on day 0.
The cell survival (CS) percentage is presented as the mean ± SD of three replications. Ellipticine, a highly cytotoxic substance, was used as a positive control in the experiment.
Acute Toxicity Assay
Acute toxicity was conducted according to Organisation for Economic Co-operation and Development (OECD) guideline number 420 and the Guidelines for pre-clinical and clinical testing of oriental medicines and medicinal herbs issued by the Vietnam Ministry of Health under Decision No. 141/QD-K2DT dated October 27, 2015.66,67 The care and use of laboratory animals in this study were conducted according to ethical guidelines and approval from the Scientific Council of the Institute of Biotechnology, VAST for animal studies dated December 4, 2023. The nanofibers were ground into powder as the sample for the acute toxicity test.
Preliminary test: 10 white mice were completely starved for 16 h before receiving the sample at the highest dose of 10,000 mg/kg body weight. In the case where no mice died within 24 h of observation and monitoring, the official test would be immediately conducted according to the fixed-dose procedure.
Official test: 20 white mice were divided into two groups (10 mice/group) and completely starved for 16 h before receiving the sample. Group 1 (control group) was treated with distilled water. Group 2 was orally administered the sample at a dose of 5000 mg/kg body weight. Treated mice were observed continuously for the first 2 h. Their food and water consumption, body weight, death, and reaction to light and sound were monitored and recorded for 14 days.
Statistical Analysis
The data were processed by Excel software. The results are presented as mean ± SD (standard deviation). A
Results and Discussion
Characterization of the Nanofibers Loaded With Ethyl p-Methoxycinnamate
The nanofibers made from three polymers, CS, PEG, and PLA loaded with 15% EPMC (w/w), were produced by the electrospinning method. The selected content of EPMC in the nanofibers was the highest content at which the electrospun nanofiber formation process went smoothly. At contents of EPMC more than 15%, the produced nanofibers had beads and the fiber formation process was interrupted because the injector of the electrospinning machine was clogged. Figure 1 shows that the surface of the nanofibers loaded with 15% EPMC was smooth and uniform, without any beads. The average diameter of the nanofibers was 224 ± 66 nm.

SEM micrographs of the nanofibers at different magnifications: (a) ×10.000 and (b) ×500.
Figure 2 presents the FT-IR spectra of CS, PEG, PLA, andEPMC, and the nanofibers. The FT-IR spectrum of EPMC shows some typical absorption peaks at 1700 cm−1 (C=O), 1167 cm−1 (C–O), 2978–2841 cm−1 (C–H), and 1627–1508 cm−1 (aromatic C=C). The absorption at 825 cm−1 suggests the presence of para-substitution in the EPMC molecules. The FT-IR spectrum of CS reveals the strong broad absorption band around 3361 cm−1 corresponding to N–H and O–H stretching vibrations, the peak at 2869 cm−1 assigned to C–H stretching vibrations, and the strong doublet absorption peak at 1023 and 1066 cm−1 attributed to the pyranose structure. 68 The FT-IR spectrum of PEG exhibited two sharp peaks at 1089 cm−1 (C–O) and 2878 cm−1 (C–H). The FT-IR spectrum of PLA displayed characteristic stretching frequencies for C=O, C–H, and C–O at 1747, 2994, 2944, and 1080 cm−1, respectively. All the above peaks of the components were observed in the FT-IR spectrum of the nanofibers. This suggested the presence of EPMC, CS, PEG, and PLA in the nanofibers. However, there were certain shifts of these peaks on the spectrum of the nanofibers, which suggested interactions among these constituents in the nanofibers. The two strongest shifted peaks observed in the spectrum of the nanofibers were 1752 and 1085 cm−1 corresponding to the C=O and C–O bonds.
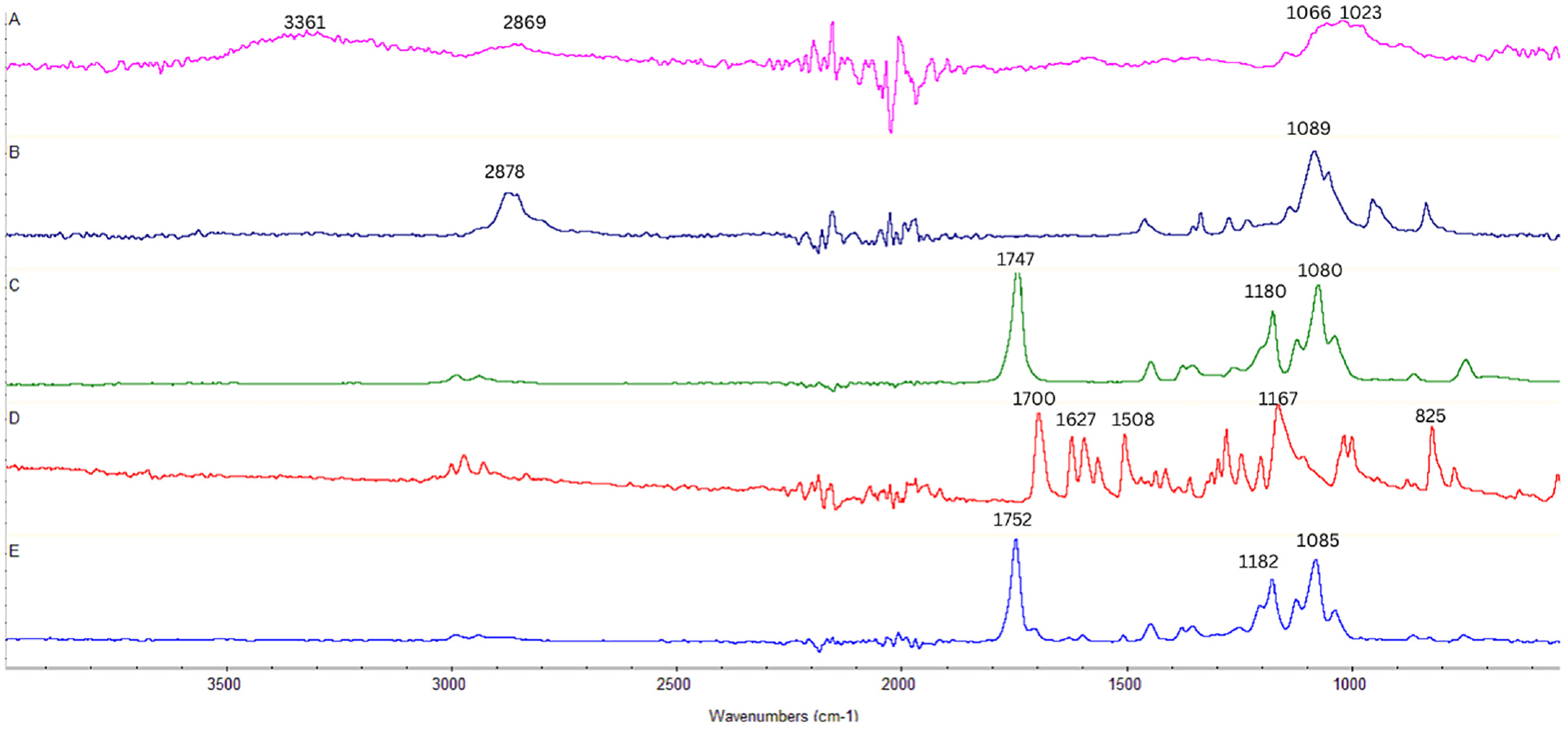
FT-IR spectra of CS (A), PEG (B) PLA (C), EPMC (D), and the nanofibers (E).
Mechanical analysis of the nanofibers (thickness of 120 ± 4.6 µm) exhibited a tensile strength of 2.07 ± 0.56 MPa, an elongation at break of 13.55 ± 2.27%, and a Young’s modulus of 106.31 ± 24.02 MPa. The stress–strain diagram of the nanofibers (Figure 3) describes two stages of the tensile process. A is considered the moment at which the bonds between fibers begin to break, whereas B is the point at which the nanofiber mat breaks completely. 69 Tensile strength is believed to be proportional to the thickness of the material. 70 Hence, it is possible to improve the mechanical properties of the nanofibers by increasing the thickness of the produced nanofibers.
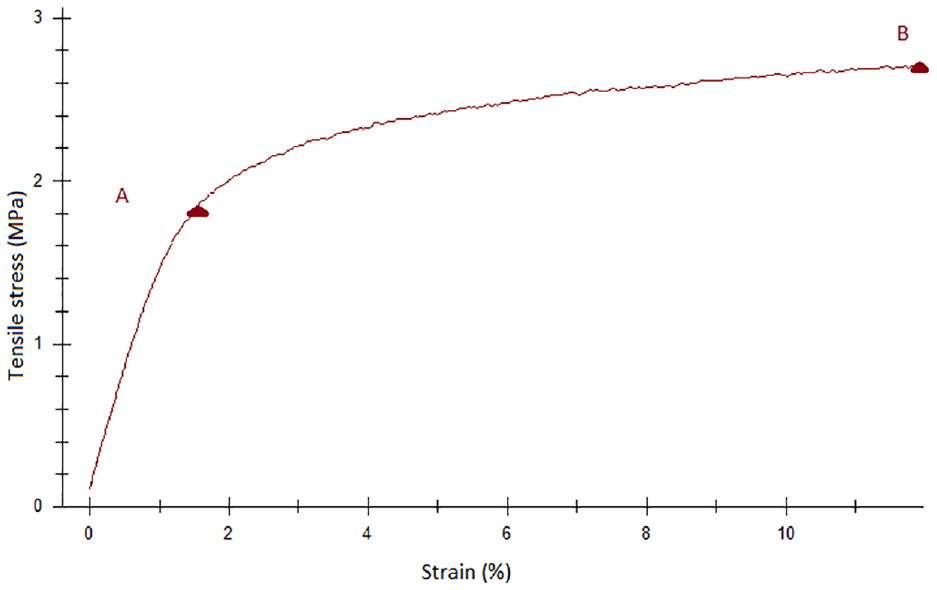
The stress–strain diagram of the nanofibers. A: the Yield point, B: the Breaking point.
The significance of wettability in enhancing the release of active ingredients and the dissolution of pharmaceuticals is widely acknowledged. 71 The water contact angle was used to evaluate the hydrophilicity of the nanofibers. The nanofibers were found to be hydrophilic with a contact angle of 73.1 ± 0.8o.
In Vitro Drug Release Study
The medium used for studying EPMC release from the nanofibers was PBS (pH 7.4), with the same pH value as the terminal ileum and plasma.
17
Three mechanisms can be used to describe drug release from nanofibers. These mechanisms include desorption from the surface of the fibers, solid-state diffusion through the fibers, and degradation of the fibers
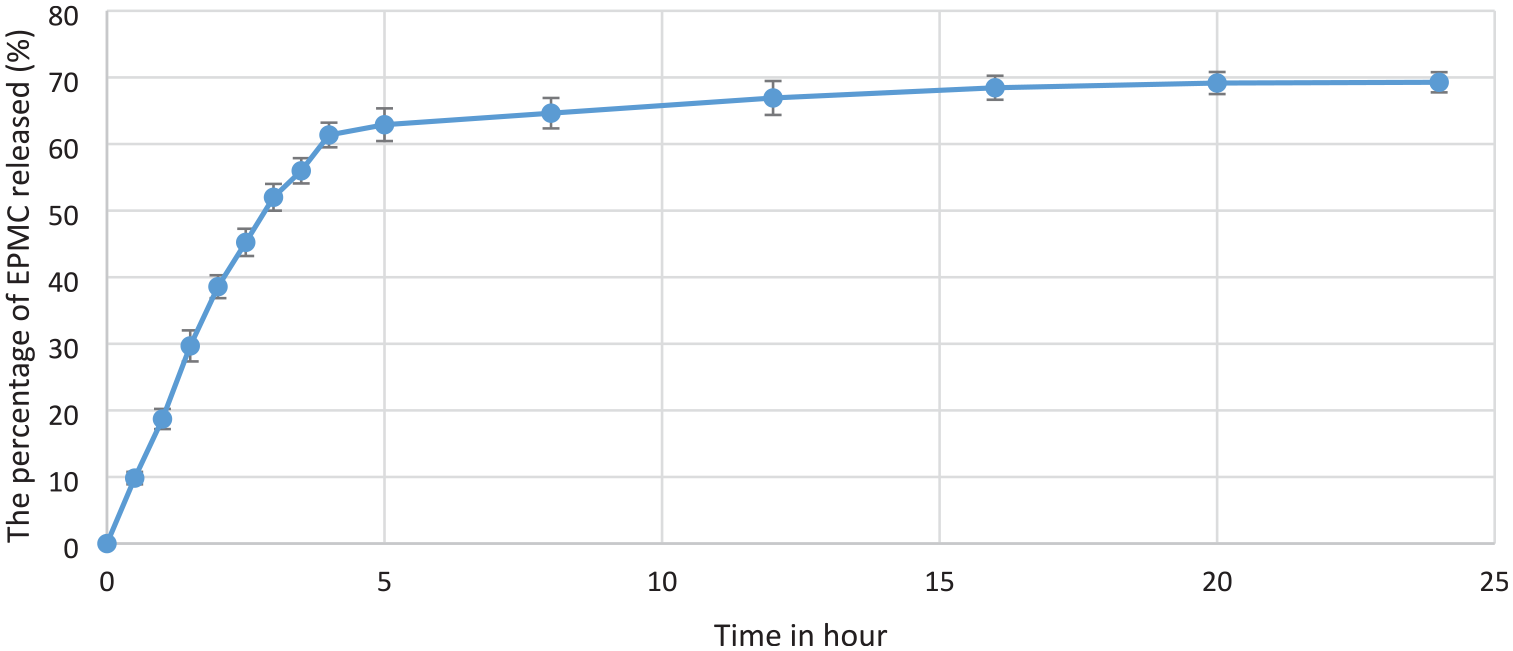
EPMC release from the nanofibers.
Anti-Inflammatory Effect
Nitric oxide (NO) is a crucial signaling molecule involved in the pathogenesis of inflammation. Under normal physiological conditions, NO can give anti-inflammatory effects. However, it is also an inflammatory mediator during inflammation. Overproduction of NO as an inflammatory mediator can lead to tissue destruction. Therefore, inhibitors of NO production are considered one of the important targets in the control of inflammatory diseases. 78 In the current study, we evaluated the anti-inflammatory properties of the nanofibers loaded with EPMC via the inhibition of NO production in LPS-stimulated RAW264.7 cells. The results (Table 1) showed that the nanofibers exhibited significant inhibitory activity on NO production with an IC50 value of 41.05 ± 1.75 µg/mL and did not affect the viability of RAW264.7 cells at test concentrations (cell survival rate > 80%). On the other hand, the inhibitory effect on NO production in human macrophage cells (U937) of free EPMC was previously reported to be 23.8% at a concentration of 200 µg/mL. 55
Inhibition of NO production of the nanofibers in LPS-stimulated RAW264.7 cells.
Anti-bacterial Effect
Natural products with MIC values below 1 mg/mL are considered to have noteworthy anti-bacterial activity.
79
The results (Table 2) showed that the nanofibers exhibited anti-bacterial effect against four types of microorganisms:
Anti-microbial effects of the nanofibers.
Note: (–): the nanofibers did not show inhibitory ability against microbial strains at the tested concentrations.
Cytotoxicity Study
Vero cells obtained from African green monkey kidneys are similar to human cells and can be easily cultured.
80
The Vero cell line is frequently utilized as a normal cell line control for assessing the
The results of cell viability assay.

Acute Toxicity
In the acute study, oral administration of the nanofibers sample at a dose of 5000 mg/kg body weight did not cause any death or signs of toxicity in mice during the experimental period of 14 days. After being treated with the sample, experimental animals still moved and ate normally and responded to light and sound well. There was no significant difference in body weight between the control group and the experimental group (
Conclusion
In the current article, the EPMC (15%, w/w) loaded CS/PEG/PLA nanofibers were fabricated with an average fiber diameter of about 224 ± 66 nm. The mechanical and physicochemical properties of the fibers were also evaluated. Research on EPMC release in PBS medium showed that after 24 h, about 70% of EPMC was released from the nanofibers. The nanofibers revealed significant anti-inflammatory activity via the inhibition of NO production with an IC50 value of 41.05±1.75 µg/mL, along with anti-bacterial activities. In addition, the nanofibers loaded with EPMC were proved to be safe via both
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was performed under the financial support of Vietnam–Czech Republic joint research project (grant no. NĐT/CZ/23/07).
