Abstract
In this paper, modified SiO2 nanoparticles were selected to improve the surface roughness and decrease the surface energy to improve the hydrophobic performance of cotton fabric. Tetraethoxysilane and octadecyltriethoxysilane were used as raw materials to synthesize octadecyltriethoxysilane-modified SiO2 nanoparticles (siloxane-SiO2), and combined with cotton fabric. The chemical composition and surface morphology of the fabric were measured. The contact angles of the modified cotton fabrics prepared under different reaction conditions were approaching or above 150°. In addition, dynamic evolutions of water droplets on the modified cotton fabric surface were discussed. The adhesive force and the energy conversion of droplet during the collision were investigated. The water droplet bounced due to the superhydrophobic properties of the cotton fabric surface producing enough residual energy. Finally, through the acid–base chemical resistance treatments, the rubbing and washing durability, and anti-fouling experiment, the modified fabric indicated excellent hydrophobic durability and self-cleaning performance.
Introduction
Cotton fabric with comfort, soft and breathable properties is widely used in our daily life. Due to the hydrophilic property of the cotton fabric,1–4 it is susceptible to contamination by water, oil, and dirt.5,6 Cotton fabric was modified to have hydrophobic properties by mist polymerization, ultrasound irradiation, and graft technique.1,7,8 The hydrophobic cotton fabric had anti-fouling, anti-stick and self-cleaning properties, which could be used in anti-fouling sportswear, military clothing, and medical textiles.9–12
Inspired by the lotus leaf, the hydrophobic properties were increased when the surface was modified with low surface energy and constructed rough structures.13–16 The cotton fabric had a microscale rough structure surface. The hydrophobic cotton fabric was obtained when the nanoscale structure and low surface energy layers were modified on its surface.17–19 Hou et al. 20 used acetone and water as solvents to prepare micro/nano scale particles of polytrifluoropropylmethylsiloxane in solution by a phase separation method; then they were arranged on fabric surface to prepare 2D and 3D porous structures. The results showed that the fabric had excellent superhydrophobicity, chemical resistance and mechanical wear resistance and high temperature resistance. Deng et al. 21 prepared octavinyl-polyhedral oligomeric silsesquioxane onto the cotton fabric surface by a one-step dipping method. The water contact angle on the modified cotton fabric was higher than 142°. Moreover, it maintained excellent stability and effective separation properties in high concentrations of acid, alkali, and salt solutions.
In previous studies, hydrophobic textiles were successfully modified by nanoparticles, especially inorganic nanoparticles comprising SiO2, ZnO, Ag and TiO2, combined with a low surface energy material such as siloxane.22–26 In particular, SiO2 had a typical Si-O-Si skeleton structure, which provided an excellent three-dimensional rough structure.27–29 As a result, it was often used as inorganic nanoparticles to increase the surface roughness.30,31 Tetraethoxysilane (TEOS) was hydrolyzed and condensed to form SiO2 with ammonium hydroxide as the catalyst. The effect of ammonium hydroxide concentration was an important influence factor due to it influencing the polycondensation rate. On the contrary, the siloxane tended to concentrate on the material surface and effectively reduced the surface energy. The low surface energy gave excellent surface hydrophobic properties. For example, octadecyltriethoxysilane (ODTES) was a low surface energy material. ODTES had three ethoxyl groups which could convert to hydroxyl groups. Polycondensation occurred when the TEOS and ODTES were reacted, and monodisperse spherical colloids of siloxane-SiO2 were obtained.
The water contact angle is an effective way to identify the hydrophobic properties of the surface. The water droplet may be trapped in the concave structure on soft surfaces such as silk and cotton fabric which could make the contact angle unobtainable. 32 As a result, the dynamic hydrophobic behavior of water droplets could solve this problem and characterize the hydrophobic properties. The impact droplets exhibited spreading, receding, or bouncing due to the hydrophobic properties of the surface, the radius of the droplets and the descent height.33,34 The morphology variation, spreading diameter, energy conversion, and adhesion of the droplet impact behaviors were tested to characterize the hydrophobic properties.
In this paper, TEOS and ODTES were used as raw materials to synthesize siloxane-SiO2. The cotton fabric was modified by the siloxane-SiO2. The surface morphology and chemical composition of cotton fabric were analyzed through scanning electron microscopy (SEM), Fourier transform infrared spectroscopy (FTIR), energy dispersive spectroscopy (EDS), and X-ray photoelectron spectroscopy (XPS). The water contact angle and water impact behavior on the modified fabric surface were investigated. The modified cotton fabric treated with strong acid, strong alkali, the rubbing and washing resistance, and the hydrophobic durability of the fabric are discussed. In addition, the self-cleaning performance of the hydrophobic fabric was analyzed. This inexpensive and fluorine-free technique was easily adaptable by industry for large-scale deployment. On the contrary, this facile synthesis method had the advantages of scalable fabrication of multifunctional fabrics for in water–oil separation and self-cleaning applications.
Experimental Section
Materials
Cotton fabrics (15 × 15 tex, 524 × 284/10 cm) were obtained from Zhejiang Guandong Textile Co., Ltd. TEOS (>99%) and n-ODTES, (>85.0%) were purchased from Aladdin biochemical technology Co., Ltd. Ammonium hydroxide (NH3), hydrochloric acid (37%), ethanol and sodium hydroxide were purchased from Sinopharm Chemical Reagent Co., Ltd. MEGAFIX® red B-2BF was supplied by Shanghai Matex Chemicals Co., Ltd.
Preparation of Hydrophobic Cotton Fabrics
The siloxane-SiO2 and hydrophobic cotton fabric were prepared through a modified Stöber method.7,35 The effect of ammonium hydroxide concentration is to promote hydrolysis, but also to promote the polycondensation rate, resulting in faster kinetics, and lager particle sizes. 36 A certain volume (6, 9, 12, 15, 18 mL) of ammonium hydroxide, 10 mL of water, and 16 mL of ethanol were put into a 100 mL flask. The thermostatic magnetic stirrer was stirred quickly while raising the temperature to 60°C. Then 2 mL ODTES and 1 mL TEOS were added to the solution. The stirring rate was at a speed of 1000 r/min for 1 min, and then was adjusted to 500 r/min for 3 h. Then the cotton fabric was added to the solution for 1 h. After that the cotton fabric was taken out, washed and dried. Finally, the solution was centrifuged (10,000 r/min). The centrifuged product was washed with ethanol and dried to obtain siloxane-SiO2 nanoparticles. The preparation of and modified cotton fabric is demonstrated in Figure 1. Unless otherwise stated, the modified cotton fabric was modified by siloxane-SiO2 which was prepared with 15 mL of ammonium hydroxide.
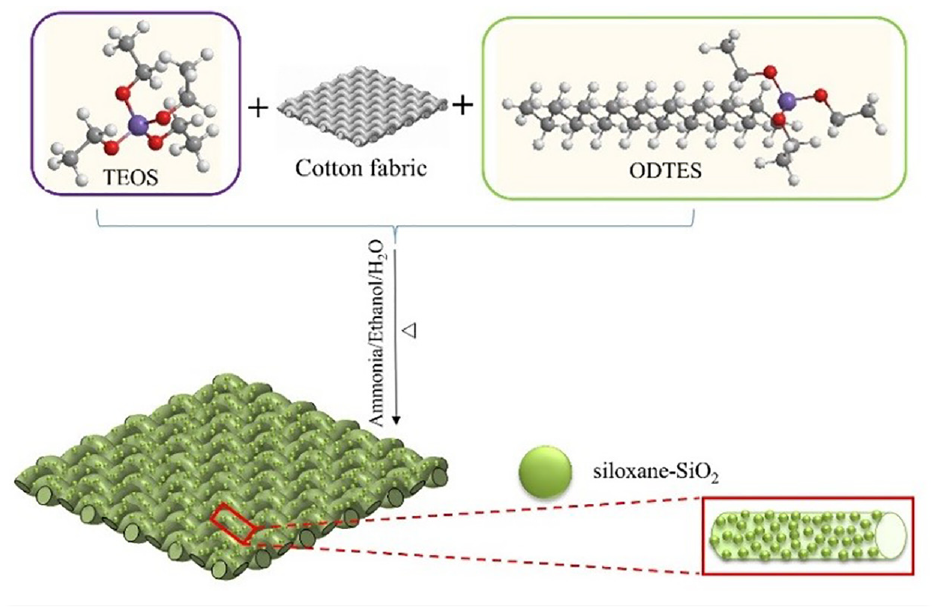
The preparation of the siloxane-SiO2 modified cotton fabric.
Dynamic Behavior and Water Contact Angle of Water Droplet on Cotton Fabric
A water droplet of about 10 μL was dropped freely onto the fabric surface from a height of 40 mm. The morphological changes of the water droplet were recorded by a high-speed camera (American Trouble Shooter HR, 10,000 fps), and the image was processed by MaxTRAQ to obtain the D(t)/D0 and H(t)/D0 values with time, where D(t) is the change value of the diameter of the droplet contact surface with time after the impact, D0 is the initial diameter of the droplet, and H(t) is the height value that changes with time after the impact.
The contact angle of the fabric was tested by the pendent drop method (PT-602A, Dong Guan Precise Test Equipment Co., Ltd), and the average of five tests was taken.
Chemical Durability of Cotton Fabric
After the hydrophobic cotton fabric was immersed in a solution of pH = 1 or pH = 13 (pH = 1 and pH = 13 were adjusted by hydrochloric acid or sodium hydroxide) for 24 h, the contact angle was tested to evaluate the chemical durability.
Rubbing Resistance of Cotton Fabric
The rubbing resistance of the fabrics was evaluated by a modified procedure based on the AATCC Test Method 8-2001 as reported previously. 37 A pristine cotton fabric was used as the rubbing partner and the modified cotton fabric was fixed onto the stainless steel column. Then, the sample was subjected to a roundtrip with a distance of 200 mm, which was defined as one cycle. After 100, 200, and 300 cycles, the water contact angle was tested.
Wash Durability of Cotton Fabric
Wash durability of the modified cotton fabrics was evaluated by AATCC Test Method 61-1996. Stainless steel canisters containing 0.15% AATCC detergent water solution (150 mL) and 50 stainless steel balls were fixed in a Launder-Ometer (Darong Textile Instrument Co., Ltd., Zhejiang, China) and rotated at 42 r/min and 49 min for 45 min. Each cycle of washing was equivalent to five machine washings in this test. The modified cotton swatches were washed for equivalents of 5, 10, 15, and 30 washing cycles. The water contact angle was tested after 5, 10, 15, and 30 washing cycles.
The Self-Cleaning Performance of the Cotton Fabric
A small amount of dye were placed on the fabric and then the water droplet was added to the dye. After 1 min, the droplet was removed to observe the surface state of the fabric, and the self-cleaning performance of the fabric was characterized.
Characterization
The surface chemical groups were tested by FTIR (Thermo Is5, Nicolet Instrument Corporation, USA) and EDS. The surface morphology of fabric was observed through SEM (HITACHI, SU-1510, Japan). The chemical elements of fabrics were characterized by X-ray photoelectron spectroscopy (XPS) (Kratos, England). The morphology of siloxane-SiO2 was observed by transmission electron microscopy (TEM) (JEM-2100PLUS, Japan).
Results and Discussion
Surface Morphology of Hydrophobic Cotton Fabric
The SEM images of cotton fabric and modified cotton fabric are shown in Figure 2.

The SEM images of the cotton fabrics: (a) the cotton fabric, (b) the modified cotton fabric with 12 mL of NH3, (c) the modified cotton fabric with 15 mL NH3, and (d) the modified cotton fabric with 18 mL of NH3.
As exhibited in Figure 2(a), the cotton fabric surface was not coated with any substance and presented a smooth state. However, the modified fabric surfaces were discovered a layer of substance in Figure 2(b)–2(d). 7 TEOS was hydrolyzed and condensed to form SiO2. On the contrary, ODTES was subsequently hydrolyzed and co-condensed on the SiO2 surface to form siloxane-SiO2. 22 Then the cotton fabric was added into the solution which was treated with siloxane-SiO2.
When the volume of ammonia was 12 mL, siloxane-SiO2 was distributed on the fabric surface to form a layer, and the surface was relatively rough, as shown in Figure 2(b). When the ammonia was 15 mL, the uneven distribution of siloxane-SiO2 on the fabric surface formed larger particles and the surface was uneven, as shown in Figure 2(c). When the ammonia was 18 mL, the distribution of siloxane-SiO2 on the fiber surface was not uniform and formed a network structure, and the surface was relatively rough, as shown in Figure 2(d).
The TEM image of siloxane-SiO2 nanoparticles is shown in Figure 3.

TEM image of siloxane-SiO2.
The siloxane-SiO2 comprised micro-nano regular spheres and had good dispersion properties. It was proved that siloxane-SiO2 was synthesized in the modified process. Meanwhile, the fabric surface was coated with a layer of siloxane-SiO2. From the perspective of the morphological structure, it was important to improve the surface roughness.
Chemical Properties of Modified Cotton Fabric
The surface chemical groups and surface elements of the pristine cotton fabric and modified cotton fabric were characterized through FTIR, EDS, and XPS. The FTIR spectra are demonstrated in Figure 4.
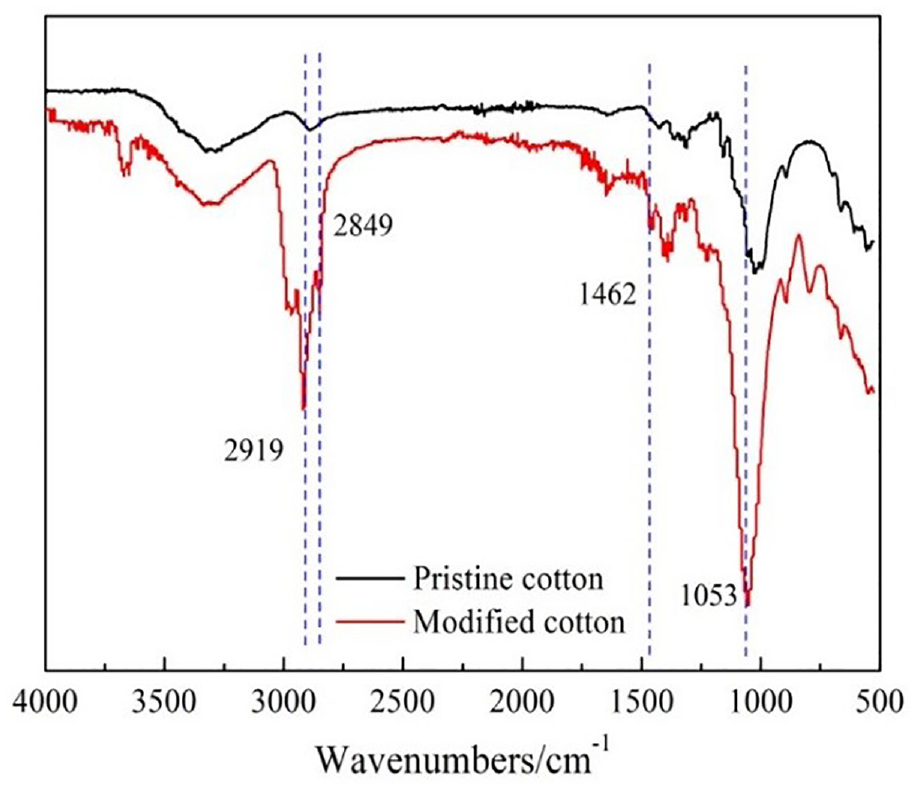
The FTIR spectra of the pristine cotton fabric and modified cotton fabric.
As shown in Figure 4, compared with pristine fabric, the modified fabric had a bending vibration peak of–CH3 at 1462 cm–1 and a stretching vibration peak of Si-O-Si at 1053 cm–1. The –CH3 and –CH2 peaks at 2849 cm–1 and 2919 cm–1 were from ODTES, which proved that siloxane-SiO2 had been successfully synthesized on the cotton fabric.
The EDS surface element analysis is displayed in Figure 5. The cotton fabric had two elements of C and O, which was consistent with the chemical composition of the cotton fabric. The weight ratios of C and O are 46.08% and 53.92% in Figure 5(a). The Si elements appear in the modified cotton fabric in Figure 5(b). The weight percentage of Si element was 20.84% which mainly came from ODTES in siloxane-SiO2 and TEOS.
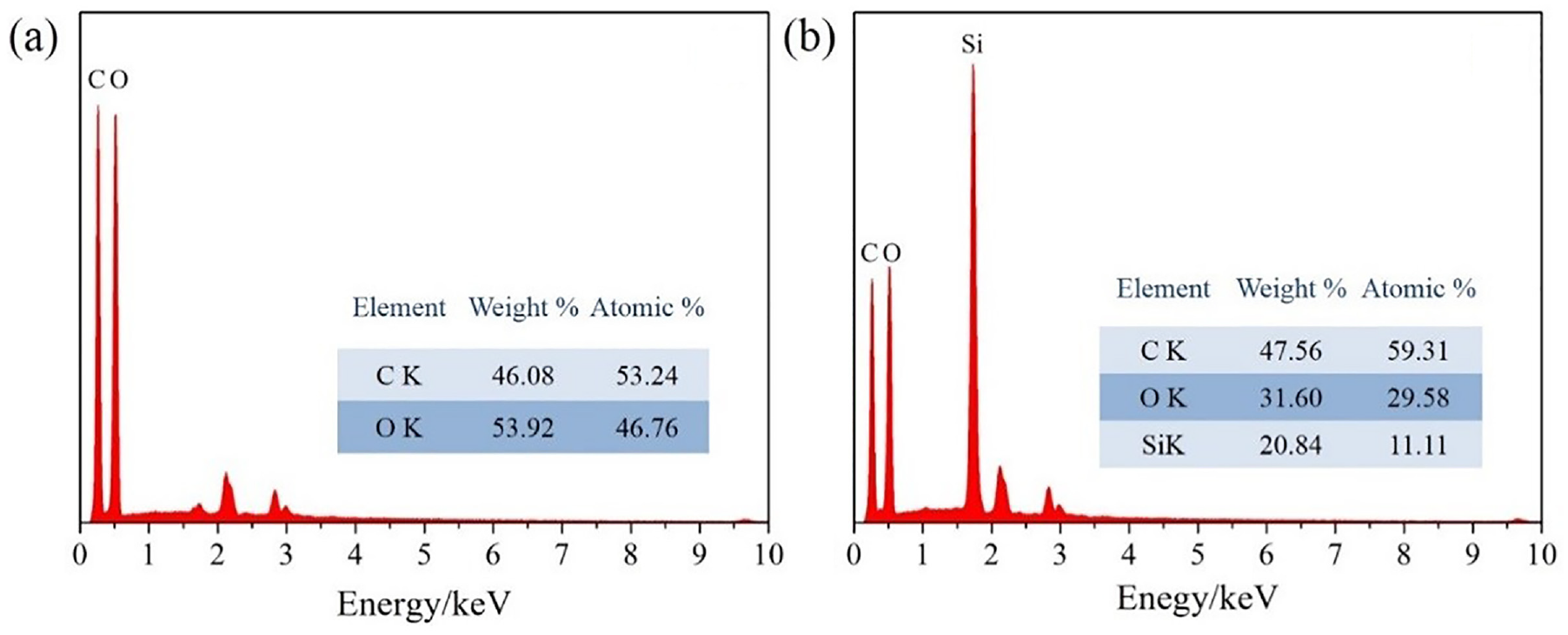
The EDS of; (a) the pristine cotton fabric and (b) the modified cotton fabric.
The point distribution of surface elements is exhibited in Figure 6; the point distribution of C and O elements on the surface of the fabric is relatively uniform. The distribution of C, O, and Si elements on the surface of the modified fabric is also relatively uniform, with an area ratio of 21:22:57%. The Si elements occupied a certain proportion, forming a continuous structure on the fabric surface. The Si element had a certain low surface energy which provided the basis for subsequent research on the hydrophobic properties of cotton fabrics.
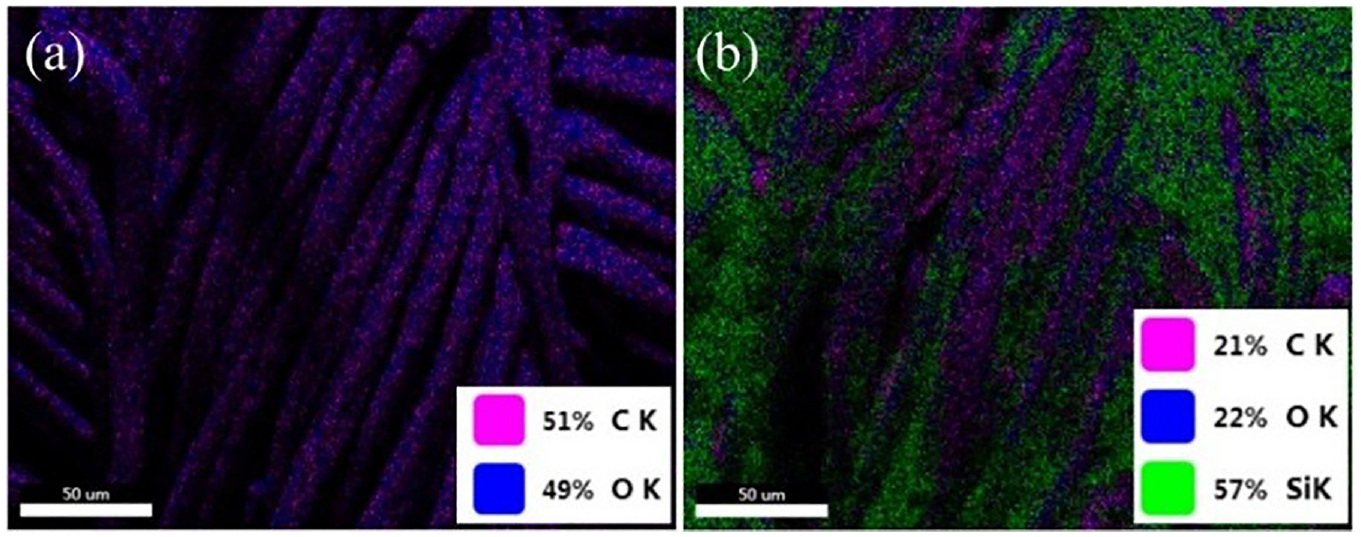
(a) Point distribution of C and O elements on the pristine cotton fabric and (b) point distribution of C, O, and Si elements on the modified cotton fabric.
There are two peaks of O 1s and C 1s on cotton fabric in the XPS spectra in Figure 7.
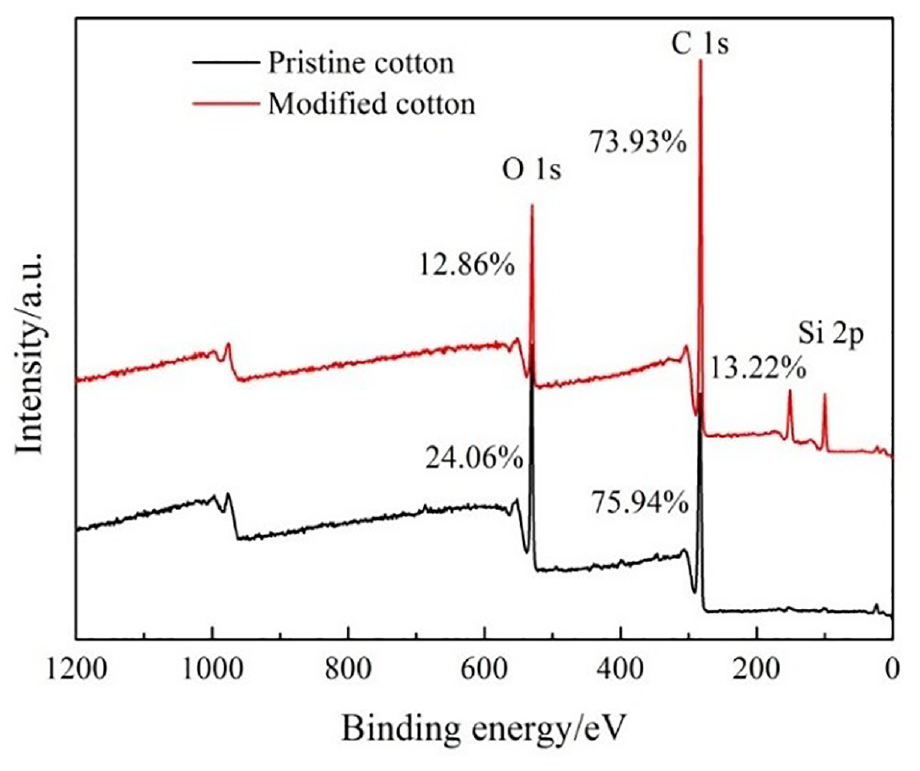
The XPS spectra of the pristine cotton fabric and modified cotton fabric.
A new peak of Si 2p with a content of 13.22% appeared in the modified fabric. The Si content came from the siloxane-SiO2. As a result, siloxane-SiO2 was successfully modified on the fabric surface.
Hydrophobic Properties of the Modified Cotton Fabric
The water contact angles on the modified fabric with siloxane-SiO2 prepared with different volumes of ammonia are exhibited in Figure 8. The dynamic impact performances of water droplets on modified cotton fabrics are demonstrated in Figure 9. The time-varying curves of D(t)/D0 and H(t)/D0 are displayed in Figure 10.
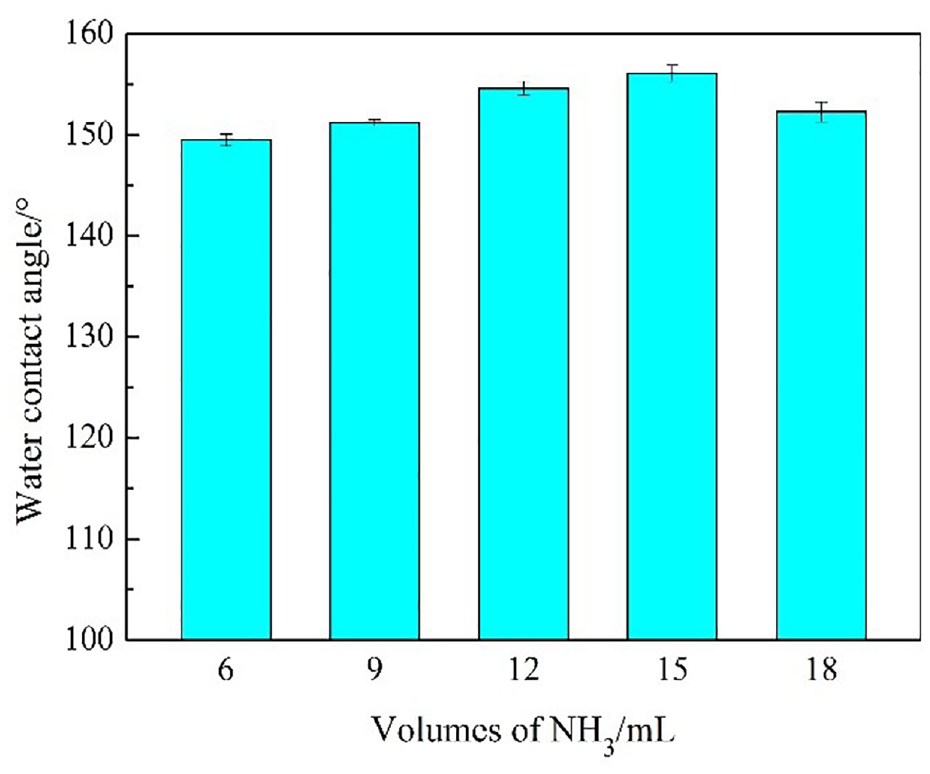
The water contact angles of the modified cotton fabric obtained with different volumes of NH3.

Surface morphology evolution of water droplet hitting modified cotton fabric with different volumes of NH3: (a) 9 mL, (b) 12 mL, and (c) 15 mL.
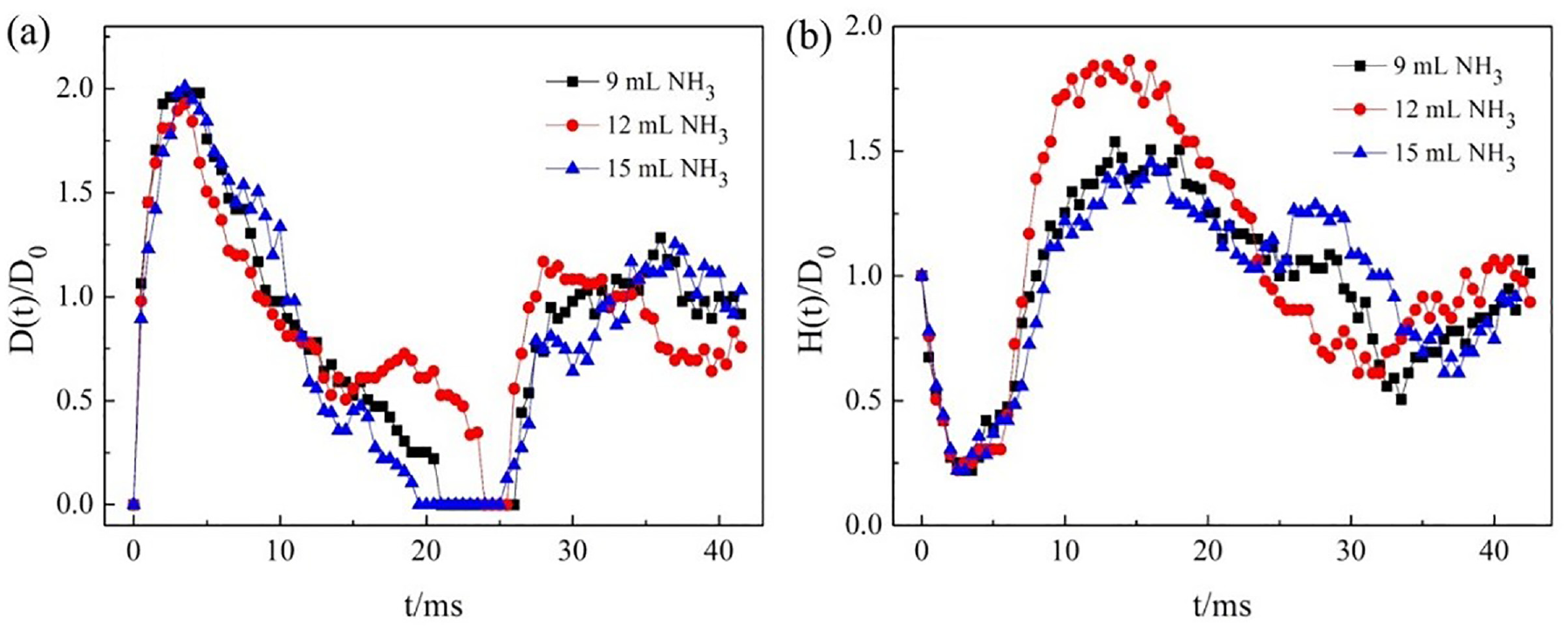
(a) D(t)/D0 values and (b) H(t)/D0 variations as a function of time.
The contact angles of the modified cotton fabrics approached 150° when the volume of NH3 was 6 mL and above 150° when the volumes of NH3 were 9, 12, 15, and 18 mL, achieving superhydrophobic properties. When the volume of NH3 was 15 mL, the water contact angle was 156.1°. The reaction of siloxane-SiO2 on cotton fabric was the rapid hydrolysis and slow condensation process. 38 Under the action of ammonia, TEOS was first hydrolyzed and underwent a condensation reaction to form SiO2 nanoparticles. As a result, the formation of SiO2 was a competitive process of hydrolysis, nucleation, and growth. With the formation of SiO2, Si-OCH3 in ODTES was hydrolyzed to form the silanol structure, which reacted with the hydroxyl groups of SiO2 and the hydroxyl groups of fabrics to form covalent bonds, forming a fastness bond on the fabric surface. As the volume of ammonia was increased, the contact angle showed a trend of increasing and then reducing. With the enhancement of the volume of ammonia to promote the hydrolysis of the TEOS and polycondensation rate, the cotton fabric was combined with more silica particles, and thus the contact angle was increased. When the volume of ammonia reached a certain value, it continued to increase to accelerate the rate of hydrolysis. The nucleation rate was faster than the condensation rate, which led to the formation of more particles. The SiO2 particles were agglomerated on the cotton fabric surface which decreased the surface roughness, so the water contact angle on the cotton fabric surface was slightly decreased.
As exhibited in Figure 9, the impact of droplets on the three modified fabric was showed several behaviors, such as spreading, diffusion, oscillation, retraction and bouncing.39–42 At 4 ms, the droplet was spread on the fabric surface and the contact area was the largest. Then the droplet was retracted to the center under the action of inertia, and the height of the droplet was increased. After the droplet reached the center, it bounced once. In this process, the gravitational potential energy was shifted to kinetic energy. During the collision with the cotton fabric interface, the kinetic energy was transformed to surface energy to cause the droplet to spread and deform. After retraction, the water droplet had enough energy to bounce. 43 The superhydrophobic properties of the modified fabric resulted in a small dissipated energy. Thus the residual energy was higher than the surface adhesion and gravitational potential energy. The droplet rebounded away from the interface and bounced up. Then the water droplet quickly hit the interface a second time, re-tiling, retracting, and reaching the center. Energy consumption also occurred during the second collision. When the water droplet retracted to the center, there was not enough energy to cause a second rebound, but it continued to oscillate until the droplet reached to static state.44,45 The contact angle on the cotton fabric was more than 150°, indicating that the modified cotton fabric was a superhydrophobic surface. Moreover, the fact that the water droplet bounced on the surface also proves the superhydrophobic properties of the fabric.
The bouncing effect can be explained quantitatively through adhesion work. The adhesion work is shown in equation (1):44,46
Here, WSL is adhesion work and γlv is the surface tension. The adhesion was high when the contact angle was low on the surface. During the collision on the fabric, the energy consumption was high due to the diffusion of water droplets. Furthermore, when the contact angle of cotton fabric was higher than 150°, the adhesion work was small, and the adhesion force of water droplets on the hydrophobic cotton fabric surface was small. The residual energy after impact was higher than the surface adhesion force and the gravitational potential energy. The droplet had enough energy to rebound, but it also had a certain energy consumption in the second collision process. At this time, the residual energy was decreased so that there was not enough energy to rebound a second time.
As revealed in Figure 10(a), the D(t)/D0 value on the three sufaces reached the maxmum value at 4 ms, which was identified in Figure 8. Then the D(t)/D0 value was decreased due to the retraction of the water droplet. The droplet did not contact the fabric surface when the water droplet bounced. During this period, the D(t)/D0 value was 0. The duration of D(t)/D0 value of 0 on the three surfaces were 11, 4, and 12 ms, which was related to the energy loss during the contact between the droplet and the interface. In addition, the maximum value of D(t)/D0 at the second impact was less than the maximum value of D(t)/D0 at the first impact, due to the energy dissipation of water droplet impacted on the fabric.
From Figure 10(b), the cotton fabric modified with siloxane-SiO2 prepared by 12 mL ammonia had the highest contact angle and the highest bounce height. Furthermore, it had the greater the energy loss with the shortest rebound time, and the second rebound did not occur. As time went on, the value of H(t)/D0 was decreased gradually due to the loss of energy. 47
The Stability Properties of the Modified Cotton Fabric
The chemical stability, rubbing, and washing durability of the modified cotton fabric (silica nanoparticles prepared from 15 mL of ammonia) were investigated, and the contact angles of the modified cotton fabric treated with acid and alkali are demonstrated in Table 1.
The CA of modified fabric treated with acid and alkali.

The contact angles of the modified fabrics treated with acid and alkali were still maintained at a good level, and the hydrophobic properties were not affected. During the reaction process, Si-OCH3 of ODTES was hydrolyzed into a silanol structure and covalently condensed with hydroxyl groups on the surface of siloxane-SiO2 particles and cotton fabrics, resulting in a three-dimensional network cross-linked structure. In addition, during the modified process, siloxane-SiO2 was relatively uniform in modified cotton fabrics and had covalent bonds.
The water contact angles of the modified cotton fabric surfaces did not show an obvious decrease after 100, 200, and 300 cycles of rubbing as shown in Figure 11(a). The water contact angles on the modified cotton fabric washed for 5, 10, 15, and 30 cycles are revealed in Figure 11(b). With increasing washing up to 15 cycles, the modified cotton fabric had a small decrease in water contact angle. However, after 30 cycles of washing, a decrease of about 15° was observed. Nevertheless, it remained above the 140°.
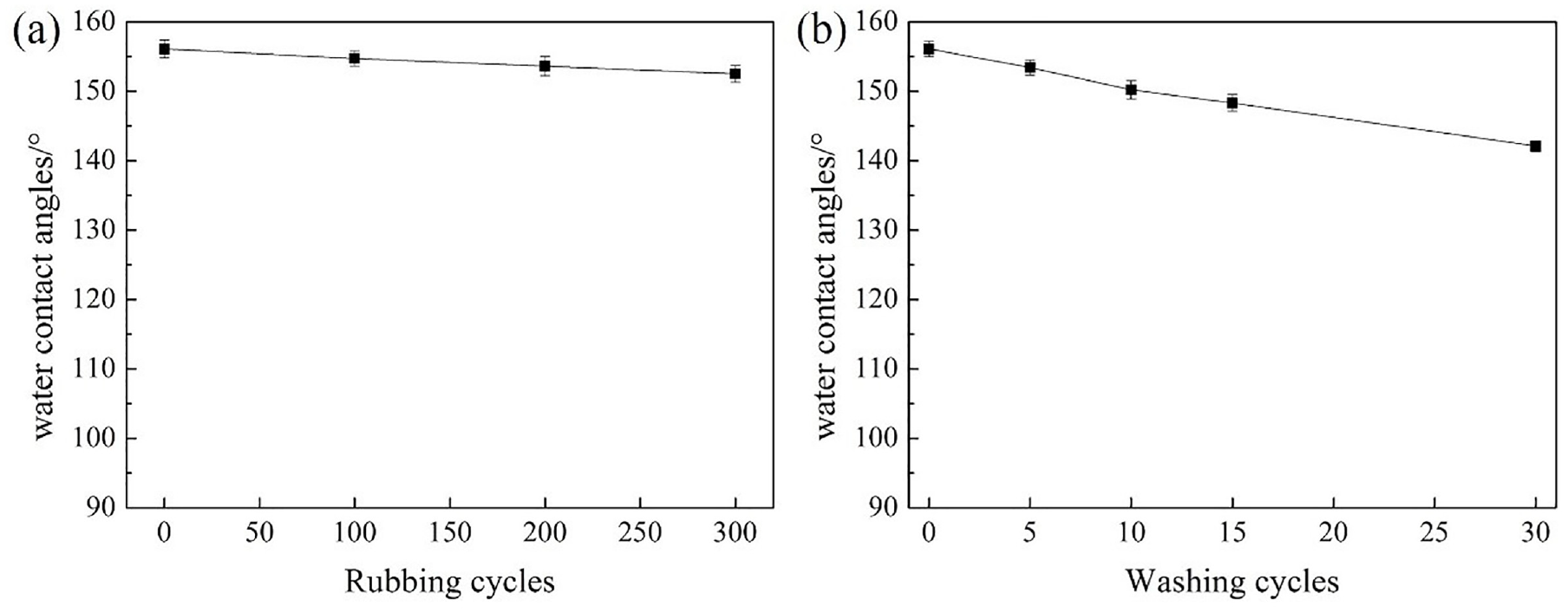
The water contact angle on the surface after: (a) rubbing cycles and (b) washing cycles.
Therefore, the hydrophobic properties of the cotton fabrics had excellent resistance to strong acid, strong alkali corrosion, and physical wear resistance.
Zhang et al. 6 prepared superhydrophobic coating on cotton fabric. It was developed for the synthesis of silica nanoparticles functionalized with octyltriethoxysilane and their in situ incorporation into cotton fabrics. With increasing washing for up to 10 cycles, the coated fabric had a small decrease in water contact angle. However, after 15 cycles of washing, a decrease of about 10° was observed in the water contact angle.
The Self-Cleaning Properties of the Cotton Fabrics
The self-cleaning properties of cotton fabric were analyzed with dyes as soil, and the results are demonstrated in Figure 12.

The self-cleaning property photographs of: (a) cotton fabric and (b) the modified cotton fabric.
From Figure 12(a), after the water droplet contacted with the dye on the fabric surface, the droplet was compatible with the dyes and quickly wet the fabric surface. It was shown that the pristine fabric had good hydrophilic properties, resulting in the dyeing of the cotton fabric.
When the water droplet was dropped on the modified cotton fabric surface, the dye was dissolved in the water droplet in Figure 12(b). The water droplet stayed on modified fabric for a long time with a red spherical state. After 1 min, the dyed water droplet rolled off from the fabric surface. There was no dyeing phenomenon on the fabric surface, which proved that the modified fabric had good self-cleaning performance.
Conclusion
In this paper, the cotton fabric was modified by ODTES-modified SiO2 nanoparticles. The surface morphology showed that the surface and the gap between the fibers were full of siloxane-SiO2. EDS and XPS analysis showed that Si element was added to the modified fabric surface. The contact angle of the modified fabric approached or was higher than 150°, resulting in a rebound phenomenon when water droplets hit the surface. The adhesive force between the water droplet and the surface was small, and the energy was consumed during the collision of water droplets. The residual energy after impact was higher than the surface adhesive force and gravitational potential energy, and the water droplets had enough energy to bounce. The modified fabric had distinct resistance to rubbing and washing, and strong acid and strong alkali corrosion. Furthermore, the modified fabric had excellent self-cleaning properties.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by the Project funded by Project funded by China Postdoctoral Science Foundation (grant no. 2023M743127), Qing Lan Project of Jiangsu University, Nantong Basic Science Research Program (grant nos JCZ2022100, JC2023068, and JCZ2022109), Scientific Planned Projects for Jiangsu College of Engineering and Technology (GYKY/2024/3), Science and Technology Guiding program of China Textile Industry Federation (grant nos 2022032 and 2022030), Hubei Key Laboratory of Biomass Fibers & Eco-Dyeing & Finishing(Wuhan Textile University) (grant no. STRZ202312), and Jiangsu Advanced Textile Engineering Technology Center Funds (grant no. XJFZ/2022/3)

