Abstract
In this research work, the antibacterial activity of inoculated cotton fabric in various metallic salts was investigated. Copper chloride, nickel nitrate, silver nitrate, cobalt oxide, antimony oxide, titanium oxide and tin chloride were used as metallic salts. Very good antibacterial activity for silver-, copper-, nickel- and cobalt-treated fabrics were achieved, however, the effect of titanium and tin on antibacterial efficiency of cotton fabrics was moderate. The scanning electron microscope was used for morphological study. Also, the crystallinity and size of crystals for inoculated and untreated samples were studied by using X-ray diffraction method. The results are truly discussed in full paper.
Introduction
In recent years, much attention has been paid to creating more comfortable living conditions, especially for health and hygiene. For example, fabrics treated with antimicrobial agents are used to suppress bacteria, molds, algae and microorganisms that cause some health-concerned problems such as corruption, spots, and odor [1].
Natural fibers are available in abundance, biodegradable and renewable resources in opposition to synthetic fibers. Cotton is a cellulosic fiber. Cellulose is a biological and functional material, made of glucose with β- 1, 4-glucoside bonds, and has more than ten thousand of these D-glucopyranose sections without branching. Chemical and physical properties of cellulose involve not only the strength and elasticity of the fiber but also water affinity (due to its many hydroxyl groups) and permeability. Cotton is made of cellulose molecules organized in fibers and its physical and chemical properties, as well as its low cost, make the textile with more importance and utility, in our days [2].
Textiles, especially those made of natural fibers such as cotton, are an excellent medium for the growth of microorganisms when the basic requirements such as nutrients, moisture, oxygen and appropriate temperature are present. The large surface area and ability to retain moisture of textiles also assist the growth of microorganisms on the fabric. Therefore, there is a great demand for antimicrobial finishes of textiles to control the growth of microorganisms and prevent the textile from deterioration of strength and quality, staining, odors and health concerns caused by microorganisms [3].
Inorganic, organic and natural antibacterial finishes have been applied on textile goods in recent years. Inorganic antibacterial agents mainly refer to some metallic compounds based on metals like silver, copper and mercury, which could cause inhibition of the active centers of enzymes (inhibition of metabolism) in terms of control-release mechanism [4–6].
Use of antibacterial materials in protective clothing is a good means to improve protection against the biological threats [7–9].
Many heavy metals are toxic to microbes at very low concentrations either in the free state or in compounds. They kill microbes by binding to intracellular proteins and inactivating them. Although some other metals, such as copper, zinc and cobalt, have attracted attention as effective antimicrobial agents for textiles, silver is by far the most widely used in general textiles as well as in wound dressings. It has a MIC value of 0.05– 0.1 mg/L against
Some concerns have been expressed about the development of bacterial resistance to silver.
For synthetic fibers, silver particles can be incorporated into the polymer before extrusion or before nanofiber formation using electro spinning [10–15].
The treatment of natural fibers with metals can only be undertaken at the finishing stage and various strategies have been devised to enhance the uptake and durability.
Cotton has been pretreated with succinic acid anhydride, which acted as ligand for metal ions to enhance the subsequent adsorption of metallic salts (Ag+ and Cu2+) and to provide very effective antibacterial activity [15].
Preparation of nano-sized metals and metal oxides, mainly silver (Ag), titanium dioxide (TiO2), zinc oxide (ZnO) and cooper II oxide (CuO) has enabled the development of a new generation of biocides [16–18]. But there is lack of knowledge about antibacterial activity of the other metallic salts.
In this research work, the antibacterial activity of inoculated cotton fabric in various metallic salts was investigated. Also the morphology and crystallinity of the fabrics after inoculation are studied.
Experimental part
Materials
The samples used in this investigation were plain weave 100% cotton fabrics supplied by the Baft Azadi Co. (Tehran, Iran), they were washed with 1 gL−1 nonionic detergent dried in a laboratory oven. CuCl2, SbCl3, TiO2, Co3O4, SnO2, Ni (NO3)2 and AgNO3 were purchased from Merck.
Inoculation of fabrics in metallic salts
The samples were incubated in a 0.01 M solution of metallic salts, protected from light, and mordanting of them was started at room temperature, the temperature was raised for 2°C/min to boil and heated for 60 min. After this period expired, the samples were rinsed three times with deionized water, subsequently analyzed by the various surface characterization techniques and submitted to bacterial activity test. Immediately before surface analysis, the samples were dried in the laboratory oven.
Morphological study
The morphology of the fabrics was observed using a scanning electron microscope (SEM; TESCAN Brno, Czech Republic). All of the samples were gold coated before conducting the SEM examination.
Testing of antibacterial efficiency
In the bacteria counting test, Luria-Bertani media (LB) broth was used as the growing medium for
X-ray diffraction
The percentage of crystallinity of the samples was determined by SEIFERT (PST-3003) X-ray diffract meter. By X-ray diffraction (XRD) measurement, the percentage of crystallinity and the size of crystals and also the distance between them were investigated.
Results and discussion
The antibacterial activity of samples was tested. The results of the counting test showed more reduction of survival of bacteria in the case of loading samples with metal salts.
The result of counting test is shown in Figure 1. As it can be seen, no colony of bacteria was found in agar culture for Ag- and Cu-loaded samples. It means that the bacteria were killed by silver and copper loading of cotton fabric and causes 100% reduction of bacterial growth.
The antibacterial activity results for untreated and inoculated cotton fabrics.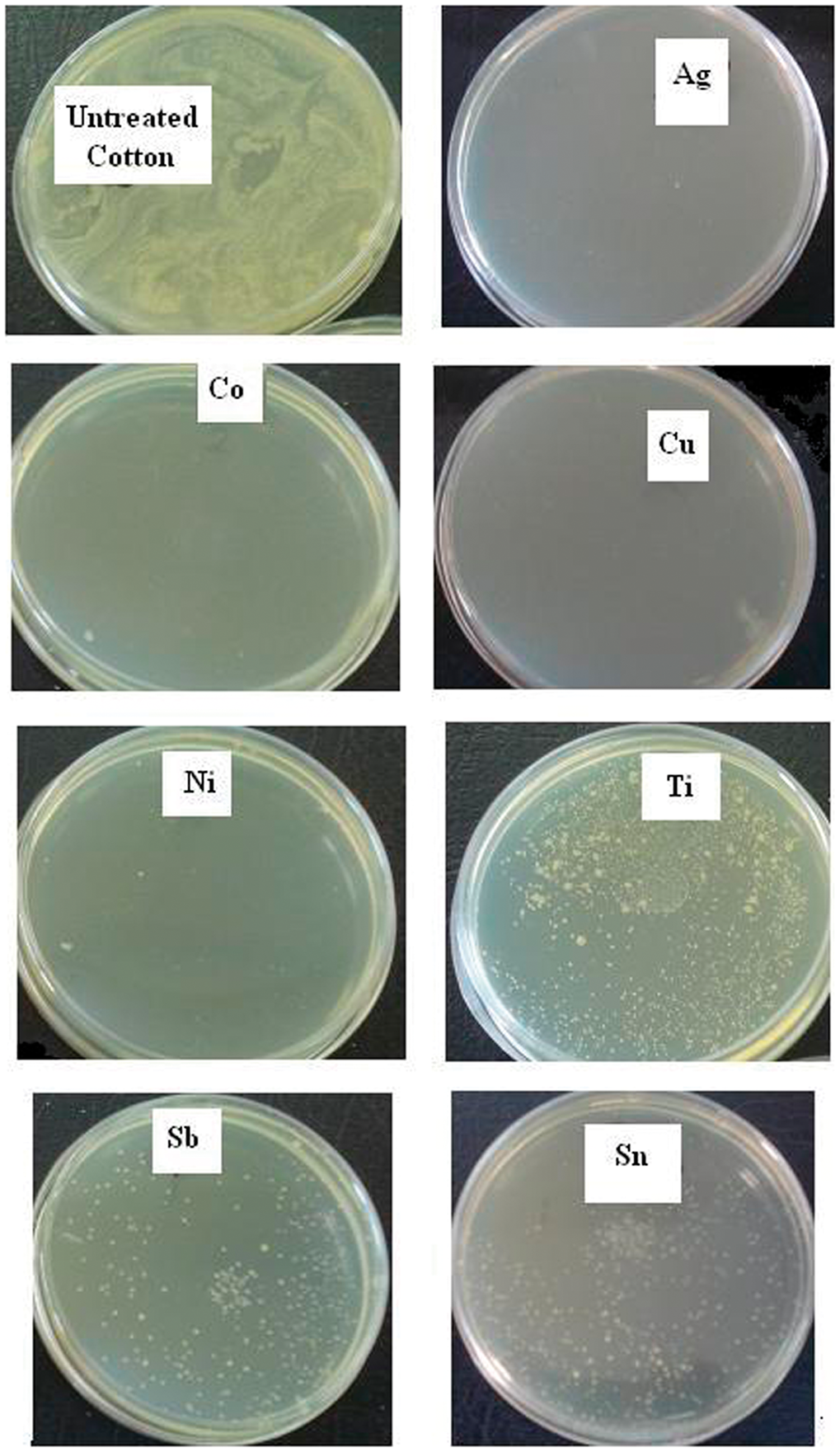
The interaction between silver and copper ions with bacteria can change the metabolic activity of bacteria and eventually causes death.
Also, the results related to nickel- and cobalt-loaded samples show a few amounts of bacteria spread over the agar plate. But, in case of Ti, Sn and Sb loading, it is seen that, more survival bacteria remain and growth in agar culture.
The counting test results related to Sb-loaded samples as compared with Sn and Ti showed better result and caused fewer bacteria to growth, although the amount of survival bacteria for Sn-loaded sample as compared with Ti-loaded one are less. When TiO2 is exposed to ultraviolet light (λ ≤ 400 nm), holes (h+vb) and excited electrons (e−cb) are generated. The hole is capable of oxidizing water or hydroxide anions into hydroxyl radicals (UOH). UOH is known to be powerful, indiscriminate oxidizing agents to degrade a wide range of organic pollutants, including aromatics and aliphatics, dyes, pesticides and herbicides. In 1985, Matsunaga et al. reported the antibacterial properties of TiO2 for the first time, which attributed to the high redox potential of the surface species, affording non-selective oxidation of bacteria [19]. Since then, TiO2, as the photo-induced antibacterial agent, has attracted increasing interest. With high photo-reactivity, cheapness, non-toxicity and chemical stability, TiO2 is promising for eliminating microorganisms in self-cleaning and self-sterilizing materials [13]. In this research work, no ultraviolet light were used before bacteria counting test for Ti-loaded sample and all the samples were analyzed in same condition without UV light as it was mentioned in experimental part. So the results related to bacterial counting test for Ti-loaded sample shows moderate reduction percentage of bacteria; however, by using proper UV light, more reduction of bacterial colonies can be maintained.
It can be concluded that, silver and copper salts causes killing of bacteria and percentage reduction of bacteria reach 100%. It means that no bacteria can spread over the agar plate. Also, the results of antibacterial efficiency for Cu−, Ni− and Co-loaded samples are very good.
And the antibacterial activity for Sn and Ti is moderate as compared with the mentioned elements. However, better antibacterial efficiency was achieved for Sb-treated sample as compared with Ti− and Sn−treated samples.
SEM is the best known and most widely used tool for morphological analyses. SEM micrographs of untreated cotton fabric and metal salt-loaded samples are shown in Figure 2. As shown, some new particles were created on the surface of treated cotton fabrics that did not exist on the surface of untreated one. As it is seen, the particles size that appears on the surface of Sb− and Sn-loaded samples are larger than the others, but it does not mean that large size of these particles made our samples with more antibacterial efficiency.
Scanning electron microscope (SEM) micrographs of untreated and inoculated cotton samples.
XRD is a crystal structure analysis method using the atomic arrays within the crystals as a three-dimensional grating to diffract a monochromatic beam of X-rays.
The angles at which the beam is diffracted are used to calculate the inter-planer atomic spacing (d-spacing) giving information about how the atoms are arranged within the crystalline compounds. X-ray diffraction is also used to measure the nature of polymer and extent of crystalline present in the sample. The results of XRD analysis are reported in Figure 3 and Table 1.
X-ray diffraction results for untreated and inoculated cotton fabrics. The X-ray diffraction (XRD) data for untreated and inoculated cotton fabrics. FWHM: full half maximum width.

X-ray diffraction patterns are used for estimating crystallinity, crystallite size and orientation and occasionally for identification.
It should be possible to determine accurately the percentage of crystallinity by comparing the intensity of diffracted X-ray. Intensity of the diffracted is shown in Figure 3. Peaks around 2 thetas of 14.7, 16.6 and 22.8 are associated to cellulose crystallite. As it can be seen in case of Ag− and Cu-loaded sample, no new sharp peaks appear on the samples, according to the study of standard cards for silver and copper, it was concluded that the important crystal peak for Ag and copper is around 2 thetas of 22 and it means that the crystal of cellulose and these 2 elements cover each other. And it is seen that the intensity of the peaks in mentioned areas are decreased and show that the percentage of crystallinity is decreased; however, this reduction of crystallinity is not significant.
The XRD results for Co-loaded sample show that some new peaks around the 2 thetas of 36 and 64 appear that they are attributed to cobalt crystals. Also, the intensity of main mentioned peaks decreased and it is shown again the reduction of crystallinity after cobalt loading.
In case of nickel-treated samples, it should be said that, no changes can be seen in XRD results and no mentionable reduction on crystallinity can be found. However, for Ti−, Sb− and Sn-treated samples, too many new peaks of crystals were found. According to standard XRD cards for these elements, it can be seen that the peaks around 2 thetas of 25.3, 36.8, 37.84, 48.0, 53.9, 55.1 and 63 are attributed to Ti that appeared on the surface of Ti-loaded cotton samples. For Sb-treated sample, new peaks around 2 thetas of 28.8, 33.7, 40.2, 42.1, 46.0, 51.28, 48.3, 63.2 and 68.9 are related to Sb crystals. Also, in case of Sn-treated samples according to standard cards new peaks around the 2 thetas of 25, 38, 43, 58, 63, 68, and 80 are crystals of tin.
These results confirm the results of SEM and it can be seen that the amounts of particles on the surface of Ti, Sb, Sn and Co are more than the other loaded samples. Especially in case of Sb-loaded samples, some cubic particles cover the surface of cotton fibers and more new crystalline peaks can be found in XRD patterns. Although it is noticeable that the intensity of main peaks around 2 thetas of 14 and 22 are reduced after Ti, Sn and Sb, respectively, it means that by loading the cotton sample in the solution of these metallic salts, the percentage of crystallinity of cotton is decreased. Also by studying the data of full half maximum width (FWHM) in Table 1, it can be see that the amount of FWHM for Ti-loaded sample around the 2 thetas of 22 is increased and it shows that the size of crystals as compared with untreated sample is decreased and by loading the sample with Ti the size of crystals are decreased. But in the case of Sb− and Sn-treated cotton, it can be seen that the FWHM of main peaks (2 thetas of 14, 16 and 22) are decreased as compared with untreated cotton and it means that the size of crystals are increased.
According to d-spacing results, it can be seen that no mentionable changes can be found on the inter-planer atomic spacing of the treated and untreated cotton fabrics.
Conclusion
In this research work, cotton fabrics were inoculated with various metallic salts. Copper chloride, nickel nitrate, silver nitrate, cobalt oxide, antimony oxide, titanium oxide and tin chloride were used as metallic salts. The SEM images show that, after inoculation, some new particles appear on the surface of cotton fibers. In case of antimony treatment, these particles have cubic shape. The antibacterial activity of untreated and inoculated samples was investigated by bacterial counting test method. The results show very good antibacterial activity for silver, copper, nickel and cobalt inoculated fabrics; however, the efficiency of antibacterial activity for Sb−, Sn− and Ti-treated samples were decreased, respectively. The results of XRD pattern show that, the percentage of crystallinity of cotton fabrics after loading them in metallic salts are decreased and this matter in case of Ti, Sn and Sb is more pronounced.
Footnotes
Acknowledgment
Author would like to thank Islamic Azad University, Karaj branch, for grant of members.
