Abstract
A robust superhydrophobic organosilica sol-gel-based coating on a cotton fabric substrate was successfully fabricated via a cost-effective one-step method. The coating was prepared by modification of silica nanoparticles with siloxane having long alkyl chain that allow to reduce surface energy. The coating on cotton fabric exhibited water contact angle of 151.6°. The surface morphology was evaluated by scanning electron microscopy, and surface chemical composition was measured with X-ray photoelectron spectroscopy. Results showed the enhanced superhydrophobicity that was attributed to the synergistic effect of roughness created by the random distribution of silica nanoparticles and the low surface energy imparted of long-chain alkane siloxane. In addition, the coating also showed excellent durability against washing treatments. Even after washed for 30 times, the specimen still had a water contact angle of 130°, indicating an obvious water-repellent property. With this outstanding property, the robust superhydrophobic coating exhibited a prospective application in textiles and plastics.
Introduction
Superhydrophobic surfaces, with static water angles larger than 150°, have aroused great attention in the past few decades due to their wide application in water repellency, anti-contamination, antifouling self-cleaning, and anticorrosion. 1 –4 To fabricate this superhydrophobic surface, two key factors, namely, the surface chemical composition and the surface geometrical microstructure, must be considered. 5 –7 According to Young’s equation γs = γsl + γl cosθ (where γs, γsl, γl and θ are the solid surface tension, solid–liquid surface tension, liquid surface tension, and water contact angle, respectively), it is easily deduced that lowering the surface tension is favorable for enhancing the superhydrophobicity because of an inverse correlation between the water contact angle and the solid surface tension. In addition, Wenzel stated that surface roughness could enhance the wettability caused by the chemistry of the surface. 8 The relationship between roughness and wettability could be depicted as the equation cosθw = rcosθ γ , where θw is the measured contact angle, θγ is the Young contact angle, and r is the roughness ratio. The roughness ratio r is defined as the ratio between the actual and projected solid surface area (r = 1 for a smooth surface and >1 for a rough one). Both micro and nanoscale roughness have been shown to have influence on surface wettability. If the surface is chemically hydrophobic, it will become even more hydrophobic when surface roughness is added. It confirmed that the rough surface with the low surface energy contributed to enhance the superhydrophobicity. 9 –11 Note that liquid wetting of a surface is omnipresent in nature. The interaction between the liquid and underlying surfaces is a transient, dynamic process involving multiple time and length scales, which affected by the surface roughness, wettability, temperature of the substrate, and the ambient conditions. In spite of significant advances, our understanding of wetting statics and dynamics still remains elusive. Much studies confirmed that there are two most important parameters to characterize drop dynamics. One of the most important parameters is the contact time, defined as the time duration between the moment of a drop impacting and bouncing off the surface. The other term that hinders the dynamic spreading of a partially wetting droplet during the initial stages far from equilibrium is the surface structure. The spreading speed under typical conditions of surface roughness and liquid viscosity has been shown to be determined by dissipation associated with contact line friction. 12 –15 Inspired by this principle, biomimetic artificial surperhydrophobic surface has been produced via various processing procedures, such as dip coating, 16 padding, 17 sol–gel method, 18,19 plasma etching, 20 electrostatic layer-by-layer, 21 chemical vapor deposition, 22,23 and so on. Among these techniques, sol–gel method was regarded as the most important and promising one due to its easy operation, cost-effectiveness, low processing temperatures, and its ability to give a large and uniform film on various kinds of substrates.
To prepare the robust superhydrophobic surfaces, most of the studies have focused on introducing silica particles, 24,25 ZnO nanorods, 26 carbon nanotubes, 27 gold particles, 28 or copper crystallites 29 onto the surfaces to improve the roughness. Fluoroalkyltrimethoxysilanes or organosilicon polymers were usually employed to control the chemical properties of surface, and hence their use is beneficial for providing a low energy surface and making the surface more hydrophobic. Until now, the fabrication of super-liquid-repellent surfaces had been researched in extensive work. 17,30 –33 Zhao et al. 30 had prepared a superhydrophobic coating on fabrics using polydimethylsiloxane filled with fluorinated alkyl silane (FAS)-functionalized silica nanoparticles and FAS. Results showed that the water contact angle was up to 171°, which exhibited an excellent water-repellent property. The surperhydrophobic surfaces consisted of roughened silica and heptadecafluorodecyltrimethoxysilane (FAS-17) were fabricated by Nakajima et al. 31 Daoud et al. 17 had applied hexadecyltrimethoxysilane (HDTMS), tetraethyl orthosilicate (TEOS) , and 3-glycidyloxypropyltrimethoxysilane (GPTMS) via a cohydrolysis and polycondensation reaction to produce the transparent and durable superhydrophobic silica-coating films on cotton substrates. However, the procedures of these techniques were complicated and time consuming.
Herein, we report a facile route to prepare water-repellent surface, in which tetraethyl orthosilicate (TEOS) and 3-azido-propyltriethoxysilane were cross-linked with HDTMS (C16) in an ethanol solution at the low temperature. Compared with the previous report, the preparation method for producing the superhydrophobic surface in this research is very simple and effective with the contact angle >150°. Although the contact angle was little smaller than those of the treated surfaces mentioned above, the prepared coating in our study could withstand 30 cycles of machine wash without apparently changing the superhydrophobicity. This simple method may show a great potential in a wide range of applications.
Experimental
Materials
Sodium azide (NaN3) was purchased from Tianyu Chemical Agent Company (Shanghai, China). 3-chloropropyltriethoxysilane ((C2H5O)3Si(CH2)3Cl), triethoxyoctylsilane (C8, as shown in Figure 1), dodecyltriethoxy silane (C12, as shown in Figure 1), and HDTMS (C16, as shown in Figure 1) were procured from Nanjing Chengong Organic Silicon Material Co., Ltd (Nanjing, China). Tetra-n-butylammonium (C16H36BrN), acetonitrile (C2H3N), petroleum ether, ethanol (C2H5OH, 99.7 wt%), ammonia (NH3·H2O, 28 wt%), tetraethyl orthosilicate [(C2H5O)4Si, TEOS)], and soap flake were purchased from Shanghai Chemical Agent Company (China). Nonionic surfactant was provided by Guangdong Dymatic Chemical Agent Co., LTD (China). The scoured and bleached twill weave cotton fabric with grammage of 304 g m−2 was purchased from a common shop.
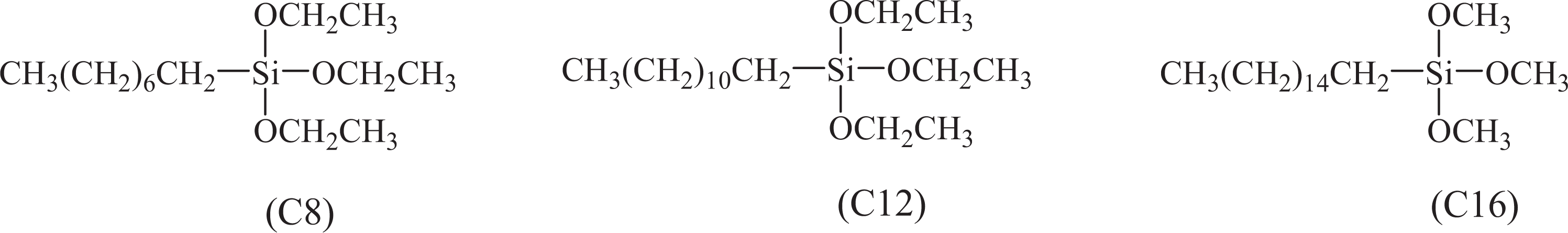
The structure of long-chain alkane siloxane.
Preparation of modified UV-curable SiO2
The ultraviolet (UV)-curable SiO2 sol was prepared by hydrolysis of tetraethyl orthosilicate (TEOS) and 3-chloropropyl triethoxysilane ((C2H5O)3Si(CH2)3Cl) using ammonia as the catalyst. Briefly, C16H36BrN (1.288 g, 4.0 mmol), NaN3 (2.160 g, 33.2 mmol), (C2H5O)3Si(CH2)3Cl (4.000 g, 16.6 mmol), and acetonitrile (100 mL) were added into the flask in sequence with vigorous stirring to obtain a homogeneous solution. Then, the mixture was heated to 40°C for 18 h under a nitrogen atmosphere. After removing the solvent through the filtration and rotary evaporation process, the colorless transparent 3-azido-propyltriethoxysilane ((C2H5O)3Si(CH2)3N3) was obtained for further use. This procedure was also reported by other authors. 34
IR (KBr, cm−1): 2975–2887 (CH3, CH2: C-H), 2097 (-N3), 1081(Si-O).
1H nuclear magnetic resonance (NMR) (500 MHz, CDCl3): δ0.65 (t, 2H, CH2-Si), 1.19 (t, 9H, CH3-CH2-O), 1.68 (q, 2H, CCH2C), 3.20 (t, 2H, CH2N3), 3.78 (q, 6H, OCH2); 13C nuclear magnetic resonance (NMR) (500 MHz, CDCl3): δ7.61 (SiCH2), 18.21 (CCH2C), 22.50 (CH2N3), 53.86 (CH3), 58.13 (OCH2).
To obtain the modified UV-curable SiO2 sol, a certain amount of NH4OH was added into 100 mL of ethanol under vigorous stirring. The used amounts of NH4OH were 3, 3.5, 4, 4.5, and 5 mL, and the resulting sols were denoted as B1, B2, B3, B4, and B5, respectively. After stirring for 30 min at 60°C, 5 mL of TEOS was introduced dropwise. Upon further stirring for 60 min at 60°C, 2 mL of (C2H5O)3Si(CH2)3N3 was added. Finally, the mixture was placed at 60°C and stirred for another 30 min. After that, the long-chain alkane siloxane ethanol hydrolysis solution that involved C8, C12, or C16 was added to modify the UV-curable SiO2. The resulting solution was further stirred for 30 min.
Preparation of water-repellent cotton fabric
The cotton fabric was properly degreased using the nonionic surfactant solution to remove finishing chemicals and impurities from the cotton fabric before they were coated. The treatment process was performed for 30 min at 60°C. After that, the cotton fabric was washed with deionized water and dried at room temperature for further use.
Water-repellent cotton fabric was successfully prepared by a simple one-step penetrant method. The cotton fabric was immersed in the modified UV-curable silica sol with a concentration of 10 mg mL−1 for 60 min. The coated fabric was prepared by the double-nip-double-dip type dye padder to ensure the coating uniformity. Then, the resulted specimen was dried at 80°C for 5 min and cured for 30 min with UV (8 W) light.
Characterization
FTIR analysis of 3-azido-propyltriethoxysilane was recorded on a Nicolet 670 FTIR spectrometer (Woodland, California, USA) with KBr pellets. Spectra in the optical range 400–4000 cm−1 were obtained by averaging 16 scans at a resolution of 4 cm−1.
1H NMR and 13C NMR spectra were collected on a Bruker Avance spectrometer (Carnation, Washington, USA) (400 MHz) using deuterated dimethyl sulfoxide as solvent.
A nano-ZS nanoparticle and potentiometric analyzer was used to measure the average particle size of UV-curable SiO2.
Scanning electron microscopy (SEM) was carried out on a JSM-5600LV scanning microscope (JEOL, Japan) to determine the morphology of coatings at a microscale.
Atomic force microscope (AFM) images were recorded in air at room temperature. Large-scale images are obtained in noncontact mode regime, using Ultralevers silicon conical tips with a typical radius of curvature of 100 Å (cantilever thickness = 2 μm; cantilever width = 28 μm; cantilever length = 85 μm; force constant = 17 N m−1; nominal resonant frequency = 320 kHz).
X-ray photoelectron spectroscopy (XPS) spectra were recorded by using an XSAM800 (Kratos Company, England) XPS system under ultrahigh vacuum. Samples were mounted on a standard sample holder and analyzed using the microfocused, monochromated Al Kα X-ray source.
Water contact angles were measured using an OCA40 Optima (Stuttgart, Germany) surface analysis/goniometry system with water droplets size of 5 μL. All values were obtained by averaging all the data measured from five different regions of each specimen.
The thermogravimetric analyses were carried out on a TG 209 F1 thermal analyzer (NETZSCH, Germany) both in nitrogen with a heating rate of 10°C min−1 between 30°C and 700°C. The weight of all samples was kept within 4–5 mg. The washing fastness of coated fabric was evaluated according to American Association of Textile Chemists and Colorists (AATCC) evaluation procedure 61-2007.
Results and discussion
The superhydrophobic coating on cotton fabric was prepared via modification of UV-curable silica nanoparticles with low surface energy long-chain alkane siloxane. The facile route to prepare water-repellent surface, in which tetraethyl orthosilicate (TEOS) and 3-azido-propyltriethoxysilane were cross-linked with HDTMS (C16) in an ethanol solution at low temperature. The schematic description of our approach is depicted in Figure 2.

Schematics for the preparation of the water-repellent films on cotton fabrics.
Effect of diameter of modified UV-curable SiO2 particle on water contact angle
According to the preparation method of superhydrophobic coating, the modified UV-curable SiO2 sol of different diameters could be obtained by controlling the mass of ammonia. It is clear that the ammonia amount has an important effect on the water contact angle. To study the significance of ammonia amount, C16 was selected as the repellent and its dosage was set as 2 mL. The relationship between ammonia amount and particle size of modified UV silica sol is presented in Table 1. The average particle sizes were found to increase gradually with a rise in the ammonia amount, while polydispersity index (PDI) was first decreased and then increased. The PDI of modified UV-curable SiO2 particles was reached the smallest value 0.077 when the ammonia amount was 4 mL. Figure 3 depicts the effect of modified UV silica sol particle sizes on water contact angle, and Figure 4 shows the images of water drop on cotton fabric surface with and without modified UV-curable silica sol. The water contact angle of original cotton fabric was 0° because of its hydrophilic property, as shown in Figure 4(a). The cellulose hydroxyl groups of cotton fabric make the water droplets spread instantly when placed on the surface of the substrate. From Figure 3, it was clear that the water contact angle revealed a contrary trend of PDI along with the increase of ammonia amount. The contact angle of the cotton fabric only immersed in C16 ethanol solution was the smallest 134.9° among these samples, as shown in Figure 4(b), while that of cotton fabric coated with modified UV-curable SiO2 sol increased to 143.5°. It confirmed that the introduction of ammonia catalyst was helpful to reduce the water contact angle. When the ammonia amount was 4 mL, the water-resistant capacity of coated cotton fabric was optimum with the water contact angle 151.6°, as shown in Figure 4(c). In addition, the water contact angle first increased then decreased with the increasing of the particle size of UV silica sol. This was affected by the PDI value. As we all know, in physical and organic chemistry, PDI is a measure of the heterogeneity of sizes of molecules or particles in a mixture. The smaller the PDI value, the more uniform the particle. In this study, it is easy to find that the water contact angle of treated cotton was high, that is, 151.6°, when the particle size of UV silica sol was 104.8 nm. Because PDI was small to the minimum value 0.077 at this experiment which indicated the UV silica sol has the most uniform particle size. This was possibly that the surface roughness of cotton fabric was improved by the uniform UV silica sol. This phenomenon was consist with the relation between the roughness of hydrophobic surface and contact angle reported by Wenzel 35 and Cassie and Baxter. 36 Therefore, the ammonia amount was hence optimized at 4 mL (B3) and further studies were carried out with this amount.
Effect of ammonia amount on particle size of UV silica sol.

UV: ultraviolet; PDI: polydispersity index.

Effect of size of UV silica sol particle on contact angle. NP represented that cotton fabric only immersed in 2 mL of C16 ethanol solution. NP: no particle.

Images of water drop on (a) original cotton fabric, (b) cotton fabric coated with C16, and (c) cotton fabric coated with modified UV-curable silica sol.
Effect of structure and amount of alkane siloxane on the water contact angle
A series of the repellent modifier (C8, C12, C16) solutions with different concentrations were prepared to study the effect of structure and amount of alkane siloxane on the water contact angle, and the corresponding result is shown in Figure 5. Apparently, the water contact angle of coated cotton fabric increased with a rise in the carbon chain length. The reactive hydroxyl groups of (C2H5O)3Si (CH2)3-N3 condensed with the -OH groups of hydrolyzed alkane siloxane resulting in the formation of siloxane networks thus lowering the surface energy of the coating. The long carbon chain of alkane siloxane was helpful for shielding hydrophilic groups, 37 which resulted in a good hydrophobic property. In addition, the water contact angle of coated cotton fabric with C8 or C12 repellent modifier showed an upward trend and then trended to be stable upon increasing the concentration of alkane siloxane, while that of C16-coated cotton fabric drop curve fell down after it ascended first. It was evident that the water contact angle of coated fabric was up to 151.6° when the dosage of C16 was 2 mL, which exhibited a good water-repellent property. However, continuing to increase the dosage of C16 to 3 mL and 4 mL, the water contact of coated cotton fabric decreased. It might be that unreacted C16 hydrolysate would compete with modified UV-curable SiO2 on the interaction with -OH of the cotton fabric surface.

Effect of siloxane structure and dosage on water-repellent property.
To obtain the structural information of the cotton surface before and after coating treatment, typical SEM images are reported in Figure 6. It was found that the surface of original cotton fiber (Figure 6(a) and (b)) had peculiar uneven stripes, showing regular natural curvature. After being coated with modified UV silica sol (Figure 6(c) and (d)), the fiber was covered by a waxy layer, which incorporated the modified UV-curable silica sol with the random distribution of nano silica particles all over the surface. The coating thickness was about 190 nm as shown in Figure 7. This granular and rough surface was helpful to improve the surface roughness of the fiber. Higher magnifications showed silica particles of size about 100 nm. In addition, the introducing of waxy material the long-chain alkane compound could be significantly reduced the surface energy of cotton fabric, which was also favorable for enhancing the water-repellent effect.

SEM images of the cotton fabric before and after being coated with modified UV silica sol: original cotton fabric ((a) ×1000 magnification; (b) original cotton fabric ×5000 magnification); cotton fabric coated with modified UV silica sol (B3) ((c) ×5000 magnification; (d) ×50,000 magnification). SEM: scanning electron microscopy.

SEM image of cross section of coated fabric with modified UV silica sol. SEM: scanning electron microscopy.
In addition, the AFM measurement was also used to investigate the morphology and the coating roughness of fibers. The obtained results on pure cotton fiber and modified UV silica sol-coated cotton fiber are shown in Figure 8(a) and (b). In particular, it is noticed that the sample surface after coating with modified UV silica sol presents a more rough morphology compared with that of pure fiber. The increased value of roughness, as compared to the pure fiber, can be explained with the presence of modified UV silica sol thin layer on the surface of the fiber. This result was confirmed by the SEM measurement and water contact angle measurement. The root mean square roughness (RMS) of the cotton fabric before and after being coated with modified UV silica sol was 1.4 and 18 nm, respectively.

AFM images of the cotton fabric before and after being coated with modified UV silica sol: (a) original cotton fabric; (b) cotton fabric coated with modified UV silica sol (B3). AFM: atomic force microscope.
There was no doubt that the elemental composition of cotton fabric had a close relationship with the water-repellent performance. Therefore, the change of element on the surface of cotton fabric was analyzed by XPS, and the spectra are presented in Figure 9. As shown in Figure 8(a), there were two intense peaks around 274.6 and 533.2 eV, which correspond to the C and O elements. After coated with modified UV silica sol, additional peaks at 399, 150.5, and 103 eV were attributed to N1s, Si2s, and Si2p, respectively. It confirmed that modified UV silica sol was successfully coated on the surface of cotton fibers.

XPS analysis of (a) original cotton fabric and (b) cotton fabric coated with modified UV-curable silica sol. XPS: X-ray photoelectron spectroscopy.
To investigate the amount of UV silica particles deposited on the cotton fibers and the thermal stability of treated fibers, thermogravimetric analysis was performed in air flow. There was an intense weight loss in air between 250°C and 360°C due to the decomposition of cotton fibers, as shown in Figure 10. Heated to 700°C, the residue was 5.88 wt% in the cotton fibers due to the impurities, while that of modified UV silica coated fabric was up to 9.65 wt% because of the existence of C16-functionalized SiO2 nanoparticles. In addition, the introduction of modified UV-curable SiO2 has no effect on the thermal stability of cotton fiber due to the similar thermal degradation curves. From these results and the SEM and XPS analyses (before described), the presence of a UV silica particle layer on the fiber surface is confirmed.

TGA curves of cotton fabric before and after being coated. TGA: thermogravimetric analysis.
Based on the analysis mentioned above, the superhydrophobictity of coating on cotton fabric was attributed to the surface roughness created by silica nanoparticles and low surface energy imparted by long-chain alkane siloxane.
The washing durability test was made to assess the stability of thin film on the fabric surface. Repeating the washing process for 5, 10, 15, 20, 25, and 30 times, the water contact angle value of the specimens was measured and water-repellent durability of the coated fabrics was evaluated. Figure 11 shows the washing fastness of cotton fabrics coated with modified UV silica sol (B3). After washing for 30 times, the specimen still had the water-repellent property, the water contact angle was over 130°. As shown in Figure 6(c) and (d), the fiber was covered by a waxy layer, which incorporated the modified UV-curable silica sol with the random distribution of nano silica particles (about 100 nm) all over the surface before being washed. Even after washed for 30 times, the surface roughness of cotton fabric was obvious and the surface was still covered by a thin and uneven waxy film as shown in Figure 12. Although the coated cotton fabric was washed for 30 times, the XPS peaks at 150.5 and 103 eV, attributed to Si2 s and Si2p, were detected, as shown in Figure 13. It was because that the cross-linking degree between the modified UV-curable silica sol and cotton fabric was high. In addition, the excellent covalent bonding between azide and cotton fabric was also favorable for the washing durability.

Variation in water contact angle and the number of wash cycles on cotton fabric coated with modified UV silica sol.

SEM image of after 30 times soaping on cotton fabric: (a) ×10,000 magnification; (b) ×30,000 magnification. SEM: scanning electron microscopy.

XPS spectra of coated cotton fabric after 30 times soaping. XPS: X-ray photoelectron spectroscopy.
Conclusions
The superhydrophobic coating on the cotton fabric was produced via a simple and environment friendly as no fluoro chemicals method. The coating was obtained through cohydrolysis and polycondensation of a tetraethyl orthosilicate, 3-azido-propyltriethoxysilane and long-chain siloxane. The results indicated that the ammonia amount and the structure of alkane siloxane play an important role in reducing the water contact angle. The coating on cotton fabric exhibited a water contact angle up to 151.6° when the ammonia amount was optimized at 4 mL and C16 dosage was set as 2 mL. Moreover, this organosilicon-based coating showed an excellent washing durability. Even after washed for 30 times, the specimen still had a water contact angle of 130°, displaying a super water-repellent performance. With this superhydrophobic property, contaminates adhered to the surface can be removed easily by the passage of flowing liquids. Therefore, this robust superhydrophobic coating with the water contact angle 151.6° in the study exhibited a prospective application in emerging technologies such as flexible wearable capacitors, inkjet printing of electronics, boiling enhancement, droplet repulsion, material patterning, and design and adhesion.
Footnotes
Acknowledgments
The authors gratefully thank the Jiangsu Lianfa Textile Co., Ltd, for providing the funds and acknowledge the funding received from the Research Innovation Funds of Shanghai Municipal Education Commission and the Fundamental Research Funds for the Central Universities. The authors would also like to thank the funding by the Collaborative and Innovation Program for Education and Research.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was supported by Jiangsu Lianfa Textile Co., Ltd, Research Innovation Funds of Shanghai Municipal Education Commission, the Fundamental Research Funds for the Central Universities (14D210504), and Collaborative and Innovation Program for Education and Research (1021ZK162001).
