Abstract
Worldwide, there is growing concern over the toxicity of arsenic in drinking water. The US Environmental Protection Agency and Central Pollution Control Board (India) have set a safe limit of 50 parts per billion for arsenic in drinking water. Millions of people are at immediate risk due to high concentrations of arsenic in drinking water, particularly in developing nations where arsenic poisoning symptoms have been observed. One of the essential techniques described in this article is the absorption of arsenic from contaminated water by sustainable cellulosic materials like jute and wood sawdust after suitable modification. Jute and sawdust are cheap and abundantly available in South Asia. They are functionalized with polypyrrole coating by in situ chemical polymerization of pyrrole in aqueous medium in the presence of ferric chloride. Polypyrrole-coated jute fabric (with 11.28% polypyrrole add-on) and polypyrrole-coated sawdust (with 5.28% polypyrrole add-on) are found to be excellent adsorbers of arsenic ion (As3+) from water. The effect of dosage of polypyrrole-coated adsorbents, treatment time and pyrrole concentration on the As removal efficiency have been investigated in this work. The arsenic concentration in water is determined using a microwave plasma atomic emission spectrometer with the highest level of accuracy. Maximum As removal efficiency by the polypyrrole-coated sawdust and polypyrrole-coated jute fabric is found to be 80.90% and 97.3%, respectively, from a 50 mL, 7.5 parts per million As solution with a dosage of 1 g at neutral pH. The polypyrrole-coated jute and sawdust are characterized by Fourier-transform infrared spectroscopy to understand the chemical interaction between cellulose and polypyrrole molecules. The scanning electron microscope studies reveal a uniform coating of polypyrrole around the surface of jute fibre and sawdust particles. Thermo-gravimetric analysis shows good thermal stability of jute and sawdust after polypyrrole coating.
Introduction
Among the world’s most hazardous environmental contaminants are heavy metals. Heavy metals and their derivatives are considered a unique class of contaminating substances that have been linked to daily detrimental impacts on people. The respiratory and digestive systems are two places where they can enter the body, and the results can be disastrous. Heavy metal ions are hazardous to living things primarily because of their toxicity and capacity to build up within the body. The environmental problems relating to the effects of human activity on the environment receive increased attention each year. The likelihood of developing a number of illnesses, including genetic mutations, cardiovascular illnesses, malignant tumours, weakened immunity, poisoning, allergies and neurological disorders, rises with the level of pollution of aquatic media. It is important to note that diseases and anomalies brought on by metal ion exposure do not yet have an effective cure. Among the heavy metal ions, arsenic appears to be the most common species in groundwater. Worldwide, there is growing concern over the toxicity of arsenic in drinking water. It is highly carcinogenic and known as one of the world’s most hazardous chemicals. 1 The sources of this contamination include fly ash disposal, geothermal discharge, arsenical pesticides, mineral dissolution in mining sites, agricultural drainage waters from certain arid soils and pyritic sedimentary rocks (mine drainage) in contact with the aquifer. Many South Asian countries are facing the problem of high arsenic concentrations in drinking water. Signs and symptoms of arsenic poisoning have been linked to high concentrations of arsenic in drinking water, particularly in developing nations where millions of people are at immediate risk. The toxic characteristics of inorganic arsenic species, such as As(III) and As(V), have long been recognized. 2 Cancer of the bladder, lungs, skin, kidney, nasal passages, liver and prostate can result from prolonged exposure to arsenic in drinking water. The non-cancerous consequences of arsenic consumption encompass pulmonary, immunological, neurological and endocrine (like diabetes) disorders. In addition to its ability to cause tumours, arsenic has been demonstrated to be genotoxic. According to World Health Organization (WHO), the US Environmental Protection Agency (USEPA) and the European Commission (EC), the standard value of maximum contaminant level (MCL) for arsenic in drinking water is 10 μg/L.1,3
In natural waterways, arsenic is mostly found in inorganic form as oxyanions of trivalent arsenite [As(III)] or pentavalent arsenate [As(V)]. As(III) is approximately 10 times more toxic to animals and plants than As(V). 4 The reduced form [As(III)] is usually found in groundwater (anaerobic conditions) under reducing conditions, and the oxidized form [As(V)] is mostly found in surface water or oxidizing waters. Depending on pH, arsenic (arsenite, arsenate) can exhibit anionic forms in aqueous systems. Nowadays, great attention has been paid to developing cost-effective technologies to remove arsenic species from water. Technologies such as adsorption, 5 co-precipitation, 6 ion exchange, 7 iron-coated sand, 8 membrane-based techniques including reverse osmosis 9 and nano-filtration, 10 along with iron-based materials 11 and activated carbon have been extensively used to remove arsenic species from water. The adsorption technique has drawn considerable attention recently from researchers due to its simplicity and cost-effectiveness in the process.
Binding free molecules of a gas, liquid or solid solute to the surface of a liquid or solid sorbent is known as adsorption. On the surface of the adsorbent, the adsorbed molecules, or adsorbate, will accumulate into a molecular or atomic layer. Adsorption techniques have several uses in a variety of sectors, including the treatment of wastewater, where they are used to remove contaminants. All suspended pollutants in wastewater can be adsorbently removed by an appropriate porous adsorbent.12–15 Adsorbate molecules bind to the adsorbent surface in different ways, leading to the classification of the process into two categories: physisorption and chemisorption. In the physisorption process, adsorbents and adsorbates interact non-covalently, whereas in the chemisorption phenomena, strong chemical bonds are formed between the adsorbates and adsorbents. Non-covalent bonds, like Van der Waals forces, are weak intermolecular forces that occur during the physisorption process.
The type of adsorption material employed determines how well different impurities are removed. Such materials are classified in a way that suggests a difference between natural and manufactured materials. The first category consists of sand, zeolites, vermiculites, chalk and clay rocks, ash, peat, slag and carbons of different natural origins. The second class of adsorbents includes activated carbons, ion exchange resins, titanium and zirconium hydroxides or phosphates, synthetic zeolites, silica gels made from mineral raw materials treated in various ways and polymers containing chelate. 16 The majority of the adsorption materials already on the market are unable to sufficiently cleanse wastewater to remove the necessary amounts of dangerous contaminants, making the current condition of anthropogenic aquatic media essential. Therefore, using innovative materials with a substantially higher specific surface area and further modified with different chemically active groups to boost adsorption activity and selectivity is required to raise the effectiveness of the adsorption. Many scientists are focusing their research efforts on creating materials whose adsorption capacity is several times higher than that of traditional adsorbents like activated carbon, zeolites and clays, in response to the industry’s rapid development in nanotechnology.17,18 The primary members of this class of materials are carbon-based modifications, specifically graphene nanostructures and carbon nanotubes (CNTs). A lot of research has been done on the adsorption characteristics of CNTs. It was observed that these methods suffer from certain drawbacks, such as long adsorption time and varied pH. 16 Therefore, these cannot be used under natural water conditions. Therefore, the authors of the present work have proposed the use of novel conductive polymers.
Among the different conducting polymers, polypyrrole (PPy) has been found to be one of the most appealing. It is characterized by its high conductivity and environmental stability.19–21 PPy can be synthesized by chemical polymerization from its precursor (pyrrole) in the presence of ferric chloride (oxidant) in aqueous solution. 19 A new field of application for this conductive polymer is ion adsorption and wastewater treatment, which demonstrates its credible candidature. 14 PPy molecules can interact and adsorb various organic and inorganic molecules such as Cr(VI), Cu(II), Pb (II), As and Cd(II), because of the presence of abundant amino groups in their molecular structure. 14 PPy is solely used for the removal of arsenic ions from water by adsorption technique, and it has been discovered that the adsorption performance of PPy is highly dependent on the preparation conditions of the mole ratio of monomer to oxidant. 5 PPy is coated over various substrates to prepare composite adsorbents for arsenic removal. The As(III) removal efficiency by PPy-coated nickel and Fe3S4 has also been reported.22,23 However, very low removal efficiency is obtained from these two PPy nanocomposites. Ansari et al. 4 investigated the use of PPy, polyaniline and poly 3-methylthiophene coated with wood sawdust (SD) on As(III) removal from water with 74.5%, 82.6% and 85% arsenic removed. Effects of various parameters like dosage, treatment time, temperature and pH on removal efficiency have been reported. However, there is a lack of studies available on the As(III) removal efficiency of PPy-coated textile substrates and optimization of the treatment process to obtain maximum removal efficiency. Jute is a natural cellulosic textile fibre which is cheap and abundantly available in south Asia. This fibre has not explored been much for PPy coating and diverse applications for water purification.
Therefore, in this research, PPy is coated on jute and SD to prepare sustainable adsorbents for the removal of As anions from contaminated water.
Experimental
Materials and Chemicals
Textile jute fabric (ends per inch—15, picks per inch—13 and areal density—300 g−2) was collected from a local shop in Kanpur, U.P. (India). The chemicals used are ferric chloride (FeCl3) as oxidant, pyrrole as monomer and arsenic trioxide for making As solution. Wood SD (obtained from a furniture shop in Kanpur, U.P. (India)) has a particle size between 64 and 265 µm. All the chemicals used are of laboratory grade and procured from Sigma-Aldrich, India.
Preparation of PPy-Coated Jute Textile and SD for As Removal
PPy is coated on the surface of jute fabric and SD by in situ chemical polymerization. The overall polymerization process is shown in Figure 1. 19 Textile jute fabric and SD samples are scoured first for better fixation of PPy on the jute surface and SD. Then, a two-stage double bath process is employed for in situ chemical polymerization of pyrrole, as shown in Figure 1. In the first stage, the sample (jute and SD) is soaked in a monomer bath containing monomer solution, and in the second stage, in situ polymerization of pyrrole is conducted in an oxidant bath. The monomer bath is prepared by dissolving pyrrole in 0.25 M concentration in de-ionized water. The oxidant bath is prepared by dissolving FeCl3 in 0.50 M concentration in de-ionized water. The material-to-liquor ratio for both the baths is kept at 1:40. Scoured jute fabric and the SD sample are allowed to soak in monomer bath for 15 min at 5°C. The oxidant bath is cooled to 5°C, and the pyrrole-enriched jute and SD samples are taken out of the monomer bath and dipped into the oxidant bath for in situ polymerization. The time of in situ polymerization is 1 h. After the polymerization, the samples are taken out of the oxidant bath, thoroughly rinsed and dried at room temperature for 72 h before measurement.
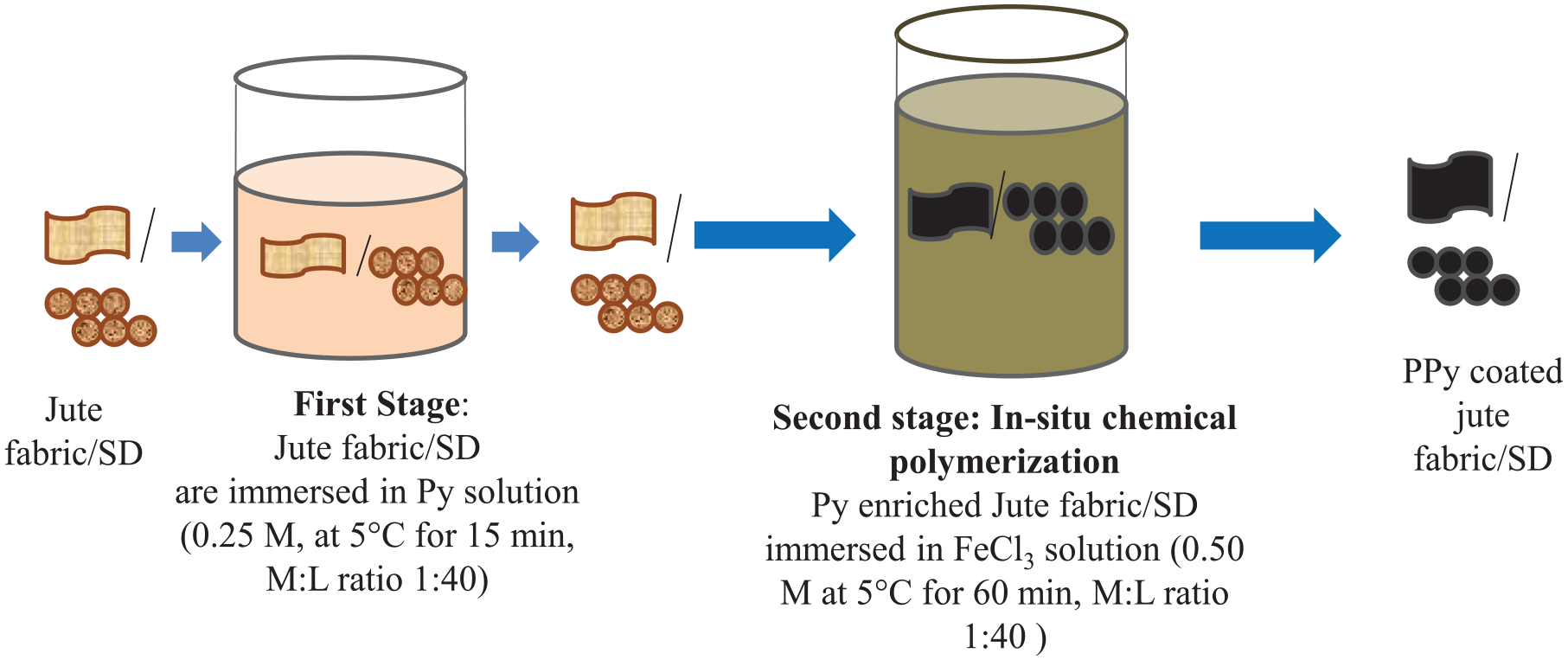
Coating the jute fabric/SD with polypyrrole by in situ chemical polymerization.
Method of As Removal Process from As Solution
Arsenic solution is prepared by dissolving As2O3 in water (7.5 parts per million (ppm)). PPy-coated adsorbents of predetermined weight are agitated with 50 mL As2O3 solution at pH 7 at 30 ± 0.5°C for a predetermined time, as shown in Figure 2. After that, the sorbent is separated from water, and the reduction in the concentration of As is determined using a microwave plasma atomic emission spectrometer (MPAES, Agilent 4200).
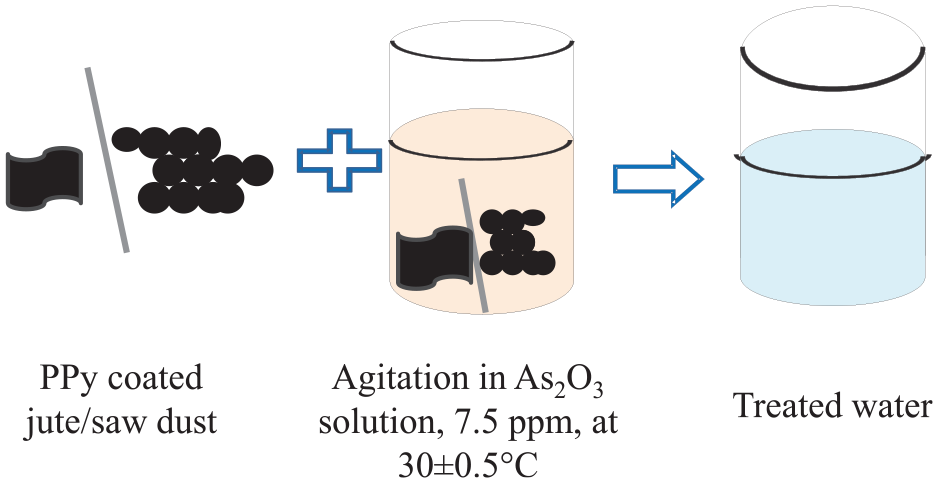
Arsenic removal process.
Analysis of PPy-Coated Jute and SD by Fourier Transform Infrared Spectroscopy
Fourier-transform infrared (FTIR) spectroscopy analysis of uncoated and PPy-coated jute fabrics and SD is conducted to identify the chemical interactions between jute fibre and SD and PPy molecules. The test is conducted using a Bruker-Alpha FTIR spectrometer for the wave number range of 4000–500 cm−1.
Surface Morphology Analysis
The morphology of uncoated and PPy-coated fabrics and SD is investigated using a scanning electron microscope (SEM) (CARL ZEISS EVO-50) at two different magnification levels of 500× and 2500×.
Thermal Analysis
The thermal stability and degradation behaviour of uncoated and PPy-coated jute fabrics and SD are evaluated using a TGA instrument (Perkin Elmer TGA-4000) for a temperature range of 30–600°C with a heating rate of 10°C/min with purging nitrogen gas. Differential scanning calorimetry (DSC) is used to investigate the thermal characteristics of uncoated and PPy-coated jute and SD using DSC (Perkin Elmer) for a temperature rate of 10°C/min with the range of temperature −10–300°C with purging nitrogen gas.
Result and Discussion
PPy Add-on Percentage on Jute and Wood SD
Although PPy has excellent ion adsorption capability, it is challenging to prepare fibres or films due to its poor mechanical properties. Therefore, PPy is coated over cellulosic substrates like SD and jute fabric owing to its good substantivity to cellulosic materials. As a result, PPy-coated SD and jute composite adsorbents are achieved where jute and SD are acting as reinforcements and PPy as the matrix. The composites are supposed to provide excellent durability, good mechanical properties, better handling and excellent ion adsorption. SD is a waste from wood industries that has already been explored by researchers for wastewater remediation treatment. Jute, as an abundant natural fibre, is taken into consideration for this study to diversify its use. The jute fabric and SD are coated with PPy by in situ chemical polymerization following the same process and recipe. After the polymerization, the coated substrates are dried in a hot air oven and the PPy add-on is measured. The PPy add-on percentage achieved for both substrates is shown in Figure 3. It can be seen that jute fabric achieves a PPy add-on of about 11.28%, which is almost double that of SD (5.31%). Jute fibres consist of 72% cellulose, 13% hemicellulose and 13% lignin. 24 Hydroxyl and carboxylic groups present on cellulose molecules act as anchoring groups for attaching PPy molecules on the jute surface. However, SD contains about 48% cellulose, followed by 33% lignin and 17% hemicellulose.25,26 Due to the presence of a higher amount of hydrophobic lignin on the SD surface and low cellulosic content, its PPy add-on is found to be significantly lower than that of jute.
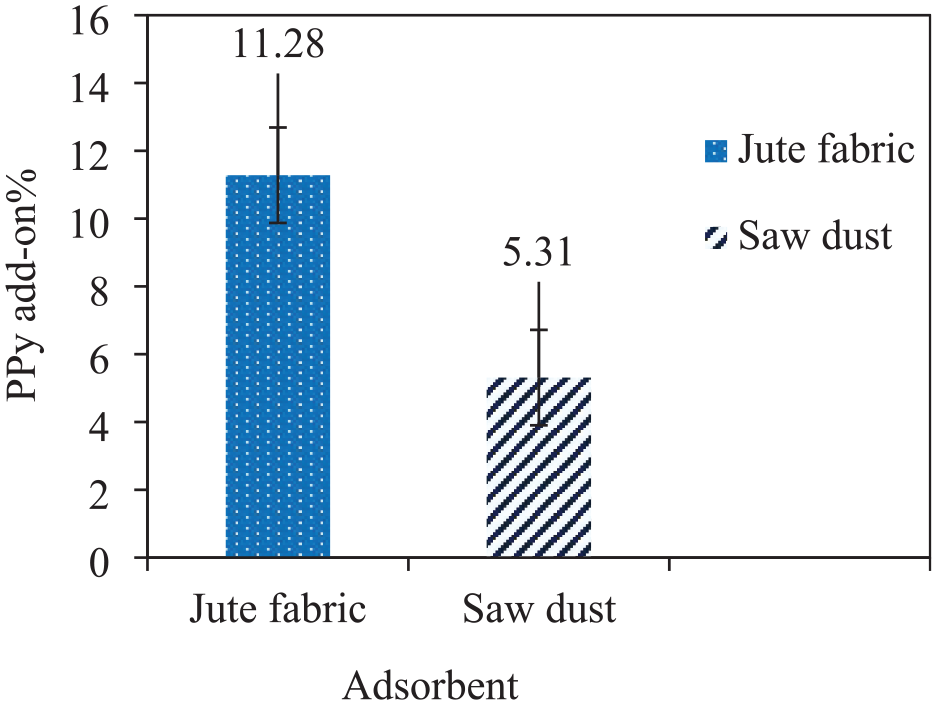
Polypyrrole add-on percentage of jute and wood sawdust.
SEM Images of Jute and SD
SEM images of the uncoated and PPy-coated jute fabrics and SD are shown in Figure 4. It can be seen that all individual jute fibres and SD particles are coated quite uniformly with PPy polymer. The granular morphology of PPy is observed on their surface. This finding confirms that the polymerization proceeded smoothly, resulting in the formation of a uniform layer of PPy around the adsorbents.

SEM images of uncoated and PPy-coated adsorbents.
FTIR Analysis of PPy-Coated Jute Fabrics
FTIR analysis of uncoated and PPy-coated jute fabrics is done to determine the chemical interaction between PPy and the substrate jute fibres. The FTIR spectra of uncoated and PPy-coated jute fabrics are shown in Figure 5. In the FTIR spectra of uncoated jute, a band observed at 3750 cm−1 is due to the non-bonded hydroxyl group, the band at 3321 cm−1 is due to –OH stretching, the band at 2925 cm−1 is due to CH and CH2 stretching of the aliphatic group, and the band at 1420 cm−1 is due to the –C=O inorganic carbonate group. The FTIR spectra of the PPy-coated jute fibre show bands at 1200 and 1473 cm−1, which can be attributed to C–N stretching vibration and C=C ring stretching vibration of PPy, respectively. Bands at 958 and 903 cm−1 may be due to the chemical bonds between cellulose and PPy polymer molecules at these positions. The new bands in these positions depict a significant chemical interaction between cellulose and PPy by which fixation of PPy occurs on the jute surface.
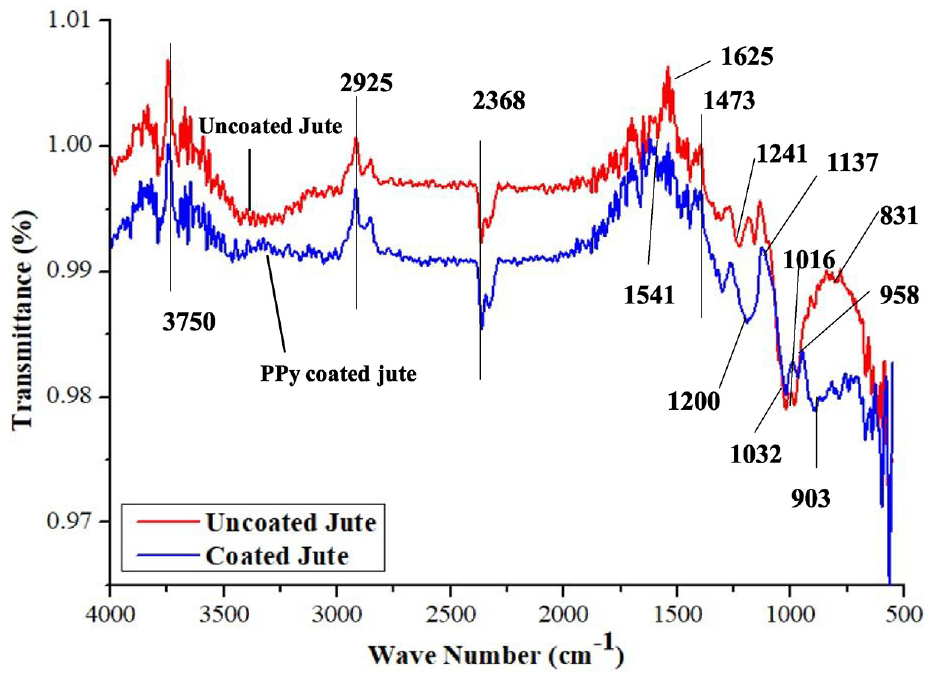
FTIR analysis of uncoated and PPy-coated jute fibres.
FTIR Analysis of SD
Figure 6 displays the FTIR spectra of SD that is uncoated and coated with PPy. Uncoated SD’s FTIR spectra exhibit peaks at 3746, 2993, 1560, 1229, 1135 and 2370 cm−1. These peaks can be attributed to the following: C=N stretching, alkyl ketone, alkyl amine, carboxyl acid polyphenol skeleton and OH stretching, in that order. Peaks in the PPy-coated SD that are found around 3750, 3165, 2888, 2368, 1648, 1197, 1123, 1276, 1026 and 999 cm−1 wave numbers can be attributed to O–H stretching, carboxyl acid, C=N stretching, C=O alkyl amine, alkyl ketone C–O in the hydroxyl group and C–O, respectively.
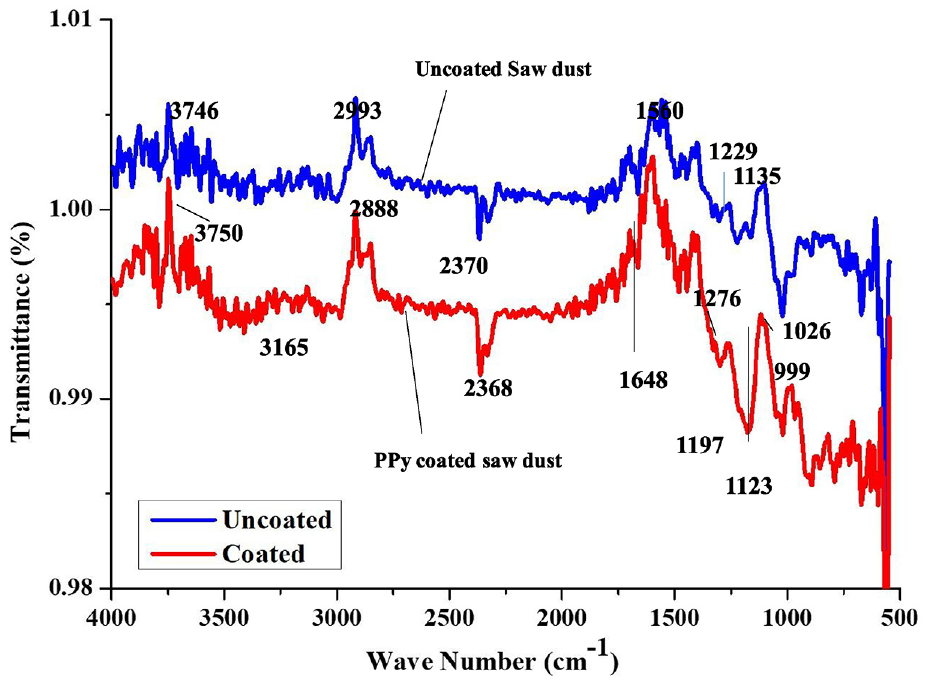
FTIR analysis of uncoated and PPy-coated sawdust.
TGA Analysis of SD and Jute
A thermogravimetric analysis was performed to investigate the weight loss and thermal stability of SD and jute exposed to elevated temperatures. All of the SD and jute samples have degradation behaviour between 30 and 500°C, as shown in Figures 7 and 8, respectively. There are two stages of mass loss in the case of SD samples. The first stage of weight loss occurs when the moisture content is lost, which is observed between 30 and 150°C. The second stage of mass loss is caused by the breakdown of cellulose components, which takes place between 250 and 380°C. The first drop in weight of SD due to loss of moisture increased from 7.5 to 11.9% due to the PPy coating. Afterwards, the total loss was around 55.8 and 39% for uncoated and PPy-coated SD, respectively. It has also been noted that both PPy-coated and uncoated jute degrade in two steps, as shown in Figure 8. The initial weight loss resulting from moisture evaporation is observed to be 8.1% for uncoated jute and 3.6% for PPy-coated jute. At approximately 280–380°C, the final stage of weight loss resulting from thermal degradation is found to be 57.3 and 50.6% for uncoated and PPy-coated jute, respectively. Therefore, it can be concluded that after PPy coating, the thermal stability of SD and jute has not deteriorated.
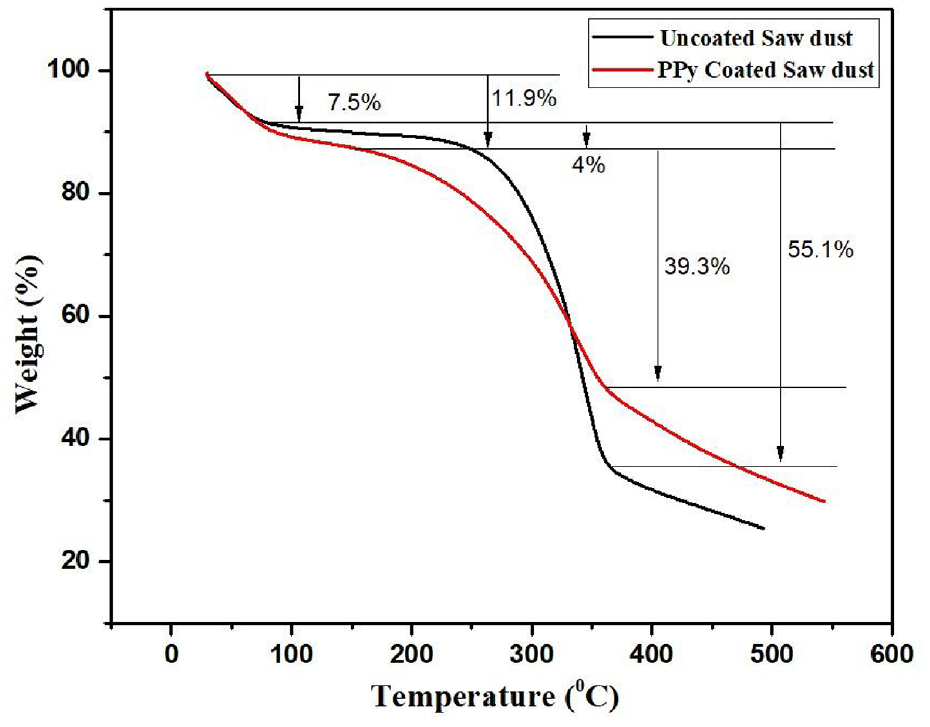
TGA curves of uncoated and PPy-coated sawdust.
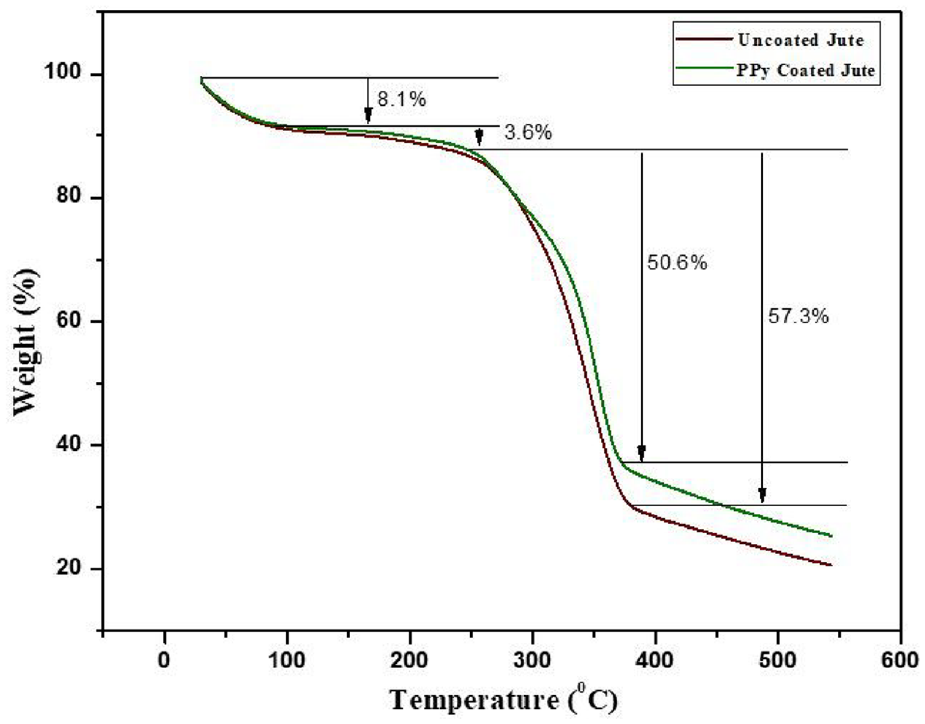
TGA curves of uncoated and PPy-coated jute fibre.
DSC Analysis of SD and Jute
DSC is used to investigate the thermal characteristics of uncoated and PPy-coated SD, as shown in Figure 9. The uncoated and PPy-coated SD show endothermic second-order transition peaks at 90.13 and 88.45°C, respectively. This is the glass transition of the material, which is shifted at a higher temperature after PPy coating along with a higher enthalpy of 12.15–64.44 J/g. This denotes significant intermolecular interaction between cellulosic SD and PPy molecules.
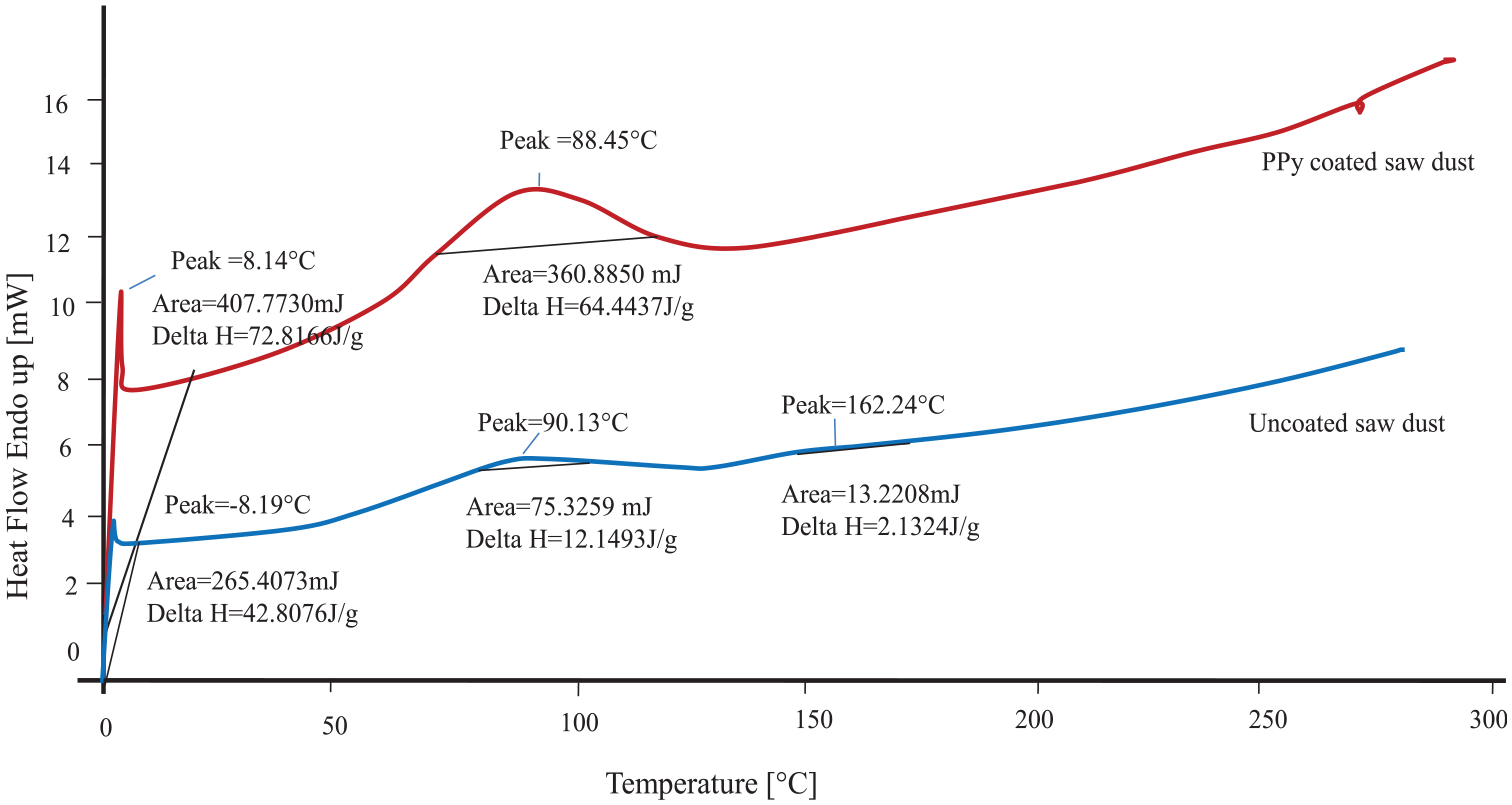
DSC curves of uncoated and PPy-coated sawdust.
The uncoated and PPy-coated jute show endothermic second-order transition peaks at 81.63 and 85.58°C, respectively, as shown in Figure 10. This is the glass transition of the material, which is shifted at higher temperatures after PPy coating along with higher enthalpy from 64.05 to 73.79 J/g. This denotes significant intermolecular interaction between cellulosic jute and PPy molecules.
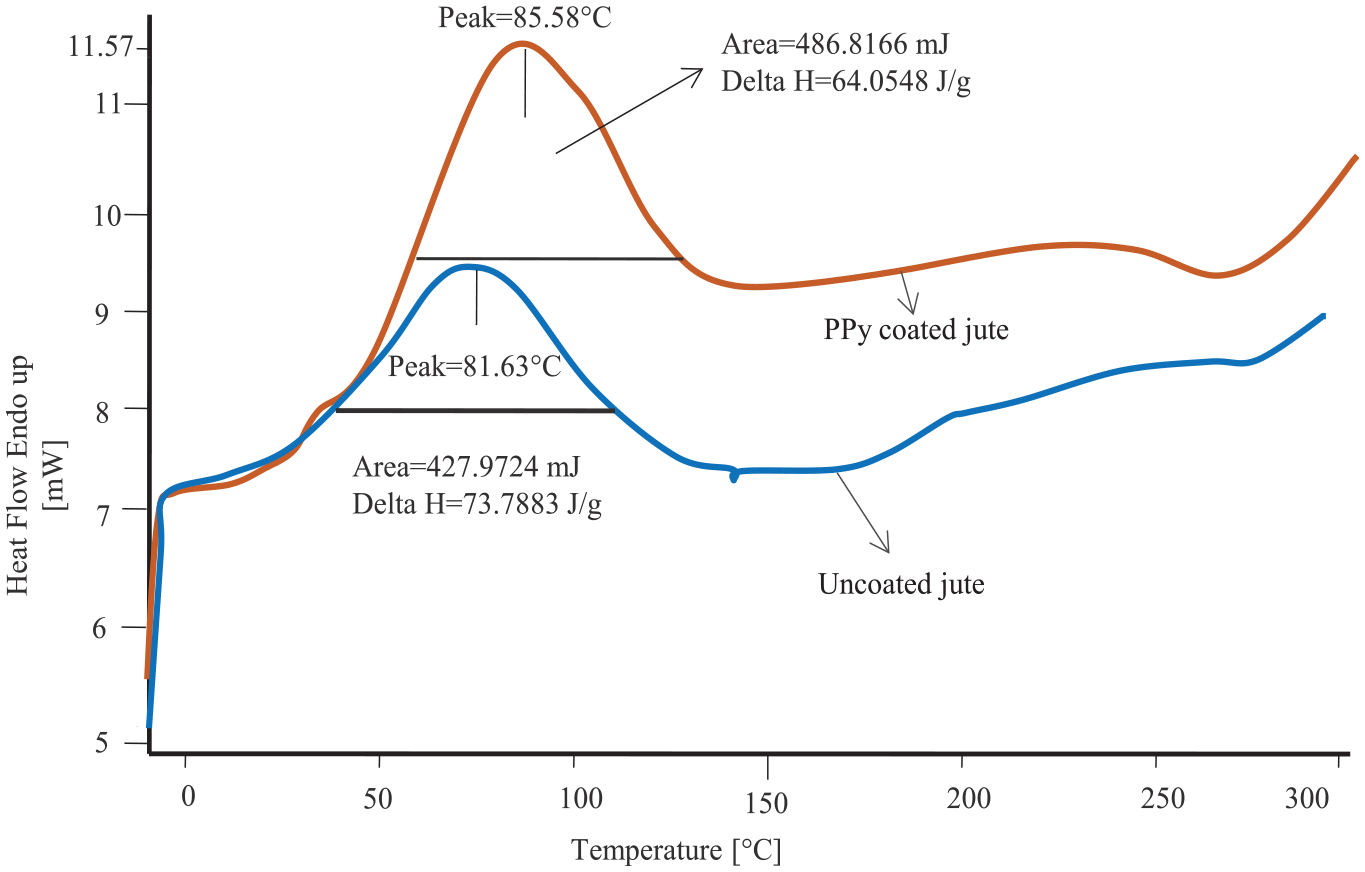
DSC curves of uncoated and PPy-coated jute.
Effect of Pyrrole Concentration on As Removal Efficiency
Three different pyrrole concentrations (0.125, 0.25, 0.50 and 1.0 M) are used during in situ chemical polymerization, keeping the FeCl3 concentration fixed at 0.5 M for coating both jute and SD. These PPy-coated adsorbents are used for water treatment for different treatment times ranging from 1 to 15 min. The effects of the change in pyrrole concentration on the As removal efficiency of PPy/jute and PPy/SD are shown in Figures 11 and 12, respectively. It can be seen that the As removal efficiency increases as the pyrrole concentration increases for both the adsorbents. The maximum removal efficiency of about 93.36% has been achieved in the case of PPy/jute with 1 M pyrrole concentration, whereas, in the case of PPy/SD, the maximum As removal efficiency is observed at about 78.36%, which is significantly lower than that of the PPy/jute. The higher level of removal efficiency at a higher level of pyrrole concentration may be due to a higher level of PPy add-on, which can adsorb more amount of As from water. PPy/jute has a higher level of PPy add-on than PPy/SD, which leads to higher As adsorption from water.
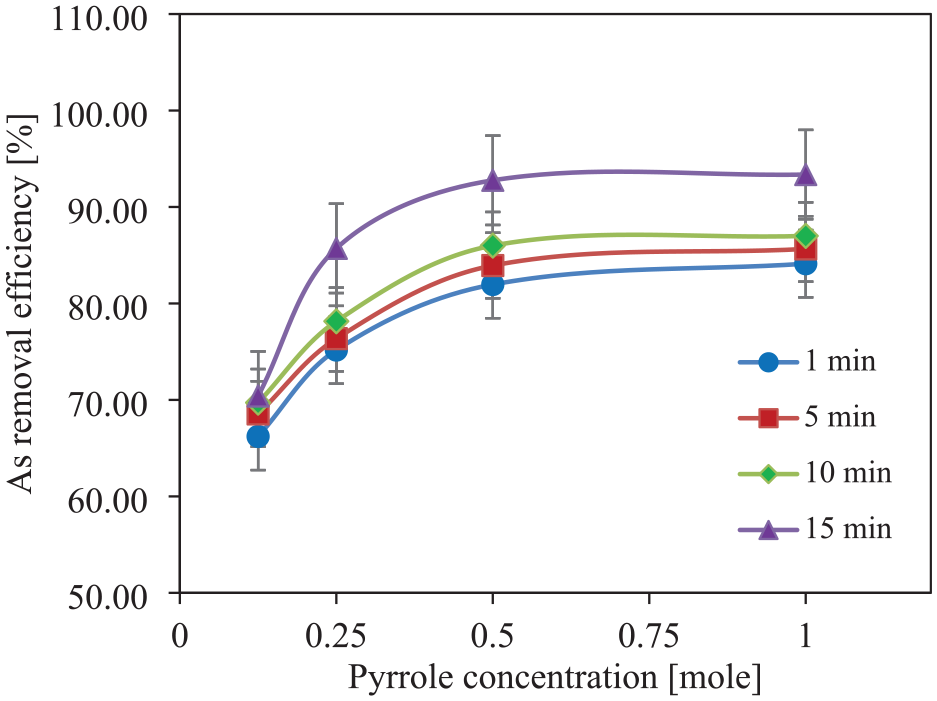
Effect of pyrrole concentration on As removal efficiency by PPy-coated jute.
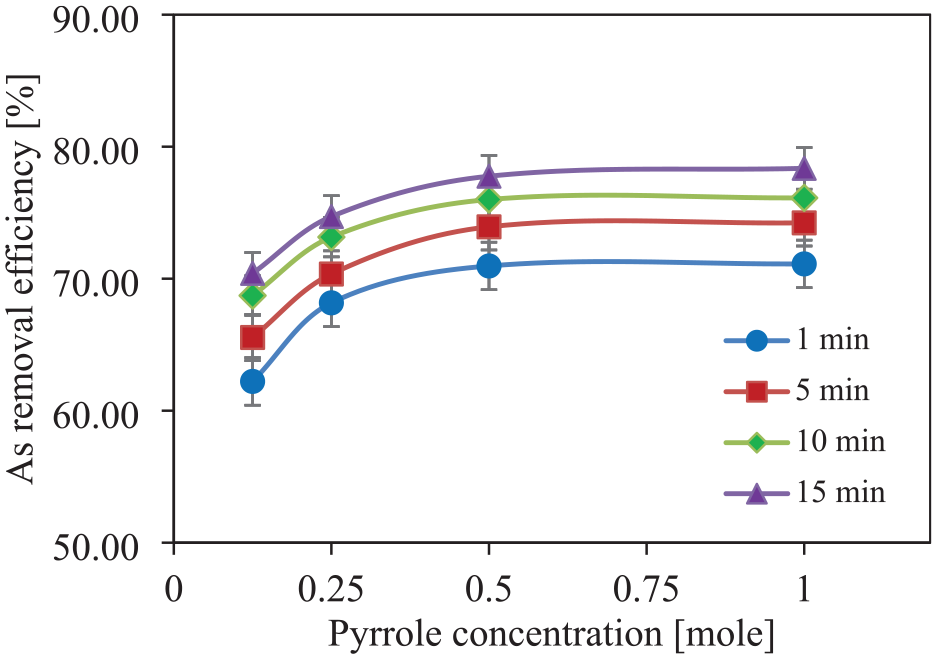
Effect of pyrrole concentration on As removal efficiency by PPy-coated sawdust.
Effect of Treatment Time on As Removal Efficiency
Water treatment time is thought to have a significant impact on the effectiveness of As removal. Figure 13 displays the outcomes of using PPy/jute and PPy/SD adsorbents (prepared with 1 Py, 0.5 M FeCl3) for varying treatment times, which range from 1 to 20 min, to remove As from water. It is observed that the majority of the As is removed from the water within 5 min, with the adsorption of As starting very quickly. Once the adsorption reaches a high level, it then slows down and becomes flattened after 15 min. Due to a higher PPy add-on, PPy/jute exhibits a significantly better As removal efficiency than PPy/SD.
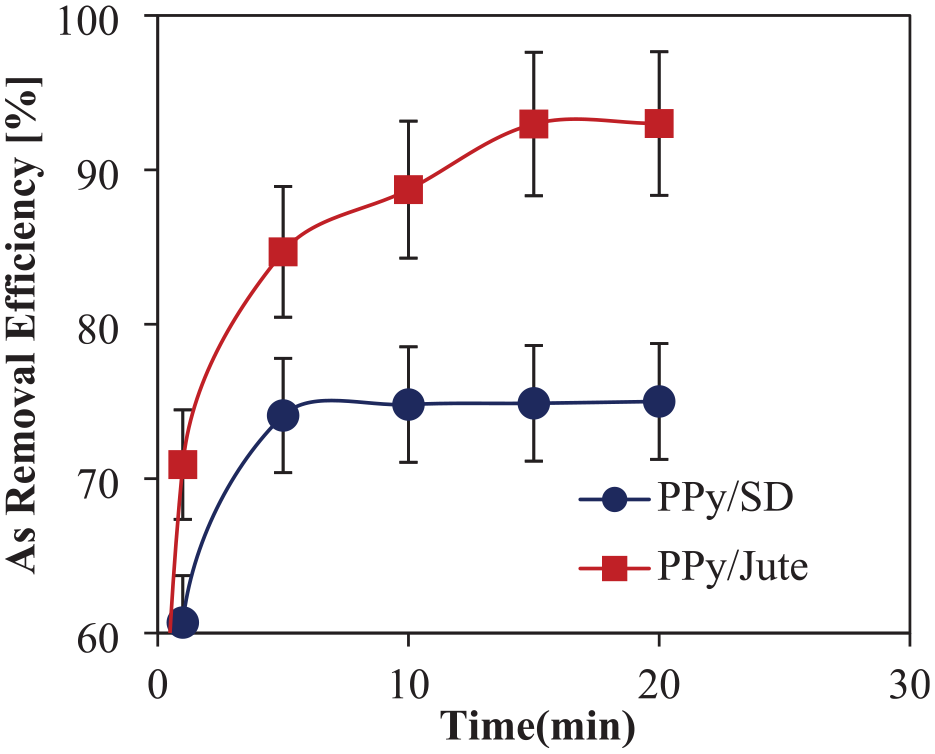
Effect of treatment time on removal efficiency of As.
Effect of Dosages on Removal Efficiency of Arsenic
The effect of dosage amount of PPy/jute and PPy/SD on the removal efficiency has been studied by varying dosage from 0.2 to 1.2 g during water remediation treatment. The PPy-coated adsorbents are prepared with 1 M pyrrole and 0.5 M FeCl3, and the water treatment time is 15 min. The removal efficiency is calculated for both the adsorbents, and the results are shown in Figure 14. It can be seen that as the dosage increases the As removal efficiency increases and attains an equilibrium after 0.8 and 1.0 g dosages for PPy/SD and PPy/jute, respectively, which are found to be their optimum dosages for the water remediation treatment. The highest As removal efficiencies of 97.3 and 82.5% are found in the case of PPy/jute and PPy/SD, respectively, after a treatment time of 15 min. The As removal efficiency is found to be better than the previously reported results of 85% observed with poly 3-methylthiophene coated SD. 4 There are probably more active sites available for adsorption, which explains why the removal efficiency increases as the adsorbent dosage increases. The removal efficiency is not enhanced by an additional increase in PPy dosage, which may be because nearly all of the arsenic ions are adsorbed on the adsorbent and reach equilibrium with the solution. Numerous other researchers have demonstrated a strikingly similar pattern of how the dosage of an adsorbent affects various heavy metal ions. 27
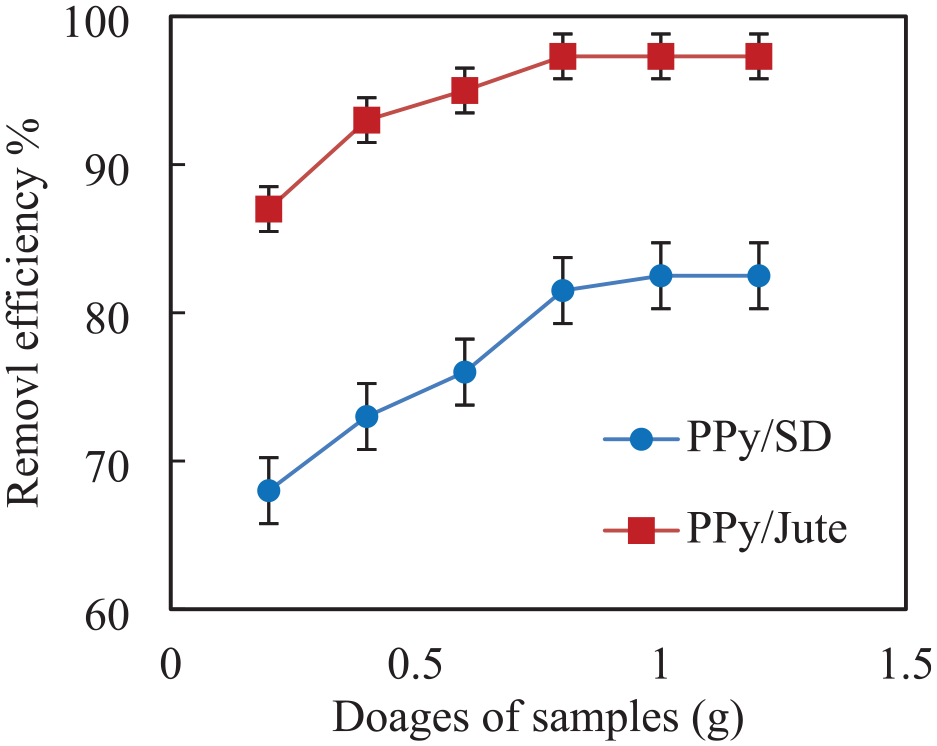
Effect of PPy/jute and SD dosages on the adsorption of As from aqueous solution.
PPy intercalated with Cl− ion possesses great ion exchange capability due to the smaller size and high mobility of the Cl− ions. In the presence of FeCl3 as an oxidant, the in situ polymerization reaction is initiated by the oxidation of pyrrole monomers into radical cations, which combine to form bipyrrole. Continuation of the process leads to the formation of PPy, which is insoluble in water. As a result, PPy precipitates from the solution, and in the process, a part of it is deposited on the surface of the textiles by adsorption. This intercalation of anions of Cl− has the ability to perform anionic exchange of AsO2− ions which are present in water. The scheme of adsorption of As by PPy is shown in Figure 15.
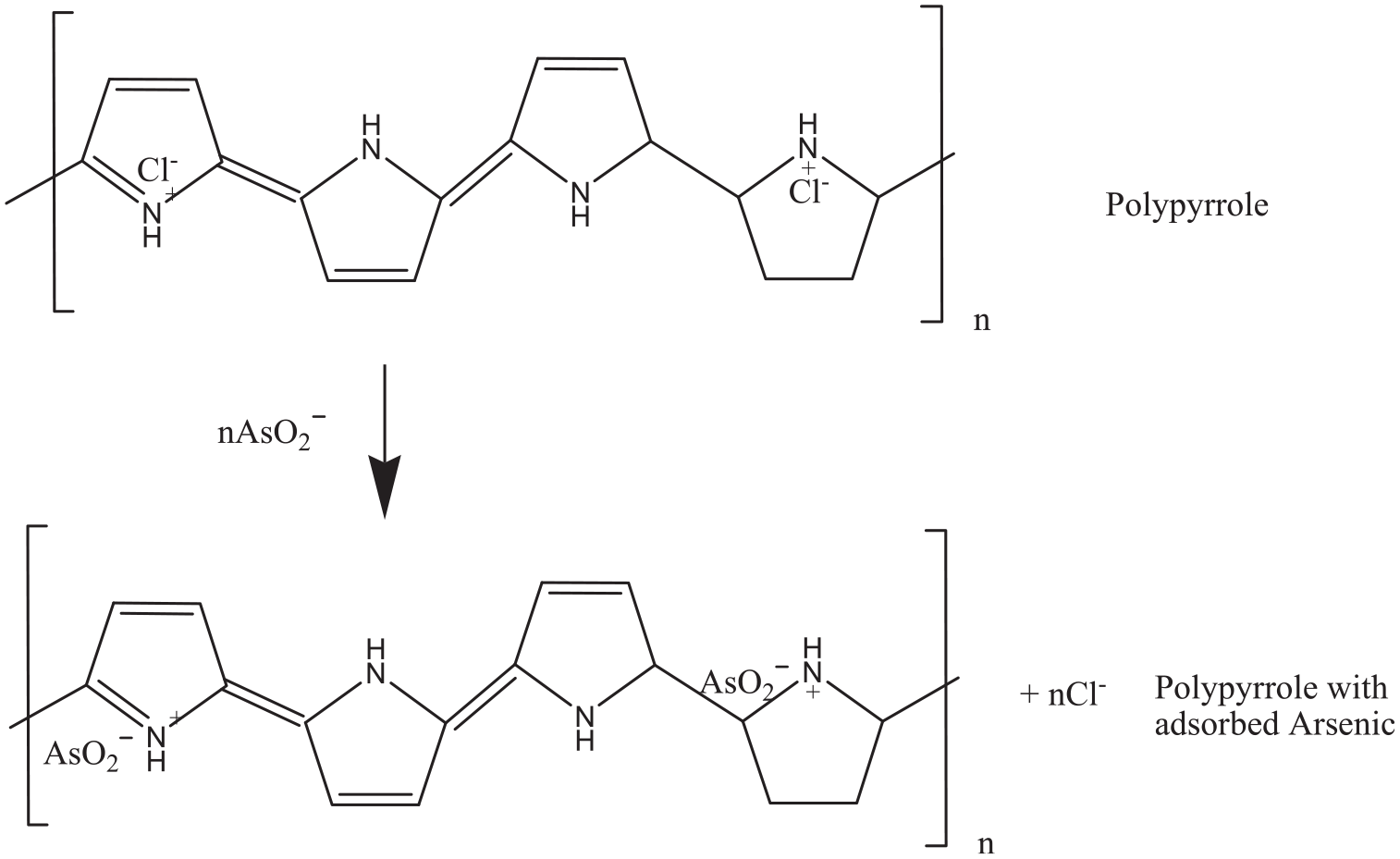
Adsorption of AsO2 – ions by PPy.
Conclusion
PPy-coated adsorbents (jute and SD) can be prepared easily by in situ chemical polymerization in aqueous media. PPy/Jute has demonstrated a significantly higher adsorption efficiency than PPy/SD. As the pyrrole concentration during in situ polymerization increases, the As removal efficiency increases for both the adsorbents. The majority of the As ions are observed to be adsorbed from the water within 5 min, showing a rapid adsorption trend initially followed by a plateau. It is evident that the removal efficiency increases with dosage and reaches equilibrium at 0.8 and 1.0 g/50 mL, for PPy/SD and PPy/jute, respectively, which are determined to be their optimum dosages for the water remediation treatment. The maximum adsorption efficiencies of PPy/jute and PPy/SD for arsenic (As) from contaminated water are found to be 97.3 and 82.5%, respectively, from a 7.5 ppm As solution after a treatment time of 15 min with a dosage of 1 g/50 mL water. The thermal analysis of the PPy-coated jute and SD shows good thermal stability. DSC and FTIR studies reveal significant intermolecular interaction between PPy molecules and cellulosic jute and SD, which is responsible for the durable coating of PPy on their surface. The SEM images show uniform coating of PPy on an individual jute fibre surface and the SD surface in granular form.
Therefore, it is observed that this water remediation technique is fast, sensitive and beneficial in reducing heavy metals like arsenic contamination from water. It is also cost-effective and environmentally benign. It can be concluded that PPy/jute and PPy/SD can be useful in removing harmful As from normal domestic and municipal water.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
