Abstract
Manganese-oxidizing fungi support bioremediation through the conversion of manganese ions into manganese oxide deposits that in turn adsorb manganese and other heavy metal ions from the environment. Manganese-oxidizing fungi were immobilized onto nanofiber surfaces to assist remediation of heavy metal–contaminated water. Two fungal isolates, Coniothyrium sp. and Coprinellus sp., from a Superfund site (Lot 86, Farm Unit #1) water treatment system were incubated in the presence of nanofibers. Fungal hyphae had strong association with nanofiber surfaces. Upon fungal attachment to manganese chloride–seeded nanofibers, Coniothyrium sp. catalyzed the conformal deposition of manganese oxide along hyphae and nanofibers, but Coprinellus sp. catalyzed manganese oxide only along its hyphae. Fungi–nanofiber hybrids removed various heavy metals from the water. Heavy metal ions were adsorbed into manganese oxide crystalline structure, possibly by ion exchange with manganese within the manganese oxide. Hybrid materials of fungal hyphae and manganese oxides confined to nanofiber-adsorbed heavy metal ions from water.
Introduction
The release of heavy metal contaminants into groundwater poses a serious threat to human and environmental health.1,2 Techniques used for heavy metal removal from water include chemical precipitation reactions, ion exchange, and adsorption. 3 Chemical precipitation is most widely used industrially because the reactions are simple and its cost is relatively low. Heavy metal cations react with hydroxide or sulfide anions to form water-insoluble salts. However, toxic by-products, such as low-density sludge and hydrogen sulfide (H2 S) fumes, can persist upon reaction. 3 Ion exchange is a highly efficient, high-capacity heavy metal removal technique. Heavy metal ions attach to polymeric beads or fibrous filter media as they replace protons belonging to sulfonic acid (–SO3 H) or carboxylic acid (–COOH) groups on the surface. 4 Adsorption has many advantages over other technologies due to its effectiveness, flexible design, and low cost.2,3
Biogenic manganese oxide (Mn(III/IV)O) 5 and fungal cells 6 are well-known bioadsorbents for heavy metals. Manganese (Mn(II))-oxidizing fungi are found throughout aquatic and terrestrial environments; biogenic Mn(III/IV)O naturally participates in the environmental remediation.7–10 These ascomycetes were found to oxidize Mn(II) 11 —Acremonium, Alternaria, Cladosporium, Coniothyrium, Curvularia, Penicillium, Phoma, Verticillium—along with wood-rotting basidiomycetes (e.g. Coprinellus). 12 The exact mechanism of Mn(III/IV)O formation by fungi is not clear; fungi may produce multicopper oxidases to aid Mn(II) oxidation. 11
In general, biogenic Mn(III/IV)O has superior adsorption properties compared to synthetically produced Mn oxides. Unit cell forms of MnO6 octahedra reassemble layered (e.g. birnessite) or tunneled structures (e.g. todorokite). 8 Most biogenic birnessite contains a significant amount of Mn(III) in the octahedral layers. 8 Mn(III/IV)O has an abundance of vacancies, which lends to its highly adsorptive nature.7,13 Dissolved salts are often found throughout the interstices of layered Mn(III/IV)O. 8
Heavy metal removal is influenced by the surface area of adsorbents. Thus, nanoscale particles are expected to have greater adsorption efficiency per mass than microscale adsorbents that are 100–10,000 times their size. 2 Nanoparticle separation and contamination is a major challenge to its use in water purification. 14 Therefore, high surface area nanofibers are presumed the best platforms for particle confinement and next-generation hybrid materials. 15
Electrospinning technique produces nanofibers from polymeric solutions that are placed under high voltage. Nanofibrous adsorbents are formed by electrospinning dopes containing nanosize particles 16 or dopes containing sol–gel precursors for their synthesis.17,18 Also, inorganic particles are synthesized at the surface of nanofibers hydrothermally 19 or by the sol–gel approach for particle synthesis along nanofibers. 20 For example, Park et al. 21 seeded electrospinning dopes with polymerized micelles to nucleate the hydrothermal growth of calcium carbonate (CaCO3) spheres—which threaded the nanofibers. The CaCO3-coated nanofibers adsorbed anthraquinone, acid blue dye from water. Wu et al. 18 fabricated composite nanofibers of silicon dioxide (SiO2), functionalized with thiol groups, that were capable of >400 mg/g of copper ion (Cu(II)) adsorption.
Thus, hybrids of Mn(III/IV)O-yielding fungi and nanofibers are posed as environmentally sound approaches for heavy metal removal from water. This novel approach immobilizes bioadsorbents along the surface of nanofibers. The implications of this innovative approach are to support the scalable use of nanotechnologies toward groundwater treatment. Mn(III/IV)O morphologies, as catalyzed by Coprinellus species (sp.) and Coniothyrium sp., were investigated. Coprinellus sp. and Coniothyrium sp. are plant pathogens, but neither of them poses serious threats to human health. 22 The adsorptive capacity of Coniothyrium sp. hybrids was tested against Mn(II), Cu(II), and lead (Pb(II)). The US Environmental Protection Agency’s (US EPA) secondary maximum contaminant levels for Mn(II) is 0.05 mg/L. 23 US EPA action levels are 1.3 mg/L Cu(II) and 0.015 mg/L Pb(II) in drinking water. If one-tenth of tap water exceeds either action level, then additional treatments must be used to amend water systems. 24
In this study, heavy metal adsorbents were fashioned from hybrids of electrospun nanofibers and Mn(II)-oxidizing fungi. Mn(II) oxidation and deposition along the surface of immobilized fungi and nanofibers were tested against growth conditions for Coprinellus sp. and Coniothyrium sp. After-ward, the efficacy of nanofibrous hybrids was tested for heavy metal adsorption in water charged with high concentrations of Mn(II), Cu(II), and Pb (II).
Materials
Strains designated C1CAP-d1AYA and ASB2-d1KA, classified as Coprinellus sp. and Coniothyrium sp., respectively, were isolated from the activated carbon column (CC) and air stripper (AS) of Superfund site, Lot 86, Farm Unit #1 remediation system in Raleigh, NC.25,26 Dextrose, yeast extract, casamino acids, (4-(2-hydroxyethyl)-1-piperazineethanesulfonic) acid (HEPES), agar, calcium chloride (CaCl2), magnesium sulfate (MgSO4), manganese(II) chloride (MnCl2), iron(III) chloride (FeCl3), copper(II) sulfate pentahydrate (CuSO4·5H2O), zinc sulfate heptahydrate (ZnSO4·7H2O), CoCl2·6H2O, Na2MoO4·2H2O, and 88% hydrolyzed poly(vinyl alcohol) (PVA, having a molecular weight of ~130 kDa), 70% glutaraldehyde (GA) in water, and 1 mol/L sulfuric acid (H2SO4) were purchased from Sigma-Aldrich. Groundwater samples were donated by Wake County government in North Carolina.
Methods
Nanofiber preparation
Neat PVA and PVA/MnCl2 nanofibers were electrospun from aqueous dopes. Therefore, 11 wt% of PVA was dissolved in 100 mL of water under constant stirring for 1 h at 60°C. PVA/MnCl2 was prepared from a 100 mL solution of 11 wt% PVA and 0.1 g of MnCl2.
PVA and PVA/MnCl2 nanofibers were electrospun at 15 kV, horizontal tip to collector distance of 8 cm, and feed rate of 0.8 mL/h using an 18-gauge needle. Nanofibers were collected onto aluminum mesh at room temperature and 40%–50% relative humidity.
Crosslinking nanofibers
PVA and PVA/MnCl2 nanofibers were crosslinked in GA solution. A 300 mmol/L GA in water/acetone was prepared from 70% aqueous GA stock and 50 mL of acetone. One drop (~50 µL) of H2SO4 catalyst was added. Nanofibers of 4 × 5 cm2 were immersed into GA solution for 24 h at room temperature. Afterward, nanofibers were air-dried at room temperature for at least 24 h before use.
Media preparation
Liquid Leptothrix media, with and without Mn(II) ions, were prepared in three steps. In Step 1, 1 g of dextrose, 0.5 g of yeast extract, 0.5 g of casamino acids, and 2.38 g of HEPES acid were added to 1 L of distilled water. Afterward, in Step 2, this solution was autoclaved for 30 min and cooled to 60°C using a room-temperature water bath. Finally, in Step 3, 0.5 mmol/L CaCl2, 0.83 mmol/L MgSO4, 1 mmol/L MnCl2, 3.7 mmol/L FeCl3, and 1 mL of trace metal mix were added to prepare Leptothrix media having Mn(II) ions. Leptothrix media without Mn(II) did not have 1 mmol/L MnCl2 added in Step 3.
For solid leptothrix media preparation, 15 g agar was added in Step 1 with the same steps. Trace metal mix was prepared by adding 10 mg CuSO4·5H2O, 44 mg ZnSO4·7H2O, 20 mg CoCl2·6H2O, and 13 mg Na2MoO4·2H2O in solution. Afterward, the trace metal mix was filtered through sterile 0.2 µm porous filters.
Fungal treatment of nanofibers
Leptothrix culture media were inoculated with Coprinellus sp. and Coniothyrium sp. in the presence of nanofibrous sheets (of 1 × 1 cm2 or 4 × 5 cm2). Nanofibers and fungal plugs were simultaneously added to liquid media. On solid plates, fungal plugs were placed on top of the nanofibers, under sterile conditions. PVA and PVA/MnCl2 nanofibers were immersed in 20–30 mL of Leptothrix culture media in 75 mL Erlenmeyer flasks. Fungal growth over 4 weeks occurred at room temperature in the dark.
The fungi were removed from the fungi-nanofiber hybrids by immersion in 20 mL of deionized water, stirring at 500 r/min for 30 min, or sonication in 20 mL of water or ethanol for 2 h. Table 1 summarizes the samples used throughout this study and their descriptions.
Summary of nanofibers with and without fungal treatment.

PVA: poly(vinyl alcohol).
Microscopic analysis
Confocal micrographs of fungi incubated on Leptothrix solid plates and in the presence of nanofibers were imaged on the LEXT OSL4000 3D. The morphology of fungi-nanofibers and nanofibers were observed using field emission scanning electron microscopy (FE-SEM, FEI, Verios 460L) at an operating voltage of 2 kV. Specimens were sputter-coated with gold and palladium before SEM analysis. Image analysis of SEM micrographs was used to calculate the average fiber diameter from at least 50 nanofibers.
Spectroscopic analysis of Mn(III/IV)O
The surface chemistry of nanofibers and their mycogenic hybrids were characterized by energy dispersive spectroscopy (EDS, attached to the Verios 460L). The operating voltage was 20 kV. EDS spectra were used to confirm the locale of metal oxides and absorbed heavy metals.
The NomadicTM Raman Microscope by BaySpec was used to analyze Mn(III/IV)O nanofibers. Raman spectra were collected using the 532 nm laser at 150 mW power, 10X objective lens, 20 scans, and 1 s exposure time.
Heavy metal adsorption
Heavy metal sorption by PVA/MnCl2, Mn(III/IV)O-#17, Mn(III/IV)O-#17-PVA/MnCl2, and Mn(III/IV)O-PVA/MnCl2(-#17) were measured. In total, 3 mg of each sample was added to 20 mL of the heavy metal ion solution.
Specimens were immersed in heavy metal solutions (of 0.55 mg/L Mn(II), 2.63 mg/L Cu(II), and 0.029 mg/L Pb(II)) at pH 7 for 24 h at room temperature. Inductively coupled plasma optical emission spectrometer (ICP-OES, Perkin Elmer 8000) was used to measure unabsorbed concentrations of heavy metal ions.
Adsorption

Results and discussion
Fungal growth and Mn(III/IV)O deposition
Figure 1 summarizes the processes of fungal growth and Mn(III/IV)O formation. Photographs of fungi grown in Leptothrix media revealed white, cotton ball-like mycelia (Figure 2(a) and (b)). Dark brown deposits, indicative of Mn(III/IV)O,9,27 formed after 1 week of Coprinellus sp. and Coniothyrium sp. incubation in Mn(II)-containing media (Figure 2(a) and (b)). Adding PVA/MnCl2 nanofibers into media without Mn(II) did not yield the Mn(III/IV)O deposits (Figure 2(a) and (b)). The average diameter of PVA nanofibers without MnCl2 was 186 ± 116 nm, and PVA/MnCl2 nanofibers was 126 ± 46 nm (Figure 2(c) and (d), respectively).

Fungus grows from (a) spores that germinate into (b) fungal cells; fungal cell grows into elongated cells of (c) hypha; (d) then hyphae aggregate to form mycelium that can reproduce fungi through the release of spores. Mn(II)-oxidizing fungi secrete enzymes that catalyze the conversion of Mn(II) into Mn(III/IV), as observed by dark brown mineral deposits of Mn(III/IV)O.

Fungi (a) Coprinellus sp. and (b) Coniothyrium sp. were grown for 1 week in Leptothrix media (a(i)–(iii), b(i)–(iii)) without Mn(II) or (a(iv)–(vi), b(iv)–(vi)) with Mn(II). Fungi were incubated (i, iv) without nanofibers or (ii, v) with PVA or (iii, vi) with PVA/MnCl2 nanofibers (of 1 × 1 cm2). SEM images of (c) PVA and (d) PVA/MnCl2 nanofibers are shown. Photographs show (e) Coniothyrium sp. grown for up to 4 weeks in Mn(II)-containing media the 4 × 5 cm2 sheet of nanofiber (either (i)–(iv) PVA or (v)–(viii) PVA/MnCl2). This growth pattern was also observed for Coprinellus sp.
Fungal growth in the presence of nanofibers and Mn(II) was monitored over 4 weeks (Figure 2(e)). Incubation beyond 2 weeks did not enhance the spread of Mn(III/IV)O deposits along nanofibers. Nevertheless, nanofibers were maintained in culture media for more than 4 weeks to obtain fully grown fungi. 9 PVA/MnCl2 nanofibers were more densely covered with mycelia.
PVA/MnCl2 nanofibers yielded more mycelia growth, and hyphal structures tended to adhere more strongly to nanofibers in comparison to neat PVA nanofibers that were immersed in Mn(II)-containing media (Figure 2(a) and (b)). The role of Mn(II) for Mn(III/IV)O was evident from fungi grown on the solid plates which are placed in supporting information. Likewise, more mycelia and Mn(III/IV)O grew on PVA/MnCl2 nanofibers when grown on Leptothrix solid plates (see Supplemental Figure S1). Confocal microscopy images (see Supplemental Figure S2) showed Mn(III/IV)O deposits only on PVA/MnCl2 fibers cultured in Mn(II)-containing media.
The attachment of Coprinellus sp. and Coniothyrium sp. mycelia to nanofibers was confirmed by inspection of SEM images (Figure 3). Mn(III/IV)O morphology can differ between each fungal species. 9 Coprinellus sp. catalyzed the growth of spherical Mn(III/IV)O particles when cultured in Mn(II)-containing media. Spherical Mn(III/IV)O particles were more prominent along the hyphae of PVA/MnCl2 nanofibers than on neat PVA nanofibers. Coniothyrium sp. catalyzed the growth of sheet-like Mn(III/IV)O in Mn(II)-containing media. Extracellular matrix appeared at the interface of nanofibers and Coniothyrium sp. hyphae. Round spores were observed as seen in Figure 3. Thus, nanofibers did not inhibit fungal reproduction by spores.
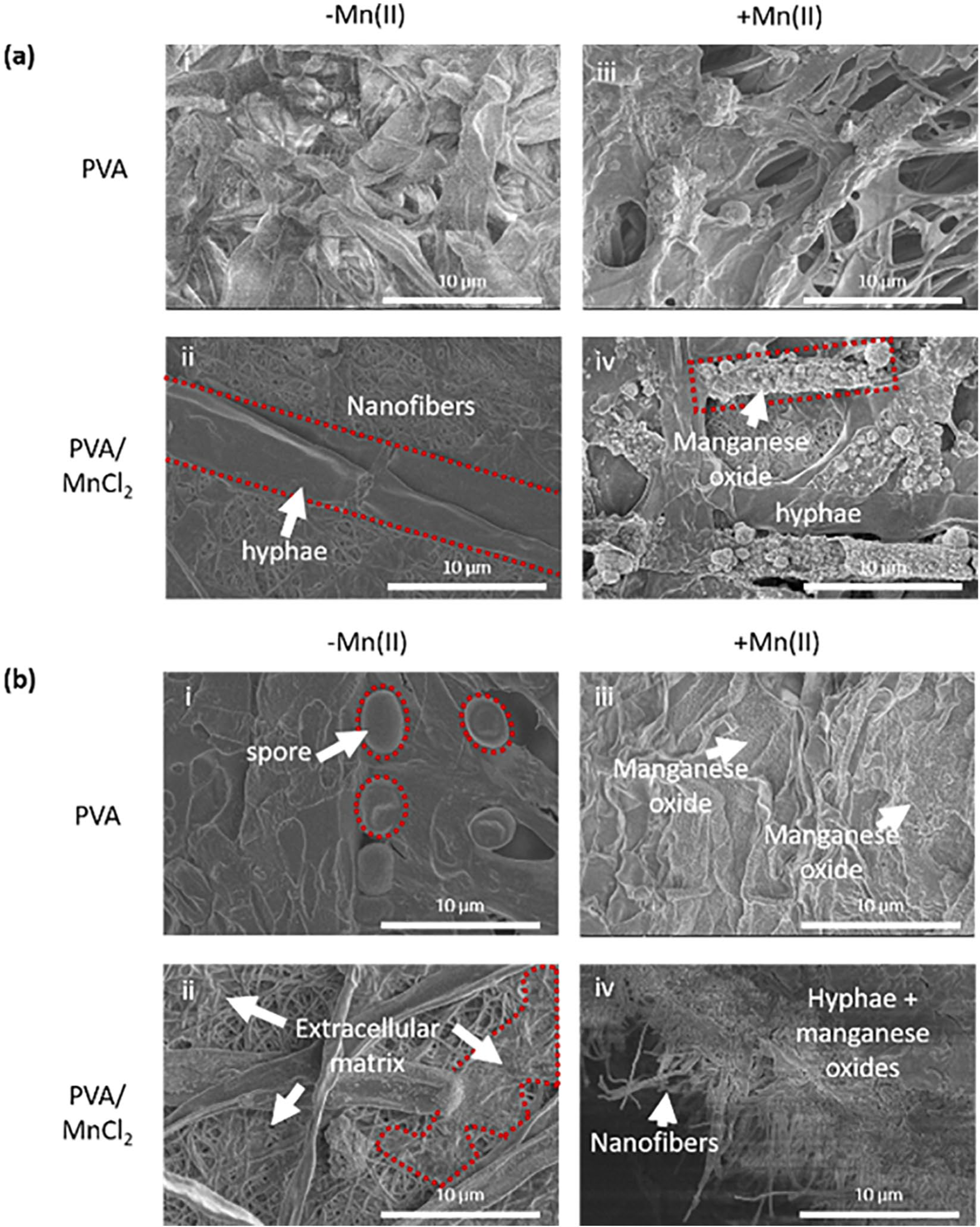
Micrographs show (a) Coprinellus sp. (b) Coniothyrium sp. grown on (i, iii) PVA and (ii, iv) PVA/MnCl2 nanofibers immersed in liquid media (i, ii) without Mn(II) (−Mn(II)) or (iii, iv) with Mn(II) (+Mn(II).
Fungi can secrete various oxidative enzymes as their hyphae continue to lengthen. This results in the cycling of nutrients throughout the environment. 28 Mn(II)-oxidizing fungi express extracellular oxidative enzymes, such as Mn(II) peroxidase. This group of proteins is capable of oxidizing Mn(II) to Mn(IV) at accelerated rates of Mn biomineralization: several orders of magnitude faster than either abiotic catalysis on mineral surfaces or homogeneous oxygenation in aqueous solution.12,29 Coprinellus sp. and Coniothyrium sp. apparently have different modes of Mn(II) oxidation. Mn(II)-oxidizing enzymes associated with the hyphae cells of Coprinellus sp. In contrast, enzymes were secreted from Coniothyrium sp. hyphae. Oxidizing enzymes were captured by the extracellular matrix, which exuded from Coniothyrium sp. and onto the surrounding nanofibers (Figure 3(b)). Santelli et al. 9 reported that locale of Mn(III/IV)O deposition (i.e. on hyphal surfaces or extracellular polymer adjacent to cells) varies among different ascomycete fungi probably due to primary placement of enzymes.
Elemental analysis of fungal Mn(III/IV)O hybrids
Mn(II) within PVA/MnCl2 fibers was not detected by EDS because Mn(II) at the surface of PVA/MnCl2 nanofibers was below the EDS detection limit, that is, 0.1 wt%. Therefore, any Mn(II) detection must result from the oxidation of Mn(II) in solution. With both fungal types, Mn(II) content as measured by EDS was qualitatively higher along PVA/MnCl2 than neat PVA nanofibers (Figure 4). EDS spectra showed impurities of Fe, S, Mg, and Ca within Mn(III/IV)O. As expected, Mn(III/IV)O can adsorb other metal ions from Leptothrix media. Ivarsson et al. 27 detected non-Mn(II) metal ions within the vacancies of biogenic Mn(III/IV)O by EDS. Coniothyrium sp. can release enzymes coupled with P, S, K, and Ca.

EDS graphs of (a) Coprinellus sp. and (b) Coniothyrium sp. on PVA (i, ii) or PVA/MnCl2 nanofibers (iii, iv) in liquid media (i, iii) without Mn(II) (−Mn(II)) or (ii, iv) with Mn(II) (+Mn(II)).
Mn(III/IV)O deposition on nanofibers
A layer of fungal hyphae intermeshed with nanofibers is illustrated in Figure 5. As hyphae are grown on the surfaces of nanofibers, their filament-like structures are entangled with each other to form a layer. However, cracks formed in the topical layer of Coniothyrium sp. after stirring hybrids immersed in water (Figure 5(a)). The cross-sectional view shows hyphae attached to both sides of nanofibers in a sandwich structure (Figure 5(b)). After sonicating the sample for 1 h in water, some fungal hyphae remained on the nanofibers (Figure 5(c)). But sonicating the fungi-nanofiber hybrids for 2 h in ethanol removed most of the fungal hyphae (Figure 5(d)).

SEM images of Mn(III/IV)O-#17-PVA/MnCl2 after water immersion and magnetic stirring for 30 min at 500 r/min: both (a) longitudinal and (b) cross-sectional views are shown. Hybrids were further treated with (c) 1 h of sonication in water or (d) 2 h of sonication in ethanol.
This latter technique was used to remove fungus from hybrids that were incubated in Mn(II)-containing media. A conformal Mn(III/IV)O coating was found on the fiber surface after removing hyphae by 2 h of ethanol sonication (Figure 6(d)). Mn was not detected on the surface of PVA and PVA/MnCl2 nanofibers that were incubated with Coprinellus sp. (Figure 6(a) and (b)). Also, Mn was not identified on the surface of neat PVA nanofibers that were incubated with Coniothyrium sp. (Figure 6(c)).

Fiber surface after removing fungal hyphae by 2 h of sonication in ethanol was observed by (i) SEM and (ii) EDS spectra. Coprinellus sp. incubated with (a) PVA and (b) PVA/MnCl2 nanofibers are shown. Coniothyrium sp. incubated with (c) PVA and (d) PVA/MnCl2 nanofibers are shown.
Heavy metal ion removal by Coniothyrium sp.-nanofiber hybrids
Coniothyrium sp.-nanofiber hybrids with and without fungal hyphae were compared with Mn(III/IV)O-coated fungi and PVA/MnCl2 nanofibers in terms of heavy metal removal in laboratory charged water. Water samples were charged with 0.55 mg/L Mn(II), 2.63 mg/L Cu(II), and 0.029 mg/L Pb(II) at levels twice as high as the US EPA’s “secondary maximum contaminant levels” for drinking water. Each nanofiber sample was capable of removing Mn(II), Cu(II), and Pb(II) from laboratory charged solutions (Table 2). The lowest amounts of heavy metal adsorption occurred by PVA/MnCl2 nanofibers. It is noteworthy that hyphae-nanofiber derivatives removed heavy metals as well if not better than Mn(III/IV)O-coated hyphae alone (Table 2). Both Mn(III/IV)O-#17-PVA/MnCl2 and Mn(III/IV)O-PVA/MnCl2(-#17) removed more than 90% of the Mn(II) from the water (i.e. > 150 mg/L) in 24 h, then charged water was well below the US EPA limit. The nanoscale dimensions of Mn(III/IV)O-coated nanofibers appear to enhance Mn(II) sorption capacities. Taffarel and Rubio 30 experimented with hybrid nanotechnologies and reported Mn(III/IV)O-coated zeolite at 4 g/L of water removed 90% of the Mn(II) from water within 2 h.
Comparing aqueous heavy metal adsorption Coniothyrium sp. derived Mn(III/IV)O coatings and nanofibers after 24 h.

PVA: poly(vinyl alcohol).
Hybrid nanofibers were capable of removing the largest amount (mg/g) of Mn(II) from water but were least capable of removing Pb(II) from water. Molecular size could potentially exclude larger heavy metal ions from absorption by Mn(III/IV)O. Laus and De Fávere 31 also noted the importance of molecular size because Cu(II) was adsorbed by chitosan better than Cd(II). Nelson et al. 32 reported Pb(II) adsorption by bacteriogenic Mn(III/IV)O and synthetic β-MnO2 were 1344.67 mg/g and 0.078 mg/g, respectively. Droz et al. 12 noted that bacteriogenic Mn(III/IV)O (from Pseudomonas putida GB-1) had more structural defects than mycogenic Mn(III/IV)O (from Coprinellus sp.). However, extended X-ray absorption fine structure (EXAFS) spectra examinations reported by Duckworth et al. 26 indicated mycogenic manganese oxides from Lot 86 biofilm (containing these particular isolates) were structurally comparable to bacteriogenic Mn(III/IV)O and with similar binding of Ba(II), Co(II), and Zn(II).
In the interest of having nanofibers scaffold Mn(II)-oxidizing fungi in nature, Cu(II) adsorption by hybrid Mn(III/IV)O-#17-PVA/MnCl2 was tested in the laboratory. Over time, Cu(II) concentration decreased as Mn(II) concentration increased (Figure 7). However, the diffusion of Mn(II) from samples of nanofiber was not a significant source of Mn(II) leaching. Figure 8 shows lower levels of Mn(II) released from samples immersed in deionized water over time. Other heavy metal ions appear to replace Mn(II) among the mineral deposits. But in their absence, Mn(II) is readily adsorbed by Mn(III/IV)O (Table 2).

Cu(II) adsorption and Mn(II) release into Cu(II) solution (at 2.6 mg/L) by Mn(III/IV)O-PVA/MnCl2 (-#17) and PVA/MnCl2 nanofibers over time.
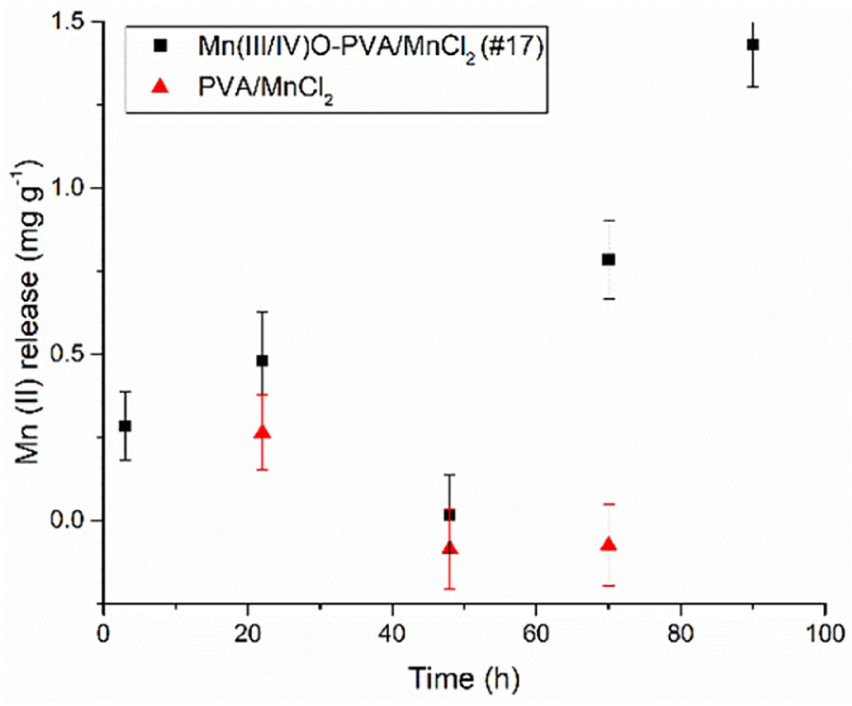
Mn(II) release to deionized (DI) water with nanofibers.
Groundwater is a natural example of water having multiple types of heavy metal ions. Composition of groundwater is shown in Supplemental Table S1 (see supporting information). Therefore, Coniothyrium sp.-nanofibrous hybrids were immersed in groundwater. The concentration of Zn(II), Fe(II), and Pb(II) decreased in water due to Mn(III/IV)O adsorption. But, Mn(II) increased in the presence of other heavy metal ions (Table 3). Thus, it is important to understand the limitations of Mn(II) removal. Biogenic Mn(III/IV)O formation reduces Mn(II) from the environment; nevertheless, ion exchange between heavy metal ions and Mn(II) within the metal oxides can in turn release Mn(II) into the environment. Therefore, Mn(II)-oxidizing microbials must remain active to continually convert Mn(II) to metal oxide adsorbents.
Comparing heavy metal adsorption over 24 h from groundwater between the control and hybrid Coniothyrium sp. nanofiber.
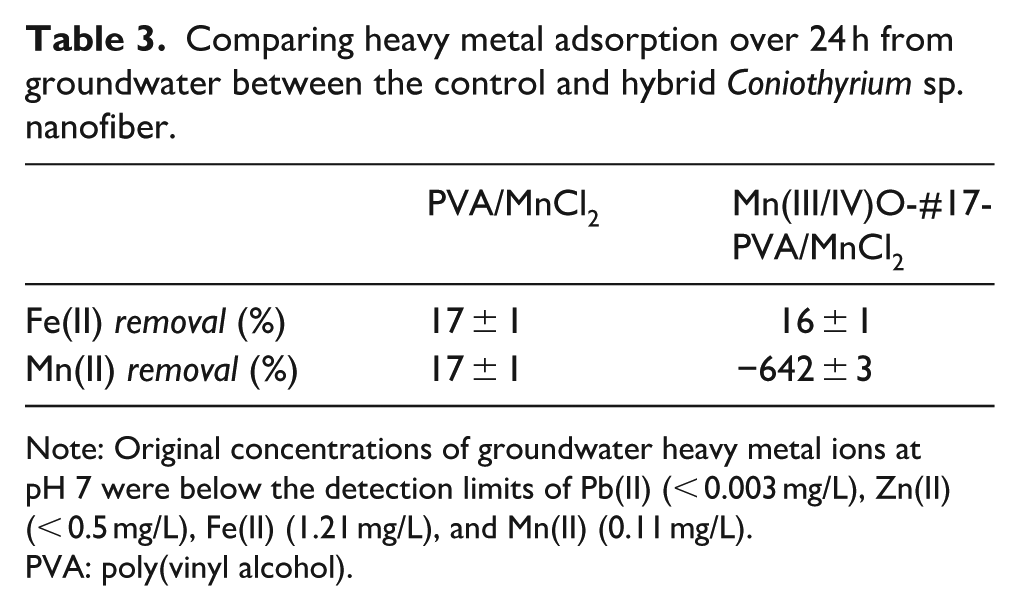
Note: Original concentrations of groundwater heavy metal ions at pH 7 were below the detection limits of Pb(II) (< 0.003 mg/L), Zn(II) (< 0.5 mg/L), Fe(II) (1.21 mg/L), and Mn(II) (0.11 mg/L).
PVA: poly(vinyl alcohol).
Conclusion
Having the nanofibers scaffold Mn(III/IV)O mitigates contamination from unconfined fungus and inorganic particulates in the water. Oxidative enzymes secreted from Coniothyrium sp. and onto the surface of nanofibers. As a result, Mn(II)-oxidizing enzymes coated the surface of nanofibers and catalyzed the nucleation and growth of Mn(III/IV)O along them. For synthetic mineralization, nanofibers are typically seeded with polyelectrolytes to nucleate the growth of inorganic minerals.21,33 Mn(III/IV)O-coated nanofibers and fungi-nanofiber hybrids effectively reduced Mn(II) contamination from laboratory charged water. However, Mn(II) leaching was noticed when Mn(III/IV)O nanofibers were exposed to groundwater containing various heavy metal contaminants.
Supplemental Material
Supplemental_material – Supplemental material for Biohybrid nanofibers containing manganese oxide–forming fungi for heavy metal removal from water
Supplemental material, Supplemental_material for Biohybrid nanofibers containing manganese oxide–forming fungi for heavy metal removal from water by Yaewon Park, Shuang Liu, Terrence Gardner, Drake Johnson, Aaron Keeler, Nathalia Ortiz, Ghada Rabah and Ericka Ford in Journal of Engineered Fibers and Fabrics
Footnotes
Acknowledgements
Yaewon Park’s current address is The Advanced Science Research Center (ASRC) at The Graduate School and University Center of the City University of New York (CUNY). A portion of this work was conducted at Smithsonian Institution. We thank Bruce Stewart, Cara Santelli, Dominique Caput and Owen Duckworth for their contributions at various stages of the overall biogeochemical study. We appreciate Evan Kane at Wake County Department of Environmental Services for the groundwater sample.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Water Resources Research Institute – North Carolina Sea Grant (WRRI-NCSG) provided support for this graduate research (WRRI Project No. 17-02-SG & Sea Grant Project No. R/MG-1708) Work from project 14-177 NC with The Nonwovens Institute was featured in this study. This work was performed in part at the Analytical Instrumentation Facility (AIF) at North Carolina State University, which is supported by the State of North Carolina and the National Science Foundation (award number ECCS-1542015). AIF is a member of the North Carolina Research Triangle Nanotechnology Network (RTNN), a site in the National Nanotechnology Coordinated Infrastructure (NNCI). Support was received from the National Science Foundation Environmental Chemical Sciences Program (award CHE–1407180) and NCSU Research Innovation and Seed Funding program.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
