Abstract
With the development of industrialization and human activities, discharge of waste water containing heavy metals in the environment is increasing day by day. It causes serious threats to the human civilization and the flora and fauna in this earth. Conducting polymers like polypyrrole and polyaniline can be used for treating wastewater due to their inherent ion absorption properties. This article has been focused on the development of polypyrrole coated bio-composites and their potential about the removal of heavy metals from industrial wastewater. Adsorption process can be successfully employed to remove heavy metals from the wastewater by the treatment of water with polypyrrole-coated composites. It was reported in literature that the polypyrrole and polyaniline-coated adsorbents had good adsorption capacities for Mg, Fe, Cu, Cd, Pb, Zn, and Ni. Polypyrrole-coated saw dust, rice husk, chitin, and cellulosic materials could be able to remove Cr, Fe, Cu, and Zn from wastewater. This method would be a replacement for costly conventional methods of removing heavy metal ions from wastewater. It is expected that this method would be an alternative for waste water treatment which will benefit the industries in future.
Introduction
Contamination of water with heavy metals is a new form of water pollution and becoming a serious threat to all living beings day by day. If the density of any metal exceeds 5 g cm−3, considered as heavy metals [1]. However, any toxic metal can be called as heavy metal irrespective of its density or atomic number. Various industries like mining, leather, textiles, paper mills, fertilizer, pesticide, metallurgy, and surface finishing generate and discharge waste water into the environment that contained various heavy metals. There are three kinds of heavy metals contaminating water viz. (i) toxic metals (such as Cr, Pb, Hg, Cd, As, Ni, Co, Zn, Cu, and Sn), (ii) precious metals (such as Au, Ag, Pt, Pd, and Ru), and (iii) radio nuclides (such as U, Th, Am and Ra). After consuming the contaminated water, the metals are accumulated in human body and cause serious health effects like damage to vital organs, nervous system failure, hormonal imbalance, and in extreme cases cancer. Also, it impairs growth and development of natural vegetation and all other living beings. Thus, metals as contamination bring serious environmental pollution, threatening human health and ecosystem when they are consumed beyond a tolerance limit [2]. Some metals are carcinogenic in nature, which highly damage our body. If the waste water is discharged directly into the natural water, it causes high risk for the aquatic ecosystem. The direct discharge into the soil highly ruined the fertility of soil. The metals which are mainly present in waste water are Zn, Cr, Fe, Ni, Cu, Pb, Cd, and Mg. These metal-containing waste water are directly or indirectly discharged into streams, lakes, rivers, or oceans, especially in developing countries [3,4]. Moreover, the soils surrounding military bases offer a potential risk of certain heavy metals, which can be a threat to groundwater and surface water. The presence of chromium, lead, and cadmium are cited as the most often accruing soil contaminants in military base. These are due to practices in military such as electro-plating, mechanical work, aircraft rebuilding, battery acid disposal, leaking tanks, firing ranges, pistol ranges, open burning of ordnance, and detonation. These heavy metal ions are not biodegradable in nature and when consumed are easily accumulated in living organisms [5,6]. Although consuming a limited amount of different heavy metals is extremely necessary for human beings for the synthesis of vitamins or utilizing as co-factors, excessive exposure or intake can have dangerous consequences. Zinc (Zn) is an essential trace element good for human health. However, excessive Zn can cause serious health issues like stomach cramps, vomiting, nausea, skin irritations, and anemia [7]. Copper (Cu) performs vital work for human metabolism. But the excessive consumption of copper is leading to serious toxicological issues like vomiting, convulsions, cramps, and even death [8]. Chromium (Cr) is available in the aquatic environment mainly in two states: Cr(III) and Cr(VI). In general, Cr(VI) is more toxic than Cr(III) and it can easily penetrate through cell membrane. It accumulates in food chain and consumed by animals and human beings naturally. It therefore affects animal and human physiology and causes severe health issues like skin irritation and lung carcinoma [9–11]. Iron (Fe) can turn clear water to brown and gives water a foul odor, causing sedimentation and turbidity in water. Its excessive exposure is also toxic for our health affecting our taste and skin appearance.
Permissible limits of some heavy metals in drinking water [1].

Therefore, the removal of traces of heavy metals from contaminated water has been a special concern for researchers and environmental scientists. Conductive polymers such as polythiophene (PTh), polypyrrole (PPy), and polyaniline (PANi)-coated sorbents can be the potential materials for the removal of various heavy metal ions from contaminated water [6,14]. Use of these conductive polymers for this purpose is gaining popularity day by day due to their ease of synthesis, biocompatibility, redox, and ion exchange properties. This review article is therefore focused on the physico-chemical properties, adsorption characteristics, mechanism, and efficiency of different PPy-based adsorbents for waste water treatments.
Existing methods of waste water treatment
There are several methods available to reduce heavy metals concentration from waste water. These are physical and chemical precipitation, ion exchange, cementation, coagulation, reverse osmosis (RO), adsorption, etc. as shown in Figure 1 [15–17].
Various methods of waste water treatments.
Precipitation
Precipitation is the popular method for removing highly toxic heavy metals from water up to parts per million (ppm) levels [18]. Some metal salts are precipitated in water when correct anion is added making them insoluble [18]. Upon formation the precipitate can be separated from the water either by sedimentation or by filtration. The treated water is then decanted and approximately discharged or used. This process is economic in nature. Its efficiency is affected by decreasing the pH and the presence of other salts which are ionic in nature. In this process, chemicals are added to the bath, which results in the formation of a high water content sludge. Precipitation with lime water, ion exchange, or disulphide lacks the specific maturity and is ineffective in removal of the metal. It is widely accepted in the mining and metallurgical industrial sector for heap leaching and acid mine drainage. It is also used in various chemical operations related to the metal transformation, electronics, and electrical industries for removal and recovery of metals [19].
Ion exchange method
Ion exchange is one of the best methods used in the industrial sectors for the removal of heavy metals from waste water effluents though it is costlier than other methods. By this method it is possible to clean up the water in parts per billion (PPB) concentration levels of contamination with handling a relatively large volume of water. An ion exchanger equipment is capable of exchanging either cations or anions from the surrounding materials. Commonly used materials for the ion exchange are synthetic organic resins [20]. However, this process is not suitable for ion removal from concentrated metal solution as the matrix gets easily fouled by contaminants and other solids present in the waste water. Moreover, the process is nonselective and is highly sensitive to pH of the solution.
Cementation
In cementation method, an electrochemical mechanism is employed. It is a type of precipitation method, where a metal having a higher oxidation potential is employed to remove a metal of lower redox potential from the solution. The process involves in removal of the toxic metal ions from the waste solution by replacing with a more active nontoxic metal. As an example, Cd++ can be removed from waste water by cementation on zinc according to the reaction:

Copper and noble metals such as Ag and Au are most frequently separated by this cementation method in manufacturing sector. In case of waste water treatment point of view, Pb, As, Cd, Ga, Pb, Sb, and Sn can be removed from contaminated water by this method [21].
Coagulation
The coagulation mechanism is based on zeta potential (ζ) measurement between coagulant and polluting agent. Coagulation process reduces the net surface charge of the colloidal particles to stabilize them by electrostatic repulsion process. Hydrophobic particles present in waste water and the suspended particles present in the solution of hard water are generally removed by coagulation process. Both soluble heavy metal and insoluble substances are efficiently removed by coagulation. Chemicals having the sodium xanthogenate group are grafted to polyethylene and this coagulant has an amphoteric polyelectrolyte that is very reactive and has the property to react with both acid and bases. At lower pH level of water, it can coagulate the colloidal substances having negative charges [22].
Coagulation and flocculation are two different processes; however, they are often used interchangeably. In the coagulation process, coagulants are added to the water to neutralize the suspended particles that are possessing opposite charges that of the coagulant and hence settle at the bottom of the water. In the flocculation process, the size of suspended particle in water is continually increased by adding some inorganic polymers such as flocculants by the mechanism of additional collisions and interaction. When the suspended small particles are flocculated into larger particles, they are separated or removed from water by the method of filtration, straining, or floatation [23]. The demerit of both these process is production of sludge, application of chemicals, and transfer of toxic compounds into solid phase.
Electro-coagulation
Electro-coagulation is an electrochemical process, which uses the phenomenon of flowing of electrical current to remove metallic ions from solution. Electro-coagulation system is also capable in removing dissolved metals, suspended solids, tannins, and dyes present in the solution. The metals present in waste water are in ionic forms possessing electrical charges. These metal ions and other charged particles present in the solution are neutralized by ions of opposite electrical charges provided by this system and hence become destabilized and precipitate from the solution [24]. In this process, an electro-chemical cell is formulated where an electrode assembly, i.e. anode and cathode are connected to an external DC power supply. The efficiency of ion removal depends on mode of combination and material of anode and cathode. There are many physical and chemical reactions occurring in this electro-coagulation process that consumes electrodes to supply ions into the pollutant solution. The coagulating ions are produced in-situ on cathode by dissolution of anode, formation of OH− ions and H2 gas. Electrolytic reactions occur at electrode surfaces only with the adsorption of coagulant on colloidal pollutants and the removal of colloids by sedimentation or flotation. For example, when aluminum is used as anode, Al3+ ions are released from anode and many ionic monomeric hydrolysis species are formed, depending on the pH of the solution. In the process, the reactions take place at electrodes, which are shown in equations (1), (2), and (3):
Oxidation reaction takes place at the anode

Reduction reaction takes place at the cathode
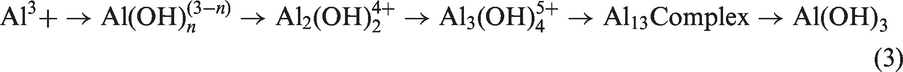
Overall reaction during electrolysis
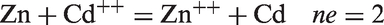
Reverse osmosis
RO is widely used in treating the hardness of water. This technique is also used in purifying the water at home. This is the process in which the pore size of membrane is less than 2 nm and it works based on the principle of size exclusion and solution diffusion with semi permeable membrane in which the passing water has to follow the pathway to escape, resulting from the membrane structure. There are many reports about the removal performance of RO. Under 5 atmospheric pressure, RO can give 99.5% removal efficiency of Cu and Ni [25]. While under 1100 kPa operation pressure, the removal efficiency of Ni and Zn can increase up to 99.3% and 98.9%, respectively [7]. The demerit of this process is the use of high power and membrane regeneration is difficult.
Adsorption
Adsorption is the process where free molecules are forced to accumulate on the surface of the sorbent. The suspended molecules which are present in bulk form in a solution are being adsorbed into the pores of a suitable adsorbent [26–28]. The driving force for adsorption is the ratio of the concentration to the solubility of the compound. Adsorption can be divided into two groups which are physical sorption and chemisorption. Physical adsorption creates weak intermolecular forces, while chemisorption forms essential chemical bond between the sorbent molecule and the surface of the adsorbent [27–29]. On the view of the demerits of other waste water treatment processes as shown in Figure 2, this adsorption process may be a suitable alternative process which would be efficient and economic [30]. This review article covers this adsorption process suitable for heavy metal removal in a more effective way as compared to other processes as mentioned in Table 2.
Scheme of formation of polypyrrole in presence of FeCl3. Various demerits of the existing waste water treatment processes.

PPy as a material of ion absorber
PPy, a conducting polymer has been extensively explored for various potential applications due to its good environmental stability, high conductivity, redox properties, and ease of processability [31–43]. PPy synthesized in aqueous solutions with small size dopants, such as Cl−, ClO4−, and NO3−, exhibits anion exchanging behavior due to the high mobility of these ions through the PPy matrix [44]. PPy intercalated with Cl− ion is possessing great ion exchange capability due to the smaller size and high mobility. Coating with PPy on textile surface is possible by in-situ chemical polymerization in aqueous solution in the presence of oxidant and dopant. In presence of FeCl3 as oxidant, the in-situ polymerization reaction is initiated by the oxidation of pyrrole monomers into radical cations (Figure 2), which combine to form bipyrrole. Continuation of the process leads to formation of PPy which is insoluble in water. As a result, PPy precipitates from the solution and in the process part of them are deposited on the surface of the textiles by adsorption. This intercalation of anion of Cl− has the ability of cationic exchange which is present in waste water as shown in Figure 3.
Scheme of adsorption of metal ion on the surface of nitrogen functional groups in PPy.
However, under certain conditions, cationic exchange is also found to occur with large dopants, such as polyvinyl sulfonate and polystyrene sulfonate, due to immobility of these ions in the polymer matrix [45]. PPy has also exhibited good prospects in adsorption applications because of the nitrogen atoms present in the polymer chains as shown in Figure 2 [46]. PPy-coated porous carbon has improved affinity for heavy metal ions than that of carbon alone. Ions of metals like mercury, lead, and silver are proved to more loving to adhere with PPy-coated carbon surface due to the presence of amine group in PPy. The absorption efficiency of PPy-coated carbon is reported 20 times higher than that of uncoated carbon adsorbents [47].
FeCl3.6H2O is used as oxidant during polymerization of PPy in an aqueous solution using a molar ratio of monomer to oxidant of 1:1. The PPy is explored for ion removal from water by adsorption. In the ion removal process, effects of various parameters like pH of the solution, dosage of the adsorbent, and treatment time on removal efficiency have been investigated. It has been reported that 100% removal of Fe, Cu, and Zn ions from aqueous solution can be possible when the solution is treated with PPy at pH 7.0 [48]. In another study, Fe3O4-coated magnetic PPy nanocomposite is prepared and that can adsorb 100% of Cr(VI) with a dose of 200 mg/L aqueous solution at pH 2 [49]. Carbon nanotubes (CNTs) are also explored for the same purpose, where PANi, PPy, and their nanocomposites with CNTs are prepared. The nanocomposites are found as excellent absorber of heavy metals, anions, and COD from paper mill waste water [50].
Therefore, waste water treatment is one of the novel applications of conducting polymers, particularly of PPy. Various researchers have tried in many ways to remove heavy metal ions from contaminated water by adsorption using PPy prepared via the chemical oxidative polymerization of pyrrole in the presence of different substrates and recipes. The ion adsorption efficiency is found to be largely dependent on the substrate on which PPy is deposited and conditions of in-situ synthesis of PPy onto the substrate.
PPy-coated bio-adsorbents
The adsorption of heavy metal ions using various PPy-coated bio-adsorbents is a very promising method in terms of low cost, renewable sources, ecofriendly, and bio-compatibility [51]. The major advantage of these bio-adsorbents is their excellent removal efficiency at cheaper cost. As inexpensive materials the mostly researched bio-materials for this purpose are saw dust (SD), rice husks, chitin, coconut coir, carbon, etc., which are coated with PPy and have been explored as potential adsorbents for heavy metals [52–54].
PPy-coated wood SD composites
SD is an abundant by-product of wood industry and easily available at low cost. It is primarily a cellulosic material that contains various organic compounds with polyphenolic groups. Therefore, it can be easily coated with PPy to obtain PPy/SD composites, which can adsorb heavy metal ions from water [55]. Preparation of PPy-coated SD is demonstrated in Figure 4. First, oxidant and monomer solutions are prepared separately by adding 5 g FeCl3 and 1 mL pure pyrrole to 100 mL of deionized water in two separate beakers and stirring by a magnetic stirrer. Then, 1 g of SD is added to oxidant solution and monomer solution is added. The mixture is kept on stirring for 4 h at room temperature. As a result, oxidative polymerization of pyrrole commences to form PPy which is coated on the surface of SD. Finally, the PPy-coated SDs are filtered from the solution and washed several times with deionized water and dried at room temperature [56].
Preparation of PPy-coated SD composites as bio-adsorbent.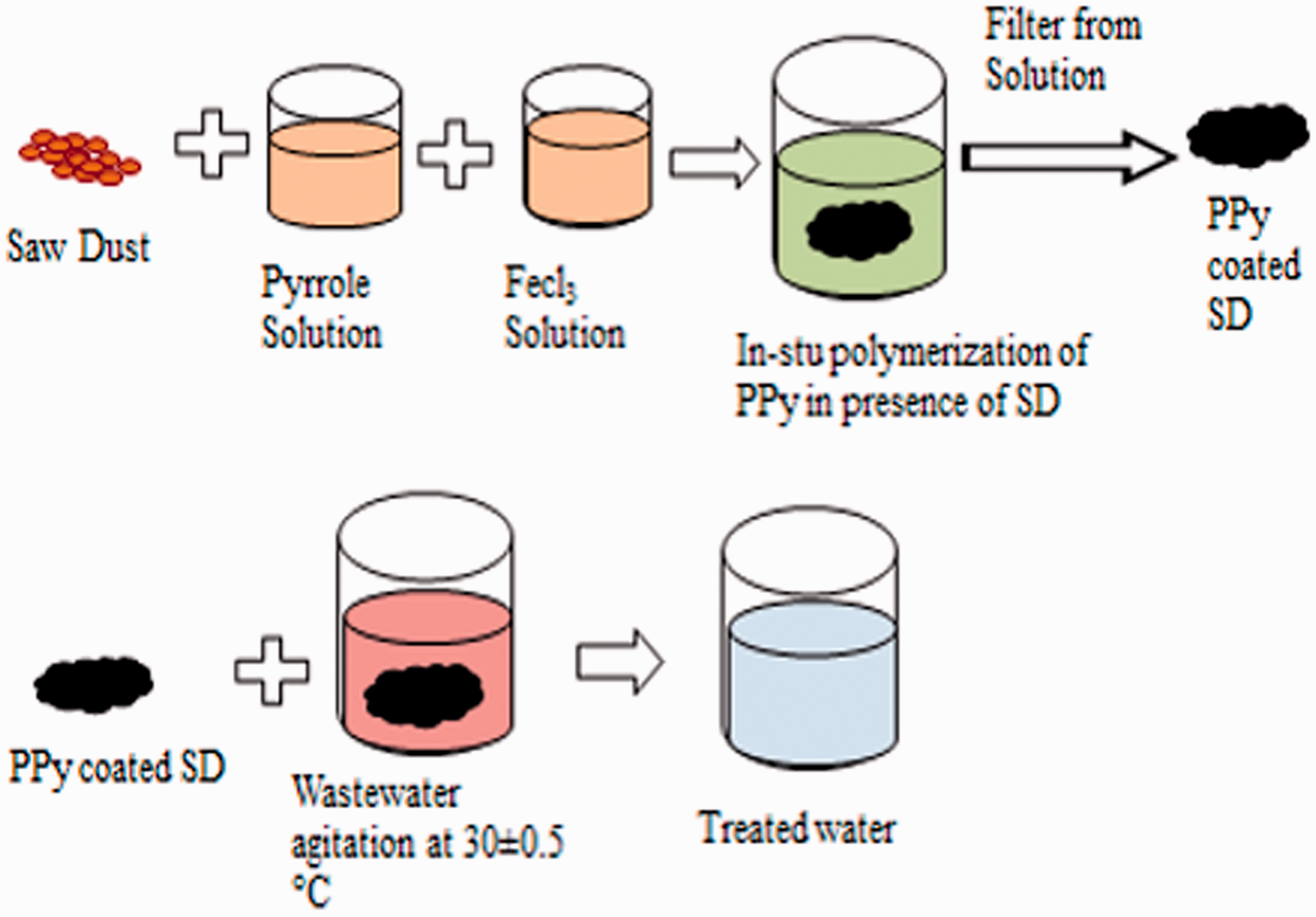
Several experiments are conducted on the removal of Zn(II) and Cr(VI) ions from water by PPy/SD composite. It is observed that PPy/SD composite has considerable potential for the removal of Zn(II) ions from water using a batch method [56–59]. The optimum conditions for sorption are found to be a dose of 0.5 g in 100 mL of contaminated water, a contact time of 14 min, pH 3.0, and at temperature of 40℃. Under these conditions, the maximum removal efficiency of Zn(II) is found to be 94.4%. The study also demonstrated about 96.4% of effective adsorption of Cr(VI) from water. Ansari and Fahim have prepared PPy/SD composite, where FeCl3 is used as oxidant for the oxidative polymerization of pyrrole [59]. They demonstrate that PPy/SD composite can remove Cr(VI) ion from aqueous solutions by a batch or column adsorption method. In the batch method, the maximum removal efficiency is reported to be at least 98% by the dose of 1.0 g PPy/SD in 25 mL water containing 100 ppm Cr(VI) within 15 min at pH 5.0 and 25℃ temperature. In the column method, a maximum of 98.6% removal efficiency is reported by the dose of 1.0 g PPy/SD in a glass column at 2 mL min−1 flow rate.
The Cr(VI) removal is proposed to be mainly due to the anion exchange properties of PPy, which is formed as PPy+/Cl− where Cl− ions which are intercalated in PPy backbone are responsible for the ion exchange mechanism [59]. In another study, PPy, PANi, and poly 3-methyl thiophene (P3MTh) are coated on the surface of SD and explored for removal of arsenic ions (As3+) from aqueous solutions. It is reported that PTh-based conducting polymers are better sorbent for removal of As(III) ions from aqueous solutions [60]. Polyaniline coated on saw dust (PANi/SD) is very easy and simple to prepare and can be used as an effective adsorbent for removal of Cd(II) ions from aqueous solutions [61].
PPy, PANi, and P3MTh are synthesized chemically and in-situ coated on the surface of wood SD, then used for removal of arsenic ions (As3+) from aqueous solutions. They propose complex processes such as anion exchange, chemical oxidation, chelation as possible reactions which are occurred during As(III) removal. In this investigation, 25 mL of As(III) solution containing 100 ppm concentration is treated with different amounts of sorbent ranging from 0.1 to 1.0 g for 1 h at room temperature with shaking. The effect of variation of sorbent dosage on the removal of As(III) is shown in Figure 5 [62].
Effect of SD, PPy/SD, PANi/SD, and P3MTh/SD dosage on the adsorption percentage of As(III). pH = 11, and C0 = 100 ppm [62].
Polypyrrole/rice husk ash (PPy/RHA) composites
Rice husk ash (RHA) is a solid obtained after burning rice husk, which is another natural abundant by-product of rice milling, low cost, and easily available. A PPy/RHA nanocomposite is prepared by coating the RHA with PPy using chemical oxidative polymerization [63-64]. Their ability in the removal of heavy metals from waste water is investigated by several researchers. Ghorbani and Eisazadeh have prepared fixed bed column of PPy/RHA composite of various length 10, 20 and 30 cm and tested for metal removal efficiency with a flow rate of 1.0 mL min−1 [50]. Another study reports the percentage efficiency of Fe, Cu, and Cd ions removal is 95.33%, 96.4%, and 92.4%, respectively by PPy/RHA composites. Color (absorbance, at 600 nm) and COD removal efficiency of the same composites are found to be 97.54% and 97.15%, respectively, using 0.5 g adsorbent dosage with only 25 min of contact time at room temperature [65].
Polypyrrole/cellulosic fiber (PPy/CF) composites
The fully bleached softwood kraft pulp (CF) is coated with PPy to prepare PPy/CF composites [65]. These composites are explored for detoxification of Cr(VI)-contaminated water. The desorption results and X-ray photoelectron spectroscopy analyses show that the highly toxic Cr(VI) is reduced to less toxic Cr(III) and then adsorbed onto the composite [64,66]. The Cr(VI) concentration of the treated water decreased significantly with the increase of the amount of coated PPy (on CF); it is also evident that PPy can play an important role in lowering the concentration of the Cr(VI) as shown in Figure 6.
Effect of PPy concentration on chromium removal from water [65].
Polypyrrole/Chitin (PPy/Ch) composites
The use of chitin alone as an adsorbent for heavy metals appears to be unsatisfactory due to its poor solubility in common solvents, poor stability, and low sorption capacity. Therefore, it has been introduced to PPy structure to enhance the sorption capacity of this conducting polymer as well as preparation of bio-adsorbent of metal species. In the line, polypyrrole functionalized chitin (PPy/Ch) is synthesized by in-situ polymerization and its adsorption capacity for Cr(VI) ions under certain conditions is measured by using batch experiments. This study reports a maximum adsorption capacity of 35.22 mg Cr(VI) by 1 g of PPy/Ch from a contaminated water of 50 mg L−1 concentration of Cr(VI) at 50℃, pH 4.8 with 60 min of contact time. Another study is carried out to investigate the possibility for the removal of Pb(II) and Cd(II) ions from an aqueous solution by the same adsorbent [67]. The maximum removal was obtained at pH 6 for both Pb(II) and Cd(II) ions with an adsorption capacity of 9.14 and 6.49 mg g−1, respectively.
Heavy metal adsorption capacity on polypyrrole-based materials.

For a comparison among all these PPy-coated bio-absorbents the following conclusion can be made. The RHA is a solid obtained after burning rice husk, which creates air pollution [68]. In PPy/Ch method, as chitin is a poor adsorbent for heavy metals and has poor stability PPy/Ch composite does not give expected results. Therefore, PPy/SD composites are more effective for heavy metals removal.
Effect of process parameters on removal efficiency
Effect of pH of the contaminated water
The chemistry of metals in aqueous solutions is pH dependent. Therefore, the effect of pH on adsorption of metals is subject of investigation in order to achieve maximum removal efficiency. High efficiency in metals removal is observed only under acidic conditions (pH = 1–3). Ansari and Fahim have reported the best pH value of 3 for metal removal using PPy/SD [59]. However, Katal et al. have found a sigmoidal decreasing trend of removal efficiency with increase in pH level from acidic to alkaline as shown in Figure 7 [69]. It can be seen from Figure 7 that up to pH 9, metal removal is not much affected by the pH of the test solution.
Effect of pH on heavy metal removal efficiency from water [69].
Under alkaline conditions, metal removal is significantly affected as alkaline pH causes significant loss of intercalated Cl− ions in PPy. Metal removal by PPy/SD composites is suspected to occur via anion exchange process between the polymer (Cl−) and the toxic metal anions present in the contaminated water. Under alkaline conditions, it is assumed that PPy/Cl is changed into PPy/OH, where Cl− is replaced by OH−. Therefore, the poor removal of Cr(VI) under alkaline conditions (pH > 10) can be due to the weak anion exchange properties of PPy/OH compared to PPy/Cl. Hydroxide anions are more strongly adhered to the positively charged polymer matrix compared to chloride ions, and as a result the anion exchange phenomenon is adversely affected [70]. Due to the dependence of the chemistry of arsenic species to pH, sorption percentage of arsenic by SD/PPy composites is a pH-dependent process. Higher removal of As(III) is observed at alkaline pH values [60]. In another study, Lei et al. [65]have found different results from what is discussed above. This difference in the effect of pH may be due to the use of different substrates onto which the PPy is coated. For example, the lignin and extractive contents of SD are substantially higher than those of the CF used by them. At a pH of lower than about 3, the Cr(III) concentration in the treated water increased significantly with the decrease of pH, indicating the chemical conversion of Cr(VI) to Cr(III) as shown in Figure 8. The enhanced Cr(VI) reduction at low pH is due to the high redox potential of Cr(VI) as a result of the presence of a large amount of hydrogen ions in the aqueous system.
Effect of initial pH of Cr(VI)-contaminated water on Cr(VI), Cr(III), and total Cr concentrations of treated water [65].
Effect of treatment time
For the analysis of the effect of treatment time, a fixed amount of the sorbent (1 g PPy/SD) is immersed into 25 mL of metal solution with concentration of 100 ppm at pH 5 at different equilibration times (5–45 min). As per in a recent study, the rate of metal sorption by PPy/SD is high and the removal is occurred effectively. More than 50% of metal was removed within 10 min. Although increasing the exposure time generally leads to a higher rate of metal removal, the majority of metal removal occurred at the initial contact time (within 10–15 min) [71]. With increasing reaction or exposure time, a gradual increase in arsenic removal is observed by SD, PPy/SD, PANi/SD, and P3MTh/SD as shown in Figure 9 [60]. An exponential decrement trend of Cr(VI) with time has been reported in case of PPy/CF composites [65].
Effect of contact time on the adsorption of As(III) by SD, PPy/SD, PANi/SD, and P3MTh/SD [60].
In the initial stages, a higher rate of metal removal efficiency by the conductive polymer-coated SD sorbent is observed. This is attributed to the abundant availability of active binding sites on the sorbent, and with gradual occupancy of these sites, the adsorption gradually reduced in the later stages [60].
Effect of temperature on adsorption
Effect of solution temperature on adsorption of total chromium by PAni-coated jute fibers is investigated by conducting a set of experiments [57]. It is reported that at 298 K temperature 43% of total chromium is removed from water, whereas at 325 K only 17% total chromium is removed. Decrease in chromium removal with the rise in temperature attributed to an exothermic reaction of total chromium adsorption by the adsorbent [57].
Effect of size of saw dust
For studying the effect of SD size on removal efficiency, SD in different sizes (20–50 µm, 100–150 µm, 1–2 mm, 20–30 mm) are coated with conductive polymers and explored for waste water treatment. It has been reported that with decrease of SD size from 20–30 mm to 1–2 mm the removal efficiency gradually increased up to a certain limit and afterwards decreases [56]. A SEM image of SD is shown in Figure 10. When the size of SD particles decreases specific surface area increases and as a result capacity of ion adsorption by PPy increases. However, for very small SD particle amount of PPy add-on would be affected due to lack of penetration inside core, as a result, adsorption capacity reduces.
SEM image of (a) uncoated and (b) PPy-coated SD [56].
Effect of dosage on removal efficiency
It has been reported that 98% removal of total chromium can be possible from a contaminated solution of 100 mg/L of Cr(VI), when the amount of dose of PANi/jute composite used is 30 g/L [57]. In another study, it is reported that 100% removal efficiency can be achieved when 1 g PPy/CF composite is added to 100 mL of 100 mg/L Cr(VI)-contaminated water [65]. The required dosage of PPy/DS for a quantitative removal of Cr(VI) by 50 mg L−1 is 24 g [59].
Regeneration of PPy-coated adsorbent
The concept behind regeneration of these bio-adsorbents is to regain their capacity of ion absorbency so that they can be reused and their life cycle can be extended. Researchers proposed three chemical treatment processes of regeneration till date based on the desorption mechanism of adsorbed metal ions. These three regeneration operations are treating PPy/SD composites using (i) 1 M NaOH solution, (ii) simply deionized water, or (iii) 1 M H2SO4 solution. After these regeneration processes, composites are utilized for waste water treatment and regeneration efficiency is measured [56]. It has been observed that desorption efficiency using NaOH solution in general is better than desorption efficiency achieved either by treatment with deionized water or by treatment with H2SO4 solution. However, color and COD desorption efficiencies using distilled water is found to be best among these three methods. Desorption efficiencies for metals like Fe, Mg, and Cu are found better after treatment with H2SO4 solution. Whereas, desorption efficiency of Zn is best found after treatment with NaOH solution [56]. In another study, desorption of Cr(VI) from water is performed at different concentrations of NaOH ranging from 0.1 to 1.0 M. In the first cycle, only 14% of the adsorbed Cr(VI) is extracted using 0.5 M NaOH. However, upon treatment with 2 M HCl, the remaining amount of Cr(VI) are reduced to less harmful Cr(III) [49]. Ninety-nine percent of Cr(VI) have been removed from PPy/SD by a column prepared with 0.1 M HCl solution. The column is again regenerated by treating with alkali solution [59].
Conclusions
This review article offers the extensive information on the removal of different heavy metal ions from waste water effluents using a wide range of PPy-based adsorbents. The effects of various parameters associated with the adsorption of heavy metal ions from aqueous solutions are compiled and discussed. They have some potential advantages and limitations. They exhibit various advantages, such as fast kinetics, high capacity, and preferable sorption towards heavy metal ions in aqueous streams/waste water. Nevertheless, to further promote the practical applications of PPy-based polymers in the abatement of heavy metal pollution, there still exist some technical constraints to be solved. To date, from a commercial point of view, no greater success has been achieved using conducting polymer-based adsorbents, particularly, PPy and its composites. Extensive research is imperative to materialize the commercial success of conducting polymer-based adsorbents, which will provide a new dimension in adsorption technology towards mitigating the environmental pollution problem.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
