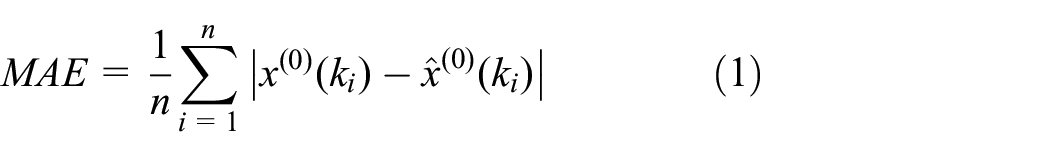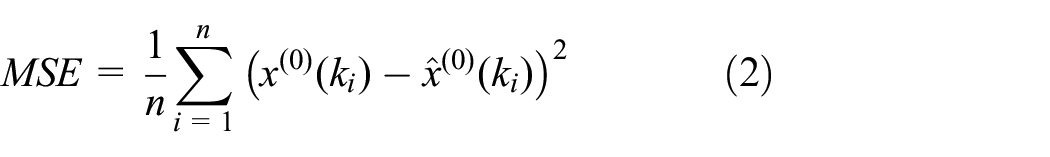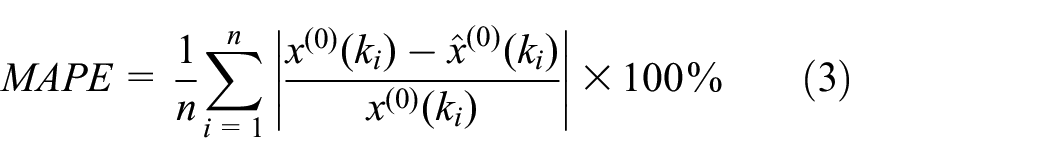Abstract
Despite the mandatory use of medical disposable protective clothing to protect against infectious hazards, how to determine optimal safe working hours induced by physical fatigue while wearing medical disposable protective clothing remains unknown. Driven by these questions, here we quantify the extent of physical fatigue experienced by medical staff wearing medical disposable protective clothings in isolation wards to identify a safe work duration. Eight healthy males were subjected to light (2.1 km/h) and moderate (4.3 km/h) treadmill exercises while wearing two different ensembles. Four physiological (tympanic temperature Tcore, mean skin temperature Tsk, heart rate, and sweat loss) and two subjective indicators (thermal sensation and humidity sensation) were measured. We then introduced a discrete gray model(1,1) to determine safe working hours. The study indicated that even at lower temperatures, prolonged wearing of medical disposable protective clothing could significantly affect physiological indicators such as Tcore and Tsk (p < 0.001), with Tcore being the predominant factor limiting safe working hours. Regarding medical disposable protective clothing-1-light, medical disposable protective clothing-1-moderate, medical disposable protective clothing-2-light, and medical disposable protective clothing-2-moderate intensity activities, the safe working hours were 6.33, 2.83, 2.83, and 2.25 h. This article presented a new approach to determining safe working hours for wearing medical disposable protective clothing from physiological thermal limits with small sample data. However, this is a preliminary study, and further validation of the prediction model through additional experiments is necessary.
Keywords
Introduction
Medical disposable protective clothing (MDPC) is essential during public health emergencies to curb the spread of infectious diseases, including the severe acute respiratory syndrome (SARS) (2003), H1N1 (2009), Middle East respiratory syndrome (MERS) (2012), Ebola (2014), Zika (2016), and coronavirus disease 2019 (COVID-19) (2020) pandemics.1,2 Despite their widespread use, we lack a quantitative understanding of the fatigue experienced by medical staff when wearing airtight MDPCs during prolonged, high-intensity work, which often lasts up to 6 h or more. This is of significant concern since prolonged use of MDPCs may induce metabolic fatigue, cause physiological strain, or even lead to pathophysiological incidents, even within climate-controlled operating rooms where isolation gowns cannot prevent sweating.3,4 Given that traditional shift schedules do not always align with physiological needs and the increasing concern regarding medical staff fatigue, optimizing work durations to minimize fatigue under specific environmental conditions becomes paramount. However, limited research exists to establish safe wearing hours for MDPCs based on thermal limits, and a validated threshold for identifying early warning signs of fatigue remains unknown.
MDPC-induced heat strain among medical staff in hot environments has attracted much attention in recent years; however, few studies have explored their thermal fatigue in air-conditioned hospital environments. Various heat stress indices have been proposed to assess heat exposure across different climates, including prominent indicators such as the wet bulb globe temperature (WBGT),5,6 predicted heat strain (PHS),7,8 predicted mean vote (PMV), 9 Universal Thermal Climate Index (UTCI), 10 thermal work limit (TWL), 11 total heat loss (THL), 12 and heat tolerance limits (HTL). 13 While WBGT is a widely used index in hot environments worldwide, it does not account for wind speed when calculating the sweat evaporation rate. Further research must elucidate its relationship with human core temperature and physiological variations. Some scholars14–16 have proposed predictive methods based on extensive laboratory and field research data to address this issue, but these methods are constrained by their lack of continuous monitoring capability, objectivity, and high cost. Personal monitoring of physiological strain, encompassing parameters such as body core and skin temperatures, heart rate (HR), and sweat rate, offers a real-time means of quantifying heat strain and has been recommended for providing individualized protection against heat-related fatigue. Recent technological advancements in wearable physiological monitors have spurred subsequent research into their utility.
It is evident from the studies above that research on protective clothing in hot environments has made significant strides. Nevertheless, several research gaps persist.
First, the majority of previous studies have focused on high-temperature and high-humidity outdoor settings, leaving us with little insight into fatigue experienced by medical staff working in air-conditioned, indoor hospitals. Unlike the outdoor environment, medical staff operate within confined spaces controlled by climate, whereas the ambient indoor environment and body heat dissipation affect the microclimate temperature of medical staff clothing, causing fatigue and decreased work efficiency. A recent investigation 17 has suggested that for heavy work, wearing MDPC should not exceed 40 min for continuous safe work, while for light work, this duration can be extended to 110 min to ensure safety.
Second, determining safe working hours remains challenging due to limited data availability, as obtaining physiological measurements under diverse clothing and environmental conditions is often costly and yields small sample sizes. Machine learning techniques14,18 are widely employed for predicting heat stress levels through real-time physiological data monitoring. However, these algorithms require a substantial quantity of high-quality data samples. Unfortunately, the available physiological data are frequently limited and suboptimal, making accurate predictions difficult. Physiological data are typically characterized by inherent uncertainties and influenced by human variability, clothing variations, and fluctuating environmental conditions, which contribute to temporal changes. As such, physiological data are often regarded as a gray system.
Finally, existing heat stress indices are intricate and have strict usage conditions, limiting their practicality in real-world scenarios. PHS models, for example, predict changes in key thermal responses such as sweat rate, sweat volume, and core temperature based on height, weight, labor intensity, and environmental variables such as temperature, humidity, wind speed, thermal radiation, and clothing properties. Nonetheless, metabolic rate, body posture, and clothing also influence thermal stress measurements, but the metabolic rate is typically estimated using empirical values, which may introduce calculation errors in high-insulating clothing and humid environments. 7 An appealing avenue of research lies in investigating the possibility of using a smaller set of widely recognized physiological parameters (e.g. Tcore, Tsk, and HR) to accurately gauge thermal comfort levels.
To address these questions, we present a novel approach to determining safe working hours using the discrete gray model (DGM)(1,1) model based on physiological thermal limits while wearing MDPC at varying MDPC ensembles and work intensities. Gray system theory, proposed by Ju-Long Deng 19 in the 1980s, characterizes black as completely unknown, white as entirely known, and gray as partially or unknown information. Safe working hours forecasting can be considered a typical gray system due to its uncertainty and poor information. Gray systems are viable for uncertain systems with limited and nonlinear data, requiring only a least four data inputs. As such, gray prediction models have been widely employed in diverse fields, such as economics, materials, engineering, and manufacturing, because they can represent unknown systems without statistical assumptions based on limited data.20,21 However, the accuracy of the traditional gray model (GM)(1,1) model requires further improvement. In response, Xie and Liu 22 developed the DGM(1,1) model, enhancing prediction accuracy in uncertainty fluctuation data. For example, Wang et al. 23 applied a DGM to forecast periodic variations in ambient air temperature in laying hen houses. Since physiological fluctuations generally affect the trend of vital signs, adopting the DGM(1,1) model may improve the trend-catching ability of physiological data and extend its application to the prediction of safe working hours.
To our knowledge, there is a paucity of studies investigating safe working hours when wearing MDPCs, which are commonly worn by medical staff in air-conditioned hospital settings. We conducted experiments that involved two MDPC ensembles and two levels of exercise intensity (light at 2.1 km/h and moderate at 4.3 km/h) to create four distinct working conditions. In these conditions, we measured various physiological parameters. Subsequently, we applied the gray prediction model to evaluate physiological responses in an indoor hospital environment (specifically, a cool setting while wearing MDPCs). Considering that the physiological parameters used in the gray prediction model are straightforward and can be measured with limited data, we believe that the DGM(1,1) model is suitable for real-time monitoring in actual clinical workplaces. Under small sample data conditions, this study provides a new approach to determining safe working hours for wearers of MDPC, allowing for rational scheduling of work shifts based on physiological thermal limits.
Materials and Methods
Participants
To control for the confounding effects of gender on physiological responses to heat fatigue, exclusively male, nonsmoking subjects free from cardiovascular, metabolic, and respiratory ailments were selected for this study. A total of eight healthy males (age: 22.5 ± 1.5 years; height: 1.77 ± 0.01 m; body mass: 66.2 ± 1.1 kg; and body mass index: 21.0 ± 0.5 kg/m2 (means ± standard deviations)) volunteered for the experiment, following the ethical guidelines outlined in the Declaration of Helsinki. 24 All participants provided written informed consent and completed a medical history questionnaire prior to study participation, with regular weekly engagement in low- to moderate-intensity physical activity. Twenty-four hours before their laboratory visit, subjects were instructed to refrain from vigorous exercise and consume caffeine, tobacco, or alcohol. Participants were free to withdraw from the study at any time without penalty.
Protective Ensembles
The study used two commercially available MDPC ensembles, DuPont Tyvek 1422 (polyethylene) and Lakeland AMN428E (polyethylene/polypropylene), commonly worn by health care staff over shorts. The test garments had different properties, with Type A MDPC having 1.4 clo insulation, 43.2 g/m2 weight, 0.19 mm thickness, 1.69 mm/s air permeability, and 46 Pa.m2/W evaporative resistance, while Type B MDPC had 1.6 clo insulation, 55.7 g/m2 weight, 0.24 mm thickness, 0.85 mm/s air permeability, and 50.8 Pa.m2/W evaporative resistance. To minimize the risk of infection, participants wore protective gear, including polycarbonate/vinyl goggles, a polypropylene N95 face mask, latex sterile gloves, disposable medical caps made of 28 g nonwoven fabric, and disposable protective shoe covers made of 70% polypropylene/30% polyethylene-coated film, underneath the MDPC garments. We tested each participant four times for each ensemble (ensemble 1 (E1) and ensemble 2 (E2)) under both light- and moderate-intensity conditions.
Outcome Measures
Participants were weighed without clothing except for briefs before and after engaging in treadmill exercise, utilizing an electronic scale (Electroscale Model 921, ±10 g). To determine sweat production and weight loss due to sweat evaporation alone, sweat loss (SL) values were deducted from weight loss measurements obtained while participants were nude and clothed. Due to participants’ unwillingness to undergo rectal temperature measurements through a probe inserted above the anal sphincter, tympanic membrane temperature was utilized as a substitute for direct measurements. A simple and validated method for accurately reflecting rectal temperature was found to be tympanic membrane temperature when wearing encapsulated personal protective clothing. 25 However, as the association between tympanic and rectal temperature is indirect, rectal temperature was estimated by adding 1.04°C 26 to the tympanic membrane temperature. Tympanic membrane temperature was measured every 5 min using an infrared tympanic electronic ear thermometer (IRT6520, Braun, Kronberg, Germany). Mean skin temperature was measured using an 8-point method 27 involving weighted individual temperatures from the forehead, chest, scapula, upper arm, forearm, dorsal hand, thigh, and calf via eight iButton temperature monitors (Model DS1923, logging rate of 1 s, Maxim Integrated, San Jose, CA, USA). HR was continuously monitored using Polar HR monitors (Polar H10, data sampling rates of 400 Hz, Polar Electro, Finland). Upon test completion, participants were weighed again in briefs immediately after removing the experimental clothing.
After each phase, subjective responses, encompassing thermal and humidity sensations (HSs), were evaluated every 5 min. Participants were requested to provide numerical ratings that best corresponded to their sensations. Ratings for thermal sensation (TS) were recorded using a 7-point scale, ranging from (−3) cold, (−2) cool, (−1) slightly cool, (0) neutral, (1) slightly warm, (2) warm, to (3) hot. HS ratings were documented using a 6-point scale, with options including (0) dry, (1) slightly wet, (2) neutral wet, (3) wet, (4) very warm, and (5) extremely wet.
Experimental Protocol
The experiments were conducted in a controlled artificial chamber in a randomized order, with an indoor relative humidity of 50 ± 5%, air temperature of 21 ± 0.5°C, 28 and wind speed of less than 0.4 ± 0.1 m/s. The dimensions of the climate-controlled room were 5.0 × 5.0 × 2.5 m (length × width × height), with air conditioners uniformly supplying air at an accuracy of ±0.5°C. Medical staff predominantly engage in light-intensity activities during their workday, with comparatively little time spent on moderate-to-high-intensity activities. To address the inconsistencies in prior research on the physiological tolerance of medical staff wearing MDPCs, we utilized diverse exercise duration and walking speed protocols at varying intensity levels to emulate real-world conditions. The experiment incorporated four working conditions, combining two levels of clothing thermal resistance with two metabolic rates. Brisk walking at 4.3 km/h (around 3.0 – 4.0 MET) simulated activities of moderate intensity (e.g. emergency treatment and major surgery), while slow walking at 2.1 km/h (around 1.8 – 2.5 MET) simulated light-intensity activities (e.g. consultations, conventional surgery, and miniature surgery).28,29 Water consumption was documented for later analysis, with participants consuming bottled water as desired. Indeed, all subjects did not choose to eat or drink anything during the experiment until it finished.
After preparation, the participants were required to intermittently walk at a speed of 2.1 km/h for 175 min under light intensity, with a 5-min rest following each 30-min exercise, and at a speed of 4.3 km/h for 105 min under moderate intensity, with a 5-min rest after each 30-min exercise. During the test, the subjects could consume water at their convenience. Treadmill exercise was terminated under the following circumstances: (1) self-withdrawal due to reasons such as fatigue or nausea, (2) Tcore ≥ 39°C, (3) HR ≥ 90% of maximum, or (4) additional safety time was provided at the discretion of the researcher. At the end of each trial, the garments were removed, and the postexperimental nude body mass was recorded after completely drying the body with a towel to remove surface sweat. Figure 1 provides a detailed description of the test protocol and measurement outcomes.
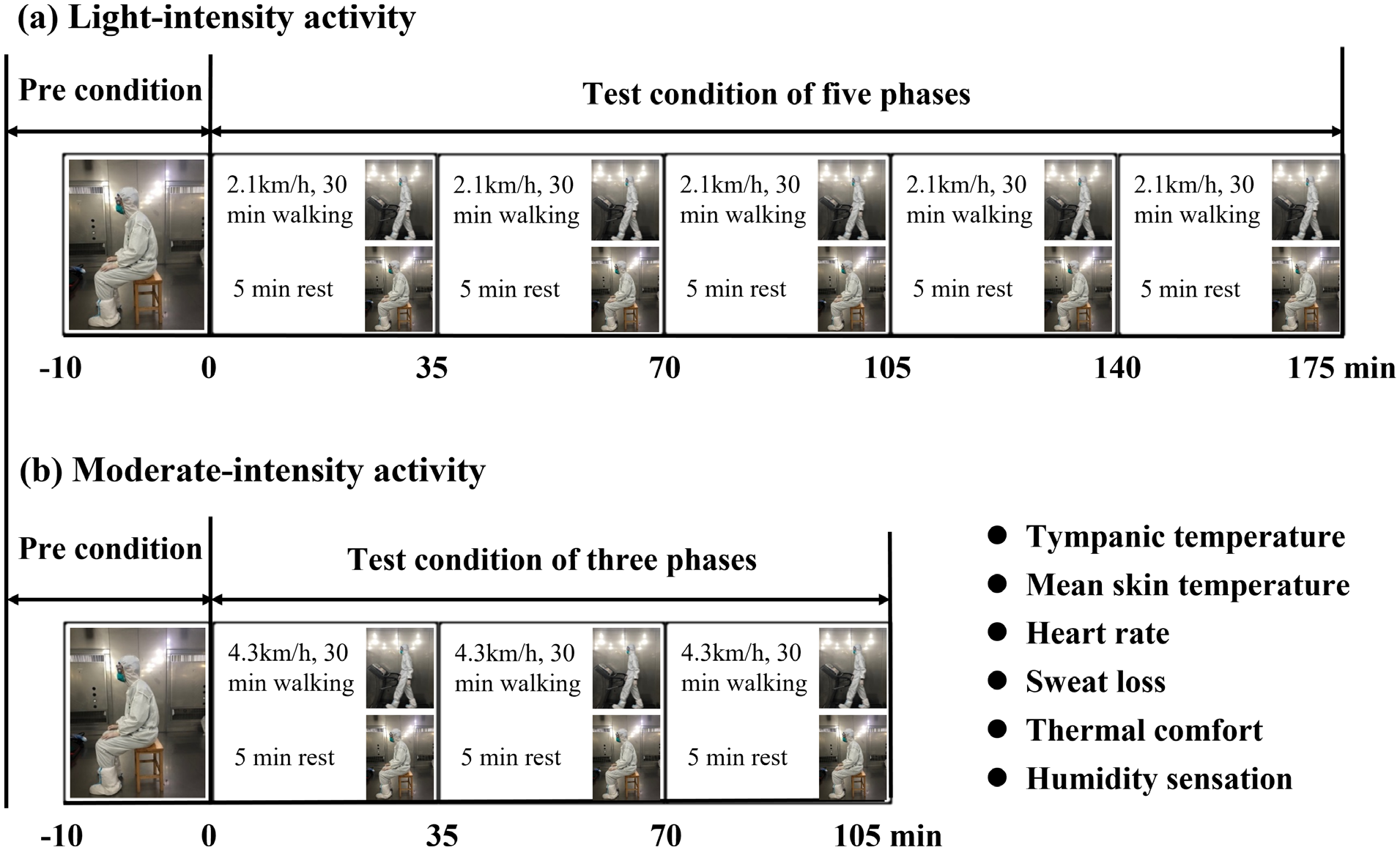
Test protocol and outcomes.
Data Analyses
The statistical data analysis was conducted using IBM SPSS software 26.0 (SPSS Inc., Chicago, IL, USA). Gray Modeling software (GSTA V7.0, Nanjing University of Aeronautics and Astronautics, Nanjing, China) was utilized to generate the DGM(1,1) models, while GraphPad Prism 9.4.1 software (GraphPad Prism Software Inc., San Diego, CA, USA) was employed to create the graphs. We employed a 2 × 4 (work intensity × ensemble) repeated measures analysis of variance (ANOVA) design to analyze the data. We assessed the dynamic fluctuations in four physiological parameters (Tcore, Tsk, HR, and SL) and two subjective indicators (TS and HS) for the E1 and E2. Using the DGM(1,1) model, we simulated the physiological indices and established their temporal relationships, following the methodology established by Xie and Liu. 22 Statistical significance among groups was determined using a p value of less than 0.05.
Upon establishment of the final model, we evaluated its performance utilizing commonly accepted metrics, 30 namely the mean absolute error (MAE), mean squared error (MSE), and mean absolute percentage error (MAPE). The accuracy of the model was assessed through the implementation of the four criteria, where MAPE values below 10% were deemed exceptional, values between 10 and 20% were considered favorable, values between 20 and 50% were deemed acceptable, and values surpassing 50% were regarded as erroneous.
Results
Thermophysiological Responses
For both light- and moderate-intensity levels (see Figure 2), there was a statistically significant difference (p < 0.001) in the Tcore and Tsk indicators among groups. The Tcore was 37.86 ± 0.06°C and 37.94 ± 0.07°C for the light activity level and 37.78 ± 0.10°C and 37.85 ± 0.11°C for the moderate-intensity level for E1 and E2, respectively. The Tsk was 32.87 ± 0.19°C and 33.04 ± 0.18°C for the light activity level and 32.28 ± 0.18°C and 32.81 ± 0.17°C for the moderate-intensity level for E1 and E2, respectively. At moderate-intensity levels, the HR under E1 showed a significantly higher value (p < 0.01, 105.15 ± 6.96 bpm) than that below 1.6 clo (99.95 ± 8.58 bpm); however, there was no significant difference in HR at light-intensity levels between E1 and E2 (p > 0.05). The amount of produced sweat was 0.25 ± 0.07 kg and 0.28 ± 0.11 kg for the light activity level and 0.41 ± 0.13 kg and 0.47 ± 0.13 kg for the moderate-intensity level for E1 and E2, respectively.
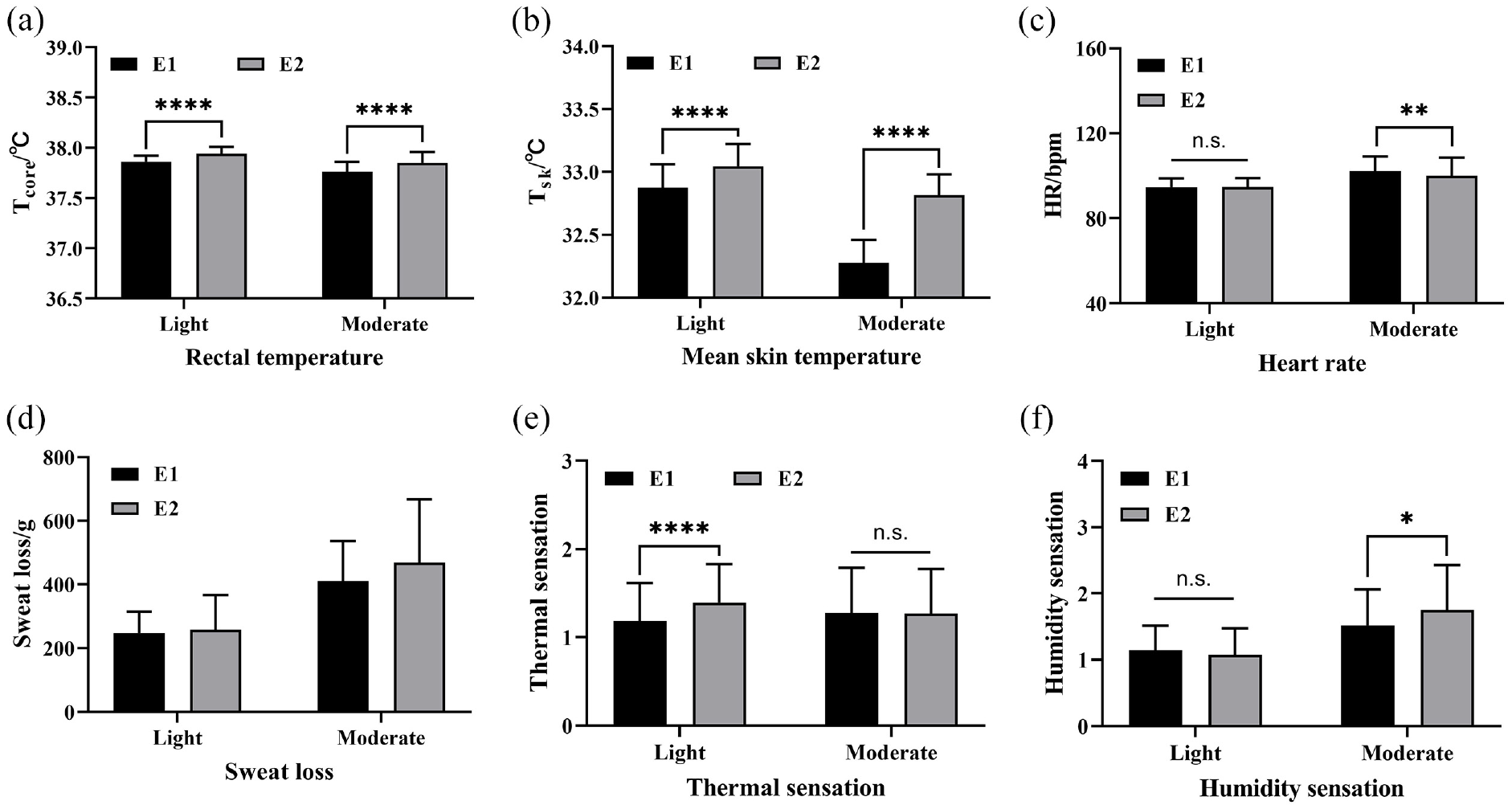
Comparison of thermophysiological responses under different intensity levels: (a) Tcore; (b) Tsk; (c) heart rate; (d) sweat loss; (e) thermal sensation; and (f) humidity sensation.
The average TSs for the two ensembles with thermal resistances of E1 and E2 were 1.19 ± 0.43 and 1.39 ± 0.44 for the light activity level and 1.28 ± 0.51 and 1.27 ± 0.51 for the moderate-intensity level, respectively. The reported light activity levels of E1 versus E2 were significantly different (p = 0.0003) (see Figure 2(e)). However, in terms of moderate-intensity levels, there was no significant difference between them (p = 0.916). In terms of average HS, participants reported that the two garments were statistically significant (p < 0.05) at moderate-intensity levels (see Figure 2(f)) but not significantly different at light-intensity levels (p > 0.05).
Selection of Physiological Indices
To establish a quantitative basis for determining safe working hours, it is imperative to meticulously select sensitive physiological indicators that ensure the reliability of physiological data. This study employed Pearson’s correlation analysis to assess human physiological states and subjective responses, providing a comprehensive understanding of their relationship. Four physiological parameters (Tcore, Tsk, HR, and SL) and two subjective sensations (TS and HS) were included in the analysis. The outcomes of Pearson’s correlation analysis are presented in Table 1, indicating that the Tcore, Tsk, and HR variables passed the significance test and exhibited a statistically significant correlation (p < 0.05). The robust correlation between physiological and subjective states suggests that information from either state can be analyzed and elucidated by the other. Therefore, we opted for the Tcore, Tsk, and HR parameters as sensitive physiological indicators, which aligns with prior research.31,32
Pearson’s correlation analysis.
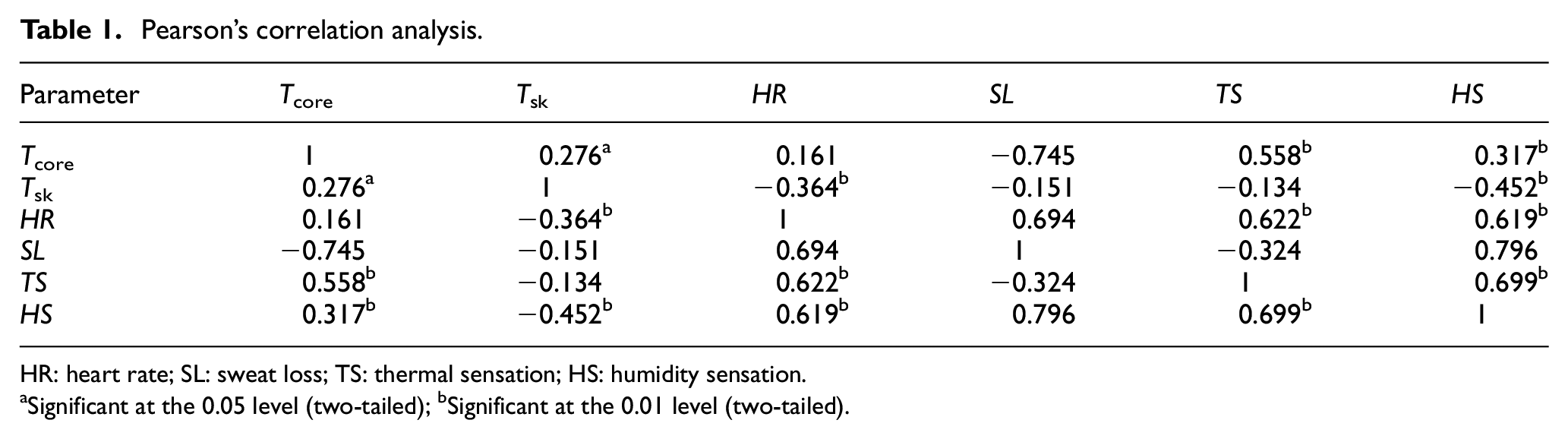
HR: heart rate; SL: sweat loss; TS: thermal sensation; HS: humidity sensation.
Significant at the 0.05 level (two-tailed); bSignificant at the 0.01 level (two-tailed).
Prediction of Safe Working Hours
Gray system theory possesses unparalleled advantages in addressing low-information and small sample fuzzy systems. A DGM(1,1) model, the first-order univariate model, provides a theoretical foundation for transforming the DGM(1,1) model into a continuous form, thus increasing its prediction capacity. Consequently, this model can be regarded as an accurate model. We obtained physiological indicators from six time points (0, 30, 65, 100, 135, and 170 min) and four time points (0, 30, 65, and 100 min) at light and moderate levels. Subsequently, we quantified and comprehensively calculated the safe working time of medical staff. Utilizing the aforementioned DGM(1,1) model, the physiological indicators of eight participants were monitored and averaged across four and six time nodes to simulate and analyze the three physiological indices of Tcore, Tsk, and HR.
Physiological indicators are the most straightforward indicators of heat stress and should be compared with tolerance thresholds to ensure the safety of medical staff. By utilizing the DGM(1,1) model and three sensitive physiological indicators, we determined the safe working hours for medical staff. A fitting curve predicted the value of physiological indicators at future time points, enabling an analysis of the entire curve of the fitting and prediction points, which revealed a slow trend over time for different physiological indicators. Given that the work environment of medical staff is stressful, it triggers physiological reactions, which result in relevant indicator changes. Figure 3 illustrates the evolution of physiological indicators among medical staff over time.
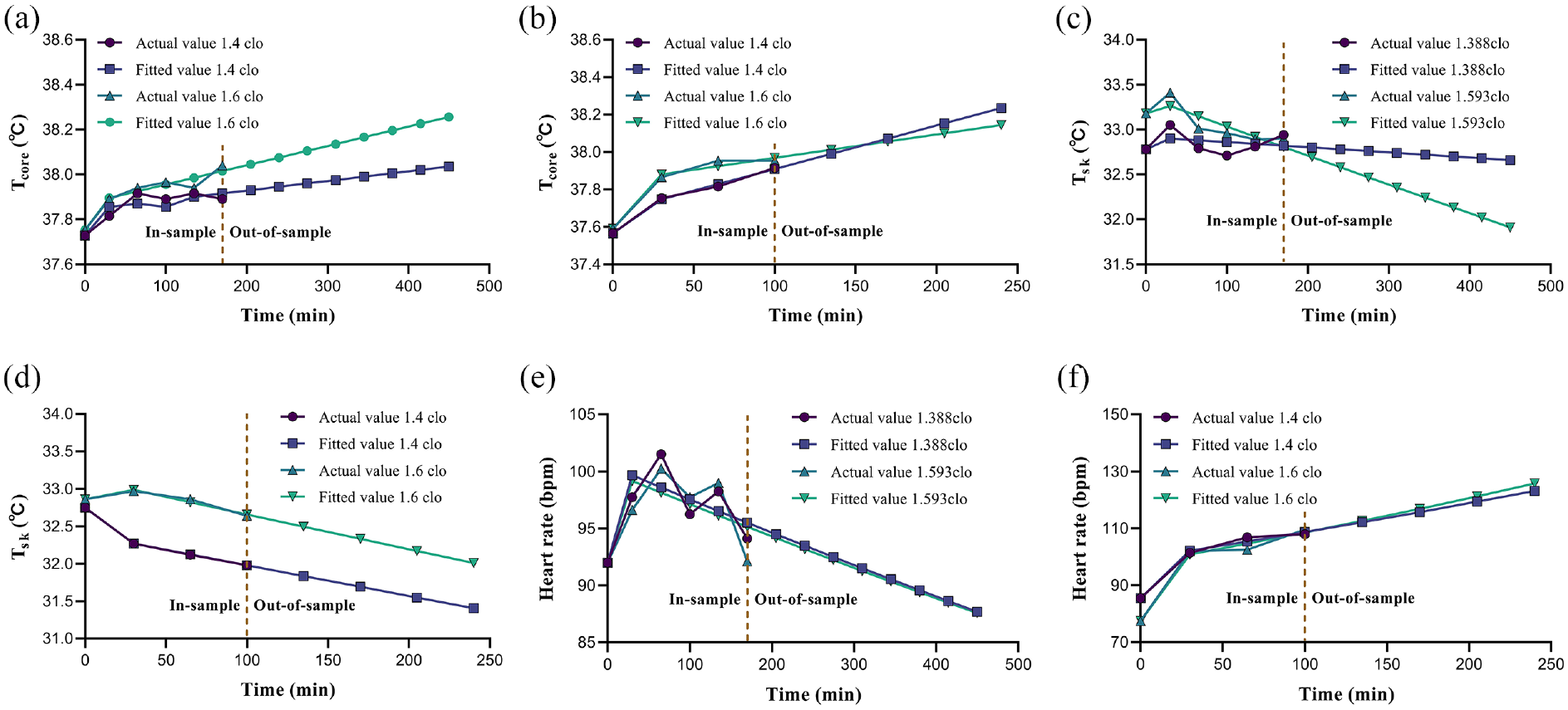
Predicting safe working hours for Tcore, Tsk, and HR indicators at different activity levels: (a) Tcore at light activity; (b) Tcore at moderate activity; (c) Tsk at light activity; (d) Tsk at moderate activity; (e) HR at light activity; and (f) HR at moderate activity.
Comparative Analysis of the Physiological Indices
We assessed the performance of the DGM(1,1) model by comparing simulated results with actual values of the three physiological indicators and computed the MAE, MSE, and MAPE. The physiological indicators of medical staff were examined using the DGM(1,1) model as a function of time throughout the experiments. The physiological data were fitted to a curve, and the equation derived from the curve was employed to predict the physiological data at future time points. A detailed comparison of the physiological indicators using the DGM(1,1) model is presented in Table 2.
Comparative analysis of the physiological indices by the DGM(1,1) model.
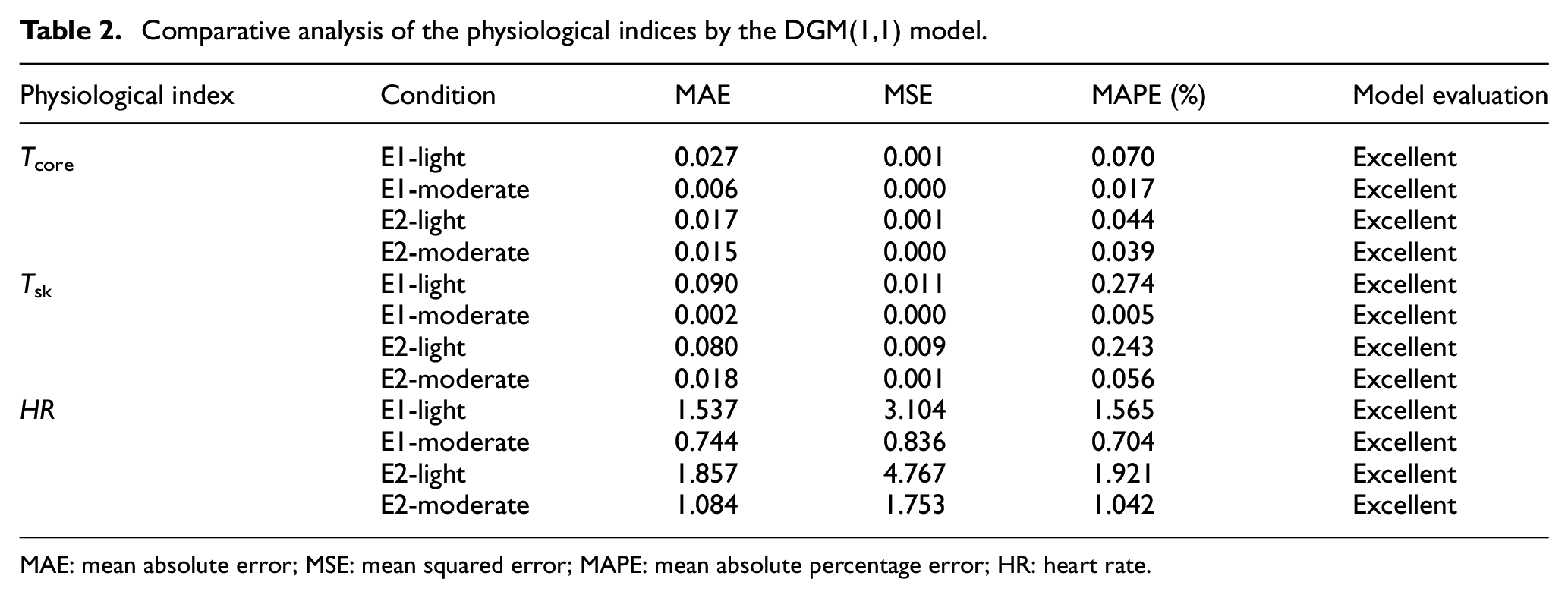
MAE: mean absolute error; MSE: mean squared error; MAPE: mean absolute percentage error; HR: heart rate.
In addition, the DGM(1,1) model was compared with the published FGM(1,1) model (fractional GM(1,1)) 33 and the linear regression model. 34 Figure 4 compares the actual and simulated data forecasting performances of the three predictive models. It is evident from the figure that all three prediction models demonstrated high accuracy in this study, with the DGM(1,1) model producing the lowest MAPE, thus confirming its superiority over the other two prediction models. Therefore, the DGM(1,1) model is considered the most appropriate for predicting safe working hours.
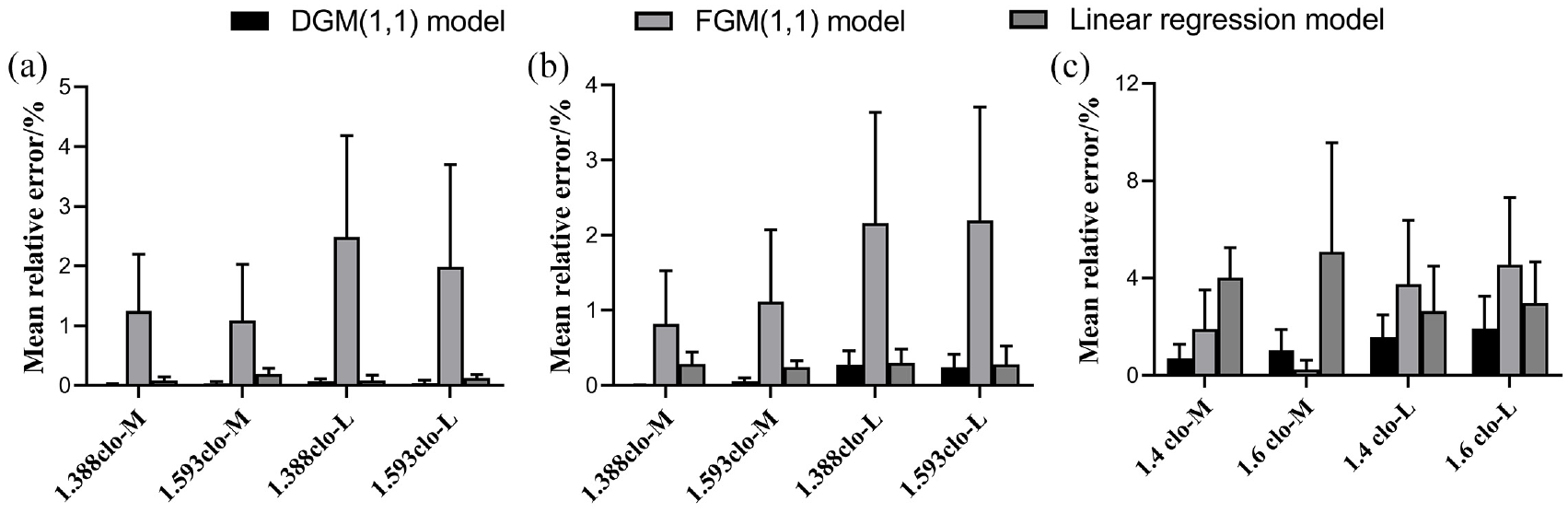
A comparison of three predictive models. (a) Tcore; (b) Tsk; (c) HR.
A high level of simulation accuracy was observed in this study, with the MAPE not exceeding 10%. This finding supports the suitability of the DGM(1,1) model for predicting safe working hours. To determine the maximum acceptable working duration, we adopted a specific protocol. After considering these sensitive physiological indicators, we identified the warning range of the Tcore, Tsk, and HR indicators as Tcore ≥ 38.0°C, Tsk ≥ 37.0°C, and HR ≥ 120 bpm.35,36 Medical staff exceeding these threshold values are considered to be at risk while working. Table 3 shows how the working environment affects the safe working hours of the medical staff.
Safe working hours under different clothing and intensity levels.
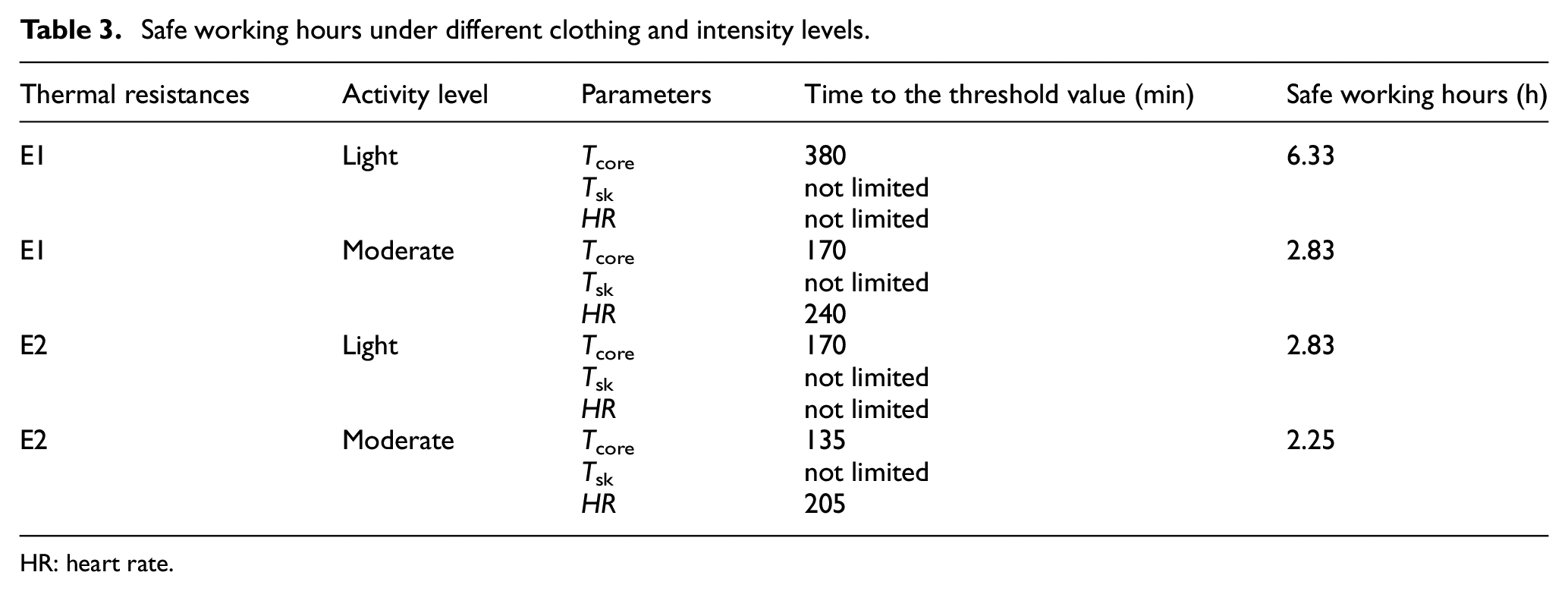
HR: heart rate.
Table 3 illustrates that different types of work intensity and physiological thermal indicators correspond to varying safe working hours. The maximum safe working hours corresponding to Tsk, which do not induce heat stress, suggest that working in a cooler environment reduces Tsk. Conversely, the minimum safe working hours corresponding to Tcore were 6.33 h and 2.83 h for light activity levels and 2.83 h and 2.25 h for moderate-intensity levels of E1 and E2, respectively. Following the minimum value triggered by the thresholds of physiological indicators, the reasonable safe working hours for medical staff were found to be 6.33 h and 2.83 h for light activity levels and 2.83 h and 2.25 h for moderate-intensity levels of E1 and E2, respectively. As such, a prolonged workload could lead to further increases in these physiological indices, which could increase physical fatigue and harm work productivity.
Discussion
Findings Concerning the Experiment
This study introduces a novel method for assessing safe working hours for medical staff wearing MDPC, contributing uniquely to the field of clothing thermal comfort. We evaluated the physical fatigue characteristics of two common MDPC ensembles under nonheat stress conditions, closely simulating typical medical tasks and environments. Our findings have implications for medical workloads and underscore the need for more reasonable work shifts and rest schedules.
While fatigue among medical staff during events like the COVID-19 pandemic has been largely attributed to long hours and psychological stress, the role of MDPC in exacerbating fatigue has been understudied. Our results reveal that even at comfortable ambient temperatures (e.g. 21°C), MDPC still may induce heat stress, altering physiological indicators significantly. We confirm that human core temperature is a more critical determinant of safe working hours than HR. Based on thermal stress characteristics, we establish that safe working durations are 6.33 h for light activities and 2.83 h for moderate activities at E1 and 2.83 h and 2.25 h at E2, respectively. Although we observed variations in metabolic heat production, thermal strain levels remained relatively constant, suggesting self-pacing behavior among medical staff. Further research is needed to understand how work patterns might change with increasing environmental heat loads.
Long work hours are widely recognized as a risk factor for various health conditions among medical staff, including cardiovascular disease, musculoskeletal issues, and mental health disorders.37,38 Despite this awareness, the exact threshold of work hours posing a risk remains undefined. Previous studies1,39,40 have focused on heat strain in occupational settings like hot environments, emphasizing the need to regulate work hours and physiological states in medical staff wearing MDPC. Standard labor practices suggest a maximum of 40 h per week or 8 h per day, but these guidelines lack a scientific basis. MDPC, commonly used by medical staff, complicates this scenario. Our study finds that even under cool conditions, wearing MDPC can significantly impact the physical fatigue of medical staff, causing increased stress levels and reduced work efficiency. This is exacerbated by the personal protective equipment’s limited vapor permeability, inhibiting sweat evaporation and thermal comfort. 12 Our study evaluated thermophysiological responses in eight healthy males, revealing significant differences in how MDPCs of varying thermal resistances affect heat stress tolerance. Existing epidemiological data indicate that health care professionals typically wear MDPCs for an average of approximately 194 ± 3.71 min, which our findings suggest could lead to increased physiological stress and reduced work efficiency. 41
While various indices and models aim to predict human thermal responses, they often lack real-world applicability. Existing metrics, like the WBGT index, are limited by their failure to account for individual physiology. Our study underscores the need for personalized work–rest schedules, urging further research to establish guidelines based on reliable heat stress markers such as core and skin temperature. We introduce the DGM(1,1) method as an alternative to WBGT and PHS index, expanding the toolkit for evaluating heat strain. Heat strain is a complex interplay of physiological and psychological responses to thermal stress. We quantified key markers, including core body temperature, skin temperature, HR, and perspiration-induced mass loss, to offer a nuanced understanding of medical staff fatigue. Our data demonstrate that the DGM(1,1) model can reliably establish safe durations for wearing MDPC, even with limited data sets. While the PHS model could assess heat strain risks, its effectiveness is compromised by factors like intermittent rest periods and garment insulation. In addition, PHS has its limitations; it is accurate only for specific wind speeds and clothing insulation ranges, necessitating caution when applied to specialized attire.7,8 Moreover, PHS requires input on posture, which can influence other variables such as solar radiation exposure and clothing characteristics.
Our study innovatively employs the DGM(1,1) model to quantify safe work durations in disposable protective attire, mitigating physical fatigue risks. Using limited physiological data, we build a DGM(1,1) model that foregoes statistical assumptions, outperforming linear regression and FGM(1,1) models (see Figure 4). This advantage partly arises from the model’s efficacy with limited and raw data sequences where gray models excel. 42 However, given the fluctuating nature of real-world physiological data, DGM(1,1) proves useful mainly for short-term work-hour predictions. Notably, the DGM(1,1) model is less accurate for E2-moderate-intensity activities than the FGM(1,1) model. Caution is needed before generalizing DGM(1,1) applications, as its efficacy varies by domain. Our findings can inform occupational heat exposure guidelines and optimize work durations for medical personnel in disposable protective gear.
Recommendations
Safe working hours are crucial for reducing heat strain in medical staff wearing MDPCs for extended periods. Our study calls for measures to mitigate heat stress responses, as they could potentially influence patient outcomes. Despite the importance of this relationship, studies investigating real-time clinical impacts of staff physical fatigue are sparse. Future research should focus on the link between heat stress and cognitive performance.
Implementing coping mechanisms and control measures can enhance both work productivity and safety, minimizing errors caused by heat-induced cognitive shifts. Recommended strategies include MDPC and heat stress training, as well as work-to-rest ratios designed to prevent dehydration and physical fatigue. Medical staff benefits from shorter MDPC use times and longer breaks, reducing susceptibility to overheating, fatigue, and dehydration during extended shifts. Real-time physiological warning systems can provide further safeguards by alerting staff to dangerous conditions. Psychological factors, which may also affect physiological responses, should not be overlooked.
Additional interventions to alleviate heat strain include localized ventilation and heat mitigation tactics, such as wearable cooling devices39,43 or periodic cool drink consumption every 15–20 min. 44 However, the water intake should be moderated, considering the potential inaccessibility to restrooms during work. Employing these strategies, medical staff can minimize physical fatigue risks and perform their duties more effectively and safely. These measures also enable potentially longer yet safer work periods.
Limitations
The study has some limitations that must be acknowledged. First, the sample size was relatively small, comprising only eight young male participants, and excluded females. Variability in medical staff—due to age, sex, acclimation, fitness, alcohol use, shift duration, and chronic disease—could affect physiological and psychological responses to high temperatures. Given the small sample size, these studies are best described as pilot studies; thus, our findings should be interpreted cautiously when generalized to workers of different ages or experience levels. Future studies should include a more varied sample to assess the cognitive performance of medical staff under physical fatigue.
Second, regarding the experimental protocol, this environment was milder than high-temperature and high-humidity settings to prevent excessive fatigue and burnout in the nursing workforce. Future work should consider the influence of different factors on cognitive function in high-temperature environments. Our experiment employed only two common levels of MDPC protection; however, the pandemic has necessitated many different levels, some higher than those used here. 4 As higher protection levels are typically used in specific situations, like surgery, they were not included in this study.
Third, ethical restrictions limited the equipment used for recording physiological variables, leading to participant hesitation toward rectal temperature measurements. Consequently, tympanic membrane temperature was used as a surrogate, adjusted by 1.04°C. 26 This compromise may have led to overestimation or underestimation of results. Although work intensity was stratified based on routine medical activities, the simulation did not precisely mimic actual clinical practice. Additional research is needed to investigate the thermophysiological impact of MDPC on medical staff wearers.
Finally, this study introduced a new DGM(1,1) model for determining safe working hours based on physiological thermal limits. However, the DGM(1,1) model, while providing better precision, still contains errors, partly due to the variability in human thermoregulation that no model can fully account for. The practical and general application value of the DGM(1,1) model necessitates careful interpretation and verification. Deep-level modeling mechanisms were not utilized to optimize prediction accuracy. Given its preliminary nature, the interpretation of this model should be approached with caution. Future research may employ alternative methods for optimizing traditional gray prediction models.
Conclusion
This study introduced a novel DGM(1,1)-based approach to determining safe working hours while wearing MDPC based on physiological thermal limits. The DGM(1,1) model was chosen due to its suitability for handling small sample data, requiring only a minimum of four data points to model real-world physiological variables without the need for adherence to statistical assumptions. Besides, this model is compared with FGM(1,1) and linear regression models to demonstrate its superiority. The results of this study not only elucidate the impact of MDPC on the physical fatigue of medical staff but also provide a foundation for medical administrators to comprehend and manage staff fatigue, enabling the adjustment of workloads to ensure work safety. The following findings can be drawn:
Even in cooler, air-conditioned hospital environments, extended periods of wearing MDPC significantly affected physiological indicators such as Tcore and Tsk (p < 0.001). Tcore was identified as the predominant factor limiting safe working hours for medical staff, emphasizing the importance of implementing mandatory breaks, workload adjustments based on worker conditions, and ensuring safe working hours to promote workplace safety.
The introduced DGM(1,1) model exhibited superior fitting and forecasting accuracy compared with the other two models, making it a valid method for determining safe working hours based on changes in physiological variables under physiological thermal limits. This model provides a foundation for medical administrators to understand and manage fatigue levels, enabling them to arrange work content for medical safety effectively.
Prediction results based on physiological thermal limits suggest maximum safe working times for medical staff wearing MDPC. Specifically, for light- and moderate-intensity levels at E1 and E2, reasonable safe working hours were estimated at 6.33, 2.83, 2.83, and 2.25 h, respectively. Adapting medical staff shifts to actual conditions and safe working hours in air-conditioned hospital environments is advisable.
Future studies should encompass diverse heat environments, a broader range of age groups, and subjects acclimated to heat. In addition, incorporating additional indicators such as body composition, respiratory minute volume, blood pressure, and metabolism is essential. Leveraging intelligent methods such as data mining and machine learning may aid in evaluating the effects of MDPC usage on heat stress and developing strategies to mitigate heat-related fatigue.
Footnotes
Acknowledgements
The authors would like to thank Professor Tamaki Mitsuno (Shinshu University, Japan) for her thoughtful and stimulating Textile Bioengineering and Informatics Society (TBIS) conferences. They would also like to thank the subjects who willingly participated in this study.
Author contributions
J.L.: conceptualization, methodology, software, data curation, writing—original draft, review & editing; X.C.: performed experiments, review & editing; Q.H.: validation, writing—review & editing; J.L.: conceptualization, supervision, review & editing.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Fundamental Research Funds for the Central Universities (grant no. 2232023G-08), the Fundamental Research Funds for the Central Universities and the Graduate Student Innovation Fund of Donghua University (grant no. CUSF-DH-D-2023047).