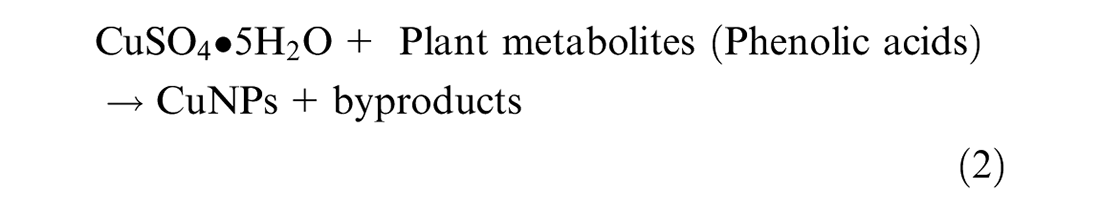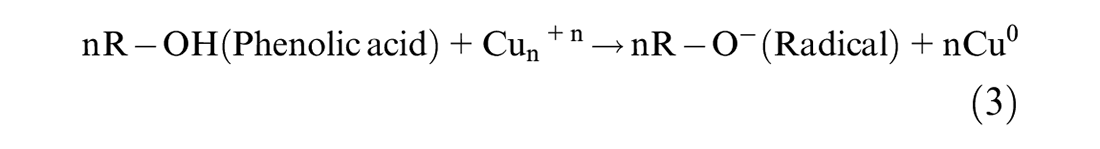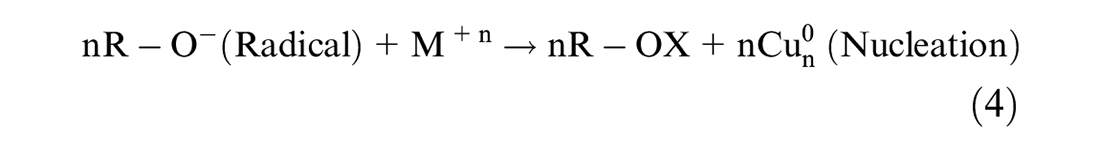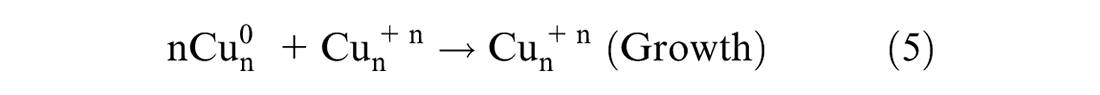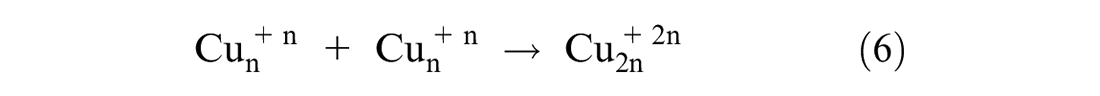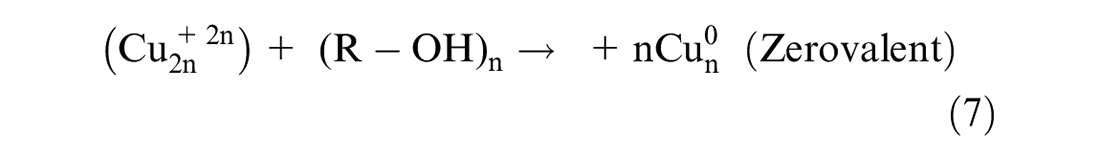Abstract
Cotton fabric samples were treated with copper acetate mordant, in addition to green synthesized copper nanoparticles extracted from the Conocarpus erectus leaves, with two different concentrations of 10% and 20%, respectively. Two dyeing techniques, continuous dyeing and exhaust dyeing, were employed. X-ray diffraction confirmed the synthesis of copper nanoparticles with an approximate size of 30 nm. Scanning electron microscopy showed copper nanocrystals with a size range of 30–70 nm. The tensile strength showed an approximately 60% rise in the warp direction and an approximately 20% rise in the weft direction for both continuous and exhaust dyeing techniques. The color strength (ratio of absorption co-efficient (K) and scattering coefficient (S); K/S) showed an improvement of 100% and 20% for the exhaust and continuous dyeing techniques, respectively. Both the dyeing techniques showed a rising trend in color fastness to wash and color fastness to rubbing with an increment up to grade 5. The light fastness showed an improvement up to grade 6 for both the techniques. Fastness to rubbing showed an increment from 1 to 5 for both techniques.
Introduction
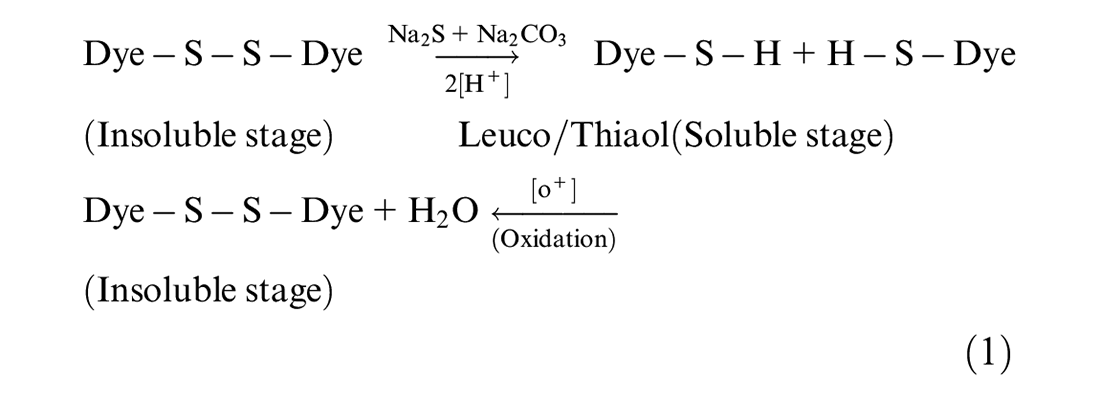
Cotton fabric can be dyed with different categories of dyes such as direct, sulfur, vat, and reactive dyes, as well as with azoic combinations. Among these, the sulfur dyes are most popular due to their being cheaper. However, the sulfur dyes show poor affinity toward cotton fabric due to their insoluble nature in water. They can be converted into water soluble form for cotton denim fabric by employing a chemical reduction process, which results in hydrogen removal. The free hydrogen combines with dye molecules, resulting in hydrogen bonding with a water molecule. Thus, the cotton fabric is then treated with a dye with the help of water medium. The reduction and oxidation mechanism is shown in equation (1). First, the sulfur dye molecules get converted into soluble form in the presence of sodium hydrosulfite and sodium hydroxide acting as reducing agents. The reduced dye then gets oxidized to produce water. The dye becomes insoluble again after the removal of hydrogen and as a result the dye molecule gets physically trapped inside the fabric fiber. This results in improved color fastness properties of the cotton fabric. 1
The main drawbacks of sulfur dyes involve poor wash fastness properties along with the crock and rubbing fastness. This usually results because of improper color dissolution, color precipitation, poor solubility of the dyes, poor and insufficient washing after dyeing of unexhausted dyes, and insufficient soaping treatments. 2 However, sulfur dyes offer darker shades and are inexpensive, thus, despite poor washing fastness properties, they are still widely used for dyeing cotton fabrics. 3 Thus, they have shown unexpected growth in the garment area where heavy depth shade is often required and the competitive cost factor results in compromised washing fastness properties.4,5 However, in the denim dyeing industry, sulfur dye use is limited due to the requirement of better wash fastness properties. 6 To overcome this drawback of sulfur dyes, modification of the fabric surface is required to allow more dye uptake (dye fixation) at low temperature and wettability. Different dyeing procedures have been proposed in literature to fix the impregnation of dye on the fabric such as paid dry, paid cure, and cation–tannin protective system based treatments.7,8 However, nowadays textile chemists are largely focusing on eco-friendly treatment methods for permanency of color on textile fabric using sulfur dyes. 5
The use of nanotechnology and nanomaterials in textiles has been reported to enhance the mechanical properties of the textile material. Due to their small size, nanoparticles can easily penetrate in the fabric surface and play a role of cross-linking fillers between polymer molecules and dye molecules. This results in increased bond strength between dye molecules and fabric to enhance the fiber, yarn, and processing technology to meet the global demands, as well as the needs of the society.9,10 The application of metallic nanoparticles (NPs) is the most scientific and versatile industrial technique used for the impregnation of textile reactant solution to enhance the dye-ability and dyeing properties. 11 The preparation of the NPs from plants is inexpensive and easy to scale up as well as energy-intensive and eco-friendly. Many studies have proposed different green synthesis methods of metallic NPs for textile applications. For example, the study by Sivakavinesan et al. 12 used peels of Garcinia mangostana (Mangostan) along with gold solution to prepare gold NPs and reported an improvement in color fastness of the copper-dyed fabrics. Another study by Afifi et al. 13 reported that there are phenolic acids found in the plant extract and these may be used as reducing agents. Similarly, the Ghidan et al. study reported the green synthesis of copper NPs (CuNPs) using Punica granatum peel extract. Punica granatum fresh peels were obtained and washed several times. After drying the peels, they were turned into powder and mixed with sterile distilled water and boiled until the color of the solution changed to yellow. 14
The CuNPs are moderately toxic materials in comparison to their precursor material, where LD50 values of CuNPs are approximately 413 mg/kg body weight. 15 Thus, the NP preparation from the plant extract requires controlled concentration of the solution to avoid crossing the LD50 limit. This ensures that the prepared NPs are free of toxic contaminants as required in biomedical and therapeutic applications. 16 Many earlier studies have also explored different leaf extracts where cytotoxicity studies are largely done on live cells or bacteria; however, to the best of our knowledge, no such study has been reported on the physicochemical attributes of sulfur-dyed cotton fabric.17–19
Therefore, inspired by earlier studies on green synthesis of NPs from plants without using any external energy, this work aimed to study the impact of sulfur dyeing on fabric with a green synthesized CuNP deposition. In our study, we used a very low amount (almost a quarter of limit) of CuNPs to avoid any toxicity danger by exceeding the LD50 limit. We used Conocarpus erectus leaves, which are commonly available, for the CuNP preparation with 10% and 20% concentration of copper acetate, respectively. The experimental results show that the NP growth improved fabric tensile strength, color adherence strength, the and washing fastness of sulfur-dyed cotton fabric, and gave significant color yield without severely affecting the physical characteristics of the fabric.
Materials and Methods
Materials
In this experiment, 100% bleached cotton plain fabric is dyed with Diresul Indiblue RDT-R (liq) sulfur dye. Copper (II) sulfate (98% pure) from Merck and Conocarpus erectus leaves were used for synthesis of CuNPs. Hydrogen peroxide (H2O2) and acetic acid of commercial grades were used to produce copper acetate. Acrylic Binder (Appretan N 9211) was used for nano-coating. Glauber’s salt, Leonil EHC, Reducer RDT, Caustic soda 48° Be, sodium hydrosulfite, Dekol 1097, Direfix SD, and Ladiquest 2005 liquor was used.
Methodology
Green Synthesis of CuNPs
A total of 100 g of powdered dried leaves of Conocarpus erectus were added to 500 mL distilled/deionized water, methanol, and ethanol in a 1000 mL flask and shaken well. The mixture was then heated by a hot plate/magnetic stirrer at 50°C for 60 min. After this, the plant extract was filtered by vacuum filtration assembly and the filtrate was kept safely for further use.
A total of 50 mL of 0.01 M solution of copper sulfate was introduced in a 250 mL conical flask and then 10 mL of the plant extract prepared in distilled/deionized water, methanol and ethanol was added drop by drop along with vigorous shaking on a hot plate until the color of mixture became dark brown. After the completion of the reaction, the solution was allowed to settle overnight and the supernatant solution was then discarded carefully. The remaining solution was centrifuged at 10,000 ×g for 10 min and the supernatant was discarded. The obtained NPs were washed three times using distilled water. Washing was carried out to remove the byproducts and the excessive extract that were bound with the NPs. After washing, the prepared NPs were dried in a vacuum at 60°C for 12 h for following characterization. The prepared and dried CuNPs are as shown in supplementary information file (SI) Figure S1. To determine the surface features of treated and untreated fabric, analysis was done using scanning electron microscope (SEM). For the identification of the crystalline structure of grown CuNPs, powder X-ray diffraction (XRD) was used.
Treatment with Copper Acetate
About 0.5 g CuNPs were dissolved in a boiling mixture of 1:2 ratios of H2O2 and acetic acid until all the particles were converted into copper acetate. The final volume of the solution was 100 mL. This solution was used for making up different concentration solutions for padding of the fabric.
Preparation of Coating Bath
Two baths were prepared for exhaust (170 mL) and continuous (Pad-Ox; 100 mL) dyeing containing 10% and 20% solutions of copper acetate in tap water. Acrylic binder (1%) was also added to the solution.
Application of Copper Acetate on Fabric
After preparing the coating baths, the four fabric pieces of size 25 × 25 cm were immersed and soaked for 5 min in the bath at room temperature and passed twice through a padding mangle, while rest of the four samples were passed through the padding mangle without immersing in the coating bath solution. A 100% wet pick-up was maintained for all the treatments and padding pressure maintained at 1 bar. After padding the fabrics, they were air dried and then cured for 3 min at 140°C in a hot air oven. Samples were treated with two different concentrations of copper acetate solution for assessing their impact on color fastness properties. 20 The coated samples are shown in SI file Figure S2.
Exhaust Dyeing of Cotton Plain Weave Fabric with Sulfur Dye
The dye bath was prepared with a 1:10 liquor ratio and set at room temperature. Diresul Indiblue RDT-R dye was taken as per the required shade of 15%. The dye bath was based on a recipe compring 2.0 g/L Leonil EHC, 4.0 g/L Dekol 1097, 10% Reducer RDT, 10% Caustic soda 48° Be, 25 g/L Glauber’s salt, 2 g/L sodium hydrosulfite, and 15% Diresul Indiblue RDT-R dyestuff. The samples of pretreated fabric as well as an untreated fabric were introduced into the dye bath and immersed well. The dye bath was treated at 75°C and maintained at this temperature for 30 min. After 30 min, the dye baths were cooled down to 50°C. Fabrics were then taken out of the dye bath, squeezed, and rinsed using tap water. IMeanwhile, the oxidation bath was ready for oxidation of sulfur dye to fix it on the fabric in its insoluble form. The oxidation bath recipe comprised 10 mL/L Oxidant BRI, 6 mL/L Direfix SD, and acetic acid for pH 4–4.5 The dyed samples were then introduced to the oxidation bath and treated at 70°C for 10 min. The fabrics were then taken out of the oxidation bath and due to their darker shade they were first warm rinsed then cold rinsed to remove unfixed dye. Fabrics were later dried under shade. Nano-coated dyed fabric sample is as shown in SI file Figure S3.
Continuous (Pad-OX) Dyeing of Cotton Fabric with Sulfur Dye
The copper acetate and nano-copper treated and untreated fabric were padded with 70–80% pick-up with a padding liquor temperature maintained at 70°C. The padding liquor recipe comprised 75 g/L Diresul Indiblue RDT-R, 1–7 g/L Leonil EHC liq c, 2–3 g/L Ladiquest 2005 liq, 20–25 g/L Reducer RDT powder, and 20–25 g/L caustic soda 50%. After padding, the fabric was air oxidized for 60 s, then introduced to the oxidation bath for another 60 s. The bath pH was adjusted to 4.5 and oxidation occurred at 70°C. The oxidation bath recipe comprised 15–20 g/L Diresul GPA, 15–20 g/L acetic acid, and 7–12 g/L Direfix SD. Later, on oxidation, the dyed fabrics were cold washed for 2–3 min to clear the unfixed dye. Then the fabrics were air dried. 21
Results and Discussion
Total Phenolic Contents
Total phenolic compounds in Conocarpus erectus leaf extract was studied and results are as shown in Figure 1.
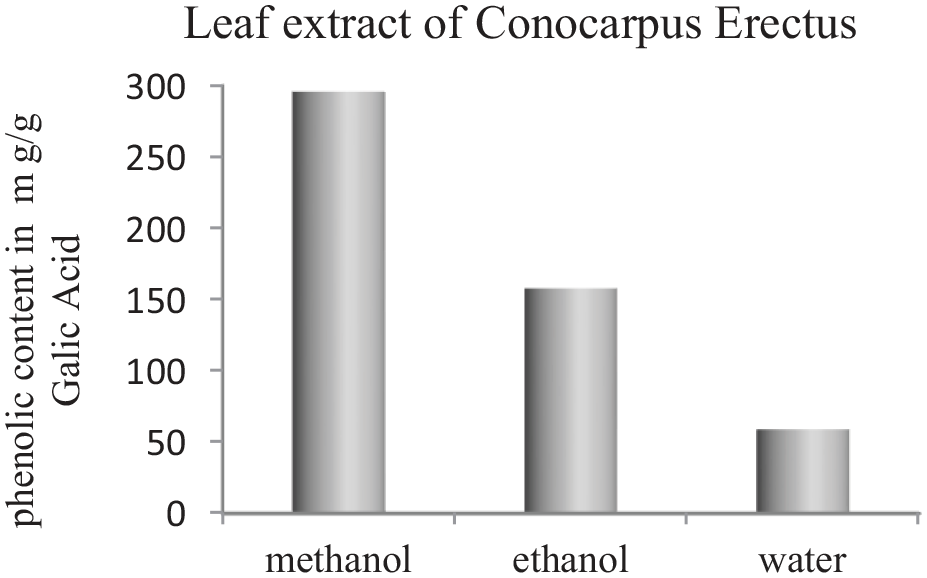
Phenolic contents in leaf extract of Conocarpus erectus.
It was observed that total phenolic or reducing compounds in methanol extract of Conocarpus erectus leaves are higher than its extract in water and ethanol. Because methanol possessed high polarity and substantial solubility behavior for phenolic compounds present in Conocarpus erectus leaves, it assures a high magnitude of extraction.22,23 The value in methanol extract is noted as 296±9 mg/g, while in ethanol extract, it is noted as 158±5 mg/g. and in water extract, it is noted as 59±8 mg/g. The methanol leaf extract of Conocarpus erectus consisted of greater values of total phenolic content, so it was used for the green synthesis of CuNPs in this study. We selected Conocarpus erectus leaf extract for the green synthesis of CuNPs as a raw material because of their availability in any season.
Green Synthesis of CuNPs
In this investigation, CuNPs were synthesized by reducing metal ions from their salt using phenolic compounds from Conocarpus erectus leaves extract as a reducing agent. The mechanism of reduction is given in equation (2) as follows: 24
This study followed the approach reported in the study by Amini and Akbari 25 for in situ synthesis of phenolic acid-coated CuNPs carried out in an aqueous solution. Applying methanol extract of Conocarpus Erectus leaves, containing phenolic acids as a reducing agent for synthesizing metal NPs, was performed using a thermodynamic equilibrium approach as evident from equation (3). Therefore, in this synthesis approach, nucleation is initiated by injecting reducing agent (phenolic acids) at the copper ion supersaturation concentration as shown by equation (4). This is followed by the subsequent growth of CuNPs through gradual ion reduction as shown in equation (5). The high oxidation tendency of phenolic acids facilitates the initiation of nucleation process. The oxidation of the caffeic acid hydroxyl functional group would provide the electron (e–) required for neutralizing ions. The bio-reduction potential of phenolic acids is directly related to the number of their functional hydroxyl groups. The growth process of CuNPs, as shown in equations (6) and (7), was mediated by the oxidized phenolic acids attached to the surface of the CuNPs. The absorbance of the phenolic acid attached to the surface of the CuNPs resulted from the formation of an adsorbent bond between the carboxyl group and the metal atom as evident from equation (7).
X-Ray Diffraction and Fourier Transform Infrared Study
XRD analysis was used to identify the CuNPs. Figure 2 illustrates the XRD pattern of CuNPs for the prepared sample in Conocarpus erectus extract and agreed with the reported XRD pattern of CuNPs. 26
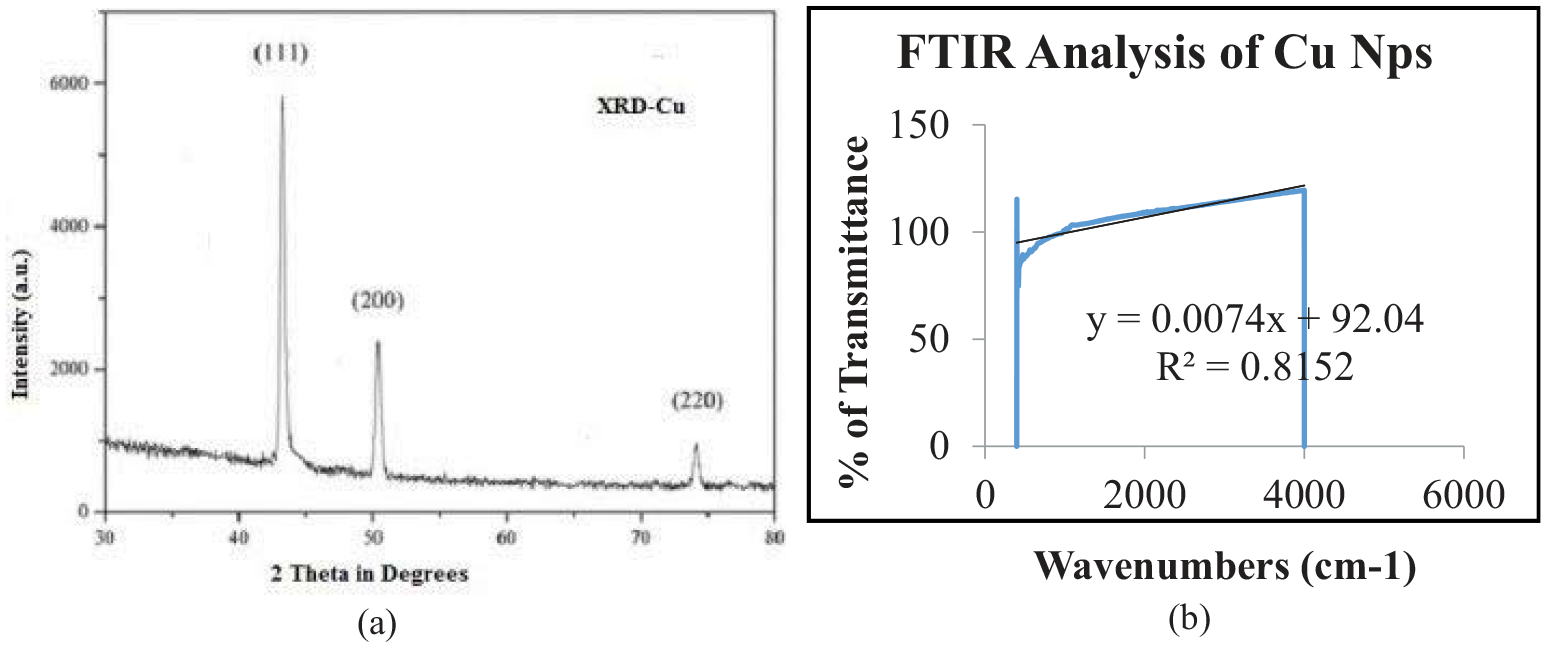
(a) XRD of copper nanoparticles. (b) FTIR of copper nanoparticles.
Three peaks at 2θ values of 43.71, 50.96, and 74.60 degree corresponding to {111}, {200}, and {220} planes of copper were observed and compared with the standard powder diffraction card of JCPDS, copper file No. 04-0836. 27 It shows an agreement with the experimentally obtained XRD angle and the standard diffraction angle of the copper specimen. 28 In addition the Fourier transform infrared (FTIR) study was also conducted to confirm the purity of CuNPs being produced as shown in Figure 2(b). The significant coefficient of determination, R2 = 0.8152 endorses the purity of CuNPs.
Scanning Electron Microscopy Study
The nature of the synthesized CuNPs and the cotton fabric surface before and after treatment was analyzed by SEM. Well-dispersed distribution of particles was seen by this analysis. The SEM images shown in Figure 3(a), revealed a number of cluster of copper particles. This image also disclosed that the size of the CuNPs was in the range of diameter 30–70 nm and a few larger particles have a diameter above 100 nm. As per standard ISO 13320:2022, the CuNP size distribution percentage has a significant coefficient of determination (R2 > 0.8), which is reported in Figure 3(b). In addition, from the SEM images, as shown in Figure 4(a) and (b), it was evident that the CuNPs were distributed almost evenly on the surface of the cotton fabric.
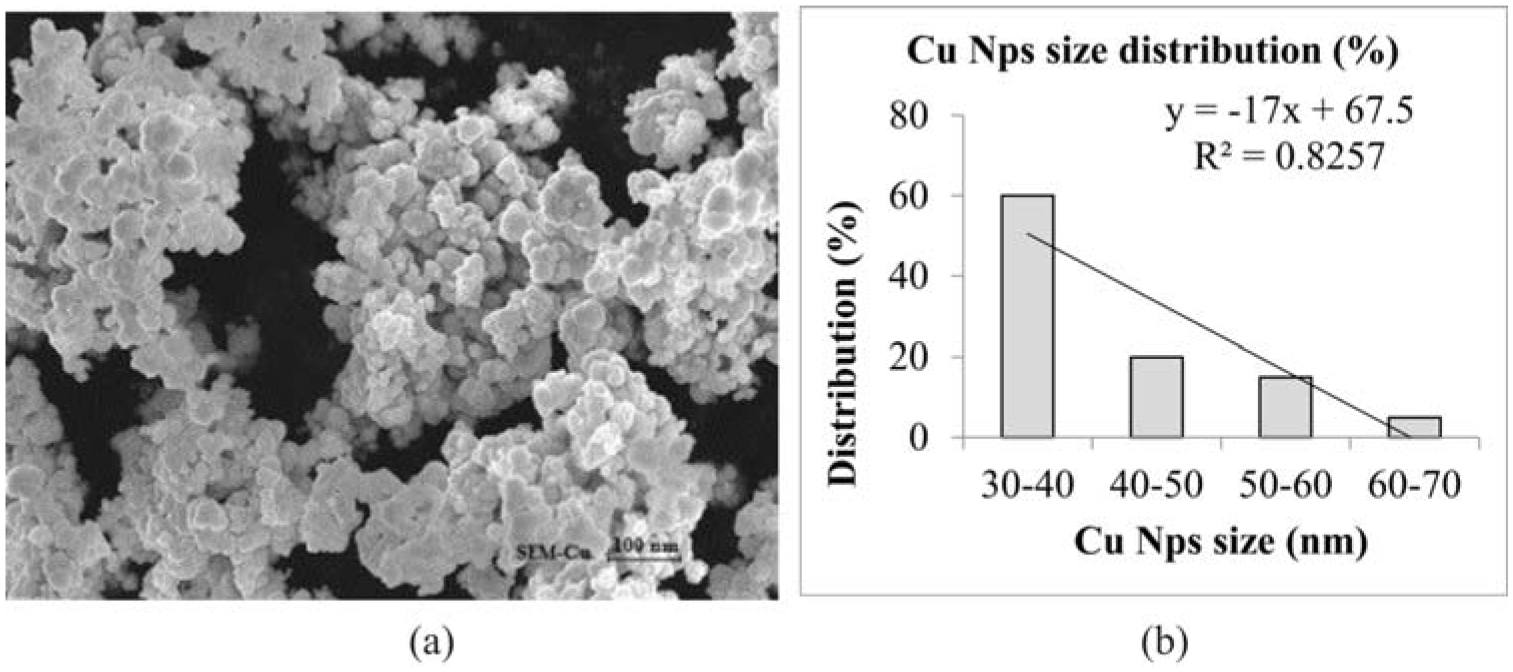
(a) SEM image of copper nanoparticle. (b) Laser diffraction method, CuNP size distribution percentage.

(a) SEM image of untreated cotton fabric. (b) SEM image of CuNP-treated cotton fabric.
Tensile Strength
As per BS-EN ISO 13934-2, the tensile strength was tested using a Titan III Machine as in similar studies. 29 Five warp and weft direction specimens were cut to dimensions of 32.5 × 6 cm and material was raveled on either side to ½ cm. The tensile strength was computed on an average of five samples in the warp and weft directions in the exhaust and continuous dyeing techniques. An increasing trend was observed with the copper acetate concentration and CuNPs. The results indicated that treatment of copper acetate and CuNPs before dyeing added strength for dyeing with the exhaust and continuous processes, respectively. It is revealed by Figure 5 that dyeing techniques have significant coefficient of determination (R2 > 0.9) with the tensile strength of treated fabric in warp and weft direction.
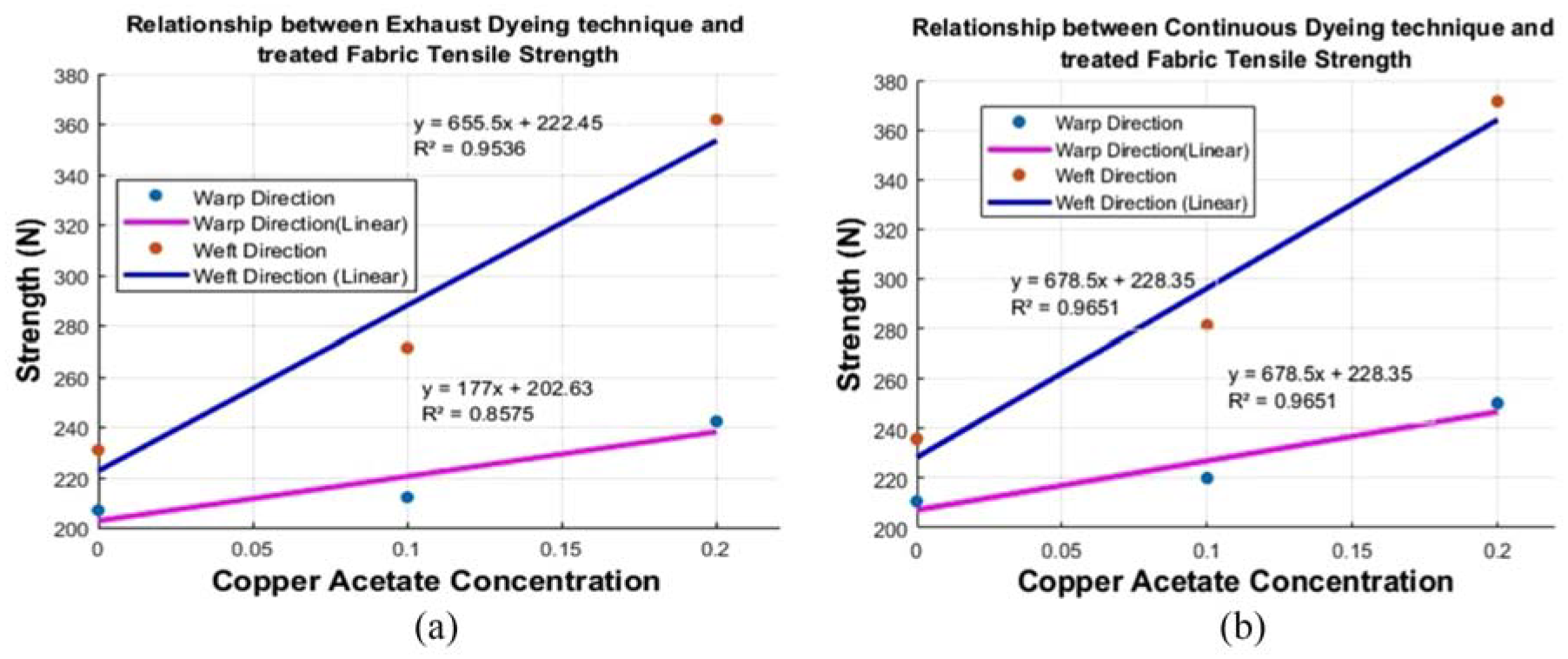
(a) Exhaust dyed treated fabric tensile strength. (b) Continuous dyed treated fabric tensile strength.
In addition, tensile strength was increased because of the geometrical properties of the samples by treating with CuNPs, as it is known that heavy fabrics are stiffer, stronger, and thicker. 30
Color Strength
The study findings indicate that the relative strength and K/S (ratio of absorption co-efficient (K) and scattering coefficient (S)) values were influenced by copper acetate concentration when the fabric was dyed with exhaust and continuous dyeing process. The relative color strength and K/S increased with the increase in concentration of copper acetate but percentage reflectance values were decreased. The reflectance magnitude depended on the surface properties of the dyed treated fabric with regard to the light reflectance value in comparison with the depth of shade due to dyeing techniques. 31
It is shown in Figure 6(a) that the reflectance percentage of treated samples has a significant coefficient of determination (R2 > 0.95) with both dyeing techniques. It is shown in Figure 6(b) that both dyeing techniques of treated samples show a significant coefficient of determination (R2 > 0.90) of the K/S value of the dyed treated sample. So the greater concentration of copper acetate with copper nano-coated fabric shows more color depth and dye uptake. The increase in color strength leads to darker shades of the samples. The increases in the K/S values were due to the presence of CuNPs and copper acetate that acted as a mordant. The highest relative color strength and K/S value were observed at 20% copper acetate concentration.
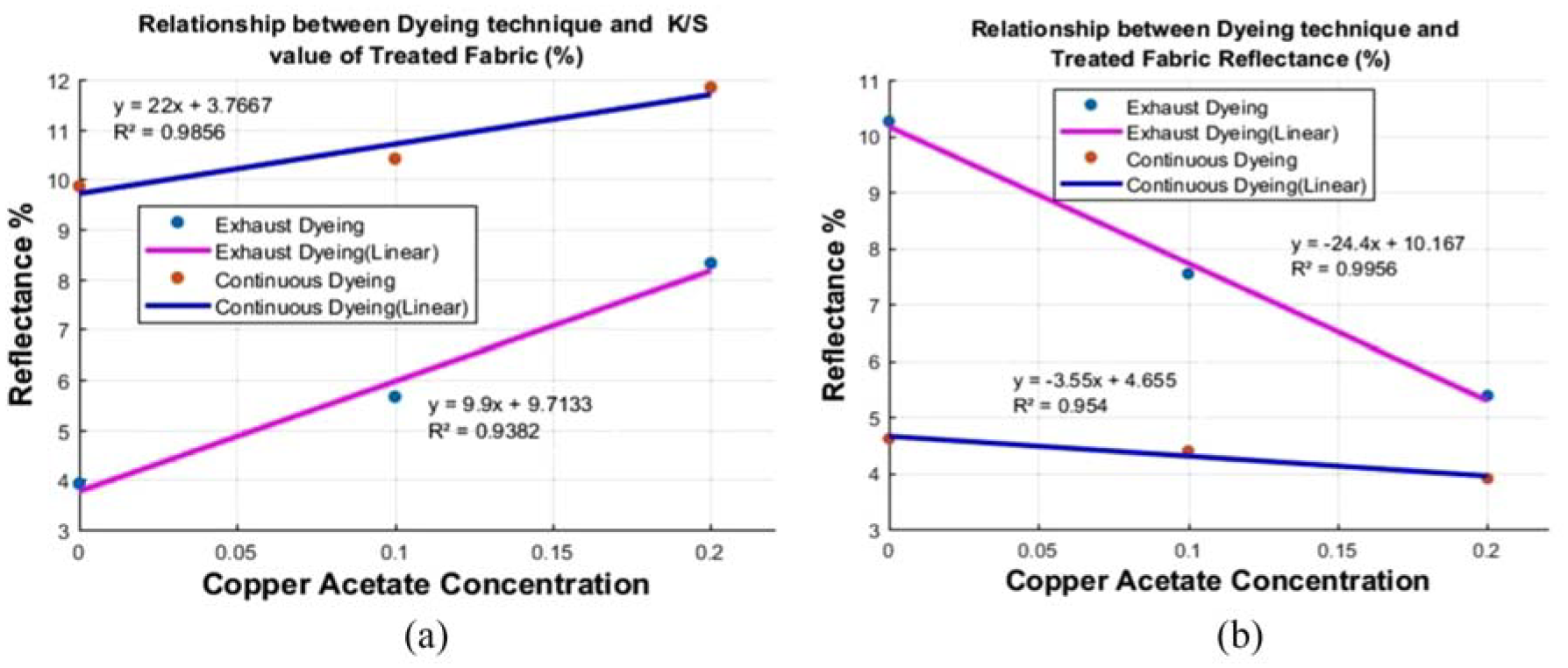
(a) Dyeing technique and K/S value of treated fabric. (b) Dyeing technique and treated fabric reflectance (%).
Color Fastness to Wash
Pretreatment of copper acetate with CuNPs improved the fastness properties of the exhaust and continuous dyed samples as is evident in Table 1.
Comparison of fastness properties of standard and coated NP samples.
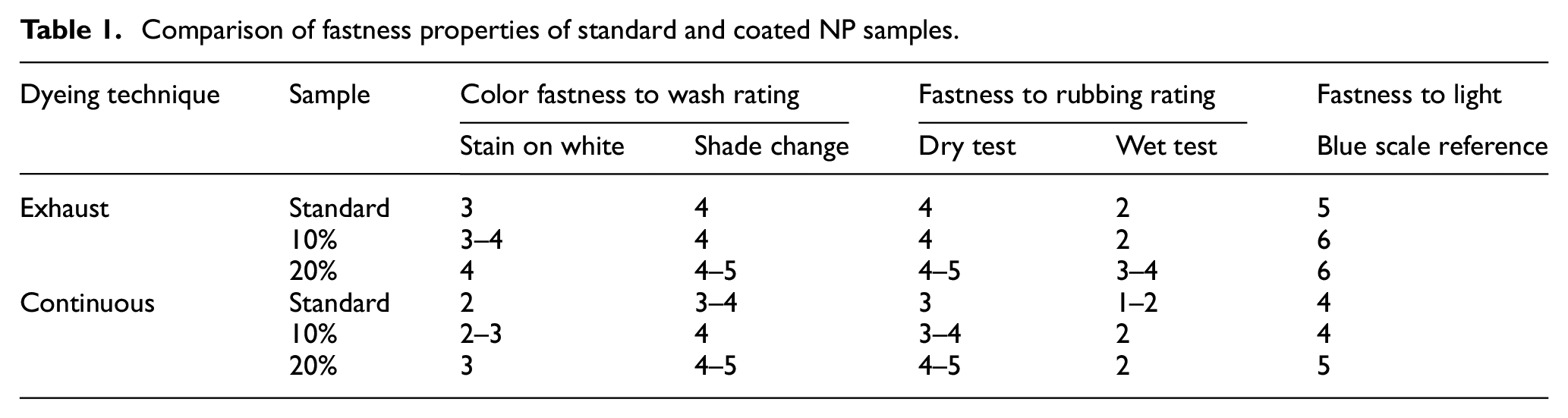
As per standard ISO 105 C06, color fastness to wash, the wash fastness ratings are 1: Very poor, 2: Poor, 3: Moderate, 4: Good, 5: Very Good, and 6: Excellent. The wash fastness value of the sample treated with 20% copper acetate solution was found to be one grade higher and better than the standard sample. There was a gradual increase in all the wash fastness values as compared to the standard, ranging from 2 to 5, which endorsed the established results. 32 The washing fastness values were improved due to the modification of the surface of the fabric, which strengthens the linkage between the dye molecule and the fiber.
Fastness to Rubbing
As per standard ISO 105 X-12, rubbing fastness ratings are 1: Very poor, 2: Poor, 3: Moderate, 4: Good, 5: Very Good, and 6: Excellent. The rubbing fastness values of standard and treated samples are given in Table 1. It was observed that the dry rubbing fastness values were better than the wet rubbing fastness values due to treatment of copper acetate, which influenced the rubbing fastness results. It was found that treatment with 20% copper acetate showed the highest dry and wet rubbing values as compared to the standard sample, while in the case of 10% copper acetate, there was no change observed.
Fastness to Light
The light fastness test was conducted by the Xenon test 450 procedure prescribed by ISO 105 BO2. Fabric samples of size 1 × 6 cm were cut from different areas of the fabric. The specimens were mounted on a the chart of size 3 × 8 cm leaving 1.5 cm above and below, to allow it to be fixed in the machine. The fabric was exposed to light for 48 h using a in xenon arc lamp of 500 watt. The distance between specimens and light was 8–12 inches. Then the fabrics were unmounted and assessment was carried out for shade variation using a blue reference scale. It is evident from Table 1 that untreated samples of copper acetate and CuNPs were influenced equally to raise the light fastness value up to 5–6 and 4–5 for exhaust and continuous dyeing techniques, respectively. These results show that the increase in light fastness due to the polarity developed in the fabric by CuNPs or by using copper acetate, which acts as a mordant, resulted in better attachment of the dye and fiber.
Conclusion
The nano-sized copper particles were synthesized successfully and applied to cotton fabric. The average particle size was found to be about 70 nm. For comparison, along with CuNPs, copper acetate solutions of 10% and 20% concentration were also prepared for the treatment of the fabric. Copper acetate and CuNP treatment on cotton enhanced the dye-ability of sulfur dye and led to improvements in the tensile strength of the cotton fabric. An improvement in the color strength (K/S) values of sulfur dye was also observed. Fastness properties such as wash, rub, and light fastness of sulfur dyed cotton fabric were also improved due to copper acetate and CuNP treatment. Thus, the copper nano-treatment upgraded the effective durability of the treated fabric in terms of their strength and dye uptake. These results show the potential application of the proposed process in the dyeing industry for cotton fabric in an eco-friendly manner.
Supplemental Material
sj-docx-1-aat-10.1177_24723444231161742 – Supplemental material for Non-Covalent Bonding of Green Synthesized Copper Nanoparticles to enhance Physicochemical behavior of Sulfur-Dyed Cotton Fabric
Supplemental material, sj-docx-1-aat-10.1177_24723444231161742 for Non-Covalent Bonding of Green Synthesized Copper Nanoparticles to enhance Physicochemical behavior of Sulfur-Dyed Cotton Fabric by Muhammad Amir, Kashif Ahmed, Syed Farhan Hasany and Rizwan Aslam Butt in AATCC Journal of Research
Footnotes
Acknowledgements
The authors acknowledge the financial and laboratory support of NED University of Engineering and Technology for conducting this research work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.