Abstract
In this study, recycling of solid textile wastes into activated carbon and the potential use of these activated carbons in color removal were investigated. Cotton and cotton/polyester-blended fabric wastes and textile wastewater treatment sludge were selected as solid textile wastes. A two-stage method, low temperature carbonization, and chemical activation with ZnCl2 were applied to prepare large surface area and rich-pore structure activated carbon from fabric wastes and sludge in textile industry. The activated carbons were characterized by elemental analysis, Fourier-transform infrared spectroscopy, X-ray fluorescent, and Brunauer–Emmett–Teller analysis. The reactive dye (Reactive Red 141) adsorption capacity of the activated carbons was investigated by the batch adsorption method. Activated carbon yields were found in the range of 28–34%. Cotton textile waste-based activated carbons were found to have the highest surface area (~1380 m2/g), followed by cotton/polyester and wastewater treatment sludge-based activated carbons. Wastewater treatment sludge-based activated carbons showed the highest dye adsorption capacity (161.29 mg/g), possibly due to its higher mesoporosity. The obtained results showed that the adsorption of the reactive dye onto textile waste-based activated carbons produced by two-step process is favorable. The developed activated carbons fit the circular economy approach well, offering a promising potential for solid textile waste management, as well as for water remediation.
Introduction
The textile industry is one of the world’s largest industries in terms of production and employment. It is one of the most polluting sectors with its wastes in various forms that are released during the industrial production and consumption phase. For this reason, sustainable production and waste management in the textile industry are among the issues that need attention. With the effects of fast fashion, textile consumption is constantly increasing; therefore, significant amounts of textile wastes are generated. It was reported that in the European Union (EU), the consumption of textiles has increased by 40% during recent decades, and 2–10% of the environmental impact is originated by the textile industry. 1 A range of waste is generated from the activities such as spinning, warp preparation, knitting/weaving, dyeing, printing, washing/drying, finishing, quality and process control, and warehousing depending on the production route. The major solid wastes generated by the textile industry are fibrous wastes. Some of them are recycled into low value-added products such as filling materials, blankets, and felts, and the rest is disposed of. In addition, wastewater treatment sludge is also an important source of solid waste in this industry. It contains high amounts of hazardous materials such as heavy metals, dyes, and toxic organic chemicals originating from various textile finishing processes. Combustion and landfilling are the two conventional techniques that are frequently used for sludge disposal; however, the risk of secondary pollution in incineration by flue gas and the risk of water and soil pollution in landfilling still remain a problem for textile wastewater sludge.2,3 Besides solid wastes, the textile industry is one of the largest producers of dye effluent. Adsorption process seems one of the attractive methods in textile wastewater control in terms of cost, simplicity of design, and operation.
An alternative approach to pre-consumer textile wastes and textile wastewater treatment sludge (TWS) is to recycle these wastes into high value-added products. In this sense, the use of huge amounts of wastes as precursors of carbon adsorbents is one of the strategies that enables the problem of waste disposal and recycling to be dealt with. Activated carbons are amorphous carbonaceous materials with high porosity, large surface area, and high surface reactivity. 4 They are excellent and versatile adsorbents for the removal of dyes, odorous substances, and other pollutants from air or aqueous medium. Adsorption can be either physical or chemical, or both, depending on the properties of the adsorbent and adsorbate. The pore development and surface characteristics of activated carbons such as surface area, microporosity and pore volume, and the molecular dimensions of the adsorbate are of great importance on the performance of the physical adsorption process. Although microporous structure is generally required for adsorption, mesopores are also of interest in the case of adsorbates with large molecules such as dyes. Besides, the chemical structure of the dyes and surface functional groups determine the availability of chemical adsorption (chemisorption). In the case of adsorption from solutions, the concentration of the solution, the amount of the adsorbent, and solution pH are also important additional factors.4,5
Activated carbons are produced from a wide range of carbonaceous materials by physical and chemical activation. In the physical activation, carbonized raw material is exposed to activation in carbon dioxide or water vapor medium at high temperature. The chemical activation, on the contrary, includes impregnation with chemicals such as zinc chloride, potassium hydroxide, and phosphoric acid followed by simultaneous carbonization and activation in one step. In comparison with physical activation, two important advantages of chemical activation are lower activation temperature (400–600°C) and an activated carbon with high yield and low ash content.
In recent years, there have been studies on the production of activated carbon from fabric wastes. In these studies, cotton (CO) fabric wastes were observed to be one of the most used precursors attributing the high cellulose content of CO fibers. For instance, Boudrahem et al. 5 produced activated carbons from CO cloth waste by chemical activation with H3PO4 and investigated the adsorption of pharmaceutical compounds. Their study highlighted that activated carbon prepared from the CO cloth waste using phosphoric acid is a promising adsorbent. Similarly, the activation of cellulose-derived denim residues by H3PO4 yielded an activated carbon having well-developed porous structures and good performances for Pb2+6 and textile dye 7 removal. In another study, the activation behavior of CO textile wastes in the presence of ZnCl2, H3PO4, and FeCl3 was comparatively investigated by Xia et al., 8 and it was reported that activation with ZnCl2 produced the activated carbon having the largest specific surface area of 1854.70 m2/g with mesopores domination. Recently, the magnetic activated carbon was also produced from CO textile using FeCl3 and/or FeCl2 as activating agent.9–13 In addition to CO fiber, there are also studies on activated carbon production from other cellulosic fiber wastes such as flax, 14 jute, 15 waste polyester textiles,16,17 and waste acrylic textiles.18–20
Since most waste textiles are composed of a mixture of different fibers to achieve excellent clothing performance, the activated carbon production from mixed waste textiles is also important. There are a few studies regarding activated carbon production from mixed fibers. Zhong et al. 21 investigated the production of activated carbon from CO/polyester waste textile by activation with ZnCl2. They concluded that the CO/polyester-based carbon had great application potential to remove Cr (VI) due to the synergistic effect of CO and polyester. In contrast, Carraro et al. 6 reported that activated carbons prepared using cellulose- and cellulose/polyester-based denim had similar textural properties and surface chemical composition. They suggested that there is no influence of precursors’ composition in properties of activated carbon.
There are numerous studies regarding activated carbon production from sewage sludge; however, only a few studies have been found on the production of activated carbon from textile wastewater sludge. Kaçan and Kütahyalı 22 produced activated carbons from textile wastewater sludge by chemical activation with KOH and investigated the strontium adsorption capacity of the obtained activated carbons. In another study, Kacan 23 investigated reactive and disperse dye adsorption of textile wastewater sludge-based activated carbons, which were chemically activated by KOH. Wong et al. 2 studied reactive black and methylene blue adsorption of activated carbons produced from textile wastewater sludge by chemical activation with H2SO4, ZnCl2, and KCl. They showed that using H2SO4 as an activating agent led to higher adsorption performance compared with the others.
Textile wastes are generated from different types of raw materials in many different forms such as fibers, yarns, fabrics, and garments. Moreover, especially in recent years, fabrics can be produced from a mixture of many different fibers in order to improve usage and functional properties. Therefore, the resultant waste is quite heterogeneous both chemically and physically. Because of this, technical problems—such as the different carbonization behaviors of the fibers within the mixed waste, the differentiation of the amount of chemical uptake depending on the fiber type and fabric structure during impregnation, and the difficulties in the impregnation of activation agent—limit the direct use of waste textiles as feedstock in activated carbon production. These disadvantages can be overcome by the production of biochar by low temperature pyrolysis as a first process. During this process, volatile decomposition products are removed from the wastes and a homogeneous carbon-rich biochar is obtained in powder form. Therefore, the biochar production as a first step in chemical activation is a viable way.
The subject of this study is the production of activated carbons from textile industry generated solid wastes by two-step process (carbonization of a carbonaceous precursor followed by the chemical activation). Besides there being very limited research on the production of activated carbon from textile solid wastes, there is no study, to our knowledge, regarding activated carbon production from solid wastes of the textile industry by two-step chemical activation. The novelty of this study is that it is the first study to produce activated carbon from textile industry wastes in a two-stage process and to compare properties of activated carbons according to the type of waste fiber. As solid wastes, CO and cotton/polyester (CO/PES) fabric wastes, which are the most common in the market today, 24 and TWS were chosen. Moreover, the reactive dye adsorption capacities of activated carbons obtained were also examined and compared. In this study, the reactive dye adsorption performance of activated carbons obtained from CO and CO/PES wastes was reported and compared with the textile wastewater treatment sludge (TWS)-based activated carbons at the same conditions for the first time. In this way, it was aimed to show that multi-faceted benefits can be achieved to ensure waste elimination, to obtain value-added products from textile industry wastes, and to use the resultant adsorbents in textile wastewater treatment.
Experimental
Materials
All wastes were supplied by a textile finishing company (Batı Basma A.Ş., Turkey) where dyeing and printing processes were carried out. Since they are common fiber types in the textile industry, CO and CO/PES (1:1)-based fabric wastes (white fabrics) were chosen as textile fabric waste. They were cut into 10 mm sizes. The waste sludge was dried in an oven at 105°C and then ground below 200 μm. Some properties of wastes are given in Table 1.
Elemental analysis and mass yield of resultant chars and activated carbons.
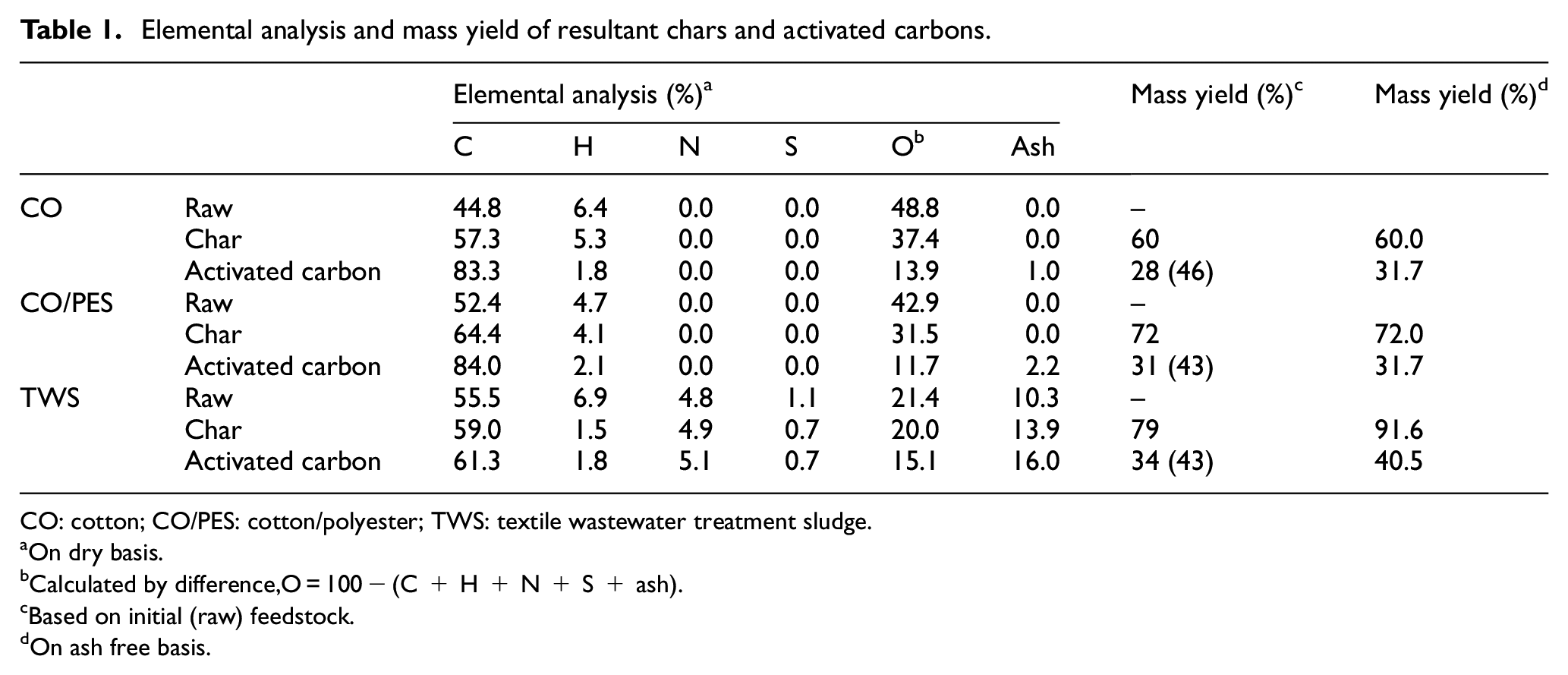
CO: cotton; CO/PES: cotton/polyester; TWS: textile wastewater treatment sludge.
On dry basis.
Calculated by difference,O = 100 − (C + H + N + S + ash).
Based on initial (raw) feedstock.
On ash free basis.
C.I. Reactive Red 141 (RR141) dye which is extensively used in the textile industry (chemical formula= C52H26O26S8Cl2N14Na8, and
Activated Carbon Production
Textile wastes were first converted into biochar, which is carbon-rich and homogeneous structure by low temperature pyrolysis. Then, biochar was activated with ZnCl2.
A stainless steel vertical bed reactor (
In activation, biochars were impregnated with ZnCl2 at a ratio of 1:1 (w:w) and kept in an oven at 105°C for 24 h. Then, the samples were loaded into the reactor, and the reactor was heated up to 550°C at a rate of 10°C/min, under nitrogen gas stream (25 mL/min), and held at this temperature for 1 h. After cooling, activated carbons were boiled with 10% HCl solution for 1 h, and then washed with hot distilled water for the removal of chlorides and other inorganics. After post-treatments, activated carbon samples were milled in a blade grinder (Grinding mill, A11 basic, IKA, Germany) and then sieved through 200 mesh screen, weighed and stored in sealed containers until further analysis.
The mass yields (

Characterization
Elemental analysis of the solid products (biochar and activated carbon) was carried out using LECO CHNS 932 elemental analyzer (St. Joseph, MI, USA) according to ASTM D5291-96. Also, X-ray fluorescent (XRF) spectrometry analysis was applied to TWS, TWS-based biochar, and activated carbon considering the high inorganic content of TWS in contrast to the negligible inorganic content of textile fibers.
Surface functional groups were analyzed using a Fourier-transform infrared spectroscopy (FT-IR) spectrometer coupled with attenuated total reflectance (ATR) (PerkinElmer Spectrum Two, Waltham, MA, USA). Moreover, acidic and basic surface functional groups were determined according to the Boehm titration method. 27
Multi-point BET (Brunauer, Emmett, and Teller) analysis by N2 adsorption at 77 K was employed by Quantachrome, Autosorb iQ Station 2 (Boynton Beach, FL, USA) to determine the surface area and pore characteristics of the resultant activated carbons. The pore size distributions of activated carbons were analyzed by the DFT (Density-Functional Theory) method.
Reactive Dye Adsorption Experiments
Since the reactive dyes are found in the wastewater mainly in their hydrolyzed form, in order to simulate a reactive dye liquor effluent, hydrolysate solution was prepared by the treatment of 2 g/L of RR141 with 50 g/L salt (Na2SO4) and 20 g/L Na2CO3 at 70°C for 1 h.
Adsorption experiments were carried out using the batch adsorption technique in conical flasks stirred on an orbital shaker (Nüve, ST 402, İstanbul, Turkey) at 25°C. A total of 50 mL of dye solutions of different initial concentrations (10, 50, 100, 150, and 200 mg/L) were prepared, and three different adsorbent dosages (0.5, 1, and 1.5 g/L) were used for each type of activated carbon at contact times of 0–24 h. Considering the pH of a typical textile wastewater, the adsorption experiments were conducted at pH 9. After adsorption, the samples were filtered and the supernatant liquids were analyzed by a UV-Vis spectrophotometer (PerkinElmer lambda 25) to calculate the residual dye concentration.
The decolorization efficiency was determined by the following equation:
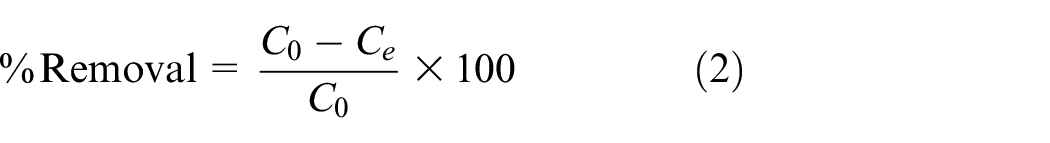
The amount of reactive dye adsorbed onto activated carbons (
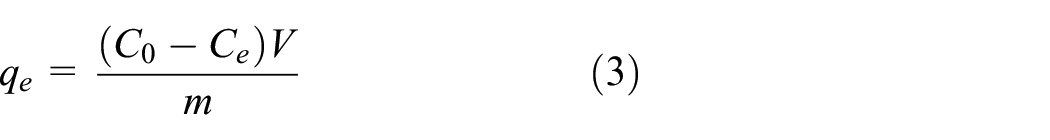
Isotherm studies were used to describe the equilibrium states of the produced activated carbons at different adsorbent concentrations after adsorption. For this purpose, the adsorption test data made with the adsorption time of 24 h and the adsorbent dosage of 1.5 g/L were used. In this context, experimental results were analyzed with the Langmuir and Freundlich isotherms.
Equations (4) and (5) express the Langmuir equation in a linear form and the dimensionless separation factor (

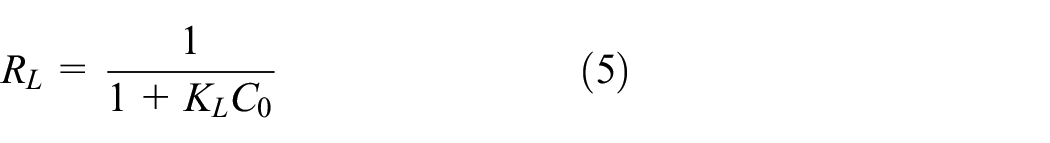
The Freundlich isotherm is expressed by equation (6) in a linear form, where

Results and Discussion
Mass Yield and Elemental Analysis
The mass yields of biochars and activated carbons obtained from CO, CO/PES, and TWS wastes are shown in Table 1. In the case of fibrous textile wastes, the mass yield of CO/PES-based biochars was found to be higher compared with CO-based samples. This was attributed to the higher thermal decomposition temperature of polyester fibers, which possibly did not breakdown under the operating temperature of biochar production. This result was in general agreement with the studies of Hanoğlu et al. 29 and Zhong et al. 21 TWS-based biochar had the higher mass yield compared with fibrous wastes, which may be due to the fact that it has different chemical composition from both natural and synthetic fibers. The major components of the organic fraction of activated sludge are extracellular polymeric substances, which are complex aggregates consisting of various microorganisms (mainly bacteria) embedded in a polymeric network, besides protein, polysaccharides, and lipids as well as organic micropollutants. 30 The yield of TWS char was similar with the other types of sludge reported in the literature. 31
As in biochar, the mass yield of wastewater treatment sludge-based activated carbon (TWS-AC) was higher than that of fibrous textile wastes. The yield of cotton textile waste-based activated carbon (CO-AC) was comparable with the yield of activated carbon obtained from jute waste 15 and waste CO fabrics 32 by ZnCl2 activation in one step. However, Carraro et al. 6 reported the activated carbon yields of 46.0% and 48.0% from CO and CO/PES, respectively, by H3PO4 activation in one step. Similarly, activated carbon from denim fabric waste with H3PO4 produced with a yield of 53% which was higher compared with activated with ZnCl2. 7 Compared with ZnCl2, H3PO4 strongly retards the evolution of volatiles, which lead to the promoting of the carbon yield. 33 In this study, ZnCl2 was chosen as the activation agent, because, at the activation temperature, phosphoric acid can combine with organic species in the carbon precursor and cause the phosphorus content by forming polyphosphate esters. 34 In addition, in case of TWS, the use of H3PO4 may lead to a mixture of P-metal complexes including Fe–P, Al–P, and Ca–P, with the various metal in the TWS.
Although the chars were free of inorganics, the fiber-based activated carbons contained little ash, probably due to the incorporation of zinc into the carbon matrix via C–O–Zn bonds. As expected, sludge-derived activated carbon contained high ash content, even about half of the ash in sludge was removed by demineralization with HCl. The reason probably is the existence of water- and HCl-insoluble compounds in raw sludge. Supplemental Table S1 shows the XRF analysis results of TWS-based samples. Silicon, sodium, and calcium were found to have the highest content in TWS. By acid washing, 60–95% of the inorganics (except Cl and Zn) in TWS were removed from the activated carbon, while Mg and P were completely removed. It should be noted that TWS-based activated carbon contains more Cl and Zn than predicted, due to the use of ZnCl2 for activation, despite repeated washings after activation.
Elemental analyses of the resultant biochar and activated carbons are also given in Table 1. Pyrolysis and activation led to an increase in the carbon content and a decrease in the oxygen content of CO and CO/PES samples as expected. Regarding elemental analysis composition, TWS-AC had much lower carbon and higher nitrogen and ash contents whereas its contents of ash, hydrogen, nitrogen, and oxygen were higher compared with the fiber-based activated carbons. However, only a slight difference in the carbon, oxygen, and nitrogen ratio was observed for the TWS-AC compared with the raw TWS. We can assume that the removal of carbon and non-carbon species occurs at the same extent during carbonization.
Surface Chemistry of Activated Carbons
FT-IR analyses were applied to precursor wastes and resultant biochars and activated carbons to detect the structural changes and surface functional groups. Supplemental Figure S1 illustrates the FT-IR spectra of CO waste-based samples. It was observed that the O–H stretching peak at 3100–3600 cm−1 weakened after biochar production to a great extent and completely disappeared by the activation process. In addition, the peak at 2900 cm−1 corresponding to the aliphatic C–H stretching disappeared by activation, which indicates the aromatization of the structure. In the spectrum of CO fiber, characteristic peaks of carbohydrates were observed in the range of 900–1200 cm−1, such as the C–O–C stretching in pyranose ring at 1160 and 1109 cm−1 and C–O stretching at 1030 cm−1. 35 The peak intensity of carbohydrate bands was significantly reduced by pyrolysis and activation, which denotes the dehydration and depolymerization of cellulose. Furthermore, new peaks were appeared at 700–900 cm−1 and 1556 cm−1 assigned to aromatic C–H deformation and C=C vibration of aromatic rings, respectively.36,37 A peak characterizing carboxylic acid groups emerged at 1697 cm−1 in activated carbon samples. As a result, it can be reported that along with aromatization, acidic functional groups emerged by an activation process.
Supplemental Figure S2 shows the FT-IR spectra of CO/PES-based samples. As in CO-based samples, the O–H stretching band in the range of 3100–3600 cm−1 for CO/PES wastes disappeared as a result of the pyrolysis process. Carbonyl (C=O) stretching at 1713 cm–1, aromatic ester asymmetric stretching at 1241 cm–1, and aromatic C–H out-of-plane vibration at 722 cm−1 can be seen as the characteristic peaks of polyester fiber. 16 It was observed that there was no change in the bands characterizing polyester in the spectrum of the CO/PES biochar. This indicated that the polyester part of the blend did not decompose during the biochar production process. The high mass yield of CO/PES-based biochar (Table 1) also supports this result. However, the peaks that characterize the polyester disappeared after the activation process, indicating that the polyester part was also carbonized. As in CO-based samples, the intensity of the peaks in the range of 900–1200 cm−1 that characterizes CO fibers decreased by pyrolysis and new aromatic bands appeared (i.e. aromatic C=C stretching at 1555 cm−1). It was investigated that the spectra of cotton/polyester-based activated carbon (CO/PES-AC) have the same peaks as CO-AC.
The FT-IR spectra of TWS-based samples came up with many functional groups as shown in Supplemental Figure S3. The wide peak in the range of 3100–3600 cm−1 corresponds to the O–H stretching associated with water, alcohols, and carboxylic acids 38 and the N–H stretching associated with organic compounds. 39 It was observed that this peak disappeared after pyrolysis and activation. The peaks found at 2919 and 2851 cm−1 indicated asymmetric and symmetrical C–H stretching in the alkyl groups. After pyrolysis and activation, these peaks were largely eliminated, which indicates that aliphatic C–H chains decomposed into carbon dioxide, methane, or other gases or turned into aromatic structures. 38 Peaks in the range of 1400–1750 cm−1 were assigned to COO, C=O, and C–N stretching, while peaks in the range of about 1000–1050 cm−1 corresponded to C–O–C and C–O stretching, probably due to carbohydrates in the structure. 40 The peak at 874 cm−1 attributed to aromatic rings. After pyrolysis and activation, these peaks were observed to be largely eliminated and new aromatic structures emerged (i.e. aromatic C=C stretching at 1576 cm−1 and aromatic C–H stretching at 880 cm−1).
Besides textural properties, the oxygen functional groups are also important characteristics of the carbons, which determine their quality as adsorbents. The amount of acidic oxygen groups (carboxyl, lactonic hydroxyl, carbonyl groups) and basic oxygen groups (pyrones, ethers, carbonyls) on the activated carbon surfaces were determined by the Boehm titration. The amount of acidic surface oxides in TWS-AC was higher than those of other activated carbons, and no basic group was encountered (Table 2). CO-AC involved both acidic and basic groups. In the case of CO/PES-AC, acidic surface oxides were found to be predominant and traces of basic groups were identified. These results for CO and CO/PES-based wastes are in parallel with the studies of Silva et al. 7 and Carraro et al., 6 in which activated carbons were produced from CO and CO/PES denim fabrics, respectively.
Surface oxygenated groups on activated carbons and BET analysis results.
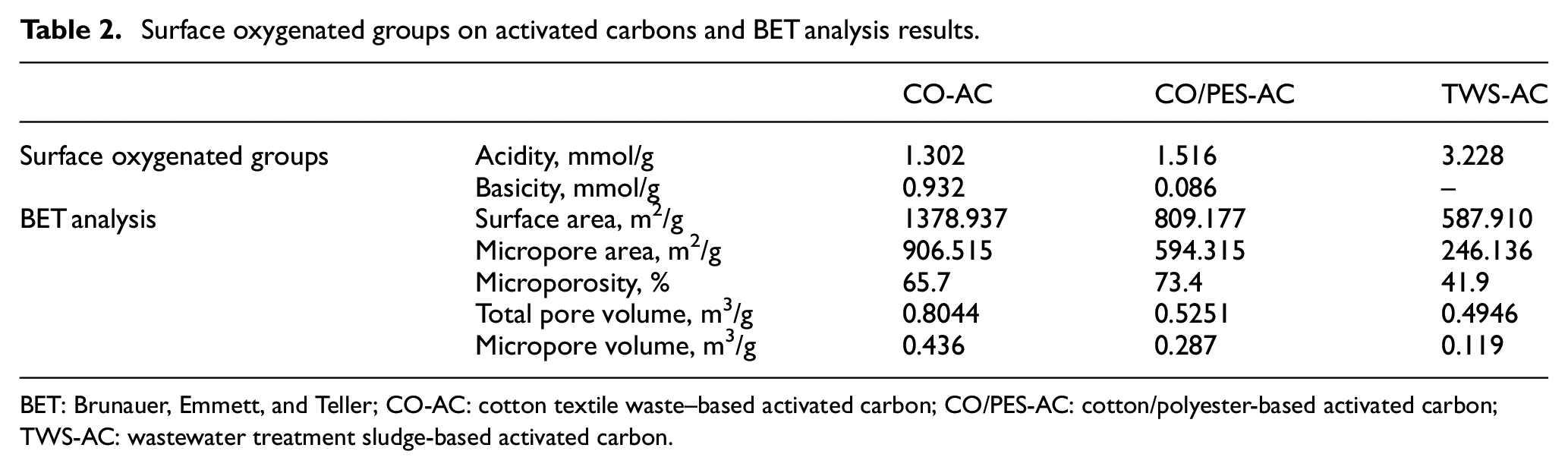
BET: Brunauer, Emmett, and Teller; CO-AC: cotton textile waste–based activated carbon; CO/PES-AC: cotton/polyester-based activated carbon; TWS-AC: wastewater treatment sludge-based activated carbon.
BET Surface Area and Pore Size Distribution
BET surface area and pore size distribution of the produced activated carbons are given in Table 2. The use of CO wastes as precursor led to the highest surface area, followed by CO/PES and TWS, respectively. The pore size distribution according to the DFT method is illustrated in Supplemental Figure S4. The results indicate that all of the activated carbons produced have pores distributed over a broad range, including micropores and mesopores. And the DFT distributions showed that the pore structure in CO-AC and CO/PES-AC was mainly micropores, while it was mainly mesopores in TWS-AC.
To explain the observed surface area and porosity, we can speculate that the molten ZnCl2 acted as templates inside the carbon to form pore structures. The obtained surface areas of CO-AC and CO/PES-AC were comparable with the fabric-based activated carbons reported in the recent literature obtained by one-stage activation as given in Table 3. Literature includes a few references regarding the activated carbon production from textile sewage sludge. In those references, reported BET surface areas were 135 m2/g 22 and 336 m2/g 23 by KOH activation and 221.52 m2/g2 by H2SO4 activation. However, in the literature, the use of sludge-derived char to produce activated carbon has been less studied. Li et al. 41 produced activated carbon from sludge-derived char by ZnCl2 activation. They obtained activated carbon having BET surface areas of 765.88 m2/g. In another study by Alvarez et al., 42 the activated carbon with a BET surface area of 440 m2/g was produced by CO2 activation of the sewage sludge char. As can be seen, TWS-AC had higher or comparable BET surface area compared with textile sewage sludge and/or sludge derived char-based activated carbons. It is worth noting that the adsorption performance of activated carbons depends on not only the surface area but also the pore size, target adsorbate, and surface functional groups, as discussed in the following section.
Recent reported properties of activated carbons produced from solid textile wastes in the literature.
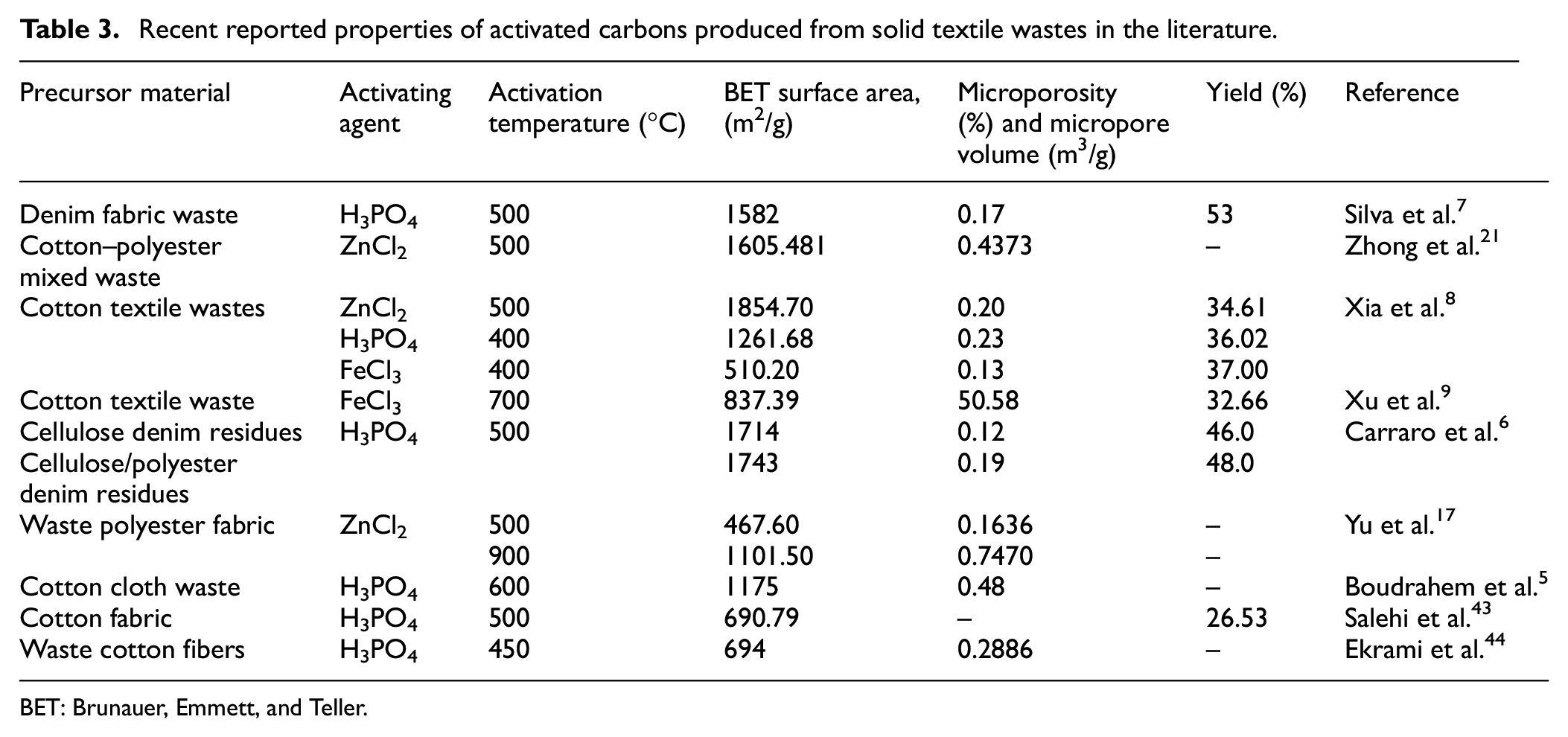
BET: Brunauer, Emmett, and Teller.
Reactive Dye Adsorption
The decolorization efficiency of TWS-AC, CO-AC, and CO/PES-AC for the adsorbent dosage of 1.5 g/L is illustrated in Figure 1 according to the adsorption time (0.5–24 h) and dye concentration (10–200 mg/L). The results for all adsorbent dosages (including 0.5 and 1 g/L) are also given in Supplemental Figures S5–S7 for TWS-AC, CO-AC, and CO/PES-AC, respectively. The decolorization efficiency of the activated carbons was ranked in the order of TWS-AC > CO-AC > CO/PES-AC. In all cases, by increasing the initial dye concentration, the percentage removal decreased, while it increased with increase in adsorbent dosage, as expected. For instance, in case of TWS-AC, depending on the contact time, 65–80% of dye was adsorbed for the 200-mg/L initial dye concentration and adsorbent dosage of 0.5 g/L. When the adsorbent dosage increased up to 1.5 g/L, 81–95% of adsorption was observed even for the 200 mg/L initial dye concentration, and 83–99% for 10 mg/L.
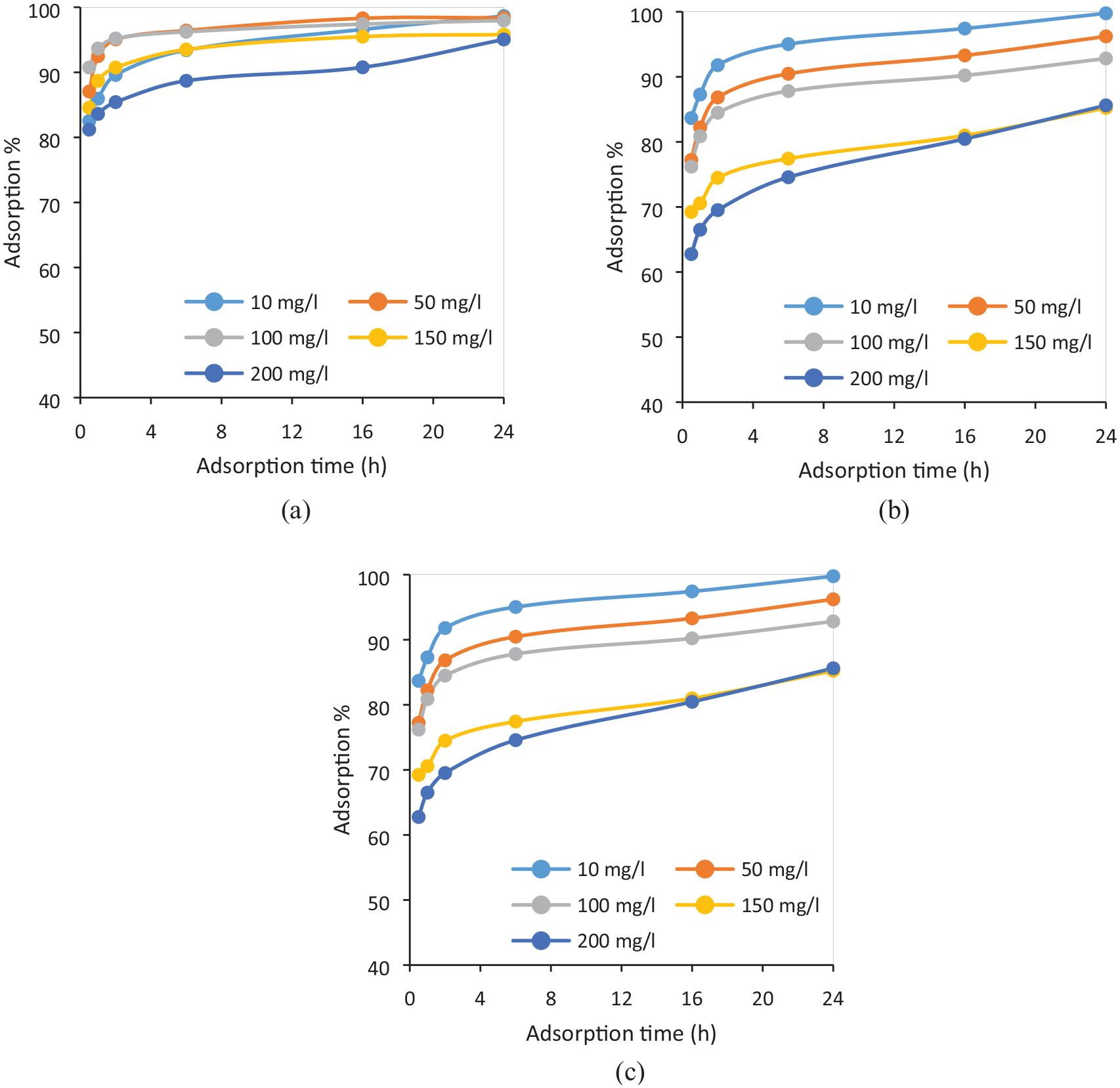
RR141 adsorption % on (a) TWS-AC, (b) CO-AC, and (c) CO/PES-AC for the adsorbent dosage of 1.5 g/L.
The dye adsorption was rapid in the first stage of the adsorption process, followed by a gradual adsorption. As known, the adsorption rate varies depending on several parameters such as stirring rate, structural properties of adsorbent, adsorbent dosage, and adsorbate properties. The high adsorption rate in the first stage of the adsorption process might be that the dye adsorption takes place first on the exterior surface of the adsorbent. When saturation is reached on the outer surface, the dye molecules begin to diffuse from the pores to the inner surfaces of the adsorbent, which takes a relatively longer time. 45 Although, most of the dye was adsorbed within the first 2 h of contact between the adsorbate and the adsorbent, an equilibrium time of 24 h could be proposed to make sure that fully equilibrium was attained.
Among the activated carbons, the highest dye removal was achieved by TWS-AC. The three main parameters affecting the adsorption behavior of an adsorbent are surface area, surface groups, and porosity. Reactive dyes are the large molecules (e.g. the dimensions of RR141 is 1.72 nm × 2.68 nm × 1.66 nm); 46 therefore, mesopores play a large part in adsorption. Although TWS-AC has the lowest surface area in this study, the reason for its high adsorption performance might be related to its higher mesoporous structure. In addition, it was thought that the higher inorganic content of TWS-AC might contribute to the higher adsorption capacity. However, CO/PES-AC showed similar adsorption capacity compared with CO-AC, although it had lower surface area.
Table 4 shows the Langmuir and Freundlich isotherm parameters for each type of activated carbon. Adsorption isotherms were also illustrated in Supplemental Figure S8. The Langmuir isotherm assumes that the adsorption occurs on a homogeneous surface containing sites with identical sorption energies.
47
The Freundlich isotherm, on the contrary, is used for heterogeneous systems. The results obtained from adsorption of RR141 on sludge-based activated carbons conformed well with both the Langmuir and Freundlich equations; however, higher
The Langmuir and Freundlich isotherm parameters.
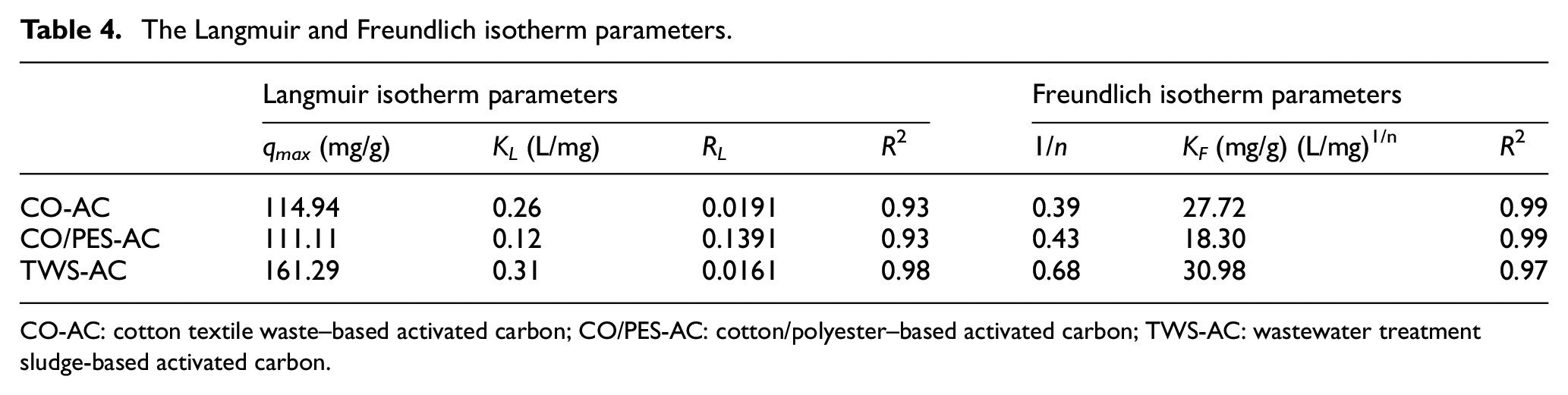
CO-AC: cotton textile waste–based activated carbon; CO/PES-AC: cotton/polyester–based activated carbon; TWS-AC: wastewater treatment sludge-based activated carbon.
As is well known, both the surface charge of adsorbent and the ionic forms of the dye in solution play an important role in adsorption process. One can think that surface charge of adsorbent becomes negative at higher pH values, and therefore, the adsorption of anions is not favored. In this study, the adsorption experiments were conducted at pH 9 by hydrolysate RR141 solution to simulate a typical textile wastewater. The fact that sorption capacity of activated carbons produced in this study is comparable with that of other activated carbons from different type of sources,26,49,50–53 even though their surfaces are negatively charged could be explained by the adsorption of dye by non-electrostatic. As discussed in the study of Ip et al. 54 who investigated the Reactive Black dye adsorption onto different adsorbents, the effect of the van der Waals attraction and π–π interactions seems to be dominant and controls the adsorption. Similarly, although CO/PES-AC had lower meso- and macro-porosity but higher surface area than CO-AC, its dye adsorption capacity was found almost similar with that of CO-AC. This showed that the RR141 adsorption on these adsorbents was controlled mainly by the van der Waals attraction and π–π interactions, as in the case of TWS-AC.
Conclusion
Activated carbons were produced from CO and CO/PES fabric wastes and TWS by chemical activation method and comparatively investigated. The main experimental findings are listed as follows:
The highest surface area was obtained with CO-AC (∼1380 m2/g), followed by CO/PES-AC and TWS-AC, respectively. Fabric waste-based activated carbons had higher microporosity, while TWS-AC was found to be more mesoporous. The microporosity is followed the order of CO/PES-AC > CO-AC > TWS-AC.
The RR141 removal performances were determined as in the order of TWS-AC > CO-AC > CO/PES-AC. Despite having the lowest surface area, the best color removal with TWS-AC was associated with the structure being more mesoporous. Since the reactive dyes are large molecules, the adsorption performance of activated carbons decreased as the microporosity increased.
The results of the study showed that textile solid wastes, which have non-uniform size and handling difficulty, could be transformed into a high value-added product through upcycling into activated carbon by two-step processing. Furthermore, these activated carbons can be successfully used as an adsorbent for color removal in textile wastewater.
Supplemental Material
sj-docx-1-aat-10.1177_24723444221147983 – Supplemental material for Valorization of Solid Wastes from Textile Industry as an Adsorbent Through Activated Carbon Production
Supplemental material, sj-docx-1-aat-10.1177_24723444221147983 for Valorization of Solid Wastes from Textile Industry as an Adsorbent Through Activated Carbon Production by Emire Ülkü Parmakoğlu, Ahmet Çay and Jale Yanık in AATCC Journal of Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ege University Scientific Research Projects Coordination Unit (Project no.: FYL-2019-20618).
Data availability
The authors can confirm that all relevant data are included in the article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
