Abstract
The fast fashion trends in the textile industry have resulted in high consumption of fiber with concomitant generation of waste. Awareness of environmental pollution resulting from textile production and disposal has increased significantly. This increase has pushed research activities toward more sustainable recycling alternatives to properly handle the end-of-life of textiles. This review provides an overview of existing technologies, the latest developments, and research studies on the recycling technologies employed in the textile industry. Different types of recycling—mechanical, chemical, and biochemical recycling of standard fabrics used in garments, cotton, wool, polyester, polyamide 6 6, and acrylic—are explored. Recent advances in recycling technologies such as advanced sorting techniques, innovative chemical processing, and emerging biochemical processes are revealed. The review also highlights efforts being made by various agencies and companies in facilitating and employing the technologies on a commercial scale. Various methods for efficient textile waste sorting and identification are also discussed. The reviewed studies revealed that most recycling technologies were conducted on post-industrial textile waste, which tends to be homogenous in the types of dyes and fibers present in the waste. It also suggested that post-consumer textiles could be recycled using chemical and biological options that have the potential to valorize the waste into high-value products.
Keywords
World fabric manufacturing has continuously risen over recent years, and production is expected to increase. Clothing has been subjected to fashion trends and styles worldwide 1 that change frequently. However, this dramatically reduces the clothing life cycle before disposal in landfills. Although the fast change in clothing results in the growth of the textile industry, its downside is that it generates significant textile waste. 2 The disposal of waste textiles has since become undesirable and costly due to the unavailability of space for continual landfilling, the environmental risks, and the costs associated with waste disposal in landfills. Thus, a reduction in the use or recycling of textile waste will benefit the environment and reduce the reliance on non-renewable fossils used in the production of fibers. Existing reviews provide an overview of various textile waste recycling technologies to recover fibers from waste into different products.3 –6 This review focuses on recent advancements in the recycling technologies employed in the recycling of textile fabrics. Detailed information on the type of current recycling, emerging technologies, and recent developments in the identification and sorting of waste is provided. Finally, challenges in implementing these technologies and potential solutions are discussed.
The textile industry contributes about 5% of worldwide waste. 7 According to the European Environmental Agency, European consumers discard about 11 kg of textiles per person every year. The industry produces many waste streams, including post-industrial, pre-consumer, and post-consumer waste. The post-industrial waste consists of waste fibers or cutting scraps from industrial processing, such as spinning, weaving, or apparel manufacture. 8 Pre-consumer waste is any material that is discarded before it reaches the consumer, such as faulty items, overstocked raw materials, or excess inventory. Products such as clothing disposed of by consumers at their end of use are classified as post-consumer waste. Generally, textile fabrics are knitted or woven from natural, regenerated, or synthetic fibers,9,10 and are further dyed and finished to improve their aesthetics. However, when discarded, textiles result in challenges of metals leaching from dyes and finishing chemicals into the environment, posing environmental and health hazards to the ecosystem and underground water sources. 11
The most dominant synthetic fibers are from petrochemicals: polyethylene terephthalate (PET), polyamide 6 6, and acrylic fibers. Synthetic textiles have a significant carbon footprint, producing around 15–35 tonnes of carbon dioxide (CO2) equivalent per tonne of textiles produced. 12 In Europe alone, 654 kg of CO2 equivalent per person emissions were recorded in 2017 from the production and handling of clothing, footwear, and household textiles. 12 Likewise, a study in Sweden estimated emissions from clothing consumption to be 330 kg CO2 equivalent per person annually, which is 3% of the estimated carbon footprint per person. 13 To reduce the emissions of greenhouse gases and exploitation of fossil fuels, textiles have been produced from renewable sources, such as cotton, wool, silk, and viscose. However, cotton, the second largest dominant fiber in textile waste, requires large amounts of water and pesticides during its cultivation. A possible intervention could be reducing the cultivation of new cotton by replacing it with recycled regenerated cellulose fibers from the cotton waste that is being thrown in landfills. Recent developments have resulted in the production of regenerated fibers such as Lyocell and Ioncell fibers, which are derived from renewable wood cellulose.14 –16 These are sometimes blended with synthetic fibers to reduce the amount of synthetic portion in the textiles.
Recent reports have estimated that 75% of materials used in the production and after the use of clothing are landfilled or incinerated.4,11 Approximately 23% of discarded clothing is collected for recycling, with less than 1% of the recovered fibers used to reproduce clothing, closing the loop in fiber-to-fiber recycling. 17 Figure 1 illustrates the flow of waste throughout the production and end use of textiles.
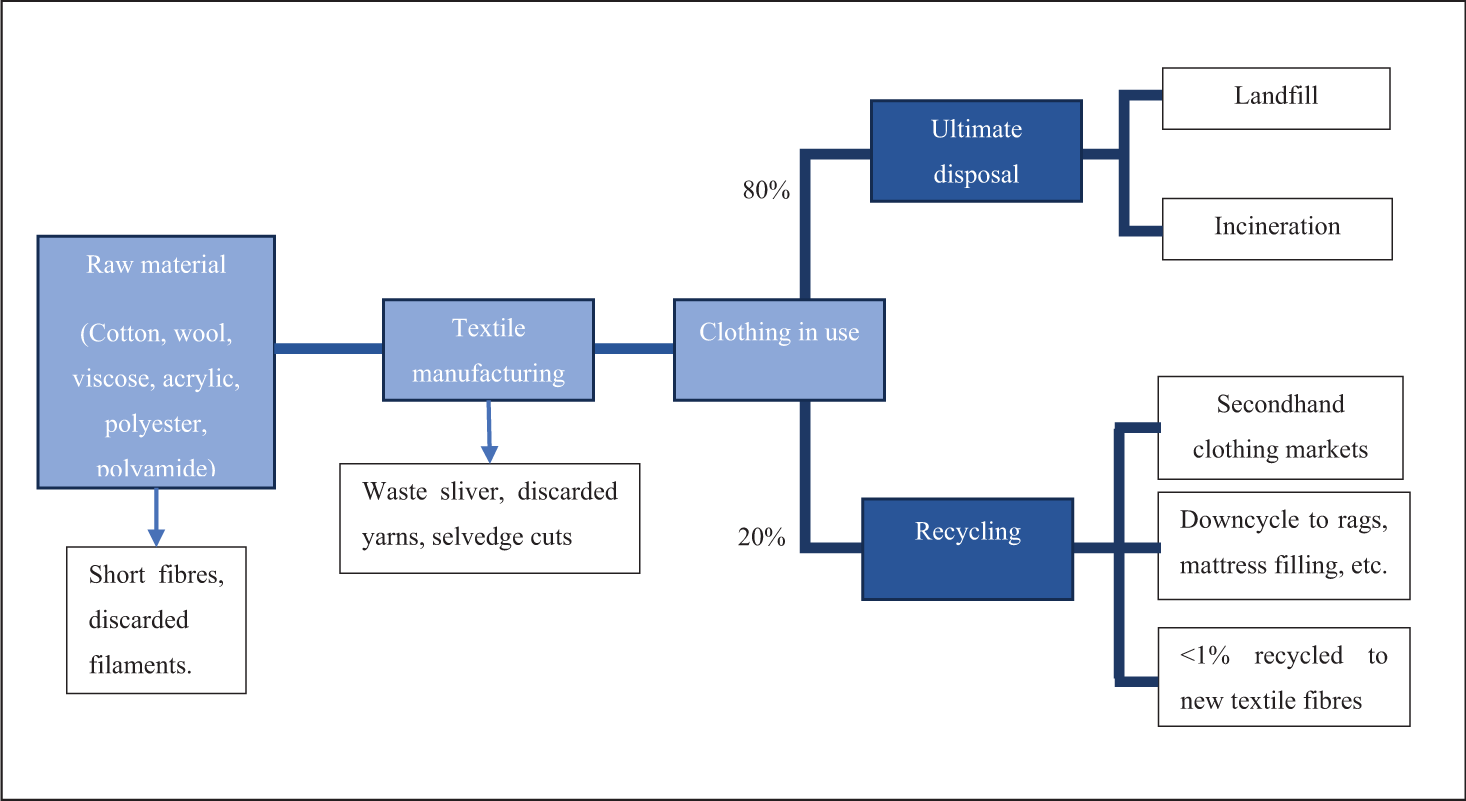
Textile waste flow from raw material to disposal.
According to Environmental Protection Agency data, the USA recorded 11.3 million tonnes of textiles disposed of in landfills in 2018, making up 7.7% of the total municipal solid waste generation that year. 18 The data relating to the total number of tonnes of textiles generated, recycled, combusted with energy recovery, and landfilled from 1960 to a projection to 2050 is shown in Figure 2. It is evident from Figure 2 that textiles generated over the years end up in landfills. Recycling of textile waste has been reported since the 1980s. However, recycling has not improved significantly since 2000. Production of textiles has skyrocketed, showing over 80% growth since 1980, but the recycling of the generated waste has not been growing at the same rate. The generation of textiles is expected to rise to approximately 28 million tonnes by 2050 in the USA alone. This is estimated to generate approximately 19 million tonnes in landfills and continues to result in the accumulation of waste.
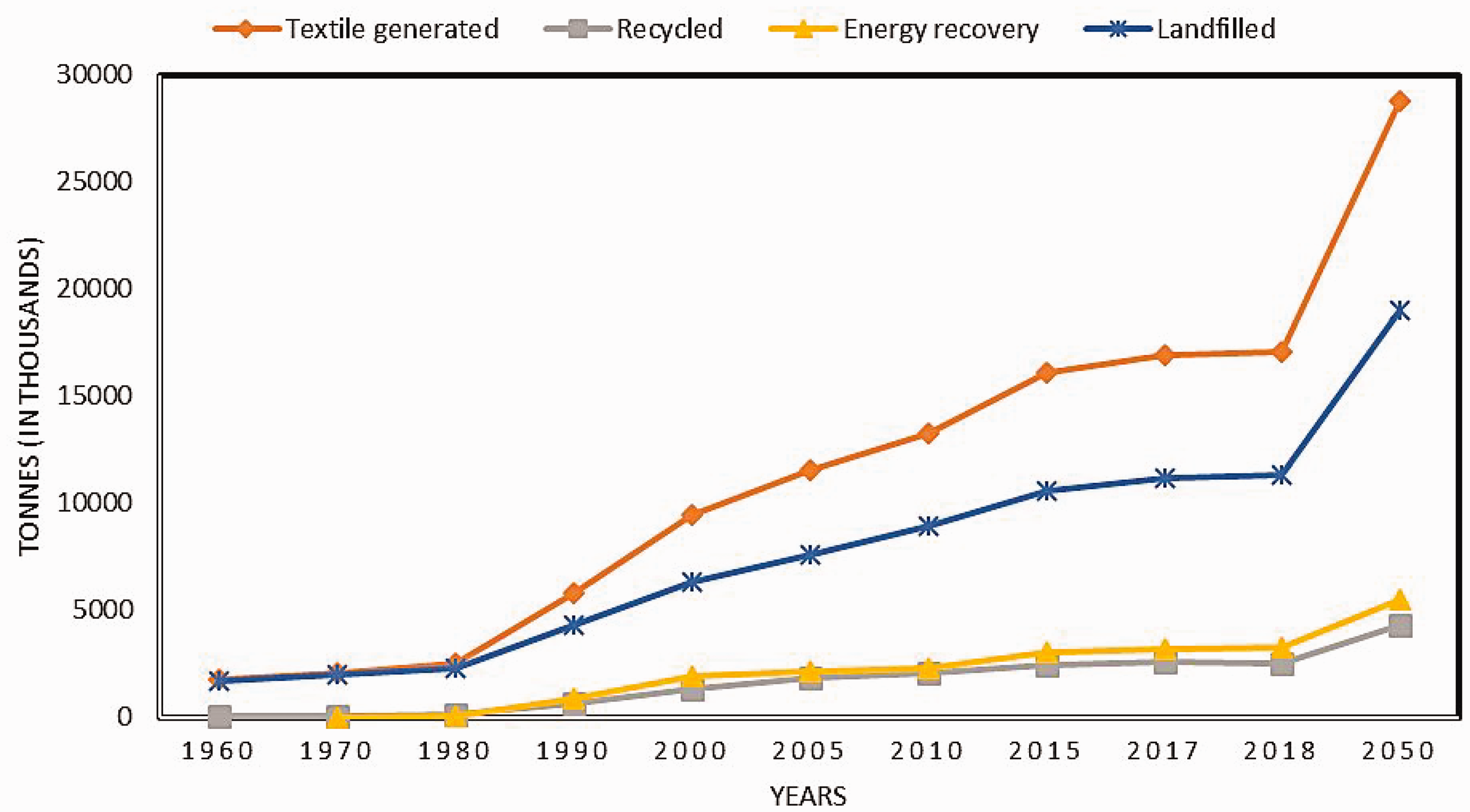
Data on textiles generated and the waste streams generated in the USA. 18
Waste generation has changed since the COVID-19 pandemic in 2020. The COVID-19 pandemic greatly affected global spending patterns and fabric production, resulting in reduced waste generation. With the world on lockdown, buying patterns and waste production of textiles were reduced. Many individuals purchase clothing considering special events, such as weddings, parties, and holidays. However, as many of these occasions were cancelled, the impulse to purchase new clothing was no longer there. In addition, the closure of shipping operations from European and Asian countries reduced the supply of second-hand clothing to African countries, leading to decreased waste flow to landfills. Research done by Vladimirova et al. 19 provides a comparative overview of fashion consumption practices and the changes that occurred during COVID-19 across nine countries. Among the countries surveyed, 56% of respondents reported that spending and shopping frequency for clothing decreased, 33% remained the same, and 11% reported the patterns increased. 19 In contrast, the outbreak of COVID-19 forced the textile sector to focus on developments that enhanced human beings' protection by producing personal protective clothing materials, such as protective gowns, masks, and surgical shoes, which are made from polypropylene, polyacrylonitrile (PAN), polyurethane (PU), or blends of these. 20 This personal protective equipment (PPE) has generated a waste mountain of PPE waste worldwide since 2020, which is already causing environmental concern. A study in Peru reported the generation of waste from face masks to be roughly 74.9 tonnes of plastic waste daily (27,344.7 tonnes per year) based on the population of the region and an average mass weight of 5g per mask. 21 Waste generation estimated per continent is shown in Table 1.
The estimated quantity of masks used during the COVID-19 pandemic 20
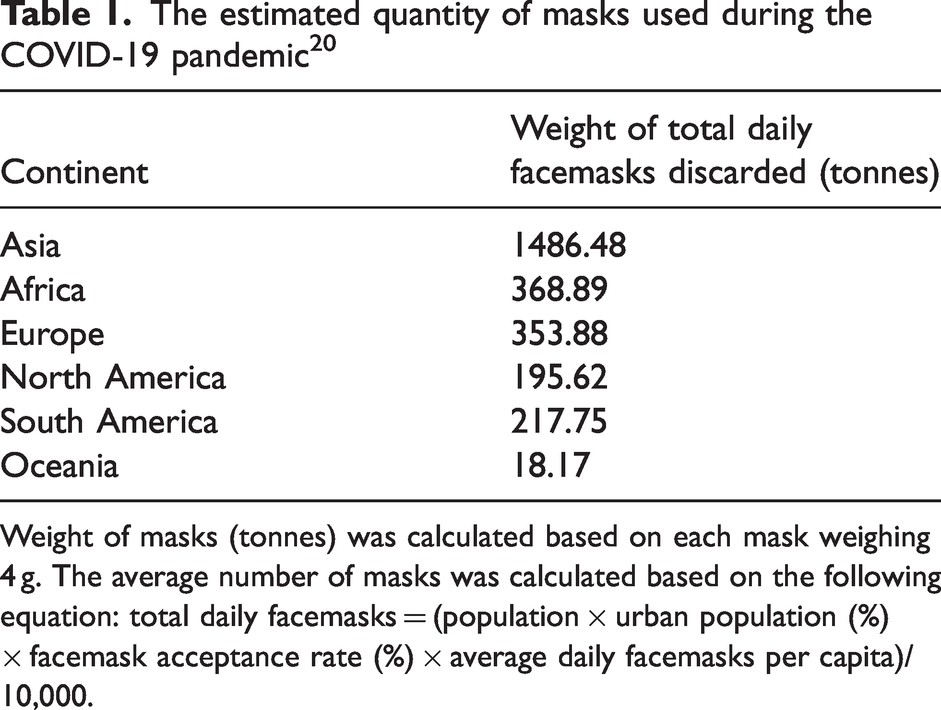
Weight of masks (tonnes) was calculated based on each mask weighing 4 g. The average number of masks was calculated based on the following equation: total daily facemasks = (population × urban population (%) × facemask acceptance rate (%) × average daily facemasks per capita)/10,000.
Environmental concerns about textile waste
The demand for fabrics has grown and, as a result, the adverse environmental impact of fiber production and disposal operations has also increased. Decomposition of synthetic textiles can take up to 200 years.10,22 –24 Furthermore, these synthetic fibers are not biodegraded but rather photodegraded; such fabric waste produced from these fibers has been in our environment for centuries. Natural fibers are degraded by living organisms into water, carbon dioxide, methane, and other non-toxic residue. During their decomposition in landfills, they leach residual toxic chemicals from dyes into groundwater and generate greenhouse gases into the air. Dyes in waste textiles consist of heavy metals and organic compounds. 25 Heavy metals from dyes and textile accessories typically include lead, copper, chromium, arsenic, nickel, silver, mercury, and zinc. 26 Such metals risk public health as they can bioaccumulate in the human body and pollute the environment through water bodies, aquatic life, and soils. 26
Discarded synthetic fabrics from polyester, acrylic, and polyamides mainly cause microplastic fiber pollution.27 –29 Tiny fragments from waste fabrics are released into drains and end up on the coastlines of densely populated areas. Waste synthetic textiles in landfills located near the water source, domestic drainages, rivers, and oceans are a secondary source of microplastics. Microfibers are prone to be absorbed or ingested by aquatic animals, which can also harm humans when consumed. Flying pieces of microfibers inhaled by humans deposit in the lung tissue and may lead to respiratory inflammation. 27 With the emergence of the COVID-19 pandemic, the production of non-woven masks from cotton and synthetic fibers increased, and with their disposable nature, many of these were thrown away. Due to their loose non-woven structure, the fibers are easily degraded to microplastic as the structure's integrity is weak. 30 Textile production should focus more on designing fabrics with a recycling mindset toward the reduction of microfiber pollution. For instance, fabrics should be constructed with a dense structure, since this tight structure is less prone to microfiber release.
Although incineration reduces waste piles in landfills, this has adverse health and environmental effects. The burning of textile fibers contributes to the emissions of carbon dioxide, methane, nitrogen oxides, and sulfur oxides. 31 These gases are reported to contribute to fog formation, create acidity in freshwater bodies, soils, and forests, and lead to the deterioration of metal structures. Furthermore, fabric chemical treatments during textile manufacturing increase the risk of environmental effects when incinerated or left to decompose in landfills. There is a need for a sound textile waste management system that reduces the environmental effects caused by waste textiles.
Textile waste management
Various textile waste management activities are being practiced worldwide to divert waste from landfills. Reuse, incineration, and recycling are the primary textile waste management techniques. Other recent waste management technologies include pyrolysis, and source management methods such as prolonging the life of textiles, buying less, and the production of recyclable textiles.
Reuse
Textile reuse is a waste management technique that focuses on various means for prolonging textiles’ helpful service from the first owner to another. 32 As a result, this extends the textile life, and there are reduced water, chemical, resource, and waste paths. 33 Reuse is commonly practiced by renting, trading, swapping, borrowing, and inheriting, facilitated by second-hand stores, garage sales, flea markets, and charities, known as the second-hand reuse market. 4 Retailers, such as Madewell and Hennes & Mauritz (H&M), have started a global garment collection program and will take used clothing and second-hand clothing for resale or use in housing insulation to keep items out of landfills. Organizations such as the Salvation Army and Goodwill have been recovering textile waste for decades, distributing second-hand clothes freely in the developing world. 17 The reuse procedure can be done from one person to another through sharing, repair, or resale in second-hand markets. Quite a big example of this system is the exporting of second-hand clothing for resale to Eastern Europe, Asia, or Africa. 34 Although the reuse of textiles has been practiced, there is a need for the industry to expand awareness campaigns to include information on reusing textiles, since most people are unaware of the extent of textile waste being produced globally. However, there is increasing criticism against this waste management model as it destroys local businesses and cleaning/hygienic conditions are disregarded.
Incineration
Incineration with/without energy recovery is the dominant textile waste treatment method. According to Ryu et al., 35 the incineration of 1 tonne of household textile waste can recover 15,800 MJ of energy, generating 27 kg of ash. Incineration of waste containing textile material/fibers is normally carried out in a muffle or electric incinerator at temperatures ranging from 500°C to 800°C for a period of 4–5 hours. 36 However, the incineration of textiles is considered a controversial waste treatment method because of the irregular behavior of the ignition rate and the temperature profile produced by blended textile waste. 35 In addition, textile waste emits toxic greenhouse gases into the air during incineration and accumulates ash. 13 According to Youhanan, 37 burning 1 tonne of textiles releases 785 kg of carbon dioxide into the air considering only the combusted cotton and polyester fraction. Textile waste, particularly for fibers with higher nitrogen content, such as polyamides and acrylics, emits nitrous oxide gas into the air.
Textile recycling
Recycling of textile materials has been practiced in open and closed loops. However, factors such as high prices, volume, and availability of raw materials have limited the ability to establish economically viable operations. 38 Similarly, open-loop recycling has been proven possible in the fashion industry. Open-loop recycling is a recycling process where textile waste can be channeled into different products/applications, whereas in closed-loop recycling, the waste products are recycled to produce the same type of product as the original one. In most textile waste, varieties of fiber types and colors result in shoddy fibers that are unattractive and unsuitable for respinning into textile quality yarn (closed-loop recycling). Hence, recycled textiles are only suitable as a raw material for products of lesser value, such as carpet underlay or insulation lining. More recently, research has focusing on using post-consumer waste to produce higher-value products, such as regenerated fibers,14,39 composites,40 –42 microcrystalline cellulose (MCC),43,44 and raw materials for synthetic fiber regeneration. 45 Waste management through recycling is hindered by fabric chemical treatments, methods of fiber identification, and the different types of fiber blends. As will be discussed in the following sections, these are critical research gaps that need to be addressed.
Effects of textile blends on recycling
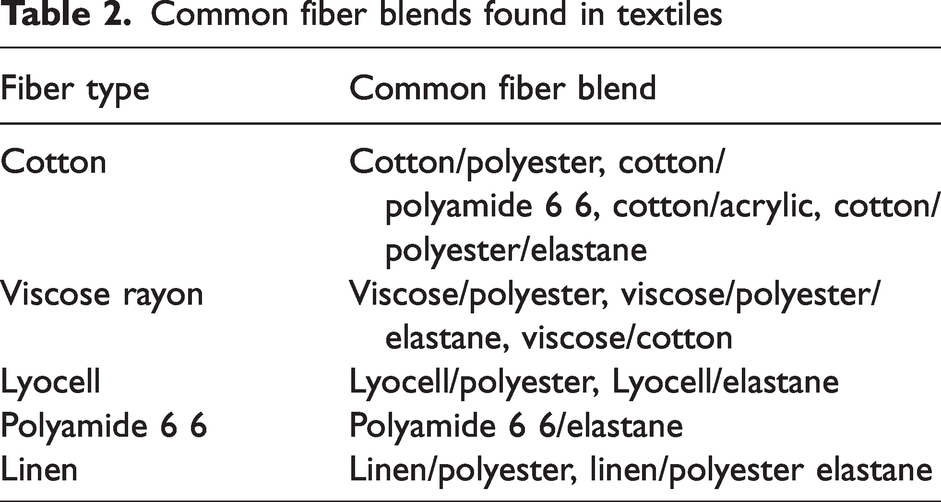
For the fabric to meet desirable requirements for clothing application, natural fibers are commonly blended with regenerated or synthetic fibers. Common fibers that are blended with natural or regenerated fibers are listed in Table 2.
Common fiber blends found in textiles
Although blending is an advantage in the textile production industry, fiber blends in the textile waste make recycling complex and challenging, as methods for separating and sorting waste into constituent fibers are required. This has resulted in most textile waste being landfilled. Most end-of-life textiles often consist of multi-material fiber compositions, which poses a complexity when attempting to recycle. Different fibers require sorting and separation, which primarily results in fiber degradation. Elastane (EL) is one common fiber that is found in small quantities in most blended textiles. EL is mainly used to impart elastic properties to clothing. Although the production volume of EL is not very high, this fiber is contained in many textiles. Some 2–10% of this fiber is commonly blended with other fibers to give an elastic effect on the fabric. Many recycling processes are made difficult or impossible by the presence of EL. Removing the fiber is a challenge since it is a PU and polycondensation polymer that cannot be removed by any solvolysis methods, making such complicated blended waste used as feedstock for energy recovery, although this is inefficient resource handling.
Effects of fabric chemical treatments on recycling
Textile production processes use a massive array of chemicals and water to assist the processes employed to achieve the specific desired textile properties. Dyeing, printing, and finishing treatments of fabrics are the major processes that impart chemicals that remain on fabrics throughout their use. Dyeing and printing are processes of imparting color and prints onto fabrics from dyes and pigments. Functional properties such as antimicrobial finishes, anti-wrinkle finishes, fabric softeners, and flame retardants can be applied on textile fabrics using chemical finishes to impart a certain property on the material. Although fabric chemical treatments impart dyes or functional properties, some chemicals may remain in textile products at the end of their useful life, even though they have undergone several launderings.46,47 Information concerning the chemicals used or traceability of remaining content in textile products from usage to end-of-life is still lacking in post-consumer textiles. Further concerns on chemical recycling processes are fabric chemical contents reacting with process chemicals. Researchers have reported technical problems such as low solubility and decreased dyeability. 48 In such cases, there may be a need to add process steps for purification. 47
Most finishing chemical substances are washed off during the use of the textile before they become waste. However, with fast-trending clothing styles, most textiles reach the end-of-life after a few washing cycles. Hence, some of the chemicals could still be on the material, with limited knowledge of the effects of certain chemicals in chemical recycling processes. Moreover, new technologies to improve the wash fastness of dyes and finishes are being employed, posing more difficulty in removing them.
Mechanical recycling has little effect on the molecular structure of textile fibers; hence, substances in textile wastes are carried on to the outgoing material produced. However, chemicals affect chemical recycling; therefore, pre-treatment processes are required. Dyes and other contaminants have been reported to coagulate or form insoluble particles, which give rise to problems during respinning processes.
49
Björquist
50
discovered that cotton fabric treated with an easy care finish resulted in low solubility in solvent
Waste identification and sorting
The primary task of waste recycling is to accurately recognize and sort items according to their material content. Identifying textile materials is the most challenging task in the textile recycling process due to the complex structure of textile fabrics and the presence of garment accessories, such as zippers, buttons, or other decorations. 6 Currently, textiles are sorted and identified manually, hindering the processing of vast amounts of material and the possibility of complete automation. Microscopy has been reported as an identification tool for textile materials. 54 However, the morphological differences among the same species are not always sufficiently distinctive, for instance, identifying cotton from viscose.
Furthermore, microscopy cannot accurately distinguish between synthetic fibers since they are very similar in appearance, and the increase in the number of varieties makes it more difficult to distinguish the fibers. In addition, the method requires intensive sample preparation and is too slow for the automated recognition and sorting of textile materials needed for recycling. 55 Scaling up textile recycling and automatic online characterization techniques are needed. Recently, near-infrared (NIR) technology has been used to identify textiles. Zhou et al. 56 demonstrated a textile fiber identification model with the prediction accuracy of cotton fiber higher than 90% and 100% for polyester, polyamide, acrylic, silk, and wool fibers. Mäkelä et al. 57 reported 100% actual favorable rates of using NIR imaging spectroscopy to classify cotton, viscose, and Lyocell fibers. However, NIR has been reported to have drawbacks when detecting textile waste with core yarns, decorative threads, and coated fabrics. 58 On the other hand, particle size, brightness, color, moisture content, and coating chemicals significantly impact the NIR signals.6,59
Fourier transfer infrared spectroscopy (FTIR) has been applied to identify textile fibers in light of this drawback. Peets et al. 60 identified wool, silk, and polyamide 6 6 fibers from different sources using two instrumental approaches: reflectance Fourier transform infrared spectroscopy (r-FTIR) and attenuated total reflectance Fourier transform infrared spectroscopy (ATR-FTIR). They concluded that r-FTIR was more successful in differentiating among polyamide fibers. Similarly, Riba et al. 61 successfully identified seven different fibers with 100% accuracy using the ATR-FTIR spectrum of the textile samples, which were processed using several algorithms. Several search studies have demonstrated the use of the thermal behavior of textile fibers in trying to identify them.26,62,63 However, the destructive nature and the time taken to process samples make it inefficient to identify fibers before recycling.
Textile recycling technologies
Several technologies have been demonstrated in textile recycling, but only a few full-scale post-consumer recycling systems exist within the value chain. This is primarily due to the infrastructural barriers and economic feasibility of scaling up processes. Current recycling textile technologies can be divided into mechanical and chemical processes. New technologies are also venturing into biological recycling processes. For recycling technologies to be efficient, the identification and separation of constituent materials, fiber blends, and textile chemical treatments must be employed. In addition, future recycling technologies must be less expensive, more energy efficient, and less polluting than conventional processes for producing virgin fibers. Various textile recycling technologies include mechanical, thermomechanical, chemical, and biochemical recycling.
Mechanical recycling
Mechanical recycling is the processing of textile waste into secondary raw material without significantly changing the polymer chemical structure of the fibers. Mechanical recycling represents a partially variable combination of principal processing steps, such as collection, identification, sorting, grinding, carding, blending, and respinning into new fibers. Mechanical recycling can regenerate textile wastes into usable products without significantly changing the material's chemical structure. 64 Recycling post-industrial waste, such as cutting fabric scraps, produces the most significant volume of mechanically recycled textiles. Waste from post-consumer sources is more challenging to sort due to various color shades and fabric blends, resulting in a more labor-intensive process.
Waste from natural fibers is commonly shredded to produce non-woven sheets through heat pressing or needle punching for applications such as noise and thermal insulation, composite reinforcing materials, and agricultural geotextiles.65,66 On the other hand, synthetic fabrics such as polyester or polyamide 6 6 are converted into flakes or pallets by shredding and melting them, followed by respinning into fibers through extrusion. 65 Mechanical recycling has existed for decades. However, the products obtained are of lower value due to the harsh mechanical tearing process, which shortens the fiber length. 67 This method is best used for homogeneous component fabrics. However, when used on blended waste, the process seems to cause more damage to natural fibers than to synthetic ones; hence, the technology is unsuitable for blended fabrics. 68
The main advantage of mechanical recycling is that there is minimum need for chemical processing such as dyeing, as fibers are sorted and recycled with color from their previous dyes, making the entire process waterless. However, mechanical recycling requires high-purity feedstock. The main drawback of mechanical recycling is the inter-fiber friction, which gradually decreases the quality (shorter length) of recycled fibers, reducing their performance during yarn and fabric production. In addition, the short fiber content limits the type of yarn spinning method employed for rotor and friction spinning. 67 According to Khan et al., 69 the blend ratio significantly affects rotor spun yarns. However, a higher percentage of recycled waste can be achieved using a higher cylinder speed during spinning. Aronsson and Persson 70 investigated the effect of used garment conditions on the quality of recycled cotton fibers from waste denim and single jersey fabric. They argued that the yarn and fabric construction affect the degree of fiber quality to a greater extent. Knitted fabrics gave recycling efficiencies higher than 75% because of the loosely spun fibers used. However, woven fabric is more compact, with dense yarns, resulting in the tearing of most fibers during the shredding process and poor recycling efficiency.
Consequently, the mechanically recycled fibers collected are usually mixed with virgin fibers with higher fiber lengths to counter this loss. These virgin fibers can be the same as the recycled fiber or blended with a different fiber type. However, fabrics that include mechanically recycled material can only use 20–30% of recycled fibers before the fabric quality is significantly reduced. 70 Lubricants have also been shown to reduce fiber length after shredding by decreasing friction during the process; hence, modern recycling methods can now break down fabrics with improved quality. 71 Lindström et al. 67 used polyethylene glycol 4000 aqueous solution as a lubricant to treat fibers, resulting in reduced fiber cohesion. Zheng et al. 72 investigated the application of soybean oil and 10% liquid detergent with water. The results showed that the tensile strength of fine and pristine cotton yarns with soybean oil treatment decreased by 44.62% and 39.78%, respectively.
Recycling of textile substrates composed of natural fibers
The mechanical recycling of natural cellulosic and protein fibers is well-established and is applied to various waste streams within the value chain. Fibers mechanically recovered from recycled cotton and wool fabrics could be respun into new fibers using the conventional ring or rotor spinning process or developed into non-wovens, which can be incorporated into composites. 73 Cotton and wool fabrics are mechanically recycled, following the illustration in Figure 3.
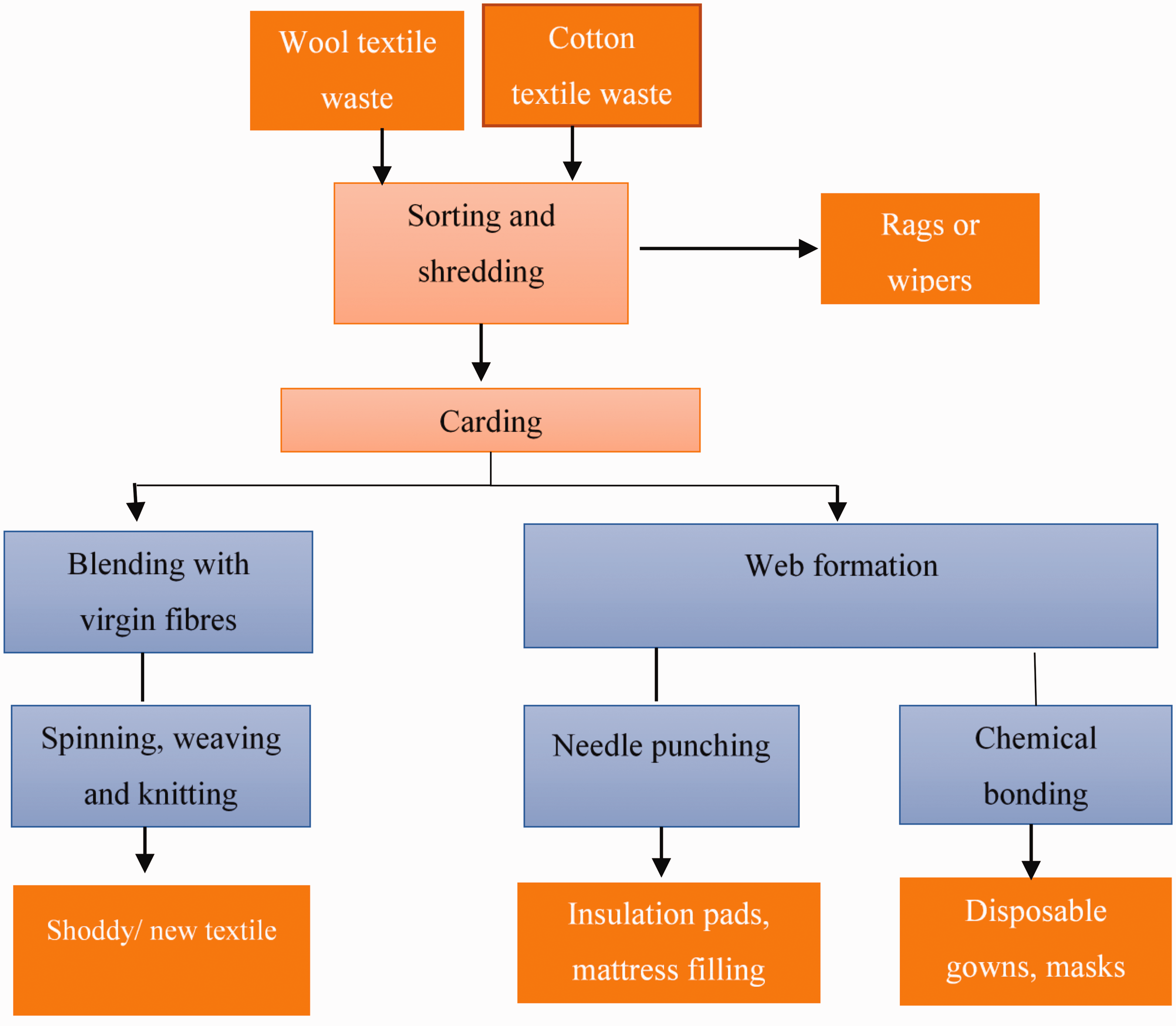
Mechanical recycling technologies for natural origin fiber.
Various companies have mechanically recycled cotton waste that can produce yarns with a higher recycled fiber ratio. In most cases, the recycled fibers are mixed with virgin fibers to improve the strength properties of the yarns. For example, Hilaturas Ferre (Spain) employs a new mechanical recycling technology that recycles post-industrial and post-consumer cotton (100%) waste. 74 The technology maintains the staple length of the fiber at 15 mm as opposed to below 10 mm, which is the distinctive maximum length of mechanical waste processing. 74 The length of virgin cotton is typically 20–25 mm. Approximately 50% recycled cotton blend yarn can be processed using their technology. Similarly, Geotex™ also recycles 100% post-consumer fabrics to produce yarn with 75–90% recycled cotton blend yarn blended with polyester fibers, and the ratio varies depending on the required application.
The production of non-woven fabrics from recycled cotton waste has been reported.75 –77 According to Islam and Bhat, 78 non-woven production results in new materials that are less costly and contribute to the reduction of the environmental impact as they require fewer production steps compared to yarn and fabric production. Non-woven fabrics from recycled cotton fibers have been used for oil spill clean-up, 79 cotton mulching films, 80 thermal insulation, and as an acoustic absorber. 81 Some composites of denim fabric waste have been manufactured using epoxy resin as a matrix, and their properties were suited to different applications, such as flooring, plywood, and partition boards. 41
Wool is considered one of the most recycled fibers, with a recycling practice dating back over 200 years. 82 Wool fibers are relatively long, and their length remains acceptable after mechanical recycling. Hence, they can be used in the production of high-value textiles. The recycling process is similar to that for cotton fabrics. However, blending with a proportion of virgin fiber is inevitable when mechanically recycling textile fibers. The respun yarn is generally blended with a proportion of virgin wool or synthetic fiber to aid processing efficiency and meet the final product's required performance specifications.
Alternatively, the recycling of wool fiber side-steps the traditional spinning, weaving, and knitting processes. It is used to make a variety of non-woven textiles that are used to produce insulator pads for automotive felts, furniture mattress stuffing, or geotextiles.83 –86 Wazna et al. 86 explored non-woven non-acoustic properties, consisting of 100% wool waste fibers thermally bonded with polyester bi-component fibers. The density, absorption coefficients, and thermal conductivity results were comparative, meaning they could be a valid alternative for building applications.
Recycling of textile substrates composed of polyamide 6 6 fibers
Mechanical recycling of polyamide 6 6 is less common compared to other textile fibers. Waste from synthetic textiles is recycled using thermomechanical recycling technology. The process involves sorting, melting, and remolding, usually done by adding some virgin material for better mechanical properties. 87 According to Mondragon et al., 88 thermomechanical recycling for polyamide 6 6 products, such as fishing nets, has similar main properties to virgin polyamide 6 6. While polyamide 6 6 recycling is still in the early stages of development, several companies are involved in pre- and post-consumer recycling of polyamide 6 6 fabrics. One company, Patagonia, uses recycled polyamide 6 6 made from post-industrial waste polyamide 6 6 fabrics and discards from weaving mills and post-consumer fishing nets to make clothing and gear that contains recycled polyamide 6 6 yarn. ECOALF, a Spanish company, collects disposed fishing nets from the ocean and mechanically recycles them into yarn used in ECOALF swimwear, which is 100% recycled polyamide 6 6. 89 Although polyamide 6 6 recycling prevents the petrochemical waste from landfilling or incineration that can release toxic emissions into the atmosphere, the polymer melts at low temperatures; hence, it is susceptible to contamination from microbes or bacteria pollutants, which can remain in the substance after melting. Hence, it makes the complete cleaning of the material before its melting an important process, although this makes the process longer and expensive.
Recycling substrates composed of polyester (polyethylene terephthalate) fibers
Polyester is a synthetic or man-made fiber that contains the ester functional group in every repeat unit of the main chain. It is commonly shortened from its technical name, PET, which is made by mixing ethylene glycol (EG) and terephthalic acid (TPA). PET is used as fibers for clothing or molded into containers for liquids and foods. Mechanical recycling of polyester has been practiced where PET flakes obtained from PET bottles are utilized to produce staple polyester fiber and used as raw material in the textile industry. However, the industry is lacking a way to deal with the waste generated in the textile value chain, that is, the 'fiber-to-fiber' process. Degradation of the molecular weight during extrusion and injection molding is one of the main problems in PET recycling. Compared to virgin PET fibers, post-consumer PET fabrics show more degradation from alkaline detergents that can cause flake segregation, resulting in poor mechanical properties. 90 However, cross-linking is done during extrusion to improve the mechanical properties. Sanches et al. 91 produced new polyester knitted fabrics using polyester fabric scraps discarded by a clothing company. From the study it was evident that there is feasibility in the production of new PET fabrics from mechanically recycled PET fabrics.
The drawbacks of recycled polyester include the difference in the glass transition property, which leads to uneven dyeing behavior. Remelting results in the reorganization of the polymer structure; hence, the crystallinity of recycled polyester was reported to be lower than that of virgin polyester by 13.85%.92 –94 Fibers with lower molecular weight will show lower breaking stress and elongation, and a higher Young's modulus. The alignment of molecules after each extrusion process leads to less shrinkage. To cater for this, various authors have studied ways to improve polymer degradation during recycling, including blending with virgin PET polymers 80 ; using chain extenders that reverse some of the damage caused by polymer chain degradation; and blending with other virgin fibers or clays. Lee et al. 95 investigated the properties of recycled PET fibers after different blend ratios with virgin PET. The results showed that thermal degradation and melting temperatures increased, and the crystallization temperature and crystallization rate decreased, showing that the blended fibers had better thermal stability but lower crystallization, resulting in comparable mechanical properties to virgin PET fibers.
Recycling substrates composed of acrylic fiber
Acrylic fiber closely resembles the look and feel of wool fiber and is commonly used in place of wool or blended with other fibers to make sweaters and other knitted products. Discarded acrylic fabrics cannot be used for energy recovery, and incineration of this fiber should only be carried out with special equipment as it emits highly toxic gases such as ammonia and hydrocyanic acid. Unlike polyester or polyamide fibers, PAN fiber is not a thermoplastic polymer and does not melt when heated. 64 At temperatures above 180°C, the polymerization of the PAN polymer chain begins, making it rigid and dark due to the high carbon content. Due to the cyclization of the chain with the formation of solid waste of high carbon content in the reactor, fabrics containing acrylic fibers cannot be depolymerized by thermal processes. 49 On the other hand, polyacrylic fabrics are blended with other types of fibers, such as cotton, wool, polyester, or polyamide. Mechanical recycling to separate the various fibers of the fabric is very difficult and expensive due to the small diameter of the filaments, a slight difference in density, and the interweaving between them. A few companies have successfully recycled 100% acrylic textile waste into brands, such as Acrycyle and Regel fibers. These recycled brands are mixed with virgin acrylic fiber to increase their strength. Poggio and Faranda 96 reported the successful recycling of acrylic fabric waste by developing a 100% recycled yarn using conventional open-end and ring spinning. The tenacity of the yarns reached the highest values of 12.7 and 27.88 cN/tex, respectively, with an 89% recycling efficiency. They disputed that virgin-identical qualities could be achieved; however, to guarantee sufficient quality, it is necessary to add virgin fibers in a mixture with recycled fibers. The recycled yarn is also prone to an increase in coarse stitches and neps due to the residue of unopened yarn during the mechanical opening and shredding of the waste fabrics. Similarly, Gun and Kuyucak 97 investigated the mechanical properties of plain knitted fabrics made from recycled rotor spun and virgin acrylic yarns. Results show that acrylic fiber waste can produce plain knitted fabrics of acceptable quality as a cheap alternative to virgin acrylic fiber. Adding an elastic yarn can improve the quality and performance properties of the fabric.
Recycling blended fabrics
Blends of cotton and polyester fabrics predominate textile waste streams. Recycled material from blended fabrics seldom finds application in the apparel industry, but has been applied in manufacturing non-wovens for the automotive and construction industries. Sharma and Goel 75 developed and characterized non-woven fabrics developed from waste cotton/polyester fabrics using the needle punching method. They concluded that the characterized non-woven fabrics had good properties for various applications. Non-woven fabrics made from recycled fibers are more environmentally friendly than conventionally used PU foams, which cannot be recycled and are produced with environmentally damaging manufacturing methods. 98 Santhanam et al. 98 used recycled cotton/polyester fiber blends to make reclaimed fiber web with spun-laid techniques to produce non-woven fabrics for sound insulation. The results showed that the porous recycled non-wovens perform excellently, absorbing high-frequency sound waves above 2000 Hz. Recycled blended fabrics have also gained interest in thermal insulation applications.81,99,100 Sakthivel et al. 42 developed chemically bonded non-wovens from mechanically recycled cotton/polyester selvedge waste for automotive interior applications for thermal insulation purposes, and found that 50/50 cotton/polyester non-wovens showed better thermal insulation performance. Similarly, Sakthivel et al. 81 characterized the insulation and acoustic properties of recycled PET/cotton non-woven mats and reported that more than 70% good resistance was achieved. Other reports found in the literature are listed in Table 3.
Summary of recent mechanical recycling of various fabrics wastes
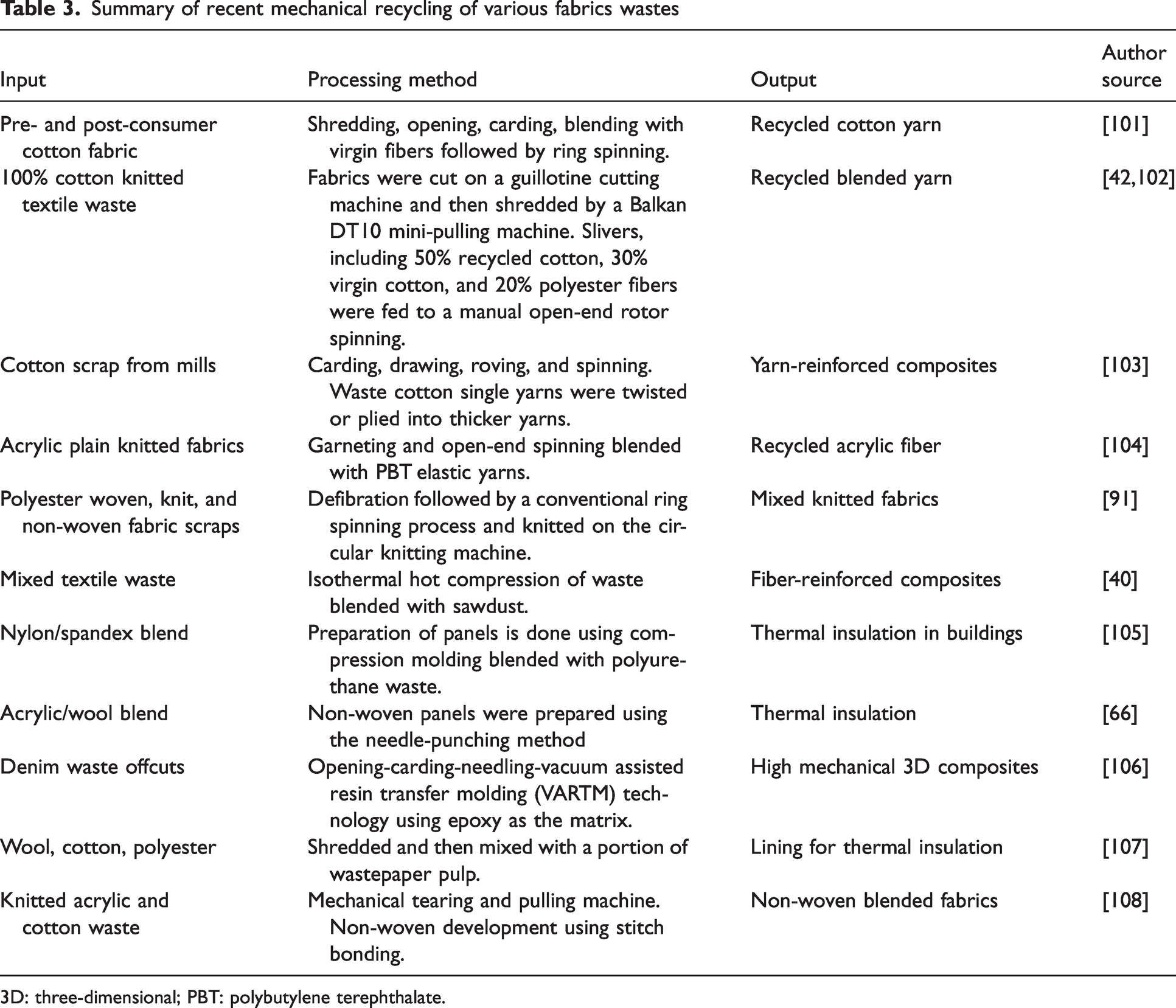
3D: three-dimensional; PBT: polybutylene terephthalate.
Chemical recycling
Chemical recycling of textile waste provides a promising alternative as it can overcome some of the shortcomings of mechanical recycling. Chemical recycling is the process of converting polymeric waste by changing its chemical structure and turning it back into monomers that can be used as raw materials for the manufacturing of new polymers. Chemical recycling allows the degradation of fibers into monomers or building blocks, then re-polymerizes them into new fibers of comparative or superior quality. However, chemical recycling risks keeping or spreading hazardous substances found in dyes, softeners, anti-wrinkle agents, and other common additives. Chemicals present in textile waste have the potential to obstruct recycling processes; however, the knowledge of specific impacts and to what extent they affect processes is limited.
Color stripping is one of the most convenient ways to rectify the various shade faults that occur during the printing or dyeing process of textiles and is widely employed as a decolorization technique in the textile industry. However, these processes have not been employed on a large scale for recycling. Technologies for color removal from textile wastes include dye destruction or extraction.52,53 Dye destruction methods, such as oxidation and photodegradation, may damage polymers and change the dyeability of the regenerated textiles. 109 Dye extraction causes minor damage to fibers but it requires finding solvents with high dye solubility and, usually, dye extraction removes the partial dye from the textiles. 109 Stripping of reactive dyes with 89–94% has been achieved using a combined reductive treatment of sodium hydroxide and sodium hydrosulfite at elevated temperatures of 80°C and 100°C. 109 Määttänen et al. 52 reported that vat, reactive, and direct dyes could be stripped and removed from textiles by combining different chemical treatments, such as alkaline, acid, hydrogen peroxide, ozone, and dithionite. However, processing caused damage to the fabric, including but not limited to losses in strength, weight, and pilling resistance ratings. Researchers have looked at ways to reduce fiber damage during dye stripping. Mu and Yang 110 demonstrated reducing fiber density by solvents and temperature control, completely removing dispersed dyes, acid dyes, and direct dyes from PET, polyamide 6 6, and cotton fibers, respectively. Various research on dye removal has been done with recent work that carries out the decolorization process of textile fabrics at a pilot scale using an ozone-assisted process. 111 Powar et al. 111 decolorized 90% and above pigment-printed cellulosic textiles using the ozone-assisted process in an acidic solution. He et al. 112 concluded that ultraviolet (UV)/Na2S2O4 with a photocatalytic system could effectively strip fixed reactive dyes, with a relatively high tensile strength retention observed from cotton fibers. In addition to effective dye stripping, sequential alkali–acid stripping of reactive dyes from cotton waste has been reported to remove cross-linked textile finishes. 113
Recycling substrates composed of cellulosic fibers
Chemical recycling of cotton fabrics into regenerated fibers is a promising solution. However, cotton fibers are difficult to dissolve in common solvents due to the strong intra- and inter-molecular hydrogen bonding in cellulose macromolecules. 39 The chemical recycling process of cotton, which has been explored, is based on cellulose dissolution, either polymer dissolution in solvents 49 or depolymerization of glucose monomers. In dissolution by solvents, the process may recover a chemically modified or pure cellulosic fiber product, which can also be incorporated as feedstock for regenerated/recycled man-made cellulosic fibers. This process can be achieved via different systems such as the Lyocell process, 51 alkali/urea, 39 or ionic liquid process. 15 In recent years, the Lyocell process has become a simple physical alternative to the dominating viscose technology or regeneration of cellulosic fibers without producing hazardous by-products (CS2, H2S, heavy metals). The Lyocell and ionic liquid processes are the dominant technologies researched for chemically recovering cellulosic fibers. 49 The overview of the chemical recycling of cotton is illustrated in Figure 4.
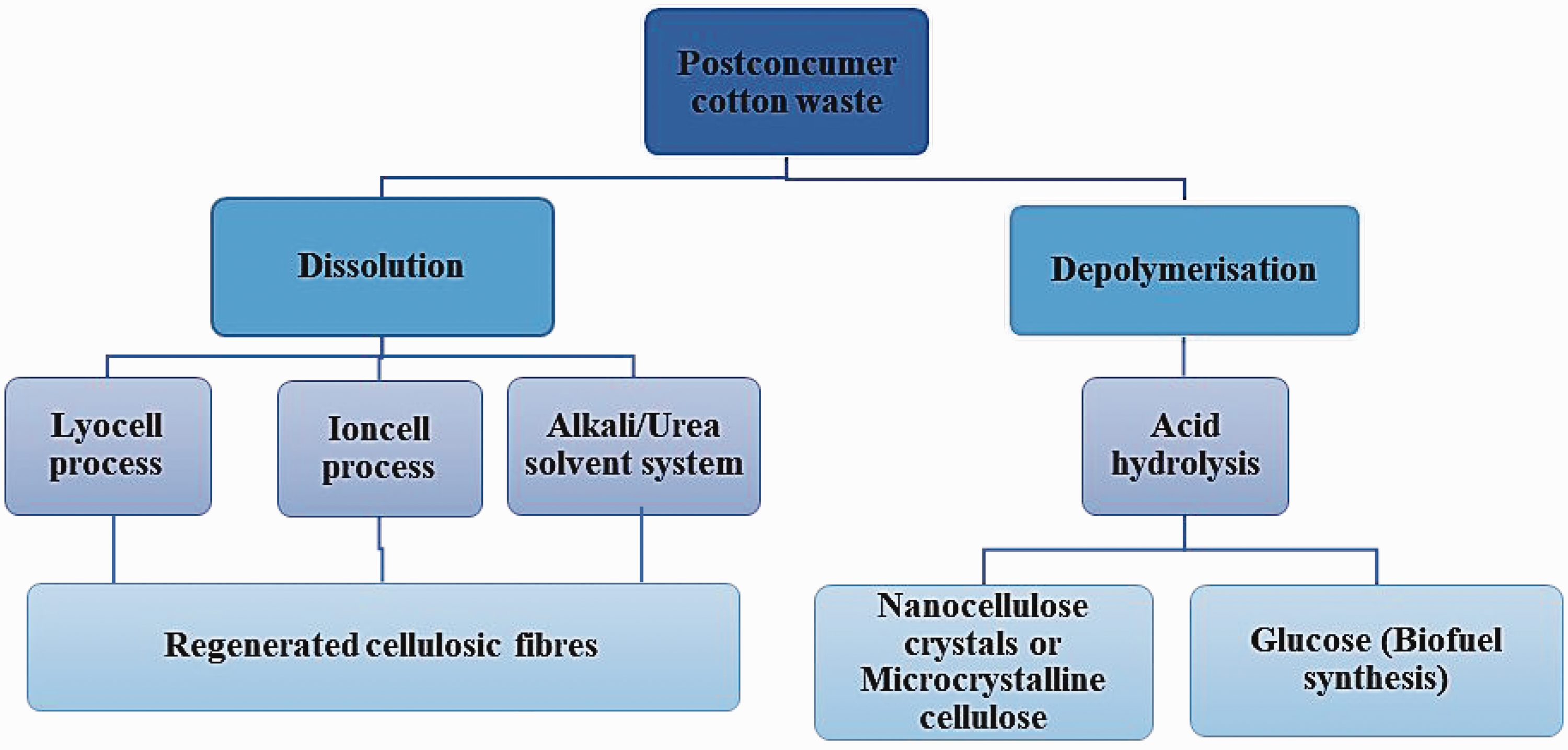
Chemical recycling technologies of cotton. 46
Various researchers have reported using NMMO to regenerate cellulosic fibers from cotton waste.16,114,115 However, this process has shown challenges in high thermal instability and energy consumption during spinning. Furthermore, regenerated Lyocell fibers fibrillate under wet abrasion, giving an undesirable hairy appearance that can make the dyeing of Lyocell fibers more difficult. 16 This Lyocell process avoids the need for toxic chemicals and the generation of harmful by-products, which ultimately implies a greener process for recycling materials. However, the process is associated with high costs in developing a highly efficient recycling system. To reduce the cost of this process, the solvent can be recycled up to 99%, which lowers the cost of the raw material used. As an alternative to NMMO technology, the NaOH/urea solvent system shows noticeable results regarding similar fiber properties but with lower production costs. 116
Alkali/urea aqueous has been utilized as an environmentally friendly alternative for cellulose dissolution systems. Liu et al.
39
compared two solvent systems, LiOH/urea and NaOH/urea, for their efficacy in dissolving cotton waste and regenerating cellulose fibers. Results showed that the regenerated fibers had tensile properties comparable to current commercial regular rayon fibers made from wood pulp. However, the complexity of dissolving cellulose pulp in this NaOH/urea solvent system at cryogenic temperatures prevents the commercialization of this wet-spinning technique.
116
Another common solvent used in recycling cotton fabric waste is a system of lithium chloride/
Recycling cotton waste fabrics has shifted to more environmentally friendly ionic liquid solvents. 49 Ionic liquids give a more energy-optimized process as they allow the complete pulp dissolution at lower temperatures and less time than the Lyocell process. De Silva and Byrne 117 and Asaadi et al. 118 both used the cellulose-dissolving ionic liquid 1-allyl-3-methylimidazolium chloride (AMIMCI) as a solvent to recycle cotton waste and produced regenerated fibers with outstanding fiber properties. Sixta et al. 15 reported the development of Ioncell-F, which is a novel cellulose spinning process that uses non-toxic super-base ionic liquid, 1,5-diaza-bicyclo [4.3.0] non-5-enium acetate ([DBNH][OAc]). Haslinger et al. 14 investigated this solvent's use in recycling cotton waste and concluded that cotton-dyed fabric waste could be recycled into colored man-made cellulosic fibers using DBNH][OAc]. However, with ionic liquids it is necessary to verify the viscosity of the waste cotton before dissolution to spin the resulting dope into fibers. Natural cotton has a higher degree of polymerization; however, this tends to reduce during garment use. Hence, to spin regenerated fibers, the dissolved cotton dope is usually blended with high-viscosity material or chemically treated to decrease the viscosity to levels that are fit for regenerated fibers. 49
Post-consumer waste has also been utilized to extract MCC, which has applications in various compositional reinforcements. 119 MCC has been prepared from waste cotton fabrics using dilute inorganic acids.43,120 However, inorganic acids are corrosive and result in environmental issues. Many researchers have been developing various methods to prepare MCCs to address the mentioned problems. Hou et al. 44 developed a novel and green process to prepare MCC from waste cotton by the catalytic hydrolysis of phosphotungstic acid. Ruiz-Caldas et al. 121 isolated MCCs from cotton, polyester/cotton, and acrylic/cotton waste fabrics through acid hydrolysis with sulfuric acid. A yield of 51 − 62 wt.% was achieved from the cotton fractions.
Cotton waste from the textile industry has been a viable feedstock for cellulosic ethanol production. Cotton waste can be pre-treated and depolymerized by acid hydrolysis to convert cellulose to reducing sugars. 6 Sanchis-Sebastiá et al. 122 evaluated the possibility of using acid hydrolysis to directly depolymerize the cotton fibers in waste textiles to produce a glucose solution of around 40 g/L with a yield over 90% by using a two-step procedure in which concentrated and dilute sulfuric acid were combined to exploit the benefits of both concentrations. Binczarski et al. 123 reported a model procedure for thermally assisted acidic hydrolysis of cotton into glucose and subsequent oxidation of the glucose into calcium gluconate over a Pd-Au/SiO2 catalyst. Acid hydrolysis into glucose seems a better solution than biological methods to the problem of textile waste that cannot be used to produce regenerated fibers. Table 2 shows the various recycling processes applied to cotton fabric waste.
Recycling substrates composed of wool fibers
Chemical recycling of wool waste is not practiced. However, the recovery of wool keratin for use in biomaterials, adhesives, and resins has been researched and demonstrated on a small scale. Chemical recycling of wool keratin to make regenerated protein fibers has not been demonstrated successfully; however, extracted keratin from wool fibers has been reported. Wool fiber comprises 82% keratin with a uniquely high proportion of cysteine in its structure (10–15%) compared to other keratin sources. 124 The cysteine in the protein chains is cross-linked via strong disulfide linkages into a distinct, tightly packed hierarchical structure, resulting in low wool keratin solubility. 125 The structure of wool keratin consists of the alpha helix form rather than the other keratin structure of beta sheets. Various methods of breaking keratin linkages have been reported, including reduction, 126 oxidation, sulfitolysis or oxidative sulfytolisis, 127 cuprammonium, 128 and ionic liquid reagents. 129 After breaking the linkages, keratin may be kept in solution to be blended with other materials and then spun directly. Wet spinning is widely used to regenerate keratin fibers blended with other polymers, and electrospinning has been reported to create ultrafine fibers for non-woven textiles. 130 However, the mechanical properties of electrospun nanofibers are often lower than those produced by other spinning techniques. The usage of toxic chemicals compromises the usage of such fibers in intended non-woven biomedical applications; hence, the obtained fiber is not adaptable for the wearable textiles industry.
Regenerated keratin is too brittle to form a fiber because of the β-structure, which consists of parallel sheets of polypeptide chains, unlike fibrous alpha keratin; therefore, it is blended with other polymers such as hydroxyethyl cellulose (HEC), polyvinyl alcohol (PVA), polybutylene succinate (PBS), or polyamide 6 to improve mechanical stability.128,131 Cross-linking between the keratin chains and other materials can improve the mechanical properties of fibers. 132 Cao et al. 132 reported an example where reductive extraction of keratin from wool was used and blended with hydroxyl propyl cellulose (H-HPC). The solution was cross-linked with glutaraldehyde followed by 4,4-methylenebis and then wet spun into filaments. Cross-linking with just glutaraldehyde and 4,40-methylenebis-(phenyl isocyanate) imparted a favorable tenacity to the fibers. 132 The chemical regeneration of keratin from wool is currently concentrated in non-textile industries and its application in textile industries have not been reported.
Recycling substrates composed of PET fiber
Chemical recycling of polyester through depolymerization has been extensively researched due to the large volume of waste from PET bottles.133,134 Chemical recycling methods allow PET recycling through depolymerization into monomers without degrading its quality. Three degradation methods of PET are hydrolysis, alcoholysis, and glycolysis. However, degradation of PET monomers, TPA, and EG can only be achieved through hydrolysis. 135 Chemical recycling of PET from waste fabrics is not practiced; however, a few researchers have reported about this technology. Li et al. 45 developed an effective method combining hydrolysis, reactive processing, and decoloration to convert colored PET fabrics into high-purity TPA. The disadvantage of PET chemical recycling is that the methods require high-pressure and temperature conditions and the employment of toxic degrading agents for depolymerization. Furthermore, these methods generally entail purification and separating steps for product recovery, thus posing environmentally hazardous issues. Mechanical recycling is less energy intensive and has a relatively low demand for resources compared with chemical recycling, making the overall cost for mechanical recycling low. Technical requirements for infrastructure are low for mechanical recycling, as the process has been fully developed compared to the new chemical methods, which are currently technically challenging and uneconomic. Mechanical recycling shreds plastic waste and does not use toxic chemicals, such as acids and solvents, to dissolve the plastics. For these reasons, mechanical recycling is preferred for PET. Figure 5 illustrates some of the recycling technologies for PET.
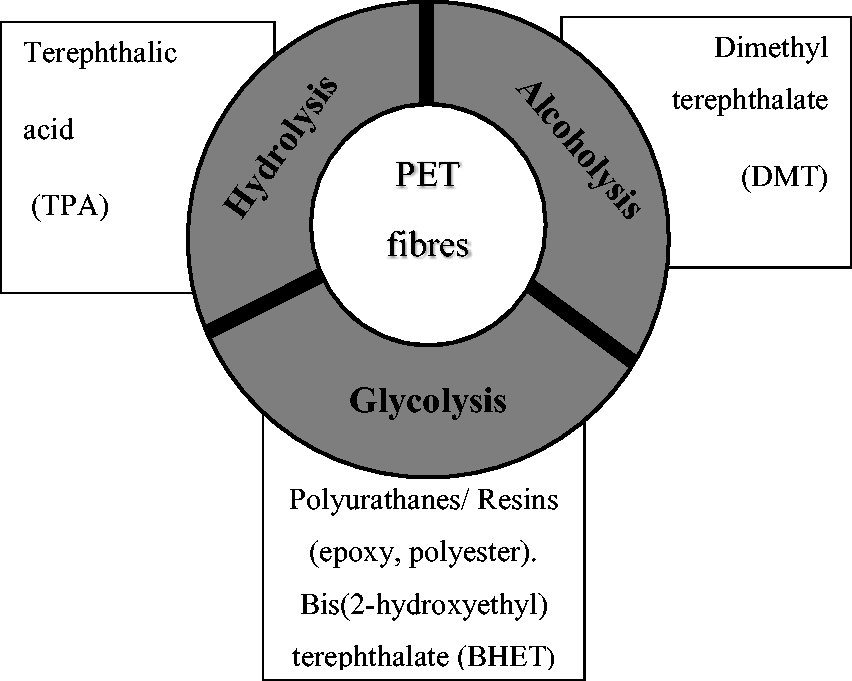
Different methods of polyethylene terephthalate (PET) chemical recycling and the value-added products derived.
Hydrolysis of waste PET is usually carried out under acid, neutral, and alkaline conditions. 136 Neutral and acidic hydrolysis of PET gave high yields of TPA. However, the TPA is usually of low purity and requires purification of the reaction product, which is solvent-consuming and imposes environmental issues. Alkaline hydrolysis is preferable and is conducted with excess metal hydroxide at temperatures between 70°C and 150°C. Although alkaline hydrolysis is preferred to neutral and acid reactions, reaction products also require separation and purification after a complete reaction. Sun et al. 137 explored a novel alcohol alkali hydrolysis method for preparing TPA from waste PET. Over 98% of PET can be depolymerized into the target TPA with the purity and yield reaching over 97% and 94%, respectively. Methanolysis for de-polymerization of PET was developed to limit the drawbacks of hydrolysis.
Although chemical recycling through depolymerization of PET to monomers enabled value-added re-usability of reaction products, such as the synthesis of virgin PET, the methods have a drawback due to high-pressure and high-temperature equipment. In addition, contamination from water, dyes, and heavy metals poses one of the greatest problems to recycling post-consumed PET.6,133 Moreover, it causes deterioration of physical and chemical properties during reprocessing. Consequently, large amounts of chemicals are consumed in the de-polymerization, successive separation, and purification stages. Thus, such chemical recycling methods impose toxic and ecological issues.
Chemical recycling of blended fabrics
Polymer recycling by melting and spinning into new textile fibers is less achievable due to contamination, leaving monomer recycling as the only option for blended textiles. Blended fabrics typically consist of hydrolysable synthetic fibers, such as polyacrylics, polyesters, and polyamides, and natural fibers, such as cotton and wool. Chemical recycling of waste textile fabrics is not well developed commercially due to the complexity of the constituents present in the material, which requires costly separation of the synthetic polymer from the natural fiber. Furthermore, various pre-processing steps must be done to remove chemicals and contaminants from the fabric waste, increasing costs. Consequently, pyrolysis to form liquid fuels is unfavorable due to the high oxygen content of biomass in natural fibers.
A few recycling technologies targeting blended textile waste have been patented and employed; however, they are only small-scale projects.
135
Robello
138
developed a method of recycling polyamide fibers from textile waste blends of polyamide 6 6 and EL, such as sportswear and lingerie. The process employs temperature control, allowing selective degradation and washing of the PU fibers, leaving the polyamide 6 6 fibers intact. The process follows a controlled washing in a polar solvent such as ethanol to separate the fibers. Both polyamide and PU fibers are recovered in this process. Walker
139
developed a recycling system used by a company, Worn Again, which focuses on separating the polyester (PET) from cotton by dissolving dyes and polyester in a solvent-based system containing 1,3-dimethyl-2-imidazolidinone (DMI). Another company, Trash-2-Cash, focuses on cellulose regeneration from cellulose obtained in polyester and cotton blends. The method uses an ionic liquid, [DBNH][OAc], which dissolves cotton and can be separated from the polyester.
14
Regenerated cellulosic fibers are spun directly from the extractant using dry-jet wet spinning, and the undissolved polyester is washed and reused as polyester raw material. Barla et al.
140
developed a method for recycling blended cotton/polyester waste fabrics to regenerate the cellulose portion and convert the polyester into TPA and EG. The technology focuses on using a subcritical water reactor as a medium to break down the textile waste at temperatures between 105°C and 190°C. Palme et al.
135
investigated the separation of cotton and PET from mixed textiles using a straightforward process of 5–15 wt.% NaOH in water and temperature between 70°C and 90°C for the hydrolysis of PET, resulting in TPA and EG and recovered cotton fibers. Gong et al.
141
developed a simple method to dissolve PU in
Summary of recent chemical recycling employed to blended textile fibers
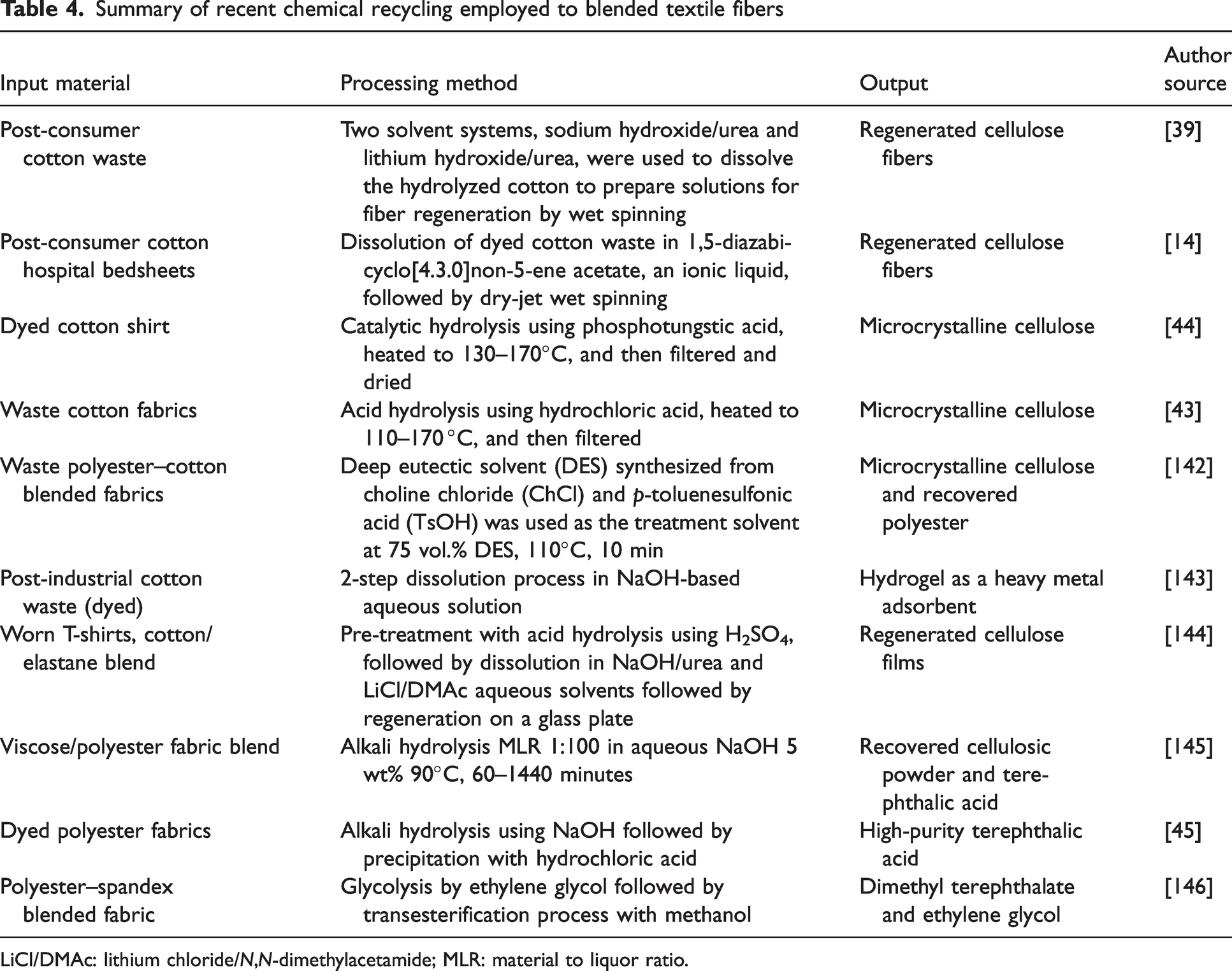
LiCl/DMAc: lithium chloride/
Biochemical recycling
Enzymes can be used to depolymerize end-of-life textiles into their monomeric building blocks, which can be used in the production of value-added products such as sugars (glucose) and bioethanol.147,148 The advantage of biological processes is that they typically have a low energy demand, use benign solvents and chemicals, and are based on renewables rather than fossil carbon. Furthermore, this process is highly selective, making them a good option for separating blended textile waste. 9 However, the technology can only be applied to natural polymers, is costly, and reaction times are much slower than chemical recycling.
Cellulase enzymes that can potentially hydrolyze cellulose-based textile materials to sugars have been developed using solid-state fermentation. Various value-added products derived from cellulosic waste using biochemical recycling are shown in Figure 6. Hu et al.
147
investigated the enzyme hydrolysis of cotton from cellulose enzymes produced by the
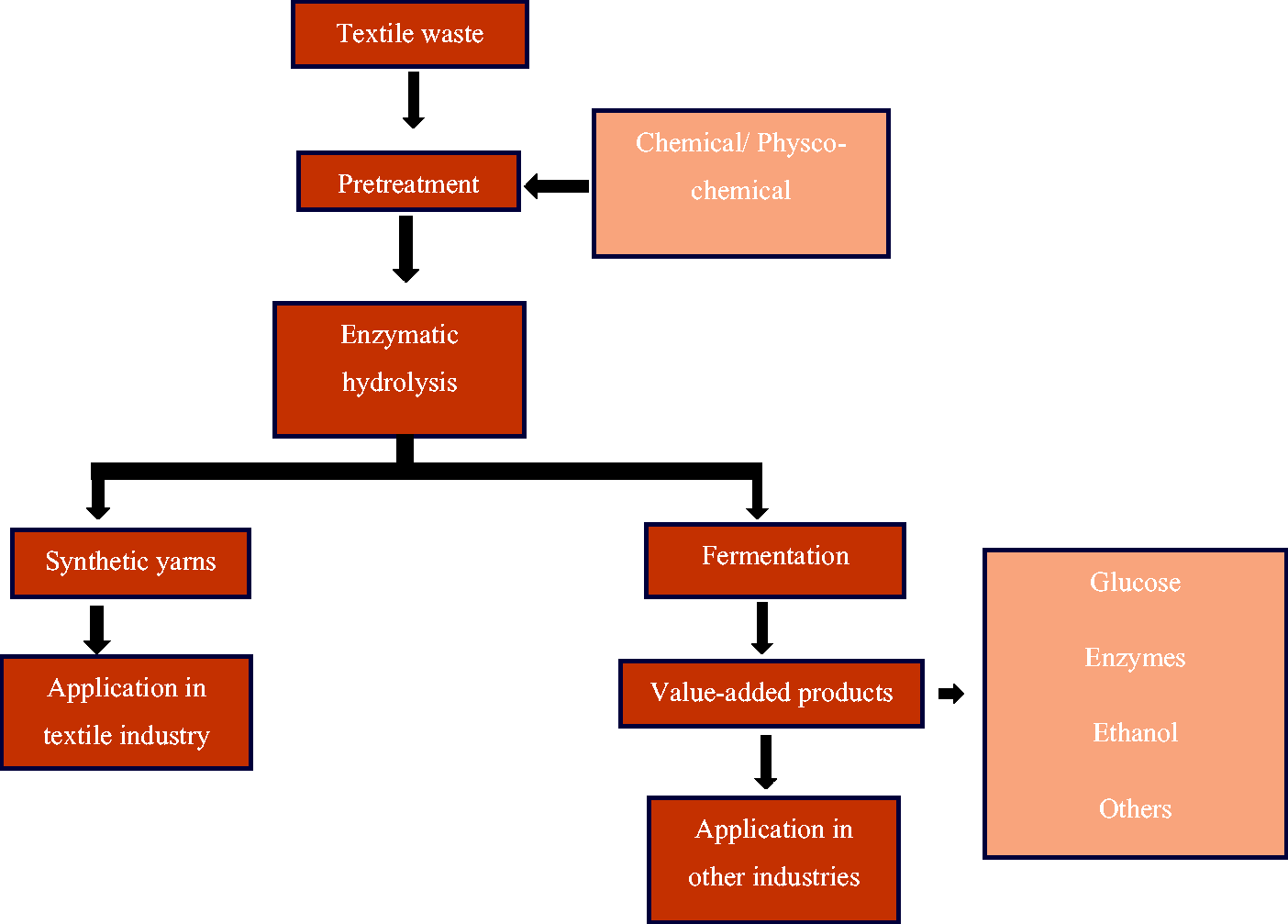
Biochemical recycling process and value-added products derived.
Biochemical separation of blended textiles has been employed with synthetic fibers. Quartinello et al. 153 investigated separating wool/cotton/polyester textile waste. The textile waste was sequentially incubated with protease for extracting amino acids from wool components and cellulases to recover glucose from cotton and rayon constituents with efficiencies of 95% and 85%, respectively. Engagement with engineering enzymes that could depolymerize PET used for plastic bottles has been seen in various research studies.154,155 However, the productivity of this process is yet to be improved and it has not been applied to textile fibers. Penetration of the large protein molecules is difficult; hence, it significantly limits the reaction speed. The resulting hydrolysis can only take place at the surface of the PET material. Potential enzymes that could hydrolyze polyamides156,157 have also been researched, but they have similar limitations to polyester enzymatic hydrolysis. 9
Studies have demonstrated that post-consumer textile wastes can be recycled into ethanol using enzymatic hydrolysis. 158 However, reports have demonstrated that the yield is insufficient; hence, pre-treatment stages are required before enzymatic hydrolysis can occur. In theory, biochemical recycling could remove textiles from the environment naturally. However, the current fiber mix consists mainly of synthetic materials and, to a lesser extent, natural polymers that are susceptible to biological degradation. Hence, the applicability of this technology remains limited. To date, commercial biochemical recycling of textile fabric is non-existent.
Challenges and the future of textile recycling
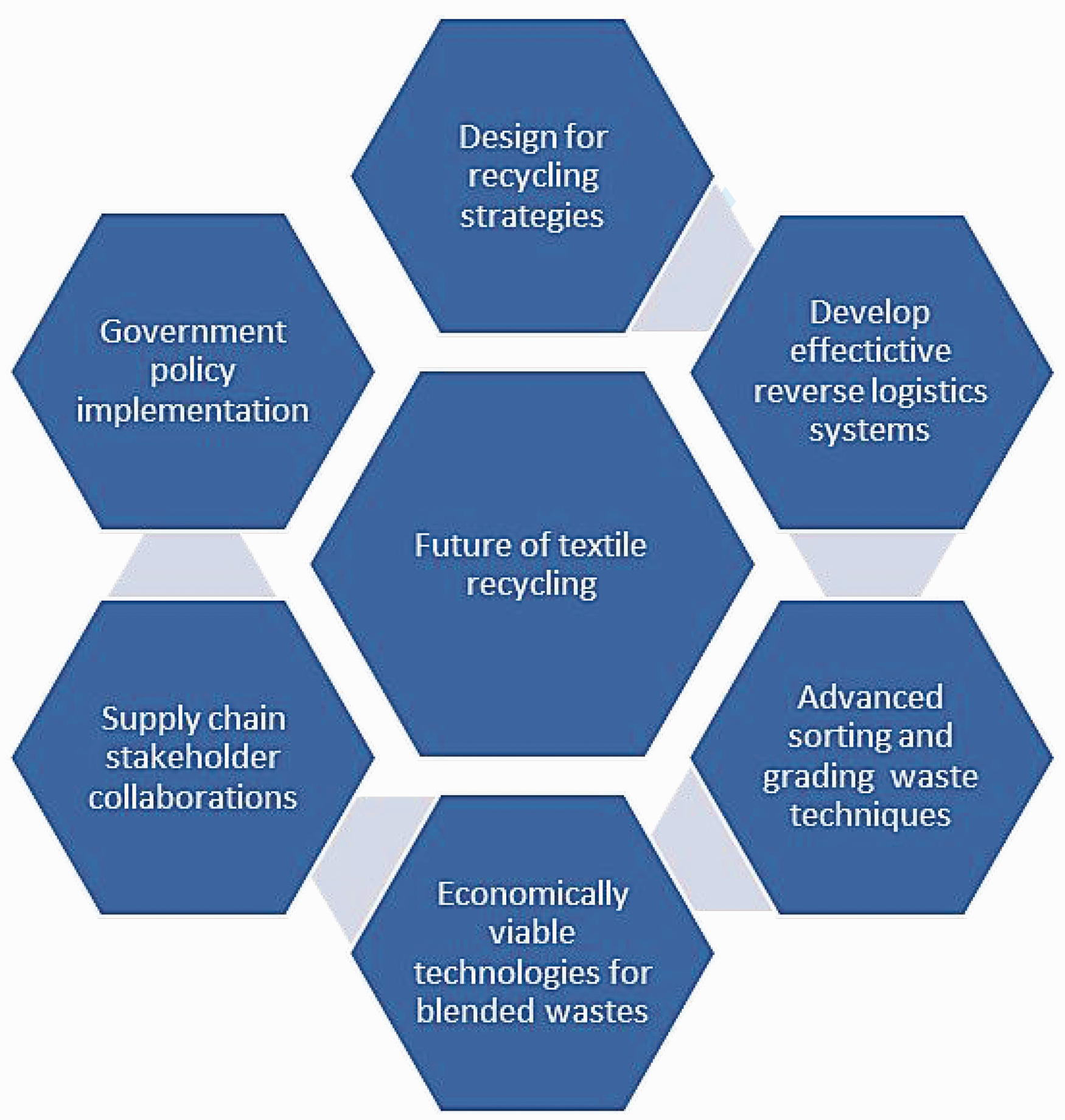
Recycling of textile waste materials has been demonstrated for quite a long time. Still, fiber-to-fiber closed-loop recycling remains a challenge. Open-loop recycling is currently the dominant technology that recycles waste fabrics to other industrial products rather than back to textile fibers. Whereas recirculation of raw materials is maintained in an open-loop recycling system, fabric-to-fiber recycling is becoming essential in moving toward a circular textile economy. However, the textile materials' complexity and the recycling systems' economic feasibility hinder the realization of a circular economy. The waste collection and an economically viable sorting infrastructure remain challenges. Chemical and biochemical recycling technologies are currently not economically viable, as most of the work is still on a small scale or under research. There is a need to create new opportunities for processes that operate under milder conditions and use less energy and less recycling time. Figure 7 shows possible routes for the future of textile recycling.
Future research directions for textile fabric recycling.
Fabric recycling is mainly based around consumer markets in developed countries, yet waste issues are more prevalent in developing countries. With the issue of the second-hand clothing market, Africa has become a dumpsite for most European and Asian countries where most of the production occurs, disconnecting the reverse supply chain from the forward supply chain. Most suppliers in the forward supply chain are reluctant about recycling. Only a few retailers are commercially collecting waste for recycling, for example, H&M. Collecting fabric waste is only handled by a few commercial waste collectors and is not regulated. It is essential to implement and operate effective take-back systems and redirect waste to appropriate sorting and recycling centers. Governments can also intervene with appropriate policy implementation to improve textile recycling, for instance, tax relief policies or rewarding technological innovations for recycling businesses. It should be mandatory for a certain number of textile products in government procurement to come from recycled materials as part of an action plan to boost the textile waste value chain. Governments could also introduce textile recycling clusters to ensure industry workers have access to the latest recycling technology. Textile industries should also participate in collaborations, research, and development of innovative recycling technologies. To ensure a successful circular economy, collaboration among manufacturers, recyclers, policymakers, and researchers is crucial for developing efficient collection systems, improving recycling technologies, and establishing a robust infrastructure.
Consumers can also play an important role in achieving a circular economy. They can make choices that support circular processes, such as choosing products that are durable, repairable, and recyclable. In addition, buying clothes made from recycled or renewable materials can help stimulate demand for these materials and promote a circular economy. Also, customers should be mindful of how waste impacts the environment and should be educated on how items are disposed of at the end of their life.
To implement fiber-to-fiber recycling, there should be an improvement in how textile materials are produced and consumed. Currently, fabrics are not designed with recycling in mind. In particular, producers must extensively focus on 'design for recycling' strategies for textile materials. For instance, increasing the use of mono fibers would stimulate recycling. The issue of blended textiles is currently the greatest problem in reducing the rate of recycling. While blending of textile fibers is inevitable, less complex blending can be achieved. For instance, instead of mixing at the fiber spinning stage, blending can be introduced during weaving and knitting so that yarns are single components. This reduces the complexity and the processing conditions when recycling, as only the yarns are blended. It is less complex to separate blended yarns than blended fibers. In fabric finishing, producers should avoid hazardous chemicals, dyes, or coatings that make recycling impossible.
Proper material selection, sorting, and grading techniques are required to facilitate larger-scale recycling. However, due to the large variety and complexity of blended materials, developing a standard system to handle textile waste is challenging. In addition, the absence of technologies to identify fiber composition and separate fibers makes recycling blended textiles impossible. Future research should focus on time- and cost-effective, automated sorting and grading systems that quickly sort fabrics based on fiber type, composition, and colors to reach full-scale recycling.
The way textiles are disposed of or recycled has an impact on most of the Sustainable Development Goals (SDGs), a collection of 17 global 2030 goals that were introduced by the United Nations in 2015. 159 SDG 12, ‘Responsible consumption and production,’ links most of the other SDGs and is structured over eight targets, addressing the use of natural resources, chemical waste, and fossil fuels and the integration of sustainable practices into production cycles, all of which apply to textile recycling. SDG 6 relates to ‘Clean water and sanitation.’ The release of hazardous chemicals and materials from dyes in textile waste can be linked to this goal. SDG 13 focuses on taking urgent action to combat climate change and its impacts. The production of fibers and incineration of textile waste is one source of greenhouse gases. Keeping materials in use longer and regenerating waste plays an important role in reducing carbon emissions and addressing the targets of this goal. SDG 14 stresses the issue of marine pollution from waste, such as dyes and microfibers released into the oceans from cloth washing and textile degradation in landfills. Textile waste recycling and deploying cleaner technologies have huge positive effects on the ‘Life below water,’ Clean water and sanitation,’ ‘Industries, innovation and infrastructure,’ and ‘Climate action’ goals. Since most of the SDGs are linked to the textile industry, recycling stakeholders need to integrate the SDGs into business models and strategies to ensure long-term sustainability.
Conclusions
In this work, current and emerging textile recycling technologies were reviewed. Although mechanical recycling is well-established, it cannot continue forever because it lowers the fabric's quality. Hence, most mechanical recycling technologies are down-cycling processes, as products are of lower value. Most textile fabrics are produced from blended natural and synthetic fibers, which cannot be easily separated using mechanical recycling methods. Chemical and biological recycling offer recycling options for blended fabrics while retaining high fiber quality. However, they have not yet been appropriately scaled up compared to mechanical recycling. Biological recycling of textile fabrics is not yet used in a closed-loop recycling format in the textile industry (textile to textile). This technology has been demonstrated in open-loop recycling, such as feedstock for succinic acid and ethanol production. The advantage of chemical and biological methods to deconstruct end-of-life textile waste is a viable way to overcome the issues associated with the multi-material nature of textiles such as polycotton. However, pre-treatments are required to remove dyes and finishes, hindering recovery. Current pre-treatments are expensive, and improved techniques must be developed for the technology to be sustainable and economically viable.
Chemical recycling is a promising approach for closed-loop recycling of cellulosic textile waste, as the process maintains the inherent properties of fibers throughout the recycling process. The most suitable method for recycling polyester fibers is degradation to TPA, which can be re-polymerized to produce new polyester fibers. Mechanical recycling is the only viable option for wool fibers, as keratin from wool has not proven helpful in generating fibers for the textile industry. Although various researchers report recycling polyester to separate polyester from its blends, there is limited research on recycling polyester fabrics, providing a research gap for future work. This review has shown that recycling technologies as an approach to utilize textile waste as a raw material resource are viable. In conclusion, recycling textile fabrics is a possible reality that needs proper investigation and appropriate resources.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Waste Research, Development and Innovation Roadmap (grant number CSIR/BEI/WRIU/2021/047).
