Abstract
The release of microplastics from the textile industry is a significant environmental concern. While the release of microplastics during the laundering process and within wastewater treatment plants has been extensively studied, there is still a pressing need for a standardised sampling protocol to assess microplastic release across various matrices involved in different textile production processes. In this report, we propose a comprehensive sampling protocol targeting four types of samples derived from textile production processes: sludge, wastewater, dust, and air. We also provide a detail method for the extraction and analysis of microplastics from these samples, including the separation of microplastics from collected textile samples through cellulose dissolution, oxidation, and oleophilic extraction processes, as well as heat treatment for air samples. This is followed by automated methods for quantifying microplastic fibres. The proposed protocol provides an efficient, effective, straightforward, cost-effective, and safe approach for analysing and monitoring textile-related microplastics.
Keywords
Introduction
Global textile fibre production has been dominated by synthetic fibres, which accounted for 65% of the total global production in 2021. Among these, polyester production alone made up 54%, while nylon and other synthetic materials contributed the remaining 11%. 1 Although the extensive use of synthetic materials has brought significant benefits to our standard of living, it also poses serious challenges that cannot be ignored – one of which is the release of microplastics.
Microplastics are, as the name suggests, tiny pieces of synthetic material. They are typically defined as pieces of plastics with dimensions under 5 mm, down to 1 µm. For particles even smaller than 1 µm, the term ‘nanoplastics’ has been coined.2–4 Microplastics are classified into two categories: primary and secondary. Primary microplastics are those that are directly released as small particles, such as scrubbing agents intentionally added in personal care products or synthetic fibres released during home laundering or erosion of tyres. Secondary microplastics, on the other hand, are plastic fragments generated through the degradation of larger pieces of plastic to the environment. 5
Mechanical processes that may lead to the release of microplastics are inevitable in textile production. Some textile production processes, such as dyeing, washing, and cutting have been widely reported to contribute significantly to microplastics release in the industry; 6 other mechanical processes, such as yarn spinning, fibre drawing, winding, weaving, knitting, embroidering, etc. less studied but may also contribute to the release of microplastics, and are worth studying.
Studies have reported on microplastics found in natural environments, such as in seawater, rivers, beaches and soil, and their effect on marine life. 7 However, compared to the extensive studies on microplastic release from laundry effluents,8–17 microplastic release during other textile processes, such as yarn making, fabric production, or garment making, are less reported.
Samples collected from textile production sites can vary vastly in form and composition. A range of sample purification and extraction methods have been proposed based on different sample matrices, and some efforts have been made to develop a general procedure applicable to a wide variety of samples.18–20 Recently, the International Standard ISO-4484 21 was published, offering a general protocol for the qualitative and quantitative analysis of microplastic release from textile sources. These recent developments provide a strong foundation for the future advancement of more comprehensive protocols for addressing textile-sourced microplastics.
Given the absence of a standardised protocol for assessing microplastic release across various sample matrices, 22 we propose sample collection protocols for four types of samples from textile production facilities: sludge, wastewater, dust, and air. These protocols are designed to facilitate future studies on textile-associated microplastics. Additionally, we outline methodologies for the extraction and analysis of microplastics from samples collected at textile production sites, following by an algorithm for automated quantification of microplastic fibres. The overarching objective is to present a comprehensive protocol that is efficient, effective, straightforward, cost-effective, and safe for analysing textile microplastics. Finally, we compare our approach with ISO 4484-2 to evaluate the similarities and differences in methodology.
Sample Collection and the Extraction of Microplastic Fibres for Quantification
The protocol begins with identification of sampling sites and concludes with quantification of microplastic fibres. The protocol covers four common types of samples that can be collected from a typical textile production facility: sludge, wastewater, dust, and air samples, arranged in order of increasing complexity of sample matrix, as illustrated in Figure 1.

Possible material within the sample matrix of air, dust, wastewater, and sludge samples.
The protocol can be segmented into three stages: sample collection, sample purification and extraction of microplastic fibres, and finally microplastic fibre quantification, as shown in Figure 2.

An overview of the protocol presented in this study.
An overview of sample collection process, beginning with the identification of suitable sampling sites is shown in Figure 3.
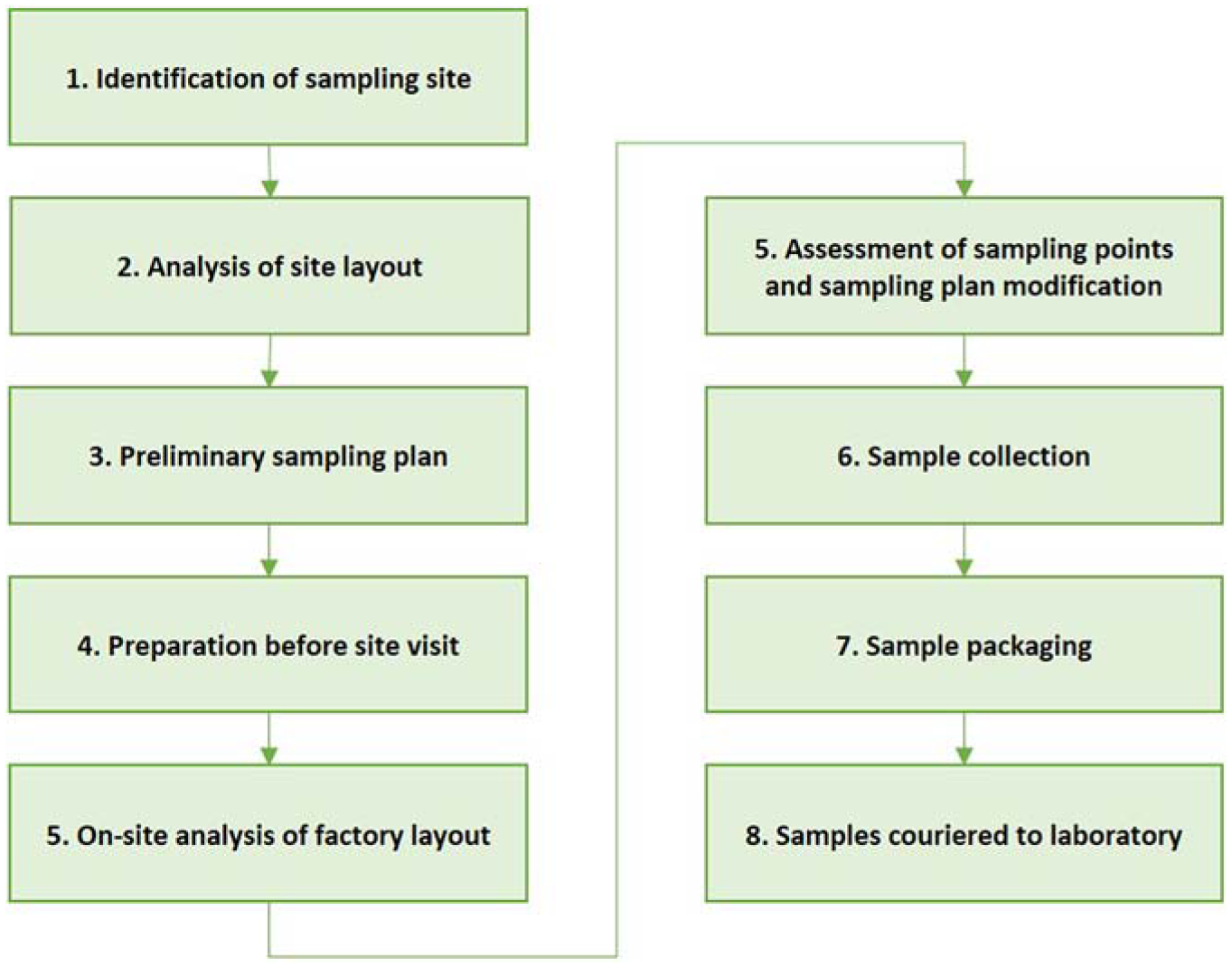
An overview of the sample collection process.
Two textile production facilities in Asia were selected as appropriate locations for the development of the sampling protocol. These two sites were chosen due to their representation of large-scale facilities in the region that specialise in the production of garments using synthetic materials. They provide an opportunity to investigate the release of microplastics across various production processes (Figure 4), beginning with yarn spinning and concluding with garment production. This vertical integration of the production process offers valuable insights into microplastic release associated with the garment manufacturing practices employed in the industry.
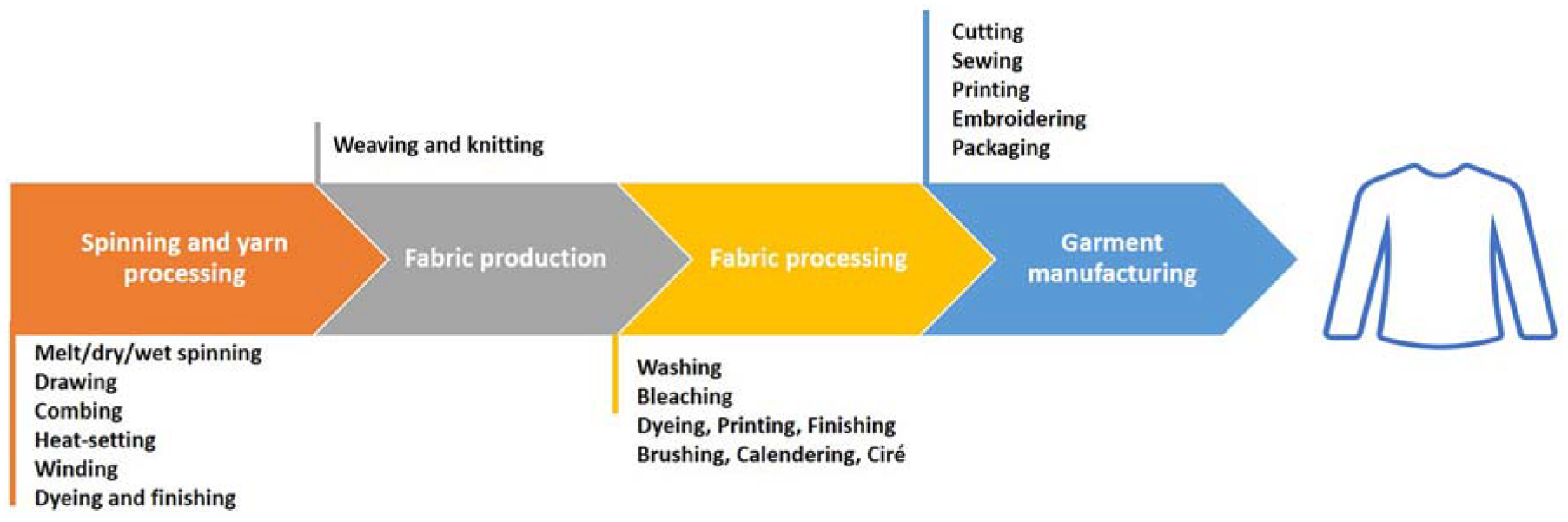
Major processes involved in the production of a piece of garment from raw materials.
Upon selection of the sampling sites, preliminary information was gathered for each location, including the factory layout, types of processes involved, production volume, and the types of textile materials handled. Although the types of samples to be collected from each site remain consistent, the sampling plan for each location must be tailored to account for variations in available sampling processes and the quantity of each sample type. Furthermore, upon arrival at the sampling site, additional details may emerge, requiring revisions to the preliminary sampling plan based on on-site inspections.
The nature of the various processes involved in textile production leads to differences in the release of microplastic fibre. For example, processes such as melt spinning are not expected to release significant amounts of microplastic fibres, as the fibres do not undergo heavy mechanical abrasion. In contrast, processes that involve significant mechanical movement and abrasion, such as weaving, are expected to lead to substantial microplastics fibre release in the form of airborne microplastics and dust on the ground. As a result, air and dust samples were collected for these processes.
On the other hand, dyeing and other wet processes are not expected to produce significant airborne microplastic fibres, as the fibres tend to be retained within the solution and are subsequently expelled in wastewater. For these processes, wastewater and sludge samples are anticipated to provide more meaningful insight into impact of wet processes, as illustrated in Figure 5.
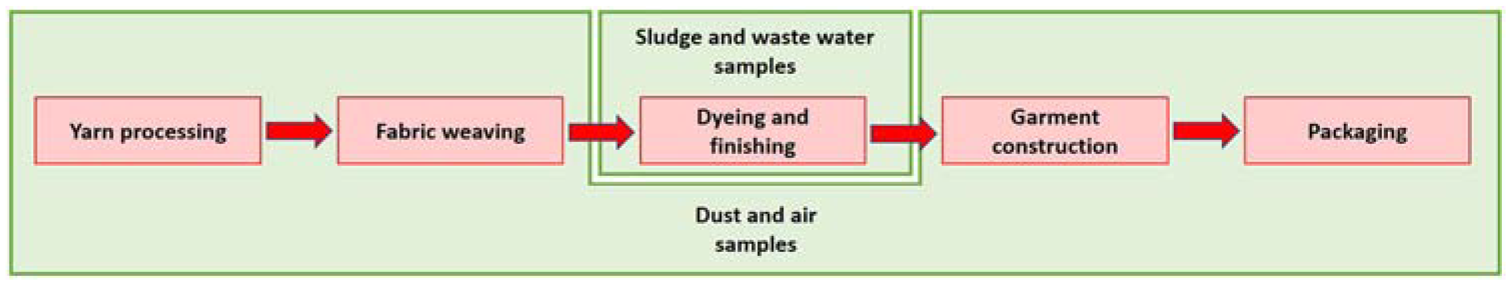
A general schematic of the garment production process, from yarn to garment, and the type of samples which can be collected at each step.
Of the four types of samples discussed in this study, sludge samples are considered the most challenging to purify and extract microplastic fibres due to the complexity of the sample matrix. To address this, we analysed the samples in terms of their potential constituents, as illustrated in Figure 1. In this context, microplastic fibres refer to synthesis fibres in general, while natural fibres primarily include cotton, along with other minor natural fibres. Textile waste may also contain partially degraded fabric, chemicals, or dyestuff. Additionally, debris refers to larger pieces of materials such as pebbles, leaves, fabric pieces, or even human hair.
The following sections provide a detailed examination of the four types of samples. Each section outlines the development of the sampling plan, the methods used for sample collection, the purification processes applied, the extraction of microplastic fibres, and the subsequent quantification of these fibres. This comprehensive approach will ensure a clear understanding of the methodologies employed in this study.
General Considerations and Precautions
Microplastic samples are highly susceptible to contamination from environment sources, including unclean glassware, fibre shedding from garment worn by researchers, and airborne fibres. As such, a series of precautions were taken to minimise sample contamination, as well as to reduce sample loss during sample manipulation and extraction. General precautions, such as avoiding plastic labware and possible contamination, were implemented during sampling to minimise sample contamination. 21
An overview of the full protocol is shown in Figure 6, which includes sample collection, purification and extraction, as well as microplastic fibre quantification. This systematic approach is crucial for ensuring the integrity of the samples and result reliability.
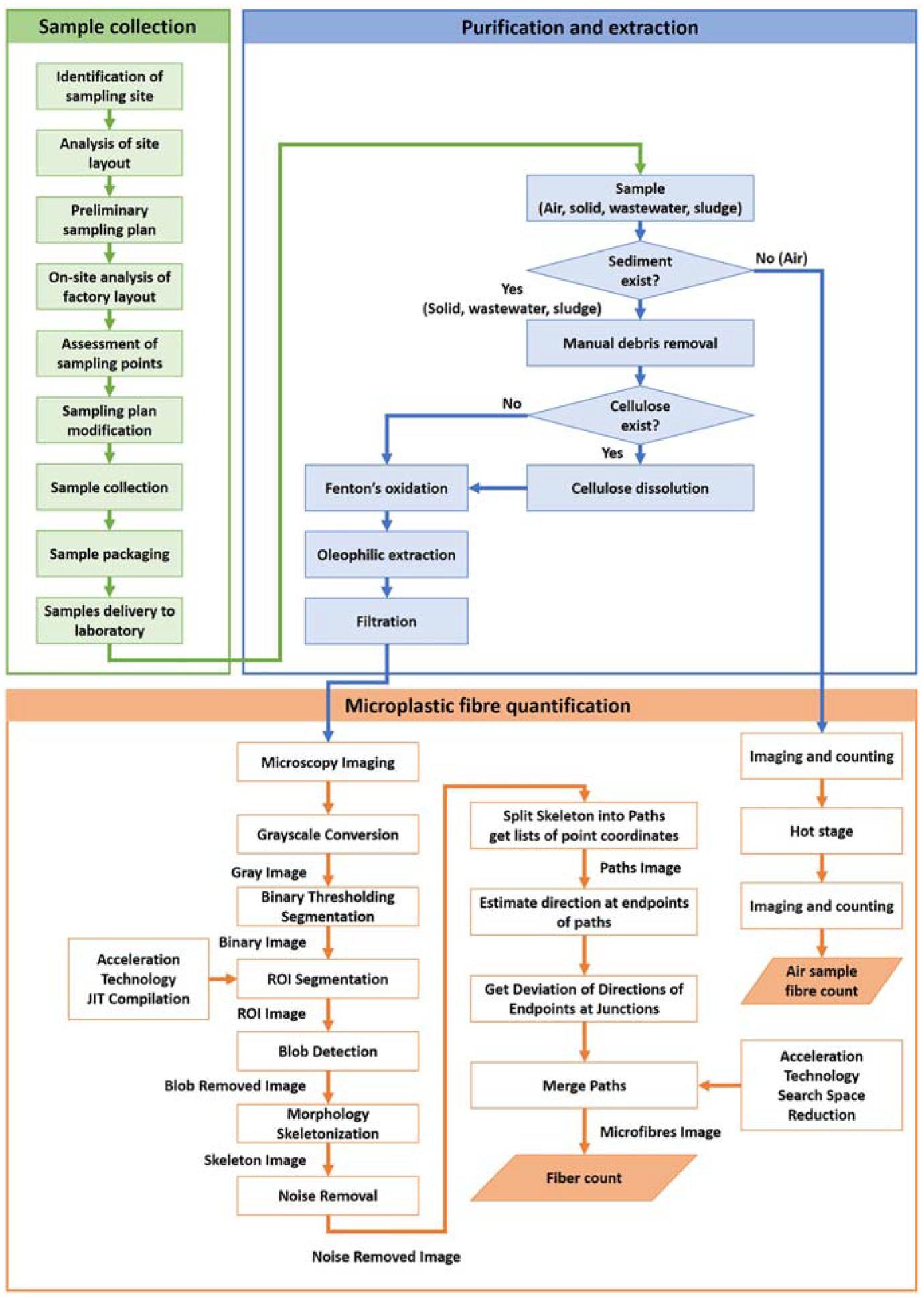
An overview of the protocol.
Sample Types
Sludge
Sludge generated in wastewater treatment process is usually cleared up periodically, following by compression or drying for easy storage and handling. The sludge will then be collected for further treatment according to local regulations. Figure 7 shows a typical sludge compression facility and storage area.

(Left) sludge compression facility; (right) storage facility for dried sludge, the farther in, the older the sludge.
Since sedimentation tank clearing typically occurs periodically – depending on the wastewater capacity of the site and the availability of sludge storage facilities – it is impractical for industrial sites to continuously monitor and compare sludge composition with the production process. In this study, sludge samples were collected from multiple locations within the sludge storage facilities to provide a representative overview of the microplastic content. Multiple replicates were taken to ensure accuracy.
Sludge samples were collected using a stainless-steel spatula and stored in glass reagent bottles with PTFE-coated caps. The collection date and location were recorded for each sample. The samples were then transported to the laboratory for analysis. Upon arrival, they were stored in a cool, dry place and kept sealed until further processing to prevent contamination.
Wastewater
Wastewater is generated as a result of wet processes, such as dyeing and finishing. Typically, the wastewater is directed to the treatment system, where it undergoes a series of physical and chemical purification processes, including sedimentation, oxidation, and disinfection. Sludge is regularly removed from the sedimentation tank for disposal. The water then passes through a series of filtration stages and finally undergoes reverse osmosis (RO), allowing the treated water to be reused in the factory.
Wastewater sampling points are highlighted in red in Figure 8. These include:
raw wastewater from the production process;
treated water from the RO system (reused in production); and
wastewater discharged from the RO system.
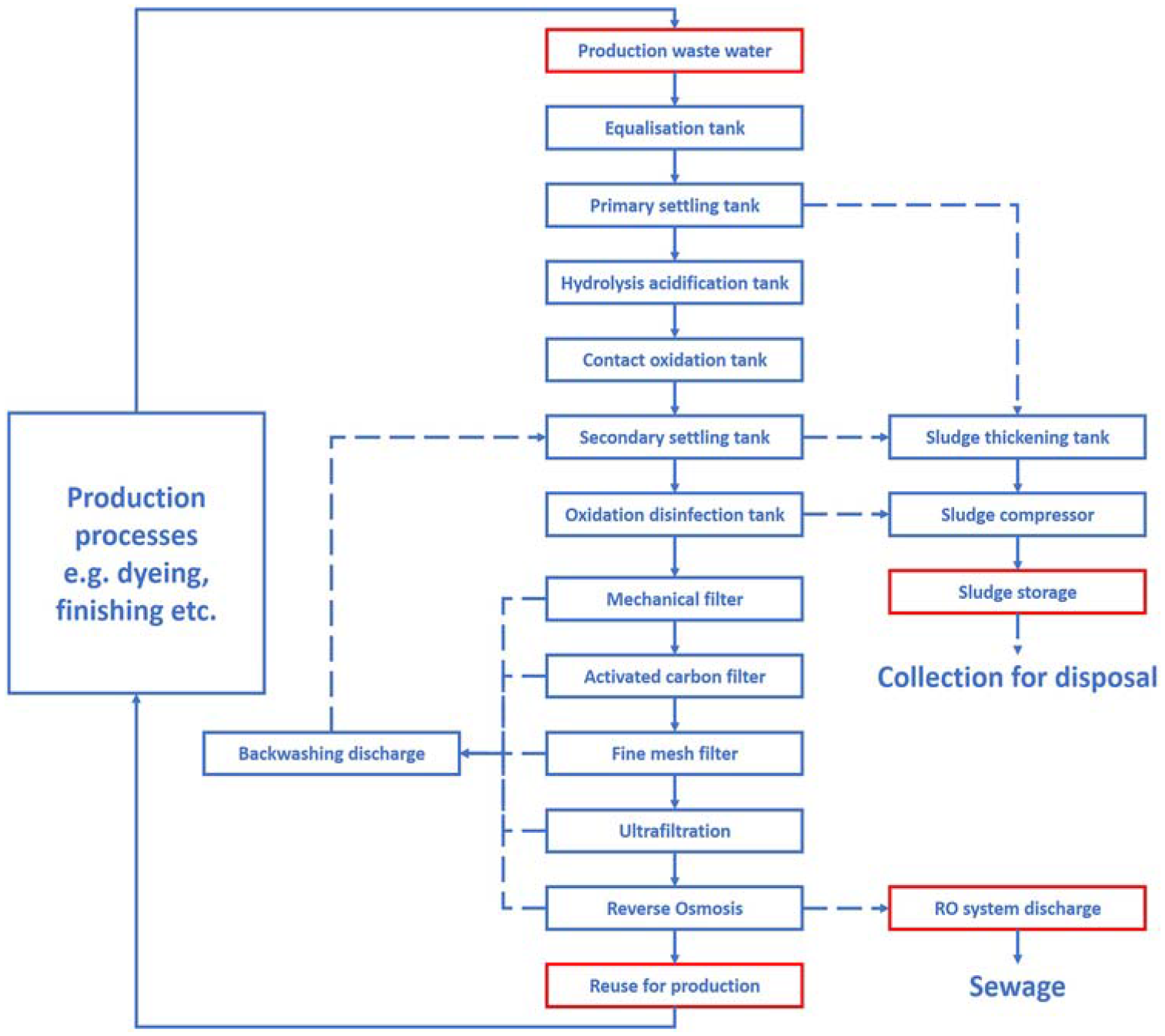
Schematic of the wastewater treatment and recycling system at the sampling site.
A comparison of the microplastic fibre counts from these samples allows an assessment of:
the quantity of microplastics released during manufacturing; and
the efficacy of the wastewater treatment system.
Wastewater samples were collected with the assistance of production site personnel, as access to the treatment tanks is often restricted due to safety regulations. Samples were collected in glass reagent bottles fitted with PTFE-coated caps to minimise contamination risk. The collection date and location of each sample were meticulously documented. Upon arrival to the laboratory, samples were stored in a cool, dry environment and remained sealed until analysis to prevent potential contamination. This rigorous handling protocol ensures sample integrity for accurate microplastic content assessment.
Dust
‘Dry’ processes involving significant mechanical movement can cause the release of microplastics. This occurs due to dynamic forces and abrasion both between mechanical parts and fibres/yarns, and between fibres/yarns themselves, leading to damage and fragmentation of longer fibres into shorter fibres.23–25
Dust samples consist of airborne microplastic fibres that have settled on floors near operating machinery. Sampling was performed on pre-cleaned floor areas in front of the active machines for a standarised duration of 1 hour. A hog bristle brush or other natural-fibre brush was employed for collection.
The sampling area dimensions varied according to site layout. All collected solid particles and fibres were swept onto clean white paper before being transferred to glass vials and capped. Documenting the exact sampling area size is essential, as this parameter is required for precise microplastic concentration calculations during subsequent analyses.
To ensure accurate sampling of microplastics during the production process, the following protocol should be followed:
It is recommended to take a minimum of two samples simultaneously from the same process or to conduct repeats from identical production lines. Background data should also be collected from non-operational areas to serve as a reference for comparison.
Air Sample
Production processes which involve significant mechanical movement of synthetic materials can lead to significant microplastic release in the form of airborne microplastic particles. Some studies have reported airborne particles to be a significant contribution to microplastic pollution; 23 however, the share of microplastic fibre release in the form of airborne particles is still inconclusive and more research is needed to assess the significance.
Air samples were collected using a Sensidyne GilAir Plus Personal Air Sampling Pump, set to an airflow rate of 5 L/min, which corresponds to the average minute ventilation of a typical adult. Employing a specialised air sampling pump enables precise control over the airflow rate, and ensures consistent sampling conditions. Three-piece polyvinyl chloride filter cassettes of 25 mm diameter containing 25 mm quartz fibre filters were utilised. To enhance the reliability of the results, a minimum of two samples should be taken simultaneously from the same process.
Air samples were collected at locations identified as being prone to higher levels of airborne microplastic release, typically near machines that exhibit significant physical movement. These machines include, but are not limited to, cutting, weaving, knitting, and embroidery machines.
The air sampler was positioned as close to the machine as practical, taking into account safety concerns and the presence of moving parts that may restrict placement directly next to the source of airborne microplastic release. To mimic the average worker’s height, the samplers were fixed at a height of 1.5 m from the ground. An example of an air sampler positioned near a knitting machine is illustrated in Figure 9. This strategic positioning is crucial for obtaining accurate measurements of airborne microplastics in the working environment.

An example of the air sampling set up near a knitting machine, with the cassette opening pointing towards the knitting machine.
Air samples were taken during factory production near the operating machineries, and from non-operational areas for reference. After collecting for a predetermined amount of time (0.5–4 hours), the air sampler was switched off, and the cassette was sealed .
The literature has reported that microplastics in sludge can be resuspended into the atmosphere by wind due to their low density and hydrophobic surface. 26 Therefore, air samples were also taken at other non-operational locations, such as sludget storage facilities, which may be prone to greater microplastics release. However, our air samples taken from the sludge storage facility showed that the airborne microplastics count is at a similar level with other environmental samples.
Sample Purification and Microplastic Fibre Extraction
Quantification of microplastic fibres in environmental samples is one of the major challenges in microplastic studies. The composition of environmental samples collected from different sources can be vastly different and this makes the purification and extraction of microplastic fibres from the sample matrix very difficult. Many methods have been proposed and used by different research groups to target specific components, with various degrees of success, but consensus has yet to be reached when tackling matrices as complicated as sludge material.22,27–30
An overview of the sample purification and microplastic fibre extraction process is shown in Figure 10. For sludge sample, a drying process is needed as sample weight is greatly affected by the water content.
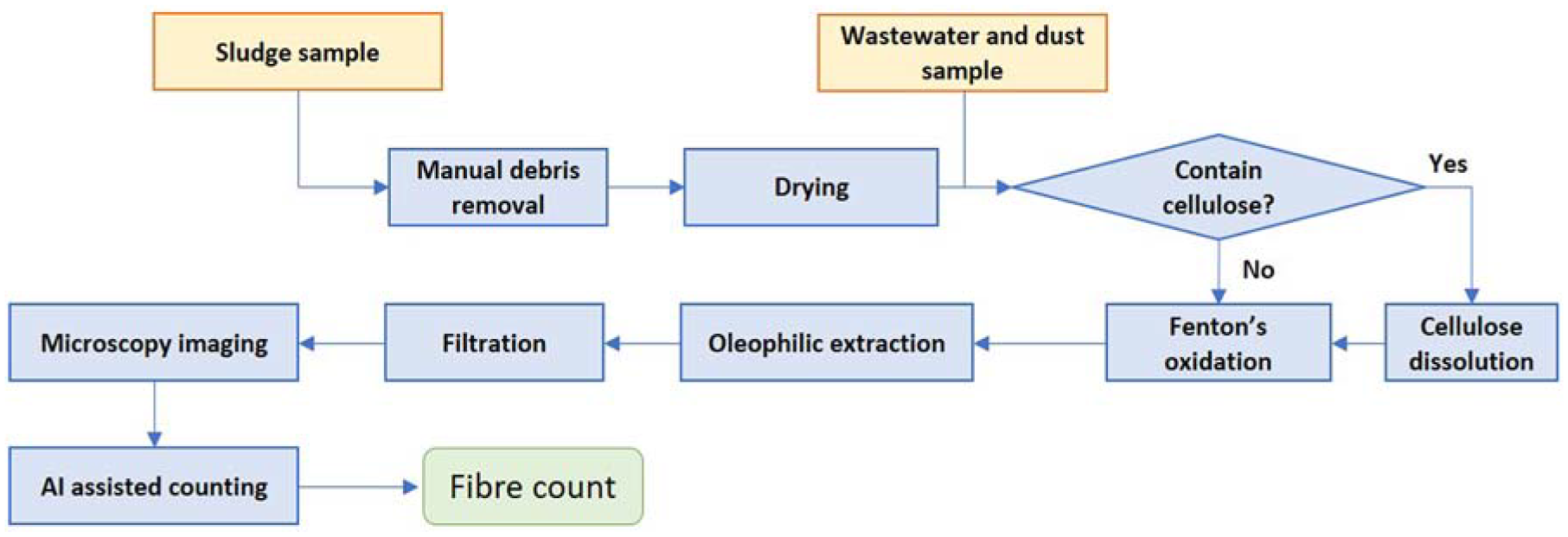
Sample purification and microplastic fibre extraction process for sludge, wastewater and dust samples.
Manual Debris Removal
Large pieces of non-microplastic fibres may exist in the samples, especially if the sludge storage facility is located in an outdoor area or not completely enclosed, such as depicted in Figure 7.
Manual debris removal is a step recommended by NOAA 31 for analysing microplastic samples which may contain large pieces of irrelevant matter. Samples taken for microplastics studies are often taken directly from the environment, and any large debris may have an effect on the chemical treatment and filtration processes that follow.
Another common approach is sieving or filtration using a series of increasingly finer mesh,31,32 exploiting the difference in size to separate out the microplastic particles of interest from the sample matrix. Microplastic particles in environmental samples are often in the form of beads, small pieces, or pellets. To a certain degree, the dimensions of these particles are similar (in the same order of magnitude) and can somewhat be approximated as ‘spherical’. 21 In contrast, the microplastics of interest in this study are in fibre form. These fibres are often around 8–20 µm in diameter but can range from 20 µm up to 5000 µm (5 mm) in length, meaning that sieving may not offer the same selectivity when separating microplastic fibres of interest from the sample matrix. 33
As such, though less efficient, manual removal using tweezers was chosen over sieving. Indeed, this is particularly suitable when faced with samples collected from textile mills since much of the solid matter in the sample is very fine.
Drying
Microplastic count per dry weight is commonly quoted for predominantly solid sample matrices. As such, sludge samples have to first be dried thoroughly to obtain their dry weight. 31
Common methods of drying quoted in the literature include freeze drying, 18 heating, and air-drying. 34 Freeze-drying requires the use of specialised equipment which may not be available in an average laboratory, and as such was not considered. Although more time-consuming, air-drying was adopted in this study. In order to minimise air-borne contamination, samples were loosely covered using aluminium foil to allow evaporation while minimising the introduction of any possible air-borne microplastic fibres.
Heating reduces the drying time significantly, especially if the matrix is high in water content. However, it has been reported that microplastic fibres exhibit changes in morphology at temperatures as low as 70ºC,35,36 so heating has been avoided in this protocol to avoid any potential damage to the microplastic fibres of interest. Care should be taken if gentle heating is to be employed to reduce drying time, such as by indirectly heating over a warm water bath.
Cellulose Dissolution
Cellulose-based material like cotton represents a major component in textile industry, so it is inevitable that microfibres generated from textile processes comprise a significant proportion of cellulose-based materials. The cellulose fibres should be removed or differentiated from synthetic materials in order to have meaningful insight into the microplastic fibre content of a sample.
Different novel methods have been used in different studies to differentiate cotton fibres from microplastic fibres, but there is not yet a consensus for the standard method to do so. One approach involves observing the difference in fluorescence by the fibres after Nile-red staining.37–39 However, our attempts to use this method as a quick and simple way to differentiate between the fibres was unsuccessful, in terms of allowing a clear indicative differentiation between synthetic and natural fibres. This is probably due to the complexity of sample matrix in textile mills compared to laboratories prepared samples.
Likewise, methods which make use of optical techniques, with the aim of fibre identification without having to go through extensive purification and extraction techniques, have largely remained viable only on laboratory-prepared standard samples. As such, in order to extract the microplastic fibres from the samples, it is necessary to include an additional step to eliminate the cellulosic fibres which will also likely be present in the sample.
ISO 1833-11:2017 40 provides a method for dissolution of cellulose within a mixture of fibres. However, the use of 75% sulphuric acid would not be compatible with nylon fibres which readily react and hydrolyse under acidic conditions. Considering the need for the reaction conditions to be relatively mild, as well as requiring a short time frame, Schweizer’s reagent, a reagent which is well known in the preparation of regenerated cellulose fibres, is used. 41
Different cotton and cellulose samples were prepared and subjected to different dissolution methods reported in the literature; their dissolution performances are shown in Table 1.
Cellulose dissolution using selected methods reported in literature.
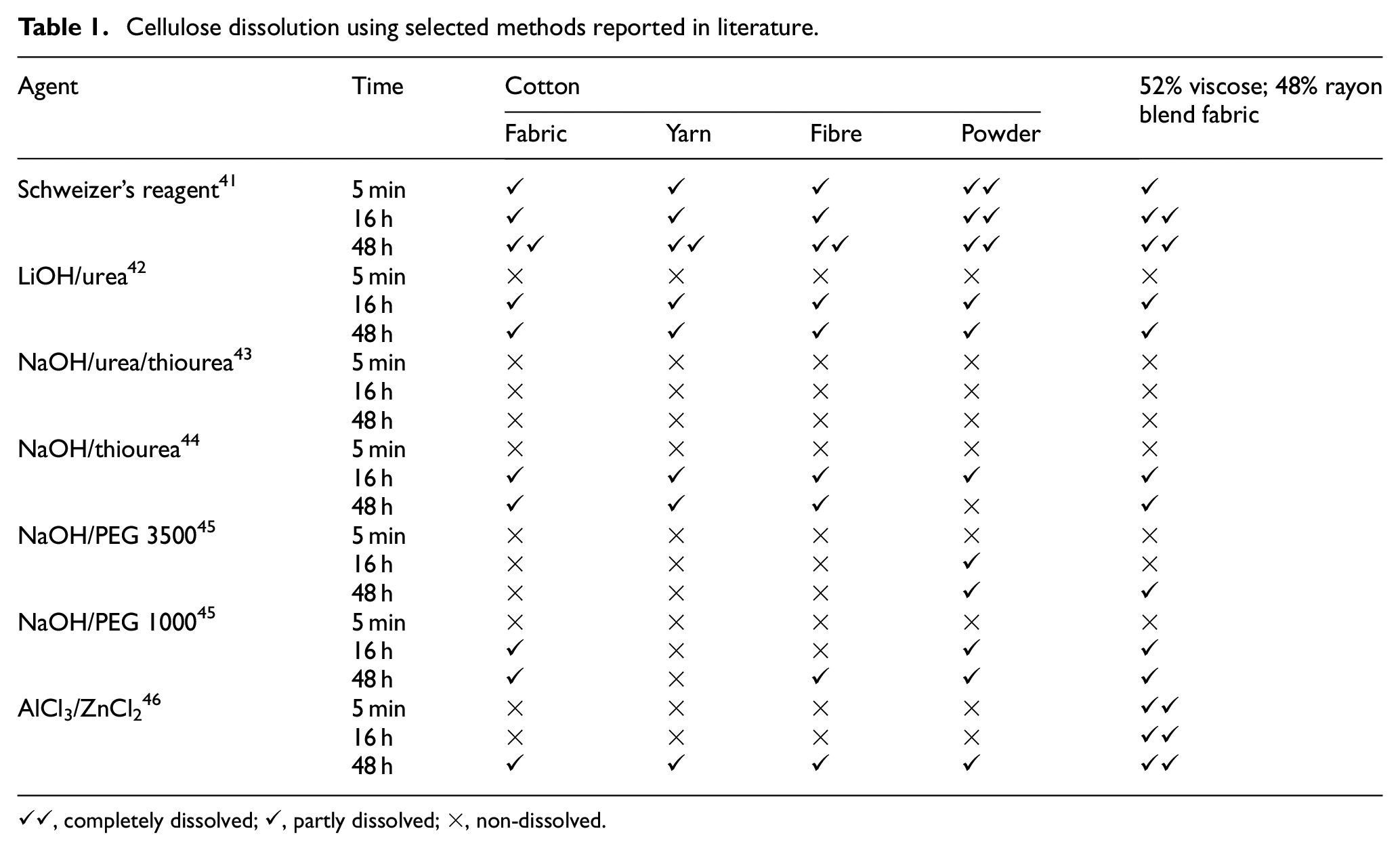
✓✓, completely dissolved; ✓, partly dissolved; ×, non-dissolved.
Schweizer’s reagent demonstrated a quick dissolution of cellulose based materials. Figure 11 shows that the dissolution of a cotton yarn can be observed within seconds upon the addition of Schweizer’s reagent, and it is nearly completely dissolved after 60 s. Trials using polyester, acrylic, and nylon fibres indicated that they were not affected by Schweizer’s regent, thereby confirming its suitability for dissolving cotton and other cellulosic material in the collected sample.

Dissolution of cotton in Schweizer’s reagent.
Schweizer’s reagent is a dark blue metal ammine complex ([Cu(NH3)4(H2O)2](OH)2) once widely used in the production of rayon and cellophane. It is very effective in dissolving natural cellulose sources such as cotton and wood pulp, and works by being inserted in between and stabilizing cellulose chains, thereby allowing it to dissolve in aqueous solution.
Dissolution of cellulose-based materials is included in the protocol as cotton constitutes a major source of natural fibre present in textile waste, which is also the only type of natural fibre involved in production sites in this study. Due to the modular nature of this protocol, it is possible to introduce additional steps specific to the type of contaminant targeted for removal.
Oxidation Using Fenton’s Reagent
Beside cotton fibres, organic matter in general makes up the majority of the sample matrix of textile samples; these organic matters are often digested with either strong acid/base or strong oxidant.
Hydrogen peroxide (H2O2) is one of the most common used oxidants. Fenton’s reagent,
47
which is formed by adding iron catalyst and acid to hydrogen peroxide, is a hydrogen peroxide-based reagent that can be used to oxidise organic matter in environmental samples. By using Fenton’s reagent, oxidation occurs at a much higher rate in a lower temperature to prevent possible heat damage to microplastic fibres. The ferrous ion catalyses the breakdown of hydrogen peroxide to generate hydroxyl and hydroperoxyl radicals
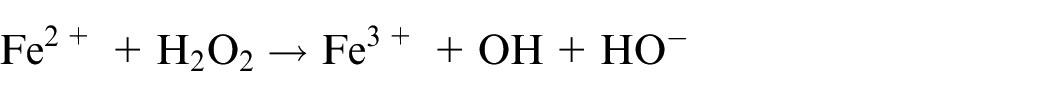
The reaction has been found to be relatively mild and does not cause significant morphological change on microplastic particles, nor the size of fragments.35,47

(Left) colour of the solution before adding H2SO4; (right) colour of the solution after adding H2SO4.
Oleophilic Extraction
A large amount of solid matter presents a challenge to microplastic studies. The most cited methods to remove solid contaminants follow two main approaches, one of which is physical separation based on the density difference between synthetic fibres and other fibres as well as other non-fibres, 32 while the other method makes use of the oleophilic interaction between oils and synthetic fibres. 48
Density separation also presents a major drawback, being that it is a very time-consuming process. Seventy-two hours is commonly cited for the time required for density separation. Our preliminary study indicated that it could take up to a week for our samples to separate effectively.
On the other hand, the oleophilic interaction between oils and synthetic fibres 48 introduced a faster and more effective approach. The basis for this extraction method is that the long carbon chains in the polymers and those in oils show significant intermolecular interaction and attract the microfibres from the aqueous phase into the oil phase .
Addition of sodium chloride (NaCl) into the extraction mixture increases the ionic strength of the aqueous phase, and therefore promotes the migration of microplastic fibres into the oil phase. This method is low-cost, easy to handle and much quicker, and does not make use of any toxic materials, compared to using zinc chloride in density separation.

The solution is shaken with oil until well mixed and allowed to settle upside down.

Filter membrane is sandwiched between Petri dishes for microscope imaging.
Imaging and Counting
Microscope Imaging
Images of the filter were taken using an optical microscope (Nikon Ti2). Brightfield images of the filter membrane were taken. The following recommendations result in high quality images:
Cellulose dissolution and oxidisation process are recommended for complicated samples like textile waste. Figure 15 compares the optical microscope images of a sludge sample which has undergone these processes, and a sample which has not. It is possible to count the microplastic fibres manually for both samples; however, the one which has undergone these two processes provides a cleaner image, which reduces the complexity of the counting process.

Filter membrane (top) with and (bottom) without the oxidisation process. Organic matter greatly affects the image quality.
Artificial Intelligence Assisted Counting
Quantification of microplastics in a sample is commonly performed via manual counting,19,49 which can be labour-intensive and time-consuming, especially when a larger number of samples is involved, and the strain on the researcher can lead to counting errors and subjective counting.
Standard protocols for manual counting are available for cell counting and fibre counting, 50 minimising biased or inconsistent counting; however, it still relies on the researcher conducting the tedious work of manual counting.
To varying degrees of success,37,38 attempts have been made by other research groups to automate the counting process. Due to the irregular dimensions of microplastic fibres, the algorithm involved will be much more complex compared to counting microplastic particles or beads. We have developed an AI-assisted microplastic fibre counting algorithm to reduce the time for the counting process. Our method presents three advantages: first, the algorithm can remove background noise and large ‘blobs’, and is therefore more tolerant to contaminants which would otherwise be miscounted as microplastic particles; second, the use of morphology skeletonization allows overlapping fibres to be resolved and counted as individual fibres; and finally, the algorithm is tolerant of very large image sizes.
The full view of the microfibre image is composed of 12 × 12 sub-microscopic images, with height and width dimensions of 7.7 × 108 pixel 2 and three RGB channels. The computational complexity of the microfibre image is resolved by developing specific algorithm and acceleration technology. Figure 16 depicts the flowchart of the AI assisted microfibre counting algorithm, which is illustrated in the following sections. Figure 17 shows parts of the original image and processed image; identified microplastic fibres are highlighted in different colours for demonstration.
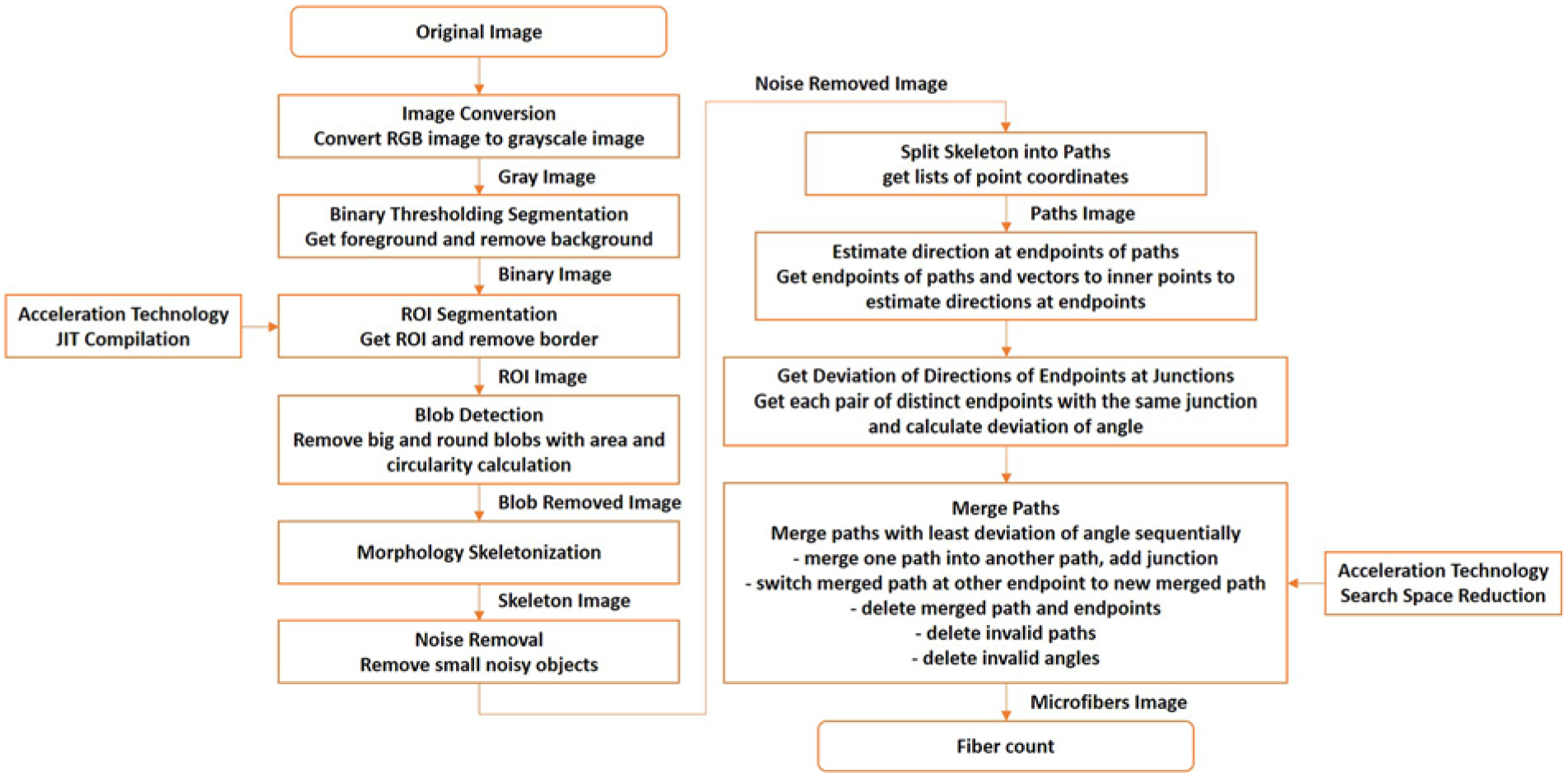
An overview of the automated image processing and counting algorithm.

(a) Original image and (b) identified microplastic fibres marked in different colours.
Image Pre-processing
Image pre-processing consists of image conversion, binary thresholding segmentation and region of interest (ROI) segmentation. The original image is RGB format, which is converted into greyscale image. To obtain the foreground, the background is removed through binary thresholding segmentation. ROI segmentation is operated to remove the border of the microfibre image.
Path Identification and Microfibre Detection
The main steps in microfibre detection consist of morphology skeletonization, splitting the skeleton into paths, estimating the direction at the endpoints of paths, getting the deviation of directions of endpoints at junctions, and merging paths.
The morphology skeletonization technique is first utilised to extract single-pixel-width-based shape features representing the general form of microfibres. Since microplastic fibres can easily overlap, a split and merge method is then applied to distinguish the separate fibres. The fibre skeleton can then be split into paths to extract a list of point coordinates. The endpoints of paths and deviation vectors to inner points can be used to estimate directions at endpoints. The deviation of angle of each pair of distinct endpoints with the same junction can then be calculated and finally paths with least deviation of angle can be merged sequentially.
Verification of AI-Assistant Counting
A paired sample
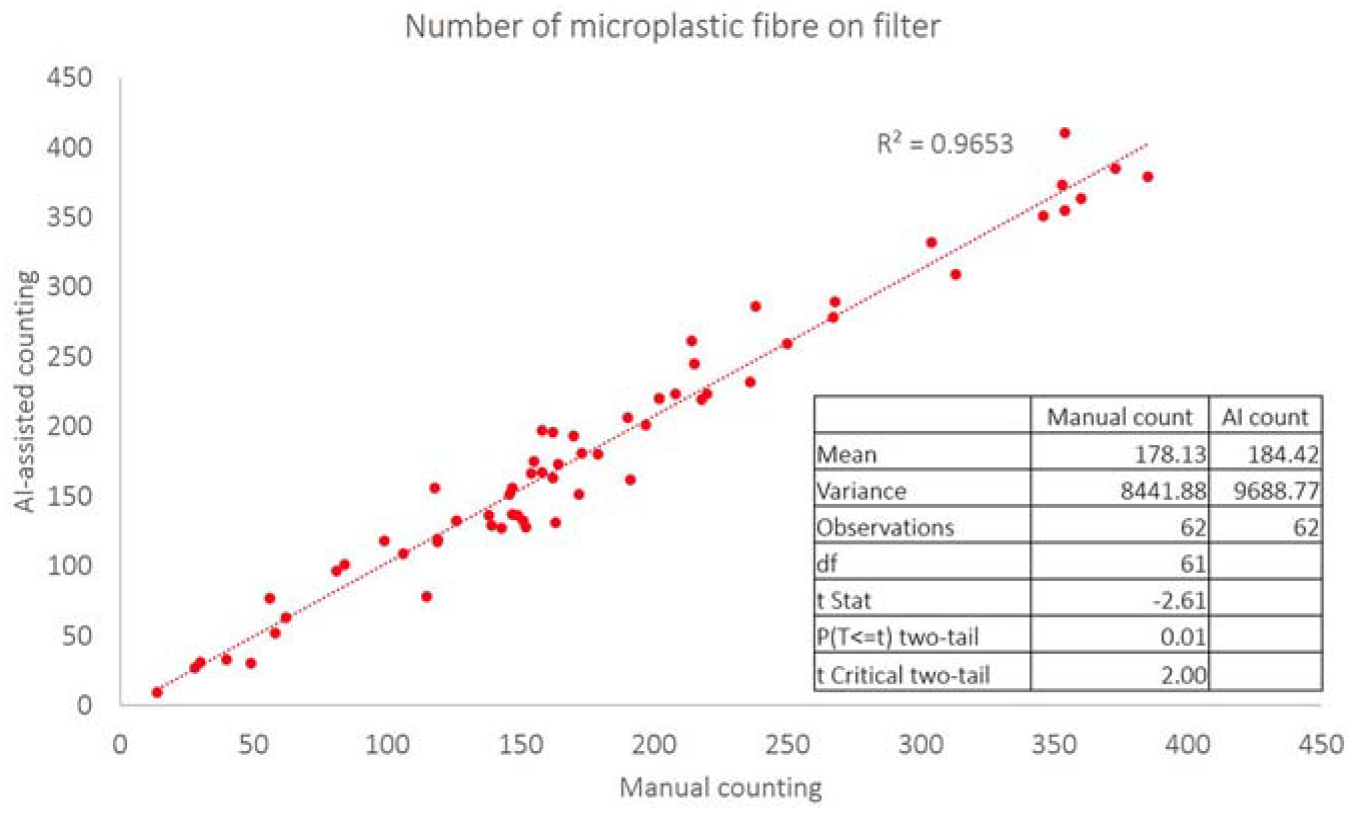
Statistical comparison between manual counting and AI-assisted counting.
Sample Recovery Rate
In order to assess the recovery rate of microplastic fibre after the whole extraction process, a standard solution of concentration 7.2 mg mL−1 (obtained from dispersing 250 µm milled nylon fibres (Goonvean Fibres Ltd, UK) in DI water and 0.0002% Tween-20 to ensure homogenous dispersion) was prepared and subjected to the entire protocol to assess the recovery rate of microplastics fibres from a sample after undergoing the entire protocol.
An average of 214.6 ± 9.1 fibres were counted from eight aliquots of 25 mL of solution. Another eight aliquots of solution were then subjected to the entire extraction and purification protocol, resulting in 177.4 ± 6.2 fibres. A recovery rate of 83% was determined for the process illustrated above.
Air Sample Purification, Microplastic Fibre Extraction, and Quantification
Since airborne microplastic fibres originate directly from the operating machinery, they can be directly observed on the glass fibre filter without extraction and purification. 51 The air samples were directly observed using a Leica M205C microscope following by an AI-assisted counting algorithm as described. However, since the air sample has to be observed directly on the filter as collected, it is possible for filter membranes to be overcluttered, thus preventing the effective use of automated counting. To minimise the possibility of such cases, sampling pre-trials should be conducted prior to sample collection to determine the suitable length of time for sample collection. Nonetheless, in cases where the filter membrane is overcluttered, manual counting was performed by dividing the filter membrane into suitable portions and randomly sampling these portions for manual counting.
Differentiate Synthesis and Natural Materials by Hot Stage
Air samples contain microplastic and natural fibres, so a further step for the differentiation of synthetic and natural fibres is necessary. The use of hot needle tests is common in microplastic studies. 52 This technique relies on the difference in response shown by fibres of different compositions towards the application of heat. Significant deformation or melting is a clear indicator that a fibre is synthetic, while colouration is indicative of natural fibres such as cotton undergoing thermal decomposition.
In this study, we focus on providing a tool to analyse the microplastic fibres produced in textile processes, so the major concern is the number of synthetic fibres but not their detail composition. Therefore, instead of the hot needle treatment to differentiate fibres, an INSTEC hot stage was used to heat the whole filter to 280ºC. The morphology changes of various type of fibres are shown in Figure 19, which corroborate the comprehensive study by Beckingham et al. 52 It is suggested that a hot stage can be used to differentiate between natural and synthetic fibres for air sample in this protocol. Details of the operation are shown below:

Various kind of fibres before and after hot stage treatment.
The number of synthetic fibres can be obtained simply by comparing the microscope images of the filter membrane before and after heating. The fibres which have significantly deformed (melting or shrinking) are synthetic fibres, whilst those which have not displayed a significant difference (colouration) are cotton or other natural fibres. In other words, the difference between the total number of fibres counted on the membrane and the number of fibres which have remained undeformed is the number of synthetic fibres collected.
Comparison with ISO 4484-2:2023
ISO 4484-2:2023 provided a clear framework for the qualitative and quantitative evaluation of the amount of microplastics within a sample, and is applicable to matrices such as wastewater, air and textile process solid waste samples.
In ISO 4484-2, samples (air, dust, wastewater, and sludge) are collected and then treated as necessary to remove unwanted contaminants so that only microplastic fibres are retained for counting. Preliminary checking such as conductivity, chemical oxygen demand (COD), and optical microscopy or a combination thereof can first be conducted to determine whether dilution or further purification steps are necessary. Purification steps, namely the use of acetic acid for salt removal and 15% hydrogen hydroxide for organic removal, were suggested. After this, filtration, microscope imaging and analysis, and finally quantification can be conducted.
There are two main areas which the present work differs from ISO 4484-2:2023. One of these is the procedure for removal of organic matter, and the other is the removal of cotton fibres.
Duration for Organic Removal
The use of hydrogen peroxide is common for removing organic contaminants when processing microplastic samples. Hydrogen peroxide is a relatively mild oxidising agent and therefore causes less damage to microplastic fibres compared to other alternatives such as strong acids/bases or chlorine-based oxidisers.
However, using 15% hydrogen peroxide to removal organic contaminants can take 7–30 days. Mild heating may be required due to trace amounts of stabilisers in commercially available hydrogen peroxide solution. 53 Alternatively, we proposed to use Fenton’s reagent instead of 15% hydrogen peroxide. The usage of Fenton’s reagent can greatly reduce the duration of organic removal from days to hours, without significant damage to common microplastic fibres.35,48,54
Cotton Fibre Removal
Cotton fibres represent a major challenge in the accurate quantification and assessment of microplastic fibres, and it is worth performing additional treatment specifically targeted towards the material. We believe that a protocol for the analysis of samples of a textile origin necessitates the inclusion of a dedicated cotton removal process to be comprehensive.
The procedure suggested in this present study makes use of a cuprammonium solution to dissolve cotton and other cellulosic fibres. Results show that cotton rapidly dissolves in the solution and undissolved microplastic fibres can then be easily filtered out for further processing.
Conclusion
This study presents a comprehensive protocol for analysing microplastic emissions from textile processes, encompassing air, dust, wastewater, and sludge samples. The modular nature of the protocol allows for flexibility in its application; when combined with a prior review of the production statistics for a specific site, it facilitates modifications and adaptation to best fit the nature of the sample collected from the production site.
Compared with typical advanced instrumentation such as micro-FTIR, micro-Raman or SEM, the current quantification methodology is based on optical microscopic analysis, with a recommended range of 100–400 microplastic fibres per filter, which could be easily adopted in a standard laboratory. An in-house developed automated counting algorithm has also been described, enhancing the efficiency of the analysis. Further advancements could improve accuracy and enable integration with spectroscopic techniques for material characterization.
Fibres present a greater analytical challenge compared to microplastic fragments or beads due to their geometric characteristics. The proposed counting algorithm significantly reduces the time required for quantifying microplastic fibres in microscope images, decreasing the process from hours to minutes.
In comparison to ISO 4484-2:2023, the current protocol uses Fenton’s reagent as an oxidation agent instead of 15% H2O2. It utilises oleophilic extraction in place of density separation, thereby eliminating the use of harmful ZnCl2 and reducing processing time. Furthermore, a dissolution procedure specific to cellulose-based fibres has been proposed. The use of Fenton’s reagent significantly reduces the reaction time from several days to several hours, without substantially affecting the microplastic content. In contrast to density separation, which requires ZnCl2, the oleophilic extraction process using olive oil and NaCl and reduces the extraction time from several days to 1 day, without the use of hazardous chemicals.
Microplastic studies remain a challenging field where a lot of research is yet to be conducted, and a general consensus on a set of standard procedures protocols for sample handling and analysis is still lacking. The publication of ISO 4484-2:2023 represents a landmark in the field of microplastic research. Certainly, this study is not the end or a final conclusion in microplastic analysis and quantification, rather it serves as a foundation upon which further refinements can be built upon towards evermore comprehensive protocols for microplastic sample analysis and quantification, so that the effect of microplastics on the environment and the human health can be better understood.
Limitation of this Study
Compared with environmental samples, sample taken in a production site contain much higher concentrations of microplastic fibres. Preliminary site analysis or sampling is recommended to estimate a suitable sample size, dilution factor and sampling time. The protocol is designed to provide a quick and easy-to-follow method to monitor the microplastic release in factory. It is assumed that the major type of material handled by the factory is known, and the area of interest is the number of microplastic fibres released instead of microplastic composition.
Footnotes
Author Contributions
Lapyan Yeung: Methodology, writing – review & editing, formal analysis, visualization. Haigang Yu, Investigation, validation. Derek Lai: Writing – Original Draft, visualization, project administration, resources. Tingyu Zhou: Investigation. Ka Ho Li: Investigation, project administration. Bin Li: Investigation, methodology, software. Yik Ming Kwan: Investigation, software. Hing Leung Chan: Formal analysis, validation. Yammy Yan Yi Cheng: Conceptualization, investigation. Ka Hei Chan: Conceptualization, investigation. Chi Hung Chow: Conceptualization, investigation. Erika Kit Shan Ngan: Conceptualization, supervision, project administration; Lei Yao: Conceptualization, writing – review & editing, supervision, funding acquisition.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the project ‘A Management Tool for Microplastic from Textile Production Process (Ref: ITP/048/19TP)’ from the Hong Kong Innovation and Technology Fund.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
