Abstract
Water repellency and flame retardancy have applications in many fields, such as automotive interiors, indoor decorations, upholstery, outdoor clothing and tents. Cotton, having superior qualities, is considered one of the best raw materials for tent manufacture. The study has undertaken the application of different water repellent and flame retardant chemicals on 100% cotton canvas fabric to be used as bivouac tents. The purpose was to find suitable water repellent and flame retardant chemicals that can be applied in a single bath or in a two-step process to give the desired repellency and retardancy effect. Initially, two screening sets were carried out, which aided in the identification of best-performed chemicals to be used in the combined application of water repellent and flame retardant. In the combined application, the fabric was finished using two approaches. In the first approach, both repellent and retardant chemicals were prepared in a single bath, and in the second approach, the fabric was first finished with flame retardant chemicals and then finished with water repellent chemicals. In both the approaches, the finished fabric was evaluated by a cone test, a hydrostatic pressure head test and a flammability test against locally set requirements. It is concluded that the procedure of single bath water repellent and flame retardant finishes (SBWRFR1b) showed reduced pickup percentage (60%) and resulted in better water repellency and flame retardancy properties, in comparison to other procedures and series of recipes.
Keywords
Introduction
Cotton fibre is one of the most reputable and preferred fibres used for many textile applications. Cotton’s superior mechanical qualities, biodegradability and air permeability make it a popular choice in civilian and military applications, including tent cloth, bedding, wall-hanging curtains, draperies, upholstery, firefighting, garments and apparel. 1 On the contrary, cotton fabric is not suitable for high-altitude applications because of its inherent characteristics such as good air–water permeability and high flammability.2–4
Finishing is the key way to impart functional properties in textiles, as it is the final phase and a crucial step in the production process. Layer-by-layer (LBL) assembly,5,6 chemical grafting,7,8 and the sol-gel process9–11 have all been used to create functional textiles with antibacterial, 12 flame retardant, 13 water repellent, 14 ultraviolet (UV) protection, or self-cleaning qualities.15,16 However, most of the flame retardant chemicals used in sol-gel procedures are hydrophilic, making them susceptible to fouling and water, resulting in the loss of flame retardancy and a reduction in the mechanical qualities of substrates after many laundering cycles. 17
Nowadays, there are two popular ways of improving the water repellency of fabric without losing its flame retardant characteristics. One technique to is to use adhesives such as polyurethane and epoxy resin, 18 but the results for flammability are comparatively not better than other techniques such sol-gel and the use of primary and synergistic flame retardants.9–11,13
The other, more promising option5–16 is to add water repellency to flame retardant coatings.19–22
Flame retardants are chemicals added to materials to prevent combustion and slow the spread of fire. Flame retardants can have a variety of compositions, including halogens (bromine and chlorine), phosphorus, nitrogen, metals, minerals based on aluminum and magnesium, or borax and antimony trioxide. 18
Cotton can also be thermally damaged, ignited and burned. As a result, numerous studies have been carried out to modify the surface of cotton to improve the fabric’s properties to match the requirements of the application area. The literature review demonstrated that coating the cotton fabric with hydrophobized ZnO nanorod array film, 23 hydrophobized TiO2, 24 hydrophobized SiO2 25 or organic–inorganic hybrid film, 26 or modification with silane, 27 improves the liquid repellency. Wilson et al. 28 reported that increasing the surface roughness of the cotton and altering it with a hydrophobic material simulates the surface phenomena seen on the lotus leaf.
Numerous studies have been reported to modify the cotton fabric with specific functionality. However, studies on multifunctionality are rarely reported. The sol-gel process has been recently used as a coating on cellulosic fibres. It gives water- and oil-repellent, antibacterial and flame retardant effects. 29 Previously, a plasma-induced graft polymerization (PIGP) process was employed to treat the cotton fabric to obtain flame and waterproof properties. 30 Concentrated ammonium polyphosphate (APP), sodium montmorillonite (MMT) and vinyl trimethoxysilane (VTMS) nanocoatings were created using self-assembly and in situ sol-gel methods, and they were then applied to cotton fabrics to create hydrophobicity and flame retardancy. 31
There are many applications such as outdoor clothing, automotive, bivouac tents and so on which require the functionality of water repellency and flame retardency. A literature review revealed that in most of the applications, cotton is used as the basal raw material. In this study, cotton is used to produce a water repellent and flame retardant bivouac fabric for local industry. A bivouac tent is a portable shelter of canvas fabric, supported by one or more poles or a frame. A bivouac tent is usually used by alpinists/soldiers during any expedition on the mountainside not only to protect them from the atmospheric conditions but also provide a comfortable zone for sleeping and resting. Different chemical finishes are applied to bivouac fabrics to enhance their properties, in which water repellency and flame retardancy are the most desirable finishes.29,30
Unlike other related research work, in this study, the hydrophobicity and flame retardancy of the bivouac fabric (developed by padding or coating process) are compared. The chemicals used in this work are less expensive than those used in the sol-gel process (e.g., the fluoro silane used by Vasiljević et al. 29 ) and similarly the coating procedure is significantly more accessible than the PIGP process. 30
Materials and Methods
Fabric
Canvas tents are available in a variety of thicknesses, but the general principle is that a heavier canvas correlates to a stronger tent with superior insulating capabilities. 32 As the weight of canvas increases, the resistance to tearing (tensile strength) and durability also improves. A 2-ounce increase in canvas weight, for example, has been shown to improve tensile strength by 25%. Conventionally, tents of canvas fabric with a GSM up to 350 are considered appropriate for occasional (1–2 days) summer use. For long-term use, it is recommended to choose a heavier canvas fabric having a GSM of greater than 350. 32
In this study, 100% cotton canvas fabric (plain) of 560 GSM was used. This fabric is made of 10/3 Ne (English count) cotton yarn used in both warp and weft. The 10/3 Ne means: three yarns of 30/s plied to form a single yarn of 10s English count. The numbers of ends and picks per inch are 44 and 26, respectively. The fabric is produced by Haji Nizam Din and Sons Pvt Ltd 33 and was purchased commercially from the market.
Application Procedures
The fabric was finished as per the guidelines provided by the chemical supplier in its data sheets. The procedures of water repellency and flame retardancy are given below:
One-step application procedure: single bath water repellent and flame retardant finish.

Two-step application procedure of water repellent and flame retardant finish.

*FR: flame retardant; **WR: water repellent.
Chemical and Auxiliaries Used
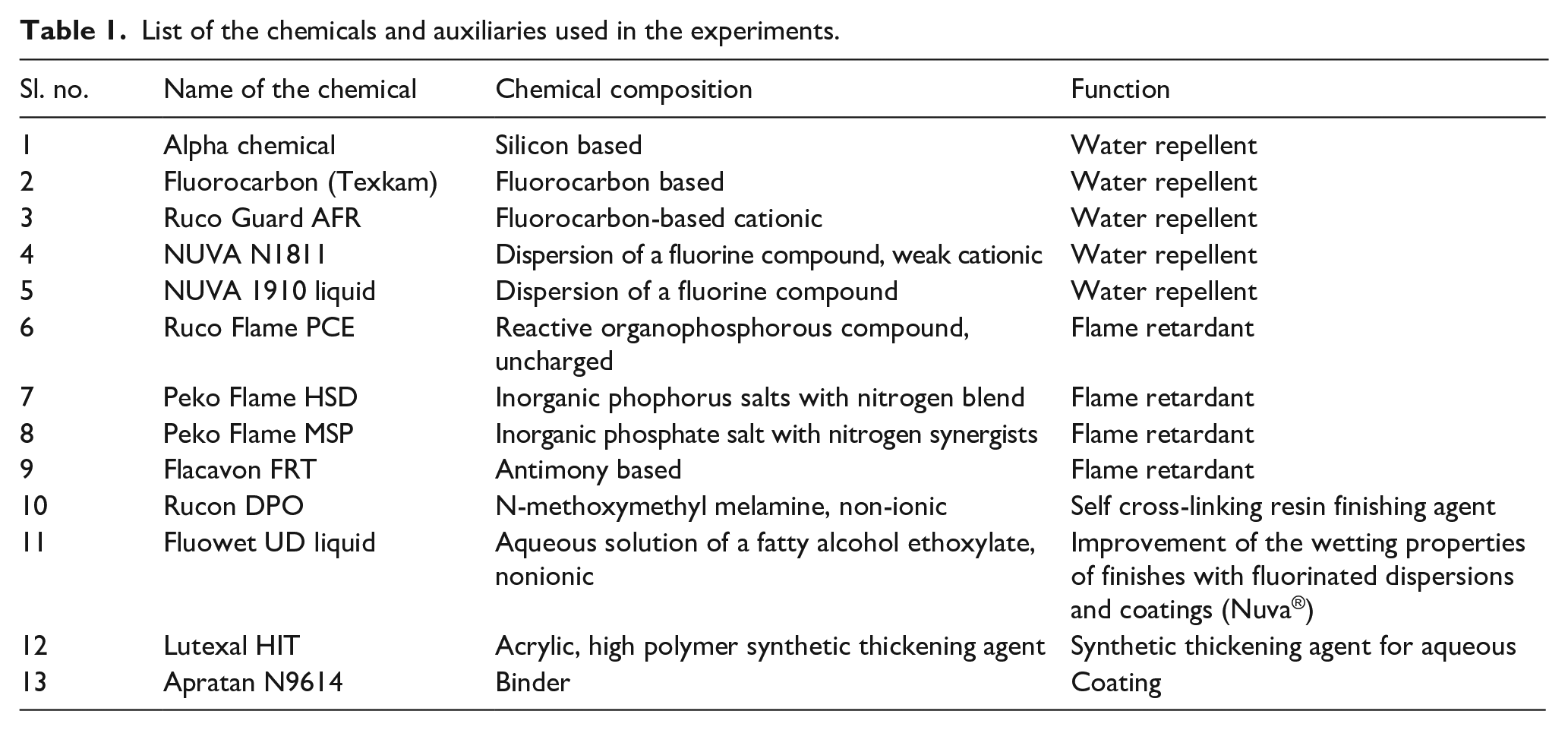
The chemicals and auxiliaries used in the experiments are mentioned in Table 1. Two types of repellent were considered; one is silicon based and the other is fluorocarbon based. Both are durable flame retardant chemicals but fluorocarbon gives oil repellency too. For flame retardancy, a primary retardant based on phosphorous (Ruco Flame PCE, antimony-based retardant (Flacavon FRT)) and a primary retardant along with a synergistic retardant (PekoFlame HSD & Peko Flame MSP) were used, and their chemical composition is mentioned in Table 1. The addition of synergistic retardant when added with primary retardant provides enhanced retardency. 34 Binder was added in some recipes to increase the adhesion of finish on to the fabric. Also in some recipes, a wetting agent was added to reduce the surface tension of the finishing liquor.
List of the chemicals and auxiliaries used in the experiments.
Evaluation of Finished Fabrics
Cone Test
A 31 cm × 31 cm piece of waterproof canvas was folded in the form of cone, so as to fit into a metal funnel as shown in Figure 1. The cone was filled with 400 cm3 of clean cold water and left inside the funnel for 18 h. During the period mentioned, there should be no leakage and no wetting of the outer surface of the cone. Condensation of moisture between the folds and the cone shall be ignored.

Cone tester used for the water repellency test.
Hydrostatic Pressure Head
A 205 mm diameter piece of waterproofed canvas was subjected to a constant head 30.5 cm of water for 1 h in a way that it shall not permit any leakage as shown in Figure 2. Leakage up to three tiny drops which may appear during the first 5 min and which do not grow in size shall be ignored.

Pressure head tester used for water repellency test: (a) clamp (manual), (b) hydrostatic pressure head tester (manual), (c) clamp (automatic), and (d) hydrostatic pressure head tester (automatic).
Flammability Test
A 51 mm × 318 mm treated sample was cut from at least three widely spaced sections of the sample. A total of 10 specimens were tested: five in warp and five in the weft directions. Before testing, the specimens were conditioned for at least 16 h at a temperature of 70 ± 2°F (21 ± 20°C) and relative humidity of 65 ± 2. A sample was suspended vertically 20 mm above the top gas burner, with 13 mm of the upper end in the clamp, so that a 305 mm length was exposed to flame. The flame was applied for 12 s vertically near the middle of the width of the lower end of the specimen. Then specimen was withdrawn, and time was recorded until the specimen self-extinguished. The burning time for any given specimen after removal of heat source shall not exceed 2 s or extinguish immediately. The afterglow time of the tested sample must be checked after removing the flame, which shall be limited to 45 s.
Once the sample was removed from the clamp, hooks were inserted in the specimen on each side of the charred area within 13 mm from the adjacent outside edge and 7 mm from the lower end of the specimen. Holes may be punched in the cloth for the hooks with an office paper punch. After that a tearing weight was attached to one of the hooks at approximately 10% of the unburned tear cloth (including the hook). It is important to apply the weight to the specimen gently without impact by raising the other hook. Char length shall be at a distance from the end of the specimen which was exposed to the flame, to the end of the tear made lengthwise in the specimen through the centre of the charred area. The average char length for a specimen shall not exceed 114.3 mm. The char length is the distance from the end of the specimen exposed to flame to the end of the tear made lengthwise of the specimen through the centre of the charred area (Figure 3).

Flammability tester.
Experimental Design
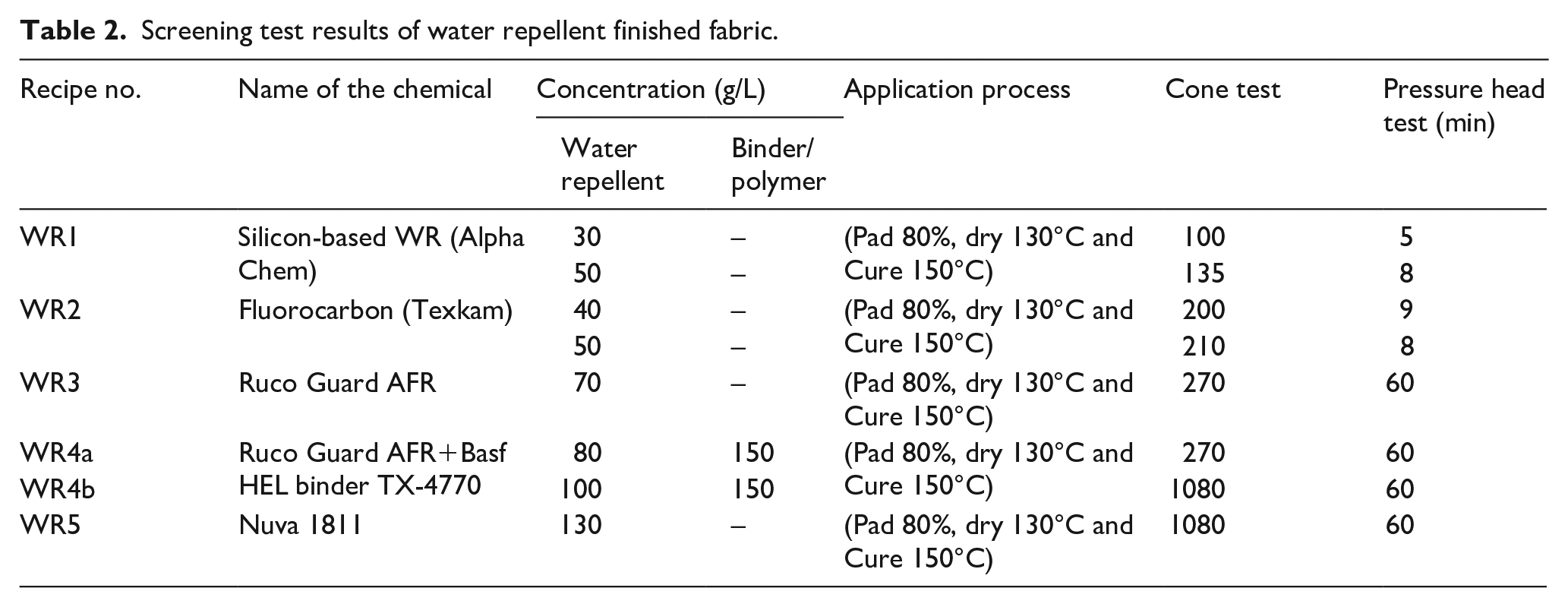
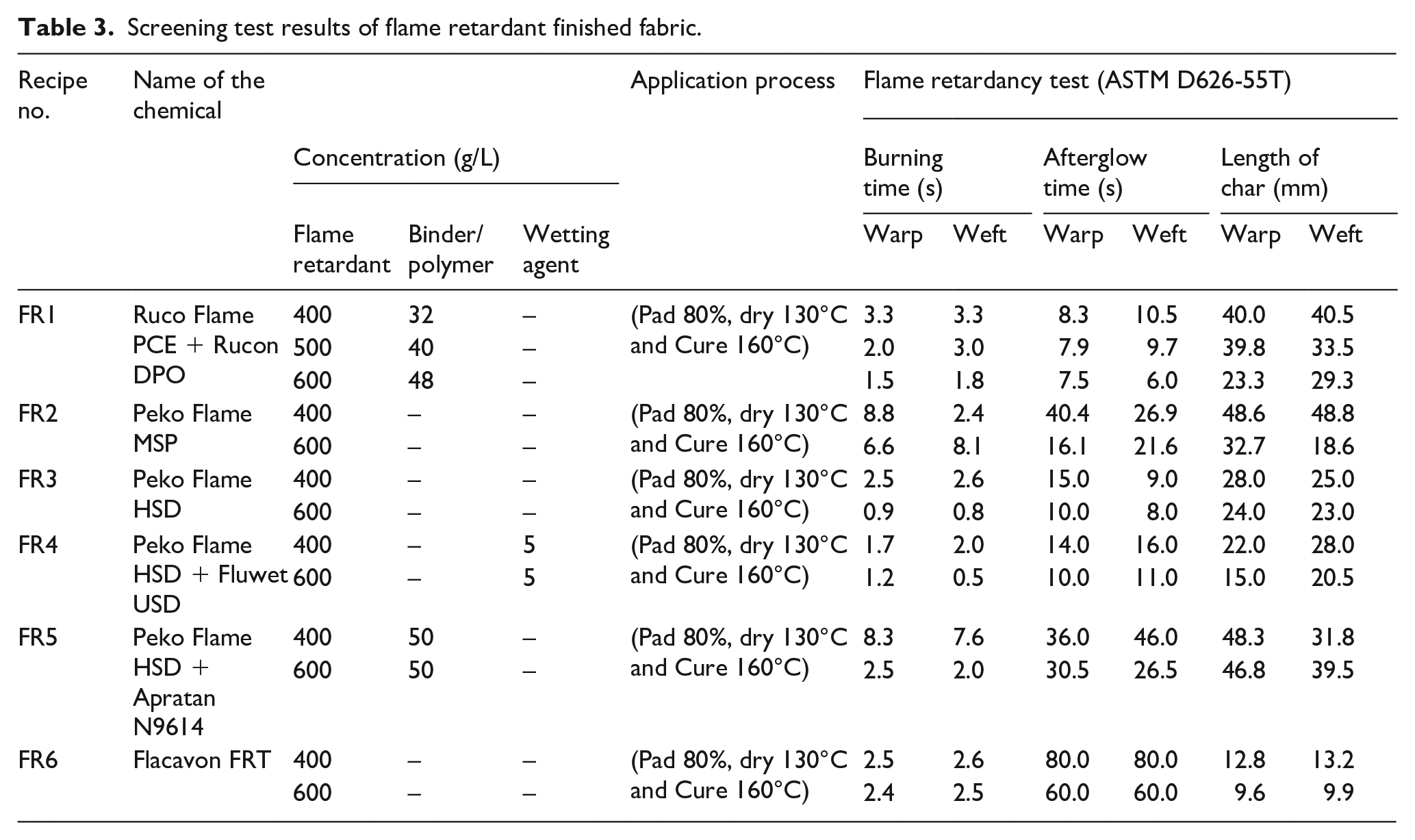
In order to impart a good water repellent and flame retardant finish, two screening sets were designed as mentioned in Tables 2 and 3. In set 01 (see Table 2), the fabric was treated with the different water repellent agents of varying concentrations. Similarly, in set 02, the fabric was treated with the flame retardant chemicals as listed in Table 3. The performance of the treated fabric was evaluated using a cone test, a hydrostatic pressure head test and a flammability test. Based on the screening test results, the fabric was then treated with water repellent and flame retardant chemicals. The finishing was carried out using two approaches. In the first approach, the fabric was finished with homogeneous solution of water repellent and flame retardant recipes, and in the second approach, the fabric was finished initially with a flame retardant recipe, then with a water repellent finish. In both approaches, the concentration of the chemicals and application methods were different as presented in Tables 4 and 5. In Tables 4 and 5, the recipes are coded as per the following scheme:
SBWRFR = single bath water repellent and flame retardant finish.
FRWR = flame retardant and water repellent finish.
Screening test results of water repellent finished fabric.
Screening test results of flame retardant finished fabric.
Recipes and application procedure of water repellent and flame retardant finished fabric.
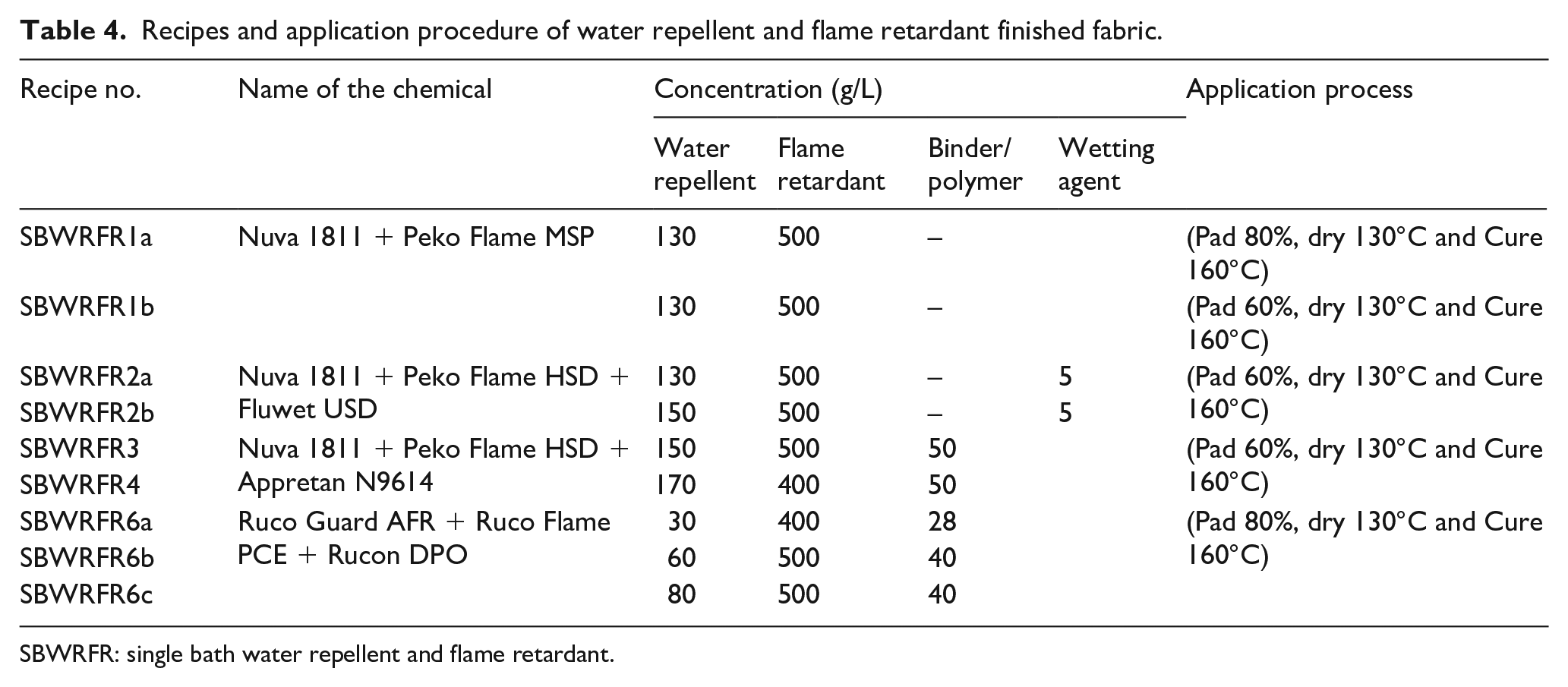
SBWRFR: single bath water repellent and flame retardant.
Recipes and application procedure of flame retardant and water repellent finished fabric.

FRWR: flame retardant and water repellent (finish in two steps; flame retardant finish then water repellent).
The evaluation of these finished fabrics was done using a cone test, a hydrostatic pressure head test and a flammability test.
Results
For the fabrication of water and flame retardant bivouac tents, two screening sets were primarily designed. The test results of these sets are mentioned in Tables 2 and 3. For simplicity, the recipes are coded as per the following scheme:
WR = water repellent.
FR = flame retardant.
The purpose of screening sets was to find suitable water repellant and flame retardant chemicals that can be used to give the best combination effect of water repellency and flame retardency.
Based on the findings of the screening test, the combined water repellent and flame retardant finishing was done using the chemicals that performed well during the screening test. The chemicals, their concentrations and processes are presented in Tables 4 and 5. The recipes in Tables 4 and 5 are coded as per the following scheme:
SBWRFR = single bath water repellent and flame retardant.
FRWR = flame retardant and water repellent (finish in two steps; flame retardant finish then water repellent).
Figures 4–7 represent the results of water repellent tests of both SBWRFR and FRWR. Similarly, flame retardant results of SBWRFR and FRWR (see Figures 8(a)–(c) and 9(a)–(c)) show the test results of flame retardant and water repellent (FRWR) finished fabrics (in a separate bath).
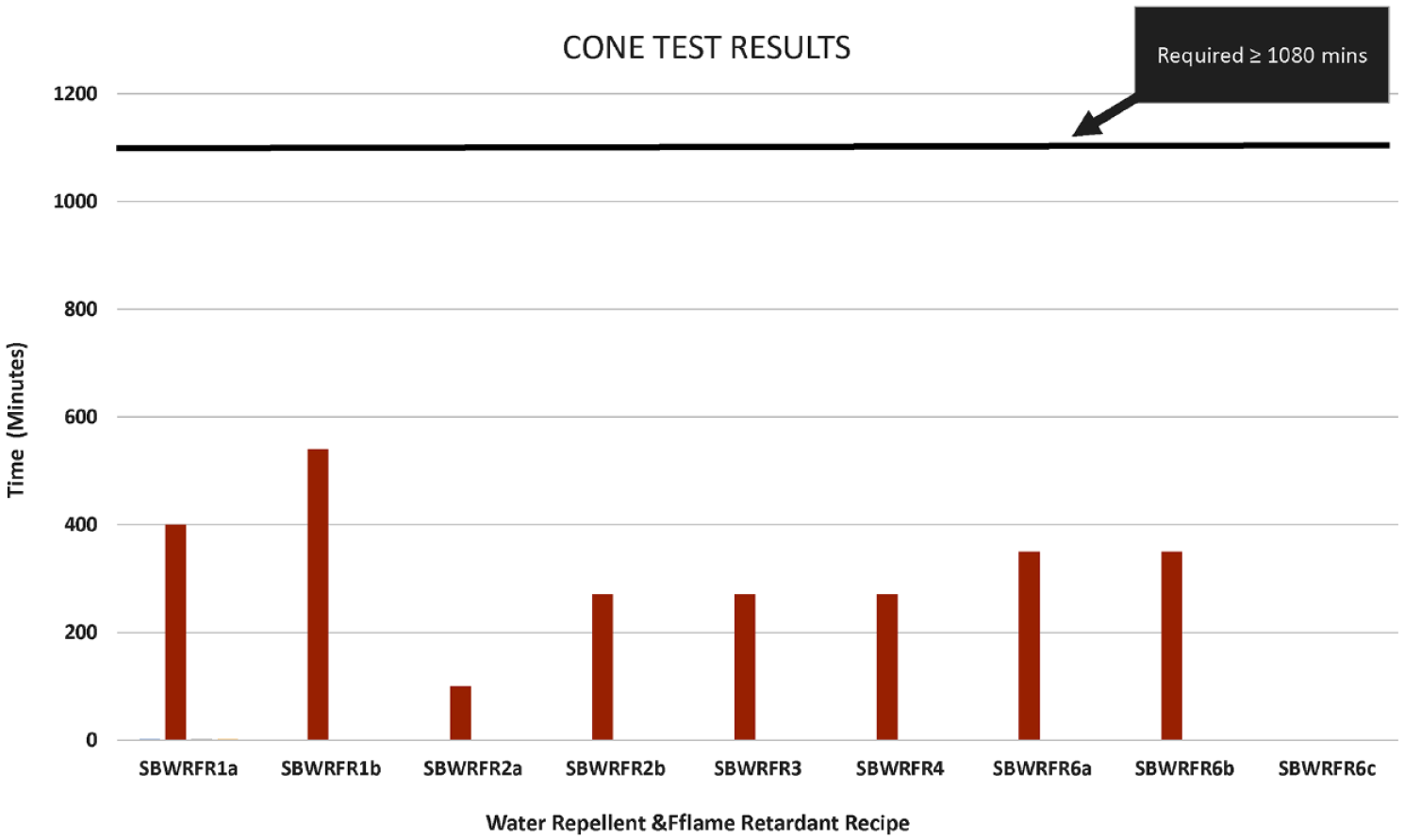
Cone test results of water repellent and flame retardant bivouac (SBWRFR).
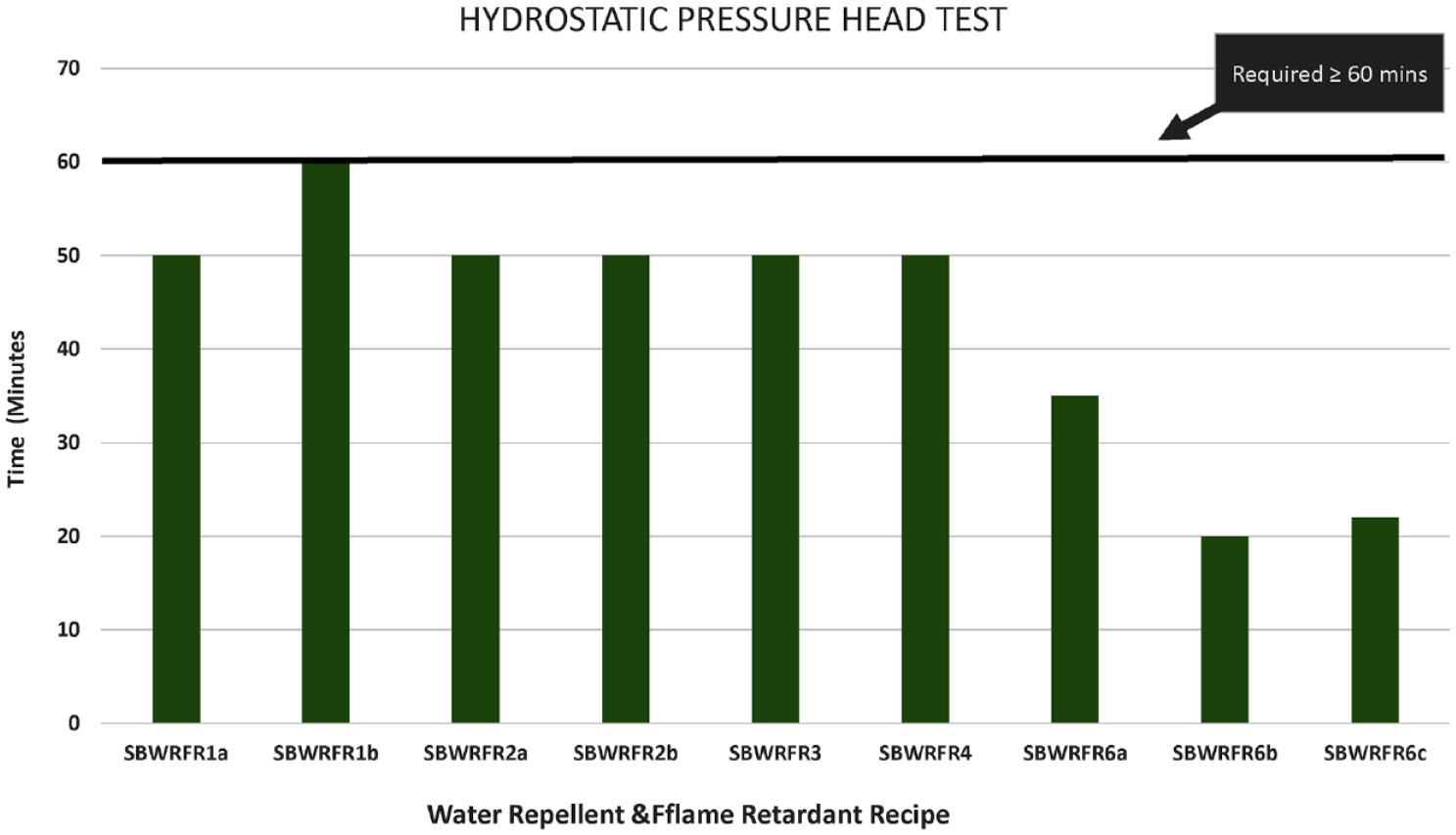
Hydrostatic pressure head test results of water repellent and flame retardant bivouac (SBWRFR).
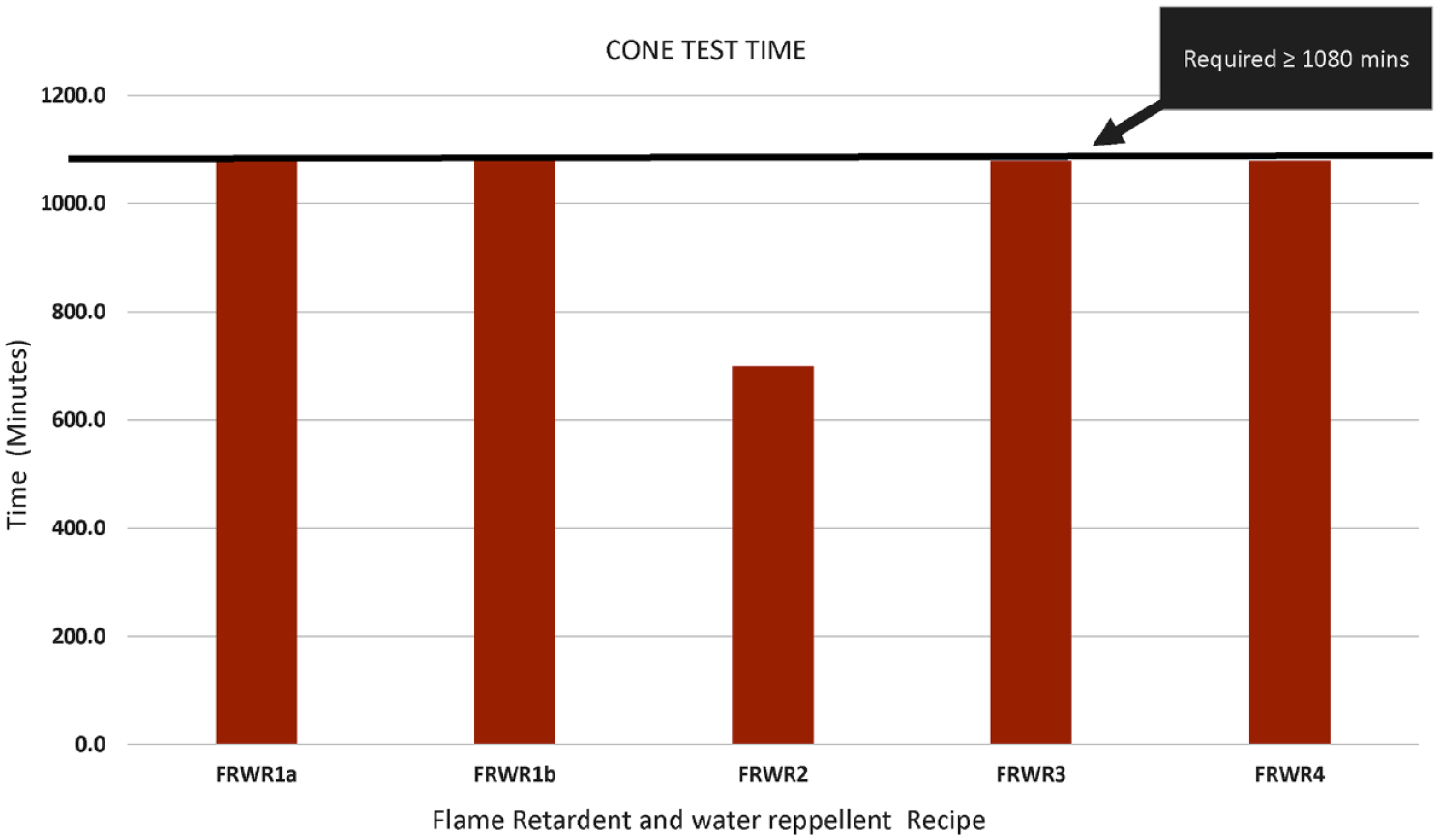
Cone test results of flame retardant and water repellent bivouac (FRWR).
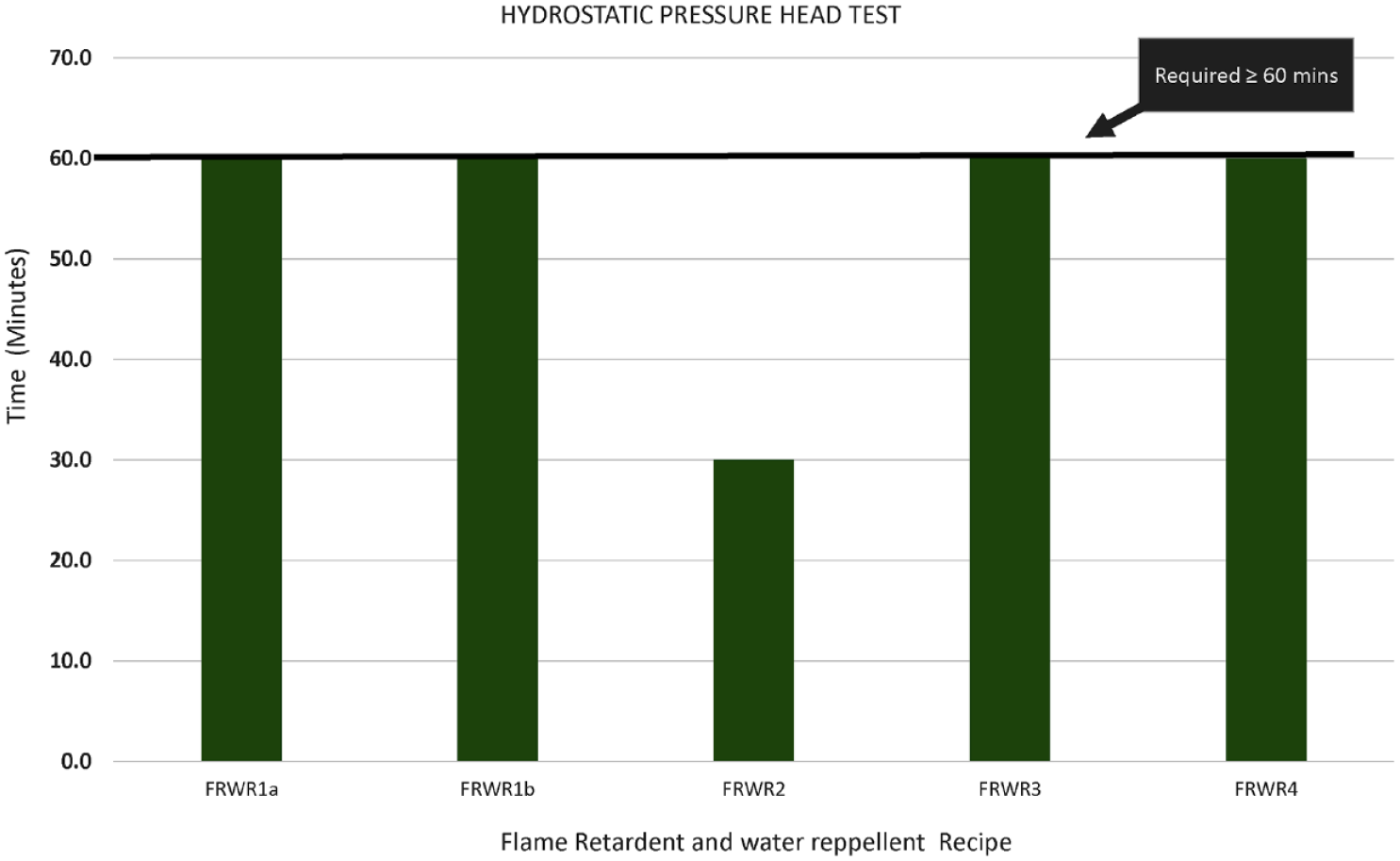
Hydrostatic pressure head test of flame retardant and water repellent bivouac (FRWR).
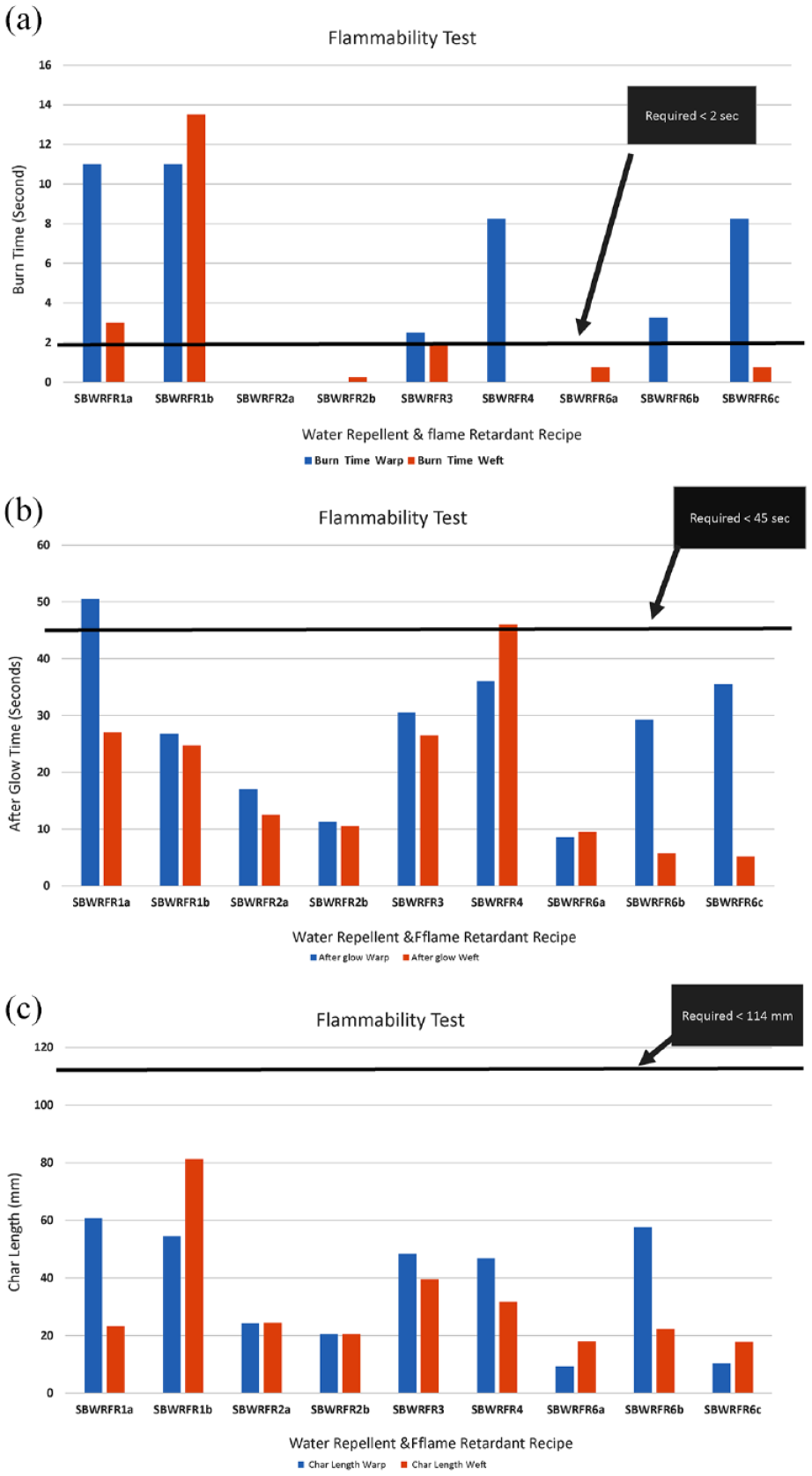
(a) Flammability test (burn time) results of water repellent and flame retardant bivouac. (b) Flammability test (afterglow) results of water repellent and flame retardant bivouac. (c) Flammability test (char length) results of water repellent and flame retardant bivouac.
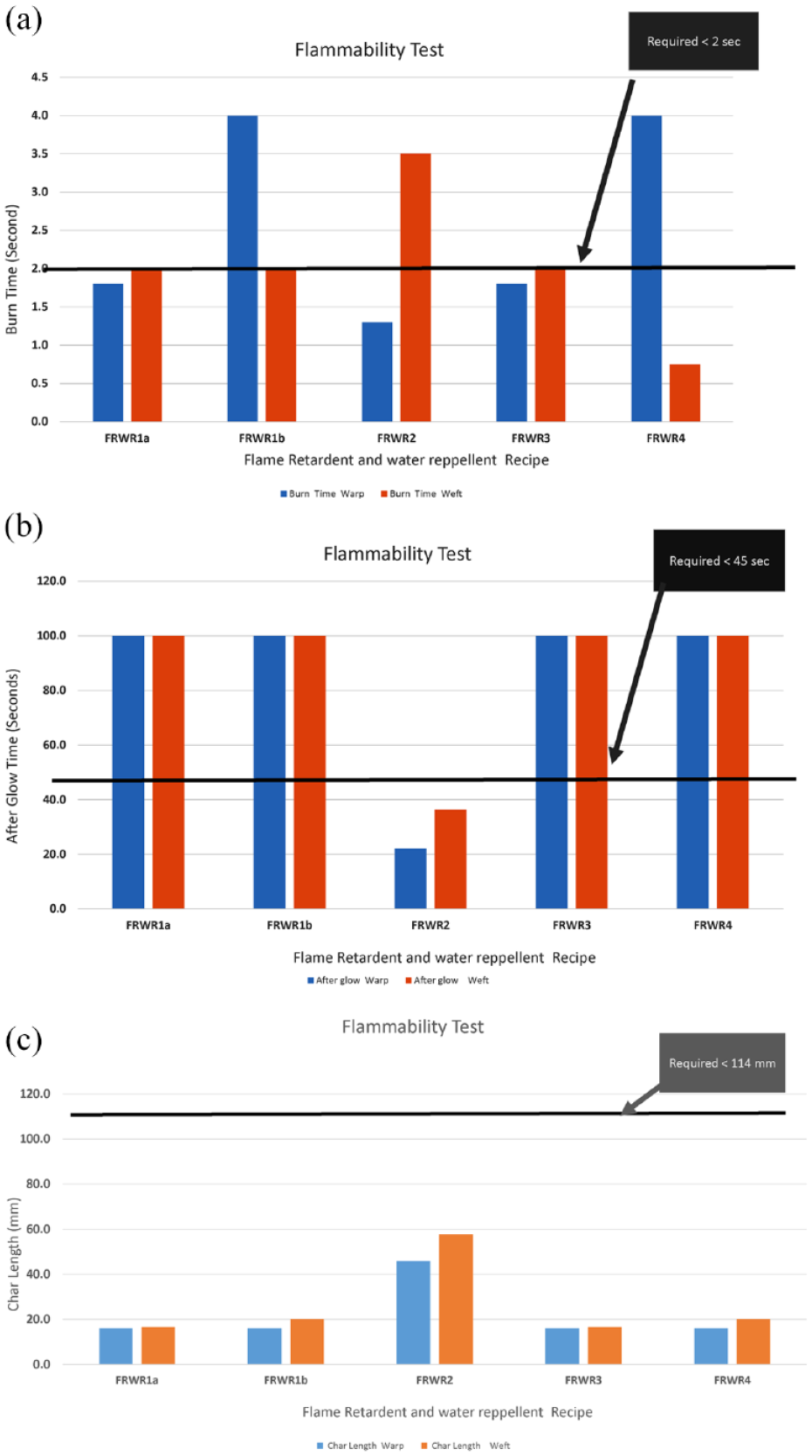
(a) Flammability test (burn time) results of flame retardant and water repellent bivouac. (b) Flammability test (afterglow) results of flame retardant and water repellent bivouac. (c) Flammability test (char length) results of flame retardant and water repellent bivouac.
Discussion
In this study, application of different water repellent and flame retardant chemicals was investigated for the purpose of finding the most suitable water and flame retardant recipe for bivouac fabric.
For imparting water repellency, frequently used repellents such as silicon and fluorocarbon-based water repellent were selected. The durability and price of fluorocarbon-based water repellent are comparatively high. 31 For flame retardency, primary, synergistic and combined retardants were selected.
The screening experiments were conducted to find the performance of locally available water repellent and flame retardant chemicals, so that the best-performed chemicals can be combined. The other reason for screening is to gauge the performance of the finishing chemicals against some specialized test methods such as the cone test and flammability test. Also different water repellent and flame retardant chemicals were used to test the chemistry of each chemical, and their linkages to fabric were different, which might have an effect on the performance of water repellency and flame retardancy. To gauge the water repellency, the finished fabric should pass the mark of 1080 min in the cone test, and 60 min in the hydrostatic pressure test. Similarly in the flammability test, three parameters were selected, namely burn time, char length and afterglow. For the flame retardant chemical to be effective, the burn time should be 2 s or self-extinguished; the char length should be less than 114 mm, and the afterglow time should be 45 s or less. These requirements were set to meet the local client’s protocols.
The screening results for water repellent chemicals are presented in Table 2. It can be seen in Table 2 that five water repellent recipes were applied to the bivouac fabric. These recipes varied by the type of the water repellent from silicon type to fluorocarbon, the type of manufacturer and concentrations. WR1 is a silicon-based water repellent and WR2 to WR5 are fluorocarbon-based water repellents. In WR3, WR4a and WR4b recipes, the chemical is Ruco Guard AFR, which is used as water repellent, but it is evident in the table that WR3 was applied without a binder and failed to give the desired results. Therefore, a cross-linking agent BASF HEL binder TX-4770 was added to WR4a and WR4b, to improve the finish adherence on the fabric. It is evident from Table 2 that WR4b and WR5 are the only recipes that met the requirements of the cone test (1080 min) and hydrostatic pressure head test (60 min). The higher concentration of Ruco Guard AFR (100 g/L) and BASF HEL binder TX-4770 (150 g/L) gave the best results (WR4b).
Similarly, for flame retardantancy of the bivouac fabric, seven basic recipes were selected using different retardant chemicals as presented in Table 3. It can be observed from Table 3 that six recipes, FR1 to FR6, were evaluated on the fabric using different concentrations. The different concentrations were taken as per the supplier recommendations. In FR1, the flame retardant chemical Ruco Flame PCE (organophosphorous based) was used along with the binder Rucon DPO (N-methoxymethyl melamine, non-ionic). The cross-linker was added to have good finish adhesion on the fabric. Three different concentrations of both the chemicals were applied. The concentrations of both the chemicals were increased by around 25% each as per manufacturer recommendation. It is evident in the FR1 recipe (Table 3) that by increasing the concentrations of flame retardant and cross-linker, the burning time decreased. The results were found to be better than the requirements (2 s) in both the warp and weft directions at 400 g/L Ruco Flame PCE and 32 g/L Rucon DPO. The same trend was found for the afterglow and length-of-char results. A similar trend was also found in FR2 to FR6, as shown in Table 3.
Based on screening test results, the best water repellant and flame retardant chemicals were combined, and the finished fabric was investigated for three response variables, namely the cone test, hydrostatic pressure head test and flammability test. Tables 4 and 5 present the result of each response as discussed. 31
Water Repellency
The results of the cone test and hydrostatic pressure head test of SBWRFR are shown in Figures 4 and 5, respectively. Similarly the results of the cone test and hydrostatic pressure head test of FRWR are shown in Figures 6 and 7, respectively. It is evident in Figure 4 that none of the recipes met the requirements of the cone test, that is, 1080 min. However, recipe SBWRFR1b, which is based on a fluorocarbon water repellent and inorganic phosphate salt with nitrogen synergists flame retardant, showed slightly better results than the rest of the recipes and failed after 540 min. Similarly, SBWRFR1b is the only recipe that met the hydrostatic head test requirement of 60 min as shown in Figure 5 from SBWRFR1a to SBWRFR1b; in these recipes, the pickup was reduced from 80% to 60%. The high pick up indicates that a greater amount of finish would be applied, which in turn required proper drying before fixation to avoid migration of the finish. Since the results were not met at the high pickup, the pickup was reduced to analyse the responses. The results improved at reduced pickup, indicating that finish was properly cross-linked with the fibre.
It is shown in Figures 6 and 7 that except for FRWR2, all the other recipes (FRWR1a-1b and FRWR3 to 4) met the cone test and hydrostatic pressure head test requirements. Since the recipes were applied in two steps, this ensured the proper fixation of the finish, hence producing a good quality water repellent bivouac. In FRWR2, the flame retardant used was based on phosphorous, whereas in rest of the recipes, it was based on antimony. The water repellent used in all the recipes was based on fluorocarbon.
Flame Retardency
The flammability test results are presented in Figure 8(a)–(c) for SBWRFR and Figure 9(a)–9(c) for FRWR. It is shown in Figure 8(a) that SBWRFR2a and 2b met the requirement of burn time in both the warp and weft directions, whereas the other failed in either the warp or in weft direction. The recipes SBWRFR2b and SBWRFR6a, presented in Figure 8(b), met the requirement of afterglow. It is exhibited in Figure 8(c) that for char length, all the recipes met the requirements.
For FRWR, the graph in Figure 9(a) shows that FRWR1a FRWR3 are the two recipes, which met the minimum requirement for burn time in both the warp and weft directions. However, it can be noticed from Figure 9(b) that only FRWR2 met the minimum requirement of afterglow. However, it can be observed in Figure 9(c) that all the FRWR recipes met the requirement of length of char.
Conclusion
This study investigated the most suitable water and flame retardant recipe for bivouac fabric by applying different water repellent and flame retardant chemicals. Single bath padding of fluorine compound dispersion (Nuva 1811) along with the combination of inorganic phosphorus salts (Peko flame MSP) provides better results of water repellency and flame retardancy on the cotton canvas fabric (bivouac tent fabric) as compared to the other chemical finishes. The compatibility of fluorine-based water repellent and phosphorus-based flame retardant showed optimum efficacy towards the cotton canvas fabric. Overall, this research provides an economical recipe that provides an opportunity to the local manufacturer to create multi-functional fabric to be used at high altitudes. The introduced hydrophobicity led to considerable wash-durability as the tent fabric is less likely to encounter home laundering. The single-bath water repellent and flame retardant recipe with reduced pick-up percentage (SBWRFR1b) fulfils most of the testing requirements, including hydrostatic water repellent testing and flame retardant testing (afterglow time and char length) on the cotton canvas fabric. However, the results of the cone test (water repellency) and burning time (flame retardancy) were found to be slightly offset from the required values. Nevertheless, the performance result of cone testing was found to be better than other chemical finishes opted in this study. Furthermore, it is also concluded that improvement in burning time for flammability is required for practical applications.
Footnotes
Acknowledgements
The authors express their earnest thanks to ARCHROMA Pakistan and RUDOLF GROUPS for providing the finishing chemicals and auxiliaries.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The research work was supported by NED University of Engineering & Technology.
