Abstract
This study concerns with the preparation of flame retardant and hydrophobic cellulosic fabric by using hexagonal boron nitride nanosheets (h-BNNs). h-BNNs were prepared from hexagonal boron nitride (h-BN) using two different exfoliation methods. These methods include direct sonication (aq-BNNs) and sonication after pretreatment with Hummers method (Hum-BNNs) in aqueous medium. The characterization of h-BNNs was carried out by x-ray diffraction (XRD), Fourier transform infrared spectroscopy (FT-IR), thermogravimetric analysis (TGA), limited oxygen index (LOI), and water contact angle (WCA) analysis. The surface morphologies of h-BNNs were investigated via atomic force microscope (AFM). The coating with the h-BNNs was proved by scanning electron microscopy (SEM). Hummers method is considered to be more effective reaction by gained functionality to h-BN structure. In this way, it will easily provide physical or chemical interaction between the functionalized h-BN and cellulosic structure. A nanometric-sized large layers and slightly functionalized h-BNNs were obtained using Hummers method. Hum-BNNs dispersions were sprayed onto the surface of cellulosic tent fabric to show flame retardance properties. However, it was observed that the flame retardant effect of nanolayered h-BNNs prepared by both methods were insufficient. In addition, ultrahydrophobic surfaces were almost obtained using aq-BNNs and Hum-BNNs. It was conclusively proposed that a few amounts of Hum-BNNs can be used as hydrophobic coating for cellulosic fabric surface with this way.
Introduction
Tent is a place often used for the purposes of social, touristic, sports related events, etc. and it is considered to be a priority for workers and disaster victims. Although tents are made of different fibers such as polyester and PVC, the cellulose constitutes an important part in making them. Many parameters play a pivotal role during the use of a tent. The properties of flame retardance and hydrophobicity are prominent among these parameters in terms of risk factors.
There are several methods to gain flame retardant features to textiles. The prevalent methods to produce flame retardant fabric are using special fibers or applying flame retardant operations to the fabric. The functional fibers are quite limited due to their expensive production costs. Although finishing method is less expensive, it cannot provide satisfactory efficiency, even if it can, it cannot show much resistance to washing, rubbing, etc. Therefore, the studies are progressed about cheaper, more efficient, and durable resistance issues [1–3]. Superhydrophobic surfaces promise a wide range of applications in functional textiles, paints, corrosion inhibition, anti-freezing, etc. Thermal stability and flame retardance are primarily required properties for superhydrophobic materials. However, there are limited studies concerned with producing materials having these features simultaneously [4]. Currently, the functional finishings on textile fabrics are critical importance for improving the multi-functional properties of textile products [5].
Boron nitride has two crystal forms particularly used: hexagonal boron nitride (h-BN) and cubic boron nitride (c-BN). Hexagonal boron nitride nanosheets (h-BNNs) are most stable two-dimensional (2D) crystalline form and is also called graphitic boron nitride because of its similarity to the analog structure of graphene. h-BN has extraordinary properties such as high thermal conductivity, mechanical strength, highly chemical stability, superhydrophobicity, and insulator [6–8], and they make it preferable for certain applications such as surface coatings and composites. Boron and nitrogen atoms are bound by strong covalent bonds within each layer, whereas the weak Van der Waals forces are held together interlayers [9]. Therefore, h-BN can be exfoliated layer-by-layer in several nanometer-sized using proper combinations of solvent, sonication, and centrifugation.
In this work, we present a novel approach related to the preparation of h-BNNs by proposed methods for investigation of the flame retardance and hydrophobic properties onto the surface of cellulosic tent fabrics. The h-BNNs dispersions prepared via these methods were coated on the surface of cellulosic tent fabrics by spraying and obtained almost ultrahydrophobic surfaces.
Experimental
General
All chemicals were purchased from Sigma Aldrich. The unbleached tent fabric (cellulosic 100% - 530 g/m2) and analytical grade h-BN were provided from Turkish Red Crescent and Bortek Company in Turkey, respectively.
Preparation of h-BNNs and applications to cellulosic tent fabric
Preparation of h-BNNs in ultra-pure water (aq-BNNS)
The preparation method was carried out as similar to that of Lin et al. [10]. Briefly, h-BN (100 mg) was exfoliated via sonication in ultra-pure water (100 mL) for 2 and 4 h. The suspension was centrifuged at 400 rpm to remove any aggregated and unexfoliated h-BN. The filtrate (aq-BNNs) was utilized to coat the surface of cellulosic tent fabric using an airbrush spray with 0.3 mm nozzle at 3.0 atm of nitrogen pressure. The suspension of aq-BNNs and cellulosic tent fabric coated with aq-BNNs were kept in a vacuum oven at 60℃ for further characterization processes.
Preparation of h-BNNs via Hummers method (Hum-BNNs)
h-BNNs were efficiently produced from h-BN by the classical Hummers method [11]. Twenty-five milliliters of cooled sulfuric acid were placed into 1 g of boron nitride in a two-necked flask. After carefully adding 3 g of KMnO4, the mixture was stirred in a water bath at 25℃ for 30 min. Forty-five milliliters of deionized water were carefully poured into the mixture. After stirring for 15 min, 150 mL of deionized water and 8.5 mL of 30% (V/V) H2O2 solution were added to finish the oxidation reaction of BN. Metallic contaminants such as Mn ions were removed by washing with 5% HCl solution, and then the precipitate of Hum-BNNs was washed with deionized water and separated by centrifugation at 5000 rpm for 3 min. The washing procedure was repeated until it reached its supernatant pH value which was almost neutral. Hum-BNNs of 1 mg/mL solution were prepared to coat the surface of cellulosic tent fabric using an airbrush spray with 0.3 mm nozzle at 3 atm of nitrogen pressure. The suspension of Hum-BNNs and cellulosic tent fabric coated with Hum-BNNs were kept in a vacuum oven at 60℃ for further characterization processes.
Instrumentation and measurements
The sonication process was applied by Sonorex with 35 kHz of ultrasound frequency (Bandelin Electronic). Fourier transform infrared spectroscopy (FT-IR) spectra were recorded by IR Affinity-1 with ATR attachment (Shimadzu Inc.). The thermal measurements were performed by a DTG-60H Thermogravimetry/Differential Thermal Analyzer (Shimadzu Inc.). X-ray diffraction (XRD) analysis was performed by APD 2000 Pro diffractometer. The layer thicknesses of aq-BNNs and Hum-BNNs were conducted under tapping mode with atomic force microscope (AFM) (Nanosurf Easyscan 2). The scanning electron microscopy (SEM) images were recorded by JSM-6060 LV (JEOL Comp.). For flame retardancy, the limited oxygen index (LOI) measurements were carried out using Dynisco Polymer Test-Limiting Oxygen Index Chamber according to TS 11162 EN ISO 4589-2 [12]. The measurements of water contact angle (WCA) were carried out by DSA 100-optical contact angle meter (KRUSS Comp.). The final value of WCA was determined by averaging the measurements taken from at least three different positions on each sample at ambient conditions.
Results and discussion
The layer thicknesses of aq-BNNs and Hum-BNNs were measured by AFM and the AFM images represent after 2 and 4 h of sonication time (Figures 1 and 2). The layer thicknesses of Hum-BNNs were determined to be between the range of 9–12 and 1.6–5 nm for 2 and 4 h of sonication time, respectively. The corresponding values for aq-BNNs were in the ranges of 6–16 and 3–5 nm for 2 and 4 h of sonication time, respectively. The results showed that the layer thicknesses decrease by increasing the sonication time, and the layers of Hum-BNNs are thinner compared to the layers of aq-BNNs. According to the fact that the thickness of single layer h-BN is 0.33 nm, 5 and 15 number of layers were obtained by Hummers method for 4 h of exfoliation. Besides, 9 and 15 numbers of layers were measured at aq-BNNs for 4 h of exfoliation. Hummers method was found as a more efficient way to reduce the thickness of h-BN layers. Hum-BNNs and aq-BNNs after 4 h sonication process were chosen for coating of cellulosic tent fabric. Molar concentrations of the aq-BN and Hum-BN solutions used in coating process are both 1 mg/mL. Weight gain rate on the fabrics was observed as 21.76% and 21.81% for aq-BNNs and Hum-BNNs, respectively.
AFM images of Hum-BNNs for sonication of 2 h (a) and 4 h (b). AFM images of aq-BNNs for sonication of 2 h (a) and 4 h (b).

Figure 3(a) and (b) shows the characteristic XRD patterns of h-BN and Hum-BNNs, respectively. The diffraction peaks observed at 2θ = 26.8°, 41.6°, 44.1°, 50.2°, and 55.4° correspond to the (002), (100), (101), (102), and (004) of h-BN plane, respectively. The peak observed at 2θ = 38.0° was caused from the sample holder at XRD instrument [13–15]. Figure 3 indicates that the diffraction peaks of Hum-BNNs were slightly shifted in their positions and intensities, which confirm the Hummers method was effective concerned with the functionalization of h-BN.
XRD patterns of h-BN (a) and Hum-BNNs (b).
SEM images of cellulosic fabric uncoated, and coated by Hum-BNNs and aq-BNNs were shown in Figure 4. Figure 4(b) and (c) clearly reveals that Hum-BNNs and aq-BNNs were coated onto the surface of cellulosic fabric.
SEM images of uncoated cellulosic fabric (a), Hum-BNNs coated fabric (b), and aq-BNNs coated fabric (c).
In a similar study, it has been previously reported that cellulosic fabric was coated by a chemical reaction of
The functional groups of h-BN, aq-BNNs, and Hum-BNNs were confirmed by FT-IR spectroscopy in Figure 5. The two characteristic peaks observed at about 1400 and 800 cm−1 related to the sp2-bonded BN (crystalline h-BN or amorphous BN) [17,18]. The corresponding peaks at 1400 and 800 cm−1 in the spectra of h-BN, aq-BNNs, and Hum-BNNs were attributed to in-plane B–N stretching and the out-of-plane B–N–B bending vibrations, respectively. The O–H stretching band at 3200 cm−1 was slightly observed in the zoomed-spectrum of Hum-BNNs which verified to form B–OH and N–H groups by the hydrolysis of B and N atoms at the edge of h-BN [8,10].
FT-IR spectra of h-BN (a), Hum-BNNs (b), and aq-BNNs (c).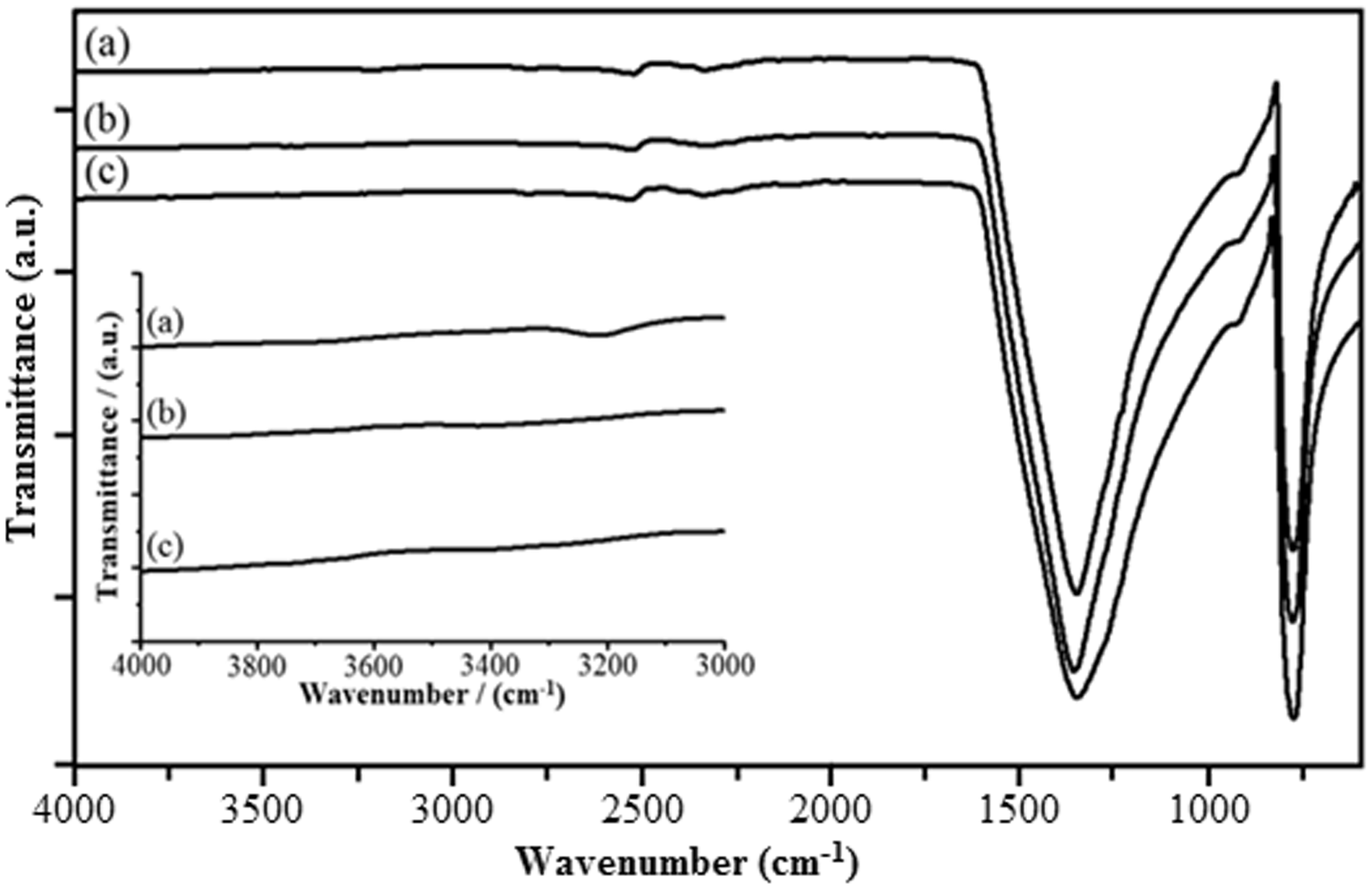
The FT-IR spectra of uncoated, aq-BNNs and Hum-BNNs coated cellulosic fabrics were shown in Figure 6. The broad band at 3330 cm−1 and the band at 2900 cm−1 belong to the characteristic stretching of –OH and aliphatic C–H. All absorption signals of cellulosic fabric were given in the related study [19,20]. In addition, extra two weak signals appeared about at 1370 and 810 cm−1 in cellulosic fabrics coated with aq-BNNs and Hum-BNNs. These peaks were attributed to in-plane and out-of-plane vibrations of h-BN, respectively [21].
FT-IR spectra of uncoated cellulosic fabric (a), Hum-BNNs coated fabric (b), and aq-BNNs coated fabric (c).
The band around at 3300 cm−1 was clearly seen in all spectra of BNNs-coated and uncoated samples in Figure 6. This band is known as belonging to the cellulosic –OH stretching. However, it was thought that the peak around at 3300 cm−1 of Hum-BNNs contributed the signal intensity of –OH stretching in cellulosic fabric coated with Hum-BNNs. The signal at 3250 cm−1 indicates the functionalization of h-BN as suggested in some studies where it has been postulated to occur by additions of hydroxyl groups to B atoms and protons to N atoms [9,11]. According to us, however, the signal should essentially belong to cellulosic OH because the band of Hum-BN at 3200 cm−1 is very weak and questionable. In addition, this peak cannot be seen even in the zoomed spectrum of aq-BN in Figure 5. At the light of these, it seems more realistic to suggest that the aforementioned peak belongs to hydroxyl groups existing in cellulose, but not resulting from the functionalized boron nitride.
Thermal stabilities of h-BN, aq-BNNs, and Hum-BNNs were investigated by TGA. The samples were heated up to 850℃ at a heating rate of 10℃/min in the nitrogen atmosphere.
As seen from TGA curves in Figure 7, the total mass loss of 11%, 15%, and 20% were recorded for aq-BNNs, h-BN, and Hum-BNNs at final temperature, respectively. The data shows that extraordinary thermal stability of h-BN was decreased due to the functionalization by Hummers reaction. The TGA curves of uncoated cellulosic fabric coated by aq-BNNs and Hum-BNNs were shown in Figure 8. The TGA curve of uncoated cellulosic fabric exhibits a minor weight loss (4%) at low temperatures. It is probably due to the removal of moisture content. The onset of decomposition was raised gradually from 250℃. After 300℃, the major decomposition started, and it reached the decomposition transition at 380℃. Although the exact temperature ranges of pyrolysis vary depending on the composition of cellulosic material and experimental conditions, it is carried out at three stages always [22,23].
TGA curves of h-BN, Hum-BNNs, and aq-BNNs. TGA curves of uncoated cellulosic fabric, Hum-BNNs coated fabric, and aq-BNNs coated fabric.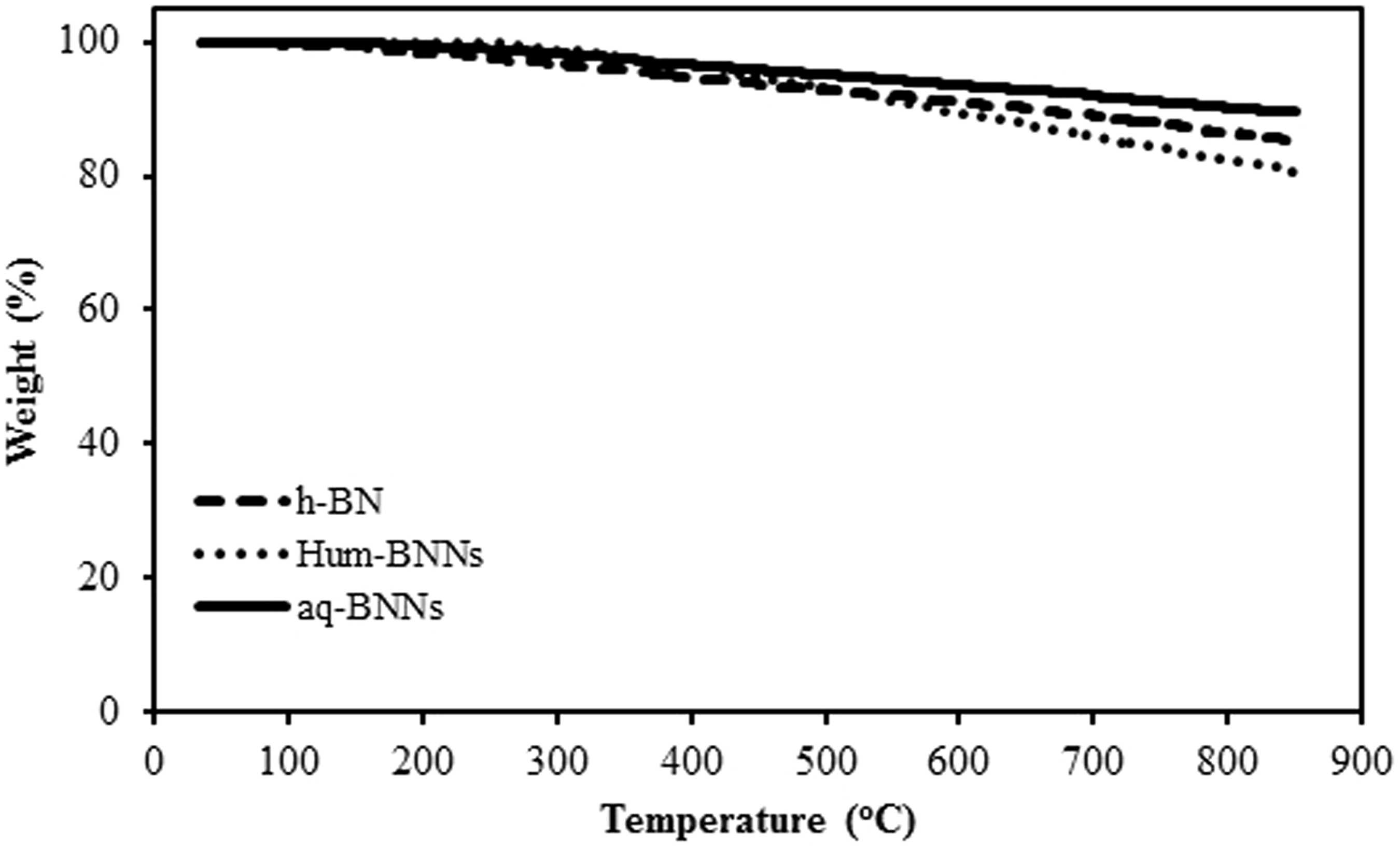

As seen from Figure 8, the decomposition curves of coated Hum-BNNs fabric and coated aq-BNNs fabric were observed to be similar compared with uncoated fabric. The total weight loss of uncoated fabric was found to be 70%, when the temperature reached 380℃. The related values were 73.2% and 79.0% for coated with aq-BNNs and coated with Hum-BNNs, respectively. The losses up to 600℃ were determined as 86.0%, 90.8%, and 93.8% for uncoated cellulosic fabric, coated with aq-BNNs, and Hum-BNNs, respectively. The results show that thermal stability of cellulosic tent fabric was not considerably changed after even applying chosen two processes. In addition, the decomposition of coated samples remarkably accelerated at especially high temperatures. This state may be attributed to the thermal conductivity of h-BN and shows that it may not be used as a flame retardant for cellulosic tent fabric. At this stage, the LOI tests were also performed to determine the flame retardancies of all samples. It was observed that there is no difference in LOI values between coated and uncoated fabrics with the LOI value of 21.0%.
In order to assess the hydrophobicity of the surface, the WCAs were measured on cellulosic tent fabrics uncoated and coated with BNNs. As seen from Figure 9, the WCAs were found to be 118.2° and 108.2° for fabrics coated with Hum-BNNs and coated with aq-BNNs, respectively, but cannot be measured for uncoated fabric due to instant penetration of the water through the fabric surface. These results showed that the hydrophobic surfaces were obtained by coating with BNNs, but uncoated cellulosic fabric is hydrophilic [5]. The difference between coated aq-BNNs and Hum-BNNs fabrics in WCAs may be attributed to functional –OH groups occurred by the high oxidation effect of Hummers’ method.
The water contact angles of uncoated cellulosic fabric (a), aq-BNNs coated fabric (b), and Hum-BNNs coated fabric (c).
A surface is approved as ultrahydrophobic if WCA of water is greater than 120° [24,25]. At the light of these, the surface coated with Hum-BNNs may be assumed as ultrahydrophobic in experimental error limits.
Overview and conclusion
h-BN was exfoliated with two different methods. When the Hummers method was used for a prolonged time of sonication, the thinner sheets were produced. A weak functionalization was observed during exfoliation of h-BN by Hummers method. However, it is a well-known fact that Hummers method is considered as a strong oxidation process and especially used in the synthesis of graphene oxide. When cellulosic fabric was coated with BNNs, its LOI value did not change so much and was insufficient for flame retardancy. However, the surface coated with BNNs, especially Hum-BNNs, showed almost an ultrahydrophobic efficiency. The modification of cellulosic fabric surface with BNNs in water is promising alternative method for providing water repellency.
Footnotes
Acknowledgment
The authors would like to thank the Scientific and Technological Research Council of Turkey (grant no. 112M456) for financial support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
