Abstract
Most effective and durable finishes for cotton fabrics typically contain toxic and sometimes carcinogenic chemicals. Although certain bio-based alternative finishes have been reported in literature but these alternatives are usually less effective than conventional toxic finishes. To improve the performance of bio-based finishes, this paper develops and improves a novel and ecofriendly finishing process for 100% cotton fabric, based on ozone treatments in very controlled amount for the first time. Four types of bio-finishes including citric acid, stearic acid, diammonium hydrogen phosphate and fatty acid amide, for easy care, water repellency, fire retardancy and softness properties respectively were used in this research. For each finish, three ozone-based finishing processes; pre-ozonation, in-situ ozonation, and post-ozonation were developed and optimized. For all four types of bio finishes, fabrics with 20% post ozonation exhibited superior finish performance. Crease recovery angle increased from 121° to 194°, water repellency rating improved from rating 0 to 90, char length decreased from 26.6 cm to 10.8 cm and bending length decreased from 2.16 mm to 1.1 mm by using bio-based finishes with ozone exposure without compromising mechanical and comfort properties of cotton fabrics. SEM, FTIR and XRD analysis were also performed on ozone exposed and finished fabrics.
Introduction
Cotton fibers are abundantly available, natural, and ecofriendly textile raw materials in the world. Despite many advantages, cotton fabrics have certain disadvantages such as high flammability, high hydrophilicity, and easy wrinkling. 1
Flame retardancy of cotton fabrics is a fundamental consumer’s requirement. 2 The limiting oxygen index of cotton fabric is approximately 18%, which classifies it as a combustible material. As a result, it ignites easily and its fire spreads quickly. To prevent the damage from fire, a range of flame retardants have been developed for cotton fabrics. Most effective and stable flame retardants are halogen and formaldehyde-based products. 3 Halogen and formaldehyde-based flame retardants release toxic emissions. 4 Hence, they are under severe scrutiny and propose to be banned soon. Recent halogen-free alternative flame retardants are based on phosphorus and silicon. Phosphorus-based flame retardants, especially commercial Proban and Pyrovatex, release carcinogenic formaldehyde during finishing and consumer use.5,6 Silicon-based flame retardants are less effective for fire retardency. 7 Consequently, scientists are continuously improving the existing flame-retardants or searching for novel and sustainable flame-retardants. Di ammonium hydrogen phosphate has been reported as halogen and formaldehyde free alternative, but its performance is lower than conventional fire retardants. 8
Oil and water repellency in cotton fabrics is another important customer requirement. Conventional oil and water repellents are based on toxic fluorocarbon chemistry. 9 C6 and C8 based fluorocarbon are banned while C4 and lower fluorocarbon are under strict scrutiny and expected to be banned by regulatory agencies. Fluorinated oil and water repellents are the most effective and durable products. 10 In this regard, a high dosage of toxic fluorocarbons is required for the desired level of water and oil repellency. Consequently, literature has reported the ecofriendly alternatives of toxic water and oil repellents, but their performance is much lower than fluorocarbon-based repellents.
Softeners are one of the commonly used finishes for textiles. Silicone based textile softeners imparted superior performance as compared to other softeners. 11 However, silicone softener exhibits high sensitivity for skin irritation, non-biodegradable, and difficult to recycle, thus contributing to environment pollution. While certain bio-based softeners are less effective as compared to silicone softeners.
Resin treatment is another mostly used finishing process in the textile industry for the enhancement of wrinkle-resistance of cotton fabrics. Many resin finishes are available in the markets, but N-methylol compounds, such as dimethylol-dihydroxy-ethylene urea (DMDHEU), are commercially available, most effective, affordable, and durable resin finishes. Unfortunately, it releases a high amount of carcinogenic formaldehyde. While modification of DMDHEU exhibited lower emission of formaldehyde but it also leads to much lower wrinkle resistance as compared to without modified resin. 12 Formaldehyde free, carboxylic acid based crosslinkers are good alternative for formaldehyde-based resins, but their performance is typically lower than the formaldehyde-based resins. 13 Finishing agents based on natural resources require high add-on which results in comfort issues. 14 In addition, application of these bio-based finishing agents does not result in required finishing performance. 15 Consequently, more effective finish application techniques would offer competitive performance.
It can be concluded from the above literature that the most effective and durable flame-retardants, water repellents, softeners and resin finishes contain toxic and carcinogenic chemicals such as formaldehyde and fluorocarbon. 16 While the traditional harmful finishing agents are being banned or they will be gradually phased out with the availability of ecofriendly alternatives. 17 In addition, application of these toxic finishes consumes substantial fresh water and chemicals, resulting in more cost, freshwater depletion, and toxic effluents discharge. Textile processing is constantly seeking cost effective, performance effective, and ecofriendly finish application techniques. In this regard, conventional pad-finishing is a widely used technique in the textile industry, but it consumes considerable amounts of water and chemicals. Substantial use of water and chemicals has enforced the attention of scientists toward the development of efficient and ecofriendly methods. 18 Many advanced finishing technologies have emerged for the reduction in water and chemical consumptions. Recently, foam finishing has been proposed as a more ecofriendly alternative as compared to pad finishing. 19 However, performance of foam finishing is still inferior to conventional finishing. In addition, foam finishing did not reach the level of economies of scale like other advanced finishing techniques. 20 Hence, there is a need for novel, more efficient, and performance effective finishing techniques for cotton fabrics. 21
Ozone molecule (O3) is a very strong oxidizing agent and is made up of three oxygen atoms. It has been reported for both textile and non-textile applications. Air purification techniques utilize this gas, 22 wastewater can be treated by it, 23 while in agriculture sector, soil remediation is carried out by this gas. 24 It is also utilized for cleaning of surfaces 25 and disinfections. 26 Ozone molecule is very unstable and reactive, therefore, when ozone is used at higher intensity and dosages then it leads to breaking apart of pigments and dyes structures. 27 Utilization of optimized ozone in the textile industry can lead to saving chemicals, time and energy, which can enable it to be an environmentally friendly alternative. Aerosol spray technology was combined with ozonation to produce fading out effect by scientists, along with saving of water and chemical consumption. 28
Surface modification has been performed by scientists in literature using low temperature plasma to achieve desired functionalities in textiles. 29 Literature showed application of plasma finish for surface modification of acrylic fabrics for cleaning and waterproofing properties. 30 Surface modification of textile fabrics has been performed by using nanotechnology and UV light source as reported in literature. 31 Low temperature plasma has been used for modification of surface to improve chrome tanning properties of natural leather. 32 Breathability of polypropylene non-woven fabrics has been improved by atmospheric plasma treatment in another study. 33 Jute fabrics have been coated with enzymes for providing ultraviolent protection properties. 34 While in another study in-situ protein enzymes have been formed for improvement of shrinkage resistance of wool fabrics. 35
The application of ozonation is an ecofriendly alternative for many applications. Most prominent application of ozonation for textile is the chemical and color removal from textile products.36,37 A prominent use of ozone treatment is the achievement of special effects in denim garments. Cotton fabric is less active and needs a larger finish quantity to achieve the desired effects. 38 Subsequently, high temperature or higher chemical dosage is required for the achievement of desired performance. A novel and sustainable dyeing process using controlled amount of ozone gas has been reported recentrly. 39 Ozone generates free radicals on the surface of fabrics which activates the surface of fabrics and introduces new reactive points which help in better crosslinking between fabric and finishes. Similarly, it can provide better absorptivity for dyes and finishes. Therefore, ozone when used in controlled amount, can provide better modification of surface which improves functionality and bonding. Use of ozone in color removal needs a high dosage of ozone gas typically 40-60 g/h, which degrades the dyes and finishes. 40 In contrast, this paper uses ozone for finish application in a control manner like 0.5 g/hr to 2.5 g/hr. For the first time, this paper reports the fabric surface activation through controlled ozonation for cotton finish application. This is a challenging problem due to ozone dosage control, ozonation time optimization, and recipe optimization with different finishing agents. The key objective of this paper is to improve the performance of bio-based finishes using ecofriendly ozone treatments on cotton fabric.
Materials and Methods
100% bleached cotton woven fabric having GSM of 155 was selected for finishing in this research. The cotton fabric was procured from Gohar Textile Mills. All the lab. grade chemicals were purchased from Sigma-Aldrich while the conventional finishes were provided by Archroma. Four bio-based finishes were prepared separately. Each finish or chemical percentage reflects the percentage of the chemical or finish on weight of the solution.
Preparation of bio-based easy care finish
For the bio resin finishing recipe, 5% bio citric acid was the main ingredient, and 4% sodium hypophosphite (SHP) was used as catalyst as reported by the researchers. 19
Preparation of bio-based oil and water repellent finish
The bio-based oil and water repellent finishing recipe was prepared by mixing 8 g bio stearic acid, 3.5 g citric acid, with 3 g SHP and heating it for 1 hour at 160°C as reported in the literature. 10 The final recipe was dissolved in 50:50 water and isopropyl alcohol solution and it was diluted and applied onto the fabric.
Preparation of bio-based flame-retardant finish
Similarly, flame retardant finish was prepared by mixing 20% diammonium hydrogen phosphate, 5% bio citric acid, and 4% SHP in aqueous solution of water as reported in literature. 8 This solution was heated for 1 hour at 95°C. Then the final recipe was applied onto the cotton fabric.
Preparation of bio-based softener finish
1% Fatty acid-based bio softener was used as bio softener.
While in the case of conventional versions of the above finishes, all the below conventional finishes were provided by Archroma, and supplier recommended recipes were used. Fixapret ELF (5%) with catalyst of magnesium chloride (1%) were used as conventional resin containing formaldehyde. Pyrovatex CP New (25%) with Knittex CHN catalyst (15%) were used as conventional fire retardant containing formaldehyde and halogen. Phobotex (2%) was used as water repellent. While Siligen (1%) was used as silicone-based softener.
Conventional and bio-based finishing application on fabric
All four finishes of both types, including conventional finishes and bio-based finishes were applied on the fabric samples. Both types of the four finishes were applied onto the cotton fabric separately with lab. scale padding machine as per above recipes having 70% pick up, dried at 100°C for 4 minutes and cured at 170°C for 3 minutes, except for softener finish which was only dried and not cured as per supplier recommendation. These finishes were applied on the samples in the presence and absence of ozone treatment. The samples prepared in the absence of ozone treatment were kept as benchmarked samples.
Three novel ozone-based finishing processes (i.e. pre-ozonation, in-situ ozonation, and post-ozonation) were optimized for each finish. For each process, ozone concentrations of 10% (0.5 g/hour), 20% (1 g/hour), 30% (1.5 g/hour), 40% (2 g/hour) and 50% (2.5 g/hour) were investigated. For ozonation, ozone gas was generated through the ozone generator (model OZ-5G from PurePro Ltd) by using corona discharge technology with the capacity to produce 5 g/hour of ozone. The ozone generator has a power of 80W and frequency of 4 kHz. The ozone generator has the ozone control knob to control the percentage of the output ozone. Ozone treatment time was optimized before experimentation. When the ozone exposure time for the cotton fabric is increased beyond 10 minutes then there is only minor change in absorbency and wicking length. While there was greater loss of tensile strength due to longer exposure time of the cotton fabric with ozone. In addition, it would have led to more cost and slow production speed. Therefore, it was identified that 10 minutes of exposure offered the best results. Hence, ozone treatment time was fixed at 10 minutes for the whole experimentation. In the pre-ozonation, untreated fabric was exposed to ozone in a closed glass chamber, and then each finishing recipe was applied through padding machine. In the in-situ ozonation, each finishing recipe was exposed to ozone in the recipe solution, and then the fabric sample was padded with the recipe as exhibited in Figure 1. In the post-ozonation, each finishing recipe was applied on the fabric, and then the resultant fabric was dried. Then, this fabric was treated with ozone in a closed glass chamber. Conventional and finishing with ozonation at various stages.
Analysis and evaluation
Testing was carried out for the evaluation of each sample and for each treatment including untreated, conventionally treated and ozone exposed finished fabric. For resin finishing, crease recovery angle was determined with the help of standard test method AATCC 66 and appearance of the fabric after repeated home laundering was assessed by AATCC 124-1996. Spray test (AATCC 22), aqueous liquid repellency (AATCC 193), and oil repellency (AATCC 118) standard test were adopted to evaluate the fabric desire performance. Flame retardancy was assessed by vertical burning test by following BS 5438:1989, Test 2B and limiting oxygen index test was performed by following ASTM D 2863 method. Softener treated fabric softness was assessed by assessing bending length as per ASTM-D1388 standard test method. Absorbency was measured by AATCC 79 standard test method. XRD analysis was performed on XRD (Bruker D8) by using advanced step scan, step size 0.02° with time per step is less than 0.2 sec. FTIR analysis was performed on FTIR machine by Tensor 27-Bruker. Moreover, SEM analysis was performed using scanning electron microscope (model AIS 1800 C). Air permeability retention (percentage) was assessed by ASTM D737 test method using air permeability apparatus. In addition, tensile strength was measured using BS EN ISO 13934-1 standard test method.
Results and discussion
Bio and conventional resin finishing for crease recovery property
Conventional (DMDHEU) and bio based (citric acid) cross linkers were applied onto fabrics for achieving crease recovery properties. The crease recovery properties were evaluated by the measurement of crease recovery angle. Crease recovery finishes work on the mechanism of crosslinking of cellulose chains with covalent bond and that will lead to better crease recovery angle and appearance of fabric after washing. Figure 2 presents the crease recovery angle of the different samples. By the application of easy-care finish, crease recovery angle increased. Samples treated with citric acid provided better results as compared to DMDHEU. It could be due to three carboxylic acid groups present in citric acid which might have provided better crosslinking as compared to DMDHEU finish. Similar kinds of results are reported in literature.
41
The crease recovery angle of the finished benchmarked samples (i.e. without ozonation) for both finishes type (DMDHEU based and bio citric acid) was less than ozone-treated samples. It could be due to the reason that control ozone exposure generated the free radicals, which enhanced and accelerated the reaction of functional group of the finish with cotton fabric. As a result, more cross-linking occurred leading to the better crease recovery angle. Similar free radical generation and improvement in crease recovery angle for carboxylic acids on cotton fabric has been reported in case of only low dosage of 0.1% nano titanium oxide in literature.
40
In pre-ozonation, ozone activated the fabric surface. For this purpose, free radicals of the ozone activated the fabric surface through the generation of active sites on fabric surface. This phenomenon resulted in the attachment of more molecules of textile finish to the fabric surface. In post ozonation, ozone offered the better fixation of already applied finish on the cotton fabric. Recently, dyeing with direct and reactive dyes on cotton fabric and then exposure of the dye treated fabric using lower dosage of ozone at post ozonation stage exhibited improvement in shade depth.
39
Subsequently, it further clarifies the positive impact of controlled ozone on the treated cotton fabric. Overall, the samples treated with 20% ozone concentration resulted in superior crease recovery angle for all the three ozonation processes. Improvement in crease recovery angle from 121° to 194° could be seen as compared to benchmark samples by using bio citric acid. DMDHEU provided a crease recovery angle of 186° using the same concentration of ozone. It could be due to the reason that initially free radicals will support positively in the post finishing process but later excessive ozone as well as subsequent higher free radicals leads to destruction or competition with the finish. In literature, a similar pattern was reported by the researchers in the case of nano titanium oxide application with citric acid, where nano titanium oxide dosage was increased by more than 0.1%.
26
In the post-ozonation process, the samples treated with 20% ozone concentration exhibited best results as compared with all other samples. Crease recovery angle for different samples and ozonation processes.
Durable press rating for different samples with and without ozonation.

Bio and conventional water repellent finishing
Oil and water repellency rating for different samples with and without ozonation.

Figure 3 presents the results of the water repellency by shower test for the different samples. In the case of pre-ozonation and post-ozonation, water repellency was improved by one level from 70 to 80 as compared to without ozonation. According to the rating scale, water repellency of value 70 interprets that all the samples were only partially wet beyond the spray points and only upper surface. In pre and post ozonation, ozone offered the comparatively better results of the water repellent finishes for the sample treated with 20% ozone concentration due to ozone free radical generation and activation of recipe carboxylic acids. In-situ ozonation offered the comparatively less performance for the 20% ozone concentrations mainly due to possible hindrance of the free radicals as well as the competition with the finish in liquid form. Overall, the samples treated with 20% ozone concentration resulted in better water repellency for post-ozonation in case of bio-based finish of stearic acid as well as conventional phobotex finish. Water repellency by spray test rating for different samples and ozonation processes.
Bio and conventional fire-retardant finishing
Cotton fabric exhibits no fire retardancy without the application of fire retardant as it is inherently non-fire-retardant fabric and catches fire easily.
43
Therefore, cotton fabric was completely burnt when tested for char length as per standard, thus reflecting the need of the fire-retardant finishing for cotton fabric. In this research nontoxic, halogen and formaldehyde free fire retardant named DAHP in Figure 4 was applied onto the cotton fabric. Since the fire-retardant recipe includes the carboxylic acid based citric acid, improvement through ozonation was expected as it was the case for resin as well as oil and water repellent finishing due to free radical generation by ozone. Figure 4 presents the char length of the different samples. Char length is the length of burned fabric in flame retardancy test. The lesser char length reflects the better flame retardancy. The samples treated with 20% and 30% ozone concentrations exhibited better flame retardancy than other samples and it is true for bio-based fire retardant as well as conventional (halogen and formaldehyde based) fire retardant. The samples treated with 20% ozone concentration exhibited slightly better flame retardancy than the samples treated with 30% ozone concentration. Char length for different samples and ozonation processes.
Limiting oxygen Index value for different samples with and without ozonation.

Bio and silicone softener finishing
Softener finish is one of the most widely used finishes for textiles.
44
Softener application onto textile leads to lower bending length which reflects softer fabric due to better lubrication of the cellulose chains. Silicone softener typically exhibited superior softness as compared to other softeners, but they are not biodegradable. While bio based fatty acid softener is an eco-friendly option. Ozone exposure imparted improvement in treated fabric softness by exhibiting lower bending length for both the silicone softener as well as fatty acid amide softener when exposed to ozone as shown in Figure 5. Highest softness was reflected by the lowest bending length of 1.1 mm, which is exhibited by bio based fatty acid softener treated fabric for 20% post ozonation process and this trend is in line with above finishes results. Bending length for different samples and ozonation processes.
Absorbency and wicking
Ozonation process for textile can lead to surface cleaning and removal of foreign matters on the fabric surface. Therefore, ozone exposure to the cotton fabric without any finish at different percentages of ozone was accessed and absorbency as well as wicking length of exposed fabric was measured in this research paper. It is quite clear from Figure 6 that average absorbency time is reduced when cotton fabric is exposed with 0% to 30% ozone from 7.2 seconds to 6.3 seconds respectively. Similarly, wicking length is increased from 5.7 to 9 cm when cotton fabric is exposed with 0% to 30% ozone. The decreasing trend of absorbency time and increasing trend of the wicking length on increasing the ozone % clearly reflect that ozone has imparted fabric surface activation and better functionalization. Therefore, the positive impact of the ozone on the finished fabrics mentioned in earlier sections could be due to the better surface functionalization and creating better conditions for finish and cotton fabric. Average absorbency and wicking length for different samples and ozonation processes.
Tensile strength retention
Ozone exposure at higher percentages can lead to decrease in tensile strength of the exposed fabric. Therefore, tensile strength of the ozone exposed, and non-ozone exposed finished fabric was assessed. Tensile strength retention for untreated sample was considered as 100% for each finish. However, untreated sample does not offer any other property like better crease recovery angle, flame retardancy, water repellency and better softness. For these performance tests, samples treated with 20% ozone concentration were identified as the best sample. Hence, the tensile strength testing was conducted for the best samples treated with 20% ozone concentration as well as the tensile strength retention was measured for the benchmark samples without ozonation process. Decrease in tensile strength during cotton fabric finishing is expected due to acidic degradation of cotton as typically finishes are applied at acidic pH. Secondly, bonding as well cross-linking due to finish application and curing will make the cotton fabric more rigid, thus leading to lower tensile strength of the treated fabric. While ozone exposure also decreased the strength of the fabric, this decrease was only 3% to 5% in this research for various finishes as compared to the finished and non-ozone exposed fabric as shown in Figure 7. This decrease in the strength of the fabric is comparatively less due to the less and controlled exposure of the ozone on the finished fabric. Overall, the decrease in tensile strength of the treated fabric for any finish was in acceptable range with ozone exposure, thus reflecting the suitability of the controlled and lower ozone dosage process for the textile finishing. Tensile strength retention of different samples and post ozonation processes.
Air permeability retention
Fabric comfort properties are crucial performance parameters. Typically, application of finish coating leads to lower air permeability. As 20% post ozonation process demonstrated the superior crease recovery, fire retardancy, softness and water repellency, therefore, it was selected for the further comparisons. There was only a 2% air permeability difference in the case of resin and fire-retardant finish samples with 20% post ozonation, 1%–2% for the water repellent and 2%–3% for softener treated and ozone exposed samples as compared to finish treated and non-ozone samples as shown in Figure 8. It clearly reflects that ozone only imparted negligible negative impact on the air permeability and comfort propertied of they treated fabric. Air permeability retention of different samples and post ozonation processes.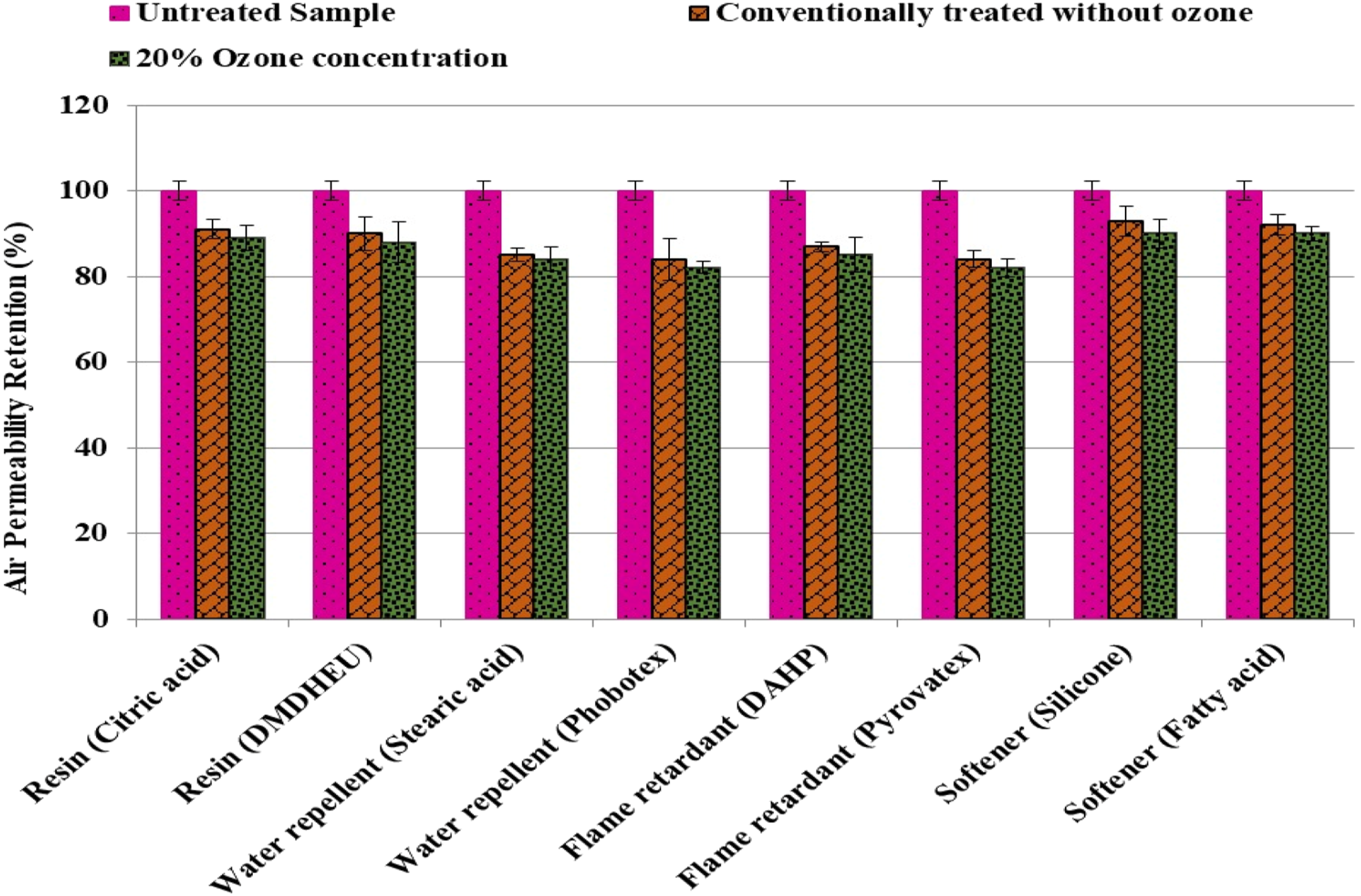
SEM analysis
Figure 9 presents the scanning electron microscope (SEM) analysis of the different samples. Untreated samples did not show any application of a finish as expected which is shown in Figure 9(a). Only water repellent finished cotton fabric as sample representative of different finished fabric and untreated cotton fabric was selected for the SEM analysis. A water repellent finished sample but without ozonation indicated moderate level of finish and cross-linking of a finish on fabric surface as shown in Figure 9(b). Samples finished with bio based and conventional water repellent finish i.e. stearic acid and phobotex and exposed to 20% post ozonation exhibited more coating and presence of finish on the fabric surface as compared to non-ozone fabric surface as shown in Figures 9(c) and 9(d). It clearly supports the superior performance of the ozone exposed and finish treated samples as compared to non-treated as well as finish treated but non ozone exposed samples. SEM analysis of cotton fabric samples treated with water repellent finish and exposed to post ozonation.
XRD analysis
In X-ray diffraction (XRD) analysis, higher intensity peaks for the softener, water repellent, fire retardant and resin finished cotton fabrics were observed as compared to untreated cotton fabric, Figure 10. Nevertheless, ozone exposed and finished cotton fabric demonstrated even higher intensity peaks in case of all four finishes than both untreated cotton fabric and respective only finished cotton fabric. It clearly reflects the effectiveness of the ozone exposure for all the finished cotton fabrics, and it can also supports the superior performance of the ozone exposed fabric for various finishes. X-ray diffraction (XRD) analysis of untreated cotton fabric, conventionally finished fabric and finished fabric with 20% post ozonation process was carried out. Nevertheless, cotton fabric is composed of 70% crystalline region with cellulose I crystal. Characteristic peaks of cellulose present in cotton fabrics were depicted by peaks at 2θ of 14.8° to 16.5°, 22.7° and 34.5°, which were the characteristic peaks of cellulose I crystalline form. These results are exhibited in Figure 10 and in line with previously reported literature.
45
Peak related to carboxylic acid was observed at 2θ of 16°, 22° and 32°, which is also in line with earlier reported literature.46,47 XRD analysis of untreated, different finish treated and post ozonated cotton samples.
Fourier-transform infrared spectroscopy (FTIR)
FTIR spectra of untreated cotton fabric and fabric treated with four types of finishes with ozone treatment is shown in Figure 11. Cotton fabrics exhibited a 1313 cm−1 band associated with the bending vibration mode of hydrocarbon structures, as well as 1354 and 1459 cm−1 bands associated with symmetric stretching of carboxylates, which originate from cellulosic molecules. The broad peak at around 3300 cm−1 was due to the surface hydroxyl groups (–OH) present on the cellulose chains of the cotton fabrics. The peaks at 2900 cm−1, 1314 cm−1 and 1036 cm−1 were due to C–H stretching vibrations of the cellulose alkyl chains, C–O, C–H bending vibrations, and C–O, O–H stretching vibrations of the polysaccharide in the cotton fabrics respectively. The greater peak intensity at above bands especially when finished fabric was exposed to ozone reflected the greater presence of the finish on the treated cotton fabric surface and hence the superior relative finish performance was achieved as described in previous sections. FTIR analysis of untreated, different finish treated and post ozonated cotton samples.
In the case of bio-based easy-care finishes i.e. citric acid, characteristic peaks were exhibited in FTIR analysis. A peak at 652 cm−1 is related to the rocking of CH2 group. The peak at 1021 cm−1, exhibited the indication of C-OH group. Characteristic peak of stretching of carboxylic acid (R-C(O)-OH) group was identified at around 2905 cm−1 and 3328 cm−1. In the case of DMDHEU, peaks appeared at wavenumber of 1026 cm−1 which was the indication of C-O stretching of ether group. The peak at wavenumber of 2898 cm−1 was the indication of C-H stretching of methyl group, while the peak at wavenumber of 3280 cm−1 was related to N-H stretching, which was related to linkage of urea.
By using silicone-based softeners, a strong peak at wavenumber of 1030 cm−1 was achieved which was the indication of siloxane (Si-O-Si) bonds which were present in the backbone of polymers with additional peak in the region of 780 cm−1. Si-CH₃ groups depicted a peak at around wavenumber of 1313 cm−1 which was the indication of stretching and bending vibrations of this group, which were common in polydimethylsiloxane. Symmetric and asymmetric C-H stretching vibrations of methyl groups were represented by peak at 2901 cm−1, while silanol groups were represented by peak at 3332 cm−1. In the case of fatty acid amide softeners, symmetric and asymmetric C-H stretching in the hydrocarbon chains were represented by peaks around 2890 cm−1, which were the indication of long alkyl chain presence in the fatty acid amide softener. Primary or weak secondary amides were represented by peak around 3298 cm−1, which were due to the N-H stretching vibrations.
FTIR analysis of oil and water repellent finishes i.e. stearic acid and Phobotex was carried out and it was found out that in case of stearic acid spectra, C-O bonds stretching of COOH groups was indicated by peak around 1025 cm−1. The strong peak which is characteristic (C = O) stretching of the carbonyl in the carboxylic acid group is represented at the wavenumber of 1699 cm−1. Another peak, which indicated stretching of O-H group of carboxy groups was represented at the wavenumber of around 2916 cm−1. Phobotex, a fluorocarbon-based oil and water repellent finish represented a peak at the wavenumber of 1028 cm−1, which was the indication of C-F stretching in fluorocarbon chain. A peak at 2899 cm−1 represented the symmetric and asymmetric stretching of C-H in the alkyl chains.
In case of DAHP finish, broad peaks, indicative of stretching of P-O bonds characteristic of phosphate group is represented at wavenumber of 935 cm−1. Peak at around 1680 cm−1 represented bending vibrations of N-H bonds, which are present in ammonium groups. Peak at 3335 cm−1 represented stretching vibrations of the N-H bonds in the ammonium groups. FTIR spectra of pyrovatex represented a peak at wavenumber of 1026 cm−1, which indicated P-O bond stretching which is present in phosphate-based fire retardants. At wavenumber of 2900 cm−1, peak appeared which indicated symmetric and asymmetric stretching of C-H bonds which were present in the chains of hydrocarbons. A peak at 3335 cm−1 represented stretching vibrations of N-H bonds present in the amide functional groups.
Ozonation is a cost-effective procedure as compared to conventional vacuum plasma and other similar surface activation techniques. In current research, cost effective and small ozone generator is proposed to be used as only attachment with existing textile application machines. Due to the small attachment of ozone generator with significant performance advantages, it can be aligned with existing machines leading to better scalability and industrial viability. Figure 12 exhibited the sample images of the cotton fabric treated with all four finishes including conventional and bio-based finishes at three levels of ozone and without ozone exposed fabrics. There was hardly any difference in appearance of the samples. Images of finished samples.
Conclusion
This work has successfully applied the novel controlled ozone treatment on bio-based and conventional finishes to improve the crease recovery angle, water repellency, softness and flame-retardancy of the cotton fabric. During the comparison of three processes, i.e. pre ozonation, in-situ ozonation and post ozonation, it was found that best results were achieved by 20% concentration of ozone in post ozonation process in terms of crease recovery angle, flame-retardancy, softness and water repellency. It could be because ozonation leads to better surface functionalization, greater free radical generation and activation thus imparting improvement in the conventional and bio-based finished fabric performance. Among the three stages of ozonation; pre-ozonation, in-situ ozonation and post ozonation, all four finished fabrics in the case of conventional and bio-based finishes exhibited superior performance with the post ozonation process. In the case of bio resin finish application, the highest crease recovery angle of 194° was achieved for 20% post ozonation process as compared to the low crease recovery angle of 168° for without ozonation and 121° for non-resin finished cotton fabric. Water repellency (shower test) rating of 90 was exhibited for 20% ozonation of water repellent finished fabric whether treated with bio stearic acid or conventional phobotex as compared to the rating of 70 and 0 for non-ozonated bio finished fabric and non-finished fabric respectively. While the best fire retardancy performance in terms of lowest char length of 10.4 cm and highest limiting oxygen index value of 26.6 was achieved at 20% post ozonation process of bio based DAHP based fire-retardant finish treated fabric. There is a negligible decrease of about 3%–5% in tensile strength and 1%–3% in air permeability for the 20% ozone exposed finished fabric as compared to any performance finished fabric without ozone. It clearly reflects that ozone did not impart any major negative effect on strength and air permeability due to controlled exposure of ozone. Ozone exposure also leads to a decrease in the absorbency time and increase in the wicking length thus reflecting the exposed surface functionalization and activation. SEM analysis showed the successful application of the bio and conventional finishes on the cotton fabric surface. Greater surface coating on the finished and ozone exposed fabric as compared to non-ozone fabric reflected the effectiveness of the control ozone exposure. XRD and FTIR analysis also confirmed the more finish presence on fabric when exposed to 20% ozone as compared to non-ozone and untreated fabric. Typically, a greater quantity of expensive and toxic conventional functional finishes is required for effective textile performance. However, considering the benefits of the control exposure of ozone especially for bio-based finishes, this novel process can provide a suitable and effective alternative for eco-friendly finishes and processes.
Footnotes
Declaration of conflicting interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
