Abstract
Nylon fibers were the first synthetic fibers to be produced on an industrial scale, and they are one of the most consumed synthetic fibers due to their visual and usage properties. Reactive dyes have been commercially produced since the mid-20th century, and they are mostly preferred in dyeing cellulosic fibers. The most important property of reactive dyes is their ability to make covalent bonds within the fiber. Mordants are used mainly in the application of natural dyes to hold and retain the dye molecules in the fiber. In this paper, nylon fabrics were dyed with three reactive dyes having different reactive groups (monochlorotiazine, trifluoropyrimidine and monochlorotriazine/vinylsulfone) in the presence of three chemical mordants (iron (II) sulfate, potassium aluminum sulfate and tannic acid) to obtain better exhaustion and coloration properties. Pre-mordanting, meta-mordanting, and post-mordanting processes were used for dyeing. The dyeings were performed at two dyeing concentrations (0.5% and 2% owf), and at three pH levels (pH 2, 4, and 6). The percentage of dyebath exhaustion (%E), the percentage fixation of the dye which exhausted (%F) and the overall percentage fixation (%T) were calculated after pyridine extraction. Wash and light fastness tests were applied on the dyed samples. The results were discussed via %
Introduction
In 1938, Du Pont publicly announced the discovery of “super-polyamides” to which the general term nylon had been ascribed; nylon 6,6, the first commercially available wholly synthetic textile fiber, was marketed in 1939. The term nylon now refers to members of a generic class of synthetic polyamides derived predominantly from aliphatic monomers. Polyamides are polymers whose structural units contain amide groups (—CONH—) as recurring integral parts of the main polymer chain. Although a variety of polyamides are now manufactured and are marketed under various trade names, the two most important nylon textile fibers are nylon 6,6 and nylon 6. 1 Nylon 6,6 fibers, spun from polyamide polymer poly-hexamethylene-diamine-adipate, have been always required to be dyed in deep shades of high wash fastness. 2
The presence of terminal amino groups in nylon fibers imparts substantivity toward ionic dyes, especially acid dyes, direct dyes and reactive dyes. Of the various types of dye that can be used to dye nylon fibers, acid dyes are the most popular, with 1:2 pre-metallized acid dyes being used when high wet fastness is required.1,3,4 The coloration of nylon is usually achieved with acid dyes. 5 These attack the nylon via ion–ion linkages between cationic, protonated, amino end groups of the nylon (NH3+) and the anionic sulfonate residue of the dye (Dye-SO).3–6
In the case of dyes for cellulosic fibers, high wet fastness has most often been achieved by the use of fiber-reactive dyes: these function by reacting chemically with the cellulose, thus forming covalent links between fiber and dye.5,7–9
Reactive dyes for cellulose resemble acid dyes in their basic structure, but additionally possess one or more fiber-reactive groups, their name being derived from their ability to react chemically with groups on the fiber. In the mid-1950s Rattee and Stephen showed that dyes containing a dichlorotriazinyl reactive group are capable of reacting with cellulosic fibers, in the presence of alkali, to form covalent dye to fiber bonds. Subsequently, research into the chemistry and the application of reactive dye focused overwhelmingly on cellulosic fibers.4,7,8,10 Considerably less attention has been centered on reactive dyes for polyamide fibers. Thus, compared with the vast number of reactive dyes for cellulose, relatively few reactive dyes have been introduced specifically for polyamide fibers. 11 It is known that some reactive dyes which were developed for cellulosic fibers can be covalently fixed at the boil to nylon at slightly acidic pH. Under these conditions, covalent bonds form between the dye and the amino groups of nylon, without the need for an alkaline fixation step. 4
The most noticeable commercial ranges of reactive dyes for nylon have been reported as the Stanalan and Erifast dyes.4,5 Other water soluble dyes possessing electrophilic reactive groups such as heterobifunctional (MCT/VS), 3 chlorofluoropyrimidies, 12 α-bromoacrylamido dyes, 13 chlorotriazines and vinylsulfones, 14 as well as some reactive disperse dyes,15,16 have also been evaluated.
Although acid and metal-complex (premetallized) dyes are mostly preferred for the coloration of nylon fibers, dyeing nylon fibers with reactive dyes holds an alternative class of dyes having the possibility of strong dye–fiber bonds which could lead to higher fastness properties, for example, especially wash fastness.
Lewis and MacDougall researched dyeing nylon 6,6 with vinyl sulfone reactive dyes and revealed that only about 30% of the terminal amino groups were available for covalent reaction with the dye and concluded that fiber crystallinity and build-up of fixed negative charges were responsible for the effect. 2
Soleimani-Gorgani and Taylor focused on different dyeing properties of nylon with reactive dyes and concluded that fixation levels appeared to be independent of the number of either reactive groups or chromophore units and the major determination of dye fixation appeared to be associated with the shape of the molecule and the type, not the number, of reactive groups; 4 covalent bond fixation of the four derived monofunctional reactive monochlorortriazinyl dyes, differing only in degree of sulfonation, increased with increasing levels of sulfonation but their fixation efficiency reduced with increasing concentration of dye applied; 5 one or two cationic groups including dyes with different reactive systems were synthesized and they all fixed efficiently to nylon under alkaline conditions with fixation and build-up being fully comparable to markets’ leading anionic reactive dyes. 17
Burkinshaw and Gandhi researched the dyeing of conventional decitex and microfiber nylon 6,6 with reactive dyes and concluded that commercial chlorodifluoroprymidinyl reactive dyes were found to yield optimum color yield under acidic conditions (pH 4), and good build-up and level dyeings were obtained on both fabrics; 12 only one of the three commercial α-bromoacrylamide reactive dyes displayed good build-up on both conventional decitex and microfiber nylon 6,6 fabrics at pH 4, but it was observed that a large proportion of adsorbed dye was unfixed and removed by wash-off. 13
Burkinshaw and Wills researched the optimum pH value for dyeing nylon 6,6 with vinyl sulfone and chlorotriazine dyes and concluded that it lay between pH 5 and pH 6 where good exhaustion and high fixation were achieved. However, the results implied that the dyeing behavior of the dyes was pH-dependent and also varied between dyes. 14 Two of the studies12,13 were performed in the absence of electrolyte and proprietary leveling agent, and generally syntan treatment did not improve the wash fastness of the dyeings.
Kim et al. 18 researched the dyeing characteristics of nylon, cotton and N/C (nylon/cotton) mixture fabrics with reactive-disperse dyes containing a sulfatoethylsulfone group and concluded that washing fastness and rubbing fastness of dyed nylon, cotton and N/C fabrics with the reactive-disperse dyes were excellent, while light fastness was moderate. Kim et al. 19 (the same research team) also researched dyeing properties of nylon, polyethylene terephthalate (PET), and N/P (nylon/polyester) mixture fabric with reactive-disperse dyes having a sulfatoethylsulfone group and concluded that the reactive-disperse dyes were found to be adequate for the one-bath, one-step dyeing of N/P mixture fabric when applied at pH 5 and 120ο.
Baig et al. evaluated the effect of three organic salts on the dyeing of cotton fabric using four reactive dyes. They studied the effect of organic mordants on different dyeing parameters such as fastness properties, color yield and K/S value and concluded that the organic salts including sodium citrate, potassium acetate and ammonium acetate showed comparable fastness properties to those obtained in the case of conventional salts. Out of these three salts sodium citrate showed better fastness properties as compared to other salts, and organic salts showed higher K/S values when lower concentrations were applied. 20
Hosen et al. utilized turmeric (
Repon at al. compared the dyeing properties of cotton knitted fabric dyed with banana floras stem (BFS) sap and reactive dye. The specimens dyed with BFS sap showed excellent color level and durability characteristics similar to the reactive dye except for light fastness, and the cost of natural dyeing was almost half that of the reactive dyeing. 22
The research about the usage of metallic mordants in reactive dyeing of nylon fabrics failed and no useful scientific data about the subject were found during the literature research.
This research focused on the usage of two metallic and one bio-mordant in reactive dyeing of nylon fabrics. The purpose was to research better dyeing, coloration and fastness properties with the usage of mordants with commercial reactive dyes which were specially used for cellulosic dyeing. The results showed that the percentage exhaustion and percentage dye fixation properties of reactive dyes were improved depending on the type of mordant, dyeing pH and the selected mordanting method. Each of the selected reactive dyes had its own best dyeing performance with different combinations of mordant, dyeing pH and mordanting method.
Materials and Method
The dyeing properties of commercial reactive dyes for cellulosic fibers were investigated on nylon fabric for different mordants, three dyebath application pH levels and three mordanting methods via absorbances of dyeing liquors and color measurements of the dyed samples. The results were reported as percentage exhaustion (%
Hundred percent nylon 6,6 fabric which was woven from 140/136 (denier/filament number) yarns in warp (49 threads/cm) direction and from 70/68 (denier/filament number) yarns in weft (39 threads/cm) direction was used. The fabric had a 2/1 twill weave and had a unit weight of 176 g/m2. The fabric was scoured under mill conditions in a pretreatment bath (pH 9) including a nonionic washing agent (4 mL/L) at 60°C for 60 min to remove the impurities. The fabric was dried and stentered in the mill at 195°C for 60 s prior to dyeing.
Dyeing experiments were carried out using a laboratory type sample dyeing machine (Ataç, Turkey). In all, 5 g pieces of fabrics were dyed at a liquor ratio of 20:1, using stainless steel dye pots. Dyebaths contained fabric sample, dyestuff, pH chemicals and mordants when necessary. Two different dyeing concentrations, that is, 0.5%, and 2% owf (on weight (mass) of fiber/fabric) were applied. Reactive dyes which had three different reactive groups; monochlorotriazine (MCT), trifluoroprymidine (TFP) and MCT/vinylsulfone (VS) were used (no C.I. names were allocated). All the dyes had a navy-blue hue. Dyeing profiles (as mordanting methods) given in Figure 1 were used. The initial dyeing temperature was selected as 30°C and a 102°C maximum dyeing temperature was used. In each dyeing process, the temperatures were increased by 2°C/min where necessary. Dyeings continued at 102°C for 60 min and later the temperature was decreased to 40°C by 4°C/min. At the end of dyeing, the dyed fabric samples were removed and rinsed in cold tap water at a 100:1 liquor ratio for 10 min to remove the unfixed dye which was left on fiber surfaces. Dyed and rinsed samples were not treated further and the cleared samples were left to dry under laboratory conditions prior to color measurements, and further extraction and fastness tests.
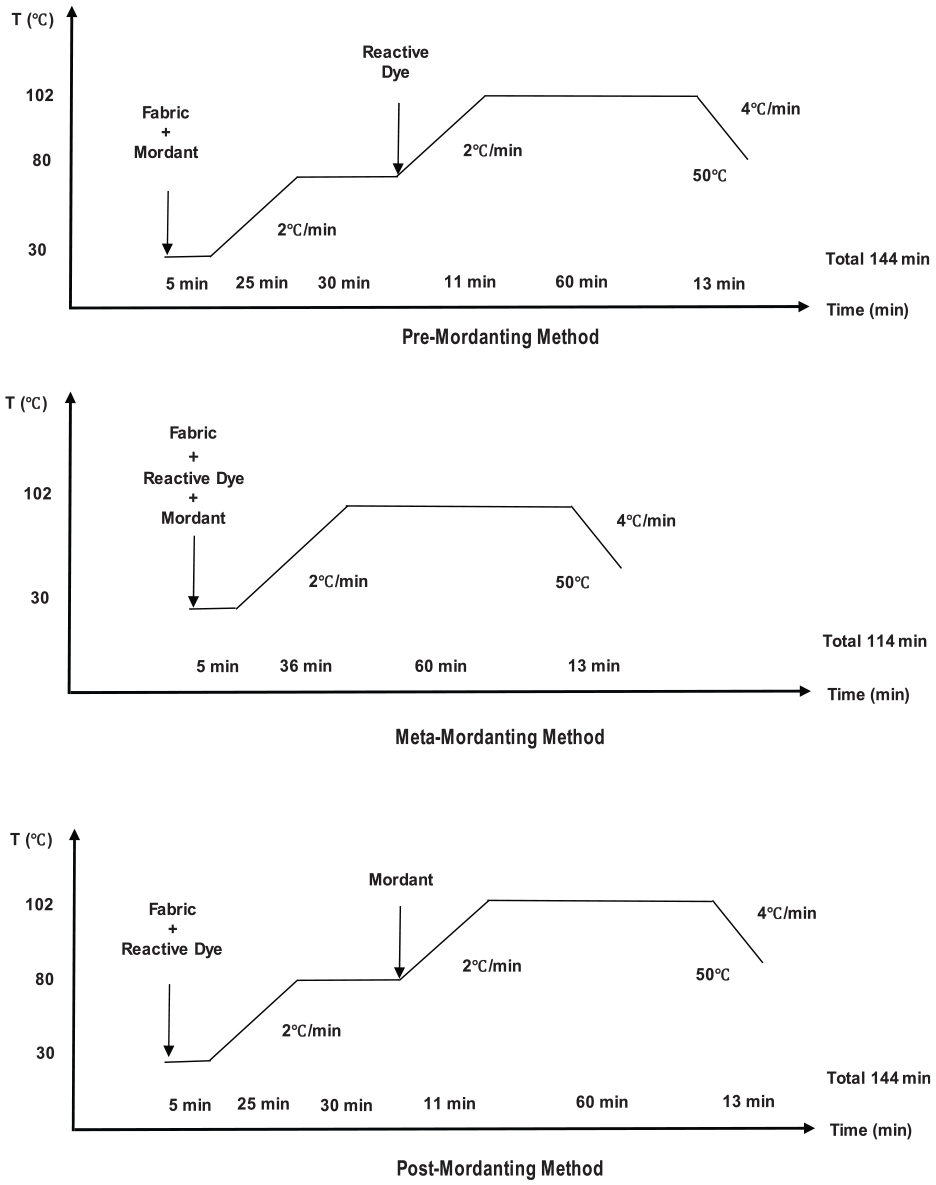
Dyeing profiles used in the experimental part.
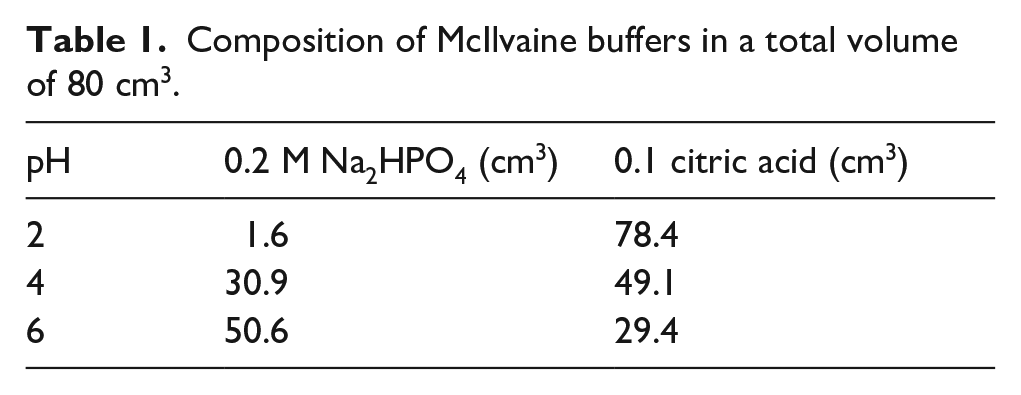
The pH of the dyeing liquors was adjusted according to McIlvaine buffers (Table 1). The pH of the dyeing liquors was carefully checked and adjusted when necessary with a pH meter. Three different pH levels of pH2, pH4 and pH6 were employed. Alkaline dyeings were not performed depending of the unfavorable results in former papers3,13,14 and also the mordants used in the experimental research were stable in acidic medium.
Composition of McIlvaine buffers in a total volume of 80 cm3.
Two metallic mordants; iron (II) sulfate heptahydrate (FeSO4·7H2O), potassium aluminum sulfate dodecahydrate (KAl(SO4)2·12H2O) (potassium alum, or alum), and one bio-mordant, tannic acid (C76H52O46), were used as mordants. They were supplied in laboratory grade and used as received from their packs with only very short contact with air.
The wash fastness test was performed according to ISO 105-C06:2010 A2S test (4 g/L ECE reference detergent WOB, 1 g/L sodium perborate, 150 mL liquor volume, 40°C, 45 min.). 23 The wash fastnesses of fabric samples were evaluated with color measurements in order to present a clearer understanding.
The light fastness test was performed according to the ISO 105-B02:2014 test. 24 The test lasted for 96 h in order to observe the behavior of the fabrics under long term exposure conditions. The duration of 96 h could be matched to L6 to L4 (blue wool standard) fading exposure. During the light fastness test, the test specimens were cut in 4 cm × 10 cm dimensions and they were placed in the exposure chamber attached to the appropriate sample holders. Similar to the wash fastness test, the light fastness of fabric samples was evaluated with color measurements in order to present a clearer understanding.
Color measurements on the dyed samples and transmittance measurements of the dyeing liquors were performed by using a reflectance/transmittance spectrophotometer (Datacolor 600, Switzerland) between 400 and 700 nm under D65/10° illuminant and specular component included (SCI) mode. Fabric samples were folded twice to achieve a four layers of fabric thickness. Four reflectance measurements were performed at different positions on the dyed fabric samples and the average percentage reflectance was recorded. Color coordinates (
Color properties of the nylon fabric prior to dyeing were as follows:
The extent of dye exhaustion was determined by transmittance measurements. The absorbance of each dyebath solution before and after dyeing process was measured using a 1 cm quartz cell housed in a Datacolor 600 spectrophotometer at the

Determination of the degree of dye fixation was performed via Soxhlet extraction by aqueous alkaline pyridine solution and color measurement. A sample of 1 g of dyed fabric was immersed in 150 cm3 of aqueous 20% (prepared by using distilled water) pyridine solution. The temperature was rapidly raised to the boiling point and treatment continued at the boil under reflux for at least 6 h until no further dye was removed from the fiber.
The stripped dyeings were thoroughly rinsed in tap water for 10 min (at 100:1 liquor ratio) and allowed to dry under laboratory conditions. The percentage fixation of the dye which exhausted, %

Equation (3) represents the overall percentage fixation, %

Color difference (Δ
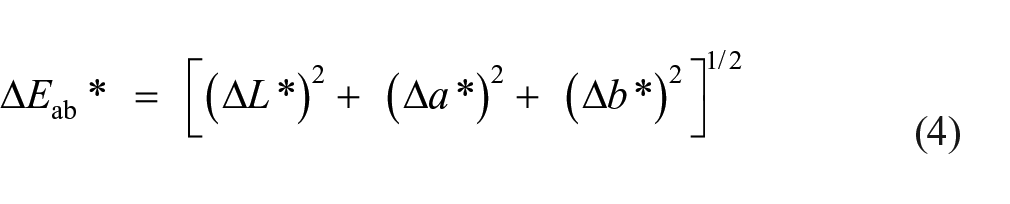
The dyeing profile of meta-mordanting (Figure 1) was used to perform dyeings which did not contain any mordant. The results were presented with the term “blind dyeing” in the tables and figures where necessary.
Results and Discussion
% Exhaustion and % Fixation Values
All the ionic dyes that penetrate inside nylon fibers do not have to form strong ionic (in case of acid dyes) or covalent bonds (in case of reactive dyes) with the ionized groups of the macromolecules and some amount of dye could stay in the amorphous regions of the fibers via relatively weaker bonds such as hydrogen and van der Waals forces. Under these circumstances, the dyes could stay in a loose state in the amorphous regions. Under wet conditions (washing or laundering medium) water molecules which penetrate into the fiber could break the weak bonds (such as hydrogen bonds) between the ionic dyes and fiber molecules; the loosely bonded dyes could become water soluble again. These dyes could migrate outside the fiber and can lower the wash fastness properties of the dyed goods; they could stain the adjacent fabrics. Both results are because of dye loss and these losses could cause color change.
The application of metallic mordants could have the possibility of improving the wash fastness properties of reactive dyed nylon fabrics. These mordants could retain the loosely bonded dyes in fibers which results in an increase in wash fastness properties.
Nylon 6,6 fabric samples were dyed at two dyeing concentrations (0.5% and 2% owf) and at three pH levels (pH2, pH4, pH6) with three reactive dyes that had different reactive groups (MCT, TFP and MCT/VS) via three mordanting methods (pre-, meta-, and post-mordanting). Color strength (K/S), percent exhaustion (%
Experimental results at 0.5% owf dyeing concentration.
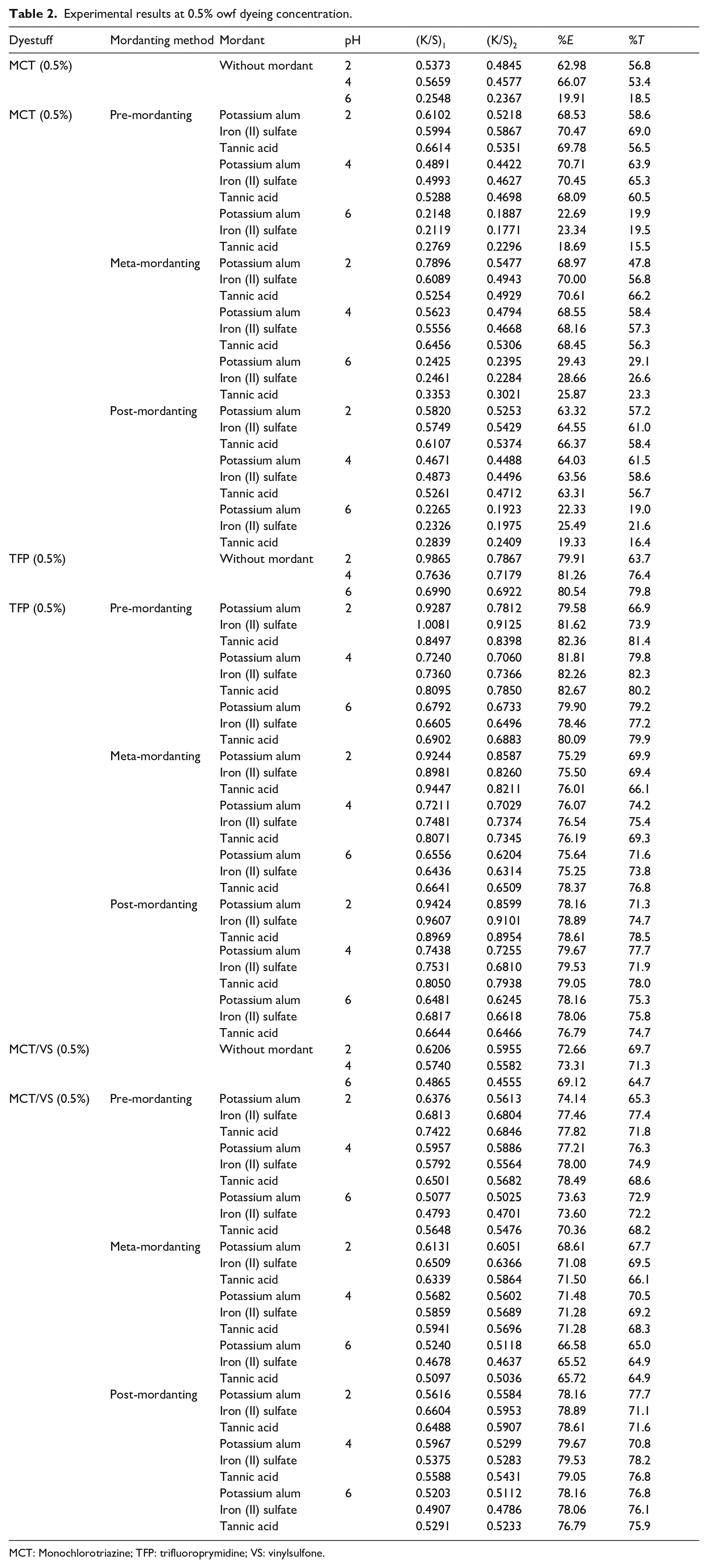
MCT: Monochlorotriazine; TFP: trifluoroprymidine; VS: vinylsulfone.
Experimental results at 2% owf dyeing concentration.
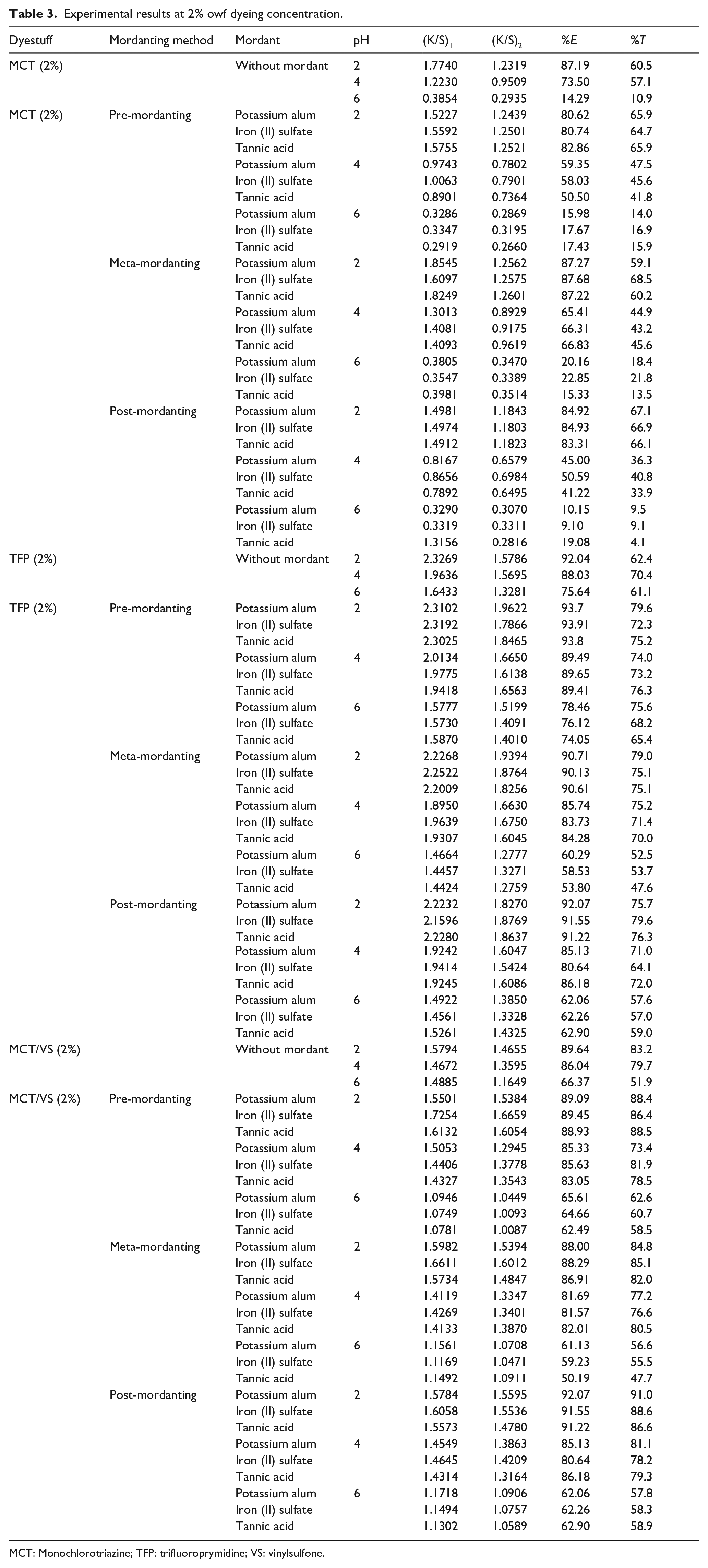
MCT: Monochlorotriazine; TFP: trifluoroprymidine; VS: vinylsulfone.
Color strength (K/S) values before ((K/S)1) and after ((K/S)2) dyeing besides percentage exhaustion (%
The experimental results of the MCT reactive dye applied at the dyeing concentration of 0.5% owf are presented in the upper part of Table 2. When the blind dyeing was performed at pH 2 and pH 4, the MCT reactive dye had similar corresponding %
When the mordanting methods were compared with each other, it was observed that the highest %
When the mordant chemicals were compared with each other, it was observed that tannic acid gave the lowest %
When the application pH levels were considered with regard to the mordant chemicals, the lowest %
An overall discussion of the %
The experimental results of the TFP reactive dye applied at the dyeing concentration of 0.5% owf are presented in the middle part of Table 2. When the blind dyeing was performed, the TFP dye had close corresponding %
When the mordanting methods were compared with each other, it was observed that almost the highest %
When the mordant chemicals were compared with each other, it was observed that all the mordants gave similar %
An overall discussion of the %
The experimental results of the MCT/VS reactive dye applied at the dyeing concentration of 0.5% owf are presented in the bottom part of Table 2. When the blind dyeing was performed, the MCT/VS dye had different %
When the mordanting methods were compared with each other, it was observed that the highest %
When the mordant chemicals were compared with each other, it was observed that their performances changed with application pH levels. All the mordant chemicals resulted almost the same %
An overall discussion of the %
Color strength (K/S) values before ((K/S)1) and after ((K/S)2) dyeing besides percentage exhaustion (%
The experimental results of the MCT reactive dye applied at the dyeing concentration of 2% owf are presented in the upper part of Table 3. When the blind dyeings were performed, the MCT dye had similar %
When the mordanting methods were compared with each other, it was observed that %
When the mordant chemicals were compared with each other, it was observed that iron II sulfate usually gave the highest %
When the application pH levels were considered with regard to the mordanting chemicals, the lowest %
An overall discussion of the %
The experimental results of the TFP reactive dye applied at the dyeing concentration of 2% owf are presented in the middle part of Table 3. When the blind dyeing was performed, the TFP reactive dye showed the best %
When the mordanting methods were compared with each other, it was observed that the highest %
When the mordanting chemicals were compared with each other, it was observed that all the mordanting chemicals resulted in high exhaustion and fixation values at pH 2 and pH 4. When pH 6 was discussed against pH 2 and pH 4, pH 6 caused low %
When the application pH levels were considered with regard to the mordanting methods and mordanting chemicals, the lowest %
An overall discussion of the %
The experimental results of the MCT/VS reactive dye applied at the dyeing concentration of 2% owf are presented in the lower part of Table 3. When the blind dyeing was performed, the MCT/VS reactive dye had different %
When the mordanting methods were compared with each other, it was observed that the highest %
When the mordant chemicals were compared with each other, it was observed that their performances changed with the application levels of pH, similar to the MCT and TFP reactive dyes in Table 3. The %
When the mordanting chemicals were compared with each other, it was observed that all the mordanting chemicals resulted in high exhaustion and fixation values at pH 2 and pH 4, similar to the MCT and TFP dyes in Table 3. The highest %
An overall discussion of the %
The %
ΔE* Color Difference Values
The Δ
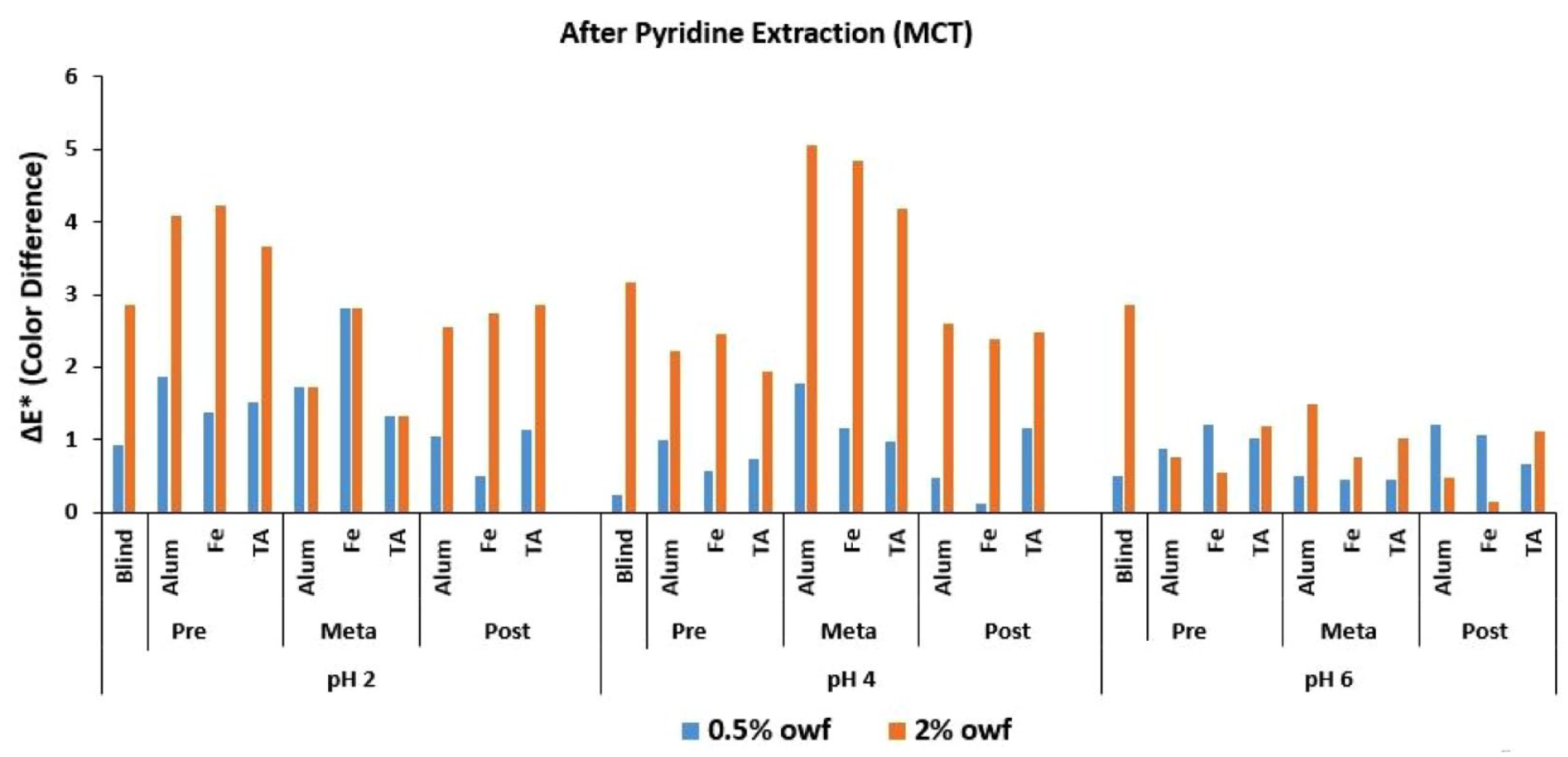
Δ
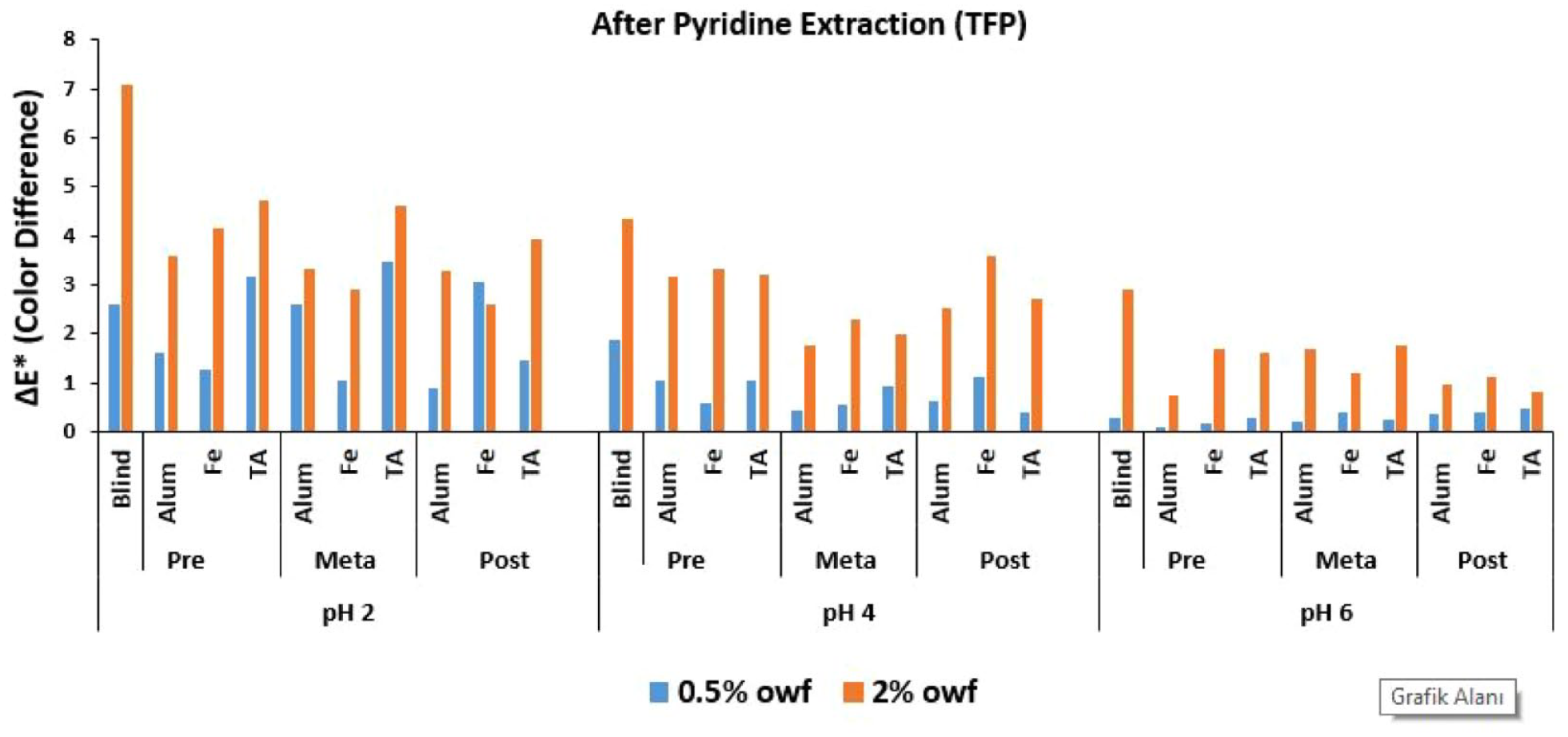
ΔE* values of TFP reactive dye after pyridine extraction.
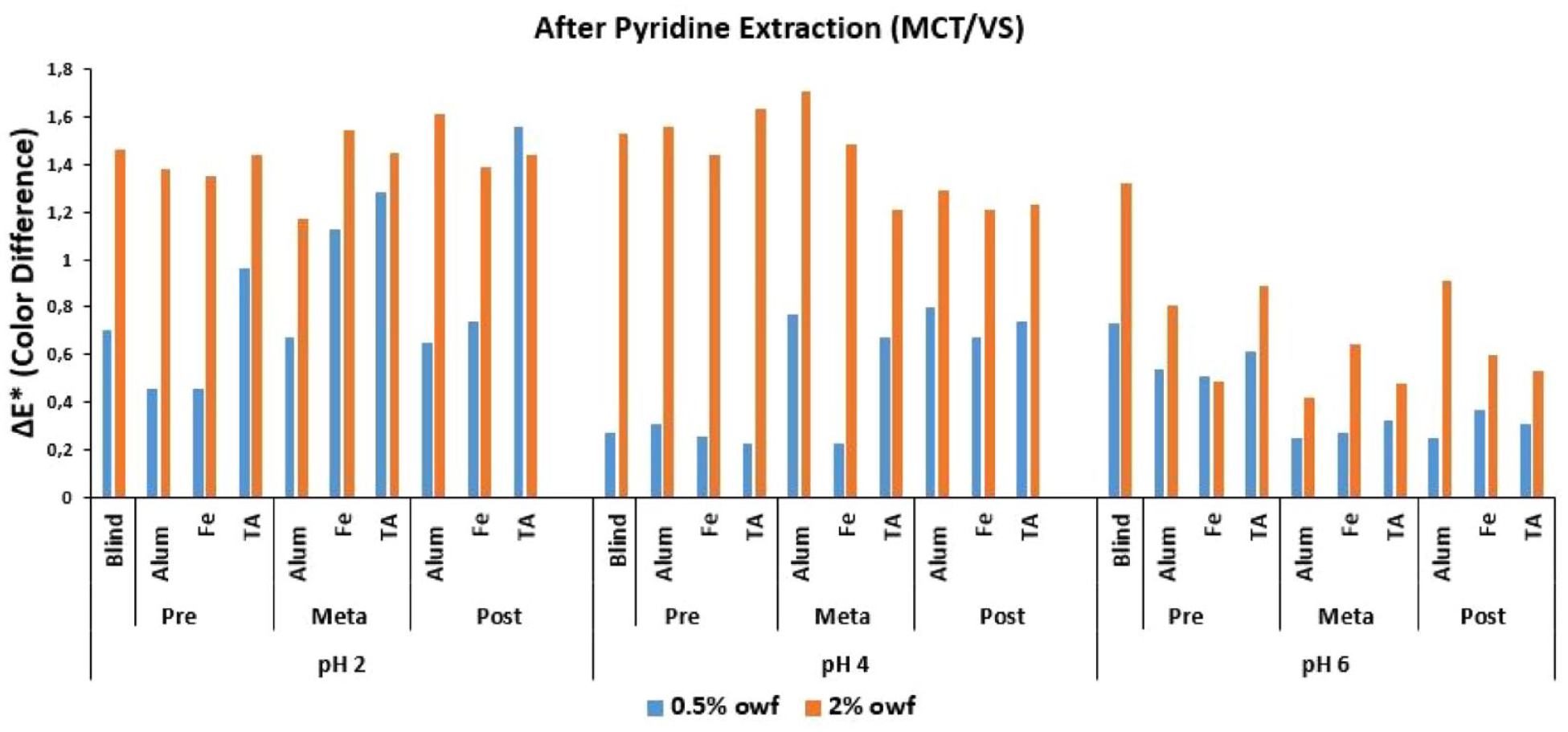
ΔE* values of MCT/VS reactive dye after pyridine extraction.
In Figure 2, where the Δ
In Figure 3 where the Δ
In Figure 4 where the Δ
An overall discussion of Figures 2–4 revealed that the lowest Δ
The Δ
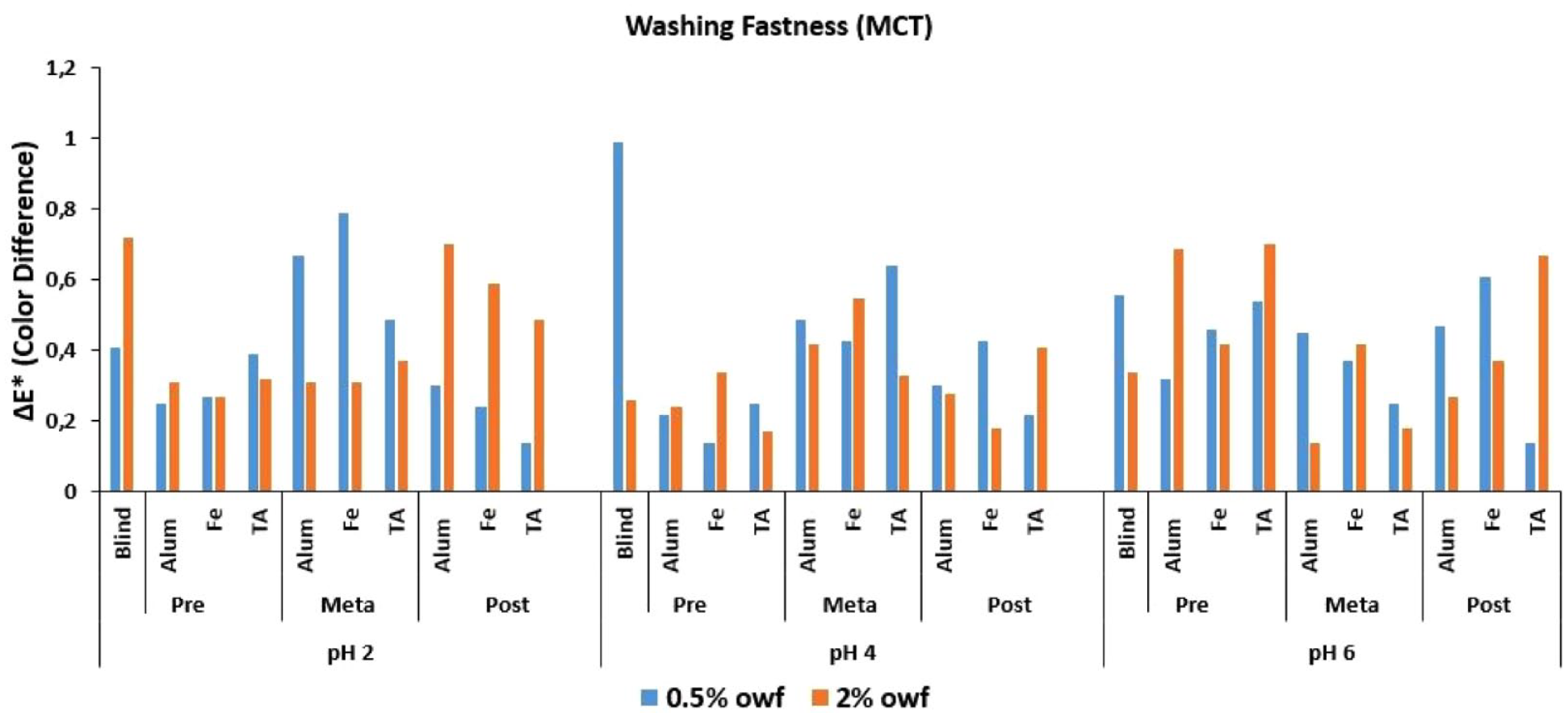
ΔE* values of MCT reactive dye after wash fastness test.
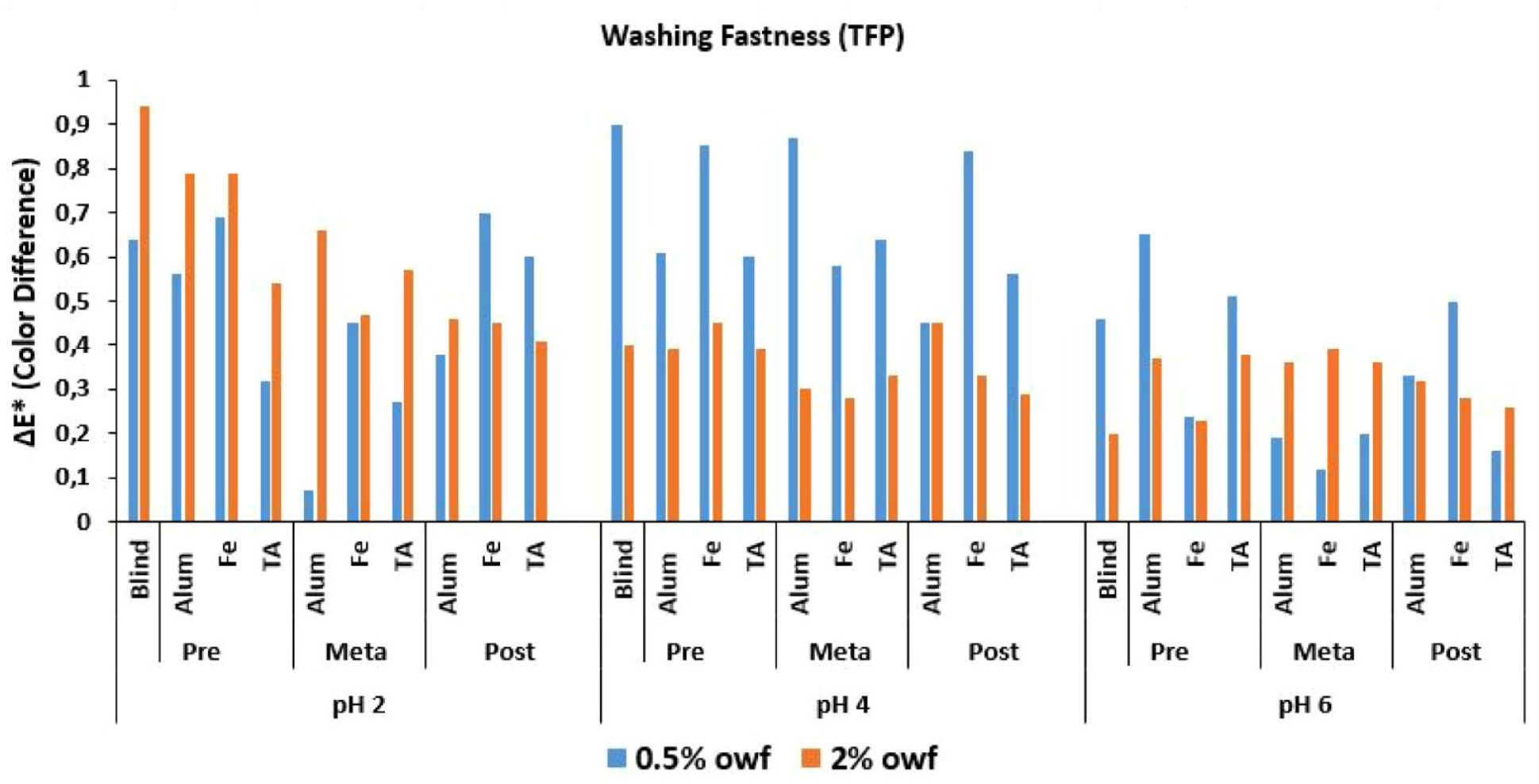
ΔE* values of TFP reactive dye after wash fastness test.
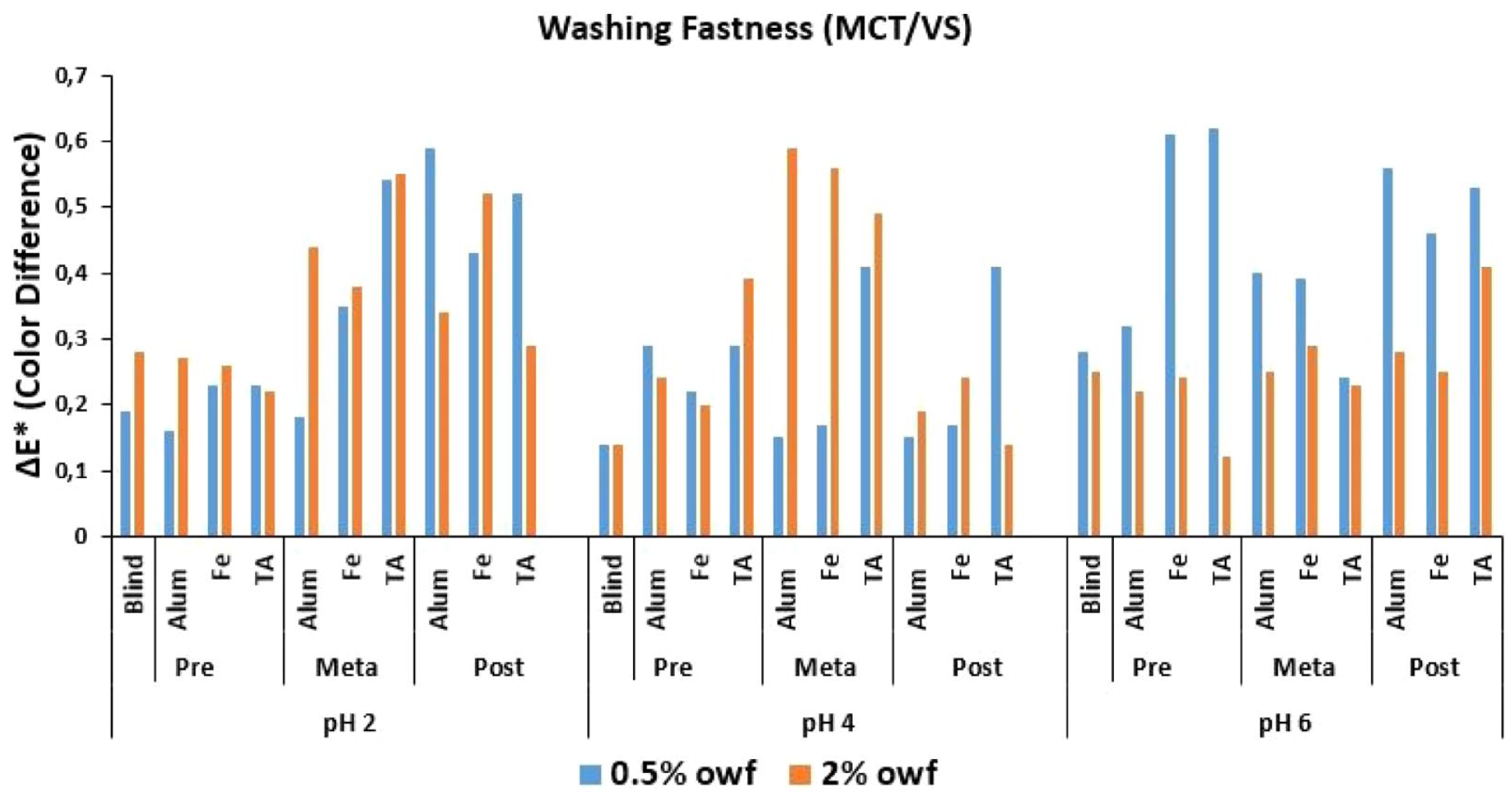
ΔE* values of MCT/VS reactive dye after wash fastness test.
The Δ
The Δ
The Δ
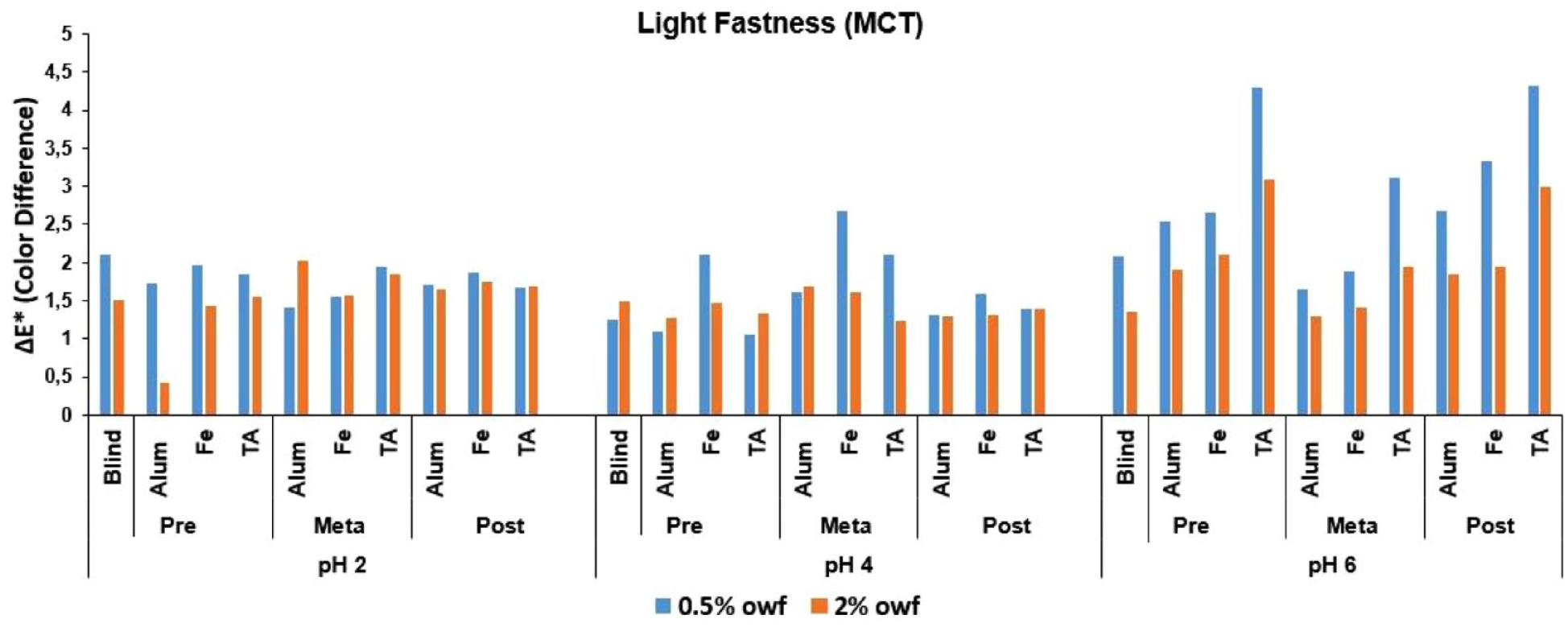
ΔE* values of MCT reactive dye after light fastness test.
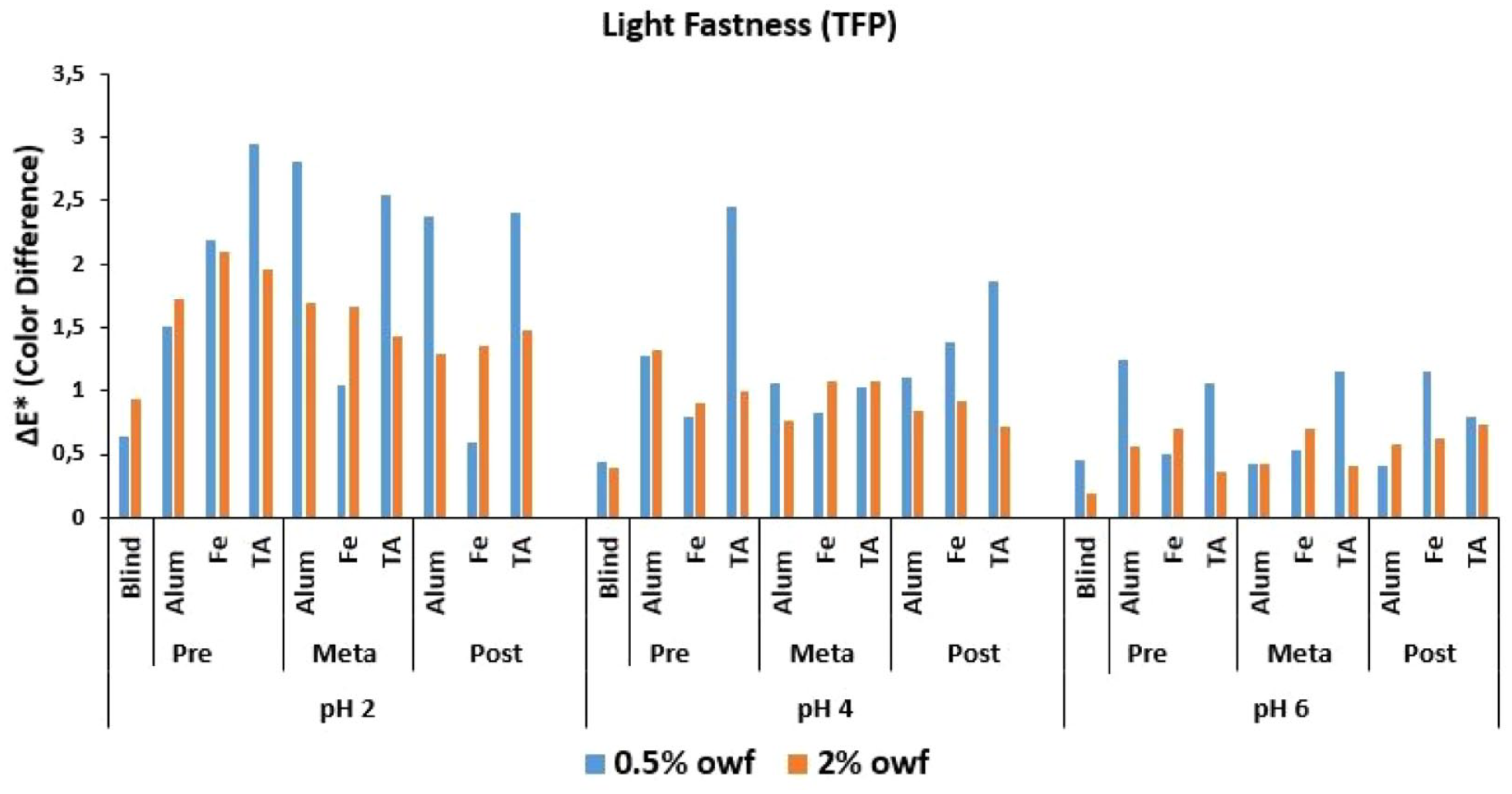
ΔE* values of TFP reactive dye after light fastness test.
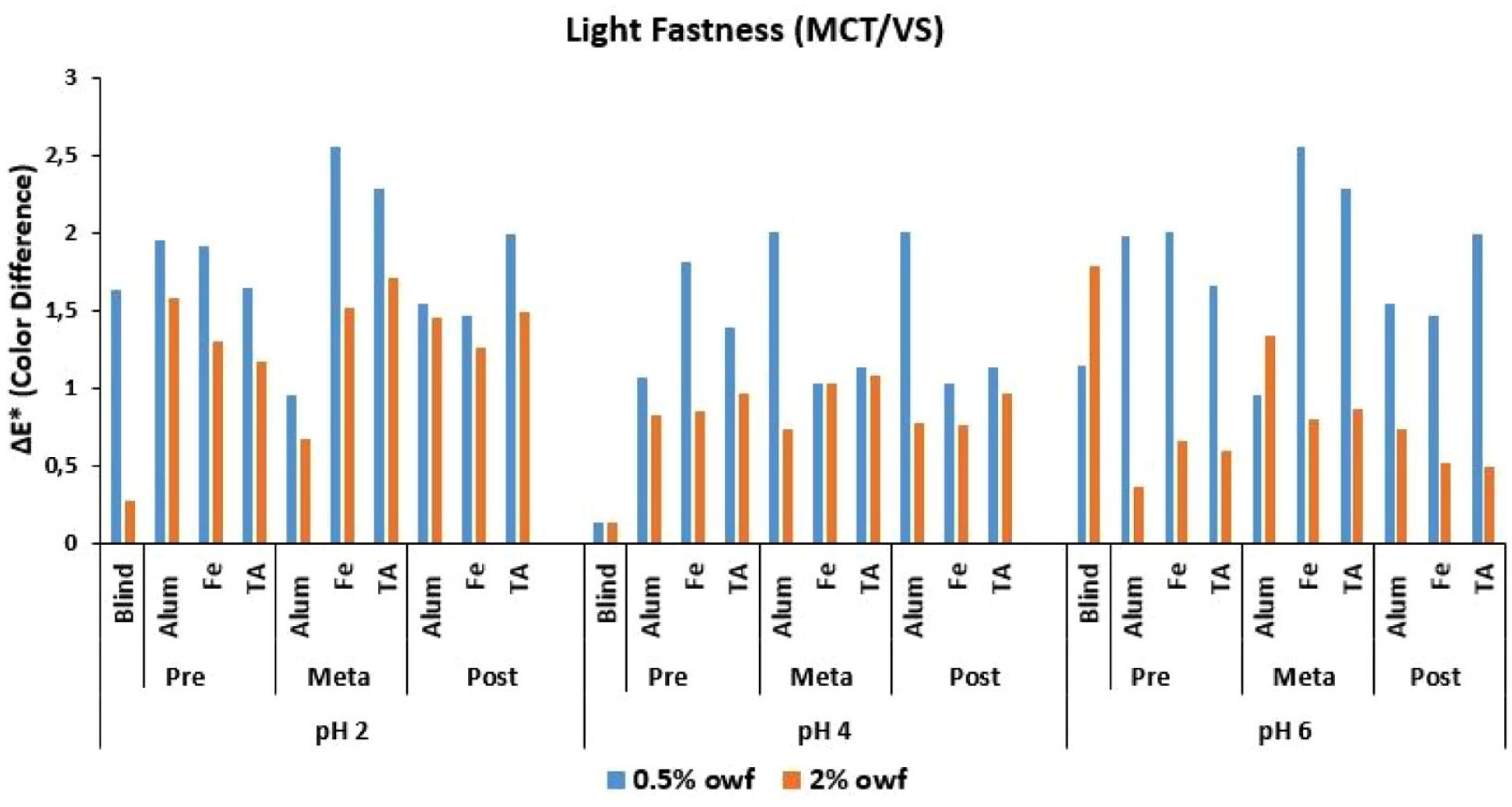
ΔE* values of MCT/VS reactive dye after light fastness test.
The Δ
The Δ
An overall discussion of the Δ
Low numerical Δ

It was stated in the Introduction part that some reactive dyes, which were developed for cellulosic fibers, can be covalently fixed to nylon fibers at slightly acidic pH dyeing medium at the boil. Under these conditions, covalent bonds form between the dye and amino groups of nylon, without the need for an alkaline fixation step.9,12,28 As indicated by Burkinshaw and Gandhi12,13 and Burkinshaw and Wills, 14 under acidic conditions the terminal amino groups in the nylon fiber will be protonated according to Equation (5). Under these conditions an equilibrium will be established according to the number of hydrogen protons in the dye bath and in the fiber. Being in close contact with the amino groups in the fiber, the anionic reactive dyes will be adsorbed on the protonated terminal amino groups in the fiber via ion–ion forces under acidic dyeing conditions. Taking the equilibrium presented in Equation (5), it could be discussed that at any given time, the amino group in the fiber will exist in its non-protonated form, and can react with the adsorbed dye. With decreasing pH of the dye bath, the equilibrium will move to the right and the number of available nucleophilic amino groups will decrease. Three pH levels were used in this study, being pH 2, 4, and 6, and in the case of pH 6, the extent of dye fixation would be expected to be higher than that at pH 2 and pH 4.12,14 Besides, the same conclusion could be made between pH 2 and pH 4. Under low pH conditions in the dye bath, it can be considered that the anionic dye can have the chance to be attracted strongly to the protonated amino end group in the fiber, and an unprotonated amino end group can attack the reactive group of the adsorbed dye molecule which results in a dye–fiber reaction. The experimental exhaustion, fixation and color difference results presented in this study revealed that in order to make a clearer discussion and conclusion, all the experimental variables should be considered together. The exhaustion and fixation levels of the dyes changed mainly according to their reactive groups and application pH levels. The mordanting method and the mordant chemical used in dyeing gave correlating results with the reactive group and dyeing pH level.
The experimental results obtained in this research revealed that metallic mordants could be used in reactive dyeing of nylon fabrics. However, the success of the functional reactive groups changes with correlation of the mordanting method, mordant chemical and application pH level, besides % owf dyeing concentrations. The mordant concentration was chosen as the possible lowest one considering the environmental aspects of commercial dyeings. Better exhaustion and fixation results were obtained with higher mordant concentrations in the unpublished part of the research and higher mordant concentrations could be used in conventional dyeings whenever environmental restrictions allow.
Conclusion
Nylon fabric was dyed with three reactive dyes having different functional groups in the presence of three chemicals mordants at two different dyeing concentrations by applying three mordanting methods and three dyeing pH levels. Exhaustion and fixation properties were researched along with wash fastness and light fastness by the extraction and color measurements. The results revealed that nylon fabric could be dyed with reactive dyes in the presence of chemical (metallic) mordants. However, the independent variables (mordanting method, mordant chemical, application pH level, and dye concentration) applied in dyeing experiments showed that the dependent variables (%E, % T, ΔE* and fastness) do not change uniformly. The dyeing results were prone to the experimental variables. TFP and MCT/VS reactive dyes performed better than MCT reactive dye. A complex correlation was obtained among mordanting methods, mordant chemicals and pH levels. Usually lower exhaustion and fixation levels were obtained at meta-mordanting than with the other two methods. An application pH level of 6 generally caused lower exhaustion and fixation at 2% dyeing concentration. Alum and iron II sulfate resulted in similar values but the results obtained with tannic acid changed very much with experimental variables. Environmentally accepted metallic mordant concentration could be used in reactive dyeing of nylon fabrics with optimized mordanting methods and pH levels.
Footnotes
Acknowledgements
The authors thank Bursa Uludag University Scientific Research Projects Coordination Unit for the financial support (Project No. FYL-2021-332) which made this research possible. The authors thank Bursalı Tekstil San. ve Tic. A.Ş. for its kind support in the usage of laboratory facilities. The authors thank Dr Mehmet Tiritoglu for his kind support in the experimental part.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Bursa Uludag University Scientific Research Projects Coordination Unit (Project No. FYL-2021-332).
