Abstract
Reactive dyes in nylon dyeing have been associated with utilization inefficiencies despite their high color fastness. A potential solution to this issue involves combining the benefits of reactive dyes and milling acid dyes to obtain low-water-soluble reactive dyes (LWSRDs), which can stain and fix to nylon fibers under neutral conditions. To identify a suitable functional group for LWSRDs, six model dyes comprising one vinyl sulfone and five monochloro-
Keywords
Polyamide, known as nylon, is a synthetic fiber polymerized from dicarboxylic acid and diamine. Dyed polyamide products are used in various fields, such as textiles and packaging.1 –3 Nylon fibers can be classified into traditional fibers and microfibers based on their fineness, with nylon microfibers referring to those with monofilament fineness of <1 dtex. The terminal amino groups in nylon possess high reactivity, making acid dyes and reactive dyes the common choice for dyeing nylon. Acid dyes create ionic bonds between the colored anions generated from the acid dyes and the protonated amino terminals in nylon under acidic conditions. Milling acid dyes with low water solubility usually exhibit good exhaustion toward nylon fibers under weakly acidic or neutral conditions. 4 However, these ionic bonds become unstable under moist or alkaline conditions, resulting in inadequate wet color fastness for nylon fibers dyed with acid dyes.5,6 Nylon fibers dyed with acid dyes often need additional treatments to improve their color fastness.7 –9 Compared with acid dyes, reactive dyes exhibit superior color fastness owing to their functional groups,10,11 which react with the amino groups in nylon to form covalent bonds.12 –14
Extensive studies have been conducted on various commercial reactive dyes with different functional groups for dyeing nylon fibers. Reactive dyes with single-type functional groups, including
To overcome the issue of insufficient utilization of commercial reactive dyes, numerous cationic reactive dyes and reactive disperse dyes have been synthesized for nylon fibers.21 –32 These dyes mainly feature sulfate ethyl sulfone and chlorotriazine functional groups, exhibiting excellent exhaustion and fixation properties. However, the choice of coupling agents (aniline derivatives) limits the chromatography of cationic reactive dyes and reactive disperse dyes, resulting in their limited applications, mainly in yellow and orange colors.
To address this challenge, one potential solution is to introduce functional groups into the structures of milling acid dyes to prepare low-water-soluble reactive dyes (LWSRDs). These dyes can stain and firmly adhered to nylon fibers under nearly neutral conditions to prevent the destruction of ionic bonds between the dyes and nylon by an alkali. Therefore, the reactivity of the functional groups is a critical factor in the fixation progress of nylon, where rapid reaction ability toward nylon and good stability under neutral pH conditions are essential. This study was focused on selecting a suitable functional group for LWSRDs based on the reactivity and stability of model dyes with monovinyl sulfone (model-monovinyl sulfone; M-MVS) and five typical monochlorotriazine (model-monochlorotriazine; M-MCT) dyes. The fixation between reactive dyes and nylon fibers is a complex process in a solid–solid system; therefore, the fixation progress is considerably influenced by the exhaustion of reactive dyes. To quantitatively evaluate the reactivity of different functional groups, the fixation progress should be simplified into a homogeneous reaction. Glycine, a molecule similar to nylon with an amino and a carboxylic group, was used as a substitute for nylon to simulate the reactions between functional groups and nylon fibers in an aqueous medium. Reactivity and stability tests were performed by referring to the hydrolysis kinetic study of the functional groups.33,34 The functional group with sufficient reactivity and good stability was considered suitable for LWSRDs. Furthermore, four low-water-soluble monovinyl sulfone dyes (LWS-MVSDs) containing the superior functional group were synthesized and used to dye nylon 6 microfibers to obtain excellent fixation and color fastness under neutral conditions.
Experimental details
Chemicals and instruments
1-Acetylamino-8-hydroxynaphthalene-3,6-disulfonic acid (acetyl H-acid) was synthesized in the laboratory. The following chemicals were used in this study: 1-amino-8-hydroxynaphthalene-3,6-disulphonic acid (H-acid), acetic anhydride, cyanuric chloride (TCT), 4-(ethylsulfurate sulfonyl) aniline, 2-amino-5-naphthol-7-sulfonic acid (J-acid), 3-methyl-1-phenyl-2-pyrazolin-5-one, 3-aminobenzene sulfonic acid, 4-aminoacetanilide, 4-butylaniline, 4-
The ultraviolet-visible (UV-Vis) absorption spectra were collected using an Agilent 8453 UV-Vis spectrophotometer. High-performance liquid chromatography (HPLC) was performed using an Agilent 1100 series HPLC system. The mobile phase comprised 3.0‰ (volume contents) triethylamine and 3‰ (volume contents) acetic acid in aqueous solution as solvent A and methanol (HPLC grade) as solvent B. The solvents were pumped through a 20-cm reverse column with A/B gradient ratios ranging from 90:10 to 10:90 over 30 min, followed by a 10-min hold at a constant flow rate of 1.0 mL/min. The samples were analyzed using a UV-Vis spectrophotometer at a wavelength of 500 nm. Atmospheric pressure ionization-electrospray-mass spectra (API-ES-Mass) were recorded using an Agilent 1100 HPLC-MS. Fourier transform infrared (FTIR) spectra were obtained using a Jasco FTIR-430 spectrometer with KBr pellets.
Preparation of dyes
Model-MVS
Preparation of chromophore: 4-(ethylsulfurate sulfonyl) aniline (2.89 g; 0.0102 mol) was dissolved in 20 mL of water at pH 6.0–7.0 by adding an aqueous sodium carbonate solution (10%; w/v). Sodium nitrite (0.70 g; 0.0102 mol) was dissolved in 5 mL of water, then added to the solution. The mixture was rapidly added to cooled concentrated hydrochloric acid (37%, 0.03 mol) under vigorous stirring. The diazotization reaction was maintained for 30 min at 0–5°C and the excess nitrous acid was decomposed using a small amount of sulfamic acid. The diazonium salt was slowly added dropwise to acetyl H-acid solution (3.47 g; 0.01 mol) within 2 h at 0–5°C, and the pH of this solution was maintained at 7.0–7.5 using aqueous sodium carbonate solution (10%; w/v). The reaction solution was maintained under the aforementioned condition above for 2 h.
Vinylation reaction: the pH of the solution was adjusted to 12.0–12.5 by adding aqueous sodium hydroxide solution (10%; w/v) at 0–5°C. The pH was maintained until all the sulfate groups were removed, as confirmed via thin-layer chromatography (mobile phase:
Aftertreatment: after adding potassium acetate (20% of solution volume), the product was isolated, separated by filtration, washed with 100 mL of ethanol thrice and dried in a vacuum oven at 40°C. The product yield was 90.1% and the product purity was 94.1% at 500 nm as measured via HPLC (retention time (RT) = 19.1 min).
IR spectrum (KBr): –NH and –OH stretching 3429 cm−1; N–H bending 1595 cm−1; N=N stretching 1565 cm−1; S=O bending 1315 cm−1; C–N bending 1295 cm−1; –SO3H stretching 1050 cm−1. MS: m/z calculated for C20H17N3O10S3 [M–H]–, 554.01, found 554.10.
Model-MCT1
A mixture of TCT (1.84 g; 0.01 mol) and absolute ethanol (30 mL) was cooled to 0–5°C in an ice bath. Subsequently, sodium carbonate (0.53 g; 0.005 mol) was added to the mixture, followed by stirring for 7 h. 35 The resulting solution was added to 50 mL of water, and then mixed with 20 mL of aqueous H-acid (3.19 g; 0.01 mol) solution. The mixture was stirred for 2 h at 30°C, and the pH of this mixture was maintained at 6.5–7.0 using an aqueous 10% (w/v) sodium carbonate solution. Furthermore, aniline-2-sulfonic acid (1.76 g; 0.0102 mol) was diazotized using the same method as a para base ester. The resulting diazonium salt was slowly added to the aforementioned solution within 2 h at 0–5°C, while maintaining the pH at 7.0–7.5 using an aqueous 10% (w/v) sodium carbonate solution. The reaction solution was maintained under these conditions for an additional 2 h. Subsequently, the pH of the solution was adjusted to 6.0–7.0. The aftertreatment of M-MCT1 was the same as for M-MVS. The product yield was 85.3% and the purity was 97.2% at 500 nm, as measured via HPLC (RT = 17.7 min).
IR spectrum (KBr): –NH and –OH stretching 3449 cm−1; N=N bending 1565 cm−1; triazine stretching 1500 cm−1; C–N bending 1295 cm−1; C–O bending 1200 cm−1; –SO3H stretching 1050 cm−1. MS: m/z calculated for C21H17ClN6O11S3 [M–H]–, 658.98, found 659.00.
Model-MCT2
TCT (1.84 g; 0.01 mol) and ice cubes (20 g) were stirred for 30 min at 0–5°C. The first condensation reaction was started by adding aniline-4-sulfonic acid solution (1.73 g; 0.01 mol) at 0–5°C, while maintaining the pH at 4.0–4.5 for 2 h. Afterward, the reaction solution was mixed with 20 mL aqueous H-acid (3.19 g; 0.01 mol) solution. The mixture was stirred for 2 h at 30°C, while maintaining the pH at 6.5–7.0. The subsequent steps of diazotization, coupling progress and post-treatment were the same as those of M-MCT1. The product yield was 80.2% and the purity was 97.5% at 500 nm, as measured via HPLC (RT = 17.9 min).
IR spectrum (KBr): –NH and –OH stretching 3426 cm−1; N=N bending 1565 cm−1; triazine stretching 1500 cm−1; C–N bending 1323 cm−1; –SO3H stretching 1050 cm−1. MS: m/z calculated for C25H18ClN7O13S4 [M–H]–, 785.95, found 786.00.
Model-MCT3
M-MCT3 was synthesized by replacing aniline-4-sulfonic acid with dimethylamine solution (33%; 0.01 mol) as for M-MCT2. The product yield was 75.8% and the purity was 90.0% at 500 nm, as measured via HPLC (RT = 19.4 min).
IR spectrum (KBr): –NH and –OH stretching 3441 cm−1; N=N bending 1565 cm−1; triazine stretching 1495 cm−1; C–N bending 1295 cm−1; –SO3H stretching 1050 cm−1. MS: m/z calculated for C21H18ClN7O10S3 [M–H]–, 658.00, found 658.00.
Model-MCT4
M-MCT4 was synthesized by replacing aniline-4-sulfonic acid with ammonium hydroxide (35%; 0.025 mol) as for M-MCT2. The product yield was 88.5% and the purity was 93.7% at 500 nm, as measured via HPLC (RT = 16.8 min).
IR spectrum (KBr): –NH and –OH stretching 3445 cm−1; N=N bending 1565 cm−1; triazine stretching 1495 cm−1; C–N bending 1300 cm−1; –SO3H stretching 1050 cm−1. MS: m/z calculated for C19H14ClN7O10S3 [M–H]–, 629.97, found 630.00.
Model-MCT5
M-MCT5 was synthesized by replacing aniline-4-sulfonic acid with aniline (0.93 g; 0.01 mol) as for M-MCT2. The product yield was 61.2% and the purity was 97.3% at 500 nm, as measured via HPLC (RT = 22.3 min).
IR spectrum (KBr): –NH and –OH stretching 3445 cm−1; N=N bending 1550 cm−1; triazine stretching 1491 cm−1; C–N bending 1321 cm−1; –SO3H stretching 1050 cm−1. MS: m/z calculated for C25H18ClN7O10S3 [M–H]−, 706.00, found 706.10.
Low-water-soluble monovinyl sulfone reactive yellow (LWS-MVSRY)
Preparation of the chromophore: 4-(ethylsulfurate sulfonyl) aniline was prepared using the same method as that used for preparing M-MVS. 3-Aminobenzene sulfonic acid (1.75 g; 0.01 mol) was dissolved in 20 mL of water and the pH was adjusted to 7.0–7.5. Subsequently, diazonium salt was added dropwise to the aforementioned solution within 2 h at 0–5°C and a pH of 7.0–7.5. Following that, concentrated hydrochloric acid (37%; 4.0 g; 0.04 mol) and sodium nitrite (0.70 g; 0.0102 mol) were sequentially added to the first coupling solution. In a separate step, 3-methyl-1-phenyl-2-pyrazolin-5-one (1.78 g; 0.01 mol) was dissolved in 50 mL of water, and the pH was adjusted to 9.0–9.5 with sodium hydroxide aqueous solution (10%, w/v). Finally, the diazonium salt was slowly added dropwise to 3-methyl-1-phenyl-2-pyrazolin-5-one solution within 2 h at 0–5°C and a pH of 7.0–7.5.
The acetylation and aftertreatment were the same as for M-MVS. The product yield was 84.3% and the purity was 88.2% at 440 nm, as measured via HPLC (RT = 31.2 min).
IR spectrum (KBr): –NH and –OH stretching 3441 cm−1; C–H stretching 3059 and 2963 cm−1; N=N bending 1556 cm−1; C=C stretching 1502 cm−1; S=O bending 1331 cm−1; C–N bending 1252 cm−1; –SO3H stretching 1029 cm−1. MS: m/z calculated for C24H20N6O6S2 [M–H]–, 551.09, found 551.00.
Low-water-soluble monovinyl sulfone reactive orange (LWS-MVSRO)
Preparation of the chromophore: 4-(ethylsulfurate sulfonyl) aniline was prepared using the same method as that used for preparing M-MVS. J-acid (2.59 g; 0.01 mol) and NaOH (0.40 g; 0.01 mol) were dissolved in 40 mL of water and added dropwise to diazonium salt for 2 h. The reaction solution was maintained at 0–5°C for 8 h; subsequently, its pH was adjusted to 7.0. The diazotization of 4-butylaniline (1.51 g; 0.0101 mol) was conducted using the same method as that used for the diazotization of 4-(ethylsulfurate sulfonyl) aniline; the resulting diazonium salt solution was added dropwise to the first coupling solution at 0–5°C, while maintaining a pH of 7.0–7.5 using an aqueous sodium carbonate solution (10%; w/v).
The acetylation and aftertreatment were performed using the same method as for M-MVS. The product yield was 95.9% and the purity was 91.2% at 440 nm, as measured via HPLC (RT = 31.5 min).
IR spectrum (KBr): –NH and –OH stretching 3376 cm−1; C–H stretching 2927 and 2857 cm−1; N=N bending 1580 cm−1; C=C stretching 1481 cm−1; S=O bending 1370 cm−1; C–N bending 1299 cm−1; –SO3H stretching 1048 cm−1. MS: m/z calculated for C28H27N5O6S2 [M–H]–, 592.14, found 592.20.
Low-water-soluble monovinyl sulfone reactive red (LWS-MVSRR)
Low-water-soluble monovinyl sulfone reactive red (LWS-MVSRR) was synthesized by replacing 4-butylaniline with 4-aminoacetanilide (1.52 g; 0.0101 mol) as for LWS-MVSRO. The product yield was 98.3% and the purity was 92.0% at 500 nm, as measured via HPLC (RT = 26.4 min).
IR spectrum (KBr): –NH and –OH stretching 3388 cm−1; N=N bending 1618 cm−1; C=C stretching 1489 cm−1; S=O bending 1333 cm−1; C–N bending 1284 cm−1; –SO3H stretching 1043 cm−1. MS: m/z calculated for C26H22N6O7S2 [M–H]–, 593.10, found 593.10.
Low-water-soluble monovinyl sulfone reactive blue (LWS-MVSRB)
LWS-MVSRB was synthesized by replacing J-acid and 4-butylaniline with H-acid (4.00 g; 0.01 mol) and 4-
IR spectrum (KBr): –NH and –OH stretching 3434 cm−1; C–H stretching 2927 and 2948 cm−1; N=N bending 1571 cm−1; C=C stretching 1489 cm−1; S=O bending 1333 cm−1; C–N bending 1284 cm−1; –SO3H stretching 1043 cm−1. MS: m/z calculated for C36H43N5O9S3 [M–H]–, 784.22, found 784.30.
Reactivity study of model dyes
Glycine (0.75 g, 0.01 mol) and phosphate buffer solution (pH 6.86, 100 mL) were mixed and stirred in a 250-mL flask equipped with a thermometer, a pH meter and an Allihn condenser. The common dyeing temperature (75°C and 90°C) was chosen to simulate the fixation progress of reactive dyes. After reaching the desired test temperature, model dyes (∼1 × 10−4 mol) were added into the flask quickly. Samples were collected at suitable intervals, quenched and maintained in an ice bath before HPLC analysis. The absorption peaks were measured at 500 nm to determine their respective peak area.
Stability study of model dyes
Phosphate buffer solution (pH 6.86, 100 mL) was stirred in a 250-mL flask with a thermometer, a pH meter and an Allihn condenser at 90°C. Subsequently, model dyes (∼1 × 10−4 mol) were added into the flask quickly. Samples were collected at suitable intervals, quenched and maintained in an ice bath before HPLC analysis. The absorption peaks were measured at 500 nm to determine their respective peak area.
Dyeing nylon 6 microfibers with the M-MVS and LWS-MVSDs
As shown in Figure 1, the M-MVS and LWS-MVSDs [2% (o.w.f.), on weight of fabric] were dissolved in a phosphate buffer solution (pH = 6.86; 40 mL). Nylon 6 nonwoven microfibers (2.000 g) were immersed in the aforementioned dye solution in a dyebath with a liquor ratio of 20:1. The dyeing progress was carried out in the sealed, stainless steel dye pots with 100 cm3 capacity of an XW4000B Sample Dyeing Machine (Jingjiang Xinwang Dyeing Finishing & Machinery Factory). The temperature for exhaust dyeing was increased to 90°C at the rate of 1°C·min−1, and maintained for 60 min to achieve dye adsorption equilibrium and sufficient fixation time. After dyeing, the dyebaths were cooled to room temperature at the rate of 2°C·min−1. The dyed nylon microfibers were cleaned in soap solution at 95°C until the solution was colorless.
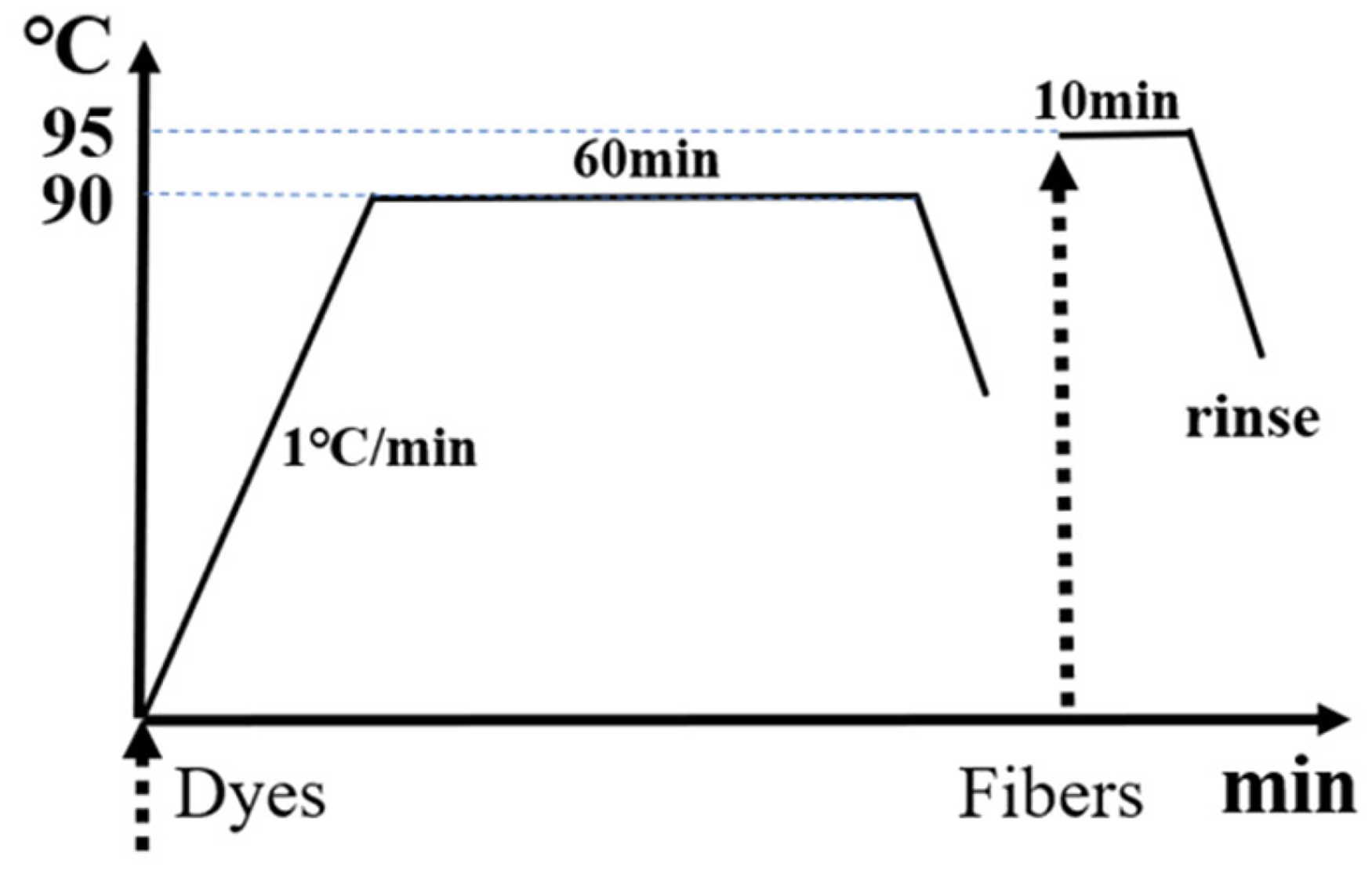
The dyeing progress of the monovinyl sulfone dyes.
Exhaustion (
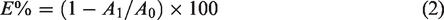
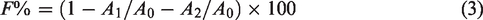

Measurements of color strength and fastness
The 
The washing fastness of the samples was tested according to the ISO 105-C10:2006 test. Rubbing fastness was estimated using a Y(B)571-II crockmeter (Darong Standard Textile Apparatus Co., Ltd, China) according to ISO 105-X12:2016. Light fastness was determined using a YG(B)611-V light-fastness tester (Darong Standard Textile Apparatus Co., Ltd, China) according to ISO 105-B02:2014.
Results and discussion
Model dyes and model reactions to simulate the fixation process
To quantitatively evaluate the reactivity of different functional groups, six model dyes were prepared using one vinyl sulfone and five common monochloro-
Structures and properties of the model dyes
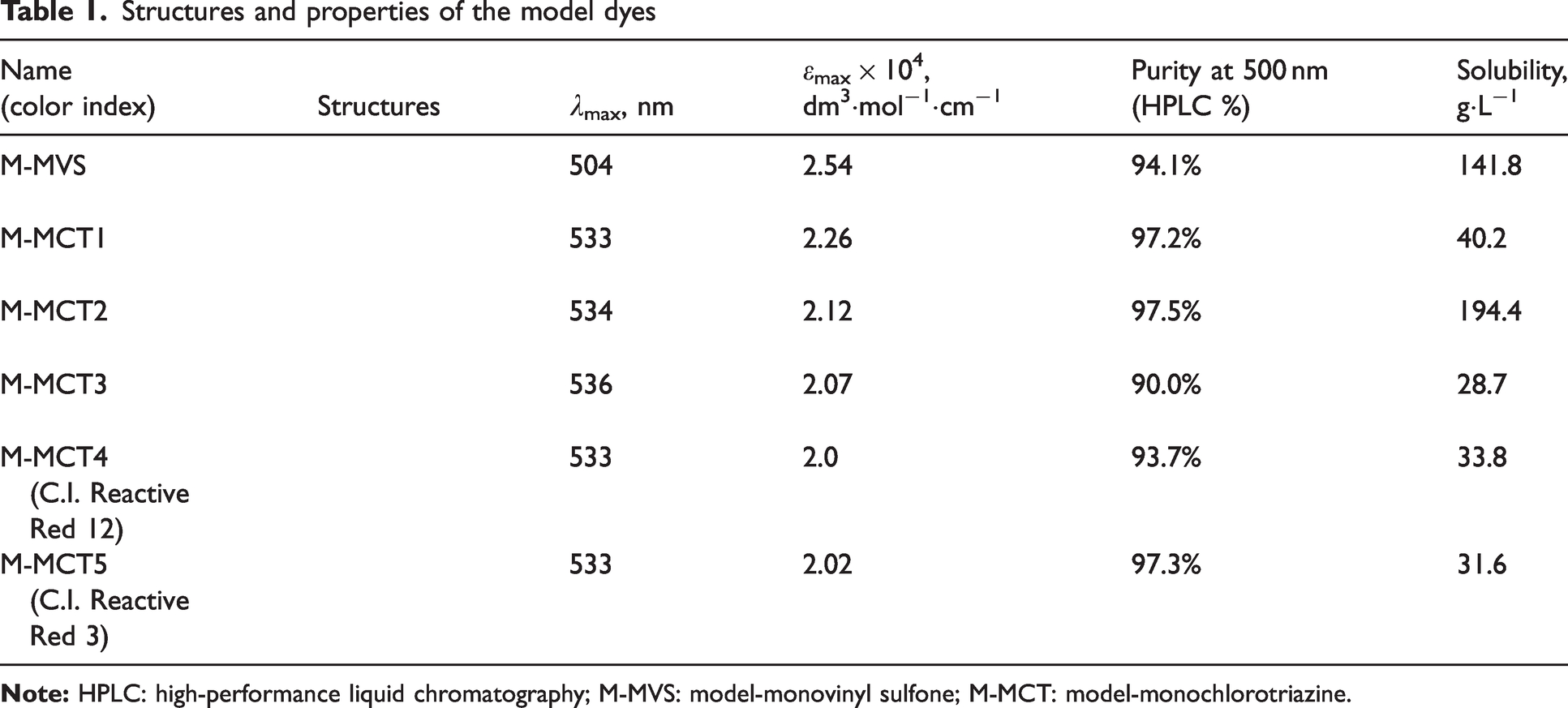
All the model dyes were red monoazo compounds derived from H-acid. M-MVS has the same functional group as C.I. Reactive Red 35. Vinyl sulfone was obtained by removing the sulfate group from sulfate ethyl sulfone to enhance fixation.
18
The monochloro-
Generally, the reactions between M-MVS and the glycine solution followed nucleophilic addition mechanisms, as presented in Figure 2(a). Conversely, the reactions involving M-MCT dyes proceeded via nucleophilic substitution mechanisms, as shown in Figure 2(b). During these reactions, the hydrolysis products of model dyes were the only byproduct because hydroxyl ions could compete with glycine as nucleophilic groups.
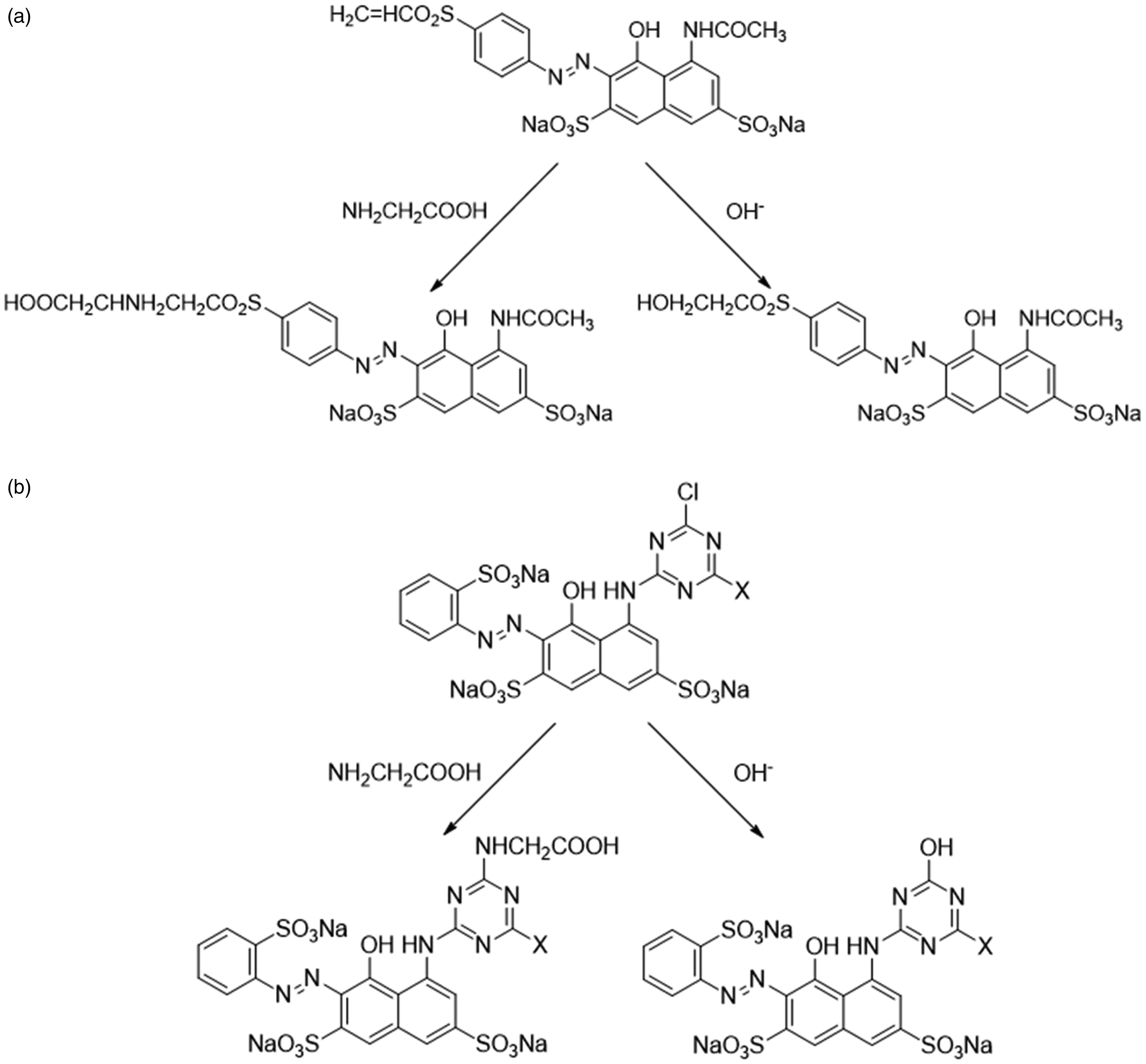
(a) Reaction between model-monovinyl sulfone and glycine and (b) Reactions between model-monochlorotriazine dyes and glycine.
Furthermore, among the five chlorotriazines, ethoxychloro-s-triazine possessed a special functional group. Taylor et al.
36
found that alkoxy could be replaced by hydroxide ion in the hydrolysis of alkoxychloro-
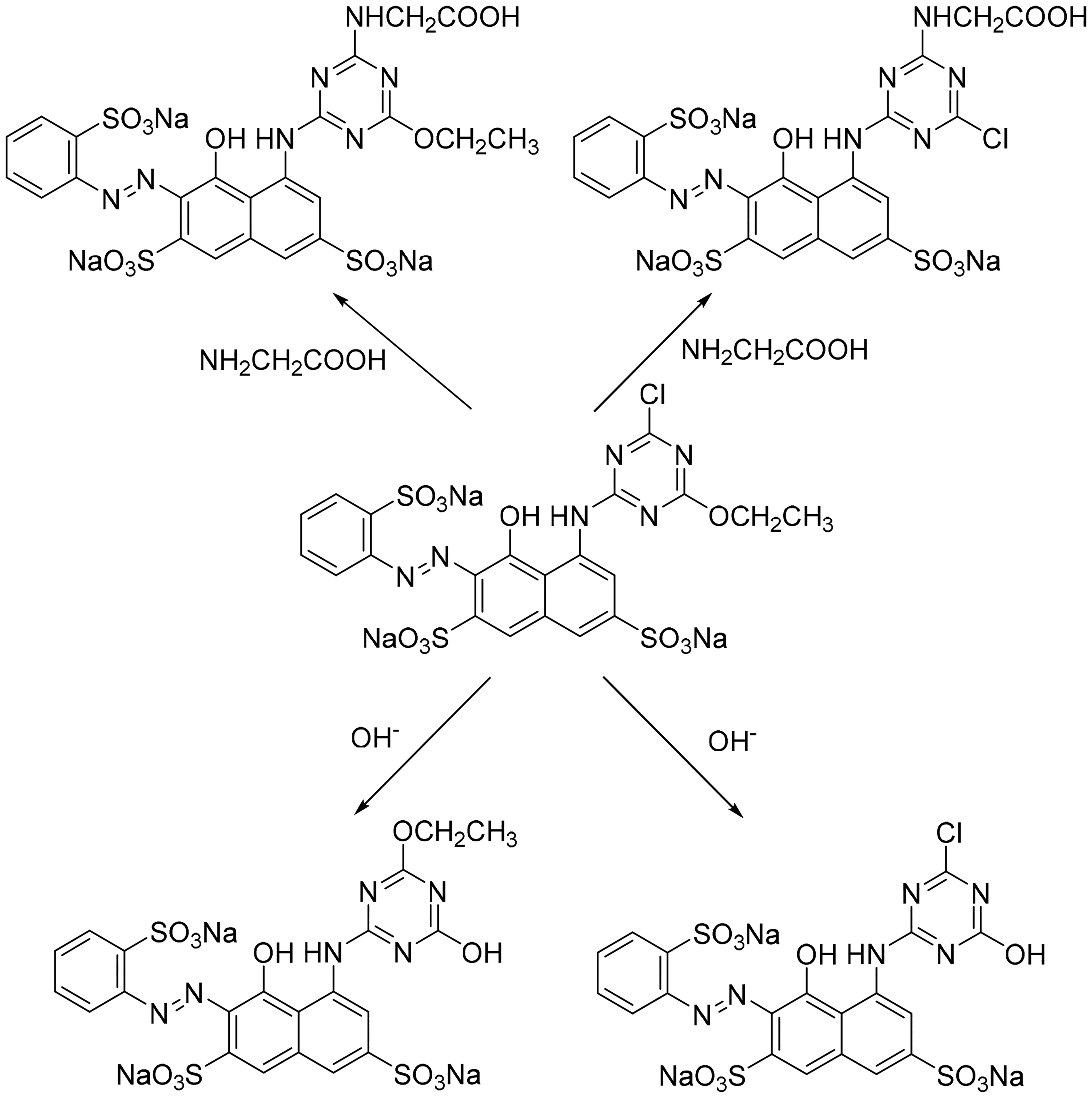
Potential reactions of M-MCT1 in glycine solution. M-MCT: model-monochlorotriazine.
Reactivity study of model dyes
Experiments were conducted in a phosphate buffer solution (pH = 6.8) to measure the reactivity of functional groups under neutral conditions. The model dyes we synthetized had only one functional group; therefore, the reaction rate equation between the dyes and glycine was expressed as in Equation (6):
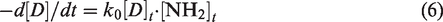
The molar amount of glycine is 100 times that of dye. Glycine in water always exists in dynamic equilibrium, as shown in Figure 4.

Dynamic equilibrium of glycine in water.
The pH of the solution remains stable due to the buffer solution, which ensures that the concentration of the free amino ([NH2]
The plots of ln(
Pseudo-first-order reaction rate of dyes with glycine at different temperatures
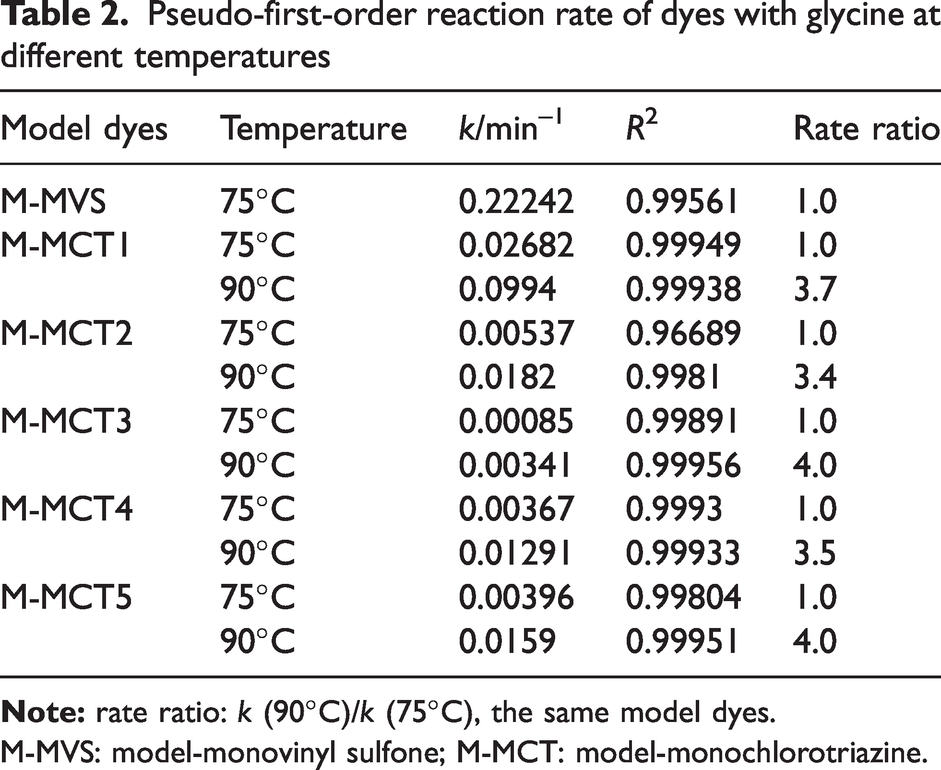
M-MVS: model-monovinyl sulfone; M-MCT: model-monochlorotriazine.
To directly observe the difference in reactivity among the model dyes, the reaction rates at 75°C are collected in Table 3. The reaction rates of ethoxychloro-
The rate ratios of model dyes at 75°C, pH 6.86
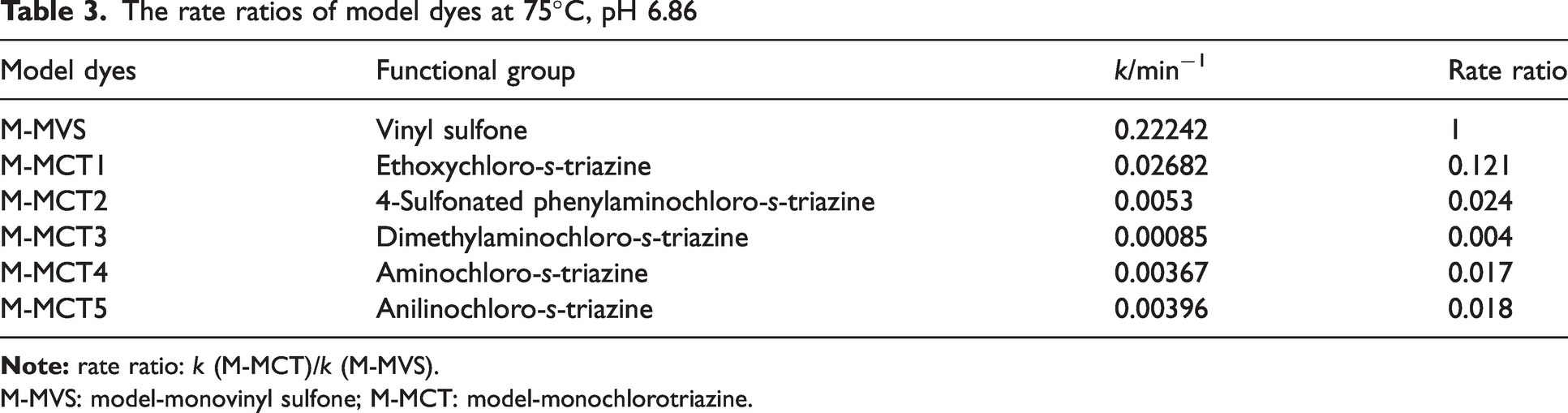
M-MVS: model-monovinyl sulfone; M-MCT: model-monochlorotriazine.
To evaluate the progress of the reactions between model dyes and glycine, plots of percentage content as a function of time were plotted for all the model dyes, and these plots are presented in Figure 5. The reactions were tested up to the 90th minute, because the fixation time for nylon remained within 90 min. The reaction between M-MVS and glycine was found to be completed within 30 min at 75°C owing to its high reactivity. The reaction rate of ethoxychloro-
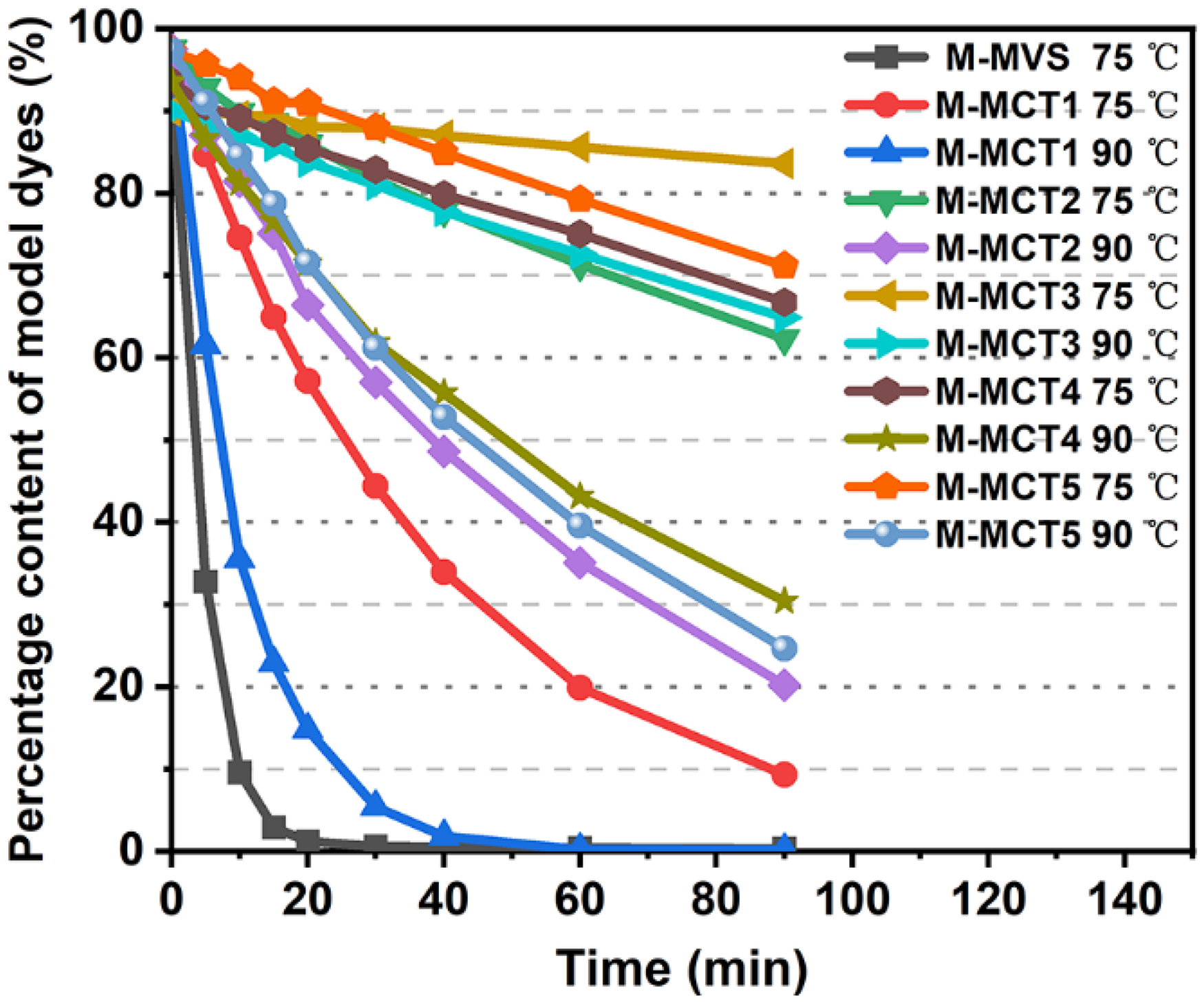
Plots of the percentage content as a function of time for model dyes in a glycine solution. M-MVS: model-monovinyl sulfone; M-MCT: model-monochlorotriazine.
Stability study of model dyes
The stability of the functional groups is also important in dyeing progress. To analyze their stability, all the model dyes were subjected to the same condition as the reactivity study, but without glycine. This stability study measured the hydrolysis of the model dyes at pH 6.86 and 90°C.
The plots of percentage content as a function of time for all the model dyes are presented in Figure 6. Both M-MVS and M-MCT1 hydrolyzed only 3% in 90 min at 90°C. This result was acceptable under these conditions, and the hydrolysis percentage in the actual dyeing progress would be lower because of the competition between amino groups in nylon fibers and hydroxyl ions. The other dyes barely hydrolyzed owing to their low reactivities. Notably, although the reactivity of M-MCT1 was only 0.121 times that of M-MVS, M-MCT1 still hydrolyzed ∼3%. Therefore, the hydrolysis rate constants of M-MVS and M-MCT1 were analyzed using a similar approach to the reactivity study. Owing to the existence of the phosphate buffer solution, the hydroxyl ion content remains nearly constant. Thus, the pseudo-first-order hydrolysis equation can be expressed as follows:

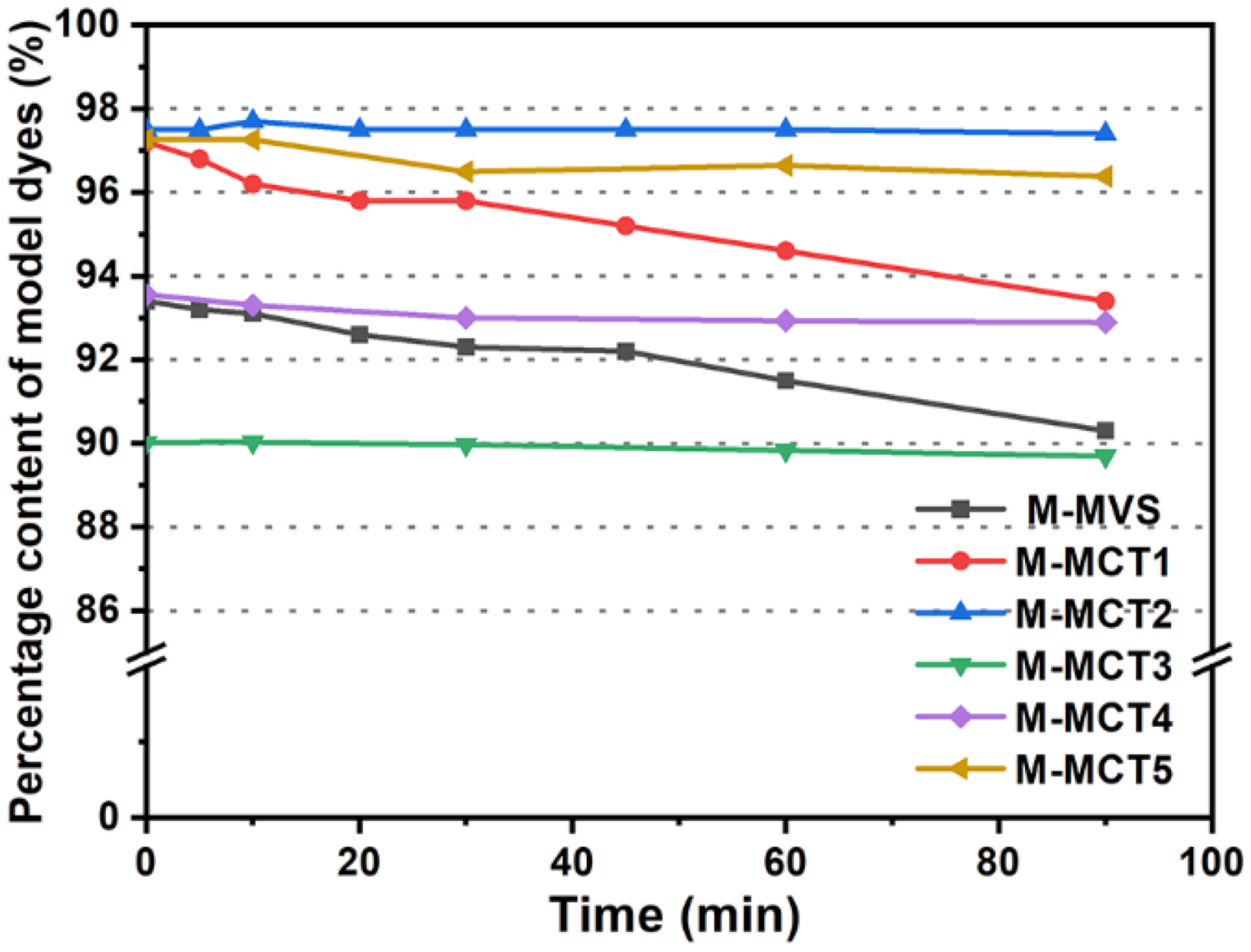
Plots of the percentage content as a function of time for model dyes in a pH 6.86 buffer solution. M-MVS: model-monovinyl sulfone; M-MCT: model-monochlorotriazine.
The plots of ln(
Pseudo-first-order hydrolysis rate of dyes

Structures and properties of LWS-MVSDs
In the reactivity and stability studies, vinyl sulfone demonstrated superior characteristics as a functional group for LWSRDs. Therefore, to synthesize four LWS-MVSDs with a large relative molecular mass, traditional dye intermediates, such as 3-methyl-1-phenyl-2-pyrazolin-5-one, J-acid and H-acid, were used as coupling components. The synthesis of LWS-MVSRY dyes involved ingenious utilization of the coupling compound comprising 3-aminobenzene sulfonic acid and 4-(ethylsulfurate sulfonyl) aniline as diazonium salts, which were coupled with 3-methyl-1-phenyl-2-pyrazolin-5-one (Figure 7).
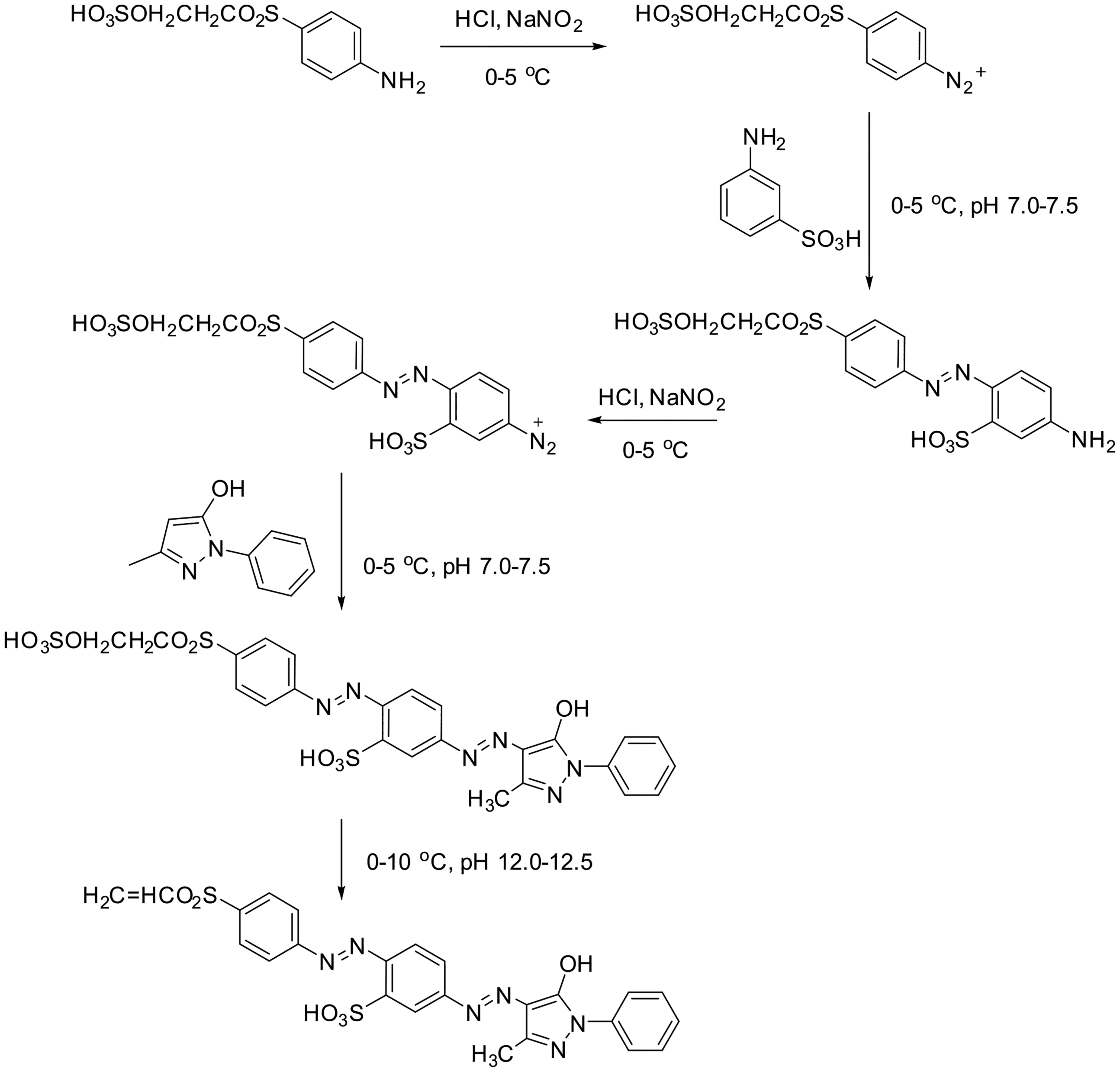
Synthesis of LWS-MVSRY.
As shown in Figure 8, LWS-MVSRO, LWS-MVSRR and LWS-MVSRB had a similar synthesis procedure. LWS-MVSRO and LWS-MVSRR were bisazo dyes derived from J-acid. The first coupling reaction was carried out between 4-(ethylsulfurate sulfonyl) aniline and J-acid, and the second diazonium salts were 4-butylaniline and 4-aminoacetanilide, respectively. The final step involved the vinylation reaction. LWS-MVSRB was synthesized by introducing 4-
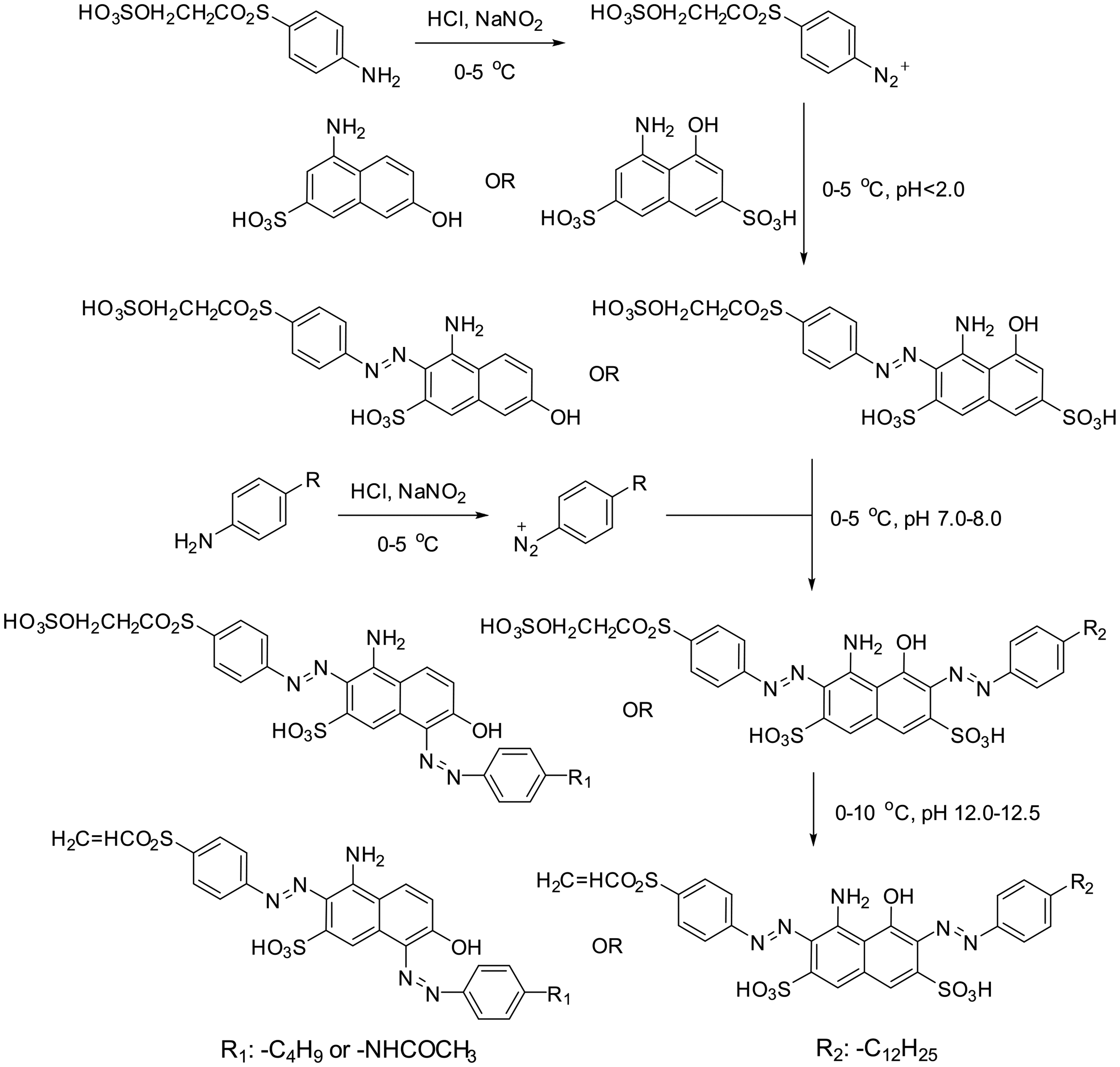
Synthesis of LWS-MVSRO, LWS-MVSRR and LWS-MVSRB.
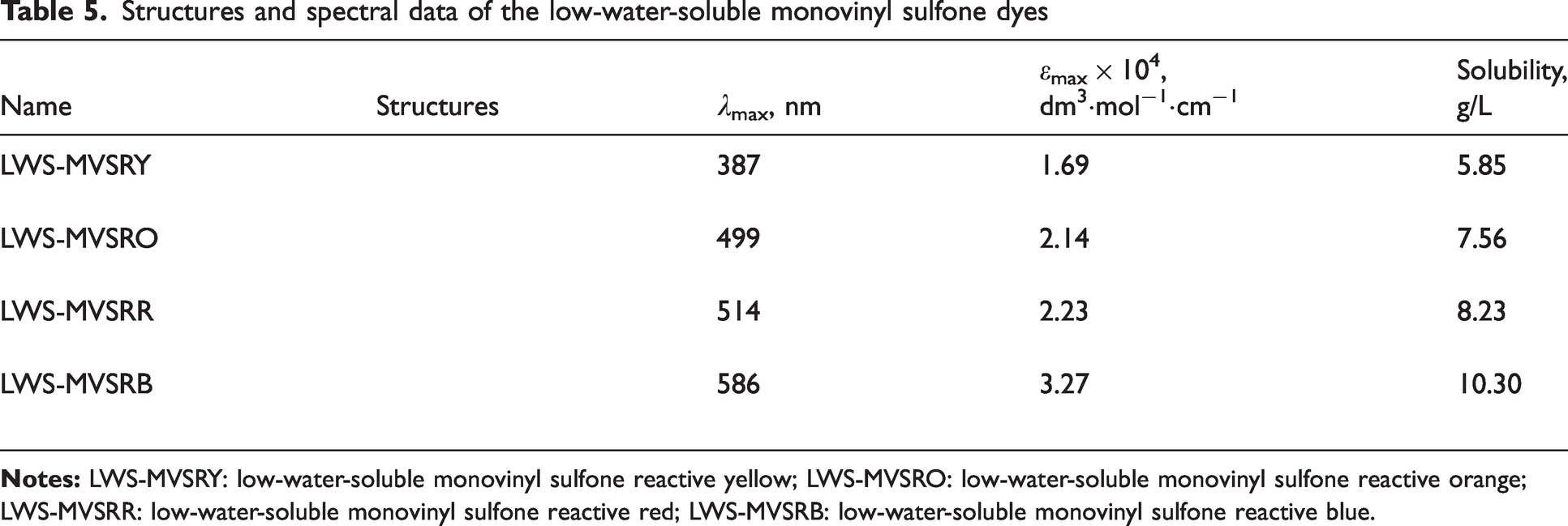
The structures and spectral data of the LWS-MVSDs are presented in Table 5. LWS-MVSRY exhibited a bright yellow color with a maximum absorption wavelength at 387 nm, accompanied by a molar absorption coefficient (
Structures and spectral data of the low-water-soluble monovinyl sulfone dyes
Dyeing results and color fastness of the M-MVS and LWS-MVSDs
Figure 9 illustrates the dyeing results of the M-MVS and LWS-MVS dyes [2% (o.w.f.)] on nylon 6 microfibers. The exhaustion of M-MVS was only 41.0%, which accounts for the low utilization of M-MVS. Furthermore, the fixation of M-MVS was only 36.6%, indicating a substantial wastage of M-MVS dyes. However, the reactivity of M-MVS was 88.0%, demonstrating the favorable affinity of vinyl for nylon 6 microfibers. Conversely, the four LWS-MVSDs performed excellent utilization. LWS-MVSRY achieved a fixation of 91.8% with a reactivity of 94.1%. The other three LWS-MVSDs displayed fixations of >97.9%. This exceptional performance was attributed to the ability of the LWS-MVSDs to effectively stain nylon 6 microfibers under neutral conditions and the high reactivity of vinyl sulfone in the LWS-MVSDs with amino groups on the nylon microfiber. Overall, the dyeing results of the LWS-MVSDs were highly satisfactory, with almost no colored wastewater produced during the dyeing process.
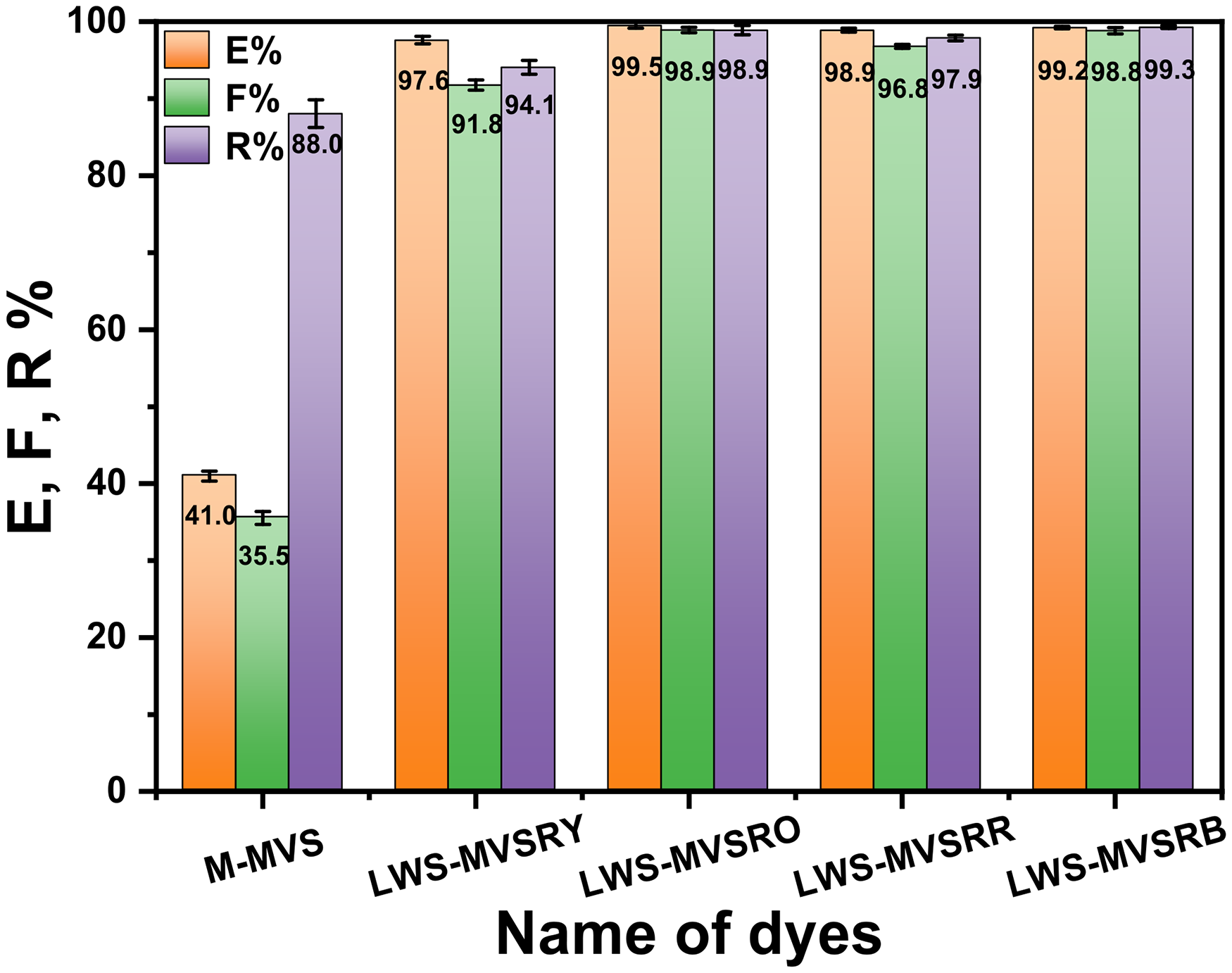
Dyeing results of nylon microfiber with the model-monovinyl sulfone (M-MVS) and low-water-soluble monovinyl sulfone dyes (LWS-MVSDs).
Digital photos of the dyed nonwoven nylon 6 microfibers are shown in Figure 10. As show in this figure, the four dyed fibers exhibited bright colors, but the

Photographs of nylon microfiber dyed with low-water-soluble monovinyl sulfone dyes (LWS-MVSDs).
Color fastness of the model-monovinyl sulfone (M-MVS) and low-water-soluble monovinyl sulfone dyes on nylon 6 microfibers
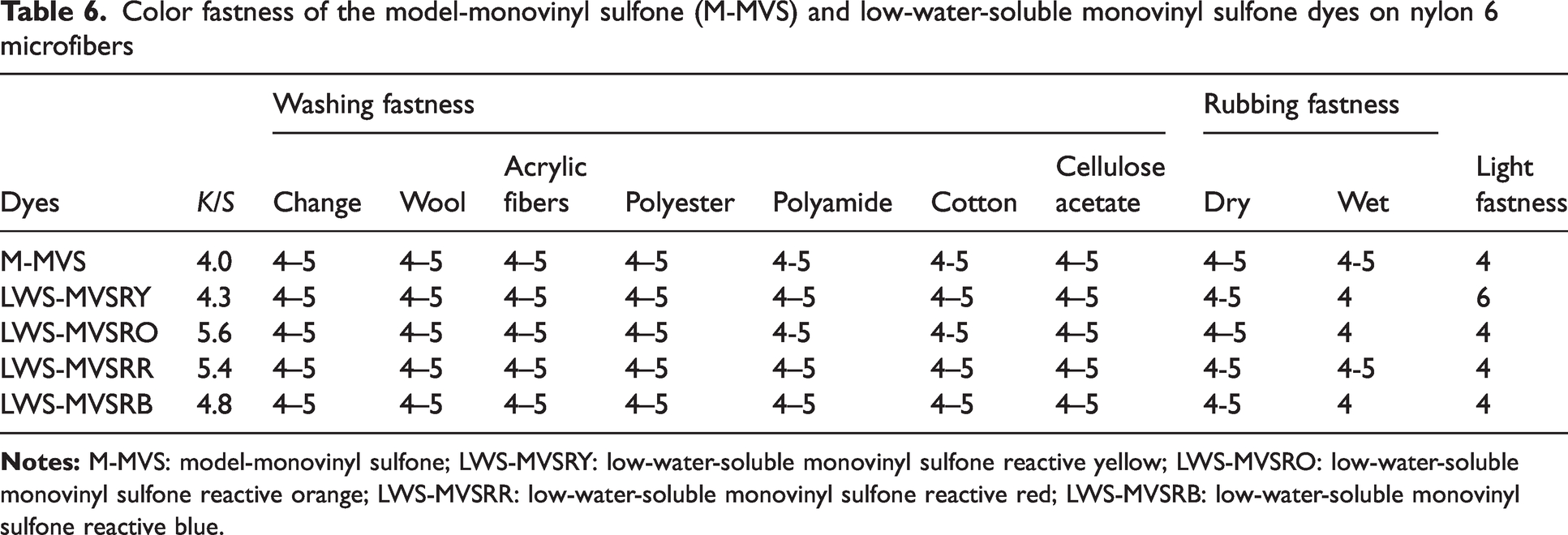
The LWS-MVSDs exhibited good dyeing results for nylon 6 microfibers; moreover, these dyes exhibited excellent color fastness with
Conclusions
LWSRDs offer a potential solution to enhance the low utilization of traditional reactive dyes on nylon fibers. To quantitatively evaluate a suitable functional group for LWSRDs, a M-MVS and five M-MCT red dyes were synthesized, possessing 4-substituents such as ethoxy, 4-sulfonated phenylamino, dimethylamino, amino and anilino. These dyes were used to simulate the fixation progress of nylon fibers using glycine as a model compound at 75°C and 90°C and a pH of 6.86. The reactivity study of the model dyes with glycine demonstrated that only vinyl sulfone and ethoxychloro-
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by the Program of the National Natural Science Foundation of China (22238002 and 21978041) and Dalian University of Technology (DUT22LAB610, DUT2022TB10).
