Abstract
Being the first commercially available wholly synthetic textile fiber which was introduced to the market in 1939, nylon fibers are one of the most frequently consumed synthetic fibers. Direct dyes were commercially produced in the early 20th century and had been the preferred method of dyeing of cellulosic fibers together with vat dyes until the discovery of reactive dyes. The most important property of direct dyes is their easy dyeing but with a disadvantage of building up weak bonds within the fiber. Chemical mordants were used mainly in the application of natural dyes to hold and retain the dye molecules in textile fibers. In this research, nylon 6,6 fabric was dyed with four direct dyes of different hues in the presence of mordants (eight metallic and one bio-mordant) to obtain better exhaustion, fixation, and coloration properties. Pre-mordanting, meta-mordanting, and post-mordanting methods were used and the dyeings were performed at 1% owf at pH 5. Percentage of dyebath exhaustion (%E), percentage fixation (%F), and overall fixation (%T) were calculated after pyridine extraction. Wash and light fastness tests were applied on the dyed samples. The results were discussed via color strength (K/S), %E, %F, %T, and CIELAB color differences (ΔEab*). The results revealed that mordant usage improved the coloration properties of direct dyes on nylon fabrics. The %E, %F, and %T results changed according to the independent variations of dye hue, mordant, and mordanting method. Certain mordants and mordanting method gave the best coloration and fastness results for the dyes under test.
The first commercially available wholly synthetic textile fiber nylon 6,6 was marketed in 1939 after the announcement of the discovery of “super-polyamides” by DuPont in 1938. “Nylon” was chosen as the general term for these new fibers. 1 The term “nylon” refers generally to any fiber-, bristle- or film-forming polymeric amide. 2 Polyamides are polymers whose structural units contain amide groups (—CONH—) as recurring integral parts of the main polymer chain. 1 A variety of polyamides are presently manufactured and are marketed under several different trade names; the two most important textile representatives being nylon 6,6 and nylon 6. Although generally quite similar, nylon 6,6 has a more compact and crystalline structure than nylon 6. Nylon fibers contain both terminal amino (–NH2) and carboxylic acid (–COOH) groups. The amino end groups impart substantivity to acid, metal-complex, direct, and reactive classes of dye; the fiber is also readily dyed with disperse dyes. In addition, cationic dyes may be applied to nylons with modified dyeing characteristics (i.e., basic-dyeable nylon). 2
The presence of terminal amino groups in nylon fibers imparts substantivity toward ionic dyes, especially acid dyes, direct dyes, and reactive dyes. Of the various types of dye that can be used to dye nylon fibers, acid dyes are the most popular, with 1:2 metal-complex (pre-metallized acid) dyes being used when high wet fastness is required.
1
However, the coloration of nylon is usually achieved with acid dyes. These dyes attack the nylon via ion–ion linkages between cationic, protonated, amino end groups of the nylon (
Direct dye is an anionic dye having substantivity mainly for cellulosic fibers, normally applied from aqueous dyebaths containing an electrolyte.5,6 Direct dyes derive their name from the fact that they can be applied directly to cotton (and other cellulosic fibers) owing to their inherent substantivity toward such fibers rather than indirectly via the use of a mordant (metal salt or tannin), such processes being commonly used until the introduction of synthetic dyes in the latter half of the nineteenth century. The first synthetic direct dye which was introduced in 1884 was Congo Red (C.I. Direct Red 28). Direct dyes are, characteristically, long, planar molecules that, invariably, are solubilized by sulfonate/sulfonic acid groups. As a consequence of their amphiphilic nature, direct dyes display a marked propensity to aggregate in aqueous solution, this dye aggregation is favored by high dye concentrations and by the addition of electrolyte (NaCl or Na2SO4) that is used to promote dye adsorption to direct dyebaths. Direct dyes furnish inexpensive pale to medium, bright shades of moderate/good light fastness but poor/moderate wet fastness, especially to washing, water, and perspiration. Marked improvement in the wet fastness of direct dyeings on cellulosic fibers can be achieved through the use of an after treatment of the dyed material with a cationic fixing agent. 6
The exhaust dyeing behavior of direct dyes on cellulosic fibers varies markedly depending on dye structure, which is of major significance with respect to compatibility in admixture; also, the dyes vary markedly in terms of the effect of added electrolyte on dye uptake. Such differences in dyeing behavior have received attention in terms of characterization of the dyeing properties of direct dyes on cellulosic fibers and resulted in the Society of Dyers and Colourists (SDC) classification of direct dyes that takes into account four parameters, namely the leveling (migrating) ability of the dyes, the influence of liquor ratio, as well as their response to both temperature and added electrolyte during exhaust dyeing. 6
SDC classification of direct dyes comprises Class A, Class B, and Class C dyes. Generally, dye–fiber substantivity increases in the order Class C > Class B > Class A as a consequence of increasing molar mass:sulfonate group ratio.6–8 The chemical potential of anionic dyes in dyebath is of paramount importance for the uptake of dye molecules by the fiber amorphous structure and the concept of chemical potential is introduced to help explain the transfer of direct dyes or other anionic chemicals from dyebath to the fiber, particularly in the presence of an excess of oppositely charged ions such as sodium cation, Na+, which is introduced by the addition of simple sodium salts. 7
Direct dyes are normally applied (or around) pH 7. Most of the direct dyes are resistant to dyeing conditions at pH 4–6 for dyeing blended fabrics and could be used in combination with disperse or acid dyes. Maximum exhaustion temperatures of direct dyes vary from 20°C to 100°C. However, most of the direct dyes are exhausted when dyeings are performed at the boil. 9
The affinity of dyes on nylon were studied mainly with acid dyes which are anionic dyes in order to obtain exact explanation of the dye–fiber system. Acid dyes have much higher substantivity to nylon than direct dyes because of their different molecular and solubilization properties. There is no exact data for the absorption of direct dyes on nylon because acid dyes have been much more preferred for dyeing nylon fibers than direct dyes. 8
The studies about dyeing properties of nylon fibers with anionic dyes other than acid dyes were mainly focused on the investigations of the dyeing properties of reactive dyes on nylon.10–21 In addition, in order to improve the dyeing results of reactive dyes, mordant usage possibilities were also investigated.22–25
Mordants have been used since ancient times and the term mordant comes from the Latin word “mordere” which means “to bite.” 26 Traditionally, mordants have been used as dye fixatives for natural dyes which were used to dye natural fibers. In other words, mordants were used to set (i.e., bind) natural dyes on natural fibers. A mordant is often a polyvalent transition metal ion, and one well-known and widely used example is chromium. 27 With the development of acid dyes which are dyes with anionic functional groups and with understanding the behavior of chromium ion with anionic dyes, mordant dyeing had begun in wool dyeing. The resulting coordination complex of dye and ion is colloidal and can be either acidic or alkaline. Mordants can be classified into three categories as metallic, tannins, or oil type. 28 The possible interactions among polyvalent metal mordant, anionic dye, and nylon structure under acidic conditions in fiber structure during dyeing in a dye bath is presented in Scheme 1.
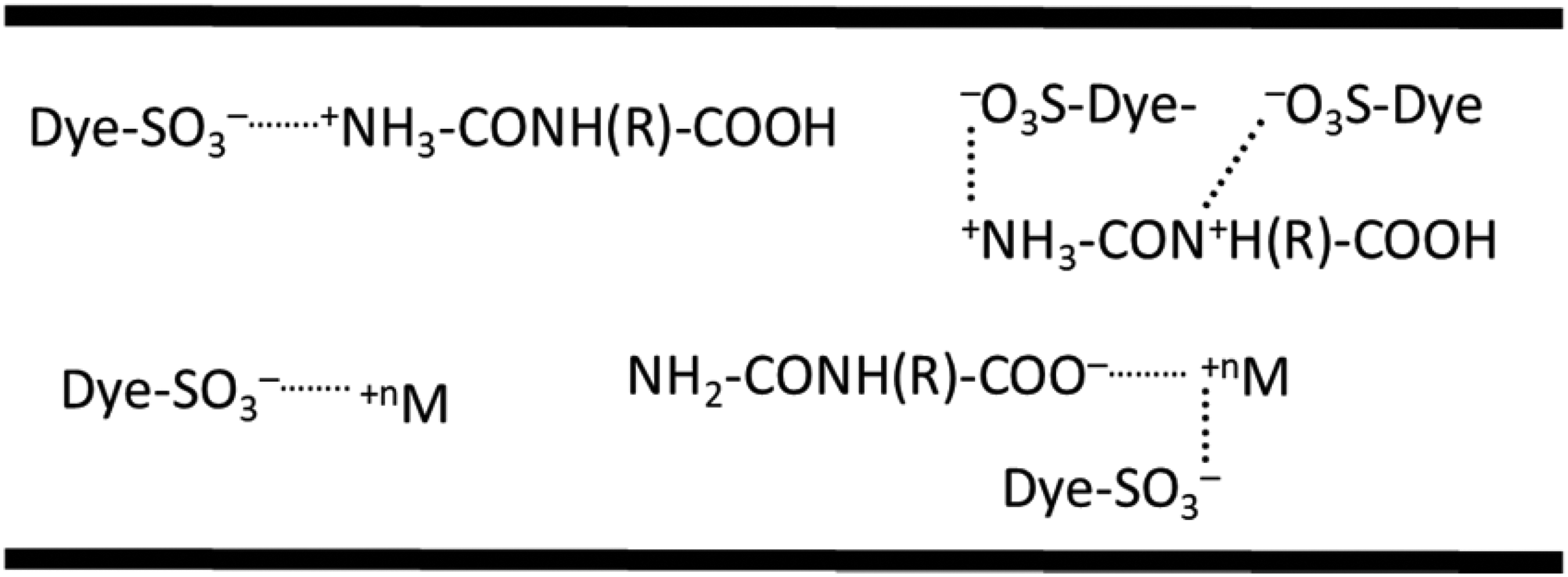
Possible interactions among polyvalent metal mordant, anionic dye, and nylon structure under acidic conditions in fiber structure during dyeing in a dye bath.
During dyeing, the mordant–dye complex can be created via pre-mordanting (applying the mordant first, then dye, later continuing dyeing), meta-mordanting (applying the dye and the mordant simultaneously and dyeing), or post-mordanting (adding the dye first and to perform dyeing the material, then mordant for complex formation). Dyeing results can also rely on the mordant chosen as the introduction of the mordant into the dye molecule and the electronic interaction will have a marked effect on the final color. Each dye can have different reactions to each mordant resulting different shades.
Mordant usage enhances the fastness properties of the colored fabrics by aiding in the absorption and fixing of dyes. The most well-known mordants are alum (potassium aluminum sulfate), copper sulfate, ferrous sulfate, potassium and sodium dichromate, zinc sulfate, and tannic acid. The metal ions of mordants can act as electron acceptors for electron donors to form coordination bonds with the dye molecules, making them insoluble in water. 29 Usage of alum is safe; however, dichromates are hazardous. The environmental impact of mordants was discussed and safer applications on different fiber materials with alternative mordants were researched.29–33
The research about usage of metallic mordants in direct dyeing of nylon fabrics failed and no useful scientific data about the subject were found during the literature research.
This research focused on the usage of eight metallic and one bio-mordant in direct dyeing of nylon fabrics. The purpose was to research the better dyeing, coloration and fastness properties with the usage of mordants with commercial direct dyes which were used for cellulosic dyeing. The results revealed that percent exhaustion (%E), percent dye fixation (%F), and overall dye fixation efficiency (%T) properties of direct dyes were improved depending on the type of mordant, and the selected mordanting method. Each of the selected direct dyes had its own best dyeing performances with different combinations of dye hue, mordant, and mordanting method. Success of dyeings performed varied with dye hue–mordant–mordanting method triplet.
Materials and method
Dyeing properties of commercial direct dyes for cellulosic fibers were investigated on nylon fabric for different mordants, dye hues, and three mordanting methods via absorbances of dyeing liquors and color measurements of the dyed samples. The results were reported as color strength (K/S), percentage exhaustion (%E), percentage fixation (%F), and overall percentage fixation (%T) in addition to color differences (ΔEab*) of the resultant dyeings after extraction, and wash and light fastness. The methodology which was presented and used successfully in the former papers was used.11–13,15–19,25
We used a 100% nylon 6,6 fabric woven from 310/140 (denier/filament number) yarns in warp (32 threads/cm) direction and from 150/72 (denier/filament number) yarns in weft (36 threads/cm) direction. The fabric had a 2/1 twill weave and had a unit weight of 165 g/m2. The fabric was scoured under mill conditions in a pretreatment bath (pH 9) including a nonionic washing agent (4 mL/L) at 60ᵒC for 60 min to remove the impurities. The fabric was dried and stentered in the mill at 195ᵒC for 60 seconds prior to dyeing.
Dyeing experiments were carried out using a laboratory type sample dyeing machine (IRDyer, South Korea). We dyed 5 g pieces of fabrics at a liquor ratio of 20:1 using stainless steel dye pots. Dyebaths contained fabric sample, dyestuff, pH chemicals, and mordants when necessary. Dyeing experiments were performed at a single concentration of 1% owf. Commercial direct dyes (Class B) of different hues were used in order to a better understanding of the experimental procedure in CIELAB color space. C.I. Direct Black 22 (C.I. 35435, multiazo class), C.I. Direct Blue 74 (C.I. 34146, Trisazo class), C.I. Direct Red 23 (C.I. 29160, double azo class) and a direct yellow dye (C.I. name and number was not allocated, no details were shared by the producing firm other than an azo class mix dye). All the dyes were used as obtained (in commercial form) and no purification was performed. The molecular structures of the dyes are presented in Scheme 2.
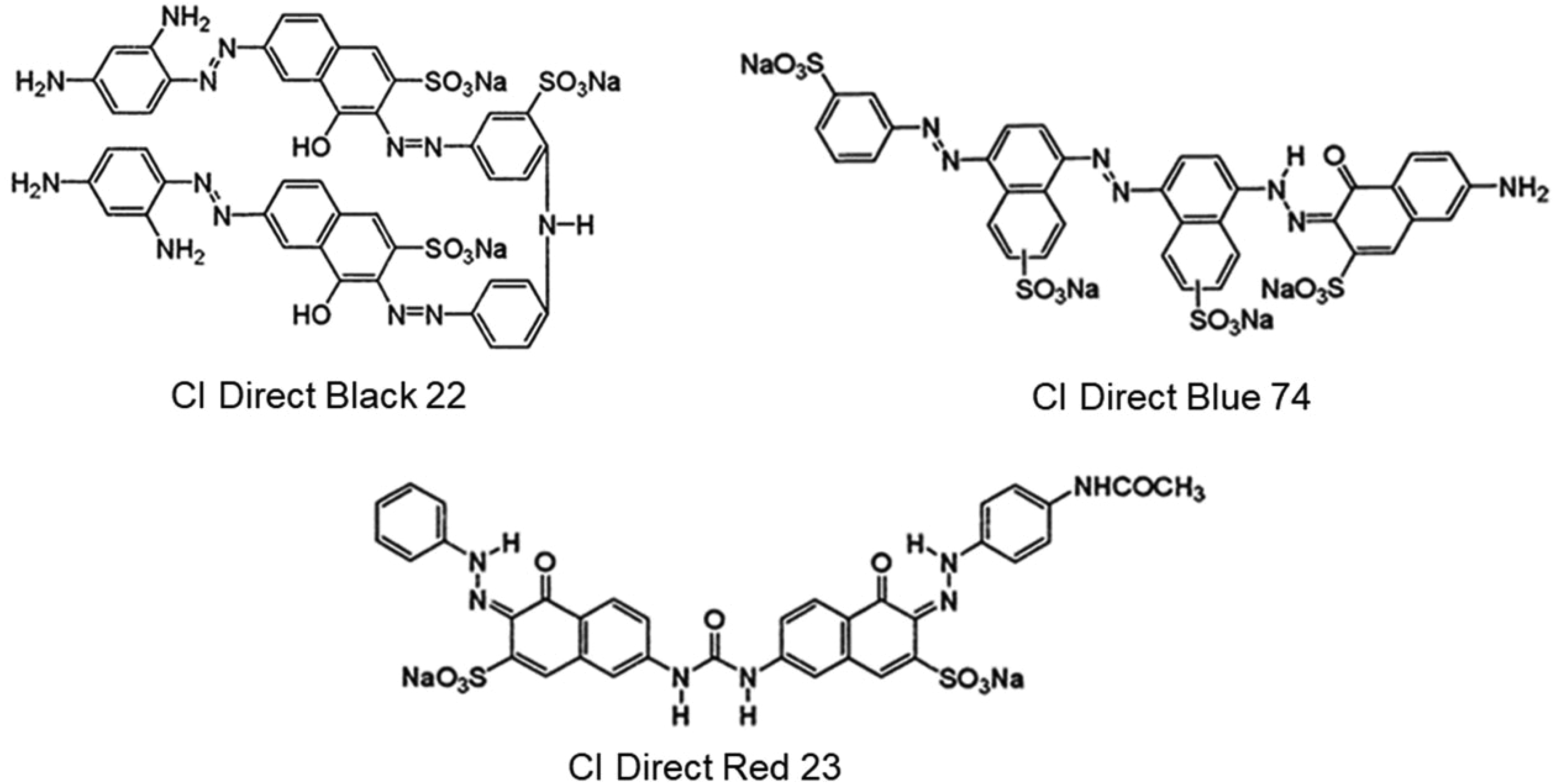
Molecular structures of the dyes.
Dyeing profiles (as mordanting methods) given in Figure 1 were used. Initial dyeing temperature was selected as 30ᵒC and a maximum dyeing temperature of 102ᵒC was used. In each dyeing process the temperatures were increased by 2ᵒC/min where necessary. Dyeings continued at 102ᵒC for 60 min and later the temperature was decreased to 40ᵒC by 4ᵒC/min. At the end of dyeing, the dyed fabric samples were removed and later rinsed in cold tap water at 100:1 liquor ratio for 10 min to remove the unfixed dye which remained on fiber surfaces. Dyed and rinsed samples were not treated further and the cleared samples were left to dry under laboratory conditions prior to color measurements, and further extraction and fastness tests.
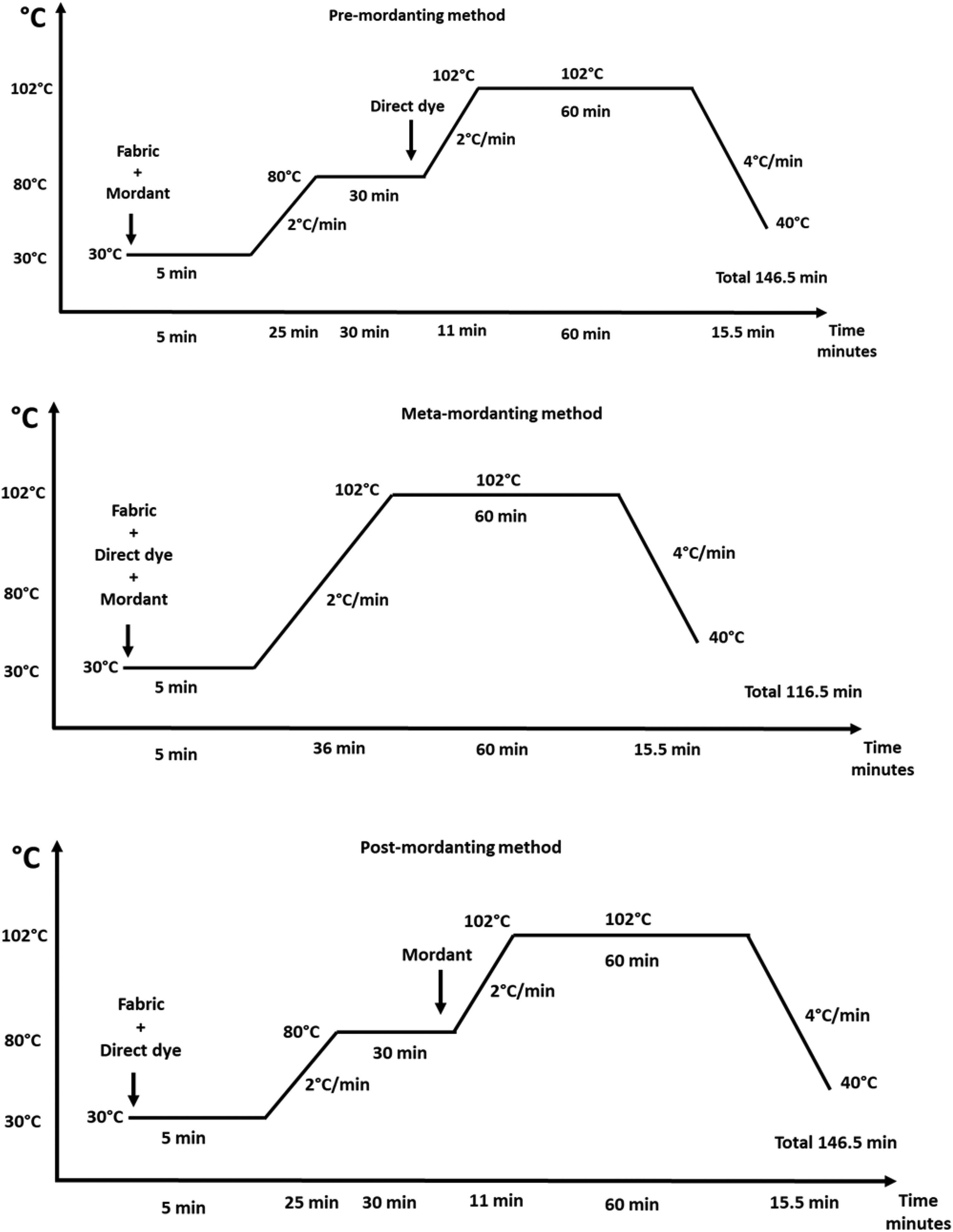
Time–temperature profiles of the dyeing experiments (mordanting methods).
All the dyeing experiments were carried out at pH 5 which was adjusted by acetic acid and sodium acetate buffer. The pH of the dyeing liquors was carefully checked and adjusted when necessary with a pH meter. All the mordants, metallic mordants, and bio-mordant used in the experimental research were stable in weak acidic medium.
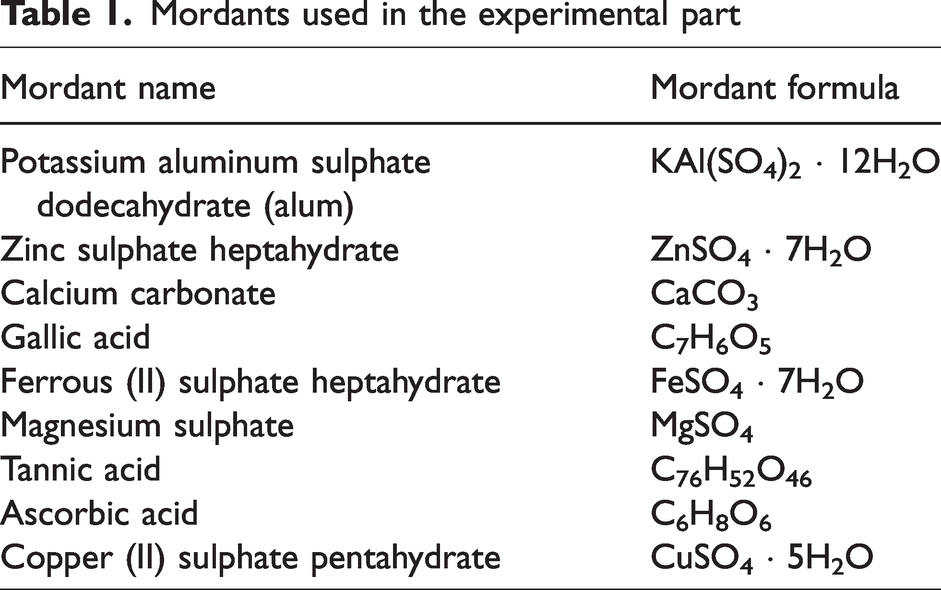
The mordants, which were used in the experimental part, are listed in Table 1. They were supplied in laboratory grade (sealed laboratory pack) and used as received from their packs with only very short contact with air. Among the mordant chemicals presented in Table 1, tannic acid is a bio-mordant while the others are referred as chemical/metallic mordants.
Mordants used in the experimental part
Wash fastness tests were performed according to ISO 105-C06:2010 A2S test (4 g/L ECE reference detergent WOB, 1 g/L sodium perborate, 150 mL liquor volume, 40°C, 45 min). 34 The wash fastness of fabric samples was evaluated with color measurements in order to present a clearer understanding.
Light fastness tests were performed according to ISO 105-B02:2014 test. 35 The test lasted for 20 hours in order to observe the behavior of the fabrics under short-term exposure conditions as nylon fiber is prone to the effects of light. The duration of 20 hours could be matched to L6 to L5 (blue wool standard) fading exposure. During the light fastness test, the test specimens were cut into 4 × 10 cm2 dimensions and the samples were placed in the exposure chamber attached to the appropriate sample holders. Similar to the wash fastness test, the light fastness of fabric samples was evaluated with color measurements in order to present a clearer understanding. Neither gray scale (wash fastness) nor blue wool standard (light fastness) comparisons were carried out. Objective color results were tried to be obtained by spectrophotometric measurements and complimentary software.
Color measurements on the dyed samples were performed by using a reflectance spectrophotometer (KonicaMinolta 3600d, Japan) between 400 and 700 nm under D65/10ᵒ illuminant and specular component included (SCI) mode. Fabric samples were folded twice to achieve a four-layer fabric thickness. Four reflectance measurements were performed at different positions (90° of sample rotation) on the dyed fabric samples and the average percentage reflectance was recorded. Color coordinates (L*, a*, b*, C*, and h°), color strength (Kubelka-Munk values, K/S; λmax for each dye: black dye 630 nm, blue dye 620 nm, red dye 540 nm, yellow dye 460 nm) and color difference values (ΔEab*; CIELAB 1976) were obtained from the color assessment software.
Color properties of the nylon fabric prior to dyeing were as follows: L*, 91.06; a*, –0.34; b*, 1.13; C*, 1.18; h°, 107.00; YI Yellowness Index E313-15, 1.62; CIE WI Whiteness Index, 73.29; CIE Tint, 0.5; % Opacity, 4.70; K/S 0.05 at 400 nm; all the measurements were taken and reported under D65/10°; YI C/2°).
The extent of dye exhaustion was determined by transmittance measurements. The absorbance of each dyebath solution before and after dyeing process was measured using 1 cm quartz cell housed in an ultraviolet–visible (UV-VIS) transmittance spectrophotometer (Shimadzu UV-1280, Japan) at the λmax of each dye after appropriate dilution. The percentage of dyebath exhaustion (%E) was calculated using
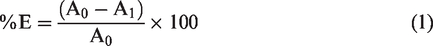
Determination of the degree of dye fixation was performed via Soxhlet extraction by aqueous alkaline pyridine solution and color measurement. A sample of 1 g of dyed fabric was immersed in 150 cm3 of aqueous 20% (prepared by using distilled water) pyridine solution. The temperature was rapidly raised to the boiling point and treatment continued at the boil under reflux for at least 6 hours until no further dye was removed from the fabric samples.
The stripped dyeings were thoroughly rinsed in tap water for 10 minutes (at 100:1 liquor ratio) and allowed to dry under laboratory conditions. The percentage fixation of the dye which exhausted, %F, was determined according to

The following equation represents the overall percentage fixation, %T, (overall fixation efficiency of dye) obtained from percentage of dyebath exhaustion (%E) and percentage fixation of the exhausted dye (%F):

Color difference (ΔEab* or ΔE*) values were calculated according to the CIELAB1976 color difference formula
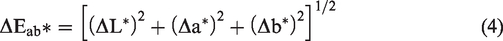
The dyeing profile of meta-mordanting (Figure 1) was used to perform the dyeings which did not contain any mordant (blind dyeing-dyeing without mordant), i.e. conventional temperature-time profile for nylon direct dyeing. The results were presented with the term “Blind Dyeing-Without Mordant” in the Tables, Figures and where necessary.
The pictures from the selected dyeings are presented in Plate 1. Successful colorings could be observed.
Results and discussion
One of the most well-known and well-studied advantages of nylon fibers in wet processing is their ability to be dyed with different dye classes, i.e., mainly with dyes which disassociate into anionic colored molecules in aqueous medium. 1 These dyes could be summarized as acid (including metal-complex dyes), reactive, and direct dyes. Although nylon fibers could also be dyed with disperse dyes because of their thermoplastic character, these dyes are mainly used to avoid barré dyeing problems when necessary. The main dye class which is used in coloration of nylon fibers/fabrics is acid dyes (including 1:2 metal-complex [pre-metallized] dyes). However, dyeing and coloration properties of other dye classes have also been studied but could not find broad industrial experience and usage.
Direct dyes are readily water soluble anionic dyes with their distinguishing property of having large planar molecules with high affinity especially to cellulosic fibers. Their planar structures are their advantages to place themselves parallel to the polymer macromolecules in the fibrous-amorphous structure of fibers, e.g., cotton fibers. In this way, the direct dye molecules could establish mainly hydrogen bonds and van der Waals forces within polymer structure. However, these dyes could not establish strong bonds in any fiber structure which is an unfavorable property for their further behaviors in water medium during domestic usage, e.g., laundering. In addition, it is not possible to confirm a hydrogen-bond network between all direct dye molecules and polymer molecules in the fibrous structure of the textile fibers. Although the dye molecules can diffuse into the polymer structure, they could not form efficient bonds within the amorphous material and they can be considered as loosely bonding/staying molecules. These loosely bonded-staying or unbonded molecules are prone to the effects of water medium and with the water absorption of fibers, e.g., in laundering, they could easily become water soluble again and migrate outside the fibers. In this way, the loss of dye molecules could lower the wash fastness properties of the dyed goods and could result heavy staining in the adjacent fabrics/goods. This could result color loss or color change during and after laundering.
Metallic mordants have been used to fix the natural dyes in textile fibers since ancient times. Their success in complexity with natural dyes are well known but as it could be easily understood that every metallic mordant is not successful with every natural dye. The same comment could be also possible when metallic mordants are used in dyeing of textile materials with synthetic dyes, e.g., direct dyes.
In this research, the application of metallic mordants in direct dyeing of nylon fabrics has been investigated experimentally and the success of mordanting the direct dyes in nylon fiber structure was researched. The main target was to get better wash fastness results and to consider direct dye usage with metallic mordants in nylon coloration to obtain better fastness properties. These mordants could retain the loosely bonded anionic direct dyes in fibers, which could results an increase in wash fastness properties. In addition, full exhaustion of the dye molecules in the dyebath could be achieved.
Exhaustion, fixation, and overall percent fixation efficiency values
Nylon 6,6 fabric samples were dyed at 1% owf dyeing concentration at pH 5 level with four direct dyes with different hues (red, blue, yellow, and black) via three mordanting methods (pre-, meta-, and post-mordanting). Color strength (K/S), percent exhaustion (%E), % dye fixation (%F), and overall percent fixation efficiency (%T) of the dyeings are presented in Tables 2–5. The corresponding results of blind dyeing (without mordant usage, performed according to the meta-mordanting dyeing profile in Figure 1) were presented above the corresponding values of each direct dye (Tables 2–5).
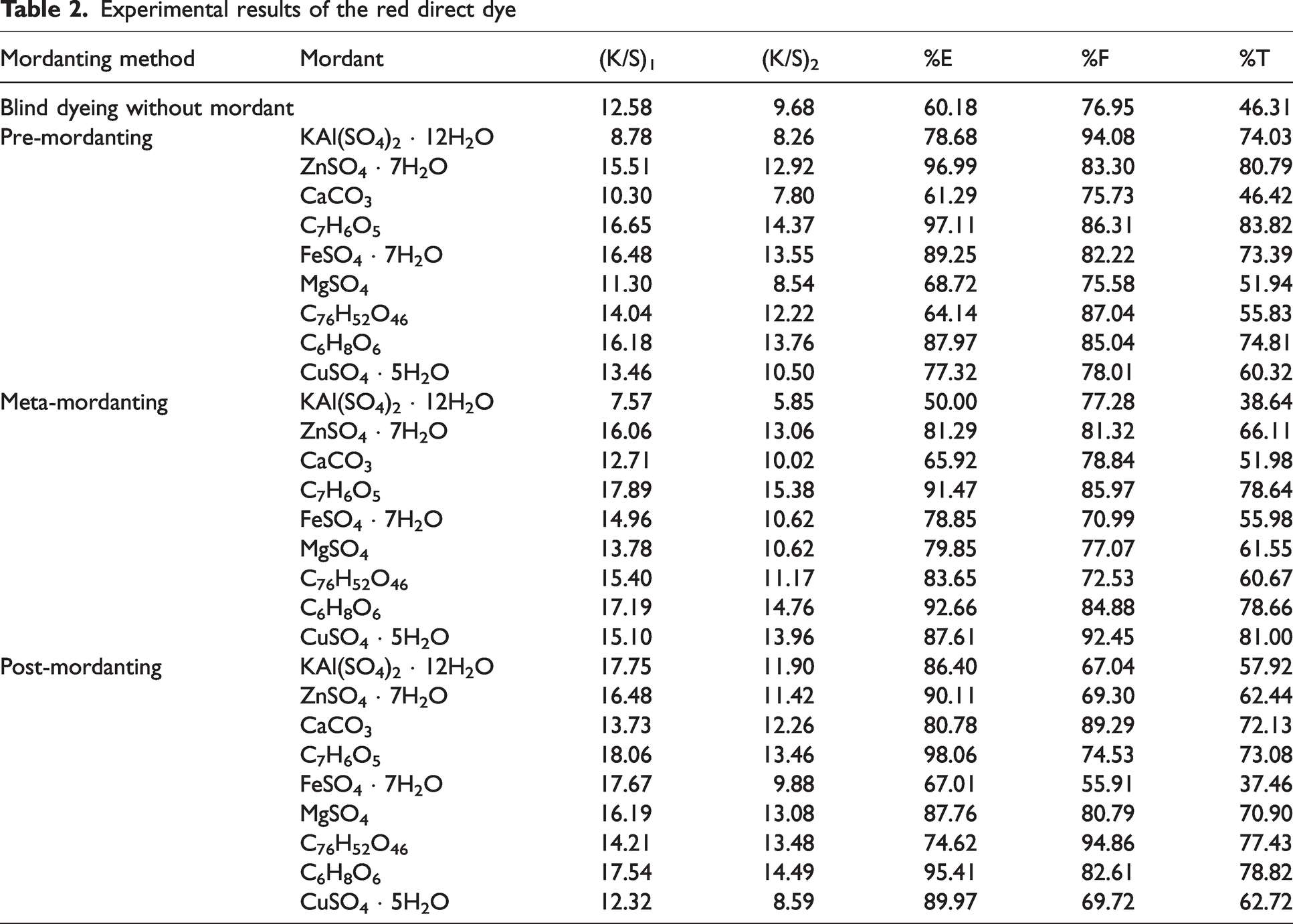
Experimental results of the red direct dye
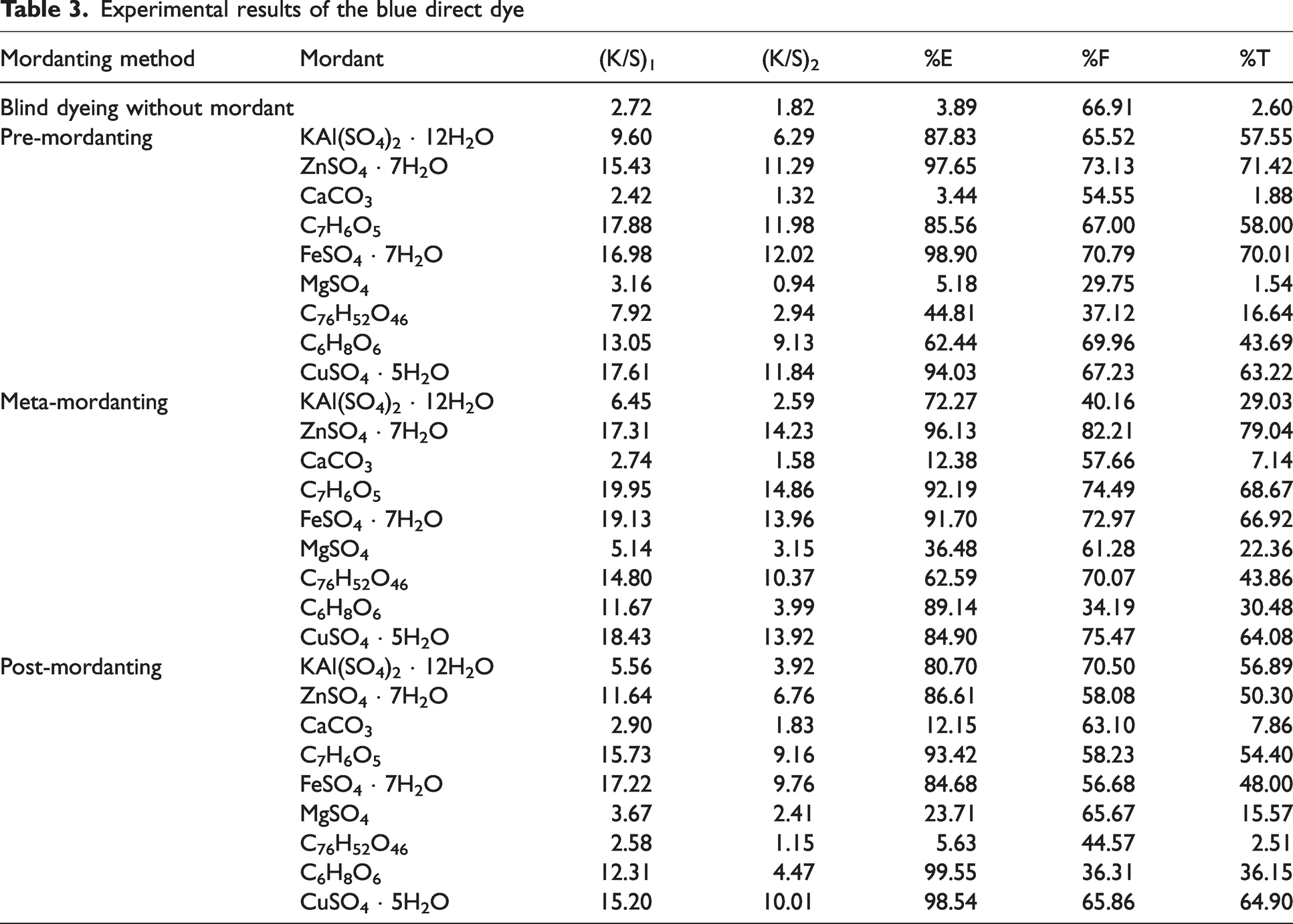
Experimental results of the blue direct dye
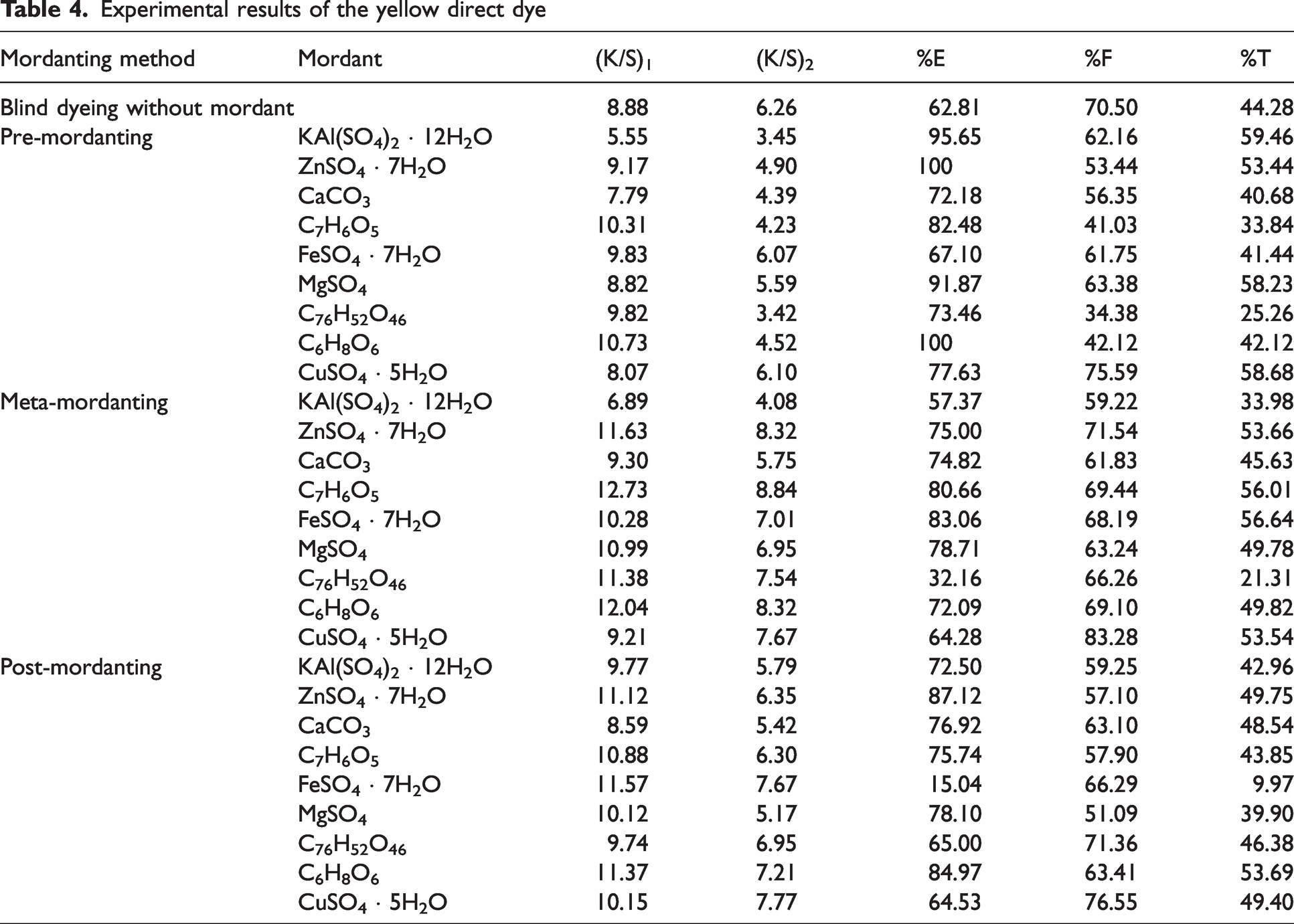
Experimental results of the yellow direct dye
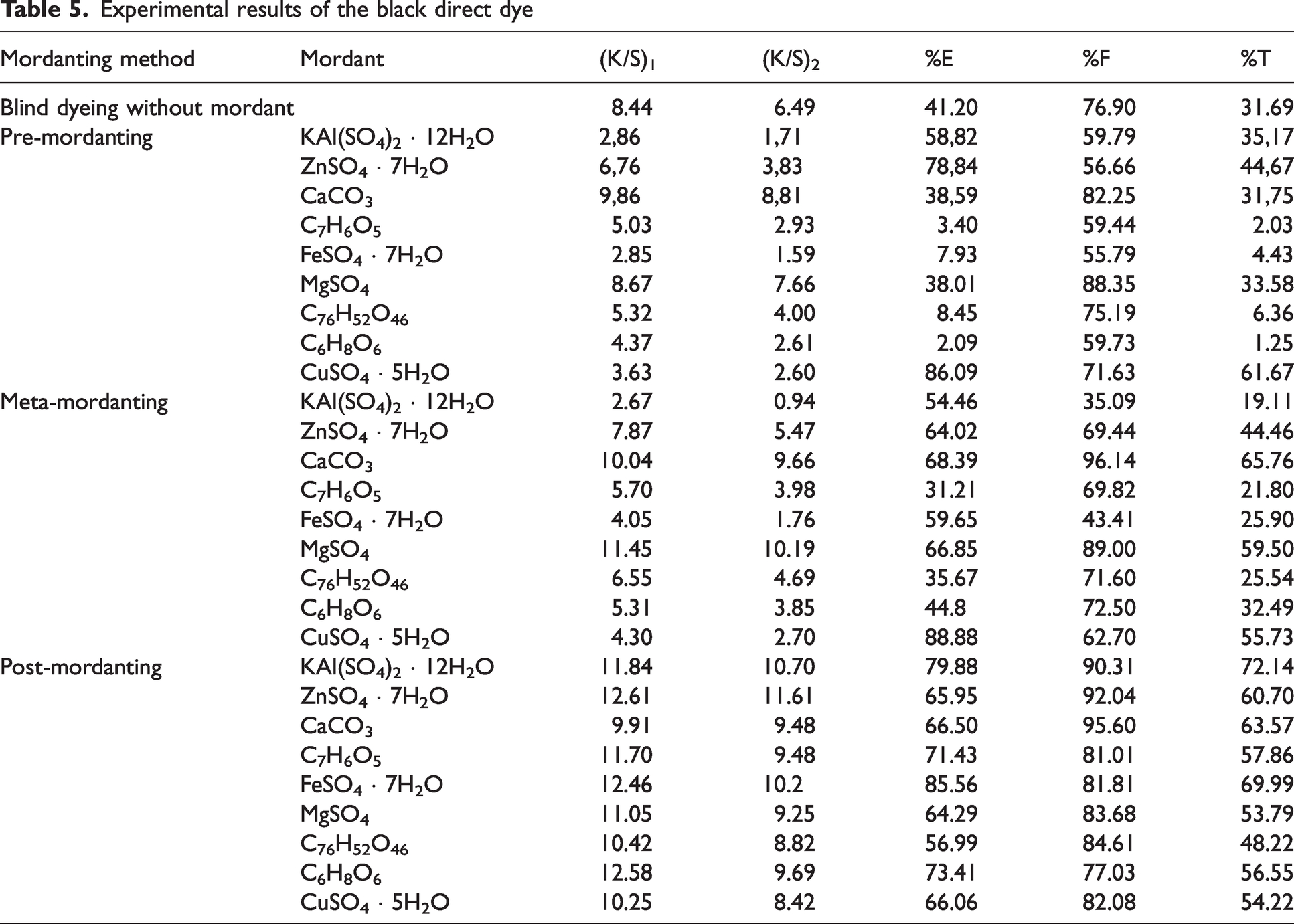
Experimental results of the black direct dye
The success of the dyeings is first shown by the high %T values. High %E values indicate that much of the dye in the dyebath was taken by the fibers but %E values could not give information about fixation. High %F values indicate that the dye molecules diffused and later they were well retained in the fiber mass whatever the %E was. High %E values could produce low %F values and low %E values could produce high %F values depending on the nature of the dyeing system besides dyeing parameters such as the dye itself, % owf dyeing, temperature–time profile, duration at maximum dyeing temperature, pH stability of the dyebath, material properties, etc. However, high %T values could be obtained only if the dye–fiber system had resulted in high %E and %F values.
Color strength (K/S) values before [(K/S)1] and after [(K/S)2] pyridine extraction besides percentage exhaustion (%E), percentage fixation (%F), and overall percentage fixation (%T) of the red direct dye (C.I. Direct Red 23) were presented in Table 2. The highest %E and %T values were obtained in pre- and post-mordanting methods. The highest %F values were obtained in pre- and meta-mordanting. In pre-mordanting method dyeing in the presence of zinc sulphate, gallic acid, ferrous II sulphate, and ascorbic acid presented the highest K/S, %E, and %T values while dyeing without mordant and dyeing with calcium carbonate presented the lowest %E and %T. All the mordants showed very high %F values while alum presented the highest. The very high %F values indicated that whether high or low %E values obtained, the build-up of the dye–mordant complexes were well retained in the fiber structure. The low %T values were obtained because of the low %E of the dyeings. Pre-mordanting with gallic acid resulted in the highest K/S values before and after pyridine extraction, i.e., (K/S)1 and (K/S)2, in addition to the highest %E and %T. All the mordants presented higher %E and %T values than blind dyeing (dyeing without mordant).
When the results obtained in meta-mordanting method were considered in Table 2, it was observed that similar to the results obtained in pre-mordanting, almost the highest K/S, %E, and %T values were obtained in dyeing with gallic acid and ascorbic acid. However, dyeing with copper sulphate also resulted very similar %E and higher %T values when compared with gallic and ascorbic acids. Different from the results presented in pre-mordanting, alum showed the lowest %E and %T in meta-mordanting method. The %F values were high but not as high as those obtained in pre-mordanting. In addition, similar to pre-mordanting, the highest (K/S)1 and (K/S)2 values were obtained by meta-mordanting with gallic acid.
In dyeing with the red direct dye according to post-mordanting method, similar to the results presented in the former two methods, the highest K/S, %E, and %T values were obtained with dyeing with gallic acid and ascorbic acid. However, different from the former methods, tannic acid (a bio-mordant) also resulted in high %E, %F, and %T. The lowest %E, %F, and %T values were obtained in post-mordanting with ferrous II sulphate. In addition, great numerical differences were obtained between (K/S)1 and (K/S)2 values in post-mordanting which revealed that the dye–mordant complex was accumulated on the fiber surfaces but could not diffuse well into the fibrous volume of the fibers. Also, lower %F values than the two former methods accompanied these results showing depletion of dye molecules to the bulk solution. However, due to the lesser extent of dye–mordant complex diffusion into the fibrous bulk, i.e., much lower (K/S)2 than (K/S)1, the %T values decreased.
An overall consideration of the results presented in Table 2 implied that any mordanting with gallic acid and ascorbic acid with the red direct dye under examination resulted in high percentage exhaustion (%E), high fixation (%F), and high overall percentage fixation (%T). Almost all the mordanting methods with mordants gave better fixation results than dyeing without mordant. In addition, the mordants presented their individual relations with the red direct dye depending on the mordanting method. For example, alum presented very poor values in meta-mordanting but excellent results in pre-mordanting. The same conclusion could also be given for ferrous II sulphate for post-mordanting and pre-mordanting. Tannic acid, being a bio-mordant, performed the best with the red dye in post-mordanting. Almost all the lowest %T values were obtained in meta-mordanting while copper sulphate performed best with this procedure.
Color strength (K/S) values before ((K/S)1) and after ((K/S)2) pyridine extraction besides percentage exhaustion (%E), percentage fixation (%F), and overall percentage fixation (%T) of the blue direct dye (C.I. Direct Blue 74) were presented in Table 3. Showing great differences from the results of the red direct dye presented in Table 2, blue direct dye produced very poor percent exhaustion (%E) and overall percent fixation (%T) when applied without mordant. This difference could be attributed to the molecular differences between the two dyes. In addition, some of the mordants presented similar poor results for the blue direct dye in the three different mordanting applications. The highest %E, %F, and %T values were obtained in pre- and meta-mordanting methods. In the pre-mordanting method, zinc sulphate, alum, gallic acid, ferrous II sulphate, and copper sulphate resulted in very high %E results but only zinc sulphate and ferrous II sulphate resulted in corresponding high %F and %T values. Pre-mordanting with calcium carbonate and magnesium sulphate resulted in very poor %E and %T values similar to dyeing without mordant. In addition, tannic acid presented the lowest %F values because of the very low (K/S)2 than (K/S)1 although its %E was moderate. When compared with the results presented in Table 2 for the red direct dye, gallic acid and ascorbic acid presented moderate %E and %T values.
The values obtained with meta-mordanting method showed a much more complex relation with regard to K/S, %E, and %T. The highest %E, %F, and %T values were obtained by meta-mordanting with Zinc sulphate. However, Gallic acid, Ferrous II sulphate and Copper sulphate resulted in high %E and %F but moderate %T values. Also, (K/S)2 values of the dyeings with these mordants were much lower than their corresponding (K/S)1 values. Ascorbic acid presented a very high %E but poor %F which led to low %T. These results implied that although a very high exhaustion was obtained from the dyebath during meta-mordanting, much of the dye–mordant complexes were adsorbed on fiber surfaces but could not diffuse into the fibrous bulk. They were moved by pyridine extraction. The great numerical differences between (K/S)1 and (K/S)2 values, e.g., gallic acid, ferrous II sulphate, and copper sulphate, revealed this result. The highest (K/S)1 results were obtained in meta-mordanting indicating darker dyeings were obtained than the other two methods. A comparison of the (K/S)2 values showed that darker shades were obtained when meta-mordanting method was applied and meta-mordanting with gallic acid produced the darkest shade while meta-mordanting with calcium carbonate produced the lightest shade. Similar to the results obtained in pre-mordanting, alum, calcium carbonate, and magnesium carbonate presented the lowest %E, %F, and %T.
In post-mordanting, calcium carbonate and tannic acid resulted very poor %E and %T values similar to the corresponding values presented in pre-mordanting. Similar to the values presented in pre- and meta-mordanting, very high %E but moderate %F values and %T values were obtained in post-mordanting with alum, zinc sulphate, gallic acid, ferrous II sulphate, and copper sulphate. The highest %E but the lowest %F were obtained with ascorbic acid almost similar to the corresponding results of this mordant in meta-mordanting. Almost all the dye was exhausted from the dyebath in the presence of ascorbic acid. When compared with its (K/S)1 value, the (K/S)2 value of this mordant was very low which led to poor %F and %T. The highest (K/S)2, %E and %T values were obtained in post-mordanting with copper sulphate. Tannic acid presented the lowest (K/S)2, %E, and %T values in post-mordanting of the blue direct dye.
An overall consideration of Table 3 showed that metallic mordants presented distinctly different dyeing behaviors when dyeing with the blue direct dye under investigation. Although some mordants, i.e., gallic acid, ferrous II sulphate, ascorbic acid, and copper sulphate, had high (K/S)1 values which indicated effective depletion of the dye molecules from the dyebaths, low (K/S)2 values accompanied them resulting in low %F and low overall fixation (%T). When the results presented in Table 2 for the red direct dye was compared with the corresponding ones presented in Table 3, it was observed that the two dyes were compatible with different mordants in different mordanting methods and a common conclusion could not be given. Tannic acid being a bio-mordant presented acceptable %E, %F, and %T results when applied with red direct dye but its corresponding results with the blue direct dye were very poor. For the blue dye under investigation via three mordanting methods, only zinc sulphate, ferrous II sulphate, and copper sulphate presented consistent %E, %F, and %T results.
Color strength (K/S) values before [(K/S)1] and after [(K/S)2] pyridine extraction besides percentage exhaustion (%E) and overall percentage fixation (%T) of the yellow direct dye (mix dye; no C.I. name was allocated) were presented in Table 4. Similar to the values of red direct dye presented in Table 2, yellow direct dye presented moderate %E, %F, and %T values in dyeing without mordant. The highest %E, %F, and %T values were obtained in pre-mordanting and then in meta-mordanting methods. However, %E, %F, and %T values obtained in post-mordanting method were correspondingly close to each other in their individual separation; the only exception was the values obtained with Ferrous II sulphate. The similarity of the values of the yellow direct dye presented in Table 4 with the corresponding values presented in Table 2 for the red direct dye and in Table 3 for the blue direct dye was that the (K/S)1 and (K/S)2 values showed distinct numerical differences in their corresponding subclasses. The distinct differences of these color strength values in subclasses were mostly similar to the corresponding values for the blue direct dye in Table 3.
In pre-mordanting method, although %E of alum, zinc sulphate, gallic acid, magnesium sulphate, and ascorbic acid were near maximum, i.e., even 100% (%E) for zinc sulphate and ascorbic acid, their corresponding %F and %T values were moderate. These results could be easily matched by the fact that when compared with (K/S)1 values, (K/S)2 values were much lower. None of the mordants showed very poor %T with the exception of gallic acid, ferrous II sulphate, ascorbic acid, and tannic acid (a bio-mordant). As could be easily observed, these results produce a contradiction with the corresponding values of these mordants in Table 2 for the red direct dye. Magnesium sulphate, which presented moderate to good %T with red direct dye in Table 2 but poor %T with the blue direct dye in Table 3, presented one of the best %T values in pre-mordanting with the yellow direct dye.
A comparison of pre-mordanting and meta-mordanting method revealed that the highest K/S values were obtained in dyeing with gallic acid and ascorbic acid. In addition, %E and %T values of gallic acid was among the highest. When these values were considered, it was observed that gallic acid–dye complex stayed on the fiber surfaces but the complex was easily extracted with pyridine extraction after pre-mordanting, i.e., very low (K/S)2 value. However, gallic acid-dye complex diffused into the fibrous bulk in meta-mordanting producing a high (K/S)2 value. As a result, a much higher %T was obtained in meta-mordanting than in pre-mordanting for gallic acid.
The highest (K/S)1 and (K/S)2 values were obtained with zinc sulphate, ferrous II sulphate, and ascorbic acid in post-mordanting. In addition, zinc sulphate and ascorbic acid presented the highest %E values. However, ferrous II sulphate had the lowest %E value so that it also derived the lowest %T. This was an interesting result because although ferrous II sulphate resulted the lowest %E, the post-mordant dyed sample had the highest color strength values. The results of Ferrous II sulphate implied that although a low exhaustion was obtained, the metal–dye complex on the fiber surfaces were strongly bonded and formed complexes with a high tinctorial strength. A dark color was obtained although overall percentage fixation (%T) was very low. Another exceptional result was obtained with magnesium sulphate in post-mordanting. Magnesium sulphate presented very high %E and %T values (and moderate %F values) in pre- and meta-mordanting methods. However, because of its comparatively low (K/S)2 value in post-mordanting, a low %T was calculated. These changing results of the metallic mordants with the direct dyes and applied mordanting methods showed the complex relations among them because it becomes impossible to derive an exact conclusion for any mordant and with any mordanting method.
An overall discussion of Table 4 implied that post-mordanting of the yellow direct dye process level results for all the mordants, except ferrous II sulphate. However, ferrous II sulphate produced moderate to good %E, %F, and %T results in pre- and meta-mordanting. An opposite coloration behavior was obtained when magnesium sulphate was used as mordant. Full exhaustion of dyes from the dyebath was obtained, e.g., zinc sulphate and ascorbic acid in pre-mordanting, but because of the poor penetration of the dye–mordant complex into the fibrous structure, [(K/S)2] values, the corresponding high %F and %T values could not be obtained. When compared with the corresponding results of the red direct dye in Table 2 and blue direct dye in Table 3, the %T values obtained with yellow direct dye were moderate and did not show great numerical differences among the mordants as it was obtained for the blue direct dye in Table 3. In addition, if an average could be considered, the %T values obtained with the yellow direct dye were lower than the ones obtained with the red direct dye.
The results in Tables 2–4 for the red, blue, and yellow dyes revealed that only ascorbic acid, zinc sulphate, and copper sulphate presented consistent %E, %F, and %T results as the hues and mordanting methods changed. The %E, %F, and %T values of these mordants were always among the best in corresponding tables.
Color strength (K/S) values before ((K/S)1) and after ((K/S)2) pyridine extraction as well as percentage exhaustion (%E), percent fixation (%F), and overall percentage fixation (%T) of the black direct dye (C.I. Direct Black 22) are presented in Table 5. Similar to red (Table 2) and yellow (Table 4) direct dyes, a moderate dyeing behavior was obtained for the black direct dye when dyeing without mordant. The best %E and %T values were obtained with zinc sulphate and copper sulphate in the pre-mordanting method. However, (K/S)2 values of these dyeings were very low and they were unable to produce dark colors. The highest %F values were obtained with calcium carbonate and magnesium sulphate but their corresponding %E and %T values were very low. In addition, the highest K/S values were obtained with calcium carbonate and magnesium sulphate in pre-mordanting which led to the highest %F values. When compared with zinc sulphate and copper sulphate, calcium carbonate, and magnesium sulphate produced dark colors although their %E and %T values were much lower. This implied that the dye–mordant complexes of these two mordants produced a new molecule with very high molar extinction. In pre-mordanting the color strength, %E and %T values of the other mordants were poor so that almost no fixation was obtained. An interesting result is the very low %E and %T values of gallic acid and ascorbic acid when compared with their corresponding results in Table 2 (red direct dye). These two mordants produced the lowest %T values in pre-mordanting of the black direct dye.
Calcium carbonate and magnesium sulphate presented the highest %E, %F, and %T values in meta-mordanting despite their corresponding values in pre-mordanting. Also, they presented the highest color strength (K/S) values in both pre- and meta-mordanting. Copper sulphate presented the highest %E, moderate %F but low %T because of its low K/S values before and after pyridine extraction. This result was similar with the corresponding ones presented in pre-mordanting. Alum presented the lowest K/S and %T values in meta-mordanting and although it had a moderate %E value, almost no coloration was obtained.
In Table 5, K/S, %E, %F, and %T values of the dyeings performed according to post-mordanting method were much better than the corresponding ones in pre- and meta-mordanting. In particular, the ((K/S)2) values which were obtained after pyridine extraction were high and almost in the same numerical range for all mordants. In addition, %T values were consistent without minimum and not under 48% (tannic acid). As a result, %T values were high and consistent with each other showing that all the mordants gave good dyeing and coloration results when post-mordanting applied. It could be stated that among all the experimental results presented in Tables 2–4, the best consistent and uniform results of K/S, %E, %F, and %T were obtained in post-mordanting dyeing procedure of the black direct dye. All the mordants resulted in K/S, %E, %F, and %T values over an acceptable level without extreme highs and lows. Alum, presenting unsuccessful results in pre- and meta-mordanting methods, performed the best in post-mordanting showing the highest %E and %T values which were similar to the corresponding ones of ferrous II sulphate. Being different from its corresponding results in pre- and meta-mordanting, copper sulphate presented high color strength, %E, %F, and %T values in post-mordanting.
An overall consideration of the different mordanting process results of the black direct dye which were presented in Table 5 implied that black direct dye was the most suitable to be used with the metallic mordants via post-mordanting method, and alum and ferrous II sulphate could be chosen to be used in dyeing according to this method. Although copper sulphate resulted the highest %E and %T values in pre- and meta-mordanting, the obtained K/S values, especially (K/S)2, were so low that the obtained color could not be considered as a deep dyeing. Opposite to that, calcium carbonate and magnesium sulphate showed moderate %E and %T values in meta-mordanting but with high K/S values that could be considered as deep colors. These two mordants showed low %E in pre-mordanting but satisfactory %E in meta- and post-mordanting. However, both of these mordants presented almost the highest %F values in all the mordanting methods. In particular, calcium carbonate resulted in a %F value over 95% in meta- and post-mordanting which showed that almost all the dye molecules which exhausted during dyeing were fixed in the fibrous bulk and resisted against the pyridine extraction application.
The K/S, %E, %F, and %T values of the four selected direct dyes of different hues which were used in dyeing of nylon fabric according to pre-, meta-, and post-mordanting methods are presented in Tables 2–5. An overall consideration of the results presented in Tables 2–5 revealed that the blue direct dye among the selected dyes was the least suitable for use in dyeing of nylon fabric samples without mordant application. The other three dyes, i.e., red, yellow, and black, could be used in dyeing of nylon fabric without mordant but resulting only in limited percent exhaustion, percent fixation, and percent overall fixation with moderate color strength values. However, the selected blue direct dye resulted in the highest (K/S)1, %E, and %T with some certain mordants, i.e., alum, zinc sulphate, gallic acid, ferrous II sulphate, and copper sulphate, in certain mordanting methods. But the calculated %T values were moderate because their corresponding (K/S)2 values were not as high as their (K/S)1 values which decreased the calculated %F values. Magnesium sulphate in pre-mordanting, calcium sulphate in meta-mordanting, and Tannic acid (bio-mordant) in post-mordanting presented the lowest experimental results for the blue direct dye (Table 3). This implied that the blue direct dye formed complexes on fiber surfaces that had high light adsorption properties. But the low (K/S)2 values indicated that the complexes formed on the fiber surfaces could not diffuse completely into the fibrous bulk volume of the fibers and later were easily extracted by pyridine solution.
It was revealed from the results presented in Tables 2–5 that an exact match of direct dye–mordant–mordanting method could not be possible because the given triplet showed distinct differences when any of them changed. However, some general conclusions could also be derived. Gallic acid and ascorbic acid presented high K/S, %E, %F, and %T results when dyeing with red direct dye according to any of the mordanting methods. Some other mordants also presented high values, e.g., ferrous II sulphate, magnesium sulphate, copper sulphate, and tannic acid, depending on the mordanting method applied. The best match of the highest K/S, %E, and %T values for the red direct dye were obtained in post-mordanting method. However, %F results were relatively lower than the corresponding ones in the other two mordanting methods.
Extremely high %E values were obtained for the yellow direct dye in Table 4 but K/S, %F, and %T values did not accompany the high %E values. In contrast to the (K/S)2 results of the blue dye in Table 3, extremely high or low corresponding (K/S)2 results were not obtained in dyeing with yellow direct dye in any mordanting method. This situation was similar to the corresponding results presented in Table 2 for the red direct dye. The evenly changing (K/S)2 results of the yellow direct dye implied that good dyeings were performed with yellow dye with the presence of any mordant and with any mordanting method applied. Some %E values were very high revealing that almost all the dye molecules were exhausted from the dyebath. However, very high (K/S)1 values similar to the corresponding ones presented in Tables 2 and 3 were not obtained and their computation with evenly changing (K/S)2 values resulted in moderate %F. As a result, moderate %T values were revealed. It could be concluded from the (K/S)2 values that the dyed samples did not show great appearance differences after pyridine extraction among themselves. Depending on the numerical results presented in Table 4 for the yellow direct dye, it was difficult to give an exact match of dye–mordant–mordanting method but it could be concluded that zinc sulphate, ferrous II sulphate, and copper sulphate could be taken into consideration for a better triple match. Among all the mordants used with the yellow dye and copper sulphate presented the highest %F values with moderate %E values which revealed much of the adsorbed dyes were fixed within the fiber bulk.
Similar to the results of the blue direct dye presented in Table 3, the most uneven dye–mordant–mordanting method match was obtained for the black direct dye in Table 5 but with an exception in post-mordanting method. Black direct dye performed the worst in pre-mordanting method but it performed the best in post-mordanting method with the mordants under research. Some mordants, e.g., gallic acid, ferrous II sulphate, tannic acid, and ascorbic acid, performed distinctly different as mordanting methods changed. Copper sulphate was the only mordant which showed similar %E, %F, and %T but different K/S values with different mordanting methods. It could be concluded that the black direct dye could be used with any of the mordants only when dyeing was performed according to post-mordanting method.
ΔE* color difference values after fastness tests
ΔE* color difference values (ΔE* according to CIELAB1976; see (4)) after extraction (Figures 2–5), after wash fastness tests (Figures 6–9), and after light fastness tests (Figures 10–13) are presented in the following figures. An overall discussion of Figures 2–13 implied that the highest ΔE* values were obtained after extraction as would be expected. The ΔE* values after extraction, wash and light fastness tests of the yellow direct dye were the highest. Black direct dye presented the lowest ΔE* values after extraction and wash fastness tests.
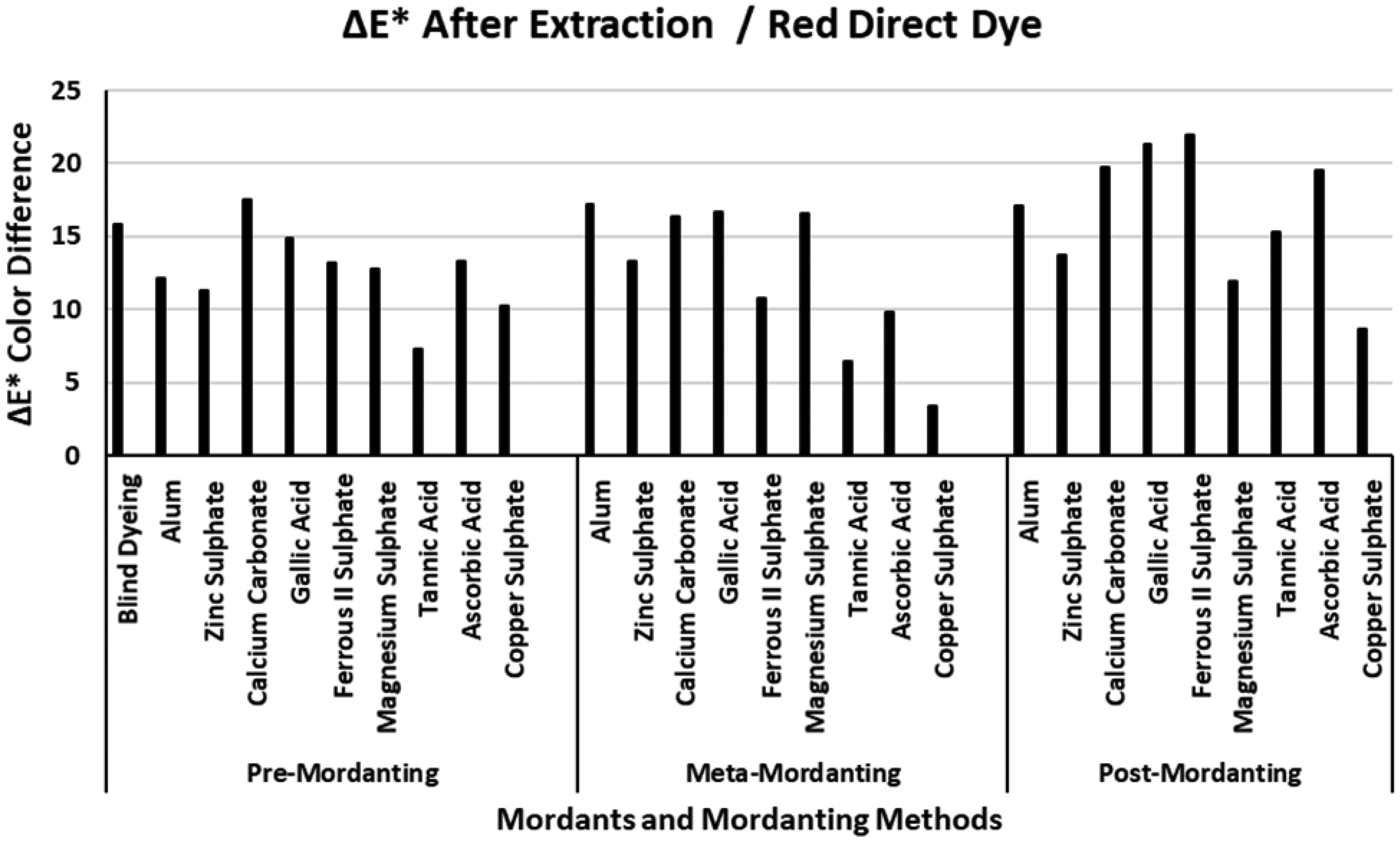
The ΔE* values of the red direct dye after extraction.
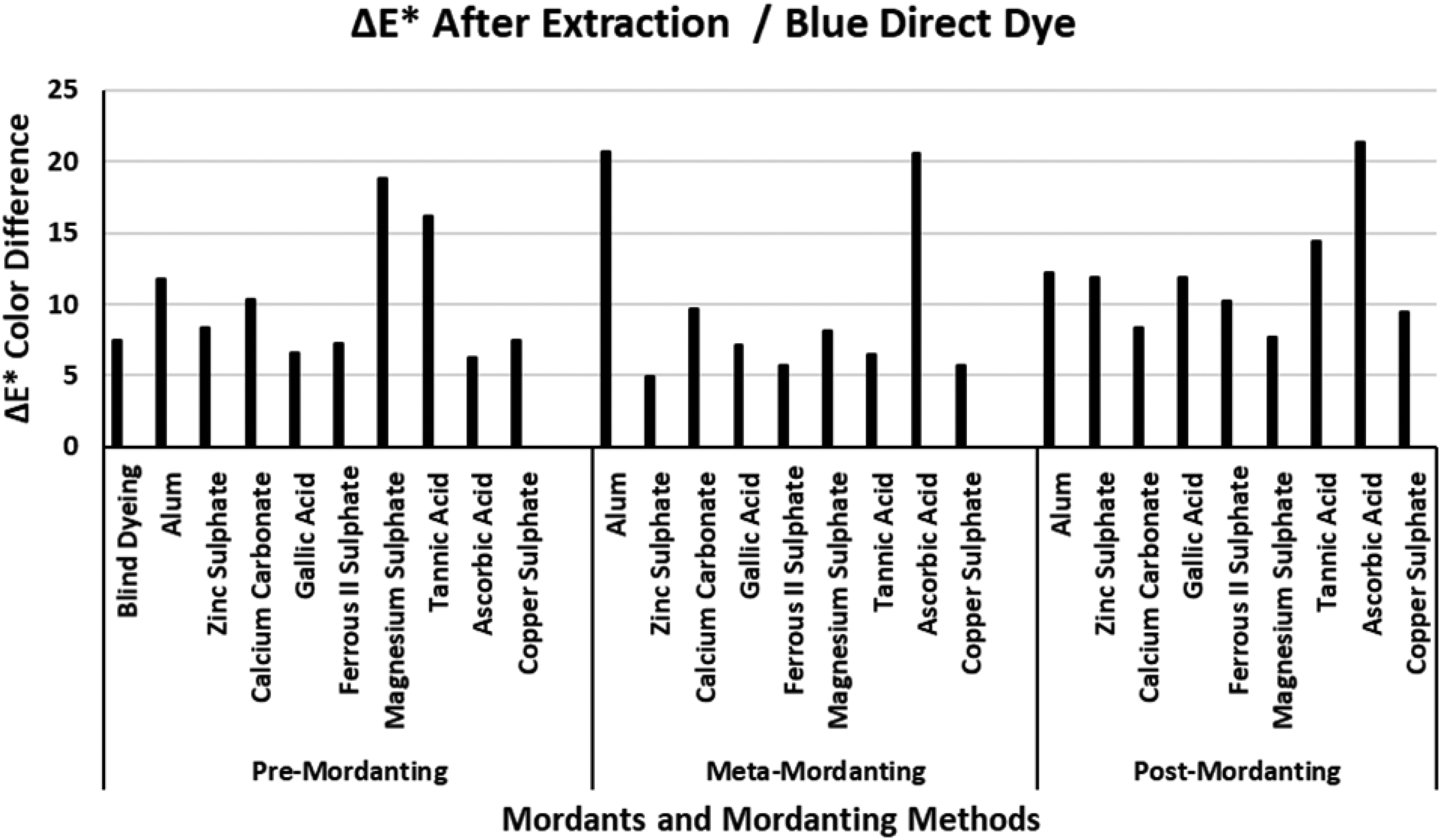
The ΔE* values of the blue direct dye after extraction.
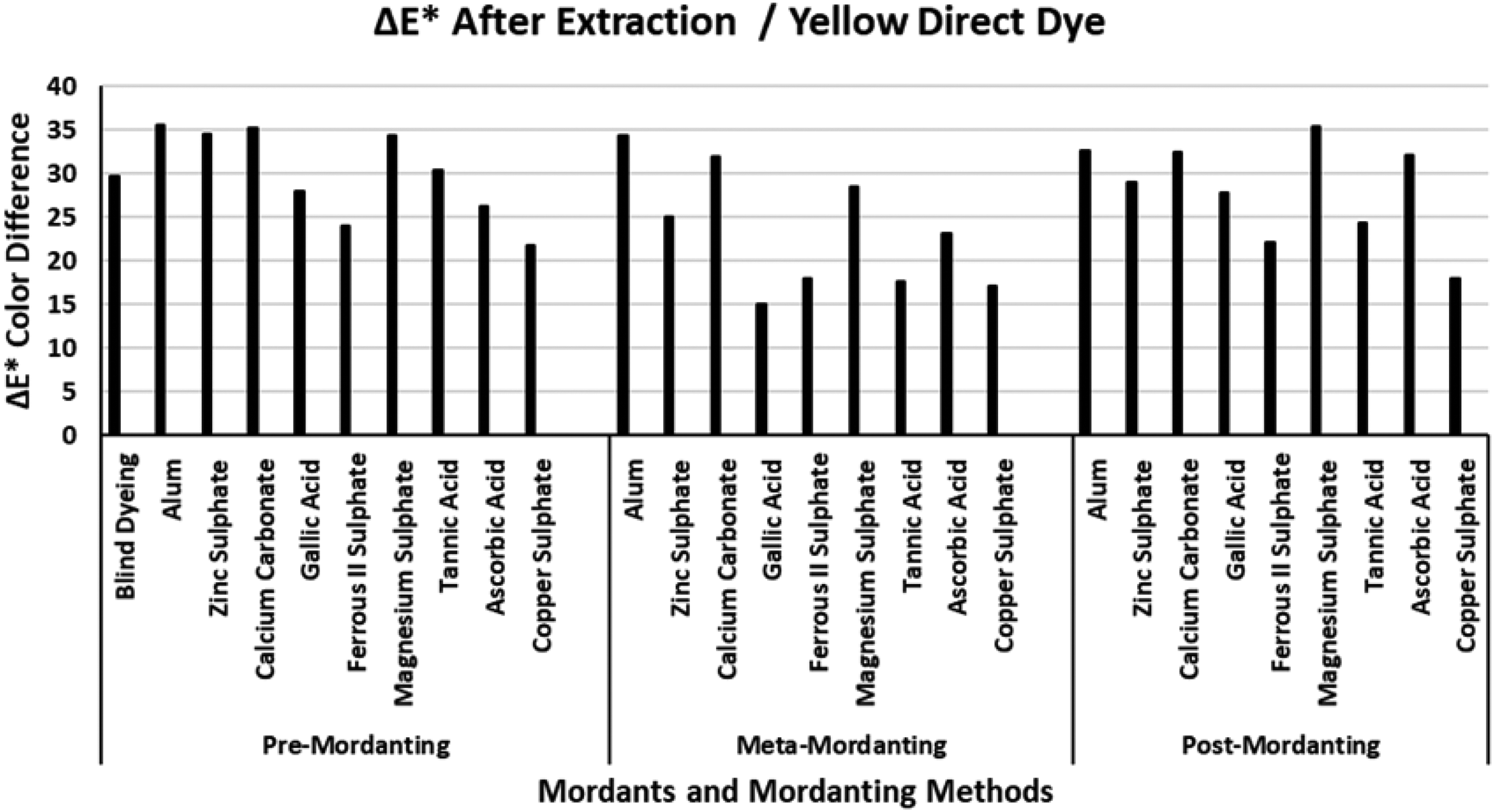
The ΔE* values of the yellow direct dye after extraction.
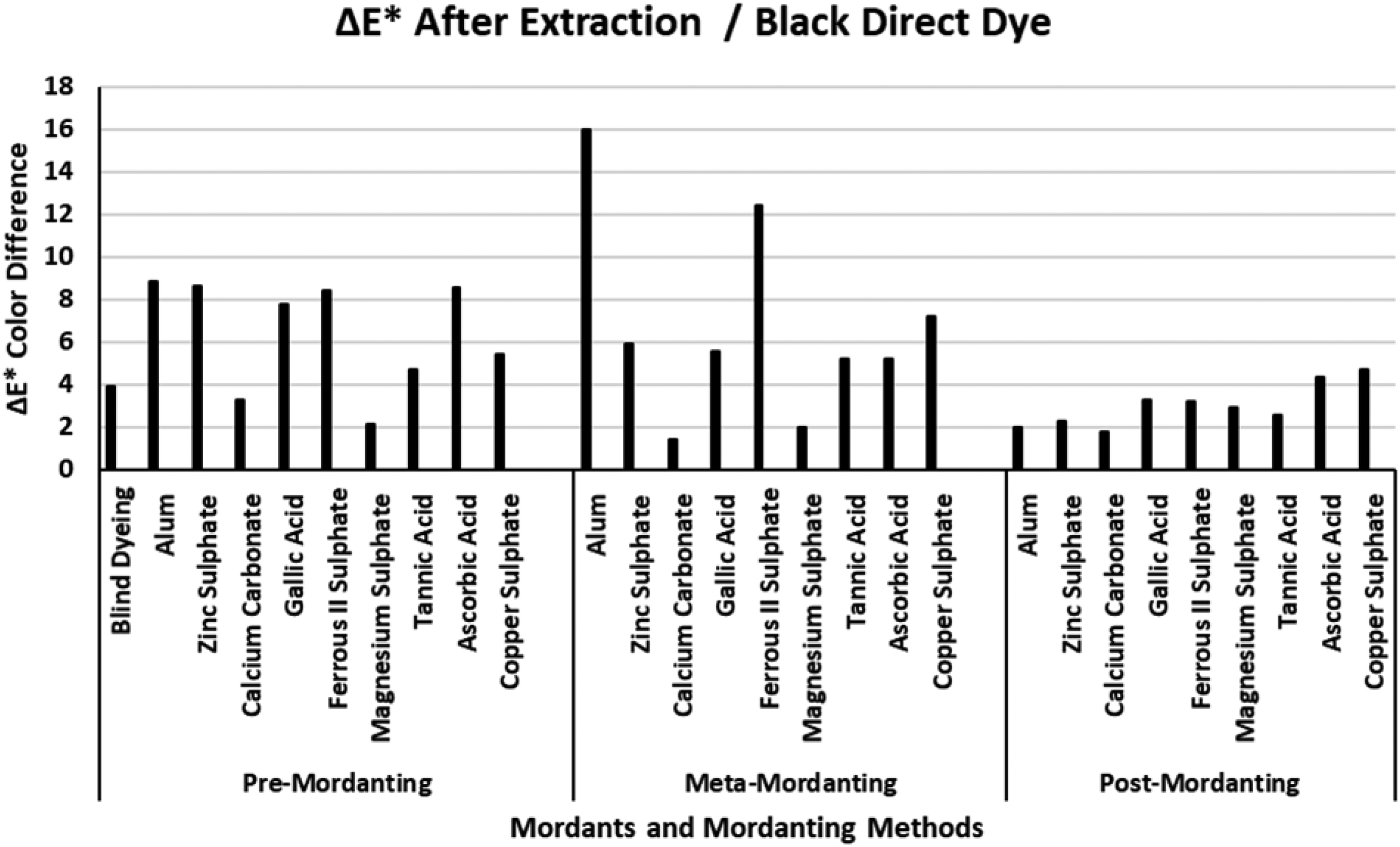
The ΔE* values of the black direct dye after extraction.
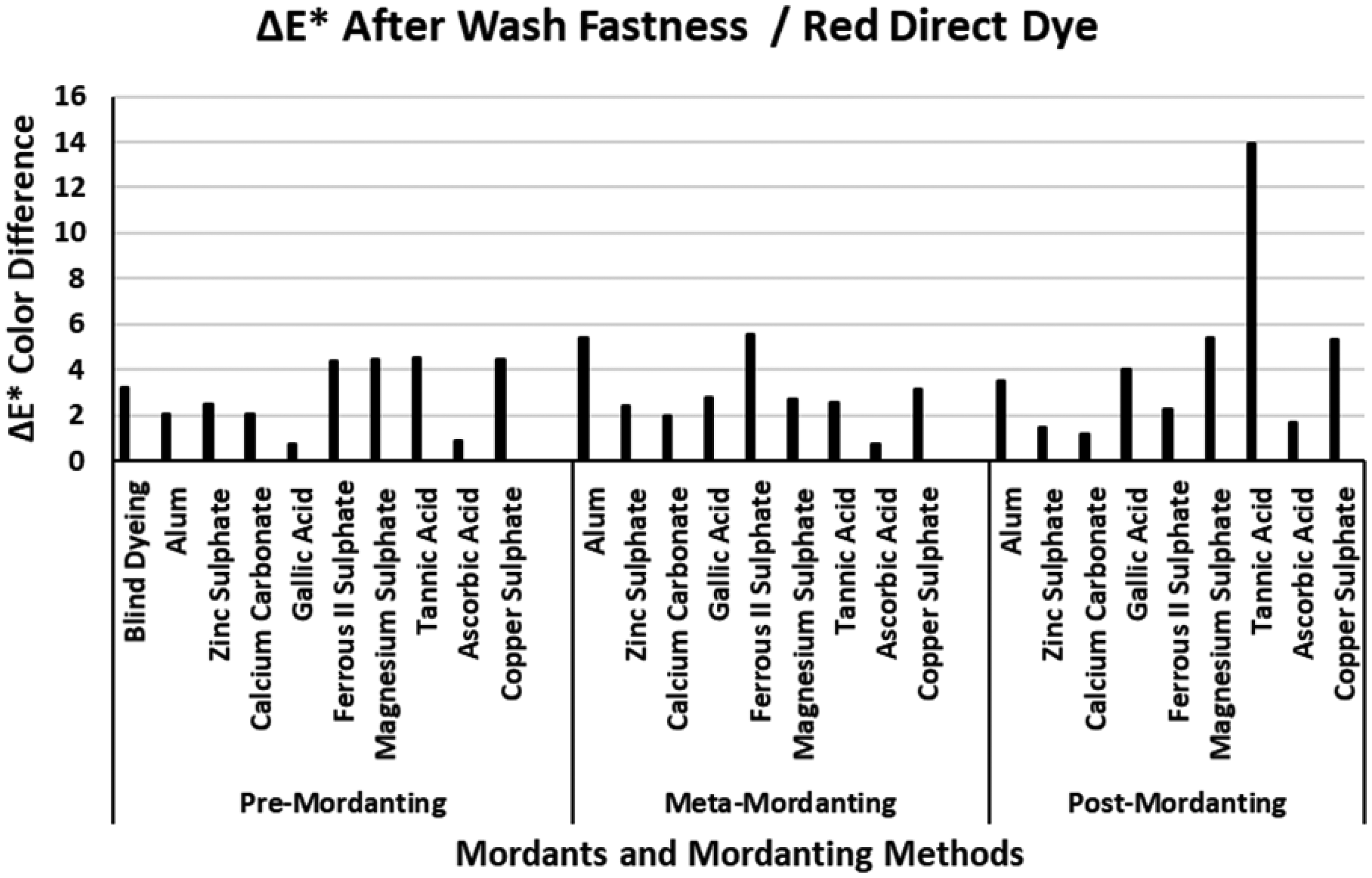
The ΔE* values of the red direct dye after wash fastness.
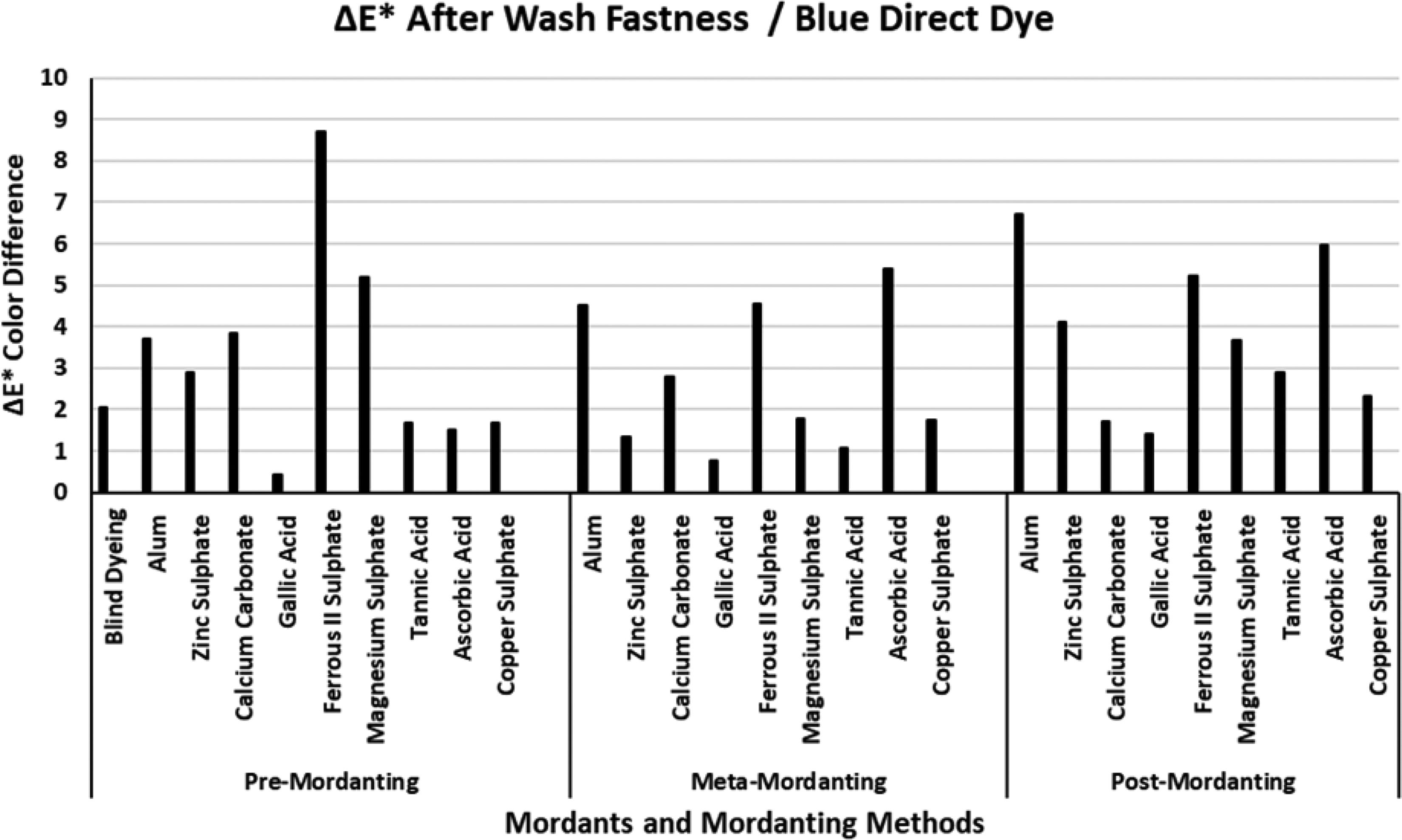
The ΔE* values of the blue direct dye after wash fastness.
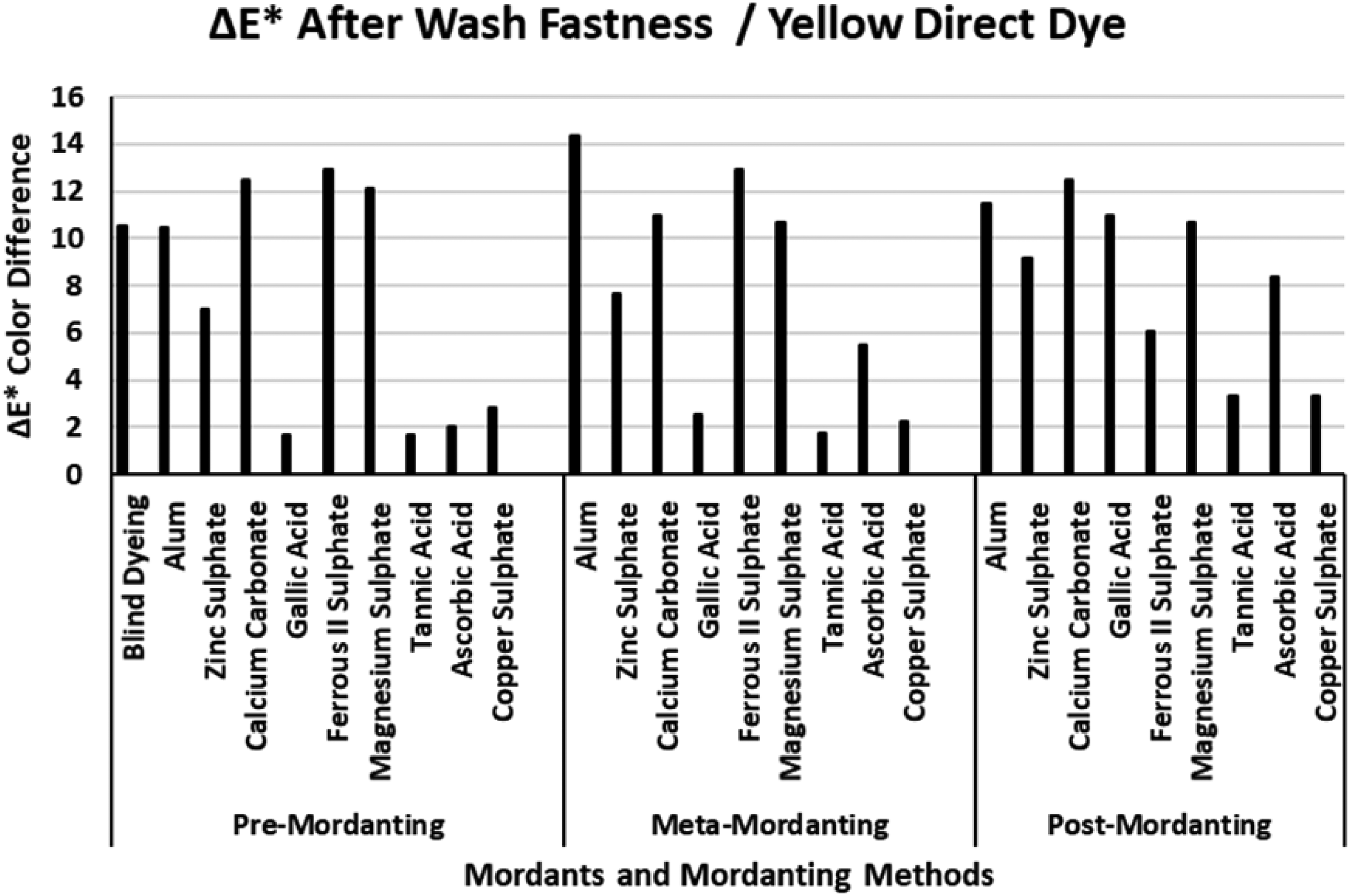
The ΔE* values of the yellow direct dye after wash fastness.
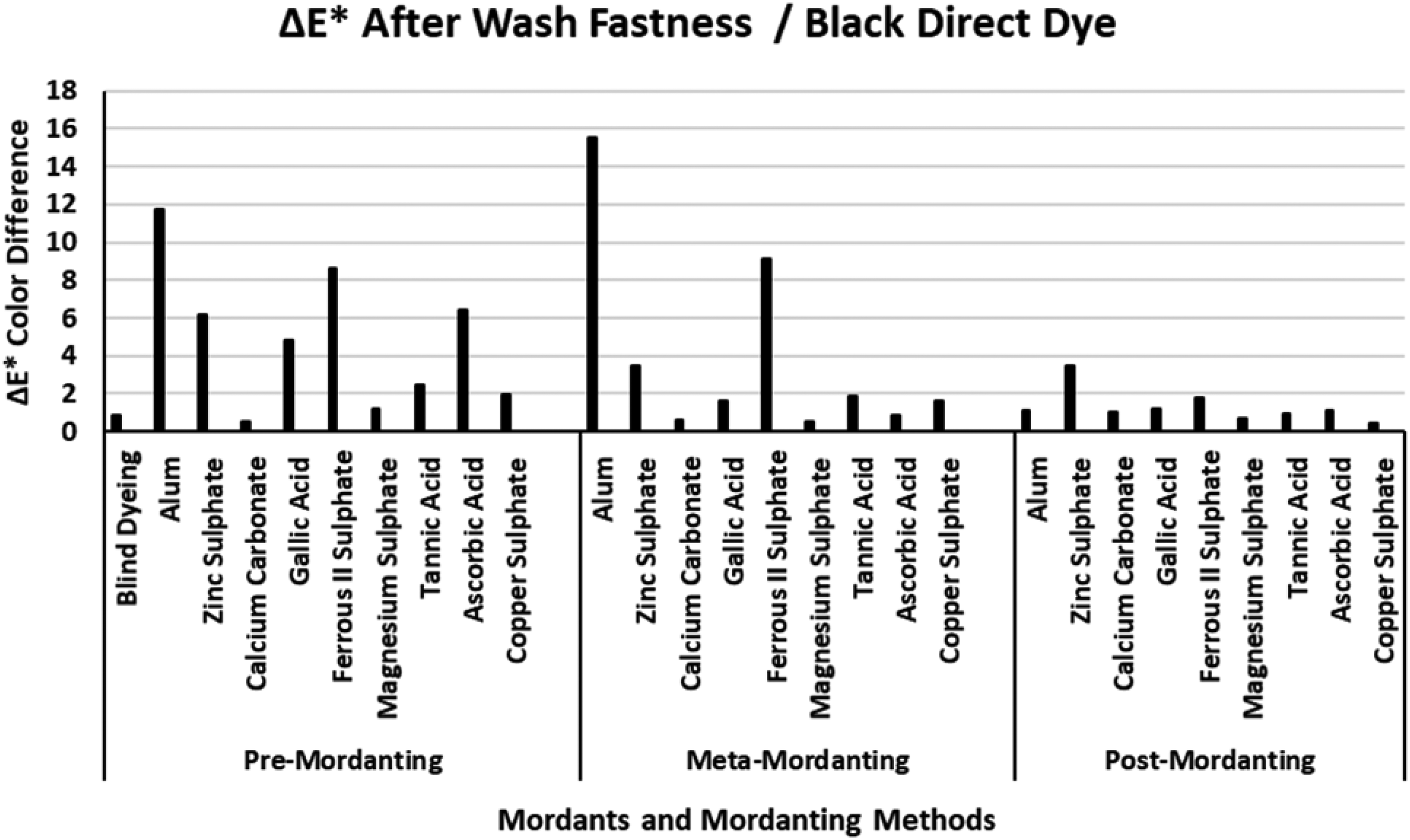
The ΔE* values of the black direct dye after wash fastness.
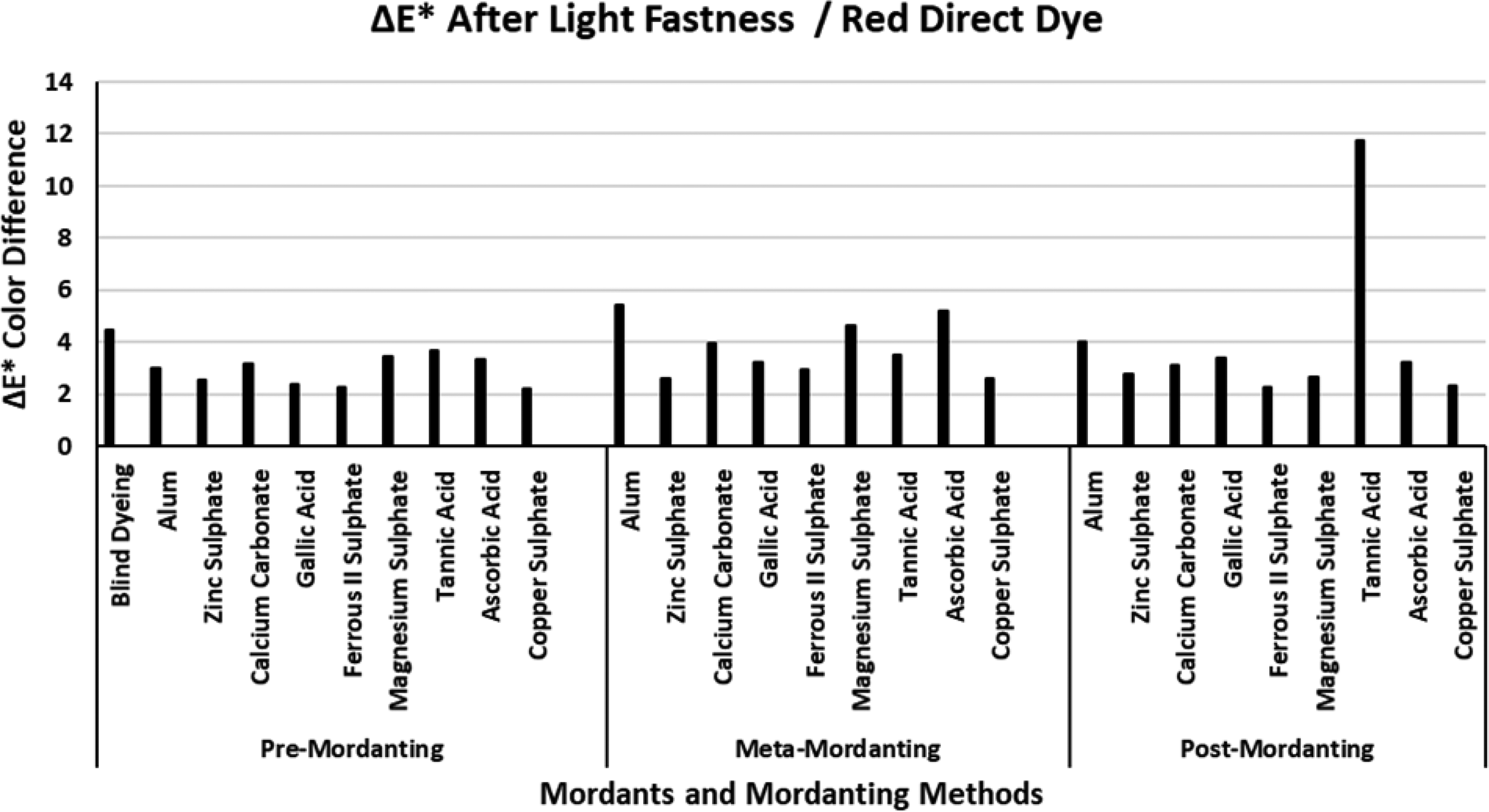
The ΔE* values of the red direct dye after light fastness.
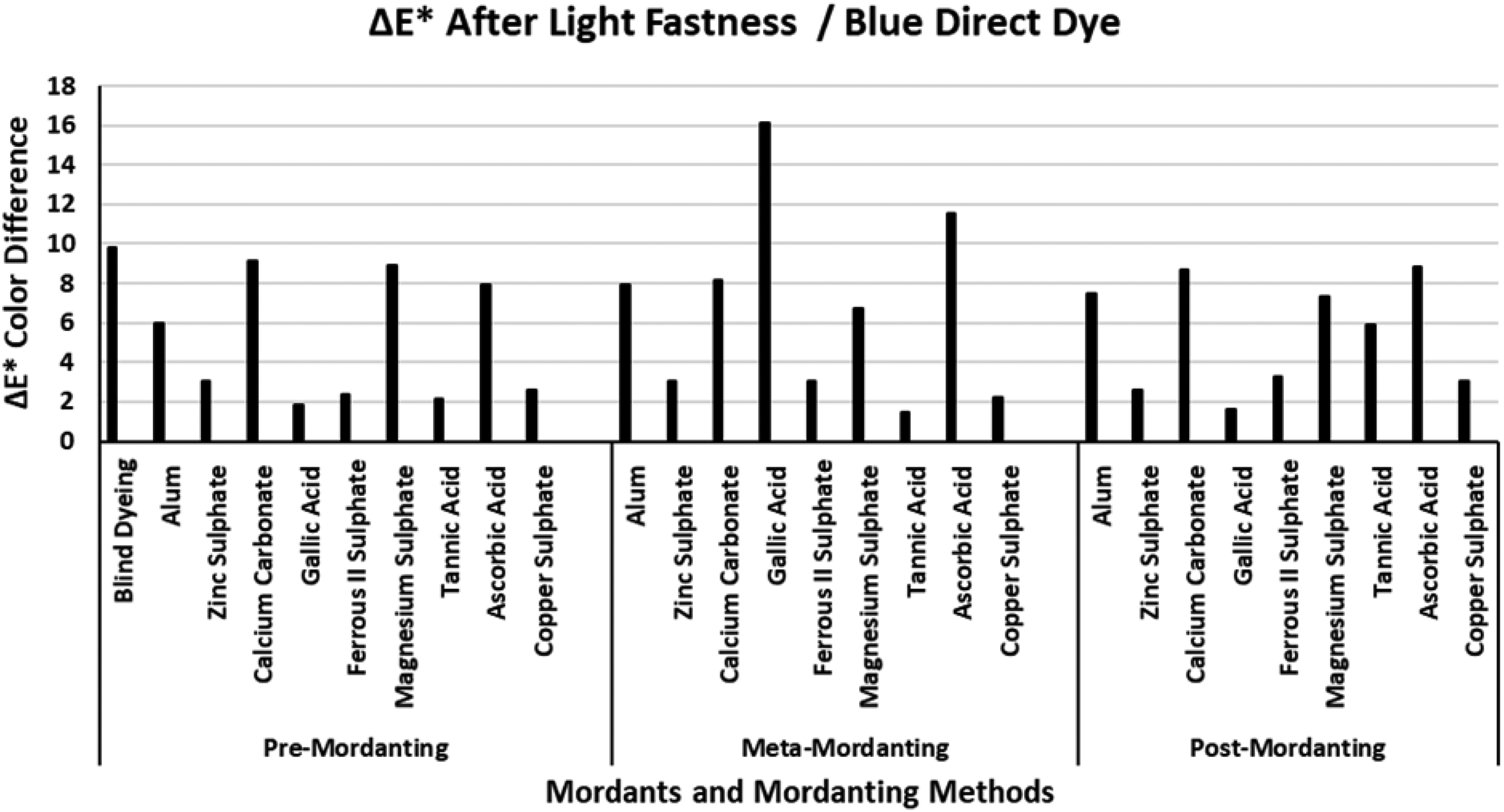
The ΔE* values of the blue direct dye after light fastness.
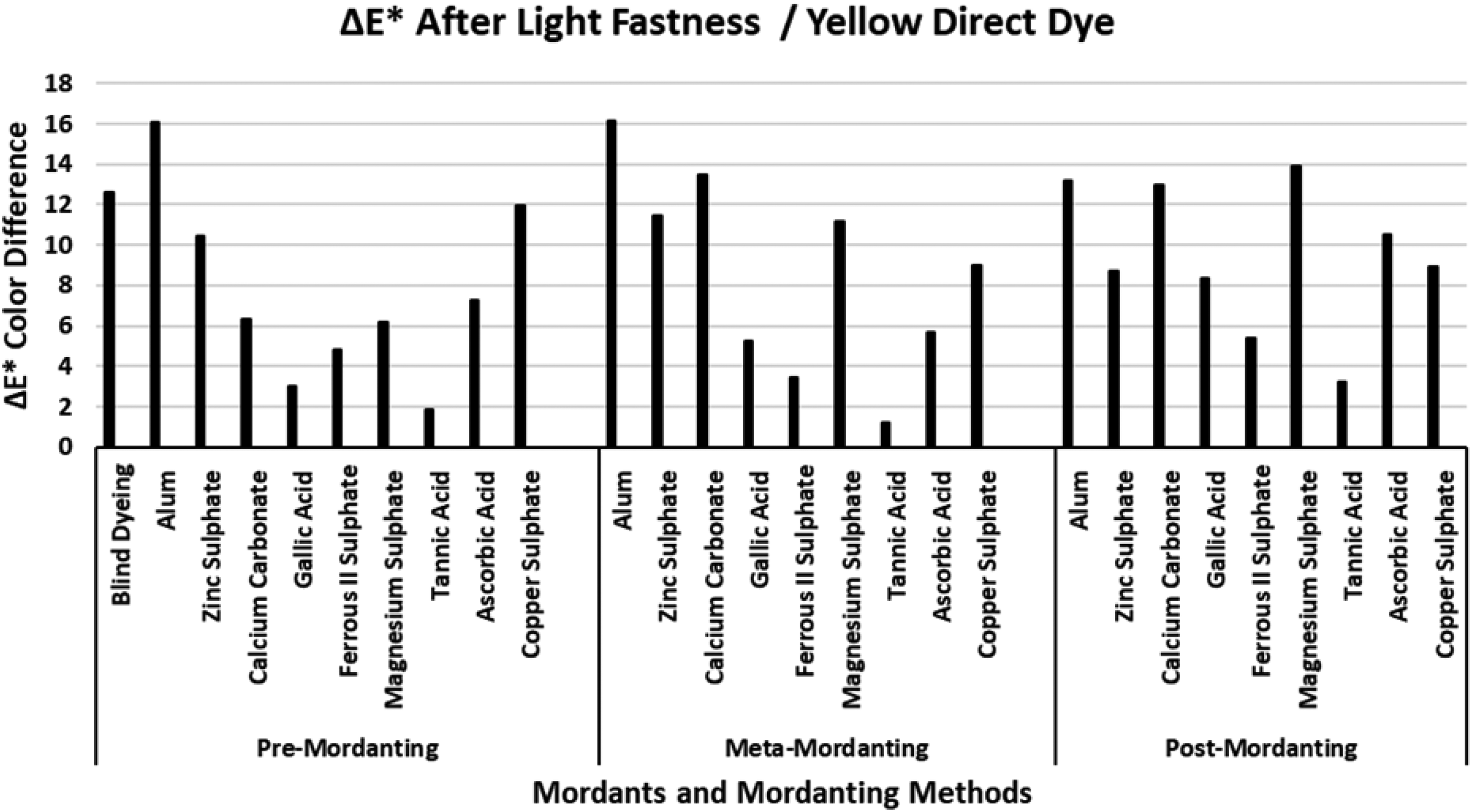
The ΔE* values of the yellow direct dye after light fastness.
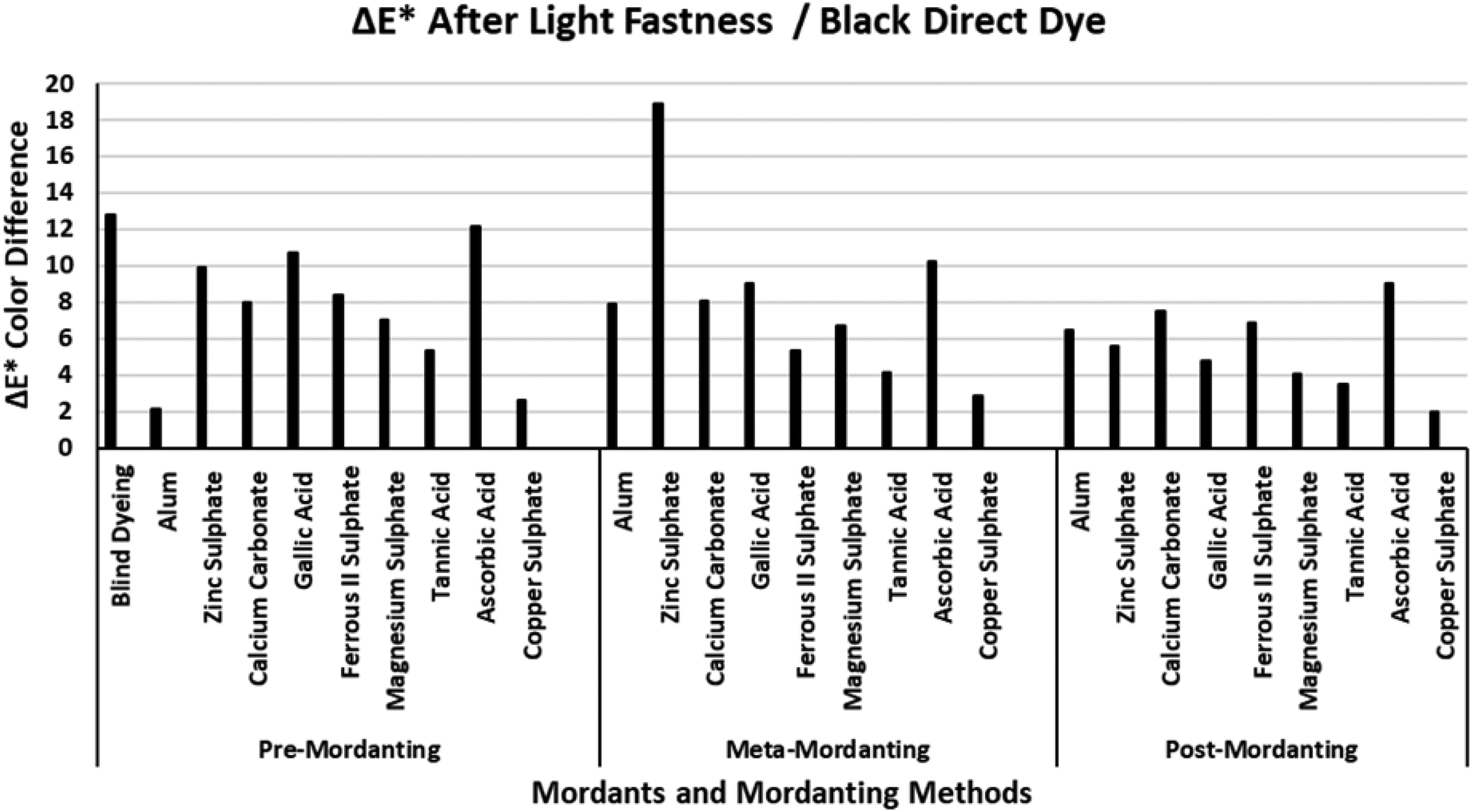
The ΔE* values of the black direct dye after light fastness.
ΔE* color difference values of the direct dyes under investigation could be discussed with their corresponding %E, %F, %T, and (K/S) data presented in Tables 2–5. Generally, dyeings with the lowest %F and %T values besides the lowest [K/S]2 values together with the lowest %E values resulted in the highest ΔE* color differences. However, it must be stated that a complex relationship among ΔE*, %F, %T, and (K/S)2 was obtained in the resultant dyeings after extraction, wash and light fastness tests. Experimental results changed considerably depending on the corresponding mordants and mordanting methods.
It must be stated that the corresponding CIELAB coordinates, e.g., L*, a*, b*, C*, and h°, were not presented in the paper due to the enormous amount of data. However, it will be stated in the coming discussions that the reason for the extremely high ΔE* color difference values stems from the huge changes in chroma (C*) values of the dyeings besides the changes in lightness (L*) coordinate. The effects of L* coordinates on ΔE* values could be estimated by the differences between (K/S)1 and (K/S)2 values. However, the effect of C* values on ΔE* were discussed in the text.
The ΔE* color difference values of the four direct dyes after extraction are presented in Figures 2–5. Figure 2 presents the ΔE* values of the red direct dye after extraction. Among the ΔE* values presented in Figures 2–5, the highest ΔE* values were obtained for the red direct dye after yellow direct dye (Figure 4). The ΔE* of the blind dyeing of the red direct dye was about 15 units indicating that dyeing with the red direct dye had a low success in terms of color difference. Although blind dyeing with the red direct dye had a high %F (76.95%) and a low %T (46.31%) with a moderate low %E (60.18%) (Table 2), the great numerical difference between the (K/S)1 and (K/S)2 values of the red direct dye revealed the high ΔE* color difference. In Figure 2, the highest ΔE* values were obtained with dyeing according to post-mordanting. The ΔE* values according to dyeing with pre- and meta-mordanting methods changed in accordance with the individual mordants under test. Dyeing in the presence of tannic acid and copper sulphate presented the lowest ΔE* values in pre- and meta-mordanting methods. Copper sulphate also presented the lowest ΔE* value in post-mordanting. All the other remaining mordants presented very high ΔE* values independent from the mordanting methods. Although gallic acid and ascorbic acid had the highest %E, %F, %T, and (K/S)2 values according to meta-mordanting method in Table 2, the dyeings with these two mordants presented high ΔE* values after extraction. The reason was observed as the great changes in chroma (C*) values of the corresponding dyeings. It must be reminded again that the color coordinate results were not presented in order not to lengthen the paper extremely. Although successful dyeings were performed with the two mordants, chroma (C*) of the resultant dyeings showed huge changes in chroma after extraction which led to high ΔE* values.
Figure 3 presents the ΔE* values of the blue direct dye after extraction. When compared with the ΔE* values of the red direct dye presented in Figure 2, lower ΔE* values were obtained after extraction of the dyeings performed with the blue direct dye. Similar to the ΔE* values of the red direct dye presented in Figure 2, the highest ΔE* values after extraction were obtained according to post-mordanting method. The ΔE* value of the blind dyeing with the blue direct dye was about 5 units which indicated that dyeing with the blue direct dye had a moderate to low success in terms of color difference. Although blind dyeing with the blue direct dye had a high %F (66.91%), it had very low %E (3.89%) and %T (2.60%) values in addition to low K/S values (Table 3). As stated in the former discussion of Table 3, blue direct dye presented very low %E, %T and K/S values with some of the mordants in the corresponding mordanting methods. It must be recalled that low %E values in accordance with low %T and K/S values were obtained in Table 3. While ascorbic acid presented the lowest ΔE* in pre-mordanting, it presented the highest ΔE* values in meta- and post-mordanting. The moderate and low ΔE* values of the other mordants were in close relationship with the data presented in Table 3. When the data in Figure 3 and Table 3 were cross discussed, it could be observed that the best dyeings with the blue direct dye were obtained in meta-mordanting. In addition, the best dyeings were obtained in pre- and meta-mordanting methods with zinc sulphate, gallic acid, ferrous II sulphate, and copper sulphate. The lowest ΔE* values of calcium carbonate and magnesium sulphate in post-mordanting method is decisive because of their corresponding %E, %F, %T, and K/S values in Table 3. An overall discussion of Figure 3 in accordance with Table 3 revealed that best dyeings with the blue direct dye were performed in the presence of ferrous II sulphate and copper sulphate in the three mordanting methods.
Figure 4 presents the ΔE* values of the yellow direct dye after extraction. When compared with the ΔE* values of the red and blue direct dyes presented in Figures 2 and 3, respectively, much higher ΔE* values were obtained after extraction of the dyeings performed with the yellow direct dye. Figure 4 indicates that extraction after meta-mordanting method resulted lower ΔE* values than pre-and meta-mordanting methods. The ΔE* value of the blind dyeing with the yellow direct dye was about 30 units which indicated that total dye fixation was very poor (%T: 44.28; Table 4). When the great color difference was evaluated with the data presented in Table 4, it was observed that blind dyeing with the yellow direct dye had moderately low %E (62.81%), and moderate %F (70.50%), but extremely low %T (44.28%) data. The difference between K/S values could not explain the great ΔE* values and it was observed from color coordinates that chroma (C*) of the dyeings changed considerably after extraction which resulted in a very high ΔE* value. All the mordants and all the mordanting methods which were used in the dyeing experiments resulted very high ΔE* indicating that successful dyeings had not been performed with the yellow direct dye. Gallic acid, ferrous II sulphate, tannic acid, and copper sulphate resulted lower ΔE* values than the other mordants in meta-mordanting method although these mordants had moderate success (%T data in Table 4) in dyeing. It could be stated that the chroma (C*) values of these dyeings changed in a lower extent than that of the remaining ones. The lowest ΔE* values after extraction were obtained by mordanting with ferrous II sulphate and copper sulphate in the three mordanting methods (except ΔE* value of gallic acid in meta-mordanting) which revealed that dyeing experiments of the yellow direct dye with these two mordants showed the lowest chroma (C*) changes after extraction. The highest ΔE* values were obtained in the presence of alum, calcium carbonate, magnesium carbonate, and ascorbic acid because of the extreme chroma changes after extraction although dyeings with these mordants presented moderate and high %E, %F, and %T data in Table 4.
Figure 5 presents the ΔE* values of the black direct dye after extraction. When compared with the ΔE* values of the red, blue, and yellow dyes presented in Figures 2–4, the lowest ΔE* values after extraction were obtained with the black dye. Figure 5 indicated that the lowest ΔE* values in mordantings after extraction were obtained in the post-mordanting method. The result corresponded with the data for post-mordanting in Table 4. The ΔE* of the blind dyeing with the black dye was about 4 units which is the lowest for the four dyes under investigation. Even lower ΔE* values were obtained in post-mordanting method. The ΔE* values obtained after extraction indicated that dyeing success of the direct dye with mordants depended on the consistency of the mordant and mordanting method. However, it must also be stated that dyeing with the black dye according to post-mordanting method was the most suitable when compared with the other two (Table 5). The lowest ΔE* values after extraction were obtained via mordanting with calcium carbonate and magnesium sulphate depending on the mordanting method. The highest ΔE* values were obtained via mordanting with alum, ferrous II sulphate, ascorbic acid, and copper sulphate. Gallic acid and tannic acid presented consistent ΔE* values in pre- and meta-mordanting methods. When ΔE* values were discussed with the data presented in Table 5 for the black dye, it was revealed that the best %E, %F, and %T values were obtained in the post-mordanting method which corresponded to the lowest ΔE* values in Figure 5. It must also be stated that a* and b* color coordinated of the black dyeings, in general, are very near the gray point (L* = 50; a* = b* = 0) in CIELAB color space. Because of that reason the obtained ΔE* values stem from the L* coordinate changes, in accordance with (K/S)2 and (K/S)1 values in Table 5, and huge chroma changes, i.e., huge changes in a* and b* coordinates, were not obtained.
An overall discussion of the ΔE* values after extraction presented in Figures 2–5 in accordance with the data presented in Tables 2–5 revealed that the success of mordant usage with the four direct dyes of four different hues changed with each mordant and mordanting method interaction. Mordants usually showed distinct dyeing and ΔE* values depending on the dye used and the mordanting method applied. The highest color differences after extraction were obtained with the yellow dye while the lowest color differences were obtained with the black dye. Depending on the ΔE* values (Figures 2–5) and data (Tables 2–5) presented, it could not be concluded on a single mordant and mordanting method to be used for successful dyeing performances. However, when ΔE* values of each mordant in each mordanting method for the four dyes were examined, it could be stated that ΔE* values resulted via mordanting with alum, zinc sulphate, and calcium carbonate were almost among the highest. None of the mordants could be given as an example that always resulted among the lowest ΔE* values. Interpreting the ΔE* values with the data presented in Tables 2–5 revealed that it could not be possible to relate directly %E, %F, and %T data with high or low ΔE* values. The only exception was the dyeing data (Table 5) and color differences obtained in post-mordanting method of the black direct dye. The high %F values of all the mordants in post-mordanting contributed the low ΔE* values of the black dye after extraction. However, the success of direct dye in post-mordanting was not only related with the corresponding %F values but with color coordinates in the CIELAB color space. As color coordinates of the black dyeing were very close to the gray point of the CIELAB color space, huge chroma shifts were not obtained or were not observed after extraction. However, the main reason of the very high ΔE* values of the other remaining three direct dyes were their corresponding hue changes in especially a* and b* coordinates which resulted huge chroma shifts on a*–b* color plane in the CIELAB color space.
When hues (hue angles) of the dyeings before and after extraction were taken into account, it was observed that necessary hue differences were not obtained which could contribute to the high color differences. After extraction, the most important changes were obtained in chroma values of the dyeings. Under these circumstances, it could be concluded that dyeings with black direct dye were the least sensitive and dyeings with the yellow direct dye were the most sensitive to chroma changes after extraction. The minor changes in hue angles of the dyeings revealed that extraction did not have an effect on hue as it could be expected. However, the results revealed that a conclusion which states high %F data would result in low ΔE* values could not be derived. As stated previously, the experimental results presented in Tables 2–5 and in Figure 2–5 showed that the success of dyeings which could be interpreted as obtaining low ΔE* values were dependent on individual interactions among dye, mordant, and mordanting method.
The ΔE* color difference values of the four direct dyes after wash fastness tests are presented in Figures 6–9. Figure 6 presents the ΔE* values of the red direct dye after wash fastness test. The ΔE* value of the blind dyeing of red direct dye was about 3 units indicating that dyeing with the red direct dye had a moderate–low success in terms of color difference. The ΔE* values after wash fastness test in Figure 6 varied according to the mordanting methods. However, the ΔE* values of ascorbic acid were remarkably low and even close to 1 in pre- and meta-mordanting methods. With reference to Table 2, %E, %F, %T, and K/S values of ascorbic acid were always among the top values when compared with the remaining mordants. Ascorbic acid resulted very high ΔE* values after extraction in Figure 2. However, the very low ΔE* values after wash fastness test in Figure 5 revealed that the dye and dye–mordant complex on the fiber surface or in the fiber bulk volume resisted the effect of wash fastness conditions and their fixation on or in the fiber was successful. Figure 6 revealed that calcium carbonate and zinc sulphate also had similar ΔE* values to ascorbic acid revealing that these two mordants performed medium-to-good dyeings with the red direct dye. These two mordants presented high ΔE* values after extraction (Figure 2) but it could be derived that they formed strong dye–mordant complexes on or in the bulk volume of the fibers resistant to the effects of the wash fastness test. Wash fastness tests with the remaining mordants resulted in unacceptable ΔE* values and the highest ΔE* value was obtained via mordanting with tannic acid in post-mordanting method. Although mordanting with tannic acid had the highest %F (94.86, Table 2) and %T (77.43; Table 2) together with ascorbic acid (82.61 and 78.82 respectively; Table 2) as well as (K/S)2 values, the effect of washing liquor considerably changed the color coordinates of the sample resulting in very high ΔE* value. The ΔE* values of gallic acid, alum, and ferrous II sulphate increased regularly from pre-mordanting to post-mordanting. Although gallic acid presented the lowest ΔE* value in pre-mordanting, the dyeings with this mordant were not successful in terms of wash fastness in meta- and post-mordanting.
Figure 7 presents the ΔE* values of the blue direct dye after wash fastness tests. The ΔE* value of the blind dyeing of the blue direct dye was about 2 units indicating that dyeing with the blue direct dye had a moderate-to-good success in terms of color difference. However, the opposite occurred when the data of blind dyeing in Table 3 were considered. The blind dyeing of the blue direct dye presented very poor data (%E 3.89; %F 2.60; (K/S)1 2.72; and (K/S)2 1.82) in Table 3 representing an unsuccessful dyeing. Low ΔE* values of the blind dyeing of the blue direct dye after wash fastness test were the result of unsuccessful coloration. Similar to the results presented for the wash fastness ΔE* values of the red direct dye in Figure 6, ΔE* values of the blue direct dye after wash fastness test changed in different directions with the mordanting methods. The lowest ΔE* values after wash fastness tests were obtained via mordanting with gallic acid. In particular, gallic acid presented the lowest ΔE* values after wash fastness tests for both red (Figure 6) and blue (Figure 7) direct dyes in pre-mordanting method. The results revealed that fabric samples pre-mordanted with gallic acid did not show considerable changes in color coordinates after wash fastness test. Alum and ferrous II sulphate always presented high ΔE* results after wash fastness test. When a correlation was made with the data presented in Table 3 for the blue direct dye, it was observed that alum performed unsuccessful results especially in meta- and post-mordanting.
Figure 8 presents the ΔE* values of the yellow direct dye after wash fastness tests. The ΔE* value of the blind dyeing of the yellow direct dye was about 10 units indicating that the dyeing with the yellow direct dye had an unacceptable color difference. Gallic acid, tannic acid, ascorbic acid, and copper sulphate presented the lowest ΔE* values in different mordanting methods. In particular, the lowest wash fastness ΔE* values of some certain mordants were obtained in the pre-mordanting method. Alum, zinc sulphate, calcium carbonate, ferrous II sulphate, and magnesium sulphate always presented high ΔE* values after the wash fastness test. The ΔE* values of the yellow direct dye after extraction (Figure 4) did not correspond with the ΔE* values after the wash fastness tests. However, relatively smaller ΔE* values of gallic acid, tannic acid, and copper sulphate in Figure 4 correlated with their corresponding low ΔE* values in Figure 8. When the ΔE* results presented in Figure 8 were correlated with the data given in Table 4 for the yellow direct dye, the (K/S)2 values of the mordants with low ΔE* were 4–5 units lower than their corresponding (K/S)1 values. This indicated that the dye–mordant complexes, which deposited on fiber surfaces, were prone to the effect of extraction but they were resistant to the action of wash fastness. Stable dye–mordant complexes of these mordants with the yellow direct dye could not be washed from fiber surfaces and low ΔE* values were obtained although the original dyeings were not successful.
Figure 9 presents the ΔE* values of the black direct dye after wash fastness tests. The ΔE* value of the blind dyeing of the black direct dye was about 1 unit which indicated that dyeing with the black direct dye made a successful coloration in terms of color difference. This finding was supported with the data of the black direct dye presented in Table 5. The blind dyeing of the black direct dye has moderate %E and %T values but a high %F value with acceptable (K/S)2 and (K/S)1 values when compared with the corresponding results of mordant application. In the interpretation of the ΔE* values presented in Figure 9 for the black direct dye, it should be recalled that the color coordinates of such dyeings and colors (hues) were near the gray point of the CIELAB color space (L*=50; a*=b*=0) accompanied by very low chroma (C*) values and absence of a definite hue. The lowest ΔE* values among Figures 2–13 were obtained in wash fastness data of the black direct dye according to the post-treatment method which indicated that the best coloring properties were obtained in the experimental segment. The lowest ΔE* values of the black direct dye were supported by the data presented in Table 5. In Table 5, among all the data presented in Tables 2–5, the best %F and %T values in addition to Δ(K/S) [(K/S)2 – (K/S)1] were obtained for the post-mordanting method with the black direct dye. It was clear that the most successful %F, %T, and Δ(K/S) data resulted in the best wash fastness results with the lowest ΔE* values. In Figure 9, the highest ΔE* values were obtained according to dyeing with the pre-mordanting method. Any mordanting with alum, zinc sulphate, and ferrous II sulphate presented the highest ΔE* values. Any mordanting with calcium carbonate presented low ΔE* values in Figure 9. The ΔE* wash fastness values of any mordanting with calcium carbonate were very near or less than 1 unit and these results were in accordance with the data presented for calcium carbonate in Table 5. In Table 5, calcium carbonate presented very high %E, %F, (K/S)2, and (K/S)1 values.
High wash fastness ΔE* values of alum and ferrous II sulphate were distinguished in Figure 9 in pre- and meta-mordanting. When the data presented in Table 5 for these two mordants were considered, it was observed that the two mordant dyeings had the lowest (K/S)1 and (K/S)2 values in pre- and meta-mordanting. However, the dyeings with these two mordants according to post-mordanting method had high %E, %F, and %T values accompanied by high K/S and low Δ(K/S) values indicating they both showed better coloration properties with the black direct dye according to the post-mordanting method.
An overall consideration of the wash fastness ΔE* values presented in Figures 6–9 revealed that best coloration properties among the four dyes were obtained by black direct dye according to the post-mordanting method. The wash fastness ΔE* results corresponded with the extraction ΔE* results. The highest ΔE* values were obtained dyeing with yellow direct dye which indicated that the yellow direct dye was not suitable for dyeing nylon fabric with the mordants under investigation according to any mordanting method. However, when Figure 8 was considered with Figure 4, together with the data presented in Table 4 for the yellow direct dye, dye–mordant combination of yellow dye–copper sulphate presented correspondingly similar behavior in terms of color differences for extraction and wash fastness tests. The %F and %T data of the yellow dye–copper sulphate pair (Table 4) were always among the highest values and the highest %F of this pair could be taken in accordance with extraction ΔE* and wash fastness ΔE*. No other mordant showed such consistent relation with the yellow direct dye. Another aspect obtained from wash fastness test could be stated as the hue of the dye itself also changed the observed results of the experimental procedure. It could be revealed from the data presented in Figures 6–9 that yellow hue was more prone to hue changes with mordants than the other three direct dyes. Similarly, black direct dye seemed less prone to hue changes with mordants. However, as the chroma of the dyeings with the black dye were very near the gray point of the a*–b* color plane, the hue changes obtained with it presented lower importance. According to real data of color coordinates, black dye also showed huge hue changes but because of the very low chroma of the dyeings, those changes presented very minor effects on ΔE* values.
Figures 10–13 present the ΔE* values after light fastness tests of the four direct dyes. Prior to the consideration of the ΔE* light fastness values of the direct dyes and their related dyeings with mordants via mordanting methods, it should be recalled that light fastness properties of the dyed textile materials change in accordance with the applied dyeing concentration. Light fastness properties of the dyed textile materials usually improve with the increase in applied dyeing concentration. When the amount of dye molecules in the fibers increase, the light fastness properties of the dyed materials improve. The property is in direct contrast to wash fastness. 37 However, only one dyeing concentration (1% owf) was applied in the experimental research. The determination of the light fastness properties of the mordant dyed samples were made according to reflectance measurements between 400 and 700 nm under D65/10° illuminant and SCI included mode. The color coordinates (L*, a*, b*, C*, and h°) in CIELAB color space were computed and later related color differences (ΔE* values) were determined according to CIELAB1976 formula (4).
Figure 10 presents the ΔE* values of the red direct dye after light fastness tests. The ΔE* value of the blind dyeing of the red direct dye was about 5 units which indicated that dyeing with the red direct dye could not present a successful coloration in terms of dyeing and light fastness. The first impression of Figure 10 revealed that the lowest ΔE* values were obtained via mordanting according to pre-mordanting method. In the pre-mordanting method almost all the ΔE* values were below 4, and zinc sulphate, gallic acid, ferrous II sulphate, and copper sulphate presented the lowest ΔE* values in three of the mordanting methods. The highest ΔE* values were obtained with mordanting via tannic acid and alum in all the mordanting methods and these results were in accordance with their corresponding ΔE* values after wash fastness tests in Figure 6. According to the data given in Table 2 for the red direct dye, successful dyeings were performed with the three mordants. In addition, among all the ΔE* values of the mordants under test, tannic acid presented the highest ΔE* values both after wash (Figure 6) and light (Figure 10) fastness tests which revealed that although dye–mordant combination of red direct dye and tannic acid presented successful data in Table 2, the produced compound was prone to the effects of wash and light fastness.
Figure 11 presents the ΔE* values of the blue direct dye after light fastness test. The ΔE* value of the blind dyeing was among the highest ΔE* values of all the mordants under investigation. Distinctly different ΔE* values were obtained for the mordants and mordanting methods in Figure 11. However, it could be stated that the highest ΔE* values in all the mordanting methods were obtained via mordanting blue direct dye with calcium carbonate, magnesium sulphate, and ascorbic acid. Mordanting with gallic acid presented the lowest ΔE* values both in pre- and post-mordanting while it presented the highest ΔE* values in meta-mordanting. Similar to the ΔE* values presented for the red direct dye in Figure 10, the lowest ΔE* values were obtained with mordanting via zinc sulphate, ferrous II sulphate, and copper sulphate. In addition, an interesting correlation was observed between Figure 7 (wash fastness ΔE* values) and Figure 11 (light fastness ΔE* values). The ΔE* values presented with ferrous II sulphate in Figure 7 were among the highest while ΔE* values presented in Figure 11 were among the lowest. A similar but less-correlated behavior was observed for mordanting with tannic acid in Figures 7 and 11. It could be concluded that the ΔE* values after light fastness were in good correlation with the data presented in Table 3 for the blue direct dye. However, both successful dyeings (high %E, %F, and %T values) and unsuccessful dyeings (low %E, %F, and %T values) presented high ΔE* values after wash and light fastness tests. This observation revealed that the success of the data presented in the tables which include data of %E, %F, and %F could not be taken as an example for good success in wash and light fastness.
Figure 12 presents the ΔE* values of the yellow direct dye after light fastness tests. The ΔE* value of the blind dyeing was among the highest ΔE* values of all the dye–mordant pairs and the high ΔE* value corresponded to that presented wash fastness ΔE* in Figure 8. Although distinctly different ΔE* values were obtained for the red direct dye–mordant interactions in Figure 6 (wash fastness ΔE*) and Figure 10 (light fastness ΔE*), very close ΔE* values were obtained for the yellow direct dye–mordant interaction in Figures 4 and 12, respectively. However, some dye–mordant pairs presented distinctly different results as was pointed in the previous discussions. The ΔE* values of gallic acid, ferrous II sulphate, and tannic acid presented the lowest ΔE* values after light fastness tests. Tannic acid presented the lowest ΔE* values but the results did not correspond with its’ poor data in Table 4. Alum, magnesium sulphate, calcium carbonate, ascorbic acid, and copper sulphate presented high ΔE* values after light fastness test. The high ΔE* values of alum, calcium carbonate, and magnesium sulphate corresponded with their high ΔE* values after wash fastness test (Figure 8). However, the ΔE* values of ferrous II sulphate in Figures 8 and 12 presented completely different results which revealed that mordanting with ferrous II sulphate behaved distinctly different against wash and light fastness tests. Yellow dye–ferrous II sulphate interaction was resistant to the effect of light but it was prone to the effect of washing. According to data presented in Table IV, ferrous II sulphate presented the highest K/S values in post-mordanting while the lowest %E, %F, and %T values. This finding pointed that yellow dye–ferrous II sulphate pair performed an unsuccessful dyeing but with successful K/S. Although ferrous II sulphate presented much better %E, %F, and %T data in pre- and meta-mordanting, the resulting ΔE* values after light fastness test were among the highest in Figure 8 (wash fastness ΔE*) and among the lowest in Figure 12 (light fastness ΔE* values). These findings implied that it could not be possible to draw exact conclusions among data presented in Table 4 and wash and light fastness ΔE* values (Figures 8 and 12, respectively).
Figure 13 presents the ΔE* values of the black direct dye after light fastness tests. It must be remembered that the lowest ΔE* values of all the experimental research were obtained after wash fastness tests of the black dye in Figure 9. In Figure 13, blind dyeing with the black dye presented one of the highest ΔE* values although it presented one of the lowest ΔE* values in Figure 9 (wash fastness ΔE*). The ΔE* values of the black direct dye after light fastness test in Figure 13 did not correspond with the ΔE* values after wash fastness tests in Figure 9 when compared according to numerical results. However, the level of numerical difference corresponded with that of given in Figure 9, because they both had the lowest ΔE* in their own fastness category. It must be stated that the best data of %E, %F, and %T were obtained for the black direct dye in post-mordanting method in the whole experimental research. However, the same conclusion could not be stated for the ΔE* results after light fastness test. The highest and the lowest ΔE* values of the mordants changed in different manners according to the mordanting method applied. All the mordants other than copper sulphate presented high ΔE* values after light fastness test. The highest ΔE* values were obtained via mordanting with zinc sulphate and ascorbic acid in all the mordanting methods. The lowest ΔE* values after light fastness tests were obtained in post-mordanting and these results corresponded with the data given in Table 2 for black dye and post-mordanting method interaction. The best fit of data in Tables 2–5 and Figures 2–13 was obtained with the black direct dye. Low %E, %F, and %T values produced high ΔE* values according to extraction, wash fastness, and light fastness. This could be because of the low chroma of the dyeings produced dyeing with black direct dye and low lightness coordinate (L*).
A brief discussion of the ΔE* color differences obtained after light fastness test implied that similar to the discussions presented for the ΔE* values after wash fastness test, it could not be possible to point regularly changing results for all the dye–mordant pairs after light fastness test. Certain dyes (dye hues) showed low ΔE* values with certain mordants according to certain mordanting applications but the magnitude of the ΔE* values obtained changed considerably when dye–mordant pairs or the mordanting methods changed. All the direct dyes presented the lowest ΔE* values after light fastness with some certain mordants depending on the nature of the dye–mordant interaction. For example, mordanting blue and red direct dyes with zinc sulphate and ferrous II sulphate always showed among the lowest ΔE* values in all the mordanting methods when light fastness test was considered.
Yellow direct dye presented the lowest ΔE* values with mordanting via tannic acid in all the mordanting methods. Black dye presented the lowest ΔE* values via mordanting with copper sulphate in all the mordanting methods. In addition, copper sulphate presented interesting ΔE* values with blue, red, and black direct dyes after light fastness. In all the mordanting methods, mordanting those three direct dyes with copper sulphate always resulted in one of the lowest ΔE* values. The only exception for the copper sulphate was mordanting with yellow direct dye. The copper sulphate–yellow direct dye pair always resulted in one of the highest ΔE* values in all the mordanting methods. These findings revealed that the color and color difference properties of the dye–mordant complexes were influenced by the intrinsic color of the mordant under acidic medium. For example, the intrinsic colors of ferrous II sulphate and copper sulphate in mild acidic medium (pH≈5) are yellow and blue tinted, respectively.
An overall discussion of the %E, %F, and %T data presented in Tables 2–5, wash fastness ΔE* values presented in Figures 2–6, and light fastness ΔE* values in Figures 9–12 revealed that it was not possible to derive exact good dyeing and/or good wash and light fastness conclusions for any dye–mordant pair according to any mordanting method. In addition, high %E, %F, and %T values together with K/S values do not reveal considerably good results after wash and light fastness tests. Some dye–mordant pairs presented high %E, %F, and %T data in Tables 2–5 but presented poor and/or light fastness results according to the mordanting method. The results presented in Tables 2–5 and Figures 2–12 did not point to an exact mordant or mordanting method to show the poorest results (%E, %F, %T, K/S, and ΔE* values) with the four direct dyes of different hues under investigation. However, when ΔE* values presented for the yellow direct dye in Figure 4 for extraction, in Figure 8 for wash fastness, and in Figure 12 for light fastness were considered in accordance with the ΔE* values of the other four direct dyes, it could be revealed that dyeing with yellow direct dye was more prone to the effects of mordanting with different mordants. The ΔE* values of the blind dyeing of yellow dye were almost among the highest ΔE* values in Figures 4, 8, and 12 indicating that yellow dye was not successful in the fastness tests after dyeing. However, the data presented in Table 4 consisted of above-average %E, %F, and %T values. It could be concluded that %E, %F, and %T values, although representing exhaustion, fixation, and overall fixation yield success of the dyeings must be discussed with the ΔE* results of extraction, wash fastness, and light fastness tests. Depending on the coloration properties of the dye under investigation; the black direct dye in this research, presented consistent %E, %F, and %T data with ΔE* values of wash fastness test in particular.
As indicated by Burkinshaw and Gandhi14,15 and Burkinshaw and Wills
16
, under acidic conditions the terminal amino groups in the nylon fiber will be protonated according to

Under these conditions, an equilibrium will be established according to the number of hydrogen protons in the dye bath and in the fiber. In this research, all the dyeing experiments were performed at pH 5 for two needs. The first need was to keep the internal fiber pH below 7 and to introduce protonated amino groups for the possible coulombic attraction between the protonated amine groups and anionic direct dyes. The second need was that the mordant chemicals which were used in the experimental part were stable under acidic conditions. Direct dyes are not preferred in nylon dyeing because when compared with acid dyes, their molecules are larger than acid dyes, their substantivity to nylon fibers is somewhat lower than acid dyes, and although direct dyes are anionic dyes and nylon fibers hold positive charges under acidic conditions the possibility of ionic bond formation is lower than that of corresponding acid dyes. The effect of dyeing pH and effect of the protonation of the nylon fiber structure on the obtained K/S, %E, and %T results were discussed in another paper. 25 The pH level in this research was chosen at pH 5 but the higher (K/S)2, %E, %F, and %T results obtained in different mordant–mordanting matches than the dyeings performed without mordant (especially for the blue direct dye) revealed that application in acidic pH contributed to better dyeing results.
Except for the blue direct dye, the (K/S)1 values which were obtained in post-mordanting were generally higher than the corresponding values obtained in pre- and meta-mordanting. This could be attributed to the better absorption properties of some certain mordants in fiber structure. The experimental results revealed that high (K/S)2 values generally accompanied high %T values in all mordanting methods. Of course there were a few exceptions which originated from the different molecular behavior and absorption properties of some certain dye–mordants pairs, but this was the general behavior.
The experimental results obtained in this research revealed that metallic mordants could be used in direct dyeing of nylon fabrics. However, the success of the dyeings changes in correlation with the dye (dye molecule and dye hue), mordanting method, and mordant chemical. The dyeings were performed at only one dyeing concentration of 1% owf, so it was not possible to draw any conclusion on the effect of dyeing concentration on the results.
The results also highlighted that metallic and bio-mordant could be used in the dyeing of nylon fabrics with success and could present an alternative dyeing method to be applied. However, the fact, which must be taken into consideration, was that dyeing and fastness performances the triple of dye–mordant–mordanting method changed considerably.
The main conclusion of this research depending on the experimental procedure and the related experimental results would be that successful dyeings on nylon fabrics with direct dyes of different hues could be obtained by using mordant chemicals and applying the classical mordanting methods. However, the success of the dyeings of dye–mordant pairs could not be assured for any mordant or mordanting method. Each direct dye under research presented peculiar behavior of its own with any mordant and mordanting method. It was not possible to derive a general useful conclusion, which revealed a certain dye–mordant pair or any mordanting method for the best dyeing and fastness results. Depending on the dyes and mordants used, in addition to applied mordanting methods, only copper sulphate presented continuing and consistent ΔE* values with all the dyes and mordanting methods.
Conclusion
In the experimental part, four commercial direct dyes, namely, red, blue, yellow, and black, were chosen to investigate their dyeing properties in the presence of different mordants, eight metallic (chemical) and one bio-mordant, according to different mordanting methods. The dye–mordant–mordanting method interactions were studied with four direct dyes in different hue regions of the CIELAB color space. Success of the dyeings was evaluated by extraction after dyeing and the related K/S, %E, %F, and %T data in addition to ΔE* values after extraction, wash fastness, and light fastness tests were calculated. The experimental results revealed that derivation of a general and a comprehensive conclusion on the subject was not possible because each direct dye and mordant interaction under investigation produced different results when mordanting according to different methods. The independent variables (mordanting method, mordant chemical, dye molecular structure associated with dye hue) applied in dyeing experiments showed that the dependent variables (%E, %F, %T, ΔE*, and fastness) do not change uniformly. Each dye–mordant interaction showed its peculiar coloration and fastness property according to different mordanting methods. Although the poorest results were obtained in yellow dye–mordant pairs, some ΔE* values of wash and light fastness were promising. The lowest ΔE* values were obtained with the black direct dye after wash fastness test regardless of the mordant used. Extreme high and low ΔE* values were obtained in the experimental part but changing the mordanting method for certain dye–mordant interactions. Among all the mordants, only mordanting with copper sulphate according to any mordanting method presented more uniform ΔE* values than the others. It must be pointed that the experimental results of this research were only valid for the direct dyes under investigation but they could be taken as a starting stage. Any generalization could not be derived from the experimental results but the only general result could be stated as the direct dyes under investigation gave dyeings with mordants and mordanting methods being peculiar to the dye–mordant interactions.
Footnotes
Acknowledgment
The authors thank Almaxtex Tekstil Sanayi ve Ticaret A.Ş. for their support in light fastness tests.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
