Abstract
The source for reactive dye transfer of cotton fabric varies in different studies, and there is limited information on the washing factors. This study explored the source of Reactive Red 195 colored cotton fabric dye transfer and reported the effects of three key washing factors and their interactions with two types of dye transfer inhibitors. First, the high-performance liquid chromatography results show that the peak of completely hydrolyzed Reactive Red 195 matches the peak of the washing residue of the fabric colored by Reactive Red 195. The dye transfer process is positively related to the hydrolyzed dyes. Based on this, response surface method models for the three factors, water consumption, water hardness, and dye transfer inhibitors concentration for polyvinylpyrrolidone and lauryl betaine were built. Both response surface method models show that due to the presence of Ca2+ and Mg2+, the increase in water hardness reduces the zeta potential on the fabric surface, and causes a decrease of electrostatic repulsion between the dye and the fabric. The dye transfer inhibiting effect is reduced. The increase in dye transfer inhibitor concentration strengthened the effect of inhibiting the transfer of hydrolyzed Reactive Red 195. In addition, there is a cross effect between water hardness and dye transfer inhibitor concentration. There is optimal water hardness and dye transfer inhibitor concentration to maximize the inhibition of hydrolyzed Reactive Red 195 dye transfer. The water consumption shows a limited effect because the washing time is long enough to reach a balance in the fabric and washing fluids. Polyvinylpyrrolidone with many hydrophilic groups is easy to combine with hydrolyzed dyes. While lauryl betaine is a small molecule surfactant, the structural differences make the response surface method model show that polyvinylpyrrolidone has a better effect than lauryl betaine. The discoveries can be used to guide dye transfer inhibitor development. Further investigations, including dye transfer inhibitor synthesis and modification, are needed.
Introduction
The dye transfer of cotton fabric for household laundry researches is dye transfer of cotton fabric for household laundry.1 –5 The current studies have explored the effect of dye transfer inhibitors (DTIs) according to their structure, 6 molecular weight and concentration, 7 and the water hardness 8 on reactive dyes.4,5,9 However, the source of dye transfer is still not precisely identified. Different researchers still use various dye transfer sources to develop their DTIs,10 –13 including reactive dyes and their hydrolyzed forms.
Some researchers have discussed the effect of polymer concentration and molecular weight based on the direct dye. 7 The DTI development also widely uses the reactive dyes.4,5,9 R Rathinamoorthy explored the effect of different DTIs on reactive dyes in inhibiting dye transfer in 2016 and 2019. In 2020, SJ Boardman et al. 3 developed and synthesized a variety of DTIs through a series of methods to inhibit the deposition of reactive dyes on various fibers with excellent effects. In 2015, MN Amin and RS Blackburn 6 explored the effect of different types of DTIs on inhibiting dye transfer for hydrolyzed reactive dyes. The source of dye transfer in cotton fabric washing is so diverse that it needs to be identified.
Also, both the single factors and the interaction effects of water consumption, water hardness, and the concentration of different DTIs, even at the statistical level with a complete design of experiment, have not been systematically explored.
Some experimental studies have reduced water and energy consumption by optimizing the washing parameters. 6 In 2014, FJ Carrion 8 tested the water hardness effect with PVNO (vinyl pyridine and nitrogen oxide, Chromabond S-403E, 9000–17,000 g/mol) and PVP (PVK-30, 66800 g/mol) for household fabric washing. Hard water has a low inhibition effect on the dye transfer in the fabric and has an excellent soft water effect. PVP (PVK-30) is the worst in hard water washing.
A thorough literature review3 –6,9 suggests that very limited work has been carried out on the source of the cotton fabric washing dye transfer. The effects of water hardness, water consumption, and dye transfer inhibitor concentration also need further exploration.
In this study, to determine the source of dye transfer in household fabric washing, we washed fabric dyed with Reactive Red 195 (RR195). Then we collected its residue and analyzed it using the high-performance liquid chromatography (HPLC) system. The dye transfer source was determined by comparing the HPLC chromatograms of the washing residue with the hydrolyzed dyes. In this way, it was possible to initially obtain the necessary information on the source of dye transfer for household fabric laundry, thus laying the foundation for more substantial future work.
Finally, we performed washing experiments on dye transfer inhibition of hydrolyzed RR195 with polyvinylpyrrolidone (PVP) and lauryl betaine. The effects of water hardness, water consumption, and concentration on DTI performance on hydrolyzed dyes were statistically investigated. An empirical model was developed for the effect of different concentrations, water hardness, and water consumption.
Experimental
Materials
This study used fabric dyed with RR195 (C31H19ClN7Na5O19S6; molecular weight 1136.3). The cotton fabric was dyed using traditional dyeing methods. First, white cotton fabric (5 g), RR195 (1% o.w.f.), and Na2SO4 (60 g/L) were added to soft water; the ratio of the total solution to the fabric was 20:1 and it was dyed for 30 min at 75°C. Then, CaCO3 (20 g/L) was used for color fixation, which took 45 min. After the dyeing process, the cotton fabric was washed thoroughly with cold water, and then with soaping agent (2 g/L) at 95°C for 15 min to ensure the cleanliness of the fabric. Finally, the cotton fabric was rinsed and dried to obtain a cotton fabric dyed with RR195. The structure information of RR195 is shown in Figure 1. Table 1 shows the structure, suppliers, and molecular weight.
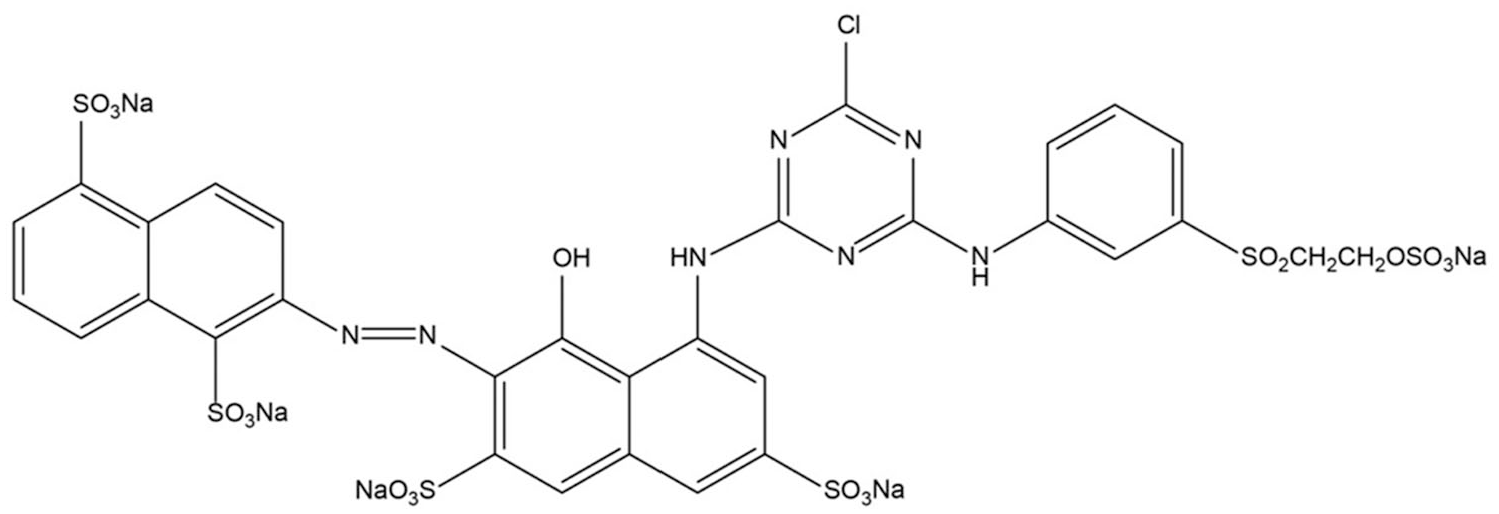
Structure of RR195.
Information about DTI.
DTI: dye transfer inhibitors; PVP: polyvinylpyrrolidone.
The dye transfer receptor is a CN-42 standard white knitted cotton (290 g/m2 weight, 130 cm width) provided by the Center for Testmaterials B.V. The laundry detergent used for the experiment was following QB/T 1224-2012 standard. Both soft and hard water were prepared according to Method B in GB/T 23119-2017.13,14
Reactive Red 195 Fabric Washing
For the RR195 fabric washing test, there were three duplicate samples to control the random error, and fabric of 5 g was used in this study. In each test, there was 0.2 g standard laundry detergent and 100 mL standard hard water, which follows the GB/T 23119-2017. The washing temperature was 60°C, and the washing time was set to 2 h. After the test finished, the residue was collected and sent to the HPLC system.
Liquid chromatography and mass spectrometry analysis
This study conducted analysis of the source of the dye transfer using the Waters e2695 High-Performance Liquid Chromatography (HPLC) system, which was made by Waters Technology (Shanghai) Co. The chromatographic column was a Waters Symmetry C18 (5 μm, 4.6 mm×250 mm) with gradient elution.
The flow rate is l mL/min, and the injection volume is 20 μL. The column temperature is 30°C, and the sensitivity of the reaction measurement is 0.04 AUFS. The gradient elution conditions for RR195 are as follows: the mobile phase A is 0.002 mol/L tetrabutylammonium bromide and 0.05 mol/L ammonium acetate aqueous solution. The mobile phase B is chromatographic grade ethanediol solution and the DAD detection is at 542 nm.
This study prepared the hydrolyzed RR195 as follows: 0.003 g dye was dissolved in 100 mL of 10% NaOH solution at 90°C (pH=13). The hydrolysis process lasted 60 min after which 5 mL samples were taken at 0, 5, 15, 30, and 60 min and injected into the HPLC system. To determine the specific composition of the red fabric laundry residue, the HPLC test conditions were the same as the washing residue of RR195 colored fabric. The HPLC results were compared when all the tests were finished. The washing residue was also sent for mass spectrum (MS) detection: acquisition SW version, 6200 series TOF/6500 series Q-TOF B.08.00 (B8058.0).
Design of Experiments for Washing Factor Analysis
The experimental factors include water hardness (
JMP is the software developed by the SAS Institute based on design of experiment (DOE) principles.15,16 We obtained the randomization and factor combinations using JMP V11, 17 as shown in Table 2. The experimental design is controlled by the algorithm of JMP and reached the criteria of the response surface method (RSM). 18 This study implemented the DOE results in Table 2 and collected the color data.
Washing experimental design.
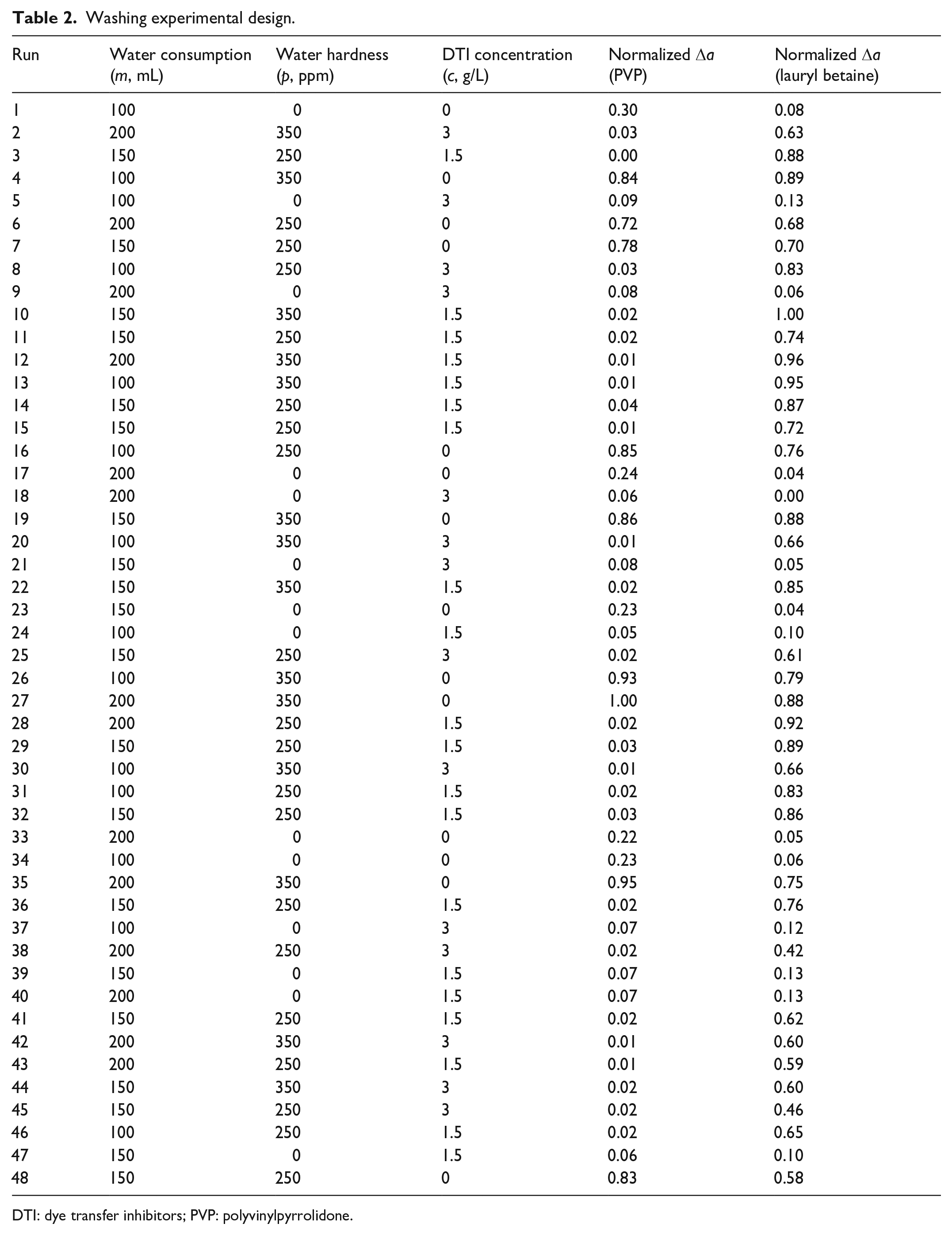
DTI: dye transfer inhibitors; PVP: polyvinylpyrrolidone.
In the experiments, the washing temperature was set at 30°C. Each washing program includes one main wash period and two rinsing periods. The main washing period lasts 20 min and each rinsing period is 2 min. The washing rotation speed is set at 40 rounds/min. All washing experiments are done using the SW-24C Type Washing Fastness Tester. The 12 cups selected for the experiment had a capacity of 550 mL and all conformed to GB/T3921.5-1997. Twelve rounds of washing could be carried out simultaneously in one experiment. An amount of 5 g of dye transfer receptor fabric sample, 2 g/L standard laundry detergent (follows the QB/T 1224-2012 standard), and 0.03 g/L hydrolyzed RR195 solution were placed in the washing tester.
This study also built the response surface of PVP and lauryl betaine by analyzing the effect of water consumption, water hardness, and concentration.
Color Testing
A HunterLab ColorQuest XE colorimeter (Shanghai New Union Electronics Co., Ltd.) was used to measure the color change of fabrics before and after washing. A small aperture of 6.3 mm was used for measurement, and the spectral range was 400–700 nm. The number of flashes in each measurement was 1 in LAV mode and 2 in SAV mode and the small time interval between measurements was 3 s.
This study used the Δa value (red direction) value to measure the dye transfer results according to the CIELAB color space. 19 Eight randomly selected points were measured on each piece of fabric to ensure data accuracy and to reduce the impact of errors. A larger Δa means a darker fabric. In other words, a better DTI will reduce the Δa.
Results and Discussion
Source of Dye Transfer
The HPLC Results Comparison
The HPLC test results, Figure 2 (top), clearly show that there are hydrolyzed dye molecules in the washing residue.20,21 The washing residue peak (87%) appears at the elution time tR = 5 min corresponding to the spectrum of the hydrolyzed RR195 of 60 min and the remaining 13% of the peak is laundry detergent. The MS spectrum from 5.277 to 5.285 min for the precursor ion of m/z 922.01 is shown in Figure 3. This result is confirmed to be hydrolyzed RR195. There is a difference of 1, and it may be that the H in the hydroxyl group is free in the aqueous solution.
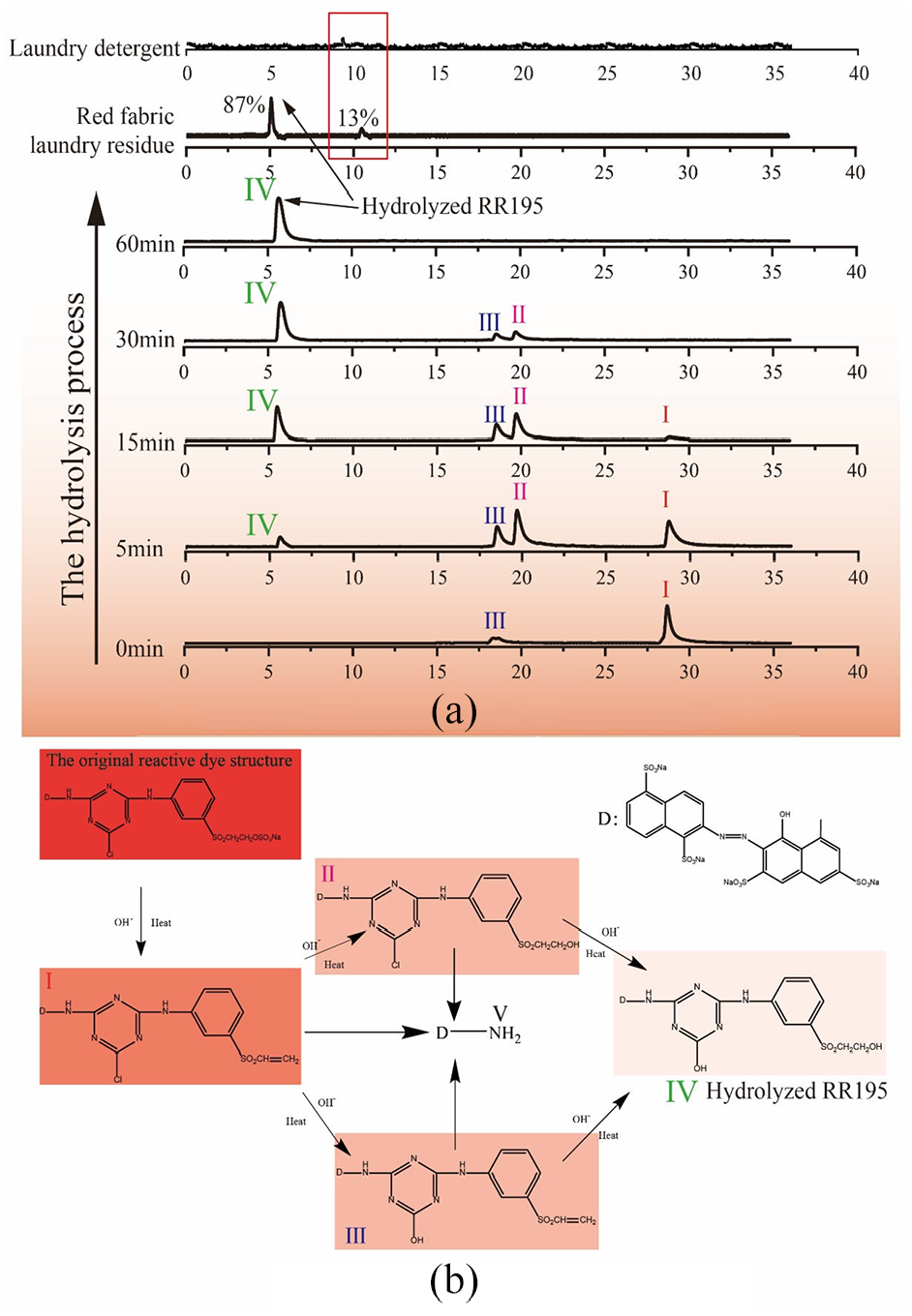
The HPLC results and hydrolytic route of RR195: (a) the HPLC results of washing residue and RR195 and (b) hydrolytic route of RR195.
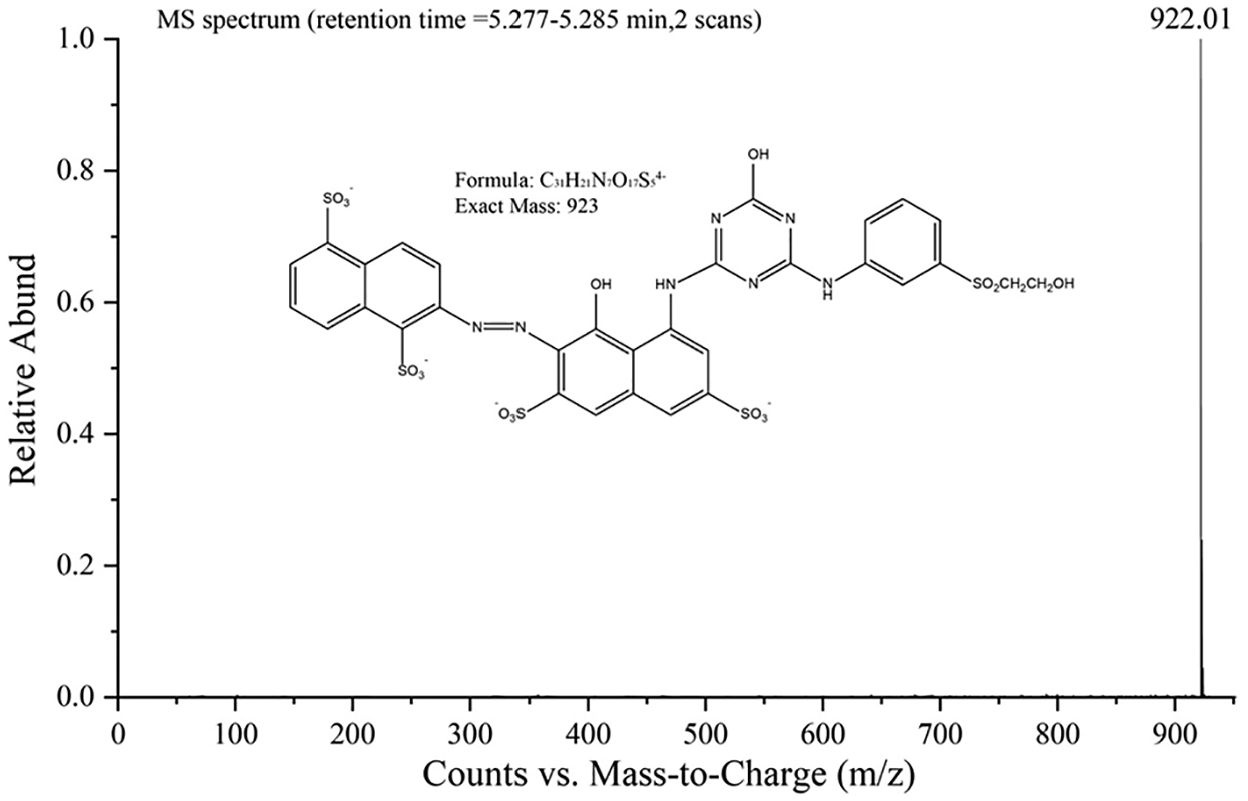
Mass spectrum of hydrolyzed RR195.
In Figure 2(a), from 0 to 60 min, the four hydrolysis peaks appear in turns of I and III, then II and IV. After 60 min of hydrolysis, the final substance that appeared in the chromatogram is IV, 22 whose structure is shown in Figure 2(b).
Figure 2(b) shows the hydrolysis process of RR195, where D is the dye parent, located on the upper right. Research by M Klančnik21,23 showed that reactive dyes with heterobireactive groups have five different forms in alkaline aqueous solutions. Since the polarity of the hydroxyl group is greater than that of the chloro group and the carbon–carbon double bond, I is the chromatographic peak of reactive dyes with heterobireactive groups. The reactive dye with vinyl sulfone structure can be hydrolyzed to obtain hydrolyzed dye (II) with a primary alcohol structure. The hydroxyl group attacks the chlorine atom to obtain the sym-triazine-hydrolyzed dye (III) having a hydroxyl group. The hydrolyzed dye (IV) contains two or more hydrolyzed structures. In addition, the bridging imino group between the dye precursor and the symmetric triazine ring will also be hydrolyzed under alkaline conditions to produce a dye (V) with an amino structure. 24
However, the hydrolysis of reactive dyes is a continuous process. Structures II and III are only intermediate products in the hydrolysis process and cannot exist stably. In the end, only hydrolyzed dye IV can stably exist in an aqueous solution. It is combined with cotton fabric through van der Waals force, so that it is easily shed during the washing process and causes dye transfer.
Effects of Different Types of DTIs
To evaluate the performance of PVP and lauryl betaine, this study analyzed the influence factors including water consumption, water hardness, concentration, and their interactions. Table 2 shows the collected color data. The analysis of variance and fitting process were done using the JMP software based on the least square regression method. 15 The data obtained by the experimental measurement were processed by min–max normalization. 25
Influence Factors and the Effect of PVP
Based on the Δa in Tables 2–4 are the ANOVA results and the RSM model terms analysis. The F-statistic in Table 3 is 199.03 at the significant level of 0.99, which means that the RSM is good enough to reflect the factors and the effects of their interactions. Table 4 shows the RSM model term values, and their error estimates, t statistics, and confidence interval. According to equation (1) and Table 4, we can plot the RSM model in Figure 4.
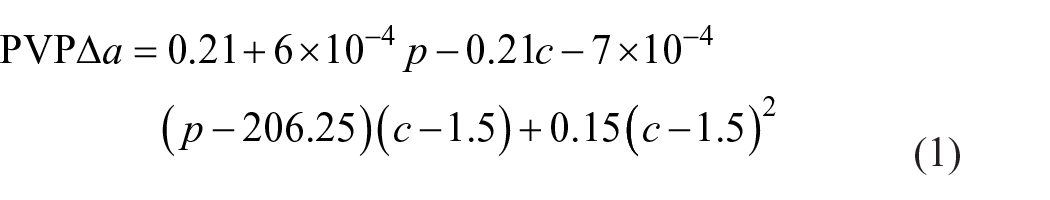
ANOVA results of PVP for response surfaces.
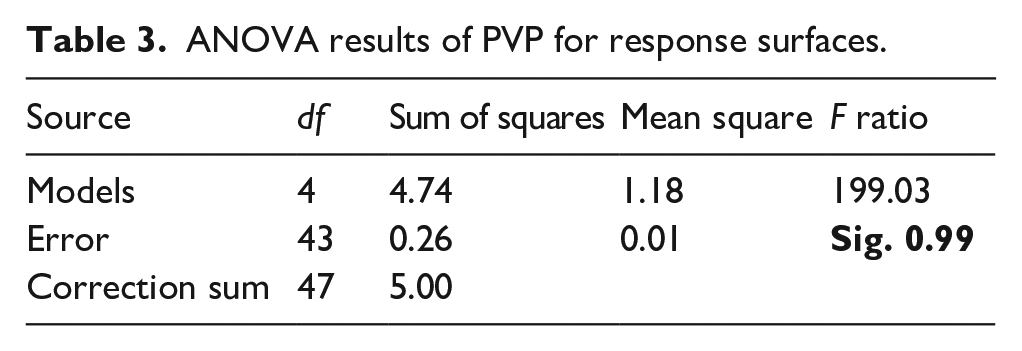
PVP factors analysis and the RSM model term coefficient.
SE: standard error; DTI: dye transfer inhibitors.
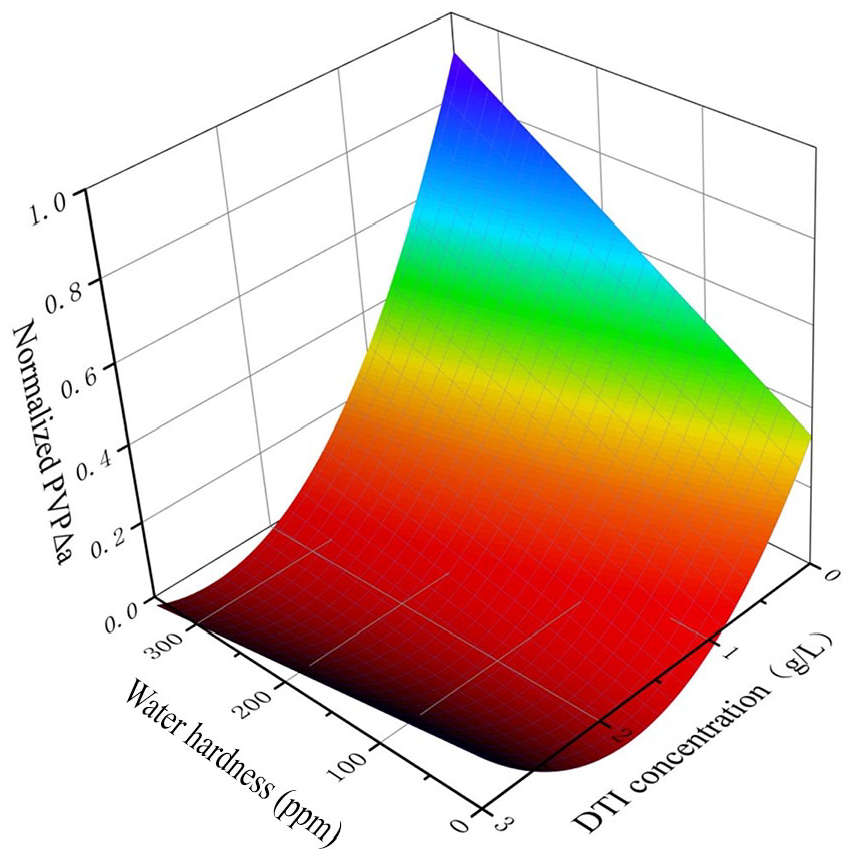
Response surface of PVP.
Both Figure 4 and Table 4 show that the DTI concentration has a significant positive effect on Δa. The lactam in the PVP molecule is a strong polar group with hydrophilic and polar group effects, which can easily form hydrogen bonds. 26 The higher the molecular weight of the hydrolyzed dye, the higher the formation of van der Waals forces and the stronger the correlation with PVP. 27 Meanwhile, many carbonyl groups are arranged continuously on the pyrrolidone ring on one side of the PVP molecular chain. They can form hydrogen bonds with the hydroxyl and amino groups in the dye molecule, strengthening the link between them. The increase in PVP concentration resulted in the formation of more H-bonds with dye and thus a stronger interaction with the dye, thus decreasing the potential for dye transfer.
Water hardness plays a significant correlation in dye transfer. For each level of water hardness, the PVP concentration effect shows a significant nonlinear effect, which corresponds to the
As shown in Figure 4, when the water hardness is constant, as the DTI concentration increases the Δa value becomes smaller. The effect of PVP on cotton fabrics in inhibiting dye transfer increases. When the DTI concentration is constant, as the water hardness increases the dye transfer inhibition effect of PVP on cotton fabrics decreases.
The effect of water hardness on the interaction between PVP and RR195 can be explained by the Derjagin–Landau–Verwey–Overbeek (DLVO) theory: 28
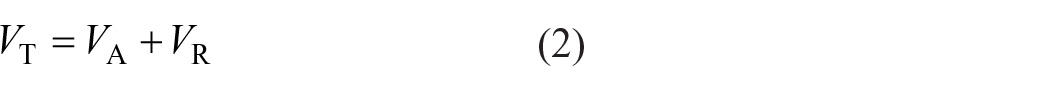
where
As shown in Figure 5, the fabric is negatively charged in the aqueous solution, and the hydrolyzed RR195 molecule is also negatively charged. When washing fabrics, the dye molecules approach the receptor from periods 1–4, as shown in Figure 5(b). In period 1, the dye molecules are far enough from the surface of the fibers,
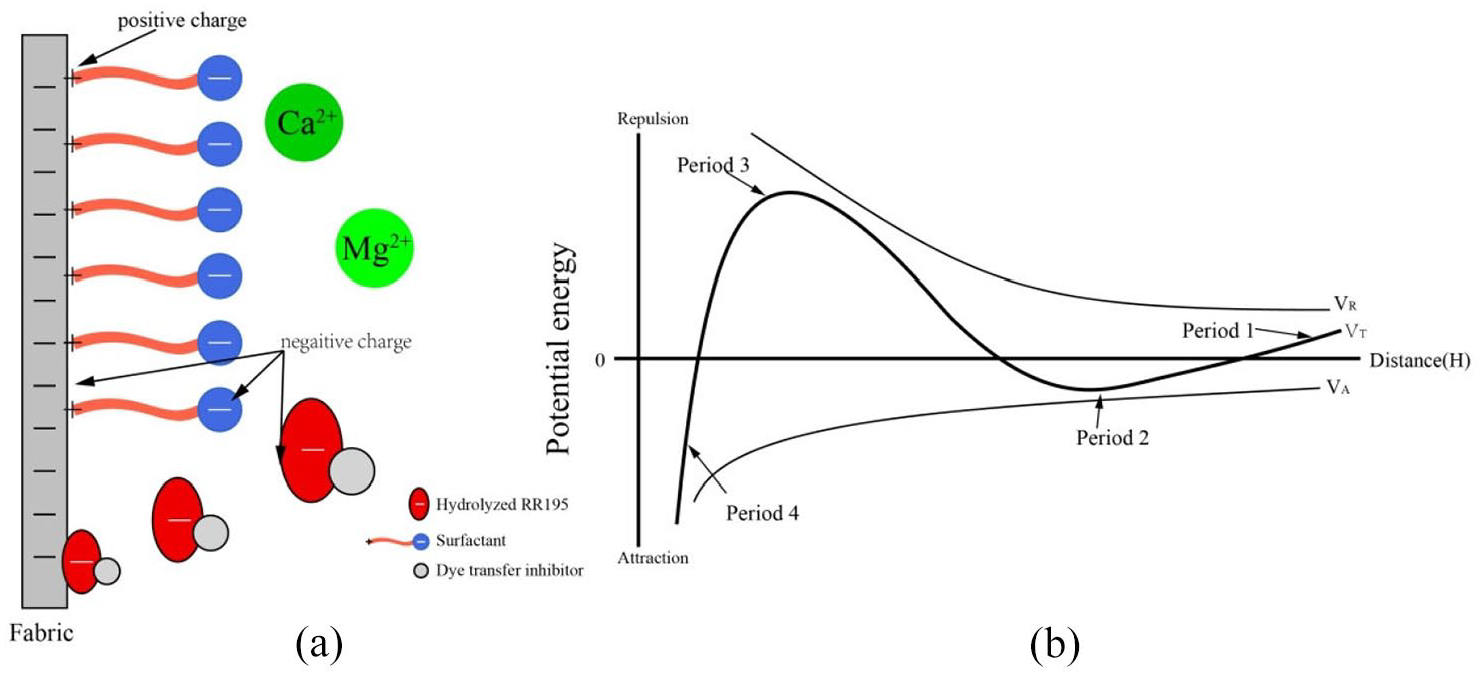
(a) The influence of Ca2+ and Mg2+ and (b) DLVO theory.
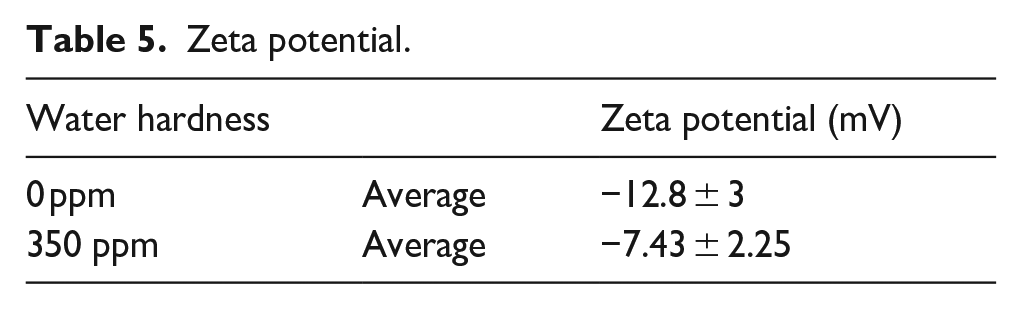
However, as the water hardness (Ca2+ and Mg2+) increases, the fiber surface zeta potential decreases, which may tend to fall into period 4 or 2, as shown in Table 5. 29 On the contrary, Ca2+ and Mg2+ combine with the sulfonate in the dye, resulting in an insoluble precipitate. 29 The insoluble precipitate will remain in the washing system and be rinsed away by the effect of fluid dynamics. 30
Zeta potential.
The experimental results also show that water consumption has no significant effect. The reason is that the washing time is long enough to achieve a concentration balance between the fluid and the fabric.
Influence Factors and the Effect of Lauryl Betaine
Tables 6 and 7 are the ANOVA results of the RSM model and its terms of analysis. The F-statistic in Table 6 is 132.00 at the significant level of 0.99. According to equation (3) and Table 7, we can plot the RSM model in Figure 6 As follows:
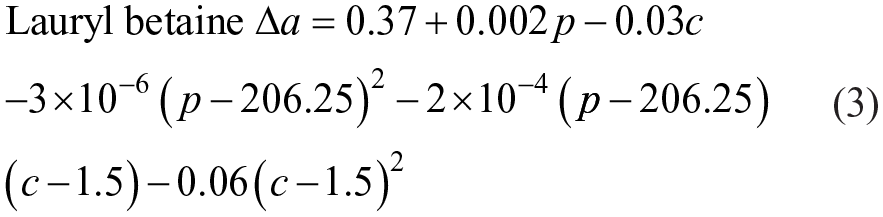
Both Figure 6 and Table 7 show that the water hardness also plays a significant negative correlation on Δa. In the same way, higher water hardness will also reduce the ability to inhibit dye transfer. The influence of water hardness is also due to the presence of Ca2+ and Mg2+.
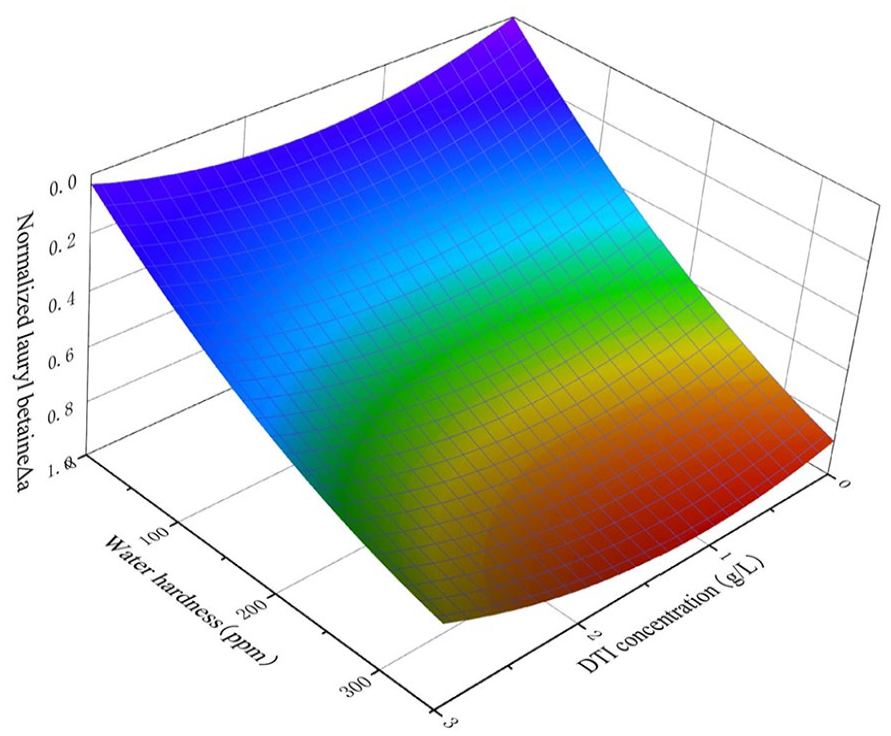
Response surface of lauryl betaine.
ANOVA results of lauryl betaine for response surfaces.
Lauryl betaine factors analysis and the RSM model term coefficient.
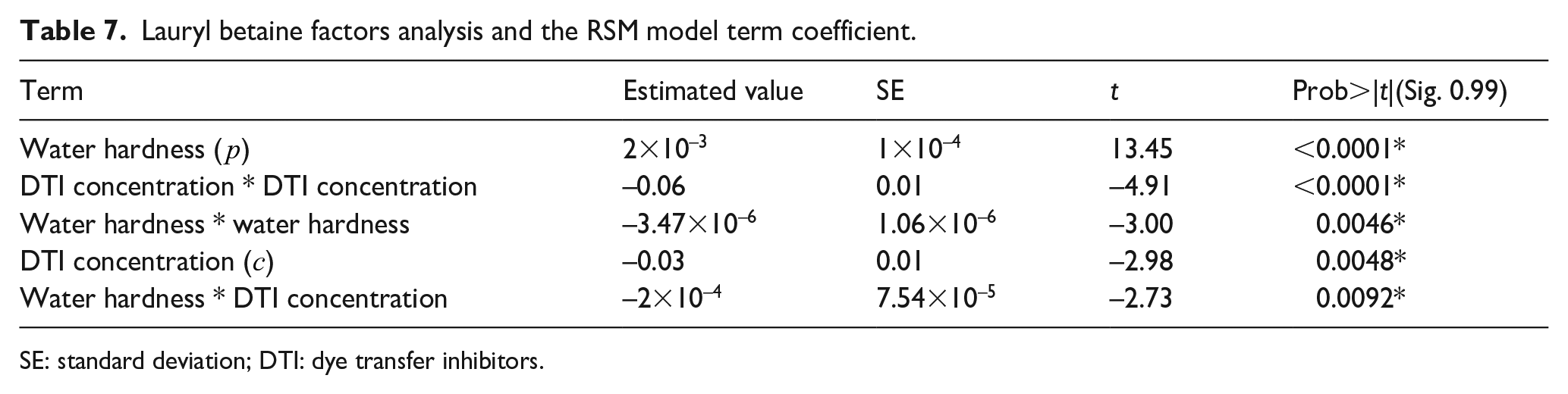
SE: standard deviation; DTI: dye transfer inhibitors.
However, for each level of water hardness, the lauryl betaine concentration effect also shows the nonlinear effect, which corresponds to the
If additional hydroxyl sites are added to PVP and lauryl betaine, the formation of H-bonds between them and the fabric may enhance the dye transfer inhibition effect. The increased hydrophilicity of DTIs indicates that their interaction with water is greater than their interaction with the fabric.
Quantum Chemistry Calculation
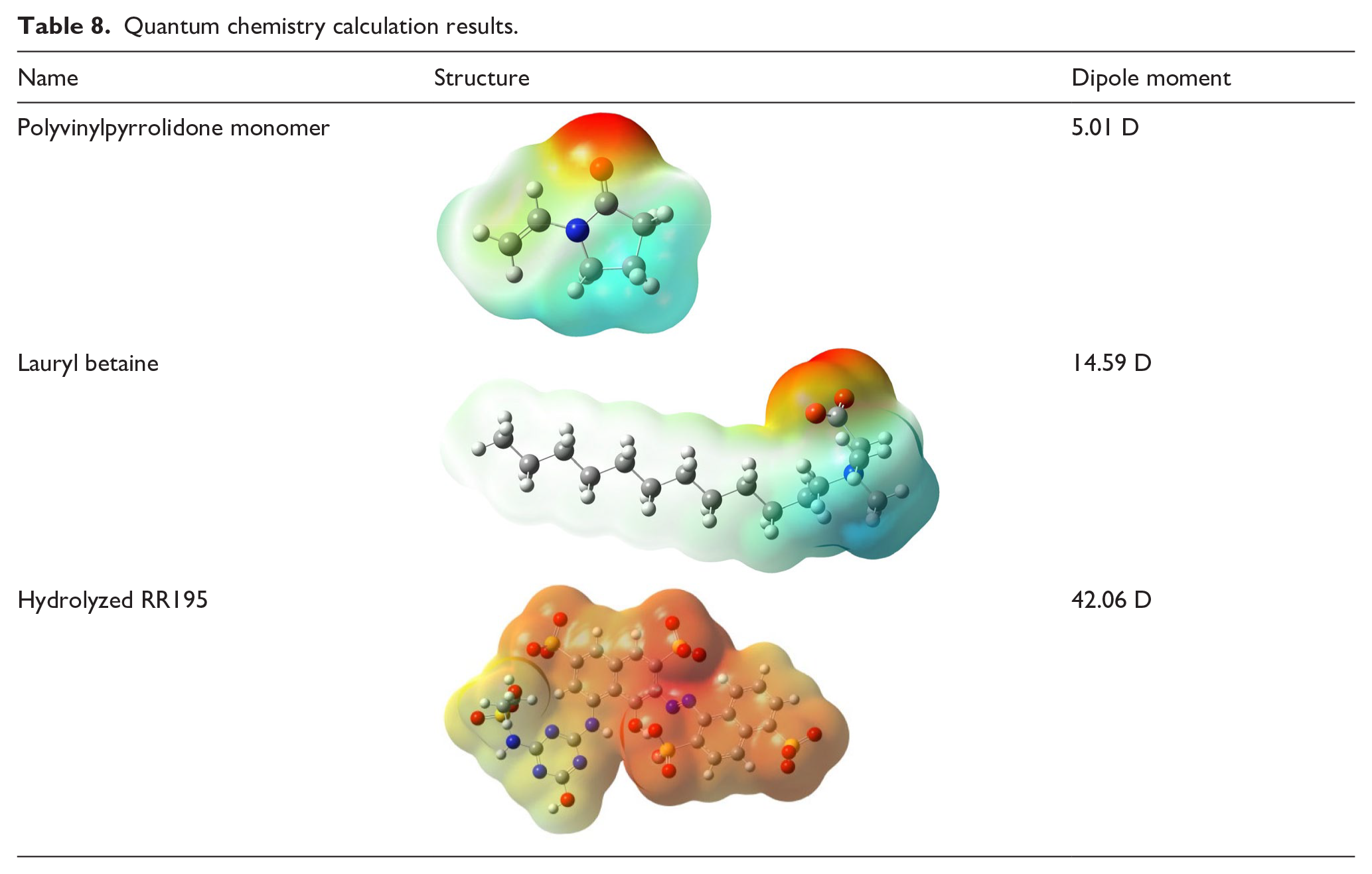
To further discuss the source of interaction between DTI and hydrolyzed RR195, we also conducted the quantum chemistry calculation. The molecule structural optimization and potential energy surfaces were completed using the WB97XD method in DFT theory and the cc-PVTZ group. 31 The calculated residual values all converged. Table 8 shows the PVP monomer and lauryl betaine calculation results and the hydrolyzed RR195 molecule.
Quantum chemistry calculation results.
The Keesom interaction describes the weak interaction force of hydrolyzed RR195 and DTI: 32
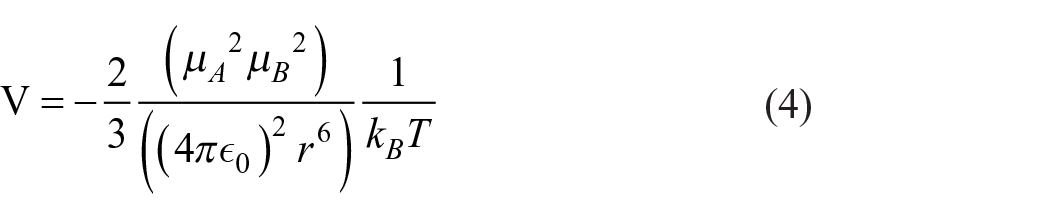
where
We assign
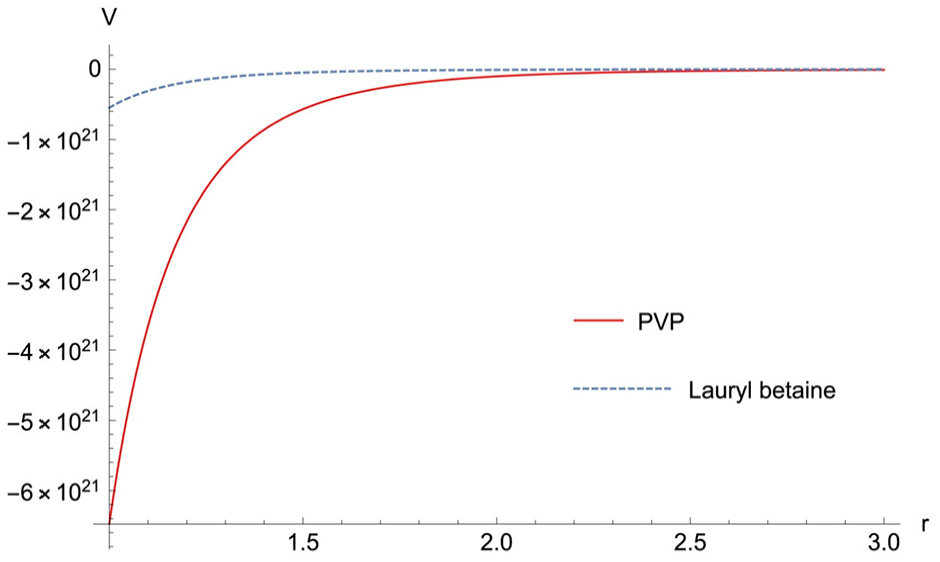
The Keesom interaction between the DTI and hydrolyzed RR195 molecule.
As shown in Figure 7, the Keesom interaction of PVP is larger than the lauryl betaine, especially when
Conclusion
In this study, a thorough investigation of the dye transfer source and influence factors was conducted.
The HPLC results show that hydrolyzed dye molecules are in the washing residue. Future experimental and theoretical studies on the reactive DTIs should be based on the hydrolyzed dyes.
Based on the water hardness and DTI concentration, the RSM model R2 reached 0.95 (PVP) and 0.93 (lauryl betaine), respectively. The experimental results show that the water hardness and DTI concentration significantly affect the dye transfer inhibition. Ca2+ and Mg2+ not only reduce the zeta potential on the fiber surface but also reduce the electrostatic repulsion between the dye and the fiber. The water consumption had a limited effect. The washing time is long enough to reach the concentration balance between the fabric and washing fluid.
The dipole moments of PVP, lauryl betaine, and hydrolyzed RR195 were obtained by quantum chemical calculations. Bringing the dipole moment into the Keesom equation can calculate the interaction between them, which proves the existence of weak interaction force between DTIs and hydrolyzed RR195.
These discoveries can be used to guide the dye transfer inhibitor development. Further investigations, including dye transfer inhibitor synthesis and modification, are needed.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
