Abstract
In this investigation, the mechanism of dye transfer during the mixed washing of natural cellulosic fabrics was elucidated through a comprehensive study of the hydrolysis of Reactive Red 195 (RR 195) and the subsequent adsorption of its hydrolysis products onto cellulose, approached both experimentally and theoretically. Initially, Reversed-phase high-performance liquid chromatography (RP-HPLC) was employed to identify the final products of Reactive Red 195 hydrolysis, collectively referred to as the unfixed dye substance (UDS). This data provided theoretical insight into the hydrolysis mechanism of the dye. Following this identification, an adsorption kinetic model was established to delineate the interaction of the hydrolysis products with cellulose. Adsorption exhibited its maximal rate at a temperature of 60°C, under conditions of a wash bath ratio of 120:1 and a pH value of 7. Furthering the investigation, a molecular dynamics model was constructed to detail the adsorption process of these hydrolysis products on the hydrophobic surface Iβ(1 0 0) of cellulose within an aqueous environment. This model offered precise insights into the adsorption volume, kinetics, and mechanistic interaction at a molecular scale. Conclusive evidence from both theoretical analyses and experimental results suggested that the hydrolysis products, devoid of reactive groups, were incapable of chemically interacting with the hydroxyl groups inherent to cellulose. Instead, it was discerned that these products underwent a physical adsorption process onto the cellulose surface. Such adsorptive behavior is postulated to be a predominant factor influencing the dye transfer phenomenon in mixed washing conditions.
Keywords
Introduction
In home laundering, as living standards improve, there is an increasing demand for fabrics with high color saturation. However, the washing of highly saturated colored fabrics often encounters issues such as low colorfastness, contamination of the wash liquor, and fabric color bleeding. 1 Reactive dyes have become the preferred choice for cotton fabric dyeing due to their vibrant color yield, and the process of preparing colored fabric has been significantly refined. 2 Despite these advancements, the high color saturation of cotton fabrics is vulnerable to various washing parameters in domestic laundering. After several wash cycles, “unfixed dyes” are released. Concurrently, some dye-fiber covalent bonds may break, causing the dyes to detach from the fabric. This results in diminished colorfastness and leads to problems such as color fading and dye transfer.
The molecular structure of reactive dyes contains reactive groups that can react with -OH in fibers. It causes covalent bonds between the dyes and cellulose, resulting in good dyeing fastness. Although reactive dyes have high dyeing fastness, the daily laundering of different color fabrics can still produce color fading and dye transfer of colored clothes. Gorenšek 3 found that the covalent bonds between reactive dyes and cellulose fibers will break and shed in water. However, color fading and dye transfer still occurred in low-temperature laundering, indicating that hydrolysis in low-temperature washing may not be the only factor for dye transfer. Abuhatab et al. 4 have shown that the different washing parameters affected the adsorption effect, such as pH, temperature, stirring time, adsorbent dosage, and initial dye concentration. Similarly, Rady et al. 5 discovered that the cogon red removal rate was influenced by the adsorbent dose, temperature, and pH. Reactive dye is easily hydrolyzed and produces the hydrolyzed dye. In the process of mixed laundering of bleached cotton fabrics (BCFs) and the cotton fabrics dyed in highly saturated color (CFSCs), dyes dropped out of highly saturated fabrics due to unconsolidated materials under the combined effect of washing parameters, which resulted from desorption and flooding of the laundering solution. 6
Currently, efforts to prevent and control the fading and color transfer issues in dark-colored cotton fabrics are primarily focused on the development of color protectants and recommendations for washing fabrics separately. However, there is a deficiency in research on the mechanisms of fading and color transfer during the laundering process of dark-colored cotton fabrics. This gap hinders the fundamental resolution of fabric fading and color transfer issues. Therefore, investigating the color mixing issue during the combined washing of dark and light-colored cotton garments with high color fastness is crucial. Such research aligns with the contemporary direction of home laundering studies.
Researchers have explored the efficacy of various nanomaterials for dye adsorption in a series of publications. A nanocomposite consisting of polyaniline, carboxymethyl cellulose, and TiO2 was used to successfully adsorb Congo Red dye from aqueous solution. 7 Polypyrrole/CMC composite nanoparticles modified with a cationic surfactant were employed to efficiently remove Reactive Red 56 and Reactive Blue 160 dyes. 8 The adsorption efficiency of Congo Red dye was significantly enhanced using zeolite particles functionalized with a cationic surfactant. 9 Fe3O4@NiO core–shell magnetic nanoparticles were synthesized for the highly efficient removal of Alizarin Red S anionic dye from contaminated effluent. 10
This study primarily achieved the theoretical and experimental determination of the structure of floating color substances released during washing. It also investigated the mechanisms behind color fading and color transfer issues in cotton fabrics dyed with high fixation rate reactive dyes. By establishing an adsorption model for the interaction between UDS and cellulose in an aqueous environment, the adsorption mechanism of RR 195 was elucidated at the molecular level. This work aims to provide theoretical references for the prevention and control of fabric fading and color transfer.
Materials and methods
Chemical reagents and materials
Bleached cotton fabric with a density of warp and weft yarn (27.8 ± 2.5)tex was provided by the CHTC DAYAO TEXTILE Company. Reactive red 3BS (RR 195) was purchased from the Runtu Company (Zhejiang, China). The A-502F type soaping agent, was supplied by Liansheng Chemical (Suzhou, China). The other chemical agents used in this experiment included sodium sulfate, sodium carbonate, glacial acetic acid, hydrochloric acid, tetrabutylammonium bromide, ammonium acetate, and acetonitrile, which were obtained from Sinopharm Chemical Reagent Company. It was worth noting that cellulose Iβ(1 0 0) considered as the adsorbent while UDS regarded as the adsorbate during the modeling process.
Preparation of highly saturated cotton fabric
First, the standard prewashing treatment is performed on cotton fabric before dyeing. The bleached cotton fabric was placed in a laundering machine at a temperature of 23 ± 2℃ and a relative percentage of 65 ± 5%. Then the cotton and linen program, was selected, spun and dried after washing, and kept it in a vacuum. To avoid affecting the testing effect, ironing before dyeing the fabric would eliminate the fabric folds and prevent swimming and staining unevenness during the dyeing process. The dyeing process was single-bath single-step dyeing, with dyeing parameters including the amount of dye being 2% (o.w.f), the fabric weight being 4.16 g, and the dyeing bath ratio set to 10:1, as shown in Figure 1.

RR 195 dyeing process.
Preparation of RR 195 hydrolysis sample
RR 195 (0.1 g) was dissolved in 100 mL of buffer solution at pH 10, 11 heated to 60℃, and immediately sampled at 7, 12, 18, 25, 35, 45, and 55 min, and 5 mL was taken and cooled on ice. After sampling, each sample was poured into pH 4 hydrochloric acid buffer until a 20 mL sample was obtained. Figure 2 shows the process preparation of the RR 195 hydrolysis sample. The absorbance of the laundering residue was tested at the maximum absorption wavelength of 542 nm. The adsorption and desorption rates were calculated using equations (1) and (2), respectively.
Where Pd is the desorption rate (%); Cd is the mass weight concentration of dye in the wash solution (mg/mL); V is the volume of the washing water solution (m/L); M is the amount of floating color on the dark cotton fabric (mg). Where Pa is the adsorption rate (%); Ca,1 is the mass concentration of dye in the washing water solution before adsorption (mg/mL); Ca,2 is the mass concentration of dye in the washing water solution after adsorption (mg/mL); Md is the amount of desorption of dark cotton fabric (mg).

Preparation of hydrolyzed samples.
The laundry residue is derived using CFSCs mixed with BCFs to qualitatively detect the type of dye shedding by CFSCs under the action of washing parameters and the type of dyes adsorbed on BCFs during mixed washing. 12 Three pieces of highly saturated fabric and three pieces of bleached fabric with 25 g were mixed launderings under bath ratios of 100:1 and 60℃. 13 The fabrics, which were highly saturated, were placed in a buffer solution with specific temperature, water volume, and pH levels. Four controlled trials were then conducted: (1) Washing at a temperature of 40℃, with a washing bath ratio of 100:1 and a solution pH of 11. (2) Washing at a temperature of 60℃, with a washing bath ratio of 100:1 and a solution pH of 9. (3) Washing at a temperature of 60℃, with a washing bath ratio of 100:1 and a solution pH of 11. Washing at a temperature of 60℃, with a washing bath ratio of 140:1 and a solution pH of 9. The rotation speed of the stirring device was set to 80 rpm. Timing began immediately after placing the fabric into the device. The washing residue was collected at 7, 12, 18, 25, 35, 45, and 55 min, respectively. A 5 mL sample was obtained and cooled on ice. It was diluted and fixed to 20 mL with pH 4 hydrochloric acid cold buffer, filtered with a 0.22 μm aqueous filter head, collected and set aside, as shown in Figure 3.

Simulated washing device for CFSCs and BCFs.
Chromatographic separation conditions
The washed-off dyes were analyzed by HPLC using reversed-phase high-performance liquid chromatography (RP-HPLC). 14 The chromatographic column was an Agilent Eclipse XDB-C18 column (5 μm, 4.6 mm × 250 mm). The mobile phase comprised solvents A and B, where solvent A was 0.002 mol/L tetra butyl ammonium bromide and 50 mol/L ammonium acetate, and solvent B was acetonitrile. The flow rate was 1 mL/min, and the injection volume was 2.0 μL. The column temperature was 30℃; DAD ultraviolet and visible (spectrophotometry) was selected. The maximum absorption wavelength of 542 nm of RR 195 was chosen as the detection wavelength for this work. 15 The sensitivity of the detection was 0.04 AUFS. The gradient elution system used in the HPLC analysis is shown in Table 1.
Gradient elution system for chromatographic analysis.

Adsorption experiments
To simulate the adsorption dye transfer phenomenon and adsorption pattern of bleached fabrics in a real mixed washing environment, the prepared specimens were cut to 16 cm × 16 cm and weighed at 4.16 g. The washing parameters were set as washing temperature (20, 30, 40, 50, 60)°C, washing bath ratio (60:1, 80:1, 100:1, 120:1, 140:1), pH of the washing solution (7, 8, 9, 10, 11). The amount of water used for washing was changed according to the set bath ratio. To serve as a parallel control experiment, the amount of dye transfer from the desorbed samples at different times was used as an indicator of the amount of dye transfer from the mixed washing system at the same time. The wash residue was collected at 3, 5, 7, 12, 18, 25, 35, 45, and 55 min after the samples were adsorbed. The adsorption rate of the dye was calculated by differentiating the amount of dye adsorbed at different times.
To obtain the variation of dye adsorption on bleached fabrics with washing time at different washing temperatures, washing time at different washing temperatures, washing bath ratios and pH of the washing water solution, the values of the adsorption variation were fitted to pseudo-first order kinetic model of the equation (3) and pseudo-second kinetic model of the equation (4).16,17


Where qt is the amount of dye adsorbed on the light colored cotton fabric at washing time t (mg), qa,e is the equilibrium amount of dye adsorbed on the light colored fabric (mg), ka,1 and ka,2 are the pseudo-first and pseudo-second adsorption rate constants respectively.
To further investigate the dynamic process of dye adsorption on bleached fabrics during the laundering process, a first-order exponential decay model is fitted using equation (5), and a second-order exponential decay model is fitted using equation (6).

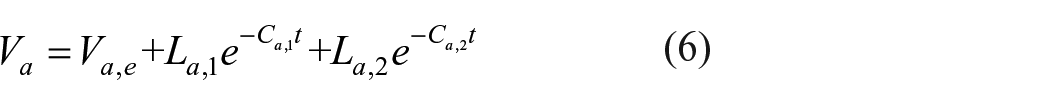
Va represents the adsorption rate of the dye on the bleached cotton fabric at a given washing time t, measured in mg/min; Va,e denotes the adsorption rate of the dye when it reaches adsorption equilibrium, also in mg/min; La,1, and La,2 refer to the adsorption rates of the dye at different stages in the exponential decay model of varying orders, quantified in mg/min; Ca,1, and Ca,2 represent the adsorption rate constants of the dye at different stages in the adsorption rate model, expressed in min−1.
Preparation of modeling and methods of molecular simulation
Modeling
In this paper, a strategy was used to investigate the adsorption between cellulose and UDS by employing the adsorption locator (AL) module and Forcite module in Materials Studio 2019.18,19 The Forcite module performs various molecular mechanics calculations on molecular and periodic systems using simulations based on classical force fields. The adsorption locator is able to simulate substrates loaded with a fixed composition of adsorbents or mixtures of adsorbents and to find low-energy adsorption sites on periodic and non-periodic substrates, or to investigate the preferential adsorption of mixtures of adsorbent components. Where the Forcite dynamic was used to calculate the interaction energy and the AL module determined the adsorption of UDS on cellulose Iβ(1 0 0) surface in simulation complex. The energy of the UDS/C system was derived from the results to elaborate the adsorption mechanism. The complex system was constructed by first building a new surface from the cellulose crystal surface, then adding unfixed dye substance to the cellulose Iβ(1 0 0) surface, and finally optimizing the structure. The adsorption of Reactive Red 195’s hydrolysis products on cellulose was modeled in aqueous environments, with water serving as the solvent. In this paper, the adsorbent in the molecular simulation was considered the hydrophobic surface of cellulose Iβ(1 0 0) by Mazeau and Wyszomirski. 20 Cellulose microfiber models with 18 glucose chains with a common four layers and 18 glucose residues on each chain were generated by a cellulose-builder. 21 The established box dimensions are 72.272 × 75.608 × 93.04 Å, α = β = 90° and γ = 86.55°. In addition, the aqueous layer was fixed at 15 Å. These parameters are available to adapt the modeling simulation. 22
Computational details
The COMPASS II force field was applied to the simulation process in this paper, such as optimizing the structure of RR 195, UDS, and cellulose.23,24 The steepest descent algorithm minimization method was used to optimize the molecular structure. The converging standard tolerance was 0.5 kcal/mol/Å and 0.001 kcal·mol−1 for energy. Monte Carlo simulation of the adsorption process showed that the surface of cellulose Iβ(1 0 0) was 41.545 × 25.556 Å. Furthermore, the atomic set was adapted to define the surface area, and the maximum adsorption distance was set to 10 Å through the Materials Studio software.
Finally, in the MD simulation, the NVT ensemble (constant atomic, constant volume, and continuous temperature) was employed at 298 k temperature with a simulation time of 2 ns, and the Nose algorithm was used to control the temperature. 25 The method of Forcefield assigned was used to evaluate the atomic charges of electrostatic interactions. The summation method for electrostatic interactions was addressed using the atom based. In addition, van der Waals interactions were computed with a spline width of 1 Å and a cutoff distance of 12.5 Å.26,27 Finally, the setting of parameters for UDS/C system were listed in Table 2.
Setting of important parameters in the molecular construction work.

Monte Carlo and molecular dynamics simulations
In the UDS/C system, the interaction between UDS and the cellulose Iβ(1 0 0) surface was investigated using MD to calculate the interaction energy and MC to determine the optimal adsorption sites. 28 The adsorption energy (Eint) on the surface of cellulose Iβ(1 0 0) was calculated using equation (7).
The interaction energy (Ecell-dye) of the adsorbate\adsorbent system was calculated using equation (8). It is used to quantify the energy fluctuations during the adsorption process in simulated aqueous environments.

where Etot denotes the energy of the whole energy, Ecellulose is the energy of Iβ(1 0 0), and Edye shows the energy of geometric optimization of the UDS.
In aqueous environments, the intermolecular interaction energy Eicell-dye was used to evaluate the relevant affinity between UDS and cellulose Iβ(1 0 0):

where Etot is the sum of energy of the energy of water (Ewater), the energy of USC (Edye), and the energy of cellulose (Ecelllulose); Edye-water is the interaction energy of UDS with water; and Ecellulose-water is the interaction energy of the cellulose with water.
Results and discussion
Qualitative analysis of hydrolysis chemical products of RR 195
In the conventional state, RR 195 is composed of a reactive group of structure I with the parent dye linked to a β-vinyl sulfate group and monochloro-s-triazine group. First, β-vinyl sulfate will cause hydrolyze and then convert into a vinyl sulfone group to obtain dye Ⅱ which is composed of sulfone and a monochloro-s-triazine anisotropic double reactive group. As Ⅱ continued to hydrolyze, vinyl sulfone reactive groups were broken with the bond generated as monochloro-s-triazine hydrolysis product Ⅲ with a beryl alcohol structure. Additionally, the monochloro-s-triazine reactive group breaks its bonds, resulting in the formation of the vinyl sulfone group hydrolysis product Ⅳ. The continuing hydrolysis of Ⅲ and Ⅳ generates bifunctional hydrolysis product Ⅴ, which loses its reactive activity. Finally, hydroxyl groups substitute all the reactive groups in Ⅴ, completely deactivating them, and rendering the reaction irreversible. The mechanism of hydrolysis of RR 195 is shown in Figure 4.
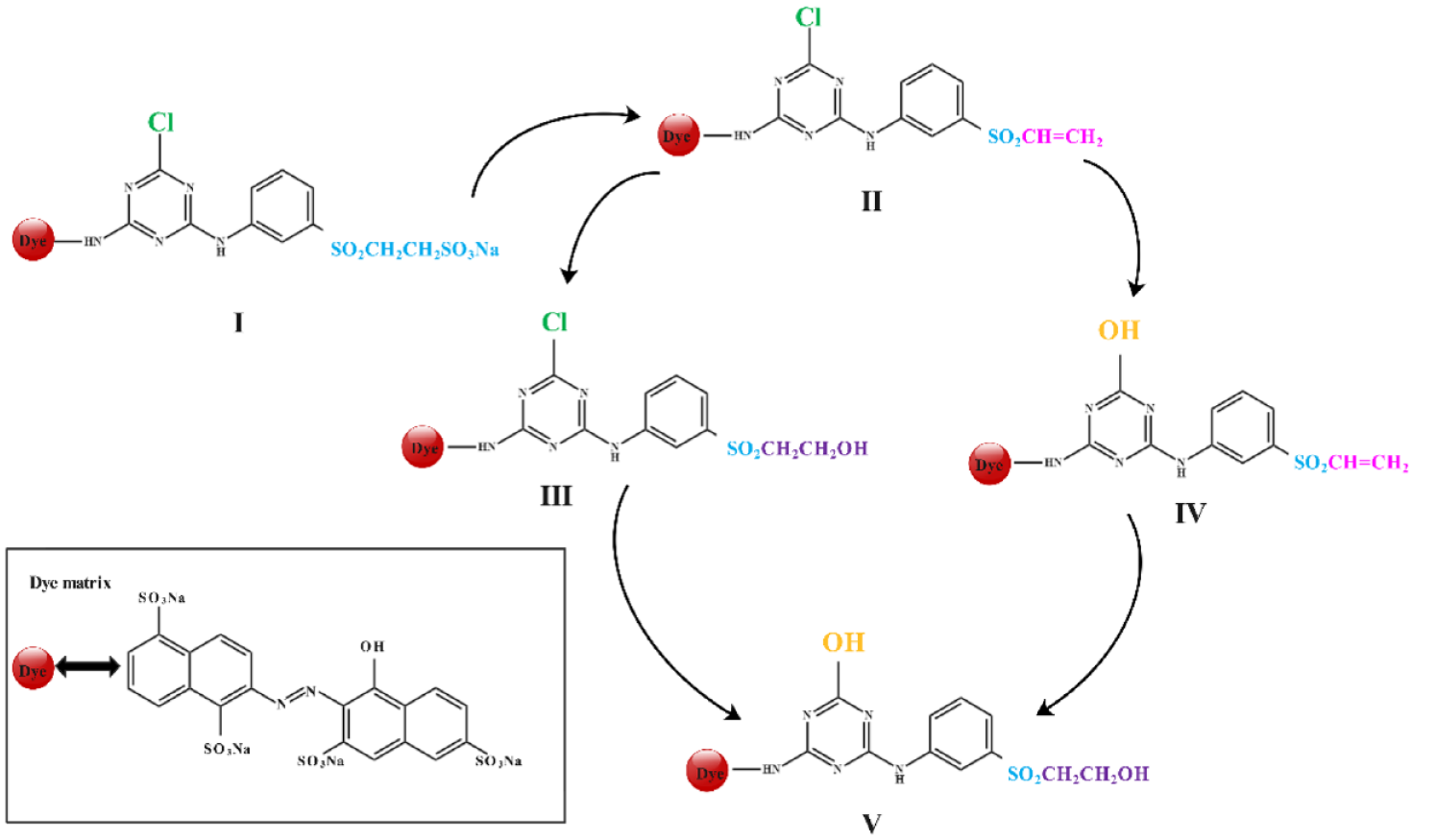
Hydrolysis process of Reactive Red 195.
An Agilent 1260 high-performance liquid chromatograph (Agilent, USA) was used to detect and separate RR 195 hydrolysis products. Figure 5 shows the hydrolysis chromatograms of RR 195 in a pH 10 and 60℃ water bath at different sampling times. Under the conditions of the gradient elution system used in this chromatography, the HPLC showed five significant peaks at tR = 1.12 min, tR = 12.55 min, tR = 15.50 min, tR = 23.41 min, and tR = 32.86 min in order.
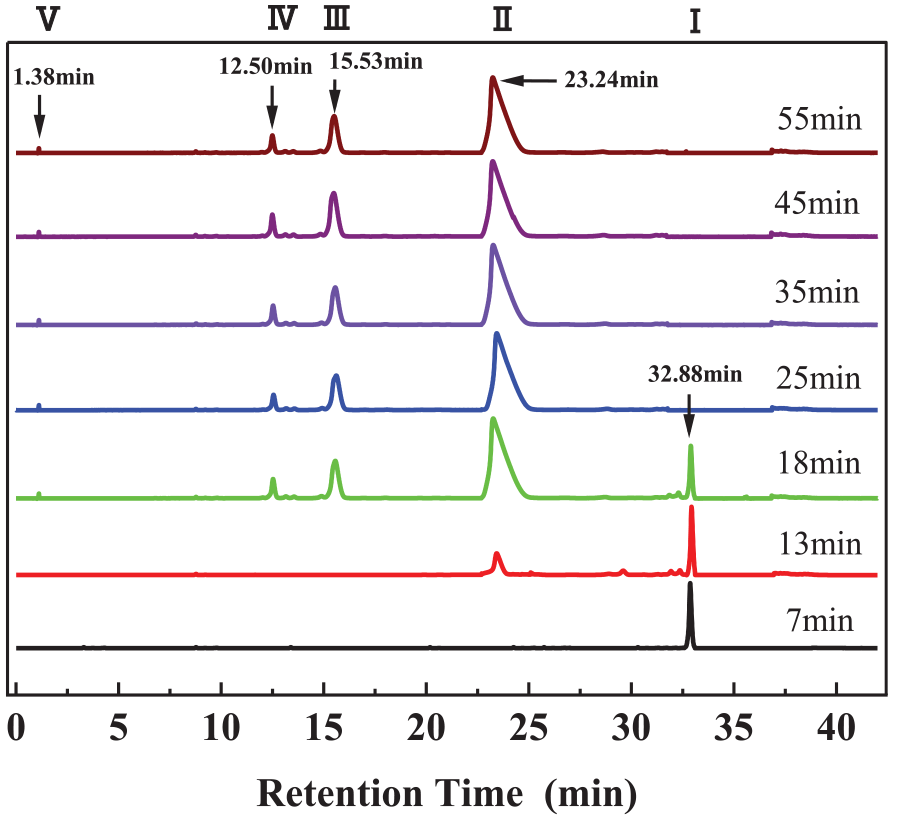
Hydrolysis chromatogram of Reactive Red 195.
Based on the principle that the earliest peaks appear for substances with high polarity. The hydrolysis products corresponding to the peaks at each retention time are shown in Table 3. In a relatively short period, the initial peak of dye Ⅰ gradually decreases in area and height. After the initial peak completely disappeared for 18 min, it was converted into intermediate hydrolysis Ⅱ. As the time continued to increase, Ⅱ generated monochloro-s-triazine hydrolysis products Ⅲ, vinyl sulfone group hydrolysis products Ⅳ, and final hydrolysis product Ⅴ.
The retention time of hydrolyzed RR 195 product structure.

Modeling the kinetics and velocity of hydrolysis of reactive red 195 dye adsorption
Adsorption kinetics and velocity modeling
Due to the interaction between the washing parameters, the dye shed on the dark fabric is free in the water and the water molecules in the light fabric are exchanged with the dye molecules. It adsorbs and diffuses inside the bleached fabric under the effect of the concentration difference inside and outside the fabric pores, leading to the phenomenon of unfixed dye transfer.
As can be seen from Figure 6 and Table 4 kinetic fit parameters, the pseudo-first order kinetic model provides a better fit to the dye sorption variation, with confidence levels greater than 0.95 for all three sets of wash parameters. Therefore, the pseudo-first order kinetic model provides a better and more effective fit to the dye sorption data.

Dye adsorption kinetics curves at different washing parameters: (a) washing temperature, (b) washing liquor ratio, and (c) washing solution pH.
Fitting parameters of pseudo-first-order and pseudo-second-order dye adsorption kinetic model under different washing parameters.

According to the adsorption profiles and the variation of the adsorption rate constants ka, 1 for the three sets of washing parameters under the quasi-primary kinetic model, the adsorption of dye on bleached fabrics can be divided into two main processes before the adsorption equilibrium, which is the fast adsorption phase and the slow adsorption phase.
Under the influence of temperature, the kinetic energy of dye diffusion and fabric expansion increases and the rate of dye adsorption to light colored fabrics increases significantly over a short period. However, the rate of dye entry into the fabric pores is strongly influenced by charge forces and drum mechanical forces. As washing time extends, the equilibrium progressively increases, though the rate of adsorption diminishes. A larger wash bath ratio leads to an increased volume of wash water. This change enhances the desorption process in dark fabrics and significantly accelerates the adsorption rate of desorbed dyes in bleached fabrics. This acceleration occurs over a short period and is attributable to the concentration gradient between the interior and exterior of the fibers, as well as to the forces of electrical charge.
As the pH of the washing solution increases, the rate of adsorption of the dye gradually decreases, along with the equilibrium adsorption amount. This further indicates that the charge force has a repulsive effect on the dye. 29
To further investigate the dynamic process of dye adsorption on bleached fabrics during the washing process, the dye adsorption data was differentiated for different washing times, and the differentiation results are the dye adsorption rates at the corresponding times, as shown in the Figure 7.

Fitting curve of hydrolyzed RR 195 adsorption velocity model under different washing parameters: (a) washing temperature, (b) washing liquor ratio, and (c) washing solution pH.
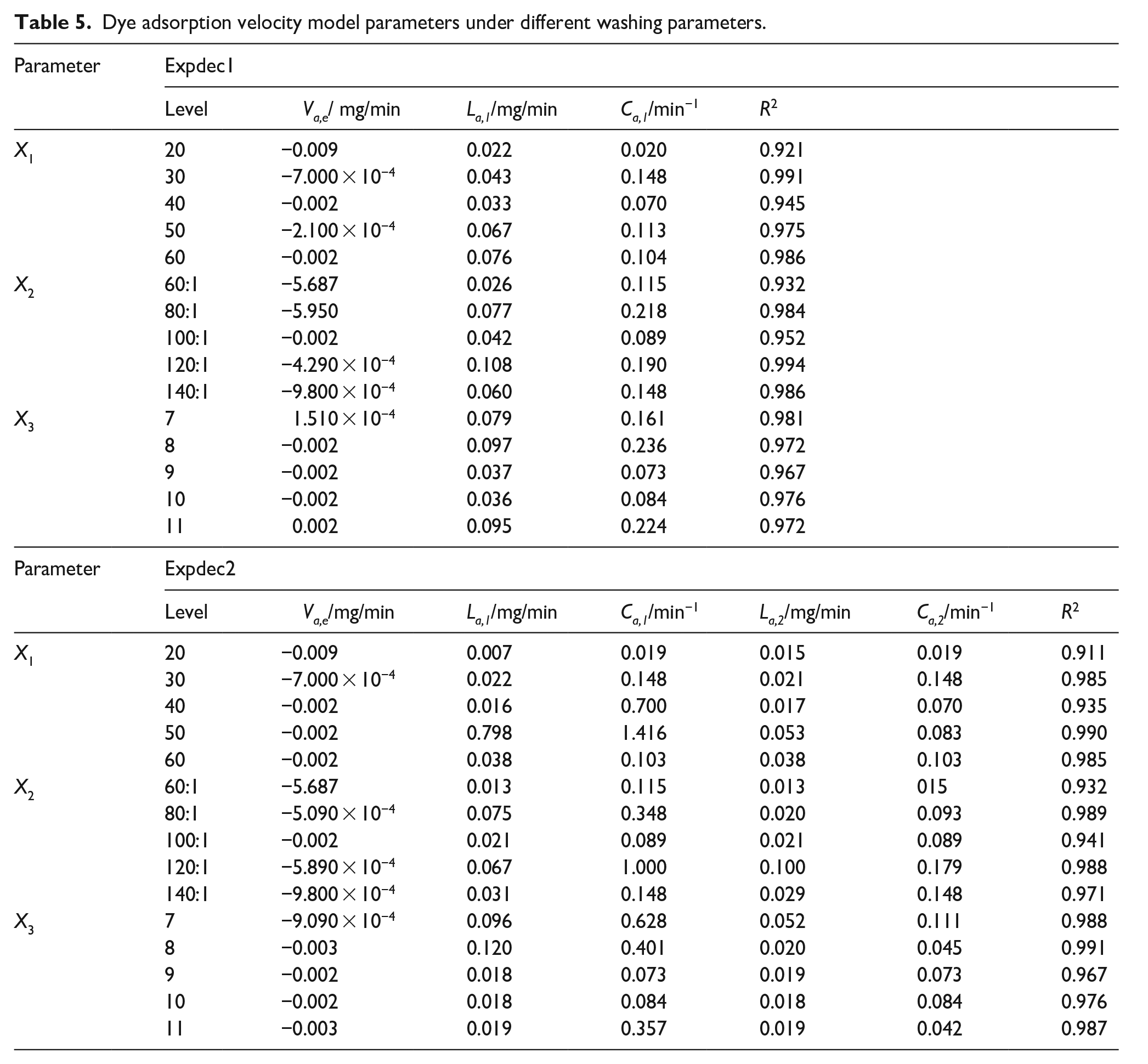
As can be seen from Figure 7 and Table 5, as the washing time continues, the adsorption rate of the light colored fabric to the dye gradually reaches equilibrium and does not change anymore. The fitting of the adsorption rate model suggests that, under three washing parameters, the adsorption of dye on light-colored fabrics before reaching equilibrium undergoes a rapid phase followed by a slower one. Therefore, the curve fitting of the dye desorption and adsorption rate shows that the process of dye desorption from dark fabrics is very similar to the process of adsorption from light fabrics. Moreover, as the washing time increases, both desorption and adsorption behavior gradually converge to equilibrium and no further changes occur.
Dye adsorption velocity model parameters under different washing parameters.
HPLC characterization of washed-off dyes
Reactive dyes form dye-fiber covalent bonds with cellulose fiber, but dye hydrolysis and covalent bond breakage often occur during the dyeing and soaping process of reactive dyes. For example, in a one-bath dyeing system, most dyes are bonded to cotton fabric under high temperature and alkaline dyeing conditions, while some dyes will be hydrolyzed.30,31 Figure 8 shows a diagram of the mechanism of dye-fiber covalent bond breakage formed by a high-temperature color fixing environment. Completely hydrolyzed dyes lose their binding ability and produce hydrolyzed dye adsorbed on the cotton fabric, which results in an “

Bond-breaking mechanism of dye-fiber hydrolysis.
An Agilent 1260 high-performance liquid chromatograph (Agilent, USA) was applied to determine the removal of dyes from the mixed washing system of highly saturated and bleached cotton fabrics for separation. As shown in Figure 9(a) to (d) present the HPLC spectra of the dyes shed from the highly saturated fabrics under different laundering parameters. As the washing time was prolonged, the peaks in the HPLC graphs were consistently positioned, and the retention time (tR) was approximately 1.12 min. Based on the retention time corresponding to the peak position of the hydrolysis product of RR 195, it can be concluded that the dyes shed onto highly saturated fabrics belong to the hydrolysis product V.

Chromatogram of shedding dye at different washing parameters. (a) The washing temperature was 40℃, the washing bath ratio was 100:1, and the washing solution pH was 11. (b) The washing temperature was 60℃, the washing bath ratio was 100:1, and the washing solution pH was 9. (c) The Washing temperature was 60℃, the washing bath ratio was 100:1, and the washing solution pH was 11. (d) The washing temperature was 60℃, the washing bath ratio was 140:1, and the washing solution pH was 9.
The shed types from CFCSs and the adsorbed dyes from BCFs were found to be identical in the gradient elution system, both being hydrolyzed dye V featuring double active groups. The hydrolyzed dye in highly saturated fabrics originates from two sources: firstly, the reactive dye not fully reacting with the cotton fabric, and secondly, the dyes that initially bonded with the cotton fabrics but subsequently underwent bond breakage.
Monte Carlo adsorption simulation
Under the high temperature and alkaline promotion conditions of the dyeing process, most of the dye bonds to the cotton fabric. However, some of the dye hydrolyzes to form unfixed dye. During the washing process, the dyes that are physically adsorbed both on the fabric’s surface and within its pores are desorbed and shed into the washing solution due to the mechanical force exerted by the washing rollers. At the same time, these dyes causes a concentration difference between the light-colored fabric and the solution inside the pores and the washing solution. As a result of this concentration difference, the dye loses its active group and adsorbs to the bleached fabric. As a result, the dye in the washing solution can no longer form a chemical bond with the cellulose fibers and can only physically adsorb to the cotton fabric, as depicted in Figure 10.

Flow chart of the detached and adsorbed hydrolyzed RR 195 on the bleached fabric during mixed laundering.
Monte Carlo simulations were used to learn more about the adsorption behavior of unfixed dye adsorbed on the surface of cellulose Iβ(1 0 0). The adsorption process was simulated using an adsorption locator. Furthermore, it was used to calculate the adsorption energy and determine the most stable adsorption configuration, as shown in Figure 11.

The lowest adsorption energy configuration of the UDS/C system.
The dye cannot counteract the chemical bonding and reaction with the fibers in the cellulose and only counteracts the physical adsorption to the cellulose surface. As can be seen from Figure 11, the molecules studied adsorb almost in a planar orientation relative to the cellulose surface or in a parallel arrangement. The interaction between the benzene ring and cellulose occurs through physical forces. In an aqueous environment, UDS are visibly present at the interface above the cellulose surface, tightly adsorbed together with water molecules. The small distances between the oxygen atoms, the carbon atoms, and cellulose suggest that the main adsorption of all studied molecules is weakly interacting physical adsorption. In conclusion, H2O can facilitate the adsorption process of hydrolysis dyes on the cellulose surface.
As shown in Figure 11, the unfixed dye substance is quickly adsorbed on the cellulose surface. A negatively charged O atom of the vinyl sulfonate group in the UDS is attached to the positively charged H atom of the cellulose hydroxyl group by electrostatic interactions. The sulfonic acid groups and naphthalene residues are tightly adsorbed on the cellulose surface, and a stable system is formed until the UDS is completely adsorbed on the cellulose.
The location of adsorption of adsorbent on the surfaces of cellulose is different. The most effective adsorption position is almost flat on the cellulose Iβ(1 0 0) surface. This is attributed to the nitrogen atom in the carbon heterocyclic ring and the sulfur and oxygen atoms in the sulfate group acting as the adsorption centers. However, the azo, naphthyl, and sulfonate groups in UDS are slightly warped, possibly due to the repulsive effect of hydrogen atoms in the adjacent central benzene ring.
The average adsorption distance between the adsorbent and the adsorbate in an aqueous environment is 3 Å, indicating that there is no covalent bonding and that adsorption between the two relies on H-bonding and van der Waals forces. Moreover, Table 6 shows the adsorption energy of UDS/C. The total energy in the UDS/C system is presented in Table 6, which represents the sum of the adsorbent compounds and the surface energy. Adsorption energy contains deformation and rigid energy. Deformation (Def) energy described release when the adsorbed adsorbate components are relaxed on the cellulose surface. Rigid energy, in kcal mol−1, released (or required) when the unrelaxed UDS components (before the geometry optimization step) are adsorbed on the cellulose Iβ(1 0 0). Def means the energy needed to relax UDS to the cellulose surface. 33
The energy of the most stable configuration for the adsorption of UDS/C (kcal·mol−1).

In aqueous environments, the presence of spontaneous exothermic processes between UDS and cellulose is indicated by negative adsorption energy values.34,35 These values are indeed negative, as shown in the analysis of the molecules in such environments. Observations from the equilibrium configuration (Figure 11) reveal that UDS molecules diffuse laterally along the cellulose surface. The average distance between these adsorbed molecules is approximately 3 Å. This distance suggests the absence of covalent bonding, pointing instead to the predominance of weak van der Waals-type interactions. Furthermore, the calculated energies listed in Table 4 are negative, confirming that the adsorption process is spontaneous.36 –38 This spontaneity is notable when compared to the energy levels in covalent bond formation and other physical adsorption systems.
Molecular dynamics simulation
The mechanism of adsorption between UDS/C in an aqueous environment is elaborated in the present kinetic adsorption simulations.
The UDS is laid flat at 10 Å above the cellulose surface, the energy fluctuations during its simulation in the periodic box by optimizing the overall system energy and geometry are shown in Figure 12 and the total energy distribution is shown in Table 7.

Total energy of fluctuations during molecular dynamic simulation of UDS/C at 2 ns.
The distribution of total energy in the process of UDS/C (kcal·mol−1).

As shown in Figure 12 the non-bond energy is the lowest in aqueous environments and plays an important role in adsorption. In particular, the van der Waals energy and the total valence energy are both positive, while the hydrogen bond energy is negative., as shown in Table 7. Accordingly, hydrogen bonding is essential in the attraction and interaction energy between the molecules of UDS and cellulose. Furthermore, the super negative electrostatic energy implies that electrostatic interactions played an major role during the adsorption process. Therefore, it is further demonstrated that the predominant relationship between UDS and cellulose is physical adsorption.
Figure 13 showed the initial configuration and final configurations of the UDS/C system with different simulation time at 298k. As we have seen, there is a tendency for the adsorbed dye molecules to become flatter, almost parallel to the cellulose surface.

The initial configurations and final configurations of the UDS/C system with different simulation time at 298k.
Molecular dynamic simulations were performed at room temperature to better understand the adsorption process and gain insight into the specific interactions between the UDS/C to calculate the interaction energy by equations (7) and (8), as shown in Table 8.
The surface interaction energy of UDS/C (kcal·mol−1).
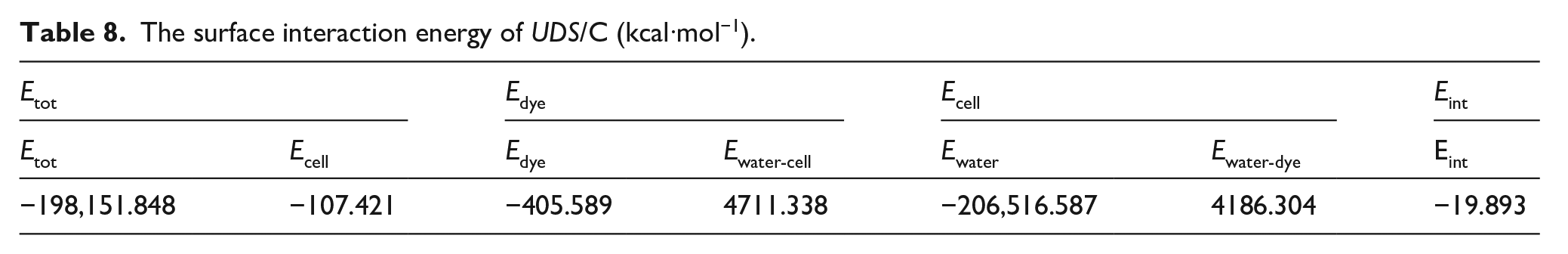
Significant interactions between UDS and the cellulose surface were observed in the aqueous environment as shown in Table 8. These findings corroborate previous experimental results and theoretical calculations. These earlier studies elucidate the interactions between the atoms in cellulose and the functional groups of the unfixed dye, further detailed in Table 9.
UDS in close contact with cellulose at 3 Å and below the structural groups.

The calculations indicate that the close contacts within the UDS/C compound system primarily involve the aliphatic groups of cellulose and the aromatic groups of UDS. Notably, the aromatic group in UDS interacts with both sides of the glucose ring in cellulose. This interaction suggests the presence of van der Waals forces, in addition to electrostatic forces, contributing to the adsorption process between cellulose and UDS. The interaction between the hydrogen atoms in cellulose and the oxygen and nitrogen atoms in the UDS indicates the role of the hydrogen bond in the adsorption process. It is worth pointing out that hydroxyl groups in cellulose form most hydrogen bonds with sulfonate, amino and azo groups in UDS. During the adsorption process, in addition to these classical hydrogen bonds, three other types of bonds were identified: (a) N-H. . .O, with lengths of 2.72 Å; (b) C-H. . .O, with lengths of 2.21 Å; and (c) C-H. . .N, with lengths of 2.88 and 2.60 Å.
Conclusion
In this study, the adsorption mechanism of the dye transfer phenomenon during the co-washing of CFSCs and BCFs using RR 195 dye was examined. Mechanisms underlying fabric fading and dye transfer were deduced via a mixed-washing simulator, followed by the analysis of the resultant dye effluent. A molecular dynamics model was developed to delineate the interactions between the dye hydrolysis products and the cellulose Iβ(1 0 0) surface. This model elucidates the adsorption mechanism at the molecular scale, offering a theoretical framework for the observed phenomenon of mixed washing color stringing. The salient conclusions drawn from the study are:
RP-HPLC analyses of the dye waste liquid revealed that the end product of RR195 hydrolysis was congruent with the dye observed on the bleached fabric.
According to the unfixed dye adsorption and rate model, dye adsorption on bleached fabrics achieves equilibrium within a specified timeframe, after which no further alterations in adsorption patterns are observed. The most rapid adsorption rate was noted at a temperature of 60°C, a bath ratio of 120:1, and a pH of 7.
Molecular dynamics simulations indicated that the interactions between the UDS and the cellulose Iβ(1 0 0) surface are dominated by robust hydrogen bonds, van der Waals forces, and electrostatic interactions. Significant adsorption sites were identified between the elements N, S, and O in the UDS and the elements C and O in cellulose.
Footnotes
Acknowledgements
All authors extend their appreciation to the National Natural Science Foundation of Jiangsu Province for funding this research work through project number BK20200608.
Author’s contributions
CYZ, XZ, and ZBH are responsible for the conception of the whole study, modeling, design and implementation of the experiment, the collection and analysis of data, and the drafting of the first draft. STZ, WT, and ZLW is responsible for the planning of the paper. SZL and BZ is responsible for coordinating the research tasks and data analysis. JLL and YZF are responsible for data analysis and the revision of the first draft. All authors read and approved the final manuscript.
Availability of data and materials
Data will be made available on request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the National Natural Science Foundation of Jiangsu Province through project number BK20200608.


