Abstract
The dyeing process in the textile industry is not only highly water intensive, but also the quality of water plays a substantial role in determining the final shade of the product. Hardness is one of the crucial factors of water quality influencing the final shade of dyed products. The influence of hard water on solubility of reactive dyes and colorimetric properties of reactive-dyed cotton were studied. It was concluded that the solubility of reactive dyes decreased with increased water hardness. Also, hard water significantly affected the colorimetric properties (L*, a*, b*, C*, h°, and DE values) of reactive-dyed cotton fabric. Lightness value increase was accompanied by the decrease in chroma values for all three primary colors studied in this work.
Introduction
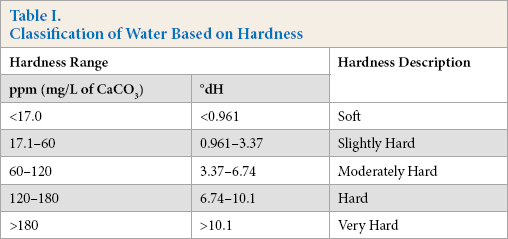
In textile wet processing, water quality plays a substantial role in determining the final shade of the products, its consistency, and its reproducibility. Water hardness is a particularly crucial factor in shade matching as water contributes significant variability to the dyeing process.1-2 In general, ground water is a solution of ionic compounds (calcium and magnesium) that may significantly reduce the performance of dyes, detergents, soaps, and laundry additives. Hardness can also be caused by several other dissolved metals, which form divalent or multivalent cations, including barium, aluminum, strontium, iron, zinc, and manganese. Generally, potassium and sodium (monovalent) ions do not cause hardness in water. 3 Hardness can be defined as the total concentration of mineral content and is expressed as German Degree Hardness (°dH) in this study. According to the Water Quality Association (WQA),4-5 water can be classified into five categories depending on its hardness (Table I).
Classification of Water Based on Hardness
Reactive dyes constitute the largest group of synthetic dyes due to their simple dyeing method, brilliancy in shades, and excellent washfastness. 6 However, despite obvious advantages, water hardness can cause dye precipitation 7 and further promotes dye aggregation. 8 As a result of this, loss in color depth and faulty dyeings (e.g., color specks) are produced. 1 Furthermore, interaction with alkali and heating during dyeing and soaping promotes calcium and magnesium ion precipitation on fabrics as whitish carbonates and hydroxide particles. 9 These precipitates are sticky, adhere readily to fabrics, and can result in unlevel dyeing, white patches, and poor shade matching. 10 Hence, it is necessary to study and assess the influence of hard water on reactive dyeing of cotton fabric.
The textile industry is a huge water consumer. 11 In a typical dyeing and finishing mill, on average for every ton of fabric processed, ∼150 m3 of water is consumed and the generally accepted range of hardness is 0-25 mg/L of CaCO3 (0-1.4 °dH). 12 The main sources of water for textile industries in Pakistan are ground water or municipal water, which both contain large amounts of mineral contents. Therefore, textile industries in Pakistan are facing an acute problem of water hardness resulting in faulty dyeings. Although the effect of water hardness has been widely explored and evaluated in several studies,2,13-19 the influence of hard water on the solubility of reactive dyes was rarely discussed. 20 Therefore, in this research, we investigated the influence of hard water on solubility of reactive dyes, along with the colorimetric properties (L*, a*, b*, C*, h°, and ΔE values) of cotton fabric dyed with red, yellow, and blue (primary colors) reactive dyes.
Experimental
Materials
Commercially scoured, bleached, and mercerized 100% cotton woven fabric (plain weave, 130 g/m2) was used in this study. Anhydrous calcium chloride (CaCl2) and magnesium sulfate heptahydrate (MgSO4·7H2O) were purchased from Merck and used as received. Drimaren Yellow HF-CD, Drimaren Red HF-CD, Drimaren Royal Blue HF-CD (cyanuric bromide reactive group), Solidokol NM, Revatol S, sodium bicarbonate, sodium chloride, sodium hydroxide (36 °Be), and soda ash were provided by Archroma Pakistan.
Methods
Preparation of Stock Solution
Stock solution A was prepared by dissolving 39 g of anhydrous calcium chloride in enough distilled water and made up to a volume of 1 L. Likewise stock solution B was prepared by dissolving 43.93 g of magnesium sulfate heptahydrate in enough distilled water and made up to a volume of 1L.
Preparation of Standard Hard Water
Solution A (100 g), solution B (50 g), and 1.24 g of sodium bicarbonate were dissolved in enough distilled water and made up to a volume of 10 L. This provided water with a hardness of 15 °dH. Hardness was checked by titrating it against standard 0.01M EDTA solution and adjusted as required. Hard water of varying hardness (10 to 50 °dH at 10° intervals) were prepared and used in this study.
Solubility of Reactive Dyes
The solubility of reactive dyes in distilled water (control sample) and prepared hard water was determined according to the method previously described by Han and Jung. 20 The solubility of 10 g of reactive dyes in 100 mL water was determined at 30 °C. The mixture was continuously stirred for 2 h and filtered after 24 h. The separated solid dyes or dye residue left as crystals or precipitates were weighed after drying at 40 °C overnight in a drying oven. Also, the filter papers were analyzed for traces of reactive dyes, whitish carbonates, and hydroxide particles.
Dyeing
Dyeings were performed with prepared hard water using Drimaren HF-CD dyes (5 and 15 g/L) and Solidokol NM (10 g/L). The wet pickup was 85% and all fabrics were dried at 120 °C. The pre-padded samples were padded again using sodium chloride (250 g/L), sodium hydroxide (36 °Be, 20 mL/L), soda ash (20 g/L), and Revatol S (10 ml/L). The fabrics were steamed at 102 °C for 60 s. They were rinsed with prepared cold hard water, soaped with 2 g/L Ladipur RSK at boil for 10 min, and then finally rinsed with cold hard water until there was no further color bleed, and then dried under standard laboratory conditions of 20 ± 2 °C and 65 ± 2% relative humidity (RH). Control samples were dyed in distilled water.
Color Measurements of Dyed Fabric Samples
Changes in colorimetric properties brought about by dyeing in hard water were measured using a Datacolor Model D650 spectrophotometer (D65 illuminant, 10° observer). CIE L*a*b* attributes were measured, where L* is the lightness value, a* indicates the degree of redness and greenness, b* indicates the degree of yellowness and blueness, C* indicates saturation, and h° indicates the hue angle of the dyed fabric samples. Total color difference (ΔE) was calculated using the DECMC(l:c) equation.
Results and Discussion
Influence of Hard Water on Solubility of Reactive Dyes
Fig. 1 shows the amount of dye residue left after filtration of dye solutions prepared in distilled (0 °dH) as well as hard water (10 to 50 °dH varied at 10° intervals) at 30 °C. The amount of residue was greatly affected by the degree of hardness of the water. As the water hardness increased, the amount of residue also increased, indicating a decrease in solubility of the reactive dyes. This could be attributed to the crystallization or precipitation of reactive dyes in hard water. 2 This decrease in solubility of dyes in hard water indicates that reactive dyes possess good affinity toward precipitates of calcium and magnesium ions, and these precipitates reduced the amount of dye transferred to the fabric surface and hence loss in color depth.2,20 From Fig. 1, the effect of water hardness on the solubility of the red and yellow reactive dyes tested was more significant as compared to the blue reactive dye tested. Visual assessment of the dried filter papers showed dark patches of precipitated reactive dyes along with whitish carbonates and hydroxide particles.
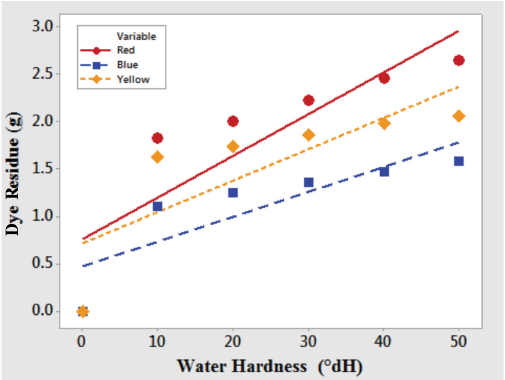
Influence of hard water on solubility of reactive dyes at 30 °C.
Influence of Hard Water on Colorimetric Properties
Tables II–IV show the changes in L*, a*, b*, C*, h°, and ΔE values induced by water hardness on cotton fabrics dyed with 0.5% and 1.5% of the red, blue, and yellow reactive dyes.
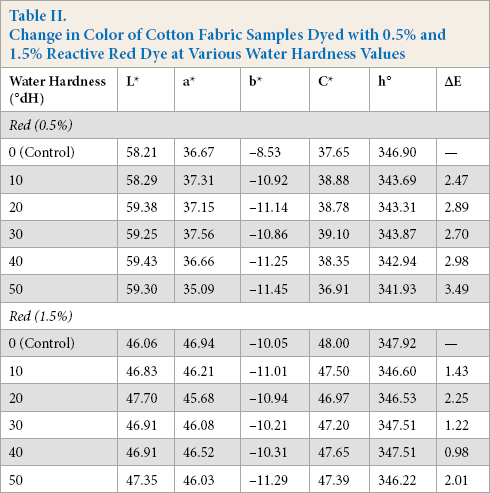
Change in Color of Cotton Fabric Samples Dyed with 0.5% and 1.5% Reactive Red Dye at Various Water Hardness Values
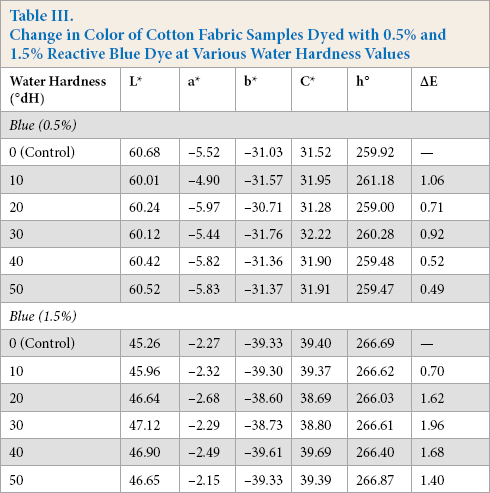
Change in Color of Cotton Fabric Samples Dyed with 0.5% and 1.5% Reactive Blue Dye at Various Water Hardness Values
Change in Color of Cotton Fabric Samples Dyed with 0.5% and 1.5% Reactive Yellow Dye at Various Water Hardness Values
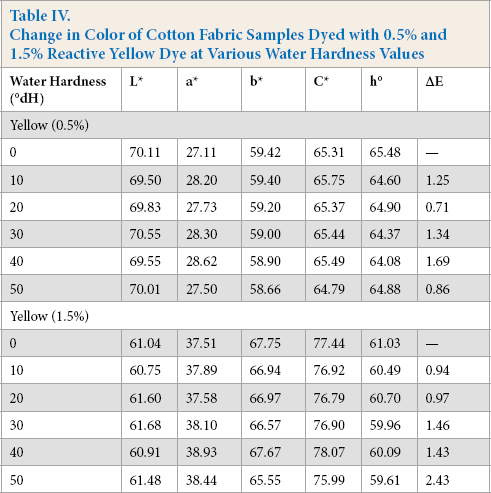
From the colorimetric values listed in Table II, the red dyes were most sensitive to change in water hardness in both pale (0.5%) and medium depths of shade (1.5%). From Fig. 2a, ΔE values increased with the increased level of water hardness, however, this effect was more pronounced in the pale shade. The resulting high ΔE values of the samples were not within the tolerance limit (ΔE ≤ 1) 21 and were unacceptable. The L* and C* values changed significantly, resulting in higher ΔE values. From Fig. 2b, for both depths of shades, the L* value increased with increased levels of water hardness, and the samples appeared lighter. This showed that dye exhaustion and fixation was reduced with increased water hardness. However, the change in C* for pale and medium shades did not follow the same trend. From Fig. 2c, the medium shade appeared duller as saturation decreased with increased water hardness. Table II also shows that the hue (h°) values for both depths of shade differed from the corresponding values with control samples and decreased with increasing hardness, although this effect was not significant.
Influence of water hardness on (a) total color difference, (b) lightness L*, and (c) chroma C* of cotton fabric after dyeing with Reactive Red dye in pale (0.5%) and medium (1.5%) shades.
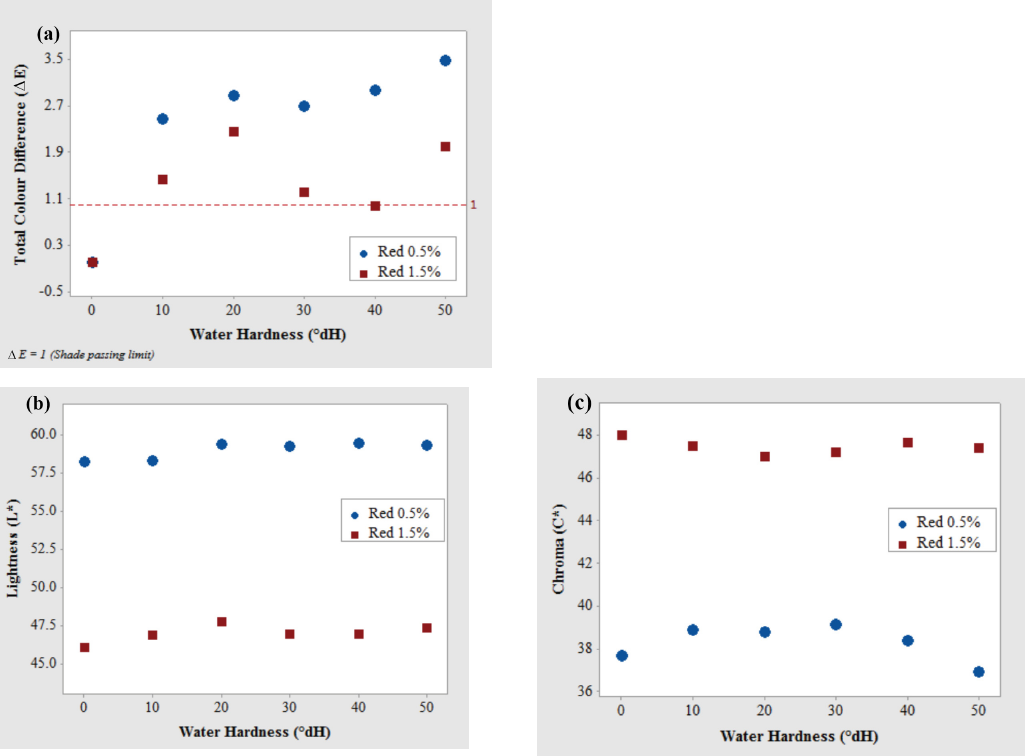
Table III and Fig. 3 show the influence of varying water hardness values on the ΔE, L*, C*, and h° values of cotton fabric dyed with the reactive blue dye in pale (0.5%) and medium (1.5%) shades. From Fig. 3a-c, for the pale shade, no significant change in L* and C* were observed and the samples were well within the ΔE tolerance limit. The pale blue color samples appeared a little darker and brighter when compared to the control sample. However, for the darker shades of the reactive blue dyed fabric, there was a significant difference in L* and C* values as water hardness increased. These samples become lighter and less saturated with increased water hardness and were unacceptable. However, the hue of these dyeings were not affected by increased hardness (Table III).
Influence of water hardness on (a) total color difference, (b) lightness L*, and (c) chroma C* of cotton fabric after dyeing with Reactive Blue dye in pale (0.5%) and medium (1.5%) shades.
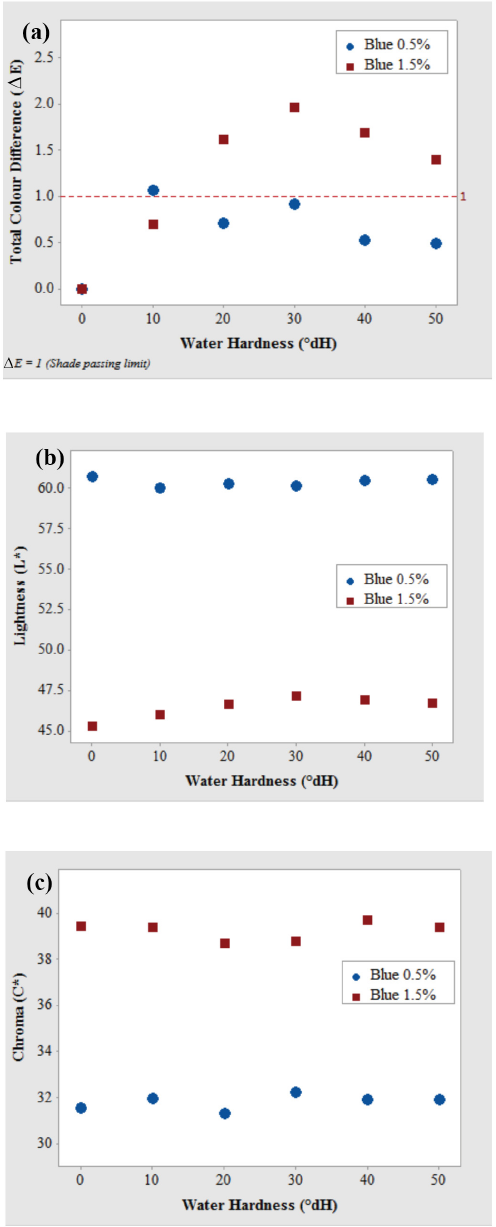
From Table IV and Figs. 4b and c, the reactive yellow dyeing was also affected by water hardness and there was a decrease in L* values, accompanied by a slight increase in saturation for the pale yellow color dyeing. However, for the medium depth shades, the samples appeared lighter and less saturated. These changes were significant at higher hardness values, which was also evident from Fig. 4a. Table IV also shows that h° value of the dyeings was slightly affected by increasing water hardness for both pale and medium shades of reactive yellow. The dyeings became redder as the hardness increased.
Influence of water hardness on (a) total color difference, (b) lightness L*, and (c) chroma C* of cotton fabric after dyeing with Reactive Yellow dye in pale (0.5%) and medium (1.5%) shades.
Changes in L*, a*, b*, C*, h°, and ΔE values of the reactive dyed samples tested could be attributed to the precipitation and aggregation of reactive dyes in the presence of high amounts of calcium and magnesium ions in water, leading to dye loss and faulty dyeings. 2
Conclusion
Water hardness is one of the prime concerns for shade matching and shade passing for reactive dyed fabrics. Under the dyeing conditions is this study, there were increased lightness values and decreased chroma values using water containing large amounts of calcium and magnesium ions. Furthermore, the solubility of reactive dyes decreased with increased water hardness. Dye exhaustion decreased with increased water hardness as the dyes preferred to form aggregates and excessive aggregation reduced the transfer of dye from dyebath to fabric surface. This work did not involve tertiary or binary colors, where the effect of water hardness on hue angle changes can be significant.
Footnotes
Acknowledgment
This work was part of an independent study project submitted by Erum Bashir in partial fulfillment of the requirements for the MS degree in Industrial Chemistry, NED University of Engineering & Technology. The authors wish to express their gratitude towards NED University of Engineering & Technology for support. The authors also wish to thank Archroma Pakistan for allowing us to carry out the necessary experimental work at their premises.
