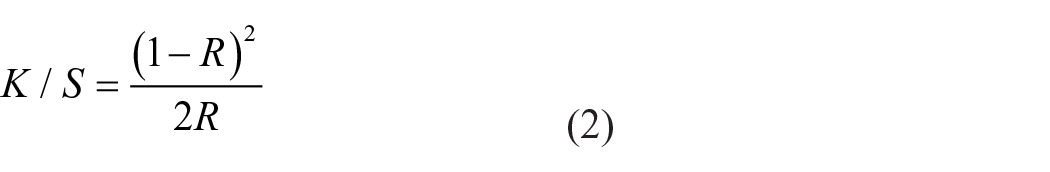Abstract
Dyeing of cotton, viscose, wool, and polyester/viscose blended fabrics has been carried out with the aqueous extract of the roots of Alkanna orientalis plant. Fastness properties and color strength values were investigated in terms of users. Phenolic compounds and cytotoxic activities of ethanol (EtOH) and water extracts were determined. It was found that cotton fabric dyed with the aqueous extract of the roots of A. orientalis plant in the presence of alum mordant gave better depth of shade (K/S). The toxicity studies reveal that the EtOH and the water extracts of the root of A. orientalis exhibited 0% cytotoxicity while the standard cancer drug 5-fluorouracil exhibited 6% cytotoxicity. The overall results of this study show that the roots of A. orientalis can be used in the textile industry, especially for dyeing cotton fabrics.
Introduction
Recently, an increase in the international awareness of environment, ecology, and health has forced people to reduce toxic effluents and stop the use of dangerous dyes. Therefore, the present trend throughout the world is toward the use of natural products over their synthetic alternatives despite their known limitations.1–4 Natural products offer more advantages such as renewable sources, minimal health hazards, mild reaction conditions, no disposal problems, and harmonization with nature. In addition, they are non-toxic, non-allergic to skin, and non-carcinogenic.5,6 Natural dyes are plant-derived phytochemicals obtained from plants and animals. Despite the disadvantages of synthetic dyes such as allergic, toxic, not ecofriendly, and non-biodegradable, the consumption of synthetic dyes is extremely high in the world. 7 Plant-derived natural dyes are generally used especially in the textile industry 8 for their bright shades and their non-toxic properties, as well. 9 Natural dyes have various biological activities—such as UV protective and antimicrobial activities—which improve the utility of natural dyes in textiles.10,11 The low substantivity problem of natural dyes can be overcome by using metal mordants which serve a bridge between the dye molecule and the textile substrate. 11 Generally, metal mordants are used in the dyeing processes in order to enhance the color strength of the dyed material and improve the staining ability of the dye, or vice versa. 12 Mordants provide an affinity between the fiber and the dyestuff of the plant by forming a coordination complex via covalent bonding. 13
Some of the widely used metallic mordants such as potassium dichromate and copper sulfate have been admitted to have toxic properties. However, alum (Al2(SO4)3) mordant was considered as the safest mordant for dyeing textile fibers with natural dyes. 14 It has the advantages of easy availability, low price, no effect on the hue, and being safe.15,16 The genus Alkanna that belongs to Boraginaceae family is useful for the preparation of dyestuffs. Thirty-four species are found in Turkey, and 76% of them are endemic to Turkey.17,18 The majority of naphthalene derivatives generally found in the roots of Boraginaceae family are not only responsible for the red coloration of the roots but also for the therapeutic behavior of the plant. 19 Flavonoids, pyrrolizidine alkaloids, 20 and γ-linolenic acid 21 have been reported from Alkanna species. Dyeing wool and cotton fibers with Alkanna tinctoria Tausch was studied, and high dye absorption and good fastness values were obtained in the presence of mordant salt for cotton fabrics. 22
It is reported that synthetic colorants might cause dermatitis, skin complaints, and cancer. To get rid of this problem, it is important to discover non-toxic natural dye sources, especially for babies’ and children‘s clothing.23,24 So, this study focused on the determination of the cytotoxic activity of A. orientalis (Figure 1) roots along with its dyeing performance on cotton, viscose, wool, and polyester–viscose blended fabrics. In addition, the phenolic constituents of the roots were determined in order to find the reasons for the different colors. To the best of our knowledge, this is the first comprehensive report on the dyeing potential of A. orientalis extract on different types of fabrics along with its cytotoxic activity and phenolic compounds.

(a) Alkanna orientalis Boiss and (b) Alkanna orientalis water extract.
Materials and Methods
Materials
Plain-weave cotton of 240 g/m2 (warp = 42, weft = 20), viscose of 121 g/m2 (warp = 44, weft = 34), wool fabric of 125 g/m2 (warp = 22, weft = 12), and polyester–viscose blended (60/40) fabrics of 120 g/m2 (warp = 160 polyester, weft = 40 viscose) were procured from Has Ozgen Textile Company (Tokat, Turkey). Analytical-grade aluminum potassium sulfate dodecahydrate (AlK(SO4)2 12H2O) was used for mordanting processes. Dulbecco’s modified Eagle’s medium (DMEM, Sigma-Aldrich, Germany), fetal bovine serum (Sigma-Aldrich, Germany), and Pen-Strep solution (Sigma-Aldrich, Germany) were purchased.
Instruments and Methods
A Termal HT 610 NHT model dyeing machine was used for mordanting and dyeing processes. A Premier Colorscan SS 6200A spectrophotometer was employed for the measurement of color strength (K/S) and CIELAB (L*, a*, b*, C*) values using illuminant D65 and 10° standard observer. The washing of the dyed fabrics was evaluated in a Rotawash washing fastness device according to ISO 15 105-C10 (2006). 25 The rubbing fastness of the dyed samples was determined using the crock fastness device 255 model Crock-meter according to ISO 105-X12 (1993). 26 The light fastness rating was determined according to the color changes of the tested fabrics based on ISO 105-B02 (1994) 27 using a Xenotest 150S+ light fastness test instrument (Atlas, Germany). The phenolic content of the plant extract was determined using Agilent Technologies 1260 Infinity HPLC System coupled with 6210 Time of Flight (TOF) LC/MS detector and Agilent Poroshell 120 EC-C18 (2.7 µm, 4.0 × 50 mm) column.
Extraction Procedures
Dried and powdered roots were cut into halves. 10 g of the plant material was extracted with 200 mL of water and ethanol (EtOH) with a Soxhlet apparatus. The solvents were evaporated under reduced pressure to give water extract (yield = 12.30%) and EtOH extract (yield = 4.54%), respectively. The extracts were used for the determination of phenolic compounds and cytotoxic activity. For the preparation of dye solution, the roots (42 g) were extracted with distilled water (1500 mL) using a Soxhlet apparatus at its boiling point, until colorless.
Determination of Phenolic Compounds
Mobile phases A and B were ultra-pure water solution with 0.1% formic acid and acetonitrile, respectively. The following conditions were undertaken for analyses: column temperature = 35°C, flow rate = 0.4 mL/min, and injection volume = 5 µL. The solvent program was adjusted as follows: 0 min 10% B; 0–1 min 10% B; 1–10 min 80% B; 10–19 min 80% B; 19–19.10 min 10% B; 19.10–29.1 min 10% B. The ionization mode of the MS-TOF instrument was negative with a gas temperature of 325°C, a gas flow of 11.0 L min−1, and nebulizer 45 (psi).
Toxicity Studies
Cell Culture
C6 (rat brain tumor) cells were amplified in Dulbecco’s modified Eagle’s medium (DMEM), supplemented with 10% (v/v) fetal bovine serum (FBS) and penicillin–streptomycin (Pen-Strep) solution at 37°C in a 5% CO2 humidified atmosphere. DMEM, FBS, and Pen-Strep solution were purchased from Sigma-Aldrich, Germany. All tests were performed in twice and triplicated using C6 cells. For the stock solution of the samples, 5-florouracil (5-FU) was dissolved in sterile dimethyl sulfoxide and diluted with DMEM (1:20). The amount of dimethyl sulfoxide was below 1% in all experiments.
Lactate Dehydrogenase (LDH) Leakage Assay
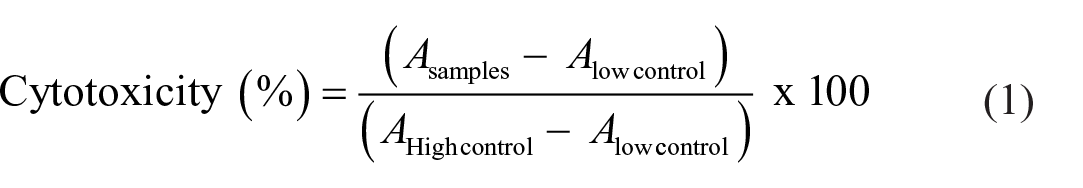
The assay was carried out using an LDH cytotoxicity detection kit by Roche Diagnostics GmbH, Mannheim, Germany, according to the protocol in the user’s manual. Antiproliferative activity tests were performed at the highest dose (100 µg/mL) and cytotoxicity (%) was determined against the C6 cell line. 5-FU was used as the positive control. Samples and 5-FU were incubated with 100 µg/mL of the C6 cell suspension having 5 × 103 cells/mL in a 96-well plate at 37°C overnight in a 5% CO2 atmosphere. 5-FU and the samples were tested in triplicate, and the cytotoxicity (%) of the samples was calculated from the following formula:
where Asamples is the absorbance of LDH released from the cells treated with samples, Alow control is the absorbance of LDH released from the untreated normal cells (=spontaneous LDH release), and Ahigh control is the absorbance of maximum releasable LDH in the cells (=maximum LDH release).
Dyeing Experiments
The meta-mordanting process was used in dyeing the fabrics for its time-saving and economic properties. 28 Dyeing experiments were performed in a Termal HT 610 NHT model dyeing machine, keeping the material-to-liquor (M:L) ratio as 1:30. Dyeing of wool and polyester–viscose blended fabrics was carried out at pH 4, while cotton and viscose fabrics were dyed at pH 8. The fabrics were put into the dye solution and 0.1 M of mordant (in solid state which is equivalent to 0.1 M mordant solution) was added. Alum was chosen as mordanting agent due to its eco-friendly property and low toxicity. The dyeing process was started at 25°C and the temperature was then raised to 75°C (2°C/min) for wool, viscose, and cotton fabrics. Dyeing processes were performed for 1 h. Dyeing of polyester–viscose blended fabrics was carried out at 75°C for 60 min and at 130°C for 30 min, respectively 29 (75°C for viscose, 130°C for polyester). After completion, the material was rinsed with distilled water and dried.
Color Measurement and Fastness Tests
The depth of shade of the dyed samples expressed as K/S values was calculated from the reflectance value using the well-known Kubelka–Munk equation
where R is the reflectance of the dyed fabric, S is scattering, and K is absorption coefficient. A higher K/S value indicates better dye receptivity of the substrates. Lightness–darkness values of dyed fabrics symbolized with “L*” and these values varied between 100 and 0, where L* = 100 shows lightness, L* = 0 shows darkness. a* values represent the color change between green (–a*) and red (+a*) while b* values represent between yellow (+b*) and blue (–b*).
Results and Discussion
Phenolic Compounds
Different colors are obtained in the presence of metal mordant. Phenolic compounds of the plant form dye–mordant complexes which are responsible for the different shades of the dyed fabric. The colors obtained from dyeing of fibers with natural dye plants under different conditions are due to the fact that when different phenolic compounds of the dye plant are combined with the mordant to form dye–mordant complexes, different shades are attained. 30 Therefore, EtOH and water extracts of the roots of A. orientalis were subjected to high-performance liquid chromatography–time-of-flight (HPLC-TOF) analysis in order to determine the phenolic compounds of the plant in terms of qualitative and quantitative studies. The phenolic compounds identified by HPLC-TOF are presented in Table 1.
Main phenolic compounds of A. orientalis (mg phenolic compound/100 g dried plant).
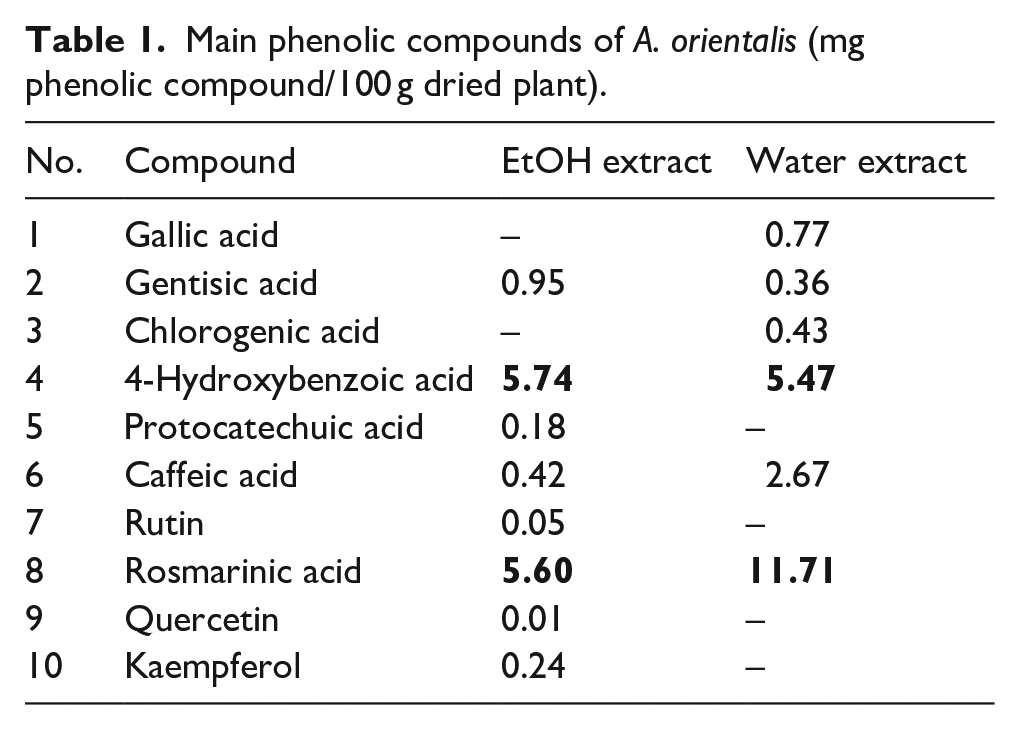
Rosmarinic acid and 4-hydroxybenzoic acid were found to be the main phenolics in the roots of A. orientalis. Rosmarinic acid content was 5.60 and 11.71 mg/100 g dried weight (dw) and 4-hydroxybenzoic acid was 5.74 and 5.47 mg/100 g dw for EtOH and water extract, respectively. The lowest value was obtained for quercetin (0.01 mg/100 g dw) for EtOH extract of A. orientalis roots.
Toxicity Assays
Toxicity can be described as the ability of a matter to produce damage in living organism when inhaled or absorbed by the skin.31,32 The prevention of the presence of skin and eye irritatation can be achieved by toxicity tests. It is possible to predict the long-term effects of the natural dyes—whether carcinogenic and mutagenic—before the usage. 33
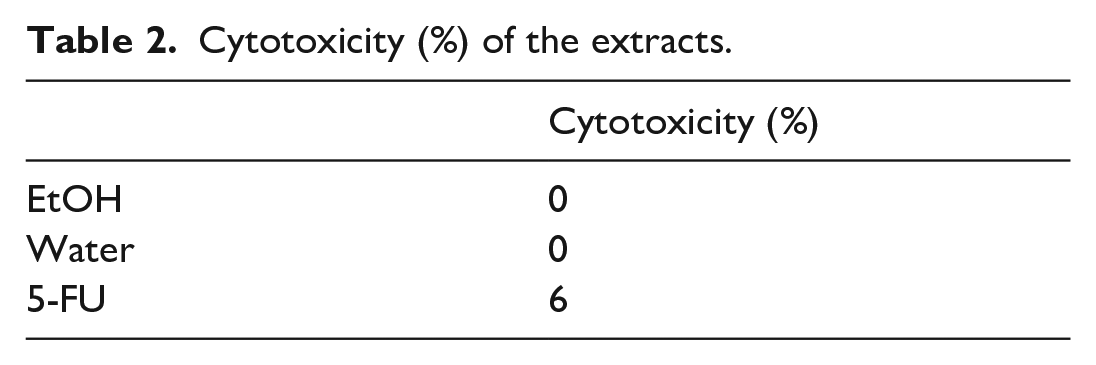
C6 cells were used to determine cytotoxicity (%). The toxicities of ethanol and water extracts of A. orientalis roots were compared with 5-fluorouracil (5-FU). 5-FU is a remarkable drug that has been on the market for 41 years and has become the mainstay of chemotherapy. 5-FU is one of the few drugs in clinical medicine whose toxicity varies depending on different doses and schedules. 34 For this purpose, 5-FU was used as the standard compound. The cytotoxicity test results are given in Table 2. As a result of the tests, at 100 µg/mL concentration, 5-FU showed 6% toxicity, while ethanol and water extracts of A. orientalis roots were not toxic when compared with 5-FU. The difference in toxicity between EtOH, water, and 5-FU mainly depends on the chemical content of the A. orientalis extracts. Water is a non-toxic inorganic solvent. Ethanol and water are preferred solvents in many studies. Besides, these polar solvents contain phenolic compounds which exhibit several biological activities—including antioxidant, antiproliferative, and antimicrobial activities. Briefly, the EtOH and the water extracts of A. orientalis roots were found to be non-toxic. According to these results, water and ethanol extracts of A. orientalis roots can be used safely in natural dyeing.
Cytotoxicity (%) of the extracts.
Dyeing Performance and Dyeing Behavior of the Fibers
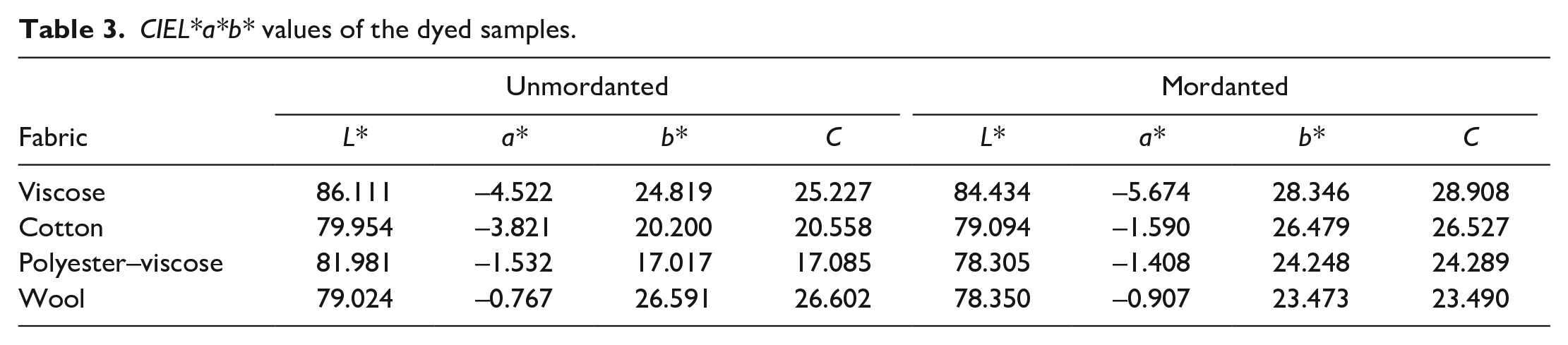
The effect of meta-mordanting on the CIEL*a*b* values of the dyed fabrics is given in Table 3. Higher values of L* led to lighter shades. In addition, positive a* and positive b* indicates red and yellow, respectively. Negative a* values indicate green while negative b* values represent blue. Cream, light pink and light green color and shades were obtained in dyeing roots of A. orientalis water extract. The pictures of the dyed fabrics—before and after washing fastness tests—are given in Figure 2. The tones of the colors became darker in the presence of alum mordant for all fabrics. The highest change was obtained for polyester–viscose fabric, that is, the L* value of polyester–viscose fabric unmordanted dyed fabric decreased from L* = 81.981 to L* = 78.305 after alum mordant. The flavonoid pigments of the dye are responsible for the yellow tones of the fabric, which indicate positive b* values. The highest chroma value was obtained for alum-mordanted viscose fabric. This can be explained by the formation of metal–dye complexes. 35 It was observed that when alum was used, the color became more intense resulting in higher K/S values. In other words, relatively high value of K/S indicates the significance of metallic mordant on the color depth.
CIEL*a*b* values of the dyed samples.

Fabrics dyed with A. orientalis extract and pictures of the fabrics after washing and light fastness tests.
The dyeing behavior of the fibers mainly depends on their chemical nature. Polyester fibers are built from multiple chemical repeating units linked together by ester (CO–O) groups. The chemical content of A. orientalis extract (Figure 3) was previously described.36,37 The dye–fiber complex occurs between the –OH (hydroxyl) groups of the dye molecules in the A. orientalis extract and the oxygen atom of the carbonyl group in polyester fabric. The proposed mechanism for dyeing polyester with A. orientalis extract is shown in Figure 4. As there are many compounds present in the extract, which can give color to fabrics, dyeing mechanisms of different types of fabrics are given with different compounds (Figures 4–6). The production of the fiber blends is the result of customers’ growing demands. Polyester–viscose blended fabrics are preferred due to their complementary properties.42,43 Different types of the fiber mixtures cause some difficulties in the dyeing process due to the differences in the chemical nature of the fibers. In addition, it is difficult to obtain high color strength values in dyeing the blended fabrics. Polyester fabrics are generally dyed at high temperatures (130°C) with disperse dyes, while viscose fabrics are dyed at 70°C–80°C. 44 Dyeing polyester–viscose blended fabrics gave a low K/S value (1.66) with A. orientalis extract. This may be due to the decomposition of dye at 130°C. 45 However, it is observed that the K/S value (3.55) increased in the presence of alum mordant.
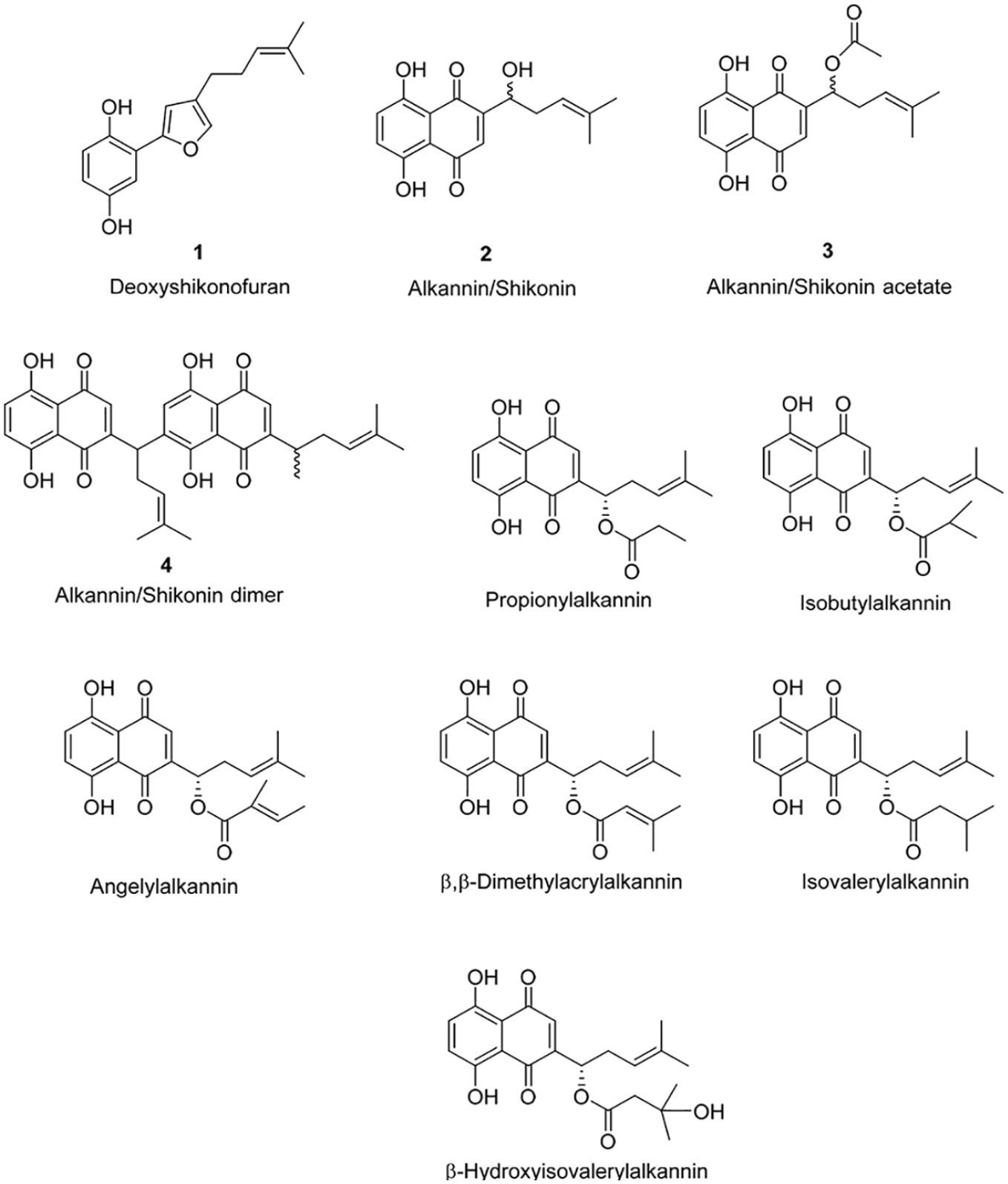
Chemical structures of the compounds of A. orientalis extract.
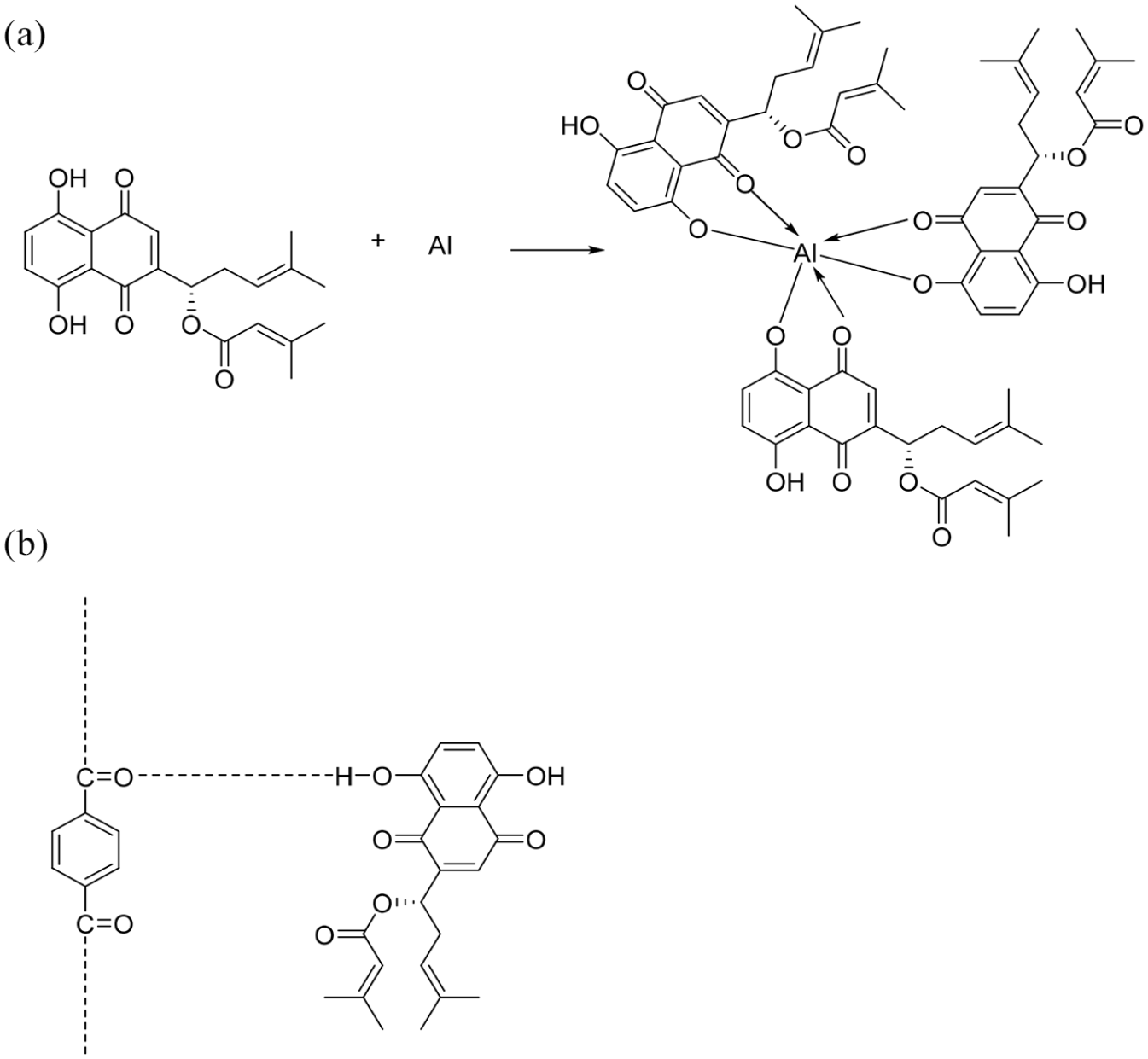
Chemical structure of the dyed cotton fabric with alkanin/shikonin acetate in the presence of metal mordant. 40
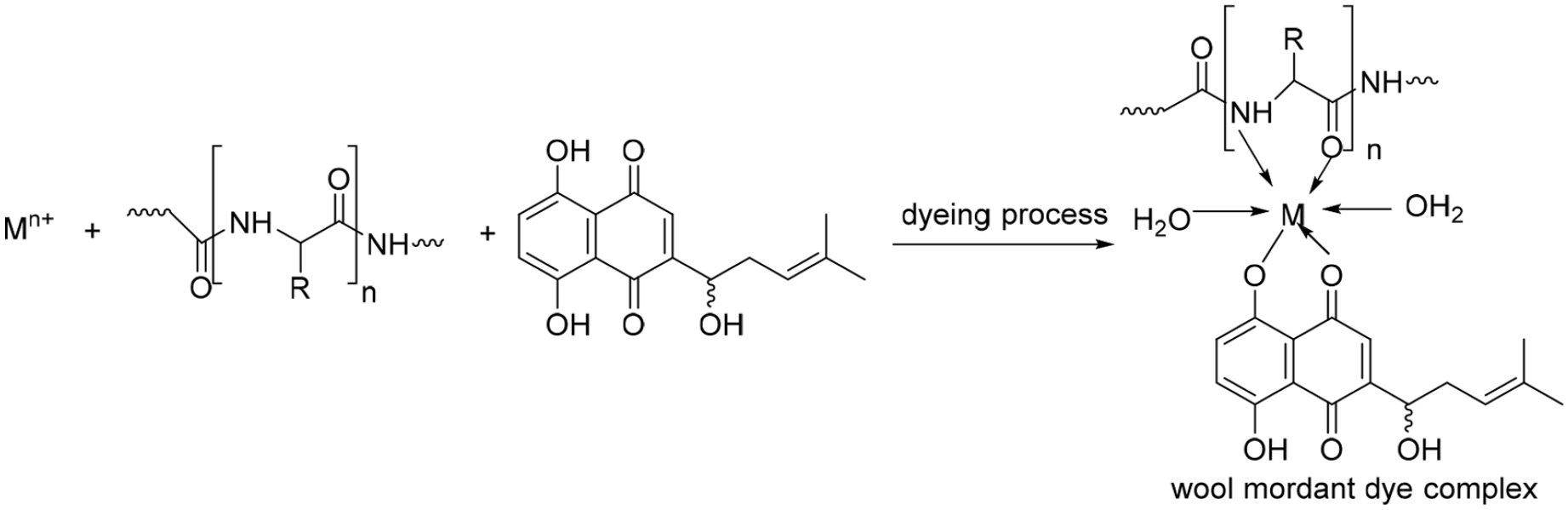
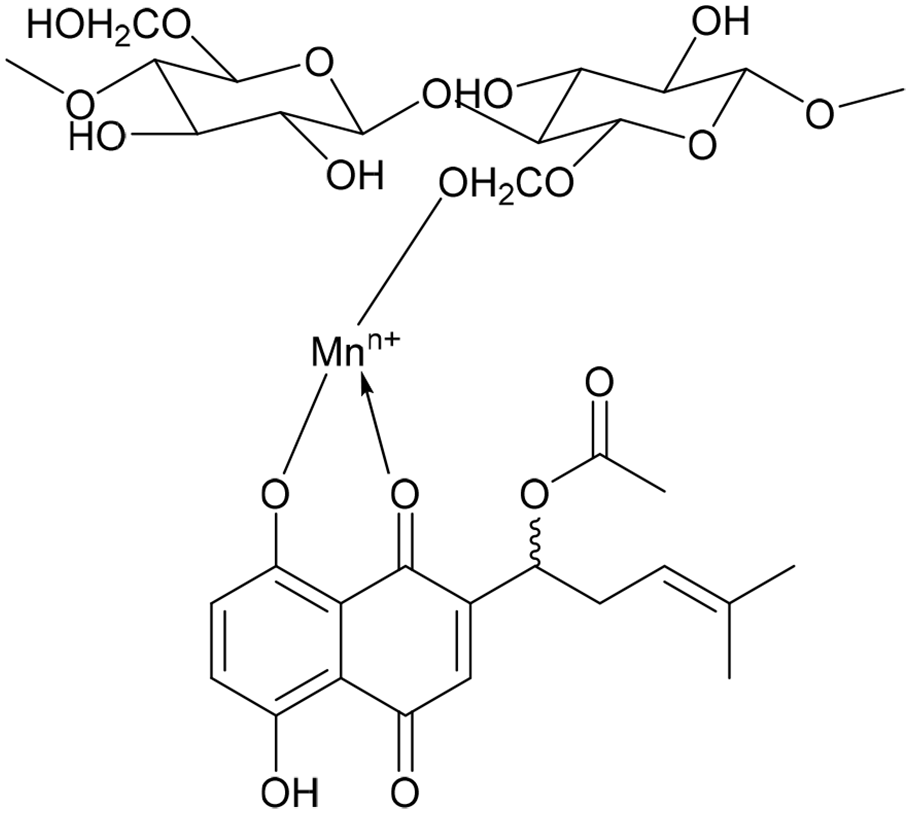
Dyeing mechanism of wool fabric with alkannin/shikonin in the presence of metal mordant. 41
Best color strength values 6.11 and 8.25 were obtained in dyeing cotton fabric, with unmordanted and mordanted experiments, respectively (Figure 7). The higher color strength value of the dyed cotton fabrics may be the result of the hydrogen bonding between the H atom of the dye and the electronegative oxygen atom of the cellulose molecule. 46 Cotton fiber contains large quantity of polar groups, which help in bonding. However, in the presence of auxochrome groups, adherence of some dyes to the fiber arises by chemical reaction. In many cases, the adherence of the dye to the fiber may arise from the dipolar interaction of these polar groups or the formation of intermolecular hydrogen bonding. 46
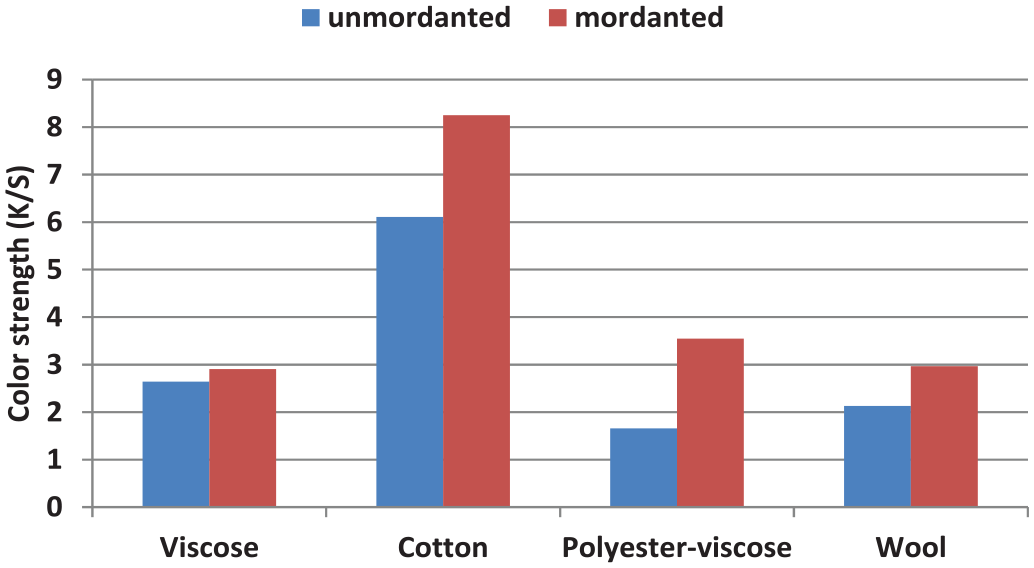
Color strength values of the dyed samples (K/S values at λmax = 420 nm).
Usage of the mordant improved the K/S value of the dyeing in all types of fibers. The higher dye uptake of the cotton fibers is the result of the strong coordination complexes between the dye molecule and the fabric. It is known that cotton fibers consist of cellulose units. Therefore, the coordination complex occurred between the –CH2O groups of the cellulose and auxochrome groups (–OH) of the dye molecule via hydrogen bonding. 47 The colors obtained with the various types of dyes vary in their hue due to the fact that when the different dyes (chemical content of A. orientalis extract) are combined with alum to form dye–alum complexes, different shades are attained (Figure 5).
Viscose fibers are chemically similar to cotton fibers, that is, they consist of cellulose. Different dyeing behaviors of viscose fabric can be explained by its capacity of the high moisture uptake. 48 However, wool fibers contain amino acid and carboxylic groups. So, the interaction occurs between the functional groups of the wool fiber and the dye molecule (Figure 6). In this study, low K/S values were obtained in dyeing the wool fabric both for the mordanted and the unmordanted form (Figure 7). This result can be explained by the decrease of the solubility of A. orientalis extract. In other words, A. orientalis extract contains water-soluble dyes due to the presence of –OH groups; therefore, it would thus interact ionically with the protonated terminal amino groups of the wool fibers at acidic pH values via an ion-exchange reaction because of the acidic property of –OH groups.49,50 The ionic interaction of the hydroxyl group of the dye and wool fibers decreases due to decreasing number of protonated terminal amino groups of wool fibers, thus lowering its dyeability, which resulted in lower K/S values. 51
Fastness Properties
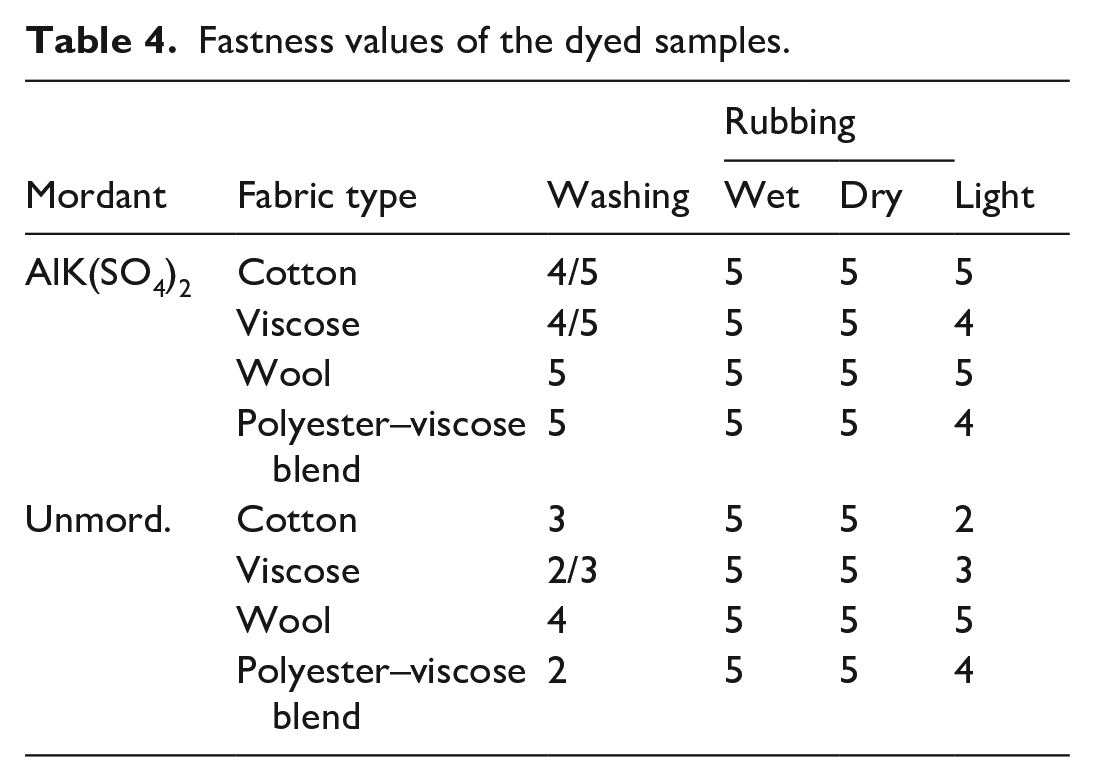
The fastness properties including washing, rubbing, and light fastness of the dyed fabrics are given in Table 4. Comparison of the washing fastness values of the dyed fabrics revealed that alum mordant improves the washing fastness levels of the dyed fabrics. The washing fastness values ranged between 4/5 and 5 (i.e. excellent to outstanding) and 2 and 4 (i.e. fair to very good) for alum mordanted and unmordanted fabrics, respectively. The higher fastness properties of the alum mordanted samples may be explained by the ability of the mordant ion to form stable complex with the dye molecule and the functional groups of the fiber. 52 All of the dyed fabrics exhibited outstanding rubbing (wet/dry) fastness scores. Light fastness values of the alum mordanted fabrics ranged between 4 and 5 (i.e. moderate and good), while the light fastness values ranged between 2 and 5 (i.e. poor to good) for unmordanted fabrics. However, the light fastness values of the alum-mordanted cotton and viscose fabrics were found to be higher than those of the unmordanted fabrics. The higher light fastness values may be due to the strong intramolecular hydrogen bonding, which helps to improve the stability of the compound via reducing the photochemical oxidation. 53 The results are in accordance with the study of Avinc et al. 54 The brightness of the colors on cotton and wool fabrics might be due to the better absorption of the A. orientalis extract along with the easy metal complex formation of mordant with the fibers. 55 It is concluded that alum mordant not only improves the washing fastness of the dyed fabrics but also light fastness.
Fastness values of the dyed samples.
The K/S values of the dyed fabrics, after washing fastness, and after light fastness are shown in Figure 8. It is observed that color strength values decreased after washing fastness and light fastness tests. The decrease in the K/S values for alum mordanted fabrics was found to be higher than for unmordanted samples both for washing and light fastness values except unmordanted viscose fabric. Different types of fabrics exhibited different degrees of color strength decrease. The highest decrease values were obtained for alum-mordanted cotton fabrics both after washing and light fastness tests.
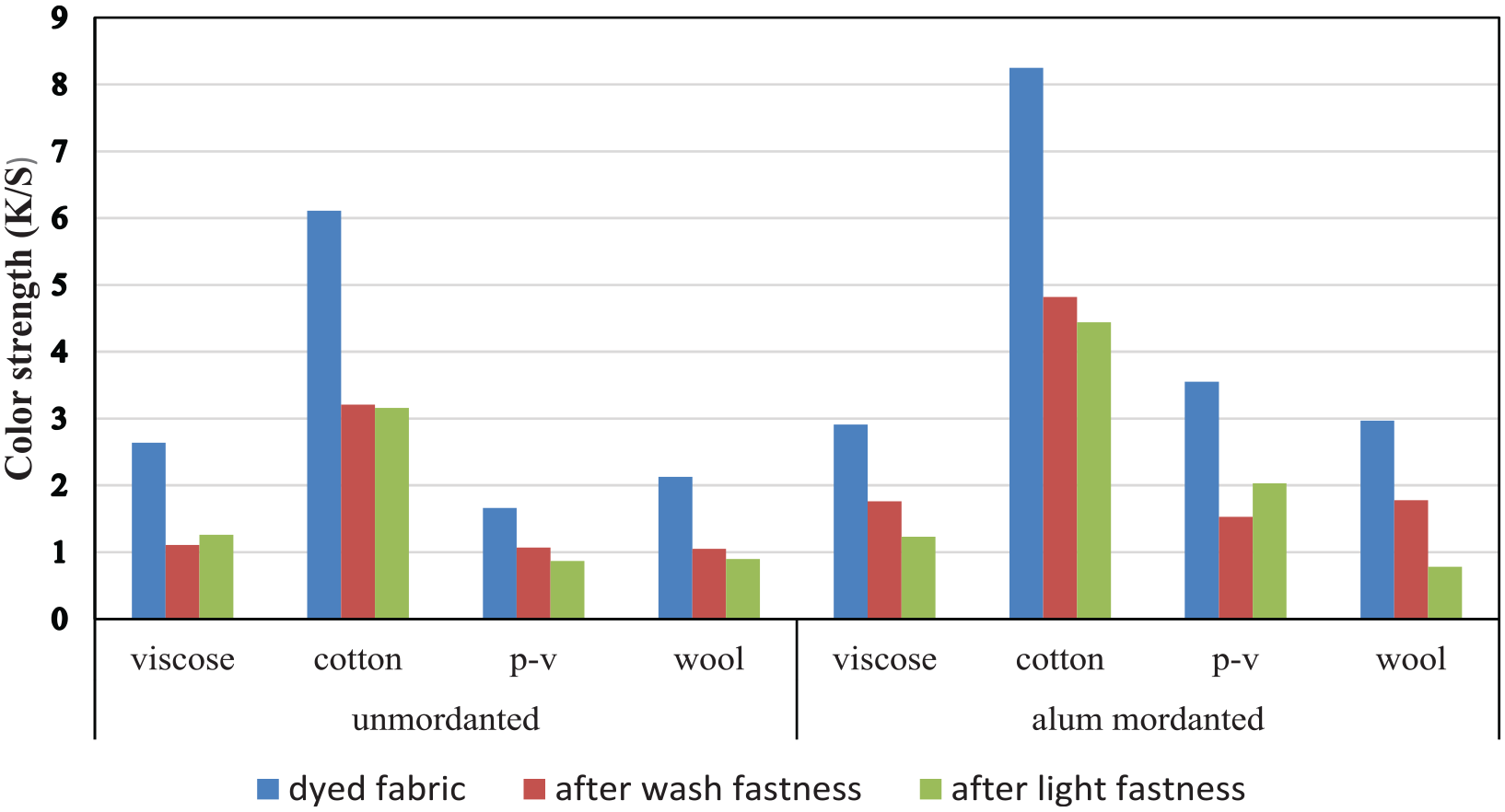
Color strength values of the dyed fabrics, after washing fastness, and after light fastness: (p-v: polyester–viscose).
Conclusion
In this study, the dyeing behavior of Alkanna orientalis was examined on the polyester/viscose blend, cotton, viscose, and wool fabrics. Ethanolic and aqueous extracts were investigated for their phenolic compounds and cytotoxicity. Ten compounds were determined from the roots of A. orientalis. Rosmarinic acid and 4-hydroxybenzoic acid were found to be the main phenolic components with the ethanol and the water extracts, respectively. Cytotoxic activity tests showed that EtOH and water extract of the root of A. orientalis exhibited 0% cytotoxicity while 5-FU exhibited 6%. In another words, the extracts were found to be less toxic than 5-FU. The best color strength value (K/S = 8.25) was obtained in the presence of alum mordant, using meta-mordanting method, for cotton fabric. From the overall results, it was concluded that the roots of A. orientalis can be used as an alternative dyestuff in dyeing cotton fabrics.
Footnotes
Acknowledgements
The plant was collected from Cankiri region in July 2016 (536999 E, 4490037 N). The identification of the plant was performed by Dr Melda Dolarslan (Faculty of Science, Department of Biology, Cankiri Karatekin Üniversitesi). The roots of the plant are deposited in the Herbarium of Ankara University (ANK60537). The roots of the plant were dried at room temperature (25°C) in the shade.
Author’s Note
Ayse Sahin Yaglıoglu is now affiliated to Amasya University, Amasya, Turkey.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.