Abstract
In the present study, berberine, a natural dye, was modified by a simple chemical conversion with the aim of improving its dyeing properties. The modified berberine was successfully applied in the dyeing and functional finishing of a variety of fabrics. The uptake of the modified berberine by acrylic fabric was more sensitive to the pH of dye bath than that of berberine. The silk, cotton, wool, polyamide, and polyester fabrics dyed with the modified berberine all showed greater exhaustion than with berberine. In addition, the textile treated with the modified berberine showed better color fastness and much better UV protection ability. This study shows that this novel modified berberine can be suitably applied to prepare multifunctional textiles for many fabrics.
Keywords
Introduction
Synthetic dye use for textile dyeing is one of the world's most polluting industries. With environmental concerns regarding the use of synthetic chemical dyes, natural dyes offer the prospect of eco-friendly dyeing of fibrous materials. In recent years, natural dyes and pigments have emerged as an important alternative to potentially harmful synthetic dyes. 1 Moreover, there has been growing interest in the use of natural dyes in fundamental textile research.2–8 Researchers are improving the technologies and strategies for extraction and application of natural dyes to natural and synthetic fabrics.9–13
Berberine, a natural cationic colorant present in the roots of barberry (
In this study, berberine was modified by a simple chemical conversion with the aim of improving its dyeing properties (Fig. 1). The modified berberine was fully characterized by Fourier transform infrared (FTIR) spectroscopy, proton nuclear magnetic resonance (NMR) spectroscopy, high-resolution mass spectrometry (HRMS), and UV-Vis absorption spectroscopy. Taking into account our longstanding research into dyeing acrylic fabrics with berberine, the dyeing properties and color characteristics of acrylic fabrics with berberine and modified berberine were performed. The UV protection performance and the overall fastness properties of the dyed fabrics with modified berberine were then investigated by comparison with berberine.
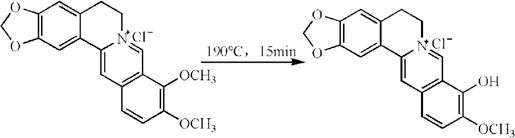
The modification of berberine.
Experimental
Materials
The rhizome powder of
Procedures
Berberine Pigment Extraction and Purification
Chemical Modification of Berberine
Berberine (2.0 g) was put into a heat resistant crucible. The system was slowly heated up to 190 °C for 15 min in a high-temperature electric furnace. 18 After cooling to room temperature, the modified berberine was stored in the dark for further experiments without further purification (yield up to 95%).
Dyeing Experiments
All dyeing processes were carried out in a laboratory-scale infrared dyeing machine (IR-24S). The liquor ratio (LR, the ratio of the volume of liquor to the weight of fabric) was 100:1. The pH values of the dye bath were adjusted from 2 to 13 using Britton-Robinson buffers (H3PO4-HOAc-H3BO3 and 0.2 mol/L NaOH). Dyeing was started at 30 °C and the temperature raised to 100 °C (130 °C for polyester fabric) at a rate of 1 °C/min; at this temperature, dyeing was performed for 60 min. At the end of dyeing, the fabrics were washed in tap water and then dried in the open air.
To evaluate the pH dependence of dyeing, a dye concentration of 2% owf was used, and the pH values of the dye bath were adjusted from 2 to 13 using Britton-Robinson buffers. In the subsequent assessment of color fastness and functionalities, the samples (dyed with 2% owf berberine and modified berberine) obtained according to the above experimental conditions were used.
Analytical Methods
Spectroscopy
The absorption spectra of berberine and modified berberine in water were measured using a Shimadzu UV-1800 UV-Vis spectrophotometer (Shimadzu Co. Ltd.) The FTIR spectra of the samples were obtained using a Is5 FTIR spectrometer (Thermo Fisher Scientific Inc.) Each sample was recorded with 32 scans at 4 cm–1 resolution. Before measurement, the berberine and the modified berberine were dried under vacuum, ground, mixed with KBr, and pressed into small pellet disks. The structure of berberine and modified berberine were confirmed using a Aurora M90-Advance HRMS (Bruker) and an Avance III HD NMR spectrometer (Bruker).
Exhaustion and Color Measurements
The absorbance of dye solutions was measured using a Shimadzu UV-1800 UV-Vis spectrophotometer. The dye exhaustion was determined by measuring the absorbance of the initial and final dye solutions. The dye exhaustion percentage
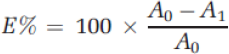
To evaluate the dyeing performance, the color strength (
Color Fastness
The wash fastness of the dyed fabrics was carried out using a SW-12A wash fastness tester (Wenzhou Fangyuan Instrument Co. Ltd.), using the standard test method ISO 105-C06. 19 Rubbing fastness was measured on a Y571N rubbing color fastness tester (Wenzhou Fangyuan Instrument Co. Ltd.) according to ISO 105-X12. 19 Color fastness to light was evaluated using a YG 611E light fastness tester (Nantong Hongda Instrument Co. Ltd.) according to ISO 105-B01. 19
Ultraviolet Protection Ability
The ultraviolet protection factor (UPF) and the UV trans-mittance of the fabrics were determined using a Labsphere UV-2000F ultraviolet transmittance analyzer (Labsphere Inc.) Each sample was tested four times at different positions, and the average of the data was used.
Results and Discussion
Modified Berberine Structure
UV-Vis Absorption Spectroscopy
The UV-Vis absorption spectra of berberine and modi-fed berberine in water are shown in Fig. 2. The modified berberine displayed a maximum absorption peak at 488 nm compared with that of berberine at 426 nm. The red shift of maximum absorption wavelength suggests that the phenolic hydroxyl group exists as the phenoxy anion in the solution.
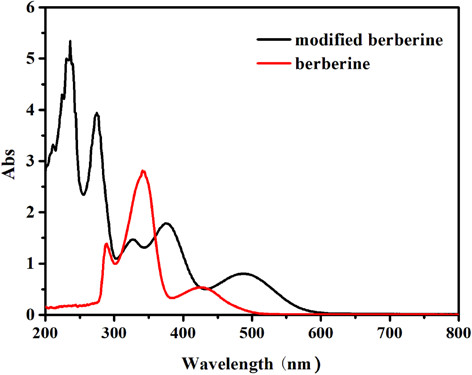
UV-Vis absorption spectra of berberine and modified berberine in water (0.10 g/L).
Modified Berberine Structure Analysis
As seen in Fig. 3, modified berberine shows a broad FTIR absorption band in the range of 3000–3500 cm–1 due to the phenolic hydroxyl group stretching vibration.
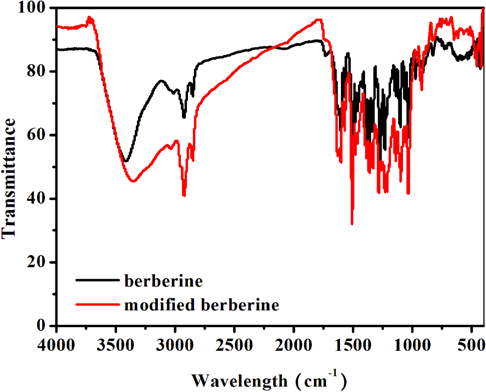
FTIR spectra of berberine and modified berberine.
To further characterize the modified berberine, HRMS analysis was performed, and the results are shown in Fig. 4. The diagnostic fragment ions at
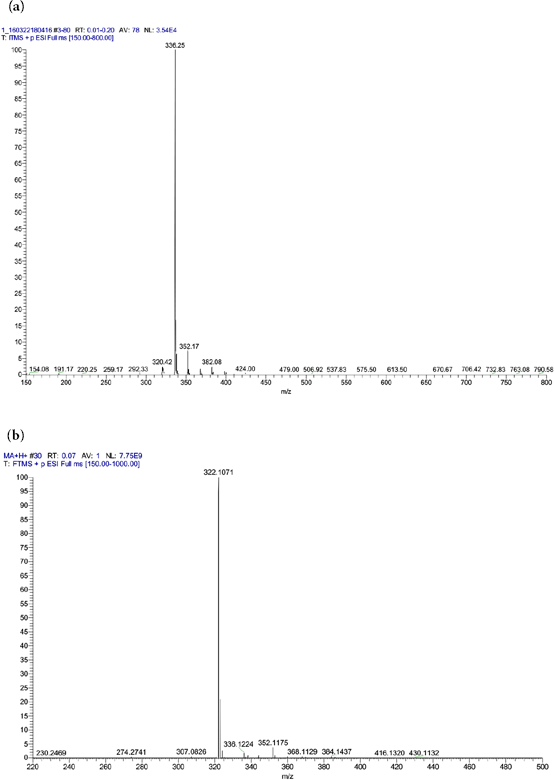
HRMS spectra of berberine (a) and modified berberine (b).
To confirm the structure of the modified berberine, the NMR spectrometer was used, and the results are shown in Fig. 5. One of the methyl groups in berberine disappeared after modification without other significant changes. For berberine: 1 H NMR (400 MHz, CD3OD) δ 3.23 (t, 2H, -CH2-), 4.13 (s, 3H, -CH3), 4.23 (s, 3H, -CH3), 4.94 (t, 2H, -CH2-), 6.13 (s, 2H, -CH2-), 6.99 (s, 1H, -Ph), 7.69 (s, 1H, -Ph), 8.01 (d, 1H, -Ph), 8.14 (d, 1H, -Ph), 8.67 (s, 1H, -Ph), and 9.79 (s, 1H, -N=CH-). For modified berberine: 1 H NMR (400 MHz, CD3OD) δ 3.14 (t, 2H, -CH2-), 3.89 (s, 3H, -CH3), 4.63 (t, 2H, -CH2-), 6.04 (s, 2H, -CH2-), 6.83 (s, 1H, -Ph), 6.94 (s, 1H, -Ph), 7.44 (d, 1H, -Ph), 7.54 (d, 1H, -Ph), 8.06 (s, 1H, -Ph), and 9.29 (s, 1H, -N=CH-). NMR results were basically consistent with the literature results, confirming formation of the isomer shown in Fig. 1. 20
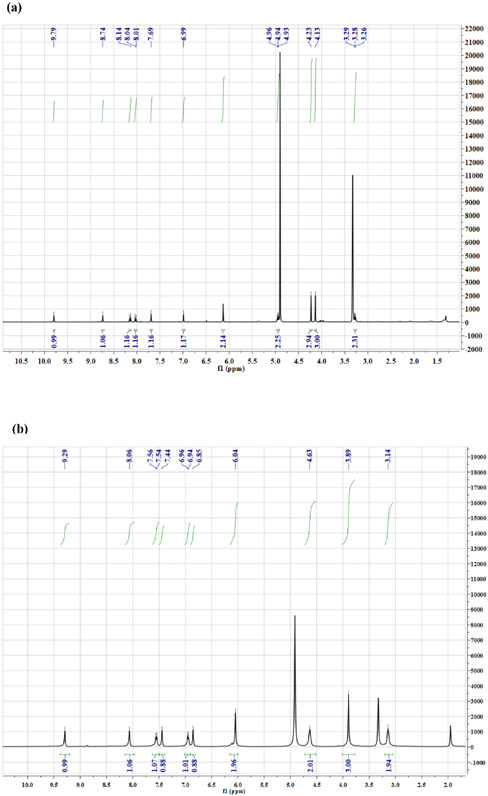
NMR spectra of berberine (a) and modified berberine (b).
Dyeing Properties of Modified Berberine
pH Dependence
It can be seen from Fig. 6 that the pH of the dye bath exerted a great influence on the uptake of both berberine and modi-fed berberine by acrylic fabric. The greatest exhaustion was obtained for berberine at pH 4.1, while the exhaustion of modified berberine decreased significantly with increased pH value (with the highest exhaustion at pH 2). Therefore, modified berberine was more sensitive to pH than berberine with regard to exhaustion on acrylic fabric. Especially at high pH values, the phenolic hydroxyl group may exist in the presence of the phenoxy anion, which led to low exhaustion. The exhaustion with modified berberine was less than that for berberine on acrylic fabric. Perhaps the phenolic hydroxyl group in modified berberine enhanced its water solubility.
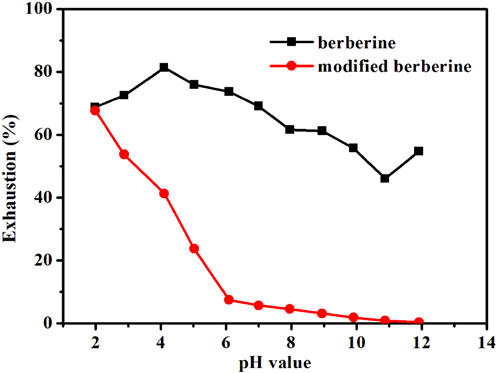
Effect of pH on exhaustion on dyed acrylic fabric.
Fig. 7 showed that the color strength on acrylic decreased when using modified berberine. The extent of adsorption of the modified berberine onto acrylic fabric decreased gradually with increased application pH. The pH value had less of an impact on the
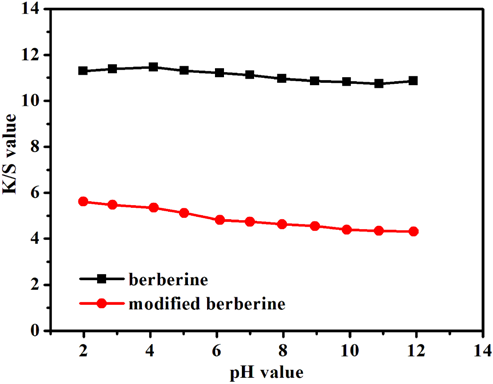
Effect of pH on
Fabric Dyeing
There was a great reduction of dyeing performance on acrylic fabric using modified berberine. Modified berberine dyeing of cotton, silk, wool, polyamide, and polyester were also investigated at pH 4.1. For all fabrics other than acrylic that were tested, the exhaustion and resulting
From Fig. 8, it can be seen that the dyeing properties of the modified berberine on these fabrics were improved to some extent, especially for silk. This may be related to the formation of hydrogen bonds between the phenolic hydroxyl group of the modified berberine and silk.
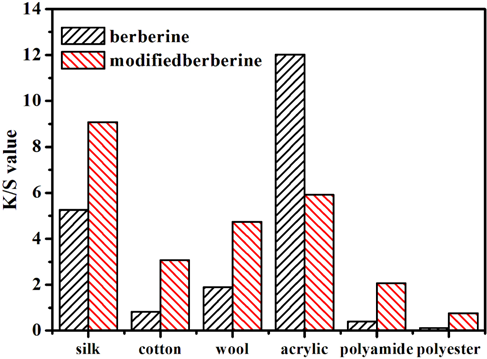
Dyeing of other fabrics with berberine and modified berberine.
Color Fastness
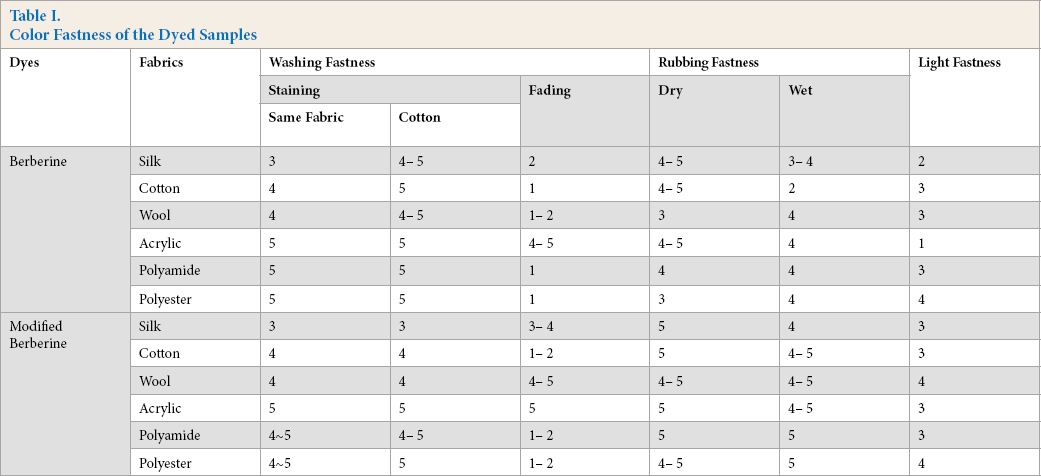
The dry and wet rubbing fastness, washing fastness, and light fastness of the dyed samples were measured (Table I). The rubbing fastness of the modified berberine showed considerable improvement. As seen in Table I, the ratings for color fastness to light in terms of the degree of color change were good to very good. The light fastness of the dyed acrylic and silk fabrics increased using the modified berberine. The significant enhancement in light fastness was mainly produced by the hydrogen bonds between the phenolic hydroxyl groups of the modified berberine and the fabrics.
Color Fastness of the Dyed Samples
UV Protection
Some natural extracts can increase the UV protection ability of textiles.21,22 The UV protection performance of undyed, and berberine and modified berberine dyed samples, were evaluated in terms of UV transmittance and UPF (Figs. 9 and 10). The results showed that the original fabrics had poor UV protection ability and that there was an effective reduction of UV light transmittance through the fabric dyed with berberine and modified berberine. Compared with berberine dyeing, dyeing with modified berberine provided cotton, wool, polyamide, and polyester fabrics tested with better UV protection ability over the UV light range of 370-450 nm. Cotton, wool, and silk fabrics dyed with modified berberine showed a UPF value of greater than 200, which is rated as “excellent protection” according to the UPF Classification system (Table II).21,23
UPF Classification System
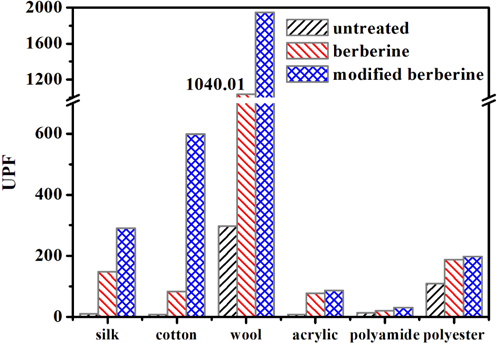
UPF of fabrics dyed with berberine and modified berberine.
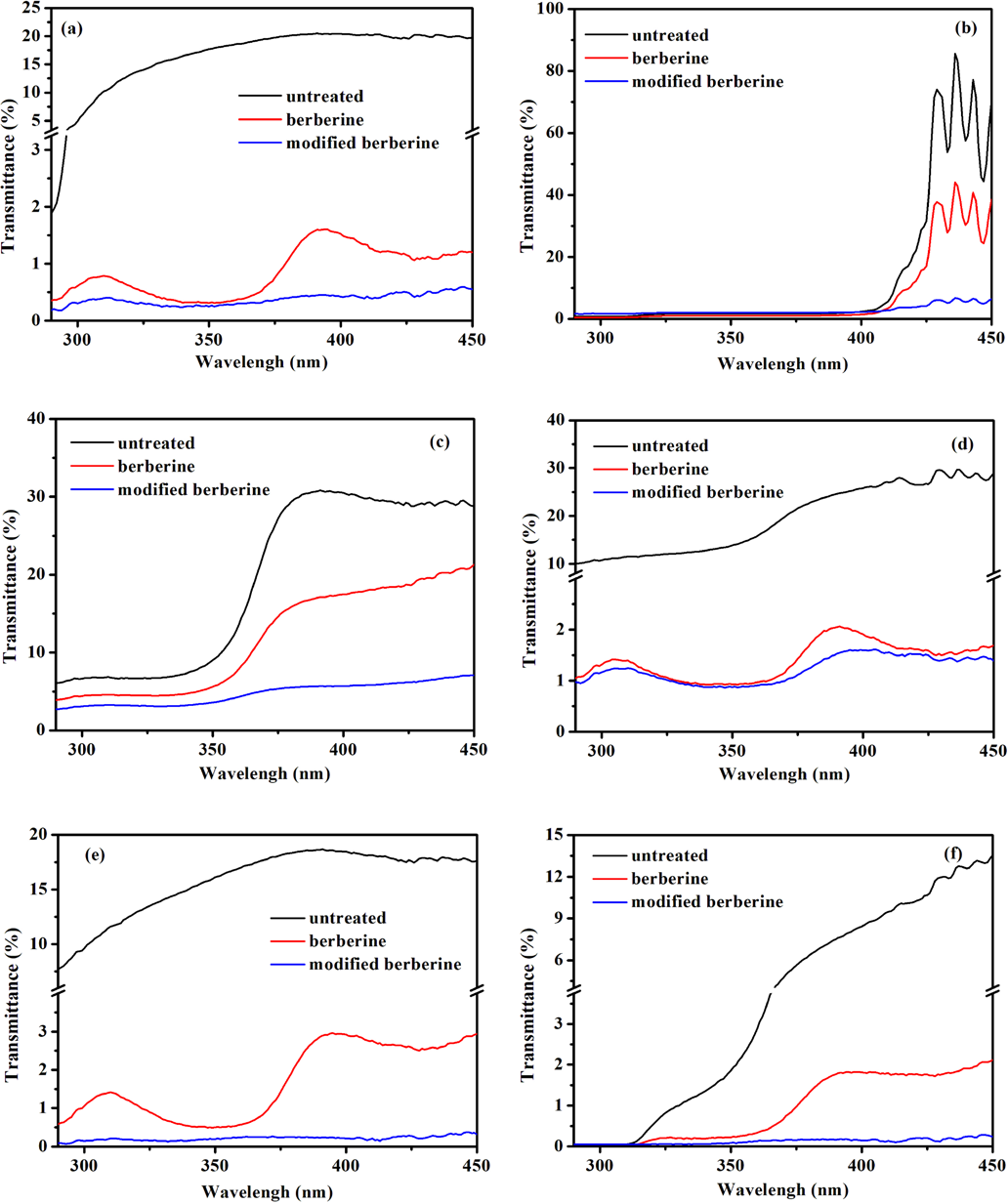
UV transmittance of fabrics dyed with berberine and modified berberine: (a) silk, (b) polyester, (c) polyamide, (d) acrylic, (e) cotton, and (f) wool.
Conclusions
This paper presented the synthesis of a modified berberine with a simple chemical conversion. The modified berberine was successfully applied to the acrylic, cotton, silk, wool, polyamide, and polyester fabrics tested, although it was more sensitive to pH and showed a reduction in uptake by acrylic fabric. The fabrics dyed with the modified berberine showed good color fastness and excellent UV protection ability. The results suggest that the present dye chemical modification provides a new approach to the preparation of colored textiles.
Footnotes
Acknowledgements
This work was partly supported by the 2017 science and technology development plans of Henan province (172102310466), and the 2015 open-funded project of the Henan Clothing Textiles Engineering Research Center (GCSYS201502).
