Abstract
Novel azo compounds were synthesized by diazotization of aniline and of 3-aminopyridine in the presence of NaNO2 and HCl, followed by coupling with 2-methylresorcinol in alkaline media. The structures were characterized using FT-IR, 1H NMR, and 13C NMR analysis. The main purpose of the present study is to investigate coloration applications, and antibacterial and fastness properties of the new azo structures in textile industry. Exhaust dyeing processes were applied on cotton fabric. Color strength (
Introduction
Azo compounds constitute the largest chemical class of commercial synthetic organic colorants, comprising approximately 70% of all dyes used in industry.1,2 They can possess an especially intense yellow, red, orange color, even a blue or green color depending on the exact structure of the molecule. As a result of providing bright color at low cost, azo compounds have great importance as dyes and also pigments for coloring of different materials in various fields such as printing inks, paints (architectural, automotive, and artists’), plastics, rubber, crayons, and colored chalks, particularly in the textile, cosmetic, and pharmaceutical industries. Azo compounds are substantial in almost whole industrial application classes of textile dyes such as acid, reactive, direct, metal complex, disperse, vat, sulfur, basic, and solvent dyes.3–6
Azo colorants include as their common structural chromophore feature the azo (-N==N-) linkage and can be classified according to the number of azo groups in the same structure such as monoazo (commercially mostly used), disazo, trisazo, polyazo, and azoic.1,7 Industrial azo compounds occur by the same two reaction steps: diazotization of a primary aromatic amine followed by coupling with electron-rich nucleophiles such as mostly used amino and hydroxyl groups.1,2,7,8 Lots of derivatives with different side groups having different features can be synthesized by this method. This versatility gives rise to many different application fields such as photoreaction sensitizers, advanced applications in organic synthesis, extraction of metal ions, acid–base indicators, liquid crystalline displays, electro-optical devices, and biological–medical studies.9–12
Azo compounds can exhibit a great variety of interesting biological activities, including antimicrobial, anti-inflammatory, anthelminthic, antiviral, antioxidant, antidiabetic, antitubercular, anticonvulsant, and anticancer effects. The medical importance of these compounds is well known for their antibiotic, antifungal, and anti-HIV properties.12–24
The majority of azo pigments are safe to use, but some of them are, especially those derived from aniline, toluidine, and benzidine diazo components, mutagenic and carcinogenic. They can undergo natural anaerobic degradation, so break down and release potentially harmful amines for humans and the entire ecosystem.9,25,26 A list of EU-restricted aromatic amines is given in the literature. 27 Replacement of aromatic diazo components with heteroaromatic ones could satisfy toxicological requirements for the design of new dyes. 28
This study is centered on synthesizing novel azo compounds and then investigating their color fastness and antibacterial properties on cotton fabric.
Experimental
Materials
All used reagents and solvents were of analytical grade quality. Indosol E 50 Liq. was obtained from Clariant Chemicals and used as a cationic agent. The bleached, hydrophilic 100% cotton fabric was in a single jersey knit structure (İzmir Basma Factory, Turkey), 150 g/m2.
Synthesis of the Azo Compounds
The synthetic route of the novel azo compounds can be seen in Scheme 1. All compounds were synthesized by the azo coupling reaction of aniline and 3-amino pyridine with 2-methyl resorcinol.
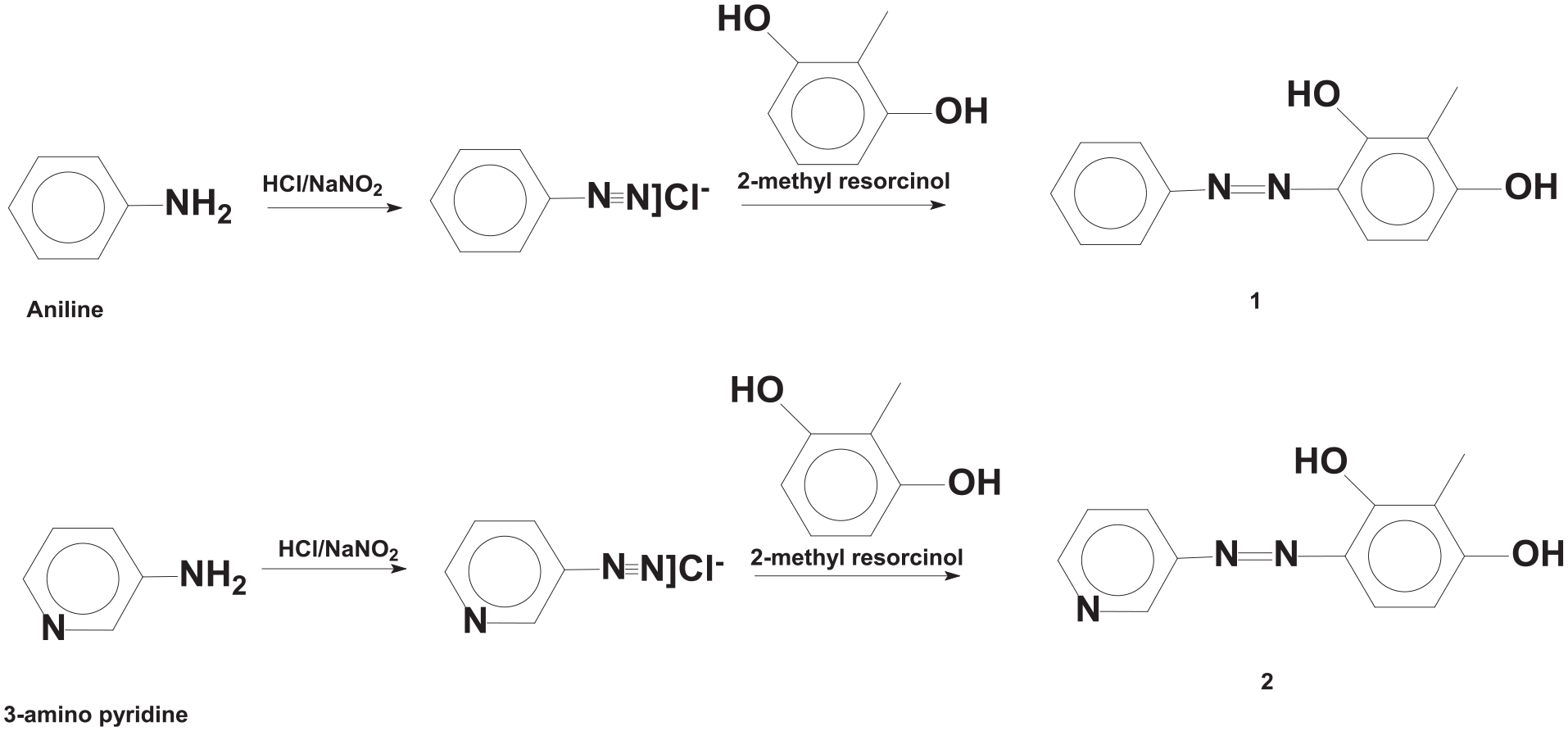
Synthetic route of azo compounds 1 and 2.
2-Methyl-4-[phenyldiazenyl]benzene-1,3-diol (Compound 1)
Aniline (1000 mg, 10.73 mmol) was dissolved in pure water (50 mL) and HCl (10 mL, 30%). Then, the solution of equivalent sodium nitrite (NaNO2) was added dropwise to this solution. The temperature was kept between 0 and 5°C during addition. Equivalent 2-methyl resorcinol (1332 mg, 10.73 mmol) in NaOH solution (70 mL, 10%) was added into this diazonium solution with efficient stirring. Then dilute HCl solution was added to the mixture until it became neutral. The formed solid material was filtered off. Finally, pure product was obtained by recrystallization from ethanol. Yield: 2100 mg (85%); m.p. 130°C–131°C.
This compound is soluble in ethanol and dimethyl sulfoxide. FTIRνmax/cm−1 3150, 2918, 2850, 1625, 1530, 1488 (N=N), 1430, 1341, 1307, 1235, 1183, 1134, 1084, 1035, 901, 794, 752, 682. 1H NMR (DMSO-d6) δ, ppm: 13.23 (1H, s, OH), 10.53 (1H, s, OH), 7.84–7.82 (2H, d, ArCH), 7.56–7.51 (3H, m, ArCH), 7.46–7.43 (1H, d, ArCH), 6.61–6.59 (1H, d, ArCH), 2.14 (3H, s, CH3). 13C NMR (DMSO-d6) δ, ppm: 161.25, 154.47, 150.61, 132.20, 130.50, 129.91, 129.83, 121.89, 111.19, 108.80, 8.21.
2-Methyl-4-[pyridin-3-yldiazenyl]benzene-1,3-diol (Compound 2)
3-Amino pyridine (1000 mg, 10.62 mmol) was dissolved in pure water (50 mL) and HCl (10 mL, 30%). Then, the solution of equivalent sodium nitrite (NaNO2) was added dropwise into this solution. The temperature was kept between 0°C and 5°C during addition. Equivalent 2-methyl resorcinol (1319 mg, 10.62 mmol) in NaOH solution (70 mL, %10) was added to this diazonium solution with efficient stirring. Then, dilute HCl solution was added into the mixture until it became neutral. The formed solid material was filtered off. Finally, pure product was obtained by recrystallization from ethanol. Yield: 2150 mg (88%); m.p. 224°C–225°C.
This compound is soluble in ethanol and dimethyl sulfoxide. FTIRνmax/cm−1 3071, 2988, 2590, 1613, 1451, 1421 (N=N), 1357, 1328, 1261, 1194, 1178, 1089, 806, 793, 806, 757, 697. 1H NMR (DMSO-d6) δ, ppm: 12.53 (1H, s, OH), 10.63 (1H, s, OH), 9.06-9.05 (1H, d, ArCH), 8.61-8.60 (H, d, ArCH), 8.22-8.20 (1H, d, ArCH), 7.57-7.53 (2H, m, ArCH), 6.61-6.59 (2H, d, ArCH), 2.02 (3H, s, CH3). 13C NMR (DMSO-d6) δ, ppm: 161.95, 154.93, 150.92, 146.74, 145.70, 132.82, 127.92, 126.89, 124.89, 111.26, 109.00, 8.28.
Cationization and Dyeing Procedures
Cotton specimens were cationized with the assistance of a cationic auxiliary (Indosol E 50 Liq, 5% conc.) at 60°C for 20 min in acidic media (via acetic acid, pH 5) by using the exhaustion method (1:10 liquor ratio). The cotton fabrics were then rinsed with tap water and air-dried.
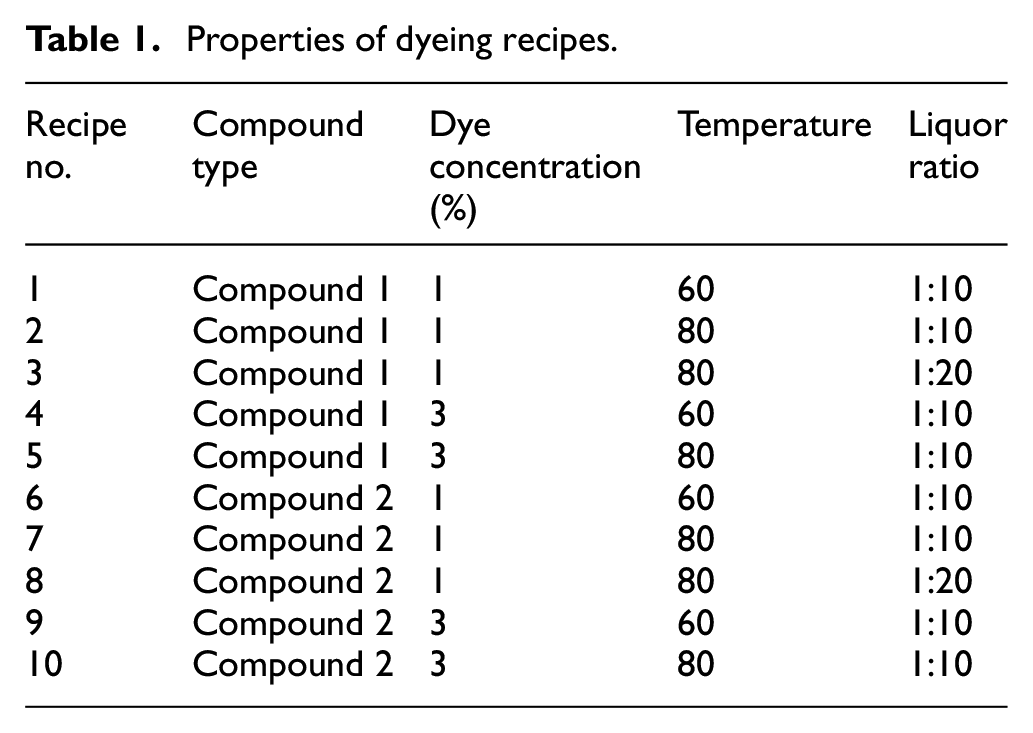
After cationization process, the cotton fabrics were dyed according to the recipes given in Table 1. Exhaust dyeing processes were carried out at different conditions according to Figure 1.
Properties of dyeing recipes.
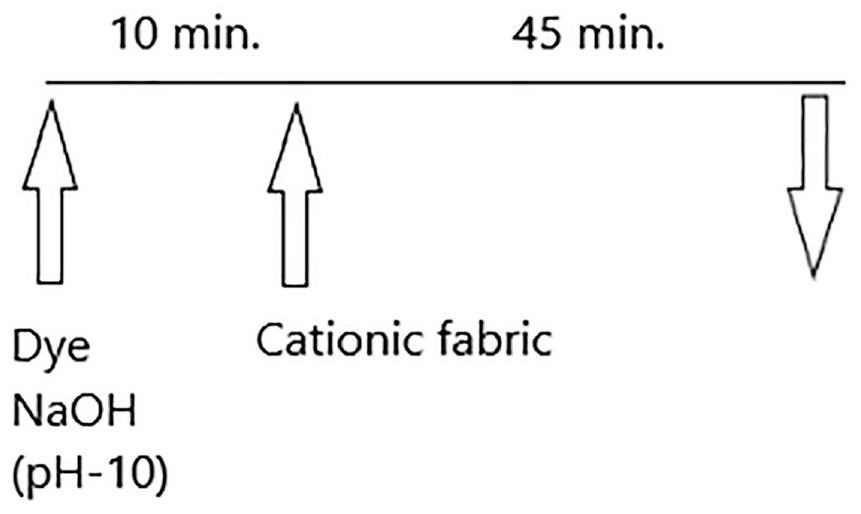
Dyeing process conditions.
The azo compounds 1 and 2 were solubilized in alkali media (via
Characterization Analyses
FT-IR spectra were recorded by Perkin-Elmer Spectrum 100 Infrared Spectrometer. 1H NMR and 13C NMR studies were performed by Agilent 400 FT-NMR. Dyeing and cationization processes were carried out in Termal Laboratory Dyeing Machine (Turkey).
Color Measurements
The color strength (

where
Antibacterial and Fastness Test Methods
Antibacterial properties of the fabrics, colored with recipes 4 and 9, were evaluated according to AATCC 147-2016 test method against two types of bacteria:
Fabrics were tested according to ISO 105-X12 for crock fastness, ISO 105-B02 for lightfastness, ISO 105-CO6 for washfastness, and ISO 105-E04 for perspiration fastness.
Results and Discussion
Synthesis and Characterization
The synthetic route of azo compounds can be seen in Scheme 1. All compounds were synthesized by the azo coupling reaction of aniline and 3-amino pyridine with 2-methyl resorcinol. Newly synthesized azo compounds were characterized by FT-IR, 1H NMR, and 13C NMR techniques.
Compound 1 exhibited the broad OH band at 3150 cm−1 in the FT-IR spectrum (Figure 2). The formation of compound 1 was clearly indicated by the appearance of two OH peaks at 13.23 and 10.53 ppm, aromatic peaks at 7.84–6.59 ppm, and an aliphatic CH3 peak at 2.01 ppm in its 1H NMR spectrum. The singlet peak at 13.23 ppm can be assigned to the proton of the OH group which is intramolecular hydrogen bonded with the nitrogen atom in the neighboring azo group (Scheme 2).29–32 The 13C NMR spectrum of compound 1 is compatible with the suggested structure (Figure 3).
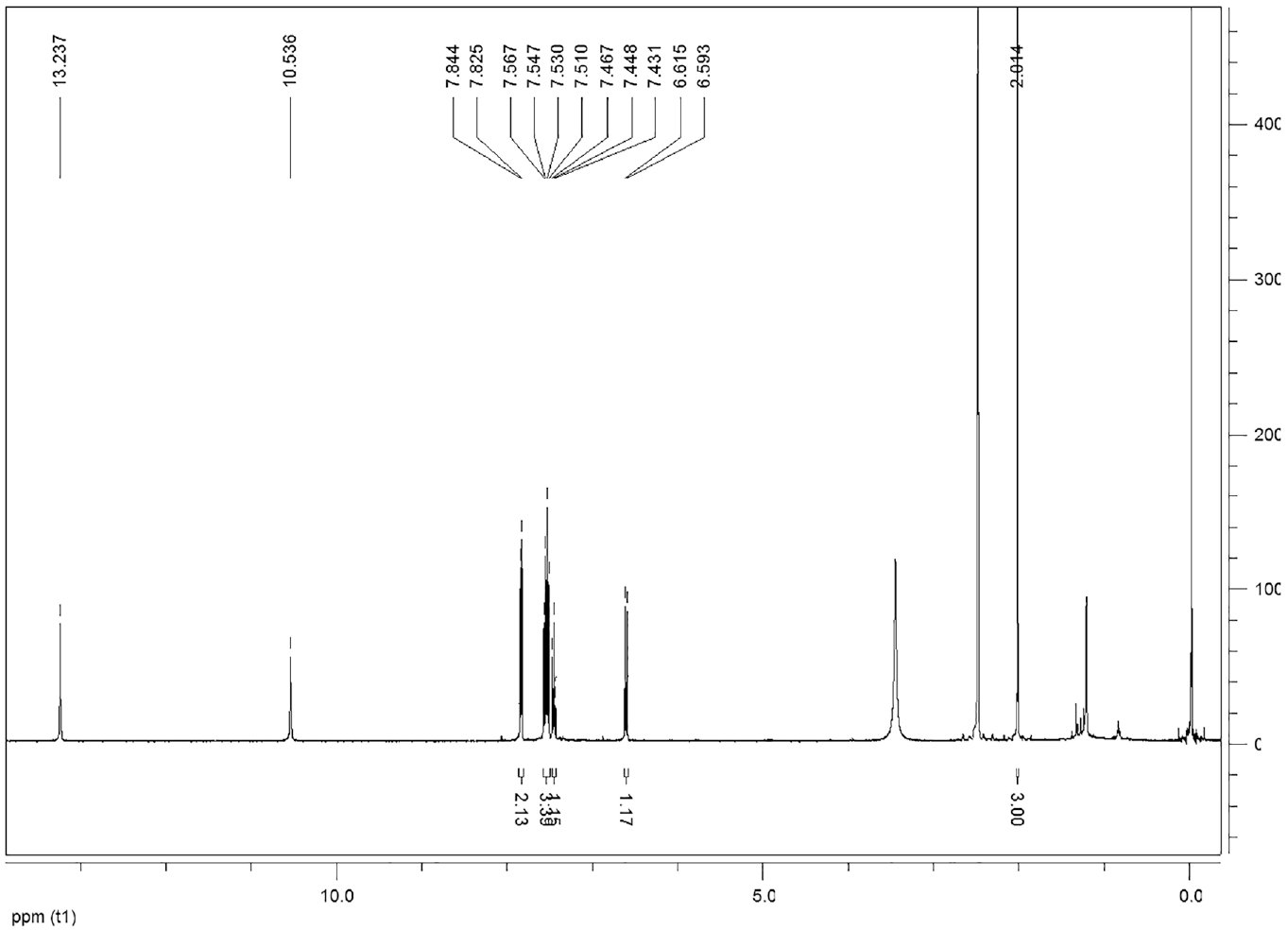
1 H NMR spectrum of compound 1.
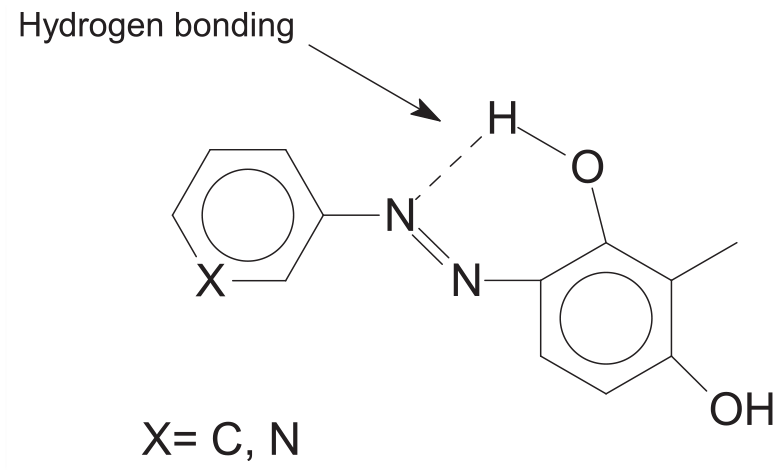
Hydrogen bonding at compounds 1 and 2.
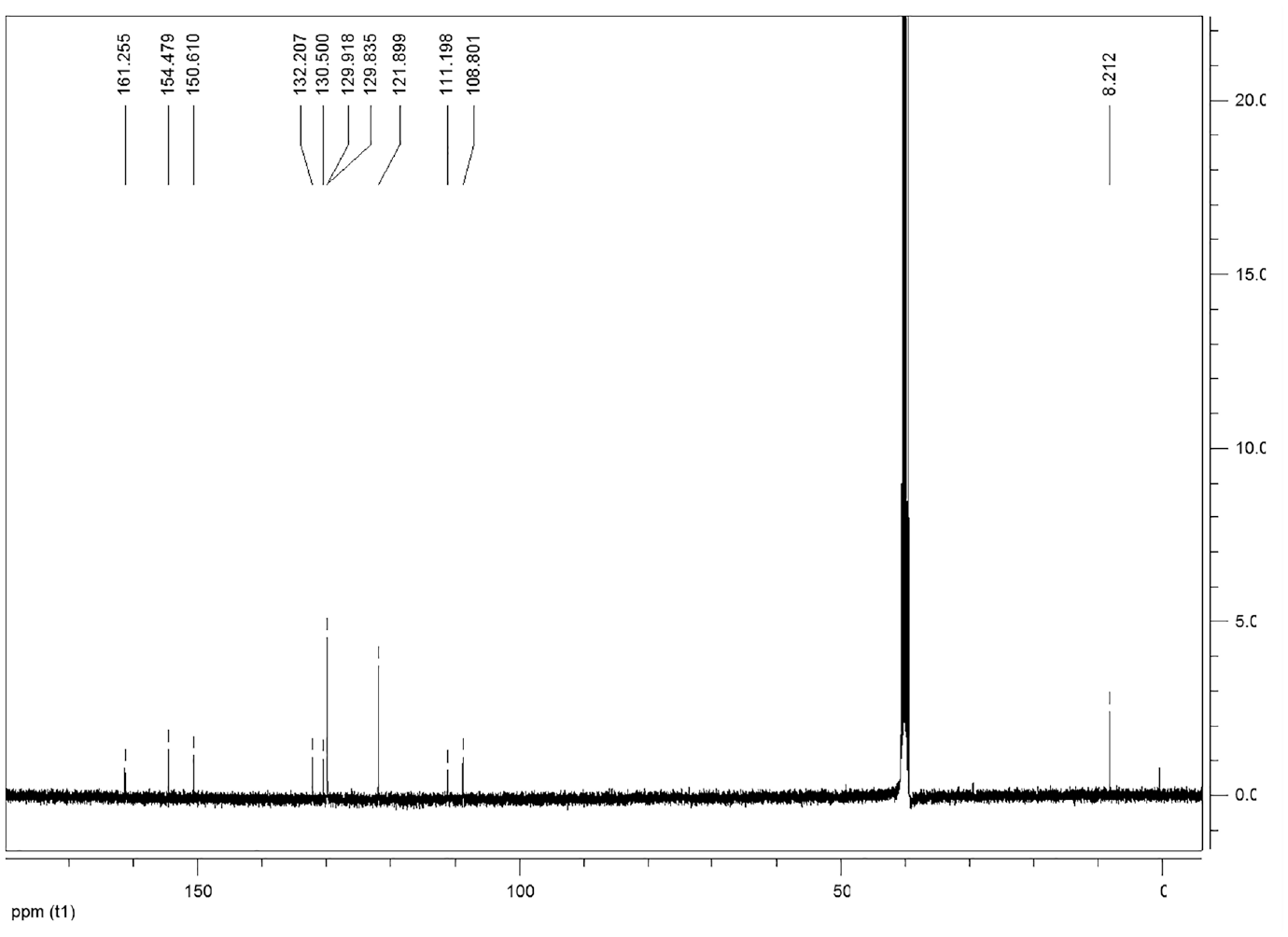
13 C NMR spectrum of compound 1.
The compound 2 signified the broad OH band in the FT-IR spectrum (Figure 4). The formation of compound 2 was clearly indicated by the appearance of two OH peaks at 12.53 and 10.63 ppm, aromatic peaks at 9.06–6.59 ppm, and an aliphatic CH3 peak at 2.02 ppm in its 1H NMR spectrum. The singlet peak at 12.53 ppm can be assigned to proton of OH group which is intramolecular hydrogen bonded with the nitrogen atom in the neighboring azo group (Scheme 2). The 13C NMR spectrum of compound 2 shows the presence of expected carbon atoms (Figure 5).
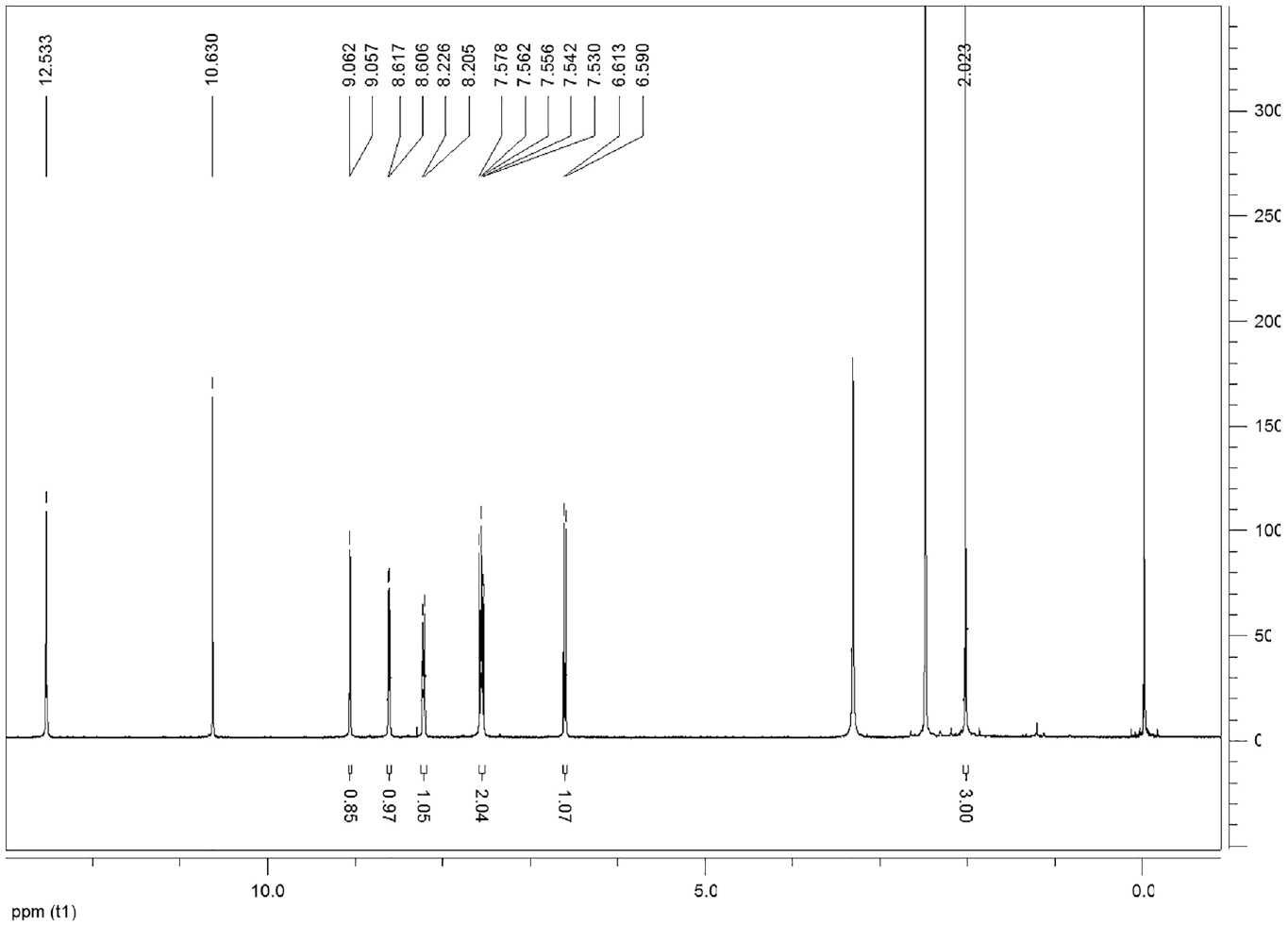
1 H NMR spectrum of compound 2.
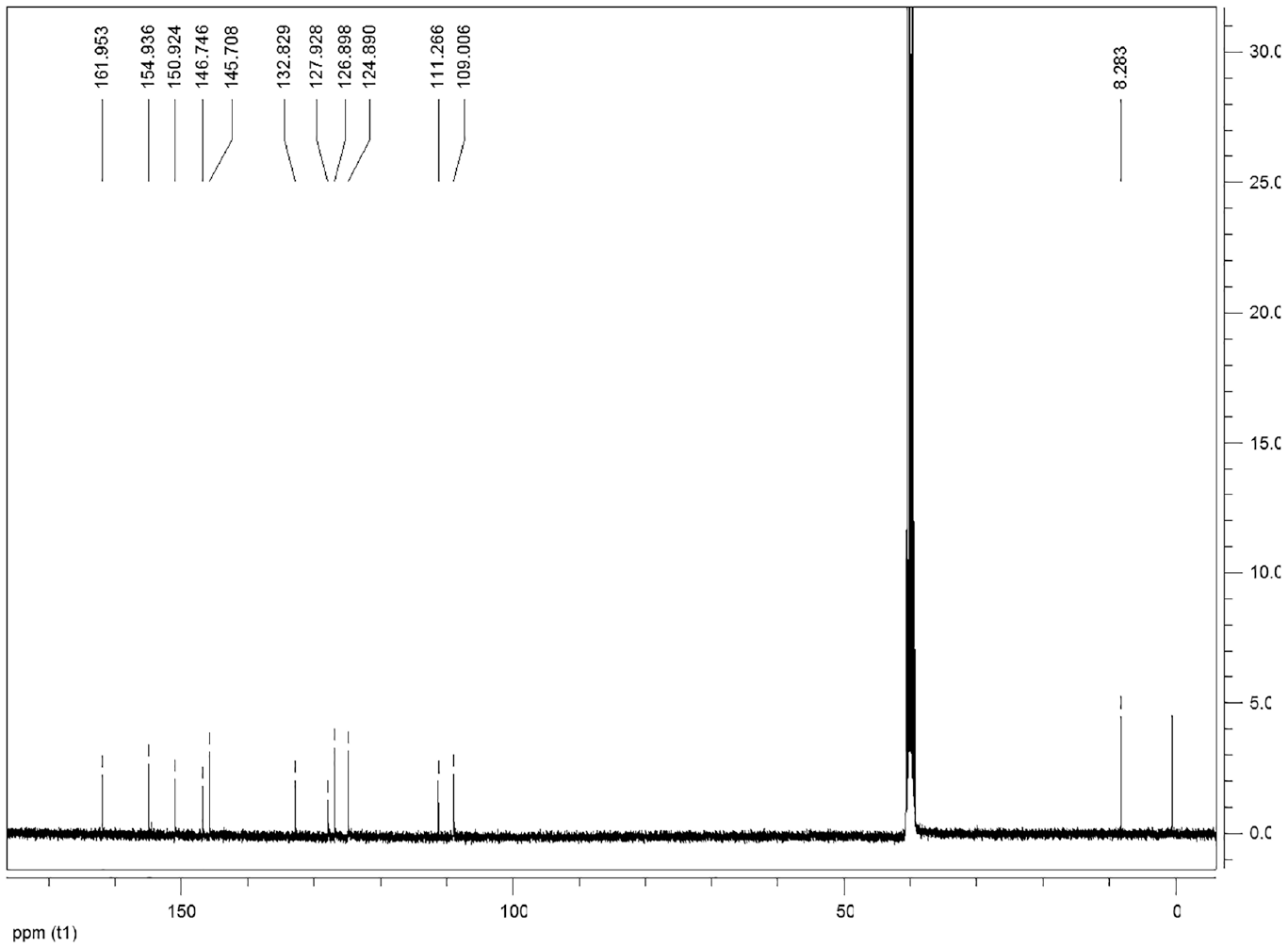
13 C NMR spectrum of compound 2.
Dyeing
Synthesized azo compounds are pigment forms and not soluble in water, but they are soluble in NaOH. In the exhaust dyeing process, first, the solubilization step in alkali media is needed (during 10 min, pH 10). Azo compounds (1, 2) become anionic in character in basic media by giving H+ ions to the medium. For more electrostatic interactions between the dye and the cotton at the main dyeing step (during 45 min), the cotton fabric was processed with cationic agent prior to the dye application. It is thought that their application methods are similar to those for direct dyes.
Colorfastness Assessments
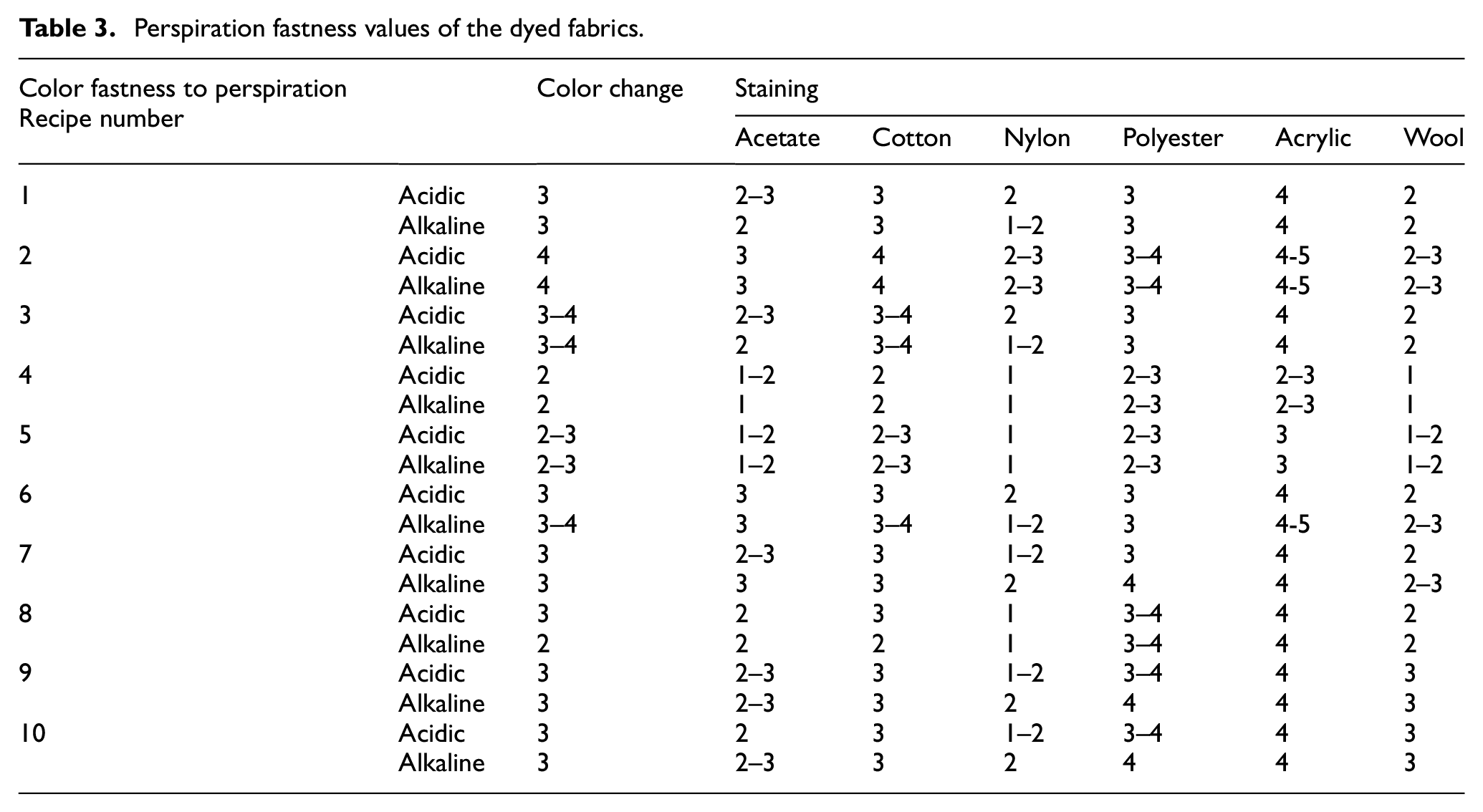
The color fastness values of the fabrics dyed with the azo compounds 1 and 2 are given in Tables 2–4 and Figures 6 and 7. The color fastness properties, except for light fastness, were measured on a scale of 1–5 (very poor, poor, fair, good, and very good). The dry rubbing fastness values of the fabrics dyed with compound 2 were highly commercially acceptable (grade 4–5, 5). The rubbing fastness (dry and wet) and washing fastness values of the fabric dyed with compound 2 were generally better than that of the fabric dyed with compound 1 (according to Figures 6 and 7, Table 2). The washing fastness results of the dyed fabrics were mostly moderate level (≥2–3 grade). When comparing the perspiration fastness values of the fabric samples treated with recipe numbers 4, 9, 5, and 10 (for 3% dye concentration with a 1:10 liquor ratio at 60°C and 80°C), the results for compound 2 were slightly higher than those for compound 1 (Table 3). The reason for the better fastness values of compound 2 could be the presence of nitrogen atoms in the benzene structure, unlike compound 1 (Scheme 1). This difference can ensure a more anionic character for compound 2 in an alkali dyeing bath and so better bonding can be achieved by more electrostatic interactions between the dye and cationic fabrics. The lightfastness is rated between 1 and 8 (very poor to outstanding) on wool (blue) scale. The light fastness values of compounds 1 and 2 were poor, being level with grade 2 (Table 4).
Washing fastness values of the dyed fabrics.
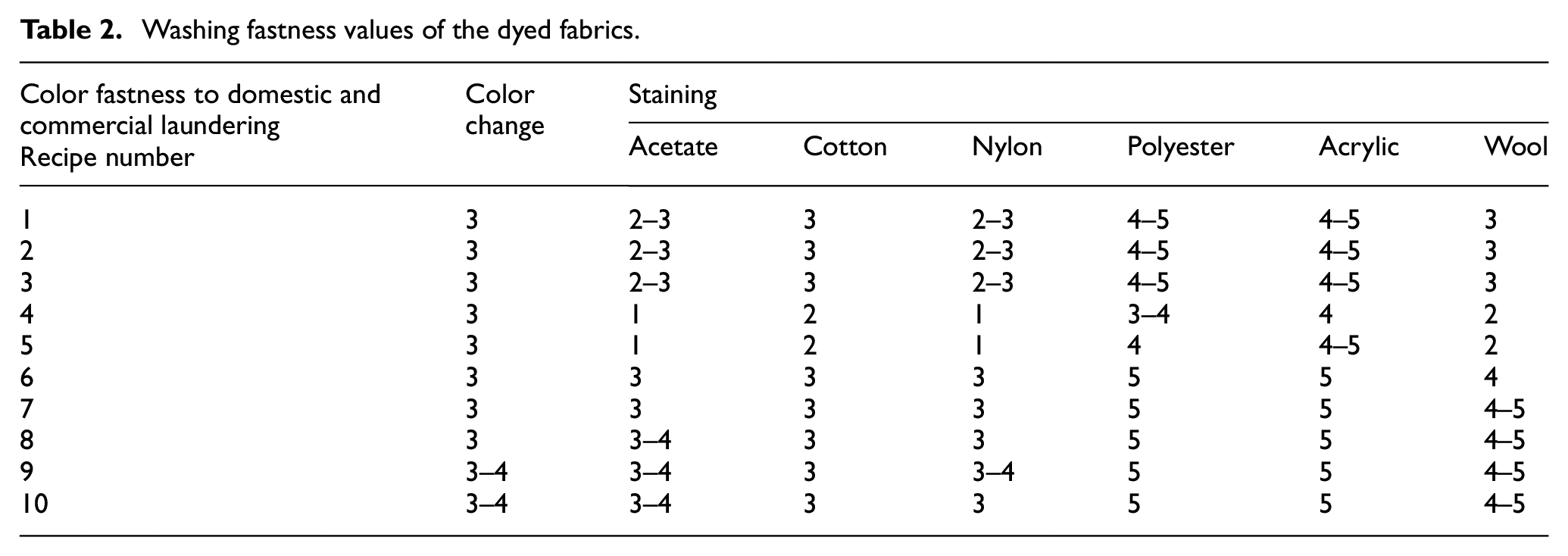
Perspiration fastness values of the dyed fabrics.
Light fastness values of compounds 1 and 2 on the dyed fabrics which treated with recipe numbers 4 and 9.

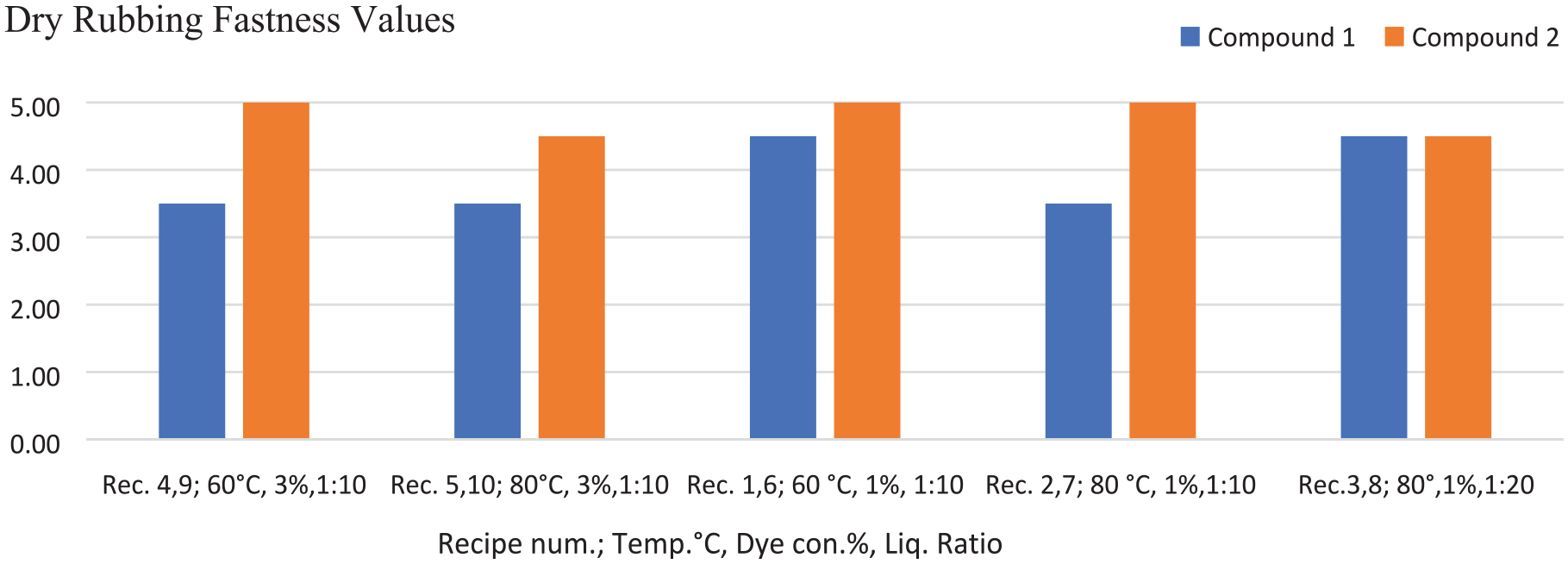
Comparison of dry rubbing fastness results of the cotton fabrics dyed with compounds 1 and 2.
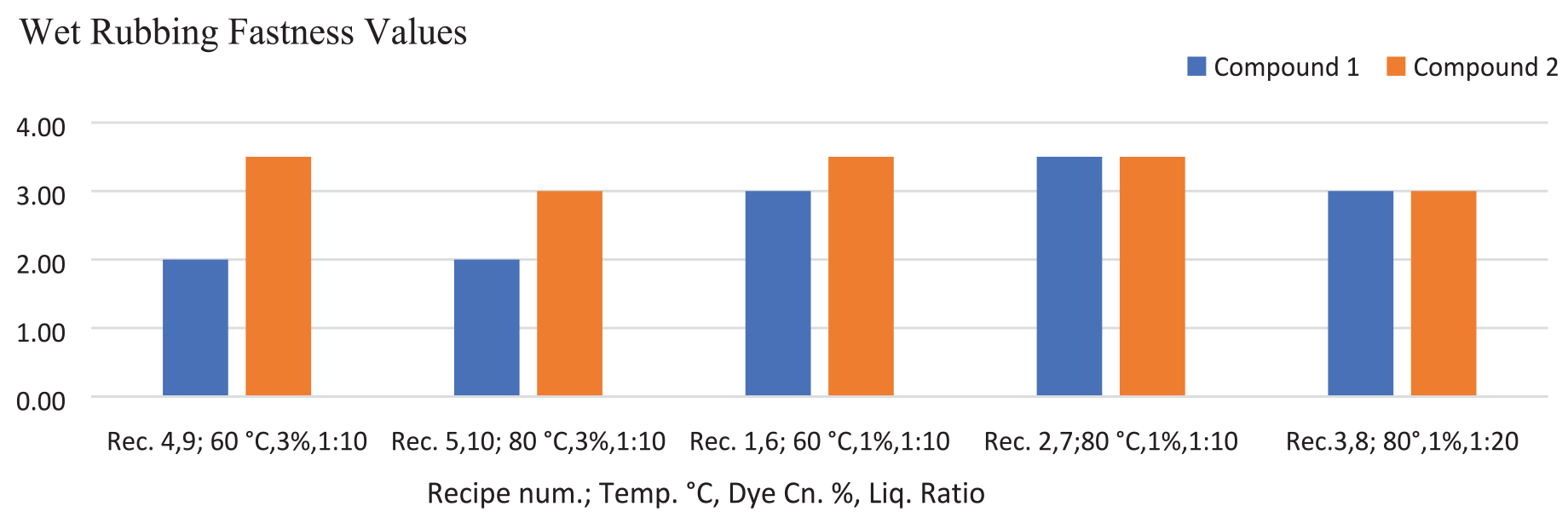
Comparison of wet rubbing fastness results of the dyed fabrics colored with compounds 1 and 2.
Color Strength
The results of the spectrophotometric measurements of the dyed fabrics are shown in Table 5. The dyed cotton samples exhibited uniformly different shades of orange color. The absorption maximum of compounds 1 and 2 on dyed fabrics (λmax) was found to be 370 nm. The fabrics dyed with compound 1 had a yellower color shade than the fabrics dyed with compound 2. Color shades changed with the change in the extent of delocalization of electrons depending on the dye structure. The presence of nitrogen atom in compound 2, the only difference between the compound 1 and 2 structures, may cause more delocalization and bathochromic shift. Bathochromic shifts make the light absorbed redder.
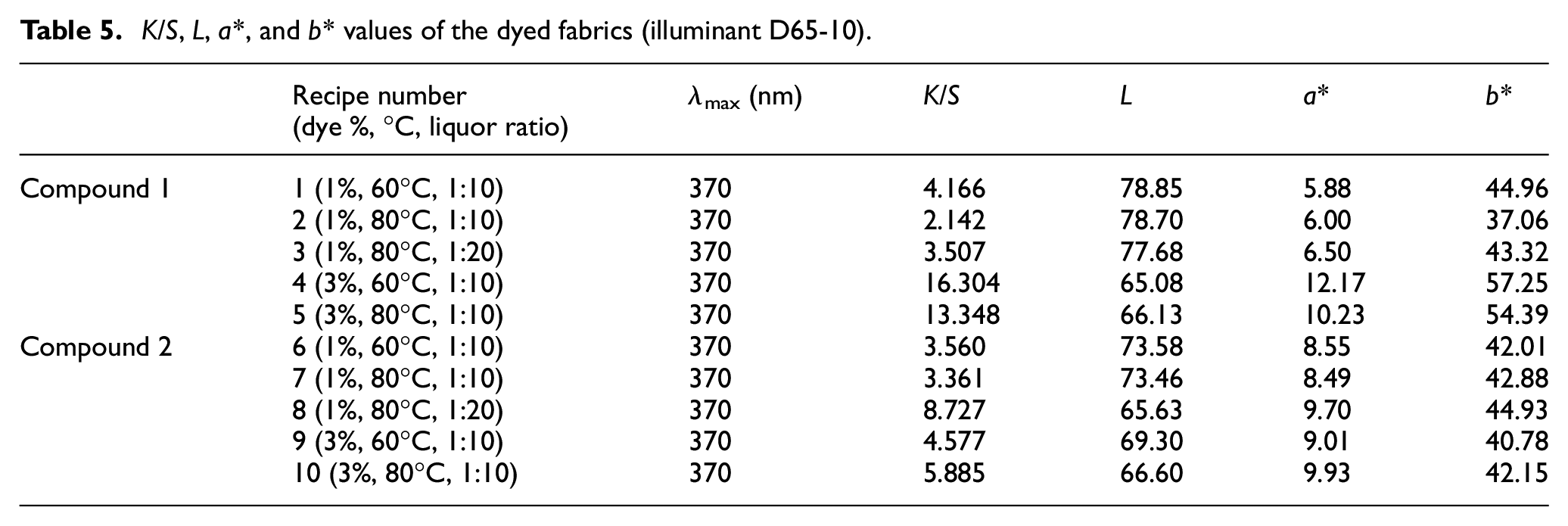
The
The
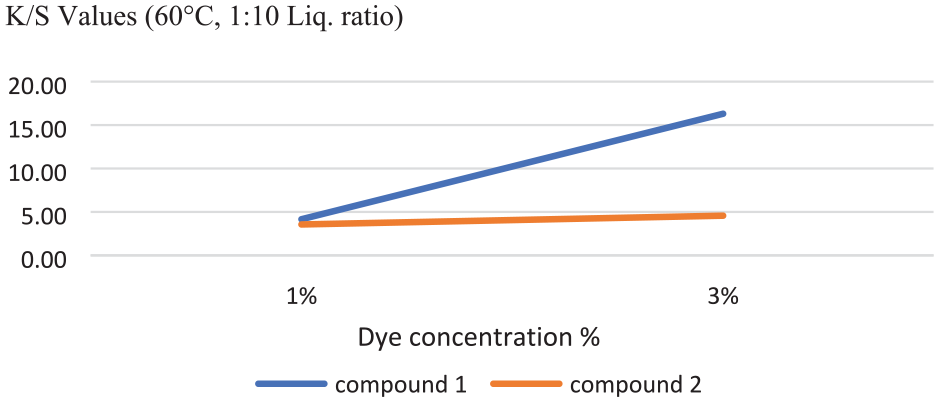
Concentration of dye solution versus
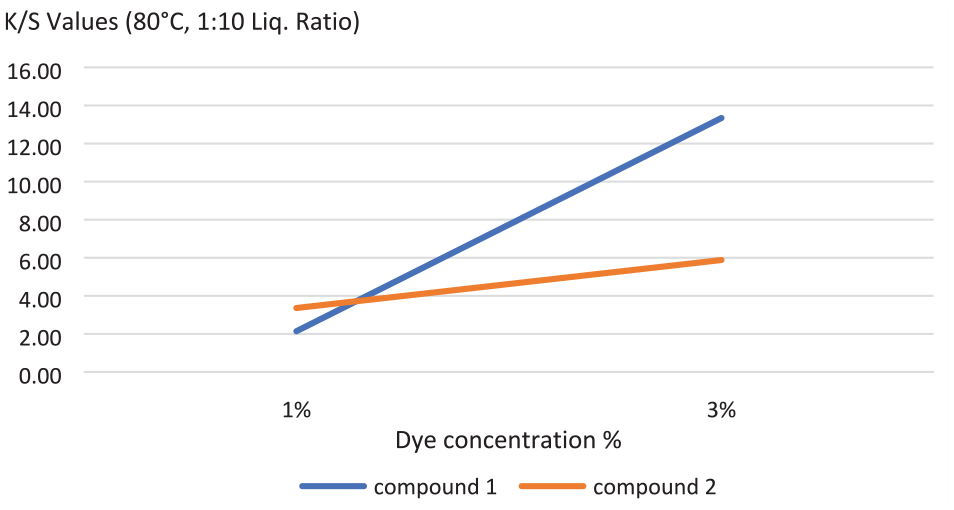
Concentration of dye solution versus
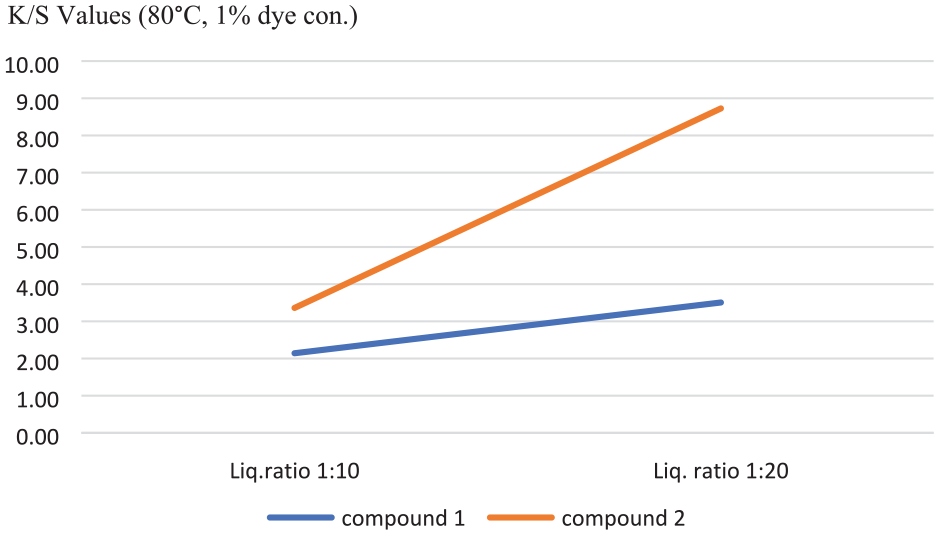
Antibacterial Activity Assessments
The antibacterial activity of the dyed cotton fabrics was tested according to Antimicrobial Activity Assessment of Textile Materials: Parallel Streak Method (AATCC 147-2016) against

where
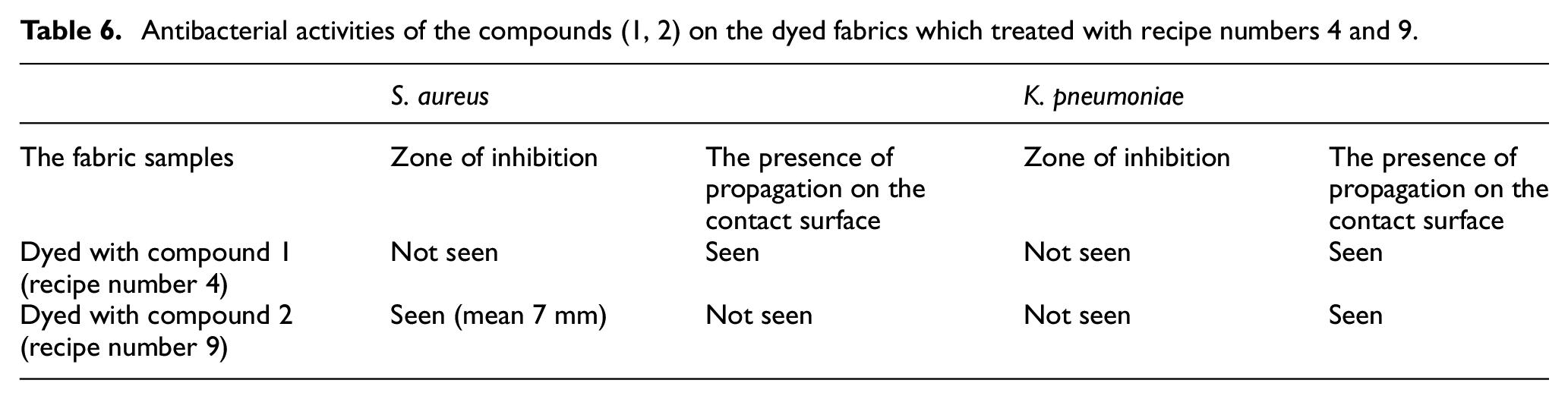
Antibacterial properties of the fabric samples treated with recipe numbers 4 and 9 (for 3% dye concentration with 1:10 liquor ratio at 60°C) are given in Table 6. Whereas the fabric dyed with compound 2 demonstrated acceptable antibacterial activity against
Antibacterial activities of the compounds (1, 2) on the dyed fabrics which treated with recipe numbers 4 and 9.

Agar plate images of the (a) untreated and (b) treated cotton fabric samples subjected to the parallel streak test (AATCC 147 Test Method) against
Conclusion
The potential use of novel azo compounds synthesized by the azo coupling reaction of aniline and 3-amino pyridine with 2-methyl resorcinol, respectively, as an antibacterial dyestuff in textile coloration was investigated. Antibacterial protection on textile material can be gained via only coloration without any additional finishing process. The color fastness features and antibacterial activities on the cotton fabric were studied. The fabric dyed with compound 2 demonstrated acceptable antibacterial activity against
In addition to coloration ability, other possible properties/uses of new azo compounds such as photo reaction sensitizers, acid–base indicators, electro-optical devices, and biological-medical studies might be worthwhile to study.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
