Abstract
Introduction:
Structural and functional abnormalities of coronary microvasculature, referred to as coronary microvascular dysfunction (CMD), have been implicated in a wide range of cardiovascular diseases and have gained growing attention in patients with chest pain with no obstructive coronary artery disease, especially in females. The central mechanisms of coronary vasomotion abnormalities encompass enhanced coronary vasoconstrictive reactivity (ie, coronary spasm), reduced endothelium-dependent and -independent coronary vasodilator capacities, and increased coronary microvascular resistance. The 2 major endothelium-derived relaxing factors, nitric oxide (NO) and endothelium-dependent hyperpolarization (EDH) factors, modulate vascular tone in a distinct vessel size–dependent manner; NO mainly mediates vasodilatation of relatively large, conduit vessels, while EDH factors in small resistance vessels. Endothelium-dependent hyperpolarization–mediated vasodilatation is more prominent in female resistance arteries, where estrogens exert beneficial effects on endothelium-dependent vasodilatation via multiple mechanisms. In the clinical settings, therapeutic approaches targeting NO are disappointing for the treatment of various cardiovascular diseases, where endothelial dysfunction and CMD are substantially involved.
Significance:
In this review, we will discuss the current knowledge on the pathophysiology and molecular mechanisms of endothelial function and coronary vasomotion abnormalities from bench to bedside, with a special reference to gender differences.
Results:
Recent experimental and clinical studies have demonstrated distinct gender differences in endothelial function and coronary vasomotion abnormalities with major clinical implications. Moreover, recent landmark clinical trials regarding the management of stable coronary artery disease have questioned the benefit of percutaneous coronary intervention, supporting the importance of the coronary microvascular physiology.
Conclusion:
Further characterization and a better understanding of the gender differences in basic vascular biology as well as those in cardiovascular diseases are indispensable to improve health care and patient outcomes in cardiovascular medicine.
Keywords
Introduction
Over the last 2 decades, we have seen a growing body of evidence that has revealed gender differences in coronary physiology and endothelial function in health and disease. Distinct gender differences have been identified in a wide range of cardiovascular diseases, such as heart failure, pulmonary hypertension, atherosclerotic vascular remodeling, spontaneous coronary artery dissection, and coronary functional abnormalities like vasospastic angina (VSA), as well as in the underlying risk factors. 1 -6 Recently, structural and functional abnormalities of coronary microvasculature, referred to as coronary microvascular dysfunction (CMD), have been implicated in a wide spectrum of cardiovascular diseases, including heart failure with preserved ejection fraction (HFpEF), a common and globally recognized form of heart failure that occurs more frequently in females. 7 Moreover, CMD has gained increasing attention in view of its unexpectedly high prevalence and significant prognostic impact in patients with chest pain regardless of the presence or absence of epicardial obstructive coronary artery disease (CAD), especially in females. 1,2,5,6,8 -10 The underlying mechanisms of CMD may be heterogeneous, including several structural and functional alterations. The central mechanisms of coronary vasomotion abnormalities are 3-fold: enhanced coronary vasoconstrictive reactivity (ie, coronary spasm) at epicardial and microvascular levels, reduced endothelium-dependent and -independent coronary vasodilator capacities, and increased coronary microvascular resistance, all of which can cause myocardial ischemia due to CMD and often coexist in various combinations even in the absence of obstructive CAD. 11 -13 The term “ischemia and no obstructive coronary artery disease (INOCA)” has been coined for this clinical condition 14 and is increasingly recognized as an important clinical entity, particularly in females. 15
Endothelial function has been recognized as an important surrogate of vascular risk. The endothelium plays a pivotal role in modulating vascular tone by synthesizing and releasing endothelium-derived relaxing factors (EDRFs), including vasodilator prostaglandins (PGs), nitric oxide (NO), and endothelium-dependent hyperpolarization (EDH) factors in a distinct vessel size–dependent manner; NO mainly mediates vasodilatation of relatively large, conduit vessels (eg, aorta and epicardial coronary arteries), while EDH factors in small resistance vessels (eg, arterioles and coronary microvessels; Figure 1). 16,17 Endothelium-dependent hyperpolarization–mediated vasodilatation is more predominant in female resistance arteries as compared with male counterparts. 17,18 In addition, estrogens have various vasoactive properties as discussed later and exert protective effects on endothelium-dependent vasodilatation via multiple mechanisms. 17,18
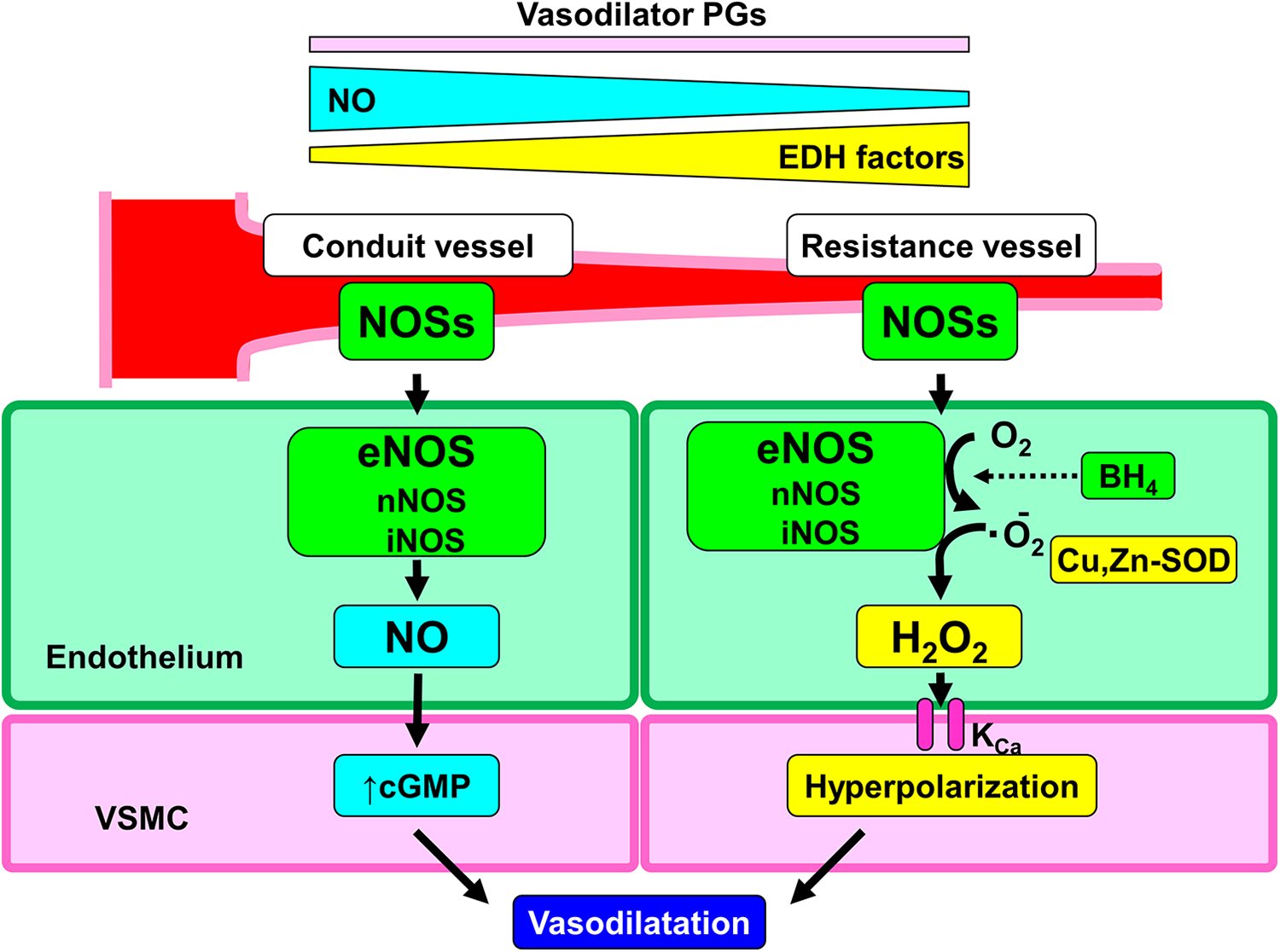
Vessel size–dependent roles of endothelial nitric oxide synthases system. BH4 indicates tetrahydrobiopterin; cGMP, cyclic GMP; Cu, Zn-SOD, copper-zinc superoxide dismutase; EDH, endothelium-dependent hyperpolarization; eNOS, endothelial nitric oxide synthase; H2O2, hydrogen peroxide; iNOS, inducible NOS; KCa, calcium-activated potassium channel; nNOS, neural NOS; NO, nitric oxide; NOSs, nitric oxide synthases; PGs, prostaglandins; VSMC, vascular smooth muscle cells.
In this review, we will give an outline of the pathophysiology and molecular mechanisms of endothelial function and coronary vasomotion abnormalities from bench to bedside with a special reference to gender differences.
Current Views on Ischemic CAD and Gender-Specific Considerations
Classically, atherosclerotic CAD was considered to be a predominantly male issue and the clinical impact of the disease burden in females is limited because previous randomized clinical trials of revascularization therapy exclusively enrolled male patients. A latest nationwide large-scale cohort study in the United States identified a total of 12 062 081 revascularization hospitalizations 19 ; among them, female patients not only remained underrepresented, merely accounting for one-third of the total cohort, but also their trend to undergo percutaneous coronary intervention (PCI) and coronary artery bypass grafting continued to decrease. 19 Moreover, as exemplified in the PROspective Multicentre Imaging Study for Evaluation of chest pain (PROMISE) trial, the prevalence of statin use was lower in female patients and they were less likely to be referred for coronary angiography compared with male counterparts. 20 Female patients with ischemic heart disease (IHD) are characterized by atypical anginal symptoms (eg, dyspnea, fatigue, and reduced activities of daily living), less prevalence of obstructive CAD on coronary angiography albeit suspected clinically, and lower rates of coronary revascularization. 21 Although there have been marked reductions in cardiovascular disease mortality in females over the last decade, 22 a recent large-scale retrospective cohort study showed that female patients have a higher mortality rate after PCI than male counterparts due to death from noncardiac causes, suggesting the need for gender-specific clinical care in the management of patients undergoing PCI. 23
The mechanisms underlying the gender differences in the characteristics of IHD appear to be multifactorial, including differences in sex hormone effects, autonomic regulation, and susceptibility to proatherogenic mediators, such as oxidative stress, endothelin-1, and angiotensin II. An autopsy study by Virmani et al of patients who died suddenly of CAD demonstrated that postmenopausal females more frequently have a ruptured plaque than do premenopausal counterparts, indicating that estrogens play protective roles against plaque destabilization through an anti-inflammatory effect on atherosclerotic plaques. 24 Considering that approximately 10 years are required for females to develop CAD to the same extent as males after menopause, 2 the aforementioned protective effects of endogenous estrogens against the development of coronary atherosclerosis might be protracted in the late postmenopausal period, leading to more advanced coronary plaque characteristics in postmenopausal females. Moreover, recently published gender-specific analyses of longitudinal blood pressure measures over 40 years revealed that compared with males females develop a steeper increase in blood pressure as early as in their 20s that persists through the life course. 25 This premature rise in blood pressure in females may affect the different presentation of various cardiovascular diseases between genders.
Previous studies focused on functional and structural abnormalities of epicardial coronary arteries; however, those of coronary microvasculature have recently gained increasing attention in many clinical settings. The Women’s Ischemic Syndrome Evaluation (WISE) study funded by the National Heart, Lung, and Blood Institute was conducted to facilitate gender-specific research on IHD, yielding significant insight into the clinical characteristics of the disorder in females. 1,2 One of the major findings of the WISE study was that the etiology of myocardial ischemia in female patients with chest pain who were found to have no obstructive CAD was attributed to the coronary microvasculature, and thus, the diagnostic evaluation of CMD and endothelial dysfunction is important using invasive or noninvasive coronary reactivity testing. Although the prevalence of CMD in this clinical entity has been shown to be not negligible in both genders, 10,11,26 the assessment and diagnosis of functional rather than structural abnormalities in the coronary circulation should be considered in light of the lower prevalence of obstructive CAD and higher prevalence of CMD in females. 27 In line with the findings from the WISE study, the results from the 2 landmark clinical trials regarding the management of stable CAD, the Objective Randomised Blinded Investigation with optimal medical Therapy of Angioplasty in stable angina (ORBITA) trial 28 and the International Study of Comparative Health Effectiveness with Medical and Invasive Approaches (ISCHEMIA) trial, 29 question the benefit of PCI and further support the importance of the coronary microvascular physiology, which PCI procedure could not improve. Although similar prognostic benefits of noninvasive computed tomography coronary angiography guided management for both female and male patients with suspected angina due to IHD, 30 a comprehensive invasive assessment of coronary physiology is feasible and of diagnostic value to detect patients with endothelium-dependent or -independent CMD (Figure 2). 11,26,31
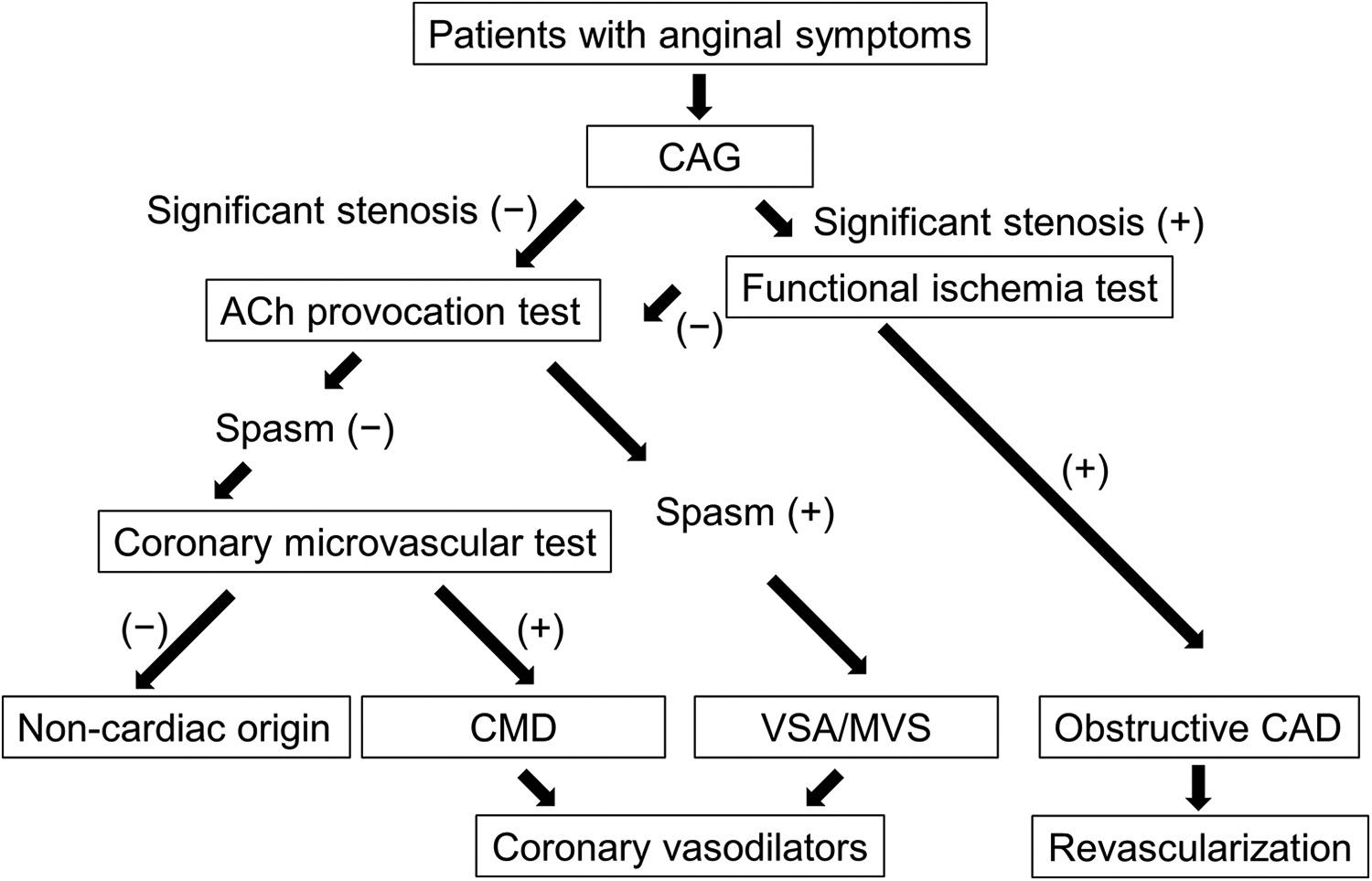
Comprehensive invasive assessment of coronary vasomotion abnormalities. ACh indicates acetylcholine; CAD, coronary artery disease; CAG, coronary angiography; CMD, coronary microvascular dysfunction; MVS, microvascular spasm; VSA, vasospastic angina.
The emerging pressure-wire-based indices, instantaneous wave-free ratio (iFR) and fractional flow reserve (FFR), are currently the gold standard in assessing whether a coronary stenosis can induce myocardial ischemia amenable to revascularization in patients with stable CAD. A post hoc analysis of the DEFINE-FLAIR (Functional Lesion Assessment of Intermediate stenosis to guide Revascularization) study aimed to evaluate gender differences in procedural characteristics and clinical outcomes of iFR- and FFR-guided revascularization strategies. 32 Female patients (n = 601) had a lower number of functionally significant lesions, a higher mean FFR value, and a comparable mean iFR value with a resultant lower rate of revascularization than male patients. 32 Although an FFR-guided strategy was associated with a higher rate of revascularization than iFR-guided strategy in males (n = 1891), but not in females, there were no differences between the 2 strategies in 1-year major adverse cardiac events in both genders. 32 These results suggest that a gender-specific cutoff value for FFR may be needed to better detect functionally significant lesions in female patients with obstructive CAD.
When assessed by reduced coronary flow reserve (CFR) and increased index of microcirculatory resistance (IMR), patients with CMD are associated with an increased risk of major adverse cardiovascular events and worse long-term outcomes. 10,13,33 Although coronary microvascular function as evaluated by IMR is similar between genders, 34,35 CFR has been shown to be lower in females because of a shorter resting mean transit time, an inverse correlate with absolute flow, and thus higher resting coronary flow than in males. 34,35 Notably, a prospective 5-year follow-up study in patients with deferred coronary artery lesions (n = 434) showed better long-term clinical outcomes in female patients compared with male counterparts. 35 These results again indicate that gender differences are important considerations when interpreting physiological indexes using resting coronary flow.
Gender Differences in Coronary Vasomotion Abnormalities
Vasospastic Angina and Coronary Microvascular Spasm
Along with endothelial dysfunction, endothelium-independent mechanisms represented by impaired coronary microvascular dilatation and enhanced coronary microvascular constriction can cause CMD. Coronary artery spasms at both epicardial and microvascular levels have been implicated in a wide variety of IHD. 16,36 The central mechanism in the pathogenesis of coronary artery spasm is Rho-kinase-induced myosin light chain phosphorylation with resultant vascular smooth muscle cells (VSMC) hypercontraction, whereas the role of endothelial dysfunction may be minimal. 16,37 Intracoronary administration of a Rho-kinase inhibitor, fasudil, is effective not only for relieving severe coronary artery spasm refractory to nitrates or calcium-channel blockers but also for suppressing coronary microvascular spasm in patients with VSA and coronary microvascular spasm, respectively. 38 -40 We have previously demonstrated that estrogens downregulate Rho-kinase, while nicotine cancels the inhibitory effect of estrogens on inflammatory stimuli-induced Rho-kinase expression, which explains in part the increasing incidence of vasospastic disorders in postmenopausal females and smokers. 41 In addition, enhanced epicardial and coronary microvascular spasms are associated with increased production of vasoconstrictive mediators, such as endothelin 42 and serotonin, 43 in patients with CMD. A potent vasoconstrictor peptide, endothelin-1, contributes to impaired coronary vasodilator responses toward CMD. Indeed, elevated plasma levels of endothelin-1 are associated with coronary microvascular endothelial dysfunction, as evaluated by the percentage fall in coronary vascular resistance after 10 minutes of rapid atrial pacing, in patients with chest pain and normal coronary arteriograms, in particular in females. 44
Coronary reactivity testing using intracoronary acetylcholine (ACh) provocation is useful in inducing coronary artery spasm with high sensitivity and specificity in the cardiac catheterization laboratory (Figure 2). A high prevalence of ACh-induced coronary microvascular spasm has been reported in one-third of patients with stable chest pain and nonobstructive CAD. 45,46 A consensus set of standardized diagnostic criteria for microvascular angina attributable to CMD including ACh-induced coronary microvascular spasm has been proposed by the Coronary Vasomotion Disorders International Study Group (COVADIS). 47 The diagnostic value of these criteria has been validated by a recent randomized clinical trial. 31 When performing ACh provocation test, it is important to note that gender differences exist in the prevalence of coronary vasomotion abnormalities and in the threshold dose of ACh required for a positive result; among patients with angina and nonobstructive CAD who undergo ACh provocation test, epicardial vasospasm and coronary microvascular spasm are more prevalent in female subjects with a higher sensitivity to ACh. 4,5
We have previously demonstrated the gender differences in the characteristics and outcomes of patients with VSA. 3,48 For example, gender is one of the significant prognostic factors in patients with VSA; the younger age (<50 years) is significantly associated with worse outcomes in females, but not in males. 3 This is also confirmed in Caucasian patients with VSA, using a prospective international multicenter cohort consisting of 1,339 Japanese patients and 118 Caucasian patients with the disease. 48
Endothelial Dysfunction
Endothelial Modulation of Vascular Tone: EDRFs
The endothelium plays a crucial role in modulating the tone of underlying VSMC by synthesizing and releasing EDRFs in an autocrine and paracrine manner. 16,17 These endothelial-derived mediators include vasodilator PGs (eg, prostacyclin [PGI2]), NO, and EDH factors, as well as endothelium-derived contracting factors (EDCFs) 16,17 (Figure 1). Endothelial dysfunction is characterized by reduced production and/or action of EDRFs, serving not only as the hallmark of atherosclerotic cardiovascular diseases but also as one of the major pathogenetic mechanisms of CMD. 49 -51
Vessel Size-Dependent Contribution of NO and EDH Factors
Endothelium-derived NO and EDH factors, regardless of sex or species, modulate vascular tone in a distinct vessel size–dependent fashion, that is, endothelium-derived NO predominantly regulates vasodilatation of relatively large, conduit vessels (eg, aorta and epicardial coronary arteries), while EDH factor-mediated responses are the major mechanisms of endothelium-dependent vasodilatation of resistance arteries (eg, arterioles and coronary microvessels; Figure 1). 16,52,53 Although NO is one of the important mediators of microvascular flow-mediated dilation (FMD) among various EDRFs, including the products of cyclooxygenase, 54 and EDH factors such as hydrogen peroxide, 55 it has been widely accepted that EDH-mediated responses rather than NO are the predominant mechanism of endothelium-dependent vasodilatation in resistance arteries. For example, the predominant effect of intracoronary nitroglycerin (an NO donor) is on the epicardial coronary arteries with only limited effects on the coronary microcirculation and resistance vessels. 56 Thus, EDH factor-mediated vasodilatation is a vital mechanism especially in microcirculations, where blood pressure and organ perfusion are determined in response to demand fluctuation in the body. On the other hand, vasodilator PGs in general play a small but constant role, irrespective of vessel size. This vessel size–dependent contribution of NO and EDH factors in endothelium-dependent vasodilatation is well preserved from rodents to humans, maintaining a physiological balance between them. 16,17 Moreover, such redundant mechanisms in endothelium-dependent vasodilatation are advantageous for the proper maintenance of vascular tone and endothelial function under pathological conditions, where one of the EDRF-mediated responses is hampered in favor of a vasoconstrictor, procoagulant, proliferative, and pro-inflammatory state. Indeed, in various pathological conditions with atherosclerotic risk factors, NO-mediated relaxations are easily impaired, while EDH factor-mediated responses are fairly preserved or even enhanced to serve as a compensatory vasodilator system. 53,57 Multifaceted mechanisms are involved in the enhanced EDH factor-mediated responses in small resistance vessels, including negative interactions between NO and several EDH factors. 58 Refer to extensive reviews for more detailed information on the regulatory mechanisms of NO-mediated responses. 59 -61
Endothelium-Dependent Hyperpolarization Factors: The Predominant Mechanism of Vasodilatation in Small Arteries
In 1988, Feletou and Vanhoutte 62 and Chen et al 63 independently demonstrated the existence of endothelium-derived, non-NO, nonprostanoid relaxing mediators, unforeseen EDH factors. By definition, the contribution of EDH factors is determined only after the blockade of both vasodilator PGs and NO. The EDH factors cause hyperpolarization and subsequent relaxation of underlying VSMC with resultant vasodilatation of small resistance vessels, finely regulating blood pressure and organ perfusion instantaneously to meet changing physiological demands in vivo. 17 The nature of EDH factors varies depending on the vascular bed, vessel size, and species, including epoxyeicosatrienoic acids (EETs), metabolites of arachidonic P450 epoxygenase pathway, 64,65 electrical communication through gap junctions, 66 potassium ions, 67 and, as we demonstrated, endothelium-derived hydrogen peroxide (H2O2) 68 (Figure 3). Several EDH factors regulate vascular tone in the coronary circulation. For instance, EETs take part in EDH-mediated relaxations in bovine, 64 porcine, 65 and human coronary arteries 69 ; potassium ions in porcine 70 and bovine 71 coronary arteries; and H2O2, at physiologically low concentrations, in human, 72 porcine, 73 and canine coronary arteries. 74 -76 Coronary vascular resistance is mostly determined by the prearterioles (>100 µm in diameter) and arterioles (<100 µm), where EDH factor-mediated responses become more prominent than NO-mediated relaxations. Considering that H2O2 has potent vasodilator properties in coronary resistance vessels, impaired H2O2-mediated vasodilatation may lead to CMD. Further comprehensive information on the role of EDH factors is available elsewhere. 77,78
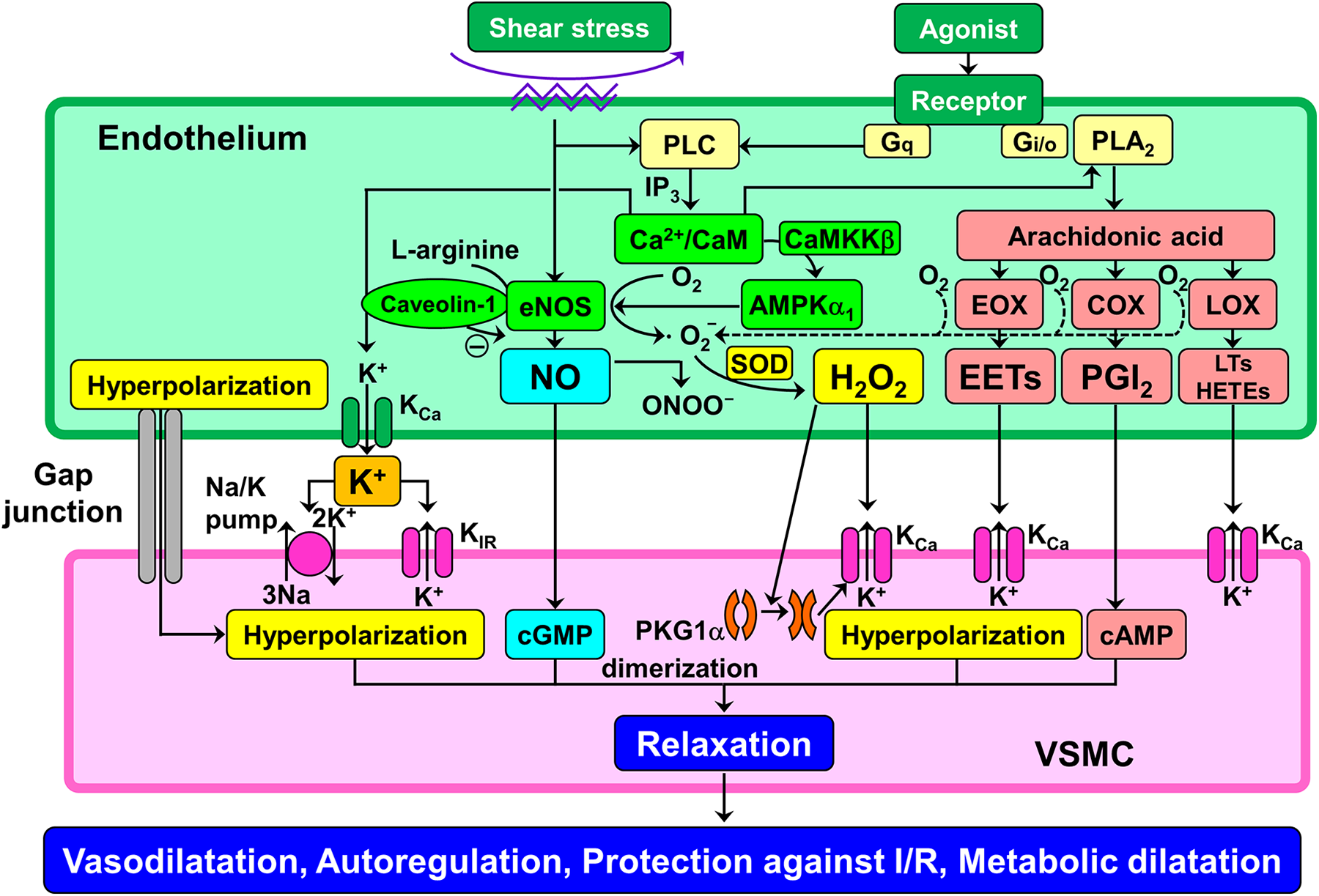
Endothelium-dependent hyperpolarization factors AMPKα1 indicates α1-subunit of AMP-activated protein kinase; CaM, calmodulin; CaMKKβ, Ca2+/CaM-dependent protein kinase β; cAMP, cyclic AMP; cGMP, cyclic GMP; COX, cyclooxygenase; EETs, epoxyeicosatrienoic acids; eNOS, endothelial NO synthase; EOX, epoxygenase; HETEs, hydroxyeicosatetraenoic acids; H2O2, hydrogen peroxide; IP3, inositol trisphosphate; I/R, ischemia–reperfusion injury; KCa, calcium-activated potassium channel; KIR, inwardly rectifying potassium channel; LOX, lipoxygenase; LTs, leukotrienes; NO, nitric oxide; ONOO−, peroxynitrite; PGI2, prostacyclin; PKG1α, 1α-subunit of protein kinase G; PLA2, phospholipase A2; PLC, phospholipase C; SOD, superoxide dismutase.
Although EDH factor-mediated responses are the major mechanism of endothelium-dependent vasodilatation in resistance arteries, EDH-mediated vasodilatation is more predominant in female resistance arteries. 79 -86 Mechanistically, a major female sex hormone, 17β-estradiol, enhances the activity of intermediate conductance calcium-activated potassium channels in endothelial cells and that of the senescence-associated proteins, silent information regulator T1, and adenosine monophosphate-activated protein kinase (AMPK) to augment EDH-mediated responses. 84,85,87,88 An indirect activator of AMPK, metformin, improves endothelial dysfunction as evaluated by digital reactive hyperemia index (RHI) in patients with polycystic ovary syndrome. 89
Effects of Estrogens on Endothelium-Dependent Vasodilatation
Endothelium-dependent vasodilatation is more pronounced in arteries from premenopausal female than in male animals and humans, while endothelial dysfunction is less prominent in premenopausal females than in males and postmenopausal females. 90,91 Multiple mechanisms are involved in the protective effects of estrogens on endothelium-dependent vasodilatation. For example, endothelial production and release of NO can be augmented by estrogens in a pleiotropic manner. 17,18,92 -94 Oral administration of tetrahydrobiopterin, an essential cofactor of NOS and a scavenger of oxygen-derived free radicals, improves FMD in estrogen-deficient postmenopausal females but has no effect in premenopausal females. 91 Activation of endothelial estrogen receptor α by estrogens also enhances the production of PGI2, 95 as well as EDH-mediated responses. 81,85,86 Estrogens also reduce the production of oxidative stress 96 and an endogenous inhibitor of endothelial nitric oxide synthase (eNOS), asymmetric dimethylarginine, 97 with a resultant increase in NO bioavailability. Moreover, estrogens reduce the production of EDCFs by endothelial cyclooxygenase-1 and vascular smooth muscle thromboxane receptors. Furthermore, estradiol induces subcellular translocation of eNOS to stimulate NO production in vascular endothelium, 98 suggesting a possible mechanism for postmenopausal endothelial dysfunction. Taken together, it is highly possible that these beneficial effects of estrogens on endothelial function as well as the braking effect of them on EDCF-mediated responses help to protect premenopausal females against the development of atherosclerotic cardiovascular diseases.
Clinical Implications and Therapeutic Approaches
Endothelial Function as a Surrogate of Vascular Risk
The assessment of endothelial function has been utilized as an excellent surrogate marker of future cardiovascular events in many clinical settings. 99 Endothelial dysfunction is manifested as reduced production and/or action of EDRFs. Although EDH factor-mediated vasodilation can be temporarily enhanced to compensate for impaired NO-mediated responses in the early stage of atherosclerotic conditions, 100 after prolonged exposure to atherosclerotic risk factors, this compensatory role of EDH factor-mediated responses is finally disrupted to cause metabolic disturbance. 101 Endothelial dysfunction, as determined by impaired FMD of the brachial artery or digital RHI in peripheral arterial tonometry, is associated with future cardiovascular events in patients with CAD and 1 standard deviation decrease in FMD or RHI is associated with doubling of cardiovascular event risk. 99 Moreover, patients with coronary vasomotion abnormalities are often complicated with peripheral endothelial dysfunction, where CMD manifests as systemic vascular dysfunction beyond the heart. 102,103 It may be speculated that female patients may benefit more from early aggressive medical management aimed at improving endothelial function and risk factors upon detection of endothelial dysfunction.
Role of H2O2 as an EDH Factor in the Pathophysiology of CAD
As discussed above, previous studies focused on structural and functional abnormalities of “epicardial” coronary arteries in patients with CAD because they are easily visible on coronary angiography and amenable to PCI procedure. However, CMD has attracted increasing attention as a novel therapeutic and research target in patients with IHD. It is conceivable that impaired H2O2/EDH factor-mediated vasodilatation is involved in the pathogenesis of CMD, given its potent vasodilator properties in coronary resistance vessels where EDH factor-mediated responses surpass NO-mediated relaxations. We have recently demonstrated that CMD caused by impaired H2O2/EDH factor is associated with cardiac diastolic dysfunction in eNOS-knockout mice. 104 It seems essential to maintain the physiological balance between NO and H2O2/EDH factor for the treatment of CAD because significant negative interactions exist between NO and several EDH factors 105,106 and nitrates as NO donors are not effective for the treatment of CMD. 107,108 Moreover, endothelium-dependent CMD is associated with low endothelial shear stress, larger plaque burden, and vulnerable plaque characteristic beyond conventional coronary risk factors in patients with INOCA. 109,110
Lessons From Clinical Trials Targeting NO: Too Much NO to Relax?
Although standard medications for the treatment of CAD share the pleiotropic effects on endothelial function by enhancing NO-mediated vasodilatation with modest antioxidant capacities, including angiotensin-converting enzyme inhibitors, 111,112 angiotensin II receptor blockers, 113 and statins, 112 the effects of isosorbide-5-mononitrate are unexpectedly neutral in patients with residual microvascular ischemia despite successful PCI. 114 Likewise, despite the high prevalence and pathophysiological relevance of CMD in patients with HFpEF, the results of systemic and long-term administrations of inorganic nitrite for those patients are neutral or even harmful in randomized clinical trials. 115,116 These lines of evidence suggest that it is important to turn our attention to avoid excessive NO supplementation. Although tachyphylaxis may be one of the reasons why nitrite derivatives are not effective vasodilators in the treatment of various cardiovascular diseases, an alternative explanation for such “paradox” of NO-targeted therapy may be nitrosative stress induced by an excessive amount of supplemental NO, 117,118 again suggesting the importance of physiological balance between NO and EDH factors in endothelium-dependent vasodilatation. Further research is needed to address how to modulate CMD to improve clinical outcomes of patients with the disorder and whether decision-making under consideration of gender-specific characteristics in the coronary physiology and endothelial function benefits them.
Summary and Clinical Perspectives
In this review, we highlighted the pathophysiology and molecular mechanisms of endothelial function and coronary vasomotion abnormalities with a special reference to gender differences. Despite the high prevalence of CMD in patients with INOCA, those patients are often underestimated and offered no specific treatment or follow-up under the umbrella of “normal” coronary arteries, especially in females. On the contrary to this otherwise common practice, patients with CMD are predisposed to future coronary events and associated worse outcomes. 33,119 Identifying CMD in this population may provide physicians with useful information for decision-making and risk stratification beyond conventional coronary risk factors. Given that “every cell has a sex” 120 and hormonal status changes throughout life, consideration of gender differences should be implemented into both basic research and clinical practice in order to improve health care and patient outcomes; a good example of this practice is available elsewhere. 121 A horizontally based gender-specific therapeutic strategy, as opposed to a vertical, hierarchical structure of the medical care for each organ, is indispensable and specialists in each field should bear the responsibility to account for sex as a biological variable to this end. In conclusion, further characterization and a better understanding of the gender differences in basic vascular biology as well as those in the pathophysiology, clinical presentation, and clinical outcomes of IHD can be an important gateway to precision medicine in cardiovascular diseases.
Footnotes
Acknowledgments
We appreciate the effort of the members of the Tohoku University Hospital Cardiac Catheterization Laboratory.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article This work was supported, in part, by the Grant-in-Aid for Scientific Research from the Ministry of Education, Culture, Sports, Science, and Technology, Tokyo, Japan (16K19383 and 17K15983).
