Abstract
Inhibition of nitric oxide (NO) synthesis induces vasoconstriction and reduction of the blood flow in the brain, indicating that basal release of NO provides a resting vasore-laxant tone in the cerebral circulation. In the present study, the contractile effect of the NO synthase blocker NG-nitro-L-arginine (100 μmol/L) in isolated rat middle cerebral arteries was attenuated markedly in the presence of the cyclooxygenase inhibitor indomethacin (5 μmol/L), the thromboxane A2 synthase inhibitor ridogrel (10 μmol/L), or the thromboxane receptor antagonist ICI 192605 (100 μmol/L). These results indicate that removal of the endogenous NO stimulates the release of thromboxane A2 in cerebral vessels and basal NO production regulates the resting cerebrovascular tone, at least in part, by suppressing thromboxane A2.
Inhibition of the L-arginine-nitric oxide (NO) pathway results in vasoconstriction and reduction of the blood flow in the brain, indicating that the basal release of NO is an important regulator of the cerebral circulation, providing a resting relaxant tone in the cerebrovascular bed (Faraci and Brian, 1994; Iadecola et al., 1994). This effect of basal NO production has been regarded generally as a direct relaxant action mediated by the activation of the soluble guanylyl cyclase in vascular smooth-muscle cells. However, recently it has been shown that, in contrast to the marked contractile effect of the NO synthase (NOS) inhibitor NG-nitro-L-arginine methyl ester, 1H-[1,2,4]oxadiazole[4,3-a]quinoxalin-1-one (ODQ), a potent and selective inhibitor of the soluble guanylyl cyclase, does not influence the tone of rat cerebral arteries (Doughty and Langton, 1997). The possibility that NO may regulate the resting cerebrovascular tone by modulation of the prostanoid synthesis has not been investigated before.
Nitric oxide reportedly stimulates the release of prostaglandins (Salvemini, 1997). A similar interaction in the regulation of the resting cerebrovascular tone is very unlikely, because inhibition of prostanoid synthesis by indomethacin does not mimic the contractile effect of NOS blockade. L-arginine analogue NOS inhibitors recently have been shown to increase the release of thromboxane A2 under in vivo and in vitro experimental conditions, suggesting an inhibitory effect of endogenous NO on thromboxane production (Stadler et al., 1993; Hellyer et al., 1997). A similar mechanism may also be involved in the maintenance of the NO-related resting cerebrovascular dilatory tone. The cerebrovascular constriction that develops after inhibition of the NO synthesis would then be a consequence of disinhibition of thromboxane release. To test our hypothesis, we investigated whether blockade of the biosynthesis or action of thromboxane A2 attenuates the contractile effect of the NOS inhibitor NG-nitro-L-arginine (L-NA) in rat middle cerebral arteries (MCAs).
METHODS
Adult male Wistar rats were ex sanguinated rapidly under deep ether anesthesia. Ring segments of the MCA were prepared for measurement of isometric force as described previously (Görlach and Wahl, 1996). Special care was taken to preserve the endothelium during preparation of the artery. The segments were transferred into 5-mL organ baths filled with a modified Krebs solution of the following composition (mmol/L): NaCl, 119; KCl, 4.6; NaH2PO4, 1.2; CaCl2·2H2O, 1.5; MgCl2·6H2O, 1.2; NaHCO3, 15; and glucose, 10. The bath solution was bubbled continuously with a humidified gas mixture (90% O2—10% CO2). The MCA segments were mounted on two L-shaped tungsten wires (50 μm diameter) one of which was fixed to the bath whereas the second one was connected to a force transducer for measuring isometric force. The vessels were allowed a 90-minute equilibration period, during which the resting tension was adjusted to 1.5 mN. Thereafter each segment was exposed to a 124-mmol/L K+ Krebs solution to elicit a reference contraction. After a 60-minute resting period, the functional integrity of the endothelium was tested by application of bradykinin (0.01–10 μmol/L, Calbiochem, Bad Soden, Germany) after precontraction induced by 100 μmol/L UTP (Serva, Heidelberg, Germany). Segments that did not show at least 20% relaxation of the precontraction were excluded from the study. The dilator effect of 1 μmol/L bradykinin in endothelium-intact vessels was 36.5 ± 17.8% of the UTP precontraction, which was 1.12 ± 0.28 mN. After a 60-minute resting period, the effect of 100 μmol/L L-NA (Sigma [Deisenhofen, Germany] dissolved in saline) on the resting tone of the vessel rings was determined 15 minutes after its application in the absence or presence of the cyclooxygenase inhibitor indomethacin (5 μmol/L, Sigma), the thromboxane A2 synthase inhibitor ridogrel ((E)-5-[[[3-pyridinil[3-(trifluoromethyl)phenyl]methylen]amino]oxy]pentanoic acid, 10 μmol/L, Janssen, Neuss, Germany), the thromboxane receptor antagonist ICI 192605 (4-(Z)-6-(2-o-chlorophenyl-4-o-hydroxyphenyl-1,3-dioxan-cis-5-yl)hexenoic acid, 100 μmol/L, Tocris, Langford, Bristol, U.K.), or their vehicles. The latter drugs, or their vehicles, were applied 12 minutes before the administration of L-NA. L-NA was used in a concentration (100 μmol/L) that completely abolishes the endothelium-dependent dilator effect of bradykinin in rat MCAs (Görlach and Wahl, 1996). Indomethacin and ridogrel were dissolved in 2% Na2CO3 and 400 mmol/L NaOH, respectively and diluted with saline. ICI 192605 was dissolved in DMSO (Aldrich, Milwaukee, WI, U.S.A.). Ridogrel is described as having both thromboxane A2 synthase inhibitor activity and antagonistic activity at the common receptor for thromboxane A2 and its precursor prostaglandin H2 (De Clerck et al., 1989). However, in the present experiments—according to the study of Descombes et al. (1993)—ridogrel is considered to act as a thromboxane A2 synthase inhibitor, because the concentration of ridogrel selected (10 μmol/L) is lower than its IC50 value at thromboxane receptors (De Clerck et al., 1989). Nevertheless, we can not exclude the possibility that ridogrel also antagonizes the action of prostaglandin H2, and therefore findings in the present study relating to the release of thromboxane A2 also refer to prostaglandin H2.
All values in the text and figure are presented as the mean (± SD) percentage of the reference contraction induced by 124 mmol/L K+ Krebs solution. The contractile effect of 124 mmol/L K+ Krebs solution in absolute terms was 2.14 ± 0.70 mN. The number of MCA segments studied is expressed by n. Results were analyzed statistically using either Student's t-test or one-way analysis of variance followed by Fisher's protected least-significant difference test. A P value less than 0.05 was considered significant.
RESULTS AND DISCUSSION
The vehicles for indomethacin, ridogrel, or ICI 192605 had no significant effect on the resting tone of the MCA segments (−0.3 ± 1.7%, −0.1 ± 0.3% and −0.2 ± 0.8%, respectively, n = 11–19). In contrast, a weak, but significant (P < 0.005), reduction of the vascular tension was observed after administration of indomethacin (−2.7 ± 3.6%, n = 21), ridogrel (−1.1 ± 1.0%, n = 22), or ICI 192605 (−3.4 ± 3.0%, n = 20). The contractile effect of L-NA was not altered in vehicle-treated segments when compared with untreated controls (Fig. 1). However, treatment with indomethacin, ridogrel, or ICI 192605, markedly attenuated the L-NA-induced contractions (Fig. 1).
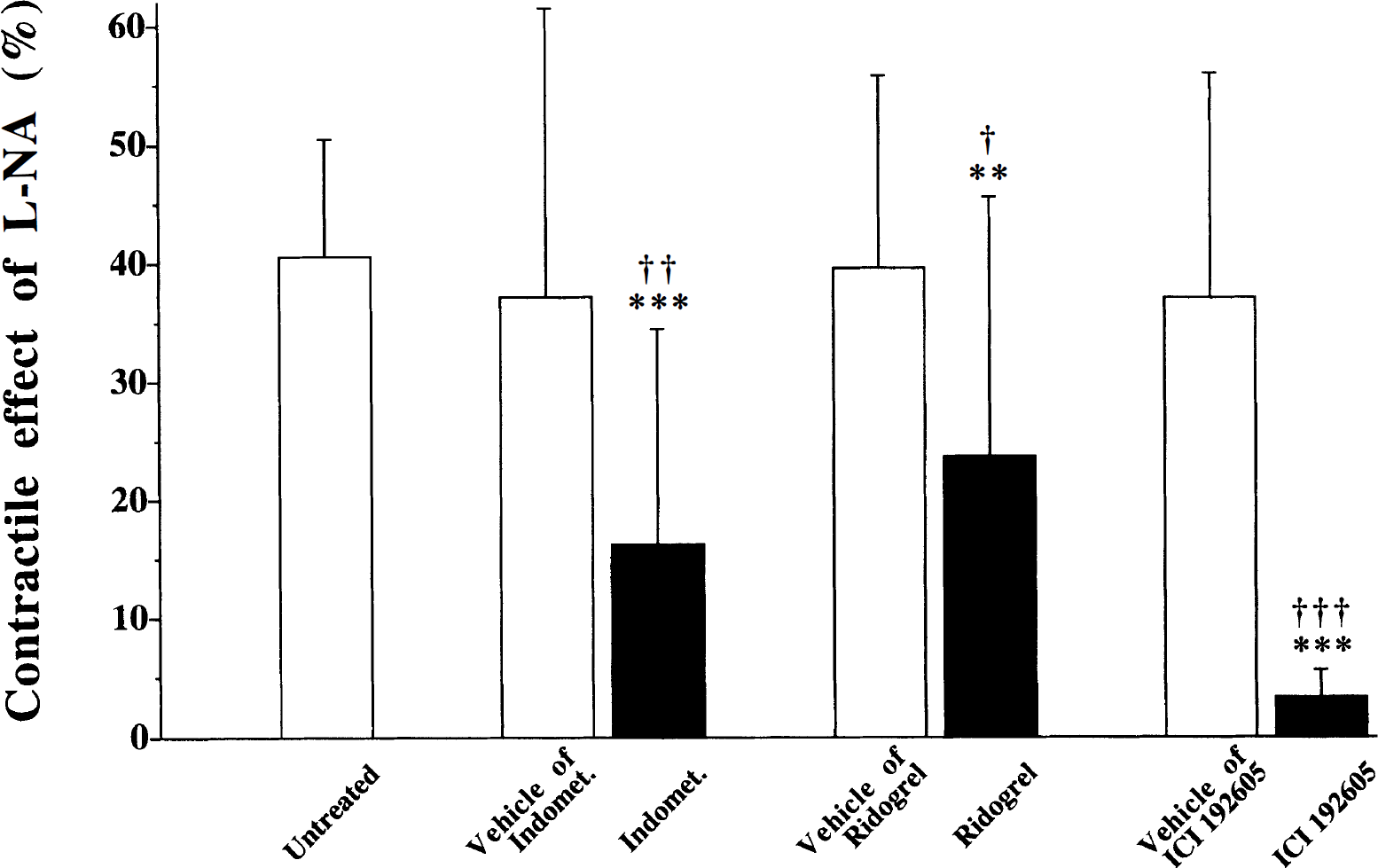
Contractile effect of 100 μmol/L L-NA in rat middle cerebral artery segments. The values (mean ± SD) are expressed as percentage of the reference contraction induced by 124 mmol/L K+ Krebs solution, n = 11–22. There was no significant difference between untreated and vehicle-treated vessels. However, in the presence of indomethacin (5 μmol/L), ridogrel (10 μmol/L), or ICI 192605 (100 μmol/L), the contractile effect of L-NA was markedly reduced. **P < 0.01, ***P < 0.001 versus untreated vessels. †P < 0.05, ††P < 0.01, †††P < 0.001 versus the corresponding vehicle-treated group. Indomet., indomethacin; L-NA, NG-nitro-L-arginine.
Our observations indicate the involvement of thromboxane A2 in the mediation of the L-NA-induced contraction of the rat MCA. These results show, for the first time, that endogenous NO may maintain the resting cerebrovascular tone by regulation (suppression) of the synthesis and/or release of thromboxane A2. The possibility that basal NO production may also have a similar regulatory action on thromboxane release in extracerebral vessels remains to be investigated. Nevertheless, the contractile effect of L-NA was recently found to be more than two times stronger in lamb coronary arteries than that of ODQ, indicating the presence of a cyclic GMP-independent component in the effect of endogenous NO (Simonsen et al., 1997). Maintenance of a resting vasodilatory tone, however, may not be the only physiologic function of NO mediated by suppression of thromboxane release. For instance, a similar mechanism may also be involved in the antiaggregatory effect of NO.
The vasoconstrictor effect of NOS inhibitors limits their potential therapeutic use in cerebral ischemia and brain trauma (Iadecola, 1997). Our present observation that this effect is mediated by thromboxane A2 suggests that the cerebroprotective action of NO blockade in these pathophysiologic states may be enhanced by simultaneous administration of thromboxane synthase inhibitors—receptor antagonists.
Previous studies have shown constitutive thromboxane release from cerebrovascular endothelial cells (Spatz et al., 1997). The present finding that inhibition of the synthesis or action of thromboxane A2 significantly reduces the resting tone of the MCA, together with our unpublished observation that ICI 192605 also relaxes rat basilar arteries, shows that basal thromboxane release induces a weak constrictor tone in the cerebral vasculature. Therefore, similarly to the effect of endogenous NO, inhibition of the constitutive thromboxane production may also contribute to the cerebrovascular action of nitrovasodilators. The recent observation that ODQ inhibits, but does not abolish completely, the dilator effect of nitroprusside in mouse cerebral arterioles (Sobey and Faraci, 1997) is consistent with this hypothesis.
In conclusion, the present results are the first indication that the basal NO production suppresses the release of thromboxane A2 in rat MCAs and that this interaction plays an important role in the maintenance of the resting cerebrovascular tone. Further studies are needed to investigate the presence of a similar action of NO in other vascular beds and platelets.
Footnotes
Acknowledgments
The authors thank Dr. John Davis for critically reading the manuscript and Josephine Specker for technical assistance.
