Abstract
The present study was designed to investigate the influence of the endothelium and that of the L-arginine pathway on the contractile responses of isolated human cerebral arteries to electrical field stimulation (EFS) and norepinephrine. Rings of human middle cerebral artery were obtained during autopsy of 19 patients who had died 3–8 h before. EFS (1–8 Hz) induced frequency-dependent contractions that were abolished by tetrodotoxin, prazosin, and guanethidine (all at 10-6 M). The increases in tension were of greater magnitude in arteries denuded of endothelium. NG-monomethyl L-arginine (L-NMMA 10-4 M) potentiated the contractile response to EFS in artery rings with endothelium but did not influence responses of endothelium-denuded arteries. L-arginine (10-4 M) reversed the potentiating effects of L-NMMA on EFS-induced contractions. Norepinephrine induced concentration-dependent contractions, which were similar in arteries with and without endothelium or in arteries treated with L-NMMA. Indomethacin (3 × 10−6 M) had no significant effect on the contractile response to EFS or on the inhibition by L-NMMA of acetylcholine-induced relaxation. These results suggest that the contractile response of human cerebral arteries to EFS is modulated by nitric oxide mainly derived from endothelial cells; although adrenergic nerves appear to be responsible for the contraction, the transmitter involved in the release of nitric oxide does not appear to be norepinephrine. The effects of L-NMMA in this preparation appear to be due to inhibition of nitric oxide formation rather than caused by cyclooxygenase activation.
Nitric oxide (or a substance containing nitric oxide) synthesized from L-arginine accounts for the powerful vasodilator effects of endothelium-derived relaxing factor (Ignarro et al., 1987; Palmer et al., 1987) and consequently plays a decisive role in determining vasomotor tone (for review see Moncada et al., 1991). Guanidino-substituted L-arginine derivatives such as NG-nitro-L-arginine methyl ester (L-NAME) and NG-monomethyl-L-arginine (L-NMMA) inhibit the formation of nitric oxide from L-arginine in endothelial cells and the endothelium-dependent relaxation of various vascular beds (Rees et al., 1989a; Rees et al., 1990; Liu et al., 1992). Histochemical studies have demonstrated the presence of nitric oxide synthase immunoreactivity in the adventitia of rat and human cerebral arteries (Bredt et al., 1990; Nozaki et al., 1993). Consistent with these observations, a series of experiments have shown that nitric oxide may be involved in neurogenic relaxation of cerebral arteries from various animal species, including humans (Toda and Okamura, 1990; González and Estrada, 1991; Toda, 1993), because nitric oxide synthase inhibitors blocked the relaxation caused by transmural electrical stimulation. However, the possible involvement of the endothelial cells and nitric oxide in the adrenergic contraction of human cerebral vessels has not been defined. Recent experiments indicate that nitric oxide from endothelial cells may account for the inhibition of the adrenergic neural vasoconstriction in human mesenteric and deferential arteries (Aldasoro et al., 1993; Martínez et al., 1994). Accordingly, the present work was performed to determine the contribution of the endothelium and that of the L-arginine pathway in the adrenergic constrictor responses of isolated human cerebral arteries to electrical stimulation and exogenous norepinephrine. Observations were made in the presence and absence of endothelium and after exposure to L-NMMA, an inhibitor of nitric oxide synthase (Rees et al., 1989a). In addition, the adrenergic responses obtained were studied in arterial preparations treated with indomethacin, to analyze in human arteries a recent proposal suggesting a key role for cyclooxygenase in the action of inhibitors of nitric oxide synthase in the brain microcirculation of mice (Rosenblum et al., 1992).
METHODS
Human cerebral arteries were obtained during autopsy of 19 patients (12 men and 7 women, ages 31–81 years) who had died 3–8 h before. The cause of death varied; nine patients had died of myocardial infarction, seven were victims of thoracic traumatism, and three had died of respiratory insufficiency. There was no relationship between age or cause of death and the ability of the vessels to develop tension in response to norepinephrine or KCl, or to relax in response to acetylcholine or sodium nitroprusside. The arteries were immediately placed in chilled Krebs-Henseleit solution, and rings 4 mm long were cut from branches of the middle cerebral artery. The outside diameter of the rings was measured using an ocular micrometer within a Wild M8 zoom microscope (Heerbrugg, Switzerland) and ranged from 1 to 1.5 mm. In ∼50% of the artery rings the endothelium was removed mechanically by inserting a roughened stainless-steel wire into the lumen and gently rolling the vessel ring on wet filter paper.
Two stainless-steel pins, 100 μm in diameter, were introduced through the arterial lumen. One pin was fixed to the organ bath wall while the other was connected to a strain gauge (model UC3 Statham). Changes in isometric force were recorded on a Beckman recorder. Each artery ring was set up in a 4-ml bath containing modified Krebs-Henseleit solution of the following millimolar composition: NaCl, 115; KCl, 4.6; MgCl2·6H2O, 1.2; CaCl2, 2.5; NaHCO3, 25; glucose, 11.1; and disodium EDTA, 0.01. The solution was equilibrated with 95% oxygen and 5% carbon dioxide to give a pH of 7.3–7.4. Temperature was held at 37°C. To establish the resting tension for maximal force development, a series of preliminary experiments were performed on artery rings of similar length and outer diameter that were exposed repeatedly to 60 mM KCl. Basal tension was increased gradually until contractions were maximal. The optimal resting tension was 1 g. The vessels were allowed to attain a steady level of tension during a 2-h accommodation period before testing.
Electrical field stimulation (EFS) was provided by a Grass S88 stimulator (Grass Instruments, Quincy, MA, U.S.A.) via two platinum electrodes positioned on either side and parallel to the axis of the vascular ring. To assess the nature of the contractile responses and avoid direct stimulation of smooth muscle, frequency-response relationships were determined on a group of arteries in the presence and absence of 10−6 M tetrodotoxin (TTX), following a procedure previously described (Liu et al., 1991; Aldasoro et al., 1993). In summary, the protocol was designed to find the optimal stimulation parameters (15 V, 0.2-ms duration) causing a contractile response that was completely eliminated by 10−6 M TTX. Frequency-response relationships were determined using 30-s trains of pulses at 1, 2, 4, and 8 Hz. A period of 10 min was allowed between stimulations. The stimulation sequence was repeated 15 min after the addition of TTX (10−6 M), guanethidine (10−6 M), or prazosin (10−6 M). As a control, a second set of stimulations was recorded on artery rings in the absence of antagonists.
The effects of L-NMMA and L-arginine or D-arginine on EFS-induced contractions were determined in a separate group of experiments. After an initial set of stimulations, the vessel rings were incubated with L-NMMA (10−4 M) for 10 min and a second set of stimulations was performed. The third set of stimulations was performed after 10-min incubation in L-arginine (10−4 M) of those arteries previously exposed to L-NMMA. AS a control, three consecutive sets of stimulations were performed at identical intervals. Less than 10% variability in magnitudes of EFS-induced contractions was observed for a given ring during three consecutive sets of control stimulations. To assess the possible intervention of relaxing prostaglandins on the neurogenic contraction, EFS was applied before and 10 min after the addition of 10−5 M indomethacin in the organ bath. At the end of each series, TTX (10−6 M) was added and electrical stimulation was repeated to confirm the neurogenic nature of the contraction.
To check whether L-NMMA inhibits acetylcholine-induced relaxation due to activation of cyclooxygenase (Rosenblum et al., 1992), the effects of L-NMMA on acetylcholine-induced responses were evaluated in the presence of indomethacin (3 × 10−6 M). One group of arterial rings with endothelium intact were precontracted with prostaglandin F2α or norepinephrine, and the effects of 10−6 M acetylcholine were determined. After a 15-min washout, the arteries were contracted again and a second application of acetylcholine was made in the absence and in the presence of either L-NMMA, indomethacin, or L-NMMA together with indomethacin. In another group of experiments, vessel rings were stimulated with fixed electrical stimuli (20 V, 4 Hz, 0.25 ms). After an initial set of stimulations, the artery rings were incubated with either L-NMMA, indomethacin, or L-NMMA together with indomethacin for 10 min and a second set of stimulations was given. At least six artery rings remained untreated and served as a control for possible time-related effects.
Concentration-response curves for norepinephrine were determined in the absence and presence of L-NMMA from separate artery preparations with and without endothelium. EC50 values (concentrations of norepinephrine producing half-maximal contractions) were determined from individual concentration-response curves by nonlinear regression analysis, and from these values the geometric means were calculated (Fleming et al., 1972).
After each experiment, the arteries were carefully opened and stained with AgNO3 to visualize the endothelium (Caplan and Schwartz, 1973). Only results from vessels with >70% of the endothelium were considered as control segments. Vessels in which the endothelium had been rubbed never showed >5% of their intima covered with endothelium either before or after the experiment. Functional integrity of the endothelium was confirmed by the presence of relaxation induced by acetylcholine (10−7 to 10−6 M) during contraction obtained with prostaglandin F2α (10−6–3 × 10−6 M) or norepinephrine (10−6–3 × 10−6M).
The following drugs were used: acetylcholine chloride, norepinephrine hydrochloride, TTX, guanethidine, L-NMMA, L-arginine hydrochloride, D-arginine hydrochloride, prostaglandin F2α, indomethacin (Sigma Chemical Co., St. Louis, MO, U.S.A.), and prazosin (Pfizer, Sandwich, Kent, England). Drugs were prepared and diluted in distilled water except for indomethacin, which was dissolved in absolute ethanol and disodium bicarbonate solution (150 mM) and readjusted to pH 7.4 with HCl before use. Stock solutions of the drugs were freshly prepared every day.
Data are expressed as means ± SD; n indicates the number of human subjects. At least eight artery rings were obtained from each case. The results were evaluated statistically by means of paired or unpaired Student's t test or one-way analysis of variance. A probability value of <0.05 was considered to be significant.
RESULTS
EFS
EFS produced frequency-dependent contractions of human cerebral artery rings at resting tension. Because the increases in tension induced by EFS were abolished by TTX (10−6 M), guanethidine (10−6 M), and prazosin (10−6 M) (Fig. 1), it is assumed that the effect was due to the release of norepinephrine from adrenergic nerves acting on α1-adrenoceptors.
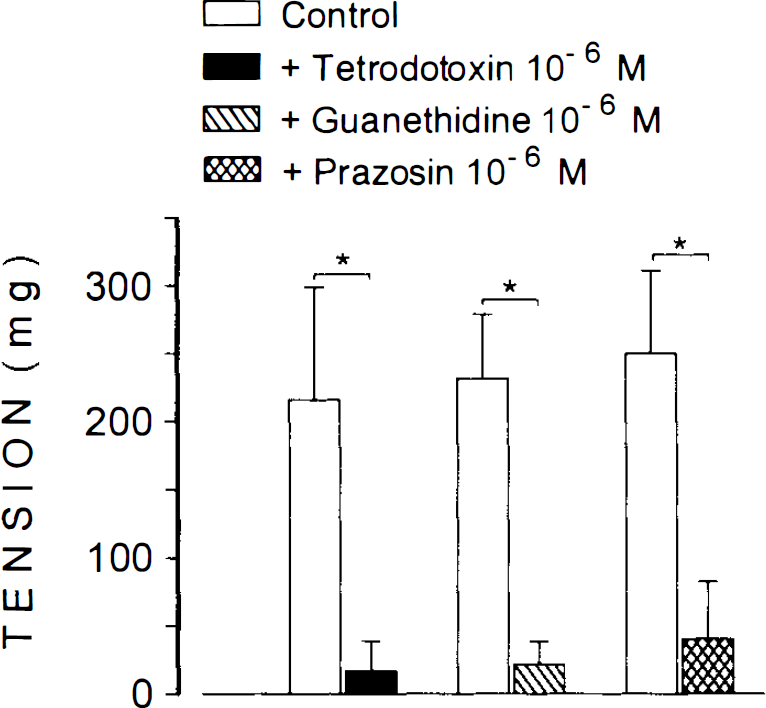
Contractile effects of electrical field stimulation (8 Hz) on human cerebral arteries in the absence (control) and in the presence of tetrodotoxin (10−6 M, n = 7), guanethidine (10−6 M, n = 5), or prazosin (10−6 M, n = 5). Values are means ± SD shown by vertical bars. *p < 0.05.
The contractile responses were of greater magnitude in arteries denuded of endothelium (Fig. 2). The increased contraction observed in vessels without endothelium does not reflect a nonspecific effect on smooth muscle because the contractile response to KCl (60 mM) was similar in intact and denuded vessels (1,546 ± 346 mg versus 1,285 ± 294 mg) (p > 0.05).
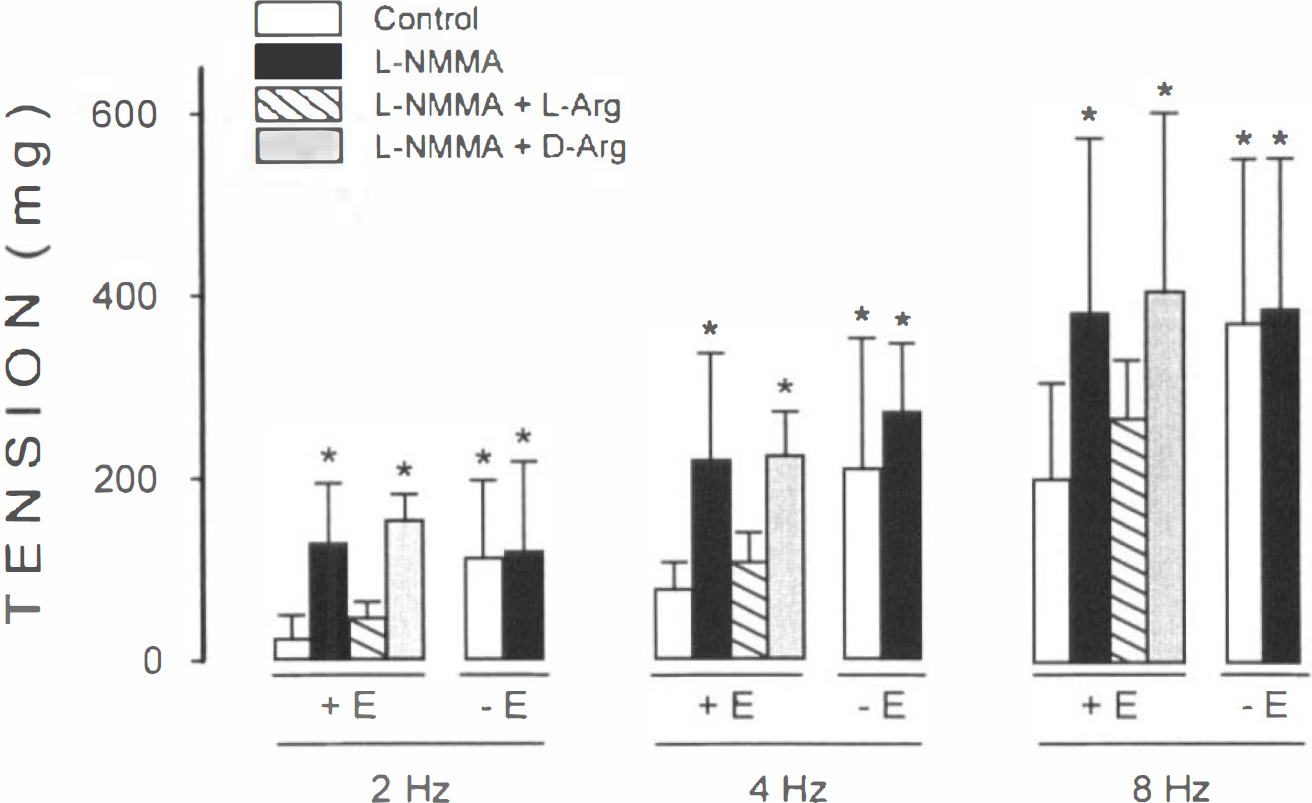
Contractile effects of electrical field stimulation in artery rings with (+ E) (n = 6) and without (−E) (n = 6) endothelium in control conditions and in the presence of NG-monomethyl-L-arginine (L-NMMA) (10−4 M), or L-NMMA plus L-arginine (10−4 M) or D-arginine (104 M). Data are the mean ± SD shown by vertical bars. *p < 0.05, compared with control rings with endothelium.
Arteries exposed to L-NMMA (10−6–10−4 M) did not show significant changes in resting tension as compared with arteries incubated in Krebs-Henseleit solution (p < 0.05). However, L-NMMA (10−4 M) enhanced significantly the EFS-induced contractile responses of artery rings with endothelium but did not influence the contractile responses of endothelium-denuded arteries (Fig. 2). The augmentation of EFS-induced contraction by L-NMMA was similar to the effect of endothelial denudation. The augmentation of EFS-induced contraction in arteries with endothelium was completely reversed by L-arginine (10−4 M), but not by an identical concentration of D-arginine.
Effects of norepinephrine
Cumulative application of norepinephrine produced concentration-dependent increases in tension. The maximal tension developed as well as the concentration of norepinephrine producing half-maximal contractions (EC50) was similar in arteries with and without endothelium (8.5 ± 3.1 × 10−7M versus 9.4 ± 3.5 × 10−7 M; p > 0.05). In arteries with endothelium, the presence of L-NMMA (10−4 M) did not change significantly (p > 0.05) the responses to norepinephrine (EC50: 6.5 ± 2.3 × 10−7 M) (Fig. 3).
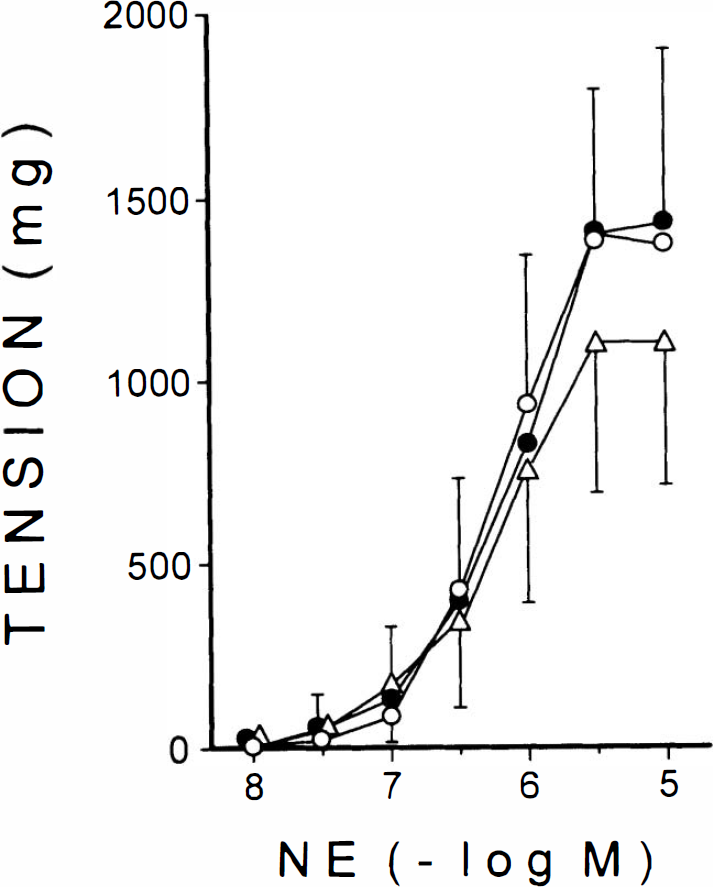
Concentration-response curves for norepinephrine (NE) determined in artery rings with (•) (n = 5) and without (n = 5) (○) endothelium. The presence of 10−4 M L-NMMA (Δ) (n = 6) did not change significantly (p > 0.05) the response to NE.
Effects of indomethacin
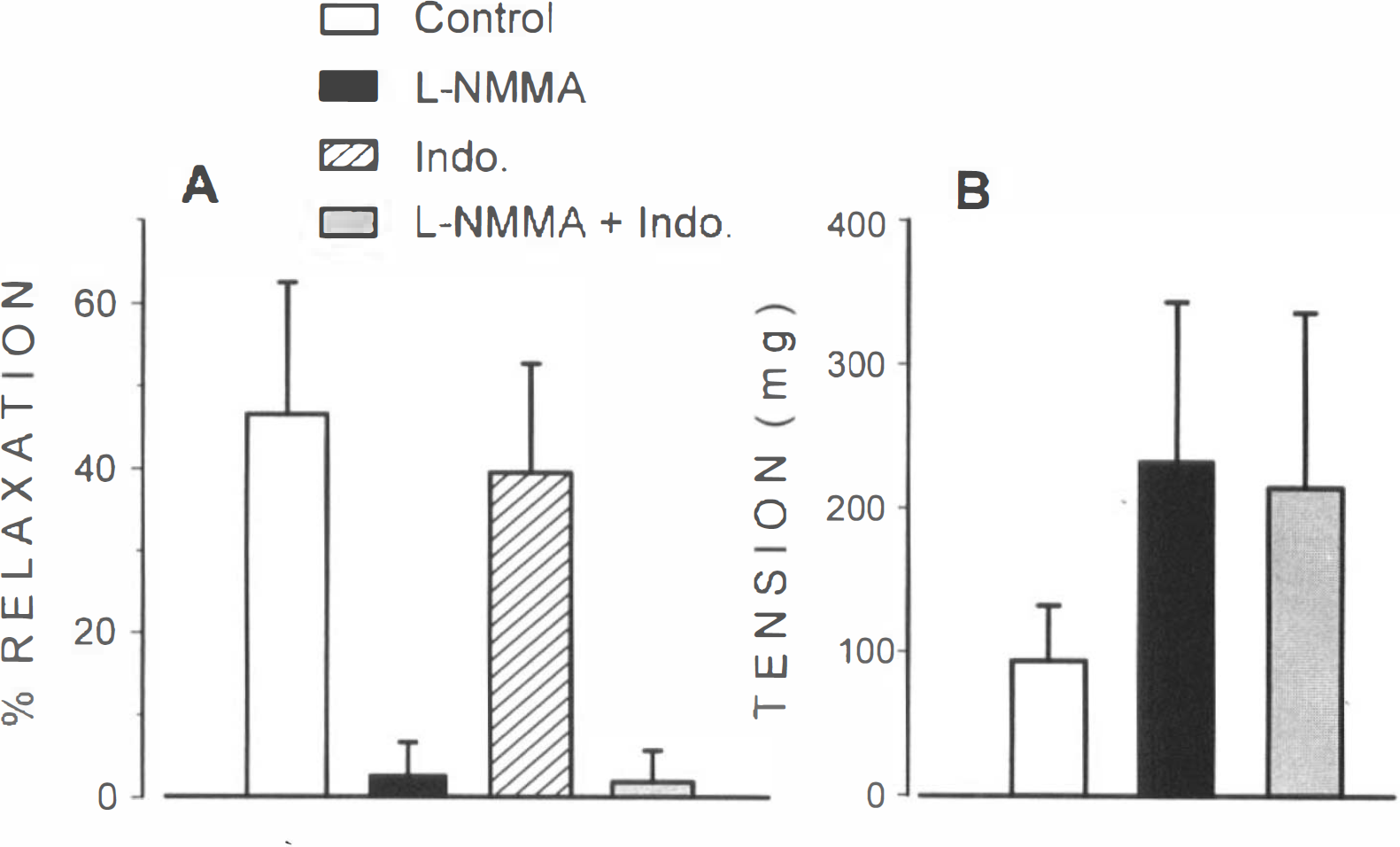
To test whether prostaglandins are involved in the contractile response to EFS, unrubbed artery rings were pretreated with the cyclooxygenase inhibitor, indomethacin (3 × 10−6 M). Contractile responses to EFS of these artery rings were unaffected (p > 0.05) after indomethacin treatment (results not shown). Figure 4A shows that L-NMMA, but not indomethacin, inhibited relaxation by acetylcholine. Treatment of artery rings with L-NMMA plus indomethacin did not affect the antidilating effects of L-NMMA. On the other hand, the potentiation induced by L-NMMA on neurogenic responses was not modified in the presence of indomethacin (Fig. 4B).
DISCUSSION
The present study shows that removal of endothelium produces a significant increase in the contractile response of human cerebral arteries to EFS. The nitric oxide inhibitor L-NMMA enhanced the contractile response to nerve stimulation in arteries with endothelium. This potentiation was reversed by L-arginine (the precursor of nitric oxide production) but not by its inactive isomer D-arginine. By contrast, L-NMMA did not affect the contractile response of endothelium-denuded arteries. The results indicate that nitric oxide released from endothelial cells is responsible for the depressant influence of endothelium on the contractile responses to EFS of human cerebral arteries. These findings agree with previous observations in rabbit pulmonary (Liu et al., 1991) and rat iliac (González et al., 1992) arteries, suggesting that the contractile response to electrical stimulation is modulated by nitric oxide derived mainly from endothelial origin. In dog and monkey cerebral arteries (Toda and Okamura, 1990) nitric oxide might be directly released from perivascular nerve fibers as a transmitter to counteract neurogenic contractions because inhibition of nitric oxide synthase potentiated contractile responses in endothelium-denuded arteries.
Our results also show that the endothelial-mediated inhibition was observed in response to EFS but not to norepinephrine. Therefore, the augmentation of neurogenic responses by endothelial removal or L-NMMA treatment do not appear to be associated with the adrenergic neurotransmitter. As postulated in previous reports, it is possible that during nerve stimulation other transmitters are released and induce nitric oxide production by endothelial cells. In the sheep, for example, there is evidence that vasoactive intestinal peptide mediates neurogenic cerebral vasodilation via cyclic guano-sine monophosphate-mediated mechanisms that involve nitric oxide synthesis (Gaw et al., 1991) whereas in the rabbit middle cerebral artery the neuroeffector mechanism mediating this response has a predominantly cholinergic endothelium-dependent component (Van Riper and Bevan, 1992). Neuropeptide Y could be another transmitter involved in neurogenic mediated responses because it coexists with norepinephrine in perivascular nerve fibers (Lundberg et al., 1982; Edvinsson et al., 1984), and it is released on sympathetic stimulation (Allen et al., 1984; Lundberg et al., 1989). Whether these or other mediators are responsible for the release of nitric oxide from human cerebral vessels is unknown.
Norepinephrine concentration–response curves were similar in arteries with and without endothelium as well as in arteries treated with L-NMMA. Thus, the effects of norepinephrine on human cerebral vessels do not appear to be modulated by release of nitric oxide. This pattern of endothelium-independent responses observed for norepinephrine also has been shown for endothelin and vasopressin in human cerebral arteries (Martín de Aguilera et al., 1990a, 1990b). From these results it can be concluded that under the present experimental conditions, the stimulation of adrenoceptors or the increase in smooth muscle tone caused by norepinephrine do not trigger the release of endothelial nitric oxide.
Recent experiments show that the inhibitory effects of L-NMMA on acetylcholine-induced relaxation of mouse pial arterioles are blocked by locally applied indomethacin. In these findings it is assumed that L-NMMA acts by causing the production of cyclooxygenase-dependent superoxide, which inactivates the endothelial factor responsible for acetylcholine-induced dilation (Rosenblum et al., 1992). No evidence of this mode of action of L-NMMA was observed in our experiments. First, indomethacin did not prevent L-NMMA from blocking dilation by acetylcholine. Second, we observe that indomethacin failed to block the L-NMMA-induced potentiation of contractile responses to EFS. Findings similar to those of the present study have been obtained in vivo, suggesting that effects of inhibitors of nitric oxide synthase are not mediated by oxygen radicals in cerebral vessels (Wei et al., 1992). In the absence of evidence that L-NMMA activates cyclooxygenase, it is assumed that L-NMMA competes with L-arginine on the pathway to nitric oxide formation. Apart from the possibility that different mechanisms may be involved in different species, care should be taken when extrapolating from in vitro to in vivo experiments. Thus, conclusions as to the possibility that L-NMMA effects on cerebral vessels are cyclooxygenase-dependent must be made with caution.
It has been reported that a continuous basal release of nitric oxide from vascular endothelium modulates responses in various beds (Rees et al., 1989b; Wiklund et al., 1990). It appears likely that inhibition of this nitric oxide release by L-NMMA would increase basal smooth-muscle tone (Rees et al., 1989b; Moncada et al., 1991). In the present experiments, L-NMMA did not increase resting tension of cerebral arteries, a finding similar to that recently reported in human mesenteric and deferential arteries (Aldasoro et al., 1993; Martínez et al., 1994). Nevertheless, the possibility of basal release of nitric oxide in the absence of contractile effects in response to L-NMMA cannot be totally excluded in the present experiments. The reason for the inability of L-NMMA to increase basal tension in these arteries is not clear. Both in vivo and in vitro experiments indicate that the vasoconstricting action of inhibitors of nitric oxide synthesis is directly dependent on vascular tone (Rees et al., 1989b). It has also been proposed that differences in basal production in nitric oxide may be due to size of the vessel, being higher in large arteries than in smaller vessels (Faraci, 1991). With regard to human vascular preparations, there are findings showing that inhibitors of nitric oxide synthesis potentiate (Yang et al., 1990) or have no effect (Woolfson and Poston, 1990) on the resting tension of human artery preparations. A critical factor in the analysis of nitric oxide activity in postmortem human cerebral arteries is the possible reduction of endothelial cell function in the face of an apparently normal ability of smooth muscle to respond to exogenous agonists (Lüscher and Vanhoutte, 1988; Onoue et al., 1993).
In conclusion, the results indicate that endothelial cells of human cerebral arteries depress the contractile response to EFS of intramural nerves by a mechanism involving the release of nitric oxide from these cells. Although adrenergic nerves appear to be responsible for the contractile response, the agent causing the release of nitric oxide does not appear to be norepinephrine. The action of L-NMMA in human cerebral arteries appears to be due to inhibition of nitric oxide formation rather than caused by cyclooxygenase activation.
Footnotes
Acknowledgment:
This work was supported by the Comisión Interministerial de Ciencia y Tecnología and Ministerio de Sanidad.
