Abstract
Introduction:
Association of serotonin transporter gene (5-HTTLPR) polymorphisms with adverse cardiovascular (CV) events in women with suspected ischemia has not yet been reported. We hypothesized an association of 5-HTTLPR polymorphisms with risk of adverse CV events in women with suspected ischemic heart disease (IHD) referred for coronary angiography enrolled in the Women’s Ischemia Syndrome Evaluation (WISE).
Method:
We studied clinical and angiographic data and DNA from a cohort of 437 Caucasian women enrolled in the WISE genotyped for the long (L) and short (S) variant of the 5-HTTLPR polymorphism. Women were followed yearly for adverse CV events (defined as first occurrence of all-cause death, myocardial infarction, stroke, or heart failure hospitalization) with data collected at WISE 10-year follow-up. Exploratory analyses compared outcomes between genotype groups.
Results:
A total of 437 women, with baseline, angiographic, and long-term follow-up data, were successfully genotyped. Their mean age was 58 ± 11 years and body mass index 29 ± 6; 54% had hypertension, 18% diabetes, 50% dyslipidemia, 20% depression history, and only 34% had obstructive CAD. At 8.9 years median follow-up, the SS genotype was associated with significantly increased risk of adverse CV event versus LL + LS (1.93, confidence interval [CI]: 1.03-3.61, P = .03). Results were not significant for all-cause death (hazard ratio: 1.63, CI: 0.91-2.93, P = .09).
Conclusion:
Among a cohort of Caucasian women with suspected IHD enrolled in the WISE, the SS homozygous genotype for the 5-HTTLPR polymorphism was associated with increased risk of adverse CV outcomes.
Introduction
Ischemic heart disease (IHD) continues as the leading cause of morbidity and mortality in women. 1 Thus, a critical need exists to improve assessment of cardiovascular (CV) risk and prognosis in women. Most women with signs and symptoms of ischemia referred to coronary angiography do not have obstructive coronary artery disease (CAD), yet such women have considerably increased risk of adverse outcomes (defined as all-cause death, non-fatal myocardial infarction [MI], heart failure hospitalization, nonfatal stroke, and cardiovascular death) versus those without symptoms of ischemia. 2,3 Thus, additional insight into the mechanisms linked with this increased risk is needed. Many of these women, with symptoms and signs of ischemia without obstructive CAD, have evidence of coronary vascular dysfunction (eg, endothelial and/or microvascular dysfunction) that has been associated with increased risk of CV events. 4 Although IHD among women has been better characterized in the recent years, the pathophysiologic mechanisms underlying this condition and links with adverse outcomes remain poorly understood.
One candidate mechanism linked to CV risk is serotonin regulation, which has been associated with several CAD syndromes. Serotonin (5-hydroxytryptamine [5-HT]) is a neurotransmitter in the brain and gastrointestinal tract involved in a variety of processes throughout the body including regulation of catecholamine release. 5 In blood, platelets possess a high-efficacy serotonin transporter, enabling uptake of 5-HT from the gut and lung and release it during a thrombotic event. 6 Platelets possess the ability to transport and store 5-HT but do not synthesize it. 5-Hydroxytryptamine levels have been found to be elevated in thrombotic and hypertensive states. 7 The function of the serotonin transporter gene (SERT), which is located on chromosome 17, is uptake and release of serotonin from cells, and this SERT function in the central nervous system (CNS) is well documented. 8,9 Also, SERT is the target of selective serotonin reuptake inhibitors used to treat many CNS disorders linked to increased IHD risk such as depression. In women, estrogen modulates 5-HT pathways responsible for CNS regulation of pain in patients with migraine headaches. 10 In post-menopausal women, recent evidence suggests that serotonergic effects directly mediate the impact of estrogen on brain activation during working memory and affective processing. 11 In patients with a history of mental stress-induced myocardial ischemia (MSIMI) and stable coronary heart disease, treatment with selective serotonin reuptake inhibitors (SSRIs) compared to placebo resulted in lower rates of MSIMI. 12
A polymorphism in the promoter region of SERT is well described. 13 –15 This polymorphism (5-HTTLPR) is located upstream from the transcription initiation site and consists of a 44-bp insertion/deletion polymorphism. This polymorphism is functional in vitro. 16 Two common alleles, long (L) and short (S), of this gene are linked to depression. The transcriptional activity of the L variant is more than twice that of the S variant, with recent studies having uncovered potential associations between expression of SERT gene promoter variants with CV diseases. 17 –19 Specifically, risk of pulmonary hypertension is increased with expression of the LL genotype, a variant associated with increased expression and function of SERT. The association of the 5-HTTLPR and risk of MI has been previously studied with suggested associations between LL genotype and adverse CV events. 20 –22 However, these studies have focused mainly on predominantly male cohorts with obstructive CAD.
The purpose of this study was to investigate possible associations of the SERT polymorphism with adverse CV events in women with symptoms and signs of myocardial ischemia enrolled in the NHLBI Women’s Ischemia Syndrome Evaluation (WISE).
Methods
Study Population and Follow-Up
The WISE was a multicenter prospective cohort study designed to optimize diagnostic evaluation and testing and identify mechanisms for IHD in women. 23 The inclusion criteria and protocol are detailed elsewhere. 23 Briefly, women had to be at least 18 years of age and referred to clinically indicated coronary angiography to evaluate symptoms and signs of IHD. Exclusion criteria included comorbidity compromising 1-year follow-up, pregnancy, contraindications to provocative diagnostic testing, cardiomyopathy, New York Heart Association class IV heart failure, recent MI, significant valvular or congenital heart disease, and inability to complete questionnaires. Outcomes data collected included all-cause death, nonfatal MI, heart failure hospitalization, nonfatal stroke, and cardiovascular death. All-cause death has been used as an additional outcome in addition to cardiovascular death in the literature, particularly in the WISE population, who were often categorized as having had a noncardiovascular event. 24 After enrollment and collection of demographic and other baseline data, the women were followed by yearly phone calls from site study coordinators for follow-up information and adverse outcomes. Follow-up data were tabulated at the data coordinating center and adjudicated by an events committee without the knowledge of woman’s clinical status or genotype. Follow-up evaluation was performed in 2 stages. Initial follow-up was performed via yearly phone calls for nonfatal CV events, and deaths were noted at those intervals and categorized as CV or non-CV by the WISE Events Committee independent of any genotype information. Additionally, in the interim between initial exploratory data analysis and final outcomes, the planned WISE 10-year follow-up data became available. These new data extended average follow-up for the cohort studied here from 5.8 to 8.9 years, which also included a National Death Index search. Thus, several women were also reclassified based on these new data (usually from lost or censored to dead).
A flow diagram summarizing the process of genotype analysis is presented in Figure 1. Initial analysis conducted with 5-year outcomes revealed an interesting trend in association between genotype and CV outcomes stratified by race; the suggested association was limited to Caucasian women, and 5-year outcomes were published in abstract form. 25 Subsequent genotyping was restricted to the remaining samples from Caucasian women. Of the total 800 samples obtained as part of the original cohort, 699 women had samples that contained adequate DNA for analysis, complete clinical data at baseline, and follow-up data for adverse outcomes analyses. Of these, 437 Caucasian women had complete psychosocial data, specifically Beck depression index, and comprise the cohort studied with long-term outcome data.
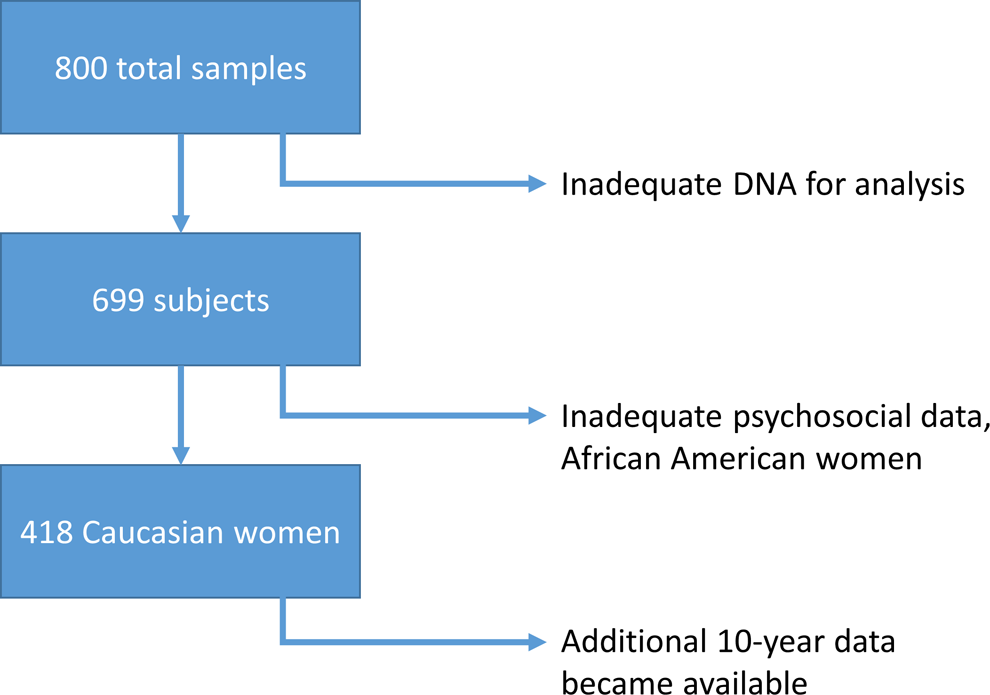
Number of women’s DNA samples available/data analysis.
Genotyping Methods
Patients who were agreeable provided informed consent for a genetic substudy that was approved by the Institutional Review Board at each participating WISE site. For analysis of the 5-HTTLPR, DNA was amplified using polymerase chain reaction (PCR) using a forward and reverse primer. Initial denaturation of DNA was done at 95°C for 10 minutes, followed by 9 cycles at 95°C for 30 seconds, then 64°C for 20 seconds, and 72°C for 30 seconds. An additional 41 cycles of 95°C for 30 seconds, 67°C for 30 seconds, and 72°C for 30 seconds were performed with a final cycle of 72°C for 7 minutes. Then, PCR products were separated on 3% agarose gel containing ethidium bromide, and visualization of the bands for the L allele (528 bp) and S allele (484 bp) was performed under ultraviolet light.
Statistical Analysis
Statistical analyses were conducted in SAS 9.3 (SAS Institute, Cary, North Carolina). Differences in patient characteristics across genotypes were assessed by F test for continuous variables, and χ2 or Fisher tests for categorical variables. Survival analysis was conducted using Cox Proportional Hazards Regression procedure. Following recommendations in Muller and Fetterman, 26 backward stepdown model selection was applied. The outcome of interest was defined as a patient experiencing a first event among CV-related death, nonfatal stroke, nonfatal MI, or heart failure hospitalization. Death from any cause was analyzed separately as all-cause death. Hardy-Weinberg equilibrium was tested for each group analysis. For genotype analysis, the SS group was compared to a composite genotype of either LS or LL (LS+LL). Covariates were sequentially tested, and those with a P > .10 were removed from the model. Covariates tested include antiplatelet drug use, depression requiring treatment, Beck depression inventory, psychoactive medication, age (divided by 10), body mass index (BMI; divided by 10), history of diabetes, history of dyslipidemia, CAD severity score (log2 transformed), history of hypertension, current smoker, and alcohol use. For the final models, after selection, hazard ratios (HRs) of these variables for the occurrence of all-cause death and CV composite outcomes were estimated, and Cox survival curves with estimated survival times plotted with log rank testing for significance. Two-tailed P < .05 were considered statistically significant.
Results
Characteristics of the Groups by Genotype
Overall, among the 437 women, genotype frequencies were LL = 30%, SS = 20%, and LS = 50%. The genotype results did not deviate from Hardy-Weinberg equilibrium. Their mean age was 58 ± 11 years and BMI 29 ± 6; 54% had hypertension, 18% diabetes, 50% dyslipidemia, 20% depression history, and only 34% had obstructive CAD. There were no differences in baseline characteristics among the genotype groups (Table 1). Baseline CV risk factors and use of antidepressants and BDI scores did not differ between groups. Notably, there was additionally no difference in obstructive CAD by coronary angiography according to genotype.
Baseline Characteristics by Genotype for Caucasian Women.a,b,c,d
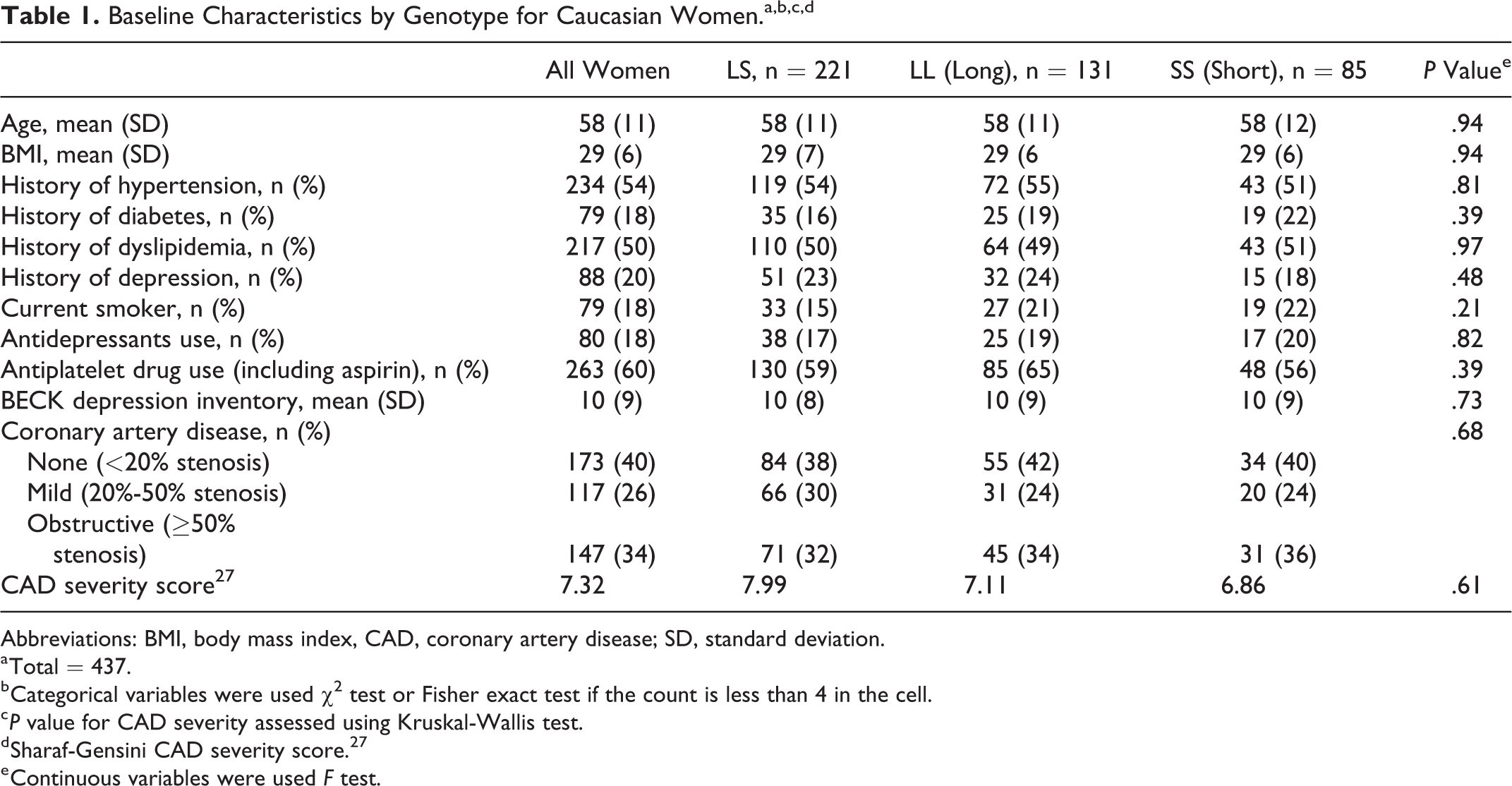
Abbreviations: BMI, body mass index, CAD, coronary artery disease; SD, standard deviation.
a Total = 437.
b Categorical variables were used χ2 test or Fisher exact test if the count is less than 4 in the cell.
c P value for CAD severity assessed using Kruskal-Wallis test.
d Sharaf-Gensini CAD severity score. 27
e Continuous variables were used F test.
During the WISE planned long-term follow-up (up to 8.9 years for the cohort studied), there were 99 women who experienced a “first” adverse CV event among all-cause death, nonfatal MI, nonfatal stroke, or hospitalization for heart failure. Risk of adverse CV event for SS versus LS+LL, adjusted for covariates, was increased (Table 2; HR 1.93, confidence interval [CI] 1.03-3.61, P = .03). Cox survival curves with estimated survival times are presented in Figure 2. Results were not significant for all-cause death alone (HR 1.63, CI 0.91-2.93, P = .09).
Cardiovascular Events at Long-Term Follow-Up (8.9 Years Median).
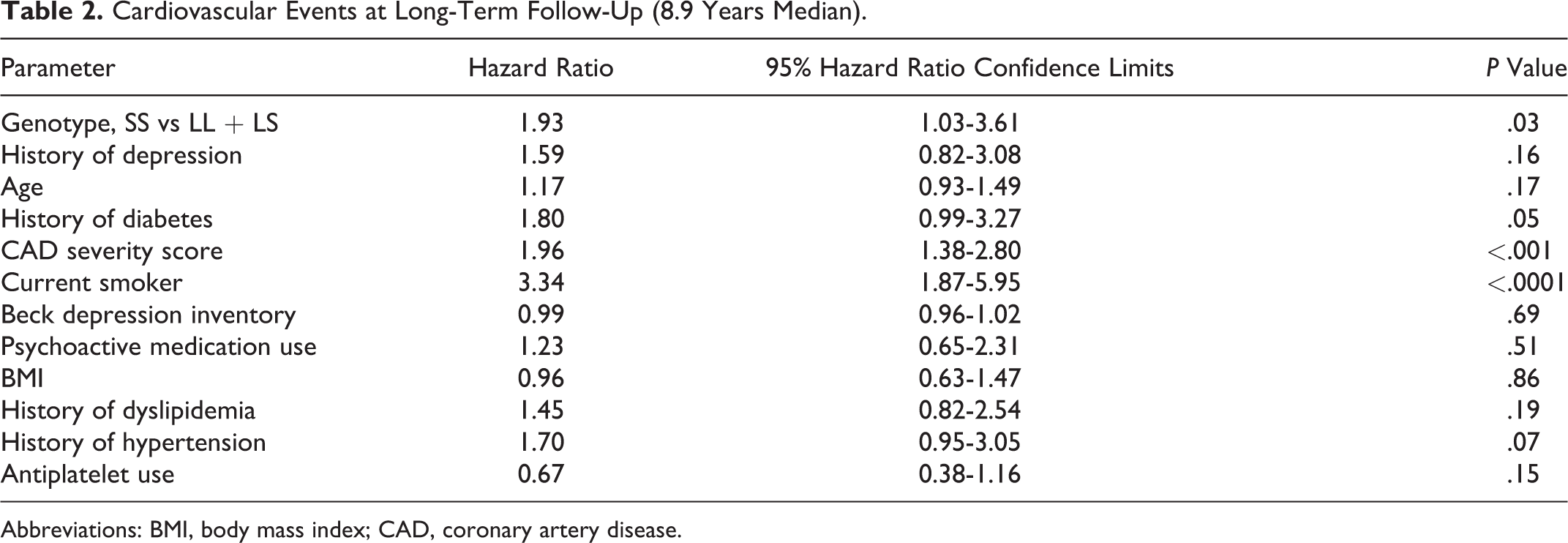
Abbreviations: BMI, body mass index; CAD, coronary artery disease.
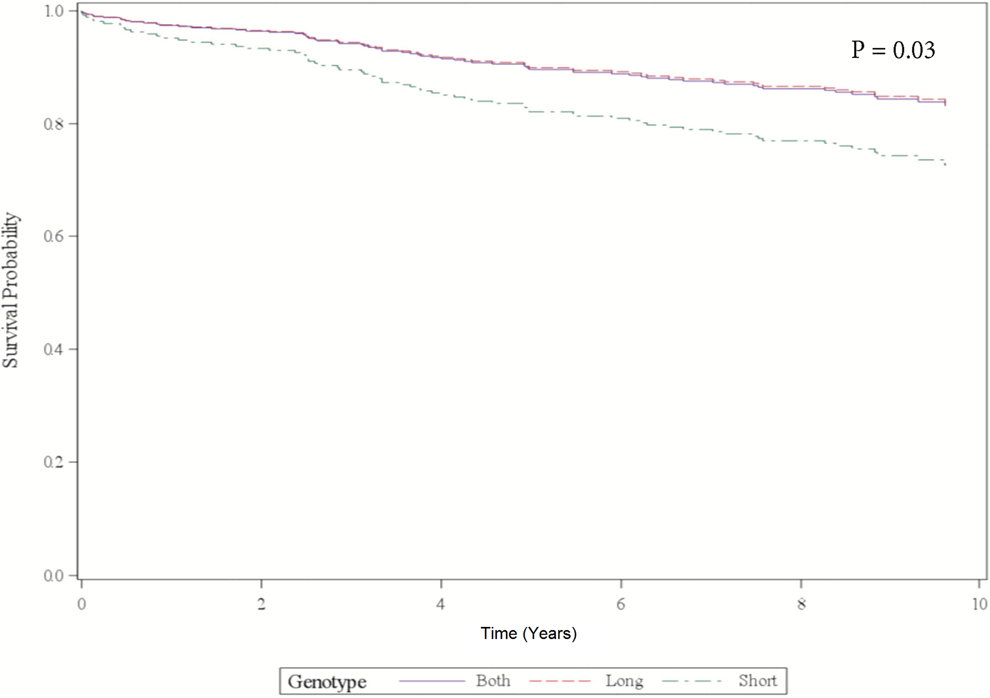
Adjusted cox survival curves for cardiovascular (CV) event at 10 years.
Discussion
This is the first report of an analysis of the 5-HTTLPR Single Nucleotide Polymorphism and association with CV-related adverse outcomes among women with symptoms/signs of IHD whose coronary anatomy was assessed by angiographic core laboratory analysis. After fully adjusting for other important clinical covariates, the SS genotype of the 5-HTTLPR was significantly associated with adverse CV events at long-term follow-up in this cohort of Caucasian women.
Our results are the first to link this polymorphism in the promoter region of SERT with adverse CV events in a specific cohort of women. These findings are potentially important, as this represents the first study performed exclusively in women with inclusion of angiographic data. Previous work from the WISE and others has suggested that women without obstructive CAD have a higher risk of adverse outcomes than predicted. 2,4 Data from the WISE and others have also documented that coronary microvascular dysfunction is associated with increased risk among those without obstructive CAD. 28,29 However, the pathophysiologic mechanisms associated with this increased risk remain poorly understood.
Our specific results differ from previous reports of associations of LL genotype with adverse cardiac events in men. One study suggested an increased risk of MI in patients with LL genotype, 15 while another suggested a protective effect with the SS genotype delaying the onset of first MI especially in smokers. 20 Others have suggested that the S allele is associated with increased risk of cardiac events mediated, at least in part, by depressive symptoms in patients after MI. 21 However, these studies were primarily performed in exclusive male cohorts with limited angiographic data. The limited data in women suggest a sex-based difference in the association between 5-HTTLPR genotypes and heart rate reactivity, depression, and interleukin (IL)-6 levels. 30,31
Several potential mechanisms could explain observed association and why results differ according to sex. When platelets aggregate, 5-HT is a primary factor that promotes continued aggregation, and blood vessels, in which platelets aggregate, are exposed to high 5-HT concentrations. As such, 5-HTTLPR genetic variants leading to altered uptake of serotonin may affect binding of 5-HT to platelets via 5HT2A receptors, which have also been implicated in mood disorders. Serotonin induces expression of tissue factor and plasminogen activator inhibitor-1, proliferation of vascular smooth muscle cells, and enhances platelet procoagulant properties and their activation. Inhibition of serotonin uptake decreases platelet 5-HT content, inhibits thrombosis, and exacerbates bleeding because of prevention of 5-HT reuptake. 5-Hydroxytryptamine also affects heart rate and can act to inhibit noradrenaline release centrally. Thus, serotonin regulation plays a key role in endothelial cell and vascular smooth muscle cell function, cardiovascular function, and platelet aggregation. Women with depression frequently have endothelial dysfunction. 32,33 In our study cohort, as well the overall WISE population, approximately 35% of women had some evidence of depressive symptoms, thus predisposing them to endothelial dysfunction. This, in turn, may affect their sensitivity to serotonin within the coronary vasculature. As discussed earlier, the effect of serotonin on coronary vessels is dependent on whether the endothelium is intact; with underlying endothelial dysfunction, serotonin induces vasoconstriction that has been shown to be associated with adverse cardiac events including MI. 34 Additionally, although the polymorphisms within this serotonin transporter genotype have been traditionally associated with depression, it was not the case in our study as depression was similarly prevalent among all genotype groups. Depression was measured by 3 different assessments including self-reported depression, use of anti-depressant drugs, and the BDI score. Therefore, this suggests that the observed association between heightened CV risk and SS genotype was not likely driven simply by a depression-mediated mechanism.
Other possible explanations for the variable association between this genotype and outcomes dependent on sex include modulating effects of chronic stress and depression, which may vary by sex and the role of estrogen. A study of the effects of environmental stress and sex on associations between depression and 5- HTTLPR suggested that the strength of this link varied according to sex and stressful life events. Specifically, women with SS genotype tended to show increased susceptibility to depression under stressful life conditions. However, in men, the LL genotype was associated with increased transcriptional activity and similar increased susceptibility to depression. 35 Others have suggested that 5-HTTLPR was associated with heart rate reactivity to psychological challenge but showed an interaction with sex. 30 Responses of women with SS genotype were greater compared to men with SS and women with LS or LL. It has also been suggested that healthy young women with SS genotype show a higher IL-6–IL-10 ratio at baseline and during stress. 36
With regard to the differential association of SS genotype and all-cause death by follow-up time period, the rationale for this finding remains unclear. As mentioned earlier, the use of all-cause death over specific CV death has been favored in the WISE group due to the frequent absence of obstructive CAD. We have observed that an angiogram report stating “normal coronary arteries” or “no obstructive CAD” often prompts the local default classification of a “non-CV death.” It may be that the mechanisms, behind the association between serotonin regulation and all-cause death in this population, are linked to early events within the first few years with relatively few late deaths. Additionally, the longer follow-up time allows for increasing number of deaths although not necessarily CV related. This association warrants further evaluation.
Limitations and Strengths
Due to the study’s initial design, detailed information about specific antidepressant medication was not collected. Tricyclic antidepressants and early SSRIs were large part of depression treatment regimens during the time of the initial WISE cohort recruitment beginning in 1997. These latter agents target the same serotonin transporter that is regulated by 5-HTTLPR polymorphisms, so the concurrent use of an SSRI could be a confounding factor. An additional potential limitation is the restriction of genotype analysis to Caucasian women. This was based on our preliminary data which suggested a genotype effect only in the Caucasian cohort. However, it is possible with a larger cohort of African American and or Hispanic women, with the extended follow-up data that a significant association may have been discovered, and this gap is worthy of further investigation. Additionally, the women available for this analysis cohort had relatively few adverse events, thus further work should focus on replicating these results in cohorts of higher risk women including minority women.
Additionally, patients without obstructive CAD are less likely to have their death classified as a CV death, so the numbers of CV deaths are possibly underestimated. Although this was addressed by an Events Committee, a defined classification of causes of death, and deaths that could not clearly be attributed to a noncardiac cause classified as cardiac, it is very difficult to ascertain a CV contribution to many noncardiac deaths associated with deaths related to other causes. So, CV deaths are likely underestimated. Along these lines, exact causes of death such as heart failure and arrhythmia in our cohort could not be determined. However, more recent work in WISE has started to provide more insight into the specific causes of mortality and morbidity among these women. Specifically, several studies from WISE have documented that, overall, the majority of deaths in WISE are CV events, specifically sudden cardiac death. 37 Additionally, heart failure hospitalization in WISE was driven primarily by women presenting with heart failure in the setting of preserved ejection fraction, 38 As such, myocardial infarction appears to be a less prominent event in this specific cohort of patients. 4 Although the exact mechanisms driving risk are not well understood, it may be that genetic predisposition, as suggested in our study, along with traditional cardiovascular risk factors, may predispose these women to microvascular dysfunction and associated CV outcomes as illustrated in Figure 3.
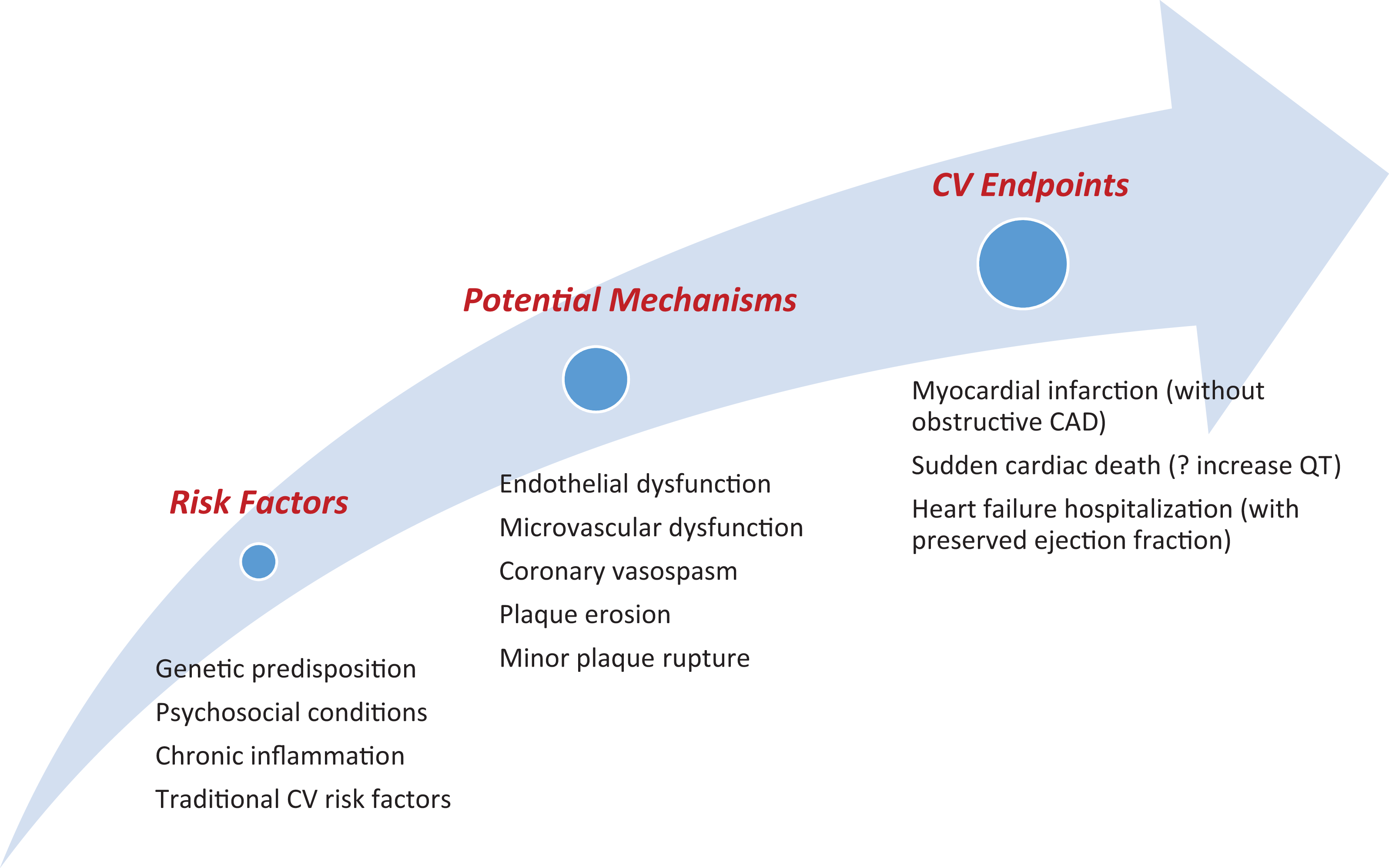
Proposed risk factors and mechanisms leading to adverse cardiovascular (CV) events in Women’s Ischemia Syndrome Evaluation (WISE).
Our study is the first to examine this polymorphism in women with signs and symptoms of ischemia in which the majority did not have obstructive CAD. Additionally, this is the first study focusing on this genotype in women, as it has been traditionally studied in predominantly male cohorts. Although the adverse outcomes risk associated with the SS genotype was relatively lower when compared to known traditional risk factors such as hypertension and CAD severity, identification of genetic risk may be critical in women without these traditional risk factors. Therefore, this work contributes to advance our understanding of the potential relevance to this polymorphism in a unique cohort of women in which traditional and genetic risk stratification studies have been limited.
Conclusion
The results of our work suggest that genetic variations in SERT may play an important role in risk of adverse cardiovascular events in Caucasian women with suspected ischemia. These findings are particularly important in this group of women without obstructive coronary disease and contribute to our understanding of the increased cardiovascular risks in this subset of women. Genotype evaluation in Caucasian women with suspected ischemia warrants further investigation in larger population studies.
Footnotes
Authors’ Note
This work is solely the responsibility of the authors and does not necessarily represent the official views of the National Heart, Lung, and Blood Institute or National Institutes of Health.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by contracts from the National Heart, Lung and Blood Institutes nos N01-HV-68161, N01-HV-68162, N01-HV-68163, N01-HV-68164, grants U0164829, U01 HL649141, U01 HL649241, K23HL105787, T32HL69751, R01 HL090957, and 1R03AG032631 from the National Institute on Aging, GCRC grant MO1-RR00425 from the National Center for Research Resources, the National Center for Advancing Translational Sciences Grant UL1TR000124 and UL1TR001427, and grants from the Gustavus and Louis Pfeiffer Research Foundation, Danville, NJ, The Women’s Guild of Cedars-Sinai Medical Center, Los Angeles, CA, The Ladies Hospital Aid Society of Western Pennsylvania, Pittsburgh, PA, and QMED, Inc, Laurence Harbor, NJ, the Edythe L. Broad and the Constance Austin Women’s Heart Research Fellowships, Cedars-Sinai Medical Center, Los Angeles, California, the Barbra Streisand Women’s Cardiovascular Research and Education Program, Cedars-Sinai Medical Center, Los Angeles, The Society for Women’s Health Research (SWHR), Washington, D.C., The Linda Joy Pollin Women’s Heart Health Program, and the Erika Glazer Women’s Heart Health Project, Cedars-Sinai Medical Center, Los Angeles, California.
