Abstract
Parkinson's disease (PD) is a neurodegenerative disease characterized by a loss of dopaminergic neurons in the nigrostriatal pathway. Apart from effective strategies to halt the underlying neuronal degeneration, cell replacement now offers novel prospects for PD therapy. Porcine embryonic neural tissue has been considered an alternative source to human fetal grafts in neurodegenerative disorders because its use avoids major practical and ethical issues. This study was undertaken to evaluate the effects of embryonic day 27 (E27) porcine mesencephalic tissue transplantation in a PD rat model using animal positron emission tomography (PET) coupled with 4-[18F]-ADAM, a serotonin transporter (SERT) imaging agent. The parkinsonian rat was induced by injecting 6-hydroxydopamine into the medial forebrain bundle (MFB) of the right nigrostriatal pathway. The apomorphine-induced rotation behavioral test and 4-[18F]-ADAM/animal PET scanning were carried out following 6-OHDA lesioning. At the second week following 6-OHDA lesioning, the parkinsonian rat rotates substantially on apomorphine-induced contralateral turning. In addition, the mean striatal-specific uptake ratio (SUR) of 4-[18F]-ADAM decreased by 44%. After transplantation, the number of drug-induced rotations decreased markedly, and the mean SUR of 4-[18F]-ADAM and the level of SERT immunoreactivity (SERT-ir) in striatum were partially restored. The mean SUR level was restored to 71% compared to that for the contralateral intact side, which together with the abundant survival of tyrosine hydroxylase (TH) neurons accounted for functional recovery at the fourth week postgraft. In regard to the extent of donor-derived cells, we found the neurons of the xenografts from E27 transgenic pigs harboring red fluorescent protein (RFP) localized with TH-ir cells and SERT-ir in the grafted area. Thus, transplanted E27 porcine mesencephalic tissue may restore dopaminergic and serotonergic systems in the parkinsonian rat. The 4-[18F]-ADAM/animal PET can be used to detect serotonergic neuron loss in PD and monitor the efficacy of therapy.
Introduction
Parkinson's disease (PD) was first described in 1817 by the British physician, James Parkinson. The loss of dopaminergic neurons, the substantia nigra and their axons, which project to the striatum, contribute the four cardinal motor symptoms of PD (tremor, rigidity, bradykinesia, and postural instability) (17,25,41,70). Allografts using fetal mesencephalic tissue is an important alternative treatment for PD based on the idea that dopamine (DA) supplied from cells implanted into the striatum can substitute for the lost dopaminergic neurons in the substantia nigra (19,37,45,71).
Transplantation of neural tissue from embryonic days 12-17 rat tissue (68), pig E27-29 (3,21,39), and 6.5- to 9-week-old human fetal ventral mesencephalic (VM) tissue (37,67) has the potential to increase striatal dopamine levels and ameliorate behavioral deficits in animal models of PD. In humans, neural xenografts not only raise new hopes and open new avenues for patients with PD but also avoid ethical constraints and create the potential for broad clinical application (59). Among different species, porcine embryonic neural tissue has long been considered to be the most suitable alternative source for transplantation into human brains due to the relative ease of pig breeding, similarities between porcine and human brain anatomy, and the potential for genetic modification of porcine tissue (71). Xenografting with E27 porcine VM tissue transplanted into the DA-depleted rat striatum is able to integrate anatomically and functionally in the striatum and produce functional recovery as measured by a reduction in drug-induced rotational asymmetry development in the immunosuppression paradigms (21,39,69).
Although the principal manifestations of PD are attributed to a decline in DA levels in the striatum, lines of evidence suggest that an impaired serotonergic system is also involved in PD (29). In a postmortem study of PD patients, Kish et al. reported that the serotonergic system was more seriously affected in the caudate nucleus than the putamen, as all the serotonin (5-HT) markers used were missing in the former (35). Serotonergic system involvement in PD has focused recently on a pivotal role in disease progression, and serotonergic neurons exert a modulatory role in neuronal transplantation (11,29,65). Taking a special interest in the functional imaging of the serotonergic system, we determined that animal PET imaging with a novel [18F]-labeled radioligand targeted to SERT (4-[18F]-ADAM; T1/2 = 110 min) (13,26,27,47,62) was capable of detecting serotonergic fiber loss (32,43), monitoring disease developing in the parkinsonian rat, and overseeing the effectiveness of allografting therapy in vivo (68).
The current study was undertaken to further investigate the effects of neural xenografting with E27 porcine mesencephalic tissue in immunosuppressed parkinsonian rats via behavioral, 4-[18F]-ADAM/animal PET imaging and immunohistochemistry (IHC) studies.
Materials and Methods
Radiopharmaceutical
The 4-[18F]-ADAM was synthesized as previously reported (52) but with slight modifications. In brief, nucleophilic fluorination of the in vitro precursor, N, N-dimethyl-2-(2,4-dinitrophenylthio) benzylamine, with K[18F]/K2.2.2 (Sigma-Aldrich, St. Louis, MO, USA) followed by reduction with NaBH4/Cu(OAc)2 (Sigma-Aldrich) and purification with high-performance liquid chromatography (HPLC; Waters Chromatography Division, Millipore Corporation, Billerica, MA, USA) produced the desired compound with ~5% radiochemical yield in a synthesis time of 120 min from the end of bombardment. The radiochemical yield of 4-[18F]-ADAM was increased to ~15% by using a different precursor (27). The chemical and radiochemical purities were >95%, and the specific activity was >3 Ci/μmol or 111 GBq/μmol.
The Parkinsonian Rat Model
The Institutional Animal Care and Use Committee (IACUC, National Defense Medical Center, Taipei, Taiwan) approved all animal study protocols. Eight-week-old male Sprague–Dawley rats (BioLASCO Taiwan Co., Ltd., Taipei, Taiwan) weighing between 280 and 300 g (mean: 290 ± 10 g) were housed in the animal facilities at the National Defense Medical Center (Taipei, Taiwan) at a constant temperature of 23 ± 2°C and a controlled light/dark cycle (light from 7:00 AM to 7:00 PM). Rats were maintained on a complete pellet diet and tap water for a week prior to the study.
The parkinsonian rat model was generated as previously reported (31,68). Briefly, male Sprague–Dawley rats were anesthetized with 7% chloral hydrate [0.5 ml/kg, intraperitoneally (IP); Riedel-de Haën, Seelze, Germany], followed by unilateral injection of 6-hydroxydopamine (6-OHDA; 20 μg in 4 μl saline containing 0.02% ascorbic acid; both Sigma-Aldrich) into the ascending nigrostriatal pathway (4.4 mm posterior to bregma, 1.2 mm lateral to the midline, and 7.8 mm below the dura).
Behavioral Testing
Two weeks after the lesion surgery, the effect of the 6-OHDA lesions was assessed by monitoring apomorphine [0.20 mg/kg dissolved in 0.1% ascorbate-saline, subcutaneously (SC); Sigma-Aldrich]-induced turning behavior over a period of 30 min (66). The drug-induced rotational response was measured in an automated rotometer system (MED Associates, Inc., St. Albans, VT, USA). The number of rotations in the ipsilateral and contralateral directions was counted and expressed as net turns per hour (the number of contralateral turns minus the number of ipsilateral turns). The parkinsonian rats (n = 12) that exhibited a net rotational asymmetry of at least four full turns per minute away from the lesioned side were selected and divided into three groups: 6-OHDA lesion group (prior to graft), mesencephalic tissue transplantation from embryonic Lanyu pig (n = 6), or transgenic pigs harboring the RFP (n = 6). At the fourth week post-E27 porcine mesencephalic tissue transplantation, the behavioral protocol was repeated. Based on previous studies, the same degree of apomorphine-induced rotational asymmetry corresponds to 90% depletion of striatal DA and 70% of dopaminergic neurons loss in the substantia nigra (66,68).
Animal PET Imaging
Two weeks following 6-OHDA lesioning and 4 weeks following E27 porcine mesencephalic tissue transplantation, 4-[18F]-ADAM (14.8-18.5 MBq; 0.4-0.5 mCi) was injected into the tail vein of parkinsonian rats used for animal PET imaging; acquisition was performed according to previous reports (47,68). In addition, six normal rats were also prepared for animal PET study. Briefly, rats were anesthetized by passive inhalation of a mixture of isoflurane/oxygen (5% isoflurane for induction and 2% for maintenance; Abbott Laboratories Ltd., Maidenhead, UK). One hour after tracer injection, animal PET imaging acquisition was started using an animal PET R4 scanner (Concorde MicroSystems, Knoxville, TN, USA). The energy window was set at 350-650 keV. The Fourier rebinning algorithm and two-dimensional filtered back projection (ramp filter with cutoff at Nyquist frequency) were employed to reconstruct the images. All images were analyzed using ASIPro VM 6.3.3.1 software (Concorde MicroSystems). The volume of interest (VOI) for the striatum and cerebellum were defined on reconstructed and summated PET images according to the MRI and rat brain atlas (51) to determine the anatomical boundaries. Considering the voxel boundaries of PET images between brain regions were not as sharp as the anatomical boundaries of MRI (42), the VOIs that were drawn manually on the reconstructed PET images were bordering within the brain structures to eliminate the partial volume effect. The SUR was expressed as (striatum – cerebellum)/cerebellum.
Mesencephalic Tissue Preparation and Transplantation
E27 porcine mesencephalic tissue was dissected from either embryonic Lanyu pig group (n = 6) (Taitung Animal Propagation Station, Livestock Research Institute, Taitung, Taiwan) (12,30,72) or transgenic pigs harboring the RFP group (n = 6) (49,53) transplanted in the lesioned striatum of PD rats. The dissection areas were selected according to the protocols of Galpern et al. (21) and Larsson et al. (40), with minor modifications as follows. The ventral half of the porcine midbrain was cut, between the rostal forebrain from the caudal rhombencephalic isthmus to include the entire VM and partially the rostral pontine raphe region. The size was about 2.0 mm2 in our measurement after VM dissection. Cyclosporin A (CyA; 10 mg/kg, IP, daily; Novartis AG, Basel, Switzerland) treatment started 2 days prior to transplantation and continued for a period of 4 weeks. Care was taken to ensure that the dissection tissue blocks contained the ventral mesencephalon and partial ventral pontine raphe, which contain both dopaminergic and serotonergic cell bodies, respectively. The tissue blocks were maintained in Hank's buffered salt solution (HBSS; Gibco, Grand Island, NY, USA) during the transplantation procedure. The grafting tissues were cut into small pieces for glass microtube (approximately 29 gage; Kimble Glass Inc., Rockwood, TN, USA) (68,75) extraction and transplanted into the ipsilateral striatum (0.5 mm posterior to bregma, 2.5 mm lateral to the midline, 5.5 mm below the dura). Each parkinsonian rat received half of a single E27 porcine mesencephalic tissue sample (~2.5 × 105 cells). All rats received both behavioral testing and animal PET imaging at the second week following 6-OHDA lesioning and the fourth week following mesencephalic tissue transplantation, then were sacrificed for IHC studies on the day after final animal PET imaging.
Immunohistochemistry
One day after the final animal PET imaging, parkinsonian rats grafted with either embryonic Lanyu pig or transgenic pig tissue harboring RFP were sacrificed for IHC studies, performed as previously reported (68,75). Briefly, the rats were deeply anesthetized with chloral hydrate prior to perfusion with normal saline followed by 4% paraformaldehyde (Sigma-Aldrich). The brains were postfixed with 4% paraformaldehyde overnight at 4°C. The tissue samples were replaced in 20% sucrose in 0.1 M PBS (pH 7.4) for 24 h and then in 30% sucrose for 24 h. These methods were used for dehydration to minimize ice crystal damage of the tissue. Coronal sections (30 μm) were first sliced by cryostat (Leica CM 3050; Leica Microsystem, Taipei, Taiwan), then rinsed with phosphate-buffered saline (PBS) and treated with 1% H2O2 (Calbiochem, Torrey Pines, CA, USA) in PBS for 0.5 h. After incubation in blocking solution [0.5% Triton X-100 (Sigma-Aldrich) and 3% normal goat serum (Vector, Burlingame, CA, USA) in PBS], sections were incubated with either rabbit anti-TH or rabbit anti-SERT antibodies (both 1:2,000 dilution; Millipore Corporation) at 4°C overnight. Thereafter, sections were rinsed with PBS and further incubated with goat anti-rabbit biotinylated IgG (1:200; Vector) for 1 h. Sections were then incubated with avidin–biotin complex (1:200; Vectastain ABC kit, Vector) for an additional hour. Subsequently, sections were visualized using 0.05% 3,3-diaminobenzidine (DAB; Sigma-Aldrich) for 6 min (TH) or 2.5 min (SERT), washed three times with PBS, and finally mounted on gelatin-coated glass slides (Thermo Fisher Scientific Inc., Waltham, MA, USA).
Additionally, the sections of RFP group were immunofluorescence stained with the same primary antibodies using the same procedure. Then the sections were also incubated with secondary antibody goat anti-rabbit IgG-conjugated FITC (1:200; Jackson Immuno Research Laboratories, West Grove, PA, USA). These sections were then stained with 4′,6-diamidino-2-phenylindole (DAPI; Thermo Fisher Scientific Inc.) to ensure the cell distribution.
The grafted area was defined based on the distribution of mesencephalic tissue in the striatum. Nine IHC sections at one-section intervals from 18 consecutive sections of the intrastriatal graft and the TH+ cell count was manually outlined using a 100× lens. Photographs of the grafted area were collected and calculated using image analysis software (ImageJ; NIH, Bethesda, MD, USA) with a color CCD camera mounted on the microscopes [OPTIPHOT-2 (10×) or MICROPHOT-FXA (100× and 200×); Nikon, Tokyo, Japan].
Statistical Analysis
The SURs of PET and TH+ cell counts from IHC-stained images in the grafted area were compared by Student's t-test for two independent groups or one-way ANOVA (analysis of variance, Bonferroni posttest) for multiple comparisons. A value of p < 0.01 was considered statistically significant. Data are presented as mean ± SD.
Results
The results of the apomorphine-induced rotation behavior of 6-OHDA-lesioned rats both prior to and 4 weeks following transplantation of E27 porcine mesencephalic tissue are depicted in Figure 1. In the parkinsonian rat, the average number of apomorphine-induced rotations was 297 ± 77 turns/hour pregrafting (>240 turn/hour), whereas the rotation number was significantly reduced to 67 ± 45 turns/h 4 weeks postgrafting (**p < 0.01).
Behavioral test of PD rat prior to and posttransplantation. Apomorphine-induced rotation behavior of 6-OHDA lesion rats both prior to and 4 weeks following E27 porcine mesencephalic tissue transplantation (**p < 0.01).
Two weeks after 6-OHDA lesions, the PET study showed that the uptake of 4-[18F]-ADAM in the 6-OHDA-lesioned hemisphere was lower compared to that in the contralateral side (Table 1, Fig. 2A). The mean SUR value of 4-[18F]-ADAM in striatal regions of the lesioned hemisphere was 44% lower than that found in the contralateral side (Table 1, Fig. 2B). At the fourth week following E27 porcine mesencephalic tissue transplantation, the uptake of 4-[18F]-ADAM significantly increased within the grafted striatum (Table 1, Fig. 2C) compared to the contralateral side. The recovery of SERT increased from 56% (at the second week after 6-OHDA lesion) to 71% (at the fourth week postgraft).
PET images of 4-[18F]-ADAM uptake in rat brain. Transverse images collected 60 to 90 min after injection of 4-[18F]-ADAM among normal distribution (A), 6-OHDA lesion rats both prior to (B) and 4 weeks following E27 porcine mesencephalic tissue transplantation (C). The arrow in (B) indicates the lesioned striatum, and the arrow in (C) indicates the grafted area. Analysis of Serotonin Transporter (SERT) Levels via PET Imaging Using 4-[18F]-ADAM as Expressed by SURs Comparing 6-OHDA-Treated (Prior to Graft) and Contralateral Striatum of Rat Brains 2 Weeks Following 6-OHDA Lesions and 4 Weeks Following Transplantation of E27 Porcine Embryonic Mesencephalic Tissue This table also demonstrates the average density of TH-ir cell in grafted area. n = 6 in normal, n = 12 in 6-OHDA lesion and 4 weeks postgraft. The values are shown as mean ± SD. p < 0.01, the 6-OHDA lesion is significantly decreased versus the contralateral side; p < 0.01, the PD rats that received E27 porcine mesencephalic tissue have markedly increased SUR value (4 weeks postgraft) compared to the 6-OHDA lesion. N/D (not detected; the striatum cannot detect endogenous TH-ir cells distribution, only the grafted TH-ir cells can be detected in striatum). N/A (not applicable; the value is not applicable in graft side/contralateral side calculation)

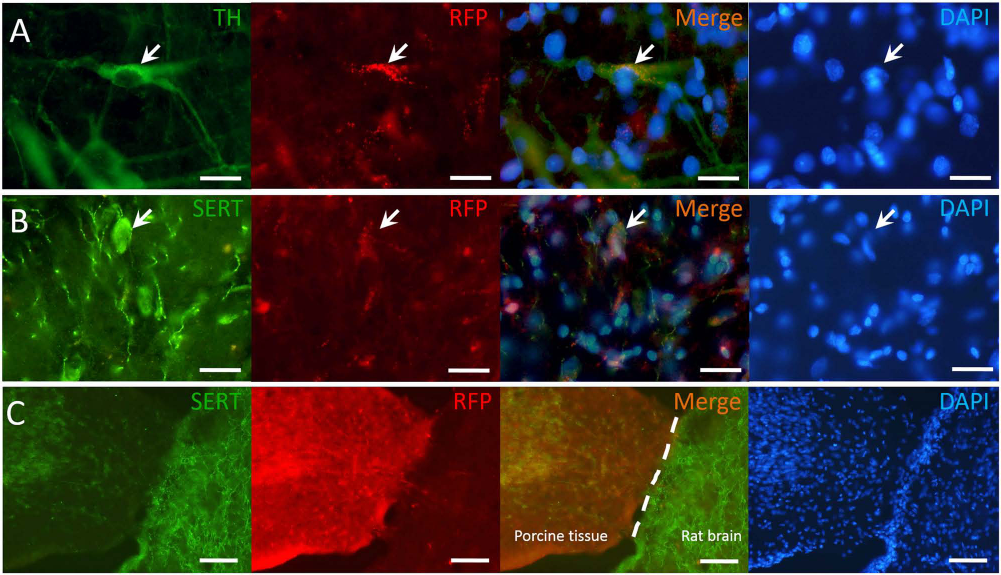
IHC results showed that the TH+ cells were found in the grafted striatum at the fourth week following E27 porcine mesencephalic tissue transplantation (Fig. 3A, B). The mean number of surviving TH cell bodies in the grafted area was 2,456 ± 450 cells, and the mean graft volume was 6.59 mm3 (Table 1). Similarly, there was also partial recovery in serotonin transporter immunoreactivity (SERT-ir) in the grafted area (Fig. 3C). Photomicrographs of SERT-ir in the grafted striatum showed abundant SERT-positive fibers with interspersion of some SERT-positive cell bodies within the grafted area (Fig. 3D, E). By contrast, SERT-positive fibers located away from the grafted area (Fig. 3H) were sparser compared to those on the corresponding contralateral side (Fig. 3E, G). Taking advantage of transgenic pigs harboring RFP, IHC photomicrographs of both TH-ir and SERT-ir in 6-OHDA-lesioned striatum following transgenic RFP E27 porcine mesencephalic tissue transplantation showed that SERT-ir fibers were found to localize in the same area with endogenous RFP (Fig. 4A, B). In the lower-magnification view, RFP of the grafted E27 porcine mesencephalic tissue was markedly distinguished from that of rat brain (Fig. 4C).
Immunohistochemical staining of PD rat with E27 porcine VM transplantation. Immunohistochemical photomicrographs of both tyrosine hydroxylase immunoreactivity (TH-ir) and serotonin transporter immunoreactivity (SERT-ir) in 6-OHDA-lesioned striatum 4 weeks following E27 porcine mesencephalic tissue transplantation. (A) TH-ir in the grafted area. (B) TH-ir cell bodies (black arrows) and TH-ir fibers in the grafted area. (C) SERT-ir in the grafted area. (D-H) SERT-ir in the stratum of the parkinsonian rat after transplantation. The SERT-ir is higher within (D) and near (F) the grafted area than that of contralateral side (E). The SERT-ir at the site far from the grafted area (H) is less than that of contralateral side (G). White arrows (D) point to SERT-ir cell bodies within the grafted area. (A, C) Scale bar: 1 mm; (B, D–H) Scale bar: 50 μm. Dopaminergic and serotonergic cells colocalized with RFP in grafted striatum of PD rat. The immunofluorescence photomicrographs of tyrosine hydroxylase immunoreactivity (TH-ir), serotonin transporter immunoreactivity (SERT-ir), and 4′,6-diamidino-2-phenylindole (DAPI) in 6-OHDA-lesioned striatum 4 weeks following E27 porcine mesencephalic tissue transplantation of transgenic pigs harboring red fluorescence protein (RFP). White arrows point to TH-ir (A) and SERT-ir (B) that are both colocalized with endogenous RFP. (C) The RFP of the grafted porcine tissue showed markedly distinct from rat brain tissue. The DAPI ensures the cell distribution. (A, B) Scale bar: 20 μm. (C) Scale bar: 200 μm.
Discussion
The results of the present study demonstrated that xenografting of E27 porcine mesencephalic tissue into the DA-depleted rat striatum is able to restore both dopaminergic and serotoninergic neurons in the brain after 6-OHDA lesioning. In the dopaminergic system, IHC results showed surviving TH cells that accompanied increased TH-ir with intrastriatal xenografting might account for partial attenuation of apomorphine-induced rotation behavior in parkinsonian rats. In serotonergic system, 4-[18F]-ADAM coupled with animal PET, and the SERT-ir was partially restored by intrastriatal xenografting in parkinsonian rats. In our previous study, the IHC (TH-ir and SERT-ir) of the sham group (administered a 0.02% ascorbic acid solution) showed no difference between the injection side and the contralateral side (68). Moreover, the contralateral side of the 6-OHDA group exhibited similar SUR and OD ratios as the sham group. Therefore, the contralateral side of the 6-OHDA group was used as the control for IHC and animal PET in this study. Endogenous RFP colocalization with both TH-ir and SERT-ir in 6-OHDA-lesioned striatum with xenografts confirms the occurrence of donor-derived partial restoration in the pig-to-rat model.
PET and single-photon emission computed tomography (SPECT) coupled with appropriate radiotracers have been used extensively to study the involvement of the nigrostriatal dopaminergic system in PD. For example, several presynaptic dopamine imaging radiotracers such as 6-[18F]-fluorodopa/PET (14,16,37,58,59), (+)-[11C]-dihydrotetrabenazine/PET (14,36), (+)-[18F]-dihydrotetrabenazine/PET (AV-133) (18,33), 123I-labeled 2b-carbomethoxy-3b-(4-[123I]-iodophenyl)tropane ([123I]-β-CIT) /SPECT (6), and a number of radioligands for postsynaptic dopaminergic receptors have been used as biomarkers for imaging PD and monitoring its response to therapy (1,8,9,50,73).
Serotonin (5-HT)-mediated neurotransmission is altered in PD and the roles of the different 5-HT receptor subtypes in disease manifestations have been investigated (29). Neuroimaging evidence for a brain serotonergic disturbance in PD is derived mainly from findings of decreased binding of various radioligands to SERT, a “marker” of serotonin neurons. Recent SPECT imaging results have found reductions in [123I]-ADAM uptake in the thalamus and striatum of parkinsonian monkeys (46) as well as PD patients (6). SPECT imaging with [123I]-β-CIT also showed a reduction of SERT density in PD midbrain (4). PET imaging with trans-(+)-1,2,3,5,6,10b-hexahydro-6-(4-([11C]methylthio)-phenyl)pyrrolo-(2,1-a)-isoquinoline ([11C](+) McN5652) (33) and 11C-labeled 3-amino-4-(2-dimethylaminomethyl-phenylsulfanyl)-benzonitrile ([11C] DASB) (24) also has shown a reduction of SERT density in the caudate and putamen of patients with PD. These results suggest that SERT density in the midbrain region may not be affected in the early stages of PD, but regionally widespread loss of brain serotonergic innervation might be a common feature of advanced PD clinically (4,24,34).
6-OHDA has been considered to be a unique dopaminergic neurotoxin that acts via the dopamine transporter to establish the PD animal model (7,66). The question of specific toxicity of 6-OHDA on catecholaminergic cells was occasionally raised when comparing the in vitro, higher concentrations of 6-OHDA administration with the in vivo, weaker intracerebral administration, which commonly guarantees the specific toxicity on the DA system (7). Truong et al. demonstrated that the injection of increasing concentrations of 6-OHDA into the medial forebrain bundle (MFB) can produce increasing extent of cell loss in the corresponding substantia nigra and well-correlated behavioral changes (66). To the best of our knowledge, no dose-dependent neurotoxicity of 6-OHDA on 5-HT system has been reported. However, it is of interest that Berger et al. demonstrated that the application of 6-OHDA into the lateral ventricles of the neonatal rats led to increasing serotonergic innervation in the striatum (5). In the adult rats, such 5-HT hyperinnervation was observed from the nigral 6-OHDA lesions (74), but a discrepancy in 5-HT innervation has been observed from the MFB-lesion rats (48,64). Several elements may explain the discrepancy regarding striatal innervation of the serotonergic fibers. First, the 5-HT striatal fibers may serve as a substitute for the DA fibers during development of the neonatal animals (5). Second, in addition to the substantia nigra, serotonergic neurons in the midbrain raphe nucleus may be responsible for the 5-HT hyperinnervation observed in the adult rats, as more than 90% of the DA terminals were degenerated after the nigral 6-OHDA lesion (73). Last, 6-OHDA is preferentially injected into the MFB, thus destroying nigral DA neurons and depleting the striatum of the DA neurotransmitter. Smith et al. demonstrated that the losses of striatal dopaminergic neurons and density of the serotonergic fibers were dependent on 6-OHDA lesions of the striatum, MFB and substantia nigra in mice, advanced at 6-OHDA MFB lesioning (63). As expected, our experiment (5 μg/μl; 20 mg in total) clearly demonstrated that partial striatal 5-HT denervation after 6-OHDA injection into the MFB is accompanied by serotonergic fibers anatomically. Similarly, Takeuchi et al. demonstrated that 6-OHDA (8 μg/μl; 8 μg in total) lesions of the MFB caused a marked decrease in serotonergic fibers in the striatum of the adult rats (64).
In our previous study, we have found that 6-OHDA lesions of the MFB affect both the dopaminergic and serotonergic systems in a monkey model of PD using [123I]-ADAM/SPECT imaging (44). After unilateral 6-OHDA lesioning of the MFB in adult rat brains, we found a 43% SERT decrease in the SUR and a 52% decrease in SERT-ir within the lesioned side 8 weeks following a single dose of 6-OHDA injected into the rat brain, suggesting that SERT expression does not recover following the lesion (68). This study clearly demonstrated that 6-OHDA was not a selective toxin for serotonergic fibers (68). After confirming the serotonergic neurotoxin of 6-OHDA and the efficacy of 4-[18F]-ADAM coupled with animal PET for imaging SERTs, we further established the allografting effects of fetal E14 mesencephalic tissue transplantation on the serotonin system in parkinsonian rats whose striatal SERT expression was restored from 57% to 77% in the SUR and 52% to 83% in SERT-ir at the fifth week postgraft (68). Subsequently, the present study demonstrated that xenografting with the E27 porcine VM tissue into the DA-depleted rat striatum was able to attenuate drug-induced rotation behavior, as well as provide partial restoration of the serotonergic system in the striatum at the fourth week postgraft, as evidenced by the 4-[18F]-ADAM/animal PET. The mean striatal SUR of 4-[18F]-ADAM was restored from 56% to 71% relative to the contralateral side. Meanwhile the abundance of surviving TH neurons also may have accounted for functional recovery at the fourth week postgraft. The current study revealed the potential impact of 4-[18F]-ADAM/PET as a clinical method for pharmaceutical research or for predicting the long-term outcome of intrastriatal fetal VM grafts in host serotonin system in PD.
Cell transplantation is one of the most practicable therapies for the treatment of PD. However, graft-induced dyskinesia (GID) poses a major obstacle to the success of cell transplantation in caudate/putamen of these patients (22,61). Politis et al. demonstrated that GID may originate from serotonin neurons that are cografted into the dopaminergic cell suspension because of the nature of the dissection process. Interestingly, buspirone, which is a 5-HT1A receptor agonist, has been shown to suppress GID in these patients, suggesting that serotonergic neurons might be involved in the etiology of GID (55,56). Following investigations for the involvement of serotonergic neurons in the appearance of GID in the rat 6-OHDA model also suggested that 5-HT neurons exert a modulatory role in the development of GID (60). Garcia et al. further characterized GID in the PD rat model after the transplantation of dopaminergic grafts containing different amounts of serotonergic neurons and showed that the impact of these neurons within the graft suspension may be minor in a parkinsonian rat in the presence of sufficient grafting dopaminergic cells (23). In the present study, we visibly validated the presence of serotonergic neurons that were cografted into the lesioned striatum at the fourth week postgraft using 4-[18F]-ADAM/animal PET. Future clinical trials of excessive inclusion of serotonergic neurons in grafted tissue may use 4-[18F]-ADAM/PET to assess the spouting of serotonergic fibers at the grafted site and monitor the effectiveness of therapy.
It is known that transplanted VM tissue, apart from DA neurons, contains additional neuronal cell types, including serotonin neurons whose cell number can vary greatly depending on the landmarks used for dissection (65). Our results showed that endogenous RFP cells localized in the same area with both TH-ir and SERT-ir in 6-OHDA-lesioned striatum over 4 weeks following E27 porcine mesencephalic tissue transplantation. Thus, our study shows that E27 porcine mesencephalic tissue grafts can be used to repair not only the dopaminergic, but also the serotonergic, system at the fourth week postgraft; these results are consistent with previous reports of porcine xenografts in the parkinsonian rat (39,40,69). It is reasonable to think that xenogafts from both mini-pig (Lanyu pig) and transgenic pigs harboring RFP demonstrated that dopaminergic and serotonergic cell bodies were found in the grafted area of the striatum. These results revealed the relative extent to which donor-derived cells were filled with RFP throughout the grafted region. Since the striatum contains dopaminergic and serotonergic fibers, we may conclude that these cells are derived from the xenografts. The main finding of this study was that the serotonergic fibers detected by 4-[18F]-ADAM can be attributed entirely to the grafted VM via endogenous RFP expression, as shown in Figure 4.
Although the brain is considered to be a site of immunological privilege for neural transplantation, immunological rejection induced by xenografts could not be avoided (2,54). The mechanisms underlying the rejection of intracerebral neural xenografts can initiate extensive host immune responses primarily mediated by T lymphocytes (20), followed by antibody-mediated immune responses (15). It is believed that immunosuppressive treatment is necessary for long-term graft survival and functional recovery (38). Grafting of tissue from a relatively distant species, such as the pig, is associated with different immunological problems compared with allogeneic transplantation into rats. CyA is an immunosuppressant that effectively inhibits immune responses against neural xenografts in rats. A previous study demonstrated that intracerebral graft survival and function can be greatly improved by CyA treatment (10). Accordingly, cessation of CyA treatment inhibited the long-term survival of xenografted tissues and the restoration of motor behavior in a PD rat model (28). Robichon et al. reported a variable degree of success of pig-to-rat xenografting; more than 40% of the grafts survived over 10 weeks (57). However, the current study examined only the short-term survival of xenografting in parkinsonian rats (4 weeks postgraft). To achieve stable long-term graft survival in discordant xenogeneic combinations, an appropriate immunosuppressive treatment or donor tissue modifications are recommended.
In conclusion, our results showed that both dopaminergic and serotonergic systems could be restored in the parkinsonian rat transplanted with E27 porcine mesencephalic tissue. Using E27 porcine mesencephalic tissue xenogafts may be optimal species for preclinical neural experimentation. Moreover, 4-[18F]-ADAM/PET can be used to detect the loss of serotonergic fibers in PD and thereby monitor the effectiveness of therapy.
Footnotes
Acknowledgments
This work was supported by the Ministry of Science and Technology of Taiwan, Grants MOST 103-2314-B-016-017-MY3. We also acknowledge technical support from the Molecular and Genetic Imaging Core supported by the National Research Program for Genomic Medicine, National Science Council, Taiwan (NSC95-3112-B-001-009 and NSC97-3112-B-010-016). We are grateful to Ms. Yu-Yeh Kuo for her technical assistance. The authors declare no conflicts of interest.
