Abstract
Short-term synaptic plasticity represents a fundamental mechanism in neural information processing and is regulated by neuromodulators. Here, using field recordings from the CA1 region of adult rat hippocampal slices, we show that excitatory synaptic transmission is suppressed by strong but not moderate activation of adenosine A1 receptors by 2-Chloro-N6-cyclopentyladenosine (CCPA) more in the dorsal than the ventral hippocampus; in contrast, both mild and strong activation of GABAB receptors by baclofen (1 μM, 10 μM) suppress synaptic transmission more in the ventral than the dorsal hippocampus. Using a 10-pulse stimulation train of variable frequency, we found that CCPA modulates short-term synaptic plasticity independently of the suppression of synaptic transmission in both segments of the hippocampus and at stimulation frequencies greater than 10 Hz. However, specifically regarding the paired-pulse ratio (PPR) and frequency facilitation/depression (FF/D) we found significant drug action before but not after adjusting conditioning responses to control levels. Activation of GABABRs by baclofen suppressed synaptic transmission more in the ventral than the dorsal hippocampus. Furthermore, relatively high (10 μM) but not low (1 μM) baclofen concentration enhanced both PPR and FF in both hippocampal segments at stimulation frequencies greater than 1 Hz, independently of the suppression of synaptic transmission by baclofen. These results show that A1Rs and GABABRs control synaptic transmission more effectively in the dorsal and the ventral hippocampus, respectively, and suggest that these receptors modulate PPR and FF/D at different frequency bands of afferent input, in both segments of the hippocampus.
Keywords
Introduction
Neuromodulation is a variety of physiological processes implicated in regulating synaptic efficacy and neuronal excitability (Katz and Edwards, 1999; Nadim and Bucher, 2014), thereby flexibly altering the flow of information in neural circuits and determining brain state and behavior (Marder et al., 2014; McCormick et al., 2020; O’Callaghan et al., 2021). A basic function of neuromodulation is to modify the strength of synapses and the properties of short-term synaptic plasticity, that is, the synaptic dynamics (Ito and Schuman, 2008; Nadim and Bucher, 2014).
Short-term synaptic plasticity is a major category of activity-dependent changes in synaptic efficacy, encompassing several phenomena of transient changes in synaptic transmission, lasting from tens of milliseconds to tens of minutes (Jackman and Regehr, 2017; Zucker and Regehr, 2002). For instance, a widely studied form of short-term synaptic plasticity is the so-called paired-pulse facilitation or depression, which consists of a change (i.e. increase or decrease, respectively) in the second versus the first response of a pair of synaptic responses evoked by pairing two stimuli (paired-pulse stimulation) applied to presynaptic fibers in fast succession. Here, we will refer to this form of short-term synaptic plasticity with the term paired-pulse ratio (PPR). The specific effect of paired-pulse stimulation, that is, facilitation or depression, and the magnitude of induced changes depends on several factors including the constitutive properties of a synapse and the specific brain region where synapses are located, the interstimulus interval, the ratio between Ca2+ and Mg2+ in the extracellular milieu, whether synapses have undergone long-term changes, the age (Dobrunz and Stevens, 1997; Dumas and Foster, 1998; Jackman et al., 2016; Manabe et al., 1993; Papatheodoropoulos and Kostopoulos, 1998; Zucker and Regehr, 2002). Another form of short-term synaptic plasticity is frequency facilitation or depression (FF/D) which is evident during short bursts of presynaptic activity of varying frequency (Abbott et al., 1997; Jackman et al., 2016; Markram and Tsodyks, 1996).
Phenomena of short-term synaptic plasticity are thought to play important roles in neural information processing performed across at a relative fast time scale, including temporal filtering, dynamic gain control, temporal selectivity, and synaptic input diversification (Dobrunz and Stevens, 1999; Lisman, 1997; Motanis et al., 2018; Rotman et al., 2011; Thomson, 2000). Furthermore, short-term synaptic plasticity is involved in processing ongoing neural activity (Klausnitzer and Manahan-Vaughan, 2008; Yang and Xu-Friedman, 2015; for recent reviews, see Abbott and Regehr, 2004; Jackman and Regehr, 2017). Therefore, the properties of short-term synaptic plasticity can critically be involved to diversify or specialize information processing in neural networks (Carrillo-Reid et al., 2015; Dayan, 2012; Giocomo and Hasselmo, 2007; Marder, 2012; McCormick and Nusbaum, 2014) and short-term synaptic plasticity may importantly be implicated in transient brain activity and related functions such as short-term memory and working memory (Devaraju et al., 2017; Le Barillier et al., 2015; Pals et al., 2020). Importantly, neuromodulation can significantly change the properties of short-term synaptic plasticity (Gonzalez-Burgos et al., 2005; Ito and Schuman, 2007; Kirby et al., 1995; Reis et al., 2019).
The hippocampus is an elongated brain structure involved in spatial and temporal navigation, memory processing and emotionality (Buzsaki and Moser, 2013; Eichenbaum et al., 2016; Gray and McNaughton, 2003). Remarkably, the functions of hippocampus are segregated along its longitudinal axis (or septotemporal axis, which corresponds to dorsal-ventral axis in rodents and anterior-posterior axis in primates). The concept of functional segregation along the hippocampus states that different segments along the hippocampus, usually represented by the dorsal and the ventral hippocampus, participate to varying degrees to hippocampus-dependent behaviors (Bannerman et al., 2014; Strange et al., 2014). More specifically, existing evidence shows that the dorsal hippocampus has an increased involvement in information processing underlying spatial learning and memory (Jung et al., 1994; Maurer et al., 2005; Moser et al., 1993), while the ventral hippocampus has been linked to anxiety-related behaviors (Bannerman et al., 2002; Kjelstrup et al., 2002; Pentkowski et al., 2006), stress-induced disfunctions and social interactions (McHugh et al., 2004; Okuyama et al., 2016). In addition to functional segregation revealed at the level of behavior, a relatively recently developed body of research shows that significant specializations exist along the longitudinal axis of the hippocampus also at the level of intrinsic neuronal network. This intrinsic diversification includes gene expression profiles (Cembrowski et al., 2016b; Dong et al., 2009; Floriou-Servou et al., 2018; Lee et al., 2017; Thompson et al., 2008), principal cell properties (Cembrowski et al., 2016a; Dougherty et al., 2012; Dubovyk and Manahan-Vaughan, 2018; Honigsperger et al., 2015; Maggio and Segal, 2009; Milior et al., 2016; Papatheodoropoulos et al., 2002), and long-term synaptic plasticity (Babiec et al., 2017; Dubovyk and Manahan-Vaughan, 2018; Grigoryan et al., 2012; Kouvaros and Papatheodoropoulos, 2016b; Maggio and Segal, 2007; Maruki et al., 2001; Milior et al., 2016; Moschovos and Papatheodoropoulos, 2016; Papatheodoropoulos and Kostopoulos, 2000a; Reis et al., 2019; Schreurs et al., 2017; Tidball et al., 2017). Moreover, remarkable dorsoventral differences have been also found in forms of short-term synaptic plasticity, namely PPR and FF/D. More specifically, dorsal versus ventral CA1 hippocampal synapses show higher scores of PPR (Babiec et al., 2017; Dubovyk and Manahan-Vaughan, 2018; Maruki et al., 2001; Milior et al., 2016; Papatheodoropoulos, 2015; Papatheodoropoulos and Kostopoulos, 2000b; Tidball et al., 2017), and the dorsal CA1 synapses prominently display FF instead of FD that characterizes the corresponding ventral synapses (Koutsoumpa and Papatheodoropoulos, 2019, 2021; Papaleonidopoulos et al., 2017).
Recent evidence shows that neuromodulation play significant roles in diversifying the functions of the local neuronal network along the hippocampus (Dubovyk and Manahan-Vaughan, 2018; Grigoryan and Segal, 2013; Maggio and Segal, 2007; Malik and Johnston, 2017; Mlinar and Corradetti, 2018; Papaleonidopoulos et al., 2018; Reis et al., 2019). Interestingly, working memory which may engage changes in short-term synaptic plasticity (Devaraju et al., 2017; Le Barillier et al., 2015; Pals et al., 2020) and is amenable to neuromodulation (Cardoso-Cruz et al., 2014; McHugh et al., 2008) may involve a distinct participation of the dorsal (posterior) and ventral (anterior) hippocampus, as recent evidence suggests (Hauser et al., 2020; Li et al., 2022). However, despite the plethora of evidence regarding dorsoventral differences in short-term synaptic plasticity, little is known regarding the actions of neuromodulation on short-term synaptic plasticity along the hippocampus. For instance, μ-opioid receptors and GABAA receptors are involved in shaping FF/D in the dorsal but not ventral hippocampus (Koutsoumpa and Papatheodoropoulos, 2019), and beta-adrenergic receptors modulate synaptic responses evoked by theta-burst stimulation only in the dorsal hippocampus (Papaleonidopoulos and Papatheodoropoulos, 2018).
Neuromodulators affect short-term synaptic plasticity mainly by regulating neurotransmitter release from presynaptic terminals (Cheng et al., 2018; Miller, 1998; Mukunda and Narayanan, 2017). In the hippocampus, transmitter release at excitatory synapses is very efficiently controlled by the neuromodulator adenosine (Cunha, 2001; Sebastião and Ribeiro, 2014) by acting at presynaptic A1 receptors (A1Rs) (Reddington et al., 1982; Sebastião et al., 1990; Thompson et al., 1992). Similarly, GABA controls excitatory synaptic transmission in the hippocampus acting at presynaptic GABAB receptors (GABABRs) (Ulrich and Bettler, 2007; Vizi and Kiss, 1998). In addition, A1Rs (Brager and Thompson, 2003; Dunwiddie and Haas, 1985; Klausnitzer and Manahan-Vaughan, 2008; Lupica et al., 1992; Trompoukis and Papatheodoropoulos, 2020) and GABABRs (Trompoukis and Papatheodoropoulos, 2020) modulate some forms of short-term synaptic plasticity in the hippocampus and other brain regions and may significantly contribute in diversify short-term synaptic plasticity along the long axis of the hippocampus. However, how adenosine receptors and GABAB receptors modulate frequency-dependent short-term synaptic dynamics in the dorsal and the ventral hippocampus remains largely unclear.
In the present study, we examined the actions of adenosine receptors and GABABRs on two forms of short-term synaptic plasticity, namely the PPR and FF/D. It should be noted that short-term synaptic plasticity is distinguished from short-term synaptic potentiation, which is an initial phase of synaptic potentiation, decays in an activity-dependent manner, can last for several minutes to hours, and is followed by a stable phase of long-term potentiation (Volianskis et al., 2015). We studied PPR and FF/D using a frequency stimulation protocol consisting of brief 10-pulse trains applied at the presynaptic fibers at different frequencies, from 0.1 to 100 Hz. PPR was studied by measuring the changes induced in the second response in a train, while the FF/D was studied by measuring the steady-state response, which was represented by the mean value of the last three responses (8th–10th). We found significant adenosine receptor-mediated and GABABR-mediated effects on basal excitatory synaptic transmission and its short-term plastic changes induced during repetitive activation.
Methods
Animals and hippocampal slice preparation
Hippocampal slices were prepared from male Wistar rats 3–4 months old. Rats were kept at the Laboratory of Experimental Animals of the Department of Medicine, University of Patras (license No: EL-13-BIOexp-04) under stable conditions of temperature (20°C–22°C) and light–dark cycle (12/12 h), and they had free access to food and water. The treatment of animals and all experimental procedures used in this study were conducted in accordance with the European Communities Council Directive Guidelines for the care and use of Laboratory animals (2010/63/EU – European Commission). Furthermore, the treatment of experimental animals and all experimental procedures have been approved by the Protocol Evaluation Committee of the Department of Medicine of the University of Patras and the Directorate of Veterinary Services of the Achaia Prefecture of Western Greece Region (reg. number: 187531/626, 26/06/2018). The number of animals that would be required in the study was determined using the G*power software. We prepared transverse 500-μm-thick slices from the dorsal and the ventral segment of hippocampus as previously described (Papatheodoropoulos and Kostopoulos, 2000a; Koutsoumpa and Papatheodoropoulos, 2019). Briefly, following decapitation under conditions of deep animal anaesthesia with diethyl-ether, the brain was removed from the cranium and placed in ice-cold (2°C–4°C) standard artificial cerebrospinal fluid (ASCF) containing, in mM, 124 NaCl, 4 KCl, 2 CaCl2, 2 MgSO4, 26 NaHCO3, 1.25 NaH2PO4 and 10 glucose. ACSF was equilibrated with 95% O2 and 5% CO2 gas mixture at a pH of 7.4. Τhen, the two hippocampi were removed from the brain and positioned on a McIlwain tissue chopper where 500-µm-thick slices were prepared by cutting hippocampus transversely to its long axis (Figure 1(a)). Slices were prepared from the two segments of the hippocampus extending between 0.5 and 3.5 mm from each end of the structure. Slices were immediately transferred to an interface type recording chamber where they were maintained at a constant temperature of 30°C±0.5°C, continuously perfused with ACSF of the same composition as above described, at a perfusion rate of ~1.5 ml/min. Slices were continuously humidified with a mixed gas consisting of 95% O2 and 5% CO2. The slices were left for at least one and a half hours to recover, and then stimulation and recording were started.

(a) Methods used to prepare dorsal and ventral hippocampal slices. Schematic drawing of the hippocampus in the rat brain and the portions of the dorsal and ventral hippocampus used to prepare slices (lines with arrowheads) transversely to the long axis of the structure are shown in the left and middle panels, respectively. In the right panel is shown a photograph of a ventral hippocampal slice illustrating the method used to stimulate Schaffer collaterals and record fEPSP (trace inside circle) in the stratum radiatum (yellow region), below stratum pyramidale (dark blue band) where pyramidal cell bodies are located. The extension of colored regions delineates the CA1 hippocampal subfield. SE, stimulation electrode; RE, recording electrode. Calibration bars: 1 mV, 5 ms. (b) Baseline measures in dorsal and ventral hippocampal slices. Input-output curves constructed by plotting fiber volley (Fv) and fEPSP as a function of stimulation current intensity (left and middle graph, respectively), and fEPSP as a function of Fv (right graph). Fv was significantly larger in dorsal than in ventral slices only at high stimulation current intensities (horizontal line in left graph; independent t-test, p < 0.05). (c) Examples of responses evoked by the stimulation frequency protocol, applied in dorsal and ventral hippocampal slices. Stimulation frequency consisted of a train of 10 pulses delivered at varying frequency. These examples illustrate synaptic responses (fEPSPs) elicited by stimulation trains delivered at three different frequencies: 5 Hz, 20 Hz, and 100 Hz. These two slices (dorsal and ventral) were obtained from the same right hippocampus of a rat. (d) Collective results, obtained under basal conditions from dorsal and ventral hippocampal slices, regarding the second and steady-state responses evoked by a stimulation train plotted as a function of stimulation frequency; the percent changes induced in the second and steady-state responses represent two forms of short-term synaptic plasticity: the paired-pulse ratio (PPR) and the frequency facilitation or depression (FF/D), respectively. The results presented in these diagrams correspond to the results for the 2nd and the average of 8th–10th responses, shown in Supplementary Figure 1 (which presents the percent changes of fEPSPs as a function of stimulation pulses). Data were obtained from 164 dorsal slices prepared from 106 rats and 140 ventral slices obtained from 92 rats. PPR ratio was significantly higher in the dorsal versus ventral hippocampus for all stimulation frequencies greater than 0.1 Hz (independent t-test, p < 0.001). Furthermore, the dorsal hippocampus showed frequency facilitation for stimulation frequencies 1–40 Hz and frequency depression at higher frequencies, while the ventral hippocampus consistently showed frequency depression; significant dorsoventral differences in FF/D were found for stimulation frequencies 1–50 Hz (independent t-test, p < 0.001). Results for additional statistical tests are given in the main text.
Electrophysiology, data processing and analysis
Evoked field excitatory postsynaptic potentials (fEPSPs) were recorded from the CA1 stratum radiatum after electrical stimulation of the Schaffer collaterals. Electrical stimulation consisted of constant current pulses of 100 μs in duration and variable amplitude (20–260 μA). We applied electrical current pulses using a home-made bipolar platinum/iridium wire electrode with a wire diameter of 25 μm and an inter-wire distance of 100 μm; wire was purchased from World Precision Instruments, USA. Recordings of fEPSPs were performed using a 7-μm-thick carbon fiber electrode (Kation Scientific, Minneapolis, USA), which was positioned 300–400 μm apart the stimulation electrode. Baseline stimulation was delivered every 30 s using a stimulation current intensity that elicited a fEPSP with a slope of about 1 mV/ms. We systematically made input–output curves between stimulation current intensity and fEPSP. We studied short-term changes of fEPSP using a frequency stimulation protocol as previously described (Koutsoumpa and Papatheodoropoulos, 2019; Papaleonidopoulos et al., 2017). Specifically, the frequency stimulation protocol consisted of a sequence of 10 consecutive pulses delivered at varying frequency between 0.1 and 100 Hz; this pattern is similar to the spike trains that normally occur in hippocampal pyramidal cells (Fenton and Muller, 1998). Stimulation trains of different frequency were applied at a random fashion during each experiment. Furthermore, consecutive trains of pulses were separated by 2-min-long intervals. We applied frequency stimulation at baseline stimulation current intensity, that is, at a stimulation current intensity producing a subthreshold fEPSP with a slope of about 1 mV/ms. In some cases, in which the first (conditioning) fEPSP in a train caused the appearance of a population spike, we slightly reduced the intensity of the stimulation current so that the fEPSP became subthreshold. Under these conditions (i.e. subthreshold conditioning fEPSP), subsequent (conditioned) fEPSPs in a train did not evoke population spike. Considering that drugs may affected the amplitude of fEPSP and that the magnitude of fEPSP significantly determines the pattern of short-term changes in conditioned fEPSPs (Koutsoumpa and Papatheodoropoulos, 2019), we applied frequency stimulation in drug condition also after adjusting conditioning fEPSP to control levels to counteract the direct effect of drugs on synaptic transmission. In this way, we can discriminate between drug actions on mechanisms of short-term synaptic plasticity and secondary drug effects on short-term synaptic plasticity through change in synaptic transmission. Signals were amplified 500 times, band-pass filtered at 0.5 Hz–2kHz using Neurolog amplifiers (Digitimer Limited, UK), digitized at 10 kHz and stored on a computer disk for offline analysis using the CED 1401-plus interface and the Signal6 software (Cambridge Electronic Design, Cambridge, UK). To quantify fEPSP, we measured the maximum slope of its initial rising phase. The effect of frequency stimulation on fEPSP was quantified as the percent change of each of the nine consecutive evoked responses with respect to the first fEPSP in a train. Steady-state response was estimated by averaging the responses evoked by the last three pulses in a train (i.e. 8th–10th). The data about fEPSP changes induced during the application of 10-pulse trains are presented either as a function of the number of stimulus pulses, in different graphs for the different stimulation frequencies (Supplementary Figures), or as a function of the stimulus frequency, only for the PPR and FF/D corresponding to second and steady-state responses, respectively (graphs in main Figures).
Drugs
The following drugs were used: the selective A1R agonist 2-Chloro-N6-cyclopentyladenosine (CCPA, 0.2–5 μM); the selective A1R antagonist 8-Cyclopentyl-1,3-dipropylxanthine (DPCPX, 150–500 nM); the selective A2AR antagonist 4-(2-[7-Amino-2-(2-furyl)[1,2,4]a][1,3,5]triazin-5-ylamino] (ZM241385, 200 nΜ); the selective agonist of GABABRs baclofen (1 and 10 μM), and the selective antagonist of GABABR 3-[[(3,4-Dichlorophenyl)methyl]amino]propyl] diethoxymethyl)phosphinic acid (CGP52432, 10 μM); the specific antagonist of N-methyl-D-aspartate (NMDA) receptors 3-((R)-2-Carboxypiperazin-4-yl)-propyl-1-phosphonic acid (CPP, 10 μΜ). Drugs were first prepared as stock solutions and then dissolved in standard medium, and bath applied to the tissue. Stock solutions of CCPA, CGP52432, CPP and baclofen were prepared in distilled water, whereas stock solutions of DPCPX and ZM241385 were prepared in dimethyl sulfoxide (DMSO) at a concentration that when diluted for bath application the final volume of DMSO was lower than 0.05%. Stock solutions in water were maintained at 4°C while solutions in DMSO were prepared in aliquots and kept at −20°C. Stock solutions were diluted in standard medium to the desired concentrations the day of the experiment. DPCPX, ZM241385, CGP52432 and baclofen were purchased from Tocris Cookson Ltd, UK; CCPA was obtained from Sigma-Aldrich, Germany.
Statistics
For statistical comparisons, we used the univariate full factorial general linear model (GLM) and the parametric two-tailed paired and independent t-test. To statistically study the action of the drugs on the synaptic transmission, we used the average value of the last 5 min under drug condition with the average value of the last 5 min of control condition, in each slice. The values in the text and figures express mean ± SEM. The number of slices and animals used is given throughout the text (slices/animals). The statistics were performed using the number of slices. The IBM SPSS Statistics 27 software package was used for all statistical analyses.
Results
Basal synaptic transmission, PPR and FF/D in the dorsal and ventral hippocampus
We compared input–output curves between the dorsal and ventral CA1 hippocampal field. Neither fiber volley (Fv) (UNIANOVA, F = 1.116, p > 0.1) nor fEPSP (UNIANOVA, F = 0.372, p > 0.5) significantly differ between the two segments of the hippocampus (Figure 1(b)). However, at relatively strong stimulation current intensities Fv was found larger in dorsal versus ventral hippocampal slices (240–260 μA, horizontal line in left graph; independent t-test at individual stimulation current intensities, F = 0.752 and F = 0.991 for 240 and 260 μA, respectively, p < 0.05). These results are similar to those reported previously (Kouvaros and Papatheodoropoulos, 2016a; Grigoryan and Segal, 2016; Milior et al., 2016). However, other studies have reported similar Fv in the two segments of the hippocampus (Kouvaros and Papatheodoropoulos, 2016b) or an increased fEPSP in the dorsal hippocampus, especially at high intensities of presynaptic stimulation (Trompoukis and Papatheodoropoulos, 2020). These discrepancies could probably result from small variations in the cutting angle that has been used in different studies to prepare hippocampal slices. The cutting angle may affect the number of fibers (expressed by Fv) that are kept intact within a slice, therefore affecting the size of Fv and fEPSP. It should, however, be noted that most studies have shown that the ratio between EPSP and Fv does not significantly differ between dorsal and ventral hippocampal slices, as also reported here.
Regarding the two forms of short-term synaptic plasticity, which we examined in this study, that is, PPR and FF/D, we found significant dorsoventral differences under basal conditions (Figure 1(c)-(d) and Supplementary Figure 1). Specifically, the dorsal hippocampus showed continuous paired-pulse facilitation across stimulation frequencies from 1 to 75 Hz (increase in PPR; paired t-test, p < 0.001). In contrast, the ventral hippocampus showed paired-pulse facilitation at stimulation frequencies 10–50 Hz (paired t-test, p < 0.001), which was significantly lower compared with the dorsal hippocampus (independent t-test, p < 0.001) and paired-pulse depression at lower (1–3 Hz) and higher (75–100 Hz) stimulation frequencies (paired t-test, p < 0.001); at 5 Hz, which signals frequency transition, we did not observe significant change in PPR (paired t-test, p > 0.05). Regarding FF/D, the dorsal hippocampus displayed significant facilitation at 1–40 Hz (paired t-test, p < 0.001) and depression at higher frequencies (50–100 Hz, paired t-test, p < 0.001). In contrast, the ventral hippocampus responded to frequency stimulation with depression of the steady-state response at 3–100 Hz but not at 20 Hz (paired t-test, p < 0.001). At the highest stimulation frequencies used (75–100 Hz), the magnitude of frequency depression was similar between the dorsal and ventral hippocampus (independent t-test, p > 0.05). The responses to the entire stimulation train delivered at different stimulation frequencies are presented in Supplementary Figure 1. These results are generally in agreement with previous observations (Koutsoumpa and Papatheodoropoulos, 2019, 2021; Miliou et al., 2021; Papaleonidopoulos et al., 2017). In should be noted that the changes induced in the conditioned fEPSPs in a train, including the second and steady-state responses, depend not only on the stimulation frequency but also on the magnitude of the conditioning (first) fEPSP (Creager et al., 1980; Dobrunz and Stevens, 1997; Harris and Cotman, 1983; Koutsoumpa and Papatheodoropoulos, 2019; Papatheodoropoulos, 2015). Therefore, some minor discrepancies in basal PPR and FF/D that may occur between studies may be due to moderately different initial stimulation conditions.
Modulation of basal synaptic transmission by endogenous adenosine
We first studied possible tonic activation of A1Rs and A2ARs by endogenous adenosine using selective receptor antagonists. We perfused slices with either 150 nM or 500 nM DPCPX. We found that 150 nM DPCPX increased fEPSP in both the dorsal (n = 32/23, paired t-test, t30 = −4.58, p < 0.05) and the ventral hippocampus (n = 21/17, paired t-test, t30 = −2.1, p < 0.05) similarly (independent t-test, t51 = −0.752, p > 0.05). Likewise, 500 nM DPCPX increased fEPSP in both the dorsal (n = 7/7, paired t-test, t6 = −2.6, p < 0.05) and the ventral hippocampus (n = 6/6, paired t-test, t5 = −2.1, p < 0.05) similarly (independent t-test, t11 = −1.95, p > 0.05). We did not find any significant difference on DPCPX effects between the two drug concentrations either in the dorsal (independent t-test, t37 = −0.887, p > 0.05) or the ventral hippocampus (independent t-test, t25 = −0.948, p > 0.05); thus, the results obtained with the two drug concentrations were pooled. Overall, we found that DPCPX significantly increased fEPSP in both the dorsal (paired t-test, t37 = −5.019, p < 0.0005) and the ventral hippocampus (paired t-test, t25 = −2.211, p < 0.05) similarly (independent t-test, t51 = 0.62, p > 0.05) (Figure 2(a), (c)). These results are consistent with previous observations (Reis et al., 2019), considering the magnitude of fEPSP to which the effect of DPCPX was studied. Application of 200 nM ZM241385 significantly increased fEPSP in the dorsal (paired t-test, t13 = −3.528, p < 0.005) but not the ventral hippocampus (paired t-test, t13 = −1.284, p > 0.05; independent t-test between the two segments of the hippocampus, F = 0.412, t26 = 2.537, p > 0.05) (Figure 2(b), (c)).

The control of synaptic transmission by adenosinergic neuromodulation differs between the dorsal and the ventral hippocampus. (a) Example fEPSP traces before and during application of the specific antagonist of A1Rs DPCPX, 150–500 nM (upper panel) and the time course of DPCPX action on fEPSP (lower panel) in dorsal and ventral hippocampal slices. (b) Example fEPSP traces before and during application of the specific antagonist of A2ARs ZM241385, 200 nM (upper panel) and the time course of ZM241385 action on fEPSP (lower panel) in dorsal and ventral hippocampal slices. (c) Blockade of A1Rs by DPCPX significantly enhances fEPSP in the dorsal and the ventral hippocampus, similarly, while blockade of A2ARs by ZM241385 significantly increased fEPSP only in the dorsal hippocampus. (d) Example fEPSP traces before and during application of 1 μM or 5 μM CCPA (upper traces) and the time course of drug action; CCPA was used at the concentrations of 0.2 μM, 1 μM and 5 μM. Calibration bars in panels (a), (b) and (d): 0.5 mV, 5 ms. Note that 0.2 μM CCPA was applied for longer time (i.e. 60 min, last 5 min shown in the two graphs) than higher drug concentrations, to reach steady state. (e) Exogenous application of CCPA produced a concentration-dependent suppression of fEPSP in both segments of the hippocampus; however, at the highest drug concentration used (5 μM), the suppression of fEPSP was significantly stronger in the dorsal than ventral hippocampus. Asterisks in (c) and (e) denote statistically significant drug effects (paired t-test, at p < 0.05), and hash symbol is denoting significant differences of drug effects between the dorsal and ventral hippocampus (independent t-test, at p < 0.05).
A1Rs control basal synaptic transmission in the dorsal and the ventral hippocampus
Then, we studied the effects of the selective A1R agonist CCPA using three different concentrations, namely 0.2 μM, 1 μM and 5 μM. The relatively lower concentrations (0.2–1 μM) fall within the range of adenosine concentrations in the brain extracellular fluid (Dunwiddie and Diao, 1994; Hagberg et al., 1987; Zetterström et al., 1982), while the relatively higher concentration (5 μM) may represent the increased brain adenosine concentration that occurs during periods of intense neuronal activity (Winn et al., 1980). We found that application of CCPA produced a concentration-dependent suppression of fEPSP in both segments of the hippocampus (Figure 2(d) & (e)). Specifically, CCPA significantly suppressed fEPSP in both the dorsal and the ventral hippocampus when applied at the concentration of 0.2 μM for 60 min (paired t-test, n = 9/5, p < 0.05 and n = 7/4, p < 0.005, in the dorsal and ventral hippocampus, respectively), when applied at the concentration of 1 μM for 35 min (paired t-test, n = 16/14, p < 0.001 and n = 18/14, p < 0.001, in dorsal and ventral hippocampus, respectively), and when applied at the concentration 5 μM for 35 min (paired t-test, n = 9/3, p < 0.001 and n = 5/3, p < 0.05, in the dorsal and ventral hippocampus, respectively). The effect of CCPA was similar in the two segments of the hippocampus for drug concentrations of 0.2 μM and 1 μM (independent t-test, p > 0.05). However, the suppressive effect of 5 μM CCPA was significantly higher in the dorsal than the ventral hippocampus (independent t-test, p < 0.01) (Figure 2(e)). The results obtained with lower CCPA concentrations, that is, 0.2 μΜ and 1 μΜ, confirm the results of a recent study (Reis et al., 2019) in which 2-chloroadenosine was used to activate A1Rs, while the higher effects of 5 μM CCPA in the dorsal compared with the ventral hippocampus are similar to those reported previously using adenosine (Lee et al., 1983).
NMDA receptors do not participate in PPR or FF/D
NMDA receptors are widely involved in phenomena of long-term synaptic plasticity (Park et al., 2014; Volianskis et al., 2015), and they may also participate in regulating forms of short-term synaptic plasticity (Bouvier et al., 2018; Davies and Collingridge, 1996; Papatheodoropoulos, 2015). Therefore, before examining the effects of adenosine receptors and GABAB receptors on PPR and FF/D, we sought to determine whether NMDA receptors are involved in these forms of short-term synaptic plasticity. We found that NMDA receptors did not significantly contribute to short-term changes of fEPSP induced during application of frequency stimulation (Figure 3). Specifically, CPP did not significantly change PPR or FF/D in either the dorsal (n = 5/3; paired t-test, p > 0.05) or the ventral hippocampus (n = 5/3; paired t-test, p > 0.05). Also, CPP did not significantly affect basal synaptic transmission either in the dorsal (paired t-test, p > 0.05) or the ventral hippocampus (paired t-test, p > 0.05).

NMDA receptors are not involved in either PPR or FF/D, in the dorsal (a) or the ventral hippocampus (b). Results for PPR and FF/D are shown under blockade of NMDA receptors by 10 μM CPP (dorsal hippocampus, n = 5/3; ventral hippocampus, n = 5/3). Blockade of NMDA receptors produced no significant change in either PPR or FF/D in either the dorsal (paired t-test, p > 0.05) or the ventral hippocampus (paired t-test, p > 0.05).
Endogenous adenosine does not modulate PPR or FF/D
Considering that endogenous adenosine controls baseline synaptic transmission more in the dorsal than the ventral hippocampus acting on A1Rs and that removal of this tonic activation of A1Rs may affect the properties of short-term synaptic plasticity, we examined whether tonic A1R activation by endogenous adenosine may also differently modulate short-term synaptic plasticity in the two segments of the hippocampus. We found that DPCPX did not significantly affect conditioned responses either in the dorsal (GLM, multivariate analysis of variance (MANOVA), F90,4276.34 = 0.846, Wilk’s Λ = 0.887, p > 0.5 and F90,4052.53 = 0.792, Wilk’s Λ = 0.889, p > 0.5, before and after the adjustment of conditioning fEPSP to control levels, respectively; see Methods) or the ventral hippocampus (GLM, MANOVA, F90,3306.47 = 1.108, Wilk’s Λ = 0.817, p > 0.2 and F90,3299.69 = 0.815, Wilk’s Λ = 0.862, p > 0.5, before and after the adjustment of fEPSP to control levels, respectively) (Figure 4, Supplementary Figure 2). We further confirmed the absence of effects of DPCPX on short-term synaptic plasticity by looking at the PPR and FF/D in the dorsal hippocampus, before (F10,461 = 0.622, p > 0.5 and F10,461 = 0.420, p > 0.5, for the PPR and FF/D, respectively) and after the adjustment of fEPSP (F10,395 = 0.466 p > 0.5 and F10,395 = 0.147, p > 0.5, for the PPR and FF/D, respectively), and in the ventral hippocampus before (F10,351 = 1.421, p > 0.1 and F10,351 = 0.919, p > 0.5, for the PPR and FF/D, respectively) and after adjusting fEPSP to control levels (F10,349 = 1.148, p > 0.1 and F10,349 = 0.617, p > 0.5, for the PPR and FF/D, respectively) (Figure 4). Similarly, application of A2AR antagonist ZM241385 (200 nM) did not significantly influenced short-term synaptic plasticity either in the dorsal (F90,2044.96 = 0.701, Wilk’s Λ = 0.814, p > 0.5 and F90,2044.96 = 0.724, Wilk’s Λ = 0.808, p > 0.5, before and after the adjustment of fEPSP to control levels, respectively) or the ventral hippocampus (F90,2044.96 = 0.934, Wilk’s Λ = 0.761, p > 0.5 and F90,2044.96 = 0.942, Wilk’s Λ = 0.759, p > 0.5, before and after the adjustment of fEPSP to control levels, respectively), (Figure 4, Supplementary Figure 3). Accordingly, 200 nM ZM241385 did not significantly affect the PPR and FF/D in the dorsal hippocampus (F10,308 = 0.183, p > 0.5 and F10,308 = 0.098, p > 0.5, for the PPR and FF/D, respectively, after response adjustment) and the ventral hippocampus (F10,308 = 0.204, p > 0.5 and F10,308 = 0.293, p > 0.5, for the PPR and FF/D, respectively, after response adjustment).

Neither A1Rs nor A2ARs tonically modulate PPR or FF/D in either the dorsal (a) or the ventral hippocampus (b). Results on PPR and FF/D are shown under blockade of A1Rs by 150–500 nM DPCPX (dorsal hippocampus, n = 18/16; ventral hippocampus, n = 17/16) or under blockade of both A1Rs and A2ARs (by 200 nM ZM241385) (dorsal hippocampus, n = 15/15; ventral hippocampus, n = 15/15). Data under drug conditions were obtained before (open triangles) and after (filled circles) adjusting fEPSP to control levels.
Then, considering the possible interaction between A1Rs and A2ARs (Cunha, 2001), we further examined the effects of DPCPX in the presence of ZM241385 and found that blockade of A2ARs did not reveal any significant effect of subsequent application of DPCPX either in the dorsal or the ventral hippocampus (GLM, MANOVA, F90,1895.75 = 0.727, Wilk’s Λ = 0.794, p > 0.5 and F90,1373.51 = 0.703, Wilk’s Λ = 0.737, p > 0.5, after the adjustment of fEPSP) (Figure 4, Supplementary Figure 3). These results were corroborated by observations on the PPR and FF/D in both the dorsal (F10,308 = 0.401, p > 0.5 and F10,308 = 0.106, p > 0.5, for the PPR and FF/D, respectively, after response adjustment) and the ventral hippocampus (F10,307 = 0.426, p > 0.5 and F10,307 = 0.453, p > 0.5, for the PPR and FF/D, respectively, after response adjustment).
A1Rs modulate PPR and FF/D in the dorsal and ventral hippocampus
Then we studied the effects of application of CCPA on short-term synaptic plasticity at three drug concentrations, 0.2 μM, 1 μM and 5 μM (Supplementary Figure 4, Supplementary Figure 5, and Supplementary Figure 6, for 0.2, 1 and 5 μM, respectively). Considering all conditioned responses in a train, without adjusting conditioning responses, we found that all CCPA concentrations significantly modulated conditioned responses both in the dorsal (GLM, MANOVA, F90,1366.73 = 2.11, Wilk’s Λ = 0.414, p < 0.001, F90,3265.78 = 2.342, Wilk’s Λ = 0.654, p < 0.001 and F90,3360.73 = 2.038, Wilk’s Λ = 0.697, p < 0.001 for 0.2 μM, n = 11/5; 1 μM, n = 24/18; and 5 μM, n = 14/6, respectively) and the ventral hippocampus (GLM, MANOVA, F90,993.70 = 1.895, Wilk’s Λ = 0.341, p < 0.001, F90,3672.72 = 1.234, Wilk’s Λ = 0.817 p < 0.05 and F90,2119.56 = 1.943, Wilk’s Λ = 0.584, p < 0.001 for 0.2 μM, n = 9/5; 1 μM, n = 26/15; and 5 μM, n = 10/6, respectively). We confirmed these results by examining the PPR and FF/D in both the dorsal and the ventral hippocampus (Figure 5). Specifically, all three CCPA concentrations significantly increased the facilitation of the PPR in the dorsal (GLM, MANOVA, F10,208 = 8.404, p < 0.001, F10,488 = 5.882, p < 0.001 and F10,502 = 2.800, p < 0.005 for 0.2, 1 and 5 μM, respectively) and the ventral hippocampus (GLM, MANOVA, F10,154 = 3.169, p < 0.005, F10,548 = 4.385, p < 0.001 and F10,319 = 4.164, p < 0.001 for 0.2, 1 and 5 μM, respectively). Similarly, all three CCPA concentrations significantly modulated FF/D in the dorsal hippocampus (GLM, MANOVA, F10,208 = 3.14, p < 0.005, F10,488 = 7.251, p < 0.001 and F10,502 = 7.505, p < 0.001 for 0.2, 1 and 5 μM, respectively). However, in the ventral hippocampus, CCPA significantly modulated FF/D at 1 μM and 5 μM (GLM, MANOVA, F10,548 = 2.689, p < 0.005 and F10,502 = 7.505, p < 0.001, for 1 and 5 μM, respectively) but not 0.2 μM (F10,154 = 0.722, p > 0.5).
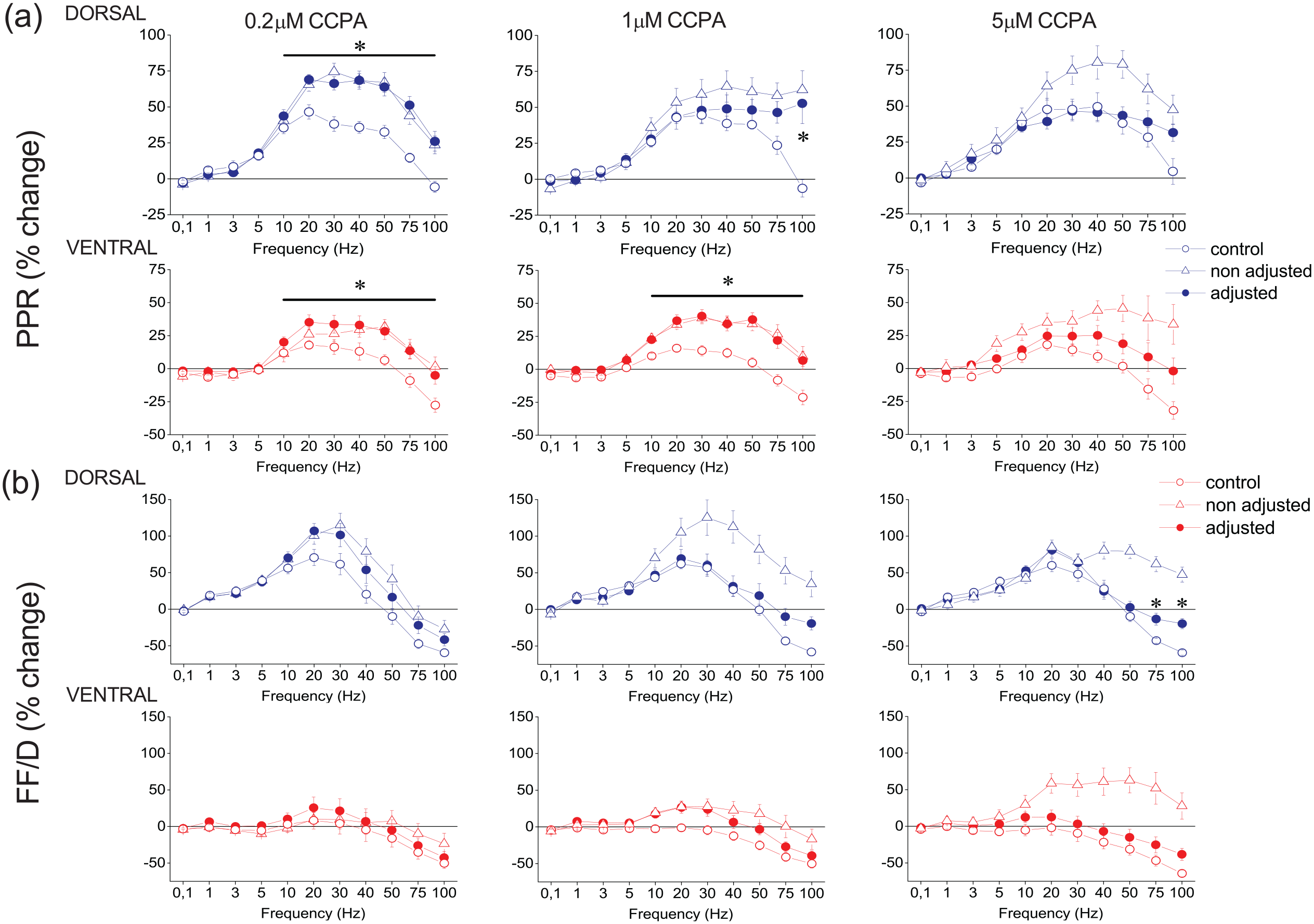
CCPA modulates PPR and FF/D in the dorsal and the ventral hippocampus. The effects of CCPA on PPR and FF/D are shown in panels (a) and (b), respectively. Data under drug conditions were obtained before (open triangles) and after (filled circles) adjusting conditioning fEPSP to control levels (after their reduction by CCPA). CCPA was applied at the concentration of 0.2 μM (dorsal hippocampus, n = 11/5 and ventral hippocampus, n = 9/5), at the concentration of 1 μM (dorsal hippocampus, n = 24/18 and ventral hippocampus, n = 26/15) and at the concentration of 5 μM (dorsal hippocampus, n = 14/6 and ventral hippocampus, n = 10/6). Asterisks indicate statistically significant differences between control and drug conditions after adjusting conditioning fEPSP (paired t-test, at p < 0.05). Note that significant drug effects occur at stimulation frequencies greater than 10 Hz. The results of the statistical comparison between control and “non-adjusted” condition are described in the main text.
The significant modulatory effect of CCPA on short-term synaptic plasticity was maintained in the dorsal hippocampus even after increasing the stimulation current intensity to counteract the depressant effect of CCPA on synaptic transmission. Specifically, after adjusting the conditioning fEPSP to control levels, CCPA significantly modulated conditioned responses in the dorsal hippocampus (GLM, MANOVA, F90,1298.90 = 2.098, Wilk’s Λ = 0.398, p < 0.001, F90,2302.69 = 1.459, Wilk’s Λ = 0.687, p < 0.005 and F90,1882.18 = 1.619, Wilk’s Λ = 0.603, p < 0.001 for 0.2 μM, n = 11/5; 1 μM, n = 24/18; and 5 μM, n = 14/6, respectively). In the ventral hippocampus, however, CCPA significantly modulated conditioned responses after adjustment of the conditioning fEPSP and at relatively lower drug concentrations, that is, 0.2 and 1 μM (F90,1149.69 = 1.587, Wilk’s Λ = 0.452, p < 0.005 and F90,2777.45 = 1.35, Wilk’s Λ = 0.748, p < 0.05 for 0.2 μM, n = 9/5 and 1 μM, n = 26/15, respectively) but not at 5 μM (F90,1570.20 = 1.238, Wilk’s Λ = 0.628, n = 10/6, p > 0.05). Furthermore, when we adjusted conditioning fEPSP to control levels, the effects of CCPA depended on both the CCPA concentration and the stimulation time. More specifically, after adjusting fEPSP, 0.2 μM and 1 μM but not 5 μM CCPA, significantly modulated the PPR in the dorsal (F10,198 = 7.370, p < 0.001 and F10,346 = 4.349, p < 0.001, for 0.2 μM and 1 μM, respectively) and the ventral hippocampus (F10,176 = 1.915, p < 0.05 and F10,416 = 3.725, p < 0.001, for 0.2 μM and 1 μM, respectively) (Figure 5). In contrast, CCPA did not significantly affect FF/D either in the dorsal (F10,198 = 1.389, p > 0.1 and F10,346 = 1.396, p > 0.1, for 0.2 μM and 1 μM, respectively) or the ventral hippocampus (F10,176 = 0.110, p > 0. 5 and F10,416 = 1.254, p > 0.1, for 0.2 μM and 1 μM, respectively). Yet, 5 μM CCPA significantly modulated FF/D in the dorsal hippocampus, at high stimulation frequencies (75–100 Hz), (F10,284 = 2.274, p < 0.05). Summarizing, we found that generally CCPA significantly modified conditioned responses in both segments of the hippocampus regardless of whether conditioning responses were adjusted or not; however, specifically regarding the PPR and FF/D we found significant drug action before but not after adjusting conditioning responses to control levels.
Modulation of basal synaptic transmission PPR and FF/D by GABABRs
Then, we examined the effects of endogenous and exogenous activation of GABABRs on synaptic transmission and short-term synaptic plasticity in the dorsal and ventral hippocampus. Figure 6(a)–(b) shows that blockade of GABABRs by 10 μM CGP52432 significantly increases fEPSP in the dorsal (n = 32/15, paired t-test, p < 0.05) but not the ventral hippocampus (n = 21/12, paired t-test, p > 0.05). In contrast, exogenous activation of GABABRs by 1 μM and 10 μM baclofen led to a greater suppression of fEPSP in the ventral compared with the dorsal hippocampus (Figure 6(a) and (c)). These data corroborated previous observations (Kouvaros and Papatheodoropoulos, 2016b; Trompoukis and Papatheodoropoulos, 2020) and suggested that excitatory synaptic transmission is tonically controlled by endogenous GABA in the dorsal hippocampus only and that under conditions of relatively enhanced activation of GABABRs, synaptic transmission is curtailed more in the ventral than the dorsal hippocampus.

Tonic GABABR activation controls synaptic transmission only in the dorsal hippocampus while the effectiveness of exogenous GABABR activation is higher in the ventral than the dorsal hippocampus. (a–b) Blockade of GABABRs by 10 μM CGP52432 increases fEPSP in the dorsal hippocampus only. (c–d) Activation of GABABRs by 1 and 10 μM baclofen suppresses fEPSP more in the ventral than the dorsal hippocampus. Calibration bars in panels (a) and (c): 0.5 mV, 5 ms. Asterisks are denoting statistically significant drug effects (paired t-test, at p < 0.05), and hash symbols are denoting significant differences of drug effects between the dorsal and ventral hippocampus (independent t-test, at p < 0.05).
Considering the existence of dorsal-ventral difference in tonic GABABR-mediated action on basal synaptic transmission, we wondered whether a difference between the two segments of the hippocampus exists also for GABABR action on short-term synaptic plasticity. Considering all conditioned responses, we found that before adjusting conditioning fEPSP to control levels, CGP52432 significantly modified short-term synaptic plasticity in the dorsal hippocampus (F90,1298.90 = 1.295, Wilk’s Λ = 0.558, n = 10/4, p < 0.05) but not the ventral hippocampus (F90,695.27 = 0.890, Wilk’s Λ = 0.477, n = 6/3, p > 0.5) (Supplementary Figure 7). However, although these results were confirmed by the drug effects on PPR and FF/D in the ventral hippocampus (F90,109 = 0.777, p > 0.5 and F90,109 = 0.525, p > 0.5, for the PPR and FF/D, respectively), they could not be confirmed in the dorsal hippocampus where none of the two responses were significantly affected by the drug (F90,198 = 0.691, p > 0. 5 and F90,198 = 1.435, p > 0.1, for the PPR and FF/D, respectively). After adjusting conditioning fEPSP, we found that CGP52432 did not significantly affect short-term synaptic plasticity either in the dorsal (F90,105.21 = 0.994, Wilk’s Λ = 0.015, p > 0.5) or the ventral hippocampus (F90,105.21 = 0.856, Wilk’s Λ = 0.024, p > 0.5) (Supplementary Figure 7 and Figure 7).

GABABRs modulate PPR or FF/D in the dorsal and the ventral hippocampus. Results on PPR and FF/D to stimulation train are shown in panels (a) and (b), respectively. Graphs arranged in columns show results obtained under blockade of GABABRs (10 μM CGP52432; dorsal hippocampus, n = 10/4 and ventral hippocampus, n = 6/3), activation of GABABRs by 1 μM baclofen (Baclofen 1 μM; dorsal hippocampus, n = 7/3 and ventral hippocampus, n = 9/3), 10 μM baclofen (Baclofen 10 μM; dorsal hippocampus, n = 11/6 and ventral hippocampus, n = 13/9), and application of 10 μM baclofen in the presence of 150 nM DPCPX (DPCPX + Baclofen 10 μM; dorsal hippocampus, n = 19/12 and ventral hippocampus, n = 20/12). Data under drug conditions were obtained after adjusting conditioning responses to control levels (after their reduction by baclofen). These results were similar to those obtained without adjusting conditioning responses (see Supplementary Figures 8 and 9). Asterisks denote statistically significant difference between control and drug conditions (paired t-test, p < 0.05).
Then, we studied the effect of exogenous activation of GABABRs on short-term synaptic plasticity in the two segments of the hippocampus. We found that application of baclofen differently modulated short-term synaptic plasticity in the dorsal and ventral hippocampus, depending on the drug concentration. Low baclofen concentration, 1 μM, did not significantly change short-term synaptic plasticity either in the dorsal (n = 7/3, F90,851.27 = 1.011, Wilk’s Λ = 0.502, p > 0.1 and F90,851.27 = 0.972, Wilk’s Λ = 0.515, p > 0.5, before and after the adjustment of fEPSP, respectively) or the ventral hippocampus (n = 9/3, F90,552.85 = 1.121, Wilk’s Λ = 0.321, p > 0.1 and F90,552.85 = 0.756, Wilk’s Λ = 0.455, p > 0.5, before and after the adjustment of fEPSP, respectively) (Supplementary Figure 8). These results were confirmed by those regarding PPR and FF/D in the dorsal (F10,132 = 0.256, p > 0.5 and F10,132 = 0.599, p > 0.5, for the PPR and FF/D, respectively, after adjustment of fEPSP) and the ventral hippocampus (F10,88 = 1.587, p > 0.1 and F10,88 = 0.083, p > 0.5, for the PPR and FF/D, respectively, after adjustment of fEPSP) (Figure 7, 1 μM).
In contrast to low baclofen concentration, high baclofen concentration (10 μM) significantly modified short-term synaptic plasticity in both the dorsal hippocampus (n = 11/6, F90,2940.23 = 1.979, Wilk’s Λ = 0.671, p < 0.001 and F90,2044.96 = 1.817, Wilk’s Λ = 0.594, p < 0.001, before and after the adjustment of fEPSP, respectively) and the ventral hippocampus (n = 13/9, F90,2791.01 = 2.077, Wilk’s Λ = 0.644, p < 0.001 and F90,2635.02 = 1.907, Wilk’s Λ = 0.652, p < 0.001, before and after the adjustment of fEPSP, respectively) by dramatically enhancing frequency facilitation or reverting frequency depression into facilitation (Supplementary Figure 9). These results were confirmed by those regarding the PPR and FF/D in both the dorsal (F10,308 = 3.643, p < 0.001 and F10,308 = 5.155, p < 0.001, for the PPR and FF/D, respectively, after adjustment of fEPSP) and the ventral hippocampus (F10,395 = 3.850, p < 0.001 and F10,395 = 5.227, p < 0.001, for the PPR and FF/D, respectively, after adjustment of fEPSP) (Figure 7, 10 μM). Eventually, exogenous activation of GABABRs by relatively high but not low baclofen concentrations significantly modulated short-term synaptic plasticity in both segments of the hippocampus, regardless of whether conditioning responses were adjusted or not. Here, we could emphasize the switching of frequency depression into facilitation across a wide range of stimulation frequencies (3–100 Hz) in the ventral hippocampus, produced by activation of GABABRs.
Finally, considering that activation of GABABRs may lead to activation of A1Rs (Zhang et al., 2003), we examined whether modulation of short-term synaptic plasticity by GABABRs interferes with activation of A1Rs. Thus, we applied 10 μM baclofen under blockade of A1Rs by 150 nM DPCPX. We observed that DPCPX did not occluded the effect of baclofen on short-term synaptic plasticity and 10 μM baclofen significantly modified short-term synaptic plasticity in both the dorsal (n = 19/12, F20,1020.0 = 2.69, Wilk’s Λ = 0.902, p < 0.001, after adjusting) and the ventral hippocampus (n = 20/12, F20,878 = 3.765, Wilk’s Λ = 0.848, p < 0.001, after adjusting) (Supplementary Figure 10). More specifically, 10 μM baclofen, in the presence of DPCPX, significantly modulated the PPR and FF/D in both the dorsal (F10,510 = 3.533, p < 0.001 and F10,511 = 3.970, p < 0.001, for the PPR and FF/D, respectively, after adjustment of fEPSP) and the ventral hippocampus (F20,440 = 4.408, p < 0.001 and F10,440 = 2.479, p < 0.001, for the PPR and FF/D, respectively, after adjustment of fEPSP) (Figure 7, DPCPX + Baclofen 10 μM). However, under blockade of A1Rs baclofen failed to eliminate the depression of high-frequency steady-state responses in the ventral hippocampus.
Discussion
In this study, we compared the effects of A1Rs, A2ARs and GABABRs on baseline synaptic transmission and short-term synaptic plasticity in the dorsal and ventral hippocampus of adult rats.
The main findings of the present study are the following:
Endogenous adenosine tonically controls synaptic transmission through A1Rs in the dorsal and ventral hippocampus, similarly, and through A2ARs in the dorsal but not the ventral hippocampus; however, endogenous adenosine does not tonically modulate PPR or FF/D in either segment of the hippocampus.
Exogenous A1R activation by high CCPA concentrations suppresses synaptic transmission more in the dorsal than the ventral hippocampus.
CCPA modulates short-term synaptic plasticity in both segments of the hippocampus independently of the suppression of synaptic transmission; yet CCPA modulates PPR but not FF/D after the depressant effect of CCPA on synaptic transmission was counteracted.
Endogenous GABABR activation tonically controls synaptic transmission in the dorsal but not the ventral hippocampus without affecting PPR or FF/D in either segment of the hippocampus.
Exogenous GABABR activation (by baclofen) suppresses synaptic transmission more in the ventral than the dorsal hippocampus and modulates PPR or FF/D in the two segments of the hippocampus, similarly, and in a A1R-independent manner.
We found that A1Rs mediate a similar tonic control of excitatory synaptic transmission in the two segments of the hippocampus, a finding that is in good agreement with previously reported observations made at a comparable level of synaptic activation (Reis et al., 2019; Trompoukis and Papatheodoropoulos, 2020). In contrast, we found that A2ARs tonically control excitatory synaptic transmission in the dorsal hippocampus only. This may sound paradox given the well-established action of A2ARs to enhance excitatory synaptic transmission (Cunha et al., 1994; Sebastião and Ribeiro, 1992); however, A2ARs also promote presynaptic GABA release (Cunha and Ribeiro, 2000), are likely involved in the anticonvulsant action of adenosine (Dunwiddie and Masino, 2001) and thus, it may contribute dampening postsynaptic depolarizations (but see also Rombo et al., 2015). Present evidence of tonic activity of A2ARs in the dorsal but not the ventral hippocampus is consistent with previous findings (Kouvaros and Papatheodoropoulos, 2016a) but is inconsistent with a previous report showing absence of tonic activity of A2ARs in the mouse hippocampus (Reis et al., 2019). This discrepancy may be related with the different species of experimental animal that have been used, since the study by Reis and colleagues was performed in mice, while the present study was performed in rats. In addition to tonic activity of endogenous adenosine, we also found that application of high CCPA concentration suppresses synaptic transmission more in the dorsal than the ventral hippocampus, confirming previous results (Lee et al., 1983) and can tentatively be explained by the higher density of A1Rs in the dorsal versus the ventral hippocampus (Lee et al., 1983; Reis et al., 2019). The present study provides the first comparative results of the effects of A1R activation on short-term synaptic plasticity in the dorsal and the ventral hippocampus. We also confirm previous observations on the higher suppressive effect of GABABR activation on synaptic transmission in the dorsal versus the ventral hippocampus (Trompoukis and Papatheodoropoulos, 2020) that corroborate histochemical data (Dubovyk and Manahan-Vaughan, 2018).
Results from previous studies have suggested that adenosinergic modulation is differentiated along the longitudinal axis of the hippocampus. Thus, in addition to an increased expression of A1Rs (Lee et al., 1983; Reis et al., 2019) and A2ARs (Reis et al., 2019) in the dorsal compared with the ventral segment of the hippocampus, some functional aspects of the adenosinergic system have also been found to differ along the septotemporal axis of the hippocampus. A1Rs control excitatory synaptic transmission more effectively in the dorsal than the ventral hippocampus (Lee et al., 1983) (and present results), contribute to resting membrane properties of CA1 pyramidal cells in the dorsal but not the ventral hippocampus (Kim and Johnston, 2015), control the induction of long-term potentiation in the ventral, not the dorsal, hippocampus (Reis et al., 2019), and they also have a higher contribution to transient heterosynaptic depression in the dorsal compared with the ventral hippocampus (Trompoukis and Papatheodoropoulos, 2020). A2Rs contribute to suppression of synaptic transmission and enhancement of neuronal excitation which is induced under coactivation of NMDA receptors and metabotropic glutamate receptor-5 in the dorsal but not the ventral hippocampus (Kouvaros and Papatheodoropoulos, 2016a), control the induction of long-term synaptic potentiation in the dorsal but not the ventral hippocampus (Reis et al., 2019) and facilitate the induction of epileptogenesis in the dorsal hippocampus under conditions of A1R blockade (Moschovos et al., 2012). Furthermore, in keeping with results from other studies (Reis et al., 2019; Trompoukis and Papatheodoropoulos, 2020), we found that endogenous adenosine tonically inhibit excitatory synaptic transmission in the dorsal and the ventral hippocampus.
Adenosine is a basic modulator of neuronal activity, implicated in several normal and pathological conditions including sleep, homeostatic synaptic plasticity, hypoxia/ischemia and epilepsy (Cunha, 2001; Dias et al., 2013; Dunwiddie and Masino, 2001; Sebastião and Ribeiro, 2014). For instance, increased release of adenosine occurs under conditions of intense synaptic activity (Lloyd et al., 1993) and intense neuronal activity associated with epileptic seizures (Schrader et al., 1980; Winn et al., 1980). Therefore, the present results that show that increased adenosine concentrations suppress excitatory synaptic transmission more in the dorsal than the ventral hippocampus may suggest that under conditions of relatively strong neuronal activation A1Rs mediate a greater curtail of synaptic transmission in the dorsal than the ventral hippocampus before the local network gets very excited. In this way, adenosine and A1Rs may act in a homeostatic manner to compensate for the increases in neuronal activity and stabilize local network activity, more in the dorsal than in the ventral segment of the hippocampus.
We found that endogenous adenosine did not tonically modulate PPR or FF/D in either segment of the hippocampus, though it modulates basal synaptic transmission. For instance, we found that DPCPX did not significantly affect PPR in dorsal or ventral hippocampal slices, while it produced an increase in basal synaptic transmission in both segments of the hippocampus. These results are indicative of a tonic activity of A1Rs, which, however, does not affect paired-pulse facilitation, and contradict findings from a previous study (Reis et al., 2019) which showed that DPCPX inhibits paired-pulse facilitation in the ventral hippocampus, where A1Rs were found to be tonically activated. The apparent discrepancy between the two studies can be interpreted in terms of transmitter release probability. Activation of A1R reduces the probability of transmitter release (Manabe et al., 1993), which is inversely related to the magnitude of paired-pulse facilitation (Dobrunz and Stevens, 1997). Therefore, tonic activation of A1Rs by endogenous adenosine is expected to increase paired-pulse facilitation. However, this effect of A1Rs may be absent if the baseline transmitter release probability is already low. For instance, in our study slices were perfused with a reduced ratio Ca2+/Mg2+, which keeps the probability of transmitter release low (Manabe et al., 1993), thereby limiting the effect of endogenous adenosine on paired-pulse facilitation, which thus remains insensitive to DPCPX. On the other hand, under conditions of high Ca2+/Mg2+ ratio, as occurs in the study by Reis et al. (2019), the baseline probability of transmitter release is relatively high allowing for a contribution of endogenous adenosine (via A1Rs) to paired-pulse facilitation, which is thus reduced by blocking A1Rs with DPCPX.
In contrast to the lack of tonic action of endogenous adenosine, activation of A1Rs by low CCPA concentrations (0.2 μM), which are equal or slightly higher than endogenous extracellular adenosine concentrations in CA1 region of rat hippocampal slices (0.14–0.2 μM) (Dunwiddie and Diao, 1994), significantly modulates PPR and FF/D suggesting that a tonic control of synaptic transmission can occur at very low ambient levels of adenosine. We found that both A1Rs and GABABRs modulate short-term synaptic plasticity in the dorsal and the ventral hippocampus, by enhancing facilitation and/or reducing depression of the conditioned responses. To some extent, these effects may result from the suppression of conditioning response produced by these receptors, given that the magnitude of synaptic facilitation is inversely related to the magnitude of the conditioning response (Creager et al., 1980; Dobrunz and Stevens, 1997; Harris and Cotman, 1983). In particular, simple forms of short-term synaptic plasticity are thought to depend mainly on presynaptic calcium-dependent mechanisms that control the probability of transmitter release and the speed of recovery from transmitter depletion (Jackman and Regehr, 2017; von Gersdorff and Borst, 2002; Zucker and Regehr, 2002). However, both A1Rs and GABABRs significantly modified short-term synaptic plasticity also after adjusting conditioning synaptic response to control levels, suggesting that activation of these receptors may directly impact on mechanisms that determine short-term synaptic plasticity in the hippocampus (Dunwiddie and Haas, 1985). In addition to presynaptic mechanisms, postsynaptic mechanisms may also be involved in some of the effects of exogenous activation of A1Rs or GABABRs. For instance, activation of these receptors in CA1 pyramidal cells leads to a hyperpolarization of the resting membrane potential through activation of G-protein-coupled inwardly rectifying potassium (GIRK) channels (Kim and Johnston, 2015; Luscher et al., 1997). A hyperpolarized resting membrane potential produced by the continuous presence of an agonist for A1Rs or GABABRs (CCPA or baclofen) can lead to an increase in driving force for flow of cation current (specifically for the sodium ion) at excitatory synapses likely resulting in an increase in response amplitude during frequency stimulation. Interestingly, in the case of A1Rs, the enhancing effect of CCPA was seen with a lower drug concentration in the dorsal compared with the ventral hippocampus; A1R-mediated activation of GIRK channels is higher in the dorsal than in ventral hippocampus (Kim and Johnston, 2015).
Extending existing evidence, we show that A1Rs can modify short-term synaptic plasticity at a wide range of agonist concentrations and in a generally similar fashion in the dorsal and the ventral hippocampus. Yet, one point to note is that though CCPA modifies short-term synaptic plasticity at both low and high concentrations, however, at high levels of CCPA, resembling adenosine concentrations that are normally seen in conditions of intense synaptic and neuronal activity (Lloyd et al., 1993; Winn et al., 1980), transmission is facilitated at the beginning but not when repetitive activity reaches a steady state. This may represent a mechanism by which adenosine signals the onset of repetitive activation of afferent input and concurrently prevents the risk of runaway excitation on local neuronal network. In contrast to A1Rs, GABABRs, which also suppress excitatory synaptic transmission in the hippocampus, require an increased activation to modify short-term synaptic plasticity, suggesting that GABABR controls the transmission of “online” information only under conditions of intense neuronal activity.
In conclusion, the present finding shows that despite significant dorsal-ventral differences in the action of A1Rs and GABABRs on baseline synaptic transmission, these receptors permit the synaptic amplification of “online” neuronal information, by means of short-term synaptic plasticity, in a similar fashion in the two segments of the hippocampus. Furthermore, these modulatory actions occur in a frequency-depended manner that differs between the two neurotransmitter receptors. A1R modifies PPR and FF/D at relatively high stimulation frequencies (>10 Hz), while GABABR modulates PPR and FF/D at stimulation frequencies greater than 1 Hz. Thus, GABABRs modulate short-term synaptic plasticity at a wider frequency range compared with A1Rs. Accordingly, a specific pattern of actions of A1Rs and GABABRs on short-term synaptic plasticity can emerge from the present results. Specifically, a wide range of ambient levels of adenosine may modulate short-term synaptic plasticity of relatively high-frequency inputs via A1Rs activation. In contrast, only intense activation of GABABRs steadily amplifies synaptic input over a wide range of frequency.
Supplemental Material
sj-eps-1-bna-10.1177_23982128221106315 – Supplemental material for Septotemporal variation in modulation of synaptic transmission, paired-pulse ratio and frequency facilitation/depression by adenosine and GABAB receptors in the rat hippocampus
Supplemental material, sj-eps-1-bna-10.1177_23982128221106315 for Septotemporal variation in modulation of synaptic transmission, paired-pulse ratio and frequency facilitation/depression by adenosine and GABAB receptors in the rat hippocampus by Maria A. Samara, George D. Oikonomou, George Trompoukis, Georgia Madarou, Maria Adamopoulou and Costas Papatheodoropoulos in Brain and Neuroscience Advances
Supplemental Material
sj-eps-10-bna-10.1177_23982128221106315 – Supplemental material for Septotemporal variation in modulation of synaptic transmission, paired-pulse ratio and frequency facilitation/depression by adenosine and GABAB receptors in the rat hippocampus
Supplemental material, sj-eps-10-bna-10.1177_23982128221106315 for Septotemporal variation in modulation of synaptic transmission, paired-pulse ratio and frequency facilitation/depression by adenosine and GABAB receptors in the rat hippocampus by Maria A. Samara, George D. Oikonomou, George Trompoukis, Georgia Madarou, Maria Adamopoulou and Costas Papatheodoropoulos in Brain and Neuroscience Advances
Supplemental Material
sj-eps-2-bna-10.1177_23982128221106315 – Supplemental material for Septotemporal variation in modulation of synaptic transmission, paired-pulse ratio and frequency facilitation/depression by adenosine and GABAB receptors in the rat hippocampus
Supplemental material, sj-eps-2-bna-10.1177_23982128221106315 for Septotemporal variation in modulation of synaptic transmission, paired-pulse ratio and frequency facilitation/depression by adenosine and GABAB receptors in the rat hippocampus by Maria A. Samara, George D. Oikonomou, George Trompoukis, Georgia Madarou, Maria Adamopoulou and Costas Papatheodoropoulos in Brain and Neuroscience Advances
Supplemental Material
sj-eps-3-bna-10.1177_23982128221106315 – Supplemental material for Septotemporal variation in modulation of synaptic transmission, paired-pulse ratio and frequency facilitation/depression by adenosine and GABAB receptors in the rat hippocampus
Supplemental material, sj-eps-3-bna-10.1177_23982128221106315 for Septotemporal variation in modulation of synaptic transmission, paired-pulse ratio and frequency facilitation/depression by adenosine and GABAB receptors in the rat hippocampus by Maria A. Samara, George D. Oikonomou, George Trompoukis, Georgia Madarou, Maria Adamopoulou and Costas Papatheodoropoulos in Brain and Neuroscience Advances
Supplemental Material
sj-eps-4-bna-10.1177_23982128221106315 – Supplemental material for Septotemporal variation in modulation of synaptic transmission, paired-pulse ratio and frequency facilitation/depression by adenosine and GABAB receptors in the rat hippocampus
Supplemental material, sj-eps-4-bna-10.1177_23982128221106315 for Septotemporal variation in modulation of synaptic transmission, paired-pulse ratio and frequency facilitation/depression by adenosine and GABAB receptors in the rat hippocampus by Maria A. Samara, George D. Oikonomou, George Trompoukis, Georgia Madarou, Maria Adamopoulou and Costas Papatheodoropoulos in Brain and Neuroscience Advances
Supplemental Material
sj-eps-5-bna-10.1177_23982128221106315 – Supplemental material for Septotemporal variation in modulation of synaptic transmission, paired-pulse ratio and frequency facilitation/depression by adenosine and GABAB receptors in the rat hippocampus
Supplemental material, sj-eps-5-bna-10.1177_23982128221106315 for Septotemporal variation in modulation of synaptic transmission, paired-pulse ratio and frequency facilitation/depression by adenosine and GABAB receptors in the rat hippocampus by Maria A. Samara, George D. Oikonomou, George Trompoukis, Georgia Madarou, Maria Adamopoulou and Costas Papatheodoropoulos in Brain and Neuroscience Advances
Supplemental Material
sj-eps-6-bna-10.1177_23982128221106315 – Supplemental material for Septotemporal variation in modulation of synaptic transmission, paired-pulse ratio and frequency facilitation/depression by adenosine and GABAB receptors in the rat hippocampus
Supplemental material, sj-eps-6-bna-10.1177_23982128221106315 for Septotemporal variation in modulation of synaptic transmission, paired-pulse ratio and frequency facilitation/depression by adenosine and GABAB receptors in the rat hippocampus by Maria A. Samara, George D. Oikonomou, George Trompoukis, Georgia Madarou, Maria Adamopoulou and Costas Papatheodoropoulos in Brain and Neuroscience Advances
Supplemental Material
sj-eps-7-bna-10.1177_23982128221106315 – Supplemental material for Septotemporal variation in modulation of synaptic transmission, paired-pulse ratio and frequency facilitation/depression by adenosine and GABAB receptors in the rat hippocampus
Supplemental material, sj-eps-7-bna-10.1177_23982128221106315 for Septotemporal variation in modulation of synaptic transmission, paired-pulse ratio and frequency facilitation/depression by adenosine and GABAB receptors in the rat hippocampus by Maria A. Samara, George D. Oikonomou, George Trompoukis, Georgia Madarou, Maria Adamopoulou and Costas Papatheodoropoulos in Brain and Neuroscience Advances
Supplemental Material
sj-eps-8-bna-10.1177_23982128221106315 – Supplemental material for Septotemporal variation in modulation of synaptic transmission, paired-pulse ratio and frequency facilitation/depression by adenosine and GABAB receptors in the rat hippocampus
Supplemental material, sj-eps-8-bna-10.1177_23982128221106315 for Septotemporal variation in modulation of synaptic transmission, paired-pulse ratio and frequency facilitation/depression by adenosine and GABAB receptors in the rat hippocampus by Maria A. Samara, George D. Oikonomou, George Trompoukis, Georgia Madarou, Maria Adamopoulou and Costas Papatheodoropoulos in Brain and Neuroscience Advances
Supplemental Material
sj-eps-9-bna-10.1177_23982128221106315 – Supplemental material for Septotemporal variation in modulation of synaptic transmission, paired-pulse ratio and frequency facilitation/depression by adenosine and GABAB receptors in the rat hippocampus
Supplemental material, sj-eps-9-bna-10.1177_23982128221106315 for Septotemporal variation in modulation of synaptic transmission, paired-pulse ratio and frequency facilitation/depression by adenosine and GABAB receptors in the rat hippocampus by Maria A. Samara, George D. Oikonomou, George Trompoukis, Georgia Madarou, Maria Adamopoulou and Costas Papatheodoropoulos in Brain and Neuroscience Advances
Footnotes
Acknowledgements
The authors thank Nicoleta Spiropoulou for animal care.
Author contributions
M.S., G.O., G.T., G.M. and M.A. performed the experiments and analyzed the data to a degree expressed by their relative position appearing in the article. C.P. designed and supervised the research, supported the data analysis, performed the statistical analysis, and prepared, wrote, and edited the whole manuscript.
Data statement
All the datasets generated and analyzed during this study are kept in the Physiology Lab, Department of Medicine, University of Patras, and they are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research has been co-financed by the European Union and Greek national funds through the Operational Program Competitiveness, Entrepreneurship and Innovation, under the call RESEARCH – CREATE – INNOVATE (project code: T2EDK – 02075).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
