Abstract
Introduction
Gardenia blue is a colorant widely used in Asia in food and beverages. The objectives of the present studies were to evaluate the maternal and prenatal embryo-fetal developmental toxicity of gardenia blue in rats and rabbits.
Methods
Sprague Dawley rats and New Zealand White rabbits were administered gardenia blue daily by oral gavage at doses of 0 (deionized water vehicle), 500, 1000 or 2000 mg/kg/day on Gestation Days 6 through 20 (rats) and 7 through 28 (rabbits). Endpoints evaluated included clinical observations, body weight, food consumption, thyroid hormones (rats), thyroid weights and histopathology (rats), gross pathologic changes, ovarian and uterine observations, fetal weight and anogenital distance (rats) and fetal morphology (external, visceral and skeletal).
Results
Treatment related maternal findings attributed to the blue/dark color of the test substance included body surface staining, and dark/blue discoloration of the kidneys, gastrointestinal track and mesenteric lymph nodes at all or most doses in the rat and/or rabbit. Slight reductions in food consumption without effects on body weight were also observed in rats at all doses and in rabbits at 2000 mg/kg/day. There were no treatment related effects on maintenance of pregnancy, postimplantation loss, litter size, fetal weight and anogenital distance, or fetal external, visceral, or skeletal malformations and variations.
Conclusions
Based on these results, the maternal and developmental no-observed-adverse-effect level for gardenia blue in rats and rabbits was ≥2000 mg/kg/day.
Keywords
Introduction
Gardenia blue is a colorant widely used in Asia for food use in frozen desserts, confections and baked goods, jams, noodles, beverages, wine and liqueurs, processed seafood and agricultural products. Gardenia blue is a dark purple to blue colorant prepared as a powder from the ripe fruit of Gardenia jasminoides Ellis, an evergreen shrub known for its distinctive white flowers. The Gardenia genus is widely distributed in warm to tropical regions and is cultivated for food colorant and medicinal purposes in Southern China, Taiwan, Philippines and Vietnam.
Decoctions of gardenia fruit based on their content of geniposide and genipin have a long history of use in traditional oriental medicine for treatment of inflammation, jaundice, headache, edema, fever, hepatic disorders, hypertension, conjunctivitis, skin ulcers, cancer, etc.,1–3 as well as possessing pharmacological properties such as the inhibition of liver apoptosis, neuroprotective effects, and anti-inflammatory activity,.4–7 The gardenia fruit contains 7–8% geniposide and trace amounts of genipin.
The principal coloring component of gardenia blue is a high molecular weight polymer formed by polymerization of the aglycone from the gardenia fruit and peptides. It is produced commercially by adding β-glucosidase to a mixture of iridoid glycosides extracted from the fruits of Gardenia jasminoides Ellis (Gardenia augusta Merrill) to yield genipin, the aglycone of geniposide. The structural unit of the gardenia blue polymer is comprised of two genipin moieties containing peptide side chains crosslinked via an alkene chain (Figure 1). Gardenia blue polymer has a mean molecular weight range of approximately 15,000–30,000 Da. Chemical structure of gardenia blue.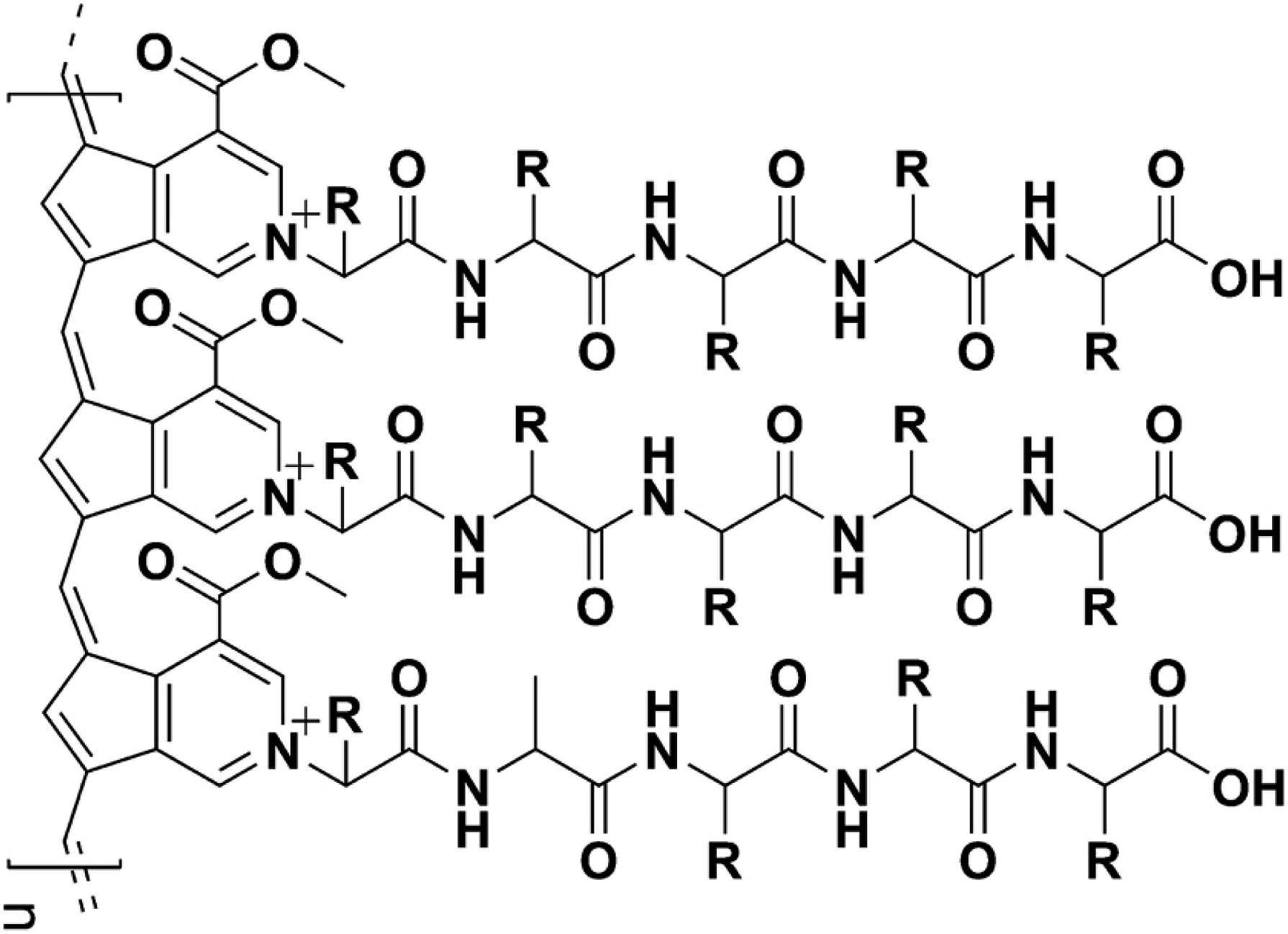
Gardenia blue is currently permitted in food in several Asian countries including China, Taiwan, Korea, and has had over 30 years of use in Japan. Contemporary interest in replacing artificial food colorants with naturally derived colorants prompted conduct of safety assessment studies prior to planned global marketing of gardenia blue as a food colorant. The objectives of the present studies were to evaluate the maternal and prenatal embryo-fetal developmental toxicity of gardenia blue colorant following oral gavage administration in Sprague Dawley rats and New Zealand White rabbits.
Materials and methods
Regulatory compliance
The design of these studies was based on the United States Environmental Protection Agency (EPA) Health Effects Test Guidelines OPPTS 870.3700 8 and the Organisation of Economic Co-operation and Development Guidelines (OECD) for the Testing of Chemicals Guideline 414. 9 These studies were conducted at Charles River Laboratories Ashland, LLC, Ashland, OH, USA, in accordance with U.S. Food and Drug Administration Good Laboratory Practice Regulations 10 as accepted by Regulatory Authorities throughout the European Union (OECD), Japan and other countries that are signatories to the OECD Mutual Acceptance of Data Agreement.11–15 These studies were approved by the Charles River Laboratories Institutional Animal Care and Use Committee.
Test substance and formulation
Gardenia blue powder (Lot Number 180910; San-Ei Gen F.F. I., Inc, Osaka, Japan) consisting primarily of 32.3% gardenia blue color (CAS:106441-42-3), 62.7% maltodextrin, 3.2% water and 1.8% other was used for these studies. Genipin was not detected in the powder.
The dose formulations were prepared weekly in deionized water (DI) and were not adjusted for purity. Previous analyses demonstrated that the test substance formulations were stable for at least 10 days at the concentrations employed and that the test substance was soluble in the vehicle when prepared under the same mixing conditions up to 200 mg/mL. The first and last dose formulation preparations were analyzed for concentration. The concentration of gardenia blue in the formulations was determined by high performance liquid chromatography (HPLC) using ultraviolet (UV) absorbance detection.
Animal husbandry
Time-mated female Crl:CD (SD) Sprague Dawley rats were received from Charles River Laboratories, Inc., Raleigh, NC on Gestation Day (GD) 1, 2, 3, or 4. The rats were approximately 11–13 weeks old and weighed between 226 and 292 g at the initiation of dosing. Time-mated female New Zealand White rabbits were received from Envigo Global Services, Inc., Denver, PA on Gestation Days 2, 3, or 4. The rabbits were approximately 7 months old and weighed between 2823 and 3662 g at the initiation of dosing. Rats were group housed, 2–3 animals of the same gestation day and dosing group together, in solid-bottom cages containing Bed-O-Cob®. Rabbits were single housed in stainless steel cages with perforated flooring elevated above ground corncob Pel-O’Cobs® bedding. The use of time-mated animals purchased from suppliers is standard practice for the conduct of rat and rabbit prenatal developmental toxicity studies, but precludes the allowance of standard acclimations periods. All animals were examined by the veterinary staff to assure acceptable health and condition prior to placement on study.
Purina Certified 5002 meal diet (Ralston Purina Co.) was provided ad libitum for rats; Certified Rabbit LabDiet® 5322 pelleted diet (PMI Nutrition International, LLC) was provided to rabbits in gradual increments during the period prior to dosing and at 180 g/day during the study. Municipal tap water treated by reverse osmosis and ultraviolet irradiation was provide ad libitum. Environmental temperature, humidity, air flow and lighting were in compliance with USDA Animal Care Animal Welfare Act and Animal Welfare Regulations, 16 and animals were handled and treated according to the Guide for the Care and Use of Laboratory Animals. 17 All animals were placed on study after an appropriate acclimation period and an acceptable physical evaluation.
Study design
Study design elements.

aThyroid hormones triiodothyronine (T3), thyroxine (T4) and thyroid stimulating hormone (TSH).
Thyroid hormones
Prior to necropsy on the morning of GD 21 blood samples were collected from the jugular vein of rats and allowed to clot for at least 30 min before centrifugation. Serum samples were analyzed for total T3 and T4 using validated ultra high performance liquid chromatography with dual mass spectroscopy. 19 Samples analyzed for TSH concentrations were analyzed using a validated Luminex Bead-Based (TSH) assay. 20 Assessment of thyroid hormones was consistent with the expectations of OECD 414 Guidelines and screening for potential endocrine effects. 9
Thyroid weight and histology
The thyroid glands of rats were excised at necropsy and weighed (post-fixation in 10% neutral buffered formalin). Thyroids from all animals in all groups were embedded in paraffin, sectioned, mounted on glass slides, stained with hematoxylin and eosin and examined histologically by a board-certified veterinary pathologist.
Necropsy and uterine examination
Following completion of the dosing periods, all surviving animals were weighed and euthanized by carbon dioxide inhalation (rats) or by an intravenous injection of sodium pentobarbital via the marginal ear vein (rabbits). All females were subjected to a gross necropsy. Laparohysterectomies and macroscopic examinations were performed blind to treatment group. The cranial, thoracic, abdominal, and pelvic cavities were opened and the contents examined. The uterus of each dam was excised and its adnexa trimmed. Corpora lutea were also counted and recorded. Gravid uterine weights were obtained and recorded. The uterus of each dam/doe was opened and the number of viable and nonviable fetuses, early and late resorptions, and total number of implantation sites were recorded, and the placentae were examined. The individual uterine distribution was documented using the following procedure: all implantation sites, including early and late resorptions, were numbered in consecutive fashion beginning with the left distal uterine horn, noting the position of the cervix and continuing from the proximal to the distal right uterine horn. Uteri which appeared nongravid by macroscopic examination were opened and placed in a 10% ammonium sulfide solution for detection of early implantation loss. 21
Fetal examination
Fetal examinations were conducted without knowledge of treatment group. External, internal, and skeletal fetal findings were recorded as either developmental variations (alterations in anatomic structure that are considered to have no significant biological effect on animal health or body conformity and/or occur at high incidence, representing slight deviations from normal), malformations (those structural anomalies that alter general body conformity, disrupt or interfere with normal body function, or may be incompatible with life), or incidental (minor changes in coloration, mechanical damage to specimen, etc.). Each viable fetus was examined in detail, sexed, weighed, tagged, and euthanized by a subcutaneous injection of sodium pentobarbital in the scapular region. Following euthanasia, anogenital distance was measured for all viable rat fetuses. The absolute and normalized (relative to the cube root of fetal body weight) values were reported. For late resorptions (advanced degree of autolysis), the degree of autolysis was recorded, a gross external examination performed (if possible), and the tissue was discarded. The sex of all fetuses was confirmed by internal examination. Approximately one-half of the rat fetuses and all rabbit fetuses in each litter were examined for visceral anomalies by dissection in the fresh (non-fixed) state. The thoracic and abdominal cavities were opened and dissected using a technique described by Stuckhardt and Poppe. 22 This examination included the heart and major vessels. Fetal kidneys were examined and graded for renal papillae development. 23 The heads from rat fetuses were removed and placed in Harrison’s fixative for subsequent processing and soft-tissue examination using the Wilson 24 sectioning technique. The heads from rabbit fetuses were examined by a mid-coronal slice. Approximately one-half of rat fetuses and all rabbit fetuses from each litter were eviscerated and fixed in 100% ethyl alcohol. Following fixation in alcohol, fetuses were macerated in potassium hydroxide and stained with Alizarin Red S 25 and Alcian Blue 26 and examined.
Statistical analysis
All statistical tests were conducted at the 5% significance level. All pairwise comparisons were conducted using two sided tests and were reported at the 1% and 5% levels, unless otherwise noted. For weights, thyroid hormone values, litter means and anogenital distance, Levene’s test was used to assess the homogeneity of group variances. The groups were compared using an overall one-way ANOVA F-test if Levene’s test was not significant or the Kruskal-Wallis test if it was significant. If the overall F-test or Kruskal-Wallis test was found to be significant, then pairwise comparisons were conducted using Dunnett’s or Dunn’s test, respectively. For ovarian and uterine content data and percent litter incidence data, the groups were compared using an overall Kruskal-Wallis test. If the overall Kruskal-Wallis test was found to be significant, then the above pairwise comparisons were conducted using Dunn’s test.
Results
Formulation analyses
The analyzed dosing formulations contained 100%–108% of the test substance for the rat formulations and 100%–110% for the rabbit formulations, which were within the protocol-specified range of target concentrations for solutions (90%–110%). The test substance was not detected in the analyzed vehicle formulations that was administered to the control groups.
Rat prenatal developmental toxicity
Maternal, reproductive, and fetal data from rats given gardenia blue.
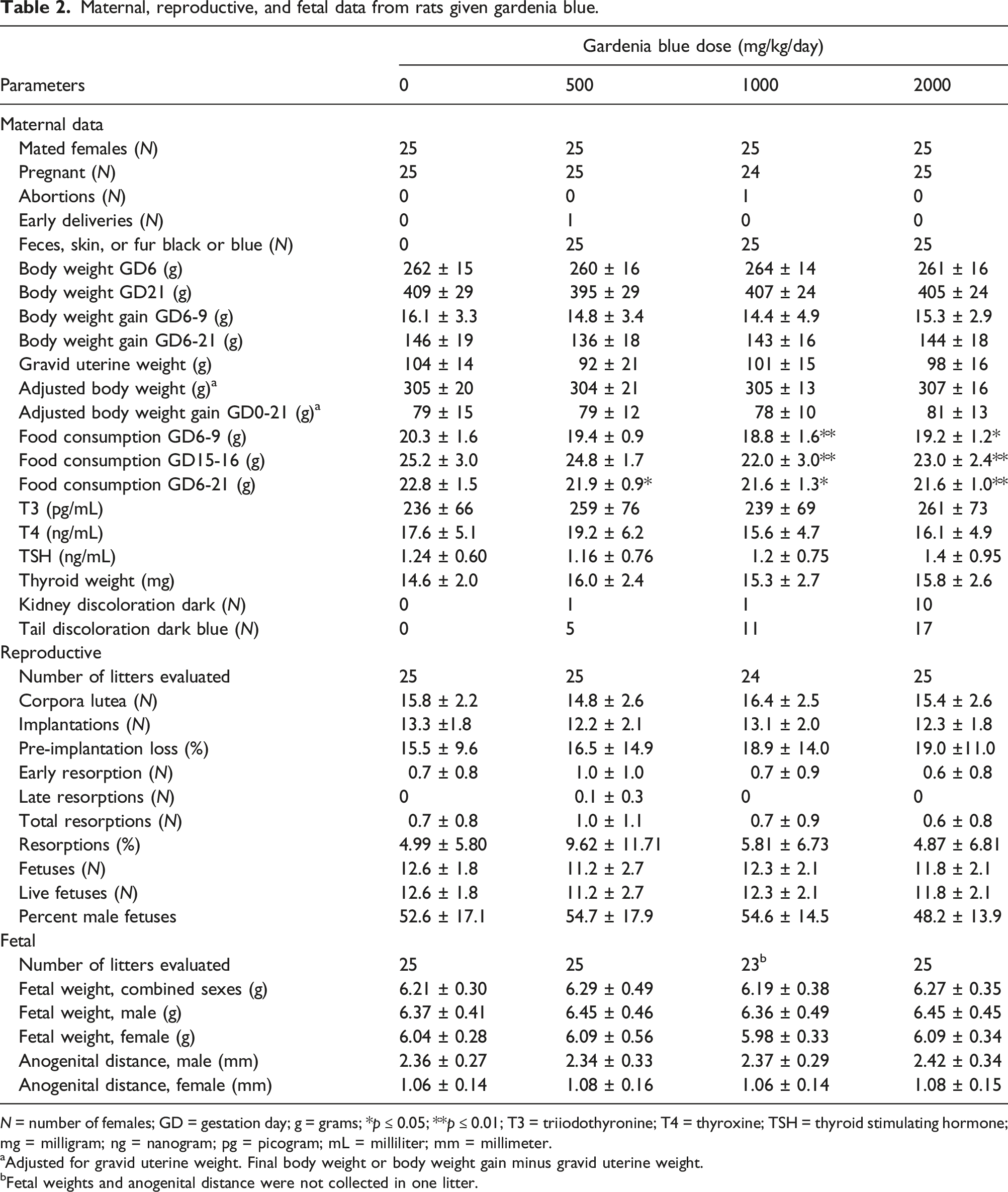
N = number of females; GD = gestation day; g = grams; *p ≤ 0.05; **p ≤ 0.01; T3 = triiodothyronine; T4 = thyroxine; TSH = thyroid stimulating hormone; mg = milligram; ng = nanogram; pg = picogram; mL = milliliter; mm = millimeter.
aAdjusted for gravid uterine weight. Final body weight or body weight gain minus gravid uterine weight.
bFetal weights and anogenital distance were not collected in one litter.
All other females survived to the scheduled necropsy. Test substance-related clinical observations, attributed to the color of the test substance, were noted at the daily examinations (Table 2). These observations consisted of black feces for all females in all test substance-treated groups during Gestation Days 7–21, and dry blue cage material for a few females in the 500 and 1000 mg/kg/day groups and a majority of females in the 2000 mg/kg/day group during Gestation Days 7–14. In addition, blue fur and skin staining on various body areas were noted in the 1000 and 2000 mg/kg/day groups at the daily examinations. Because these clinical observations were attributed to the color of the test article they were not considered adverse. No other test substance-related clinical observations were noted.
Mean maternal body weights, body weight gains, adjusted body weights, adjusted body weight gains, and gravid uterine weights were unaffected by test substance administration (Figure 2, Table 2). A statistically significantly lower mean body weight gain was noted at 1000 mg/kg/day on Gestation Day 15–16 compared to the control group; however, this difference was transient and not noted in a dose-related manner, and therefore was not considered test substance-related. Body weights of female rats during gestation. No significant differences in body weight were noted.
Slightly lower mean food consumption was noted following the initiation of dosing (Gestation Days 6–9) in all groups administered gardenia blue compared to the control group; differences were statistically significant in the 1000 and 2000 mg/kg/day groups but were not noted in a clear dose-related manner (Table 2). Mean food consumption in all test substance-treated groups was generally comparable to the control group during Gestation Days 9–15. Lower mean food consumption was noted again in the 500, 1000, and 2000 mg/kg/day groups during Gestation Days 15–21; differences were generally statistically significant in the 1000 and 2000 mg/kg/day group and was statistically significant on Gestation Day 19–20 in the 500 mg/kg/day group. Body weights of female rabbits during gestation. No significant differences in body weight were noted.
When the entire dosing period (Gestation Days 6–21) was evaluated, statistically significantly lower mean food consumption was noted in all test substance-treated groups compared to the control group. The effects on food were considered test substance-related but not adverse because there were no effects on mean body weights or body weight gains at any dosage level.
Mean maternal T3, T4, and TSH concentrations were unaffected by test substance administration (Table 2). Differences from the control group were slight, not statistically significant, and/or not noted in a dose-related manner.
Test substance-related gross pathology findings were noted in the kidneys and the tail. Dark discoloration of the kidney (entire kidney or cortex) and blue coloration of the tail were noted in all test substance-treated groups and were considered test substance-related (Table 2). The findings in the kidney and tail were considered non-adverse and due to the physical nature of the test substance and not a direct toxic effect of the test substance. No other test substance-related gross findings were noted.
No test substance-related organ weight changes were noted. Thyroid gland weights were not statistically different from their respective controls (Table 2).
No test substance-related microscopic findings were noted in the thyroid glands. The microscopic findings observed were considered incidental, of the nature commonly observed in this strain and age of rats, and/or were of similar incidence and severity in control and treated animals.
Intrauterine growth and survival were unaffected by test substance administration (Table 2). Parameters evaluated included mean litter proportions of postimplantation loss, mean number of live fetuses, mean fetal body weights, and fetal sex ratios. Differences from the control group were slight and not statistically significant. In addition, mean anogenital distances (absolute and normalized to the cube root of body weight) in the 500, 1000, and 2000 mg/kg/day groups were unaffected by test substance administration. Mean numbers of corpora lutea and implantation sites and the mean litter proportions of pre-implantation loss were similar across all groups.
Rat fetal morphology.
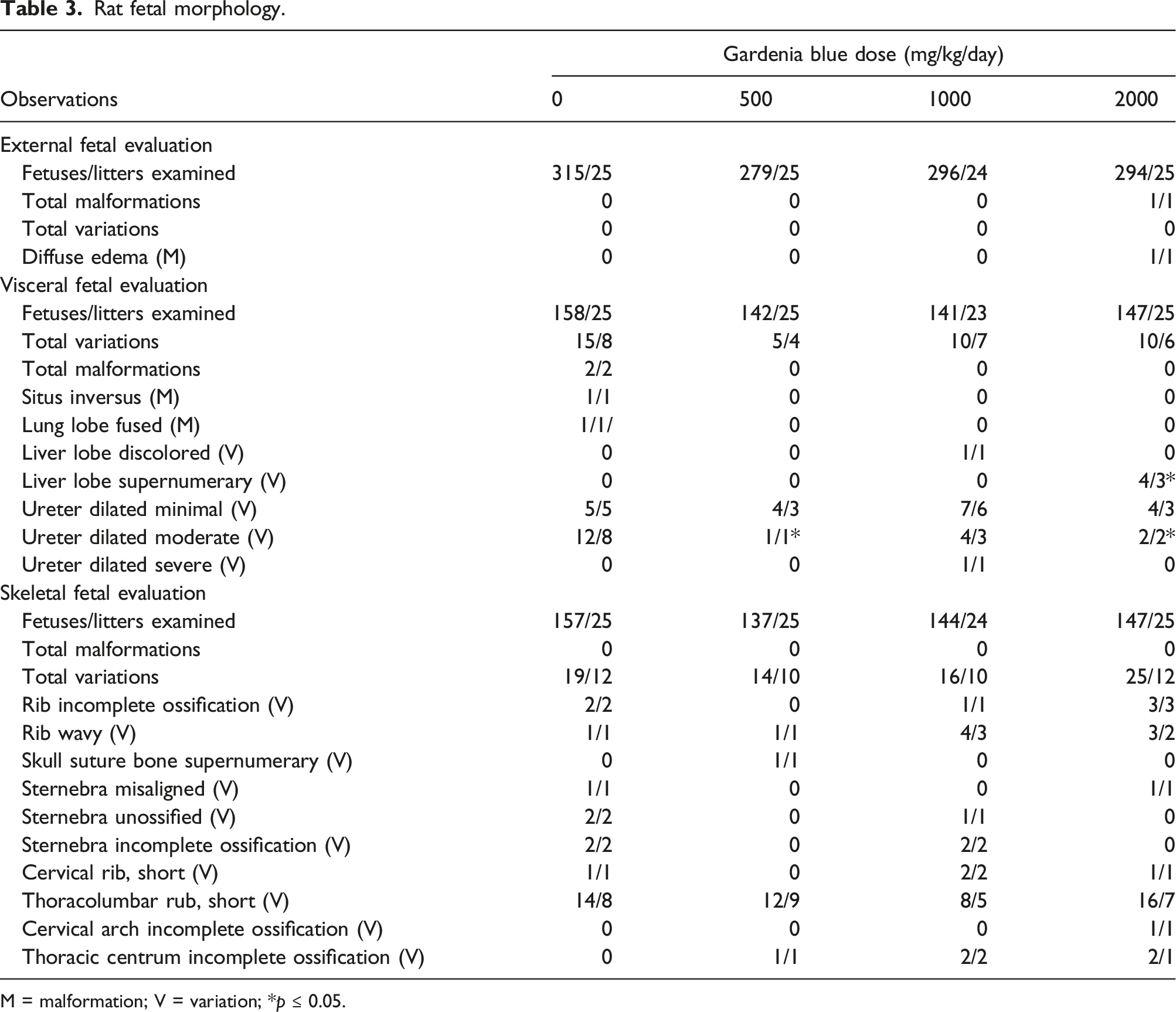
M = malformation; V = variation; *p ≤ 0.05.
No test substance-related external, visceral, or skeletal malformations or variations were noted in rat fetuses in this study (Table 3). External malformations were limited to edema of the entire subcutis in one fetus from the 2000 mg/kg/day group, which was not considered test substance-related because it was noted for a single fetus. No external developmental variations were observed. For visceral malformations, one fetus in the control group was noted with situs inversus and a second control fetus was noted with fused lobes of the lungs. No visceral malformations were noted in the other treatment groups. For visceral variations, a higher mean litter proportion of supernumerary lobes of the liver (2.38% per litter) was noted at 2000 mg/kg/day compared to the concurrent control group (0.00% per litter); however, the value was within the range of historical control values (0.000%–4.190% per litter) for the laboratory (Charles River Ashland, OH). In addition, the mean litter proportion of moderate dilation of the ureter in the 500 and 2000 mg/kg/day groups was statistically significantly lower than the concurrent control group. The lower incidences in the treatment groups were not considered toxicologically significant. Other developmental visceral variations in the test substance-treated groups were noted infrequently, similarly in the control group, were not noted in a dose-related manner, the mean litter proportions were not statistically significant compared to the concurrent control group, and/or the values were within the range of normal background historical control data. Findings for skeletal malformations and variations observed in the test substance-treated groups did not occur (malformations) or occurred infrequently, similar in incidence to the control group, did not occur in a dose-related manner, were not statistically significant compared to the concurrent control group, and/or within the range of historical control data.
Rabbit prenatal developmental toxicity
Maternal, reproductive, and fetal data from rabbits given gardenia blue.
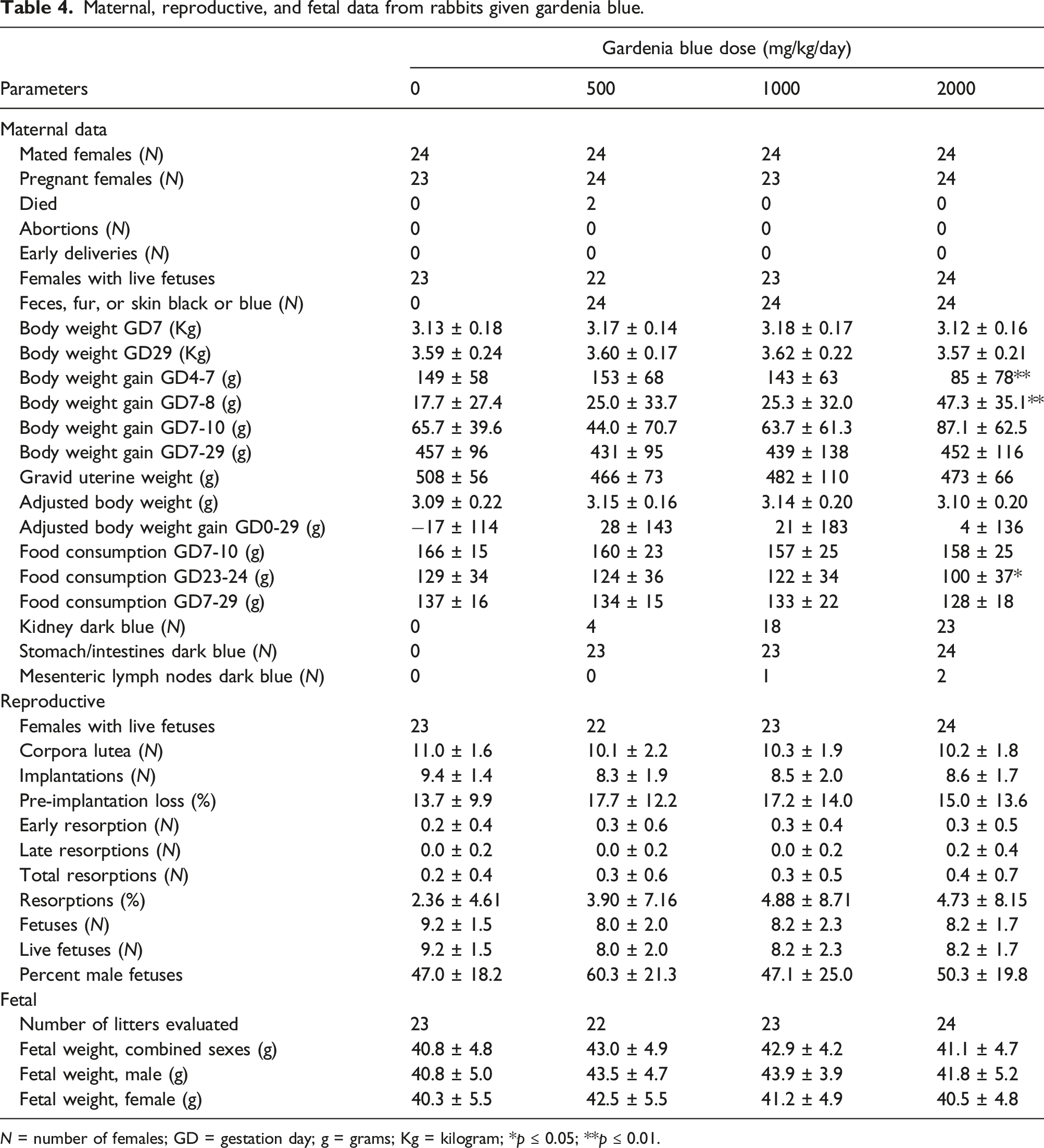
N = number of females; GD = gestation day; g = grams; Kg = kilogram; *p ≤ 0.05; **p ≤ 0.01.
Test substance-related clinical observations, related to the color of the test substance, were noted in all test substance-treated groups (Table 4). All animals in the gardenia treated groups were noted with blue and black feces beginning on Gestation Day 8 and generally lasting through the entire dosing period. In addition, blue staining of various body surfaces (primarily on the head, forepaws, hindpaws, tail and urogenital area) was observed in animals from the 500, 1000, and 2000 mg/kg/day groups generally throughout the dosing period. These clinical observations were related to the color of the test substance and were not considered adverse. All other findings noted occurred in single animals, at a similar incidence in the controls, and/or did not present in a dose-related manner.
Mean body weights and body weight gains were unaffected by test substance administration (Figure 3; Table 4). Differences from the control group were slight, transient, generally not statistically significant, and/or not dose responsive. In the 2000 mg/kg/day group, the statistically significant higher mean body weight gain following the first dose (Gestation Days 7–8) was considered compensatory to the statistically significantly lower mean body weight gain observed prior to the start of dosing (Gestation Days 4–7). Mean adjusted body weights, adjusted body weight changes, and gravid uterine weights in the 500, 1000, and 2000 mg/kg/day groups were unaffected by test substance administration. Differences from the control group were slight, not dose-responsive, and/or not statistically significant.
Mean food consumption in the 2000 mg/kg/day group was comparable to the control group during Gestation Days 7–16, but was slightly lower than the control group throughout the remainder of the dosing period (Table 4); the difference was statistically significant on Gestation Day 23–24. This lower food consumption was considered test substance-related, but non-adverse, as there were no corresponding effects on mean body weight. Mean maternal food consumption in the 500 and 1000 mg/kg/day groups was unaffected by test substance administration.
At the scheduled necropsy on Gestation Day 29, dark blue discoloration of the kidney was observed in a dose-responsive manner for females administered gardenia blue (Table 4). With the exception of 1 female each in the 500 and 1000 mg/kg/day groups, all females in all test substance-treated groups were noted with dark blue discoloration of the stomach and/or intestines (appendix, cecum, colon, rectum, duodenum, ileum, and/or jejunum). One and 2 females in the 1000 and 2000 mg/kg/day groups, respectively, were also noted with dark blue discoloration of the mesenteric lymph nodes. These findings were due to the color of the test substance and were not considered adverse.
Intrauterine growth and survival were unaffected by test substance administration (Table 4). Parameters evaluated included mean litter proportions of postimplantation loss, mean number of live fetuses, mean fetal body weights, and fetal sex ratios. Statistically significant lower mean number of live female fetuses was observed in the 500 mg/kg/day group; however, this did not present in a dose-responsive manner and did not affect the mean total number of live fetuses, and therefore, was not considered related to treatment. Mean numbers of corpora lutea and implantation sites and the mean litter proportions of pre-implantation loss were similar across all groups.
Rabbit fetal morphology.
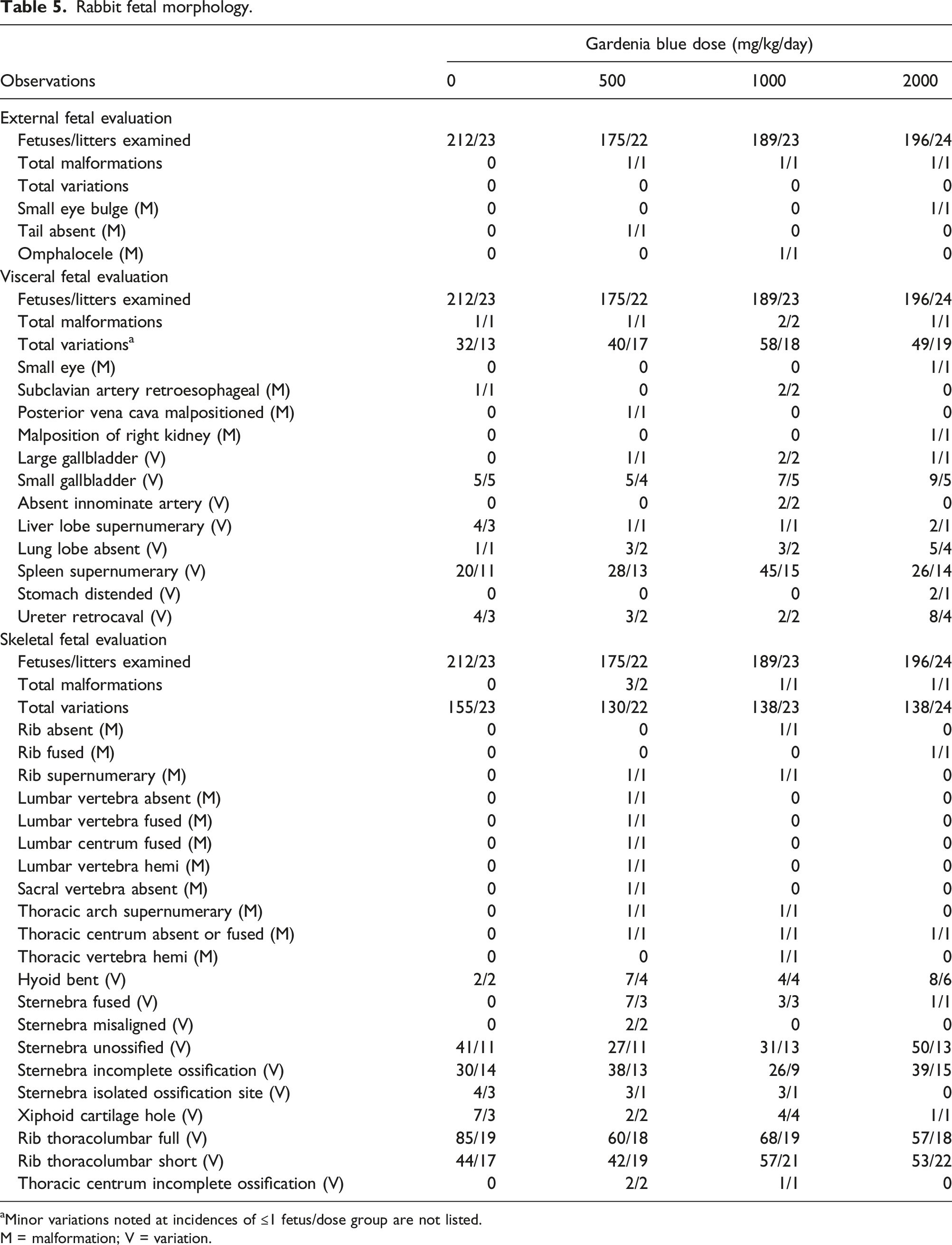
aMinor variations noted at incidences of ≤1 fetus/dose group are not listed.
M = malformation; V = variation.
No test substance-related external, visceral, or skeletal malformations or variations were noted in rabbit fetuses in this study (Table 3). External malformations were limited to single fetuses and included the following. In the 2000 mg/kg/day group, one fetus was observed with a small left eye bulge and a second fetus was observed with an omphalocele (portion of liver protruded with remnants of a membranous sac). In the 500 mg/kg/day group, one fetus had an absent tail. These external malformations presented in single fetuses and/or were not observed in a dose-related manner or did not demonstrate a common pattern. In addition, the mean litter proportions of small eye bulge and omphalocele were within the ranges of the historical control data. Therefore, these findings were not considered test substance-related. No external variations were observed for any fetuses in this study.
Visceral malformations were limited to one or two fetuses/group and included the following. In the 2000 mg/kg/day group, one fetus was observed with malformations of a small left eye (correlating externally to small eye bulge), transposition of the great vessels, and malposition of the right kidney. In the 1000 mg/kg/day group, two fetuses were observed with a retroesophageal right subclavian artery; this finding was also observed for one fetus in the control group. In the 500 mg/kg/day group, one fetus was observed with malposition of the posterior vena cava. These visceral malformations occurred in single fetuses (except retroesophageal right subclavian artery), not in a dose-dependent manner, and/or the mean litter proportions were within the range of values in the historical control data, and therefore were not considered test substance-related.
Visceral variations noted in the test substance-treated groups occurred in single fetuses, at a similar incidence in the control group, and/or did not present in a dose-related manner (Table 5). Slightly higher mean litter proportions of small gallbladder, absent lung lobe, and retrocaval ureter in the 2000 mg/kg/day group were not statistically significant from the control group and the values were within the range of values in the Charles River Ashland historical control data.
Vertebral malformations (with or without associated rib malformations) were noted for 3, 1, and 1 fetuses in the 500, 1000, and 2000 mg/kg/day groups, respectively (Table 5). One fetus in the 2000 mg/kg/day group was noted with fused ribs and an absent thoracic centra. One fetus in the 1000 mg/kg/day group was noted with absent and supernumerary ribs, fused thoracic centra, a supernumerary thoracic arch, and a thoracic hemivertebra. In the 500 mg/kg/day group, malformations included a supernumerary rib and thoracic arch and fused thoracic centra for one fetus; absent caudal and sacral vertebra (corresponding externally to an absent tail), a small lumbar arch, and absent and fused lumbar vertebra for a second fetus, and a lumbar hemivertebra for a third fetus. These skeletal malformations were not considered test substance-related because they were noted infrequently, in single fetuses, and/or were not observed in a dose-responsive manner.
Skeletal variations noted in the test substance-treated groups occurred in single fetuses, at a similar incidence in the control group, did not present in a dose-related manner, and/or the mean litter proportions were within the range of values in the Charles River Ashland historical control data.
Discussion
Gardenia blue is a dark purple to blue colorant widely used in a variety of food and beverages in several Asian countries including Japan, China, Taiwan, and Korea. In Japan, it has been used for over 30 years. The primary coloring component of gardenia blue is a high molecular weight (15,000 – 30,000 Da) polymer formed by the polymerization of the aglycone from the gardenia fruit and peptides and is produced as a purified powder combined with maltodextrin.
The genotoxicity, 13-week subchronic toxicity, 12-month chronic toxicity, extended one-generation reproductive toxicity, and rat and mouse carcinogenicity of gardenia blue have been evaluated in a series of studies. Gardenia blue was determined to be negative for mutagenic and clastogenic effects in bacterial reverse mutation, in vitro micronucleus and chromosomal aberration and in vivo micronucleus and comet assays.27,28 In a 13-week subchronic toxicity study in F344 rats, gardenia blue was administered in the diet at concentrations from 0.6% to 5%. 29 The only apparent treatment-related findings included dose-dependent decreases in platelet numbers in females only, with no histopathologic correlates. In the chronic 24-month toxicity study, no adverse effects were noted at dietary concentrations up to 5% gardenia blue. 30 Based on these results, the maximum tolerated dose was considered ≥5% in the diet. In a Fisher 344 rat carcinogenicity study, male and female rats were given diets containing 0, 2.5 or 5% gardenia blue for 104 weeks. 31 Gardenia blue was determined to be negative for carcinogenicity; there were no statistically significant increases in any type of neoplastic lesions in either sex. Subsequent Sprague Dawley rat 30 and rasH2 transgenic mice 32 were also negative for carcinogenicity after 24 or 6 months of exposure, respectively, at dietary concentration up to 5% gardenia blue. The manuscript for the extended one generation reproduction study is currently in preparation.
As part of an ongoing effort to characterize the toxicity and safety of gardenia blue using current regulatory standards, the prenatal developmental toxicity of gardenia blue was assessed in rats and rabbits under the Organisation for Economic Co-operation and Development (OECD) 414 protocol. 9
The objectives of the prenatal developmental studies were to determine the potential of the test substance to induce developmental toxicity after maternal exposure from implantation to 1 day prior to expected parturition, to characterize maternal toxicity at the exposure levels tested, and to determine a NOAEL for maternal and developmental toxicity in Crl:CD (SD) rats and New Zealand White rabbits. The dose levels of 0, 500, 1000 and 2000 mg/kg/day were based on the results of previous oral gavage dose range-findings embryo-fetal developmental toxicity studies 18 demonstrating treatment with doses as high as 2000 mg/kg gardenia blue was well tolerated. The high dose of 2000 mg/kg/day is 2-fold higher than the limit dose of 1000 mg/kg/day cited in the OECD Guideline 414 9 and was not expected to produce dose-limiting toxicity. The lower doses were selected to provide for a dose-response for any treatment-related effects and to establish a clear NOAEL.
Test substance-related clinical observations, attributed to the color of the test substance, were noted in the 500, 1000, and 2000 mg/kg/day groups at the daily examinations for both rats and rabbits. These included black feces for all females in all test substance-treated groups, as well as blue fur staining on various dorsal and/or ventral body surfaces. These clinical observations were not considered adverse.
There were no significant effects on body weight or body weight gain in either rats or rabbits. Test substance-related lower food consumption, at the beginning (Gestation Days 6–9), near the end (Gestation Days 15–21), and over the entire (Gestation Days 7–29) dosing period for rats was noted in all test substance-treated groups; differences were occasionally statistically significant. Similar reductions in food consumption at the end of dosing (Gestation Days 16–29) were noted for rabbits at 2000 mg/kg/day. Because the deficits in food consumption had no effects on mean absolute body weights and body weight gains, they were not considered adverse.
Test substance-related macroscopic findings, excluding those already described for clinical observations, included dark/blue discoloration of the kidney in rats at 2000 mg/kg/day and in rabbits at all doses, blue coloration of the gastrointestinal tract in rabbits at all doses and blue coloration of the mesenteric lymph nodes at ≥ 1000 mg/kg/day in rabbits. Discolored kidney supports the absorption of test article to the systemic circulation. However, these findings have been attributed to the color of the test material, have been demonstrated to be reversible in earlier studies following discontinuation of the test substance, and have not been correlated with any histological changes.33,34 Therefore, the tissue findings noted were not considered adverse.
In both the rat and rabbits there were no indications of developmental toxicity at any dosage level. There were no treatment related effects on maintenance of pregnancy, postimplantation loss, litter size, fetal weight, fetal anogenital distance or fetal external, visceral, or skeletal malformations and variations.
Conclusions
In conclusions, oral gavage administration of gardenia blue caused no adverse maternal or developmental effects in either rat or rabbits when administered at doses as high as 2000 mg/kg/day. Therefore, the maternal and developmental no-observed-adverse-effect level (NOAEL) for gardenia blue in rats and rabbits was ≥2000 mg/kg/day. The NAOEL for the gardenia blue colorant, corrected for purity, was 646 mg/kg/day.
Footnotes
Author’s note
These studies were conducted at Charles River Laboratories Ashland, LLC, Ashland, Ohio, US.
Acknowledgements
The authors acknowledge the contributions of staff members at Charles River Laboratories in providing technical, animal care, formulations, dosing, necropsy, analytical chemistry, report preparation and quality assurance services in support of this work.
Author contributions
William Breslin was a scientific advisor on the design, conduct, interpretation and reporting of these studies and prepared the manuscript. Jaime L Mesnard was the Study Director on these studies and wrote the final reports. Robert R Maronpot, Mihoko Koyanagi, Shuichi Chiba, Masayuki Nishino and Shim-mo Hayashi provided scientific advice and review of the manuscript.
Declaration of conflicting interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: W Breslin is a paid consultant for scientific advice on the design and conduct of these studies and for the preparation of this manuscript. J Mesnard in an employee of Charles River Laboratory. Gardenia blue is a product of San-Ei Gen F.F.I., Inc. (SEG). Mihoko Koyanagi, Shuichi Chiba, and Masayuki Nishino are employees of SEG and members of Gardenia Blue Interest Group. R Maronpot is a paid consultant of SEG. Shim-mo Hayashi was a paid consultant for scientific advice.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ministry of Agriculture, Forestry and Fisheries of Japan under the project for promotion of export environment “the project for preparation of an application of gardenia blue among those for existing additives” through the Gardenia Blue Interest Group, Osaka, Japan.
Ethical approval
Ethical approval for these studies was obtained from the Charles River Institutional Animal Care and Use Committee (protocol numbers 01339005 and 01339003).
Availability of data
Data ownership resides with the Gardenia Blue Interest Group, Osaka, Japan.
