Abstract
During the development of international standards by the Codex Alimentarius Commission, infant foods and their constituent ingredients are subject to rigorous risk analysis and are strictly regulated by many authorities. Various jurisdictions have approved only a limited number of additives specifically with regard to infant foods to fulfill specific technical requirements of quality. As part of the approval process, a rigorous safety assessment is essential to confirm that the use of additives does not pose any health risk for the consumer. An acceptable daily intake (ADI) may be derived from the toxicological databases. However, the ADI may not be applicable to infants because of the possible developmental sensitivities and potentially high exposure scenarios, leading to possible lower margins of safety than would often be determined for adult populations. There is interest in defining better food safety assessment approaches for pre-weaned infants aged less than 12–16 weeks. To confirm safe use in infants, we reviewed the suitability of the existing safety databases of six additives with historical uses in infant nutrition products. To determine further toxicity testing strategies, it is necessary to understand whether the chemical used in the additives is identical to endogenous physiological metabolites and/or whether immature organs of infants are targets of toxicity. Combined with an in-depth review of the existing relevant toxicological and nutritional studies, this integrated approach will facilitate decision-making. We propose a decision tree as a tool within this approach to help guide appropriate data requirements and identify data gaps. In cases of reasonable uncertainty, studies of targeted juvenile should be considered to investigate the safe use levels in food products.
Introduction
Additives provide solutions to a range of technical challenges in the manufacturing, processing, packaging, transport and consumer use of foods and beverages. Strict requirements on the safety data allowing the assessments of such additives for their intended uses are detailed in several guidance documents. 1 –4 Manufacturers should ensure that the use of technologically justified additives specifically in infant formula and formulas for special medical purposes intended for Infants (FSMP) is in agreement with the principle endorsed by the Codex Alimentarius Commission (CAC). The latter stipulates that wherever possible baby foods should be prepared without food additives, but where the use of additives becomes necessary, great caution should be exercised in choosing the additives and also it’s the level to be used. 5 The classical risk assessment paradigm consists of four steps: hazard identification (toxicological effect of concern), hazard characterization (analysis of dose–response relationships, mechanism of actions, derivation of guidance values), exposure assessment (actual/estimated intakes of substance being evaluated), and risk characterization (probability that adverse effect would arise from human exposure at the intended levels used). One outcome of the hazard analysis is the derivation of a health-based guidance value such as the acceptable daily intake (ADI), with which the expected exposure can be compared. 6 An ADI is defined as “the estimate of the amount of a substance in food expressed on a body weight basis that can be ingested daily over a lifetime without appreciable health risk to the consumer.” 4,6
However, the ADI applicability in the early life stage has come under much discussion. Major differences between infants and adults typically include the exposures through total food sources, maturity of the gastrointestinal tract (GIT), and susceptibility to toxicity. In many instances, the newborn infant and young child are more vulnerable to potential health effects of environmental chemicals and pharmaceuticals. 3 This is because of immature physiological functions which rapidly change during this early stage of development. 3 The toxicological database of animal feeding studies rarely includes direct exposure of juveniles from birth to weaning. Even more important however is that infants may have greater exposure than adults because of limited and less varied food sources (often a single food source for infants in early life) as well as higher relative food intake per kilogram body weight (bw). 7 –9 Taking these considerations together, an ADI derived for the general population is generally not applicable to young infants of <12 weeks of age. 10 –12
Since, generally, additives that are used in infant foods are already approved for use in foods for the adult population, with few novel additives currently being considered, this paper explores the challenges to adequately address knowledge gaps in conventional toxicological data packages to support the safety of extending the uses as an additive in infant foods. Six additives [citric acid esters of mono- and diglycerides of fatty acids (CITREM, INS 472c), starch sodium octenyl succinic (INS 1450), carrageenan (INS 407), locust bean gum (INS 410), xanthan gum (INS 415), and pectin (INS 440)] have recently been presented by the specialized nutrition industry to Joint FAO/WHO Expert Committee on Food Additives (JECFA) for evaluating their use in infant formulas, taking into account recommendations for life stage-specific animal models. We make use of the experiences gained from these cases to propose a flexible integrated approach, using a decision tree as a tool to guide relevant safety assessment data requirements for the intended uses. The principles should be applicable to new additives as well as currently authorized food additives that may undergo reevaluation.
Regulatory context
Although infant formulas are commercially available since the late nineteenth century, 13 –16 the 1970s and 1980s saw the emergence of harmonized laws to ensure their safety and nutritional adequacy. 5 The technological need for certain food additives in infant formulas was acknowledged by the CAC. 5,17 –19 In the United States, the Infant Formula Act of 1980 sets the mandate for the US Food and Drug Administration (FDA) to establish the testing and premarket notification requirements for manufacturers of infant formula. 20 As part of that Act, it was established that only ingredients, including additives, that are regarded as safe for the specific use in infant formula may be used. In Europe, although infant formulas containing food additives were already commercially available in a number of European Union (EU) member states, the first harmonized provisions on food additive use in infant formula were not established as part of the common market until 1995. 21 All of the food additives permitted for use in infant formula according to regulatory provisions in a given jurisdiction have been evaluated for safety by relevant scientific panels, that is, by the European Food Safety Authority (EFSA) for the EU (previously the Scientific Committee for Food (SCF)), by JECFA for World Health Organization/Food and Agriculture Organization of the United Nations member countries and channeled through CODEX, and by the FDA for the United States. The use of food additives in food products can be authorized only if there is a technological/functional need, if they do not mislead the consumers, and if they present no appreciable health risk to the consumers. In addition, all food additives must fulfill certain defined suitability and purity criteria to further ensure their safety. 22 –24
The Codex Infant Formula Standard (CX STAN 72-1981) 25 provides standards for infant formula (breast milk substitute to satisfy nutritional requirements of infants during the first months of life up to the introduction of appropriate complementary feeding) and for FSMPs (substitute for human milk or infant formula meeting the special nutritional requirements arising from the disorder, disease, or medical condition for whose dietary management the product has been formulated). In that standard, from 2007, a number of food additive provisions were also adopted, based on prior JECFA evaluations, technological justification, and history of safe use. Additional additives foreseen to be essential for infant formula categories were also proposed and awaited formal advice via CODEX from JECFA. The 2011 report from the 43rd Codex Committee on Food Additives (CCFA) meeting, regarding food additive provisions for use in infant formulas and FSMPs, concluded that the toxicological database should include “evidence of safety for young animals” in a comparable life stage of physiological development, including models with artificial feeding of juvenile animals, although it was acknowledged that the practical difficulties of this recommendation would be immense. 26 –28 Thus, proposals for new additives in CODEX standards for foods intended for infants below 12 weeks of age would require a separate specific evaluation by JECFA. The six food additives for use in infant formula and/or FSMPs which have been assessed by JECFA since 2011, and for which data are publicly available, are discussed in this document. These six food additives were chosen as case studies for this study, for the simple reason they are the only additives in recent years for which dossiers have been presented to JECFA. There was no other selection criteria.
General categorization of food additives
Additives used in foods including infant formulas must have a technological function according to one or more of the following general functions: (a) preserve nutritional quality; (b) provide necessary constituents for consumers with special dietary needs; (c) enhance the keeping quality, stability, or organoleptic properties; and/or (d) provide aid in the manufacture, preparation, processing, treatment, packing, transport, or storage. 24,26 Additives are further classified into functional categories or technical effects; however, many commonly serve more than one function. 29,30 The main functional classes used in formulas for infants include antioxidants, acidity regulators, emulsifiers, stabilizers, gelling agents, and thickeners. Such functions are essential to the manufacturing of modern infant formulas. FSMPs are often formulated with specialized ingredients (e.g. medium chain triglyceride fats, long chain polyunsaturated fats, synthetic amino acids, extensively hydrolyzed proteins), and methods to maintain the safety and quality of these formulas are also needed. It is essential to ensure the stability of products throughout shelf life, to deliver adequate physical and sensory characteristics for all formulas, and to enable feeding via both bottle and enteral routes when needed.
The CCFA suggested that grouping of additives in accordance with their needs for different levels of assessment could assist in evaluating substances with very different chemical profiles. 27 Such grouping may include substances which may be (i) physiological body constituents (e.g. salts of citric or phosphoric acids consisting of ions, such as sodium, calcium, and/or citrate, that may also be part of additives or minerals already permitted in infant formula), (ii) nutrients or physiological metabolites thereof derived from dietary sources (e.g. mono- and diglycerides of fatty acids [FAs]), and (iii) not naturally produced or expected to be endogenously present, such as certain emulsifiers and thickeners originating from plant material. This approach has been taken into consideration for our present discussion.
Specific toxicological considerations for infants
Many of the physiological processes that deal with the absorption, metabolism, distribution, and excretion of chemicals (toxicokinetics) are immature at birth. 12,31,32 Gastric pH is higher, gastric emptying time is slower, although motility is higher and digestive enzyme activities may be low. These factors may interfere with how chemicals become available in the gut and are absorbed. An immature mucosal barrier could raise concerns of an increased permeability. Differences in body fat and water content in adults may modify the distribution of chemicals, with altered storage of lipophilic chemicals. The binding capacity of plasma proteins may be low, so during systemic circulation, free chemicals may be available to target sites of toxicity. The phase I and phase II xenobiotic metabolizing enzymes in liver are immature; therefore, metabolic pathways may be different from adults. 33,34 Consequences may be that toxic metabolites are not produced, or if produced then not de-toxified adequately, but also that a lower detoxification capacity might be balanced by a lower formation of toxic metabolites. Clearance of chemicals may be different in infants and adults because of lower glomerular filtration rate, tubular secretion, and reabsorption in the kidney. In infants, tissue exposure to chemicals may be different compared to adults. Particularly susceptible targets include the central nervous system (CNS), the immune system, endocrine and reproductive organs, brain and neurodevelopment, and cognitive/behavior development. The net impact of all these toxicokinetic and toxicodynamic considerations is chemical specific, and no generalizations can be made. 33,34 Examples can be found in the cited literature.
Derivation of health-based guidance values and applicability to the infant
The lowest identified no observed adverse effect level (NOAEL) or benchmark dose with limits of confidence (BMDL) from appropriate toxicological studies is used as the point of departure (PoD) from which to extrapolate to the human situation. Usually the PoD is derived from in vivo studies of animals, although the preferred source would be from human data, if available the PoD used is that for the most sensitive endpoint leading to an adverse effect that is biologically relevant for humans.
4,35
An adverse effect has been defined as: Change in the morphology, physiology, growth, development, reproduction or lifespan of an organism, system or (sub) population that results in an impairment of functional capacity, an impairment of the capacity to compensate for additional stress or an increase in susceptibility to other influences.
3
The ADI is a conservative concept, which might actually be overprotective for the majority of the population including infants. For example, the real NOAEL may be higher than the recorded one due to dose selection (dose spacing, group sizes). This is one reason why the BMD modeling approach is used, with the advantage of increasing the power of risk assessment in animal studies, suggesting the use of more doses (but without increasing the number of animals used). Importantly, in many cases, an unspecified ADI may be established, reflecting the low toxicological profile of the additive under evaluation and the lack of adverse health concern under its intended uses. Good manufacturing practice should always ensure that the lowest amounts are used for the desired technological/functional effects.
The underlying basis of deriving an ADI therefore facilitates an assessment of the appropriateness of the default uncertainty (safety) factor and the case-by-case demonstration of the applicability of a specific ADI to particular circumstances, such as infants and children. If it can be demonstrated that the toxicokinetics and/or toxicodynamics are similar in the animal species considered, the humans, and also in infants, the use of an additional intra- or interspecies safety factors may not be necessary or could lead to a reduction in the standard safety factors. In addition, if the PoD can be derived from human data, and particularly in the target population of interest, then a factor for interspecies differences may not be needed. In some cases, the available data may demonstrate the applicability of the derived health-based guidance, that is, ADI, in term infants, in which case the generation of additional data would not be necessary. Consideration of the intrinsic chemical properties and the overall toxicity database may lead to adjustment of the standard safety factors applied for human health risk assessment.
Designing a safety data package for infants
Data requirements for the assessment of food additives for the general population have been detailed elsewhere 43 and as discussed, the established ADIs as a rule do not apply to infants aged <12 weeks. In their recent guidance for food additive evaluations, the EFSA provided a framework consisting of three tiers to guide requirements for toxicological studies. 43 The framework is considered suitable to address two scenarios: one for authorization of a new additive and the other for modification/extension of the existing authorizations. A minimal data set (tier 1) would be applicable to all substances and considers the identity, composition, and stability of the material and its expected fate in the body (i.e. absorption and systemic exposure). If triggered by outcomes of tier 1 testing (i.e. systemic exposure, toxicity, or genotoxicity is alerted), then tier 2 testing would generate more extensive data to better characterize potential hazards. Tier 3 is intended for case-by-case considerations, taking into account all available data and the need to further investigate findings from tier 2. So far only a few authors have specifically considered the requirements for infant formula and its ingredients. 39,44 Activities are ongoing at EFSA to develop guidance to assess the safety of substances (intentional and unintentional exposures) during the first 16 weeks of early life. Based on a general framework, an integrated testing strategy on existing guidances was developed. 39,44,45 From our own experiences, we focused on the suitability of fit-for-purpose data to assess safety in early life. Information that are to be reviewed to assess the suitability of food additives in infant formulas including FSMP are:
A detailed description and complete chemical and physical data (molecular size and forms, mixtures), nutritional compositional data, possible contaminants, impurities, allergenic residues, and so on, along with relevant specifications. Structure–activity relation or quantitative structure–activity relation databases could be of assistance in identifying alerts for potential safety concern. The threshold of toxicological concern concept could be applied to assess impurities or process-related contaminants.
Intended use levels and target population (including if for general infant population, or special cases under medical supervision), and resulting exposure assessments.
Previous/existing uses, previous authorizations and assessments, and all available post-marketing safety data.
All available biological and toxicological information (bioavailability, metabolism, biological distribution, biological effects, interactions, and toxic effects, relevant mechanisms of action).
Clinical trial data (pediatric and adult populations).
Information on in vitro and in vivo studies in animal models if they add information on nutritional adequacy, potential benefits, and safety.
When evaluating the suitability of an existing data package to assess the safety of an application in early life, it is necessary to structure this information in such a manner that decisions can be made on relevant data gaps and to provide options on the best way forward. To this end, we have developed a decision tree specifically for early life phases to complement the existing guidance. The proposed decision tree is shown in Figure 1. The tree focuses on potential differences in toxicokinetics and toxicodynamics of neonates compared to adults. Important prerequisites are to (i) fully identify the material, including physical and chemical properties, and food additive ingredient specifications and (ii) understand the intended use levels and resulting exposures and collect all toxicological data on the material in question, and available data on safe use. A first step in decision-making is to understand the fate of the material in the body and to identify similarity or differences in early life and adults with regard to gastrointestinal (GI) digestion and ADME. Several approaches exist to study digestibility and ADME and can include in vitro metabolism assays using human enzymatic systems and appropriate life stage conditions (such as specific pH values) and physical systems such as the TIM (TNO gastrointestinal model) 46 as well as standard in vivo approaches. If adequate data are available, physiologically based pharmacokinetic modeling can be done, mimicking infant conditions. If a similar metabolism is demonstrated between species and within species (adult vs. infants), and systemic exposure is negligible, or if there is systemic exposure but immature target organs in early life are not identified in the standard tox package, then there is no requirement for additional juvenile-specific toxicological data. Demonstration of nutritional adequacy in the additive-containing formula matrix can then be assessed by tolerance and nutritional adequacy in clinical studies that are appropriately designed and controlled. 24,44 If a substance is not digested or metabolized in the gut, then it is reasonable that this substance cannot be expected to be systemically available. Since the human GIT develops rapidly, and gut closure is already quite effective just before birth, there is no indication that systemic exposure would occur in the healthy term-born human infant if it will not occur in the adult. 47 In these cases, the single site of contact would be the GIT. Within the context of the actual intended uses, it is necessary to determine whether a material will have direct contact with the intestinal mucosa and what the potential effects might be. Emulsifiers for example bind to the components of infant formula and have little or no direct contact with the GIT, in comparison to a substance delivered in water (see example of carrageenan). Events at the border of nutrition and toxicology should be emphasized, such as tolerance and nutritional effects, which are often best studied in the actual target infant population 48 –50 (once the toxicological safety has been established). This can be demonstrated by historical data or with targeted clinical trials at the intended use levels (usually existing use levels). Second step in decision-making focuses on the toxicodynamics, where the overall toxicological profile of the substance should be studied and attention focused on potential target organs that are immature in early life (GIT, CNS, immune system, endocrine system). If these are identified as target organs, a study of juvenile might be considered as an appropriate solution to further characterize any potential hazard. Species of choice, target organ of concern, and the adequacy of the dose that can be achieved within nutrition/technical restrictions must be given careful consideration prior to initiating a juvenile testing program. While the conduct of juvenile animal studies may have an important role in some cases, such studies are not without practical limitations, and careful interpretation of the results is imperative. When finally concluding on the risk assessment, there must be consideration if a numerical ADI can be determined which is adequate for the infant population; if low margins of exposure can be accepted between animal and infant studies exposures; and if integrating the totality of the collected data allows one to conclude that the intended, or current, usual and maximum use levels are not of concern for the consuming population.

Decision tree to guide data requirements.
Juvenile animal models
The pharmaceutical industry has accumulated extensive experience in the use of preclinical toxicological juvenile models for pediatric drug applications, as a consequence of mandatory regulatory requirements. This experience has provided the insight that having such a mandatory requirement for toxicological studies of juvenile animals does not always provide additional knowledge and that a careful consideration of all available data is needed to decide on the value of conducting a juvenile study. 50 –53 Recent reviews in the pharmaceutical area have indicated that from a total of 241 juvenile toxicity studies, 84% were conducted in rats, 14% in dogs, and the remaining 2% in other species. 51,53 In 15% of programs reviewed, existing adult preclinical and clinical data had already been considered sufficient to support pediatric drug trials. The majority of the juvenile toxicity studies showed findings comparable to that in adults. Quantitatively, a general trend for increased sensitivity in terms of general toxicity was observed in rats but not dogs (raising the question on the relevance of any one particular animal species compared to infants). Novel toxicity (finding in an organ system not previously seen in adult animals) was seen in 14 (out of 202) rat studies and in only 2 (out of 33) dog studies. 51,53 In most cases (although not all), these could have been predicted from either known pharmacology or adult data.
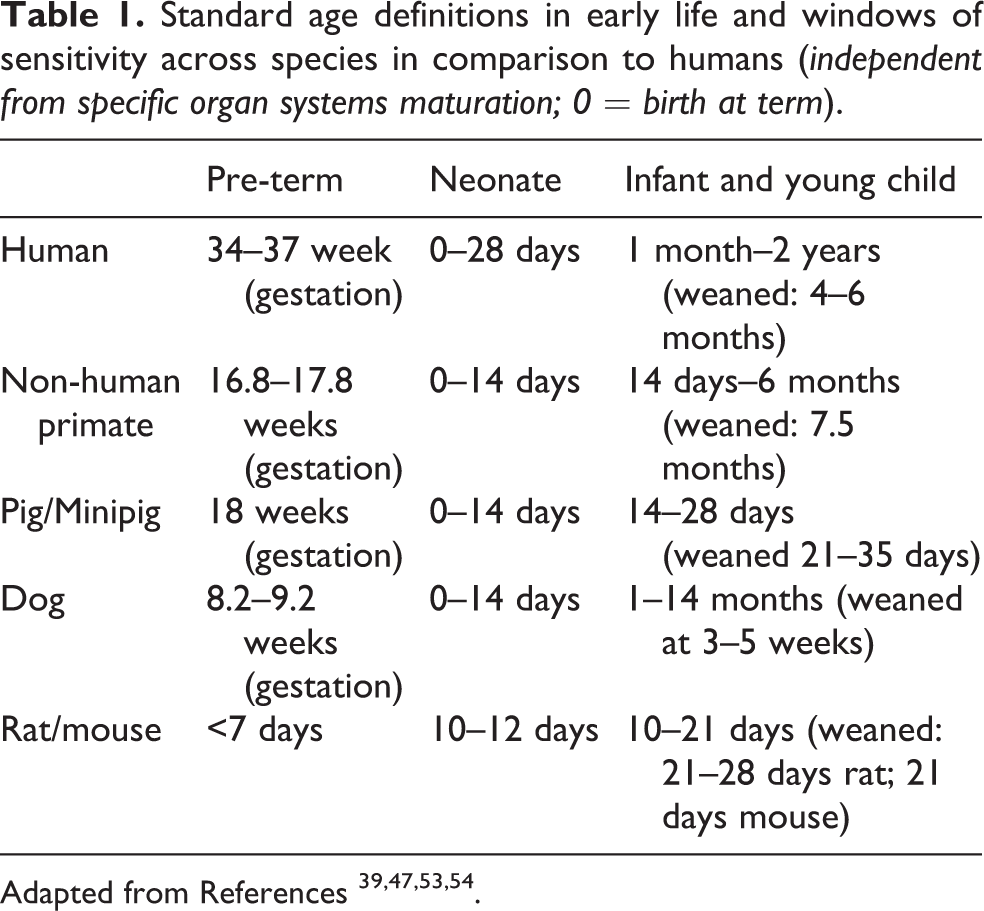
Currently there is no scientific consensus, nor guidance from regulatory bodies on the most suitable animal species to address toxicological data gaps in the early life period. A human infant develops quite differently from most animal models. The animal species to be selected depends on the time of exposure (Table 1) and expected target organ (Table 2) and requires a thorough knowledge of the difference in physiological development amongst species and how they relate to the human situation. 33,34 The time of early stage should be relative to life span. In that case, the early life stage of pigs and nonhuman primates are the closest to the human early life stage.
Standard age definitions in early life and windows of sensitivity across species in comparison to humans (independent from specific organ systems maturation; 0 = birth at term).
Comparison of critical organ systems for different juvenile animal species and human neonates [see associated references].
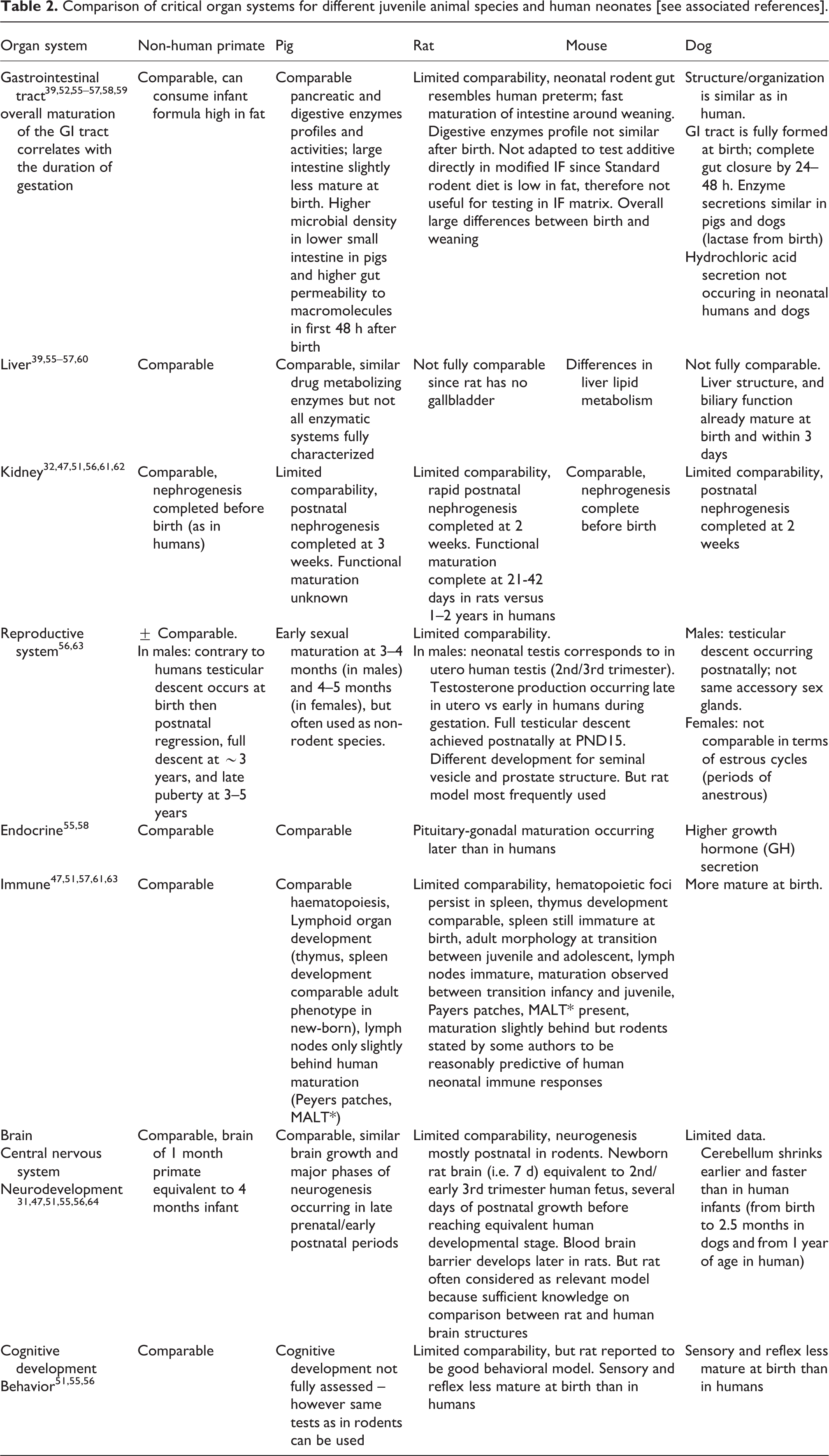
The use of juvenile studies in the food area is relatively new, compared to the use in pediatric medical applications. Nevertheless, for useful additions to the toxicological database, studies in rat pup and neonatal piglet models as well as non-human primates have been performed for new food additives or ingredients intended for infants. 65 –69
The physiological development of the piglet GIT is considered to be more similar than the developing rodent GIT to the human infant 39,55 so it could be the most relevant animal model for infant food applications. Guilloteau et al. also put forward that the early stage and exposure time relative to life span in the pig is the most comparable with early life in humans. 47 However, it is also thought that enterocyte morphology, macromolecule absorption as well as other intestinal enzyme activities are more developed in human infants compared to piglets. 47 It is also reported that the porcine lower small intestine has a much higher microbial density, which could lead to a higher fermentation rate of indigestible carbohydrates. 47 This could exacerbate some of the known physiological effects of fibers, perhaps leading to increased sensitivity and tolerance issues at lower doses than would occur in human infants. Another interesting consideration is the superior hygienic rearing conditions required for Good Laboratory Practice (GLP) studies, and one study 70 resulting in higher rate of diarrhea, lower feed efficiency and lower fiber digestibility than when the animals were reared in less hygienic conditions. All these factors suggest a certain impact of the microbial ecology (influenced by species and environment) on digestion processes and physiological outcomes.
A current weakness to using the piglet model is the lack of historical data in the pig species to aid interpretation of the results. For pigs in general, certain husbandry considerations are needed, for example, they are sensitive to stress particularly in high room temperature, as they have no sweat glands. Weaning needs to be achieved carefully to observe test-related effects in neonatal pigs as their immune system is just developing and may require the coadministration of antibiotics. A great advantage however is that they can be easily fed with (species adapted) formula in feeding bottles, although rather large amounts of test substances would be needed compared to rodent studies.
A huge database and experience have been generated for rodents over the years, although development in the use of pups is fairly recent; and experience is accumulating, but the historical database is still limited. The practicalities of exposing very small young rodent pups can be difficult. Conducting studies in neonates requires good practical skills, since handling these very young and fragile animals, especially when dosing by gavage, may result in treatment (rather than test material)-related animal losses.
Importantly, the need for any (additional) animal toxicological study, including in juveniles, must balance data requirements and ease of interpretation of the results against potential risk. Regulatory agencies and scientific expert bodies, especially in Europe, are mindful of the need for ethical considerations and animal welfare when considering animal testing strategies. 71,72 For this, the opportunities for replacement, refinement, and reduction (the three Rs) must be kept in mind when considering whether animal studies are necessary. The appropriateness of any testing strategy depends on the problem formulation and the real necessity of filling any gaps in the database. The absence of a particular study does not automatically imply that there is an absence of knowledge on which to make a scientific judgment. Modern-day toxicology is developing with more focus where possible on in silico and in vitro techniques, and integrated testing strategies which may include these alternative approaches are needed to inform on risk assessment.
Nutritional considerations
In addition to a toxicological assessment, an understanding of the potential nutritional impacts of a food additive should also be considered. The use of fibers or other bulking agents, for example, can provide technological benefits such as thickening at intended use levels, but at higher levels can increase viscosity and thus negatively impact nutrient digestibility, gut transit times, and feed intake, resulting in negative effects on growth. Thus, it is important to recognize that the use of such additives at high levels (e.g. in range-finding studies) in animal studies can induce nutritional imbalances that may ultimately impact growth unrelated to any toxicological effects. Official guidelines for testing from Organization Economic and Cultural Development (OECD) or the US FDA Redbook state that animal feeding studies should not use dosing regimens exceeding 5% in the diet to avoid unspecific nutritional imbalance effects unrelated to a compound-related toxic effect. 1,73,74 It is important however to ensure that any food substance, especially intended for infants whose nutrition is limited to breast milk or formula in the first months of life, is nutritionally adequate and will support normal growth and development and will not result in unacceptable intolerances. This can be investigated in appropriate juvenile studies, acknowledging technical limitations or even more informatively, in human infants at the actual use levels, under real in-use conditions. Such confirmatory trials in human infants should however only be performed if the preclinical assessment and total weight of evidence indicates the absence of any expected toxicity under the clinical conditions. 39,75
Case studies
We summarized the safety data on six additives that have recently been evaluated by JECFA for use in infant formula and FSMPs. 65,66,68,69,76,77 It is not the intention of this paper to perform a detailed reevaluation of the safety data for each food additive. Each additive has a history of previous use in the general population and also in infants 0–12 months old as permitted by regulations in certain jurisdictions. We summarized the details on each of the toxicological databases to run through our proposed decision tree. Emphasis is on understanding the added contribution of juvenile animal toxicity studies in the overall weight of evidence to conclude the safety of each additive in early life exposure.
A crucial aspect of a risk assessment is the exposure assessment. In this report, all examples of exposure to infant formula and FSMPs were taken from JECFA estimations. 65,66,68,69,76,77 JECFA takes the median and 95th percentile energy requirements for fully formula–fed infants (i.e. nonbreast-fed infants) between 0 and 6 months of age and age-specific body weights (highest intakes considered at 2–3 weeks of age). This approach is considered by JECFA more conservative than using mean and 95th percentile intakes of actual consumption volumes available in the literature.
In the particular case of formula prescribed under medical supervision where the volume of feeding and number of feeding are more tightly controlled by the health-care professional, one may acknowledge that there is a lower chance of infants being 95th percentile consumers, indicating that such exposure simulation leads to an overestimation of the actual exposure.
Citric and FA esters of mono- and diglycerides of FAs
CITREM (INS 472c) consists of mixed esters of citric acid and edible FAs with glycerol and may contain minor amounts of free FAs, free glycerol, free citric acid, and mono- and diglycerides. 77 CITREM is used as an antioxidant, flour treatment agent, sequestrant, and stabilizer in several food categories. An ADI of “not specified” was allocated to evaluated CITREM (and its related organic acid esters of mono- and diglycerides) by JECFA in 1973 and confirmed in later evaluations, based on studies demonstrating that it is completely hydrolyzed in the GI tract into components that are both endogenous constituents of the body and nutrients normally present in the diet, and the lack of toxicity of those components. 78 –82 CITREM of up to 5 g/kg in processed cereal-based foods for infants and young children is provided for in the relevant CODEX Standard. 83 CITREM is also authorized for use in infant formula, follow-on formula, and FSMPs in Europe, the United States, Canada, Switzerland, Turkey, Mexico, Russia, Brazil, and China. Its use up to 7.5 g/L in powdered infant formula and up to 9.0 g/L in liquid ready to feed was positively evaluated by the EU SCF in 2002. 84 Typical CITREM use levels requested for the Codex Infant Formula Standard (CODEX STAN 72-1981) were 2.7 g/L (as consumed) in reconstituted powdered infant formula, with high-end use levels for maximum technological efficacy at 7.5 g/L powdered infant formula and 9.0 g/L in ready-to-use liquid infant formula. Because the components and resulting metabolites of CITREM (glycerol, citric acid, mainly palmitic and stearic acid, other minor free FAs) closely resemble naturally occurring triglycerides, CITREM is classified into the subgroup of additives that are physiological substances or their metabolites are physiological substances. CITREM, and its subcomponents and metabolites, also have similarities to triglycerides physiologically present in human milk and in other infant formulas. 77 Exposure to CITREM was calculated using the corresponding exposure to citric acid, that is, 440 mg/kg bw/d (2.7 g/L) and 1140 mg/kg bw/d (9 g/L) at 95th percentile energy intake.
Information on the biotransformation and ADME properties of CITREM comes from in vitro and in vivo data and from data generated for other similar organic esters of mono- and diglycerides of FAs (lactic and acetic acid esters). 77 Newborn infants exhibit lingual, gastric and pancreatic (bile salt–dependent lipase) and pancreatic lipase–related protein, and lipase enzymatic activity, and they also have the endogenous capacity to absorb and metabolize FAs, glycerol, and citric acid. 77 Free glycerides have been demonstrated in vitro to have negative effects on the rate of CITREM hydrolysis, and results from an in vitro two-stage digestion model indicated that FAs are released from CITREM under infant-specific pH conditions, although the ester bond between citric acid and glycerol may not be fully hydrolyzed. 85 However, this model does not capture all enzymatic hydrolysis steps (i.e. lingual lipase), nor did it capture physiological dynamics of digestion that contributes to create micelles for fat digestion. Absorption of the contents of the micelles occurs mainly in the proximal jejunum and partly in more distal segments of the small intestine, which are also not modeled in the two-stage in vitro model. 77 The weight of evidence from in vitro and in vivo experiments thus indicates that CITREM will not be absorbed as such but is hydrolyzed into its free subcomponents (glycerol, citric acid, mainly palmitic and stearic acid, and other minor free FAs) and progressively absorbed within the intestine. 77 The safety of any remaining citric acid–glycerol esters can be supported by comparing with other organic acid esters.
Several (at least 11) pediatric clinical trials in infants have been performed from birth onward, studying the adequacy of amino acid–based infant formula (FSMP for infants suffering from cow milk allergy) containing CITREM at 0.95–1.62 g/L (142.5–324 mg/kg bw/d), with no adverse effects on GI tolerance or growth. Although these trials were performed with use levels lower than the maximum levels petitioned, further pediatric clinical data are available for high doses of the individual hydrolysis product. 61,80,81 For example, FA supplementation of 570–2340 mg/kg bw/d palmitic acid and 70–390 mg/kg bw/d stearic acid have not been associated with growth effects or other adverse effects in infants. Glycerol was shown not to be associated with tolerance and adverse effects at levels up to 1500 mg/kg bw/d. While three studies indicated that citrate at 98–500 mg/kg bw/d for 24 h to 3 weeks added to formula was not associated with safety concerns, one smaller scale study utilizing gavage (not along with food) in infants suggested that a total citric acid exposure of 400–700 mg/kg bw divided over 24 h was associated with diarrhea in 4 out of the 8 infants. 77,86 –88 Various citrate salts are routinely used per os to actually treat infantile diarrhea, urinary tract infections, and kidney stones at comparable dosages (108–430 mg/kg bw/d), with diarrhea listed as a seldom side effect in the latter two cases. 77 Citric acid was nevertheless considered to be the main limiting metabolite in driving the tolerance and safety of CITREM exposure in infants. At typical CITREM use levels (2.7 g/L), exposure to citrate in 95th percentile consumers was estimated to be at 440 mg/kg bw/d. At high end use-level (9 g/L) exposure in 95th percentile consumers was estimated to be 1140 mg/kg bw/d. It was commented by the JECFA that the gavage administration method and the osmolarity of the solution in the later infant study could largely explain the observed diarrhea, which was not retrieved in the other studies. In addition, it was largely acknowledged that in the context of ingested CITREM, the actual exposure to citric acid would be gradual as enzymatic digestion of CITREM would progressively occur. Overall, the risk of diarrhea was estimated to be low when put into perspective of the highly maximized exposure scenarios considered that would logically not be realistically applicable for such infants given formula under medical supervision. Although safety is supported by knowledge of the metabolism of CITREM and by adequate clinical studies, a toxicological package (ADME data, genotoxicity studies and subchronic feeding data) for CITREM is also available. In the subchronic feeding studies for CITREM, no compound-related adverse effects were noted with levels in the range of at least 10,500–18,750 mg/kg bw/d (highest dose tested). This information is useful for the hazard characterization and for comparison to data on related organic acid esters. 78 –82,88,89 Overall the studies indicate that CITREM has low intrinsic toxicity. It is not genotoxic and is not a reproductive or developmental toxicant, which is consistent with its chemical structure. The products of CITREM hydrolysis are not new to an infant diet since they occur physiologically in human breast milk and are normal endogenous metabolites participating in endogenous biochemical reactions.
In summary, to demonstrate the safety of the intended CITREM application in infant formula, the scientific rationale focused on the low intrinsic toxicity of the substance, the digestion and metabolism capacities of human neonate, the physiological nature of CITREM metabolites in the infant diet, and the total clinical evidence gathered for both CITREM and for its individual metabolites. As clinical evidence of safety in the target population at the proposed use levels is available, there is no need to focus on margin of exposure (MoE) calculations from animal studies, and no juvenile toxicity study is necessary. When all the evidence is taken together, it can be concluded that the intended application levels (up to 7.5 and 9 g/L) for CITREM are of no toxicological concern. Diarrhea occurring at high usage levels with high intakes (due to free citric acid) is a possibility but given that the exposure assumptions for citric acid were maximized, the probability of occurrence is considered to be low. 77 CITREM would move rapidly down the left-hand side of the decision tree.
Octenyl succinic anhydride (OSA)-modified starch
OSA-modified starch (INS 1450, CAS 66829-29-6) is produced by esterification of food starch with food grade OSA. 76 Lipophilic octenyl succinic groups are attached usually at the hydroxyl group on the sixth carbon atom of an anhydroglucose unit of the starch molecule but also either to the second or third carbon atoms of some glucose units. The final starch sodium octenyl succinate product contains no more than 3% octenyl succinyl groups, with <0.3% residual-free OSA. The OSA-modified starch is used as a stabilizer, emulsifier, and thickener in several food categories. The safety of OSA-modified starch was first considered by JECFA in 1969, where an ADI was not specified. 90 OSA-modified starch is used as an emulsifier in infant formula, follow-on formula, and FSMPs, at use levels of 9 g/100 g powdered formula (or 12 g/L as consumed) and 2 g/100 mL (20 g/L) in liquid formula. Postmarketing surveillance information on an infant formula containing 2% OSA-modified starch, which has been globally available since 2012, indicated no safety issues when used as recommended. The 95th percentile estimated exposures is 4.4 g/kg bw/d, with esterified octenyl succinate (<3%) at 130 mg/kg bw/d and free octenyl succinic acid (0.3%) at 14 mg/kg bw/d.
The metabolic fate of OSA-modified starch and its hydrolysis product OSA has been examined in several in vitro and in vivo studies. The in vitro enzyme digestibility of OSA-modified starch was comparable to other modified food starches. 91 It is partially hydrolyzed in the GIT by digestive enzymes to form OSA and native starch. The native starch undergoes typical carbohydrate digestion and absorption.
Excretion of OSA and its related metabolites was analyzed in 17 hospitalized infants and children (aged from 2 months to 6 years) fed OSA-modified starch-containing hydrolyzed protein formulas. The metabolism of OSA and excretion of these metabolites were not associated with adverse health effects. Based on the molecular weight (MW) and mass fragmentation of the nine metabolites associated with the excretion of OSA, it was proposed that OSA is metabolized by infants by a combination of ω-, ω-1, and β-oxidation steps, similar to the metabolism of another branched chain FA, valproic acid. 92
In adult rats and dogs, tricarbylate levels excreted following oral administration account for approximately 30% and 4% of the administered dose, respectively. The level of unmodified OSA eliminated in the urine was approximately 10% in the rat, 55–60% in the dog, and 10–25% in human infants. OSA-modified starch and the hydrolyzed product OSA are handled similarly in rats, dogs, and human infants with respect to enzyme hydrolysis, absorption, metabolism, and elimination with the only difference being the amount of unmodified OSA excreted in the urine. 93
The safety of OSA-modified starch is supported by clinical studies conducted in healthy adults and term infants. Five studies were performed with different infant formulas containing OSA-modified starch at levels similar to the proposed use levels (up to 20 g/L). 94 –98 The formulas were the sole source of nutrition for infants starting to feed from birth to 16 days of age and were administered for periods of 28–120 days. All studies demonstrated that formulas containing OSA-modified starch were well tolerated and supported normal healthy growth, with no health concerns reported.
The toxicological database contains two rat subchronic and a chronic rodent feeding study with up to 30% OSA starch in the diet, where an adverse effect was reported in just one of the subchronic studies. 98 –101 This effect was related to nutritional imbalance from an extremely high starch diet (30%) which is well above the recommended upper limit of 5%. 102 No genotoxic potential was evident from in vitro studies, and no carcinogenicity was observed in the chronic rat dietary study.
Studies in neonatal beagle pups (via gavage, 5 beagle dams, 4 pups of 5–9 days of age of each sex per litter) and 3-month-old pups (via the diet, n = 5 in each group of each sex) over 6–7 weeks at daily doses up to 10,000–12,000 mg/kg bw resulted in decreased body weight at the top doses, which was reported to be due to nutritional causes such as reduced calorific intakes resulting from incomplete starch digestion. No toxicologically relevant effects were noted. 103 In a recent toxicity study conducted in neonatal piglets (n = 6 in each group of each sex) to investigate the effect of OSA on GI sensitivity, dietary administration of OSA-modified starch leading to intake of up to 10,000 mg/kg bw/d for 3 weeks after birth was well tolerated, did not significantly affect growth and no toxic effects were reported. 93
Collectively, these studies demonstrated that OSA-modified starch is safe and well-tolerated in term infants at levels up to 20 g/L in infant formula and is not associated with adverse effects on growth or development. The metabolism/digestion data in rats and human infants indicate that OSA-modified starch is enzymatically hydrolyzed in the GI tract to produce OSA and starch. The starch component undergoes normal carbohydrate digestion and absorption. The hydrolyzed product OSA is handled similarly in rats, dogs, and human infants with respect to enzyme hydrolysis, absorption, metabolism, and elimination with the only difference being the amount of unmodified OSA excreted in the urine. This provides sufficient ADME data for the first step in the decision tree. This information along with the standard preclinical studies in rats and dogs allows movement along the decision tree to the next step with regard to target organ sensitivity/development in early life. Although the weight of evidence was robust regarding the safety of the additive in juvenile rat and dog studies previously performed, an additional neonatal piglet study was conducted, 93 confirming the safety of OSA-modified starch. In retrospect, with the postmarket surveillance data and the clinical studies conducted with OSA, sufficient information with early infant exposure was available to progress along the decision tree and to arrive at a safe level for infants under 12 weeks of age, without the additional neonatal piglet study. Taking into account the overall low toxicity of OSA-modified starch, the conservatism in the NOAEL (10,000 mg/kg bw/d, the highest dose tested in neonatal animals) and in the infant exposure assessments (up to 4.4 g/kg bw/d) as well as the supporting evidence from clinical trials and postmarketing surveillance, the consumption of OSA-modified starch in infant formula or formula for special medical purposes intended for infants is not of concern at use levels up to 20 g/L. 76 The neonatal piglet study helped confirm the safety and contributed to the acceptance of a low MoE (2.3) but was not an essential addition to the database.
Carrageenan
Carrageenan (INS 407, CGN) is a hydrocolloid obtained from members of the class Rhodophyceae (red seaweeds). Carrageenan consists mainly of the ammonium, calcium, magnesium, potassium, and sodium sulfate esters of galactose and 3,6-anhydrogalactose polysaccharides. These hexoses are alternately linked α-1,3 and β-1,4 in the copolymer. The prevalent polysaccharides in carrageenan are designated as kappa-, iota-, and lambda-carrageenan, each characterized by the degree and position of sulfation. Lambda-carrageenan is also characterized by the absence of anhydrogalactose. Food grade CGN has an average MW of 200–800 kDa. In the JECFA specifications, a viscosity of ≥5 cp at 75°C (1.5% solution) is required to minimize the presence of low-molecular-weight CGN. 65,104 The SCF proposed a specification of <5% for the <50 kDa fraction. 105 Its physical chemical properties make it a useful thickener, gelling agent, stabilizer, and glazing agent and CGN is approved for use in a variety of general foodstuffs as well as for foods and formula for infants of 6 months and older. There is an extensive toxicological database on CGN, indicating very low toxicity.
JECFA maintained a group of ADI not specified for the sum of CGN and processed Eucheuma seaweed in 2007. 104 The EC Scientific Committee on Food (SCF) allocated an ADI of 75 mg/kg bw/d to CGN, 105 based on an NOAEL of 750 mg/kg bw/d for increased thymidine kinase activity used as a biomarker for increased cell proliferation in the rat colon. Neither JECFA nor SCF considered that the available data were adequate to conclude on the safety of CGN for infants of <12 weeks old. 104,105 Concerns were raised over local effects on the gut and on potential consequences of systematic uptake. However, a fundamental and important issue with CGN is that much of the experimental data wrongly associates the biological effects of a low-MW material to food grade CGN. It has been demonstrated in in vitro and in vivo studies that a similar substance, poligeenan (also known as degraded CGN) but having an average MW of 20–30 kD has inflammatory properties and can cause severe GI problems and even preneoplastic colon lesions at high doses, when given orally to rodents. 106,107 Poligeenan is used in experimental research for investigation of immune processes, and for medical imaging, but has no functional use as a food additive. In fact, a food grade preparation of CGN should have as little low-MW material as possible. Poligeenan is produced by subjecting CGN to acid hydrolysis at low pH (0.9–1.3) and at high temperatures (>80°C) for several hours.
The α-1,3 and β-1,4 glycosidic bonds of CGN can be broken by galactosidases; however, these enzymes are not present in the human gut. Amylase present in the gut breaks down starch at α-1,4 bonds, and so CGN is not digested in humans. Experimental studies in animals demonstrate that ingested CGN is excreted quantitatively in the feces and is not significantly degraded by low gastric pH, or the microflora in the GI tract, and is not absorbed. 107 In addition, the conditions in the human GI tract are not severe enough to degrade CGN to poligeenan.
In vitro studies in human intestinal epithelial cell lines and in human colorectal adenocarcinoma cell lines have suggested that CGN can activate inflammatory signaling pathways resulting in an induction of proinflammatory cytokines. 106 Recently, such effects were not reproduced using commercially available food grade CGN, and in addition, it was demonstrated in a CaCO-2 permeability model that CGN is not cytotoxic and does not cross the cell barrier. 108 Feeding rodents with CGN in the drinking water did result in inflammatory effects, with an LOAEL of 1100–1300 mg/kg bw/d (1% in the diet). However, when not bound to protein in food, CGN is in a random, open-structured molecular conformation, allowing increased exposure to the intestinal mucosa, 107,109 and is not representative of the conditions of use of the material. This also helps to explain the observations when direct exposure of various forms of CGN in the in vitro cell culture systems suggested the potential for inflammation. Oral administration of CGN of up 5% in the diet, representative of its food uses, has not resulted in toxicological effects in several species (guinea pigs, rats – equivalent to 3394 mg/kg/d in males and 3867 mg/kg/d in females – mice and hamsters). 107,110,111 Some evidence of soft stools was noted at that top dose in rats, not unexpected from high doses of nondigestible dietary fiber. Only high doses (>15%) of dietary CGN produced some ulcerative effects in rats. 112 Insoluble, indigestible materials have been shown to induce proliferation of cells at high doses in the diets of rats (>5%) in the GI tract, but this is an adaptive response to high levels of bulking agents, due to reduced absorption of food and reduced digestive capacity, causing hyperplasia and inflammation in the gut. 113 As described elsewhere in this document, doses above 5%, which is the regulatory top level unless appropriate controls are included, can result in confounding results due to nutritional deficiencies. 109 Carcinogenicity studies in rats and hamsters of food grade CGN at 5% in the diet did not show evidence of carcinogenicity, tumor promotion, or ulceration. 111,112,114 Teratogenicity was not observed in mice, rats, rabbits, or hamsters, no developmental or reproductive toxicity was observed in a three generational study in rats, and no genotoxicity was identified. 115
Data exist on feeding CGN to nonhuman primates (baboons). 116,117 Newborn baboons were fed infant formula containing food grade CGN (MW 197.1–394 kDa) to stabilize the protein, at doses of 0, 255, and 1220 mg/L (equivalent to 86 and 432 mg/kg bw/d), as the sole source of food from the age of 1-day-old until 112 days of age. 116 Primates are similar to humans with regard to gut closure and maturation and so is the most appropriate animal model for human infants. No adverse effects on body weight, organ weights, urine and fecal characteristics, or hematological and blood chemical parameters resulted from CGN-containing formula. There were no abnormal findings on physical examination. Particular attention was given to the GIT, which was examined macro- and microscopically. Histopathology of the gut using standard techniques and stains (hematoxylin and eosin, periodic acid Schiff, and Prussian blue) did not indicate any lesions due to CGN exposure. The NOAEL was established at 432 mg/kg bw/d. In addition, when adult baboons having been fed with 500 mg/kg bw/d CGN in the diet over a period of 7.5 years were examined thoroughly, including in particular histopathology for inflammatory effects in the intestine, no significant findings were noted.
A retrospective study of 1418 infants from the 1988 National Maternal and Health Survey of the United States examined immunosuppressive effects of CGN by looking at the frequency of symptomatic upper respiratory tract infection during the first 6 months of life. 118 One group of infants (n = 1269) was fed exclusively with formula containing 0.03% CGN and compared with a group (n = 149) fed with formula containing no CGN. Statistical analysis showed no differences between frequencies of upper respiratory tract infection in the two groups. In a masked, randomized parallel study, groups of healthy newborns (0–9 days of age) were fed either liquid formula containing 0.1 g CGN/100 mL (n = 95) or a powdered base formula with no CGN (n = 100) until 112 days of age. 119 Tolerance to the formula, stool pattern, and anthropometric measurements were similar between the two groups.
In summary, knowledge of the chemical and physical properties of the material gives indications that it binds tightly to food proteins will not be digested or broken down in the digestive tract, even in young infants. Due to its large MW, structure, and stability when bound to protein in food, CGN is not significantly absorbed or metabolized. Conditions in the infant gut (a pH of 5) would not be sufficient to degrade CGN into smaller fragments such as poligeenan. Lack of systemic exposure indicates that we would follow the right-hand side of the proposed decision tree. Long-term studies at levels considered by OECD guidance to be the maximum amount of a substance to be tested in animal studies (5%, approx. 1000 mg/kg bw/d) demonstrated low toxicity; some evidence of soft stools was noted at that top dose, not unexpected from high doses of nondigestible dietary fiber. Only studies that did not mimic the intended uses (i.e. administered through drinking water without food, administration via routes other than oral, and nonfood grade preparations) resulted in inflammation. The infant baboon study, performed according to standard protocols, with extensive histopathology and biochemical analyses, is a suitable model for human infants, covered the actual commercial use levels and no clinical or toxicological adverse events were reported. Nevertheless, it was considered to be inadequate for concluding in the absence of local inflammation because it did not include specific staining such as toluidine blue for the presence of mast cells, which would be present if an inflammatory process had been initiated. Such a staining had actually been included in the previous long-term studies in baboons (feeding at 500 mg/kg bw/d over 7.5 years), with negative results. 109 No adverse events had been identified from the commercial use of CGN-containing infant formulas; however, it was considered that the retrospective observations on associations of CGN-containing formula with respiratory tract infections did not address possible effects on the GI tract and that the statistical power was not sufficient to have identified any immune-suppressive effect. In addition, when considering LOAELs based on inflammatory effects from rodent studies fed with CGN in the drinking water (1100–1300 mg/kg bw/d) compared to the highest expected exposure to infants from the use of 0.1% CGN in formula (160 mg/kg bw/d), the margin of exposure of about 7 was not considered sufficient to protect the health of young infants, 104 even though this study is not representative of the intended food uses.
In response to these concerns from JECFA, feeding studies using pre-weaning piglets were performed. 119 A toxicity/toxicokinetic swine-adapted infant formula feeding study was conducted in Domestic Yorkshire Crossbred Swine from lactation day 3 for 28 consecutive days at concentrations of 0, 300, 1000, and 2250 ppm CGN (leading to exposures of approx. 51, 192, and 430 mg/kg bw/d), under GLP guidelines. The top dose levels were chosen based on prior 2- and 10-day feeding studies in 2-day-old minipigs, where a top dose of 3000 ppm resulted in high-viscosity formula leading to reduced feed intake. Organ weights, clinical chemistry, special GIT stains (toluidine blue, Periodic Acid Schiff), plasma levels of CGN, and evaluation of potential immune system effects were performed. Immunophenotyping of blood cell types (lymphocytes, monocytes, B cells, helper T cells, cytotoxic T cells, mature T cells), immunoassays for blood cytokines (interleukin 6 [IL-6], IL-8, IL-1β, tumor necrosis factor α [TNF-α]), and immunohistochemical staining of the gut for IL-8 and TNF-α were conducted. No treatment-related adverse effects at any CGN concentration tested were found on any parameter. Glucosuria in a few animals was not considered treatment related. The highest dose tested, 2250 mg/L, equivalent to ∼430 mg/kg/d was considered the NOAEL. 119
In the 79th JECFA meeting, the use of carrageenan for young infants was reassessed. The committee considered that the neonatal pig and minipig were appropriate to model the human infant from 0 to 12 weeks of age, with respect to gut closure and immunological development, and were satisfied that infant formula containing CGN did not result in adverse effects on the gut or on immune parameters. The margins of exposures between the NOAEL from the pig study and human infant exposures at 2–4 weeks of age ranged from 2 to 12 on a body weight basis. Although acknowledging that the MoE is very small, JECFA noted that it was derived from a neonatal pig study without adverse effects on the gut or on immune parameters and also considered that the total toxicological database on CGN did not indicate toxicological concerns. JECFA concluded that the use of CGN in infant formula at concentrations of 300 mg/L for general use, and up to 1000 mg/L for FSMPs is not of concern. 65
If the data on the intended food grade material and intended conditions of use are considered, then we would travel down the right-hand side of the tree, which considers lack of digestibility and lack of systemic exposure. The standard toxicological data would have been adequate to conclude on the safety of CGN, even with the low MoEs for the infant situation. It can be questioned if any juvenile studies were necessary considering the totality of the data and that the existing nonhuman primate model is actually more relevant than other animal species, especially in light of the proper immunological stains. The recent piglet studies provided additional information on CGN absorption, and on immune system and GI parameters, providing more robust evidence, as required by JECFA. Nevertheless, the two juvenile studies gave exactly the same health outcomes, with the same NOAEL and MoEs. Unfortunately, proving lack of toxicity is always more difficult than proving toxicity, it is expert judgement on when sufficient data are available to exclude major uncertainties. The JECFA panel finally concluded that there is no evidence for the concerns raised. These studies also highlighted several technical difficulties when administering higher than desired use levels of materials with thickening properties. There are limitations to testing doses high enough in animal studies to obtain large margins over infant exposure due the viscosity of the material at high doses, leading to feeding, nutritional, and palatability problems. 109
Locust (carob) bean gum
Locust (also called carob) bean gum (LBG, INS 410, CAS 9000-40-2) is a galactomannan polysaccharide extracted from the endosperm seed of Ceratoniasiliqua Leguminosae Taub tree. The material of commerce is a white to yellowish nearly odorless powder. LBG consists of high-molecular-weight polysaccharides (50,000–3,000,000 Da). The polysaccharides are composed of at least 75% galactomannans consisting of a linear chain of (1→4)-linked β-
LBG is used as a thickener, stabilizer, emulsifier, and gelling agent in many different food products. LBG was allocated an ADI “not specified” by JECFA. 120 –122 In infant formula, LBG can be used as a standard technological thickener, and is already provided for in Codex Standard (CX STAN 72-1981) up to a level of 1 g/L. 25
LBG is also used in infant formula at higher levels of 5–10 g/L as a thickening agent in FSMPs for bottle feeding to provide clinically effective dietary management of gastroesophageal reflux (GER). These levels provide acceptable thickening properties for bottle feeding and clinical efficacy through dietary management. 123 The SCF in the EU previously concluded that the use of LBG in FSMP for infants for the dietary management of GER up to 10 g/L was acceptable. 123 Jurisdictions such as Europe, China, and the Russia–Kazakhstan–Belarus Customs Union currently approve the use of LBG in FSMPs at typical use levels of 5 g/L and upper end use levels of up to 10 g/L. Infant formulas containing LBG have been on the market in Europe and safely administered to many infants for more than 15 years. 123 –128 LBG may also be used in the United States as a food additive in infant formula for management of GER at a level not exceeding 5 g/L after a premarket notification. 129,130
This galactomannan polysaccharide behaves in the GIT as a standard dietary fiber and is not hydrolyzed by digestive enzymes, as demonstrated by in vitro and in vivo rat digestibility studies. 81,90,120 –122,131,132 Because of its high MW (50–3000 kDa), LBG will not be absorbed either in immature or in mature organisms and is mainly excreted unchanged in the feces. 123 A small fraction (1–2%) can be fermented by colonic bacteria as shown in animal studies by a physiological increase in cecum weight and content. 133,134 This is a common effect in animals consuming high levels dietary of fibers. 135 –140 LBG is therefore not absorbed and not systemically available, as observed for other galactomannans (i.e. cassia and tara gum) and fibers in general. 120,141,142 No major differences are expected in the ADME/toxicokinetic properties between a neonatal organism (infant) and a mature organism (adult).
In the absence of LBG digestibility and systemic availability, the single site of contact is the gut. Potential health concerns from nondigestible fibers may result from clinically relevant osmotic imbalance, gut irritation, or inflammation. 123 In infants these GI intolerances would manifest as vomiting or diarrhea, with adverse effects on growth after repeated exposure. Such clinical outcomes are logically carefully scrutinized in human clinical studies with infants, in particular, those suffering from GER (usually diagnosed 2–3 weeks after birth), who because of this condition have to be strictly monitored. To study the clinical efficacy of LBG, 13 pediatric clinical studies with 396 infants with GER from birth onward (327 infants <12 weeks old, 69 infants >12 weeks old) have been conducted, with LBG levels ranging from 3 to 6 g/Lover periods of 1 week to 3 months. 123 The totality of the pediatric clinical data demonstrates good tolerance and safety of LBG under these conditions of use. Effective clinical treatment of acute infantile diarrhea and vomiting with up to 10 g/L (and even 14–28 g/L LBG equivalents from the use of carob flour or from LBG-based thickeners) has also been reported in clinical practice. 123,143 –146
The available toxicological database for LBG is extensive (i.e. subchronic and chronic toxicity, genotoxicity, carcinogenicity, reproductive toxicity, and teratogenicity). All studies consistently demonstrated low toxicity of LBG, with NOAELs observed at the highest doses tested. NOAELs from subchronic toxicity studies range from 4500 to 12,000 mg/kg bw/d in the rat and 20,000 mg/kg bw/d in mice (i.e. up to 10% in the diet). 123 For chronic and carcinogenicity studies, the NOAELs in chronic and carcinogenicity studies were at least 2500 mg/kg bw/d in the rat and 7500 mg/kg bw/d in mice (i.e. about 5% in the diet, the top dose tested in chronic studies). The low toxicity is further confirmed by comparison to available toxicological data from closely related galactomannan substances. 121,140,141
For infants aged 0–6 months, and a typical use level of 5 g/L formula as consumed, the clinical data and history of use support a conclusion of safety for the intended use. Data from animal studies give complementary support. The absence of systemic availability (no difference in endogenous metabolism between humans), the available strong pediatric clinical data set (no evidence of adverse tolerance effects in human infants), and the absence of any characterized toxic effect in animal toxicity studies, particularly in the GIT target organ, demonstrate the safety of LBG as a functional thickener in infants with GER. There are no alerts that would indicate additional toxicological data are needed for levels up to 10 g/L in FSMP infant formula for the dietary management of GER (as currently approved in the EU). 77 This additive has been on the market for the intended application in the dietary management of GER for several years in many areas of the world, 123 without reported adverse outcomes. Persistent GER may lead to decreased nutrient intake, failure to thrive, and an increased risk of health problems such as respiratory illness which can be very detrimental in term infants, and the addition of LBG clearly reduces the number and frequency of GER episodes. 126,127
Based on the available evidence, the lack of digestibility and systemic availability justifies the assumption of a similar ADME behavior between infants and adults in the decision tree. No alert on any type of toxic or allergic effect was observed from the considerable amount of (pre)clinical studies up to the highest dose tested. The availability of at least 13 good quality clinical investigations in the actual target population, addressing the relevant effects of a fiber-like substance, that is, tolerance and nutritional adequacy, would exclude the need for an additional juvenile toxicity study or an extra safety factor for the derivation of a safe level for term infants. An MoE of at least >1 with the NOAEL from clinical data in the target population at the intended use level of 5 g/L is considered sufficient. It is however acknowledged that the reported clinical use and clinical investigations up to 10 g/L or higher in other infant target groups (i.e. acute diarrhea, vomiting) are not robustly documented in the public literature and taken alone could be considered to only partially cover part of the safety in use. However, when the full safety data set is taken together, one can consider this level as acceptable under the positioning as food for special medical purpose being prescribed under medical supervision.
Nonetheless, at its 82nd meeting, the JECFA concluded that the available studies were not sufficient for the evaluation of LBG for use in infant formula at the highest proposed use level (10 g/L) 77 and requested toxicological data from studies in neonatal animals to complete its evaluation. 77 This application of LBG provides infant formula with the functionality to support infants with GER, so to study this in a juvenile animal model without underlying GER condition would not represent the conditions of the intended use of 10 g/L. Given the physical properties of LBG and limitations to high-dose administrations in animals, it is unlikely that additional juvenile studies would contribute to establishing a larger MoE. To assure safety at 10 g/L for FSMPs, and that an MoE of <1 is acceptable, human clinical data in the target infants at this use level would be more relevant.
Xanthan gum
Xanthan gum (INS 415) is a hetero-polysaccharide with a very high MW (between one and several million) produced by fermentation with Xanthomonas campestris. Beta-
Intended use levels in both infant formula and FSMPs is 1 g/L, leading to conservative intake of up 222 mg xanthan gum/kg bw/d. Xanthan gum is used in a variety of general foods as a thickener, stabilizer, emulsifier, and foaming agent. JECFA has previously established an ADI of not specified based on the lack of toxicity in animal and human studies. 140 Results of in vitro studies, as well as studies involving oral administration of xanthan gum to rats, show that xanthan gum is largely (98%) not digested by the digestive enzymes in the upper GIT and is poorly absorbed. 147 –149 Several adult human clinical trials with doses up to 10–15 g/d for approximately 4 weeks did not raise any safety issues, other than mild GI effects which are consistent with the consumption of a largely indigestible polysaccharide. The toxicological database of xanthan gum includes a subchronic (90 days rodent feeding) study and a 2-year carcinogenicity study in rats each with an NOAEL of 1000 mg/kg bw/d, and a three-generation rat study with an NOAEL of 500 mg/kg bw/d. 150,151 These NOAELs were the highest doses tested. Although experimental mutagenicity/genotoxicity data were not identified, there is an absence of any such structural alert. Long-term studies in rats and dogs demonstrated the absence of any effects on tumor incidence. 151 Dietary dog studies over 12 and 107 weeks with up to 1000 mg/kg bw/d demonstrated a lack of toxicological effects. 151 However, a conservative NOAEL was considered at 250 mg/kg bw/d in both studies due to softer stools than controls at higher doses, and a slight reduction in growth in males, neither of which were considered treatment related.
Xanthan gum at concentrations of up to 1500 mg/L in infant formulae fed to infants from birth onward for periods of 10–112 days of age was well tolerated. 152 In particular, in a 16-week growth study, formula with xanthan gum at a concentration of 750 mg/L was shown to support normal body weights and weight gains in infants. 96 This clinical study reported no undesirable effects on stool characteristics in infants consuming 750 mg/L xanthan gum–containing formula. Postmarketing surveillance information received over a 5-year period on a xanthan gum–containing infant formula intended for use in infants with milk protein allergies indicated no safety issues when used as recommended.
Additional studies were performed in juvenile piglets specifically designed to assess the safety of xanthan gum in infant formula and formula for special medical purposes. A first study in juvenile piglets with doses of 750 and 7500 mg/L in piglet adapted formula demonstrated reduced body weight gain and feed efficiency and increased weights of the large intestine at 7500 mg/L. 153 A second study in juvenile piglets added a more realistic intermediate dose of 1500 mg/L, more in keeping with real use levels (of 1 g/L) in infant formula. 153 The NOAEL was identified at this dose of 1500 mg/L, equivalent to 750 mg/kg bw/d. This NOAEL of 750 mg/kg bw/d compared with the most conservative estimate of xanthan gum intake of 222 mg xanthan gum/kg bw/d by infants provides an MoE of 3.4. JECFA has previously commented that when all relevant uncertainties or conservatisms in the toxicological data and/or the exposure estimates were taken into account, an MoE in the region of 1–10 could be interpreted as indicating a low risk for the health of 0- to 12-week-old infants consuming the food additive in infant formula. 92
The lack of digestion and systemic absorption, the absence of toxicological effects in animal studies, and the knowledge of high-dose intake on GI tolerance in adults indicate the suitability for use of xanthan gum in infant formulas needs to be targeted on the nutritional adequacy to support growth and to be tolerated in the target population at the intended use levels. The existing clinical data, postmarketing surveillance data, and history of use should be sufficient to conclude that the consumption of xanthan gum in infant formula or formula for special medical purposes intended for infants is of no concern at use levels up to 1000 mg/L as consumed. When using the proposed decision tree, we would stop in the early section, since there are no significant differences in the toxicokinetics and target organs between adult and juvenile populations, and the safety of the intended use levels was demonstrated by clinical data (i.e. an MoE of 1 to clinical data is acceptable). The additional piglet studies did not provide additional information but rather highlighted that administering high-dose indigestible fibers in neonatal animals, especially at levels greater than intended use levels, can cause antinutritional effects and does not help to derive large MoEs.
Pectin
Pectin (INS 440, CAS No 9000-69-5) is a high MW (100–200 kD) soluble fermentable fiber originating from fruits such as citrus and apple. It is a complex hetero-polysaccharide, consisting mainly of partial methyl esters of polygalacturonic acid and their ammonium, sodium, potassium, and calcium salts. Pectin is a common constituent in the human diet and is present in virtually all fruits and vegetables.
Pectin is considered as Generally Regarded As Safe (GRAS) in the United States 154 and permitted for use as a gelling, thickening, and stabilizing agent, in general foods worldwide. It has been assessed by JECFA on six separate occasions, with an ADI established as not specified for pectin and amidated pectin originally in 1981. 122 In infant formula, pectin is used as a thickener to stabilize the product and maintain product homogeneity, by reducing protein sediments and maintaining stable emulsion both during thermal processing and throughout shelf life. Pectin is listed by CODEX 155 for use in formula for infants 6 months and above, and for young children, at a level of 1% (1 g/100 mL). Pectin is permitted for up to 1% (1 g/100 mL) in FSMPs intended from birth onward in the EU. 156
Pectin is not digested in either humans or animals but is metabolized extensively by the intestinal microbiota to oligogalacturonic acids, which are then further metabolized to short-chain FAs. 68,157,158 Pectin-derived acidic oligosaccharides (pAOS) produced by enzymatic hydrolysis of pectin have also been studied as food additive, and since pAOS is similar to products formed from pectin in the GIT, studies with pAOS are relevant to the safety evaluation of pectin. Absorption and systemic exposure of pectin as such is negligible.
Pediatric clinical studies with formulas containing pectin (up to 0.09%) or pAOS (up to 0.2%) in both preterm and healthy infants from birth onward up to 3 months support normal growth and no adverse effects at these levels were reported. 159 –163 The toxicological database on pAOS demonstrates no genotoxicity. 68 No relevant health effects were observed in a 90-day toxicity study in young rats preceded by an in utero exposure phase, at maternal doses of pAOS up to 7.1 g/kg bw/d, the highest dose tested. 68,164 Thus, all available toxicological and clinical data indicate that pectin has no systemic uptake nor toxicity. 68
Safety studies in neonatal pigs were conducted as additional support for recent submissions to JECFA for pectin evaluation at 0.5%, a level considered sufficient to meet the technological need. Neonatal pigs were fed diets containing 0%, 0.05% (0.5 g/L), 0.3% (3 g/L), or 1% (10 g/L) pectin for 21 days and evaluated for toxicological and growth end points. 68 Only minor changes in the clinical pathology parameters were observed, mostly at the highest dose of 10 g/L, and were not considered toxicologically meaningful. There were also increases in cecum and colon weights at 3 and 10 g/L in both males and females, which is consistent with previous data from pectin and other dietary fibers and is considered normal. The finding that no overt toxicological effects were observed in the neonatal pigs 68 is consistent with the toxicological data that had previously been generated and published for pectin in a variety of model systems. 68 Decreased feed consumption and decreased body weight gain were however observed at the 1% level in males. JECFA assigned an NOAEL at the 0.3% dose level (nominal value (calculated) of 847 mg/kg bw/d but demonstrated to be 1049 mg/kg bw/d using actual analyzed dietary pectin 77 based on the growth results. The available clinical studies collected at that time were mainly conducted with pectin or pectin-derived oligosaccharides at concentrations of 0.2% or less, and no target population relevant juvenile study was available at the intended use level. JECFA therefore concluded there was no evidence to demonstrate tolerance and normal growth at the proposed use level of 0.5%. 68 An additional piglet study was performed with pig adapted formula containing 0, 0.2% (2 g/L) or 1% (10 g/L) pectin for 21 days, to support a further JECFA evaluation for 0.2% levels of pectin infant formula. 77 Pectin negatively impacted feed intake and growth at 1%, as before, but confirmed the absence of effects at the 0.2% use level.
Nutritional effects at the top dose in piglet studies would not be unexpected, since 1% pectin inclusion led to an increase in dietary viscosity. As discussed for other fibers in this document, increases in viscosity and concomitant decreases in digestibility have been observed with high levels of pectin and other dietary fibers, and are linked with decrease feed intake. 165 –183 Thus, the impact of high levels of pectin on growth in the piglet trials is due to delayed gastric emptying and/or prolonged gut transit resulting from the viscosity of the material rather than a toxicological effect. In studies in older growing pigs (greater than 6 weeks old and older), growth effects were not observed at pectin exposures up to 6000 mg/kg bw per day, well above the exposures that impacted growth in the neonatal piglets. 172,175 Most of the studies in pigs that have reported an impact of pectin on digestibility have not reported a decrease in growth. Many of these studies were performed at lower exposure levels of pectin, and/or for shorter durations, and/or in older pigs that have a more developed digestive capacity than the neonatal pigs used in the present study. 172 –174,179,180,184 –187
When considering pectin in our proposed decision tree, there is an extensive literature on its toxicological, ADME, and nutritional properties, to indicate that there are no differences in digestion and target organ between juveniles and adults. The totality of the evidence with previously published toxicological data, and the demonstration of adequate growth and tolerance of pectin in clinical pediatric data already cover the safety of 0.2% use levels. Neonatal piglet studies would not have been necessary. The juvenile animal studies demonstrated adverse nutritional effects at 1% levels of pectin in formula fed to piglets which were not seen in older piglets fed higher levels of pectin. 175,182 In general, these nutritional effects could be expected at high levels of pectin based on its physical characteristics and functional properties, although the precise levels at which they occur would need to be determined by in vivo data. Differences in susceptibility in nutritional effects at the higher dose levels early in life could not be excluded based on animal data existing at the time. In the proposed decision tree, this could indicate the need for additional juvenile studies. Unfortunately, the targeted 0.5% use dose level was not included in the subsequent piglet studies. However, since there is reason to believe that juvenile piglets may be more sensitive to dietary fibers than are human infants, 47,56 and given that levels of 0.5–1% pectin area already authorized and used in some jurisdictions, specific human infant data at the petitioned use level would have been more relevant to demonstrate tolerance. There is some quite recent evidence from clinical trials in infants aged 1–12 months old (mean age at inclusion of 4.8 ± 3 months fed with formula containing fibers, consisting mainly of pectin, at 0.5 g/100 mL (5%), 188 and in other similar trials 188 demonstrating adequate growth.
Conclusions
In this article, we presented an integrated approach to assess the safety of additives for use in early life. A decision tree has been developed and is proposed as a means to help assess requirements to address data gaps and harmonize ongoing efforts. In this article, we focused on relevant, fit-for-purpose assessments, to be in a position to better assess what additional information is needed to ensure a robust evaluation for sensitive populations. This approach highlights that when significant uncertainties in the existing safety data for the intended target population are apparent, then expert judgment is needed to decide if additional data in juvenile models should be generated. A case-by-case analysis of data is important whether new additives for use are under consideration or when a gap analysis is conducted with existing data including safe history of use. The approach was tested on six additives that have been recently supported by the industry. When thought relevant, data gaps were filled to finalize the risk assessment in the context of use in infants below 12 weeks of age. Leading in the level of additional data considered necessary to assess safety in early life were the available knowledge on the type of additive, its kinetic behavior, the existence of standard (pre)clinical data, and available clinical data/postmarket surveillance data.
Several key criteria to consider when reviewing existing databases are highlighted. Some of these criteria are not specific to assessment in early life. The material must be well characterized with appropriate specifications, and any studies considered in the assessment must be related to that material. As we have seen in the case of CGN, much confusion and unnecessary testing arose because the focus was often on the wrong material, poligeenan. CGN itself is not degraded under the conditions in the infant or adult GIT, and there was no evidence of inflammatory responses when administered under dietary conditions. The digestibility and toxicokinetics of the additive also are important to consider. Experimental data indicated that CITREM and OSA-modified starch are not metabolized by the juvenile in a significantly different manner, and is comparable, to the adult organisms, and that the metabolites were not of concern with respect to the critical biological systems, leading to the conclusion that there are no gaps for which additional juvenile toxicological studies are needed. The clinical evidence and history of use supporting safety must include the top dose levels being applied for. Unfortunately, in the cases of pectin and LBG, the clinical data did not adequately cover the top dose, and marketing surveillance data were limited. In vivo data will always be required as proof of absence of concern. Ideally, this will be clinical data in the target infant population, with robust study designs to cover the infant feeding period, and relevant parameters investigated.
When reviewing animal toxicological data, the mode of administration and the technical characteristics of the additive can affect the outcome of studies. In the case of the emulsifiers and thickeners reviewed in this document, there are physical limitations to the top levels used in the food matrices, which can result in decreased palatability and feed intake, and hence reduced growth and possibly other nutritional effects in the animals. It could also be the case that the animal models chosen may be more sensitive to, for example, GI tolerance, as suggested by our case studies, and so careful interpretation and extrapolation to the infant situation is needed. As JECFA has noted, if additives are proposed at relatively high levels (e. g. >0.01%), leading to relatively high infant exposures on a body weight basis, then regular toxicological feeding studies in animals at doses of high enough magnitude to cover the traditional MoE approach, may not be possible, although much lower MoEs can be acceptable depending on the adequacy of the data. 92 This is already recognized for novel food ingredients, where traditional 100× uncertainty factors for extrapolation to the human situation may not be achievable and would lead to nutritional imbalances. 189
Although an MoE is a numerical way to express a level of safety, it needs to be interpreted within the specific context. The PoD on which it is based should be considered during the study design. It is clear that very low MoEs will often be derived from animal feeding studies compared to the human infant exposure, and so it is important that any in vivo study (clinical, animal) uses test material with concentrations that covers the use levels for the eventual additive dossier. Parameters and end points to investigate can be selected from analysis of the existing toxicological database in adults, or otherwise broadly chosen to cover important organ systems. The intended use levels should not result in any adverse effects. Derivation of an MoE from juvenile animal models should not be the only deciding factor in the safety assessment. When the MoE is used, JECFA has indicated that low MoEs (often <10) may be acceptable when considering the whole of the database on a substance.92 Provided that the total body of evidence accumulated on the additive indicates that a human neonate/immature organism will not behave significantly differently than a mature organism, for given doses and exposure times, then an MoE can be derived from standard (sub) chronic feeding studies.
Further work and recommendations
Further research may be warranted when knowledge gaps have been identified and there remains uncertainty regarding safe levels in early life compared to adults; for example, development of model systems mimicking infant conditions, other than in vivo animal investigations; determination of what specific parameters to address in each organ system relevant to address adversity in early life. Research into relevant in vitro model systems is warranted, especially in view of current ethical consideration on performing unnecessary animal studies. A further important factor to consider is to how better address the impact on later life as a result of early exposure since current juvenile studies rarely include groups of animal up to adulthood.
Further investigations can be made to determine how the approach described here can be extended to address more complex ingredients and whole formulas, novel materials with biological function, new processing conditions as well as chemical contaminants. Organization of a workshop with relevant stakeholders, research institutions, and international scientific assessment bodies to discuss the topic in a wider context would provide a good forum to harmonize approaches.
Although this document focuses mainly on technological food additives, the considerations discussed in this document, and the decision tree approach can also be applied to the assessment of nutritional ingredients in infant formula. For example, lutein 190 and lactoferrin 66 are present in breast milk, and intended levels in the final formula are within levels detected in human breast milk. 66,190 Therefore, the metabolism and biological effects of these materials in infant formula should be the same or very similar. When animal toxicological data have been generated, expert groups have considered that a standard 90-day toxicological study was sufficient, and that the final conclusion on safety, nutritional adequacy, and tolerance of formula containing these nutrients was derived from clinical studies under real feeding conditions. In recent years, some novel ingredients 64,67 have been subjected to investigation in juvenile animal studies, followed by human infant studies. 64,67 The results confirmed what was expected and provided additional reassurance of no harm.
Summary
In summary, uncertainty on safety of an additive in the infant population can be decreased when good quality pediatric clinical and postmarket surveillance data at the requested use level are available and a holistic weight-of-evidence approach is taken. Specifically in the cases discussed in this document, generating additional data in juvenile model systems has sometimes but not always brought additional, previously unexpected, information to the assessments. We consider therefore that the systematic use of juvenile animal studies for food additives should not be a standard requirement. If required, a targeted study designed on a case-by-case basis should be preferred. The metabolism of the additive, and potential target organs, and its potential nutritional impact must be part of the decision-making process. In the absence of indication that critical organs and developmental systems will be targeted, then assessment based on reliable human pediatric data should be considered as the gold standard.
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Drs. Constable, Garthoff, Mahadevan, Meunier, and Mr O’ Sullivan are employed by industry. Drs Garthoff, Mahadevan, Meunier, and Mr O’ Sullivan represent the sponsors of some of the additives submitted for JECFA review and discussed as case studies. The non-industry co-authors of this paper listed below are identified as Consultants, providing advice on toxicological and risk assessment issues to private firms. Drs. Hayes, Pressman, Schrenk and Speijers acted as expert consultants through ISDI under a cost reimbursable contract for their contribution to this manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors acknowledge the financial support from the International Special Dietary Foods Industries (ISDI) which led the project. The authors particularly thank Nuria Moreno Odero, International Special Dietary Foods Industries (ISDI), and Allison Cooke, Infant Nutrition Council of America (INCA) for diligent project management, coordination of comments on the manuscript and overall assistance with formatting/referencing for journal submission of the article.
