Abstract
The chronic intake of different medications by chronic kidney disease (CKD) patients predisposes them to extremely harmful and clinically nonbeneficial drug–drug interactions (DDIs) which can ultimately lead to increase in morbidity, mortality, healthcare cost, and frequency and length of hospitalization. This produces a negative deteriorating and counter-efficient outcome on the health, quality of life and treatment response of these patients. This was an 18-month prospective descriptive study that reviewed the medical case records of consented adult CKD patients attending the Nephrology medical outpatient clinic of a Nigerian Tertiary Healthcare Centre from January 2015 to June 2016. The Medscape drug reference database was used to evaluate patients’ medications for extremely harmful, clinically nonbeneficial DDIs. This study involved 123 consented adult CKD patients comprising of 82 (66.67%) males and 41 (33.33%) females with a mean age of 53.81 ± 16.03 years. In this study, the prevalence of extremely harmful, clinically nonbeneficial DDIs (type D or type X interaction categories only) was 24.4%, while the overall prevalence for all the observed DDIs was 95.9%. The most frequent extremely harmful, clinically nonbeneficial DDIs in this study was between α-methyldopa and metoclopramide: 16 (0.9%) interactions in eight (6.5%) patients. Furthermore, α-methyldopa decreases the antiemetic effects of metoclopramide by pharmacodynamics antagonism at the chemoreceptor trigger zone site D2-receptors (type X; pharmacodynamics). In addition, metoclopramide decreases the level of α-methyldopa by inhibition of gastrointestinal tract (GIT) absorption, as this applies to only oral formulations of both agents (type D; pharmacokinetic). The occurrence and burden of extremely harmful, clinically nonbeneficial DDIs is significantly high among these CKD patients. There is also a critical need to minimize the number of prescribed medications for these patients in order to optimize their care.
Introduction
Drug–drug interaction (DDI) can be defined as an appreciably harmful or beneficial process whereby the pharmacological effects of a drug (or object drug) is directly or indirectly influenced and modified by the presence of another drug (or precipitant drug) which can result in either treatment failure (antagonistic interaction) or drug-induced toxicity (synergistic/additive interaction). 1,2 DDIs have also been observed to be one of the most frequently occurring challenges that may alter the pharmacokinetic and pharmacodynamics profiles of drugs. They have the potential of altering the overall therapeutic response clinically. DDIs can be pharmacokinetic, pharmacodynamics, or pharmaceutical in nature. 1
Pharmaceutical interactions occur when two drugs are given together concurrently in the same infusion solution or when a drug reacts with the infusion solution (e.g. between intravenous (IV) ceftriaxone and IV calcium-containing solutions such as Ringer’s lactate, calcium gluconate, or Hartmann’s solution). While it is necessary to be aware of this type of interaction, it is relatively uncommon in clinical practice. 3
Pharmacodynamics interactions are those interactions where the effects of a drug (or object drug) are changed by the presence of another drug (or precipitant drug) at its site of action, without any alteration in the concentrations of both drugs. Sometimes one drug competes directly with another for a particular receptor, but often the reaction is more indirect and involves the interference with physiological mechanisms, making pharmacodynamics interactions more difficult to classify than pharmacokinetic interactions. In pharmacodynamics interaction, the process of interaction may involve the precipitant drug altering the sensitivity or responsiveness of tissues to the object drug by having the same (agonistic) or a blocking (antagonistic) effect; these effects usually can occur at the membrane surface receptor level but may also occur intracellularly. 4
On the other hand, pharmacokinetic interactions may be defined as those interactions in which the disposition of a drug (or object drug) is altered by the presence of another drug (or precipitant drug). As a result, the effect of the object drug is either diminished or increased. Pharmacokinetic interactions are divided into those that affect absorption, distribution, plasma protein binding, metabolism, and excretion. It therefore changes the concentration/amount and the persistence of available drug at the receptors’ sites. Pharmacokinetic interactions inevitably alter the magnitude and duration of drugs’ effects, and these effects are either predicted based on our scientific knowledge of these individual drugs or detected by therapeutic monitoring of these drugs’ concentrations in the plasma or at the specific site of action if feasible and in addition checking out for obvious or detectable clinical signs. 5
Extremely harmful, clinically nonbeneficial DDIs are those interactions that belong to the level of clinically significant categories of either major/serious interactions (type D interactions) or contraindication/avoid drug combination (type X interactions) in which the risk of adverse drug effects (ADEs) associated with the concomitant use of these two drugs usually outweighs their benefits in the patient after each individual patient-specific assessment has been done.
Chronic kidney disease (CKD) can be defined as a progressive and irreversible deterioration in the renal function of an individual over a period of at least 3 months regardless of the underlying aetiology. 1,6 In Nigeria and worldwide, poorly controlled chronic hypertension, poorly controlled diabetes mellitus, and human immunodeficiency virus (HIV)-associated nephropathy among others are the common causes of CKD. 3,4,7 CKD is associated with rising incidence and prevalence, high cost of treatment, and poor outcomes. There is evidence to suggest that early in the course of CKD, appropriate interventions may slow down its progression or completely halt the progression of the disease. Despite this, many patients with CKD still present late to the nephrologists, so that at the time of initial patient assessment, all that can be offered is preparation for renal replacement therapy. 1,3 –6,8,9 This is particularly so in resource-poor setting where, among several other factors, the lack of awareness, traditional beliefs about the cause and nature of the disease, the need to pay out-of-pocket for healthcare, and shortage of specialists combine to promote inappropriate healthcare-seeking behavior and late presentation to the nephrologist. Usually, in CKD, there is a progressive loss of kidney function occurring over several months to years and is characterized by the gradual replacement of normal kidney architecture with fibrous tissue. 3,5 The individual with the end-stage renal disease (ESRD) can no longer adequately excrete waste products, regulate acid–base balance, and maintain sodium, potassium and water homeostasis. There are other associated complications such as anemia, hyperkalemia, metabolic acidosis, inability to maintain calcium balance (hypocalcemia), erectile dysfunction, hyperphosphatemia, and increased cardiovascular disorders such as hypertension, stroke, ischemic heart diseases, cardiac arrhythmias, and sudden cardiac arrest, which can lead to increase in mortality. 1,3 –9 Patients with CKD stages 1–3 (estimated glomerular filtration rate (eGFR) > 30 mL/min/1.73 m2) are generally asymptomatic. Typically, it is not until stages 4–5 (eGFR < 30 mL/min/1.73 m2) that endocrine and metabolic derangements or disturbances in water and electrolyte balance become clinically manifest. The early stages of CKD are often completely asymptomatic, despite the accumulation of numerous metabolites. Serum urea and creatinine concentrations are measured in CKD since methods for their determination are available and a rough correlation exists between serum urea and creatinine concentrations and symptoms. These substances are, however, in themselves not particularly toxic. 1,3 –6,8 The nature of the metabolites that are involved in the genesis of symptoms is unclear. Such metabolites must be products of protein catabolism (since dietary protein restriction may reverse symptoms associated with CKD) and many of them must be of relatively small molecular size (since hemodialysis employing membranes which allow through only relatively small molecules improves symptoms). Little else is known with certainty. Symptoms are common when the serum urea concentration exceeds 40 mmol/L, but many patients develop uremic symptoms at lower levels of serum urea. 1,3 –9
Hypertension and diabetes mellitus are the commonest causes of end-stage renal disease (ESRD) worldwide; therefore, control of high blood pressure and optimization of blood glucose level are essential in delaying and retarding CKD progression. 3 –7 These necessitate the use of several medications to improve the quality of life of these patients and slowing down the progression of early CKD to full-blown ESRD. Although efficacy, compliance, and economic factors are considered in selecting drugs for treating patients with CKD, these patients are usually on polypharmacy with attendant risk for drug-related problems such as DDIs and adverse drug reactions (ADRs). 4 –6 Polypharmacy can be defined as the concurrent use of more than five different medications by a patient for the treatment of a particular disease or group of diseases. 2,10 As the disease progresses, the number of drugs prescribed usually increases substantially; sometimes, up to 10 or more different drugs are ingested daily by the time a patient is at CKD stage 5 (eGFR < 15 mL/min/1.73 m2). 2,4,5,8,10 The influences of the disease on drugs’ pharmacokinetic and pharmacodynamics mechanism increase the risk for drug-related problems such as DDIs and ADRs; these further compound the problems of patients with CKD and may further increase morbidity, mortality, healthcare cost, length and frequency of hospitalization, among them. 11 –14 ADRs can be defined as an undesirable effect of a drug beyond its anticipated therapeutic action occurring during clinical use or any noxious and unintended response to a drug that occurs at doses used in humans for prophylaxis, diagnosis, or therapy of disease. 15,16 ADRs have been classified as mild (laboratory abnormality or symptom not requiring treatment), moderate (laboratory abnormality or symptom requiring treatment or admission to hospital or resulting in nonpermanent disability), severe (laboratory abnormality or symptom that was life-threatening or resulted in permanent disability), and fatal (when it resulted in patient death). 16,17,26
DDIs are major clinical problems, accounting for 2–6% of all hospital admissions with estimated annual cost to the National Health Service of about £500 million in the United Kingdom. 15 In a meta-analysis of 39 prospective studies from hospitals in the United States, it was shown that DDIs ranked fourth to sixth as a leading cause of death. 16 Recent studies in the United States have also indicated that DDIs also increase the cost of treatment, length and frequency of hospitalization. 16,17 The prevalence of DDIs in CKD patients from previous reports ranged between 56.9% and 89.1%. 18 –23 This relatively high prevalence is related to the polypharmacy that is involved in the management of this condition. It is associated with increased morbidity, mortality, healthcare cost, and length and frequency of hospitalization. 2,9,10,24,25 These added burdens of extremely harmful, clinically nonbeneficial DDIs and their consequent hazardous adverse effects in CKD patients are preventable because of their predictable nature. Various software are available that can detect DDIs such as British National Formulary, Drug Interaction Facts, Medscape, Epocrates, Lexi-Interact, Harmavista and Stockley’s Drug Interaction Checker. 27,29
CKD is a major public health problem due to its increasing incidence, prevalence, and associated high burden. The global prevalence of CKD is estimated to be 11–13%. 15 –17,26 The prevalence of CKD in Nigeria varied between 11.4% and 18.8% from both community- and hospital-based studies. 1,3,5,6,8 Cardiovascular disease burdens in CKD patients are high and associated with increased frequency of hospitalization, morbidity, mortality, length of hospital stay, and healthcare cost. 7,18 –20 Cardiovascular risk factors such as hypertension, diabetes mellitus, anemia, calcium–phosphate abnormalities, hyperuricemia, and left ventricular hypertrophy are highly prevalent in CKD. 7,21 –23 These are largely responsible for cardiovascular disease burden and some complications in CKD patients. Management of these cardiovascular diseases and risk factors is important in retarding the progression of CKD and reducing its associated mortality. 9,24,25 This involves the use of multiple drugs’ combination therapy in the management of CKD and its attendant complications; hence, polypharmacy is often practiced. 10 The consequences of polypharmacy include poor patients’ medication adherence due to high pill burden, increased cost of healthcare, and most importantly, drug-related problems such as DDIs and ADRs which can have deleterious effects on patients’ health. 2,10,27 DDIs may result from a number of processes, which are related to either pharmacokinetic or pharmacodynamics properties of the drugs. The consequences of DDIs include increase risk for toxicity or decrease in the efficacy of treatment. The clinical pharmacologists and clinical pharmacists also have major role to play in preventing drug-related problems such as DDIs and ADRs associated with polypharmacy by thoroughly and regularly evaluating prescriptions for CKD patients before rational dispensing. 7,28,29 Therefore, integrated professional interaction should be encouraged between nephrologists, clinical pharmacologists, clinical pharmacists and nephrology nurses in order to optimize CKD patients’ care. Vigilance by healthcare workers such as nephrologists, clinical pharmacologists, clinical pharmacists and nephrology nurses in detecting, diagnosing, and reporting drug-related problems particularly in at-risk individuals such as CKD patients is vital for continued drug safety monitoring. 30 –33
This study was designed to unravel the pattern of extremely harmful and clinically nonbeneficial DDIs among CKD patients attending the nephrology clinic of a Nigerian Tertiary Healthcare Centre. To highlight the importance of prevention, early identification, correction, and monitoring of drug interactions with prompt treatment of any associated ADRs that may occur. This will create awareness concerning the occurrence and burden of extremely harmful, clinically nonbeneficial DDIs among CKD patients in renal practice and will bring to limelight the need to regularly evaluate the prescriptions of CKD patients for medication-related problems such as DDIs and ADRs. It will also help to manage any associated complications and comorbidities appropriately.
Methods
This was a prospective, descriptive study carried out at the Nephrology Clinic of a Tertiary Kidney Care Centre, University of Medical Sciences, Ondo City, Ondo State, Nigeria. The hospital receives referral from within and outside the state. One hundred and twenty-three (123) consented adult CKD patients who were being managed at the center over an 18-month period between January 2015 and June 2016 were recruited for the study. Patients below the age of 18 years, those being managed for acute kidney injury, and adult CKD patients who did not grant their informed consent were excluded from the study. The medical case records of all the adult CKD patients were retrieved after a verbal informed consent had been obtained from each of them, and the following information was extracted using a pro forma: sociodemographic data; stage of CKD; number and list of medications at the time of last clinic visit for outpatients and at the time of discharge for those who received inpatient care; number and list of comorbidities such as hypertension, diabetes mellitus, obesity, heart failure, HIV infection, and stroke. The serum creatinine level was used to calculate the eGFR using CKD Epidemiology Collaboration formula, and CKD staging was done using eGFR as follows: stage 1 (eGFR of ≥90 mL/min with evidence of kidney damage), stage 2 (eGFR of 60–89 mL/min with or without evidence of kidney damage), stage 3 (eGFR of 30–59 mL/min with or without evidence of kidney damage), stage 4 (eGFR of 15–29 mL/min with or without evidence of kidney damage), and stage 5 (eGFR <15 mL/min with or without evidence of kidney damage).
28
The Medscape drug reference database was used to evaluate patients’ medication regimen for extremely harmful, clinically nonbeneficial DDIs. Medscape drug reference database has been reported to show the best results in precision analysis.
29
The Medscape drug reference database system provides accurate information about the risk, type, mechanism, and pattern of DDIs. It also gives recommendation(s) on how to prevent and manage DDIs if they occur. The software identifies and classifies DDIs according to their level of clinical significance in categories. These categories are types A, B, C, D, and X: Type A: no known interaction Type B: minor or mild interaction Type C: moderate or significant interaction Type D: major or serious interaction Type X: contraindication or avoid drug combination
Extremely harmful, clinically nonbeneficial DDIs are those interactions that belong to the level of clinically significant categories of either major/serious interactions (type D interactions) or contraindication/avoid drug combination (type X interactions) in which the risk of ADEs associated with the concomitant use of these two drugs usually outweighs their benefits in the patient after each individual patient-specific assessment has been done. The classification of DDIs according to their level of clinical significance in categories is shown in Table 1.
Classification of drug–drug interactions according to their level of clinical significance.

The mean for all DDIs observed per prescription was obtained by dividing the total number of all DDIs observed by the total number of all prescriptions issued.
The mean for all extremely harmful, clinically nonbeneficial DDIs observed per patient belonging to these extremely harmful interaction categories was obtained by dividing the total number of all extremely harmful, clinically nonbeneficial DDIs observed by the total number of all patients belonging to these extremely harmful interaction categories.
The mean for all extremely harmful, clinically nonbeneficial DDIs observed per prescription was obtained by dividing the total number of all extremely harmful, clinically nonbeneficial DDIs observed by the total number of all prescriptions issued.
Ethical clearance for the study was obtained from the Health Research Ethical Committee of the Tertiary Kidney Care Centre. The Ethical Clearance/Protocol Research Number issued for the study was UNIMED/KCCO/NOV 2014/0007. In addition, a verbal informed consent was obtained from each of the adult CKD patients whose medical case records were used, while the medical case records for those who did not grant their informed consent were excluded from the study. Consent was sought from patient’s relative where the patient had impaired level of consciousness. Participants’ confidentiality was respected and maintained by ensuring that no unauthorized person had access to the information on the questionnaires, no information could be traced to the respondents (as coding system was used for the questionnaires instead of writing the patients’ names on them), and no unauthorized use of information was made. Those who were at risk of extremely harmful DDIs were monitored closely, and feasible alternative prescriptions were made where the concomitant risk and adverse effects outweigh the benefits.
Data collected were encoded and analyzed using the Statistical Package for the Social Sciences (SPSS) Version 17 (released 2008; SPSS Incorporations, Chicago, Illinois, USA). Results were expressed as mean ± standard deviation or using frequency and percentage values where necessary. The t-test and chi-square test were used to compare means and proportions, respectively. The level of statistical significance was set at P<0.05.
Results
This study involved 123 consented adult CKD patients comprising of 82 (66.67%) males and 41 (33.33%) females. The mean age of the study participants was 53.81±16.03 years. Forty-eight (39.0%) patients were between 20 and 49 years, 52 (42.3%) patients were between 50 and 69 years while the remaining 23 (18.7%) patients were 70 years and older (see Table 2).
Characteristics of the study population.
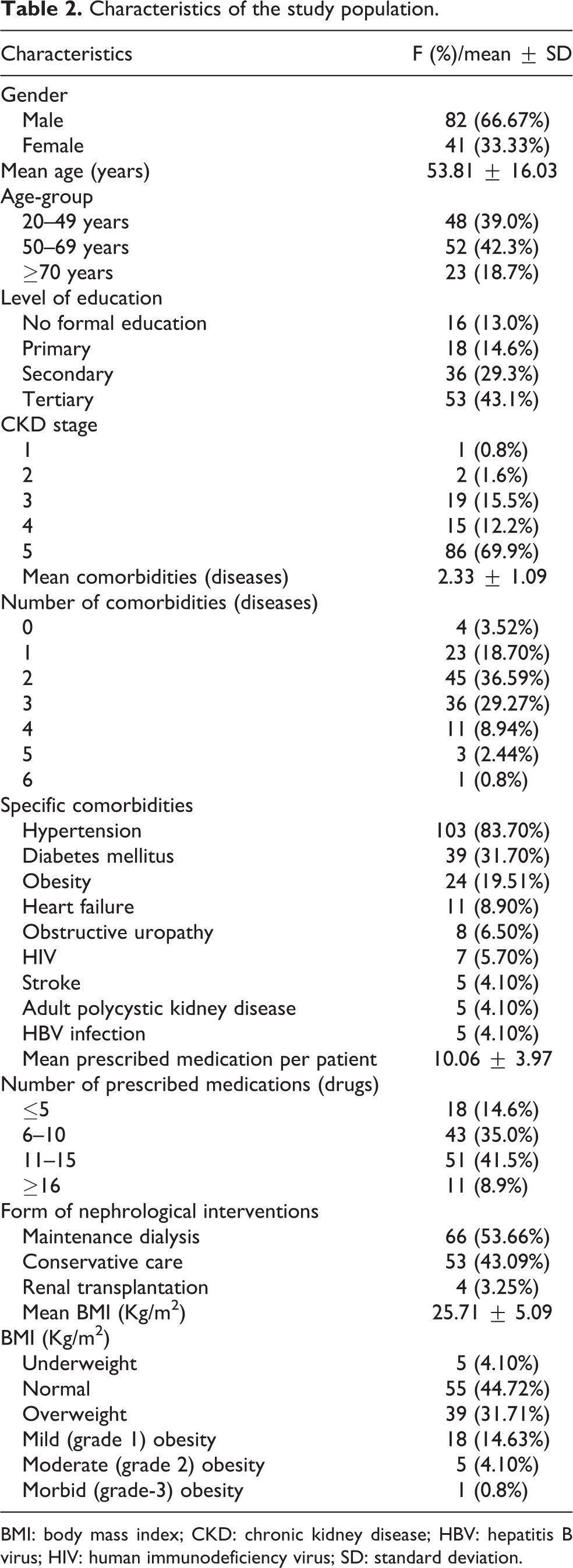
BMI: body mass index; CKD: chronic kidney disease; HBV: hepatitis B virus; HIV: human immunodeficiency virus; SD: standard deviation.
Fifty-three (43.1%) subjects had tertiary education, 36 (29.3%) subjects had secondary education, 18 (14.6%) subjects had primary education, and 16 (13.0%) subjects had no formal education. A total of 86 (69.9%) study subjects were in CKD stage 5, 15 (12.2%) subjects were in CKD stage 4, 19 (15.5%) subjects were in CKD stage 3, 2 (1.6%) subjects were in CKD stage 2, and the remaining 1 (0.8%) subject was in CKD stage 1 (see Table 2).
Regarding the form of nephrological interventions offered, the majority of the respondents, 66 (53.66%) subjects, were on maintenance dialysis, followed by 53 (43.09%) subjects on conservative care, while 4 (3.25%) subjects were on renal transplantation (see Table 2). Seventy patients were on renal replacement therapy. The prevalence of renal replacement therapy in this study was 57%.
In this study, the range for number of associated comorbidities was 0–6 diseases with a mean of 2.33 ± 1.09 diseases per patient. Most proportion of the respondents, 45 (36.59%), had two comorbidities, followed by 36 (29.27%) with three comorbidities, 23 (18.70%) had only one comorbidity, while 11 (8.94%) had four comorbidities (see Table 2). The most common specific comorbidities were hypertension in 103 (83.70%), diabetes mellitus in 39 (31.70%), obesity in 24 (19.51%), heart failure in 11 (8.9%), obstructive uropathy in 8 (6.5%), HIV in 7 (5.7%), and stroke in 5 (4.1%; see Table 2).
A total number of 1237 medications were prescribed for the study subjects, and the mean number of prescribed medications per patient was 10.06 ± 3.97. A total of 18 (14.6%) CKD subjects were on ≤5 medications, 43 (35.0%) subjects were on 6–10 medications, 51 (41.5%) subjects were on 11–15 medications, and 11 (8.9%) subjects were on ≥16 medications (see Table 2).
The prescribed medications frequently involved in extremely harmful, clinically nonbeneficial DDIs in this study were heparin in 67 (54.47%) subjects, lisinopril in 65 (52.9%) subjects, oral calcium carbonate (CaCO3) in 63 (51.2%) subjects, amlodipine in 56 (45.5%) subjects, oral ferrous sulfate (OFS) in 50 (40.7%) subjects, α-methyldopa in 32 (26%) subjects, and atorvastatin in 30 (24.4%) subjects (see Table 3). Prophylactic low molecular weight heparin (enoxaparin/clexane) was prescribed for these CKD patients with symptomatic ESRD who are undergoing maintenance hemodialysis (MHD) sections so as to prevent hemocoagulopathy disorder in them. In addition, these CKD patients with diabetic ketoacidosis/hyperosmolar hyperglycemic state (DKA/HHS), ischemic stroke, varicose veins, valvular heart diseases, ischemic heart disease, atrial fibrillation, or/and prolonged immobility may also be prescribed prophylactic enoxaparin/clexane in order to prevent the occurrence of deep vein thrombosis or cardiogenic thrombosis and subsequent thromboembolism phenomenon in them. The route of administration is subcutaneous usually at a dose of 40 mg (4000 IU) once daily prior to the commencement of hemodialysis section. Duration and frequency of administration among CKD patients are determined by the clotting profile (International Normalized Ratio [INR] value between the range of 0.8–1.3 is usually the desirable target) and also at the discretion of the nephrologist physician depending on the primary indication/purpose for usage such as for maintenance hemodialysis sections in symptomatic ESRD patients or for ESRD/pre-ESRD CKD patients with DKA/HHS, ischemic stroke, varicose veins, valvular heart diseases, ischemic heart disease, atrial fibrillation, or/and prolonged immobility.
List of the prescribed medications frequently involved in extremely harmful, clinically nonbeneficial DDIs among the study population.
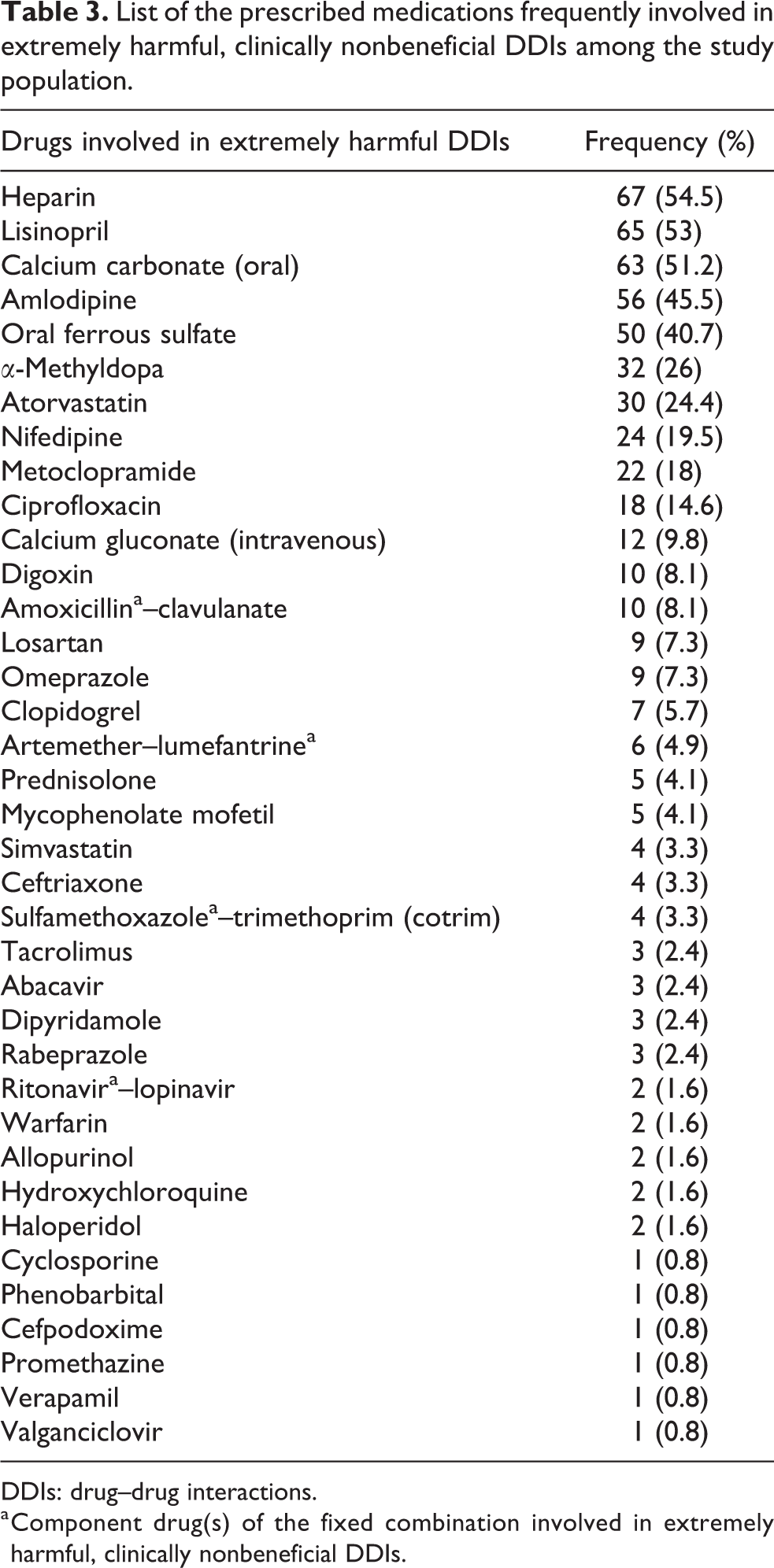
DDIs: drug–drug interactions.
a Component drug(s) of the fixed combination involved in extremely harmful, clinically nonbeneficial DDIs.
A total number of 1851 interactions were identified among 118 (95.9%) subjects, while 5 (4.1%) subjects had no known interaction. Twenty-nine (23.6%) subjects had between 6 and 10 interactions, 26 (21.1%) subjects had between 11 and 15 interactions, while 16 (13.0%) subjects had between 21 and 25 interactions; the rest are shown in Table 4.
Number of DDIs observed among the study population.a
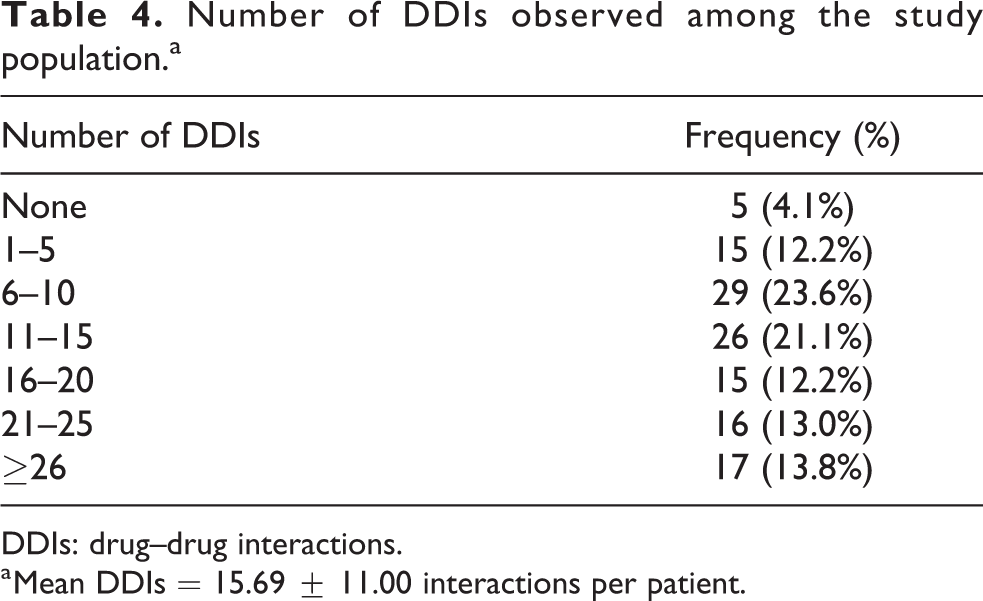
DDIs: drug–drug interactions.
a Mean DDIs = 15.69 ± 11.00 interactions per patient.
The mean for all DDIs observed per prescription in this study was 1.5, which means that for each additional medication prescription issued out to CKD patients in this study, there is additional chance of at least 1–2 DDIs occurring.
The mean for all extremely harmful, clinically nonbeneficial DDIs observed per patient belonging to these extremely harmful interaction categories in this study was 1.9; this revealed that there was likelihood of at least two extremely harmful DDIs in the prescription for each CKD patient belonging to these extremely harmful interaction categories in this study.
Also, the mean for all extremely harmful, clinically nonbeneficial DDIs observed per prescription in this study was 0.046 (equivalent to 4.6% or 46 extremely harmful interactions per a thousand prescriptions issued), which means that for every 1000 prescriptions issued out to CKD patients in this study, there was chance for the occurrence of at least 46 extremely harmful DDIs.
Furthermore, in this study, the prevalence of extremely harmful, clinically nonbeneficial DDIs (that is, type D or type X interaction categories only) was 24.4%, while the overall prevalence for all the observed DDIs was 95.9%.
The range for the number of DDIs among the 118 respondents was 1–85 interactions with a mean value of 15.69 ± 11.00 interactions per patient. A total of 639 (34.5%) DDIs were of mild severity (type B), 1155 (62.4%) DDIs were of moderate severity (type C), 45 (2.4%) DDIs were of major severity (type D), while 12 (1%) DDIs were of contraindicated/avoid drug combination (type X; see Table 5). Furthermore, a total number of 639 (34.5%) type B interactions with a mean value of 5.86 ± 4.35 number of interactions per patient were identified in 109 (88.6%) subjects. A total number of 1155 (62.4%) type C interactions with a mean value of 9.80 ± 7.37 number of interactions per patient were identified in 118 (95.9%) subjects. A total number of 45 (2.4%) type D interactions with a mean value of 1.50 ± 1.02 number of interactions per patient were identified in 30 (24.4%) subjects. Also, a total number of 12 (1%) type X interactions with a mean value of 1.20 ± 0.86 number of interactions per patient were identified in 10 (8.1%) subjects. While five (4.1%) subjects belonged to type A (no known drug interaction) category.
Level of clinical significance (severity) of DDIs among the population.

DDIs: drug–drug interactions.
A total of 960 (51.8%) DDIs were pharmacokinetic interactions (Pk), 890 (48.1%) DDIs were pharmacodynamics interactions (Pd), while the remaining 1 (0.1%) DDI was a pharmaceutical interaction (PC; see Table 6). Furthermore, a total number of 960 (51.8%) pharmacokinetic interactions with a mean value of 8.90±7.19 number of interactions per patient were identified in 108 (87.8%) subjects. A total number of 890 (48.1%) pharmacodynamics interactions with a mean value of 7.54±5.14 number of interactions per patient were identified in 118 (95.9%) subjects. While a total number of one (0.1%) pharmaceutical interaction with a mean value of 1.00±0.00 number of interaction per patient was identified in only one (0.81%) subject.
Mechanism of DDIs observed among the study population.
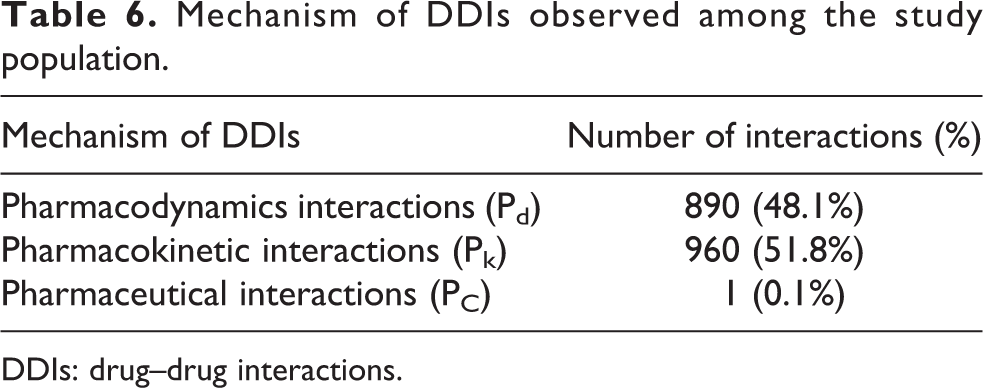
DDIs: drug–drug interactions.
The most frequent major severity interactions (type D) in this study occured between α-methyldopa and metoclopramide: eight (0.45%) interactions in eight (6.5%) patients; mycophenolate mofetil (MMF) and tacrolimus: three (0.2%) interactions in three (2.4%) patients; MMF and OFS: three (0.2%) interactions in three (2.4%) patients; while dipyridamole and heparin: three (0.2%) interactions in three (2.4%) patients; and the rest were shown in Table 7.
Pattern of extremely harmful, clinically nonbeneficial DDIs observed among the study population.
CTZ: chemoreceptor trigger zone; DDI: drug–drug interaction; MDR1: multidrug resistance transporter 1; MMF: mycophenolate mofetil; OFS: oral ferrous sulfate; PC: pharmaceutical DDIs; Pd: pharmacodynamics DDIs; Pk: pharmacokinetic DDIs.
a Total number of type D interactions = 45 interactions (2.4%) in 30 patients (24.4%). Total number of type X interactions = 12 interactions (1%) in 10 patients (8.1%). Total number of DDIs observed = 1851 interactions (100%) in 123 patients (100%).
The most frequent contraindicated/avoid drug combination interactions (type X) in this study occurred between α-methyldopa and metoclopramide: eight (0.45%) interactions in eight (6.5%) patients; α-methyldopa and haloperidol: two (0.1%) interactions in two (1.6%) patients; α-methyldopa and promethazine: one (0.1%) interaction in one (0.81%) patient; while IV calcium gluconate and IV ceftriaxone: one (0.1%) interaction in one (0.81%) patient (see Table 7).
The only one pharmaceutical DDI in this study occurred between IV calcium gluconate and IV ceftriaxone (specifically, type X; pharmaceutical interaction), which carries the potential risk of fatal particulate ceftriaxone–calcium complex precipitates being deposited in the lungs, heart, and kidneys. The administration interval between IV ceftriaxone and any IV calcium-containing solutions (such as Ringer’s solution, Hartman’s solution, and IV calcium gluconate) must be separated by at least 48 hours apart (see Table 7).
Regarding the patterns of extremely harmful, clinically nonbeneficial DDIs among the respondents, a total number of 30 different patterns of interactions were observed with the most frequent extremely harmful DDI patterns occurring between α-methyldopa and metoclopramide: 16 (0.9%) interactions in eight (6.5%) patients; MMF and tacrolimus: three (0.2%) interactions in three (2.4%) patients; MMF and OFS: three (0.2%) interactions in three (2.4%) patients; while dipyridamole and heparin: three (0.2%) interactions in three (2.4%) patients. The rest are shown in Table 7.
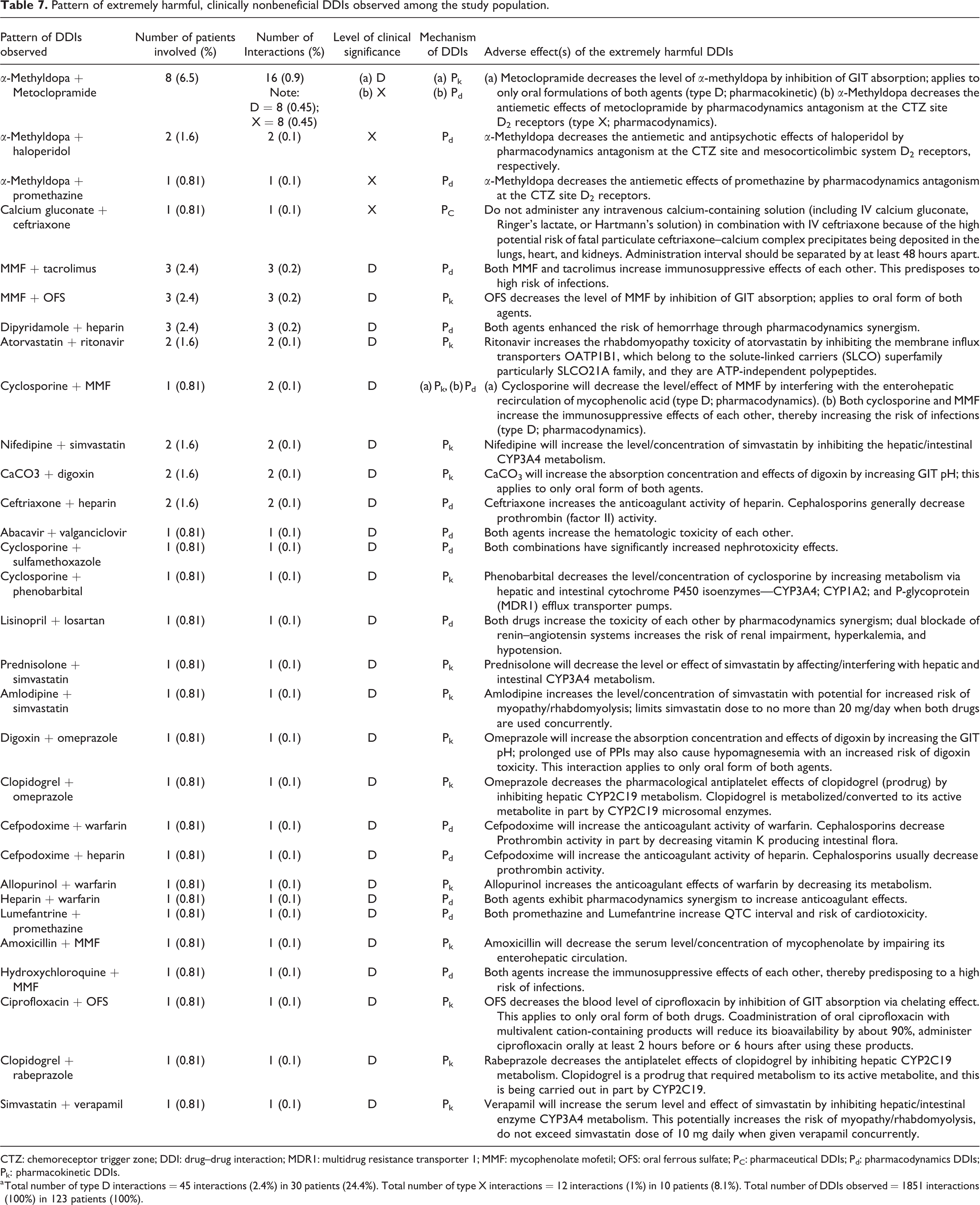
With respect to the Venn’s diagram in Figure 1 for the level of clinically significant DDIs observed among the patients, it was noticed that 10 (8.1%) subjects had all the categories of clinically significant interactions type B, type C, type D, and type X altogether. Twenty (16.3%) subjects had only type D interactions, 79 (64.2%) subjects had only type B interactions, while 9 (7.3%) subjects had only type C interactions. Five (4.1%) subjects belonged to type A (no known drug interaction) category, that is; neither had type B nor type C nor type D nor type X interactions. Furthermore, 30 (24.4%) subjects had either type D or type X interactions, 109 (88.6%) subjects had either type B or type D or type X interactions, 88 (71.5%) subjects had either type B or type C interactions, 99 (80.5%) subjects had either type B or type D interactions, 29 (23.6%) subjects had either type C or type D interactions, 108 (87.8%) subjects had either type B or type C or type D interactions while 118 (95.9%) subjects had either type B or type C or type D or type X interactions. It was also observed that the respondents with type X interactions were found to be a subset of the respondents with type D interactions, while those respondents with type D interactions were also a subset of the respondents with type B interactions. In addition, those respondents with type B interactions were also found to be a subset of the respondents with type C interactions. In summary, all the respondents with either type B or type D or type X interactions were all subsets of the respondents with type C interactions. What this implies is that an individual patient with the propensity to develop drug interactions can only be prone to type B, type D and type X interactions when he/she has first developed type C interaction. Likewise an individual patient can only develop type X interaction when he/she has first developed type C, type B and type D interactions.
Venn’s diagram for the level of clinically significant interactions among the study population.
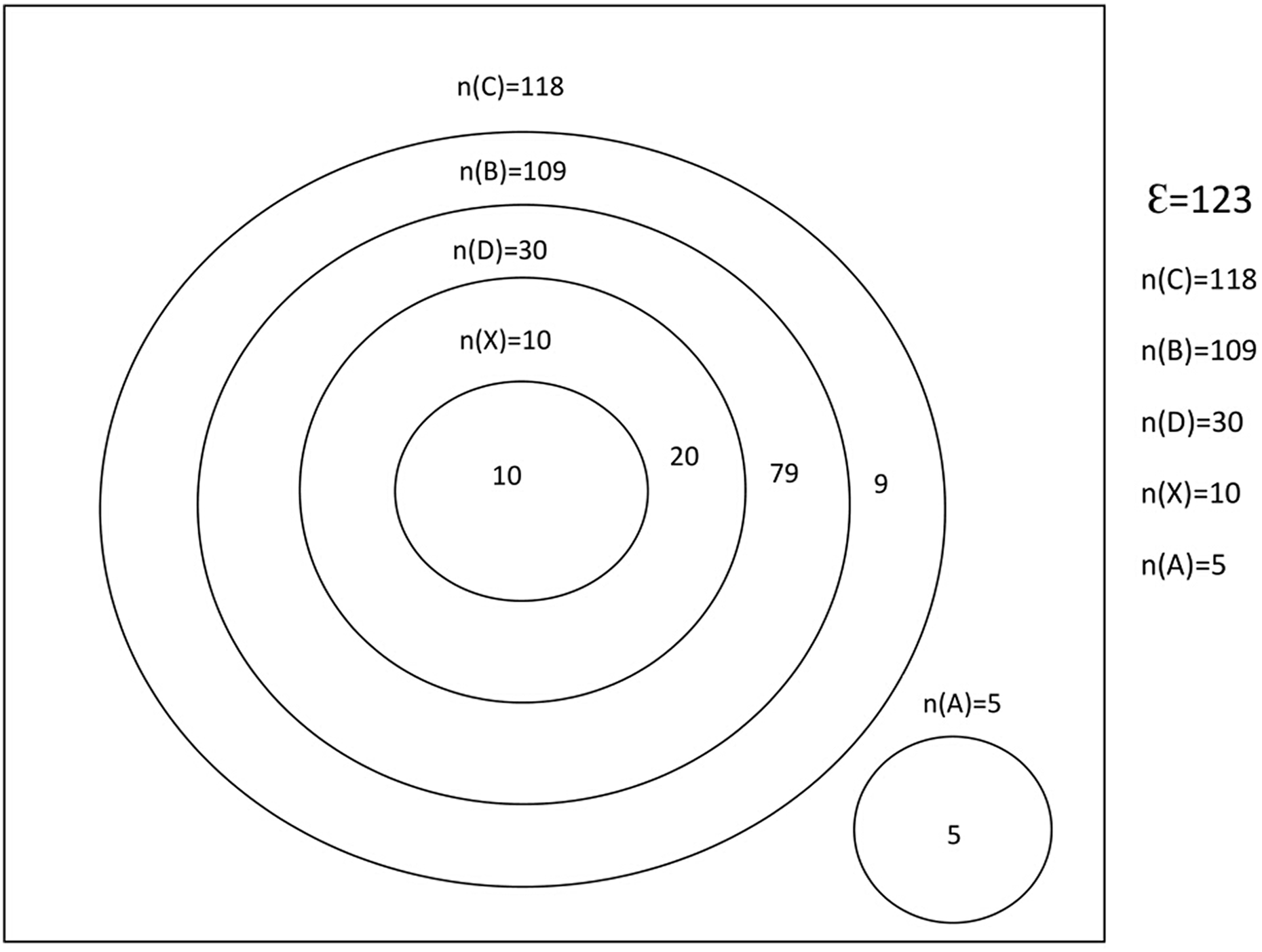
Also, with respect to the Venn’s diagram in Figure 2 for the mechanism of DDIs observed among the patients, it was noticed that only one (0.81%) subject had all the known mechanisms of pharmacodynamics, pharmacokinetic and pharmaceutical interactions altogether. One hundred and seven (87%) subjects had only pharmacokinetic interactions, while 10 (8.1%) subjects had only pharmacodynamics interactions. Five (4.1%) subjects had “no known mechanism of drug interaction”, that is; neither had pharmacodynamics nor pharmacokinetic nor pharmaceutical interactions. Furthermore, 117 (95.1%) subjects had either pharmacokinetic or pharmacodynamics interactions, 108 (87.8%) subjects had either pharmacokinetic or pharmaceutical interactions, while 118 (95.9%) subjects had either pharmacodynamics or pharmacokinetic or pharmaceutical interactions. It was also observed that the patient with pharmaceutical interaction was a subset of those with pharmacokinetic interactions, while the patients with pharmacokinetic interactions were also found to be a subset of those with pharmacodynamics interactions in this study. In summary, all the patients with either pharmacokinetic or pharmaceutical interactions were all subsets of the patients with pharmacodynamics interactions. What this implies is that an individual patient with the propensity to develop drug interactions can only be prone to pharmacokinetic and pharmaceutical interactions when he/she has first developed pharmacodynamics interaction. Likewise an individual patient can only develop pharmaceutical interaction when he/she has first developed both pharmacodynamics and pharmacokinetic interactions.
Venn’s diagram for the mechanism of interactions among the study population. PC: pharmaceutical interaction; Pd: pharmacodynamics interaction; Pk: pharmacokinetic interaction.
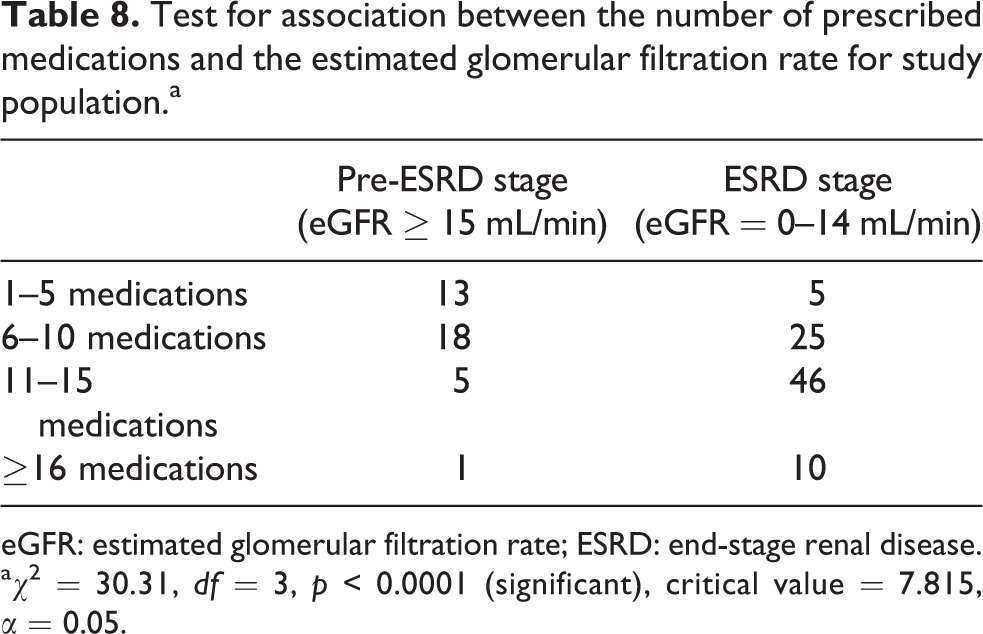
In this study, there was a statistically significant association between the number of prescribed medications and the eGFR (pre-ESRD and ESRD stages) for the respondents with a p value less than 0.0001. This implies that the number of prescribed medications increases as their eGFR decline in advance CKD stage patients (Table 8).
Test for association between the number of prescribed medications and the estimated glomerular filtration rate for study population.a
eGFR: estimated glomerular filtration rate; ESRD: end-stage renal disease.
a χ 2 = 30.31, df = 3, p < 0.0001 (significant), critical value = 7.815, α = 0.05.
Furthermore, there was a statistically significant association between the number of prescribed medications and the occurrence of DDIs among the respondents with a p value less than 0.0001. This implies that as the number of prescribed medications increases, the chance for occurrence of DDIs also increases among these CKD patients. About 77.8% of those CKD patients taking around 1–5 medications are at risk of developing DDIs, while 99.05% of those taking around 6 or more medications are at risk of developing DDIs (Table 9).
Test for the association between the number of prescribed medications and the occurrence of DDIs among the study population.a

DDIs: drug–drug interactions.
a χ 2 = 17·83, df =1, p < 0.0001 (significant), critical value = 3.841, α = 0.05.
In addition, there was a statistically significant association between the eGFR and the occurrence of DDIs among the study population with a p value less than 0.01. This implies that the chance for the occurrence of DDIs increases among these CKD patients as their eGFR decline in advance CKD stage due to the substantial increase in the number of prescribed medications being ingested (Table 10).
Test for the association between the eGFR and the occurrence of DDIs among the study population.a

CKD: chronic kidney disease; DDIs: drug–drug interactions; eGFR: estimated glomerular filtration rate.
a χ 2 = 6.755, df =1, p < 0.01 (significant), critical value = 3.841, α = 0.05.
Last, among these CKD patients recruited for this study, there was also a statistically significant association between those with diabetes mellitus and obesity with p value less than 0.0001. This implies that those CKD patients with obesity are highly predisposed and at risk of developing diabetes mellitus (Table 11).
Test for the association between diabetes mellitus status and obesity status for the study population.a

a χ 2 = 16.83, df =1, p < 0.0001 (significant), critical value = 3.841, α = 0.05.
Discussion
The most frequent extremely harmful, clinically nonbeneficial DDI in this study was between α-methyldopa and metoclopramide: 16 (0.9%) interactions in 8 (6.5%) patients. Furthermore, α-methyldopa decreases the antiemetic effects of metoclopramide by pharmacodynamics antagonism at the chemoreceptor trigger zone site D2-receptors (type X; pharmacodynamics). In addition, metoclopramide decreases the level of α-methyldopa by inhibition of GIT absorption, as this applies to only oral formulations of both agents (type D; pharmacokinetic). This was in contrast to the study done by Saleem et al. 21 where the most frequent extremely harmful, clinically nonbeneficial DDI reported was between IV calcium gluconate and IV ceftriaxone 21 (3.6%) interactions (specifically, contraindicated type X; pharmaceutical interactions) in 21 (10.01%) CKD patients, which carries the potential risk of fatal particulate ceftriaxone–calcium complex precipitates being deposited in the lungs, heart, and kidneys. The administration interval between IV ceftriaxone and any IV calcium-containing solutions (such as Ringer’s solution, Hartman’s solution, and IV calcium gluconate) must be separated by at least 48 hours apart.
The prescribed medications frequently involved in extremely harmful, clinically nonbeneficial DDIs in this study were heparin, lisinopril, oral CaCO3, amlodipine, and oral OFS. This agreed with previous studies where oral CaCO3 (Sgnaolin et al. 18 ) and lisinopril (Chinwendu et al. 30 ) were also reported as among the most frequently prescribed medications for CKD patients. 18,30
In this study, the mean prescribed medications per patient was 10.06 ± 3.97. This is slightly higher than 7.87 ± 2.44 reported by Al-Ramahi et al. 20 However, Rama et al. 19 reported a higher mean prescribed drugs per patient of 12.08 ± 6.30 compared to the finding in this present study. Our study population involved both nondialyzed and dialyzed patients unlike the study by Al-Ramahi et al. that involved only CKD patients on MHD. Patients who are on regular MHD may require less number of drugs for blood pressure control; hence, this may possibly explain the lower mean number of prescribed medications. The practice of polypharmacy in the management of CKD is however not surprising because they have a high number of cardiovascular risk factors, comorbidities, and complications such as hypertension, diabetes mellitus, heart failure, arrhythmias, anemia, hyperuricemia, and calcium–phosphate abnormalities, which are managed by a combination of drugs. The most common comorbidities in this study were hypertension and diabetes, which agreed with previous studies done. 7,22,23 This can be attributed to the fact that both conditions are the leading etiologies of CKD in Nigeria, Sub-Saharan West Africa region, and worldwide. The overall prevalence of DDIs among the studied CKD patients was 95.9% with about 62.4% showing moderate (type C) clinical significance.
The mean for all DDIs observed per prescription in this study was 1.5, which was similar to the report by Sgnaolin et al. 18 (1.0) but lower than the number reported by Rama et al. 19 (2.7) and Marquito et al. 22 (2.5). This showed that there was likelihood of at least one to two DDIs occurring for each additional medication prescription issued out to CKD patients in this study.
Furthermore, the mean for all extremely harmful, clinically nonbeneficial DDIs observed per patient belonging to these extremely harmful interaction categories in this study was 1.9; this revealed that there was likelihood of at least two extremely harmful DDIs in the prescription for each CKD patient belonging to these extremely harmful interaction categories in this study. Also, the mean for all extremely harmful, clinically nonbeneficial DDIs observed per prescription in this study was 0.046 (equivalent to 4.6% or 46 extremely harmful interactions per a 1000 prescriptions issued), which means that for every 1000 prescriptions issued out to CKD patients in this study, there was chance for the occurrence of at least 46 extremely harmful DDIs compared to the standard reference value of less than 0.00001 (equivalent to less than 0.001% or less than one extremely harmful interaction per a 100,000 prescriptions issued). This finding however leaves room for more important and nonnegotiable improvement in our rational prescriptions for CKD patients. This therefore emphasize the need for clinical pharmacologists and clinical pharmacists to regularly evaluate the prescriptions of CKD patients for extremely harmful, clinically nonbeneficial DDIs. The prevalence of extremely harmful, clinically nonbeneficial DDIs (i.e. type D or type X interaction categories only) in this study was 24.4% compared to the standard reference value of near or approximately 0% (i.e. value less than 0.001%) being required for efficient, accurate, and rational medical prescriptions in clinical practice. While the overall prevalence for all the DDIs observed in this study was 95.9% which was higher than the range of 56.9-89.1% reported in some previous studies. 19 –23 Furthermore, the estimated overall prevalence for DDIs in this study was higher than the value of 56.9% reported by Sgnaolin et al. 18 Differences in methodology, average number of medications per prescription, number of specific associated comorbidities or complications per patient, and CKD stages of the studied population could account for the variations seen in the prevalence rates for overall DDIs in the different studies. For example, in the study by Sgnaolin et al. 18 where a lower prevalence of overall DDIs was reported compared to this study, the average number of medications per prescription was 6.3 ± 3.1, but unlike this present study where the average number of medications per prescription was 10.06 ± 3.97. In fact, the number of medications per prescription and number of specific associated comorbidities or complications per patient were the major determinants of the prevalence rate for extremely harmful DDIs in this study.
Regarding the level of clinically significant interactions in this study, majority of the DDIs (62.4%) were of moderate severity (type C) and this was similar to previous reports. 18,19,22,23 In this study, the chance for the occurrence of type D (major/serious) interactions was 2.4%, which were found in 30 patients. While the chance for the occurrence of type X interactions (contraindication/avoid drug combination) was 1%, which were found in 10 patients; this is comparable to the value of 0.4% type X interactions reported by Marquito et al. 22 This finding is however at variance to that of Saleem et al. 21 who reported that 13.4% of DDIs observed in their study as contraindication/avoid drug combination.
A slightly higher proportion 960 (51.8%) interactions of the total DDIs was found to be from pharmacokinetic interactions. This is different from the reports in other studies where pharmacodynamics interactions were found to be predominant. 19,23 Also, most of the total DDIs were of delayed onset, which was similar to the report by Rama et al. 19 The clinical significance of this is that the patients may not manifest the effects of DDIs early, hence the need for long-term follow-up of these patients.
There was a statistically significant association between the number of prescribed medications and the eGFR (pre-ESRD and ESRD stages) for the respondents in this study with a p value less than 0.0001. This implies that the number of prescribed medications increases as their eGFR decline in advance CKD stage patients. This finding also agreed with previous studies done by Sgnaolin et al., 18 Rama et al., 19 and Marquito et al. 22
Furthermore, there was a statistically significant association between the number of prescribed medications and the occurrence of DDIs among the respondents with a p value less than 0.0001. This implies that as the number of prescribed medications increases, the chance for the occurrence of DDIs also increases among these CKD patients. About 77.8% of those CKD patients taking around 1–5 medications are at risk of developing DDIs (compared to the value of 50% in previous reference literatures), while 99.05% of those taking around 6 or more medications are at risk of developing DDIs (compared to the value of 100% in previous reference literatures). 18,19,22,23
In addition, there was a statistically significant association between the eGFR and the occurrence of DDIs among the study population with a p value less than 0.01. This implies that the chance for the occurrence of DDIs increases among these CKD patients as their eGFR decline in advance CKD stage due to the substantial increase in the number of prescribed medications being ingested. This finding also agreed with previous studies done by Sgnaolin et al., 18 Rama et al., 19 and Marquito et al. 22
Last, among these CKD patients recruited for this study, there was also a statistically significant association between those with diabetes mellitus and obesity with a p value less than 0.0001. This implies that those CKD patients with obesity are highly predisposed and at risk of developing diabetes mellitus.
The limitation of this study was that the extremely harmful, clinically nonbeneficial DDIs detected in this study were theoretically discovered using Medscape drug interaction checker. Also, the Medscape drug interaction checker used in this study did not take into consideration the prescribed dose, frequency of administration, route of administration, and duration of medication use.
Conclusion
In this study, the occurrence and burden of extremely harmful, clinically nonbeneficial DDIs is significantly high among these CKD patients. Nephrologists, clinical pharmacologists and clinical pharmacists should utilize available interaction detecting softwares in order to identify and avoid extremely harmful DDIs among CKD patients. There is also a critical need to minimize the number of prescribed medications for these patients in order to prevent the occurrence and burden of extremely harmful DDIs. This approach will save lives and avoid the scourge of associated morbidity, mortality, enormous healthcare cost, increase frequency and length (duration) of hospitalization. However, this study has also brought to limelight the magnitude of extremely harmful, clinically nonbeneficial DDIs among CKD patients in renal practice. In addition, there is a proper need to take proactive steps to reduce these additional burdens and optimize the care of our renal patients.
Recommendations
Nephrologists, clinical pharmacologists and clinical pharmacists should make use of available interaction detecting softwares to check all prescribed medications for CKD patients so as to identify and avoid the presence of extremely harmful, clinically nonbeneficial DDIs.
A cordial integrated relationship between healthcare professionals (Nephrologists, clinical pharmacologists, clinical pharmacists and nephrology nurses) should be encouraged in order to optimize CKD patients’ care and to reduce the occurrence and burden of extremely harmful DDIs in them.
Whenever the clinical pharmacists that are involved in the rational dispensing of CKD patients medications detect the presence of any harmful DDIs in their prescription, he/she should alert the doctors (Nephrologists and clinical pharmacologists) involved and inform them of the impending problem so that feasible alternative prescriptions can be made and the error corrected as this will involve more demanding effort in the area of clinical pharmacist and doctor’s commitment time but the hospital should make this mandatory. As this approach will save lives, reduce the morbidity, mortality, frequency of hospitalisation, length of hospital stay and healthcare costs associated with the burden and occurrence of extremely harmful DDIs in clinical practice.
Regular organisation of health education awareness program on the prevention of CKD and its associated complications among the general public should be done by healthcare professionals (Nephrologists, clinical pharmacologists, clinical pharmacists and nephrology nurses) coupled with adequate support from the government.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
