Abstract
Keywords
Introduction
Capsaicin (8-methyl-N-vanillyl-6-nonemide) is a good candidate for a functional feed additive as it has been found to both increase broiler body weight (BW) and reduce gut Salmonella enteritidis, Escherichia coli and Clostridium perfringens in broiler chickens.1–8 Capsaicin is the alkaloid giving chili its pungency. It is part of the fruit defense chemistry in members of the genus Capsicum (peppers) and ensures seed dispersal by birds by deterring mammalian fruit predators.9–11 Capsaicin accounts for c. 48% of the active substances in capsaicinoids,1,7 and the deterrent effect in mammals is caused by the mammalian vanilloid receptor TRPV1, which is able to detect capsaicin-like inflammatory substances.12,13 However, high-purity capsaicin has not been available at the cost and volumes needed for the poultry industry due to the limited production of red peppers and challenges related to capsaicin purification. These challenges were solved by the development of the commercially available synthetic capsaicin analogue phenylcapsaicin (PheCap; 7-phenylhept-6-yne-acid-hydroxy-3-mathoxylbenzylamide, CAS no 848127-67-3) (aXichem AB, Malmö, Sweden), Figure 1. Chemical structure of capsaicin and phenylcapsaicin.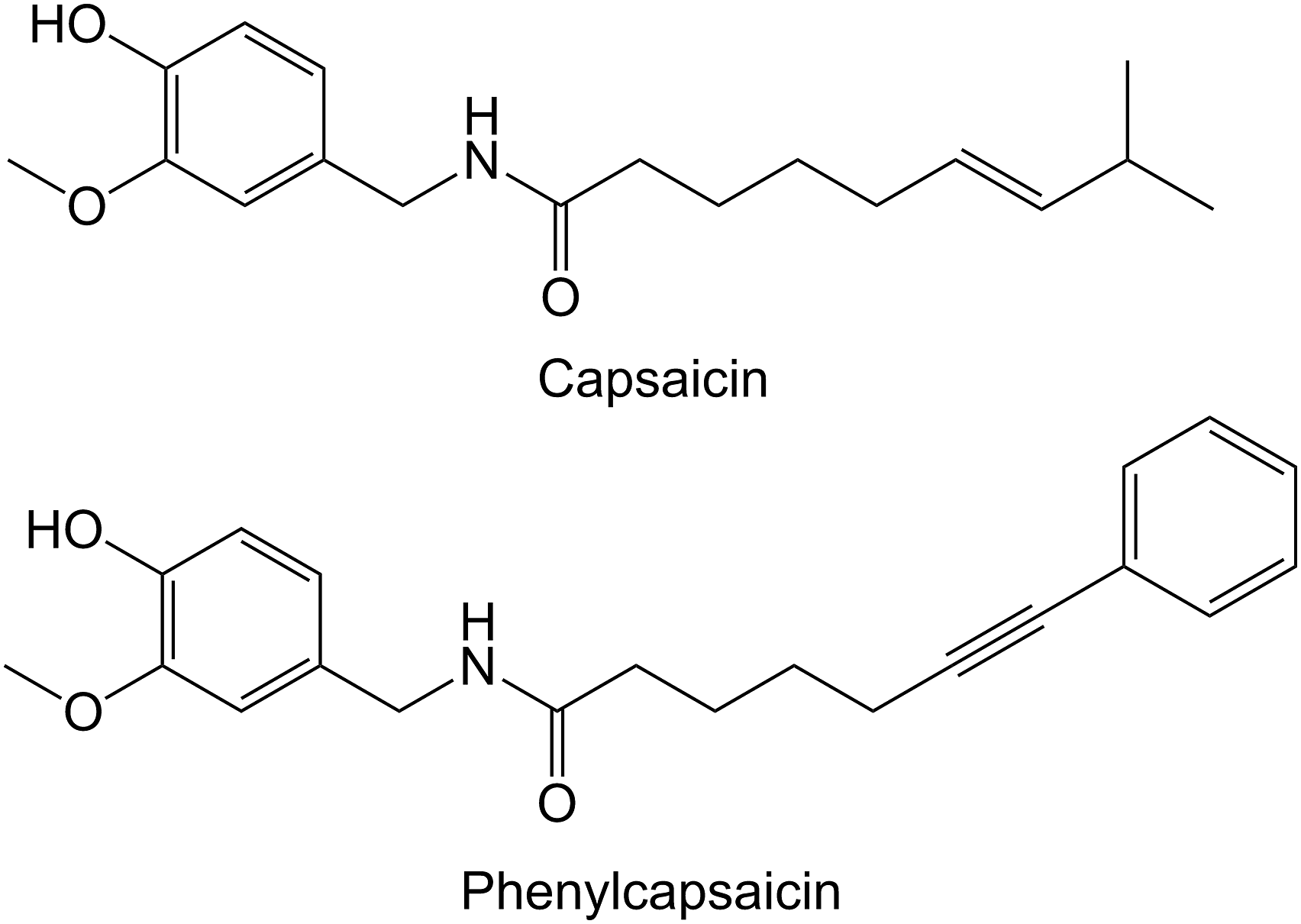
Few studies on the performance and toxicological effects of high-purity capsaicin on poultry are available in the literature 14 as most studies examined the effect of capsaicinoid powder or capsicum oleoresin. The results from these studies might not be directly transferable to the effects of high-purity capsaicin and PheCap in poultry because of effects related to differences in active substances between pepper breeding lines, purity profiles, harvest time and drying methods. 15 For a comprehensive summary of the effect of hot red pepper on carcass traits, organ weights, blood parameters, antimicrobial and intestinal histomorphology, see Abd El-Hack ME et al. 1
Nevertheless, the results from the few studies available seem to have reproduced results from studies using hot red peppers. In Arbor Acres broilers, using a purified natural capsaicin extract (2% capsaicin and 98% stearic acid as diluent), final feed concentrations of 2 and 4 mg capsaicin/kg feed were found to improve the feed to body weight ratio, breast meat quality, and small intestine, liver and immune organ development, and digestive enzyme activities. These effects were not found for 6 mg capsaicin/kg feed. 14 A concentration of 80 mg/kg feed improved meat quality, nutrient digestibility, growth performance as well as antioxidant status and immune function. 16 In female Longyan laying ducks, at a concentration of 150 mg/kg feed, egg production increased due to improved follicular growth and maturation, and higher antioxidant capacity was found. 17
To the best of our knowledge, only a single study is available in the literature on the toxicity of high-purity PheCap. In Wistar rats, degenerative, but reversible changes in the liver at 250 mg/kg BW/day, and local irritating effects in the stomach at 100 and 250 mg PheCap/kg BW/day, were found in a 90-days repeated dose oral gavage study with a 28-days recovery period. In the same study, high-purity PheCap was non-mutagenic at concentrations up to 5000 μg in the tester stains Salmonella typhimurium TA 98, TA 100, TA 102, TA 1535, or TA 1537
Here we present the first safety assessment of high-purity PheCap at doses of 10, 15, and 150 mg/kg feed in broiler chicken diets in a controlled laboratory experiment.
Materials and methods
The present study was carried out to provide an evaluation of the tolerance of PheCap (aXiphen-feed®, aXichem AB, Malmö, Sweden) over a period of 35 days in commercial male broilers. The broilers received a diet with inclusion of either 10, 15, or 150 mg PheCap/kg feed to be compared to a diet without PheCap (control). The 10 and 15 mg PheCap/kg feed concentrations were chosen based on findings from a full-scale farm pilot where 10 and 15 mg PheCap/kg feed showed significant reduction in floor Salmonella. In addition, 150 mg PheCap/kg feed was included as a tolerance level. A completely randomized block design comprising four treatments (0, 10, 15, 150 mg PheCap/kg feed) in two rooms was used. Within each room there were 16 pens (floor space: 2.15 m2) in four blocks where each block consisted of 4 adjacent pens. Each treatment was replicated eight times and the experimental unit was a pen with 20 male broilers.
Study compliance
The experiment was conducted by Wageningen Livestock Research (ISO 9001:2015 certified) at the Research Facility Carus (Wageningen, The Netherlands) according to Animal and Human Welfare Codes and laboratory practice codes relevant in The Netherlands. The experimental protocol was approved by the Animal Welfare Body of Wageningen University (IvD-WU), Wageningen, The Netherlands (2019.D- 0033.001). The study was carried out according to the EFSA guidelines for the assessment of the safety of feed additives for the target species. 19 Permission number 2020428 was granted by the Ministry of Agriculture, Nature and Food Quality, The Netherlands, prior to the study for the preparation, availability or stocking, delivery, transport and feeding of test feed and the product Phenylcapsaicin incorporated therein in accordance with Regulation EC 1831/2003.
Test substance
A two-phase starter and grower diet program were provided from 0-14 and 14-35 days of age, respectively. The experimental diets were formulated by Wageningen Livestock Research and produced by Research Diet Services (Wijk bij Duurstede, The Netherlands). The diets were formulated to meet or exceed the CVB recommendation for Ross 308 broiler chickens 20 and were prepared without inclusion of anticoccidial.
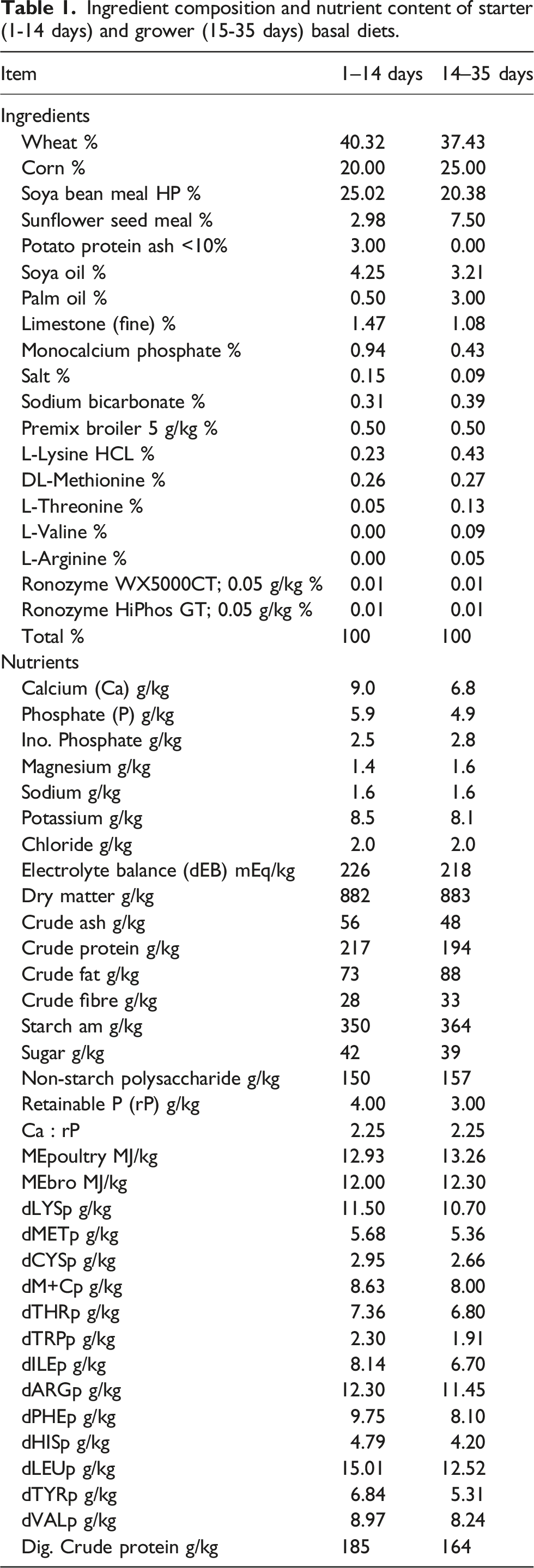
Ingredient composition and nutrient content of starter (1-14 days) and grower (15-35 days) basal diets.
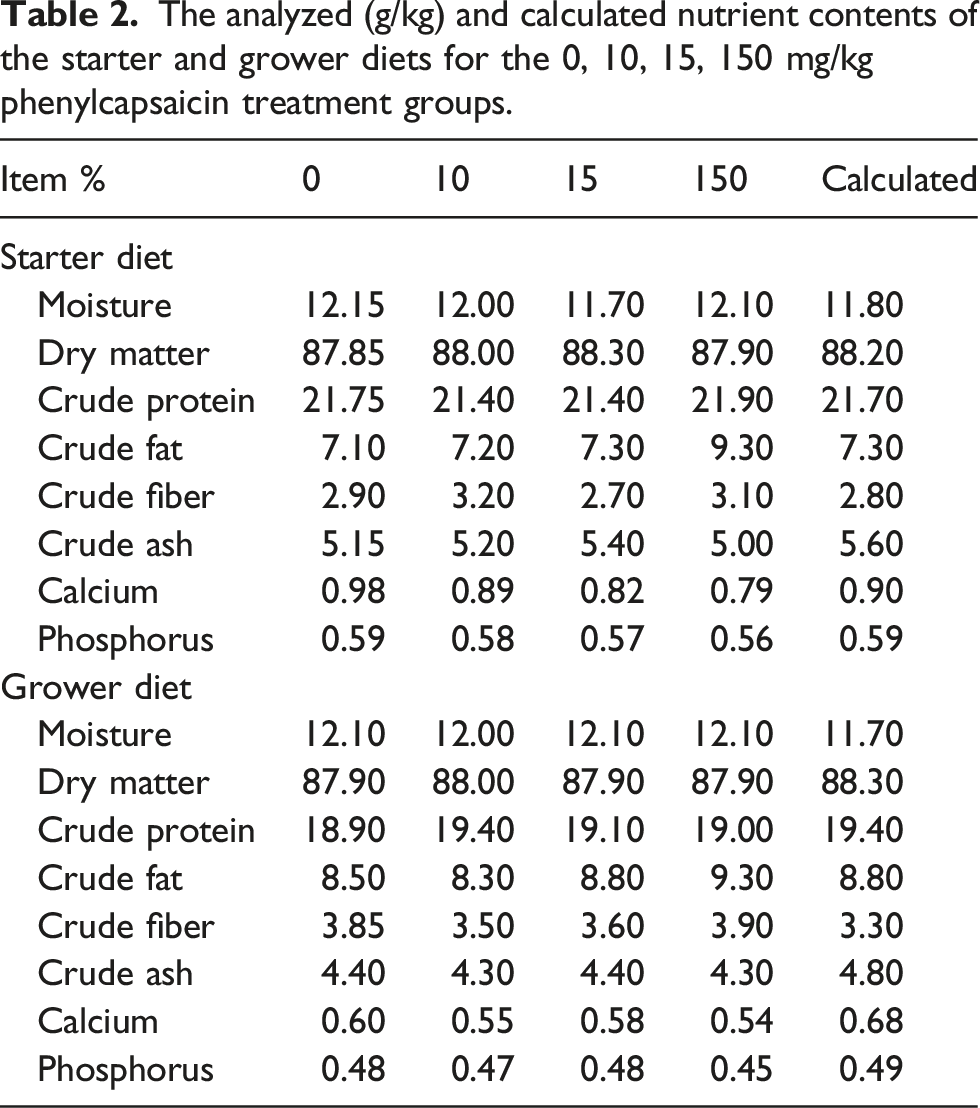
The analyzed (g/kg) and calculated nutrient contents of the starter and grower diets for the 0, 10, 15, 150 mg/kg phenylcapsaicin treatment groups.
Oral toxicity
A total of 640 1-day-old male Ross 308 broilers were purchased from a commercial hatchery (Kuikenbroederij Morren B.V., Lunteren, The Netherlands) and allocated to the 32 floor pens according to a weight class system where the mean body weight of the 20 birds per pen had to be within 3% of the mean body weight of all birds. The pens were bedded with wood shavings (±3 kg/m2) and provided with a perch, a feeding pan (Valenta, Ø 335 mm, VDL Agrotech BV, AW Eindhoven, The Netherlands) and six nipple drinkers with drip cups (Impex Barneveld BV, MA Barneveld, The Netherlands). Feed and water were provided ad libitum. The temperature at placement of the broilers was 33°C and the temperature was gradually decreased to 20°C at 28 days of age and retained until the end of the experiment. During the first 3 days, light was continuous (24L:0D) and thereafter a day/night schedule of 18 h light and 6 h dark (18L:6D) per 24 h was given. Light intensity was 20 lux during the entire experimental period.
Day-old broilers were vaccinated against Infectious Bronchitis (IB) in the hatchery (IB primer, Zoetis B.V., Capelle a/d IJssel, The Netherlands) and against coccidiosis (Paracox 5, spray, MSD Animal Health, Boxmeer, The Netherlands) at the trial facility. At 14 days of age all broilers were vaccinated against New Castle Disease (NCD, Clone 30, spray vaccination, MSD Animal Health, Boxmeer, The Netherlands).
Body weight and residual feed were recorded per pen at 0, 7, 14, 21, 28, and 35 days of age to determine average BW, BW gain, and feed consumption on weekly basis, per feeding phase (0-14 days and 14-35 days) and cumulative over the entire experimental period. Culling, mortality, and weight of the removed broilers were recorded daily.
The feed conversion ratio (FCR) was calculated as gross feed intake/total gross BW gain for a given time period and the European Production Efficiency Factor (EPEF) was calculated as (mean daily body weight gain (g)/FCR x10) x (100 - mortality (%)). Daily growth rate (%) was calculated using the compounded growth rate formula
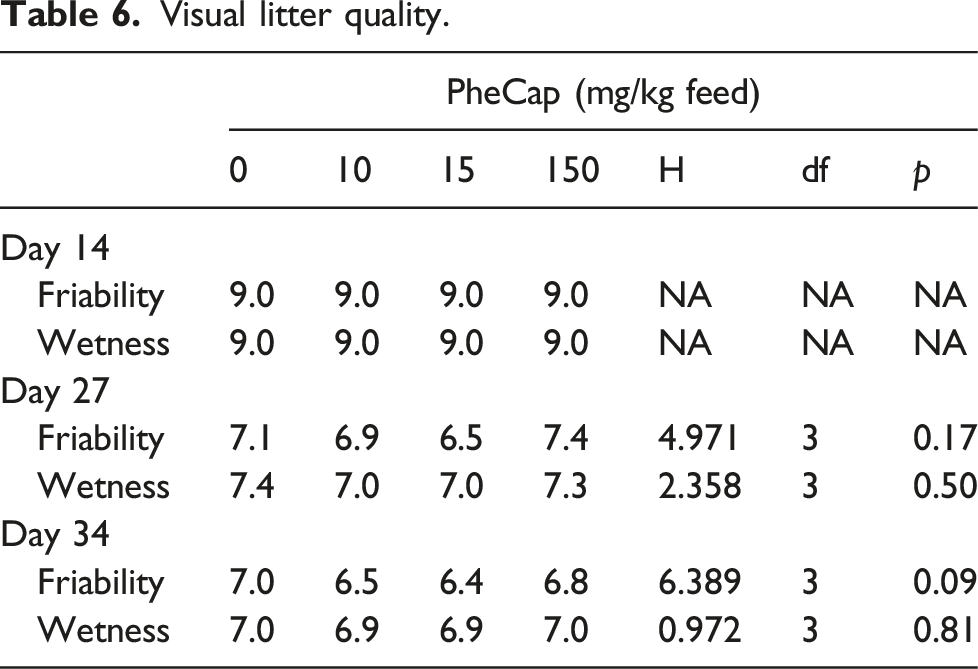
Litter quality was observed on day 14, 27, and 34 by an experienced assessor and friability and wetness of the litter in each pen was scored on a 1-to-10-point scale. Score 1 is complete caked litter: wet litter, total area by pressure on the litter water is appearing; Score 10 is friable litter, no caked litter particles: very dry litter (only observed at start of the experiment).
The occurrence of footpad lesions and their severity were determined at day 34 by an experienced assessor. All broilers per pen were assessed. Footpad dermatitis was scored for both feet according to the ‘Swedish’ classification, 21 i.e. score 0: no lesions or very small discoloration; score 1: discoloration but no deep lesion; score 2: deep lesion with ulcers or scabs, bumble foot.
The severity of footpad lesions was expressed as footpad score (FPS) per pen calculated as 100% x [(0.5 x the total number of birds with score 1) + (2 x the total number of birds with score 2)]/the total number of scored birds. The flock FPS ranges from 0 (all birds having no lesions) to 200 (all birds having score 2).
On day 35, two broilers with average weight were removed per pen, weighed, individually marked with wing tags and transported to Royal GD (Deventer, The Netherlands) for haematology, blood chemistry analysis, pathology and histology of different intestinal tissues. At Royal GD, the broilers were euthanized one by one by electrocution and 1 × 5 mL NaF blood, 1 × 5 mL EDTA blood and 1 × 5 mL serum blood were immediately drawn from the jugular vein. The blood samples were analyzed on packed cell volume (haematocrit), haemoglobin, mean corpuscular haemoglobin concentration, and total and differential counts for leukocytes. The blood plasma/serum samples were analyzed on sodium, potassium, chloride, calcium, phosphate, magnesium, total protein, albumin/globulin ratio, glucose, urea, cholesterol, creatinine, triglycerides, glutamate dehydrogenase (GLDH), haemolyse-index, bilirubin, alanine aminotransferase (ALT), aspartate aminotransferase (AST), lactate dehydrogenase (LDH), gamma-glutamyltransferase (GGT), alkaline phosphatase (ALP) and creatine kinase (CPK) concentration. Gross pathology of the liver, kidneys, spleen, lung, stomach, small intestine, colon, cecum, heart, pancreas, adrenal gland, thymus and thyroid gland was determined in the same broilers from which blood samples were taken.
Statistical analysis
All statistics were calculated using R, version 4.2.1. 22 For the response variables growth performance, feed intake, feed conversion ratio and EPEF, we used linear mixed effects models (LME) to account for the clustering of observations in the four blocks within each of two rooms. Each block consisted of four adjacent pens. The mean of 20 broilers within each pen was used in the analysis, making the pen the unit of observation. The R syntax for these models was: lme(Response ∼ PheCap, random = ∼+1|Room/Block) where ‘lme’ is a function from the nlme library of R,23, 24 ‘Response’ represents the response variable analyzed, and ‘PheCap’ was the predictor, treated as a categorical variable with the four levels; 0, 10, 15 and 150 mg PheCap/kg feed. The statement random = ∼+1|Room/Block sets the intercept of each room to be a random effect factor as well as each block nested under a room.
We used the same modelling for the blood chemistry and haematology datasets but with the nesting structure Room/Block/Pen for the random effect factors. We did this since two broilers per pen were examined separately, making a broiler the unit of observation. Due to the binary nature of the response variables of the gross pathology data, they were analyzed using generalized linear mixed-effects models using the glmmTMB library of R. 25 Room, block, and pen were included as random effect factors since two broilers per pen were examined individually. The R syntax for these models was: glmmTMB(Response ∼ PheCap+(1|Room)+(1|Block)+(1|Pen), family = binomial)
Mortality was analyzed using the survival library of R.26,27 We used a parametric survival model with a Weibull distribution and censoring. The R syntax for this model was: survreg(Surv(Age,Status) ∼ PheCap, cluster = RoomPen, dist = ’weibull’) where ‘survrg’ is the function for performing parametric survival modelling, ‘Surv’ is a function used to create the survival object, i.e., the response variable. ‘Age’ represents the age at death (in days) except when the status indicator ‘Status’ is zero
For all the above models, we first analyzed for an overall effect of PheCap using the ANOVA function of R. When an overall effect was found, treatment contrasts from the summary output of R was used to compare the different concentrations of PheCap against the 0 mg PheCap/kg feed group.
The Kruskal-Wallis test was used to analyze for differences in litter moisture and friability due to the non-normality of these data, and, due to low variance resulting from low occurrences, the Fisher’s exact test was used to analyze for the effect of dietary PheCap on footpad lesions.
Results
Growth performance results for days 1-14, 14-34 and 1-34.
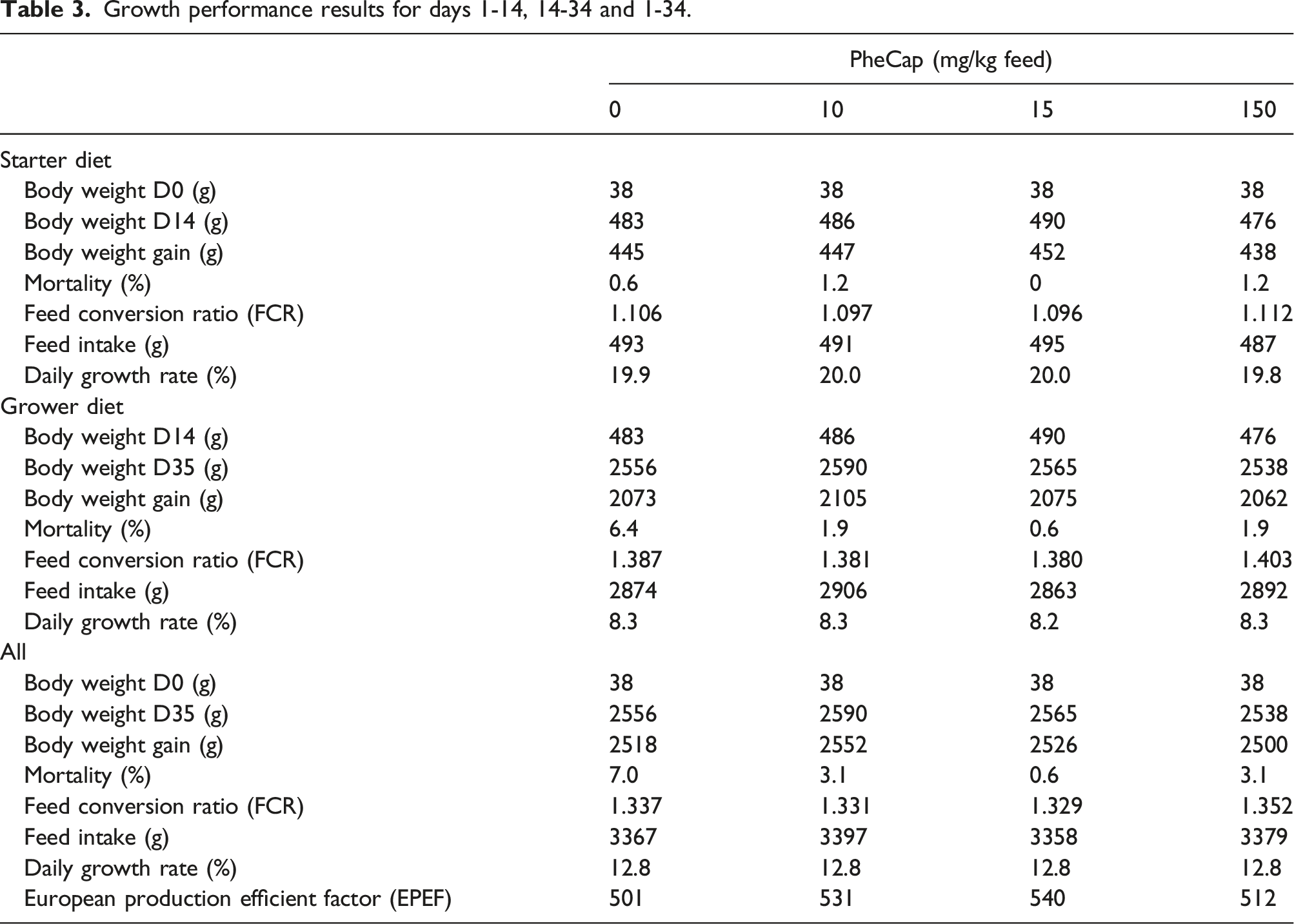
No differences in feed intake were observed between the experimental treatment groups in either the starter phase (df = (3, 21), F = 0.293, p = 0.83), the grower phase (df = (3, 21), F = 1.413, p = 0.27) or overall (df = (3, 21), F = 1.730, p = 0.19), Table 3.
No effect on feed conversion ratio (FCR) was found for either the starter diet (df = (3, 21), F = 0.380, p = 0.77), grower diet (df = (3, 21), F = 0.181, p = 0.91) or overall (df = (3, 21), F = 0.533, p = 0.66), Table 3.
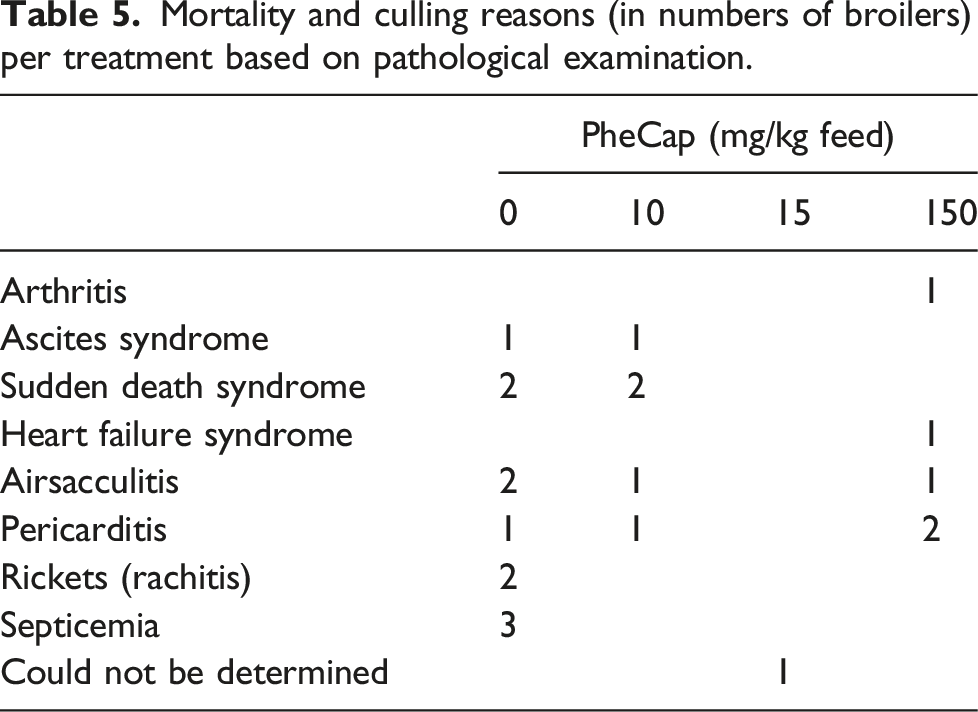
A total of 22 birds died or were euthanized over the course of this study, with statistically significant lower mortality for all three treatment diets compared to the control. Half of the dead broilers (11, 6,9%) were in the control group while 5 (3.1%), 1 (0.6%) and 5 (3.1%) broilers were in the 10 (SE = 0.251, z = 2.18, p = 0.03), 15 (SE = 0.708, z = 2.33, p = 0.02) and 150 (SE = 0.251, z = 2.18, p = 0.03) mg/kg PheCap groups, respectively. In the control group, 10 of 11 deaths occurred during the growth period, Table 3.
This reduced mortality due to dietary PheCap resulted in a statistically significant increase in the EPEF (df = (3, 21), F = 4.652, p = 0.01), and a statistically significantly increase was found in both the 10 mg/kg feed (df = 21, t = 2.579, p = 0.02) and the 15 mg/kg feed groups (df = 21, t = 3.363, p = 0.003), but not for the 150 mg/kg feed group (df = 21, t = 0.958, p = 0.35) compared to the control group, Table 3.
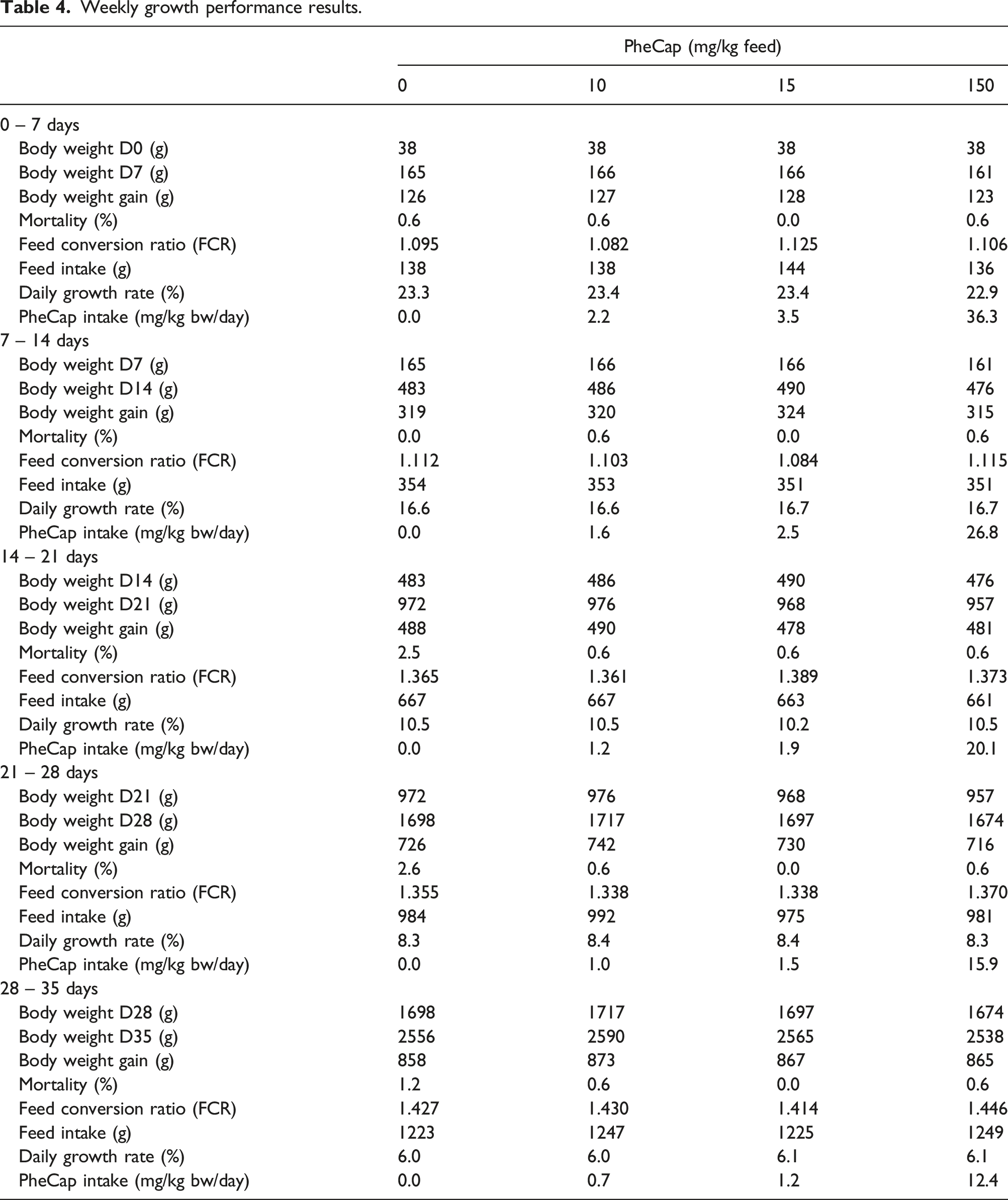
Weekly growth performance results.
Mortality and culling reasons (in numbers of broilers) per treatment based on pathological examination.
Visual litter quality.
The severity of footpad lesions was very low under the present conditions with 2, 3, 1 and 0 observations for the 0, 10, 15, and 150 mg/kg treatment groups, respectively. No differences were observed in footpad lesions between treatment groups (p = 0.33).
Twenty two of the 64 blood samples were not suitable for analysis of the red and white blood cell parameters. Some blood samples clotted immediately but most unsuitable blood samples were visually not clotted, and it is not clear why these samples could not be analyzed. This reduced the sample sizes from 16 per treatment to 10, 11, 11, and 10 for the 0, 10, 15, and 150 mg/kg PheCap treatment groups, respectively.
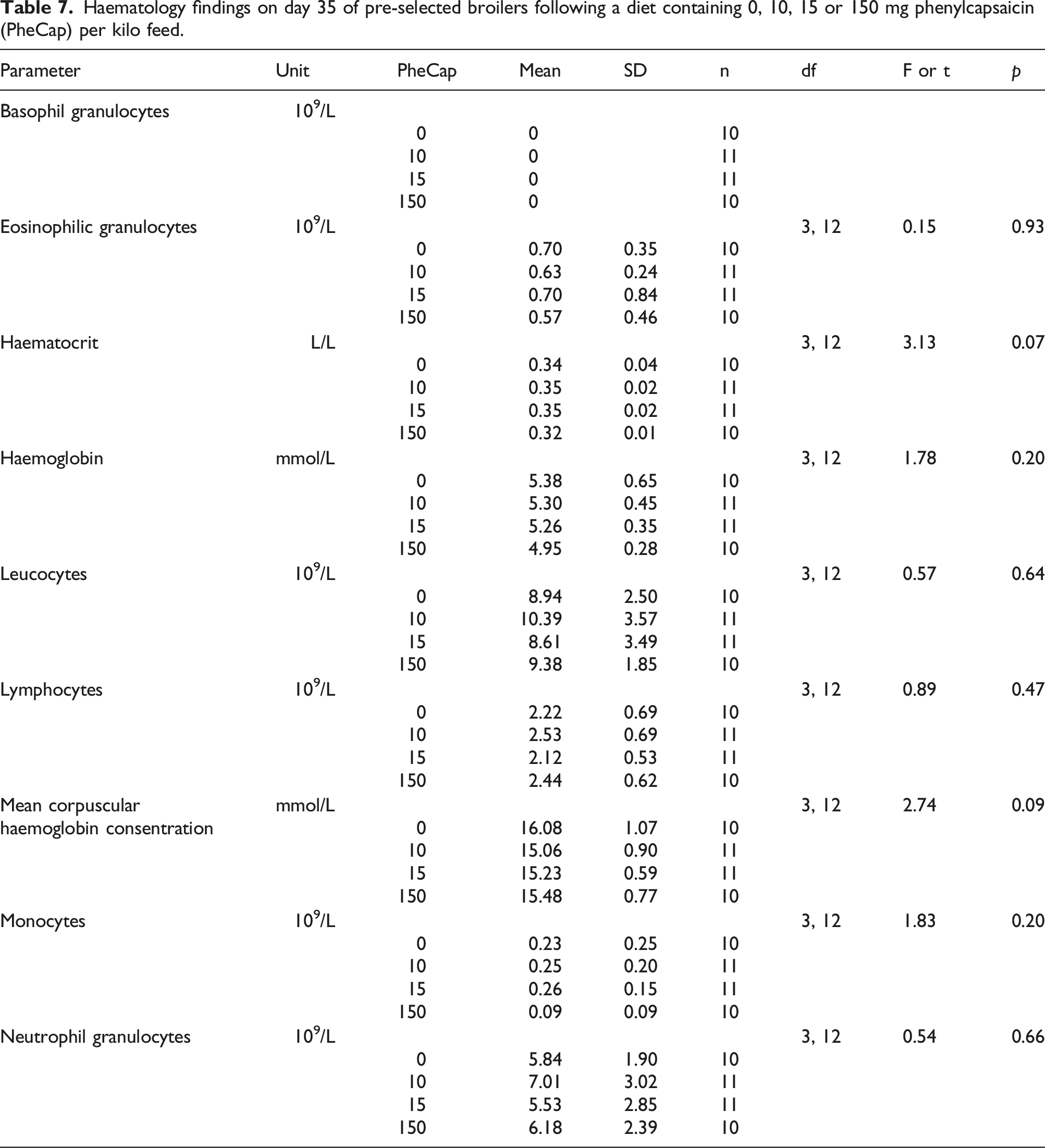
Haematology findings on day 35 of pre-selected broilers following a diet containing 0, 10, 15 or 150 mg phenylcapsaicin (PheCap) per kilo feed.
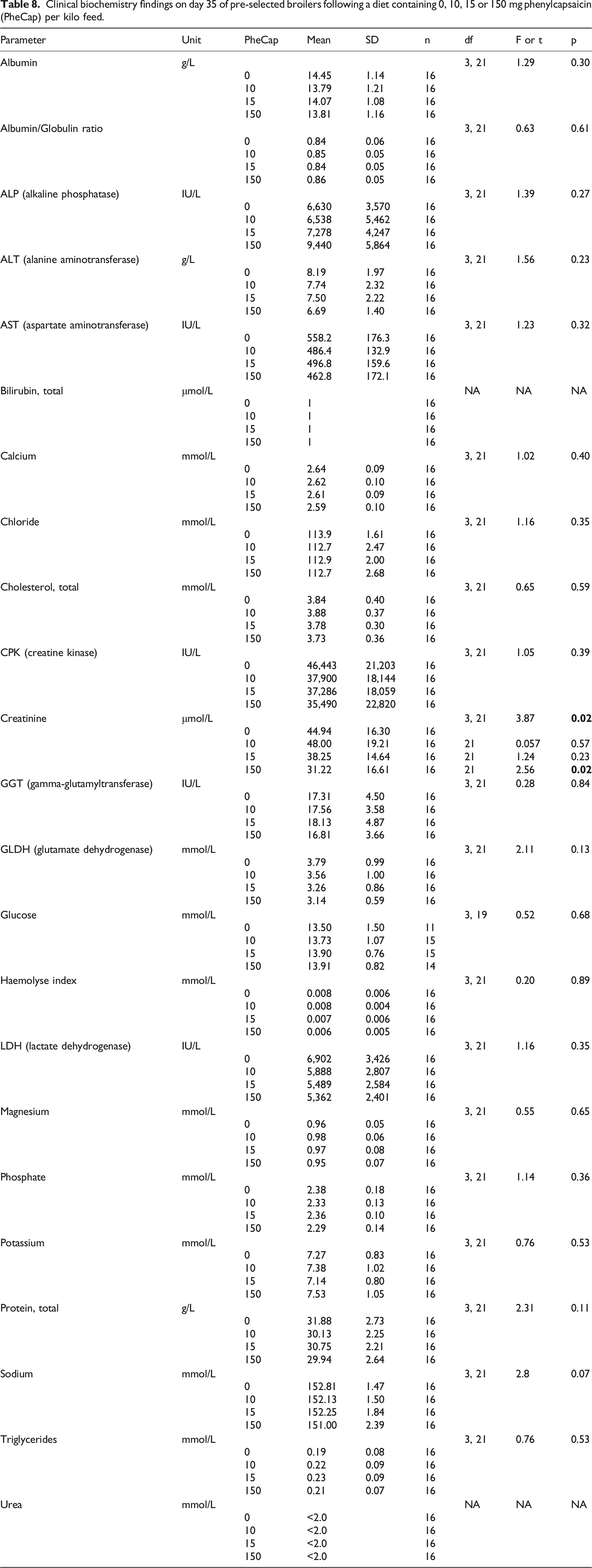
Clinical biochemistry findings on day 35 of pre-selected broilers following a diet containing 0, 10, 15 or 150 mg phenylcapsaicin (PheCap) per kilo feed.
Gross pathology
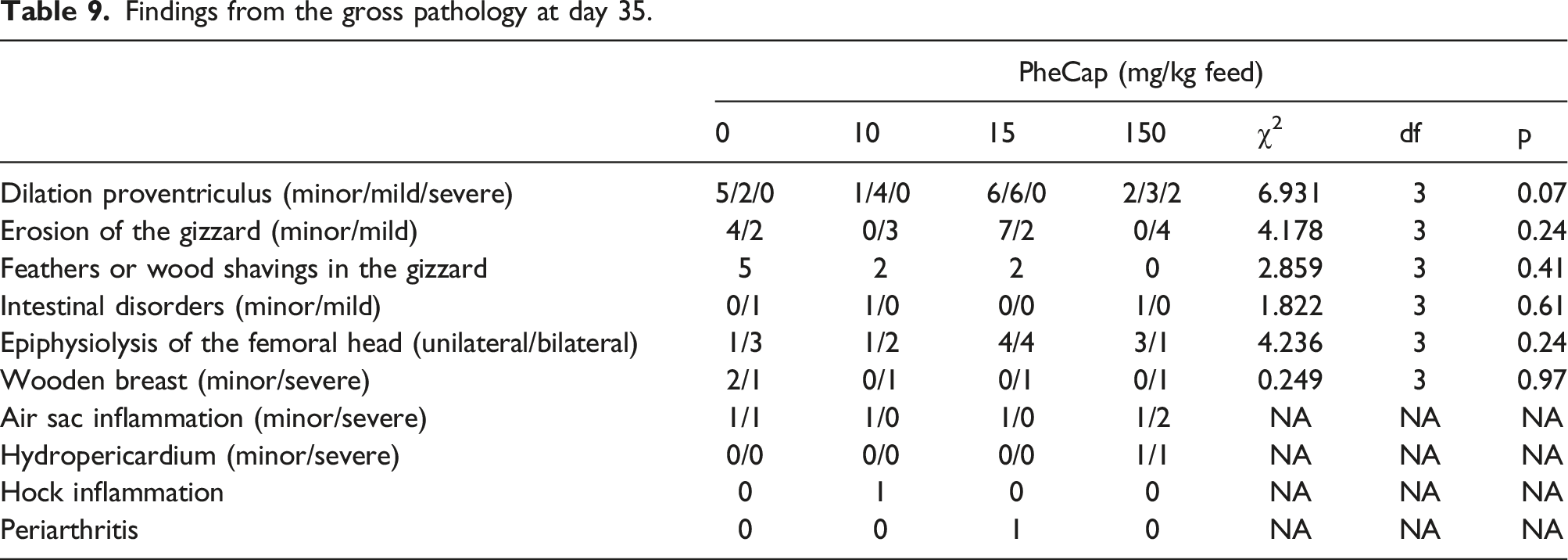
Findings from the gross pathology at day 35.
Discussion
Dietary inclusion of PheCap at levels up to 150 mg/kg feed did not affect BW, BWG, FI and FCR compared to the non-supplemented control group. This contrasts the effects found in Arbor Acre male broilers where the addition of 80 mg/kg pure natural capsaicin extract increased both averaged daily weight gain and final body weight in both the starter (d 1–21) and grower (d 22–42) periods, 16 and both 2 and 4 mg/kg decreased the FCR in the entire growth period (d 1–42). 14
Dietary inclusion of 10, 15, and 150 mg PheCap/kg feed resulted in lower mortality rates. Due to these lower mortality rates, EPEF of the 10 and 15 mg PheCap/kg feed treatment groups was significantly higher than the control group, with the highest numerical value in the 15 mg/kg feed group.
The reduced mortality found in the present study contrast the findings summarized by Abd El-Hack ME et al. 1 Although under dissimilar experimental conditions with hot red pepper (HRP) added to broiler feed at different concentrations, none of the five studies summarized showed reduced mortality. Reduced mortality was, however, found following a diet supplemented with both 0.1 g HRP, 1 g thyme and 1 g garlic per kg feed. 1
It is worth noticing that three dead broilers in the control group were diagnosed with septicaemia while no septicaemia was found in the PheCap treatment groups. Since capsaicin is used as a phytobiotic in broiler nutrition as a possible alternative to antibiotics in poultry, and the number of dead broilers in general was small in this study, it should be worthwhile to address in future studies to examine if PheCap could reduce septicaemia in broiler chicken and as such act as a phytobiotic.
Although no statistically significant differences were found between treatment groups and control at day 35, the average body weight of control group broilers was 180 g higher (2556 vs 2376), and the average feed conversion ratio 0.146 g/g lower (1.337 vs 1.463) than the Ross 308 male broiler performance objectives, 28 probably caused by a lower stocking density and better rearing conditions compared to farming conditions.
Dietary inclusion of Phenylcapsaicin did not negatively affect litter quality or footpad lesions.
No statistically significant effects of PheCap on the red and white blood cell parameters were found under the conditions of the present study. However, the numerical dose dependent decrease in the eosinophilic granulocytes and monocytes seen in the 150 mg PheCap/kg feed group compared to the control (Table 7.) could indicate positive effects on the immune system of higher doses of dietary PheCap in broilers, and this should be addressed in future studies. These effects were also seen following dietary red hot pepper supplementation. 1 A numeric dose dependent decrease was also seen for haemoglobin and mean corpuscular haemoglobin, and further research should explore whether high doses of PheCap can lead to anemia.
No toxicological relevant dose dependent effects were found for basophil granulocytes, haematocrit, leucocytes, lymphocytes, or neutrophil granulocytes.
Of the clinical biochemistry parameters, statistically significantly dose dependent response was only found for creatinine. Being a byproduct of skeletal muscle phosphocreatine breakdown, creatinine is an indicator of protein metabolism and an increase in serum creatinine is often used as an indicator of kidney damage.
29
This is not the case here
The dose dependent decrease found for ALT, CPK, GLDH, Haemolyse index, and LDH is not considered toxicologically relevant, and no toxicologically relevant dose dependent effects were found for sodium, potassium, chloride, calcium, phosphate, magnesium, total protein, albumin/globulin ratio, glucose, urea, cholesterol, triglycerides, bilirubin, AST, GGT or ALP.
During gross pathology, the examined broilers of the 15 mg/kg treatment group had the numerically highest occurrences of proventriculus dilatations, gizzard erosions and locomotion disorders. Because the pen was the experimental unit, individual PheCap consumption is not available to allow for comparison with the individual pathological findings under the present design. Furthermore, the occurrence of these findings is not consistent with the occurrence of these findings in the 10 and 150 mg/kg PheCap treatment groups. Thus, it is assumed that the numerically highest occurrences in the 15 mg/kg feed treatment group are coincidental and not caused by the inclusion of PheCap.
Only male broilers were used in the present study. However, we argue that because feed intake, and as such also the intake of the test substance, is higher in male than in female broilers, the toxicological conclusions presented are also valid for female broilers.
Because no adverse effects were found in either the performance results, haematology and clinical chemistry blood parameters or pathology, it is concluded that inclusion of phenylcapsaicin in broiler feed at doses up to 15 mg/kg feed has no negative effect on broiler chicken health, and that the tolerance dose of 150 mg/kg feed also has no negative effect on broiler chicken health. The No Observed Adverse Effect Level (NOAEL) of PheCap for systemic toxicity in Ross 308 broilers is therefore considered to be higher than 36.3 mg/kg BW/day, the highest weekly averaged daily intake found in the first week for the 150 mg PheCap/kg feed group in the present study, Table 4.
It is not surprising that no adverse effects were observed even in the tolerance level group receiving 150 mg PheCap/kg feed (150 ppm). The amounts used in the present study are much lower than the 1000 ppm capsaicin found in wild capsicum peppers fruits, fruits that birds commonly feed on.10,32
Intestinal colonisation of Salmonella is a major concern in the poultry industry, and low dose PheCap has shown promising effects as a phytobiotic alternative to antibiotics. In an unpublished full-scale pilot using a pre- and posttest design with paired-sample, the inclusion of 15 mg PheCap/kg feed in a regular starter diet reduced the the number of Salmonella-positive broiler chicken houses from 57 to 18 during the test period. If this result is confirmed in future studies, low dose inclusion of PheCap in regular poultry feed is a simple and efficient alternative to antibiotics for reducing Salmonella in the poultry industry.
Based on the low incidence of global findings, the non-pathological character of findings, and the absence of significant dose-response relationships for main parameters, it can be assumed that the inclusion of PheCap in broiler feed does not have any negative effects on animal health even at the tolerance dose level of 150 mg/kg.
Footnotes
Author contributions
JvH and TV conducted the study. TRP, JvH and TV drafted the manuscript and KHJ performed the data analyses. All contributed in the presentation and interpretation of the data and critical revisions of the manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: TRP hold shares in aXichem at time of submission.
Data availability
Data sharing is not applicable; the data is the property of aXichem AB.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was sponsored by aXichem AB.
Ethical approval
Not applicable, the study is a broiler feeding trail. The experimental protocol was approved by the Animal Welfare Body of Wageningen University (IvD-WU), Wageningen, The Netherlands (2019.D- 0033.001).
