Abstract
Environmental water sources are constantly polluted by anthropogenic compounds, not always minimized by conventional water treatment methods to remove these compounds at the micro- and nano-range. The absolute concentrations of a suite of seven representative environmental micropollutants were compared pre- and post-treatment with both ozone and microbial biofilms, in terms of removal efficiencies and toxicity assays. Both synthetic micropollutant mixes and environmental water samples were evaluated. The study started with two representative micropollutants (carbamazepine, CBZ, and sulfamethoxazole, SMX), and broadened into a suite of pollutants, evaluating whole-sample eco-toxicological footprints. An ozone concentration of 4.24 ± 0.27 mg/L in tap water, resulted in an 87.9% and 96.5% removal of CBZ and SMX, respectively, within 1 min. Despite almost immediate removal of parent micropollutants by oxidation, endocrine disruption potential (anti-estrogenicity) of CBZ and SMX required up to 240 min of ozone treatment to show no assay effect. A broader suite of micropollutants in more complex environmental matrices showed scavenging of ozone (2.95 ± 0.17–0.25 ± 0.03 mg/L) and varying micropollutant recalcitrance to oxidation. Lower matrix pollution led to lower reduction in eco-toxicity. Microbial degradation of CBZ and SMX (56% and 70% versus 19% and 79%, respectively, in duplicate biofilms) by nutrient-limited biofilms showed less removal than ozonation, with marked variation due to the stochastic nature of biofilm sloughing. Microbial degradation of CBZ and SMX resulted in an increase of >90% in both estrogenicity and Aliivibrio inhibition. The results obtained from this study address a gap in understanding the removal efficiency of micropollutants, where the removal process often receives more attention than the comparative reduction of toxicological effects. This shift from a controlled laboratory environment to real-world scenarios also provided comparative insights into the removal of micropollutants and the eco-toxicity of the transformation by-products of each process.
Keywords
Introduction
Freshwater systems are constantly contaminated by increasing anthropogenic activity, leading to the wide distribution, accumulation and persistence of various types of organic and chemical pollutants. Some of these pollutants have been described as micropollutants due to their occurrence and persistence at micro- and nano-grams per litre in the environment. Micropollutants are known to originate from various sources, such as domestic waste, agricultural practices and industrial processes. 1 The full effect of long-term exposure to complex mixtures of micropollutants on the environment, and subsequently people, is largely unknown. Chronic exposure to various micropollutants has been linked to human and wildlife endocrine disruption, the rapid development of anti-microbial resistance and increased risk of chronic illness.2–5 Conventional wastewater treatment works (WWTW’s) are not fully equipped to reduce the load of these chemical pollutants, as they are designed to remove organic waste, heavy metals and only a select few chemicals. This was confirmed in recent studies that found that many micropollutants and their transformation products pass through most WWTWs untreated and that WWTWs may serve as a site of accumulation for some persistent compounds, rather than removal thereof.6–8 Concentrations of micropollutants passing through WWTWs typically range from ng/L to μg/L in wastewater effluents. 9
A range of potential technologies exist with the capacity to degrade micropollutants, including UV and chlorine 10 carbon adsorption, 11 solar degradation/photocatalysis, 12 stabilisation ponds, 13 microbial degradation, 14 enzyme technology 15 and advanced oxidation processes (AOP). 16 Each technology is evaluated on two criteria: the removal efficiency of the micropollutants, as well as the toxicity of the transformation by-products after the removal process.
This study focuses on the comparison between electrochemical oxidation and biological degradation, according to these parameters. Carbon Based Electrochemical Oxidation (CabECO) technology is used as a model electrochemical oxidation process and harnesses the process of electrochemical oxidation to generate ozone in the aqueous phase of water, for disinfection and the reduction of persistent micropollutants. The CabECO cell comprises of a carbon membrane with two electrodes, encased in a plastic shell. 17 Ozone is generated in the aqueous phase due to a reaction on the anode when water is passed through the cell and the membrane is connected to a power source. Klavarioti et al. 18 summarized and evaluated various similar AOP’s for the removal of micropollutants. The degradation of compounds in wastewater treatment by advanced oxidation relies on the principle of oxidation by highly reactive hydroxyl ions.19,20 Removal efficiencies, however, vary greatly depending on the AOP’s used, or combinations thereof, and the chemical properties of the contaminants in question. 18
Biological degradation of micropollutants has also been widely investigated as a cost-effective and environmentally friendly alternative for micropollutant removal. Directed metabolism in microbial communities has achieved some success for the removal of selected micropollutants. 21 However, the process is not universal, and removal efficiencies vary with compound structure, aerobic and anaerobic processes, suspended and attached growth, as well as climatic conditions and the retention time of the treatment system.22,9,21
Although diverse treatment options exist for the removal of micropollutants, the processes are never universal for the incredibly broad array of micropollutants. The current library of micropollutants includes thousands of chemicals classified under this term, 23 and an extended list of unpredictable transformation products, depending on treatment technology and complexity of water matrices. 24 To get a grasp on this overwhelming problem, the EU Water Framework Directive has attempted to narrow this list down to 45 priority substances, based on extensive risk and persistence assessments, with Environmental Quality Standards. 25 Yet, increasing numbers of pollutants and by-products limit the feasibility of one-size-fits-all treatment technologies in real-world scenarios. Beyond the ever-widening target range of degradation products, the environment also influences the efficiency of removal technology, and thus multi-barrier, environmentally-controlled approaches are becoming popular. For instance, oxidants are effective in the removal of micropollutants, however, only when the ozone concentration is high enough to avoid depletion by non-target scavengers, such as high concentrations of chemical organic materials. 26 Because of the typically low environmental concentrations of micropollutants, low oxidation removal efficiencies have been demonstrated in water treatment systems where the COD (chemical oxygen demand) is high (> 200 mg/L). 27 The amount of energy required to produce enough oxidants to reduce such high COD loads becomes economically unfeasible. 26 A similar problem is encountered when utilizing microbial degradation to remove organic micropollutants from wastewater: effective micropollutant removal has been reported by microbial metabolism, but is inhibited when the organic load is increased with non-target carbon sources. 28 Microbial communities are unlikely to degrade recalcitrant micropollutants that are present at low concentrations, when there are high loads of other readily available organic compounds (g/L) available to drive metabolism. 29 Thus, both oxidation and the microbial degradation of micropollutants are ideal as final wastewater ‘polishing’ steps, after the bulk nutrients (carbon, nitrogen and phosphates) have been removed. 30 In terms of oxidation, this increases the rate of oxidation by-products interacting directly with the micropollutants. In terms of microbiology, this harnesses the metabolism of “hungry” biofilms, exposed to a selective evolutionary pressure with only the micropollutants as an energy source.
Thus, this study compares these two treatment processes, electrochemical oxidation and microbial degradation, for the removal of recalcitrant compounds at low concentrations, in terms of removal efficiency as well as the toxicity footprint of the by-products of each removal process. It is hypothesised that although CabECO (electrochemical oxidation) may have higher and broader removal efficiency than microbial degradation, the highly energized environment may generate free radicals and more toxic by-products. 31 In contrast, microbial degradation may be more specific and targeted, but microbial evolution may potentially also select for metabolic processes generating less toxic by-products.32–35 Similar studies often only evaluate removal efficiencies of micropollutant removal technologies, neglecting the toxicological effects of transformation products or metabolites after treatment. Furthermore, toxicological studies of micropollutant transformation products rarely compare treatment processes. It is envisioned that the findings of this study could provide valuable insights for the design of treatment strategies for downstream wastewater treatment processing as a polishing step, after the removal of the bulk nutrient loads.
Materials and methods
Electrochemical ozone transformation of CBZ and SMX, and toxicity of transformation products
Experimental setup
Two CabECO cells were connected in a stainless-steel piping system (Figure 1), where the flow paths could be adjusted to let water pass through either one of the cells individually or over both in series. Pressure and flow rate were controlled by means of piping system valves and a Watson-Marlow 630S peristaltic pump. The regular (5 min) inversion of the current flow through the CabECO cells prevented buildup of deposits on the electrodes.
36
Sampling points along the various flow paths allowed for sampling water either directly after passing over either membrane (Figure 1(a) and (b) respectively), or further downstream of both membranes (c). Schematic of the piping network, which houses and controls the passing of water over CabECO cells for the oxidative treatment of water. The position of the CabECO cells and the various sampling valves (A, B and C) are indicated.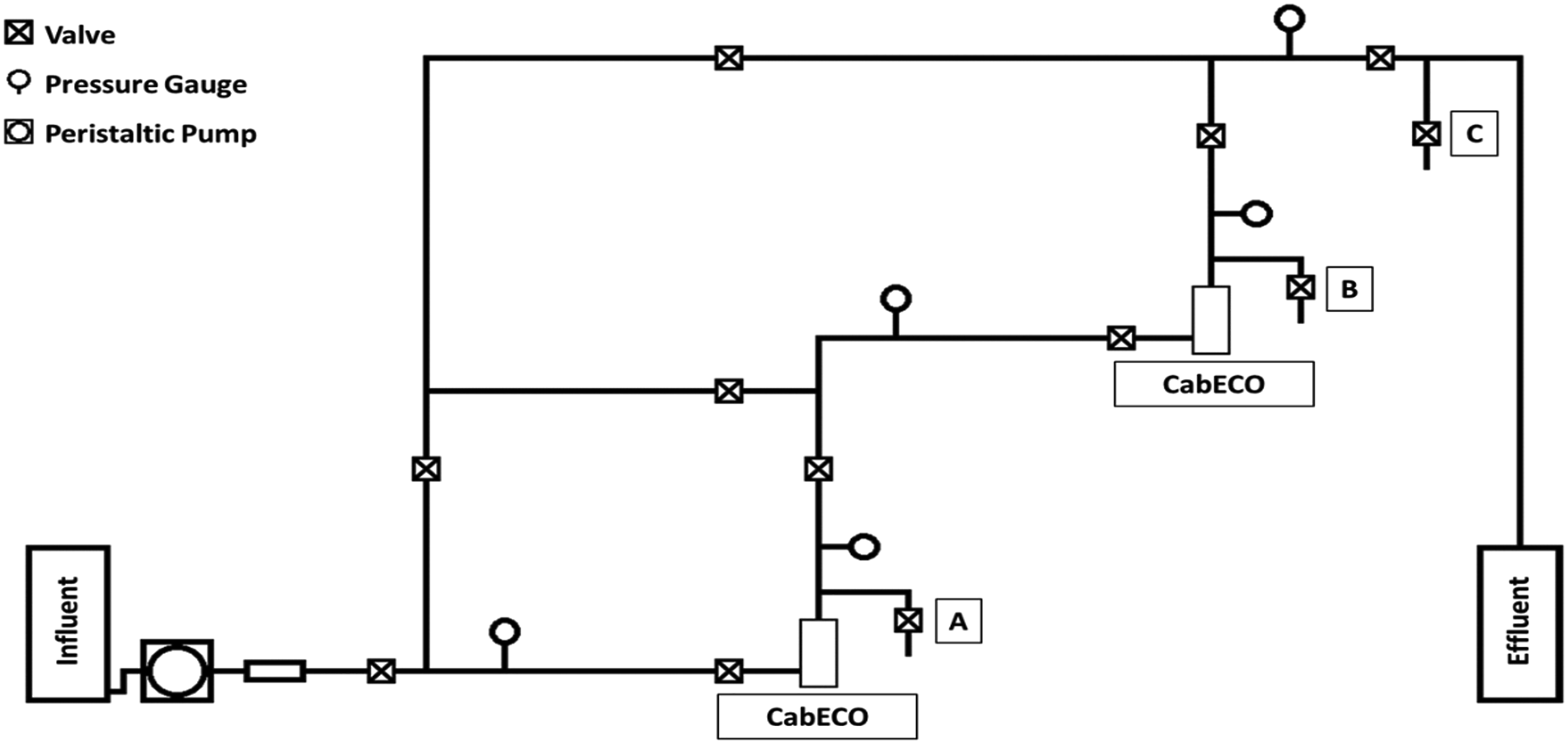
Reverse osmosis (RO) water was passed over two CabECO cells in series (Figure 1(c)) and water samples were analyzed to determine ozone concentration using an ozone concentration kit (Merck, HC850565) in combination with a Spectroquant® Pharo 300 (Merck, South Africa) until the desired ozone concentrations were achieved. Water samples were then collected in six 500 mL Schott bottles and filled to the brim to prevent gas exchange. Immediately upon collection, three bottles each were spiked to a final concentration of 500 μg/L for CBZ and 3000 μg/L for SMX respectively. These concentrations are approximately 100 times the environmental concentrations recorded for CBZ and SMX in South African waste waters, 6 and were selected for proof-of-concept. Although much higher than environmental concentrations, this is one of the few studies that did not exceed the milligrams per litre range for bench-scale evaluations of removal technologies.37–39 Although environmental concentrations are always preferable in experimental design, this is a comparative study between technologies, rather than a study of risk assessments or absolute concentrations. Since micropollutants are at such low environmental concentrations, their impacts are often at the lower end of measurement techniques and standard curves, where standard deviation is higher and data resolution lower. Thus, these technologies were compared at higher micropollutant concentrations, to comparatively assess their efficiency. Control samples consisted of RO water spiked with the same concentrations of CBZ and SMX, passed over the system without ozonation. Samples for analysis were collected from all bottles at time intervals of 1 and 4 h.
Microbial transformation of CBZ and SMX, and toxicity of transformation products
Biofilm inoculum preparation
A standardized inoculum, of wastewater origin, was prepared and stored (−80°C) for subsequent biofilm experiments. To prepare this inoculum, a wastewater grab sample was collected in a sterile 50 mL Falcon tube from a WWTW aeration tank (Western Cape, South Africa). After vortexing (1 min), 1 mL of the sample was used as initial inoculum and added to 3 L of micropollutant-enriched M63 media (M63*) in conical flasks, and grown aerobically with agitation (7 days, 26°C, 120 r/min). M63* medium was prepared as described by Elbing and Brent (2002), 40 with the only exception that the primary carbon source, glucose/glycerol, was substituted with a high dose of the respective micropollutants: CBZ (500 μg/L) and SMX (3000 μg/L). Thereafter, every 7 days, 1 mL of the culture was transferred to a sterile 3 L flask of M63* medium, continuously incubated under the same conditions. This was repeated three times, applying selective evolutionary pressure for a microbial community capable of growing on the experimental concentrations of CBZ and SMX. The final culture was used to prepare standardized freeze cultures and stored at −80°C (40% glycerol). For biofilm inoculation, 100 μL of the thawed culture was added to 100 mL of M63* media and incubated (26°C, 120 r/min, agitation) until it reached the desired inoculation concentration after 24 h (OD of 0.6 at 600 nm, ±106 CFU/mL), pre-determined with growth curves.
Biofilm exposure to micropollutants
To investigate the ability of biofilms to remove (bind or metabolically transform) the selected micropollutants (CBZ and SMX), biofilms were grown up in flowcells. 41 Modified M63* medium was aseptically passed through 1.6 mm inner diameter (3 mm outer diameter) silicone tubing to the flowcell (Perspex, glass microscope slide) at 14 mL/h. Glass bubble traps inserted upstream from the flowcells captured air bubbles in the system and reduced the risk of biofilm disruption. 42 For this experiment, 8 flowcell channels evaluated four treatments in duplicate. Channels 1 and 2 were supplied with M63* medium, with the carbon source replaced by CBZ to a final concentration of 200 μg/L. Channels 3 and 4 were supplied with M63* where the carbon source was replaced by SMX to a final concentration of 200 μg/L. Channels 5 and 6 were supplied with M63* medium where the carbon source was replaced by both CBZ and SMX, combined, to a final concentration of 100 μg/L each. Channels 7 and 8 served as un-inoculated controls and were supplied with the same combined medium as channels 5 and 6, to monitor micropollutant degradation in a sterile system. Channels 1 to 6 were each inoculated by injecting 200 μL of the culture (2.2.1) directly into the flowcells.
Prior to inoculation, the flowcell system was sterilized with a 10% sodium hypochlorite solution for 2 h, followed by sterile dH2O (distilled water) for 10 h (14 mL/hr). The respective media were then pumped through the system for 2 h before inoculation (14 mL/hr). During inoculation, flow was switched off and the silicone tubing up and downstream from the flowcells clamped off to allow the cells from the inoculum to adhere to the flowcell chamber (1 h) before flow was switched back on.
Once the flow of the medium was resumed, effluent (1.5 mL) was collected from each respective channel and the optical density was measured at an absorbance of 600 nm every 6 h using a Spectroquant Pharo300 spectrophotometer (Merck, Germany). Additionally, 1 mL of the effluent was collected from each flowcell, from which serial dilutions (10−1–10−8) were prepared and plated out on 3 g/L TSA in triplicate. All TSA plates were incubated at 26°C for 48 h, where-after the resulting colony forming units were counted.
Biofilms were grown and allowed to mature for 15 days, where-after 100 mL and 300 mL volumes were sampled in triplicate from each influent and effluent containers respectively for HPLC-MS analysis, yeast anti-estrogen screening and Aliivibrio toxicity assays, as described in the subsequent methodological sections.
Electrochemical ozone micropollutant transformation of environmental samples, and toxicity of transformation products
In order to test the technology on a broader range of environmental micropollutants, 10 L of water was collected at the following two river sites in Stellenbosch, Western Cape: (1) the Eerste river (−33.940,574, 18.889,760) which is a relatively pristine river, (2) the Plankenbrug river (−33.932,923, 18.851,525) which is an extremely polluted river, and municipal shower grey water, collected and stored at 26°C for 48 h. The shower water was collected directly from use, as was a common practice during a drought in the area, for flushing toilets. Triplicates of each sample were passed over two CabECO cells in series (60 L/h; Figure 1(c)) and collected for analysis. Between samples, the system was flushed by passing tap water over the two cells in series for 30 min. Ozone was monitored until it reached steady state, where-after the influent was switched to the respective sample. At 60 L/hr the sample would pass through the system in less than 40 s (flow rate and retention time pre-determined with food coloring in tap water) and thus samples were passed over the two CabECO cells for 2 min before collection, to eliminate any dilution effects from the tap water. Triplicate samples were collected in separate 0.5 L and 1 L bottles, filled to the brim, sealed immediately and left undisturbed for 1 h (incubation time selected based upon 2.1.1) at room temperature. All untreated and CabECO treated environmental samples were analysed for total carbon (TC), total organic carbon (TOC) and total inorganic carbon (TIC). The triplicate 0.5 L samples were analysed for micropollutant removal efficiency (HPLC-MS), measuring the absolute concentrations of the following compounds: carbamazepine (CBZ), sulfamethoxazole (SMX), acetaminophen (ACM), benzotriazole (BZT), diclofenac (DCF), caffeine (CAF) and atrazine (ATZ). The 1 L sample was analysed in triplicate for eco-toxicity (yeast estrogen screen and the Aliivibrio toxicity assay).
Measurement of micropollutant removal efficiency (HPLC-MS) and eco-toxicity (YES and Aliivibrio assays)
Solid phase extraction
Solid Phase Extraction (SPE) for High Performance Liquid Chromatography and Mass Spectrometry (HPLC-MS) was performed as described by Petrie et al. 43 Briefly, collected samples (100 mL) were spiked with the corresponding internal standards to a final concentration of 50 μg/L (in the final eluted sample) before filtering through a 0.7 μm glass fibre filter. Oasis HLB 3 cc extraction cartridges were conditioned with 2 mL of methanol (MeOH) followed by 2 mL of ddH2O (MilliQ/double distilled water) at a rate of less than 1 mL/min. The filtered sample was then passed through the cartridge at a rate of 5 mL/min. After allowing for complete drying of the cartridge, the sample was eluted from the cartridge with 4 mL of MeOH under gravity, and collected in 5 mL glass test tubes. All glassware used during SPE and subsequent HPLC-MS processing procedures were rinsed with MeOH and double distilled water, three times. The effect of glassware silanization was investigated and it was found that the compounds of interest at the concentrations used in this study showed no significant binding to the glassware surface. The eluted samples were dried under nitrogen and reconstituted in 500/1000 μL (depending on required concentration of final sample) of MeOH. After resuspension by vortexing, 175 μL of the sample was transferred to polypropylene HPLC-MS vials with glass inserts and subjected to HPLC-MS analysis (HPLC; Waters AQUITY). 6 When samples immobilized on the column were kept for later analysis, they were wrapped in parafilm and aluminium foil directly after drying, and stored in airtight plastic bags at −30°C.
Similarly, Solid Phase Extraction for the yeast estrogen screens (YES) was performed as described by Petrie et al. 43 for HPLC-MS sample preparation, with minor modifications. Briefly, Oasis HLB 6 cc extraction cartridges were conditioned with 4 mL of MeOH followed by 4 mL of ddH2O at a rate of less than 1 mL/min. Samples were collected (300 mL) and filtered through a 0.7 μm filter. The filtered sample was then passed through the cartridge at a rate of 5 mL/min. After allowing for complete drying of the cartridge, the sample was eluted from the cartridge with 6 mL of MeOH under gravity and collected in 10 mL glass test tubes. The eluted samples were dried under nitrogen and reconstituted in 600 μL of MeOH. The samples were transferred to glass vials and stored for no more than a day at 4°C before being subjected to the yeast screen.
Toxicity (endocrine disruption) assays: Recombinant yeast estrogenic screen
The yeast-based screen for estrogenic activity was performed according to Sohoni and Sumpter. 44 Briefly, a recombinant yeast strain of Saccharomyces cerevisiae was used (obtained from Prof JH van Wyk, University of Stellenbosch), which had been transfected with the human estrogen receptor (hER) gene and a plasmid containing an estrogen response element-linked lac-Z gene. The binding of EDCs and steroids in the water sample to the lac-Z reporter gene leads to the production of β–galactosidase, metabolising chlorophenol red galactopyranoside (CPRG) in the medium, which results in a measurable red-yellow colour change.
The recombinant yeast strains were inoculated in an assay medium [45 mL minimal medium, 5 mL 20% Glucose solution, 1.25 mL L-Aspartic acid (4 mg/mL), 0.5 mL Vitamin solution, 0.4 mL L-Threonine (24 mg/mL), and 0.125 mL Copper (II)Sulphate solution (0.319 mg/mL)] and incubated for 24 h on an orbital shaker at 130 r/min at 26°C. A 2 mL sample from this culture was transferred to fresh minimal media and incubated for 24 h at the same conditions.
All samples and concentration factors were analysed in triplicate in the same assay plate. A standard curve for the steroid hormone 17β-estradiol (E2; CAS 50–28-2; Sigma) was included for each assay plate in 12 serial dilutions as a positive control, ranging from 1.3 to 2724.0 ng/L. Blank wells were also included in each assay plate containing only the assay medium, without any hormone spike or sample extracts. Serial dilutions of the samples concentrated with SPE and reconstituted in MeOH (2.4.1) were prepared, and 10 μL of each dilution was transferred to a sterile 96 well flat bottom plate (Costar, 3370, Sigma) and allowed to evaporate. After evaporation, all the wells on the plate were inoculated with 200 μL of the incubated assay medium containing 0.5 mL of CPRG (10 mg/mL). The assay plates were then allowed to incubate on a shaker for 72 h at 30°C under dark conditions.
After 72 h of incubation, or when an adequate colour change was observed in the dilution range of the E2 standard curve, the absorbance of the assay plate was read with a plate reader at 570 nm (CPRG metabolism) and at 620 nm (turbidity to assess cell toxicity).
Toxicity assays: Aliivibrio luminescence
The Microtox® acute bioassay (Johannesburg, South Africa) was used to assess the toxicity of micropollutants treated with CabECO ozonation. Samples assessed were as described above: (1) pure micropollutants solubilized in distilled water (2.1), and (2) polluted and pristine water sources.
All Microtox® assays were prepared according to manufacturer’s instructions, with amendments outlined below, and measured using a MicroTox® Model 500 toxicity analyser. During the assay, the luminescent bacteria were maintained in an incubator at 16°C. Luminescence data was recorded as light emission at 0, 15 and 30 min, and recorded and analysed in the Microtox® Omni™ (version 1.18) software. The assay measures the inhibition of bioluminescence due to pollutant impact on the growth of the Gram-negative marine bacterium, Aliivibrio fischeri (strain NRRL B−11,177), according to routine methods (ISO 11,348–3 2007; ASTM Standard method D5560-96 2009). Lyophilized bacteria were obtained from Microtox® and stored at −20°C. Immediately prior to the assay, they were reconstituted in 1 mL of Microtox® reconstitution solution (ultrapure water +0.01% NaCl), and acclimatised at 8°C (1 h) before acclimatizing to room temperature (1 h). The assay included a negative control (sterile tap water), and a reference toxicant (potassium dichromate; 20 mg/L), each prepared in triplicate. All samples were prepared in duplicate.
Dose-response curves (6-point curves, at 50% dilutions, starting at 100 mg/L) were designed for CBZ, SMX and CBZ+SMX (at a 1:1 ratio). The manufacturer’s protocol was adjusted due to the toxicity of the diluent used to release the micropollutants from the columns (MeOH), and the prerequisite of solubilising the micropollutants in MeOH. The standard protocol for a dose response curve involves solubilising the toxicant in water, and adding the bacteria (500 μL) to the sample (500 μL). However, the toxicants in this study, the micropollutants, are eluted from the columns, dried under nitrogen and reconstituted in pure MeOH. The MeOH is toxic to the bacteria, and these samples cannot be used in the toxicity assays. To account for this, dose response curves were also amended. CBZ, SMX and CBZ+SMZ samples were solubilized in MeOH, added to the test tubes (500 μL) and similarly dried under nitrogen gas to evaporate the MeOH. In these tubes, 500 μL of the 2% NaCl used for the bioassay definitive tests was added, vortexed for 10 s, and the bacteria were added (500 μL) to make up a total volume of 1 mL.
Identical dose response curves (CBZ, SMX and CBZ+SMX) were prepared according to standard protocol, solubilized in ddH2O. These were compared to the evaporation method described above. The SMX did not solubilize completely in the water, but the dose response curve was generated with the precipitate, to evaluate the effect of the evaporation method.
Sample sets tested for toxicity, according to the evaporation method, included (1) the total micropollutant footprint of the Plankenbrug and Eerste Rivers, as well as (2) samples pre- and post-biological treatment (section 2.2). The same samples used in the above-mentioned section, reconstituted for the YES assay, were subjected to the Aliivibrio toxicity assay.
Statistical analysis
Unpaired Student t-tests quantified significant differences between independent means. One-way analysis of variance (ANOVA), with Tukey’s Honest Significance Difference (HSD) test as post hoc analysis, evaluated the differences between means of multiple groups. All tests, including testing for normality, were performed in Microsoft Excel, with a 95% confidence interval.
Results and discussion
Electrochemical ozone transformation of CBZ and SMX, and toxicity of transformation products
At maximum ozone generation over the CabECO system (4.24 ± 0.27 mg/L) The electrochemical oxidation removal rate of CBZ and SMX over time. (a) The percentage reduction of CBZ and SMX over time (1 and 4 h), as well as CBZ and SMX combined at 4h (4(C+S)). (b) The electrochemical oxidation removal rate of CBX and SMX individually, at more resolved time intervals. (* indicates statistically significant differences between treatment parameters and no treatment and the treatment intervals).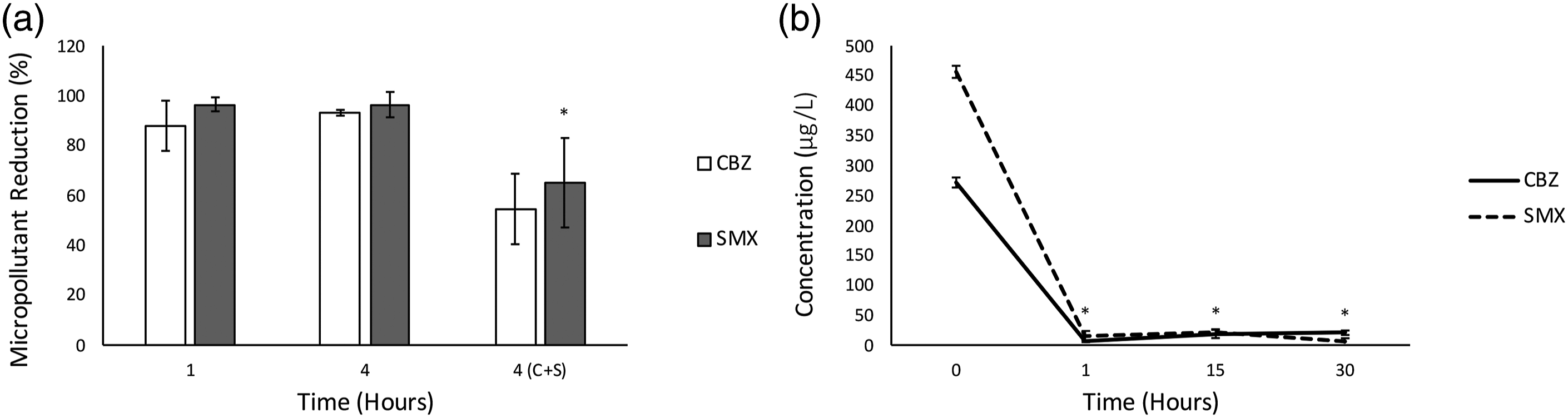
During micropollutant abatement, ozone and/or hydroxyl radicals, that can be formed by decomposition of ozone with water constituents, are responsible for driving the oxidation of compounds. 45 Although ozone is one of the strongest oxidizers, it is a selective oxidant and favours reaction with electron-rich compounds such as phenols, olefins, reduced sulfur groups and deprotonated amines. In contrast, hydroxyl radicals are less selective and react freely with almost all types of organic materials. Proportionally few hydroxyl radicals are generated in the ozonation process harnessed by the CabECO technology 46 and therefore, the majority of the micropollutant abatement in this study can be ascribed to oxidation by ozone, although hydroxyl radicals can also contribute to a small extent. 47
It is well-known that the ozonation of CBZ and SMX effectively reduces their concentration in various types of water matrices. For CBZ, various studies show 75–100% removal efficiency, depending on the method of ozone exposure and treatment time (20 min maximum).48–50 Contradictory results have been found, where ozonation resulted in only 30% removal of CBZ. 51 Good removal efficiency of SMX was reported in literature, with removal efficiencies ranging from 88 – 100%. However, SMX required longer exposure times overall in comparison with CBZ, with treatment times reaching up to 60 min52–54 In contrast to these studies, we report an immediate (1 min), almost complete (>96%) degradation of SMX with this technology. This might be due to the high concentrations of SMX in previous studies, in comparison to the concentration closer to environmental levels evaluated in this study. The CabECO cells seem to have the advantage of very high removal efficiency within a very short period of time, in comparison to other technologies. 55 The CabECO cells showed >96% removal efficiency for both CBZ and SMX after 1 min of exposure time. A possible reason for CabECO being such a good candidate for micropollutant abatement is the direct contact of the water with the ozone-generating membrane. The CabECO cell is designed to increase turbulence over the membrane as the water is passed through the cell. Additionally, CabECO has the advantage of harnessing the ozone in the aqueous phase and therefore not losing any reactive capacity by having to transfer ozone from the gas to the aqueous phase, as is typical of standard ozone technology.
Considering the efficient transformation of these two parent compounds, two questions arise: (1) what transformation products are formed, and (2) how toxic are they? However, due to a large and diverse suite of pollutants and degradation products, 23 this question becomes complex and expensive. CBZ alone has more than 30 known human metabolites, and up to 13 predicted transformation products from ozonation.32,56 Bahlmann et al. 32 reports that some of the metabolites of CBZ remain therapeutically active and some have a higher eco-toxicological concern than CBZ itself. SMX can be metabolized into five major metabolites and six predicted ozonation transformation products, all with potential for increased toxicity.33,34
In light of this complexity, and expenses associated with tracking individual parent compounds’ and transformation products’ fate post treatment, the toxicity of transformation products was investigated in this study. The extent of endocrine disruption (YES screen) and bacterial growth inhibition (Aliivibrio luminescence assays) were measured against the total micropollutant footprint of the water sample. Endocrine disruptors have been linked to a number of health indices, including decreased fertility, increased incidence of cancers, birth defects, increases in spontaneous abortions and several physiological disorders in various organisms (mostly aquatic).6,57,58 These assays were harnessed to measure the toxicity of transformation products. The effects of CabECO and biological micropollutant removal technologies were compared against the whole extraction footprint of the water, after treatment with these two engineered processes.
The estrogenic response is measured against an estradiol (E2) spike, (a predetermined hormone concentration known to disrupt endocrine activity). Any measurement lower than the half maximum effective concentration (EC50) of the E2 spike is considered anti-estrogenic, indicating that the micropollutants inhibited the expected estrogenic response of the E2 spike, whereas measurements higher than the EC50 of the E2 spike add to estrogenicity. Figure 3 shows the results of the YES screen for both CBZ and SMX treated with electrochemical oxidation. Before treatment, CBZ and SMX both show an anti-estrogenic effect. Over time, the anti-estrogenicity of CBZ is slowly decreased by exposure to electrochemical oxidation, until it exhibits no estrogenic/anti-estrogenic effect (240 min, p < .05). In contrast, the removal rate of CBZ (parent compound) is almost complete, and almost immediate (>96% removal efficiency, 1 min, Figure 2(b)). It is evident that even though the parent compound is removed efficiently, the YES results suggest that CBZ transformation products are present and resistant to immediate removal by electrochemical oxidation. Removal of these transformation products may be important to the life and health of aquatic and other organisms, including humans, through their own endocrine-disrupting effects. Estrogenic effect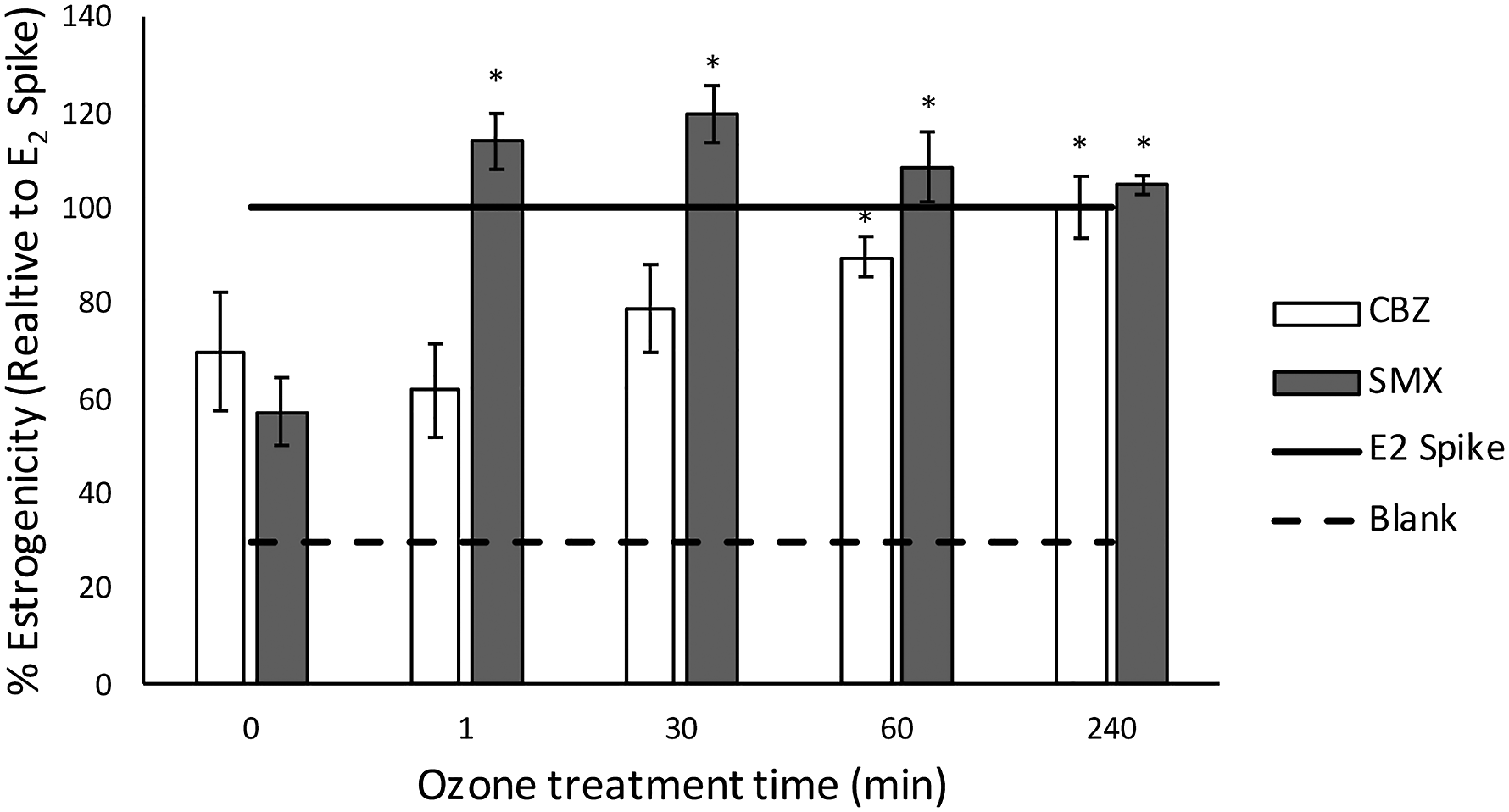
Prior to CabECO treatment, SMX also showed an anti-estrogenic effect similar to CBZ (Figure 3). However, unlike CBZ, the toxicity response rate of SMX transformation products (Figure 3) reflected the removal rate (Figure 2(b)). After electrochemical oxidation, the SMX transformation products show no anti-estrogenic activity. In contrast, by the 1 min time point, they become slightly estrogenic and decrease to almost negligible hormonal activity over time (240 min, p < .05).
Overall, the comparative data supports the implementation of a residence column in the final design of an operational CabECO system to WWTW’s. The desired residence column would allow for a longer retention time of ozone-rich water in the system allowing for adequate reaction time between the ozone and the transformation by-products of the contaminants. The longer retention times required to effectively reduce the toxicological effects could be explained by the oxidized transformation products that retain bio-active properties, potentially being less readily oxidized due to the more stable functional groups on the molecules.
Microbial transformation of CBZ and SMX, and toxicity of transformation products
In this study, planktonic cultures, exposed to the same evolutionary pressure over the same period of time (15 days), showed no micropollutant removal and no toxicity as measured by the YES (data not shown). Biofilm studies were thus selected to evaluate the potential of evolutionary selection pressure for the removal of micropollutants by microbial communities of WWTW origin. Biofilms have several advantages over planktonic cultures for bioremediation, including increased resistance to antimicrobial compounds, a wider metabolic range due to aerobic and anaerobic zones in the biofilm structure, inter-species co-operation and horizontal genetic transfer, as well as increased protection from physical forces.59–61 The channels and tunnels within the biofilm structure and EPS-sorption (extracellular polymeric substances) also increase the system retention time, allowing for longer microbe-compound contact time. 62 Within this study, the removal of micropollutants over the biofilm was assessed in post-biofilm effluent water samples, without distinguishing whether removal was due to sorption or metabolic transformation.
The biofilms in this study are termed “hungry” biofilms, as the carbon source in the M63* enrichment medium is replaced entirely with the respective micropollutant (CBZ or SMX), or both in combination. The typical sigmoidal growth curve of a biofilm grown on carbon-rich media was not observed in the effluent cells,
63
but rather, the biofilms responded to the stress of carbon limitation by periodically releasing planktonic cells, as measured by CFU’s (Figure 4). It has been demonstrated that stress responses affect the release of planktonic cells from biofilms, suggesting that the microbial communities involved in these micropollutant polishing steps are metabolizing the micropollutants, but not as comfortably as more readily accessible carbon sources.
64
Even over a longer adaptation period, the irregular cell-release (sloughing) response does not decrease. Biofilm growth curves, measured as OD, representative of the planktonic cells released from biofilms exposed to M63* medium with (a) CBZ, (b) SMX, (c) CBZ+SMX as carbon sources, with a sterile control (d). Duplicate biofilms (1 and 2) are plotted on each graph.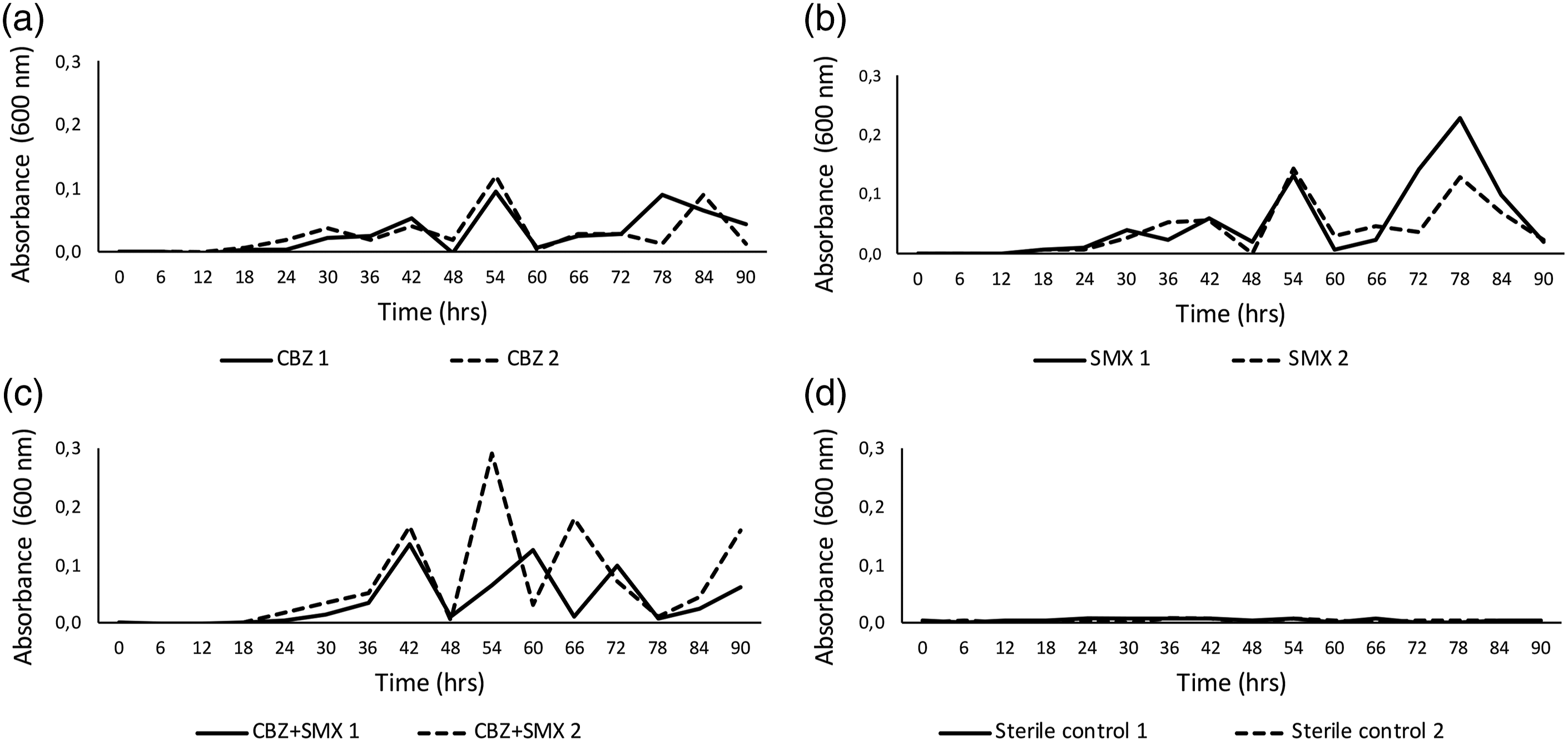
Microbial biofilm communities, originating from WWTW and adapted to CBZ and SMX exposure, showed a CBZ removal efficiency of 28% (Figure 5). There is significant variation (p < .05) between the removal efficiencies of the duplicate, which might be explained by the irregular sloughing events. During such an event, the biofilm loses most of the structure, biomass and EPS that potentially aid in CBZ degradation or absorption. Furthermore, micropollutant degradation by microbial metabolism is enzyme-mediated and the loss of biomass would presumably lead to less enzymes produced and shorter retention times. The cytochrome P450 group of enzymes (found in all lifeforms), as well as the lignin-degrading enzyme laccase, metabolize most organic compounds by adding functional groups such as –OH, -SH, -NH2 or –COOH during metabolism to form more hydrophilic products.65,66 In this study, the final micropollutant removal efficiency in the medium was measured as a snapshot in time. A future study could test the removal efficiency throughout these sloughing cycles and relate removal rate to accumulated biomass. (a) Percentage CBZ reduction by duplicate biofilms grown up on CBZ-enriched media and CBZ+SMX-enriched media and (b) Percentage SMX reduction by duplicate biofilms grown up on SMX-enriched media and CBZ+SMX-enriched media. Biofilms were grown on M63* media, with the carbon source replaced by the respective micropollutants, or combinations thereof, and the micropollutant removal rate measured with LC-MS (* indicates statistically significant differences between treatment parameters) (** indicates statistically significant differences between replicates).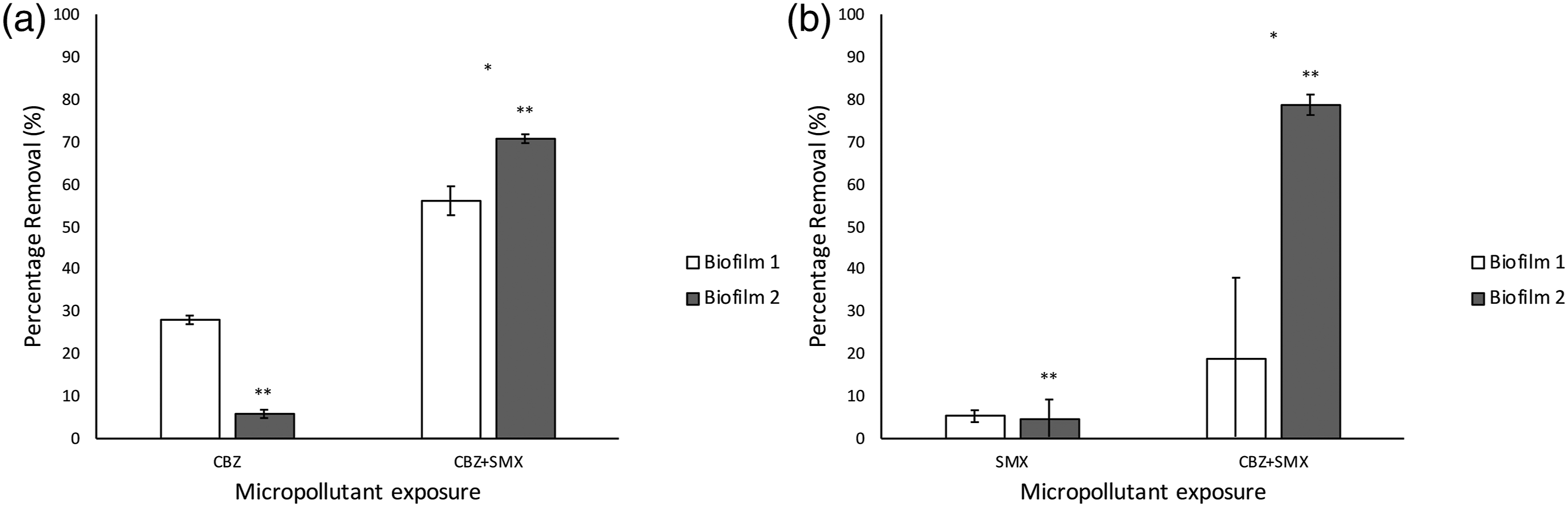
There was a significant increase in CBZ removal, when exposed to the biofilm grown on the media supplemented with the combination of CBZ and SMX (100 μg/L each) compared to the biofilm grown up on the media supplemented with only CBZ (200 μg/L) (one-tailed T-test, p < .05). The duplicates of the combined biofilms removed 56% and 70% of the CBZ, whereas the best performing CBZ biofilm only removed 28%. Although variation between the duplicates is significant (p < .05), it can be concluded that the combined biofilm is more efficient in CBZ removal. The same effect is seen in the removal of SMX by the biofilms (Figure 5). Although the variation in the combined biofilms is high, the SMX biofilms have a significantly lower removal efficiency of SMX, when compared to the biofilms grown on both micropollutants in combination (p <.05).
Several factors might account for the possible better removal performance of the combined biofilms. The higher individual concentration of CBZ and SMX in the single-compound biofilms media might have led to some inhibitory effect on the biofilm, resulting in a less active biofilm compared to the combined ones. This effect is not toxic, as there was no difference in effluent cell concentrations between the biofilms. This could be expected for the SMX biofilm, as SMX is an antibiotic. The minimum inhibitory concentration (MIC) of SMX is 25 mg/L, well above the 200 μg/L in the SMX biofilm medium, therefore an inhibitory effect on the biofilm is improbable. 67 However, MIC’s are determined based on cell death, and metabolic impact might nevertheless present at lower concentrations. Inversely, the increased pressure from the combined effect of CBZ and SMX may have led to several stress responses that promoted metabolic responses that impacted micropollutant removal. As a stress response, biofilms are known to have an increased metabolic rate,68,69 higher biomass production 70 and in the case of nutrient deprivation, produce more EPS and quorum sensing molecules, 71 as well as altered gene expression. 72 Thus, if there was a higher stress response in the biofilms exposed to the micropollutants in combination, increased removal rates may be due to increased metabolic turnover of the parent compounds, or sorption to increased EPS.
The reported efficiency of biofilms to remove CBZ and SMX is contentious. Various studies have reported that CBZ shows recalcitrance towards microbial degradation.73–75 However, Hellauer et al. 76 found that, with the addition of a labile carbon source, a previously nutrient-deprived biofilm showed increased removal of CBZ. This could be due to co-metabolism, or simply more cells producing the required enzymes. The improved removal efficiency when combining the micropollutants in the current study might be attributed to this co-metabolism phenomenon, as SMX is known to be more easily degraded by biofilms, 73 thus potentially acting as a nutrient source and facilitating the removal of CBZ. In a similar study by Zhang et al., 75 where the addition of low levels of acetate to the system enhanced micropollutant degradation, adding carbon in excess reduced the removal efficiency due to preferential metabolic pathways stimulated by redox changes. 75 The proposed link between removal efficiencies and co-metabolism 75 might be supported in this study, by the increased removal of both SMX and CBZ in the biofilm exposed to combined micropollutants. However, sorption is a viable alternative for removal. A future study exploring the ratio of labile carbon sources to micropollutants in the growth medium for biofilm cultivation could be beneficial for determining optimal micropollutant degradation by microbial co-metabolism. Similarly, co-metabolism with nitrogen or other nutrients may promote micropollutant degradation.
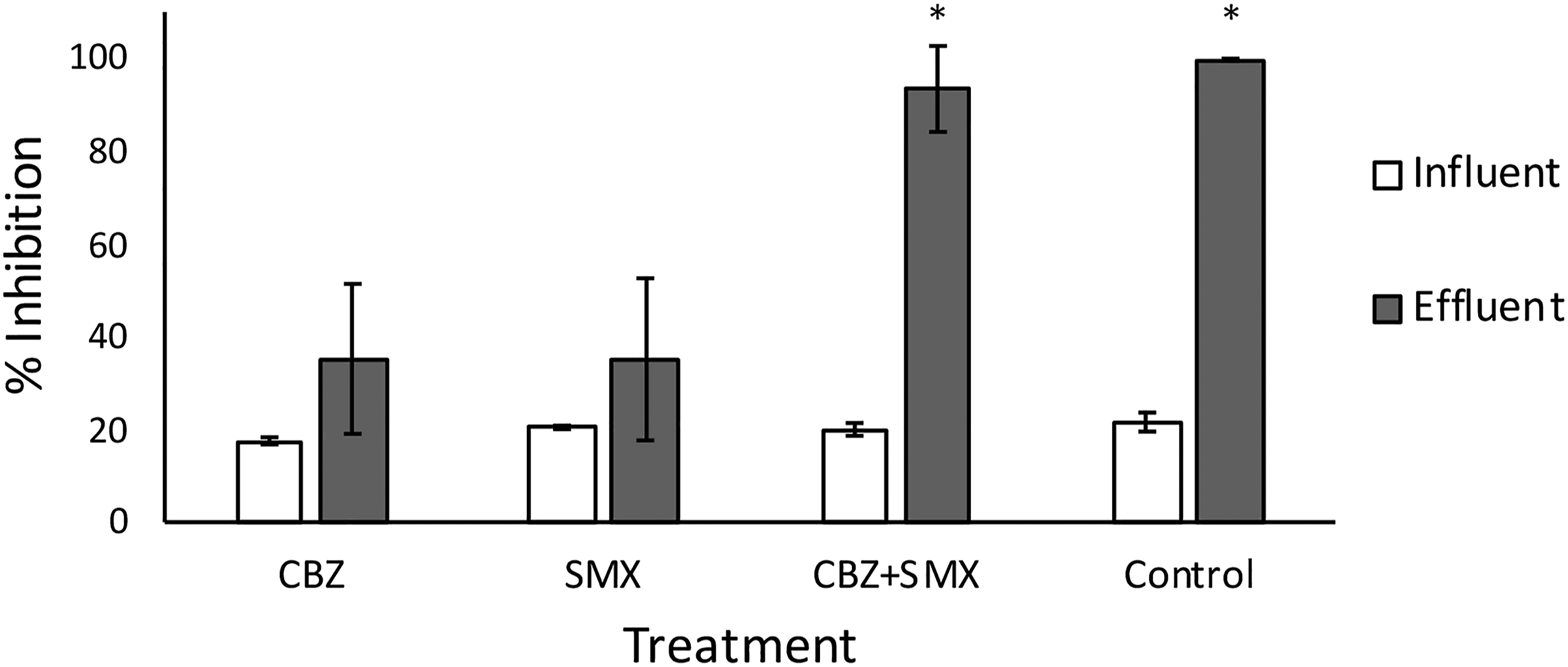
In terms of toxicity, the biofilm influent micropollutant concentrations, in isolation and in combination, do not show any estrogenic nor any anti-estrogenic effect (Figure 6), as expected due to the low influent concentrations mimicking environmental micropollutant concentrations. Similarly, the effluent CBZ and SMX biofilm samples show no change in endocrine disruption (Figure 6, p < .05), reflecting the low removal rates of each micropollutant in isolation (Figure 5). However, the transformation products in the effluent of the biofilm exposed to the combined micropollutants (CBZ+SMX) show a drastic increase in estrogenicity (p < .05). It is reported that biotransformation products by enzymatic degradation can maintain therapeutic activity, binding to proteins and other cellular components, causing disruption of cellular components. This can be ascribed to higher biological degradation in these biofilms (Figure 5). Some of the main metabolites of CBZ and SMX, carbamazepine-10,11-epoxide and 4-OH-sulfamethoxazole respectively, have been reported to be more potent than the parent compounds.77,78 The sterile control, fed with identical media to the combined biofilm, also shows an increase in estrogenicity, possibly due to estrogenic active products formed by association between CBZ and SMX over time, or due to transformation products from UV exposure over time. The Aliivibrio toxicity assay reflected the YES screen (Figure 7), supporting the presence of estrogenically active transformation products. We know from Figure 5 that co-amendment with micropollutants promotes removal of the parent compounds. Where removal rate was higher in the combined biofilms, the final estrogenicity was higher, supporting the idea that transformation products are more ecotoxic than parent compounds. However, the control increased in estrogenicity from influent to effluent, to a similar degree as the combined micropollutant biofilm. This is an unexpected phenomenon, reflected in both assays. One hypothesis is that this abiotic effect is due to physico-chemical transformation, such as UV or temperature gradients, which the mono-amended biofilms might prevent due to light inhibition and the buffering of a biofilm in terms of temperature homeostasis. In the mono-amended biofilms, there is a low removal rate, and no increase in ecotoxicity, suggesting that the less-active biofilms (in terms of micropollutant removal rate) limited both biotic and abiotic increases in ecotoxicity. Estrogenic effect of CBZ, SMX and a combination of CBZ+SMX relative to an EC50 E2 spike, before and after exposure to biofilms grown up on the respective micropollutant-enriched media. Biofilms were grown on M63* media, with the carbon source replaced by the respective micropollutants, or combinations thereof, and the estrogenic effect of the influent and effluent media measured with a yeast estrogen screen (* indicates statistically significant differences). Percentage Aliivibrio inhibition due to CBZ, SMX and a combination of CBZ+SMX before and after exposure to biofilms grown up on the respective micropollutant-enriched media. Biofilms were grown on M63* media, with the carbon source replaced by the respective micropollutants, or combinations thereof. The eco-toxicity of the influent and effluent media was measured with a bioluminescent bacterial assay (* indicates statistically significant differences).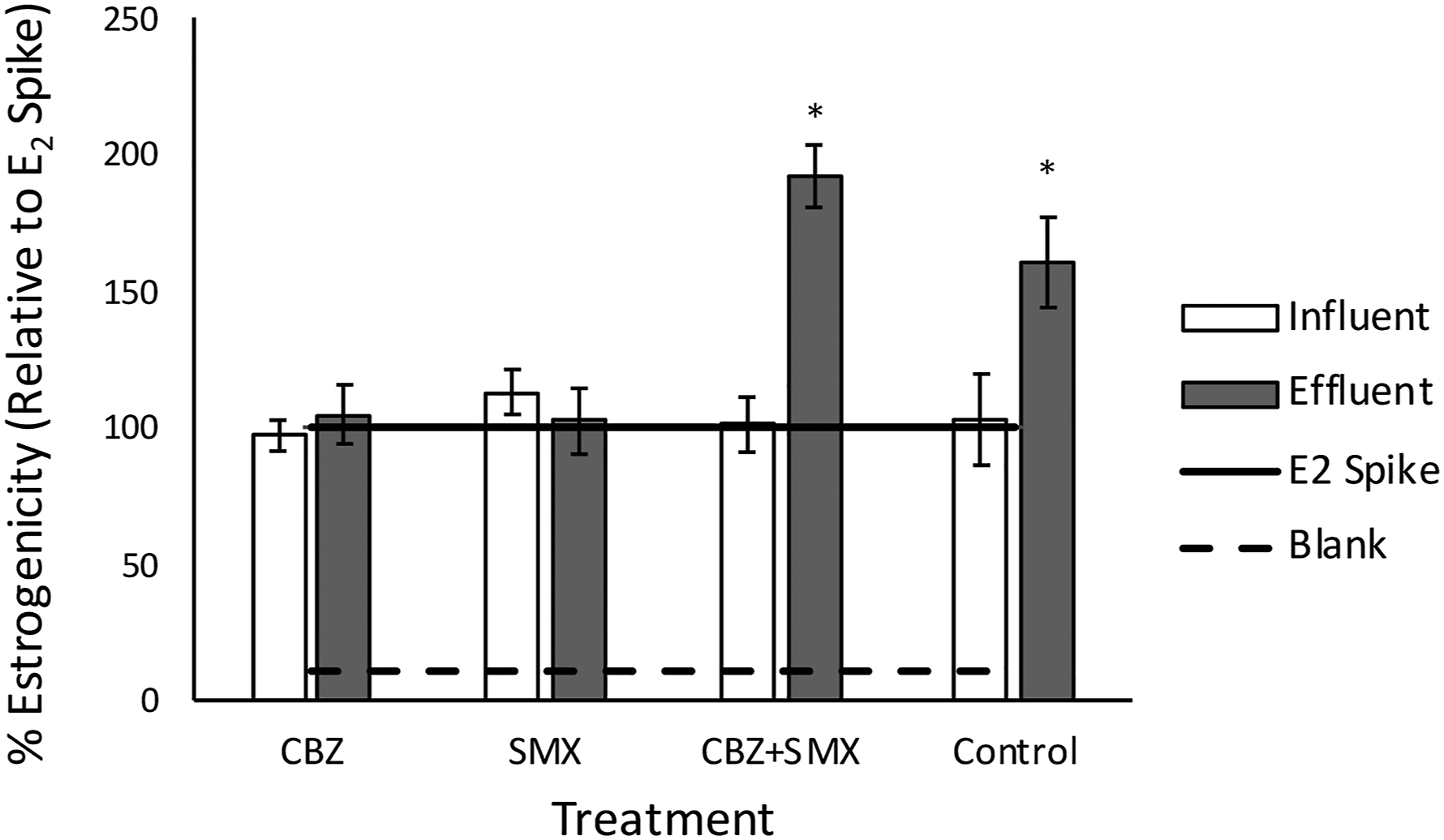
Electrochemical ozone micropollutant transformation of environmental samples, and toxicity of transformation products
Ozone concentrations after sample exposure to the electrochemical cells, measured in the CabECO system, were reduced as the organic levels of the environmental samples increased, from tap water to river water and shower greywater (Figure 8). The ozone concentration was reduced from 2.97 ± 0.17 mg/L in the Eerste river water (2.106 ± 0.099 mg/L TOC) to 0.25 ± 0.03 mg/L for the shower greywater (99.547 ± 2.777 mg/L TOC). All coefficient of correlation values fell between −0.3 and −0.7, indicating that ozone has a moderate inverse correlation with TIC, TC and TOC. These inverse correlations were not significant (p > .05), however, a clear trend can be observed in the inverse association of ozone and TIC, TC or TOC. The scavenging of ozone with increased TOC promotes the use of these technologies as a polishing step after wastewater treatment, when most organic matter has been removed. Total carbon (TC), total organic carbon (TOC) and total inorganic carbon (TIC) concentrations of untreated and CabECO treated environmental water samples, with increasing organic pollutants: Eerste river (pristine), Plankenbrug river (polluted) and greywater (shower). The secondary axis shows the aqueous ozone concentration of CabECO-treated water from the same environmental sources.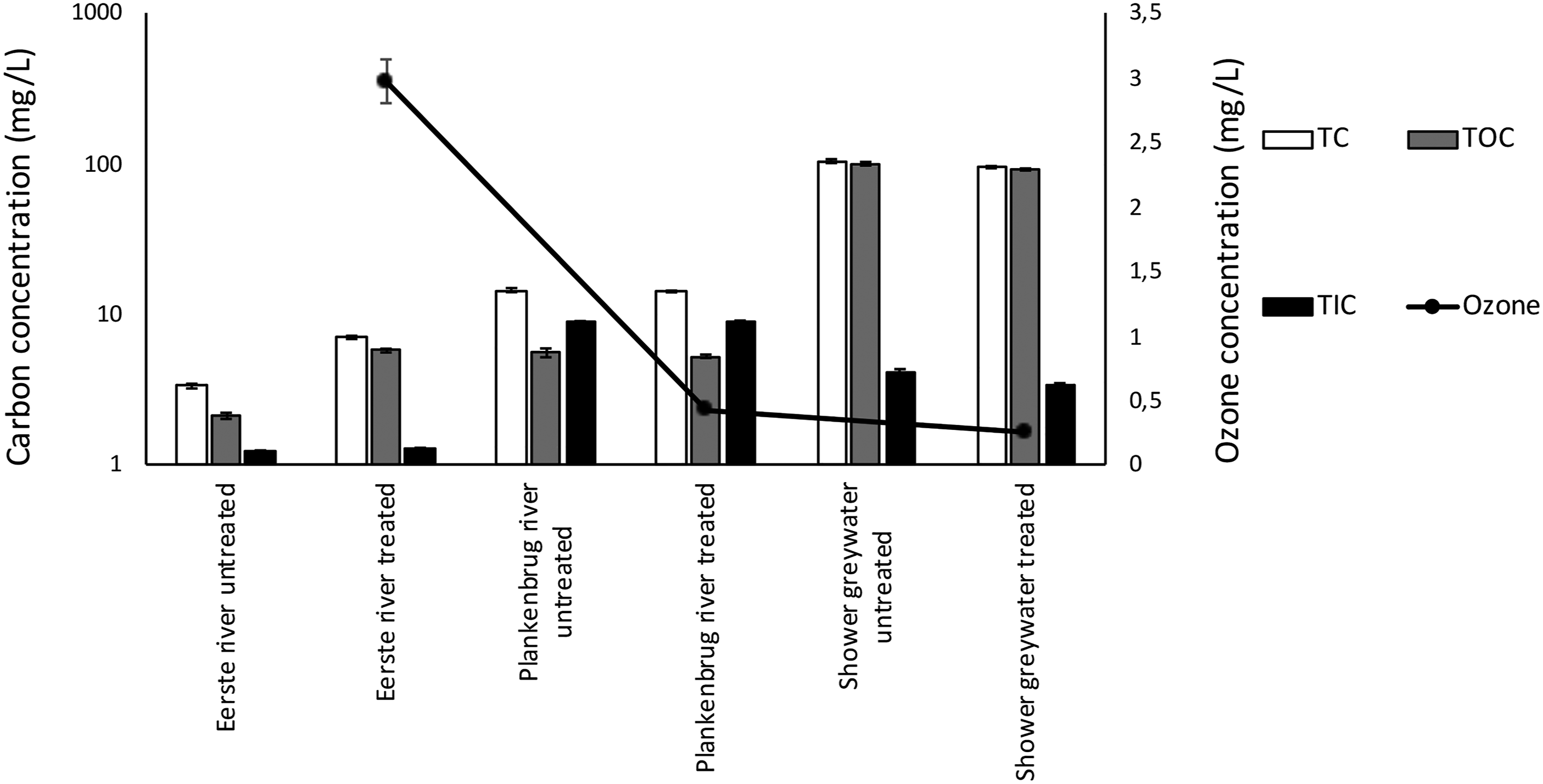
When testing the efficiency of electrochemical oxidation in the removal of a suite of micropollutants from environmental samples, the degradation of CBZ, ACM and SMX was most efficient, with removal rates upward of 90% in all three environmental matrices (Figure 9; p < .05; pristine and polluted rivers, greywater). However, in shower greywater, quantified as the most polluted (Figure 8, TOC), even the high removal efficiencies showed lower reproducibility. Less polluted samples evidenced lower variation (Figure 9), and the laboratory-based clean samples showed similar low variation (Figure 2). Other factors beyond carbon-based pollution, such as chemical pollutants and their metabolites and/or by-products, pose challenges to the evaluation of wastewater treatment modalities.
79
Electrochemical oxidation removal efficiency of a broad suite of selected micropollutants in environmental water samples. Water samples include the Eerste river (pristine), Plankenbrug river (polluted) and greywater (shower).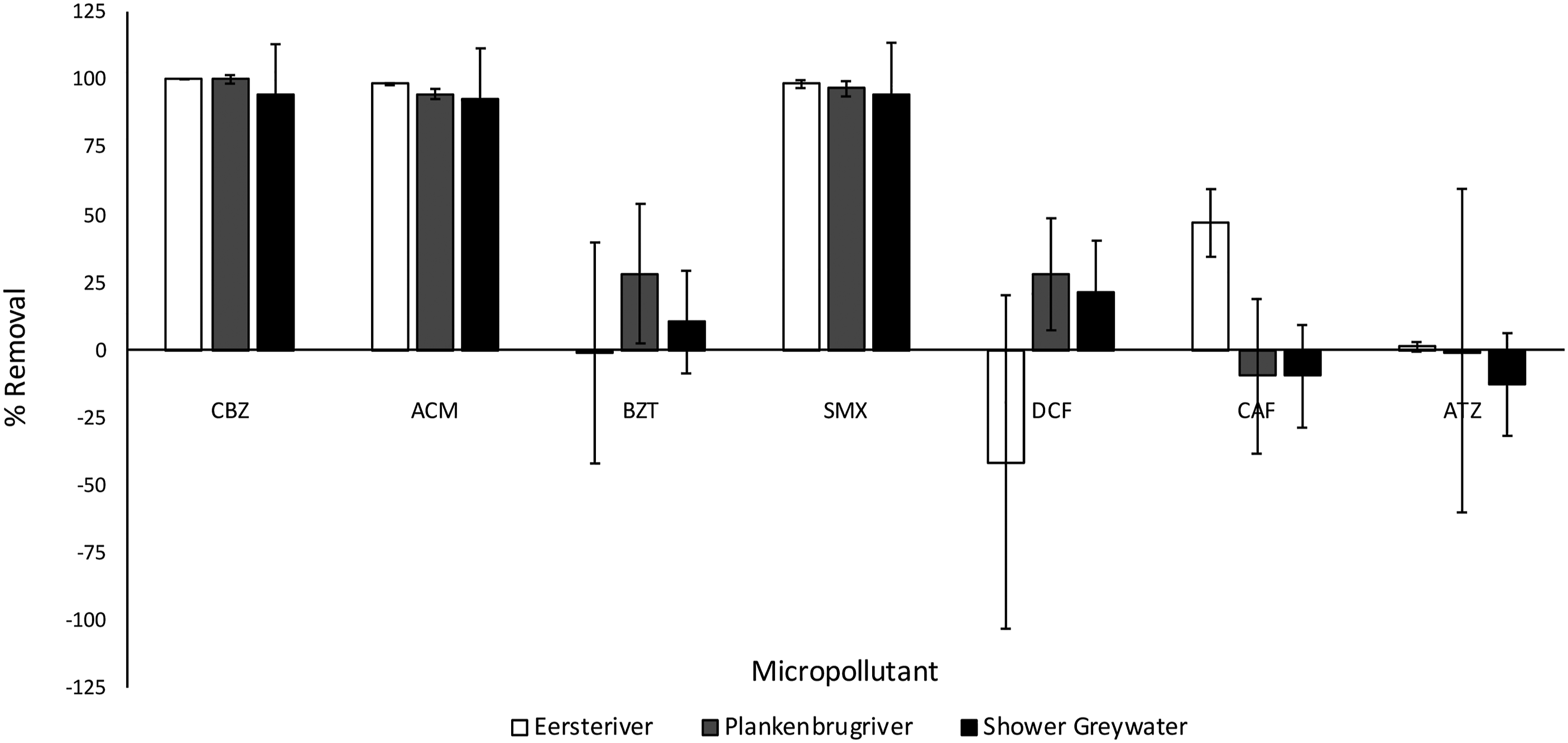
In contrast to the above-mentioned environmental pollutants, which were effectively removed by CabECO technology, BZT, DCF, CAF and ATZ were persistent during ozone exposure, with the highest removal percentages only reaching 30% and the lowest no removal (Figure 9). ATZ was the least sensitive to ozone, showing little to no removal. Hua et al. 80 also found ATZ to be a persistent compound during ozone treatment of river water in Canada.
The same samples analyzed for micropollutant removal (Figure 9) were evaluated for toxicity according to the YES screen and the Aliivibrio assay. Figure 10 shows relatively stable estrogenicity in the Plankenbrug river water upon electrochemical oxidation (one-tailed T-test, p > .05). In contrast, CabECO treatment resulted in a statistically significant decrease in estrogenicity in the Eerste river sample (one-tailed T-test, p < .05), suggesting that the eco-toxicological compounds in this sample are more easily degraded than the ones in the Plankenbrug river sample, likely due to the lower organic load in the Eerste river. Notably, the Eerste river sample has a higher pre-treatment estrogenicity than the Plankenbrug river sample, although Plankenbrug river is the more polluted river (Figure 8, TOC). Swart el al.
81
also reported high estrogenicity in the Eerste river, due to phyto- and myco-estrogens, possibly from surrounding agricultural practices. The estrogenic impact of herbicides and pesticides is well-demonstrated.
82
Toxicity effects of an Eerste river and Plankenbrug river sample at environmental micropollutant concentrations, relative to an EC50 E2 spike before and after CabECO treatment, measured with the YES assay (a) and the Aliivibrio assay (b). (* indicates statistically significant differences between treated and untreated samples).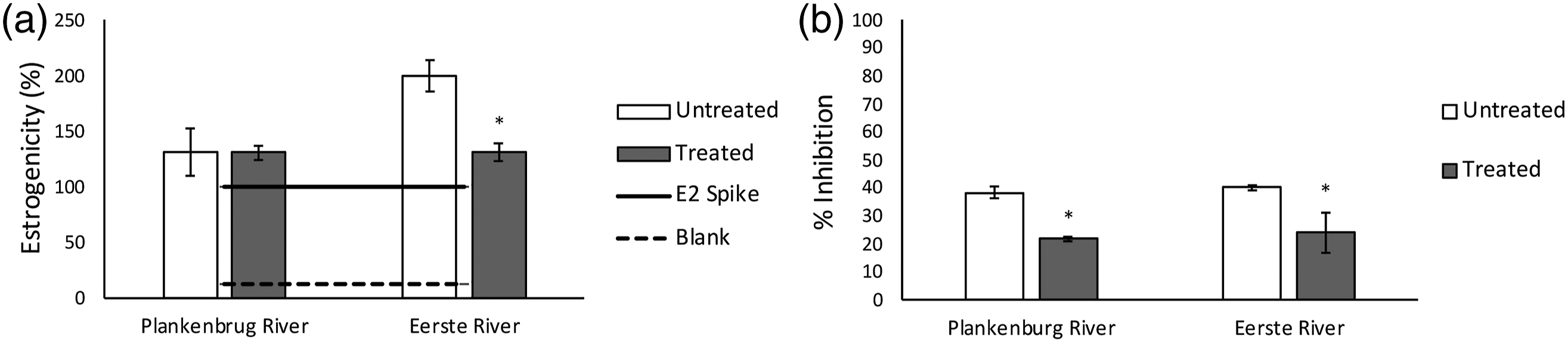
In contrast to the laboratory studies, where the YES trends were clearly reflected in the Aliivibrio toxicity assay (Figures 6 and 7), the environmental samples showed much greater variation. This is likely due to the complexity of the matrix. Environmental samples may contain naturally occurring estrogens as well, such as phyto-estrogens produced by plants and myco-estrogens from fungi, which may also contribute to the estrogenicity of the untreated sample (Kuiper, 1998). Toressi et al. 83 state that river biofilms have a high affinity to sorb organic micropollutants and from microbial disinfection trials using CabECO, 55 the conclusion can be made that biofilm structure and flocs are disturbed by the CabECO process. This is further supported by the increase in TOC in the Eerste river samples after CabECO treatment, as organic complexes are disturbed by the process of electrochemical oxidation. This opens the possibility that some compounds could have been released from bound organic complexes during ozone exposure, contributing to the estrogenicity of the sample. Similarly, it has been found that the matrix composition of the water being treated with ozone influences the transformation products formed after electrochemical oxidation, resulting in conflicting and variable toxicity results towards Daphnia magna. 24 Additionally, Dopp et al. 84 report variable toxicity results in estrogenicity, cytotoxicity, mutagenicity and genotoxicity assays when treating wastewater effluent with ozone, from three different municipal WWTW in Germany. However, toxicity assays show more reproducible and comparable results in controlled laboratory conditions with simple and pre-treated water matrices, than with complex water matrices, when treating with ozone.86,87, 85
Practical implications of this study
This study highlights the challenges associated with micropollutant degradation studies at environmental concentrations and with complex matrices. Excess non-target organic matter in environmental samples scavenge oxidizing agents during oxidation processes, resulting in less effective removal of micropollutants. Similarly, the metabolic redundancy of microbial communities leads to biofilms rather utilizing more labile carbon sources than recalcitrant micropollutants. Therefore, it is suggested that future studies design hypotheses envisioning electrochemical oxidation and microbial degradation as polishing steps in wastewater treatment, once most organic matter has been removed by the conventional treatment processes.
Conclusions
Electrochemical oxidation, as applied in the CabECO system, was shown to be effective in the reduction of two representative micropollutants (CBZ and SMX), with a >96% reduction within 1 min of treatment. However, the endocrine disrupting potential of these compounds required significantly longer treatment times (240 min) to reach a negligible effect, motivating the implementation of a retention system when using ozonation for micropollutant reduction. Microbial metabolism as driven by nutrient limited, or ‘hungry’, biofilms showed significantly lower removal efficiencies of CBZ and SMX compared to electrochemical oxidation, in contrast to planktonic cultures that showed zero micropollutant removal efficiency. These hungry biofilms were characterized by regular sloughing of planktonic cells, rather than the typical sigmoidal biofilm growth curve. The addition of labile carbon sources for co-metabolism might enhance the removal efficiency of biological degradation of micropollutants. However, where CBZ and SMX were co-metabolized by the biofilms, there was a significant increase in both toxicity assays, evaluating estrogenic activity as well as Aliivibrio inhibition. In contrast to controlled laboratory-based micropollutant treatment with electrochemical oxidation, environmental samples, analyzed for a larger suite of micropollutants, showed variable results. Some compounds were almost completely removed (CBZ, SMX, and ACM), while others (BZT, DCF, CAF and ATZ) proved more recalcitrant. Environmental toxicity results post electrochemical oxidation also showed variance between the YES and Aliivibrio toxicity assays, which could be attributed to the complexity of environmental water matrices, non-target oxidant scavengers and unpredictable formation of transformation products.
In contrast to targeted analysis of a pre-selected and analytically feasible set of compounds, an array of toxicity assays with different endpoints provides a more holistic representation of the full eco-toxicological footprint of treated water. Monitoring of only parent micropollutant removal efficiencies are not sufficiently representative safety assessments, as evidenced by the unpredictable formation of active transformation products by electrochemical oxidation and microbial metabolism. Furthermore, the results obtained from this study, as well as supporting literature, highlighted the complexity of the presence, treatment and effect of micropollutants in the natural environment.
Footnotes
Acknowledgments
The authors would like to acknowledge Dr Edward Archer for generous guidance and expertise in micropollutant analysis. Prof Marietjie Stander and Mr Erick van Schalkwyk (Mass Spectrometry Unit, Central Analytical Facility, Stellenbosch University) provided valuable assistance in method development. The authors would like to thank Dr Janine Colling (BIOGRIP SU Node) for assistance with measuring TOC. The authors would also like to thank Mercia Volschenk at the City of Cape Town, Scientific Services, for assistance with the Microtox assays.
Author contributions
JHLB, first author, was responsible for experimental design, execution, data analysis and primary authorship. WS contributed to experimental design, writing, data analysis and editing. AC contributed to experimental execution, and editing. GMW holds last authorship and was host, funder and supervisor, involved in concept design.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: JHLB and WS, as well as research running costs, were funded by the European Commission, via Horizon2020 (SafeWaterAfrica, grant number 689,925). WS was also funded by Biogeochemistry Research Infrastructure Platform (BIOGRIP), Stellenbosch University Soil and Water Node, funded by the Department of Science and Innovation of South Africa.
Ethical approval
(include full name of committee approving the research and if available mention reference number of that approval) N/A. No human, clinical or animal trials.
Informed consent
N/A. No human, clinical or animal trials.
Trial registration
(where applicable) N/A. No human, clinical or animal trials.
