Abstract
Chemical-induced pulmonary carcinogenesis in humans is usually a multi-decade process. The average age at diagnosis of lung cancer in cigarette smokers, and mesothelioma in asbestos workers, is approximately 70. The carcinogenic process consists of genetic changes to normal cells usually sequenced as initiation (mutations), promotion (clonal expansion of initiated cells), and progression (carcinoma in situ to invasive carcinoma to metastatic carcinoma). Angiogenesis, the sprouting of new vessels from preexisting capillaries, plays an important role in the progression of small avascular tumors to vascularized invasive carcinomas and metastatic carcinomas. While the overall carcinogenic process is multi-decade, the transition from avascular to vascularized carcinoma is believed to take place over the course of a much more limited time span. Tumor progression is a late-stage event in carcinogenesis. About 70% of human cancers including lung cancer express hypoxia-inducible factor (HIF)-1 in the absence of cobalt exposure. Cobalt compounds administered to transformed cell lines and primary cultures of human endothelial cells, smooth muscle cells, and mesenchymal stem cells can elicit overexpression of HIF-1. Cobalt-induced expression of HIF-1 would not be expected to interact with either the avascular initiation or promotion phases of carcinoma development. Humans exposed long-term to cobalt leaching from implants do not have an elevated cancer risk, and neither do goats ingesting up to 667 times the recommended daily human cobalt dietary requirement throughout their 15- to 18-year life span. In vitro hypoxia-mimetic characteristics of cobalt compounds do not correlate with the absence of increased risk following long-term systemic exposure to cobalt in humans.
Executive summary
Many thousands of patients with metal-on-metal hip implants that have begun to shed cobalt into the systemic circulation have not developed excess cancers over a time period of decades. Despite regularly ingesting a cobalt level up to 667 times the recommended intake for humans, over their life span of 15–18 years goats display low cancer rates overall and do not develop tumors suggestive of exposure to a carcinogen. National Toxicology Program (NTP) rodent 2-year bioassays suffer from high false-positive rates. Cobalt compounds administered via inhalation in an NTP 2-year rodent bioassay induce pulmonary tumors in rats. The well-established mechanism of reparative mitogenesis inducing mutagenesis described by Ames and Gold, Cohen and Ellwein, Moolgavkar, and Tomasetti and Vogelstein was not considered by the NTP as an underlying mechanism for the development of the rat tumors, but NTP instead posited that cobalt exposure was inducing hypoxia-inducible factor (HIF) thereby facilitating angiogenesis. Rodent pulmonary tumors differ from human lung cancers in that the human tumors tend to be highly vascularized thereby possessing metastatic potential. In contrast, rodent pulmonary tumors tend to be localized and less vascularized. Inhalation of cobalt compounds in rats and mice does not model the late final progression stage of human pulmonary carcinogenesis but rather attempts to model the earlier stages of tumor initiation and promotion not affected by the overexpression of HIF. The mouse two-stage dermal promotion assay provides a framework for conceptualizing the stages of cancer development as initiation (mutation), promotion (clonal expansion of initiated cells), and progression (angiogenesis, localized invasion, and metastasis). Angiogenesis is a late change in tumor development usually occurring decades after the beginning of exposure to a carcinogenic agent or action. Tumors usually do not undergo angiogenesis until exceeding a diameter of 1–2 mm3. Pulmonary inflammation produces a hypoxic local environment. High doses of inhaled cobalt compounds and many other pro-inflammatory agents induce pulmonary inflammation in rodents. Over 70% of all human cancers are HIF positive in the absence of exposure to cobalt compounds. In vitro, cobalt induces the expression of HIF in both transformed and normal cell lines. The majority of in vitro demonstrations of cobalt-induced HIF expression is reported in transformed cell lines. Transformed cell lines have already experienced one or more oncogenic mutations and are not analogous to normal cell lines. The mechanism by which cobalt-induced overexpression of HIF-1 would be expected to influence cancer would be an increased tendency toward angiogenesis thereby accelerating the later progression stages of the disease. Evidence suggesting that this is actually occurring is absent. Therefore, the hypoxia-mimetic characteristics of cobalt compounds seen in in vitro cell culture studies do not correlate with the risk of exposure to cobalt compounds.
Very large numbers of patients with cobalt-shedding hip implants have not developed cancer
The epidemiological associations between occupational exposure to cobalt and cobalt compounds and risk of lung cancer are potentially confounded and unclear leading the National Toxicology Program (NTP) to consider the evidence from human studies to be “inadequate.” 1 –5 In industrial environments, the primary route of exposure to cobalt is via inhalation. 6 –9
Cobalt is rarely the only agent of potential concern found in workplace air in the facilities studied. 6,7 In addition, it is very difficult in these occupational cohorts to control for the potential contribution to risk from by far the most important pulmonary carcinogen, that is, cigarette smoking. Not only does smoking account for 80–90% of lung cancer cases, 10 but the duration of smoking is more important than the intensity of smoking in terms of lung cancer risk. 11 Therefore, an accurate determination of both current and past smoking history would be essential toward disentangling a possible effect of smoking from cobalt or other inhalation exposures. A sufficient evaluation of current smoking status and smoking history is not available for the occupational cohorts exposed to cobalt compounds via inhalation. 6 –9
In contrast with occupational cohorts with exposures to cobalt confounded by other inhaled agents and indeterminate smoking histories, cobalt exposures from aging medical devices occur in the absence of co-exposure to agents other than chromium. Also, exposure to cobalt from aging medical devices has been occurring for decades. 12 More than one million metal-on-metal (MOM) prosthetic hips have been surgically implanted worldwide. 12 Several studies have demonstrated that as the surfaces of these devices wear a significant shedding into the general circulation of cobalt and chromium ions occurs. 13 A number of studies on large numbers of patients have not found an increase in cancer incidence in association with these aging implants. 14 –20 Steinberg et al. 13 studied epigenetic methylation changes in 34 osteoarthritis patients with MOM total hip replacements and 34 osteoarthritis patients with non-MOM total hip replacements and found no difference between these small groups. The results from this pilot study are consistent with the absence of cancer risk in MOM patients.
Very high lifetime (15–18 years) of ingestion of cobalt does not correlate with cancer rates in goats supporting the absence of tumors in MOM patients
Adjusted for body weight, goats have the highest mineral requirements of all ruminants. In proportion to body size, the goat’s metabolic rate is higher, and the surface area and capacity of the rumen is larger in comparison to the cow, sheep, or horse. 21 Domestic goats are at greater risk for parasitical infestation than wild goats probably because the freedom of wild goats allows them to not browse the same area twice. 21 Parasite burdens are associated with anemia in goats thereby increasing the need for cobalt to facilitate hemoglobin formation. Cobalt is directly involved in vitamin B12 formation by bacteria in the stomachs of ruminants. Sheep require more cobalt than cows. Goats have four times the cobalt requirement as do sheep. Goats have a dietary requirement for cobalt of 0.1–10 ppm per day. 22 In all animals, cobalt displays relatively low toxicity. Cobalt toxicity in ruminants is rare because toxic levels are approximately 300 times dietary requirement levels. On average, ruminants have 10 times the amount of cobalt in their system as compared with dogs, cats, rats, or man. The recommended level in humans is 0.015 ppm. 23 The high end of the recommended level for goats (10 ppm) is 667 times the recommended level for humans.
Despite a relatively high intake of cobalt, goats do not show an increase in cancer incidence compared with other animals. 24 Across mammalian species, the risk of cancer depends on both the number of cells in the body and number of years over which those cells can accumulate mutations. Cancer incidence can be calculated as a function of Mass × Life Span. The goat has a log (Mass × Life Span) = 5.4. When necropsied for tumor incidence, about 1% of goat necropsies present with a tumor. According to Abegglen et al., 24 this places the goat relatively close to the raccoon as regards the tendency toward cancer formation.
Oregon State University has studied the distribution of tumor sites in the 1% of goats that do develop tumors. At their Veterinary Diagnostic Laboratory, 1146 goat (caprine) necropsy or biopsy specimens were submitted from 1987 to 2011. Of the 1146 specimens, 100 goats (8.7%) presented with 102 tumors. 25 Histopathology was performed on all but three of the tumors, that is, 97 tumors. The most common tumor was lymphoma (n = 17) with most of the lymphomas being multicentric. 26 There were 10 cases of cutaneous squamous cell carcinoma and 9 cases of thymoma. Seven mammary neoplasms were all classified as adenocarcinomas. Seven vascular proliferations were reported with five being hemangiosarcomas. Four malignant melanomas were seen. These six tumor types constituted 55.7% of the total cancer cases in goats.
A number of rare tumors were also found among the 97 tumors in goats including the following: one choroid plexus carcinoma, two rhabdomyosarcomas, three pheochromocytomas, two histiocytomas, five mast cell tumors, one amyloid-producing odontogenic tumor, one myxosarcoma, one sebaceous carcinoma, one apocrine sweat gland adenoma, and one thyroid carcinoma. The results from this 25-year retrospective study conducted by Oregon State University demonstrate that while lymphoma and other tumors do occur in goats, the rate of tumor formation is low, and when a goat does develop a tumor, it tends to be only a single tumor. 25 The causation of lymphoma in goats is believed to have a genetic basis with a possible contribution from eating pesticides. 27 The squamous cell carcinomas in goats occur in body parts not well protected from sunlight and are consistent with the large amount of sun exposure goats receive over their life span of 15–18 years. 28,29 Despite regularly ingesting a cobalt level up to 667 times the recommended intake for humans, goats display low cancer rates overall and do not develop tumors suggestive of exposure to a carcinogen via the oral route. 30
NTP rodent 2-year bioassays suffer from high false-positive rates
Our group conducted a mechanistic and statistical analysis of the entire NTP rodent 2-year bioassay database up through 2017.
30
–36
In the 1980s, Cohen, Ellwein, and their colleagues conducted a series of studies that demonstrated that cellular proliferation could amplify the background mutation rate. This mutational amplification increased tumor formation in experimental animals.
37
–40
Studies conducted by Moolgavkar and Knudson also supported an important role for cellular proliferation during this era.
41
Throughout the 1990s, Ames and Gold incorporated these new findings on proliferation-induced amplification of background mutation rate into their thinking resulting in a series of publications, one of which we highlighted in Smith and Perfetti,
35
that is, Ames and Gold.
42
Published in Science, Ames and Gold,
42
was actually a commentary on the Cohen and Ellwein paper published in the same issue.
38
In 2017, Tomasetti and Vogelstein provided evidence for the clinical relevance in humans of the Ames and Gold mechanism regarding the amplification of the background mutation rate.
43
These authors sought to determine the relative contribution to human cancers from inherited mutations, mutations induced by environmental factors, or mutations resulting from DNA replication errors (R). They compared the number of normal stem cell divisions with the risk of 17 cancer types occurring in 69 different countries. Tomasetti and Vogelstein
43
reported the following results: The data revealed a strong correlation (median ¼ 0.80) between cancer incidence and normal stem cell divisions in all countries, regardless of their environment. The major role of R mutations in cancer etiology was supported by an independent approach, based solely on cancer genome sequencing and epidemiological data, which suggested that R mutations are responsible for two-thirds of the mutations in human cancers.
Stages of malignant tumor development
There are several separate but interrelated conceptual frameworks for ideating the life cycle of cancer from its origins as a single normal cell to its final stage as a large mass of billions of locally invasive or metastatic cells within a malignant tumor. One of the most useful conceptual frameworks has been the two-stage mouse dermal promotion assay from which the terms initiation, promotion, and progression derive. 45,46 The eponymous term “initiation” refers to the first step in the mouse dermal promotion assay. 47 A parent chemical that reacts with DNA can serve as an initiator, although metabolic enzymes are frequently required to convert a nonreactive parent chemical into an electrophilic mutagen. 48 Different tissues and different species can possess significantly different levels of metabolic enzyme activity. This differential in metabolic potential confers tissue and species specificity on many initiators. 49
It is important to understand that the effects of initiators are irreversible. After the DNA in a particular cell has been altered by a chemical initiating agent, that cell is susceptible to promotion until its death. Daughter cells produced from the division of the mutated cell will also carry the mutation. 48 A linear relationship has been observed between the dose of initiator and the quantity of tumors produced in studies of mouse skin carcinogenesis. Therefore, any exposure to an initiator increases cancer risk and this risk increases with higher levels of exposure. 49
Promotion refers to the process of enhancing cellular proliferation. 50 Cells mutated by an initiator are susceptible to the effects of promoters. Promotion gives rise to a large number of daughter cells containing the original mutation induced by the initiating chemical. 51 Promoters do not affect cells not previously treated with an initiator. 49 Unlike initiators, promoters do not covalently bind to DNA or cellular macromolecules. Promoters can be categorized as specific or nonspecific. Specific promoters interact with receptors on or in target cells of defined tissues. Nonspecific promoters alter gene expression without binding to a known receptor. 48 Tissue and species specificity results from the differential distribution of receptors on various tissue types. The effect on tumor growth of promoter application is dose-dependent. However, promotion displays both threshold and ceiling effects with both a minimum dose required and a maximum dose beyond which no further effects are seen. 49
Progression refers to the stepwise transformation of a benign tumor to a malignant tumor. Repeated application of a promoting agent onto previously initiator-treated mouse skin can produce benign papillomas. After cessation of treatment with promoter, most of these papillomas regress but some progress to cancer. 52 Benign papillomas that progress to cancer have acquired an additional, spontaneous mutation. 53 Progression is associated with karyotypic change as virtually all benign tumors that advance to malignant tumors become aneuploid (incorrect number of chromosomes). Karyotypic change is associated with increased growth rate, invasiveness, metastatic potential, and biochemical and morphological changes. 49
All of the major human carcinomas pass through histological stages of hyperplasia, dysplasia, carcinoma in situ, and invasive carcinoma. 54 Cancers of the central nervous system, bone, blood, and lymph system do not use the same terminology due to differences in pathology. In humans, molecular stages analogous to tumor initiation, promotion, and progression are concomitant with but variably overlapping with the sequential histological stages of carcinoma development, including hyperplasia (increase in cell number), dysplasia (precancerous stage), carcinoma in situ (not yet invasive or noninvasive carcinoma), invasive carcinoma, and metastatic carcinoma.
Angiogenesis is a late change in tumor development
Tumors can grow to approximately 1–2 mm3 before metabolic demands restrict growth due to the diffusion limit of oxygen and nutrients. Growth beyond this size requires the tumor to switch to an angiogenic phenotype. 55 Angiogenesis is a multistep process by which new blood vessels sprout from preexisting capillaries. During angiogenesis, activated endothelial cells proliferate and migrate to specific target locations and assemble into new capillary tubes. 56 A vascular lumen is constructed by the synthesis of a new basement membrane and maturation of vessels. Vasculogenesis differs from angiogenesis as it involves de novo differentiation of endothelial cells from in situ mesoderm-derived precursor cells. The principal process in angiogenesis is sprouting from preexisting blood vessels. Under physiological and pathological conditions, a secondary contribution to angiogenesis includes recruitment and differentiation in situ of bone marrow–derived endothelial progenitor cells. 57 Angiogenesis plays a crucial role in several normal and pathological processes including embryogenesis, the female reproductive cycle, tissue repair, wound healing, cardiac ischemia, limb ischemia, neovascularization associated with diabetic retinopathy, rheumatoid arthritis, and neoplasms. 58 Angiogenic and antiangiogenic factors are secreted by both tumor cells and host cells infiltrating tumors and interact to influence the growth and progression of cancer. 56 As tumors grow, they need additional oxygen and nutrients. In the absence of a blood supply from angiogenesis, the growing tumor can become latent or its cells can die. 59 A number of angiogenic growth factors including angiogenin, angiopoietin-1, cyclo-oxygenase, hepatocyte growth factor, and tumor growth factor are produced by tumor cells or host cells. In contrast with the angiogenic growth factors, antiangiogenic factors including angiopoietin-2, angiostatin, interferon-α, interferon-β, endostatin, and vasostatin hold the existing blood vessels in a quiescent state. 60 Hypoxia regulates the vascularity of the tumor via different cells in the tumor microenvironment including vascular endothelial cells, pericytes, and bone marrow precursor cells. 61,62
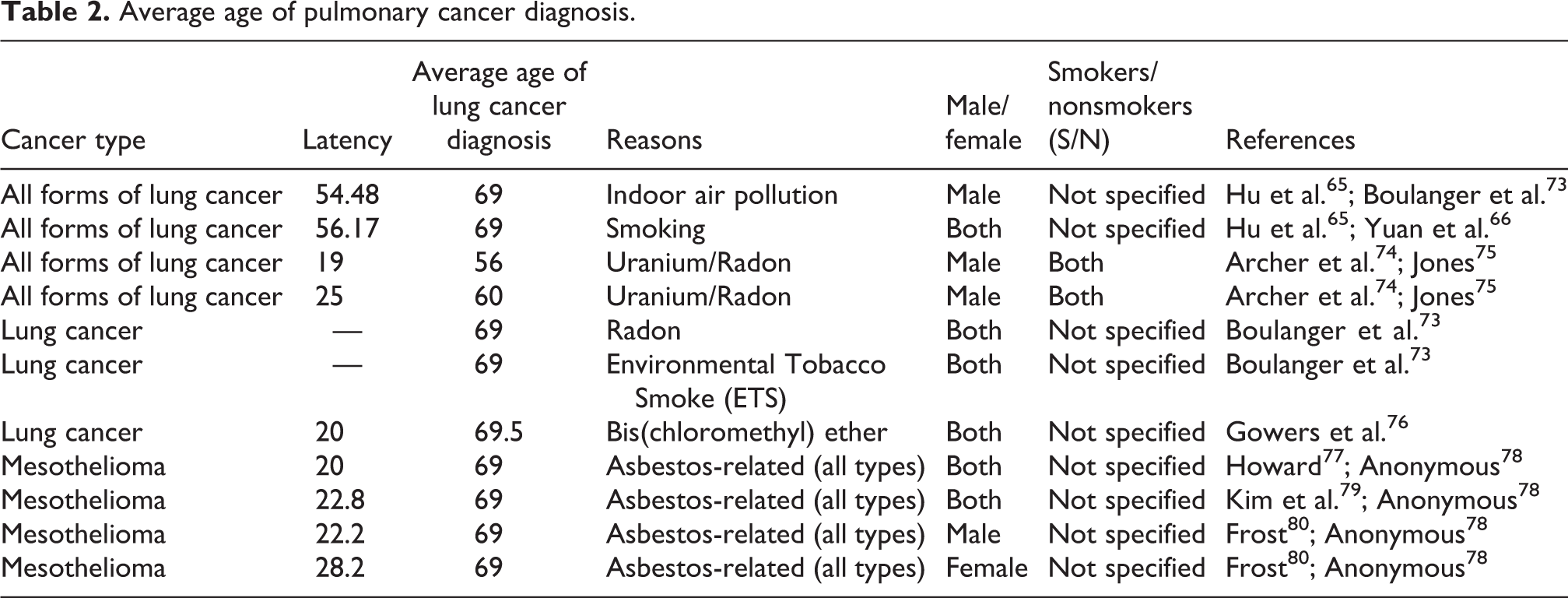
In the life cycle of a malignant carcinoma, angiogenesis is a relatively late event that frequently occurs decades following the initial exposure to mutagens. 63 Table 1 presents evidence that small previously avascular tumors enter a rapid growth phase following vascularization via angiogenesis. In contrast with the decades long time period of the overall carcinogenic process, this final phase of tumor progression appears to occur on a much more limited timeframe of a few to several years (Table 1). The extremely long lag phase between exposure to different chemical carcinogens and the clinical presentation of cancer (Table 2) supports the late introduction of the angiogenic process and its attendant exponential increase in tumor growth. 81 For example, the average age of presentation for asbestos-related mesothelioma is 69 years, 82 and the average age for lung cancer presentation in cigarette smokers is approximately 70 (Table 2). 83
Relative brevity of vascularized progression phase of squamous cell lung cancer resulting in treatment failure.

aCigarette smoking usually initiates in late teens to early 20s. Occupational exposures usually begin following graduation from high school and into the early to mid-20s.
bLung cancer diagnosis is rare prior to age 40. The average age at clinical presentation is approximately 70, meaning that half of all cases occur before age 70 and half after age 70.
cIt takes approximately 8 years for a single squamous cell carcinoma to expand to approximately 30 mm. The growth rate of cancer cells is exponential. The natural history of malignant nodules as small as 5 mm detected on computed tomography scans is not well described and even nodules only 10 mm in size containing 109 cells represent a fairly late stage in the disease process, considering that at death lung cancer tumors typically have 1012 cells (Birring and Peake). 64
dThe time from malignant transformation of a single cell to the formation of a small avascular or poorly vascularized tumor 10–30 mm is not well understood but the growth rate is believed to be relatively slow (approximately 8 years) prior to vascularization via angiogenesis.
eThis vascularized tumor is in its exponential growth phase.
Average age of pulmonary cancer diagnosis.
Inflammation is associated with hypoxia
Pulmonary inflammation can result from a wide variety of agents and actions causing injury to the cells lining the airways, including inhalation of a wide variety of chemicals, including irritants, cytotoxic compounds, and certain metals and metallic complexes. 31 The vast majority of neutrophils and macrophages found in pulmonary inflammation are not normally resident in the lung, but rather are recruited to inflammatory lesions. 84 One of the metabolic changes associated with active inflammation is development of hypoxia with concomitant accumulation of lactic acid and sometimes metabolic acidosis depending on the amount of lactic acid and the degree of buffering in the tissue environment. 85 –87 A number of factors can contribute to inflammation-induced tissue hypoxia including increased metabolic demands of cells and reductions in metabolic substrates caused by thrombosis (blood clots), trauma, compression (interstitial hypertension), or atelectasis (airway plugging). 88
Hypoxia due to inflammation is a stressor that can preferentially select for expansion of clonal populations of cells with particular mutations, for example, preferential survival of epidermal growth factor receptor (EGFR) positive clones. 89 In hypoxic mice with lung tumors induced by urethane, significant overexpression of EGFR, fibroblast growth factor receptor 2, and platelet-derived growth factor receptor were seen. 89 Similarly, using the HCC827 NSCLC cell line, Lu et al. 90 provided evidence that hypoxia/hypoxia-inducible factor (HIF)-2α activation mediates upregulation of EGFR protein levels. This observation provides a potential non-mutational explanation for the EGFR overexpression often seen in human adenocarcinomas. The authors hypothesized that “The data presented in this contribution also introduce the intriguing possibility that the tumor microenvironment may act as a universal oncogenic trigger that drives the autonomous growth of tumor cells.”
Human epidermal growth factor (EGF) is a 6 kilodalton protein and cytokine 91,92 comprised of 53 amino acid residues and containing three intramolecular disulfide bonds. 93 EGFR is a protein found on the surface of some cells and to which EGF binds. Cell division is stimulated following the binding of EGF to EGFR. EGFR is found at abnormally high levels on the surface of many types of cancer cells, so these cells may divide excessively in the presence of EGF. Alternate names for EGFR are ErbB1 and HER1. 94
Non-small cell lung cancers (NSCLs) are a group of lung cancers so named for either the normal cell of origin or the microscopic appearance of the tumor cells. The three main types of NSCL are squamous cell carcinoma, large cell carcinoma, and adenocarcinoma. NSCL is the most common kind of lung cancer. 95 Approximately 10% of patients with NSCLC in the United States and 35% in East Asia have a mutation in the EGFR gene in the DNA of their lung tumor. 96 –98 In both the United States and East Asia, EGFR mutations are more common in tumors from female never smokers (<100 cigarettes in patient’s lifetime) with adenocarcinoma histology. 96 –98 The percentage of EGFR mutation frequency rises to 60–65% in female East Asian never-smoker adenocarcinoma patients. 99 However, EGFR mutations are sometimes seen in squamous cell carcinoma and large cell carcinoma in both former and current smokers. 100
The EGFR mutations occur with exons 18–21. (Both the DNA sequence within a gene and the corresponding sequence in RNA transcripts are termed exons. 101 ) EGFR exons 18–21 encode a portion of the EGFR kinase domain. EGFR mutations are usually heterozygous and display gene amplification, that is, increase in number of copies. 102 The overwhelming majority, that is, approximately 90% of EGFR mutations, are deletions in exon 19 or point mutations in exon 21 L858R. 103 These mutations increase the kinase activity of EGFR. Kinase-induced phosphorylation activates signaling pathways that block apoptosis. 104 In the vast majority of cases, EGFR mutations seen in NSCLC do not overlap with KRAS mutations observed in the same tumors, 100 although KRAS mutations are found in 25–35% of newly diagnosed non-small cell, non-squamous cell patients. 99
Overexpression of HIF-1α and HIF-2α is common in human cancers
HIF-1 is a dimeric protein complex that plays an important role in the body’s response to hypoxia. HIF-1 is a transcription factor for dozens of target genes. The best-known function of HIF-1 is increasing vascularization in hypoxic tissues caused by ischemia or tumors. 105 Other functions of HIF-1 include immunological responses, physiological regulation of homeostasis, vascularization, and anaerobic metabolism. Due to its angiogenic properties, HIF-1 facilitates the survival and proliferation of cancer cells. 105
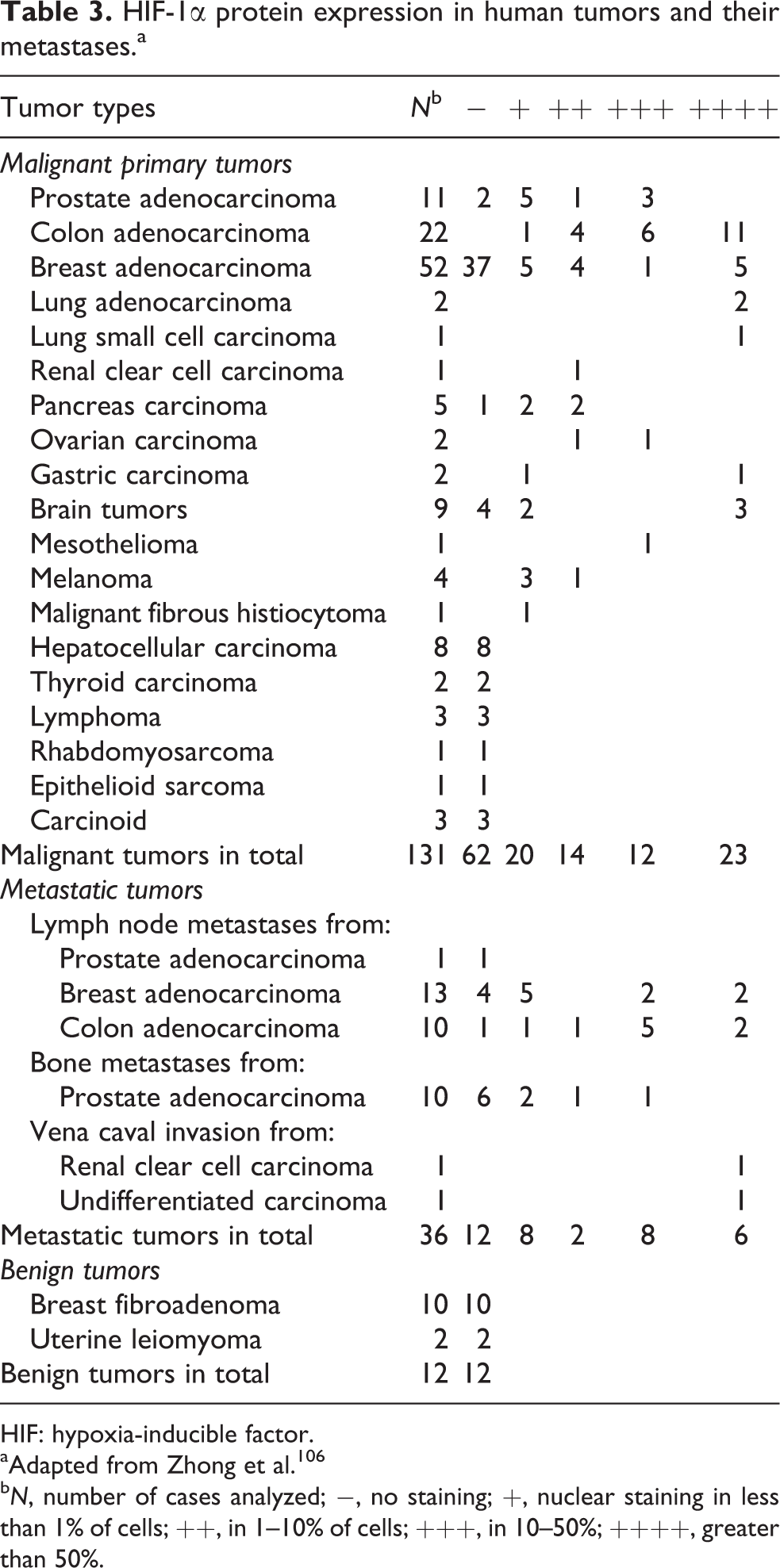
In well-developed human tumors, expression of HIF-1α and HIF-2α is common (Table 3). Over 70% of all human cancers are HIF positive. 107,108 A majority of solid tumors, including bladder, brain, breast, colon, ovarian, pancreatic, prostate, and renal carcinomas, 109 reported nuclear expression of HIF-1α and HIF-2α in subsets of the different tumor cells. As compared with the relevant normal tissues, HIF-1α was overexpressed in 13 of 19 histological tumor types including colon, breast, gastric, lung, skin, ovarian, pancreatic, prostate, and renal carcinomas. Abnormal p53 accumulation and cellular proliferation was associated with HIF-1α expression. Dysplastic lesions in breast, colon, and prostate tissue overexpressed HIF-1α, while benign tumors of the breast and uterus did not. Only 29% of primary breast cancers displayed HIF-1α expression. In contrast, 69% of breast cancer metastases showed HIF-1α expression. In brain tumors, areas of the brain vascularized by angiogenesis stained positive for the presence of HIF-1α. All of these data support an important role for HIF-1α in human cancer progression, that is, the final stage of the malignant tumor development process. 106
HIF-1α protein expression in human tumors and their metastases.a
HIF: hypoxia-inducible factor.
aAdapted from Zhong et al. 106
b N, number of cases analyzed; −, no staining; +, nuclear staining in less than 1% of cells; ++, in 1–10% of cells; +++, in 10–50%; ++++, greater than 50%.
Additional lines of evidence support a role for HIF-1α in the later and final stages of malignant tumor progression. HIF-1α overexpression seen in biopsies of brain, breast, cervical, esophageal, oropharyngeal, and ovarian cancers is positively associated with treatment failure and patient mortality. 110 In human breast and brain cancers, HIF-1α overexpression strongly correlates with tumor grade (degree of microscopic abnormality) and vascularity. 111 In metastatic osteosarcoma, HIF-1α impacts tumorigenesis, tumor metabolism, differentiation, angiogenesis, proliferation, metastasis, and ultimately patient survival. 112,113 HIF-1α is overexpressed in gastrointestinal stroma of stomach tumors and in gastric adenocarcinomas but absent in normal gastric mucosa. 114
Transformed cell lines are not analogous to pre-initiated cells
Before summarizing the data on the effect of cobalt on HIF-1 expression in cultured cells, it is necessary to understand the many differences between immortalized cell lines and primary cell cultures. The genetic characteristics of transformed cells include aneuploidy, heteroploidy, high spontaneous mutation rate, overexpressed oncogenes, and mutated or deleted suppressor genes. Structural changes of transformed cells include altered cytoskeleton, changed extracellular matrix, modified expression of cell adhesion molecules, and disrupted cell polarity. Growth characteristics of transformed cells include immortalization, loss of contact inhibition, anchorage independence, reduction in growth density limits, growth factor independence, low serum requirement, and a shorter population doubling time. Neoplastic characteristics of transformed cells include tumorigenicity, invasiveness, and increased protease secretion. In summary, transformation is associated with genetic instability, immortalization, aberrant growth control, and malignancy. 115
Cobalt can mimic hypoxia in vitro
A number of different in vitro studies have exposed primary and transformed cells to cobalt compounds and measured expression of HIF-1. Table 4 lists the cobalt source, cell lines; whether the cell line was HIF-1 positive following cobalt compound administration; and the reference. Cobalt is an essential element in cellular growth and development. The level of cobalt in cells is incredibly small (in the ppb range). In both primary and immortalized cell lines, very small amounts of cobalt is added in the growing medium. 126 Both primary and immortalized cell lines exposed to cobalt (a hypoxia mimetic agent) will induce the accumulation of HIF-1α protein in primary cell cultures under normal oxygen tension (normoxia). 127–128 The concentration of Co ions is somewhat elevated for induction of overexpression of HIF-1 to occur (>50 µM) under normoxia.
Cobalt as a hypoxia mimetic in primary and transformed cell lines.
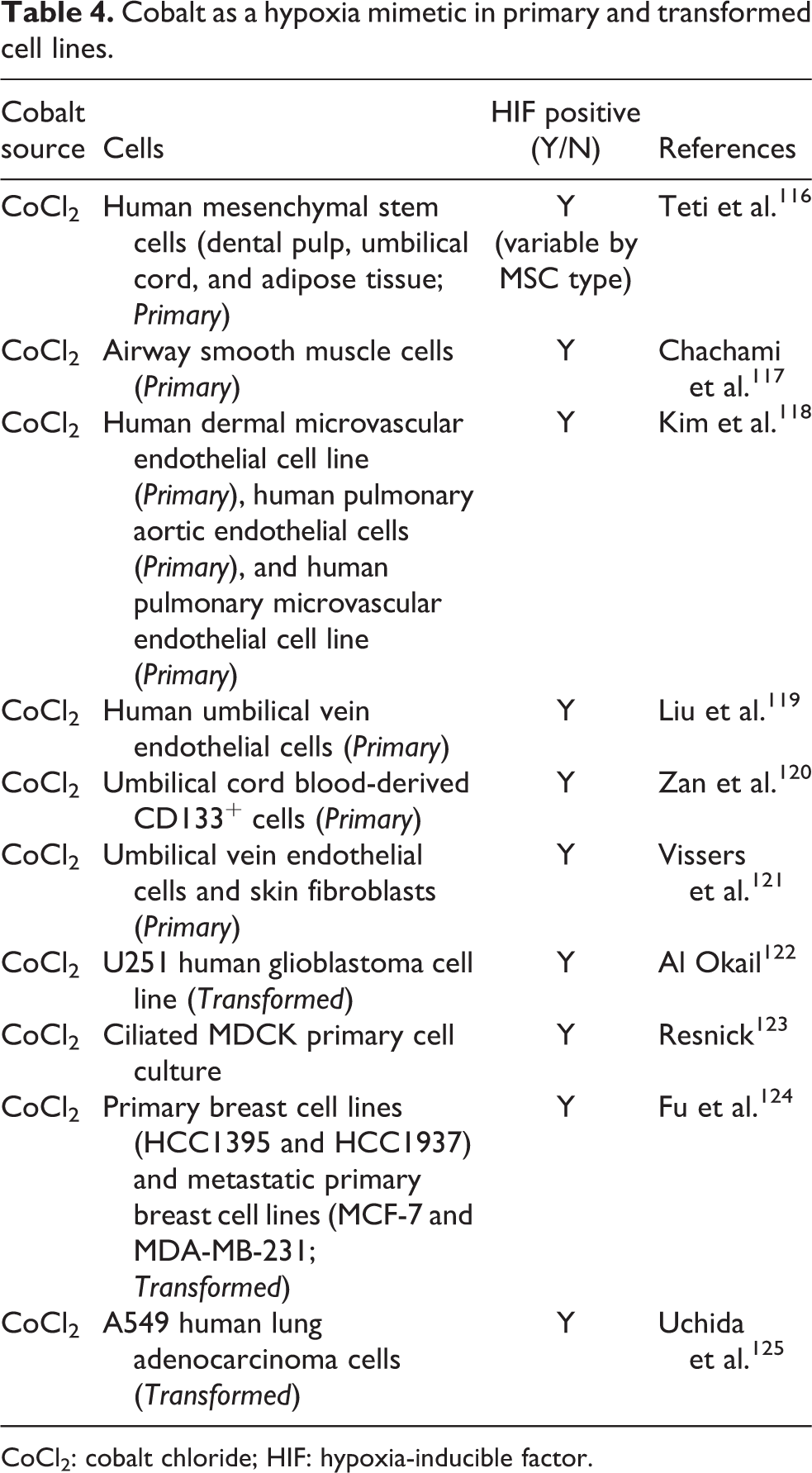
CoCl2: cobalt chloride; HIF: hypoxia-inducible factor.
Conclusions
Chronic NTP rodent inhalation studies using cobalt sulfate and cobalt metal report an increase in lung and other tumors in rats. 1,2 Previously, our group analyzed the entire NTP database 31,35,36 and supported the original conclusions of Cohen and colleagues, 37 –40 Moolgavkar and Knudson, 41 and Ames and Gold 42 regarding an important role for increased cellular proliferation rates resulting from cytotoxicity induced by high doses of test agent. The increased cellular proliferation rates amplify background mutation rates and induce tumors in rodents not related to genotoxic effects. NTP does not consider the role of mitogenesis inducing mutagenesis as an explanation underlying the formation of rat pulmonary tumors despite the pro-inflammatory effect of inhaled cobalt compounds. In contrast, one of the mechanisms invoked to explain the increase in rat lung tumors in the NTP study, and why that increase would be relevant to humans, is an increase in HIF-1 from cobalt exposure. Rodent pulmonary tumors differ from human lung cancers in that the human tumors tend to be highly vascularized thereby possessing metastatic potential. In contrast, rodent pulmonary tumors tend to be localized and less vascularized. Inhalation of cobalt compounds in rats and mice does not model the late final progression stage of human pulmonary carcinogenesis but rather attempts to model the earlier stages of tumor initiation and promotion not affected by overexpression of HIF. 36
Several lines of evidence suggest that in vitro demonstrations of cobalt-induced increases in HIF-1 are not relevant to human cancers. The mechanism by which overexpression of HIF-1 would be expected to influence cancer would be an increased tendency toward angiogenesis thereby accelerating the later progression stages of the disease. The very large number of human patients with long-term exposure to cobalt ions via leaching of MOM prosthetic implants who do not demonstrate an increased risk of cancer does not support a role for enhanced angiogenesis. Similarly, goats ingest up to 667 times the human ingestion level of cobalt throughout their lifetime of 15–18 years and demonstrate low cancer rates, that is, about a 1% incidence rate. If cobalt was inducing a clinically significant angiogenic response, the large exposures and significant life spans of goats might be expected to show at least some carcinogenic effect. In addition, 70% of all human cancers, including lung cancers, demonstrate overexpression of HIF-1 in the absence of cobalt exposure. Although cobalt can function as a hypoxia mimetic in cell culture studies, hypoxia routinely develops when tumors exceed the size at which oxygen and waste products can be exchanged via diffusion in the absence of cobalt 129 –132 rendering cobalt’s hypoxia mimetic properties redundant. In summary, the current weight of the evidence suggests that in vitro cobalt-stimulated HIF-1 overexpression does not correlate with cancer risk from cobalt exposure in humans.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
