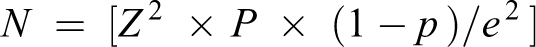Abstract
Background:
The potential effect of chronic metformin pharmacotherapy to cause vitamin B12 deficiency has been of tremendous concern especially among diabetic patients. Haematological abnormalities following vitamin B12 deficiency among diabetic patients contribute immensely to morbidity and mortality in this group of patients.
Aim:
This study was designed to elucidate the chronic haemato-toxicologic adverse profile for metformin with respect to its potential to induce vitamin B12 deficiency via reduction in the gastrointestinal absorption of vitamin B12 by performing comparative analyses between the serum vitamin B12 levels and haematological indices among metformin-treated and metformin-naive type 2 diabetes mellitus (DM) patients attending the outpatient Endocrinology Clinic of Irrua Specialist Teaching Hospital (ISTH), Irrua, Edo State, Nigeria, with the rational purpose of alleviating the associated morbidity and mortality.
Materials and Methods:
This was a case–control, prospective, analytical, and observational study of 200 adult participants (100 per group) attending the Endocrinology Outpatients Clinic of ISTH. Serum vitamin B12 levels were analysed using an immunoassay technique. Haematological indices were determined using standard methods, and patients examined for clinical features of anaemia. Data were presented using tables and charts. χ 2 and t-tests were used to compare discrete and continuous data, respectively. The receiver operating characteristic (ROC) curve was plotted graphically to determine the sensitivity and specificity of using serum vitamin B12 assay as a screening and diagnostic test for the haematologic abnormality of ovalocytosis among the metformin-treated type 2 DM patients.
Results:
A total of 200 type 2 diabetic patients comprising 100 metformin-treated and 100 metformin-naive patients with average age of 55.8 ± 9.3 years were studied. The mean serum vitamin B12 levels in metformin-treated and metformin-naive participants with frank vitamin B12 deficiency (i.e. mean serum vitamin B12 level ≤ 199 pg ml−1) were 158.29 ± 29.27 pg ml−1 and 173.95 ± 14.21 pg ml−1, respectively (p = 0.028). This was significantly lower for the metformin-treated group compared to metformin-naive group with respect to the participants with frank vitamin B12 deficiency. There were instances of statistically significant differences between the mean corpuscular volume (MCV), mean corpuscular haemoglobin (MCH) and total white blood cell (WBC) count among the metformin-treated compared to the metformin-naive type 2 DM patients. The ROC curve showed that serum vitamin B12 assay had moderate sensitivity of 72% with moderate specificity of 66% at detecting the presence and absence of ovalocytosis in the presence and absence of frank vitamin B12 deficiency, respectively, among the metformin-treated group (i.e. serum vitamin B12 ≤ 199 pg ml−1 with p = 0.002).
Conclusion:
The occurrence of vitamin B12 deficiency was high among metformin-treated type 2 DM patients. Our study showed remarkable statistically and clinically significant differences in the chronic haemato-toxicology of metformin on mean serum vitamin B12 level, ovalocytosis, MCV, MCH and total WBC count between the metformin-treated and metformin-naive participants. We advocate for vitamin B12 supplements in this group of patients via the parenteral route of administration, most preferably the intramuscular site injection; in order to prevent the occurrence of vitamin B12 deficiency among them. Lastly, we recommend the use of serum vitamin B12 assay and complete blood count (CBC) with peripheral blood films (PBFs) as a reliable way to diagnose and screen for vitamin B12 deficiency among metformin-treated type 2 DM patients.
Keywords
Introduction
Metformin is currently one of the first-line pharmacotherapeutic agents used for the treatment of type 2 diabetes mellitus (DM). It belongs to the biguanide class of antidiabetic drugs. 1 –3 Metformin has been found to significantly improve peripheral insulin sensitivity and blood glucose control via decreasing hepatic gluconeogenesis and glycogenolysis and has been associated with decreased cardiovascular mortality, which is the major cause of death among diabetic patients. 4 –6 Metformin has been found to be useful in primary prevention of ischaemic heart disease by reducing cardiomyocytes apoptosis and causing adaptation of cardiomyocytes metabolism during ischaemia as well as protection against the development of heart failure. 7 –9 Furthermore, metformin has been discovered to be useful in the treatment of diabetic nephropathy by decreasing the generation of reactive oxygen species 10,11 and polycystic ovarian syndrome (PCOS), a condition occurring among 5–15% of women in reproductive age with clinical features of menstrual disturbances, hyperandrogenism, bilateral polycystic ovaries on ultrasound and hyperinsulinemia, which makes the use of metformin unquestionably relevant in increasing insulin sensitivity. 12 –14 Metformin has also been found to cause apoptosis of cancer cells in type 2 DM patients 15,16 and has been reconsidered in the treatment of gestational diabetes mellitus in late second half of the second trimester and whole of the third trimester, following cumulative evidence of therapeutic benefits in pregnant obese women. 7 –9 It was observed to play a significant role in the regulation of circadian clock by decreasing a whole body metabolic rate, a valid process in the reduction of body weight. 17 –20 Metformin is a hydrophilic base, hence passive diffusion through plasma membrane is limited. It is absorbed into the gut, liver, and kidneys by an active process involving organic cationic transporters (OCT1/2) and excreted via the kidneys. 10 –12 Metformin causes adverse effect of lactic acidosis which will manifest mostly as myalgia or muscle cramps in affected individuals clinically. But the lactic acidosis caused by metformin pharmacotherapy has not been linked to vitamin B12 deficiency by any previous nor current study.
The prevalence of DM has been found to be on the increase worldwide. Globally, as at 2010, an estimated 285 million people had DM, with type 2 DM making up about 90% of the cases. 7 –9 As of September 2018, according to the report by International Diabetes Federation, an estimated 425 million people are living with DM and 50% of these patients were undiagnosed. Also, about 1.6 million deaths are directly attributed to diabetes each year. The global prevalence (age standardized) of diabetes has nearly doubled since 1980 rising from 4.7% to 8.5% in adult population. This will rise to about 629 million in 2045, with 79% of diabetics living in low- and middle-income countries. 10 –13 A national survey in 1992 by the Non-communicable Disease Expert Committee of the Federal Ministry of Health recorded a prevalence of 2.2% in Nigeria. 14 In 2014, The International Diabetes Federation estimated the prevalence of DM to be 4.6% in Nigeria. 15,16 In 2016, an estimated 422 million adults globally are said to be living with DM according to World Health Organisation. 17 –20
Vitamin B12 or cobalamin is a water-soluble vitamin that plays a significant role in DNA synthesis, optimal haemopoiesis and neurological function. 21 Deficiency of vitamin B12 presents predominantly with haematological and neurocognitive dysfunction. 21 Vitamin B12 is a red crystalline substance with a molecular weight of 1630 g mol−1. 22 It contains cobalt, nitrogen and phosphorus. This is an essential biomolecule with a stable metal–carbon bond, and it is an organometallic compound in which the cobalt atom can link various ligands such as a methyl group (–CH3), a 5′-adenosyl, a cyano (–CN) group or hydroxyl (–OH). The cobalt atom is situated within a corrin moiety that contains four reduced pyrrole rings that bind a central cobalt atom – a structure similar to the porphyrin from which haem is formed. The remaining coordinating positions of the central cobalt atom are occupied by a 5,6,-dimethyl benzimidazole group – below the ring and various ligands above. Vitamin B12 is an essential vitamin which cannot be synthesized by the human body and must be supplied in the diet. It is not destroyed by cooking; the only source in humans is food of animal origin. The highest amounts are found in liver and kidney but are also present in organs and muscle meats, fish, shellfish, chicken and dairy products (eggs, cheese and milk which all contain small amounts). The minimum daily requirement for cobalamin in adults is about 2.5 µg. 18 The body has a large reserve for cobalamin. This is between 2 mg and 5 mg. 19 About 2 mg of cobalamin is normally stored in the liver and another 2 mg elsewhere in the body. In view of the minimum daily requirement, about 3–6 years would be required for a normal healthy individual to be deficient in cobalamin if its absorption was to cease abruptly and completely. 18,23
The preliminary steps in the metabolism of vitamin B12 involve its release from animal sources, a process mediated by the action of pepsin and gastric acid. Following its release, it then binds to R-protein secreted by the salivary glands. At the duodenum, in the presence of an alkaline medium and pancreatic juices, the R-protein is hydrolysed to release vitamin B12 which then binds intrinsic factor (IF) secreted by gastric parietal cells. The vitamin B12-IF complex is highly resistant to proteolytic degradation. The complex attaches at a specific receptor in the mucosa of the terminal ileum, where its absorption occurs. This endocytotic process of vitamin B12 absorption is a calcium-dependent mechanism. This is the active mechanism of vitamin B12 absorption. 20,24 The intracellular vitamin B12 is released following degradation of IF. The free vitamin B12 subsequently binds a protein carrier known as transcobalamin (TC)-II. TC-II is a β-globulin (molecular weight 38,000 g mol−1) synthesized by the liver cells, macrophages and by other tissues such as the ileum and endotheliocytes. It normally carries about 20–60 ng of cobalamin and readily gives up cobalamin to bone marrow, placenta and other tissues which it enters by receptor-mediated endocytosis to form a complex known as holo-TC-II which later releases the vitamin B12 into circulation or the complex is actively taken up by the liver, bone marrow and other vital body cells. The liver is the major storage organ for vitamin B12, and it stores up to about 90% of total body vitamin B12. A disruption in the absorptive process, such as the use of metformin, proton-pump inhibitors (PPIs), inadequate intake, pernicious anaemia, chronic atrophic gastritis in elderly and chronic pancreatitis, will result in clinical or biochemical vitamin B12 deficiency. 11,12 Another mechanism exists for vitamin B12 absorption which is passive occurring equally through the duodenum and the ileum; it is rapid but less efficient as less than 1% of an oral dose can be absorbed by this process. The active mechanism discussed above, which occurs through the ileum in humans, is efficient for small oral doses of vitamin B12. This is the normal mechanism by which the body acquires vitamin B12 and it is mediated by gastric IF (molecular weight 45 000 g mol−1) which is synthesized in the fundus and body of the stomach by the gastric parietal cells. 13,14 It is worthy of note that TC-II deficiency causes megaloblastic anaemia because of the failure of cobalamin to enter the marrow (and other cells) from plasma but serum cobalamin levels in TC-II deficiency are normal. This is because most cobalamin in plasma is bound to another transport protein TC-I. 15,16 TC-III is a minor iso-protein of TC-I found in human plasma and specific granules of granulocytes, which differs from TC-I by its composition of sugars and cobalamin content. 17,18
Vitamin B12 exerts its physiological function by mediating two principal enzymatic pathways, that is, the methylation process of homocysteine to methionine and the conversion of methyl malonyl coenzyme A (Co-A) to succinyl Co-A 22 which enters into the Krebs cycle. Vitamin B12 is an essential cofactor for two enzymes in human cells, which are methionine synthase and methyl malonyl Co-A synthase. Vitamin B12 exists in two metabolically active forms as methyl cobalamin and adenosyl cobalamin. 23,25 Vitamin B12 acts as a cofactor for methionine synthase in the methylation of homocysteine to methionine, which is later activated to s-adenosyl-methionine that donates its methyl group to methyl acceptors such as myelin, neurotransmitters and phospholipids. Metabolically significant vitamin B12 deficiency thus will result in disruption of the methylation process and the accumulation of intracellular and serum homocysteine. Hyperhomocysteinaemia has been documented as potentially neurotoxic and capable of causing vascular endothelial injury. This pathway is also essential in the conversion of dietary folate (methyl-tetrahydrofolate) to its active form, tetrahydrofolate. In another important enzymatic pathway, vitamin B12 acts as a cofactor to mediate the conversion of methyl malonyl Co-A to succinyl Co-A. In the presence of vitamin B12 deficiency, this pathway is hampered, resulting in the accumulation of methyl malonyl Co-A and its metabolite methyl malonic acid (MMA). The accumulation of MMA has been implicated in impaired neuronal membrane fatty acid synthesis as a result of production of long-chain fatty acids with odd number carbon atoms, as against an even number carbon atoms in the presence of methyl malonyl CoA. 22,23 Vitamin B12 is also essential for the synthesis of hormones such as melatonin and epinephrine, as well as the monoaminergic neurotransmitters such as serotonin, norepinephrine and dopamine. 26,27 Thus, these processes are thought to be responsible for the resultant neurocognitive and neuropsychiatric manifestations accompanying vitamin B12 deficiency. The hallmark of vitamin B12 deficiency is axonal demyelination, degeneration and subsequent death following neuronal damage, which manifests as peripheral or autonomic neuropathy, subacute combined degeneration of the spinal cord, delirium and dementia. 25 –28 There is also evidence from studies that have demonstrated that hyperhomocystinaemia is associated with a high risk of cardiovascular events due to its cellular and vasculotoxic effects. 23,25,28 Vitamin B12 as an essential micronutrient is also required for DNA synthesis, cellular repairs and normal haemopoiesis alongside other micronutrients such as folic acid and iron. Deficiency of vitamin B12 is associated with overt haematological findings like oval macrocytic red blood cells (mean corpuscular volume (MCV) > 100 fl), with or without anaemia, hypersegmented neutrophils (>5% neutrophils with 5 lobes or a single neutrophil with >5 lobes) and pancytopenia. 20,29 Defective cell repair process can result in mucositis, malabsorption from villi atrophy, atrophic glossitis and stomatitis. At present, guidelines advocating for routine screening for vitamin B12 status in type 2 DM patients on metformin therapy have not been well established. However, studies that have demonstrated findings of vitamin B12 deficiency in this group of patients have advocated for the need of vitamin B12 status evaluation or screening prior to initiation of metformin and during the course of therapy among patients with a history of long-term usage of metformin (≥3–4 years), use of high doses of metformin (≥ 2 g day−1), clinically worsening diabetic distal peripheral polyneuropathy in the presence or absence of haematological abnormalities. 30 The screening approach for vitamin B12 status among diabetic patients and the general population is similar. Serum vitamin B12 level is widely accepted and relatively the preliminary screening step for vitamin B12 status among patients with type 2 DM. Serum concentrations <200 pg ml−1 are often diagnostic of vitamin B12 deficiency, while concentrations >400 pg ml−1 confirm the absence of vitamin B12 deficiency. Serum vitamin B12 concentrations between 200 pg ml−1 and 400 pg ml−1 are considered as borderline status and it is an indication to screen for serum MMA and/or homocysteine levels. 26 –28 Sensitivity of the test depends on the assay kits. Vitamin B12 assay kits are readily available and affordable compared to the screening kits for MMA and homocysteine. Measurement of serum MMA and/or homocysteine concentration(s) is/are recommended for patients with suboptimal vitamin B12 levels (i.e. serum vitamin B12 concentrations ranging from 200 pg ml−1 to 400 pg ml−1 with subtle haematological manifestations). Serum MMA measurement has a sensitivity of 90–95% and specificity of 60%, while homocysteine assay is both less sensitive and specific as values could be affected by the concentration of serum folate; these however vary with the laboratory methods employed. Serum homocysteine and MMA concentrations of 5–15 µmol l−1 and <0.28 µmol l−1, respectively, are considered to be within the normal reference range. 26 –28
The management of type 2 DM involves the use of several oral hypoglycaemic agents; however, metformin which belongs to biguanide group of antidiabetic drugs is widely used as the first-line agent alone with dietary modifications. 18,19 Despite having these advantages, its potential to cause vitamin B12 deficiency has been widely reported, thereby contributing to the problems of DM such as anaemia, immune dysfunction, distal sensory polyneuropathy and cognitive impairment. 20 –26 Metformin has been reported to lower serum vitamin B12 levels in a dose-dependent manner and based on the duration of use. But, the precise mechanism by which metformin causes vitamin B12 deficiency has not been clearly elucidated; however, proposed mechanisms include alterations in small bowel motility due to calcium deficiency that stimulates bacterial overgrowth and consequential vitamin B12 malabsorption. Metformin has also been shown to inhibit the calcium-dependent endocytotic absorption process of vitamin B12-IF of castle complex at the terminal ileum. This inhibitory effect is reversed by calcium supplementation. 23–27,30 The chronic haemato-toxicologic and neuro-toxicologic adverse profiles of metformin with respect to its potential effect to induce vitamin B12 deficiency occur via reduction in the gastrointestinal absorption of vitamin B12. Decrease in vitamin B12 absorption and plasma levels following metformin use typically starts as early as the fourth month after commencing metformin pharmacotherapy. 16,17,29,30 A study done on seven type 2 DM patients using metformin with abnormal vitamin B12 absorption attending the Royal Victoria Hospital, Belfast, Ireland, showed a normal vitamin B12 absorption within 7–28 days after substituting chlorpropamide for metformin when retested for vitamin B12 absorption by measuring percentage concentrations of radiolabelled 57Cobalt [57Co] excretion in urine. 16,17 Clinically, overt features of vitamin B12 deficiency manifest by 5–10 years after commencing metformin pharmacotherapy owing to the large body stores in the liver that is not quickly depleted. 18,24,27,31 Also, a study done in Ibadan, Nigeria, found a prevalence of vitamin B12 deficiency in metformin-treated type 2 DM patients to be 8.6%. 12 However, the study in Ibadan did not determine the presence of clinical or haematological abnormalities associated with vitamin B12 deficiency which our study will thoroughly investigate. In sub-Saharan Africa, including Nigeria, there has been an increase in the prevalence of type 2 DM lately. 5,6,12,24,31 There are, however, paucity of data regarding the contribution of vitamin B12 deficiency among metformin-treated type 2 DM patients to the development of associated complications such as megaloblastic (macro-ovalocytic) anaemia. 17,25–31
This study was designed to elucidate the chronic haemato-toxicologic adverse profile for metformin with respect to its potential to induce vitamin B12 deficiency via reduction in the gastrointestinal absorption of vitamin B12 by performing comparative analyses between the serum vitamin B12 levels and haematological indices among metformin-treated and metformin-naive type 2 diabetic patients attending the outpatient Endocrinology Clinic of Irrua Specialist Teaching Hospital (ISTH), Irrua, Edo State, Nigeria, with the rational purpose of alleviating the associated morbidity and mortality.
Materials and methods
Study location and population
This study was conducted at the ISTH. It is a Tertiary Teaching Hospital located at Irrua town, Esan Central Local Government Area in Edo State, Nigeria. The hospital is at a distance of about 87 km north of Benin City, the Edo state capital. The ISTH is a 343-bed capacity hospital involved in the management of patients and training of medical students, resident doctors and nursing students. The Endocrinology Clinic holds once a week with about 46 diabetic patients seen per clinic day. The study population comprised of adult medical patients with type 2 DM attending the Endocrinology Outpatients Clinic at ISTH. Their ages were ≥30 years and were fulfilling the inclusion criteria for recruitment, evaluation and followed up.
Inclusion and exclusion criteria
The inclusion criteria for evaluation in this study were known adult medical patients with type 2 DM attending the Endocrinology Outpatients Clinic at ISTH after commencing the study, provided they granted their informed consents to participate in the study and were ≥30 years of age. They also fulfilled the diagnostic inclusion criteria used to define type 2 DM according to the American Diabetes association (ADA).
The exclusion criteria for this study were as follow: Known type 1 DM patients. Diabetic patients <30 years of age whose diagnosis of whether type 1 DM or type 2 DM was highly ambiguous and uncertain. Patients who do not fulfil the diagnostic inclusion criteria used to define type 2 DM according to the ADA. Adult type 2 DM patients who did not grant their informed consents to participate in the study. Adult type 2 DM patients who at the time of recruitment for the study were pregnant women, purely vegetarians, receiving vitamin B12 containing supplement or chronic alcoholics (>21 units and >14 units per week for male and female for >10 years, respectively), patients taking cytochrome P450 microsomal enzyme inducer/inhibitor substances such as omeprazole and other PPIs, rifampicin, barbiturates, cimetidine, grapefruit juice, ritonavir, ketoconazole, carbamazepine or phenytoin, patients taking neomycin or having comorbidities such as undernutrition (body mass index (BMI) < 18.5 kg m−2), tuberculosis, benign/malignant neoplasms on chemotherapy, human immunodeficiency virus (HIV) infection/acquired immunodeficiency syndrome (AIDS), chronic liver disease, chronic kidney disease/acute kidney injury with estimate glomerular filtration rate < 60 mL min−1/1.73 m2, history of irritable bowel syndrome, pernicious anaemia or inflammatory bowel disorders such as Crohn’s disease or ulcerative colitis. Adult type 2 DM patients who had done previous surgeries such as bariatric surgery, partial/total gastrectomy or partial/total ileal resection. Patients without DM taking metformin for other medical reasons/conditions such as PCOS or insulin resistance syndrome.
Study design
This was a case–control, prospective, analytical, observational study.
Sample size determination
The formula for estimating sample size is 14
where N is the required sample size; Z is the critical value at 95% confidence level, which is equal to 1.96; P is the prevalence of vitamin B12 deficiency in patients with type 2 DM on metformin obtained from a previous study 5.8%; 15 e is the error margin that the investigator was willing to accept and in this instance, which is equal to 0.05. Note: (1 − p) = q, this was the proportion of the sample population not covered by the study; n = 1.96 × 1.96 × 0.058 (1 − 0.058)/0.05 × 0.05; n = 84; n = 96 (with 10% attrition). This was rounded up to 100 patients for each group.
Sampling method
Consecutively consented adults with type 2 DM who met the inclusion criteria for cases and controls were selected from the outpatient clinic at ISTH until the desired sample size was achieved. Both cases and controls were matched for age and sex.
Clinical examination
A structured questionnaire was administered to the selected participants by the researchers. The structured questionnaire contained information about socio-demographic characteristics, possible co-morbidity, surgery, alcohol ingestion, diet (vegetarian or not vegetarian) and symptoms of anaemia and gastrointestinal symptoms associated with vitamin B12 deficiency, medication information including the dose and duration of therapy.
All participants were made to undergo thorough clinical examination to document the presence of relevant haematological signs. Evidence of pallor was examined for in areas like the conjunctiva, oral mucosa and palms.
Subjects’ preparation and sample collection
The laboratory assessments comprised of baseline haematological parameters (full blood count) and serum vitamin B12. Venopuncture site was cleaned with 70% methylated spirit and allowed to dry. Ten millilitres of whole blood was collected from the antecubital vein of each participant. Of this, 6 mL of venous blood was collected into a labelled vacutainer sample bottle coated inside with potassium ethylene diamine tetraacetic acid (EDTA) for complete blood count (CBC) and peripheral blood film (PBF). The remaining 4 mL of whole blood was dispensed into a labelled plain vacutainer sample bottle, allowed to clot, centrifuged for 15 min at 3000 r min−1 and separated into another plain bottle for vitamin B12 assay. Freshly prepared sera were stored in aliquots of at least 2 mL at a temperature of −20°C and pooled until the desired sample size was met for analysis. Repeated freezing/thawing cycles were avoided due to constant power supply at the storage unit.
Baseline haematological indices
CBC including red cell indices was obtained using an autohaematology analyser that requires blood sample drawn by the instrument (PCE-210N ERMA Inc., Tokyo, Japan). Blood sample collection and preparation of PBFs were done. Viewing of PBFs was done by haematologists. The standard reference intervals for CBC parameters are as follows: Hb = 13–18 g dL−1 male, 11.5–16.5 g dL−1 female, PCV = 40–52% male, 36–48% female, MCV = 77–95 fL, mean corpuscular haemoglobin (MCH) = 27–32 pg, mean corpuscular haemoglobin concentration (MCHC) = 32–36 g dL−1, white blood cells (WBC) = 4.0–11.0 × 103 cells µL−1, neutrophil = 2.0–7.5 × 103 cells µL−1, lymphocytes = 1.5–4.5 × 103 cells µL−1, eosinophils = 0.04–0.4 × 103 cells µL−1, basophils = 0.0–0.1 × 103 cells µl−1 and platelets = 150–400 × 103 cells µL−1.
Preparation and staining of peripheral blood film
First, a spreader was made from a glass slide that had a smooth end by breaking the corner of the slide leaving a width of 18 mm as the spreader. 16 A drop of blood was placed using a capillary tube at the centre of another glass slide that was dried and degreased, about 1 cm from one end. Without delay, the spreader was placed in front of the drop of blood at an angle of about 30° to the slide, and it was then moved back to make contact with the drop of blood. The drop spreads out quickly along the line of contact. With a steady movement of the hand, a spread of the drop of blood along the slide was made. Then, the slide was left to dry. The peripheral blood film (PBF) was immediately labelled after spreading with a grease pencil in the 1 cm space at the end of the slide. The air-dried blood smear was fixed for 15 min in 100% methanol. It was then transferred without blotting to a staining jar containing May-Grünwald stain freshly diluted with an equal volume of buffer solution (phosphate buffer pH 7.2) for 15 min. The slide was allowed to dry on a filter paper without blotting and then transferred into a jar container followed by the placement of Giemsa staining solution freshly diluted with 9:1 volume ratio of phosphate buffer at pH 7.2 for 30 min. After, it was transferred into a phosphate buffer solution (pH 7.2) and agitated for 10–20 s and then removed. The slide was allowed to dry in a vertical position. The film was viewed by haematologist under a light microscope to assess the red and white cells morphology using ×40 microscope objective lens, while the proportion of platelets was viewed with ×100 microscope objective lens.
Measurement of serum vitamin B12 levels
Vitamin B12 enzyme-linked immunosorbent assay (ELISA) assay kit, Cat. No. EE-L-0010, Lot AK0017APRO1014 Elabscience Biotechnology Company Ltd, China, was used for the measurement of serum vitamin B12 levels. The vitamin B12 quantitative test is based on the principle of the ELISA technique. The lower limit of normal for serum vitamin B12 depends on the technique used for the assay and it is usually set at about 200 pg ml−1 (148 pmol L−1). 17
Serum vitamin B12 levels <200 pg ml−1 are diagnostic of vitamin B12 deficiency, values ≥400 pg ml−1 confirm the absence of vitamin B12 deficiency, while values from 200 pg ml−1 to −399 pg ml−1 are considered as borderline vitamin B12 status levels. 18
Ethical consideration
Ethical approval for this study was obtained from the Health Research Ethical Committee of ISTH and the assigned protocol identification number was ISTH/HREC/2016/March/026. Duly written informed consents were obtained from all cases and controls. These written informed consents were documented in the respective medical case file for each of these patients and then counter signed by each of them for appropriate record purpose. All the study participants were counselled before and after clinical assessment and serum vitamin B12 screening. In addition, they were assured of the confidentiality of all their records. This study also served to notify and treat those participants with vitamin B12 deficiency and borderline vitamin B12 status.
Data analysis
The statistical package software for social sciences (SPSS) version 21.0 was used for data entry, validation and analysis. Frequency distribution tables and charts were generated for the categorical variables. Tests of significance were generated for different variables. χ 2 test was used to test categorical variables, and independent t-test was used to test the significance of means between two groups. The level of statistical significance was defined by a value of p < 0.05. The receiver operating characteristic (ROC) curve was plotted graphically to determine the sensitivity and specificity of using serum vitamin B12 assay as a screening and diagnostic test for the haematologic abnormality of macro-ovalocytosis among the metformin-treated type 2 DM patients.
Results
A total number of 200 participants comprising 100 metformin-treated and 100 metformin-naive type 2 DM patients were recruited for this study from November 2016 to September 2017. The socio-demographic parameters include age with its categorization and gender distribution. The age of the participants ranged from 31 years to 80 years with a mean of 55.80 ± 9.3 years. Majority of the participants, 39 (39%) for metformin-treated type 2DM patients and 37 (37%) for metformin-naive type 2DM patients, were between the age group of 51–60 years. Both groups were matched for age and gender. This was represented in Table 1 alongside with other socio-demographic profiles of the participants. Furthermore, Table 1 shows that 49% versus 51% among the metformin-treated group were males and females, respectively; the opposite was recorded for the metformin-naive group. Majority of the participants were married (96%) and income less than US$100 per month.
Socio-demographic profiles of metformin-treated and metformin-naive type 2 DM groups.
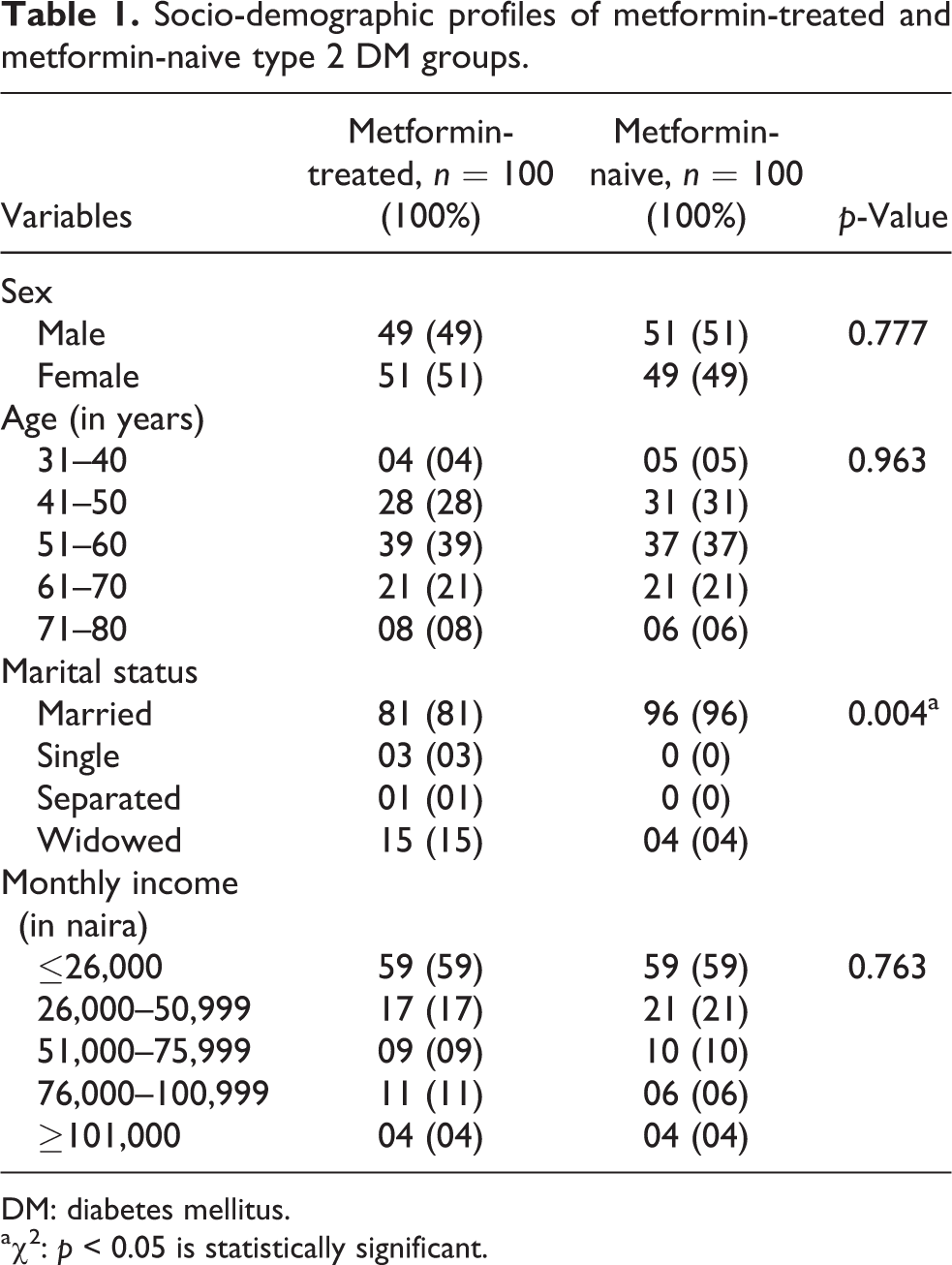
DM: diabetes mellitus.
a χ 2: p < 0.05 is statistically significant.
The mean age, height and weight were higher for male participants when compared with their female counterparts, 58.8 ± 8.9 years versus 56.1 ± 9.4 years, p = 0.316; 1.62 ± 0.08 m versus 1.59 ± 0.07 m, p = 0.512; and 67.5 ± 8.9 kg versus 66.7 ± 8.8 kg, p = 0.408, for males and females, respectively. However, the reverse was observed concerning the mean for BMI (25.7 ± 2.8 kg m−2 versus 26.4 ± 3.6 kg m−2; p = 0.491) and duration of DM diagnosis (4.92 ± 4.5 years versus 5.14 ± 4.9 years; p = 0.705) for males and females, respectively, although all their p-values were not statistically significant.
Figure 1 shows the mean serum levels of vitamin B12 among both metformin-treated and metformin-naive type 2 DM groups that were vitamin B12 deficient. The mean serum vitamin B12 levels in metformin-treated and metformin-naive participants with frank vitamin B12 deficiency (i.e. mean serum vitamin B12 level ≤199 pg ml−1) were 158.29 ± 29.27 pg ml−1 and 173.95 ± 14.21 pg ml−1, respectively (p = 0.028). This was significantly lower for the metformin-treated group compared to metformin-naive group with respect to the participants with frank vitamin B12 deficiency.
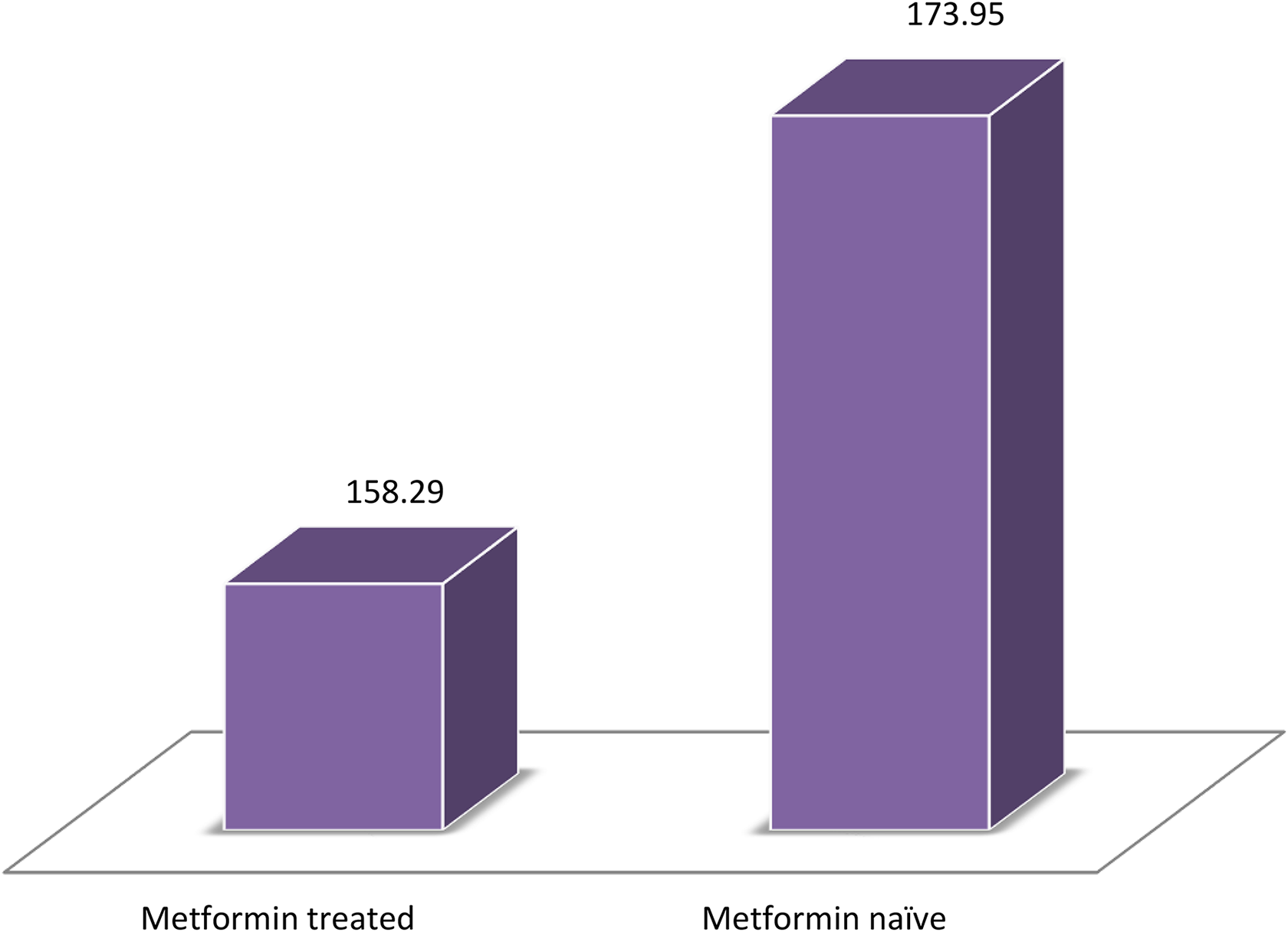
The mean serum levels of vitamin B12 among both metformin-treated and metformin-naive diabetic patients that were vitamin B12 deficient.
Table 2 presents the mean values of the hematologic parameters for both metformin-treated and metformin-naive type 2DM groups. Mean haematological parameters were lower for metformin-treated when compared to metformin-naive and were statistically significant for MCV, MCH and total WBC count with p-values of 0.01, 0.02 and 0.01, respectively.
Mean haematological parameters of the study groups.
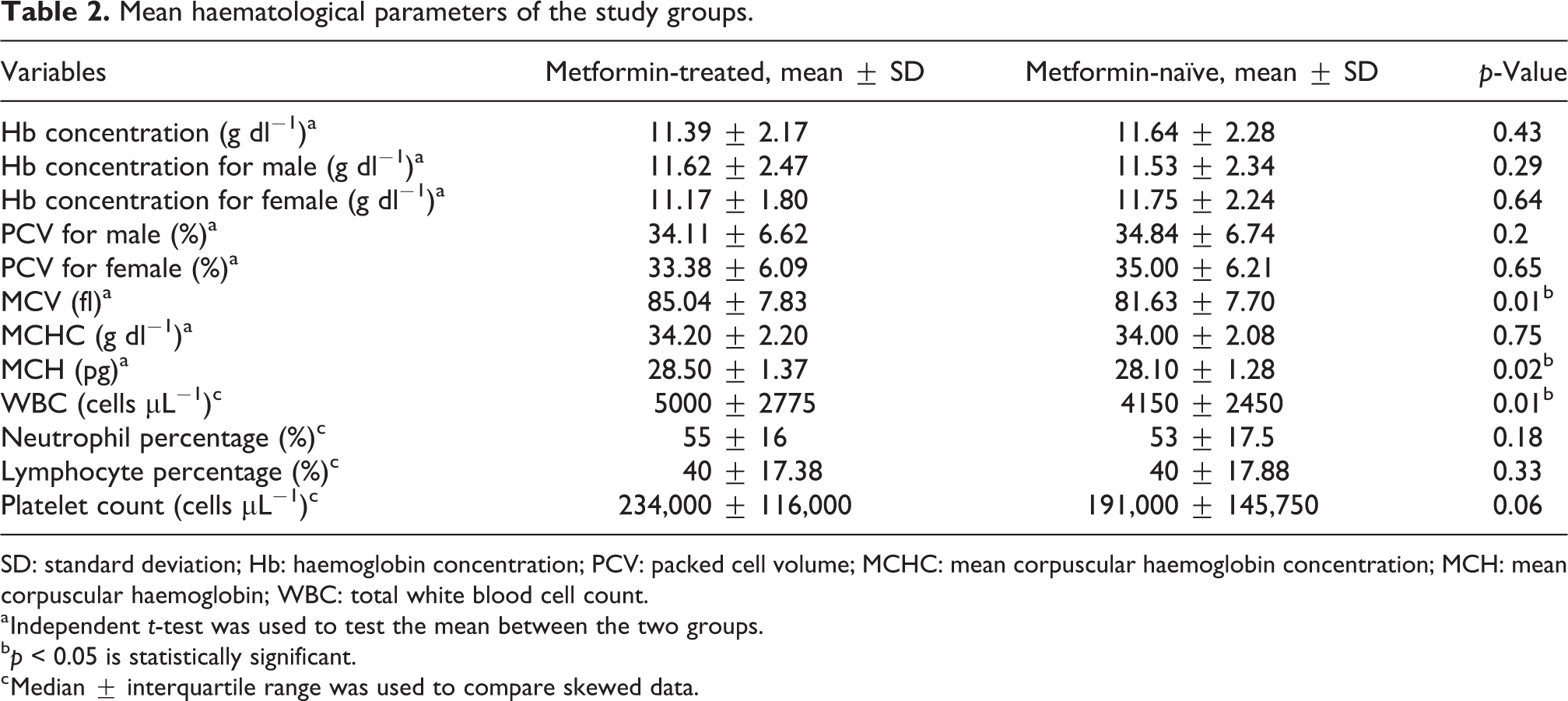
SD: standard deviation; Hb: haemoglobin concentration; PCV: packed cell volume; MCHC: mean corpuscular haemoglobin concentration; MCH: mean corpuscular haemoglobin; WBC: total white blood cell count.
a Independent t-test was used to test the mean between the two groups.
b p < 0.05 is statistically significant.
c Median ± interquartile range was used to compare skewed data.
Table 3 shows the frequency distribution for oval macrocytic red blood cells in the PBFs of metformin-treated and metformin-naive groups. Table 3 further reveals a statistically significant difference between the metformin-treated and metformin-naive participants with respect to ovalocytosis in 18% versus 6%, respectively (p = 0.009). But, there was no statistically significant difference between the metformin-treated and metformin-naive participants with respect to macrocytosis in 5% versus 0%, respectively (p = 0.06). A high MCV was recorded for very few participants (i.e. 5%) in the metformin-treated group, but it was normal for all the participants in the metformin-naive group.
Frequency distribution for macro-ovalocytes between metformin-treated and metformin-naive groups.
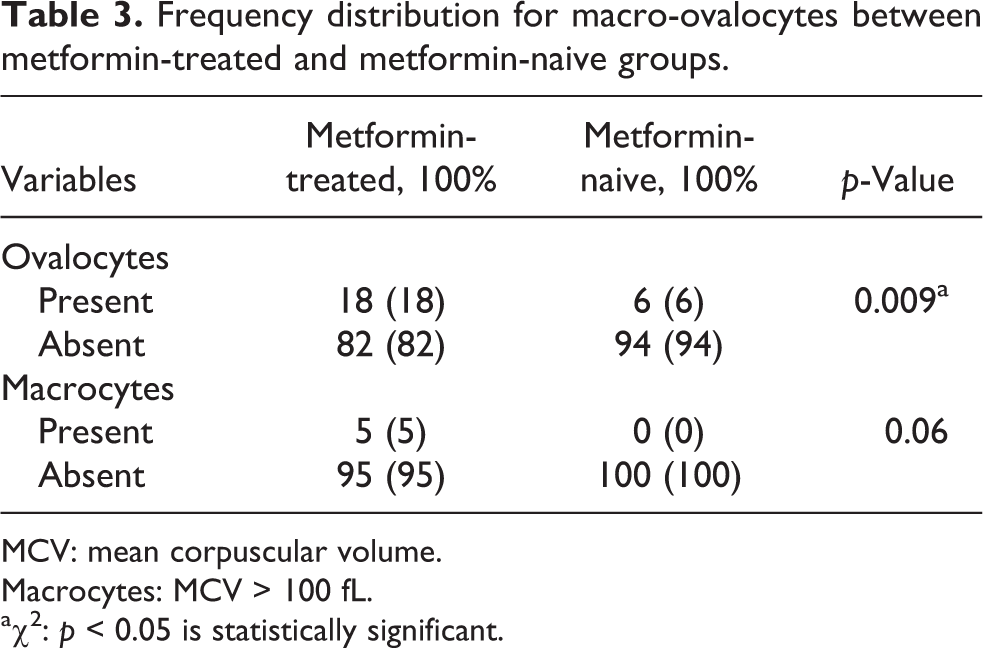
MCV: mean corpuscular volume.
Macrocytes: MCV > 100 fL.
a χ 2: p < 0.05 is statistically significant.
Table 4 presents the frequency distribution for clinical features of anaemia among metformin-treated and metformin-naive groups, respectively. There were no statistically significant differences between both groups with respect to the clinical features of anaemia such as easy fatiguability (p = 0.191), dizziness (p = 0.461), palpitation (p = 0.140), glossitis (p = 0.477), angular stomatitis (p = 0.312) and pallor (p = 0.404).
Frequency distribution for clinical features of anaemia among metformin-treated and metformin-naïve groups.
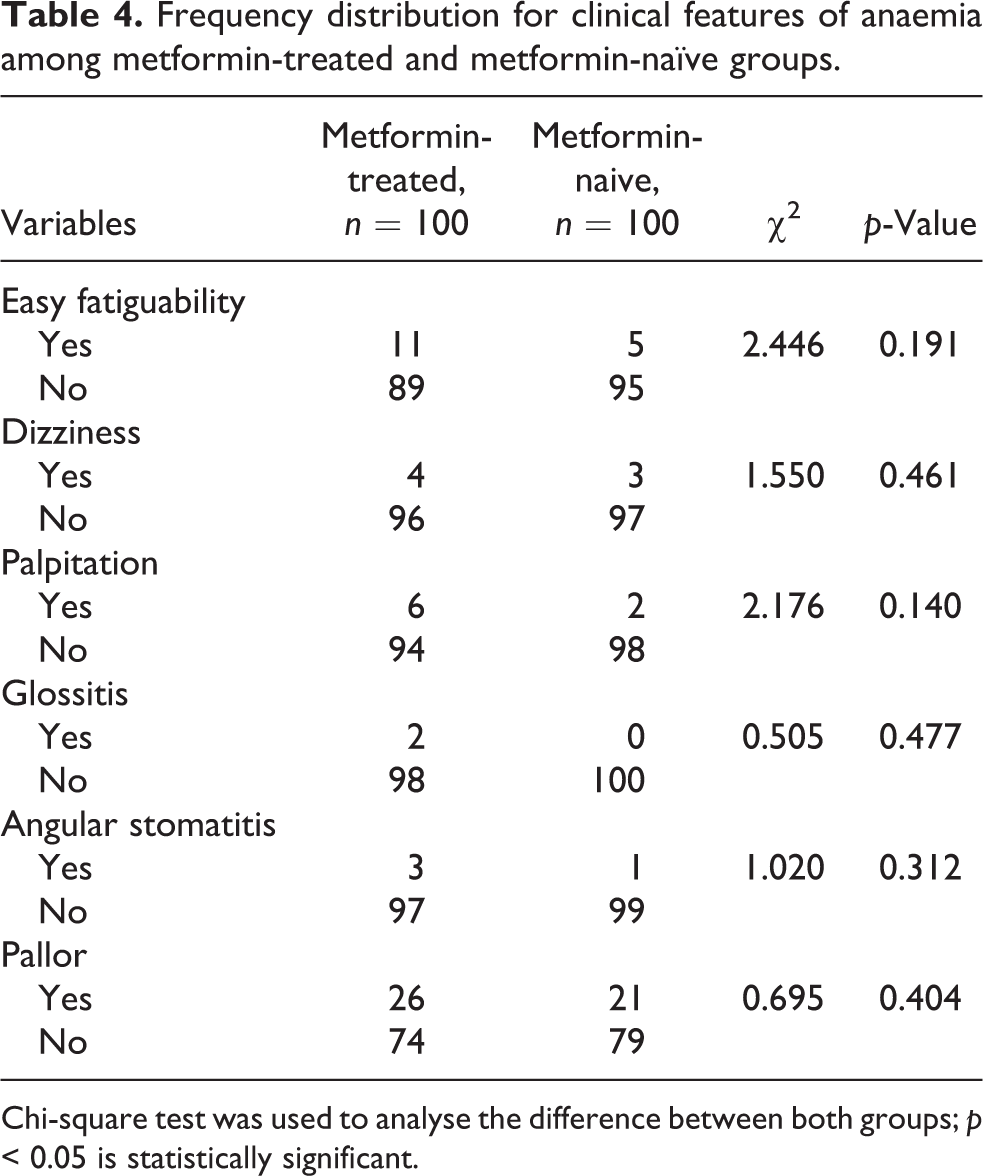
Chi-square test was used to analyse the difference between both groups; p < 0.05 is statistically significant.
Table 5 presents the relationship between serum vitamin B12 levels among metformin-treated group participants having vitamin B12 deficiency with and without the clinical features of anaemia. However, Table 5 did not reveal any statistically significant differences between these mean values with respect to the presence and absence of clinical features of anaemia such as easy fatiguability (p = 0.074), pallor (p = 0.170), palpitation (p = 0.074), dizziness (p = 0.219) and angular stomatitis (p = 0.278) within the metformin-treated group.
Relationship between serum vitamin B12 levels among metformin-treated group participants having vitamin B12 deficiency with and without the clinical features of anaemia.
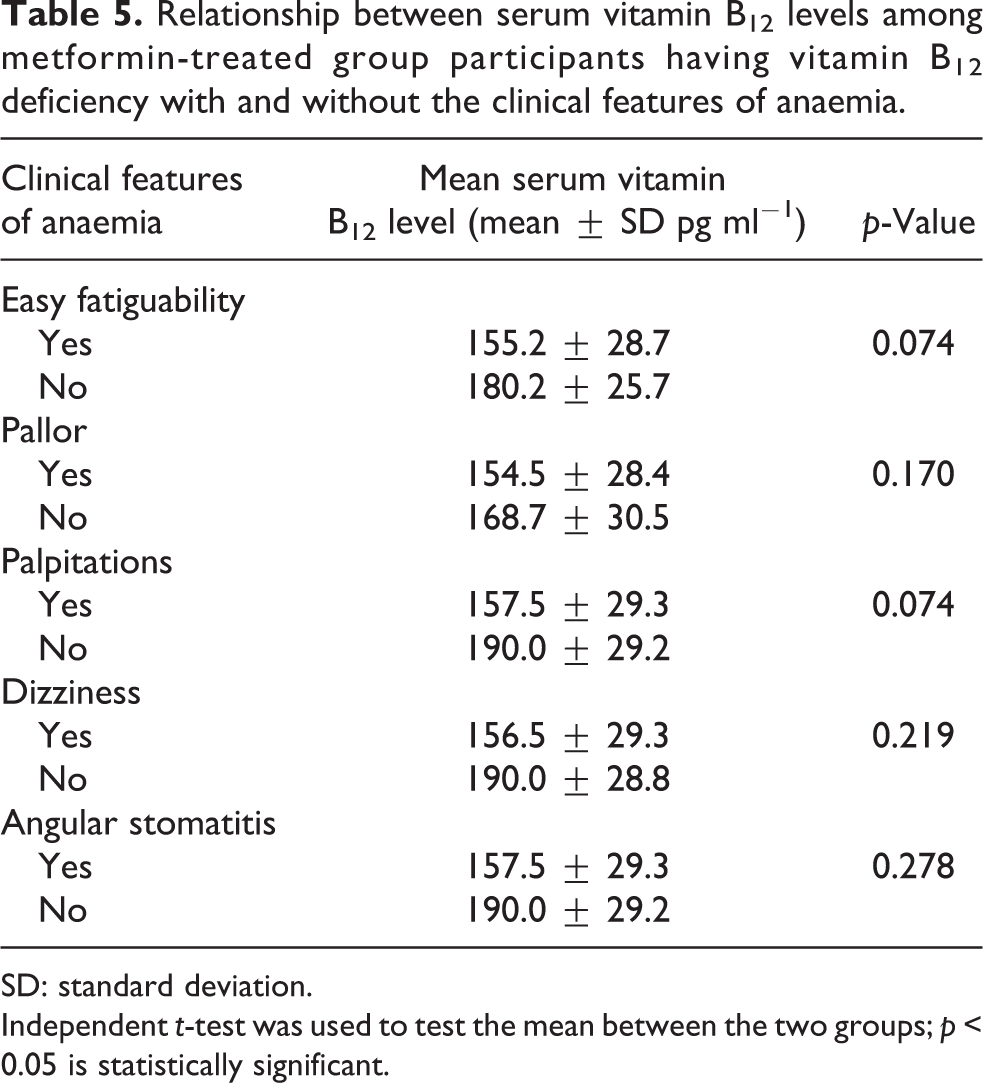
SD: standard deviation.
Independent t-test was used to test the mean between the two groups; p < 0.05 is statistically significant.
Table 6 presents the comparison between serum vitamin B12 levels among metformin-treated group participants with vitamin B12 deficiency and borderline vitamin B12 status with or without the clinical and laboratory features of anaemia. However, Table 6 reveals some statistically significant differences between the mean values with respect to the occurrence of clinical and laboratory features of anaemia, which included pallor (p = 0.001), dizziness (p = 0.038), easy fatiguability (p = 0.004), haemoglobin concentrations (Hb) for both males and females (p = 0.001 each), PCV for both males and females (p = 0.001 each) and ovalocytosis (p = 0.001). The participants with vitamin B12 deficiency had lower mean values compared to those with borderline status.
Comparison between serum vitamin B12 levels among metformin-treated group participants with vitamin B12 deficiency and borderline vitamin B12 status with or without the clinical and laboratory features of anaemia.
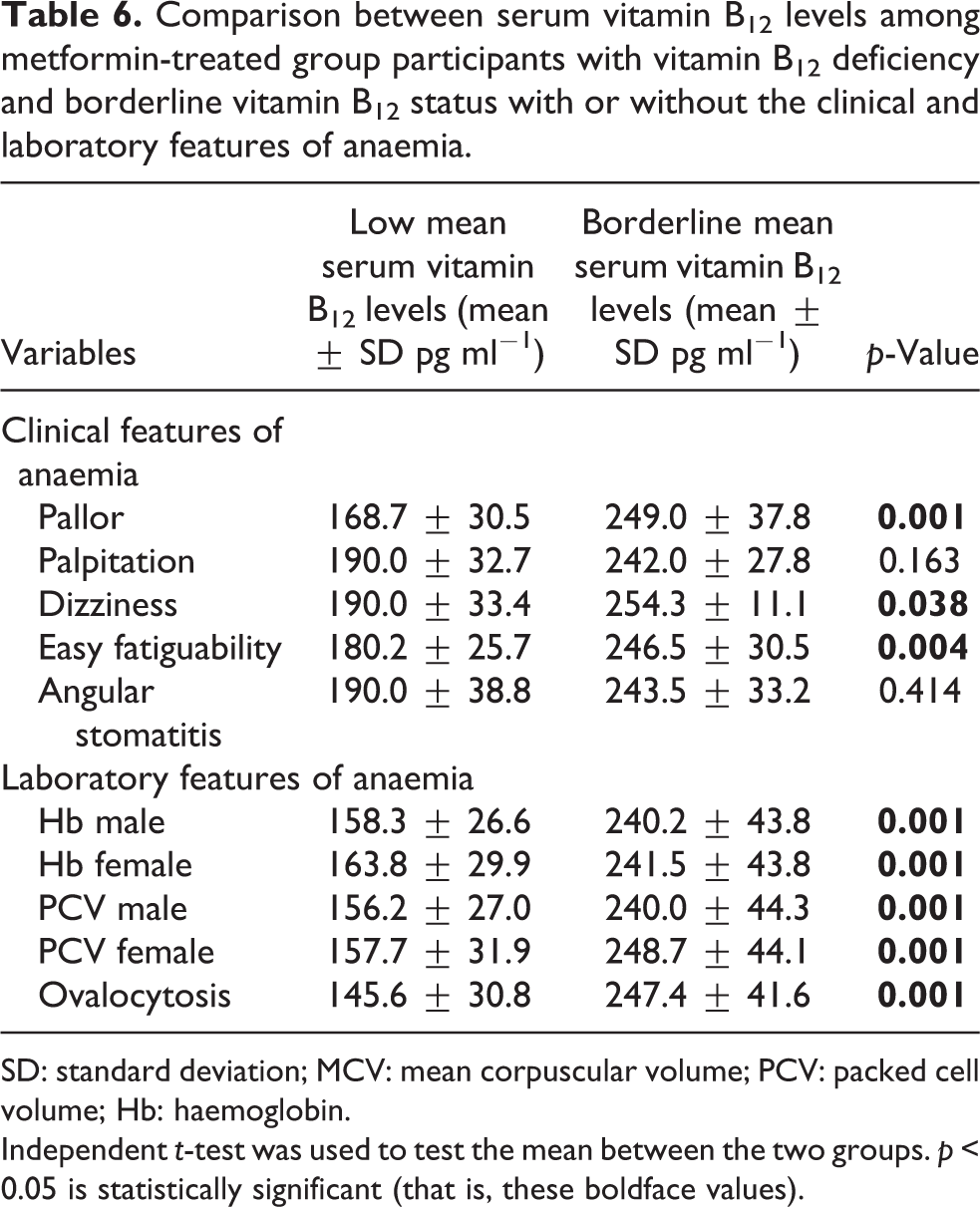
SD: standard deviation; MCV: mean corpuscular volume; PCV: packed cell volume; Hb: haemoglobin.
Independent t-test was used to test the mean between the two groups. p < 0.05 is statistically significant (that is, these boldface values).
Figure 2 illustrates the ROC curve to determine the sensitivity and specificity of serum vitamin B12 assay as a reliable screening and diagnostic test for ovalocytosis in metformin-treated (experimental) type 2 DM patients; area under the curve = 0.74; sensitivity = 72%; specificity = 66%; serum vitamin B12 ≤199 pg ml−1; p = 0.002. The curve showed that serum vitamin B12 assay value of ≤199 pg ml−1 was moderately sensitive and moderately specific at determining the presence and absence of ovalocytosis, respectively, in the presence and absence of vitamin B12 deficiency among the metformin-treated group.
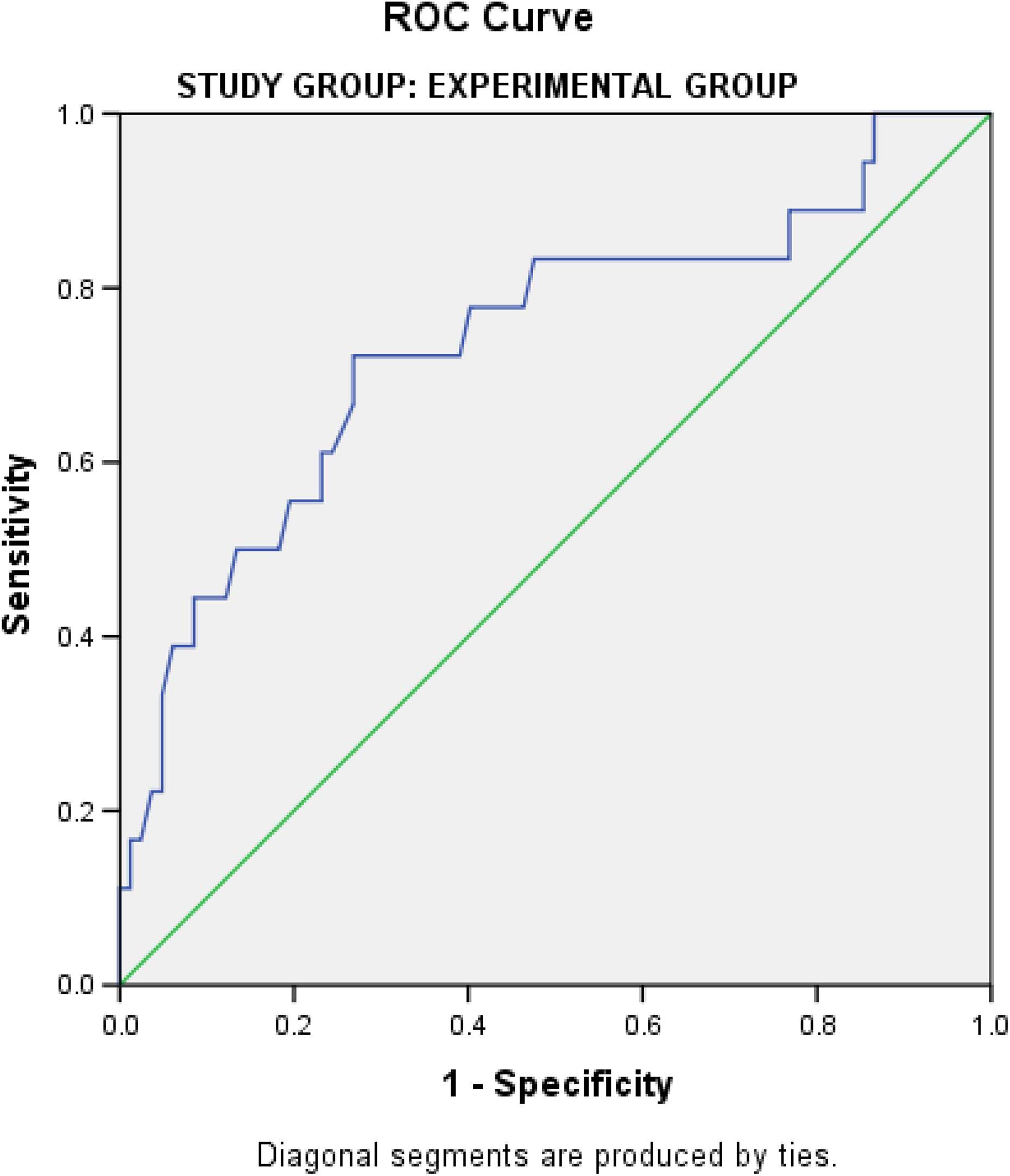
ROC curve to determine the sensitivity and specificity of serum vitamin B12 as a screening test for ovalocytosis in metformin-treated (experimental) type 2 DM patients. Area under the curve = 0.74; sensitivity = 72%; specificity = 66%; serum vitamin B12 ≤ 199pg ml−1; p = 0.002. ROC: receiver operating characteristic; DM: diabetes mellitus.
The prevalence rates for vitamin B12 deficiency among the metformin-treated and metformin-naive type 2 diabetes patients were 41% and 20%, respectively, which revealed a statistically significant difference between both groups (p < 0.001). Furthermore, the prevalence rates for frank anaemia (PCV value <30% with haemoglobin concentration <10 g dl−1) among the metformin-treated and metformin-naive type 2 diabetes patients were 29% and 14%, respectively, which revealed a statistically significant difference between both groups (p < 0.05).
In this study, regarding the metformin-naive participants, 70% were on insulin alone, 10% were on pioglitazone and vildagliptin, 10% were on pioglitazone and glibenclamide, 5% were on pioglitazone and glimepiride, while the remaining 5% were on glibenclamide alone. Concerning the metformin-treated participants, 70% were on metformin and glibenclamide, 10% were on metformin and glimepiride, 5% were on metformin and vildagliptin, 5% were on metformin, vildagliptin and pioglitazone, 5% were on metformin, glibenclamide and pioglitazone, while the remaining 5% were on metformin, glimepiride and pioglitazone. All the participants were also on dietary measures too.
Both case and control groups had fair glucose control using their mean fasting plasma glucose (FPG) at presentation during diagnosis versus recruitment contact for sample collections in this study. The mean FPG at presentation during diagnosis versus recruitment contact for sample collections in this study among the metformin-treated group was 167.8 ± 29.1 mg dl−1 versus 140.8 ± 60.2 mg dl−1 (p ≤ 0.001), respectively. While the FPG at presentation during diagnosis versus recruitment contact for sample collections in this study among the metformin-naive group was 186.8 ± 33.4 mg dl−1 versus 128.1 ± 42.1 mg dl−1 (p ≤ 0.001), respectively. Therefore, the glycaemic index for the participants at the recruitment contact for sample collections in this study may not likely have impacted the outcome of our findings since their mean FPG levels revealed that they were better controlled.
Moreover, there was no statistically significant correlation between the increasing doses of metformin and serum vitamin B12 levels (r = 0.77, p = 0.45). Lastly, there was also no statistically significant correlation between the duration of metformin use and serum vitamin B12 levels (r = 0.002, p = 0.98).
Discussion
As earlier mentioned, this study was designed to elucidate the chronic haemato-toxicologic adverse profile for metformin with respect to its potential to induce vitamin B12 deficiency via reduction in the gastrointestinal absorption of vitamin B12 by performing comparative analyses between the serum vitamin B12 levels and haematological indices among metformin-treated and metformin-naive type 2 diabetic patients attending the outpatient Endocrinology Clinic of ISTH with the rational purpose of alleviating the associated morbidity and mortality. The mean PCV was 33.38 ± 6.0% for metformin-treated type 2DM patients and was not significantly lower compared to the mean PCV of 35.00 ± 6.21% for the metformin-naive participants (p = 0.65). However, ovalocytosis and macrocytosis (high MCV > 100 fL) were present in 18 (18%) participants and 5 (5%) participants, respectively, of the metformin-treated type 2DM group; which imply the contribution of vitamin B12 deficiency to megaloblastic anaemia in these metformin-treated type 2DM participants. Six (6%) of metformin-naive participants were having ovalocytosis with normal MCV, which indeed indicated that ovalocytosis usually preceded the occurrence of macrocytosis and frank anaemia in patients with borderline vitamin B12 status and actual (real) vitamin B12 deficiency in clinical practice settings. However, other features of megaloblastic anaemia, such as hypersegmented neutrophils, did not show any statistically significant occurrence and variation among these metformin-treated and metformin-naive participants with respect to the mean serum vitamin B12 levels among them. Also, neither did MCHC, PCV, haemoglobin concentrations, relative neutrophil percentage, relative lymphocyte percentage nor absolute platelet count showed any statistically significant occurrence and variation among these metformin-treated and metformin-naive participants with respect to the mean serum vitamin B12 levels among them.
This study did reveal remarkable statistically significant difference in the haematological profiles between both groups in the case of a slightly higher mean MCV, MCH and WBC. This may be explained by the fact that clinically, overt features of vitamin B12 deficiency takes a long time of about 5–10 years before manifesting due to large amount of storage in the liver. The causes of anaemia in these patients may also be due to multiple etiological factors. 18,23
It was found that 18 (18%) participants and 5 (5%) participants of the metformin-treated group had ovalocytosis and macrocytosis, respectively. This finding corroborates the study of Kalitsa et al., who found evidence of megaloblastic anaemia among 600 type 2 DM out patients using metformin at general hospitals in Athens, Greece. 19 It has been well established that vitamin B12 deficiency in type 2 diabetic patients caused macro-ovalocytic anaemia and hypersegmented neutrophils. When both conditions coexist, it becomes a major dilemma as to the primary or major contributor to these complications. Evidence of vitamin B12 deficiency, which includes macro-ovalocytosis, low PCV and hypersegmented neutrophils, may rightly point to vitamin B12 deficiency as a contributor to anaemia. Therefore, this emphasis the need for serum vitamin B12 assay in metformin-treated type 2 DM patients to determine the contribution of vitamin B12 deficiency to these mentioned haematological abnormalities. Also, there is need to assess the sensitivity and specificity of serum vitamin B12 assay for the detection of vitamin B12 deficiency in the presence and absence of ovalocytosis, anaemia as well as hypersegmented neutrophils, respectively, among metformin-treated diabetic population. This study revealed that the serum vitamin B12 assay had moderate sensitivity of 72% with moderate specificity of 66% at detecting the presence and absence of ovalocytosis in the presence and absence of frank vitamin B12 deficiency, respectively, among the metformin-treated group. The implication is that this test has a positive potential to predict about 30 (72%) of the 41 participants in the metformin-treated group with ovalocytosis to be due to vitamin B12 deficiency, and the potential to accurately predict 39 (66%) of the 59 participants in the metformin-treated group without ovalocytosis not to be due to vitamin B12 deficiency (i.e. those without vitamin B12 deficiency among the metformin-treated group). The fact that vitamin B12 assay can predict, though having a relatively moderate sensitivity and moderate specificity to determine the presence of ovalocytosis as a consequence of vitamin B12 deficiency, should therefore serve as an indication to screen for serum vitamin B12 deficiency among type 2 DM patients taking metformin by using peripheral blood films (PBFs) to check for the presence of ovalocytosis. It also important to note that ‘serum cobalamin (vitamin B12) assay does not meet the criteria for an ideal test of high sensitivity and high specificity; but, this moderate sensitivity and moderate specificity does not invalidate serum vitamin B12 measurements as a diagnostic marker’. 26 If results are indeterminate, serum methyl malonic acid (MMA) and/or homocysteine levels may be requested. 27,28 Serum vitamin B12 concentrations between 200 pg ml−1 and 400 pg ml−1 are considered borderline, and it is an indication to screen for serum MMA and/or homocysteine levels. Measurement of serum MMA and/or homocysteine concentration(s) is/are recommended for patients with suboptimal vitamin B12 levels (i.e. serum vitamin B12 concentrations ranging from 200 pg ml−1 to 400 pg ml−1 with subtle haematological manifestations). Serum MMA measurement has a sensitivity of 90–95% and specificity of 60%, while homocysteine assay is both less sensitive and less specific as values could be affected by the concentration of serum folate; these however vary with the laboratory methods employed. Serum homocysteine and MMA concentrations of 5–15 µmol l−1 and <0.28 µmol l−1, respectively, are considered to be within the normal reference range. 26 –28
This study did not show any statistically significant correlation between the increasing doses of metformin and serum vitamin B12 levels (r = 0.77, p = 0.45). In addition, there was also no statistically significant correlation between the duration of metformin use and serum vitamin B12 levels (r = 0.002, p = 0.98). This finding was, however, in contrast and discrepancy to the cohort study in Pakistan where the duration and dose of metformin were found to have a statistically significant influence on the serum vitamin B12 levels among the participants (p < 0.05). 28 The reason behind this observational finding may be because the patients in this study were not followed up for a longer period of time to ensure drug (metformin) compliance, as this may explain the fact that clinically overt features of vitamin B12 deficiency takes a long period of time, about 5–10 years before manifesting due to large amount of hepatic storage. In addition, the serum vitamin B12 measurement was done at one particular point in time during this study as it was a short time prospective observational study of about 11-month duration.
According to the study conducted by Bauman et al., 25 the easiest way to lower the risk of vitamin B12 deficiency in diabetic patients treated with metformin is through proper and adequate supplementation with calcium, as increase intake of calcium reverses vitamin B12 malabsorption induced by metformin. The precise mechanism by which metformin causes vitamin B12 deficiency has not been clearly elucidated; however, proposed mechanisms include alterations in small bowel motility due to calcium deficiency which stimulate bacterial overgrowth and consequential vitamin B12 malabsorption. Metformin has also been shown to inhibit the calcium-dependent endocytosis absorption process of vitamin B12-IF of castle complex at the terminal ileum. This inhibitory effect is reversed by calcium supplementation.
Finally, the limitation of this present study was that the patients were not followed up for a longer period of time to ensure metformin drug compliance. In addition, the serum vitamin B12 measurement was done at one particular point in time as it was a short time prospective observational study of about 11-month duration. It also aimed to look at the effect of chronic metformin therapy on serum vitamin B12 levels but not folate. Chronic metformin therapy does not contribute to folate deficiency; however, folate deficiency may have contributed to megaloblastic (macro-ovalocytic) anaemia among some of the participants but this was not within the scope of our study design. This study did not assess for the level of HbA1C among the participants since it was not part of the scope of this research work and also the glycaemic index for the participants at recruitment contact for sample collections in this study may not likely have impacted the outcome of our findings because their mean FPG levels revealed that they were better controlled. It is also important to note that it takes quite a long duration of time, which is about 5–10 years for vitamin B12 deficiency to manifest during laboratory measurement or clinically due to hepatic storage compared to HbA1C which measures plasma glucose control over a period of 3-month duration. In addition, these other antidiabetic medications that were coadministered with metformin in this study have not been shown by any previous research to affect serum vitamin B12 levels in anyway. A long-term assessment of relationship between serum vitamin B12 level and metformin use using a prospective observational longitudinal cohort study design to monitor vitamin B12 deficiency, progression and associated laboratory (haematological) abnormalities and clinical features would have been desirable. Also, intrinsic factor (IF) assay, as well as transcobalamin profile assay, serum methyl malonic acid (MMA) and serum homocysteine levels would have been desirable.
Conclusion
In this study, the occurrence of chronic haemato-toxicologic adverse effects of metformin on mean serum vitamin B12 level, ovalocytosis, MCV, MCH and total WBC count was remarkable and clinically significant among the metformin-treated compared to the metformin-naive type 2 diabetic patients. The serum vitamin B12 assay had moderate sensitivity of 72% with moderate specificity of 66% at detecting the presence and absence of ovalocytosis in the presence and absence of frank vitamin B12 deficiency, respectively, among the metformin-treated group. Vitamin B12 supplementation would be helpful in metformin-treated type 2 diabetes patients via the parenteral route of administration, most preferably the intramuscular site injection; in order to prevent the occurrence of vitamin B12 deficiency among them. Lastly, we recommend the use of serum vitamin B12 assay and CBC with peripheral blood films (PBFs) as a reliable way to diagnose and screen for vitamin B12 deficiency among metformin-treated type 2 DM patients.
Footnotes
Authors’ note
The authors alone are responsible for the contents and writing of this research manuscript.
Acknowledgements
The authors would like to thank and appreciate all the staffs at outpatient Endocrinology Clinic of ISTH for their immense assistance during the course of this study. Their support was of paramount importance to the completion of this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.