Abstract
Pharmacokinetic studies on the undiluted insect repellent active ingredient, DEET, were assessed for relevance to exposure assessment for humans. Five studies designed to define the DEET plasma concentration profiles of systemic exposure occurring at the respective NOAELs for two laboratory species using dermal and/or oral exposure scenarios and human volunteers at the 95th percentile of consumer use were undertaken to support EPA re-registration. The studies reported herein demonstrate that the DEET plasma concentration for Sprague-Dawley rats and Beagle dogs at the respective NOAELs following oral bolus dosing is much greater in terms of Cmax and AUC than that achieved following human dermal administration at the 95th percentile of consumer use. In addition, these plasma level data confirm that a high-dose rat subchronic dermal toxicity study with DEET is the most relevant model for human safety assessment of this topically applied insect repellent active ingredient. Therefore, utilization of pharmacokinetic plasma level data is important to exposure assessment of DEET and significantly reduces uncertainty in human health safety assessment.
Research highlights
• High DEET plasma levels occur after oral bolus dosing in rats and dogs • These oral doses yield higher DEET plasma levels than possible via dermal application to humans in consumer products • Rat subchronic dermal toxicity data is most relevant for DEET human safety assessment
Introduction
N,N-diethyl-m-toluamide (DEET, CAS #134-62-3, EINECS #205-149-7) is the major component of most topically applied insect repellents used by the public. First developed by the U.S. Department of Agriculture for use by military personnel in 1946, DEET was registered for use in the general population in 1957 and re-registered in 1998.1,2 Clinical studies have demonstrated consistently that DEET is the most effective repellent for biting insects, mosquitoes, and arthropods such as ticks. 3 DEET concentrations in insect repellent products range from 5 to 100%. Estimates show that approximately 30% of the population uses one or more insect repellent products containing DEET on an annual basis. 2
This article was prepared for two reasons: 1) present previously unpublished pharmacokinetic data in multiple species from studies conducted with DEET during the EPA Reregistration program and 2) compare DEET plasma level data in multiple species following dose administration via the dermal or oral route of administration, to improve human health safety assessment. Note that the article is not intended to present a quantitative human health risk assessment nor a comprehensive review of the published toxicology or clinical safety data for DEET.
DEET has been the subject of many animal toxicology studies and sporadic human case reports over the past 50 years.4–7 A subject of interest has been whether DEET has the potential to elicit acute clinical signs of narcosis at high dose levels relative to those from normal and intended topical exposure of consumers using commercial products. Although toxicology studies have demonstrated that DEET is not a selective neurotoxicant, 8 after bolus oral administration some evidence of acute transient clinical signs was observed in the following animal studies conducted for EPA Reregistration:
The data presented herein are from studies conducted to Good Laboratory Practices (GLP) and Good Clinical Practices (GCP), which were submitted for the EPA Reregistration of DEET, and consequently underwent a rigorous regulatory scientific review process. In addition, these studies were all submitted and underwent equally rigorous regulatory scientific review processes in Canada and the European Union. The purpose of the reported pharmacokinetic studies was to assess the relevance of these dog and rat toxicology studies using oral administration to safety assessment for humans using topically applied DEET insect repellent products. Five pharmacokinetic studies designed to define the DEET plasma concentration profiles of systemic exposure occurring for three species and seven different exposure scenarios were undertaken. The species and exposure scenarios included: • Humans - Following single and repeated dermal applications of DEET at the 95th percentile of human exposure based on normal and intended consumer use;
11
• Beagle Dogs - Under the exposure conditions at which transient acute clinical signs were observed (including abnormal head movements, ptyalism, emesis and/or relaxed nictitating membrane within 60 min of dosing)9,10 following single and repeated oral dose administration via gelatin capsules;
12
• Charles River CD® (Sprague-Dawley) Rats - Under the exposure conditions at which slight transient clinical effects were observed (i.e. increased response time to heat in the thermal response test and slightly decreased rearing activity in the motor activity test at the 1-h post-treatment observation interval)
8
following acute oral administration by gastric gavage;13,14 • Charles River CD® (Sprague-Dawley) Rats - Following single and repeated dermal applications of DEET, i.e. under the exposure conditions of the 90-days dermal toxicity study considered most relevant to human safety assessment.
15
The background, methods and results of these blood level studies are discussed below.
Human plasma level study 11
Background
DEET is widely used by consumers to prevent biting by insects. The 95th percentile of consumer use is approximately 3 g/day and 4 g/day for females and males, respectively (derived from data presented in a study submitted to US EPA). 16 These doses were selected for the human plasma level study as they represent a plausible upper bound application rate for DEET.
Methods
Three healthy female and three healthy male subjects were enrolled in the 4-days study. The subjects ranged from 21-36 years and weighed between 45-55 kg (females) and 65–75 kg (males). All subjects were regular users of products formulated with DEET. 11 Obtaining ethical clearance for studies involving volunteer human subjects has been restricted for many years; therefore, the number of volunteer subjects was limited in agreement with the U.S. EPA. The intention of developing human volunteer plasma level profiles was to have representative data for comparisons to other species, not to define the pharmacokinetic profile of DEET for the consumer population using insect repellents.
The study was conducted in accordance with the clinical research guidelines established by the Health Protection Branch (HPB) Drugs Directorate of Health Canada, 17 the Medical Research Council of Canada, 18 the Basic Principles as defined in the US Code of Federal Regulations 21 CFR Part 312.120, as well as the Declaration of Helsinki. The informed consent forms and the protocol were reviewed by the laboratory’s independent Institutional Review Board (IRB). The IRB operated according to requirements outlined in the HPB Drugs Directorate “Conduct of Clinical Investigations”, Medical Research Council of Canada “Guidelines on Research Involving Human Subjects” and the US Code of Federal Regulations (21 CFR Part 56).
Undiluted DEET was applied at the 95th percentile of consumer use (as described above) 16 once per day for four consecutive days at a level of 3 g/day for females and 4 g/day for males to evaluate any differences in plasma levels following acute and repeated exposure scenarios. DEET was applied to both legs and one arm of each subject using glass syringes and spread evenly by a clinical technician wearing a polyethylene glove. DEET was not applied to the arm from which blood was drawn. The amount of DEET applied was sufficient to result in a wet appearance, much like occurs with normal consumer application of DEET insect repellent products, but not enough that dripping or runoff occurred. The application sites were left uncovered.
Subjects wore laboratory supplied white, unbleached cotton tee shirts and shorts for the duration of each daily application period (8 h). They engaged in normal activity for 8 h after the application of DEET and then showered with warm water (27–32°C) and Ivory® soap for approximately 4–16 min. Subjects resided in the laboratory’s residential facility and ate regular meals at scheduled times for the entire duration of the study.
Blood samples were drawn into Vacutainer tubes containing ethylenediaminetetraacetic acid (EDTA) on days 1 and 4, prior to DEET application and at the following times after application: 0.5, 1, 1.5, 2, 3, 4, 6, 8 (immediately before showering), 8.5 (after showering), 9, 10, 12, 14, 16, 20 and 24 h. Day 2 and 3 samples were drawn prior to DEET application and at 8 h (immediately before showering) and 8.5 h (after showering). Samples were placed in an ice bath and centrifuged under refrigeration as soon as possible. Plasma was stored at <-10°C until the time of analysis. DEET was extracted from plasma and analyzed by high-performance liquid chromatography and a UV detector (HPLC-UV) with a lower limit of quantitation of 49.6 ng/mL plasma.
The mean, standard deviation, and coefficient of variation were calculated for the following parameters: • Maximum plasma concentration after DEET application (Cmax) • Apparent first order plasma elimination rate constant (kel) • Half-life for DEET elimination from plasma t1/2el
These were calculated separately for males and females on days 1 and 4, and for males and females following pooling of day 1 and day 4 data.
Pooled male and female data were used to calculate the mean, standard deviation, and coefficient of variation for the following parameters for day 1 and day 4: • Area Under the Curve (AUC0-t) • Area Under the Curve (AUC0-inf) • Time of maximum plasma level (tmax)
Values for Cmax, kel, t1/2el for males and females were compared within a given test day and with pooled data from days 1 and 4 using a two-sample t-test. Comparisons between sexes with pooled data from days 1 and 4 were performed using a repeated measures analysis. Comparisons between day 1 and day 4 for males, females, and pooled male and female data were performed using a paired t-test.
Results
No adverse reactions were observed in the test subjects at any time during the study.
DEET was first detected in the plasma (lower limit of quantitation = 49.6 ng/mL) at 3–4 h after application on days 1 and 4 in females and between 0.5 h and 3 h after application in males. DEET plasma levels generally increased slowly during the 8-h exposure period and reached peak immediately before or after showering at 8 h. Following showering, plasma levels gradually decreased to levels near or below the limit of quantitation prior to the next application (Figure 1(a) and (b)). (a) DEET plasma profile in human volunteers administered DEET dermally at the 95th percentile of human use during and following the first of four daily 8-h dermal applications. Each value represents the mean ± SE of six individual values (three males and three females). (b) DEET plasma profile in human volunteers administered DEET dermally at the 95th percentile of human use during and following the fourth of four daily 8-h dermal applications. Each value represents the mean ± SE of six individual values (three males and three females).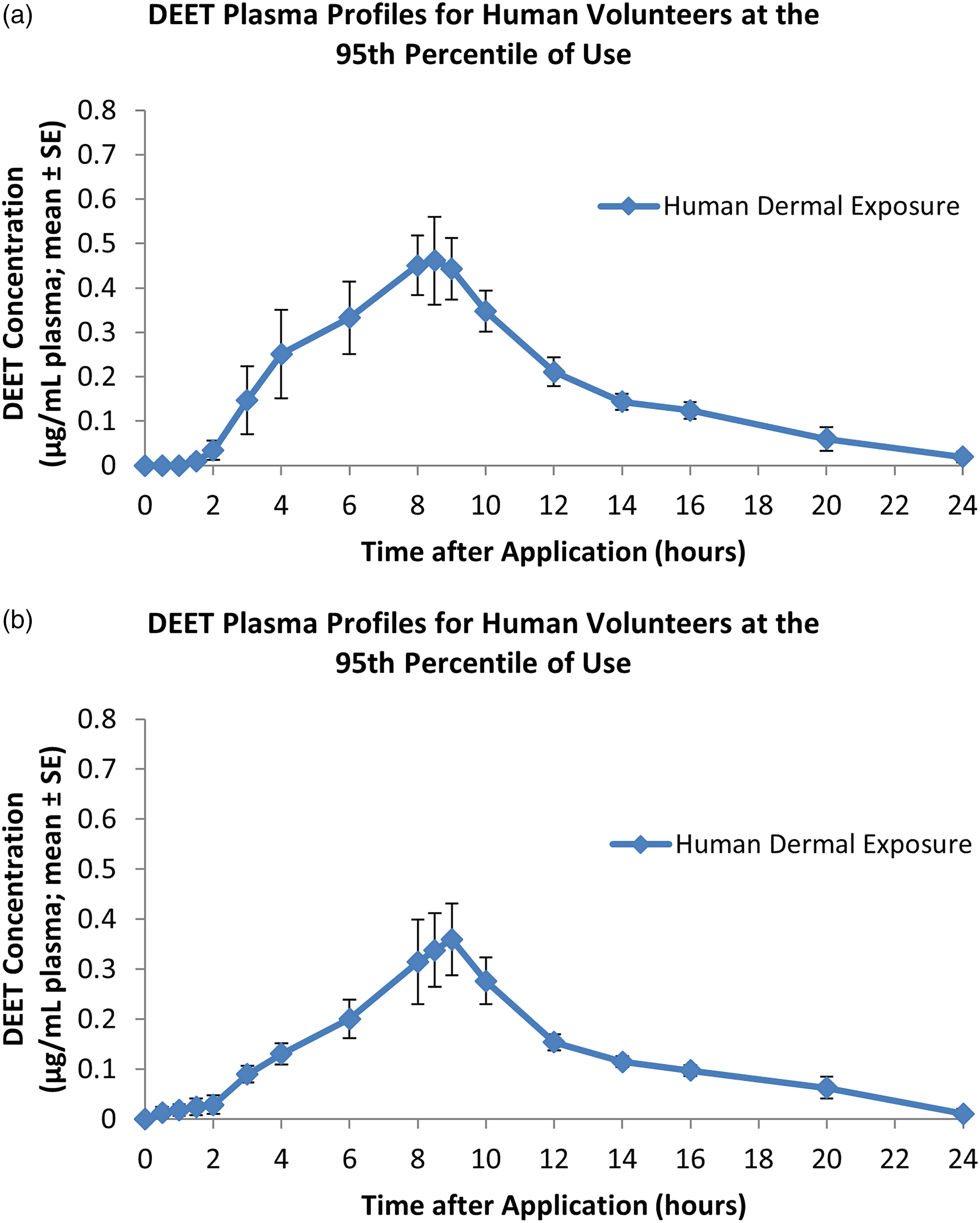
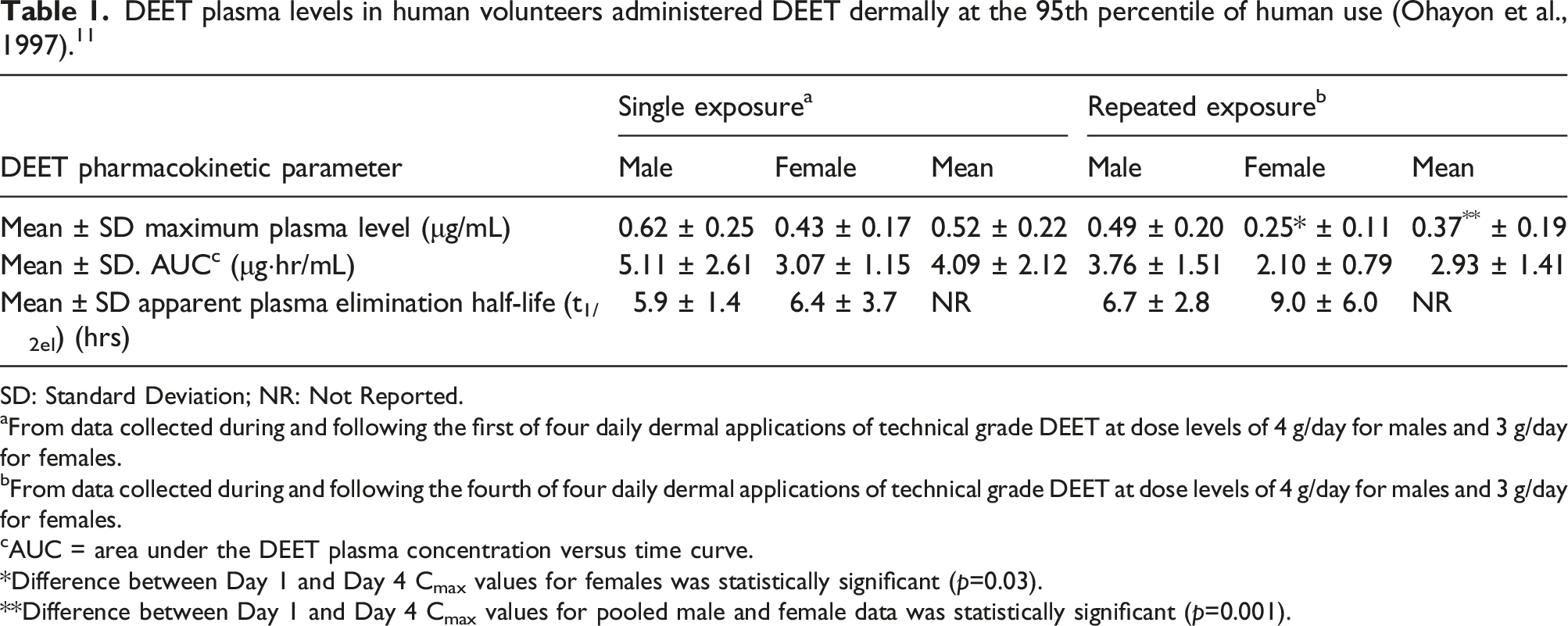
DEET plasma levels in human volunteers administered DEET dermally at the 95th percentile of human use (Ohayon et al., 1997). 11
SD: Standard Deviation; NR: Not Reported.
aFrom data collected during and following the first of four daily dermal applications of technical grade DEET at dose levels of 4 g/day for males and 3 g/day for females.
bFrom data collected during and following the fourth of four daily dermal applications of technical grade DEET at dose levels of 4 g/day for males and 3 g/day for females.
cAUC = area under the DEET plasma concentration versus time curve.
Difference between Day 1 and Day 4 Cmax values for females was statistically significant (p=0.03).
Difference between Day 1 and Day 4 Cmax values for pooled male and female data was statistically significant (p=0.001).
The mean apparent plasma elimination half-life for females was 6.4 ± 3.7 h and 9.0 ± 6.0 h on day 1 and day 4, respectively. For males, the mean apparent plasma elimination half-life was 5.9 ± 1.4 h and 6.7 ± 2.8 h on day 1 and day 4, respectively. Although plasma levels for females were lower, no statistically significant differences were found between sexes for maximum plasma concentration or between sexes or study day for the apparent plasma elimination half-life. The overall mean maximum plasma level (single and repeated exposure) was 0.45 μg/mL and the overall mean AUC was 3.5 μg·hr/mL (Table 1). 11
Dog plasma level study 12
Background
Dermal toxicity studies are not feasible with dogs; therefore, studies were conducted via the oral route of administration. Preliminary studies conducted in preparation for a 1-year oral toxicology study in Beagle dogs revealed palatability issues limiting the dose level of DEET that could be administered in the diet to approximately 75 mg/kg/day (3000 ppm). Subsequent to these findings, three dose range-finding studies were conducted to determine the possibility of achieving higher doses if DEET was administered in oral bolus doses using gelatin capsules. The numbers of dogs used per group were kept to a minimum, as with human volunteers, for ethical considerations.
In the first dose range-finding study, DEET was administered orally as a single gelatin capsule daily at levels of 0, 62.5, 125, 250, and 500 mg/kg/day for 2 weeks (one male and one female per group). 9 At 250 and 500 mg/kg/day emesis, ptyalism and nodding or twitching of the head and neck were observed occasionally. The male also exhibited ptosis, ataxia, and convulsions at 500 mg/kg/day. Clinical signs were observed shortly after dosing and were not observed after 1 hr post-dosing.
In the second dose range-finding study, DEET was scheduled to be administered daily for a period of 8 weeks as a single oral bolus dose via gelatin capsules at levels of 0, 75, 125, 175, and 225 mg/kg/day (two males and two females per group). 19 At dose levels ≥125 mg/kg/day, emesis, ptyalism, abnormal biting and scratching, and abnormal head movements were observed in one or more animals in each group during the first 5 days of the study. Ataxia and ptosis also were observed in some dogs at 175 and 225 mg/kg/day. In addition, convulsions were observed following the first dose in a female dog in the 225 mg/kg/day dose group and following the third dose in a male dog in the 125 mg/kg/day dose group. As in the first dose range-finding study, clinical signs occurred shortly after dosing, after which rapid recovery was observed. The study was terminated after these 5 days of dosing. The no observable adverse effect level (NOAEL) for the acute transient clinical signs was 75 mg/kg/day.
In a follow-up dose range-finding study also conducted in Beagle dogs (two males and two females per group), the daily dose of DEET was administered orally via gelatin capsules in two equally divided doses (one in the morning and one in the afternoon, approximately 5.5 h apart) at total dose levels of 0, 50, 100, 200, and 400 mg/kg/day for a period of 8 weeks. 10 Test-article related findings of abnormal head movements and/or pytalism were observed within 1 hr of dosing at 200 and 400 mg/kg/day.
A subsequent pharmacokinetics study was conducted to compare the systemic exposure received by Beagle dogs at the NOAEL (75 mg/kg/day) following oral bolus dosing to that of human volunteers under intended and normal conditions of use. 12
Methods
A pharmacokinetics study was conducted in four male and four female Beagle dogs aged 7 months old and weighing 6.1–7.7 kg (males) and 6.5–6.8 kg (females). Study animals were administered undiluted DEET as a daily oral bolus dose via gelatin capsules for 4 days at the NOAEL for clinical signs in the previous dog dose range-finding studies, i.e. 75 mg/kg/day. DEET plasma levels were profiled on the first and last day of this exposure with blood samples collected at 10, 20, 30, 45, 60, and 90 min, and 2, 3, 4, 6, and 24 h post-dosing. 12
Results
Peak plasma levels were observed within 30 min after dosing (based on mean Tmax for males and females) and plasma levels returned to below the limit of quantitation within 3–4 h after dose administration (Figure 2(a) and (b)). Similarly as with humans, although DEET plasma levels were lower in females, no significant differences were observed in mean maximum plasma level (μg/mL) or AUC (μg hr/mL) between male and female dogs and there was no evidence of accumulation of DEET in the blood following repeated doses (Table 2). The overall mean maximum plasma level (single and repeated exposure) was 14.7 μg/mL. The overall mean AUC was 12.67 μg·hr/mL. The overall mean apparent plasma elimination half-life (t1/2el) was 0.46 h.
12
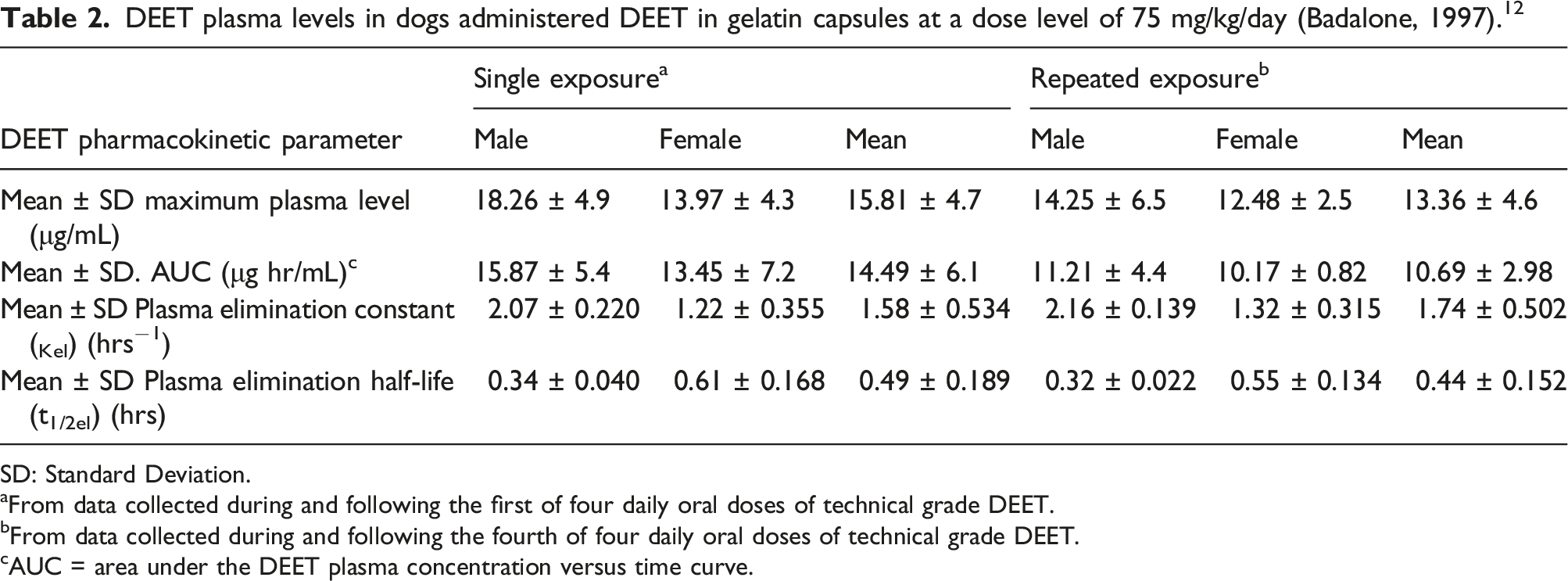
(a) DEET plasma profile in dogs during and following the first of four daily oral bolus doses of DEET at a level of 75 mg/kg/day. Each value represents the mean ± SE of eight individual values (four males and four females). (b) DEET plasma profile in dogs during and following the fourth of four daily oral bolus doses of DEET at a level of 75 mg/kg/day. Each value represents the mean ± SE of eight individual values (four males and four females). DEET plasma levels in dogs administered DEET in gelatin capsules at a dose level of 75 mg/kg/day (Badalone, 1997).
12
SD: Standard Deviation. aFrom data collected during and following the first of four daily oral doses of technical grade DEET. bFrom data collected during and following the fourth of four daily oral doses of technical grade DEET. cAUC = area under the DEET plasma concentration versus time curve.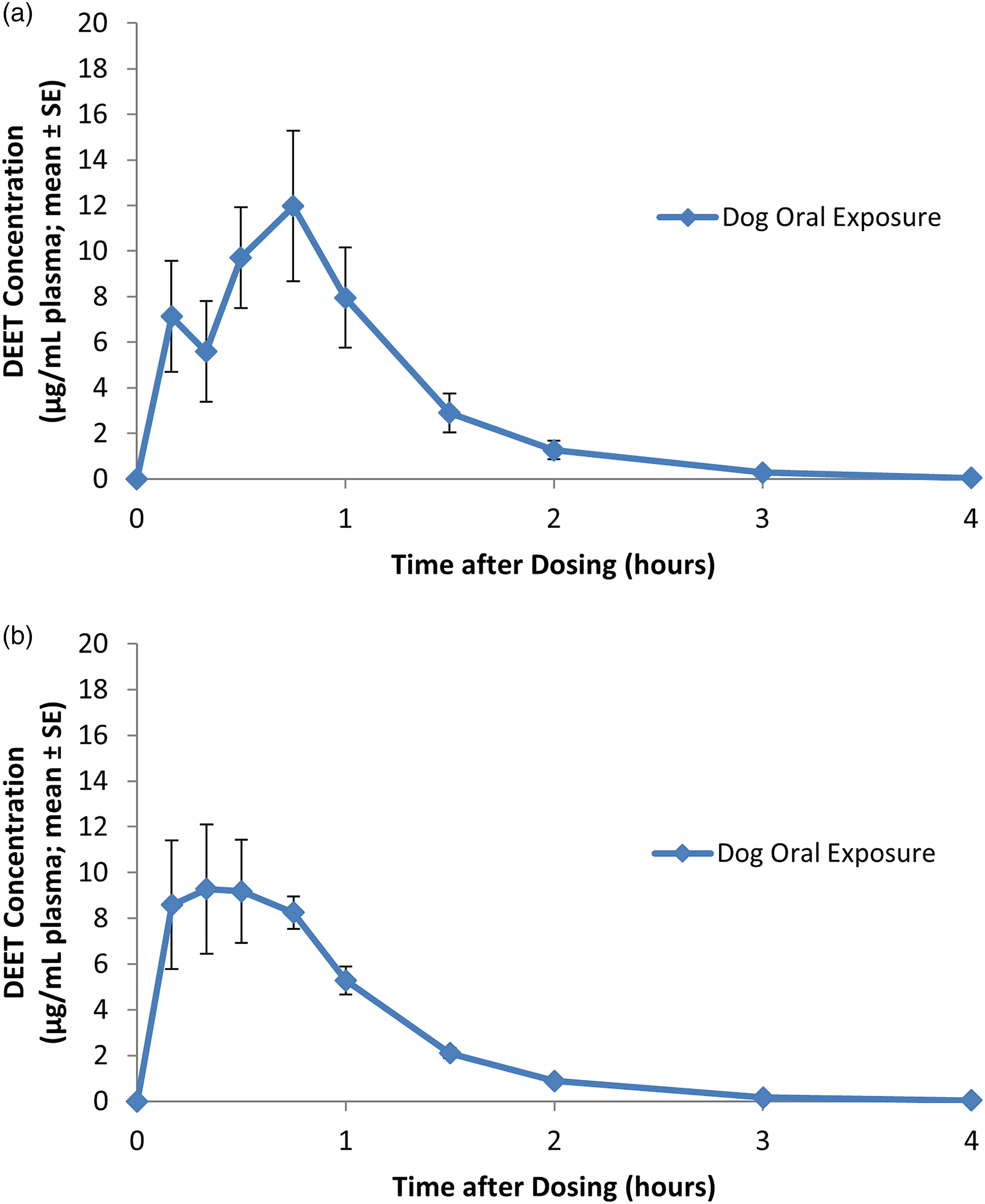
Rat plasma level studies13,14,15
Background
A pharmacokinetics study was conducted to determine the plasma profile in rats dosed with DEET under the conditions of a previous toxicology study in which transient clinical signs were observed following acute oral administration by gastric gavage. The potential effects evaluated included an increase in time to respond in a thermal response test and decreased rearing activity detected by two measures of vertical movement (vertical activity and vertical time) in a motor activity test. These findings were observed in animals of both sexes, but only at the 1-h post-treatment time point and only at the 500 mg/kg dose level. 8 The NOAEL for acute transient clinical signs in rats was 200 mg/kg. In order to compare the systemic exposure to DEET that rats received under the dosage regimen of this study to that of volunteer subjects under simulated conditions of human use, a pharmacokinetics study was conducted at the highest level at which no effects were observed in the rat study, i.e. 200 mg/kg.
In addition, since normal and intended use of DEET by consumers consists of dermal application, a pharmacokinetics study was conducted to determine the plasma profile of DEET in rats dosed under the conditions of the 90-days dermal toxicity study. Because it is recognized that rat skin is more permeable to a lipophilic substance like DEET than human skin, dermal exposure to DEET in the rat would be expected to exceed human systemic exposure by the dermal route, thus providing conservative data for safety assessment. The NOAEL for rat subchronic systemic toxicity was 1000 mg/kg/day (the highest achievable dose tested). 20
Methods
For two of the pharmacokinetics studies, rats were orally dosed once with undiluted DEET via gastric gavage at the NOAEL of 200 mg/kg.13,14 A third study was conducted with dermally applied DEET (undiluted) at a dose level of 1000 mg/kg/day. 15
Study 1
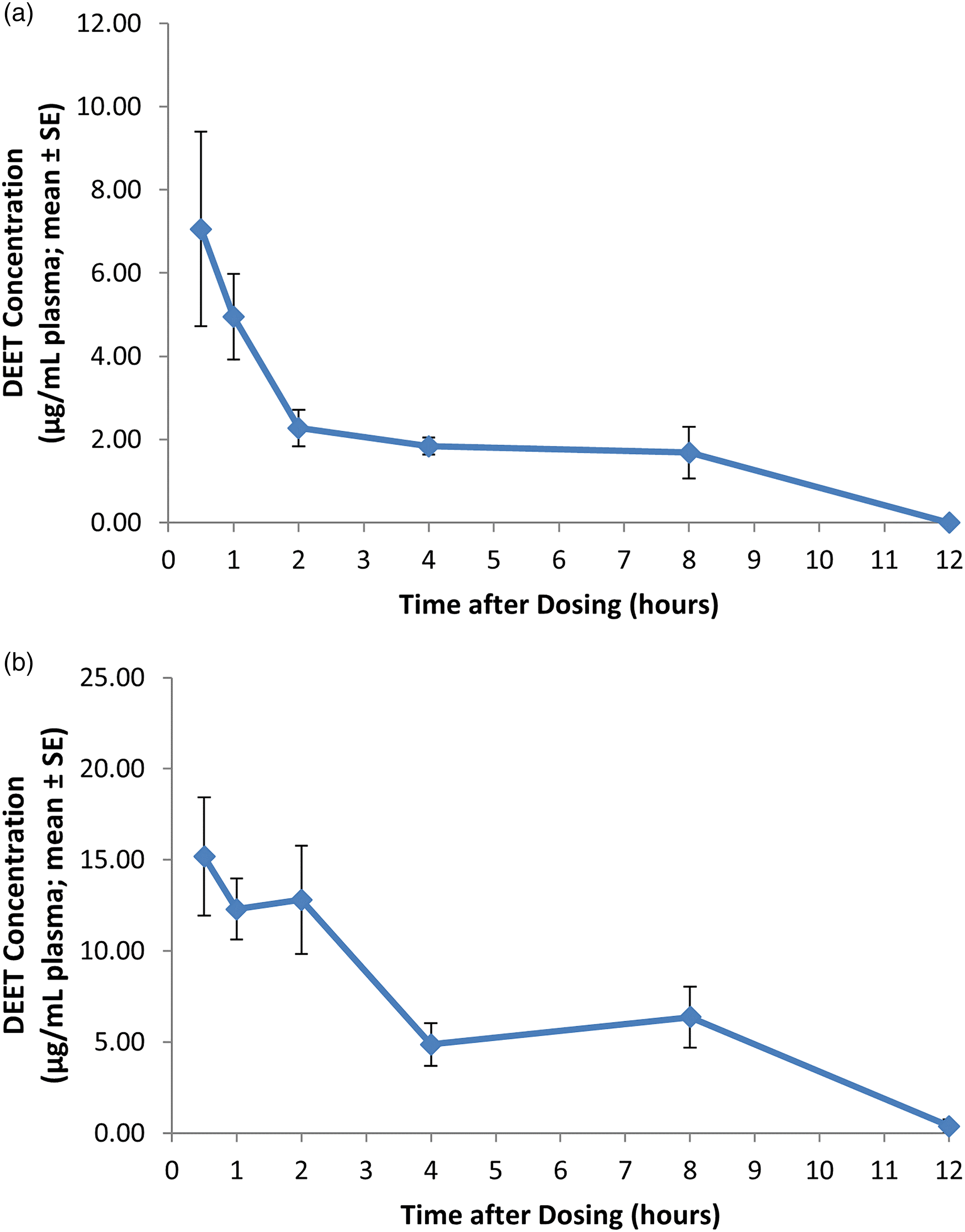
Eight groups (three male and three female Sprague-Dawley rats per group, 10 weeks of age and weighing 201–248 g (females) and 341–389 g (males) on the day of dosing), were employed in this study. One group of rats was sacrificed for blood collection and subsequent DEET plasma analysis at each of eight different time intervals over a 4-h time period (0.25, 0.5, 0.75, 1, 1.5, 2, 3, or 4 h post-dosing) following oral dose administration of DEET at 200 mg/kg. 13
Study 2
The same dose level (200 mg/kg) was administered as Study 1; however, the DEET plasma profile was evaluated over a 48-h period. Eight groups of Sprague-Dawley rats (five male and five female rats per group), 10–11 weeks of age and weighing 223–237 g (females) and 368–383 g (males) on the day of dosing, were sacrificed at each time interval (0.5, 1, 2, 4, 8, 12, 24, or 48 h post-dosing). 14
Study 3
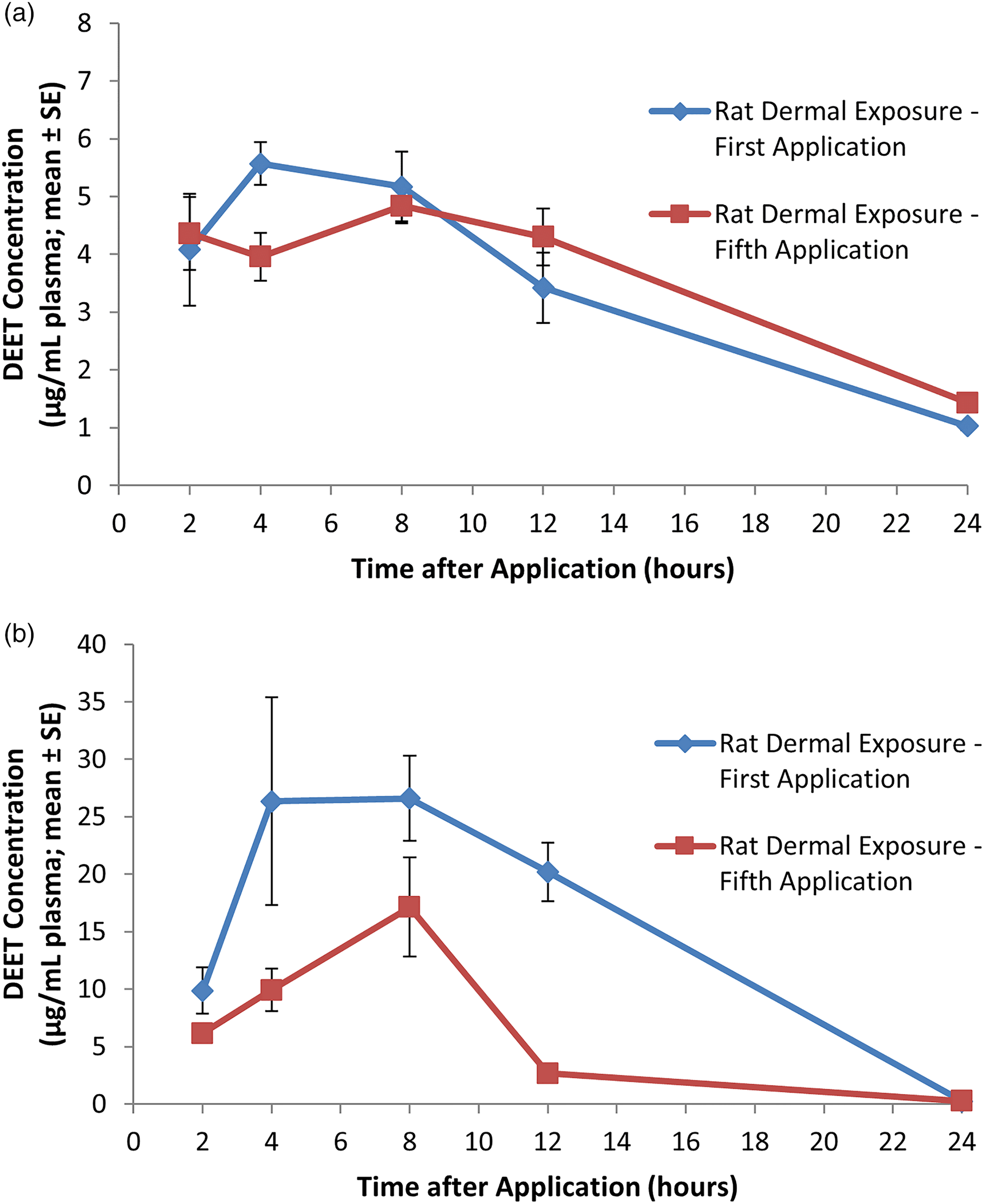
Five groups (four male and four female Sprague-Dawley rats per group) received a single application or five consecutive daily applications of undiluted DEET at 1000 mg/kg/day. 15 Study animals were approximately 9 weeks of age and weighed 195–245 g (females) and 298–362 g (males) on the first day of dosing. The application site remained uncovered and was not rinsed between repeated applications. Blood was collected from four males and four females at 2, 4, 8, 12, and 24 h following the first or fifth application for determination of DEET plasma concentration.
Results
The DEET plasma profiles for male and female rats under the scenarios described above are presented in Figures 3(a) and (b), 4(a) and (b), and 5(a) and (b) for Study 1, 2 and 3, respectively. (a) DEET plasma profile over a 4-h period in male rats following a single oral bolus dose of DEET at a dose level of 200 mg/kg. Each value represents the mean ± SE of three individual values. (b) DEET plasma profile over a 4-h period in female rats following a single oral bolus dose of DEET at a dose level of 200 mg/kg. Each value represents the mean ± SE of three individual values. (a) DEET plasma profile over a 12-h period in male rats following a single oral bolus dose of DEET at a dose level of 200 mg/kg. Each value represents the mean ± SE of five individual values. DEET levels in plasma were measured at 24 and 48 h and were lower than values at 12 h (data not shown). (b) DEET plasma profile over a 12-h period in female rats following a single oral bolus dose of DEET at a dose level of 200 mg/kg. Each value represents the mean ± SE of five individual values. DEET levels in plasma were measured at 24 and 48 h and were lower than values at 12 h (data not shown). (a) DEET plasma profile in male rats during and following the first and fifth of five consecutive dermal applications of DEET at a dose level of 1000 mg/kg/day. Each value represents the mean ± SE of four individual values. (b) DEET plasma profile in female rats during and following the first and fifth of five consecutive dermal applications of DEET at a dose level of 1000 mg/kg/day. Each value represents the mean ± SE of four individual values.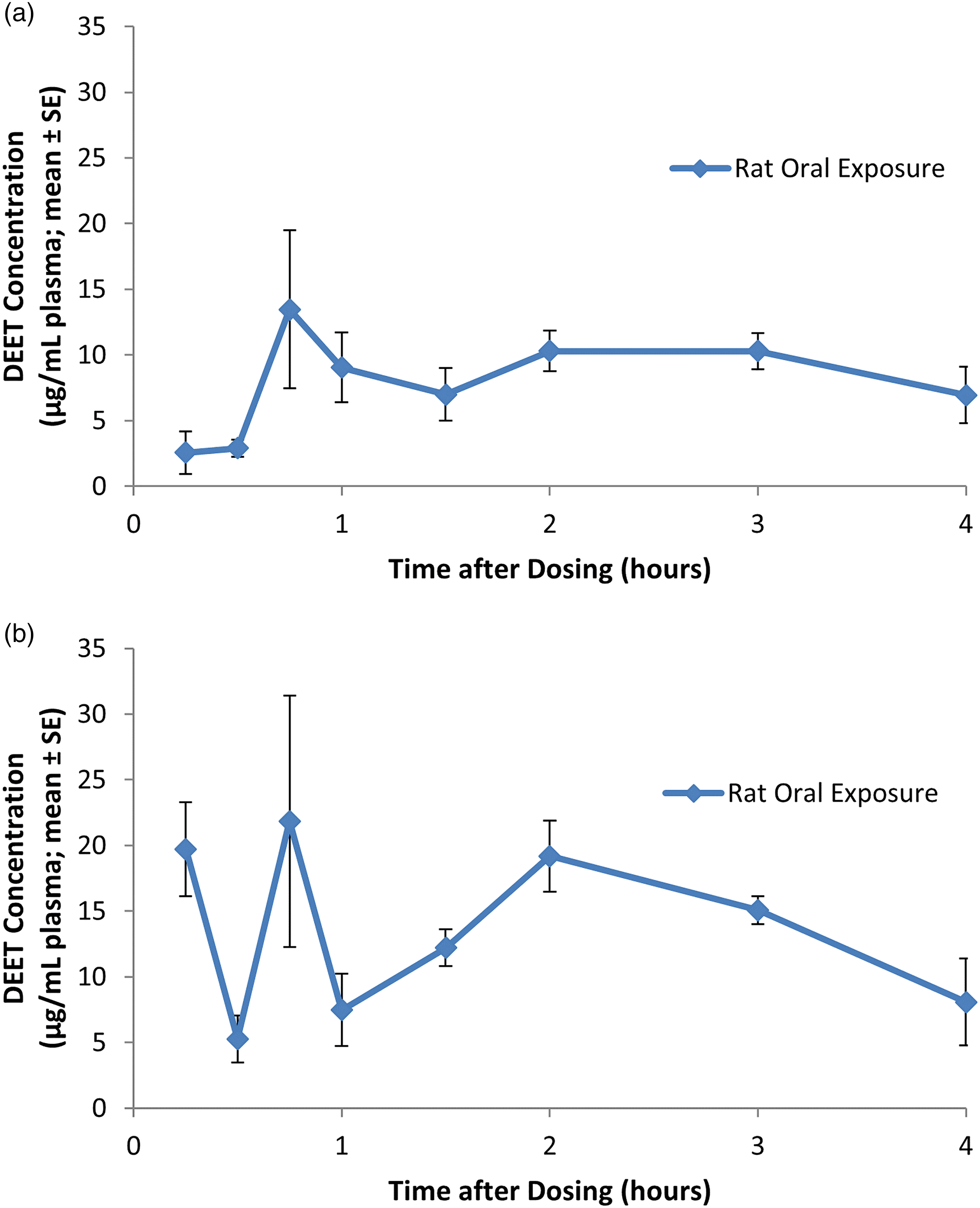
Study 1
DEET plasma levels in rats administered DEET by gastric gavage at a dose level of 200 mg/kg during a 4-h period following dosing (Laveglia, 1998a). 13
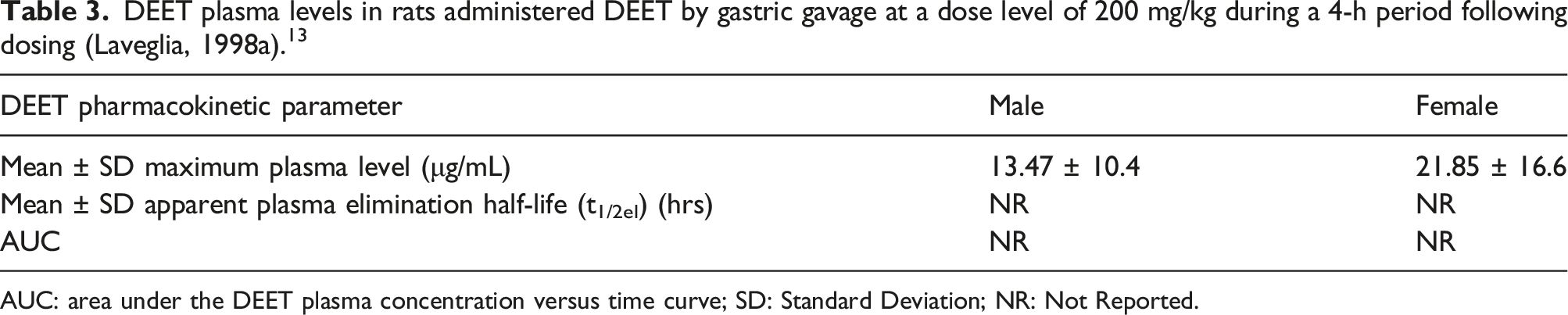
AUC: area under the DEET plasma concentration versus time curve; SD: Standard Deviation; NR: Not Reported.
Study 2
DEET plasma levels in rats administered DEET by gastric gavage at a dose level of 200 mg/kg during a 48-h period following dosing (Goldenthal, 1999). 14
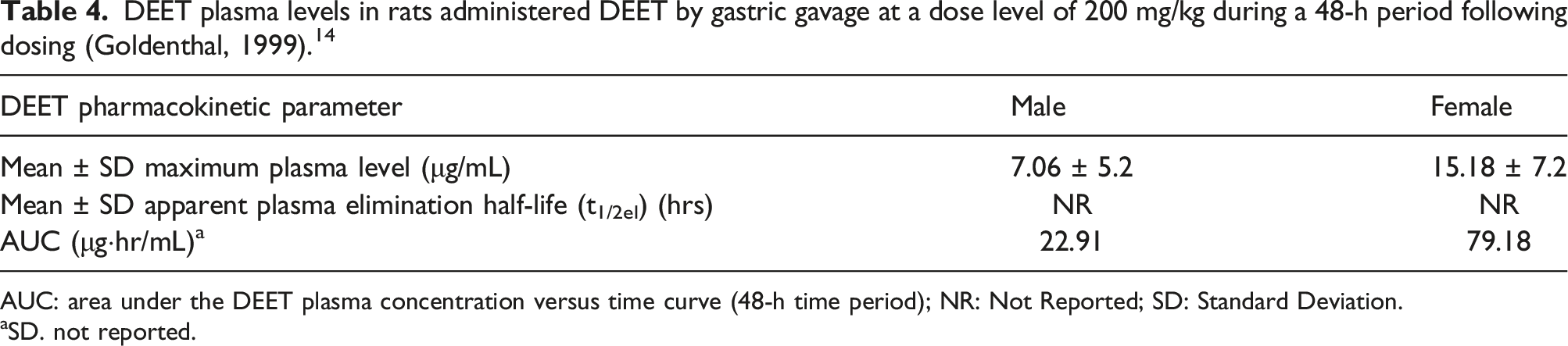
AUC: area under the DEET plasma concentration versus time curve (48-h time period); NR: Not Reported; SD: Standard Deviation.
aSD. not reported.
Study 3
DEET plasma levels in rats administered DEET dermally at 1000 mg/kg/day during a 24-h period following dosing (Laveglia, 1998b). 15
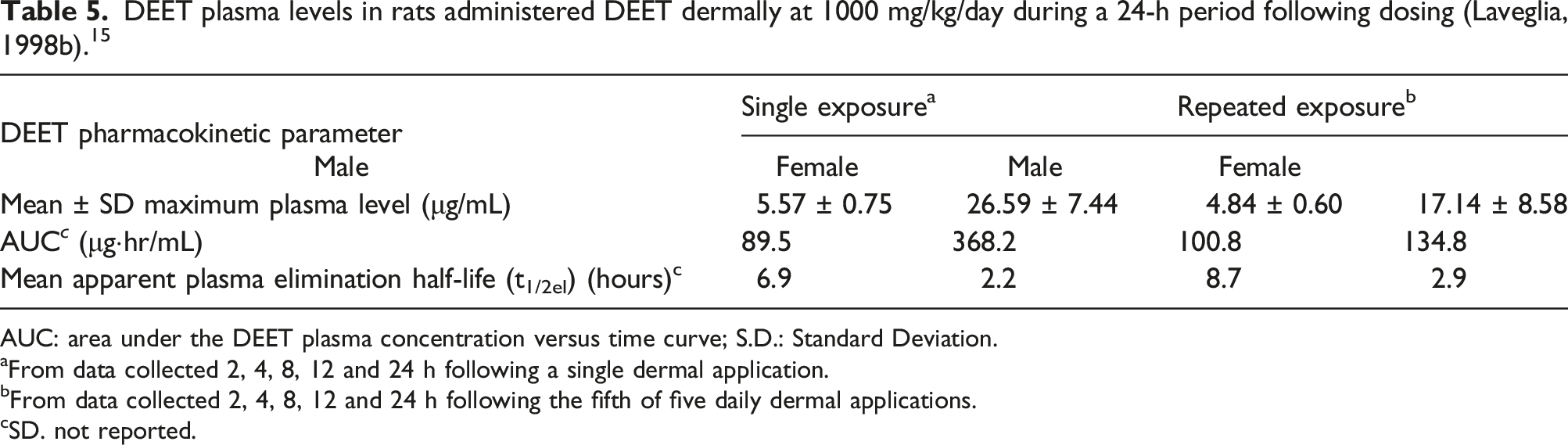
AUC: area under the DEET plasma concentration versus time curve; S.D.: Standard Deviation.
aFrom data collected 2, 4, 8, 12 and 24 h following a single dermal application.
bFrom data collected 2, 4, 8, 12 and 24 h following the fifth of five daily dermal applications.
cSD. not reported.
A different pattern was observed in the female rats that received five consecutive dermal applications of DEET. In these animals, a plateau in DEET plasma concentrations was not observed. Rather, DEET plasma concentrations increased from 6.17 μg/mL at the 2-h sampling interval to 17.14 μg/mL (Cmax) at 8 h after dose administration. At the 12-h sampling interval, the average DEET plasma levels had decreased to 2.66 μg/mL (Figure 5(b)). 15
Mean apparent plasma elimination half-life (t1/2el) was higher in male rats (6.9 and 8.7 h following single and repeated exposures, respectively) compared to female rats (2.2 and 2.9 h following single and repeated exposures, respectively). 15
Discussion and applicability of results to safety assessment
Overview of pharmacokinetics study data in humans, dogs and rats
As human volunteer studies with pesticides/biocides are no longer permitted, the data derived from the five DEET pharmacokinetic parameter studies described herein, in multiple species (humans, rats, and dogs) and via dermal and oral routes of administration, are unique from a regulatory and scientific perspective for an active ingredient dermally applied in consumer products. The characterization of the systemic exposure to DEET as indicated by plasma levels provide important insights relevant for the safety assessment of DEET as used by consumers and demonstrates two points. First, the systemic exposure that laboratory animals receive following oral dose administration (especially oral bolus dosing) is much greater than potential systemic exposure in humans by the dermal route of exposure, even at the 95th percentile of consumer use rate (see Figures 6 and 7). Second, these DEET plasma level data demonstrate that the subchronic dermal toxicity study conducted in rats provides the most relevant and meaningful data for human health safety assessment.
Appropriate toxicology study for safety assessment and rationale
Consistent with the findings of Mount et al.,
21
it is clear that the DEET-related acute clinical signs observed in the 5-day oral bolus dog study are due to the oral route-of-exposure and bolus dosing employed in this study, which results in very rapid and complete uptake of DEET into the blood (see Figure 6). Because this type of rapid and high peak blood level of DEET does not occur for humans using DEET as a topically applied insect repellent (see Figure 6), the 5-days oral bolus dog study reported herein is not appropriate to use for safety characterization following dermal application of DEET DEET plasma profile for human volunteers administered DEET at the 95th percentile of human use (50 mg/kg/day) during and following the first of four daily 8-h dermal applications; each value represents the mean ± SE of six individual values (three males and three females). DEET plasma profile for dogs following administration of a single bolus oral dose of DEET at the NOAEL (75 mg/kg/day); each value represents the mean ± SE of eight individual values (four males and four females).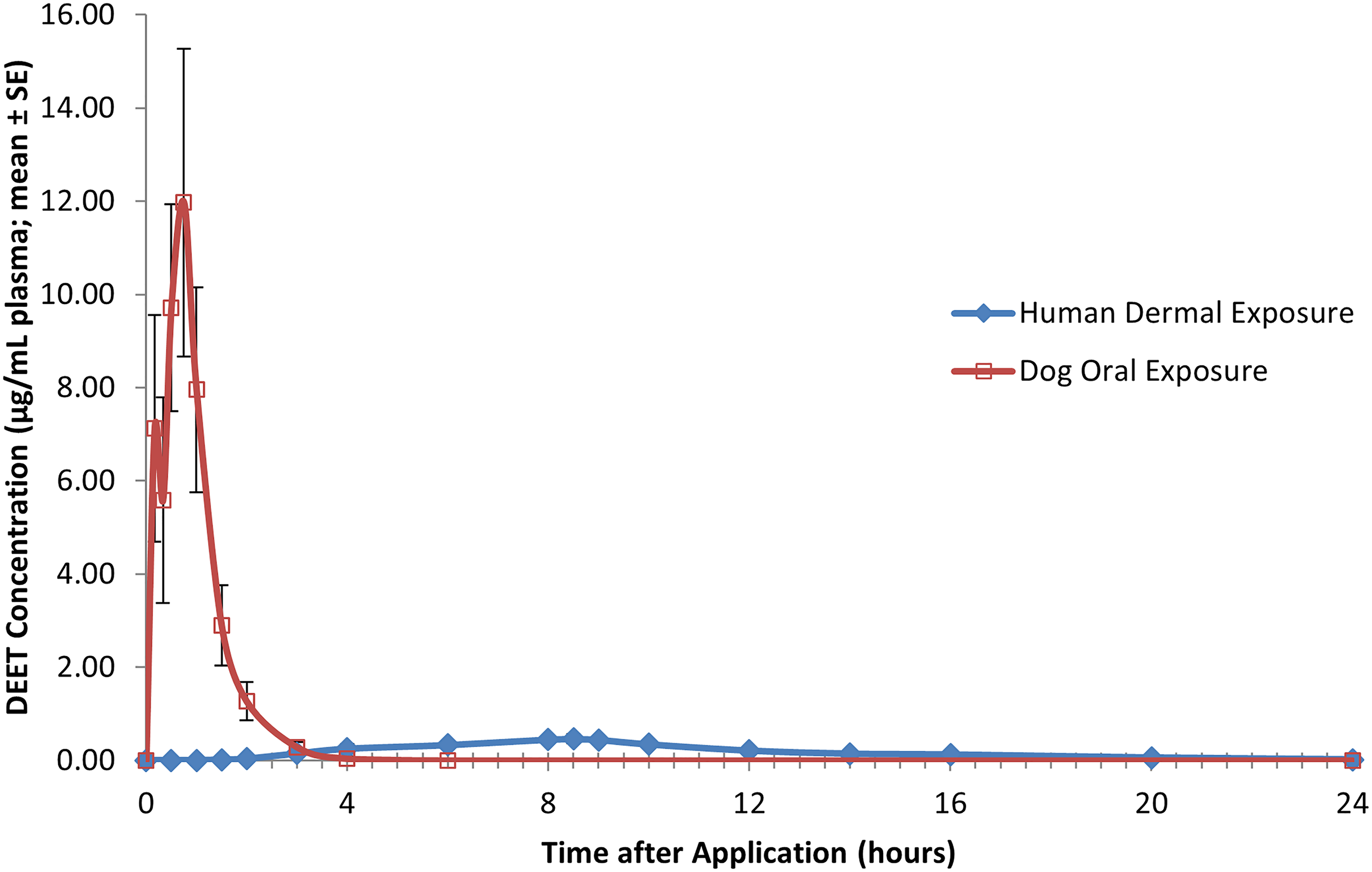
However, the rat 90-day dermal toxicity study provides a relevant, albeit conservative model for safety characterization since rats absorb about 78–85% of dermally applied DEET, whereas humans absorb on average 5.6–8.4% (conservatively estimated as 20% by regulatory authorities to account for unrecovered material) of topically applied DEET.22,23 Therefore, if rats and humans were administered equal dermal doses of DEET, the rat would absorb at least 4-fold (80% ÷ 20%) to >10-fold (85% ÷ 8.4%) more than the amount humans would. This difference between rats and humans in absorbed dermally applied dose makes the safety characterization even more conservative. DEET plasma profile for human volunteers administered DEET at the 95th percentile of human use (50 mg/kg/day) during and following the first of four daily 8-h dermal applications; each value represents the mean ± SE of six individual values (three males and three females). DEET plasma profile for rats following administration of a single bolus oral dose of DEET (200 mg/kg); each value represents the mean ± SE of ten individual values (five males and five females).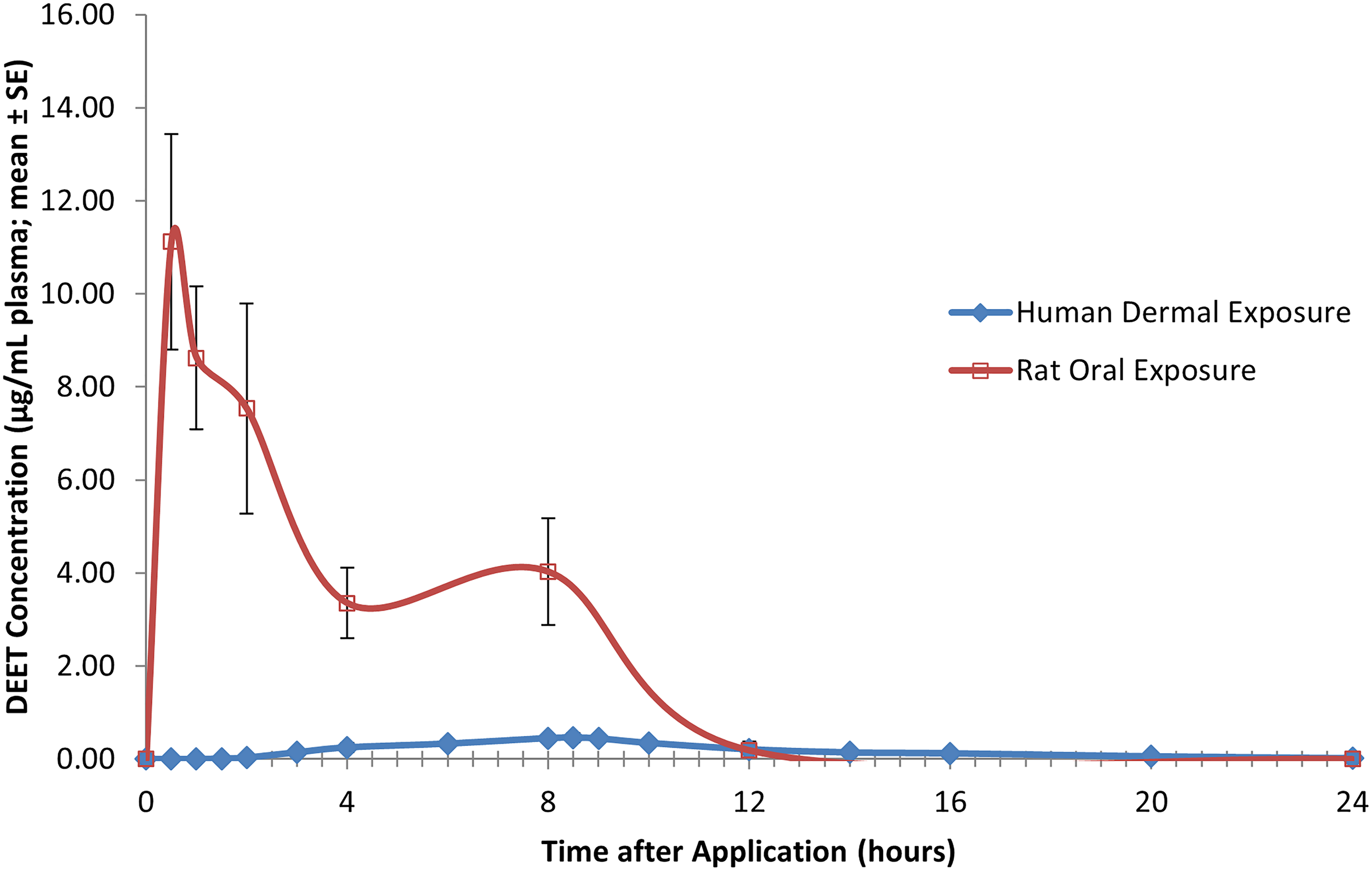
In a study conducted for regulatory submission according to a standardized testing guideline and in compliance with GLP a single dermal low dose of 100 mg/kg ring-labeled 14C-DEET was applied to the shaved backs (12.5 cm2) of fasted rats and radioactivity was measured in urine and feces collected for 7 days 22 The rats excreted 74–78% of administered radioactivity in the urine and 4–7% in the feces (total excretion 78–85%), with an additional 6.5% found on the skin surface at the application site or associated with the occlusive wrapping.
Following dermal application of 14C-DEET to male human volunteers (undiluted technical grade material or 15% w/w solution in ethanol applied to the forearm and rinsed after 8 h), total excretion of applied radioactivity in the urine and feces ranged from 3 to 8% (mean of 5.6%) in the volunteers that applied undiluted DEET and from 4 to 14% (mean of 8.4%) in the volunteers that applied 15% DEET in ethanol. 23 The recovery of radioactivity in tape strips applied to the treated skin at 1, 23, and 45 h after rinsing was very low, indicating that DEET did not accumulate in the superficial layers of the skin. Most of the applied radioactivity was recovered in the skin rinses.
Several other non-guideline, non-GLP studies designed to evaluate one or more of the pharmacokinetic properties of DEET by various routes of exposure have been published. As summarized by Schoenig and Osimitz, 6 this body of studies demonstrates that DEET is well absorbed following dermal exposure, with absorption ranging from 7.9 to 92.5% in the various laboratory animal species. The site of application, the method of application (occluded or unoccluded) and vehicle were shown to influence dermal absorption in laboratory species. In contrast, dermal absorption in human volunteers ranged from 4.6 to 16.7%. There is virtually no evidence suggesting accumulation in tissues and, for the most part, DEET was found to be quantitatively metabolized and excreted in the urine.
The normal and intended use of DEET as an insect repellent by consumers is exclusively via dermal topical application. Although negligible incidental exposure many occur via inhalation from spray products, human exposure to DEET is essentially entirely by the dermal route and is, for the most part, intermittent and seasonal typically over a 3-month period. Therefore, the rat 90-day dermal toxicity study is the most appropriate study for human safety assessment. As described earlier, there is considerable evidence that rat skin is more permeable than human skin to DEET.22,23 Thus, systemic exposure to DEET in this animal model would be expected to exceed or mimic systemic human exposure by the dermal route. No significant endpoints for systemic toxicity concern were observed at the highest dose that could be applied to the rat (NOAEL = 1000 mg/kg/day).
20
In addition, the DEET plasma profiles of human volunteers at the 95th percentile of intended use as a topically applied insect repellent and for rats at the dermal NOAEL demonstrate that because of the plasma profile similarity of the uptake and clearance curves it is most appropriate scientifically to compare these curves for the purpose of safety characterization (see Figure 8). DEET plasma profile for human volunteers administered DEET at the 95th percentile of human use (50 mg/kg/day) during and following the first of four daily 8-h dermal applications; each value represents the mean ± SE of six individual values (three males and three females). DEET plasma profile for rats during and following the first of five daily dermal application of DEET at the NOAEL (1000 mg/kg/day); each value represents the mean ± SE of eight individual values (four males and four females).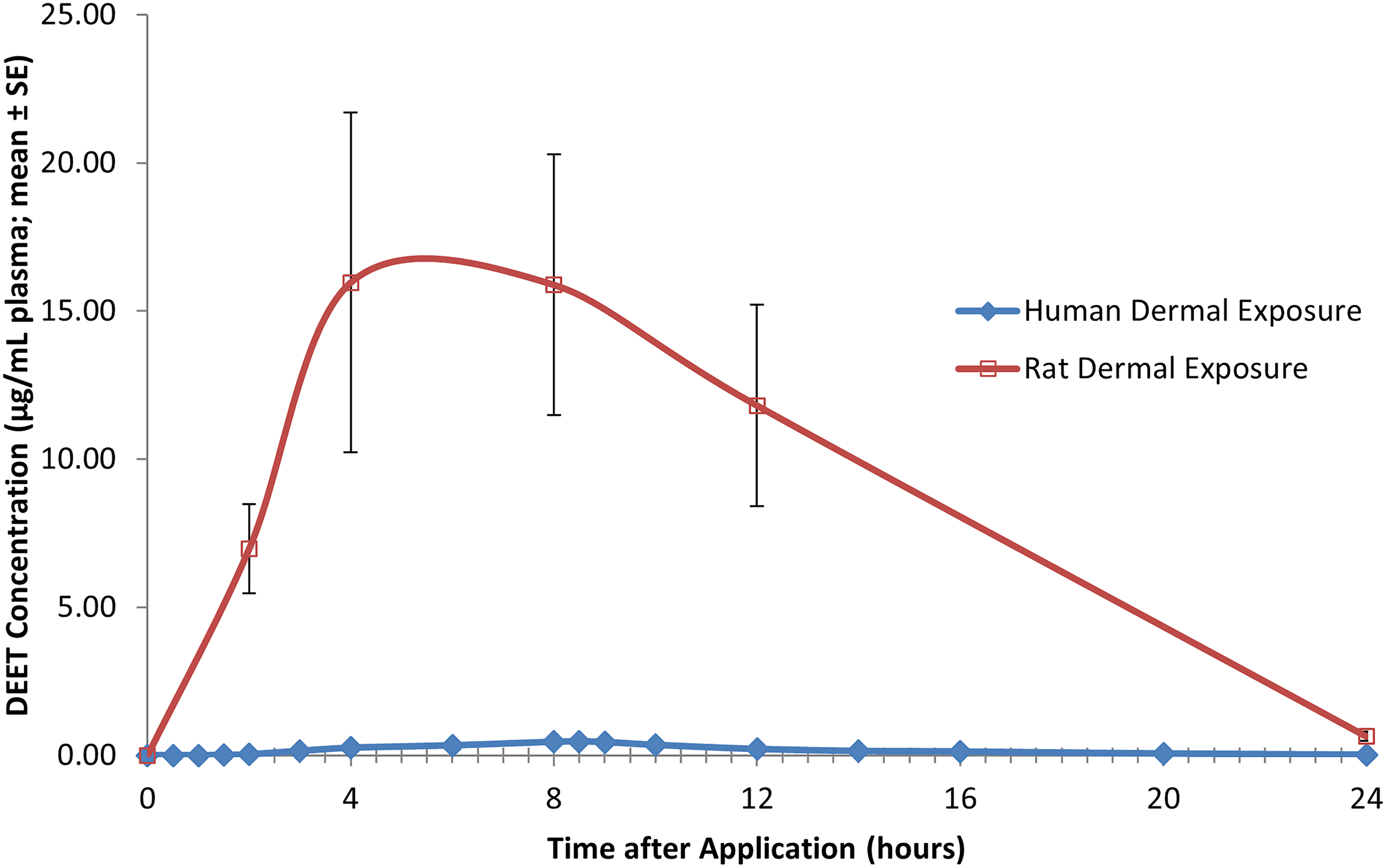
Moreover, because of the large differences in actual systemic exposure, the animal studies conducted with DEET by the oral route of administration are not suitable for defining endpoints for toxicity concern for safety or quantitative risk assessment for the normal and intended use of DEET as an insect repellent active ingredient. Nonetheless, the studies that were conducted at maximum tolerated doses (MTD) by the more rigorous oral route of administration demonstrate that DEET is not a teratogen, reproductive toxicant, selective neurotoxicant, or oncogen.4–7
Rationale for target margin of exposure less than 100
Whereas plasma level data are not usually available for pesticides and insect repellents, they are routinely developed and used for risk assessment for pharmaceutical products by U.S. FDA. A 10-fold difference in plasma levels is used as the benchmark for safety for pharmaceutical products in much the same manner as other regulatory agencies use a target MOE of 100 (uncertainty/safety factor) for administered doses for non-drug products such as pesticides.
6
Because the plasma level data for humans and laboratory animal species represent the absorbed internal dose of DEET, the uncertainties associated with making interspecies comparisons based on administered dose on an mg/kg basis have been eliminated. Because of this reduction in uncertainty, a target MOE of 10 when comparing plasma levels is considered acceptable.
6
Additionally, use of a target MOE of 100 for the rat 90-day dermal toxicity study is unduly conservative because of the abundance of DEET data relevant to risk assessment available for humans and rats as well as other laboratory species, thus eliminating uncertainty with respect to inter- and intraspecies variation. For example: • NOAEL >1000 mg/kg/day (highest dose tested) with repeated dermal exposure in rats and micropigs.20,24 • In studies conducted under comparable experimental conditions, rats have been shown to absorb ≥80% and humans ≤20% of dermally applied DEet allowing one to conclude that rats are a conservative dermal toxicology model • Dermal absorption is more rapid for rats (at the NOAEL) and slower for humans (at the 95th percentile of consumer use rate) allowing one to conclude that rats are a conservative toxicology model (see Figure 8)
A target MOE significantly less than 100 is scientifically justified in consideration of the following: 1) reliable toxicology data for multiple species, 2) more than 60 years of safe consumer experience (including children and adults), 3) similar human and rat metabolism and elimination pathways 4) extensive plasma profile data showing humans absorb DEET to a lesser degree than do rats, and 5) regulatory guidance and state-of-the-science publications (e.g. Dorne and Renwick) 25 on deriving scientifically-based uncertainty/safety factors in risk assessment. On this basis it may be concluded that a target MOE = 10 applied to DEET plasma levels at the NOAEL of the rat 90-day dermal toxicity study is appropriate to characterize risk from repeated dermal applications of DEET by humans.
Although acute clinical signs may be produced by administering DEET at high oral bolus doses in animal studies, the plasma level studies reviewed herein demonstrate that the DEET plasma profile following oral bolus dosing is much greater in terms of Cmax and AUC than that achieved following human dermal administration at the 95th percentile of consumer use. In addition, these plasma level data confirm that the rat subchronic dermal toxicity study is the most relevant model for DEET human safety assessment. Therefore, utilization of plasma level data is relevant to safety characterization of DEET and significantly reduces uncertainty in human health risk assessment.
Supplemental Material
Supplemental Material - Analysis and interpretation of pharmacokinetic studies following DEET administration to rats, dogs, and humans
Supplemental Material for Analysis and interpretation of pharmacokinetic studies following DEET administration to rats, dogs, and humansn by Andrey I Nikiforov and Thomas G Osimitz in Toxicology Research and Application
Footnotes
Acknowledgements
The authors thank Lara Hall, Marisa Rihner, Ben Burruss, and the late Conney Berger, PhD for their assistance in preparation and review of this manuscript. Also, the authors would like to acknowledge the leadership provided by the late Gerald P. Schoenig, PhD in designing and scientifically monitoring the many toxicology studies in the DEET database that form the foundation to support the worldwide registration of DEET insect repellents and their safe use by consumers.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Authors Nikiforov and Osimitz are consultants to the DEET Task Force/Joint Venture, the funding sponsor of this research.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the DEET Task Force for the authorship of this article. Research reviewed in this article was funded by the DEET Joint Venture.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
